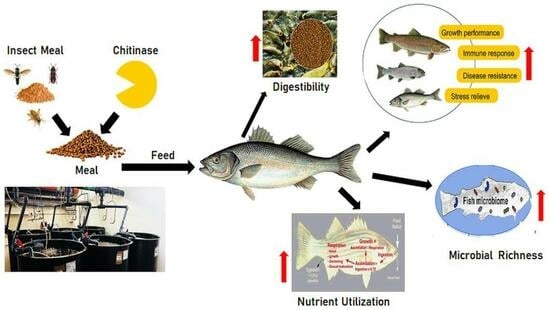
Chitinase and Insect Meal in Aquaculture Nutrition: A Comprehensive Overview of the Latest Achievements
Abstract
1. Introduction
2. Is Chitin Really a Problem in Aquaculture?
3. Chitinases from Insects and Microbial Organisms
4. Fungal Chitinases: A Key to Improved Digestibility
5. Enhanced Nutrient Utilization with Chitinase
6. Role of Chitinase in Disease Prevention and Immunity
7. Inclusion of Chitin and Chitinase in Aquaculture Feed
8. Latest Technological Advances in Chitinase Production for Aquaculture
9. Challenges in Using Chitinase in Aquaculture
10. Conclusions and Future Perspectives
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nogales-Mérida, S.; Gobbi, P.; Józefiak, D.; Mazurkiewicz, J.; Dudek, K.; Rawski, M.; Kierończyk, B.; Józefiak, A. Insect Meals in Fish Nutrition. Rev. Aquac. 2019, 11, 1080–1103. [Google Scholar] [CrossRef]
- Hasan, I.; Rimoldi, S.; Saroglia, G.; Terova, G. Sustainable Fish Feeds with Insects and Probiotics Positively Affect Freshwater and Marine Fish Gut Microbiota. Animals 2023, 13, 1633. [Google Scholar] [CrossRef] [PubMed]
- Hua, K.; Cobcroft, J.M.; Cole, A.; Condon, K.; Jerry, D.R.; Mangott, A.; Praeger, C.; Vucko, M.J.; Zeng, C.; Zenger, K.; et al. The Future of Aquatic Protein: Implications for Protein Sources in Aquaculture Diets. One Earth 2019, 1, 316–329. [Google Scholar] [CrossRef]
- Gasco, L.; Acuti, G.; Bani, P.; Dalle Zotte, A.; Danieli, P.P.; De Angelis, A.; Fortina, R.; Marino, R.; Parisi, G.; Piccolo, G.; et al. Insect and Fish By-Products as Sustainable Alternatives to Conventional Animal Proteins in Animal Nutrition. Ital. J. Anim. Sci. 2020, 19, 360–372. [Google Scholar] [CrossRef]
- Lock, E.-J.; Biancarosa, I.; Gasco, L. Insects as Raw Materials in Compound Feed for Aquaculture. In Edible Insects in Sustainable Food Systems; Springer: Berlin/Heidelberg, Germany, 2018; pp. 263–276. ISBN 978-3-319-74011-9. [Google Scholar]
- Rimoldi, S.; Antonini, M.; Gasco, L.; Moroni, F.; Terova, G. Intestinal Microbial Communities of Rainbow Trout (Oncorhynchus mykiss) May Be Improved by Feeding a Hermetia illucens Meal/Low-Fishmeal Diet. Fish Physiol. Biochem. 2021, 47, 365–380. [Google Scholar] [CrossRef]
- Rimoldi, S.; Gini, E.; Iannini, F.; Gasco, L.; Terova, G. The Effects of Dietary Insect Meal from Hermetia illucens Prepupae on Autochthonous Gut Microbiota of Rainbow Trout (Oncorhynchus mykiss). Animals 2019, 9, 143. [Google Scholar] [CrossRef]
- Terova, G.; Gini, E.; Gasco, L.; Moroni, F.; Antonini, M.; Rimoldi, S. Effects of Full Replacement of Dietary Fishmeal with Insect Meal from Tenebrio molitor on Rainbow Trout Gut and Skin Microbiota. J. Anim. Sci. Biotechnol. 2021, 12, 30. [Google Scholar] [CrossRef]
- Terova, G.; Ceccotti, C.; Ascione, C.; Gasco, L.; Rimoldi, S. Effects of Partially Defatted Hermetia illucens Meal in Rainbow Trout Diet on Hepatic Methionine Metabolism. Animals 2020, 10, 1059. [Google Scholar] [CrossRef]
- Terova, G.; Rimoldi, S.; Ascione, C.; Gini, E.; Ceccotti, C.; Gasco, L. Rainbow Trout (Oncorhynchus mykiss) Gut Microbiota Is Modulated by Insect Meal from Hermetia illucens Prepupae in the Diet. Rev. Fish Biol. Fish 2019, 29, 465–486. [Google Scholar] [CrossRef]
- Rimoldi, S.; Ceccotti, C.; Brambilla, F.; Faccenda, F.; Antonini, M.; Terova, G. Potential of Shrimp Waste Meal and Insect Exuviae as Sustainable Sources of Chitin for Fish Feeds. Aquaculture 2023, 567, 739256. [Google Scholar] [CrossRef]
- Abdel-Ghany, H.M.; Salem, M.E.-S. Effects of Dietary Chitosan Supplementation on Farmed Fish: A Review. Rev. Aquac. 2020, 12, 438–452. [Google Scholar] [CrossRef]
- Jiménez-Gómez, C.P.; Cecilia, J.A. Chitosan: A Natural Biopolymer with a Wide and Varied Range of Applications. Molecules 2020, 25, 3981. [Google Scholar] [CrossRef]
- Alishahi, A.; Aïder, M. Applications of Chitosan in the Seafood Industry and Aquaculture: A Review. Food Bioprocess. Technol. 2012, 5, 817–830. [Google Scholar] [CrossRef]
- Ringø, E.; Zhou, Z.; Olsen, R.E.; Song, S.K. Use of Chitin and Krill in Aquaculture—The Effect on Gut Microbiota and the Immune System: A Review. Aquac. Nutr. 2012, 18, 117–131. [Google Scholar] [CrossRef]
- Ahmed, F.; Soliman, F.M.; Adly, M.A.; Soliman, H.A.M.; El-Matbouli, M.; Saleh, M. Dietary Chitosan Nanoparticles: Potential Role in Modulation of Rainbow Trout (Oncorhynchus mykiss) Antibacterial Defense and Intestinal Immunity against Enteric Redmouth Disease. Mar. Drugs 2021, 19, 72. [Google Scholar] [CrossRef]
- Dawood, M.A.O.; Gewaily, M.S.; Soliman, A.A.; Shukry, M.; Amer, A.A.; Younis, E.M.; Abdel-Warith, A.-W.A.; Van Doan, H.; Saad, A.H.; Aboubakr, M.; et al. Marine-Derived Chitosan Nanoparticles Improved the Intestinal Histo-Morphometrical Features in Association with the Health and Immune Response of Grey Mullet (Liza ramada). Mar. Drugs 2020, 18, 611. [Google Scholar] [CrossRef]
- Kono, M.; Matsui, T.; Shimizu, C. Effect of Chitin, Chitosan, and Cellulose as Diet Supplements on the Growth of Cultured Fish. Nippon. Suisan Gakkaishi 1987, 53, 125–129. [Google Scholar] [CrossRef]
- Qin, C.; Zhang, Y.; Liu, W.; Xu, L.; Yang, Y.; Zhou, Z. Effects of Chito-Oligosaccharides Supplementation on Growth Performance, Intestinal Cytokine Expression, Autochthonous Gut Bacteria and Disease Resistance in Hybrid Tilapia Oreochromis niloticus ♀ × Oreochromis aureus ♂. Fish Shellfish Immunol. 2014, 40, 267–274. [Google Scholar] [CrossRef]
- Elserafy, S.S.; Abdel-Hameid, N.-A.H.; Abdel-Salam, H.A.; Dakrouni, A.M. Effect of Shrimp Waste Extracted Chitin on Growth and Some Biochemical Parameters of the Nile Tilapia. Egypt. J. Aquat. Biol. Fish 2021, 25, 313–329. [Google Scholar] [CrossRef]
- Shi, F.; Qiu, X.; Nie, L.; Hu, L.; Babu, V.S.; Lin, Q.; Zhang, Y.; Chen, L.; Li, J.; Lin, L.; et al. Effects of Oligochitosan on the Growth, Immune Responses and Gut Microbes of Tilapia (Oreochromis niloticus). Fish Shellfish Immunol. 2020, 106, 563–573. [Google Scholar] [CrossRef]
- Gasco, L.; Biasato, I.; Dabbou, S.; Schiavone, A.; Gai, F. Animals Fed Insect-Based Diets: State-of-the-Art on Digestibility, Performance and Product Quality. Animals 2019, 9, 170. [Google Scholar] [CrossRef]
- Danulat, E. The Effects of Various Diets on Chitinase and SS-Glucosidase Activities and the Condition of Cod, Gadus morhua (L.). J. Fish Biol. 1986, 28, 191–197. [Google Scholar] [CrossRef]
- Fines, B.C.; Holt, G.J. Chitinase and Apparent Digestibility of Chitin in the Digestive Tract of Juvenile Cobia, Rachycentron canadum. Aquaculture 2010, 303, 34–39. [Google Scholar] [CrossRef]
- Gutowska, M.A.; Drazen, J.C.; Robison, B.H. Digestive Chitinolytic Activity in Marine Fishes of Monterey Bay, California. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2004, 139, 351–358. [Google Scholar] [CrossRef]
- Jozefiak, A.; Engberg, R. Insect Proteins as a Potential Source of Antimicrobial Peptides in Livestock Production. A Review. J. Anim. Feed Sci. 2017, 26, 69998. [Google Scholar] [CrossRef]
- Bruni, L.; Belghit, I.; Lock, E.-J.; Secci, G.; Taiti, C.; Parisi, G. Total Replacement of Dietary Fish Meal with Black Soldier Fly (Hermetia illucens) Larvae Does Not Impair Physical, Chemical or Volatile Composition of Farmed Atlantic Salmon (Salmo salar L.). J. Sci. Food Agric. 2020, 100, 1038–1047. [Google Scholar] [CrossRef]
- Foysal, M.J.; Gupta, S.K. A Systematic Meta-Analysis Reveals Enrichment of Actinobacteria and Firmicutes in the Fish Gut in Response to Black Soldier Fly (Hermetica illucens) Meal-Based Diets. Aquaculture 2022, 549, 737760. [Google Scholar] [CrossRef]
- Gaudioso, G.; Marzorati, G.; Faccenda, F.; Weil, T.; Lunelli, F.; Cardinaletti, G.; Marino, G.; Olivotto, I.; Parisi, G.; Tibaldi, E.; et al. Processed Animal Proteins from Insect and Poultry By-Products in a Fish Meal-Free Diet for Rainbow Trout: Impact on Intestinal Microbiota and Inflammatory Markers. Int. J. Mol. Sci. 2021, 22, 5454. [Google Scholar] [CrossRef]
- Huyben, D.; Vidaković, A.; Werner Hallgren, S.; Langeland, M. High-Throughput Sequencing of Gut Microbiota in Rainbow Trout (Oncorhynchus mykiss) Fed Larval and Pre-Pupae Stages of Black Soldier Fly (Hermetia illucens). Aquaculture 2019, 500, 485–491. [Google Scholar] [CrossRef]
- Karlsen, Ø.; Amlund, H.; Berg, A.; Olsen, R.E. The Effect of Dietary Chitin on Growth and Nutrient Digestibility in Farmed Atlantic Cod, Atlantic Salmon and Atlantic Halibut. Aquac. Res. 2017, 48, 123–133. [Google Scholar] [CrossRef]
- Renna, M.; Schiavone, A.; Gai, F.; Dabbou, S.; Lussiana, C.; Malfatto, V.; Prearo, M.; Capucchio, M.T.; Biasato, I.; Biasibetti, E.; et al. Evaluation of the Suitability of a Partially Defatted Black Soldier Fly (Hermetia illucens L.) Larvae Meal as Ingredient for Rainbow Trout (Oncorhynchus mykiss Walbaum) Diets. J. Anim. Sci. Biotechnol. 2017, 8, 57. [Google Scholar] [CrossRef] [PubMed]
- Maulu, S.; Langi, S.; Hasimuna, O.J.; Missinhoun, D.; Munganga, B.P.; Hampuwo, B.M.; Gabriel, N.N.; Elsabagh, M.; Doan, H.V.; Kari, Z.A.; et al. Recent Advances in the Utilization of Insects as an Ingredient in Aquafeeds: A Review. Anim. Nutr. 2022, 11, 334. [Google Scholar] [CrossRef] [PubMed]
- Rangel, F.; Monteiro, M.; Santos, R.A.; Ferreira-Martins, D.; Cortinhas, R.; Gasco, L.; Gai, F.; Pousão-Ferreira, P.; Couto, A.; Oliva-Teles, A.; et al. Novel Chitinolytic Bacillus spp. Increase Feed Efficiency, Feed Digestibility, and Survivability to Vibrio anguillarum in European Seabass Fed with Diets Containing Hermetia illucens Larvae Meal. Aquaculture 2024, 579, 740258. [Google Scholar] [CrossRef]
- Gao, C.; Cai, X.; Zhang, Y.; Su, B.; Song, H.; Wenqi, W.; Li, C. Characterization and Expression Analysis of Chitinase Genes (CHIT1, CHIT2 and CHIT3) in Turbot (Scophthalmus maximus L.) Following Bacterial Challenge. Fish Shellfish Immunol. 2017, 64, 357–366. [Google Scholar] [CrossRef]
- Ikeda, M.; Kakizaki, H.; Matsumiya, M. Biochemistry of Fish Stomach Chitinase. Int. J. Biol. Macromol. 2017, 104, 1672–1681. [Google Scholar] [CrossRef]
- Kakizaki, H.; Ikeda, M.; Fukushima, H.; Matsumiya, M. Distribution of Chitinolytic Enzymes in the Organs and cDNA Cloning of Chitinase Isozymes from the Stomach of Two Species of Fish, Chub Mackerel (Scomber japonicus) and Silver Croaker (Pennahia argentata). Open J. Mar. Sci. 2015, 05, 398. [Google Scholar] [CrossRef]
- Pohls, P.; González-Dávalos, L.; Mora, O.; Shimada, A.; Varela-Echavarria, A.; Toledo-Cuevas, E.M.; Martínez-Palacios, C.A. A Complete Chitinolytic System in the Atherinopsid Pike Silverside Chirostoma Estor: Gene Expression and Activities. J. Fish Biol. 2016, 88, 2130–2143. [Google Scholar] [CrossRef]
- Lindsay, G.J.H.; Walton, M.J.; Adron, J.W.; Fletcher, T.C.; Cho, C.Y.; Cowey, C.B. The Growth of Rainbow Trout (Salmo gairdneri) given Diets Containing Chitin and Its Relationship to Chitinolytic Enzymes and Chitin Digestibility. Aquaculture 1984, 37, 315–334. [Google Scholar] [CrossRef]
- LeCleir, G.R.; Buchan, A.; Hollibaugh, J.T. Chitinase Gene Sequences Retrieved from Diverse Aquatic Habitats Reveal Environment-Specific Distributions. Appl. Environ. Microbiol. 2004, 70, 6977–6983. [Google Scholar] [CrossRef]
- Hamid, R.; Khan, M.A.; Ahmad, M.; Ahmad, M.M.; Abdin, M.Z.; Musarrat, J.; Javed, S. Chitinases: An Update. J. Pharm. Bioallied Sci. 2013, 5, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Basawa, R.; Kabra, S.; Khile, D.A.; Faruk Abbu, R.U.; Parekkadan, S.J.; Thomas, N.A.; Kim, S.K.; Raval, R. Repurposing Chitin-Rich Seafood Waste for Warm-Water Fish Farming. Heliyon 2023, 9, e18197. [Google Scholar] [CrossRef] [PubMed]
- Powell, A.; Rowley, A.F. The Effect of Dietary Chitin Supplementation on the Survival and Immune Reactivity of the Shore Crab, Carcinus maenas. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2007, 147, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Eggink, K.M.; Pedersen, P.B.; Lund, I.; Dalsgaard, J. Chitin Digestibility and Intestinal Exochitinase Activity in Nile Tilapia and Rainbow Trout Fed Different Black Soldier Fly Larvae Meal Size Fractions. Aquac. Res. 2022, 53, 5536–5546. [Google Scholar] [CrossRef]
- Brauer, J.M.E. Chitin and Chitosan in Aquaculture. In Avances en Nutrición Acuicola; Universidad de Sonora: Hermosillo, Mexico, 2015. [Google Scholar]
- Mohan, K.; Rajan, D.K.; Ganesan, A.R.; Divya, D.; Johansen, J.; Zhang, S. Chitin, Chitosan and Chitooligosaccharides as Potential Growth Promoters and Immunostimulants in Aquaculture: A Comprehensive Review. Int. J. Biol. Macromol. 2023, 251, 126285. [Google Scholar] [CrossRef] [PubMed]
- Cheng, A.-C.; Shiu, Y.-L.; Chiu, S.-T.; Ballantyne, R.; Liu, C.-H. Effects of Chitin from Daphnia similis and Its Derivative, Chitosan on the Immune Response and Disease Resistance of White Shrimp, Litopenaeus vannamei. Fish Shellfish Immunol. 2021, 119, 329–338. [Google Scholar] [CrossRef]
- Kamilya, D.; Khan, M.d.I.R. Chapter 24—Chitin and Chitosan as Promising Immunostimulant for Aquaculture. In Handbook of Chitin and Chitosan; Gopi, S., Thomas, S., Pius, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 761–771. ISBN 978-0-12-817966-6. [Google Scholar]
- Andriani, Y.; Pratama, R.I.; Hanidah, I.I. Chitosan Application in Aquatic Feed and Its Impact on Fish and Shrimp Productivity. Asian J. Biol. 2023, 19, 25–30. [Google Scholar] [CrossRef]
- Köprücü, K.; Özdemir, Y. Apparent Digestibility of Selected Feed Ingredients for Nile Tilapia (Oreochromis niloticus). Aquaculture 2005, 250, 308–316. [Google Scholar] [CrossRef]
- Longvah, T.; Mangthya, K.; Ramulu, P. Nutrient Composition and Protein Quality Evaluation of Eri Silkworm (Samia ricinii) Prepupae and Pupae. Food Chem. 2011, 128, 400–403. [Google Scholar] [CrossRef]
- Li, D.; Zhang, J.; Wang, Y.; Liu, X.; Ma, E.; Sun, Y.; Li, S.; Zhu, K.Y.; Zhang, J. Two Chitinase 5 Genes from Locusta Migratoria: Molecular Characteristics and Functional Differentiation. Insect Biochem. Mol. Biol. 2015, 58, 46–54. [Google Scholar] [CrossRef]
- Xi, Y.; Pan, P.-L.; Ye, Y.-X.; Yu, B.; Xu, H.-J.; Zhang, C.-X. Chitinase-like Gene Family in the Brown Planthopper, Nilaparvata lugens. Insect Mol. Biol. 2015, 24, 29–40. [Google Scholar] [CrossRef]
- Kramer, K.J.; Corpuz, L.; Choi, H.K.; Muthukrishnan, S. Sequence of a cDNA and Expression of the Gene Encoding Epidermal and Gut Chitinases of Manduca sexta. Insect Biochem. Mol. Biol. 1993, 23, 691–701. [Google Scholar] [CrossRef] [PubMed]
- Niu, S.; Yang, L.; Zuo, H.; Zheng, J.; Weng, S.; He, J.; Xu, X. A Chitinase from Pacific White Shrimp Litopenaeus vannamei Involved in Immune Regulation. Dev. Comp. Immunol. 2018, 85, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Veliz, E.A.; Martínez-Hidalgo, P.; Hirsch, A.M.; Veliz, E.A.; Martínez-Hidalgo, P.; Hirsch, A.M. Chitinase-Producing Bacteria and Their Role in Biocontrol. AIMS Microbiol. 2017, 3, 689–705. [Google Scholar] [CrossRef] [PubMed]
- Karthik, N.; Akanksha, K.; Pandey, A. Production, Purification and Properties of Fungal Chitinases—A Review. Indian J. Exp. Biol. 2014, 52, 1025–1035. [Google Scholar]
- Thakur, D.; Bairwa, A.; Dipta, B.; Jhilta, P.; Chauhan, A. An Overview of Fungal Chitinases and Their Potential Applications. Protoplasma 2023, 260, 1031–1046. [Google Scholar] [CrossRef]
- Rangel, F.; Santos, R.A.; Monteiro, M.; Lavrador, A.S.; Gasco, L.; Gai, F.; Oliva-Teles, A.; Enes, P.; Serra, C.R. Isolation of Chitinolytic Bacteria from European Sea Bass Gut Microbiota Fed Diets with Distinct Insect Meals. Biology 2022, 11, 964. [Google Scholar] [CrossRef]
- Subramanian, K.; Balaraman, D.; Panangal, M.; Nageswara Rao, T.; Perumal, E.; Kumarappan, R.A.A.; Sampath Renuga, P.; Arumugam, S.; Thirunavukkarasu, R.; Aruni, W.; et al. Bioconversion of Chitin Waste through Stenotrophomonas maltophilia for Production of Chitin Derivatives as a Seabass Enrichment Diet. Sci. Rep. 2022, 12, 4792. [Google Scholar] [CrossRef]
- Agbohessou, P.S.; Mandiki, S.N.M.; Mbondo Biyong, S.R.; Cornet, V.; Nguyen, T.M.; Lambert, J.; Jauniaux, T.; Lalèyè, P.A.; Kestemont, P. Intestinal Histopathology and Immune Responses Following Escherichia coli Lipopolysaccharide Challenge in Nile Tilapia Fed Enriched Black Soldier Fly Larval (BSF) Meal Supplemented with Chitinase. Fish Shellfish Immunol. 2022, 128, 620–633. [Google Scholar] [CrossRef]
- Liu, Y.; Zhou, Z.; Miao, W.; Zhang, Y.; Cao, Y.; He, S.; Bai, D.; Yao, B. A Chitinase from Aeromonas veronii CD3 with the Potential to Control Myxozoan Disease. PLoS ONE 2011, 6, e29091. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhou, Z.; Liu, Y.; Cao, Y.; He, S.; Huo, F.; Qin, C.; Yao, B.; Ringø, E. High-Yield Production of a Chitinase from Aeromonas veronii B565 as a Potential Feed Supplement for Warm-Water Aquaculture. Appl. Microbiol. Biotechnol. 2014, 98, 1651–1662. [Google Scholar] [CrossRef]
- Mohan, K.; Ravichandran, S.; Muralisankar, T.; Uthayakumar, V.; Chandirasekar, R.; Seedevi, P.; Rajan, D.K. Potential Uses of Fungal Polysaccharides as Immunostimulants in Fish and Shrimp Aquaculture: A Review. Aquaculture 2019, 500, 250–263. [Google Scholar] [CrossRef]
- Poria, V.; Rana, A.; Kumari, A.; Grewal, J.; Pranaw, K.; Singh, S. Current Perspectives on Chitinolytic Enzymes and Their Agro-Industrial Applications. Biology 2021, 10, 1319. [Google Scholar] [CrossRef]
- Rathore, A.S.; Gupta, R.D. Chitinases from Bacteria to Human: Properties, Applications, and Future Perspectives. Enzyme Res. 2015, 2015, 791907. [Google Scholar] [CrossRef]
- Swiontek Brzezinska, M.; Jankiewicz, U.; Burkowska, A.; Walczak, M. Chitinolytic Microorganisms and Their Possible Application in Environmental Protection. Curr. Microbiol. 2014, 68, 71–81. [Google Scholar] [CrossRef]
- Gomaa, E.Z. Microbial Chitinases: Properties, Enhancement and Potential Applications. Protoplasma 2021, 258, 695–710. [Google Scholar] [CrossRef]
- Komi, D.E.A.; Sharma, L.; Dela Cruz, C.S. Chitin and Its Effects on Inflammatory and Immune Responses. Clin. Rev. Allergy Immunol. 2018, 54, 213–223. [Google Scholar] [CrossRef]
- Kumar, M.; Brar, A.; Yadav, M.; Chawade, A.; Vivekanand, V.; Pareek, N. Chitinases—Potential Candidates for Enhanced Plant Resistance towards Fungal Pathogens. Agriculture 2018, 8, 88. [Google Scholar] [CrossRef]
- Fox, C.J. The Effect of Dietary Chitin on the Growth, Survival and Chitinase Levels in the Digestive Gland of Juvenile Penaeus monodon (Fab.). Aquaculture 1993, 109, 39–49. [Google Scholar] [CrossRef]
- Fontes, T.V.; de Oliveira, K.R.B.; Gomes Almeida, I.L.; Orlando, T.M.; Rodrigues, P.B.; da Costa, D.V.; Rosa, P.V.E. Digestibility of Insect Meals for Nile Tilapia Fingerlings. Animals 2019, 9, 181. [Google Scholar] [CrossRef]
- Zhang, Y.; Feng, S.; Chen, J.; Qin, C.; Lin, H.; Li, W. Stimulatory Effects of Chitinase on Growth and Immune Defense of Orange-Spotted Grouper (Epinephelus coioides). Fish Shellfish Immunol. 2012, 32, 844–854. [Google Scholar] [CrossRef]
- Molinari, L.M.; Pedroso, R.B.; Scoaris, D.d.O.; Ueda-Nakamura, T.; Nakamura, C.V.; Dias Filho, B.P. Identification and Partial Characterisation of a Chitinase from Nile Tilapia, Oreochromis niloticus. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2007, 146, 81–87. [Google Scholar] [CrossRef]
- Harikrishnan, R.; Kim, J.-S.; Balasundaram, C.; Heo, M.-S. Dietary Supplementation with Chitin and Chitosan on Haematology and Innate Immune Response in Epinephelus bruneus against Philasterides dicentrarchi. Exp. Parasitol. 2012, 131, 116–124. [Google Scholar] [CrossRef]
- Li, Y.; Kortner, T.M.; Chikwati, E.M.; Belghit, I.; Lock, E.-J.; Krogdahl, Å. Total Replacement of Fish Meal with Black Soldier Fly (Hermetia illucens) Larvae Meal Does Not Compromise the Gut Health of Atlantic Salmon (Salmo salar). Aquaculture 2020, 520, 734967. [Google Scholar] [CrossRef]
- Stoykov, Y.M.; Pavlov, A.I.; Krastanov, A.I. Chitinase Biotechnology: Production, Purification, and Application. Eng. Life Sci. 2015, 15, 30–38. [Google Scholar] [CrossRef]
- Meena, S.; Gothwal, R.K.; Saxena, J.; Mohan, M.K.; Ghosh, P. Chitinase Production by a Newly Isolated Thermotolerant Paenibacillus sp. BISR-047. Ann. Microbiol. 2014, 64, 787–797. [Google Scholar] [CrossRef]
- Shahbaz, U.; Yu, X. Cloning, Isolation, and Characterization of Novel Chitinase-Producing Bacterial Strain UM01 (Myxococcus fulvus). J. Genet. Eng. Biotechnol. 2020, 18, 45. [Google Scholar] [CrossRef]
- Narayanan, K.; Chopade, N.; Raj, P.V.; Subrahmanyam, V.M.; Rao, J.V. Fungal Chitinase Production and Its Application in Biowaste Management. NIScPR Online Period. Repos. 2013, 72, 393–399. [Google Scholar]
- Loc, N.H.; Huy, N.D.; Quang, H.T.; Lan, T.T.; Thu Ha, T.T. Characterisation and Antifungal Activity of Extracellular Chitinase from a Biocontrol Fungus, Trichoderma asperellum PQ34. Mycology 2019, 11, 38–48. [Google Scholar] [CrossRef]
- Baldoni, D.B.; Antoniolli, Z.I.; Mazutti, M.A.; Jacques, R.J.S.; Dotto, A.C.; de Oliveira Silveira, A.; Ferraz, R.C.; Soares, V.B.; de Souza, A.R.C. Chitinase Production by Trichoderma koningiopsis UFSMQ40 Using Solid State Fermentation. Braz. J. Microbiol. 2020, 51, 1897–1908. [Google Scholar] [CrossRef]
- Brzezinska, M.S.; Jankiewicz, U. Production of Antifungal Chitinase by Aspergillus niger LOCK 62 and Its Potential Role in the Biological Control. Curr. Microbiol. 2012, 65, 666–672. [Google Scholar] [CrossRef]
- Holen, M.M.; Vaaje-Kolstad, G.; Kent, M.P.; Sandve, S.R. Gene Family Expansion and Functional Diversification of Chitinase and Chitin Synthase Genes in Atlantic Salmon (Salmo salar). G3 Genes Genomes Genet. 2023, 13, jkad069. [Google Scholar] [CrossRef] [PubMed]
- Liao, Q.; Qin, Y.; Zhou, Y.; Shi, G.; Li, X.; Li, J.; Mo, R.; Zhang, Y.; Yu, Z. Characterization and Functional Analysis of a Chitinase Gene: Evidence of Ch-chit Participates in the Regulation of Biomineralization in Crassostrea hongkongensis. Aquac. Rep. 2021, 21, 100852. [Google Scholar] [CrossRef]
- Doan, C.T.; Tran, T.N.; Wang, S.-L. Production of Thermophilic Chitinase by Paenibacillus sp. TKU052 by Bioprocessing of Chitinous Fishery Wastes and Its Application in N-Acetyl-D-Glucosamine Production. Polymers 2021, 13, 3048. [Google Scholar] [CrossRef] [PubMed]
- Robinson, N.A.; Robledo, D.; Sveen, L.; Daniels, R.R.; Krasnov, A.; Coates, A.; Jin, Y.H.; Barrett, L.T.; Lillehammer, M.; Kettunen, A.H.; et al. Applying Genetic Technologies to Combat Infectious Diseases in Aquaculture. Rev. Aquac. 2023, 15, 491–535. [Google Scholar] [CrossRef]
- Kaczmarek, M.B.; Struszczyk-Swita, K.; Xiao, M.; Szczęsna-Antczak, M.; Antczak, T.; Gierszewska, M.; Steinbüchel, A.; Daroch, M. Polycistronic Expression System for Pichia pastoris Composed of Chitino- and Chitosanolytic Enzymes. Front. Bioeng. Biotechnol. 2021, 9, 710992. [Google Scholar] [CrossRef] [PubMed]
- Karbalaei, M.; Rezaee, S.A.; Farsiani, H. Pichia Pastoris: A Highly Successful Expression System for Optimal Synthesis of Heterologous Proteins. J. Cell. Physiol. 2020, 235, 5867–5881. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Yan, H. Cloning, Expression, Purification and Biochemical Characterization of the Recombinant Chitinase Enzyme Encoded by CTS2 in the Budding Yeast. Protein Expr. Purif. 2023, 208–209, 106294. [Google Scholar] [CrossRef]
- Oyeleye, A.; Normi, Y.M. Chitinase: Diversity, Limitations, and Trends in Engineering for Suitable Applications. Biosci. Rep. 2018, 38, BSR2018032300. [Google Scholar] [CrossRef]
- Liu, X.-Y.; Wang, S.-S.; Zhong, F.; Zhou, M.; Jiang, X.-Y.; Cheng, Y.-S.; Dan, Y.-H.; Hu, G.; Li, C.; Tang, B.; et al. Chitinase (CHI) of Spodoptera frugiperda Affects Molting Development by Regulating the Metabolism of Chitin and Trehalose. Front. Physiol. 2022, 13, 2100. [Google Scholar] [CrossRef]
- Merzendorfer, H.; Zimoch, L. Chitin Metabolism in Insects: Structure, Function and Regulation of Chitin Synthases and Chitinases. J. Exp. Biol. 2003, 206, 4393–4412. [Google Scholar] [CrossRef]
- Nurfikari, A.; de Boer, W. Chitin Determination in Residual Streams Derived from Insect Production by LC-ECD and LC-MS/MS Methods. Front. Sustain. Food Syst. 2021, 5, 795694. [Google Scholar] [CrossRef]
- Hao, Z.; Cai, Y.; Liao, X.; Zhang, X.; Fang, Z.; Zhang, D. Optimization of Nutrition Factors on Chitinase Production from a Newly Isolated Chitiolyticbacter meiyuanensis SYBC-H1. Braz. J. Microbiol. 2012, 43, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Rachmawaty, R.; Halifah, P.; Hartati, H.; Maulana, Z.; Salleh, M.M.d. Optimization of Chitinase Production by Trichoderma virens in Solid State Fermentation Using Response Surface Methodology. Mater. Sci. Forum 2019, 967, 132–142. [Google Scholar] [CrossRef]
- Adámková, A.; Mlček, J.; Kouřimská, L.; Borkovcová, M.; Bušina, T.; Adámek, M.; Bednářová, M.; Krajsa, J. Nutritional Potential of Selected Insect Species Reared on the Island of Sumatra. Int. J. Environ. Res. Public Health 2017, 14, 521. [Google Scholar] [CrossRef]
- Hahn, T.; Tafi, E.; Paul, A.; Salvia, R.; Falabella, P.; Zibek, S. Current State of Chitin Purification and Chitosan Production from Insects. J. Chem. Technol. Biotechnol. 2020, 95, 2775–2795. [Google Scholar] [CrossRef]
| Fungal Chitinases | Major Finding(s) | References |
|---|---|---|
| Aspergillus flavus | Fungal chitinases from Aspergillus flavus have been shown to have an anti-fungal effect against many plant pathogenic fungi. This indicates that Aspergillus flavus chitinases can be used to control pests and diseases in aquaculture. | [65] |
| Mucor | Chitin-degrading fungi such as Mucor have been identified in the aquatic environment. This indicates that Mucor chitinases can be used for chitin degradation in aquaculture. | [66] |
| Trichoderma | Fungal chitinases from Trichoderma facilitate mycoparasitism of other fungi. This suggests that Trichoderma chitinases can be used to control fungal infections in aquaculture. | [58] |
| Aspergillus sp. S1–13 | Aspergillus sp. S1–13 has been shown to synthesize chitinolytic enzymes when grown on a medium containing shrimp waste. This suggests that the chitinases of Aspergillus sp. S1–13 can be used to convert chitin waste from fisheries into simpler, useful components to reduce water pollution. | [66] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hasan, I.; Gai, F.; Cirrincione, S.; Rimoldi, S.; Saroglia, G.; Terova, G. Chitinase and Insect Meal in Aquaculture Nutrition: A Comprehensive Overview of the Latest Achievements. Fishes 2023, 8, 607. https://doi.org/10.3390/fishes8120607
Hasan I, Gai F, Cirrincione S, Rimoldi S, Saroglia G, Terova G. Chitinase and Insect Meal in Aquaculture Nutrition: A Comprehensive Overview of the Latest Achievements. Fishes. 2023; 8(12):607. https://doi.org/10.3390/fishes8120607
Chicago/Turabian StyleHasan, Imam, Francesco Gai, Simona Cirrincione, Simona Rimoldi, Giulio Saroglia, and Genciana Terova. 2023. "Chitinase and Insect Meal in Aquaculture Nutrition: A Comprehensive Overview of the Latest Achievements" Fishes 8, no. 12: 607. https://doi.org/10.3390/fishes8120607
APA StyleHasan, I., Gai, F., Cirrincione, S., Rimoldi, S., Saroglia, G., & Terova, G. (2023). Chitinase and Insect Meal in Aquaculture Nutrition: A Comprehensive Overview of the Latest Achievements. Fishes, 8(12), 607. https://doi.org/10.3390/fishes8120607