Eye Movements Actively Reinstate Spatiotemporal Mnemonic Content
Abstract
1. Eye Movements and Memory Encoding
2. Eye Movements and Memory Retrieval
Scanpath Theory
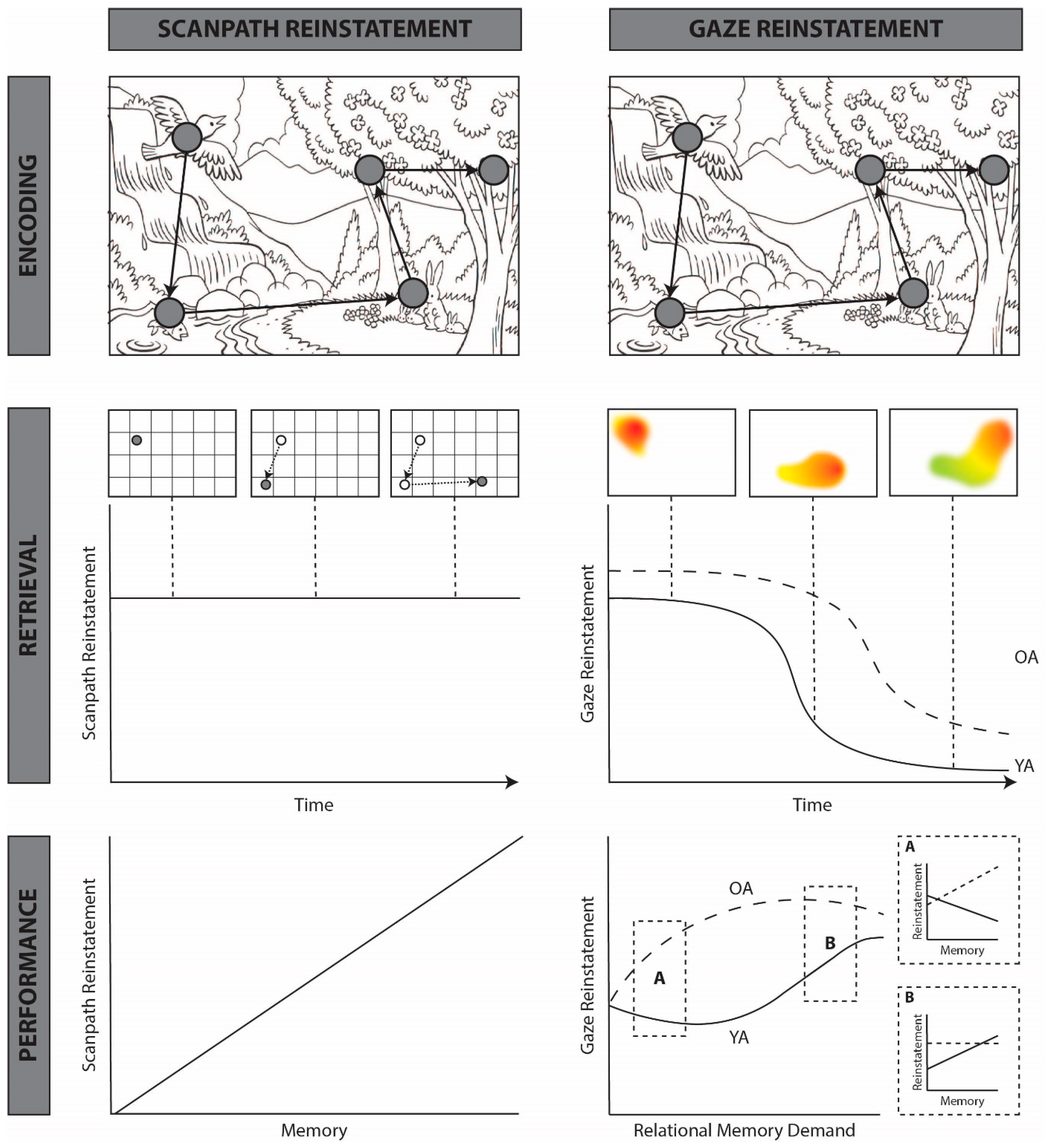
3. A New Theory of Functional Gaze Reinstatement
4. How Does Gaze Reinstatement Support Memory Retrieval?
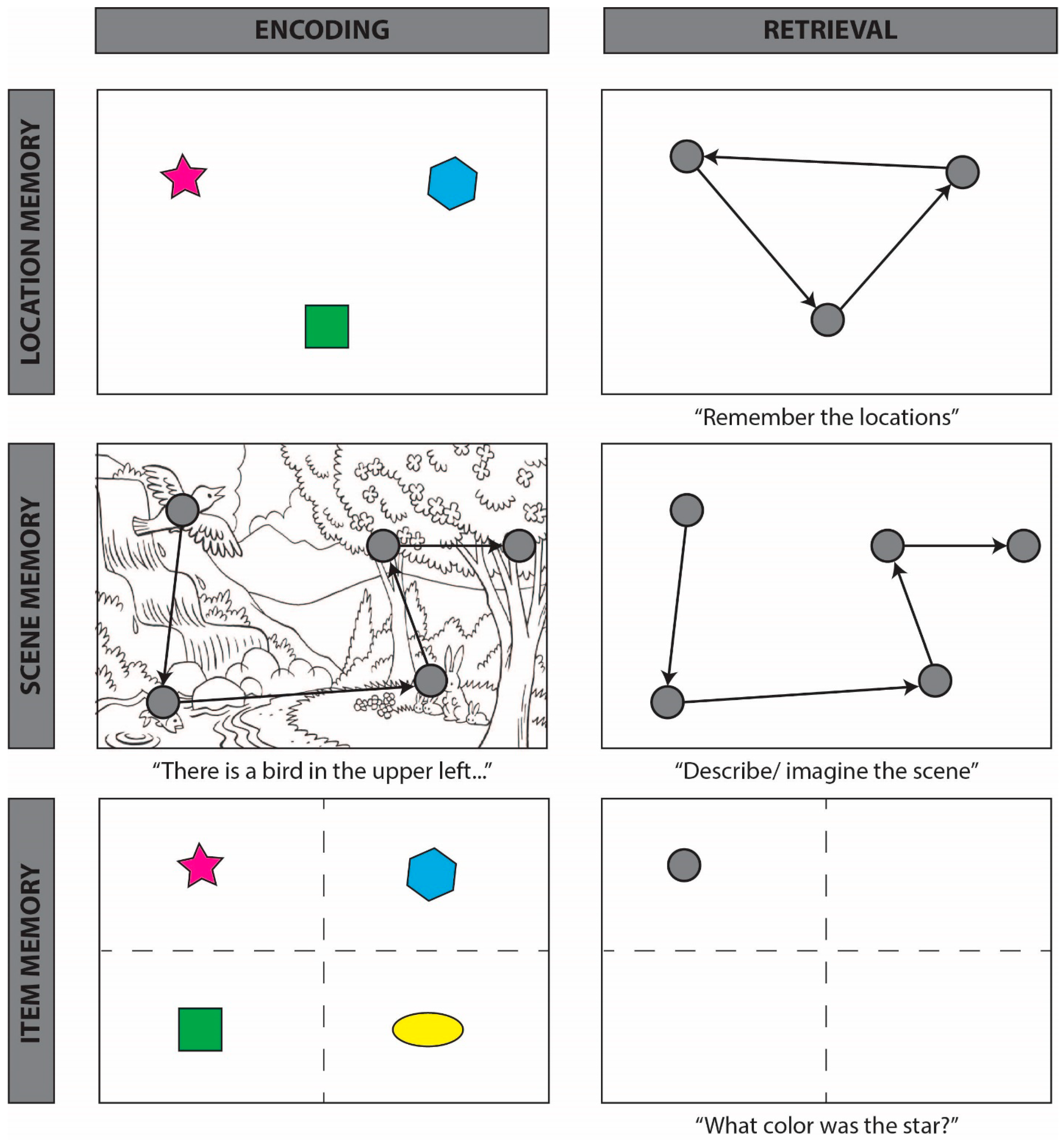
4.1. Spatial Reinstatement
4.2. Temporal Reinstatement
5. When Does Gaze Reinstatement Support Memory Retrieval?
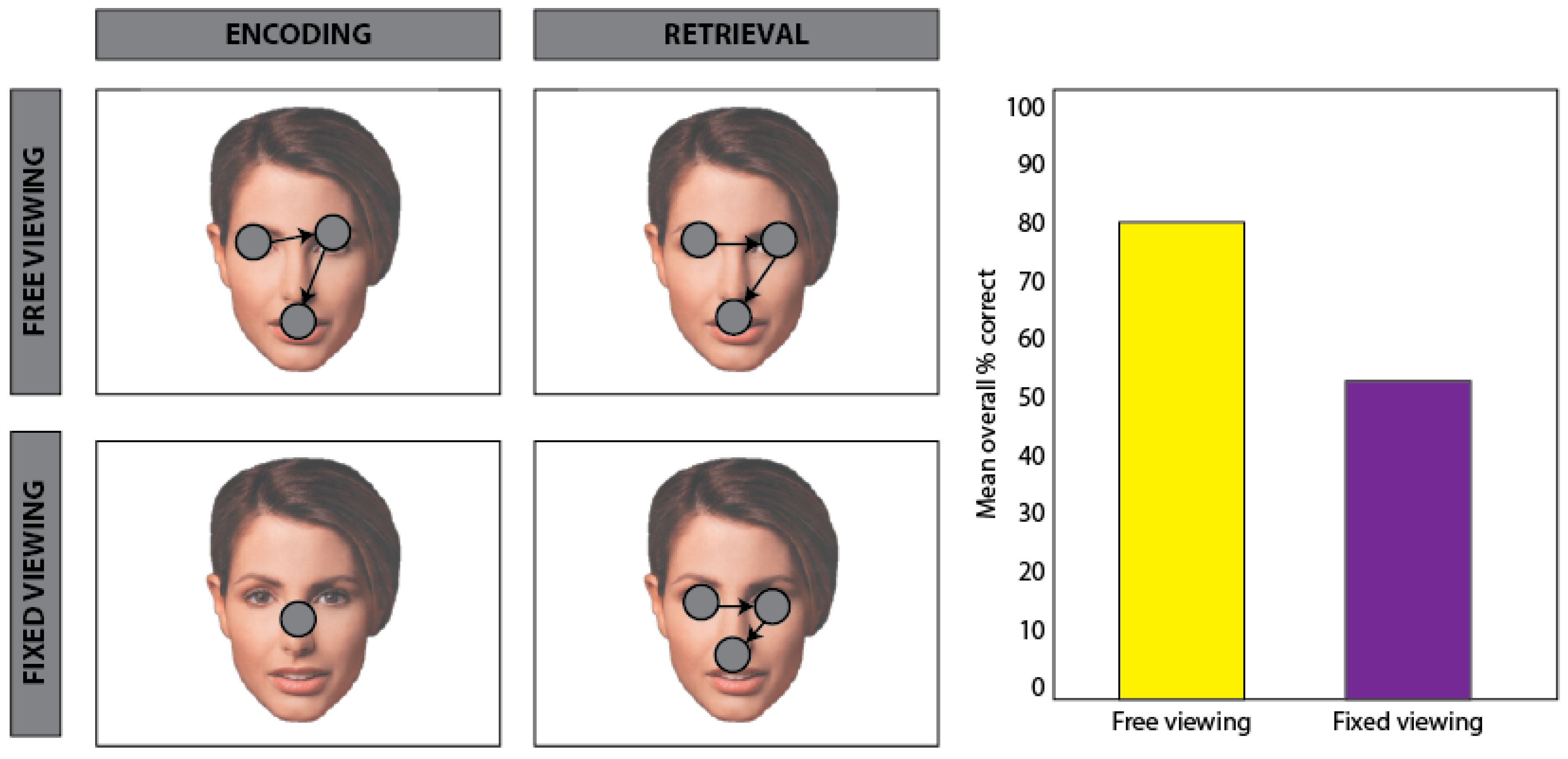
5.1. Effects of Task Demands
5.2. Effects of Memory Decline
5.3. What Are the Conditions under Which Gaze Reinstatement Supports Memory Retrieval?
5.4. What Are the Neural Correlates of Gaze Reinstatement?
6. Implications for Models of Oculomotor Control, Memory, and Aging
7. Conclusions
Funding
Conflicts of Interest
References
- Itti, L.; Koch, C. A saliency-based search mechanism for overt and covert shifts of visual attention. Vis. Res. 2000, 40, 1489–1506. [Google Scholar] [CrossRef]
- Yarbus, A.L. Eye Movements and Vision; Springer US: New York, NY, USA, 1967. [Google Scholar]
- Castelhano, M.S.; Mack, M.L.; Henderson, J.M. Viewing task influences eye movement control during active scene perception. J. Vis. 2009, 9, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Loftus, G.R.; Mackworth, N.H. Cognitive determinants of fixat ion locati on during picture viewing. J. Exp. Psychol. Hum. Percept. Perform. 1978, 4, 565–572. [Google Scholar] [CrossRef] [PubMed]
- Cerf, M.; Frady, E.P.; Koch, C. Faces and text attract gaze independent of the task: Experimental data and computer model. J. Vis. 2009, 9, 1–15. [Google Scholar] [CrossRef]
- Tatler, B.W.; Vincent, B.T. The prominence of behavioural biases in eye guidance. Vis. Cogn. 2009, 17, 1029–1054. [Google Scholar] [CrossRef]
- Einhäuser, W.; Perona, P. Objects predict fixations better than early saliency. J. Vis. 2008, 8, 1–26. [Google Scholar] [CrossRef]
- Torralba, A.; Oliva, A.; Castelhano, M.S.; Henderson, J.M. Contextual guidance of eye movements and attention in real-world scenes: The role of global features on object search. Psy. Rev. 2006, 113, 766–786. [Google Scholar]
- Henderson, J. Human gaze control during real-world scene perception. Trends Cogn. Sci. 2003, 7, 498–504. [Google Scholar] [CrossRef] [PubMed]
- Henderson, J.M.; Hayes, T.R. Meaning guides attention in real-world scene images: Evidence from eye movements and meaning maps. J. Vis. 2018, 18, 10. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.P.K.; Kamino, D.; Binns, M.A.; Ryan, J.D. Can changes in eye movement scanning alter the age-related deficit in recognition memory? Front. Psychol. 2011, 2, 1–11. [Google Scholar] [CrossRef]
- Henderson, J.M.; Williams, C.C.; Falk, R.J. Eye movements are functional during face learning. Mem. Cognit. 2005, 33, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Olsen, R.K.; Sebanayagam, V.; Lee, Y.; Moscovitch, M.; Grady, C.L.; Rosenbaum, R.S.; Ryan, J.D. The relationship between eye movements and subsequent recognition: Evidence from individual differences and amnesia. Cortex 2016, 85, 182–193. [Google Scholar] [CrossRef] [PubMed]
- Olsen, R.K.; Lee, Y.; Kube, J.; Rosenbaum, R.S.; Grady, C.L.; Moscovitch, M.; Ryan, J.D. The Role of Relational Binding in Item Memory: Evidence from Face Recognition in a Case of Developmental Amnesia. J. Neurosci. 2015, 35, 5342–5350. [Google Scholar] [CrossRef]
- Liu, Z.; Shen, K.; Olsen, R.K.; Ryan, J.D. Visual sampling predicts hippocampal activity. J. Neurosci. 2017, 37, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Killian, N.; Jutras, M.; Buffalo, E. A Map of Visual Space in the Primate Entorhinal Cortex. Nature 2012, 491, 761–764. [Google Scholar] [CrossRef] [PubMed]
- Jutras, M.J.; Fries, P.; Buffalo, E.A. Oscillatory activity in the monkey hippocampus during visual exploration and memory formation. Proc. Natl. Acad. Sci. 2013, 110, 13144–13149. [Google Scholar] [CrossRef]
- Staudigl, T.; Hartl, E.; Noachtar, S.; Doeller, C.F.; Jensen, O. Saccades are phase-locked to alpha oscillations in the occipital and medial temporal lobe during successful memory encoding. PLOS Biol. 2017, 15, e2003404. [Google Scholar] [CrossRef]
- Althoff, R.; Cohen, N. Eye-movement based memory effect: A reprocessing effect in face perception. J. Exp. Psychol. Learn. Mem. Cogn. 1999, 25, 997–1010. [Google Scholar] [CrossRef]
- Ryan, J.D.; Althoff, R.R.; Whitlow, S.; Cohen, N.J. Amnesia is a Deficit in Relational Memory. Psychol. Sci. 2000, 11, 454–461. [Google Scholar] [CrossRef]
- Hannula, D.E.; Ranganath, C. The eyes have it: Hippocampal activity predicts expression of memory in eye movements. Neuron 2009, 63, 592–599. [Google Scholar] [CrossRef]
- Bridge, D.J.; Cohen, N.J.; Voss, J.L. Distinct hippocampal versus frontoparietal-network contributions to retrieval and memory-guided exploration Donna. J. Cogn. Neurosci. 2017, 26, 194–198. [Google Scholar]
- Shen, K.; Bezgin, G.; Selvam, R.; McIntosh, A.R.; Ryan, J.D. An Anatomical Interface between Memory and Oculomotor Systems. J. Cogn. Neurosci. 2016, 28, 1772–1783. [Google Scholar] [CrossRef] [PubMed]
- Ryan, J.D.; Shen, K.; Kacollja, A.; Tian, H.; Griffiths, J.; McIntosh, R. The functional reach of the hippocampal memory system to the oculomotor system. bioRxiv 2018. [Google Scholar] [CrossRef]
- Hebb, D.O. Concerning imagery 1. Psychol. Rev. 1968, 75, 466–477. [Google Scholar] [CrossRef]
- Sheehan, P.W.; Neisser, U. Some variables affecting the vividness of imagery in recall. Br. J. Psychol. 1969, 60, 71–80. [Google Scholar] [CrossRef]
- Noton, D.; Stark, L. Scanpaths in saccadic eye movements while viewing and recognizing patterns. Vis. Res. 1971, 11, 929–942. [Google Scholar] [CrossRef]
- Noton, D.; Stark, L. Scanpaths in eye movements during pattern perception. Science 1971, 171, 308–311. [Google Scholar] [CrossRef]
- Blackmon, T.T.; Ho, Y.F.; Chernyak, D.A.; Azzariti, M.; Stark, L.W. Dynamic scanpaths: Eye movement analysis methods. In Human Vision and Electronic Imaging IV: SPIE proceedings; Rogowitz, B.E., Pappas, T.N., Eds.; SPIE Press: Bellingham, WA, USA, 1999; Volume 3644, pp. 511–519. [Google Scholar]
- Choi, Y.S.; Mosley, A.D.; Stark, L.W. String editing analysis of human visual search. Optom. Vis. Sci. 1995, 72, 439–451. [Google Scholar] [CrossRef] [PubMed]
- Hacisalihzade, S.S.; Stark, L.W.; Allen, J.S. Visual perception and sequences of eye movement fixations: A stochastic modeling approach. IEEE Trans. Syst. Man. Cybern. 1992, 22, 474–481. [Google Scholar] [CrossRef]
- Brandt, S.; Stark, L. Spontaneous eye movements during visual imagery reflect the content of the visual scene. J. Cogn. Neurosci. 1997, 9, 27–38. [Google Scholar] [CrossRef] [PubMed]
- Foulsham, T.; Underwood, G. What can saliency models predict about eye movements? Spatial and sequential aspects of fixations during encoding and recognition. J. Vis. 2008, 8, 1–17. [Google Scholar] [CrossRef]
- Underwood, G.; Foulsham, T.; Humphrey, K. Saliency and scan patterns in the inspection of real-world scenes: Eye movements during encoding and recognition. Vis. cogn. 2009, 17, 812–834. [Google Scholar] [CrossRef]
- Holm, L.; Mäntylä, T. Memory for scenes: Refixations reflect retrieval. Mem. Cogn. 2007, 35, 1664–1674. [Google Scholar] [CrossRef]
- Wynn, J.S.; Olsen, R.K.; Binns, M.A.; Buchsbaum, B.R.; Ryan, J.D. Fixation reinstatement supports visuospatial memory in older adults. J. Exp. Psychol. Hum. Percept. Perform. 2018, 44, 1119–1127. [Google Scholar] [CrossRef] [PubMed]
- Wynn, J.S.; Bone, M.B.; Dragan, M.C.; Hoffman, K.L.; Buchsbaum, B.R.; Ryan, J.D. Selective scanpath repetition during memory-guided visual search. Vis. Cogn. 2016, 24, 15–37. [Google Scholar] [CrossRef] [PubMed]
- Ryals, A.J.; Wang, J.X.; Polnaszek, K.L.; Voss, J.L. Hippocampal contribution to implicit configuration memory expressed via eye movements during scene exploration. Hippocampus 2015, 25, 1028–1041. [Google Scholar] [CrossRef] [PubMed]
- Damiano, C.; Walther, D.B. Distinct roles of eye movements during memory encoding and retrieval. Cognition 2019, 184, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Laeng, B.; Bloem, I.M.; D’Ascenzo, S.; Tommasi, L. Scrutinizing visual images: The role of gaze in mental imagery and memory. Cognition 2014, 131, 263–283. [Google Scholar] [CrossRef]
- Laeng, B.; Teodorescu, D.-S. Eye scanpaths during visual imagery reenact those of perception of the same visual scene. Cogn. Sci. 2002, 26, 207–231. [Google Scholar] [CrossRef]
- Johansson, R.; Johansson, M. Look here, eye movements play a functional role in memory retrieval. Psychol. Sci. 2013, 25, 236–242. [Google Scholar] [CrossRef] [PubMed]
- Olsen, R.K.; Chiew, M.; Buchsbaum, B.R.; Ryan, J.D. The relationship between delay period eye movements and visuospatial memory. J. Vis. 2014, 14, 8. [Google Scholar] [CrossRef] [PubMed]
- Humphrey, K.; Underwood, G. Fixation sequences in imagery and in recognition during the processing of pictures of real-world scenes. J. Eye Mov. Res. 2008, 2, 1–15. [Google Scholar]
- Bochynska, A.; Laeng, B. Tracking down the path of memory: Eye scanpaths facilitate retrieval of visuospatial information. Cogn. Process. 2015, 16, 159–163. [Google Scholar] [CrossRef] [PubMed]
- Spivey, M.J.; Geng, J.J. Oculomotor mechanisms activated by imagery and memory: Eye movements to absent objects. Psychol. Res. 2001, 65, 235–241. [Google Scholar] [CrossRef]
- Kumcu, A.; Thompson, R.L. Less imageable words lead to more looks to blank locations during memory retrieval. Psychol. Res. 2018, 0, 0. [Google Scholar] [CrossRef]
- Foulsham, T.; Kingstone, A. Fixation-dependent memory for natural scenes: An experimental test of scanpath theory. J. Exp. Psychol. Gen. 2013, 142, 41–56. [Google Scholar] [CrossRef]
- Reuter-lorenz, P.A.; Cappell, K.A. Neurocognitive Aging and the Compensation Hypothesis. Curr. Dir. Psychol. Sci. 2008, 17, 177–182. [Google Scholar] [CrossRef]
- Ferreira, F.; Apel, J.; Henderson, J.M. Taking a new look at looking at nothing. Trends Cogn. Sci. 2008, 12, 405–410. [Google Scholar] [CrossRef]
- Robin, J. Spatial scaffold effects in event memory and imagination. Wiley Interdiscip. Rev. Cogn. Sci. 2018, 9, e1462. [Google Scholar] [CrossRef]
- Rondina, R.; Curtiss, K.; Meltzer, J.A.; Barense, M.D.; Ryan, J.D. The organisation of spatial and temporal relations in memory. Memory 2017, 25, 436–449. [Google Scholar] [CrossRef]
- Tulving, E.; Thomson, D.M. Encoding specificity and retrieval processes in episodic memory. Psychol. Rev. 1973, 80, 352–373. [Google Scholar] [CrossRef]
- Kent, C.; Lamberts, K. The encoding-retrieval relationship: Retrieval as mental simulation. Trends Cogn. Sci. 2008, 12, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Parker, R.E. Picture processing during recognition. J. Exp. Psychol. Hum. Percept. Perform. 1978, 4, 284–293. [Google Scholar] [CrossRef]
- Richardson, D.C.; Spivey, M.J. Representation, space and Hollywood Squares: Looking at things that aren’t there anymore. Cognition 2000, 76, 269–295. [Google Scholar] [CrossRef]
- Altmann, G.T.M. Language-mediated eye movements in the absence of a visual world: The “blank screen paradigm”. Cognition 2004, 93, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Johansson, R.; Holsanova, J.; Holmqvist, K. Pictures and spoken descriptions elicit similar eye movements during mental imagery, both in light and in complete darkness. Cogn. Sci. 2006, 30, 1053–1079. [Google Scholar] [CrossRef]
- Johansson, R.; Holsanova, J.; Dewhurst, R.; Holmqvist, K. Eye movements during scene recollection have a functional role, but they are not reinstatements of those produced at encoding. J. Exp. Psychol. Hum. Percept. Perform. 2012, 38, 1289–1314. [Google Scholar] [CrossRef]
- Scholz, A.; Mehlhorn, K.; Krems, J.F. Listen up, eye movements play a role in verbal memory retrieval. Psychol. Res. 2016, 80, 149–158. [Google Scholar] [CrossRef]
- Martarelli, C.S.; Mast, F.W. Eye movements during long-term pictorial recall. Psychol. Res. 2013, 77, 303–309. [Google Scholar] [CrossRef]
- Parkhurst, D.; Law, K.; Niebur, E. Modeling the role of salience in the allocation of overt visual attention. Vis. Res. 2002, 42, 107–123. [Google Scholar] [CrossRef]
- Tatler, B.W.; Baddeley, R.J.; Gilchrist, I.D. Visual correlates of fixation selection: Effects of scale and time. Vis. Res. 2005, 45, 643–659. [Google Scholar] [CrossRef] [PubMed]
- O’Connell, T.P.; Walther, D.B. Dissociation of salience-driven and content-driven spatial attention to scene category with predictive decoding of gaze patterns. J. Vis. 2015, 15, 1–13. [Google Scholar] [CrossRef]
- Castelhano, M.S.; Henderson, J.M. Initial scene representations facilitate eye movement guidance in visual search. J. Exp. Psychol. Hum. Percept. Perform. 2007, 33, 753–763. [Google Scholar] [CrossRef]
- Chun, M.M.; Jiang, Y. Contextual cueing: Implicit learning and memory of visual context guides spatial attention. Cogn. Psychol. 1998, 36, 28–71. [Google Scholar] [CrossRef]
- Chau, V.L.; Murphy, E.F.; Rosenbaum, R.S.; Ryan, J.D.; Hoffman, K.L. A Flicker Change Detection Task Reveals Object-in-Scene Memory Across Species. Front. Behav. Neurosci. 2011, 5, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Myers, C.W.; Gray, W.D. Visual scan adaptation during repeated visual search. J. Vis. 2010, 10, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Shih, S.-I.; Meadmore, K.L.; Liversedge, S.P. Aging, eye movements, and object-location memory. PLoS ONE 2012, 7, 1–7. [Google Scholar] [CrossRef]
- Brandstatt, K.L.; Voss, J.L. Age-related impairments in active learning and strategic visual exploration. Front. Aging Neurosci. 2014, 6, 19. [Google Scholar] [CrossRef]
- Vieweg, P.; Riemer, M.; Berron, D.; Wolbers, T. Memory Image Completion: Establishing a task to behaviorally assess pattern completion in humans. Hippocampus 2018, 340–351. [Google Scholar] [CrossRef]
- Old, S.R.; Naveh-Benjamin, M. Differential effects of age on item and associative measures of memory: A meta-analysis. Psychol. Aging 2008, 23, 104–118. [Google Scholar] [CrossRef]
- Grady, C.L.; Ryan, J.D. Age-Related Differences in the Human Hippocampus: Behavioral, Structural and Functional Measures. In The Hippocampus from Cells to Systems; Hannula, D.E., Duff, M.C., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 167–208. ISBN 978-3-319-50406-3. [Google Scholar]
- Grady, C. The cognitive neuroscience of ageing. Nat. Rev. Neurosci. 2012, 13, 491–505. [Google Scholar] [CrossRef]
- Stern, Y. Cognitive reserve. Neuropsychologia 2009, 47, 2015–2028. [Google Scholar] [CrossRef]
- Cohen, N.J.; Eichenbaum, H. Memory, Amnesia, and the Hippocampal System; The MIT Press: Cambridge, MA, USA, 1993; ISBN 0-262-03203-1. [Google Scholar]
- Eichenbaum, H.; Cohen, N.J. From Conditioning to Conscious Recollection: Memory Systems of the Brain; Oxford University Press: New York, NY, USA, 2001; ISBN 0-19-508590-6. [Google Scholar]
- Moses, S.N.; Ryan, J.D. A comparison and evaluation of the predictions of relational and conjunctive accounts of hippocampal function. Hippocampus 2006, 16, 43–65. [Google Scholar] [CrossRef]
- Eichenbaum, H.; Otto, T.; Cohen, N.J. Two functional components of the hippocampal memory system. Behav. Brain Sci. 2010, 17, 449–472. [Google Scholar] [CrossRef]
- Ryan, J.D.; Cohen, N.J. The nature of change detection and online representations of scenes. J. Exp. Psychol. Hum. Percept. Perform. 2004, 30, 988–1015. [Google Scholar] [CrossRef] [PubMed]
- Ryan, J.D.; Cohen, N.J. Evaluating the neuropsychological dissociation evidence for multiple memory systems. Cogn. Affect. Behav. Neurosci. 2003, 3, 168–185. [Google Scholar] [CrossRef]
- Olsen, R.K.; Moses, S.N.; Riggs, L.; Ryan, J.D. The hippocampus supports multiple cognitive processes through relational binding and comparison. Front. Hum. Neurosci. 2011, 6, 146. [Google Scholar] [CrossRef] [PubMed]
- Yassa, M.A.; Stark, C.E.L. Pattern separation in the hippocampus. Trends Neurosci. 2011, 34, 515–525. [Google Scholar] [CrossRef]
- Hunsaker, M.R.; Kesner, R.P. The operation of pattern separation and pattern completion processes associated with different attributes or domains of memory. Neurosci. Biobehav. Rev. 2013, 37, 36–58. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.Y.; Gould, R.L.; Coulson, M.C.; Ward, E.V.; Howard, R.J. Tests of pattern separation and pattern completion in humans—A systematic review. Hippocampus 2016, 26, 2–31. [Google Scholar] [CrossRef]
- Hannula, D.E.; Ryan, J.D.; Warren, D.E. Beyond Long-Term Declarative Memory: Evaluating Hippocampal Contributions to Unconscious Memory Expression, Perception, and Short-Term Retention. In The Hippocampus from Cells to Systems; Springer International Publishing: Cham, Switzerland, 2017; pp. 281–336. [Google Scholar]
- Rolls, E.T.; Wirth, S. Spatial representations in the primate hippocampus, and their functions in memory and navigation. Prog. Neurobiol. 2018, 171, 90–113. [Google Scholar] [CrossRef]
- Eichenbaum, H. Time cells in the hippocampus: A new dimension for mapping memories. Nat. Rev. Neurosci. 2014, 15, 732–744. [Google Scholar] [CrossRef]
- Cohen, N.J.; Ryan, J.; Hunt, C.; Romine, L.; Wszalek, T.; Nash, C. Hippocampal system and declarative (relational) memory: Summarizing the data from functional neuroimaging studies. Hippocampus 1999, 9, 83–98. [Google Scholar] [CrossRef]
- Staresina, B.P.; Davachi, L. Mind the Gap: Binding Experiences across Space and Time in the Human Hippocampus. Neuron 2009, 63, 267–276. [Google Scholar] [CrossRef]
- Henke, K.; Buck, A.; Weber, B.; Wieser, H.G. Human Hippocampus Establishes Associations in Memory. Hippocampus 1997, 256, 249–256. [Google Scholar] [CrossRef]
- Mayes, A.; Montaldi, D.; Migo, E. Associative memory and the medial temporal lobes. Trends Cogn. Sci. 2007, 11, 126–135. [Google Scholar] [CrossRef]
- Bone, M.B.; St-Laurent, M.; Dang, C.; McQuiggan, D.A.; Ryan, J.D.; Buchsbaum, B.R. Eye Movement Reinstatement and Neural Reactivation During Mental Imagery. Cereb. Cortex 2018, 1–15. [Google Scholar] [CrossRef]
- Wolfe, J.M. Guided Search 2.0 A revised model of visual search. Psychon. Bull. Rev. 1994, 1, 202–238. [Google Scholar] [CrossRef]
- Hamker, F.H. Modeling feature-based attention as an active top-down inference process. Biosystems 2006, 86, 91–99. [Google Scholar] [CrossRef]
- Klein, R.M. Inhibition of return. Trends Cogn. Sci. 2000, 4, 138–147. [Google Scholar] [CrossRef]
- Thompson, K.G.; Biscoe, K.L.; Sato, T.R. Neuronal Basis of Covert Spatial Attention in the Frontal Eye Field. J. Neurophysiol. 2005, 25, 9479–9487. [Google Scholar] [CrossRef]
- Ipata, A.E.; Gee, A.L.; Goldberg, M.E.; Bisley, J.W. Activity in the Lateral Intraparietal Area Predicts the Goal and Latency of Saccades in a Free-Viewing Visual Search Task. J. Neurosci. 2006, 34, 880–886. [Google Scholar] [CrossRef] [PubMed]
- Shen, K.; Paré, M. Neuronal activity in superior colliculus signals both stimulus identity and saccade goals during visual conjunction search. J. Vis. 2007, 7, 15. [Google Scholar] [CrossRef]
- Fecteau, J.H.; Munoz, D.P. Salience, relevance, and firing: A priority map for target selection. Trends Cogn. Sci. 2006, 10, 382–390. [Google Scholar] [CrossRef] [PubMed]
- Bisley, J.W.; Mirpour, K. The neural instantiation of a priority map. Curr. Opin. Psychol. 2019, 29, 108–112. [Google Scholar] [CrossRef] [PubMed]
- Shen, K.; McIntosh, A.R.; Ryan, J.D. A working memory account of refixations in visual search. J. Vis. 2014, 14, 11. [Google Scholar] [CrossRef][Green Version]
- Meister, M.L.R.; Buffalo, E.A. Getting directions from the hippocampus: The neural connection between looking and memory. Neurobiol. Learn. Mem. 2016, 134, 135–144. [Google Scholar] [CrossRef]
- Hannula, D.E.; Althoff, R.R.; Warren, D.E.; Riggs, L.; Cohen, N.J. Worth a glance: Using eye movements to investigate the cognitive neuroscience of memory. Front. Hum. Neurosci. 2010, 4, 1–16. [Google Scholar] [CrossRef]
- Voss, J.L.; Bridge, D.J.; Cohen, N.J.; Walker, J.A. A Closer Look at the Hippocampus and Memory. Trends Cogn. Sci. 2017, 21, 577–588. [Google Scholar] [CrossRef]
- Lindenberger, U.; Mayr, U. Cognitive aging: Is there a dark side to environmental support? Trends Cogn. Sci. 2014, 18, 7–15. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wynn, J.S.; Shen, K.; Ryan, J.D. Eye Movements Actively Reinstate Spatiotemporal Mnemonic Content. Vision 2019, 3, 21. https://doi.org/10.3390/vision3020021
Wynn JS, Shen K, Ryan JD. Eye Movements Actively Reinstate Spatiotemporal Mnemonic Content. Vision. 2019; 3(2):21. https://doi.org/10.3390/vision3020021
Chicago/Turabian StyleWynn, Jordana S., Kelly Shen, and Jennifer D. Ryan. 2019. "Eye Movements Actively Reinstate Spatiotemporal Mnemonic Content" Vision 3, no. 2: 21. https://doi.org/10.3390/vision3020021
APA StyleWynn, J. S., Shen, K., & Ryan, J. D. (2019). Eye Movements Actively Reinstate Spatiotemporal Mnemonic Content. Vision, 3(2), 21. https://doi.org/10.3390/vision3020021




