1. Introduction
The availability of high-quality single crystals is essential for the full clarification of the novel bulk properties of quantum materials, especially of unconventional superconductors. The layered perovskite superconductor Sr
2RuO
4 [
1] is a typical example. This superconductor has attracted much attention over the last twenty years as a strong candidate of a spin-triplet superconductor [
2,
3,
4,
5,
6], as well as of a bulk topological superconductor [
7,
8]. Reflecting its unconventional pairing, its superconductivity is sensitively suppressed by even non-magnetic impurities [
9,
10]. Thus, it is challenging to consistently obtain a large homogeneous crystal of Sr
2RuO
4 with
Tc close to the intrinsic value of 1.50 K, corresponding to the mean free path exceeding 1 μm. Its superconductivity is completely suppressed when the mean-free-path becomes comparable to the superconducting coherence length (≈ 70 nm), corresponding to the impurity level of c.a. 1500 ppm, where the impurities act as strong scattering centers. As another characteristic of unconventional superconductivity, the quasiparticle density of states readily emerges within the superconducting gap even with a small amount of impurities and defects [
11]; the resulting large residual density of states often makes the determination of the intrinsic gap anisotropy a challenging issue [
12,
13,
14,
15]. Furthermore, some unusual superconducting phenomena occur only in very high-purity crystals: the superconducting transition with a magnetic field applied parallel to the RuO
2 plane becomes first order at low temperatures [
16,
17,
18]. Despite the key experimental results supporting spin-triplet pairing [
19,
20,
21,
22,
23], such a first-order transition is difficult to explain within the context of spin-triplet superconductivity. This first-order transition becomes second order when the
Tc is suppressed below 1.45 K, which corresponds to an impurity level of ~50 ppm [
10]. Thus, pristine samples with impurity levels less than 10 ppm are required in order to deepen our knowledge of the superconducting state of Sr
2RuO
4.
Recent innovations in the design of uniaxial-stress cells enables the
Tc of Sr
2RuO
4 to be enhanced up to 3.5 K when the stress is along the crystalline [100] direction [
24,
25,
26,
27]. The origin of this enhancement of
Tc is attributed to the Fermi-level crossing of the van-Hove singularity in one of the three quasi-two-dimensional Fermi surfaces. It is hoped that detailed investigations of this phenomenon will lead to the clarification of the superconducting symmetry and mechanism in Sr
2RuO
4. The
Tc enhancement has actually been known for many years in the eutectic crystals of Sr
2RuO
4 with micron-size metallic Ru platelets, which introduce strong strains in Sr
2RuO
4 near the interfaces [
28,
29,
30]. In order to investigate the strain-induced superconducting phase, high-quality Ru-inclusion free single crystals of Sr
2RuO
4 with a specific in-plane crystalline direction are in demand.
In this study, we examine how we can further improve the quality of crystals of Sr
2RuO
4 grown by the floating-zone method with an infrared image furnace. The floating-zone technique has been used to produce high-quality crystals of relatively large size useful for most experimental purposes including inelastic neutron scattering. Because the technique is essentially crucible-free, it can be used to achieve the minimum possible impurity levels. The floating-zone technique has been used for the successful crystal growth of Sr
2RuO
4 [
31], as well as other ruthenates [
32,
33,
34,
35,
36,
37]. Previous reports mainly describe optimization of the atomic compositions of the feed rod and various parameters of the final growth process. On the other hand, there are a number of processes (from powder grinding to feed-rod sintering) involved prior to the zone-melting growth process. These processes turn out to be of critical importance to minimize impurity contamination. In this study, we focus on the process of feed-rod preparation in order to further improve the quality of Sr
2RuO
4 crystals. Although the reexaminations of our previous processes were performed independently in Kyoto and Dresden, we achieved consistent results and reached a consistent set of conclusions. The improved feed-rod preparation processes have allowed for the efficient production of high-quality crystals which will help deepen our understanding of the superconducting state of Sr
2RuO
4.
2. Experimental
We first describe our standard procedure of feed-rod preparation [
31], and identify possible issues. After grinding the starting materials of SrCO
3 and RuO
2 with the molar ratio of Sr:Ru = 2:
n with
n = 1.15, the powder was pressed into pellets using stainless steel molds. The pellets were typically pre-sintered at 1150 °C for 24 h. Next, the pellets were reground and the powder was packed into a balloon to shape the feed rod. For this balloon, we typically use latex tubing with a diameter of 6 mm and a thickness of 0.2 mm. The powder-filled balloon was then pressed under hydrostatic pressure (40 MPa for 5 min) and the resulting rod was sintered at 1420 °C for 2 h. Finally, the sintered rod was suspended in the floating-zone furnace. In this method, many steps are required before arriving at the final feed rod which allows for a number of opportunities for contamination. In particular, the surface of the pellets show a greenish color after pressing in the stainless-steel molds. This contamination is also evident from a black material on the surface of the pellets when pelletizing insulating white oxide powders instead of black RuO
2. Thus, when pellets are really needed, we cover the entire interior of the stainless-steel molds with Teflon sheets and discs. In the newly-developed processes described below, we avoid using stainless-steel molds altogether in order to establish procedures that leave less opportunity for contamination of the feed rod.
We have examined the following Methods (A)–(D), in all of which we avoid using stainless-steel molds. Procedure (A) is the simplest and perhaps the least prone to contamination. Methods (B) through (D) were developed to increase the hardness of the sintered feed rod and at the same time to reduce evaporation during the melt growth. For example, in Method (A) the handling of the powders is minimized which should significantly reduce the potential for contamination. However, because there is no pre-sintering, the resulting feed rod is very delicate and contains carbon from the SrCO3 starting powder. Method (D), on the other hand, requires the most handling of the powder but results in a relatively robust polycrystalline feed rod. Through trials that have been done to date, the intermediate methods have been preferred (Method (B) at Dresden and Method (C) at Kyoto). These two methods offer the best compromise between reducing the risk of contamination of the powders and achieving polycrystalline Sr2RunO2n+2 feed rods that are not too fragile and can, therefore, be suspended in the image furnace with relative ease.
- (A)
The ground powder is packed into a balloon, then pressed, and suspended in the floating-zone furnace. Here there is no sintering process and the feed rod contains carbon from pre-reacted SrCO3.
- (B)
The ground powder is packed into a balloon, then pressed, and sintered (1150 °C for 24 h in Kyoto, 1000 °C for 2 h in Dresden). The sintered rod is then suspended in the floating-zone furnace.
- (C)
The ground powder is packed into a balloon, then pressed, and sintered at 1150 °C for 24 h and then subsequently at 1420 °C for 2 h. The sintered rod is then suspended in the floating-zone furnace.
- (D)
The ground powder is packed into a balloon, then pressed, and pre-sintered at 1150 °C for 24 h. The pre-sintered rod is reground and the powder is again packed into another balloon, then pressed, and sintered at 1420 °C for 2 h. The sintered rod is then suspended in the floating-zone furnace.
All four procedures were examined in Kyoto, with emphasis on (C); Method (B) was adopted in Dresden. From these feed rods, Sr
2RuO
4 crystals were gown by the floating-zone method as described by Mao et al. [
31]. Infrared image furnaces with double-elliptical mirrors (Canon Machinery, model SC-E15HD in Kyoto, and model SC-MDH in Dresden) were used. We note that in Dresden a part of the feed rod was used as a seed during the floating-zone growth, whereas oriented single crystals or a part of the feed rod were used as seeds in Kyoto.
We now describe the procedure used to prepare the feed rods in detail, since this is the part we particularly focus on in this study. First, two parts are ground with parts in a dry nitrogen atmosphere using a mortar and pestle for at least one hour. We used 4N+ containing only 5 ppm Ba. Since readily absorbs moisture from the air, it is first heated to °C for one hour to remove adsorbed water and then weighed before being allowed to cool back to room temperature. We used 3N RuO2 typically containing a few hundred ppm Cl and ~50 ppm Si as main impurities according to the Glow Discharge Mass Spectroscopy (GDMS) analysis. The metal impurities, such as Fe, Cu, and Zn, in the RuO2 are less than 20 ppm each. The achieved Tc of as high as 1.495 K suggests that some of these impurities do not enter into the crystal structure of Sr2RuO4 during its growth from the melt. Even though the analysis shows less batch dependence, we experience some batch dependence in sintering the rods; consequently, adjustment of the feed speed during the floating-zone growth is necessary. Before packing the ground powder into a latex tubing balloon, both the inner and outer surfaces of the balloon were thoroughly cleaned. The surfaces of the as-purchased latex tubing are coated with a fine powder which prevents the balloon surfaces from sticking to itself. This powder coating must be completely removed in order to avoid unnecessary contamination of the feed rods.
Before the powder is introduced inside, a length of balloon, with both ends open, is slide over a glass rod and its outside surface is cleaned using isopropanol or ethanol and a lint-free wipe. The clean surface of the balloon is then coated with some of the ground powder. The balloon is then inverted on the glass rod such that the opposite side can be cleaned and coated. This entire process is repeated for a second time such that the inner and outer surfaces of the balloon are each cleaned and coated twice.
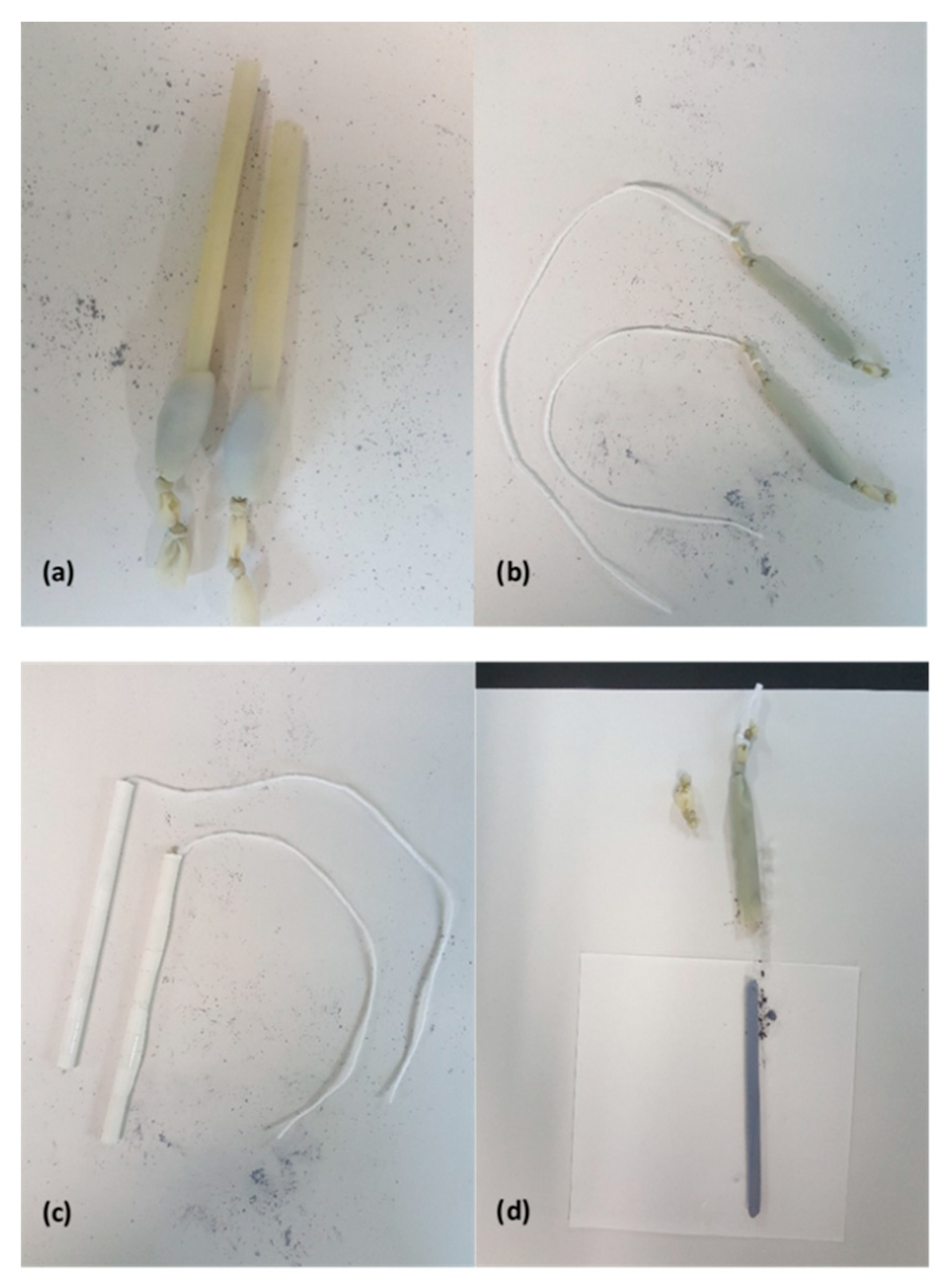
Next, one end of the balloon is tied shut with two knots separated by a few millimeters and then loaded with typically 5 g of
powder (
Figure 1a). The powder is packed into the bottom of the balloon so as to remove as much air as possible before sealing the opposite end of the balloon with a pair of knots. Once sealed, the powder is distributed evenly throughout the length of the balloon. To form a long narrow rod, the filled balloon is inserted into a tightly-rolled tube of paper. Thick paper is rolled onto a metal rod and then secured with cellophane tape before extracting the rod. The resulting tube of paper is several layers thick and has an inner diameter of 6 mm. A length of string is then tied between the pair of knots at one end of the balloon and used to pull the powder-filled balloon into the paper tube (
Figure 1b,c).
The paper tube containing the powder-filled balloon is then loaded into a stainless-steel rod which has been partially bored out from one end. The bore is next filled with water and a tight-fitting piston and hydraulic press are used to apply 40 MPa of hydrostatic pressure for five minutes. After removing and drying the paper tube, the cellophane tape is cut and the paper tube carefully uncoiled to expose the balloon. Fine scissors are used to cut one end of the balloon while one’s remaining hand lightly pinches the balloon against the pressed-powder rod near the cut. After the cut is made, the pressure applied with the fingers can be slowly released in order to allow the rod to gently slide out of the balloon (
Figure 1d). The resulting rod, if in one piece, will be 6 mm in diameter and up to 10 cm long. Typically, only feed rods that are at least 5 cm in length are suitable for floating-zone growth. The rod is very delicate and should be handled with great care.
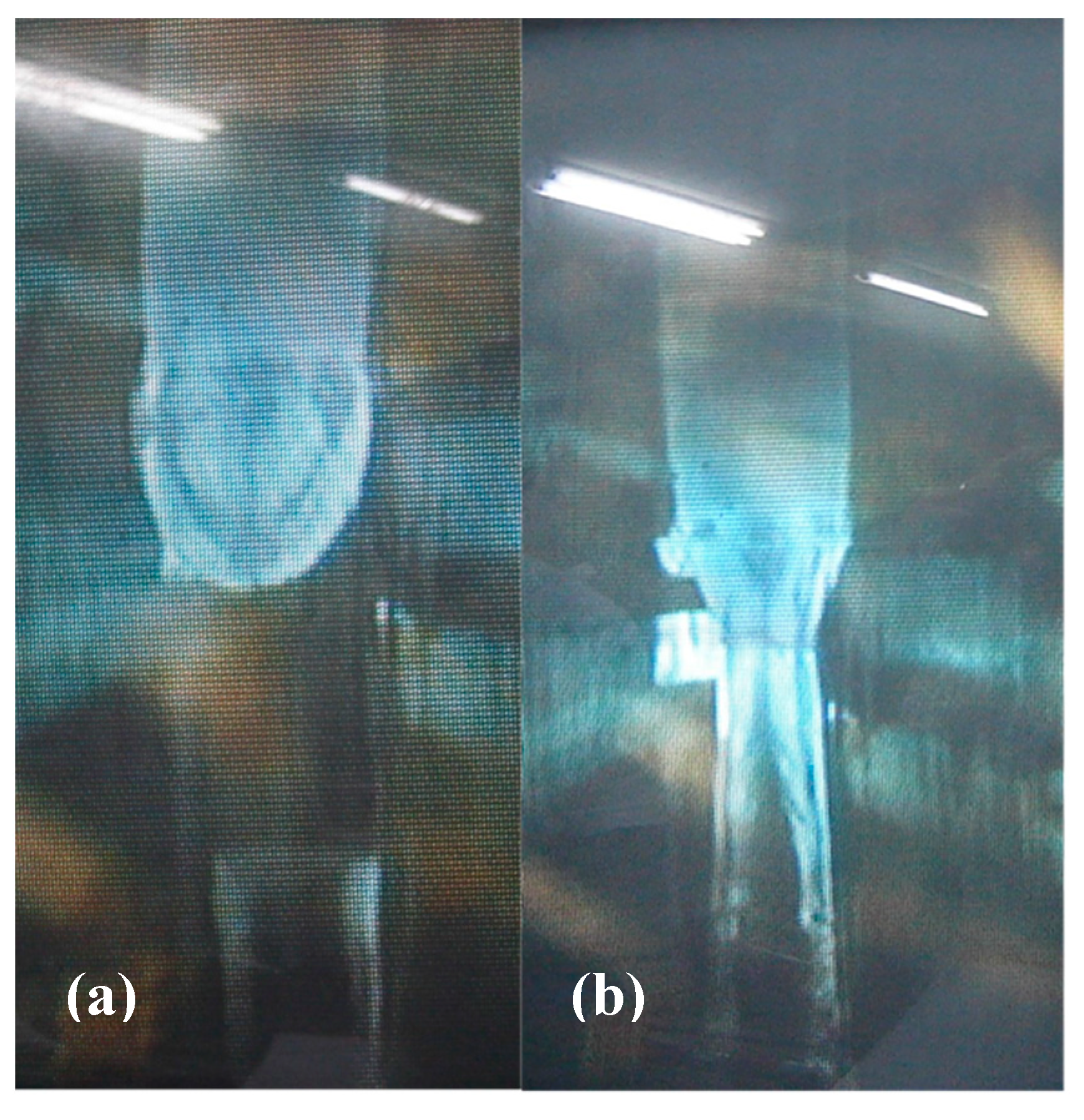
In Method (A), a wire is attached to this feed rod and one then proceeds with the floating-zone growth. A utility knife can be used to carve a narrow groove near the end of the feed rod. A length of fine chromel wire (90 percent nickel and 10 percent chromium, ϕ 0.2 mm) is wrapped around the groove in order to form a loop that can be used to suspend the feed rod in the floating-zone furnace as shown in
Figure 2. In Methods (B)–(D), the feed rod is first sintered before the chromel wire is attached. For sintering, the feed rods are first transferred onto an alumina boat that has a bedding of pre-sintered
powder. This bedding is used to prevent direct contact between the feed rod and alumina boat, which can be a source of unwanted contamination. In Method (B) the rods are sintered for 24 h at
°C (or 2 h at
°C) before the chromel wire is attached. In (C) the rods are sintered at
°C for 24 h followed by
°C for an additional 2 h, after which the chromel wire is attached to the feed rod. Finally, in Method (D) the feed rods are first pre-sintered at
°C for 24 h. These rods were then reground into powder and new feed rods were formed using the procedures described above. This second set of feed rods were then sintered at
°C for 2 h before adding the chromel wire and then suspended in the floating-zone furnace. Weight-loss measurements during sintering processes (B)–(D) show that the evaporation of the excess
is less than
= 0.01. The powder X-ray diffraction analysis indicates that the rods sintered at and above
°C contain a slight amount of
. We note that the high-temperature sinter has the advantage that the resulting feed rods are less fragile and, therefore, easier to handle and manipulate. As a seed material fixed in a seed holder at the bottom (
Figure 2), we use either a part of the polycrystalline feed rod or a crystal of Sr
2RuO
4.
During the floating-zone growth, some
is lost to evaporation via the reaction:
Therefore, it is necessary to start with
to produce single crystals with
. In
Appendix A we describe how the value of
can be estimated from the mass of the final single crystal after the floating-zone growth and the change in mass of the feed rod. The value of
is a good indicator to monitor proper growth, and for
n = 1.15 we obtain
that are slightly less than 1. So far, we have not obtained any systematic relation between
and
Tc as long as
n = 1.15, perhaps because the precision in determining
is not sufficiently high.
Figure 3a shows photographs of a polycrystalline feed rod prepared following Method (C) after its tip has been melted in the image furnace. The melted tip of the feed rod is suspended above a single-crystal seed.
Figure 3b is an image taken during stable growth of single-crystal
by the floating-zone technique. We use essentially the same growth parameters as reported previously [
31]. At Kyoto, the feed speed is typically 25 mm/h and crystal-growth speed is 43 mm/h in a gas mixture of O
2 (15%) and Ar (85%) at 0.25 MPa. The feed and seed are rotated in opposite directions, each at 30 rpm. In Dresden, the feed speed is typically 28–30 mm/h, and the crystal-growth speed is 45 mm/h in a gas mixture of O
2 (15% or 10%) and Ar (85% or 90%) at 0.35 MPa. The feed and seed are rotated in opposite directions typically at 30 rpm.
3. Results
Figure 4 shows a series of back-scattered Laue images from Sr
2RuO
4 samples along the [001] direction. All of these samples were grown using the floating-zone technique starting from feed rods prepared from Method (C). The Laue results confirm the good crystallinity of the samples. We note that high-quality samples from Method (C) have been obtained using both single crystal (C447–C453) and polycrystalline (C442 and C454) seeds. It is much preferred for some key experiments that a particular crystalline axis is along the single-crystalline rod axis. The results in
Figure 4 show a tendency of the [110] orientation along the crystalline rod if polycrystalline seeds are used. This tendency was found independently in Dresden using polycrystalline seeds. On the other hand, by using single-crystalline seeds, crystals with the axes matching with those of the seeds can be produced; this facilitates the preparation of single crystals suited for uniaxial-stress experiments. It is also noted that these Laue photos indicate low mosaicity within the X-ray beam spot size of 1 mm; namely, inclusion of crystal domains with slightly different orientations is almost negligible. We took several additional Laue photos in different positions covering the area of 2 × 2 mm
2 on the crystal piece C454B with the area of 3.2 × 3.3 mm
2 and confirmed low mosaic spread in a wider part of the crystal.
At Kyoto, low-temperature AC susceptibility measurements are used to assess the quality of the as-grown
crystals. High-quality samples exhibit a sharp superconducting transition with
Tc near 1.50 K. The AC susceptibility measurements are done using a commercial system (Quantum Design, Model PPMS) with an Adiabatic Demagnetization Refrigerator (ADR) option. The coil set and data acquisition system were designed and developed in Kyoto [
38]. The dimensions of the samples measured are typically 4 mm × 3 mm × 2 mm where the
c-axis direction is along the shortest dimension and the AC magnetic field from the primary coil is applied parallel to the
ab-plane.
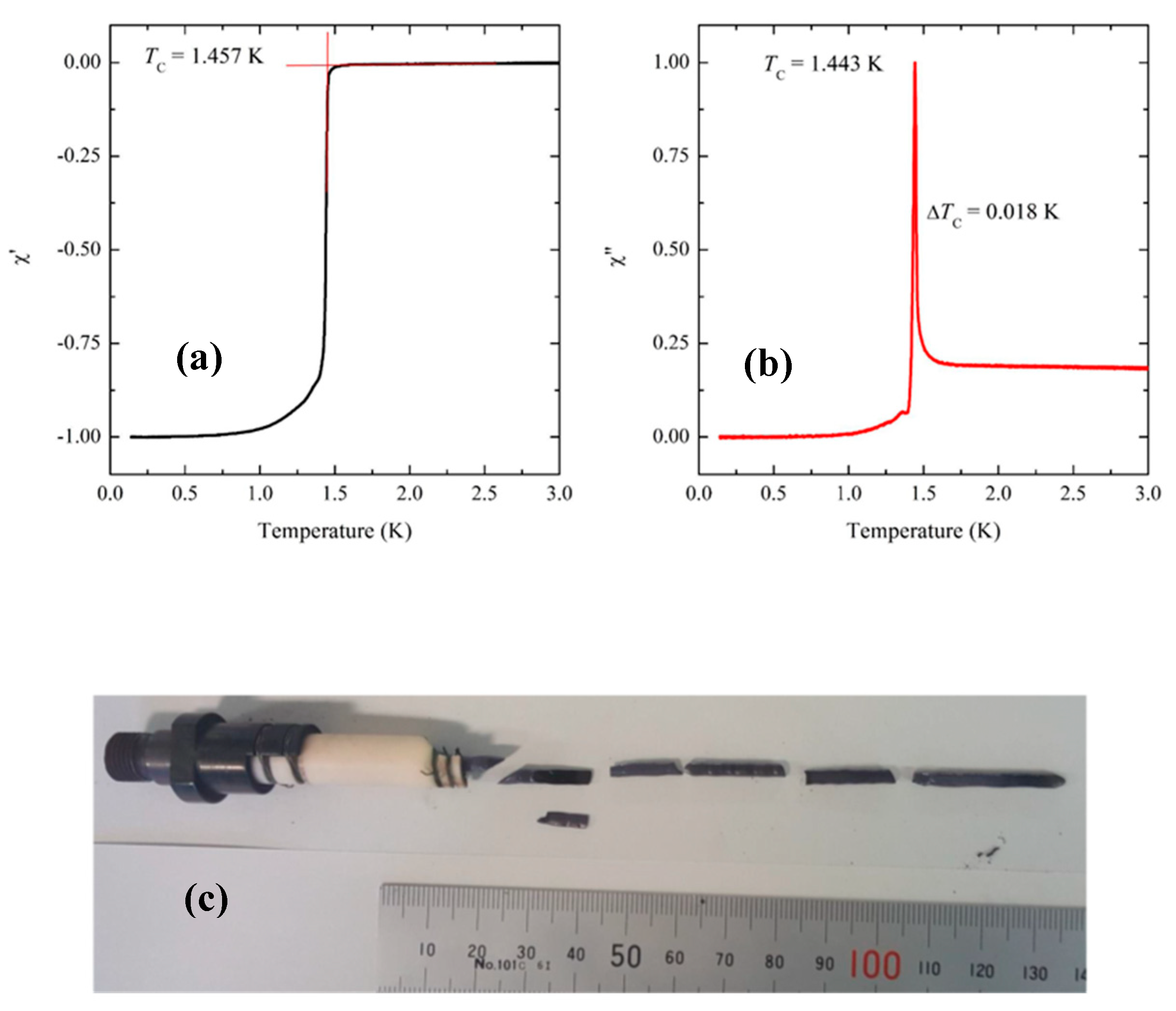
Figure 5 shows the real and imaginary parts of the AC susceptibility of the sample (batch-sample number C447-B) whose feed rod was prepared following Method (C). For this sample, a single-crystal seed was used during the floating-zone growth. The
data show an onset
that is greater than 1.45 K and the width of the superconducting transition, as measured from the FWHM of
, is less than 20 mK. As described earlier, we used as a seed either a crystal of Sr
2RuO
4 or a part of the polycrystalline feed rod. High-quality crystals are produced in both cases. However, the samples showing sharpest superconducting transitions with transition widths less than 30 mK were obtained when using single-crystal seeds. Although most of the experimental work at Kyoto has focused on Method (C), we have also successfully grown high-quality
crystals using Methods (A), (B), and (D). Method (A) has the advantage that it involves the least handling of the powders prior to the floating-zone growth and therefore is the least susceptible to contamination. However, since there is no pre-sintering the feed rods are very fragile and can be easily broken when adding the wire required to suspend the rod in the image furnace. Furthermore, these feed rods still contain carbon at the start of the floating-zone growth. Method (D) has the disadvantage that it requires the most handling of the powder prior to the floating-zone growth and these feed rods take the most time to prepare.
A photograph of a crystal grown from a feed rod prepared by Method (B) at Dresden is shown in
Figure 6a. Because we used a polycrystalline seed, rather than a single-crystalline seed, for this crystal, the growth at the first stage was necked. A back-scattered Laue-image from another batch of sample along the [001] direction is shown in
Figure 6b, after cleaving the as-grown pieces along the basal plane. It is worth mentioning that crystals grown using polycrystalline seeds have a tendency of their [110] axis along the crystalline rod within 10 degrees. This tendency has also been observed at Kyoto. We note, however, that despite this tendency there are exceptions. Sample C454 was grown at Kyoto using a polycrystalline seed and its [100] axis was found to be misaligned with the crystal-growth axis by only 4.4°. The bottom-right image in
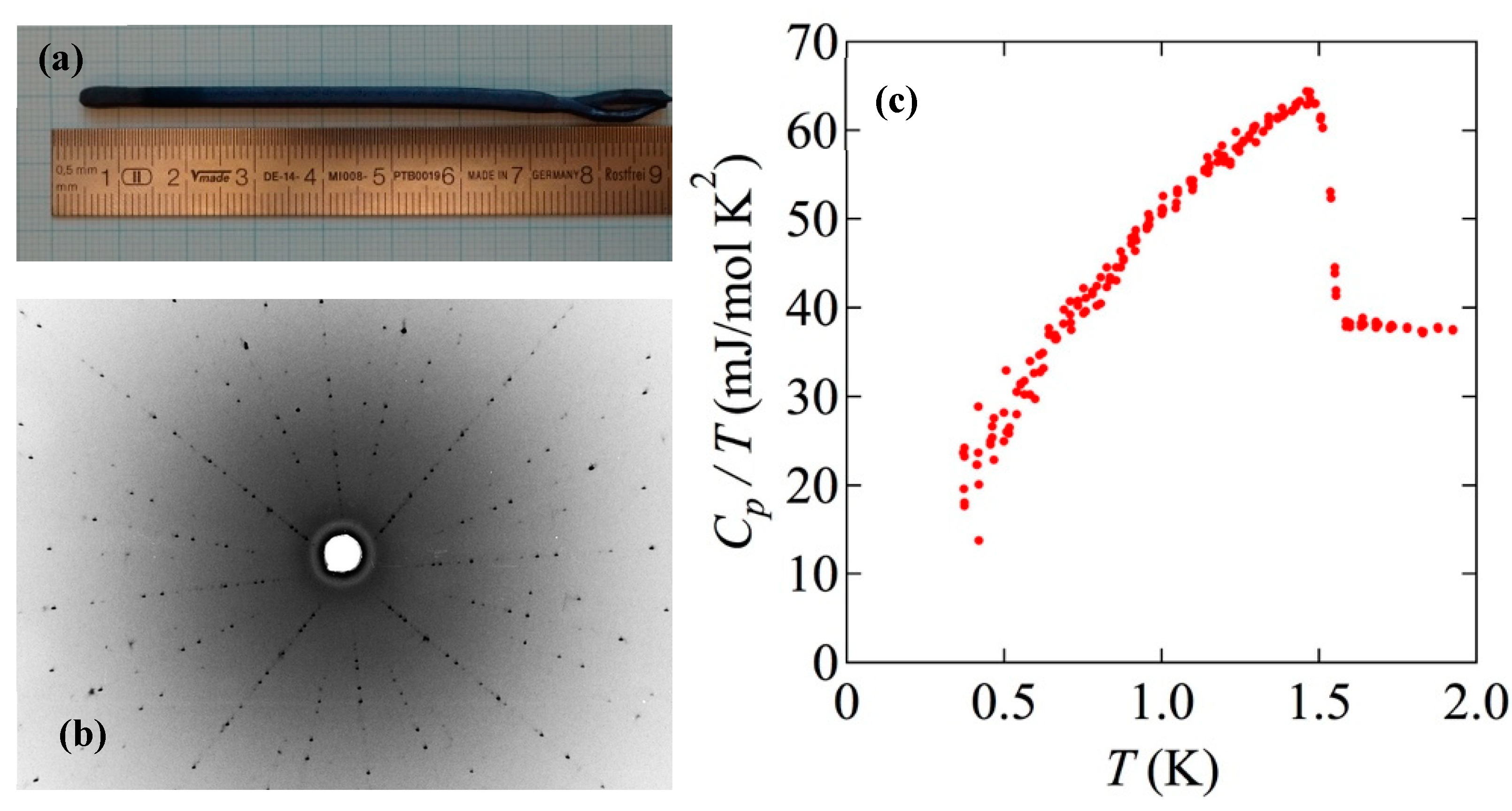
Figure 4 shows the back-scattered Laue image obtained from this sample. In order to characterize the bulk quality of the crystal prepared following Method (B) in Dresden, we measured the specific heat (
Cp) as a function of temperature (
T) below 2 K using a PPMS equipped with a
3He cooling system.
Figure 6c shows the measured specific heat divided by temperature (
Cp/T) as a function of temperature. We see a clear and sharp superconducting transition at 1.5 K, along with a relatively small residual density of states, deduced by extrapolating
Cp/T to
T = 0 K. These observations guarantee that the grown crystal is of high quality. We note that the typical sample size used for the
Cp measurement is 3 mm × 3 mm × 1 mm. This size is sufficient for a wide variety of experiments.