Prevalence and Risk of Anisakid Larvae in Fresh Fish Frequently Consumed in Spain: An Overview
Abstract
1. Introduction
2. Results
2.1. Blue Whiting (Micromesistius poutassou)
2.2. European Hake (Merluccius merluccius)
2.3. Silver Hake (Merluccius bilinearis)
2.4. Red Mullet (Mullus barbatus)
2.5. Mackerel (Scomber scombrus)
2.6. Chub Mackerel (Scomber japonicus)
2.7. Horse Mackerel (Trachurus trachurus)
2.8. Sardine (Sardina pilchardus)
2.9. Anchovy (Engraulis encrasicolus)
3. Discussion
4. Materials and Methods
4.1. Samples
4.2. Fish Inspection
4.3. Morphological Identification
4.4. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Serrano-Moliner, M.; Morales-Suarez-Varela, M.; Valero, M.A. Epidemiology and management of foodborne nematodiasis in the European Union, systematic review 2000–2016. Pathog. Glob. Health 2018, 112, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Hochberg, N.S.; Hamer, D. Anisakidosis: Perils of the Deep. Clin. Infect. Dis. 2010, 51, 806–812. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority (EFSA). Scientific opinion on risk assessment of parasites in fishery products. EFSA Panel on Biological Hazards (BIOHAZ). EFSA J. 2010, 8, 1543. [Google Scholar] [CrossRef]
- Mazzucco, W.; Raia, D.D.; Marotta, C.; Costa, A.; Ferrantelli, V.; Vitale, F.; Casuccio, A. Anisakis sensitization in different population groups and public health impact: A systematic review. PLoS ONE 2018, 13, e0203671. [Google Scholar] [CrossRef]
- Baird, F.J.; Gasser, R.; Jabbar, A.; Lopata, A.L. Foodborne anisakiasis and allergy. Mol. Cell Probes 2014, 28, 164–174. [Google Scholar] [CrossRef] [PubMed]
- Bao, M.; Pierce, G.J.; Pascual, S.; González-Muñoz, M.; Mattiuci, S.; Mladineo, I.; Cipriani, P.; Bušelić, I.; Strachan, N.J. Assessing the risk of an emerging zoonosis of worldwide concern: Anisakiasis. Sci. Rep. 2017, 13, 43699. [Google Scholar] [CrossRef] [PubMed]
- Buchmann, K.; Mehrdana, F. Effects of anisakid nematodes Anisakis simplex (s.l.), Pseudoterranova decipiens (s.l.) and Contracaecum osculatum (s.l.) on fish and consumer health. FAWPAR 2016, 4, 13–22. [Google Scholar] [CrossRef]
- Mattiucci, S.; Cipriani, P.; Levsen, A.; Paoletti, M.; Nascetti, G. Molecular epidemiology of Anisakis and Anisakiasis: An Ecological and Evolutionary Road Map. Adv. Parasitol. 2018, 99, 93–263. [Google Scholar] [CrossRef]
- Romero, M.C.; Valero, A.; Navarro-Moll, M.C.; Martín-Sánchez, J. Experimental comparison of pathogenic potential of two sibling species Anisakis simplex s.s. and Anisakis pegreffii in Wistar rat. Trop. Med. Int. Health 2013, 18, 979–984. [Google Scholar] [CrossRef]
- Levsen, A.; Svanevik, C.S.; Cipriani, P.; Mattiucci, S.; Gay, M.; Hastie, L.C.; Bušelić, I.; Mladineo, I.; Karl, H.G.N.D.; Ostermeyer, U.; et al. A survey of zoonotic nematodes of commercial key fish species from major European fishing grounds—Introducing the FP7 PARASITE exposure assessment study. Fish. Res. 2018, 202, 4–21. [Google Scholar] [CrossRef]
- Herrador, Z.; Daschner, A.; Perteguer, M.J.; Benito, A. Epidemiological scenario of anisakidosis in Spain based on associated hospitalizations: The tipping point of the iceberg. Clin. Inf. Dis. 2018. [Google Scholar] [CrossRef]
- European Commission. Commission Regulation (EU) No 1276/2011 of 8 December 2011 amending Annex III to Regulation (EC) No 853/2004 of the European Parliament and of the Council as regards the treatment to kill viable parasites in fishery products for human consumption. Off. J. Eur. Union L 2011, 327, 39–41. [Google Scholar]
- Real Decreto 1420/2006, de 1 de diciembre, sobre prevención de la parasitosis por anisakis en productos de la pesca suministrados por establecimientos que sirven comida a los consumidores finales o a colectividades. Boletín Oficial Estado 2006, 302, 44547–44549. (In Spanish)
- Sánchez-Alonso, I.; Carballeda-Sangia, N.; González-Muñoz, M.; Navas, A.; Arcos, S.C.; Mendizábal, A.; Tejada, M.; Careche, M. Pathogenic potential of Anisakis L3 after freezing in domestic freezers. Food Control 2018, 84, 61–69. [Google Scholar] [CrossRef]
- Solas, M.T.; García, M.L.; De las Heras, C.; Rodriguez-Mahillo, A.I.; Gonzalez-Muñoz, M.; Moneo, I.; Mendizábal, A.; Tejada, M. Anisakis simplex antigens in fresh and frozen-thawed muscle of anchovies in vinegar. Food Sci. Technol. Int. 2008, 15, 139–148. [Google Scholar] [CrossRef]
- Klimpel, S.; Palm, H.W.; Rückert, S.; Piatkowski, U. The life cycle of Anisakis simplex in the Norwegian Deep (northern North Sea). Parasitol. Res. 2004, 94, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Kuhn, T.; Hailer, F.; Palm, H.W.; Klimpel, S. Global assessment of molecularly identified Anisakis Dujardin, 1845 (Nematoda: Anisakidae) in their teleost intermediate hosts. Folia Parasitol. 2013, 60, 123–134. [Google Scholar] [CrossRef] [PubMed]
- Gregori, M.; Roura, Á.; Abollo, E.; González, Á.F.; Pascual, S. Anisakis simplex complex (Nematoda: Anisakidae) in zooplankton communities from temperate NE Atlantic waters. J. Nat. Hist. 2015, 49, 755–773. [Google Scholar] [CrossRef]
- Ministerio de Agricultura, Pesca y Alimentación. Informe del Consumo de Alimentación en España. 2017. Available online: https://www.mapa.gob.es/images/es/informeanualdeconsumoalimentario2017_tcm30-456186.pdf (accessed on 8 December 2018).
- Fernández, M.; Aznar, F.J.; Montero, F.E.; Raga, J.A. Endoparasites of the blue whiting, Micromesistius poutassou from north-west Spain. J. Helminthol. 2005, 79, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Molina-Fernández, D.; Rubio-Calvo, D.; Adroher, F.J.; Benítez, R. Molecular epidemiology of Anisakis spp. in blue whiting Micromesistius poutassou in eastern waters of Spain, western Mediterranean Sea. Int. J. Food Microbiol. 2018, 282, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Ojeda-Torrejón, A.J.; Collado-Jara, A.; Salasváxquez, M. Investigación de parásitos en bacaladillascapturadas en la bahía de Cádiz. Med. Gen. 2001, 38, 808–811. [Google Scholar]
- Marcogliese, D.J. Geographic and temporal variations in levels of anisakid nematode larvae among fishes in the Gulf of St. Lawrence, eastern Canada. Can. Tech. Rep. Fish. Aquat. Sci. 1995, 2029, 1–16. [Google Scholar]
- Pascual, S.; Rodríguez, H.; Pierce, G.J.; Hastie, L.C.; González, A.F. The NE Atlantic European hake: A neglected high exposure risk for zoonotic parasites in European fish markets. Fish. Res. 2018, 202, 69–78. [Google Scholar] [CrossRef]
- Cipriani, P.; Sbaraglia, G.L.; Paoletti, M.; Giulietti, L.; Bellisario, B.; Palomba, M.; Bušelić, I.; Mladineo, I.; Nascetti, G.; Mattiucci, S. The Mediterranean European hake, Merluccius merluccius: Detecting drivers influencing the Anisakis spp. larvae distribution. Fish. Res. 2018, 202, 78–89. [Google Scholar] [CrossRef]
- Cruz, C.; Vaz, A.; Saraiva, A. Larval anisakids from horse mackerel in Portugal. Helminthologia 2005, 42, 3–7. [Google Scholar]
- Abattouy, N.; Valero, A.; Lozano, J.; Benajiba, N.H.; Martín-Sánchez, J. Epidemiology and molecular identification of Anisakis pegreffii (Nematoda: Anisakidae) in the horse mackerel Trachurus trachurus from northern Morocco. J. Helminthol. 2014, 88, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Shawket, N.; El Aasri, A.; Elmadhi, Y.; M’Bareck, I.; El Kharrim, K.; Belghyti, D. Anisakis simplex (Nematoda: Anisakidae) from horse mackerel (Trachurus trachurus) in Atlantic coast of Morocco. Asian Pac. J. Trop. Dis. 2017, 7, 463–466. [Google Scholar] [CrossRef]
- Madrid, E.; Gil, F.; García, M.; Debenedetti, A.L.; Trelis, M.; Fuentes, M. Potential risk analysis of human anisakiasis through the consumption of mackerel, Scomber scombrus, sold at Spanish supermarkets. Food Control 2016, 66, 300–305. [Google Scholar] [CrossRef]
- Eissa, A.E.; Showehdib, M.L.; Ismailc, M.M.; El-Naasd, A.S.; Abu-Mharae, A.A.; Abolghaitf, S.K. Identification and prevalence of Anisakis pegreffii and A. pegreffii × A. simplex (s.s.) hybrid genotype larvae in Atlantic horse mackerel (Trachurus trachurus) from some North African Mediterranean coasts. Egypt. J. Aquat. Res. 2018, 44, 21–27. [Google Scholar] [CrossRef]
- Madrid, E.; Galán-Puchades, M.T.; Fuentes, M. Risk analysis of human anisakidosis through the consumption of the blue whiting, Micromesistius poutassou, sold at Spanish supermarkets. Foodborne Pathog. Dis. 2012, 9, 934–938. [Google Scholar] [CrossRef]
- Ministerio de Agricultura, Alimentación y Medio Ambiente. Informe Sobre Abastecimiento de Pescado Blanco. 2015. Available online: https://www.mapa.gob.es/va/pesca/temas/mercados-economia-pesquera/informesobreabastecimientodepescadoblanco_tcm39-290622.pdf (accessed on 8 December 2018).
- Shamsi, S.; Butcher, A.R. First report of human anisakidosis in Australia. Med. J. Aust. 2011, 194, 199–200. [Google Scholar] [PubMed]
- Galeano, N.A. Evaluación del Potencial Zoonótico de Contracaecum spp. (Nematoda: Anisakidae) e Hysterothylacium spp. (Nematoda: Raphidascarididae) Como Agentes de Anisakidosis Humana. Ph.D. Thesis, Universidad Nacional del Sur, Bahía Blanca, Argentina, 2017. [Google Scholar]
- Gutiérrez-Galindo, J.F.; Osanz-Mur, A.C.; Moraventura, M.T. Occurrence and infection dynamics of anisakid larvae in Scomber scombrus, Trachurus trachurus, Sardina pilchardus, and Engraulis encrasicolus from Tarragona (NE Spain). Food Control 2010, 21, 1550–1555. [Google Scholar] [CrossRef]
- Serracca, L.; Cencetti, E.; Battistini, R.; Rossini, I.; Prearo, M.; Pavoletti, E.; Fioravanti, M.L.; Righetti, M.; Di Donfrancesco, B.; Ercolini, C. Survey on the presence of Anisakis and Hysterothylacium larvae in fishes and squids caught in Ligurian Sea. Vet. Parasitol. 2013, 196, 547–551. [Google Scholar] [CrossRef] [PubMed]
- Barcala, E.; Ramilo, A.; Ortega, N.; Picó, G.; Abollo, E.; Pascual, S.; Muñoz, P. Occurrence of Anisakis and Hysterothylacium larvae in commercial fish from Balearic Sea (Western Mediterranean Sea). Parasitol. Res. 2018, 117, 4003–4012. [Google Scholar] [CrossRef] [PubMed]
- Debenedetti, A.L.; Madrid, E.; Fuentes, M.V. Study of helminth parasites in the red mullet, Mullus barbatus, from the Mediterranean Sea and acquired in greater València, Spain. Rev. Ibero-Latinoam. Parasitol. 2010, 72, 118–123. [Google Scholar]
- Suzuki, J.; Murata, R.; Hosaka, M.; Araki, J. Risk factors for human Anisakis infection and association between the geographic origins of Scomber japonicus and anisakid nematodes. Int. J. Food Microbiol. 2010, 137, 88–93. [Google Scholar] [CrossRef]
- Costa, G.; Pontes, T.; Mattiucci, S.; D’Amélio, S. The occurrence and infection dynamics of Anisakis larvae in the black-scabbard fish, Aphanopus carbo, chub mackerel, Scomber japonicus, and oceanic horse mackerel, Trachurus picturatus from Madeira, Portugal. J. Helminthol. 2003, 77, 163–166. [Google Scholar] [CrossRef]
- Rello, F.J.; Adroher, F.J.; Valero, A. Hysterothylacium aduncum, the only anisakid parasite of sardines (Sardina pilchardus) from the southern and eastern coasts of Spain. Parasitol. Res. 2008, 104, 117–121. [Google Scholar] [CrossRef]
- Sanmartín, M.L.; Quinteiro, P.; Iglesias, R.; Santamaría, M.T.; Leiro, J.; Ubeira, F.M. Nematodos Parásitos de Peces de las Costas Gallegas; Ediciones Díaz Santos: Madrid, Spain, 1994; p. 88. [Google Scholar]
- Cavallero, S.; Magnabosco, C.; Civettini, M.; Boffo, L.; Mingarelli, G.; Buratti, P.; Giovanardi, O.; Fortuna, C.M.; Arcangeli, G. Survey of Anisakis sp. and Hysterothylacium sp. in sardines and anchovies from the North Adriatic Sea. Int. J. Food. Microbiol. 2015, 200, 18–21. [Google Scholar] [CrossRef]
- López-Vélez, R.; García, A.; Barros, C.; Manzarbeitia, F.; Oñate, J.M. Anisakiasis in Spain. Report of 3 cases. Enferm. Infecc. Microbiol. Clin. 1992, 10, 158–161. [Google Scholar]
- Berger, S. Infectious Diseases of Spain; Gideon Informatics: Los Angeles, CA, USA, 2018; pp. 27–28. [Google Scholar]
- De Liberato, C.; Bossù, T.; Scaramozzino, P.; Ncolini, G.; Ceddia, P.; Mallozzi, S.; Cavallero, S.; D’Amelio, S. Presence of anisakid larvae in the European anchovy, Engraulis encrasicolus, fished off the Tyrrhenian coast of central Italy. J. Food Prot. 2013, 76, 1643–1648. [Google Scholar] [CrossRef]
- Cipriani, P.; Sbaraglia, G.L.; Palomba, M.; Giulietti, L.; Bellisario, B.; Bušelić, I.; Mladineo, I.; Cheleschi, R.; Nascetti, G.; Mattiucci, S. Anisakis pegreffii (Nematoda: Anisakidae) in the European anchovy Engraulis encrasicolus, from the Mediterranean Sea: Fishing ground as a predictor of the parasite distribution. Fish. Res. 2018, 202, 59–68. [Google Scholar] [CrossRef]
- Rello, F.J.; Adroher, F.J.; Benítez, R.; Valero, A. The fishing area as a possible indicator of the infection by anisakids in anchovies (Engraulis encrasicolus) from southwestern Europe. Int. J. Food Microbiol. 2009, 129, 277–281. [Google Scholar] [CrossRef] [PubMed]
- Gazzonis, A.L.; Cavallero, S.; Zanzani, S.A.; Olivieri, E.; Malandra, R.; Ranghieri, V.; D’Amelio, S.; Manfredi, M.T. Anisakis sp. and Hysterothylacium sp. larvae in anchovies (Engraulis encrasicolus) and chub mackerel (Scomber colias) in the Mediterranean Sea: Molecular identification and risk factors. Food Control 2017, 80, 366–373. [Google Scholar] [CrossRef]
- Rodríguez-Domínguez, H.; González, A.F.; Abollo, E.; Pascual, S. Re-evaluation of anchovies (Engraulis encrasicolus) as an important risk factor for sensitization to zoonotic nematodes in Spain. Fish. Res. 2018, 202, 49–58. [Google Scholar] [CrossRef]
- Abaunza, P.; Villamor, B.; Perez, J.R. Infestation by larvae of Anisakis simplex (Nematoda: Ascaridata) in horse mackerel, Trachurus trachurus, and Atlantic mackerel, Scomber scombrus, in ICES Divisions VIIIb, VIIIc and Ixa (N-NW of Spain). Sci. Mar. 1995, 59, 223–233. [Google Scholar]
- Silva, M.; Eiras, J. Occurrence of Anisakis sp. in fishes off the Portuguese West coast and evaluation of its zoonotic potential. bull. Eur. Assoc. Fish Pathol. 2003, 23, 13–17. [Google Scholar]
- Abollo, E.; Gestal, C.; Pascual, S. Anisakis infestation in marine fish and cephalopods from Galician waters: An updated perspective. Parasitol. Res. 2001, 87, 492–499. [Google Scholar] [CrossRef]
- Adroher, F.J.; Valero, A.; Ruiz-Valero, J.; Iglesias, L. Larval anisakids (Nematoda: Ascaridoidea) in horse mackerel (Trachurus trachurus) from the fish market in Granada (Spain). Parasitol. Res. 1996, 82, 253–256. [Google Scholar] [CrossRef]
- Valero, A.; López-Cuello, M.M.; Benítez, R.; Adroher, F.J. Anisakis spp. in European hake, Merluccius merluccius (L.) from the Atlantic off north-west Africa and the Mediterranean off southern Spain. Acta Parasitol. 2006, 51, 209–212. [Google Scholar] [CrossRef]
- Soric, R.E.; Miramontes-Sequeiros, L.C.; Miron, L. Palanca-Soler, A. Morphometrics for Anisakiasis diagnosis in the North Atlantic horse mackerel (Trachurus trachurus). Sci. Parasitol. 2012, 12, 77–81. [Google Scholar]
- Strømnes, E.; Andersen, K. “Spring rise” of whaleworm (Anisakis simplex; Nematoda, Ascaridoidea) thirdstage larvae in some fish species from Norwegian waters. Parasitol. Res. 2000, 86, 619–624. [Google Scholar] [CrossRef] [PubMed]
- De la Torre Molina, R.; Pérez Aparicio, J.; Hernández-Bienes, M.; Jurado-Pérez, R.; Martínez Ruso, A.; Morales Franco, E. Anisakiasis in fresh fish sold in the north of Córdoba. Revista Espanola Salud Publica 2000, 74, 517–526. [Google Scholar]
- Gómez-Mateos, M.; Valero, A.; Morales-Yuste, M.; Martín-Sánchez, J. Molecular epidemiology and risk factors for Anisakis simplex s.l. infection in blue whiting (Micromesistius poutassou) in a confluence zone of the Atlantic and Mediterranean: Differences between A. simplex s.s. and A. pegreffii. Int. J. Food Microbiol. 2016, 232, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Nagasawa, K. The Life Cycle of Anisakis simplex: A Review. In Intestinal Anisakiasis in Japan; Ishikura, H., Kikuchi, K., Eds.; Springer: Tokyo, Japan, 2010; pp. 31–40. [Google Scholar]
- Cipriani, P.; Acerra, V.; Bellisario, B.; Sbaragli, G.L.; Cheleschi, R.; Nascetti, G.; Mattiucci, S. Larval migration of the zoonotic parasite Anisakis pegreffii (Nematoda: Anisakidae) in European anchovy, Engraulis encrasicolus: Implications to seafood safety. Food Control 2016, 59, 148–157. [Google Scholar] [CrossRef]
- Yagi, K.; Nagasawa, K.; Ishikura, H.; Nagasawa, A.; Sato, N.; Kikuchi, K.; Ishikura, H. Female worm Hysterothylacium aduncum excreted from human: A case report. Jpn. J Parasitol. 1996, 45, 12–23. [Google Scholar]
- González-Amores, Y.; Clavijo-Frutos, E.; Salas-Casanova, C.; Alcain-Martínez, G. Direct parasitologial diagnosis of infection with Hysterothylacium aduncum in a patient with epigastralgia. Revista Espanola Enfermedades Digestivas 2015, 107, 699–700. [Google Scholar]
- Skovgaard, A.; Bahlool, Q.Z.M.; Munk, P.; Berge, T.; Buchmann, K. Infection of North Sea cod, Gadus morhua L., larvae with the parasitic nematode Hysterothylacium aduncum Rudolphi. J. Plankton Res. 2011, 33, 1311–1316. [Google Scholar] [CrossRef]
- Llarena-Reino, M.; Piñeiro, C.; Antonio, J.; Outeriño, L.; Vello, C.; González, A.F.; Pascual, S. Optimization of the pepsin digestion method for anisakids inspection in the fishing industry. Vet. Parasitol. 2013, 191, 276–283. [Google Scholar] [CrossRef]
- Grabda, J. Marine Fish Parasitology. An Outline; PWN-Polish scientific Publisher: Warszawa, Poland, 1991; p. 306. [Google Scholar]
- Anderson, R.C.; Chabaud, A.G.; Willmott, S. Keys to the Nematode Parasites of Vertebrates, Archival Volume; CAB International: Wallingford, England, 2009; p. 463. [Google Scholar]
- Gibbons, L.M. Keys to the Nematode Parasites of Vertebrates, Supplementari Volume; CAB International: Wallingford, England, 2010; p. 416. [Google Scholar]
- Bush, A.O.; Lafferty, K.D.; Lotz, J.M.; Shostak, A.W. Parasitology meets ecology on its own terms: Margolis et al. revisited. J. Parasitol. 1997, 83, 575–583. [Google Scholar] [CrossRef]
- Rózsa, L.; Reiczigel, J.; Majoros, G. Quantifying parasites in samples of hosts. J. Parasitol. 2000, 86, 228–232. [Google Scholar] [CrossRef]
- Shvydka, S.; Sarabeev, V.; Estruch, V.D.; Cadarso-Suárez, C. Optimum sample size to estimate mean parasite abundance in fish parasite surveys. Helminthologia 2018, 55, 52–59. [Google Scholar] [CrossRef]
| Fish Species | Total Sample | ||||
|---|---|---|---|---|---|
| N | Prevalence (%) | Mean Abundance | Range | Number of Larvae | |
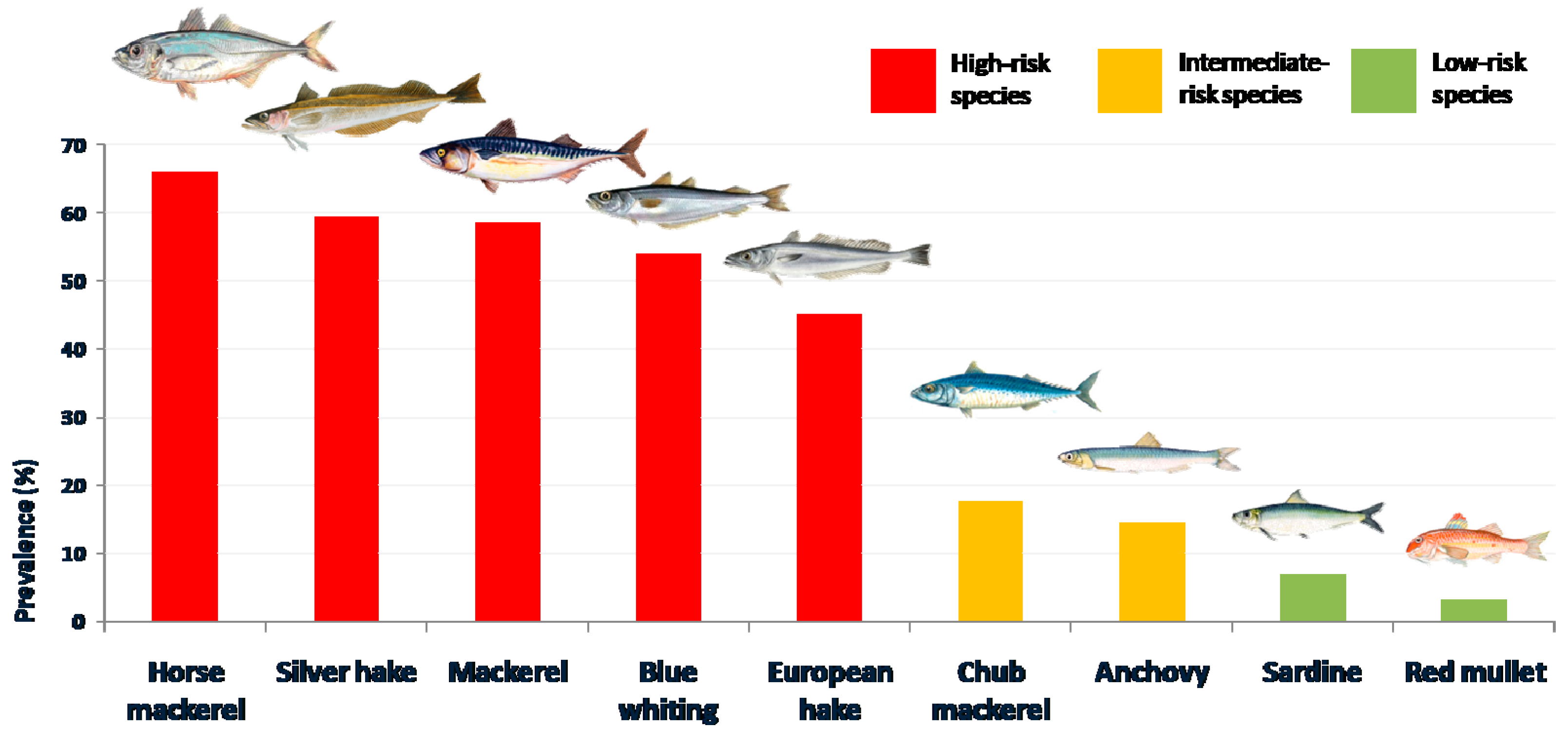
| Blue whiting (Micromesistius poutasou) | 153 | 53.87 | 3.87 (2.02–5.72) | 1–219 | 1098 |
| Hake (Merluccius merluccius) | 90 | 45.00 | 10.20 (2.66–17.73) | 1–574 | 2042 |
| Silver hake (Merlucccius bilinearis) | 69 | 59.48 | 1.42 (1.06–1.78) | 1–6 | 165 |
| Red mullet (Mulllus barbatus) | 5 | 3.23 | 0.05 (0.00–0.09) | 1–3 | 7 |
| Mackerel (Scomber scombrus) | 135 | 58.44 | 5.68 (3.67–7.68) | 1–198 | 1311 |
| Chub mackerel (Scomber japonicum) | 19 | 17.59 | 1.11 (0.00–2.85) | 1–95 | 120 |
| Horse mackerel (Trachurus trachurus) | 66 | 66.00 | 26.72 (16.29–35.73) | 1–341 | 2672 |
| Sardine (Sardina pilchardus) | 19 | 6.76 | 0.20 (0.06–033) | 1–16 | 55 |
| Anchovy (Engraulis encrasicolus) | 45 | 14.47 | 0.28 (0.16–0.39) | 1–13 | 86 |
| Total | 601 | 33.65 | 3.99 | 1–574 | 7123 |
| Fish Species | Atlantic Ocean | Mediterranean Sea | ||
|---|---|---|---|---|
| Prevalence (%) | Mean Abundance | Prevalence (%) | Mean Abundance | |
| Blue whiting (Micromesistius poutasou) | 77.51 | 4.83 (3.08–6.58) | 19.13 | 2.45 (0.00–6.25) |
| Hake (Merluccius merluccius) | 84.00 | 20.34 (5.40–35.28) | 6.00 | 0.05 (0.00–0.10) |
| Silver hake (Merlucccius bilinearis) | 59.48 | 1.42 (1.06–1.78) | - | - |
| Red mullet (Mulllus barbatus) | - | - | 3.23 | 0.05 (0.00–0.09) |
| Mackerel (Scomber scombrus) | 76.43 | 7.29 (4.16–10.41) | 30.77 | 3.20 (1.57–4.83) |
| Chub mackerel (Scomber japonicum) | 19.18 | 1.84 (0.00–5.12) | 14.29 | 0.17 (0.20–0.33) |
| Horse mackerel (Trachurus trachurus) | 100.00 | 52.50 (35.26–69.74) | 32.00 | 0.94 (0.43–1.45) |
| Sardine (Sardina pilchardus) | 11.43 | 0.34 (0.08–0.61) | 2.13 | 0.05 (0.00–0.12) |
| Anchovy (Engraulis encrasicolus) | 18.87 | 0.43 (0.21–0.66) | 9.87 | 0.11 (0.05–0.17) |
| Total | 52.90 | 4.56 | 11.92 | 0.79 |
| Fish Species | Prevalence (%) | Mean Abundance | ||
|---|---|---|---|---|
| Viscera | Flesh | Viscera | Flesh | |
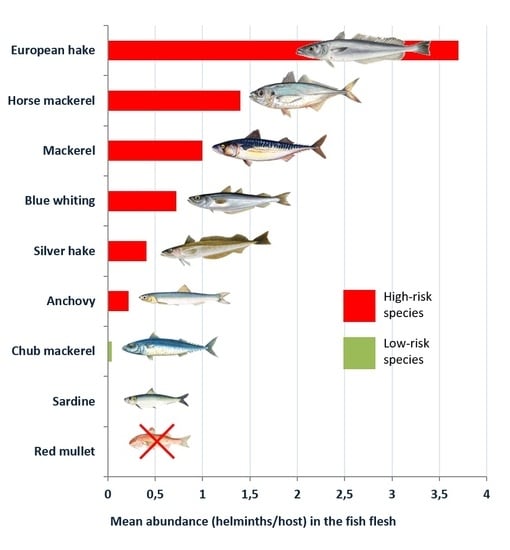
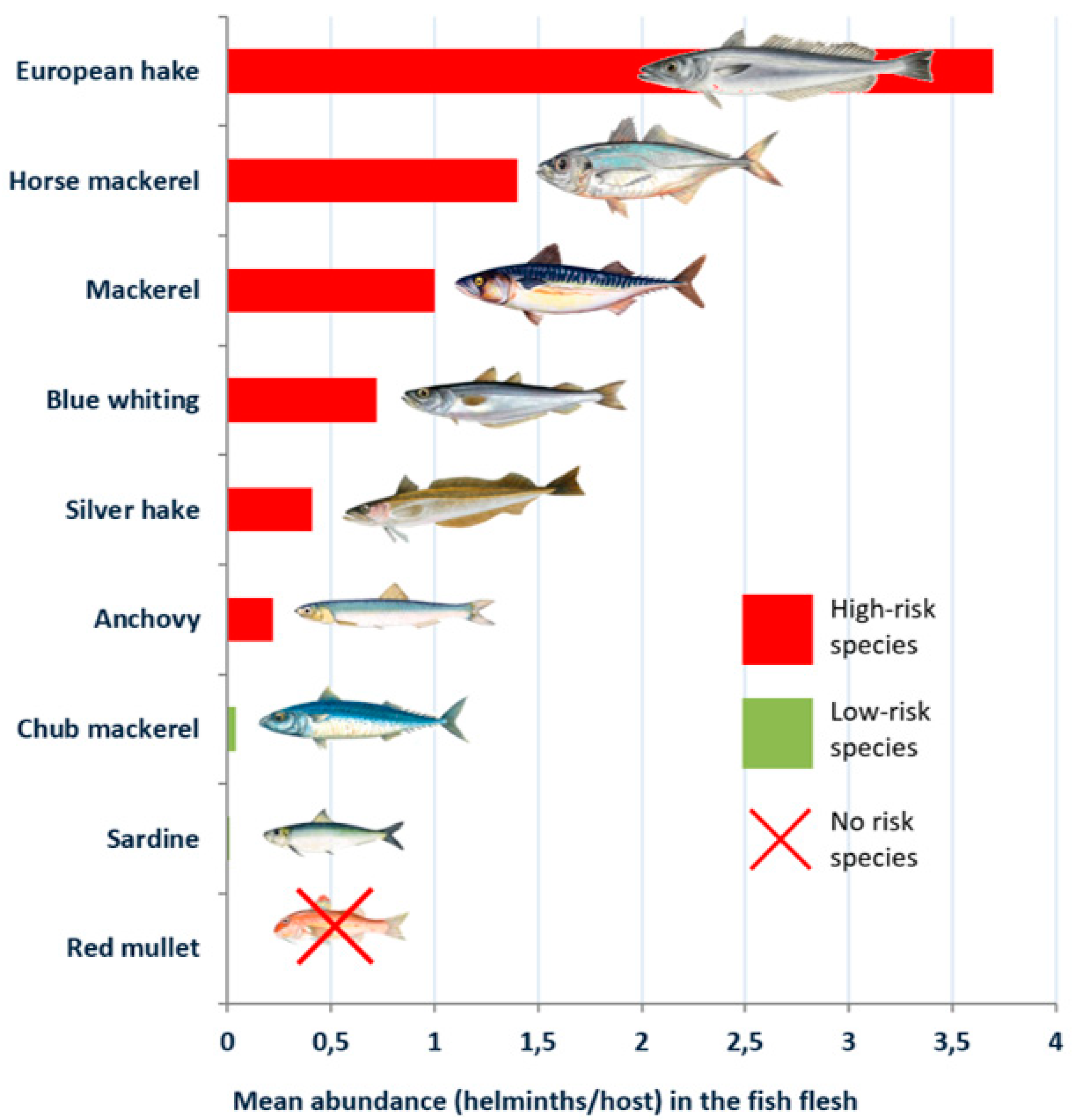
| Blue whiting (Micromesistius poutasou) | 49.60 | 26.10 | 3.15 (1.54–4.75) | 0.72 (0.40–1.03) |
| Hake (Merluccius merluccius) | 35.00 | 29.50 | 6.50 (1.15–11.84) | 3.70 (1.51–5.99) |
| Silver hake (Merlucccius bilinearis) | 46.60 | 28.40 | 1.02 (0.71–1.32) | 0.41 (0.25–0.56) |
| Red mullet (Mulllus barbatus) | 3.23 | 0.00 | 0.05 (0.00–0.09) | 0.00 |
| Mackerel (Scomber scombrus) | 55.85 | 25.98 | 4.65 (3.07–6.23) | 1.03 (0.48–1.57) |
| Chub mackerel (Scomber japonicum) | 16.70 | 2.80 | 1.07 (0.00–2.78) | 0.04 (0.00–0.08) |
| Horse mackerel (Trachurus trachurus) | 60.00 | 28.00 | 26.72 (16.81–36.63) | 1.40 (1.05–1.75) |
| Sardine (Sardina pilchardus) | 5.34 | 2.14 | 0.17 (0.04–0.31) | <0.01 (0.00–0.04) |
| Anchovy (Engraulis encrasicolus) | 11.90 | 4.18 | 0.26 (0.12–0.34) | 0.22 (0.09–0.38) |
| Fish Species | Prevalence (%) | Mean Abundance | ||
|---|---|---|---|---|
| Autumn | Spring | Autumn | Spring | |
| Blue whiting (Micromesistius poutasou) | 47.80 | 59.30 | 4.35 (2.13–6.57) | 3.43 (0.53–6.34) |
| Hake (Merluccius merluccius) | 7.50 | 54.40 | 0.36 (0.00–0.74) | 12.81 (3.29–22.33) |
| Silver hake (Merlucccius bilinearis) | 64.30 | 55.00 | 1.75 (1.15–2.35) | 1.12 (0.70–1.53) |
| Red mullet (Mulllus barbatus) | 5.30 | 1.20 | 0.08 (0.00–0.17) | 0.01 (0.00–0.04) |
| Mackerel (Scomber scombrus) | 62.35 | 56.16 | 4.47 (3.05–5.88) | 7.75 (2.85–12.66) |
| Chub mackerel (Scomber japonicum) | 19.20 | 22.00 | 1.84 (0.00–5.12) | 0.26 (0.11–0.41) |
| Sardine (Sardina pilchardus) | 3.19 | 8.70 | 0.07 (0.00–0.18) | 0.26 (0.06–0.46) |
| Anchovy (Engraulis encrasicolus) | 7.69 | 17.28 | 0.22 (0.00–0.51) | 0.30 (0.18–0.42) |
| Fish Species | N | Atlantic | Mediterranean | Autumn | Spring |
|---|---|---|---|---|---|
| Blue whiting (Micromesistius poutasou) | 284 | 169 | 115 | 134 | 150 |
| Hake (Merluccius merluccius) | 200 | 100 | 100 | 40 | 160 |
| Silver hake (Merlucccius bilinearis) | 116 | 116 | - | 56 | 60 |
| Red mullet (Mulllus barbatus) | 155 | - | 155 | 75 | 80 |
| Mackerel (Scomber scombrus) | 231 | 140 | 91 | 85 | 146 |
| Chub mackerel (Scomber japonicum) | 108 | 73 | 35 | 58 | 50 |
| Horse mackerel (Trachurus trachurus) | 100 | 50 | 50 | - | 100 |
| Sardine (Sardina pilchardus) | 281 | 140 | 141 | 94 | 187 |
| Anchovy (Engraulis encrasicolus) | 311 | 159 | 152 | 91 | 220 |
| Total | 1786 | 947 | 839 | 657 | 1129 |
| Fish Species | Weight Range (g) | Length Range (cm) | Days Range |
|---|---|---|---|
| Blue whiting (Micromesistius poutasou) | 17.58–145.20 | 14.50–28.60 | 1–8 |
| Hake (Merluccius merluccius) | 43.63–549.6 | 19.40–41.50 | 1–6 |
| Silver hake (Merlucccius bilinearis) | 51.44–215.92 | 19.00–32.50 | 1–8 |
| Red mullet (Mulllus barbatus) | 11.24–85.60 | 10.60–21.00 | 1–5 |
| Mackerel (Scomber scombrus) | 29.96–466.80 | 16.60–25.70 | 1–6 |
| Chub mackerel (Scomber japonicum) | 62.72–511.80 | 15.00–39.20 | 1–6 |
| Horse mackerel (Trachurus trachurus) | 16.10–37.00 | 29.90–360.40 | 1–7 |
| Sardine (Sardina pilchardus) | 8.80–120.61 | 10.60–25.70 | 1–4 |
| Anchovy (Engraulis encrasicolus) | 7.10–39.10 | 10.50–18.20 | 1–5 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Debenedetti, Á.L.; Madrid, E.; Trelis, M.; Codes, F.J.; Gil-Gómez, F.; Sáez-Durán, S.; Fuentes, M.V. Prevalence and Risk of Anisakid Larvae in Fresh Fish Frequently Consumed in Spain: An Overview. Fishes 2019, 4, 13. https://doi.org/10.3390/fishes4010013
Debenedetti ÁL, Madrid E, Trelis M, Codes FJ, Gil-Gómez F, Sáez-Durán S, Fuentes MV. Prevalence and Risk of Anisakid Larvae in Fresh Fish Frequently Consumed in Spain: An Overview. Fishes. 2019; 4(1):13. https://doi.org/10.3390/fishes4010013
Chicago/Turabian StyleDebenedetti, Ángela L., Elena Madrid, María Trelis, Francisco J. Codes, Florimar Gil-Gómez, Sandra Sáez-Durán, and Màrius V. Fuentes. 2019. "Prevalence and Risk of Anisakid Larvae in Fresh Fish Frequently Consumed in Spain: An Overview" Fishes 4, no. 1: 13. https://doi.org/10.3390/fishes4010013
APA StyleDebenedetti, Á. L., Madrid, E., Trelis, M., Codes, F. J., Gil-Gómez, F., Sáez-Durán, S., & Fuentes, M. V. (2019). Prevalence and Risk of Anisakid Larvae in Fresh Fish Frequently Consumed in Spain: An Overview. Fishes, 4(1), 13. https://doi.org/10.3390/fishes4010013