Non-Conventional Ingredients for Tilapia (Oreochromis spp.) Feed: A Systematic Review
Abstract
1. Introduction
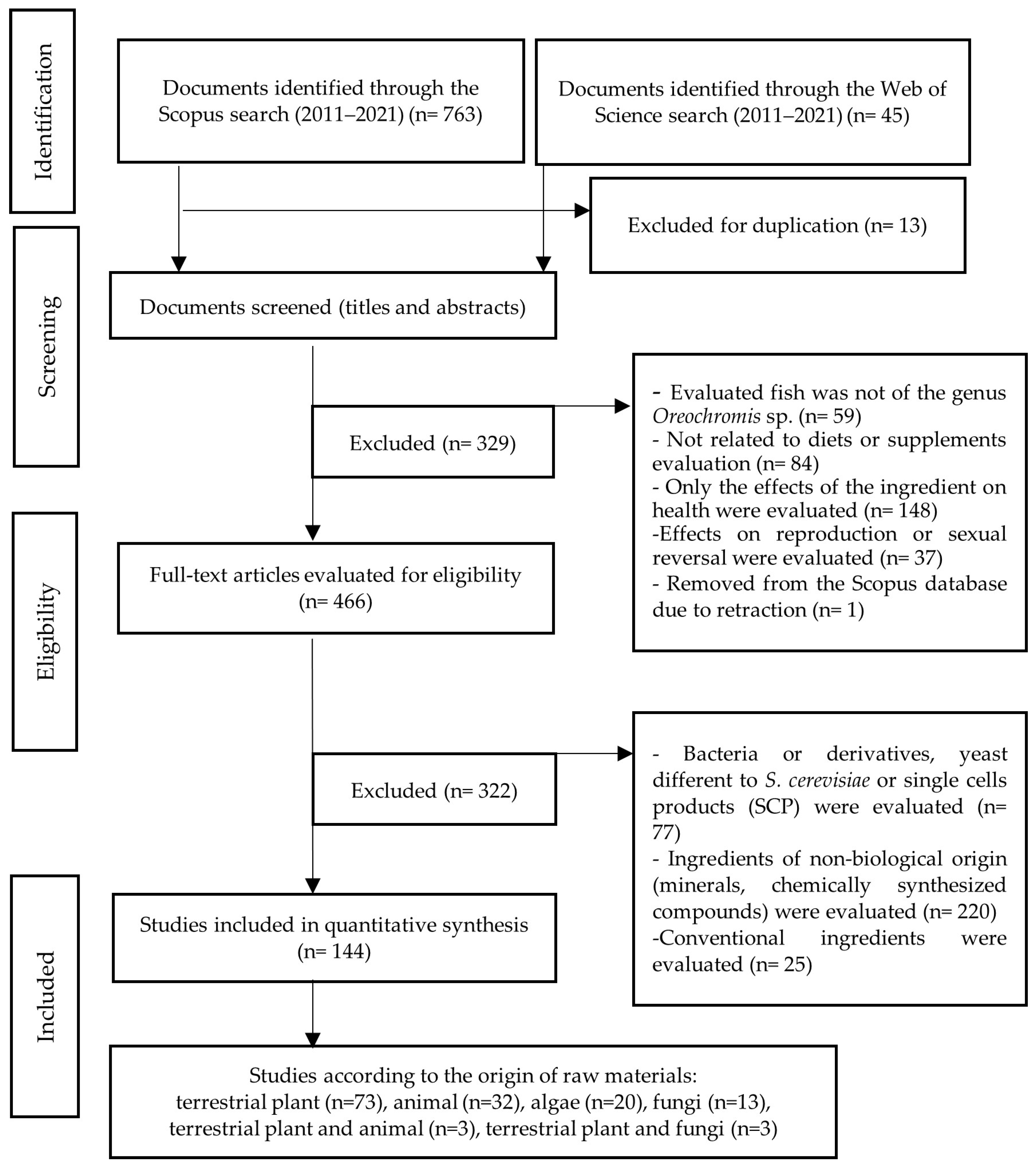
2. Materials and Methods
- -
- The fish evaluated is not of the genus Oreochromis spp.
- -
- The article is not related to diet or supplement evaluation.
- -
- Only the effects of the ingredient on fish health were evaluated in the article.
- -
- Only effects on reproduction or sexual reversion were evaluated in the article.
- -
- The ingredients evaluated are not included in the scope of the study but are bacteria, minerals, or chemically synthesized compounds.
- -
- The ingredients evaluated are conventional. In the context of this paper, the term “conventional ingredient” is used to refer to an ingredient traditionally used in the production of commercial fish feed. For example, soybean, corn, fishmeal, and fish oil, among others. After these depuration stages, a preliminary selection of articles dedicated to feeding based on terrestrial plants (embryophytes), animals, algae, fungi, and some industrial by-products was obtained. The procedure followed is outlined in Figure 1.
- Raw material origin—the food source evaluated was algae, animal, fungus, plant, or insect.
- Raw material processing method—the type of raw material evaluated in the articles: by-product, excrement, seed, meal, cake, shell, bran, extract, oil, essential oil, leaves, powder, hydrolate, protein hydrolysate, the residue of slaughtering, syrup, and nanoparticles.
- Analyses carried out—the type of evaluation carried out in the study: growth, body composition, digestibility, feed conversion ratio, health, survival, food consumption, food cost, and sensory acceptance.
3. Results and Discussion
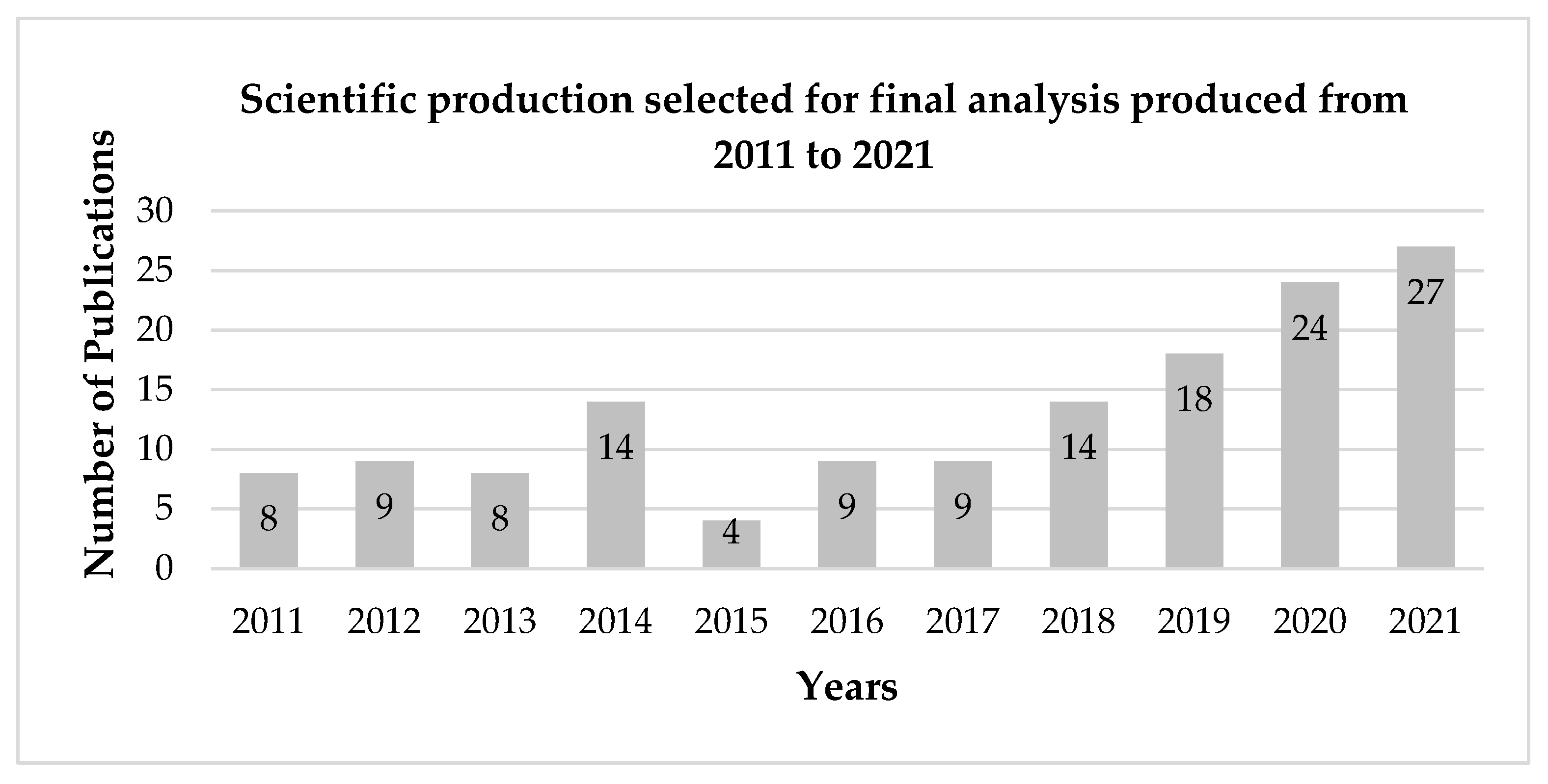
3.1. Analysis Criteria
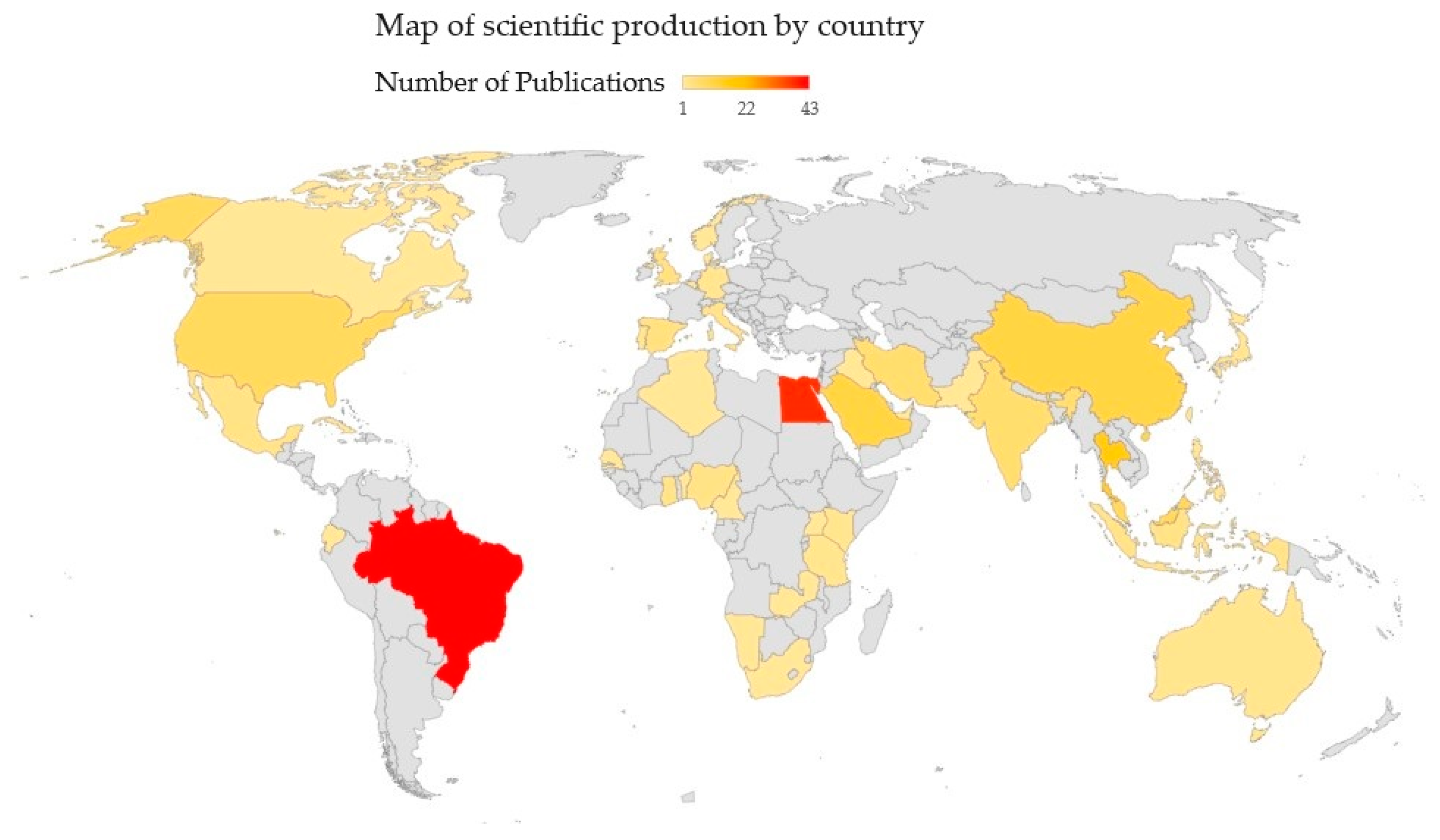
3.2. General Considerations
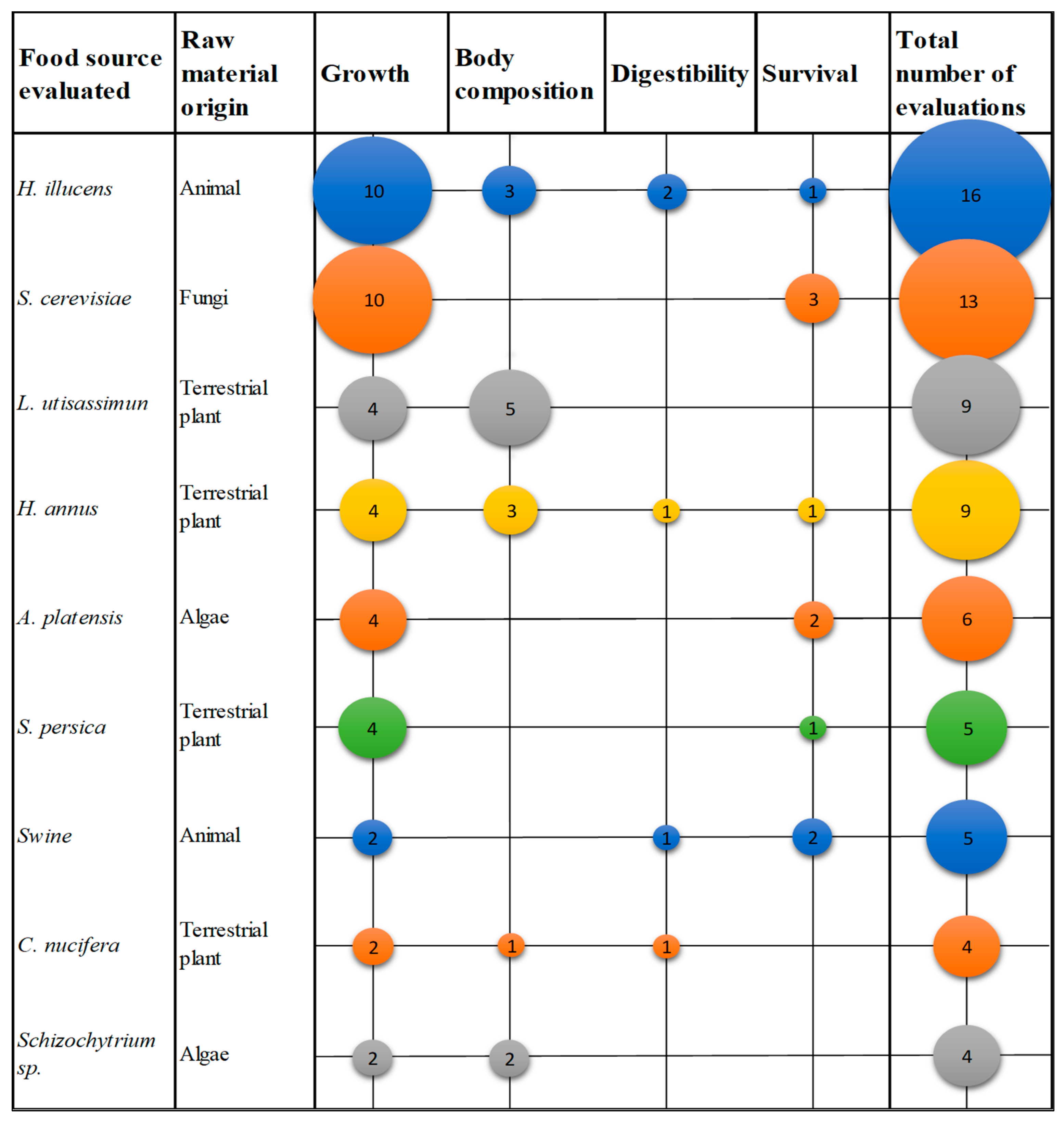
3.3. Parameters Studied in Fish
3.3.1. Growth
- Wi: initial weight
- Wf: final weight.
3.3.2. Fatty Acids Profile
| Plant Species | H. annuus, L. usitatissimum, C. nucifera, O. europaea | B. napus, Salvia hispánica L. | H. annuus, Perilla frutescens (L.) Britton | H. annuus, L. usitatissimum, B. napus, C. sativum | Borago officinalis L., Oenothera biennis L. |
|---|---|---|---|---|---|
| Raw material processing method | Oil | Oil | Oil | Oil | Oil |
| Fish species | n | n | GIFT | GIFT | n |
| Stage | J | J | - | F | - |
| Omega 3 | - | ↑ | ↑ * | = | ↓ * |
| Omega 6 | - | ↑ | ↓ * | ↓ * | ↑ * |
| SFA | = | ↑ | = | = | = |
| MUFA | = | ↑ | ↓ * | = | = |
| PUFA | = | ↑ | ↑ * | = | = |
| LCPUFA | ↓ | - | - | = | - |
| Omega 3 | - | ↑ | - | = | - |
| Omega 6 | - | ↑ | - | ↑ * | - |
| n3/n6 | ↓ | ↑ | ↑ * | = | ↓ |
| Reference | [37] | [132] | [133] | [62] | [134] |
| Raw Material Origin | Algae | Animal | Fungi | |||
|---|---|---|---|---|---|---|
| Food source | Schizochytrium sp. | N. salina | Schizochytrium sp. | H. illucens | T. molitor | Aurantiochytrium sp. |
| Raw material processing method | Meal | Meal | Dried whole cell | Meal | Meal | Meal |
| Fish species | n | n | n | n | n | GIFT |
| Stage | J | F | J | F | J | J |
| Omega 3 | ↑ | ↑ * | = | ↓ | ↓ * | ↑ |
| Omega 6 | = | ↓ * | = | = | ↑ | = |
| SFA | = | ↑ * | ↑ * | = | ↓ | = |
| MUFA | = | ↑ * | ↓ * | = | = | = |
| PUFA | = | - | = | = | = | ↑ * |
| LCPUFA | - | - | - | - | - | - |
| Omega 3 | - | - | = | - | - | ↑ |
| Omega 6 | - | - | ↑ | - | - | ↓ |
| n3/n6 | ↑ | ↑ * | ↓ | - | ↓ | ↑ |
| Reference | [102] | [104] | [107] | [87] | [90] | [124] |
3.3.3. Digestibility
| Raw Material Origin | Food Source | Fish Species | Stage | Raw Material Processing Method | ADC | Reference | |||
|---|---|---|---|---|---|---|---|---|---|
| Dry Matter | Protein | Energy | Lipid | ||||||
| Plant species | G. max | n | J | By-product: Okara | ↓= | ↓=↑ | ↓= | - | [49] |
| E. ulmoides, A. membranaceus, L. japonica, C. pilosula | n × a | J | Extract | ↑ | ↑ | = | - | [74] | |
| Unknown | n × m | F | Charcoal powder | ↑ | ↑ | - | - | [70] | |
| M. sculenta | n | J | Root meal, Leaves meal | ↑ | ↓ | ↑ | - | [137] | |
| Animal | H. illucens, C. putoria | n | F | Meal | = | ↓= | - | ↓= | [76] |
| G. bimaculatus | n | F | Meal | - | ↑ | - | ↑ | [79] | |
| H. illucens | n × m | J | Oil | - | = | - | ↓= | [80] | |
| H. illucens | n | J | Meal | - | ↑ | - | - | [82] | |
| Algae | Porphyra dioica C. Agardh, Ulva spp., Gracilaria vermiculophylla (Ohmi) Papenfuss, Sargassum muticum (Yendo) Fensholt1, | n | J | Meal | ↓ | ↓ | ↓ | ↓= | [138] |
| Chlorella sorokiniana Shihira & R.W. Krauss | - | J | Meal | = | = | = | - | [139] | |
| By-product | Poultry, Swine | n | J | Protein hydrolysates; Slaughter residues | =↑ | =↑ | =↑ | - | [140] |
3.3.4. Survival Rate
| Raw Material Origin | Fish Species | Stage | Food Source | Raw Material Processing Method | SSD | Reference |
|---|---|---|---|---|---|---|
| Plant species | n | F | C. longa | Hydrolate | ↑ | [18] |
| n | F | L. origanoides | Essential oil | ↑ | [24] | |
| n | F | S. persica | Derived | ↓= | [31] | |
| n | J | M. nigra | Syrup | =↑ | [47] | |
| n | J | P. juliflora | Meal | ↑ | [56] | |
| n | J | P. vera | Extract, Seed | =↑ | [53] | |
| n × m | F | Hevea brasiliensis (Willd. ex A. Juss.) Müll. Arg. | Seed | ↓= | [145] | |
| Algae | n | F | C. lentillifera | Meal | =↑ | [100] |
| n | J | C. vulgaris | Meal | ↓↑ | [101] | |
| n × m | F | A. platensis | Meal | ↓↑ | [108] | |
| n × m | F | A. platensis | Meal | ↓= | [111] | |
| n × m | F | U. fasciata | Meal | =↑ | [109] | |
| Animal | n | F | Oreochromis spp. | By-product; Hydrolysate | =↑ | [129] |
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAO. The State of World Fisheries and Aquaculture 2022; Towards Blue Transformation; FAO: Rome, Italy, 2022. [Google Scholar] [CrossRef]
- OECD; FAO. OECD-FAO Agricultural Outlook 2021–2030. In OECD-FAO Agricultural Outlook; OECD: Paris, France, 2021. [Google Scholar] [CrossRef]
- FAO. Versión Resumida de El Estado Mundial de la Pesca y la Acuicultura 2022; Hacia la Transformación azul; FAO: Rome, Italy, 2022. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, 71. [Google Scholar] [CrossRef] [PubMed]
- Rethlefsen, M.L.; Kirtley, S.; Waffenschmidt, S.; Ayala, A.P.; Moher, D.; Page, M.J.; Koffel, J.B.; PRISMA-S Group. PRISMA-S: An extension to the PRISMA Statement for Reporting Literature Searches in Systematic Reviews. Syst. Rev. 2021, 10, 39. [Google Scholar] [CrossRef] [PubMed]
- Pai, M.; McCulloch, M.; Gorman, J.D.; Pai, N.; Enanoria, W.; Kennedy, G.; Tharyan, P.; Colford, J.M. Systematic reviews and meta-analyses: An illustrated, step-by-step guide. Natl. Med. J. India 2004, 17, 86–95. [Google Scholar] [PubMed]
- United Nations. Transforming Our World: The 2030 Agenda for Sustainable Development; United Nations: New York, NY, USA, 2015. [Google Scholar]
- Mabrouk, H.; Labib, E.M.H.; Zaki, M.A. Response of Nile Tilapia mono-sex (Oreochromis niloticus) Fingerlings to Different Sources and Levels of Protein Using Garlic and Onion as Feed Phytophytoadditives. Arab. Gulf J. Sci. Res. 2011, 29, 146–159. [Google Scholar] [CrossRef]
- Agbo, N.W.; Adjei-Boateng, D.; Jauncey, K. The Potential of Groundnut (Arachis hypogaea L.) By-Products as Alternative Protein Sources in the Diet of Nile Tilapia (Oreochromis niloticus). J. Appl. Aquac. 2011, 23, 367–378. [Google Scholar] [CrossRef]
- Kareem, Z.H.; Abdelhadi, Y.M.; Christianus, A.; Karim, M.; Romano, N. Effects of some dietary crude plant extracts on the growth and gonadal maturity of Nile tilapia (Oreochromis niloticus) and their resistance to Streptococcus agalactiae infection. Fish Physiol. Biochem. 2015, 42, 757–769. [Google Scholar] [CrossRef]
- Van Doan, H.; Hoseinifar, S.H.; Sringarm, K.; Jaturasitha, S.; Yuangsoi, B.; Dawood, M.A.; Esteban, M.; Ringø, E.; Faggio, C. Effects of Assam tea extract on growth, skin mucus, serum immunity and disease resistance of Nile tilapia (Oreochromis niloticus) against Streptococcus agalactiae. Fish Shellfish. Immunol. 2019, 93, 428–435. [Google Scholar] [CrossRef]
- Pessoa, M.; Avelar, J.; Nascimento, A.H.; Silva, K.; Soares, A.; Camargo, A.; Filho, D.F. Desempenho de tilápias-do-nilo alimentadas com farelo da casca de pequi. Arq. Bras. Med. Vet. Zootec. 2013, 65, 547–552. [Google Scholar] [CrossRef]
- Abdel-Tawwab, M.; Samir, F.; El-Naby, A.S.A.; Monier, M.N. Antioxidative and immunostimulatory effect of dietary cinnamon nanoparticles on the performance of Nile tilapia, Oreochromis niloticus (L.) and its susceptibility to hypoxia stress and Aeromonas hydrophila infection. Fish Shellfish. Immunol. 2018, 74, 19–25. [Google Scholar] [CrossRef]
- Mombach, P.I.; Adorian, T.J.; Goulart, F.R.; Martinelli, S.G.; Dalcin, M.O.; Veiverberg, C.A.; da Silva, L.P. The Effects of Fermentable Dietary Fiber on Performance and Metabolism of Nile Tilapia (Oreochromis niloticus). Braz. Arch. Biol. Technol. 2020, 63, e20190396. [Google Scholar] [CrossRef]
- Kesbiç, O.S.; Acar, Ü.; Yilmaz, S.; Aydin, Ö.D. Effects of bergamot (Citrus bergamia) peel oil-supplemented diets on growth performance, haematology and serum biochemical parameters of Nile tilapia (Oreochromis niloticus). Fish Physiol. Biochem. 2019, 46, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Dawood, M.A.O.; Ali, M.F.; Amer, A.A.; Gewaily, M.S.; Mahmoud, M.M.; Alkafafy, M.; Assar, D.H.; Soliman, A.A.; Van Doan, H. The influence of coconut oil on the growth, immune, and antioxidative responses and the intestinal digestive enzymes and histomorphometry features of Nile tilapia (Oreochromis niloticus). Fish Physiol. Biochem. 2021, 47, 869–880. [Google Scholar] [CrossRef] [PubMed]
- Handajani, H.; Andriawan, S.; Gilang, R. Enrichment of commercial feed with plant proteins for Oreochromis niloticus diet: Digestibility and growth performance. AACL Bioflux 2021, 14, 2894–2904. Available online: https://www.bioflux.com.ro/docs/2021.2894-2904.pdf (accessed on 22 January 2022).
- Pereira, M.; Hess, J.D.; Rodhermel, J.C.B.; Farias, D.R.; Schleder, D.D.; Alves, L.; Bertoldi, F.C.; Chaban, A.; DE Andrade, J.I.A.; Jatobá, A. Curcuma longa hydrolate improves Nile tilapia survival in a recirculation rearing system, maintaining the animal homeostasis and modulating the gut microbial community. An. Acad. Bras. Ciências 2021, 93, e20210088. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, H.K.; Al-Sagheer, A.A.; Reda, F.M.; Mahgoub, S.A.; Ayyat, M.S. Dietary curcumin supplement influence on growth, immunity, antioxidant status, and resistance to Aeromonas hydrophila in Oreochromis niloticus. Aquaculture 2017, 475, 16–23. [Google Scholar] [CrossRef]
- Al-Sagheer, A.A.; Mahmoud, H.K.; Reda, F.M.; Mahgoub, S.A.; Ayyat, M.S. Supplementation of diets for Oreochromis niloticus with essential oil extracts from lemongrass (Cymbopogon citratus) and geranium (Pelargonium graveolens) and effects on growth, intestinal microbiota, antioxidant and immune activities. Aquac. Nutr. 2018, 24, 1006–1014. [Google Scholar] [CrossRef]
- Van Doan, H.; Hoseinifar, S.H.; Sringarm, K.; Jaturasitha, S.; Khamlor, T.; Dawood, M.A.; Esteban, M.; Soltani, M.; Musthafa, M.S. Effects of elephant’s foot (Elephantopus scaber) extract on growth performance, immune response, and disease resistance of nile tilapia (Oreochromis niloticus) fingerlings. Fish Shellfish. Immunol. 2019, 93, 328–335. [Google Scholar] [CrossRef]
- Srichaiyo, N.; Tongsiri, S.; Hoseinifar, S.H.; Dawood, M.A.; Esteban, M.; Ringø, E.; Van Doan, H. The effect of fishwort (Houttuynia cordata) on skin mucosal, serum immunities, and growth performance of Nile tilapia. Fish Shellfish. Immunol. 2020, 98, 193–200. [Google Scholar] [CrossRef]
- Pallaya-Baleta, L.J.; Baleta, F.N.; Magistrado-Candelaria, P.; Plantado, L.C.; Baldo, D.E.B.; Navarro, M.C.; Encinas, J.L. Growth performance and economic viability of dietary inclusion of Ipomoea batatas L. shoot powder and extracts in the practical diets of Oreochromis niloticus L. Egypt. J. Aquat. Res. 2021, 48, 273–279. [Google Scholar] [CrossRef]
- Addam, K.G.S.; Pereira, S.A.; Jesus, G.F.A.; Cardoso, L.; Syracuse, N.; Lopes, G.R.; Lehmann, N.B.; Silva, B.C.; Sá, L.S.; Chaves, F.C.M.; et al. Dietary organic acids blend alone or in combination with an essential oil on the survival, growth, gut/liver structure and de hemato-immunological in Nile tilapia Oreochromis niloticus. Aquac. Res. 2019, 50, 2960–2971. [Google Scholar] [CrossRef]
- Zhang, X.; Huang, K.; Zhong, H.; Ma, Y.; Guo, Z.; Tang, Z.; Liang, J.; Luo, Y.; Su, Z.; Wang, L. Effects of Lycium barbarum polysaccharides on immunological parameters, apoptosis, and growth performance of Nile tilapia (Oreochromis niloticus). Fish Shellfish. Immunol. 2020, 97, 509–514. [Google Scholar] [CrossRef]
- Djeziri, M.; Nouri, L.; Kacher, M. An investigation on the effects of different diets on the growth performance of Nile tilapia, Oreochromis niloticus (Linné 1758). Iran J. Fish Sci. 2020, 19, 136–150. [Google Scholar] [CrossRef]
- Melo, J.F.B.; Seabra, A.G.L.; Souza, S.A.; Souza, R.C.; Figueiredo, R.A.C.R. Substituição do farelo de milho pela farinha de manga no desempenho da tilápia-do-nilo [Replacement of corn meal by mango in the dietary in performance of fingerlings of Nile-tilapia]. Arq. Bras. Med. Veterinária Zootec. 2012, 64, 177–182. [Google Scholar] [CrossRef]
- Elabd, H.; Soror, E.; El-Asely, A.; El-Gawad, E.A.; Abbass, A. Dietary supplementation of Moringa leaf meal for Nile tilapia Oreochromis niloticus: Effect on growth and stress indices. Egypt. J. Aquat. Res. 2019, 45, 265–271. [Google Scholar] [CrossRef]
- Parveen, S.; Rasool, F.; Akram, M.N.; Khan, N.; Ullah, M.; Mahmood, S.; Rabbani, G.; Manzoor, K. Effect of Moringa olifera leaves on growth and gut microbiota of Nile tilapia (Oreochromis niloticus). Braz. J. Biol. 2021, 84, e250916. [Google Scholar] [CrossRef] [PubMed]
- Serrano, A.E.J.; Tumbokon, B.L.M.; Cabanero, P.C. Nutritional Evaluation of Rhizoclonium riparium var implexum Meal to Replace Soybean in the Diet of Nile Tilapia Fry. Isr. J. Aquac.-Bamidgeh 2016, 68, 20818. [Google Scholar] [CrossRef]
- Abd El-Naby, A.S.; El Asely, A.M.; Amin, A.A.; Samir, F.; El-Ashram, A.; Dawood, M.A.O. Miswak (Salvadora persica) modulated the growth performance, antioxidative response, and histopathological damage induced by zinc toxicity in Nile tilapia (Oreochromis niloticus). Environ. Sci. Pollut. Res. 2020, 27, 31918–31932. [Google Scholar] [CrossRef]
- Toutou, M.M.; Soliman, A.A.; Elokaby, M.A.; Abdel-Rahim, M.M.; Abouelwafa, A.E.; Yones, A.E.M. The potential antimicrobial effects of dietary supplementation with Arak, Salvadora persica, on growth, health status, and pathogenic bacterial loads in Nile tilapia, Oreochromis niloticus fingerlings. Egypt. J. Aquat. Res. 2019, 45, 251–257. [Google Scholar] [CrossRef]
- Ashade, O.; Osineye, O. Effect of replacing maize with cocoa pod husk in the nutrition of Oreochromis niloticus. J. Fish. Aquat. Sci. 2010, 8, 73–79. [Google Scholar] [CrossRef][Green Version]
- Fakunmoju, F.; Babalola, O.; Ijimakinde, B.; Anjola, O.; Orowole, P. Effect of Substituting Maize with Bambara (Voandzeia subterrenea Thouars) Waste Meal in the Practical Diets of Tilapia niloticus Fingerlings. J. Fish. Aquat. Sci. 2016, 11, 185–189. [Google Scholar] [CrossRef][Green Version]
- Zahran, E.; Risha, E.; AbdelHamid, F.; Mahgoub, H.A.; Ibrahim, T. Effects of dietary Astragalus polysaccharides (APS) on growth performance, immunological parameters, digestive enzymes, and intestinal morphology of Nile tilapia (Oreochromis niloticus). Fish Shellfish. Immunol. 2014, 38, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Whangchai, N.; Ramaraj, R.; Whangchai, K.; Nomura, N.; Pimpimol, T. Innovative biorefinery concept for biogas-based digestate with rice bran protein-rich feed ingredient for tilapia production. Biomass-Convers. Biorefinery 2020, 12, 1639–1645. [Google Scholar] [CrossRef]
- Corrêa, C.F.; Nobrega, R.O.; Block, J.M.; Fracalossi, D.M. Mixes of plant oils as fish oil substitutes for Nile tilapia at optimal and cold suboptimal temperature. Aquaculture 2018, 497, 82–90. [Google Scholar] [CrossRef]
- De Souza, E.M.; De Souza, R.C.; Melo, J.F.; Da Costa, M.M.; De Souza, S.A.; De Souza, A.M.; Copatti, C.E. Cymbopogon flexuosus essential oil as an additive improves growth, biochemical and physiological responses and survival against Aeromonas hydrophila infection in nile tilapia. An. Acad. Bras. Cienc. 2020, 92, e20190140. [Google Scholar] [CrossRef] [PubMed]
- Sakabe, R.; de Moraes, F.R.; Belo, M.A.d.A.; Pilarski, F.; de Moraes, J.R.E. Kinetics of chronic inflammation in Nile tilapia fed n-3 and n-6 essential fatty acids. Pesqui Agropecu Bras. 2013, 48, 313–319. [Google Scholar] [CrossRef]
- Limbu, S.M.; Shoko, A.P.; Lamtane, H.A.; Kishe-Machumu, M.A.; Joram, M.C.; Mbonde, A.S.; Mgana, H.F.; Mgaya, Y.D. Supplemental effects of mixed ingredients and rice bran on the growth performance, survival and yield of Nile tilapia, Oreochromis niloticus reared in fertilized earthen ponds. SpringerPlus 2016, 5, 5. [Google Scholar] [CrossRef]
- Palintorn, N.; Rujinanont, N.; Srisathaporn, A.; Gawborisut, S.; Wongkaew, P. Effects of dietary supplemented with flesh ripe fruit of local cultivated banana CV. Kluai Namwa on growth performance and meat quality of Nile tilapia. AACL Bioflux 2019, 12, 1578–1591. Available online: https://www.bioflux.com.ro/docs/2019.1578-1591.pdf (accessed on 20 February 2022).
- Kurian, A.; Van Doan, H.; Tapingkae, W.; Elumalai, P. Modulation of mucosal parameters, innate immunity, growth and resistance against Streptococcus agalactiae by enrichment of Nile tilapia (Oreochromis niloticus) diet with Leucas aspera. Fish Shellfish. Immunol. 2020, 97, 165–172. [Google Scholar] [CrossRef]
- de Rezende, R.A.E.; Soares, M.P.; Sampaio, F.G.; Cardoso, I.L.; Ishikawa, M.M.; Dallago, B.S.L.; Rantin, F.T.; Duarte, M.C.T. Phytobiotics blend as a dietary supplement for Nile tilapia health improvement. Fish Shellfish. Immunol. 2021, 114, 293–300. [Google Scholar] [CrossRef]
- Souza, R.C.; Melo, J.F.B.; Filho, R.M.N.; Campeche, D.F.B.; Figueiredo, R.A.C.R. Influencia da farinha de manga no crescimento e composição corporal da tilápia do nilo. Arch. Zootec. 2012, 62, 217–225. [Google Scholar] [CrossRef][Green Version]
- Magouz, F.I.; Mahmoud, S.A.; El-Morsy, R.A.; Paray, B.A.; Soliman, A.A.; Zaineldin, A.I.; Dawood, M.A. Dietary menthol essential oil enhanced the growth performance, digestive enzyme activity, immune-related genes, and resistance against acute ammonia exposure in Nile tilapia (Oreochromis niloticus). Aquaculture 2021, 530, 735944. [Google Scholar] [CrossRef]
- Ibrahim, R.E.; El-Houseiny, W.; Behairy, A.; Mansour, M.F.; Abd-Elhakim, Y.M. Ameliorative effects of Moringa oleifera seeds and leaves on chlorpyrifos-induced growth retardation, immune suppression, oxidative stress, and DNA damage in Oreochromis niloticus. Aquaculture 2019, 505, 225–234. [Google Scholar] [CrossRef]
- Yilmaz, S.; Ergün, S.; Yigit, M.; Yilmaz, E.; Ahmadifar, E. Dietary supplementation of black mulberry (Morus nigra) syrup improves the growth performance, innate immune response, antioxidant status, gene expression responses, and disease resistance of Nile tilapia (Oreochromis niloticus). Fish Shellfish. Immunol. 2020, 107, 211–217. [Google Scholar] [CrossRef]
- Panprommin, D.; Kaewpunnin, W.; Insee, D. Effects of Holy Basil (Ocimum sanctum) Extract on the Growth, Immune Response and Disease Resistance against Streptococcus agalactiae of Nile Tilapia (Oreochromis niloticus). Int. J. Agric. Biol. 2016, 18, 677–682. [Google Scholar] [CrossRef]
- Voss, G.B.; Sousa, V.; Rema, P.; Pintado, M.E.; Valente, L.M.P. Processed By-Products from Soy Beverage (Okara) as Sustainable Ingredients for Nile Tilapia (O. niloticus) Juveniles: Effects on Nutrient Utilization and Muscle Quality. Animals 2021, 11, 590. [Google Scholar] [CrossRef] [PubMed]
- Ismail, T.; Hegazi, E.; Nassef, E.; El-Din, M.T.S.; Dawood, M.A.; Abdo, S.E.; Gewaily, M.S. Gut immune-related gene expression, histomorphometry and hematoimmunological assays in Nile tilapia (Oreochromis niloticus) fed Aspergillus oryzae fermented olive cake. Fish Shellfish. Immunol. 2021, 117, 299–310. [Google Scholar] [CrossRef] [PubMed]
- Santo, A.H.E.; Brito, T.S.; Brandão, L.L.; Tavares, G.C.; Leibowitz, M.P.; Prado, S.A.; Ferraz, V.P.; Hoyos, D.C.M.; Turra, E.M.; Teixeira, E.A.; et al. Dietary supplementation of dry oregano leaves increases the innate immunity and resistance of Nile tilapia against Streptococcus agalactiae infection. J. World Aquac. Soc. 2020, 51, 418–436. [Google Scholar] [CrossRef]
- Freccia, A.; Sousa, S.M.d.N.; Meurer, F.; Butzge, A.J.; Mewes, J.K.; Bombardelli, R.A. Essential oils in the initial phase of broodstock diets of Nile tilapia. Rev. Bras. Zootec. 2014, 43, 1–7. [Google Scholar] [CrossRef]
- Mohammadi, G.; Rafiee, G.; El Basuini, M.F.; Abdel-Latif, H.M.; Dawood, M.A. The growth performance, antioxidant capacity, immunological responses, and the resistance against Aeromonas hydrophila in Nile tilapia (Oreochromis niloticus) fed Pistacia vera hulls derived polysaccharide. Fish Shellfish. Immunol. 2020, 106, 36–43. [Google Scholar] [CrossRef]
- Abdel-Razek, N.; Awad, S.M.; Abdel-Tawwab, M. Effect of dietary purslane (Portulaca oleracea L.) leaves powder on growth, immunostimulation, and protection of Nile tilapia, Oreochromis niloticus against Aeromonas hydrophila infection. Fish Physiol. Biochem. 2019, 45, 1907–1917. [Google Scholar] [CrossRef]
- Nascimento, A.A.; Melo, J.F.B.; de Souza, A.M.; Melo, F.V.S.T. Inclusion of mesquite pod meal (Prosopis juliflora) in diets for nile tilapia (Oreochromis niloticus) juveniles. Bol. Inst. Pesca 2019, 45, 1–9. [Google Scholar] [CrossRef]
- Silva, T.; Chung, S.; Araújo, T.; Azevedo, K.; Santos, M.; Bicudo, Á. Substituição do milho pelo farelo de algaroba (Prosopis juliflora) em dietas para juvenis de tilápia do Nilo cultivados em baixa temperatura. Rev. Bras. Cienc. Agrar. 2015, 10, 460–465. [Google Scholar] [CrossRef]
- El-Latif, A.M.A.; El-Gawad, E.A.A.; Soror, E.I.; Shourbela, R.M.; Zahran, E. Dietary supplementation with miswak (Salvadora persica) improves the health status of Nile tilapia and protects against Aeromonas hydrophila infection. Aquac. Rep. 2021, 19, 100594. [Google Scholar] [CrossRef]
- Lebda, M.A.; El-Hawarry, W.N.; Shourbela, R.M.; El-Far, A.H.; Shewita, R.S.; Mousa, S.A. Miswak (Salvadora persica) dietary supplementation improves antioxidant status and nonspecific immunity in Nile tilapia (Oreochromis niloticus). Fish Shellfish. Immunol. 2019, 88, 619–626. [Google Scholar] [CrossRef]
- Abbas, W.T.; Abumourad, I.M.; Mohamed, L.A.; Abbas, H.H.; Authman, M.; Soliman, W.S.; Elgendy, M.Y. The Role of the Dietary Supplementation of Fenugreek Seeds in Growth and Immunity in Nile Tilapia with or without Cadmium Contamination. Jordan J. Biol. Sci. 2019, 12, 649–656. [Google Scholar]
- Yunis-Aguinaga, J.; Claudiano, G.S.; Marcusso, P.F.; Manrique, W.G.; de Moraes, J.R.E.; de Moraes, F.R.; Fernandes, J.B. Uncaria tomentosa increases growth and immune activity in Oreochromis niloticus challenged with Streptococcus agalactiae. Fish Shellfish. Immunol. 2015, 47, 630–638. [Google Scholar] [CrossRef]
- Gabriel, N.N.; Qiang, J.; He, J.; Ma, X.Y.; Kpundeh, M.D.; Xu, P. Dietary Aloe vera supplementation on growth performance, some haemato-biochemical parameters and disease resistance against Streptococcus iniae in tilapia (GIFT). Fish Shellfish. Immunol. 2015, 44, 504–514. [Google Scholar] [CrossRef]
- Teoh, C.-Y.; Ng, W.-K. Evaluation of the impact of dietary petroselinic acid on the growth performance, fatty acid composition, and efficacy of long chain-polyunsaturated fatty acid biosynthesis of farmed Nile tilapia. J. Agric. Food Chem. 2013, 61, 6056–6068. [Google Scholar] [CrossRef]
- Qiang, J.; Khamis, O.A.M.; Jiang, H.J.; Cao, Z.M.; He, J.; Tao, Y.F.; Xu, P.; Bao, J.W. Effects of dietary supplementation with apple peel powder on the growth, blood and liver parameters, and transcriptome of genetically improved farmed tilapia (GIFT, Oreochromis niloticus). PLoS ONE 2019, 14, e0224995. [Google Scholar] [CrossRef]
- Brum, A.; Pereira, S.A.; Owatari, M.S.; Chagas, E.C.; Chaves, F.C.M.; Mouriño, J.L.P.; Martins, M.L. Effect of dietary essential oils of clove basil and ginger on Nile tilapia (Oreochromis niloticus) following challenge with Streptococcus agalactiae. Aquaculture 2017, 468, 235–243. [Google Scholar] [CrossRef]
- Yılmaz, S.; Ergün, S. Dietary Supplementation with allspice Pimenta dioica reduces the occurrence of streptococcal disease during first feeding of mozambique tilapia fry. J. Aquat. Anim. Health 2014, 26, 144–148. [Google Scholar] [CrossRef] [PubMed]
- Gobi, N.; Ramya, C.; Vaseeharan, B.; Malaikozhundan, B.; Vijayakumar, S.; Murugan, K.; Benelli, G. Oreochromis mossambicus diet supplementation with Psidium guajava leaf extracts enhance growth, immune, antioxidant response and resistance to Aeromonas hydrophila. Fish Shellfish. Immunol. 2016, 58, 572–583. [Google Scholar] [CrossRef] [PubMed]
- Musthafa, M.S.; Ali, A.R.J.; Kumar, M.S.A.; Paray, B.A.; Al-Sadoon, M.K.; Balasundaram, C.; Harikrishnan, R. Effect of Cucurbita mixta (L.) seed meal enrichment diet on growth, immune response and disease resistance in Oreochromis mossambicus. Fish Shellfish. Immunol. 2017, 68, 509–515. [Google Scholar] [CrossRef] [PubMed]
- Musthafa, M.S.; Asgari, S.M.; Kurian, A.; Elumalai, P.; Ali, A.R.J.; Paray, B.A.; Al-Sadoon, M.K. Protective efficacy of Mucuna pruriens (L.) seed meal enriched diet on growth performance, innate immunity, and disease resistance in Oreochromis mossambicus against Aeromonas hydrophila. Fish Shellfish. Immunol. 2018, 75, 374–380. [Google Scholar] [CrossRef]
- Yılmaz, S.; Ergün, S. Chickweed (Stellaria media) Leaf Meal as a Feed Ingredient for Tilapia (Oreochromis mossambicus). J. Appl. Aquac. 2013, 25, 329–336. [Google Scholar] [CrossRef]
- Michael, F.; Saleh, N.; Shalaby, S.; Sakr, E.; Abd-El-Khalek, D.; Elmonem, A.A. Effect of different dietary levels of commercial wood charcoal on growth, body composition and environmental loading of red tilapia hybrid. Aquac. Nutr. 2017, 23, 210–216. [Google Scholar] [CrossRef]
- Zheng, Q.; Han, C.; Zhong, Y.; Wen, R.; Zhong, M. Effects of dietary supplementation with green tea waste on growth, digestive enzyme and lipid metabolism of juvenile hybrid tilapia, Oreochromis niloticus × O. aureus. Fish Physiol. Biochem. 2017, 43, 361–371. [Google Scholar] [CrossRef]
- Sewaka, M.; Trullas, C.; Chotiko, A.; Rodkhum, C.; Chansue, N.; Boonanuntanasarn, S.; Pirarat, N. Efficacy of synbiotic Jerusalem artichoke and Lactobacillus rhamnosus GG-supplemented diets on growth performance, serum biochemical parameters, intestinal morphology, immune parameters and protection against Aeromonas veronii in juvenile red tilapia (Oreochromis spp.). Fish Shellfish. Immunol. 2019, 86, 260–268. [Google Scholar] [CrossRef]
- Botello-León, A.; Viana, M.T.; Téllez-Girón, E.; Pullés-Ariza, E.; Cisneros-Lopez, M.; Solano-Silveira, G.; Valdivie, M.; Miranda-Miranda, O.; Rodriguez-Valera, Y.; Cutiño-Espinoza, M.; et al. Fish meal substitution by protein sugar cane in diets for weight gain in red tilapia. Agrociencia 2011, 45, 23–31. Available online: https://www.researchgate.net/publication/289038710 (accessed on 12 February 2022).
- Poolsawat, L.; Yu, Y.; Li, X.; Zhen, X.; Yao, W.; Wang, P.; Luo, C.; Leng, X. Efficacy of phytogenic extracts on growth performance and health of tilapia (Oreochromis niloticus × O. aureus). Aquac. Fish. 2020, 7, 411–419. [Google Scholar] [CrossRef]
- Wachira, M.N.; Osuga, I.M.; Munguti, J.M.; Ambula, M.K.; Subramanian, S.; Tanga, C.M. Efficiency and improved profitability of insect-based aquafeeds for farming Nile tilapia fish (Oreochromis niloticus L.). Animals 2021, 11, 2599. [Google Scholar] [CrossRef] [PubMed]
- Agbohessou, P.S.; Mandiki, S.N.; Gougbédji, A.; Megido, R.C.; Lima, L.-M.W.; Cornet, V.; Lambert, J.; Purcaro, G.; Francis, F.; Lalèyè, P.A.; et al. Efficiency of fatty acid-enriched dipteran-based meal on husbandry, digestive activity and immunological responses of Nile tilapia Oreochromis niloticus juveniles. Aquaculture 2021, 545, 737193. [Google Scholar] [CrossRef]
- Alves, A.P.D.C.; Paulino, R.R.; Pereira, R.T.; Costa, D.V.; Rosa, P.V. Nile tilapia fed insect meal: Growth and innate immune response in different times under lipopolysaccharide challenge. Aquac. Res. 2021, 52, 529–540. [Google Scholar] [CrossRef]
- Amer, A.A.; El-Nabawy, E.M.; Gouda, A.H.; Dawood, M.A.O. The addition of insect meal from Spodoptera littoralis in the diets of Nile tilapia and its effect on growth rates, digestive enzyme activity and health status. Aquac. Res. 2021, 52, 5585–5594. [Google Scholar] [CrossRef]
- Perera, A.D.; Bhujel, R.C. Field cricket (Gryllus bimaculatus) meal (FCM) to replace fishmeal in the diets for sex reversal and nursing of Nile tilapia (Oreochromis niloticus) fry. Aquac. Res. 2021, 52, 4946–4958. [Google Scholar] [CrossRef]
- Abu Bakar, N.-H.; Razak, S.A.; Taufek, N.M.; Alias, Z. Evaluation of black soldier fly (Hermetia illucens) prepupae oil as meal supplementation in diets for red hybrid tilapia (Oreochromis spp.). Int. J. Trop. Insect Sci. 2021, 41, 2093–2102. [Google Scholar] [CrossRef]
- Taufek, N.; Lim, J.; Abu Bakar, N. Comparative Evaluation of Hermetia illucens Larvae Reared on Different Substrates for Red tilapia Diet: Effect on Growth and Body Composition. J. Insects Food Feed. 2021, 7, 79–88. [Google Scholar] [CrossRef]
- Tippayadara, N.; Dawood, M.A.O.; Krutmuang, P.; Hoseinifar, S.H.; Van Doan, H.; Paolucci, M. Replacement of fish meal by black soldier fly (Hermetia illucens) larvae meal: Effects on growth, haematology, and skin mucus immunity of Nile tilapia, Oreochromis niloticus. Animals 2021, 11, 193. [Google Scholar] [CrossRef]
- Yildirim-Aksoy, M.; Eljack, R.; Schrimsher, C.; Beck, B.H. Use of dietary frass from black soldier fly larvae, Hermetia illucens, in hybrid tilapia (Nile x Mozambique, Oreocromis niloticus x O. mozambique) diets improves growth and resistance to bacterial diseases. Aquac. Rep. 2020, 17, 100373. [Google Scholar] [CrossRef]
- Wu, S. The growth performance, body composition and nonspecific immunity of Tilapia (Oreochromis niloticus) affected by chitosan. Int. J. Biol. Macromol. 2020, 145, 682–685. [Google Scholar] [CrossRef]
- Toriz-Roldan, A.; Ruiz-Vega, J.; García-Ulloa, M.; Hernández-Llamas, A.; Fonseca-Madrigal, J.; Rodríguez-González, H. Assessment of Dietary Supplementation Levels of Black Soldier Fly, Hermetia illucens, Pre-pupae Meal for Juvenile Nile Tilapia, Oreochromis niloticus. Southwest. Entomol. 2019, 44, 251–259. Available online: http://www.worldbank.org/ (accessed on 3 February 2022). [CrossRef]
- Dietz, C.; Liebert, F. Does graded substitution of soy protein concentrate by an insect meal respond on growth and N-utilization in Nile tilapia (Oreochromis niloticus)? Aquac. Rep. 2018, 12, 43–48. [Google Scholar] [CrossRef]
- Devic, E.; Leschen, W.; Murray, F.; Little, D. Growth performance, feed utilization and body composition of advanced nursing Nile tilapia (Oreochromis niloticus) fed diets containing Black Soldier Fly (Hermetia illucens) larvae meal. Aquac. Nutr. 2018, 24, 416–423. [Google Scholar] [CrossRef]
- Orsi, R.O.; dos Santos, V.G.; Pezzato, L.E.; de Carvalho, P.L.; Teixeira, C.P.; Freitas, J.M.; Padovani, C.R.; Sartori, M.M.; Barros, M.M. Activity of Brazilian propolis against Aeromonas hydrophila and its effect on Nile tilapia growth, hematological and non-specific immune response under bacterial infection. An. Acad. Bras. Cienc. 2017, 89, 1785–1799. [Google Scholar] [CrossRef]
- Freccia, A.; Meurer, E.S.; Filho, J.C.; Jerônimo, G.T.; Emerenciano, M.G.C. Insect meal in tilapia fingerlings diets [Farinha de inseto em dietas de alevinos de tilápia]. Arch. Zootec. 2016, 62, 541–547. Available online: https://www.uco.es/ucopress/az/index.php/az/ (accessed on 5 February 2022).
- Sánchez-Muros, M.J.; De Haro, C.; Sanz, A.; Trenzado, C.E.; Villareces, S.; Barroso, F.G. Nutritional evaluation of Tenebrio molitor meal as fishmeal substitute for tilapia (Oreochromis niloticus) diet. Aquac. Nutr. 2016, 22, 943–955. [Google Scholar] [CrossRef]
- Sing, K.W.; Kamarudin, M.S.; Wilson, J.J.; Sofian-Azirun, M. Evaluation of Blowfly (Chrysomya megacephala) Maggot Meal as an Effective, Sustainable Replacement for Fishmeal in the Diet of Farmed Juvenile Red Tilapia (Oreochromis spp.). Pak. Vet. J. 2014, 34, 288–292. Available online: https://www.researchgate.net/publication/285988706 (accessed on 6 February 2022).
- Qin, C.; Zhang, Y.; Liu, W.; Xu, L.; Yang, Y.; Zhou, Z. Effects of chito-oligosaccharides supplementation on growth performance, intestinal cytokine expression, autochthonous gut bacteria and disease resistance in hybrid tilapia Oreochromis niloticus ♀ × Oreochromis aureus ♂. Fish Shellfish. Immunol. 2014, 40, 267–274. [Google Scholar] [CrossRef]
- Fall, J.; Ndong, D.; Thengtseng, Y.; Sheen, S.-S. The effects of different protein sources on the growth of hybrid tilapia (Oreochromis niloticus × O. aureus) reared under fresh water and brackish water. Afr. J. Agric. Res. 2011, 6, 5024–5029. [Google Scholar] [CrossRef]
- Wang, Y.; Li, J. Effects of chitosan nanoparticles on survival, growth and meat quality of tilapia, Oreochromis nilotica. Nanotoxicology 2011, 5, 425–431. [Google Scholar] [CrossRef]
- El-Asely, A.M.; Abbass, A.A.; Austin, B. Honey bee pollen improves growth, immunity and protection of Nile tilapia (Oreochromis niloticus) against infection with Aeromonas hydrophila. Fish Shellfish. Immunol. 2014, 40, 500–506. [Google Scholar] [CrossRef]
- Fadl, S.E.; El-Habashi, N.; Gad, D.M.; Elkassas, W.M.; Elbialy, Z.I.; Abdelhady, D.H.; Hegazi, S.M. Effect of adding Dunaliella algae to fish diet on lead acetate toxicity and gene expression in the liver of Nile tilapia. Toxin Rev. 2021, 40, 1155–1171. [Google Scholar] [CrossRef]
- Mansour, A.T.; Alsaqufi, A.S.; Alkhamis, Y.A.; Al-Gazar, F.F.; Zaki, M.A.; Nour, A.A.M.; Ramadan, K.M.A. The evaluation of Arthrospira platensis bioactivity and their dietary supplementation to Nile tilapia vegetarian diet on growth performance, feed utilization, body composition and hemato-biochemical parameters. Ann. Anim. Sci. 2021, 21, 1061–1080. [Google Scholar] [CrossRef]
- Abdelghany, M.F.; El-Sawy, H.B.; El-Hameed, S.A.A.; Khames, M.K.; Abdel-Latif, H.M.; Naiel, M.A. Effects of dietary Nannochloropsis oculata on growth performance, serum biochemical parameters, immune responses, and resistance against Aeromonas veronii challenge in Nile tilapia (Oreochromis niloticus). Fish Shellfish. Immunol. 2020, 107, 277–288. [Google Scholar] [CrossRef] [PubMed]
- Elabd, H.; Wang, H.-P.; Shaheen, A.; Matter, A. Nano spirulina dietary supplementation augments growth, antioxidative and immunological reactions, digestion, and protection of Nile tilapia, Oreochromis niloticus, against Aeromonas veronii and some physical stressors. Fish Physiol. Biochem. 2020, 46, 2143–2155. [Google Scholar] [CrossRef]
- Arisa, I.; Zulfikar, Z.; Muhammadar, M.; Nurfadillah, N.; Mellisa, S. Study on the addition of Caulerpa lentillifera on growth and survival rate of saline tilapia Oreochromis niloticus, L. IOP Conf. Ser. Earth Environ. Sci. 2020, 493, 012004. [Google Scholar] [CrossRef]
- Mahmoud, E.A.; El-Sayed, B.M.; Mahsoub, Y.H.; El-Murr, A.E.I.; Neamat-Allah, A.N. Effect of Chlorella vulgaris enriched diet on growth performance, hemato-immunological responses, antioxidant and transcriptomics profile disorders caused by deltamethrin toxicity in Nile tilapia (Oreochromis niloticus). Fish Shellfish. Immunol. 2020, 102, 422–429. [Google Scholar] [CrossRef]
- dos Santos, S.K.A.; Schorer, M.; Moura, G.d.S.; Lanna, E.A.T.; Pedreira, M.M. Evaluation of growth and fatty acid profile of Nile tilapia (Oreochromis niloticus) fed with Schizochytrium sp. Aquac. Res. 2019, 50, 1068–1074. [Google Scholar] [CrossRef]
- Li, Z.; Yang, H.; Shangguan, J.; Chen, Q.; Li, W.; Lu, J. Growth performance, digestive enzyme activities and serum nonspecific immunity of the red tilapia (Oreochromis mossambicus × Oreochromis niloticus) fed diets supplemented with ultrafine powder of Enteromopha prolifera. J. Oceanol. Limnol. 2018, 36, 1843–1850. [Google Scholar] [CrossRef]
- Gbadamosi, O.K.; Lupatsch, I. Effects of dietary Nannochloropsis salina on the nutritional performance and fatty acid profile of Nile tilapia, Oreochromis niloticus. Algal Res. 2018, 33, 48–54. [Google Scholar] [CrossRef]
- Younis, E.-S.M.; Al-Quffail, A.S.; Al-Asgah, N.A.; Abdel-Warith, A.-W.A.; Al-Hafedh, Y.S. Effect of dietary fish meal replacement by red algae, Gracilaria arcuata, on growth performance and body composition of Nile tilapia Oreochromis niloticus. Saudi J. Biol. Sci. 2018, 25, 198–203. [Google Scholar] [CrossRef] [PubMed]
- Quezada-Rodríguez, P.d.R.; Fajer-Ávila, E.J. The dietary effect of ulvan from Ulva clathrata on hematological-immunological parameters and growth of tilapia (Oreochromis niloticus). J. Appl. Phycol. 2017, 29, 423–431. [Google Scholar] [CrossRef]
- Sarker, P.K.; Kapuscinski, A.R.; Lanois, A.J.; Livesey, E.D.; Bernhard, K.P.; Coley, M.L. Towards Sustainable Aquafeeds: Complete Substitution of Fish Oil with Marine Microalga Schizochytrium sp. Improves Growth and Fatty Acid Deposition in Juvenile Nile Tilapia (Oreochromis niloticus). PLoS ONE 2016, 11, e0156684. [Google Scholar] [CrossRef]
- El-Sheekh, M.; El-Shourbagy, I.; Shalaby, S.; Hosny, S. Effect of feeding Arthrospira platensis (Spirulina) on growth and carcass composition of hybrid red tilapia (Oreochromis niloticus x Oreochromis mossambicus). Turk. J. Fish Aquat. Sci. 2014, 14, 471–478. [Google Scholar] [CrossRef] [PubMed]
- Saleh, N.E.; Shalaby, S.M.; Sakr, E.M.; Elmonem, A.I.A.; Michael, F.R. Effect of Dietary Inclusion of Ulva fasciata on Red Hybrid Tilapia Growth and Carcass Composition. J. Appl. Aquac. 2014, 26, 197–207. [Google Scholar] [CrossRef]
- Costa, M.M.; Oliveira, S.T.L.; Balen, R.E.; Bueno Junior, G.; Baldan, L.T.; Silva, L.C.R.; Santos, L.D. Brown seaweed meal to nile tilapia fingerlings [alga marinha marrom para alevinos de tilápia do nilo]. Arch. Zootec. 2013, 62, 101–109. [Google Scholar] [CrossRef]
- Gomes, I.G.; Chaves, F.H.; Barros, R.N.; Moreira, R.L.; Teixeira, E.G.; Moreira, A.G.; Farias, W.R. Dietary supplementation with Spirulina platensis increases growth and color of red tilapia [Efecto de la suplementación dietaria con Spirulina platensis en el crecimiento y la coloración de la tilapia roja]. Rev. Colomb. Cienc. Pecu. 2012, 25, 462–471. [Google Scholar]
- Yossa, R.; Greiling, A.M.; Basiita, R.K.; Sakala, M.E.; Baumgartner, W.A.; Taylor, A.; Gatlin, D.M. Replacing fishmeal with a single cell protein feedstuff in Nile tilapia Oreochromis niloticus diets. Anim. Feed. Sci. Technol. 2021, 281, 115089. [Google Scholar] [CrossRef]
- Ching, J.J.; Shuib, A.S.; Abdullah, N.; Majid, N.A.; Taufek, N.M.; Sutra, J.; Azmai, M.N.A. Hot water extract of Pleurotus pulmonarius stalk waste enhances innate immune response and immune-related gene expression in red hybrid tilapia Oreochromis spp. following challenge with pathogen-associated molecular patterns. Fish Shellfish. Immunol. 2021, 116, 61–73. [Google Scholar] [CrossRef]
- Selim, K.M.; Reda, R.M.; Mahmoud, R.; El-Araby, I.E. Effects of nucleotides supplemented diets on growth performance and expressions of ghrelin and insulin-like growth factor genes in Nile tilapia, Oreochromis niloticus. J. Appl. Aquac. 2020, 32, 157–174. [Google Scholar] [CrossRef]
- Divya, M.; Gopi, N.; Iswarya, A.; Govindarajan, M.; Alharbi, N.S.; Kadaikunnan, S.; Khaled, J.M.; Almanaa, T.N.; Vaseeharan, B. β-glucan extracted from eukaryotic single-celled microorganism Saccharomyces cerevisiae: Dietary supplementation and enhanced ammonia stress tolerance on Oreochromis mossambicus. Microb. Pathog. 2020, 139, 103917. [Google Scholar] [CrossRef]
- Chen, X.-Q.; Zhao, W.; Xie, S.-W.; Xie, J.-J.; Zhang, Z.-H.; Tian, L.-X.; Liu, Y.-J.; Niu, J. Effects of dietary hydrolyzed yeast (Rhodotorula mucilaginosa) on growth performance, immune response, antioxidant capacity and histomorphology of juvenile Nile tilapia (Oreochromis niloticus). Fish Shellfish. Immunol. 2019, 90, 30–39. [Google Scholar] [CrossRef]
- Abass, D.A.; Obirikorang, K.A.; Campion, B.B.; Edziyie, R.E.; Skov, P.V. Dietary supplementation of yeast (Saccharomyces cerevisiae) improves growth, stress tolerance, and disease resistance in juvenile Nile tilapia (Oreochromis niloticus). Aquac. Int. 2018, 26, 843–855. [Google Scholar] [CrossRef]
- Assem, H.; Khalifa, A.; Elsalhia, M. Physiological and microbiological indices as indicators of evaluating dietary fungi degraded date pits as a probiotic for cultured Nile tilapia Oreochromis niloticus fingerling and its effect on fish welfare. Egypt. J. Aquat. Res. 2014, 40, 435–441. [Google Scholar] [CrossRef]
- Salvador, R.; Claudiano, G.d.S.; Loureiro, B.A.; Marcusso, P.F.; Eto, S.F.; Pilarski, F.; Toazza, C.S.; de Moraes, J.R.E.; de Moraes, F.R. Desempenho e hematologia de tilápias-do-nilo alimentadas com Saccharomyces cerevisiae e vacinadas contra Streptococcus agalactiae. Pesquisa Agropecuária Brasileira 2013, 48, 892–898. [Google Scholar] [CrossRef]
- de Sousa Liranço, A.; Ciarlini, P.C.; Moraes, G.; Camargo, A.L.S.; Romagosa, E. Mannanoligosaccharide (mos) and ß-glucan (ß-glu) in Dietary Supplementation for Nile Tilapia Juveniles Dept in Cages. 2013. Available online: www.cobea.org.br (accessed on 2 February 2022).
- Abd Rahman, J.M.D.; Razak, S.A.; Sabaratnam, V. Effect of Mushroom Supplementation as a Prebiotic Compound in Super Worm Based Diet on Growth Performance of Red Tilapia Fingerlings. Sains Malays 2012, 41, 1197–1203. Available online: https://www.researchgate.net/publication/265042127 (accessed on 25 January 2022).
- Vechklang, K.; Lim, C.; Boonanuntanasarn, S.; Welker, T.; Ponchunchuwong, S.; Klesius, P.H.; Wanapu, C. Growth Performance and Resistance to Streptococcus iniae of Juvenile Nile Tilapia (Oreochromis niloticus) Fed Diets Supplemented with GroBiotic-A and Brewtech Dried Brewers Yeast. J. Appl. Aquac. 2012, 24, 183–198. [Google Scholar] [CrossRef]
- Koch, J.F.A.; Pezzato, L.E.; Barros, M.M.; Teixeira, C.P.; Junior, A.C.F.; Padovani, C.R. Levedura como pronutriente em dietas para matrizes e alevinos de tilápia-do-nilo. Rev. Bras. Zootec. 2011, 40, 2281–2289. [Google Scholar] [CrossRef]
- Nobrega, R.O.; Batista, R.O.; Corrêa, C.F.; Mattioni, B.; Filer, K.; Pettigrew, J.E.; Fracalossi, D.M. Dietary supplementation of Aurantiochytrium sp. meal, a docosahexaenoic-acid source, promotes growth of Nile tilapia at a suboptimal low temperature. Aquaculture 2019, 507, 500–509. [Google Scholar] [CrossRef]
- Abu-Elala, N.M.; Younis, N.A.; AbuBakr, H.O.; Ragaa, N.M.; Borges, L.L.; Bonato, M.A. Efficacy of dietary yeast cell wall supplementation on the nutrition and immune response of Nile tilapia. Egypt. J. Aquat. Res. 2018, 44, 333–341. [Google Scholar] [CrossRef]
- Tongmee, B.; Tongsiri, S.; Unpaprom, Y.; Ramaraj, R.; Whangchai, K.; Pugazhendhi, A.; Whangchai, N. Sustainable development of feed formulation for farmed tilapia enriched with fermented pig manure to reduce production costs. Sci. Total. Environ. 2021, 801, 149614. [Google Scholar] [CrossRef]
- Carvalho, K.V.; Luczinski, T.G.; Boscolo, W.R.; de Freitas, J.M.A.; Signor, A. Poultry byproducts and swine liver used in diets for Nile tilapia juveniles. Lat. Am. J. Aquat. Res. 2020, 48, 895–900. [Google Scholar] [CrossRef]
- Obirikorang, K.A.; Mensah, N.E.; Asiamah, E.A. Growth, feed utilization, and liver histology of juvenile Nile tilapia (Oreochromis niloticus) fed diets containing increasing levels of swine fat. J. Appl. Aquac. 2018, 30, 366–381. [Google Scholar] [CrossRef]
- Sary, C.; de Paris, L.D.; Bernardi, D.M.; Lewandowiski, V.; Signor, A.; Boscolo, W.R. Tilapia by-product hydrolysate powder in diets for Nile tilapia larvae. Acta Sci. Anim. Sci. 2017, 39, 1–6. [Google Scholar] [CrossRef][Green Version]
- Abimorad, E.G.; Castellani, D.; Gonçalves, G.S.; Romera, D.M.; Garcia, F.; Nascimento, T.M.T.D. Substituição parcial do farelo de soja pela farinha de carne e ossos em dietas para juvenis de tilápia-do-nilo. Pesqui Agropecu Bras. 2014, 49, 836–843. [Google Scholar] [CrossRef]
- Silva, T.R.M.; de Andrade, M.L.S.; Chung, S.; de Almeida Bicudo, Á.J. Substituição parcial do milho pelo resíduo de macarrão em dietas para tilápia-do-nilo [partial replacement of corn by pasta waste in diets for nile tilapia]. Bol. Inst. Pesca Sao Paulo 2014, 40, 669–676. [Google Scholar]
- Petenuci, M.E.; Schneider, V.V.; Lopes, A.P.; Gonçalves, R.M.; Dos Santos, V.J.; Matsushita, M.; Visentainer, J.V. Effect of Alpha-Linolenic Acid Sources in Diets for Nile Tilapia on Fatty Acid Composition of Fish Fillet Using Principal Component Analysis. J. Aquat. Food Prod. Technol. 2018, 27, 464–476. [Google Scholar] [CrossRef]
- Carbonera, F.; Bonafe, E.G.; Martin, C.A.; Montanher, P.F.; Ribeiro, R.P.; Figueiredo, L.C.; Almeida, V.C.; Visentainer, J.V. Effect of dietary replacement of sunflower oil with perilla oil on the absolute fatty acid composition in Nile tilapia (GIFT). Food Chem. 2014, 148, 230–234. [Google Scholar] [CrossRef]
- Visentainer, J.V.; França, P.B.; Aguiar, A.C.; Montanher, P.F.; Boroski, M.; de Souza, N.E. Incorporation and fatty acid composition in liver of Nile tilapia fed with flaxseed oil. Acta Sci. Technol. 2011, 33, 221–225. [Google Scholar] [CrossRef][Green Version]
- FAO. Fats and Fatty Acids in Human Nutrition: Report of an Expert Consultation: 10–14 November 2008; FAO: Geneva, Switzerland, 2010. [Google Scholar]
- dos Santos, L.D.; Sousa, S.M.d.N.; da Silva, L.C.R.; Bombardelli, R.A.; Meurer, F. Efeitos da peletização e extrusão sobre a digestibilidade de ingredientes alternativos do Semi-árido Nordestino para a tilápia do Nilo. Semin. Cienc. Agrar. 2014, 35, 3367. [Google Scholar] [CrossRef][Green Version]
- Damasceno, F.M.; Pezzato, L.E. Nutritional value of root and leaves of cassava for Nile tilapia. Bol. Inst. Pesca Sao Paulo 2012, 38, 61–69. Available online: https://www.researchgate.net/publication/285958520 (accessed on 17 February 2022).
- Pereira, R.; Valente, L.M.P.; Sousa-Pinto, I.; Rema, P. Apparent nutrient digestibility of seaweeds by rainbow trout (Oncorhynchus mykiss) and Nile tilapia (Oreochromis niloticus). Algal Res. 2012, 1, 77–82. [Google Scholar] [CrossRef]
- Barone, R.S.C.; Sonoda, D.Y.; Lorenz, E.K.; Cyrino, J.E.P. Digestibility and pricing of Chlorella sorokiniana meal for use in tilapia feeds. Sci. Agricola 2018, 75, 184–190. [Google Scholar] [CrossRef]
- dos Santos Cardoso, M.; Godoy, A.C.; Oxford, J.H.; Rodrigues, R.; Cardoso, M.d.S.; Bittencourt, F.; Signor, A.; Boscolo, W.R.; Feiden, A. Apparent digestibility of protein hydrolysates from chicken and swine slaughter residues for Nile tilapia. Aquaculture 2021, 530, 735720. [Google Scholar] [CrossRef]
- Fontes, T.V.; de Oliveira, K.R.B.; Gomes Almeida, I.L.; Maria Orlando, T.; Rodrigues, P.B.; Costa, D.V.D. Digestibility of Insect Meals for Nile Tilapia Fingerlings. Animals 2019, 9, 181. [Google Scholar] [CrossRef] [PubMed]
- Sirmah, P.; Mburu, F.; Iaych, K.; Dumarçay, S.; Gérardin, P. Potential antioxidant compounds from different parts of Prosopis juliflora. J. Trop. For. Sci. 2011, 23, 187–195. Available online: https://www.frim.gov.my/v1/JTFSOnline/jtfs/v23n2/187-195.pdf (accessed on 7 February 2022).
- Raja, K.; Saravanakumar, A.; Vijayakumar, R. Efficient synthesis of silver nanoparticles from Prosopis juliflora leaf extract and its antimicrobial activity using sewage. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2012, 97, 490–494. [Google Scholar] [CrossRef] [PubMed]
- Thakur, R.; Singh, R.; Saxena, P.; Mani, A. Evaluation of Antibacterial Activity of Prosopis juliflora (Sw.) Dc. Leaves. Afr. J. Tradit. Complement. Altern. Med. AJTCAM 2014, 11, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Seong, W.L.; Wee, W. Malaysian rubber (Hevea brasiliensis) seed as alternative protein source for red hybrid tilapia, Oreochromis spp., farming. AACL Bioflux 2017, 10, 32–37. Available online: https://www.bioflux.com.ro/docs/2017.32-37.pdf. (accessed on 24 February 2022).

| Fish Species | Stage | Plant Species | Raw Material Processing Method | WG | SGR | FCR | Reference |
|---|---|---|---|---|---|---|---|
| n | F | Allium cepa L. | Meal | = | = | = | [8] |
| n | F | Allium sativum L. | Meal | = | = | = | [8] |
| n | F | Arachis hypogaea L. | Cake, Husk | = | = | = | [9] |
| n | F | Azadirachta indica A. Juss. | Seed extract | = | = | = | [10] |
| n | F | Camellia sinensis L. (30 days) | Extract | =↑ | ↑ | ↑ | [11] |
| n | F | C. sinensis (60 days) | Extract | ↑ | =↑ | ↑ | [11] |
| n | F | Carica papaya L. | Seed extract | ↑ | ↑ | ↑ | [10] |
| n | F | Caryocar brasiliense Cambess. | Peel, Meal | = | - | = | [12] |
| n | F | Cinnamomum camphora L. | Extract, Seed | ↑ | ↑ | ↑ | [10] |
| n | F | Cinnamomum verum J. Presl | Nanoparticles | ↑ | ↑ | = | [13] |
| n | F | Citrus × sinensis (L.) Osbeck | Extract | ↑ | = | = | [14] |
| n | F | Citrus bergamia Risso & Poit. | Oil | =↑ | =↑ | =↑ | [15] |
| n | F | C. nucifera | Oil | =↑ | =↑ | =↑ | [16] |
| n | F | Cucurbita moschata Duchesne | Seed meal | ↑ | ↑ | - | [17] |
| n | F | Curcuma longa L. | Hydrolate | = | = | = | [18] |
| n | F | C. longa | Curcumin | ↑ | ↑ | ↑ | [19] |
| n | F | Cymbopogon citratus (DC.) Stapf | Essential oil | ↑ | ↑ | ↑ | [20] |
| n | F | Elephantopus scaber L. (30 days) | Extract | ↑ | ↑ | ↑ | [21] |
| n | F | E. scaber (60 days) | Extract | =↑ | =↑ | =↑ | [21] |
| n | F | Euphorbia hirta L. | Seed extract | ↑ | ↑ | ↑ | [10] |
| n | F | H. annuus | Seed meal | ↑ | ↑ | - | [17] |
| n | F | Houttuynia cordata Thunb. | Meal | = | = | = | [22] |
| n | F | Ipomoea batatas L. | Extract, Meal | =↑ | =↑ | =↑ | [23] |
| n | F | L. usitatissimum | Seed extract | ↑ | = | = | [14] |
| n | F | Lippia origanoides Kunth | Essential oil | = | - | = | [24] |
| n | F | Lycium barbarum L. | Meal | =↑ | =↑ | - | [25] |
| n | F | Lycopersicon esculentum Mill. | By-product, Meal | ↓ | ↓ | - | [26] |
| n | F | Mangifera indica L. | Meal | ↓= | ↓= | - | [27] |
| n | F | M. oleifera | Meal | ↑ | ↑ | ↑ | [28] |
| n | F | M. oleifera | Leaves | - | ↑ | = | [29] |
| n | F | Pelargonium graveolens L’Hér. ex Aiton | Extract | =↑ | =↑ | =↑ | [20] |
| n | F | Phoenix dactylifera L. | By-product, Meal | ↓ | ↓ | - | [26] |
| n | F | Pisum sativum L. | By-product, Meal | ↓ | ↓ | - | [26] |
| n | F | Rhizoclonium riparium var. implexum (Dillwyn) Rosenvinge | Meal | =↑ | = | ↑ | [30] |
| n | F | S. persica | Derived | =↑ | =↑ | = | [31] |
| n | F | S. persica | Meal | ↑ | =↑ | =↑ | [32] |
| n | F | Theobroma cacao L. | Meal, Husk | ↓=↑ | - | =↑ | [33] |
| n | F | Voandzeia subterránea (L.) DC. | By-product, Meal | =↑ | - | =↑ | [34] |
| n | J | A. membranaceus | Polysaccharides | ↑ | ↑ | ↑ | [35] |
| n | J | Cenchrus purpureus (Schumach.) Morrone | By-product, Digestate meal | = | - | = | [36] |
| n | J | C. nucifera (28 °C) | Oil | ↓ | - | - | [37] |
| n | J | C. nucifera (22 °C) | Oil | ↓= | - | - | [37] |
| n | J | Cymbopogon flexuosus (Nees) Will. Watson | Essential oil | = | = | = | [38] |
| n | J | Glycine max (L.) Merr. | Degummed soybean Oil | =↑ | = | = | [39] |
| n | J | H. annuus (28 °C) | Oil | ↓ | - | - | [37] |
| n | J | H. annuus (22 °C) | Oil | ↓= | - | - | [37] |
| n | J | H. annuus | Seed, Cake | = | = | - | [40] |
| n | J | Musa ABB cv. Kluai “Namwa” | Fresh | ↑ | ↑ | ↑ | [41] |
| n | J | Leucas aspera (Willd.) Link | Meal | ↓=↑ | ↓=↑ | ↓=↑ | [42] |
| n | J | L. usitatissimum | Oil | =↑ | = | = | [39] |
| n | J | L. usitatissimum (28 °C) | Oil | ↓ | - | - | [37] |
| n | J | L. usitatissimum (22 °C) | Oil | ↓= | - | - | [37] |
| n | J | Lippia sidoides Cham. | Essential oil | = | = | = | [43] |
| n | J | M. indica | Meal | ↓= | ↓= | ↓= | [44] |
| n | J | Mentha arvensis L. | Essential oil | ↑ | ↑ | =↑ | [45] |
| n | J | M. oleifera | Leaves | = | = | - | [46] |
| n | J | M. oleifera | Seed | ↑ | ↑ | - | [46] |
| n | J | Morus nigra | Syrup | =↑ | =↑ | =↑ | [47] |
| n | J | Ocimum sanctum L. | Extract | - | = | = | [48] |
| n | J | G. max | By-product: Okara | = | = | = | [49] |
| n | J | O. europaea | Cake | ↑ | ↑ | =↑ | [50] |
| n | J | O. europaea (28 °C) | Oil | ↓ | - | - | [37] |
| n | J | O. europaea (22 °C) | Oil | ↓= | - | - | [37] |
| n | J | Origanum vulgare L. | Dry leaves | = | = | = | [51] |
| n | J | Oryza sativa L. | Seed; Cake | = | = | - | [40] |
| n | J | Phragmites spp. | By-product, Digestate meal | = | - | = | [36] |
| n | J | Piper nigrum L. | Essential oil | = | = | = | [52] |
| n | J | Pistacia vera L. | Extract, Seed | =↑ | =↑ | =↑ | [53] |
| n | J | Portulaca oleracea L. | Leaves | ↓= | ↓= | = | [54] |
| n | J | Prosopis juliflora (Sw.) DC. | Meal | = | - | = | [55] |
| n | J | P. juliflora | Meal | = | = | - | [56] |
| n | J | S. persica | Meal | = | = | - | [57] |
| n | J | S. persica | Meal | = | = | = | [58] |
| n | J | Thymus vulgaris L. | Essential oil | = | = | = | [43] |
| n | J | Thymus zygis L. | Essential oil | = | = | = | [43] |
| n | J | Trigonella foenum-graecum L. | Seed | =↑ | = | - | [59] |
| n | J | T. foenum-graecum | Seed, Extract | =↑ | =↑ | - | [59] |
| n | J | Uncaria tomentosa (Willd.) DC. | Extract | =↑ | - | =↑ | [60] |
| GIFT | F | Aloe vera L. | Meal | =↑ | =↑ | =↑ | [61] |
| GIFT | F | Brassica napus L. | Oil | = | = | = | [62] |
| GIFT | F | Coriandrum sativum L. | Seed oil | = | = | = | [62] |
| GIFT | F | H. annuus | Oil | = | = | = | [62] |
| GIFT | F | L. usitatissimum | Oil | = | = | = | [62] |
| GIFT | F | Malus domestica Borkh. | By-product, Meal | ↓=↑ | ↓=↑ | =↑ | [63] |
| GIFT | F | Ocimum gratissimum L. | Essential oil | ↓=↑ | =↑ | = | [64] |
| GIFT | F | Zingiber officinale Roscoe | Essential oil | ↓= | ↓= | ↓= | [64] |
| m | F | Pimenta dioica (L.) Merr. | Seed meal | =↑ | =↑ | =↑ | [65] |
| m | F | Psidium guajava L. | Leaves, Extract | =↑ | = | = | [66] |
| m | J | Cucurbita mixta Pangalo | Seed, Meal | =↑ | =↑ | = | [67] |
| m | J | Mucuna pruriens (L.) DC. | Seed, Meal | ↑ | ↑ | =↑ | [68] |
| m | J | Stellaria media L. | Meal | ↓= | ↓= | ↓= | [69] |
| n × m | F | Unknown | Charcoal powder | =↑ | =↑ | =↑ | [70] |
| n × m | J | C. sinensis | By-product | = | = | =↑ | [71] |
| n × m | J | Helianthus tuberosus L. | Extract | ↑ | ↑ | ↑ | [72] |
| n × m | J | Saccharum officinarum L. | By-product, Meal | ↓= | - | ↓= | [73] |
| n × a | J | Mixture of A. memembbranaceus, Codonopsis pilosula (Franch.) Nannf., Eucommia ulmoides Oliv., Lonicera japónica Thunb. | Extract | =↑ | =↑ | =↑ | [74] |
| Fish Species | Stage | Raw Material Origin | Raw Material Processing Method | WG | SGR | FCR | Reference |
|---|---|---|---|---|---|---|---|
| Animal | |||||||
| n | F | H. illucens | Meal | ↓=↑ | - | = | [75] |
| n | J | H. illucens | Meal | ↓ | ↓ | - | [76] |
| n | J | Chrysomya putoria Wiedemann | Meal | ↓= | ↓= | - | [76] |
| n | J | Zophobas morio Fabricius | Meal | = | = | - | [77] |
| n | J | Spodoptera littoralis | Meal | ↓= | ↓= | ↓= | [78] |
| n | F | Gryllus bimaculatus | Meal | =↑ | = | =↑ | [79] |
| n × m | J | H. illucens | Oil | ↓=↑ | = | = | [80] |
| n × m | J | H. illucens | Meal | = | = | =↑ | [81] |
| n | J | H. illucens | Meal | = | = | = | [82] |
| n × m | F | H. illucens | Excrement | =↑ | - | - | [83] |
| n | J | Penaeus monodon | Shells | =↑ | =↑ | =↑ | [84] |
| n | F | H. illucens | Meal | = | = | = | [85] |
| n | J | H. illucens | Meal | = | = | = | [86] |
| n | F | H. illucens | Meal | = | = | = | [87] |
| n | J | Apis mellifera L. | Propolis | ↓= | ↓= | = | [88] |
| n | F | Cinereous cockroach | Meal | = | = | = | [89] |
| n | J | Tenebrio molitor | Meal | ↓ | - | - | [90] |
| n × m | J | Chrysomya megacephala | Meal | =↑ | =↑ | = | [91] |
| n × m | F | P. vannamei | Shells | = | - | = | [92] |
| n × m | F | P. vannamei | Meal | ↓= | - | = | [93] |
| n | - | P. vannamei | Shells | =↑ | - | = | [94] |
| n | J | A. mellifera | Pollen | ↑ | ↑ | = | [95] |
| Algae | |||||||
| n | - | Dunaliella | Fresh | ↓↑ | - | ↓= | [96] |
| n | F | Arthrospira platensis | Meal | =↑ | =↑ | =↑ | [97] |
| n | J | Nannochloropsis oculata | Meal | =↑ | =↑ | =↑ | [98] |
| n | J | A. platensis | Nanoparticles | ↑ | ↑ | ↑ | [99] |
| n | F | Caulerpa lentillifera | Meal | =↑ | =↑ | - | [100] |
| n | J | Chlorella vulgaris | Meal | ↓=↑ | ↓=↑ | ↓=↑ | [101] |
| n | J | Schizochytrium sp. | Meal | =↑ | - | = | [102] |
| n × m | F | Enteromopha prolifera | Meal | ↑ | =↑ | =↑ | [103] |
| n | F | Nannochloropsis salina | Meal | ↓ | ↓ | ↓ | [104] |
| n | - | Gracilaria arcuata | Meal | ↓ | ↓ | ↓= | [105] |
| n | J | Ulva clathrata | Extract | = | = | = | [106] |
| n | J | Schizochytrium sp. | Dried whole cells | =↑ | = | = | [107] |
| n × m | F | A. platensis | Meal | =↑ | =↑ | =↑ | [108] |
| n × m | F | Ulva fasciata | Meal | ↑ | ↑ | ↑ | [109] |
| n | F | Ascophyllum nodosum | Meal | = | - | - | [110] |
| n × m | F | A. platensis | Meal | ↓=↑ | =↑ | - | [111] |
| Fungi | |||||||
| n | J | S. cerevisiae | Inactivated dry | = | - | = | [112] |
| n | J | Aspergillus oryzae | Cake | ↑ | ↑ | =↑ | [50] |
| n × m | F | Pleurotus pulmonarius | Extract | = | = | ↓ | [113] |
| n | J | S. cerevisiae | Extract | ↑ | ↑ | ↑ | [114] |
| m | - | S. cerevisiae | Extract | ↑ | =↑ | =↑ | [115] |
| n | J | Rhodotorula mucilaginosa | Extract | = | =↑ | =↑ | [116] |
| n | J | S. cerevisiae | Meal | ↑ | ↑ | ↑ | [117] |
| n | F | Trichoderma reesei | Fungi-degraded seed | =↑ | - | - | [118] |
| n | - | S. cerevisiae | Meal | ↑ | ↑ | - | [119] |
| n | J | S. cerevisiae | Derived | = | = | = | [120] |
| n × m | F | Pleurotus sajorcaju | Meal | = | = | = | [121] |
| n | J | S. cerevisiae | Dried | = | - | - | [122] |
| GIFT | F | S. cerevisiae | Whole | - | - | ↓ | [123] |
| GIFT | J | Aurantiochytrium sp. | Meal | = | = | - | [124] |
| n | - | S. cerevisiae | Cell wall | ↑ | ↑ | ↑ | [125] |
| By-products | |||||||
| n | - | Swine | Excrement | = | - | = | [126] |
| n | J | Poultry | Slaughter residues, Meal | = | = | - | [127] |
| n | J | Swine | Slaughter residues, Meal | = | = | - | [127] |
| n | J | Swine | Fat | ↓= | ↓= | ↓= | [128] |
| n | F | Oreochromis spp. | By-product, Hydrolysate | ↓= | - | - | [129] |
| n | J | Meat | Slaughter residues, Meal | =↑ | - | - | [130] |
| n | J | Bone | Slaughter residues, Meal | =↑ | - | - | [130] |
| n | J | Swine | Digestate meal | = | - | = | [36] |
| n | J | Cow | Digestate meal | = | - | = | [36] |
| n | F | Feathers poultry | Meal | ↓ | ↓ | - | [26] |
| n | J | Pasta | Waste | = | - | - | [131] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zuluaga-Hernández, C.D.; Hincapié, C.A.; Osorio, M. Non-Conventional Ingredients for Tilapia (Oreochromis spp.) Feed: A Systematic Review. Fishes 2023, 8, 556. https://doi.org/10.3390/fishes8110556
Zuluaga-Hernández CD, Hincapié CA, Osorio M. Non-Conventional Ingredients for Tilapia (Oreochromis spp.) Feed: A Systematic Review. Fishes. 2023; 8(11):556. https://doi.org/10.3390/fishes8110556
Chicago/Turabian StyleZuluaga-Hernández, Christian David, Carlos A. Hincapié, and Marisol Osorio. 2023. "Non-Conventional Ingredients for Tilapia (Oreochromis spp.) Feed: A Systematic Review" Fishes 8, no. 11: 556. https://doi.org/10.3390/fishes8110556
APA StyleZuluaga-Hernández, C. D., Hincapié, C. A., & Osorio, M. (2023). Non-Conventional Ingredients for Tilapia (Oreochromis spp.) Feed: A Systematic Review. Fishes, 8(11), 556. https://doi.org/10.3390/fishes8110556







