Direct Recycling R&D at the ReCell Center
Abstract
1. Introduction
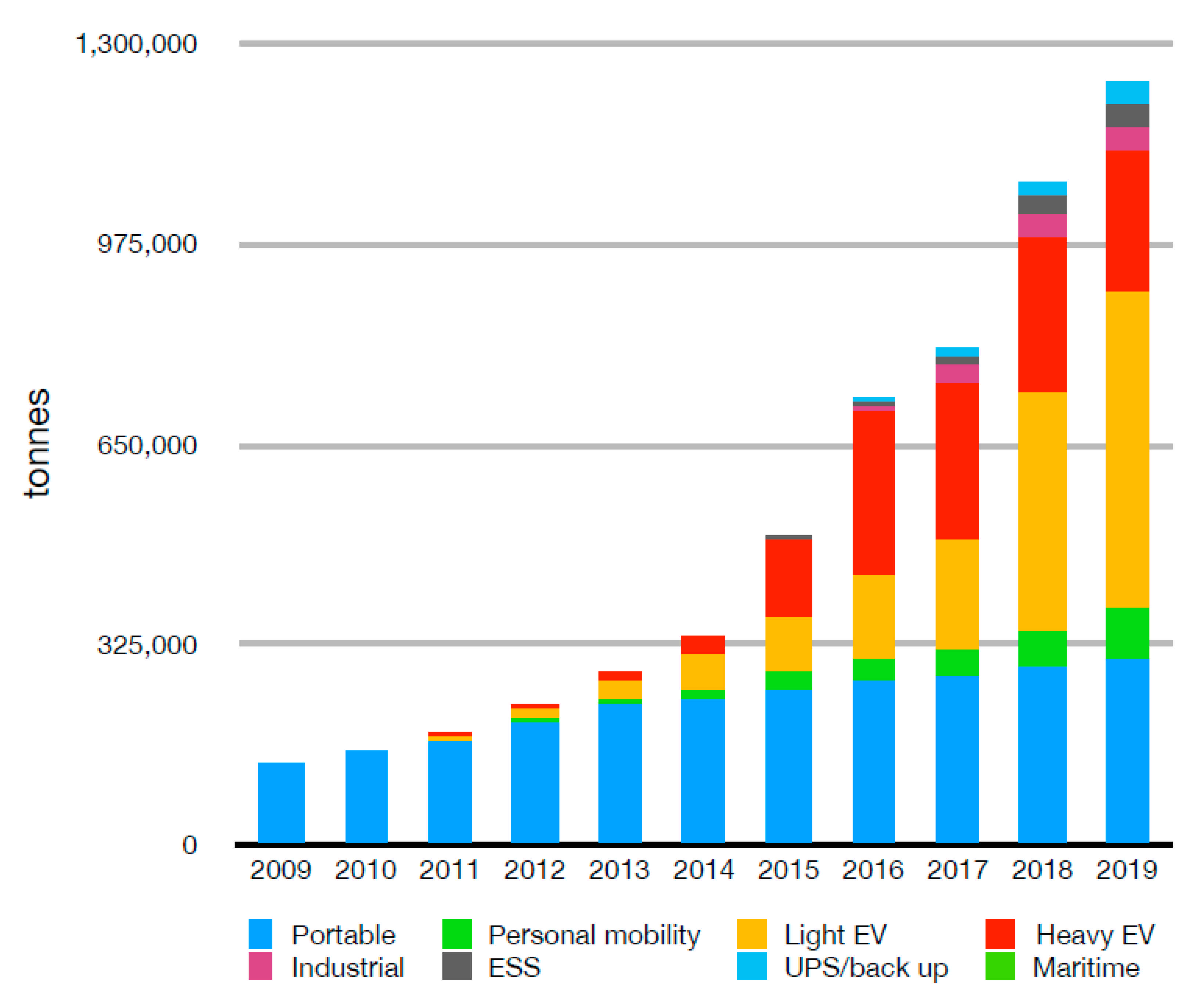
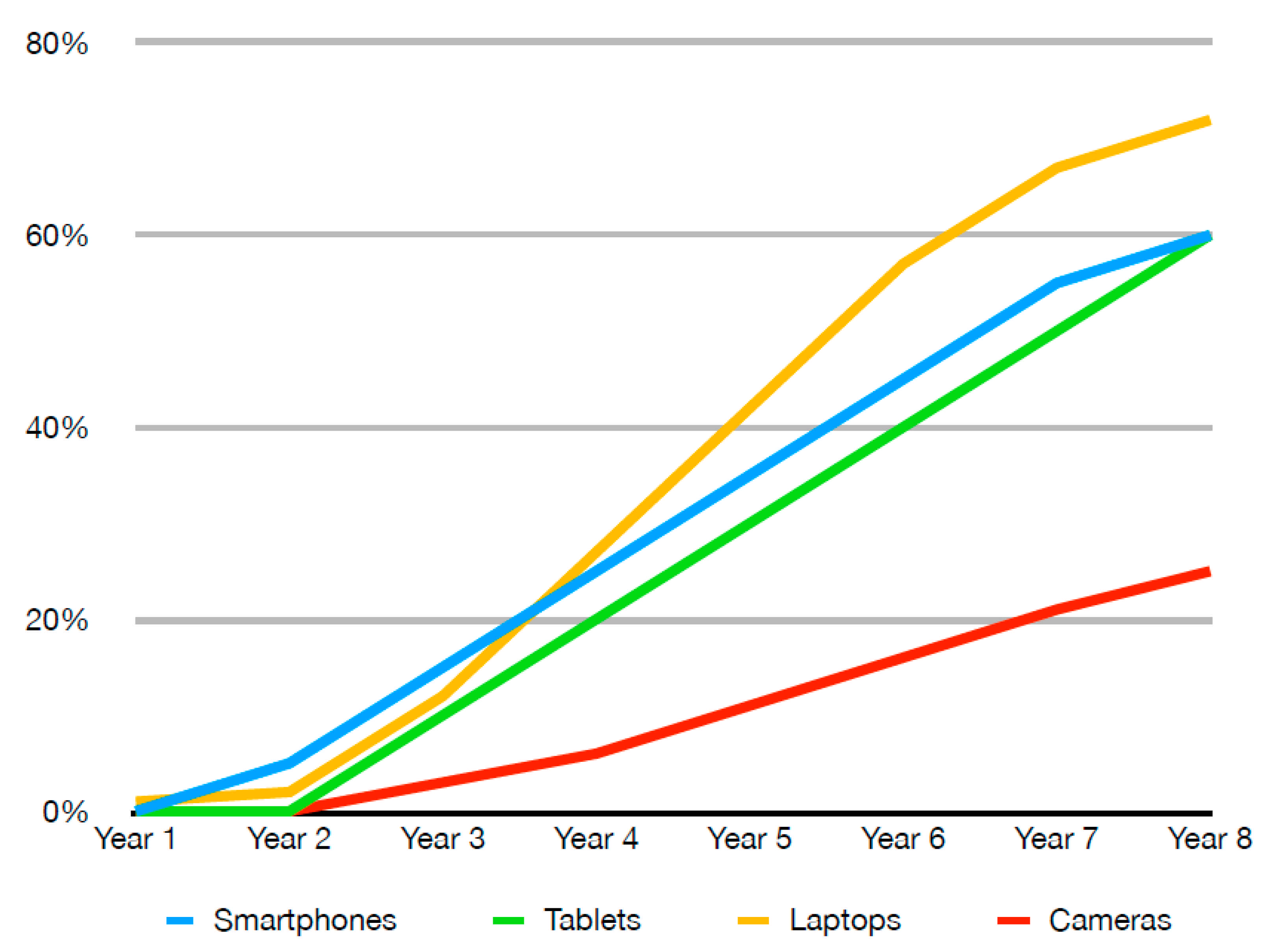
2. Material Available for Recycling
2.1. End-of-Life Batteries
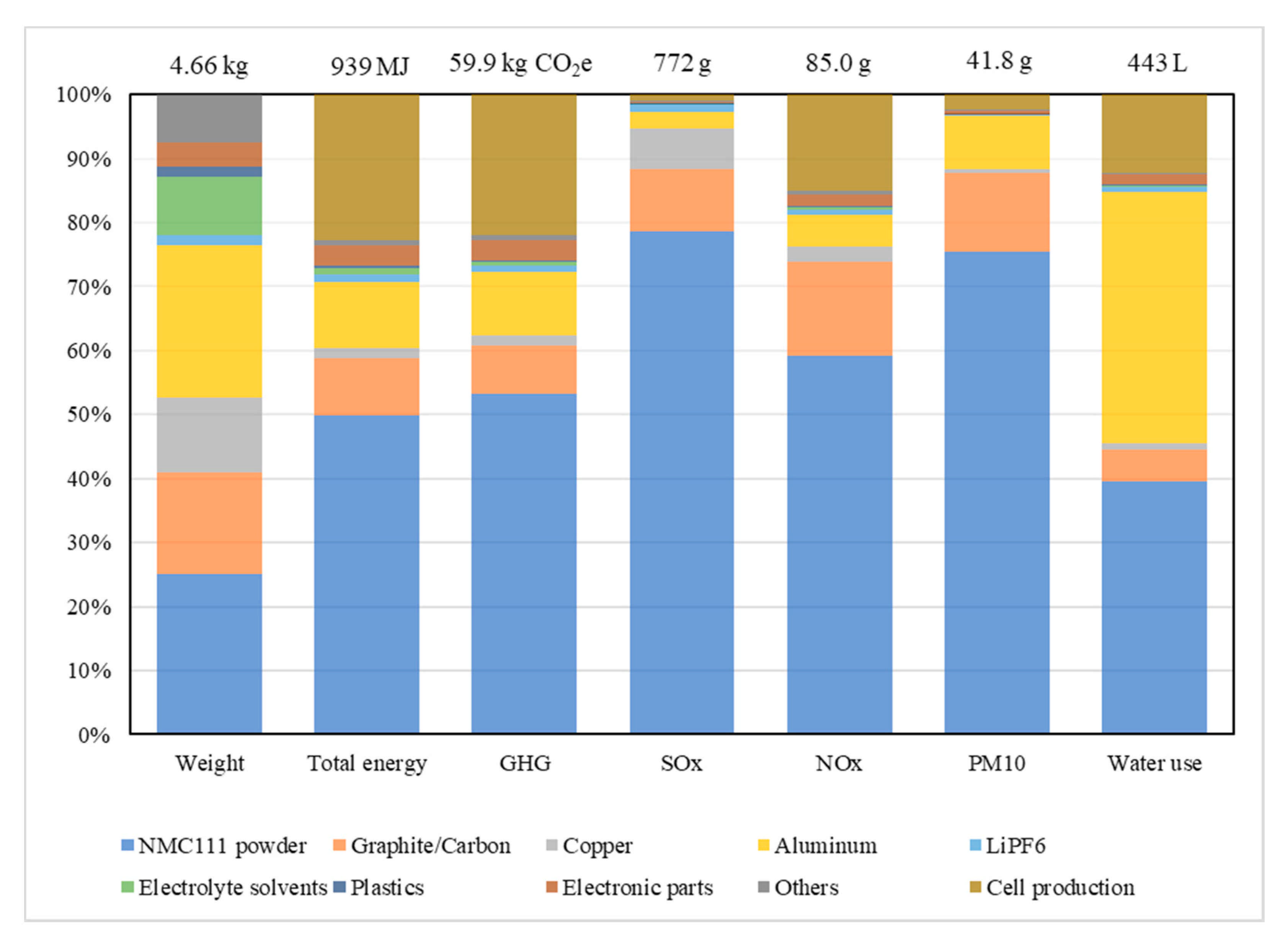
2.2. Production Scrap
2.3. Exports
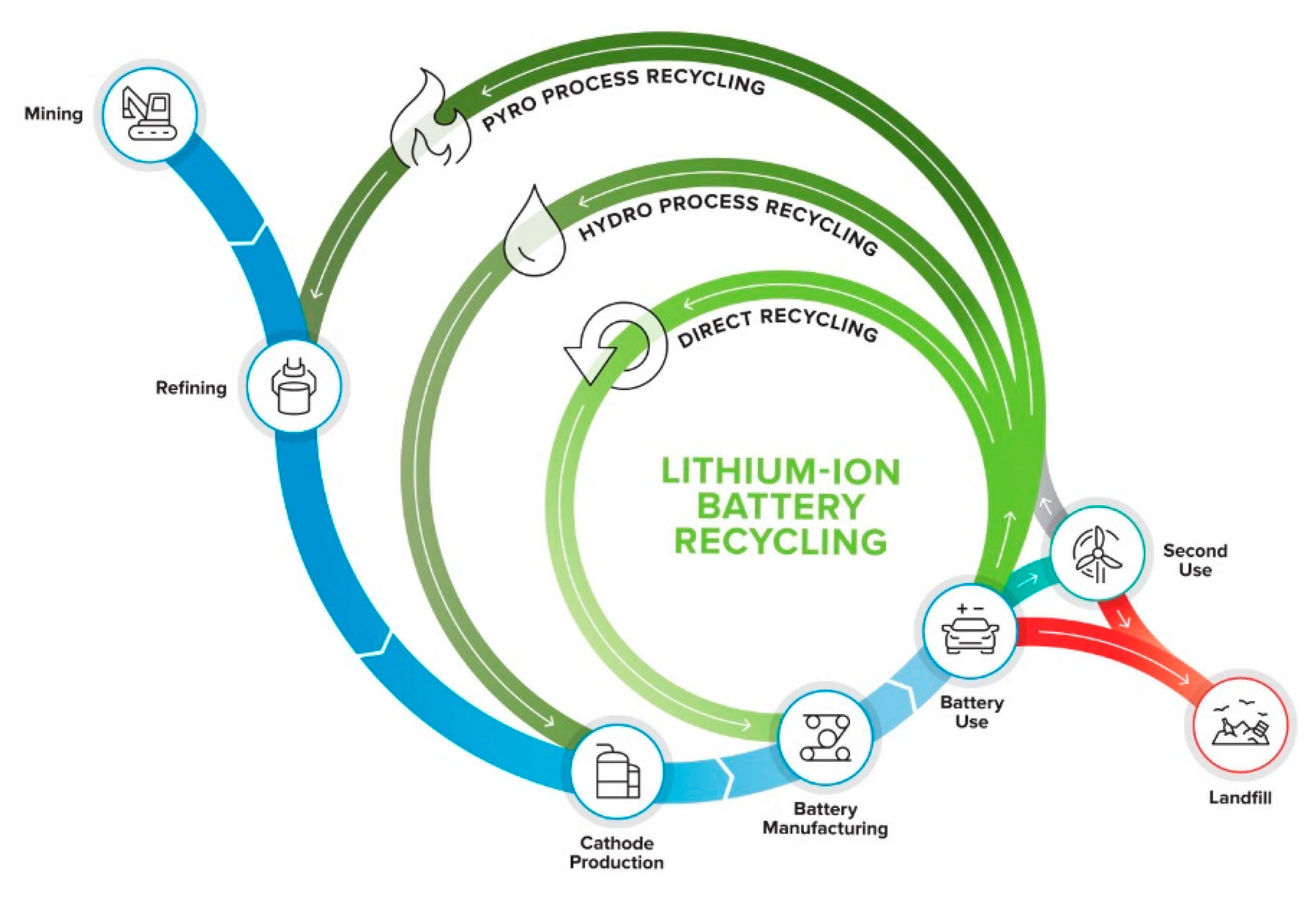
3. Recycling Processes and the Circular Economy
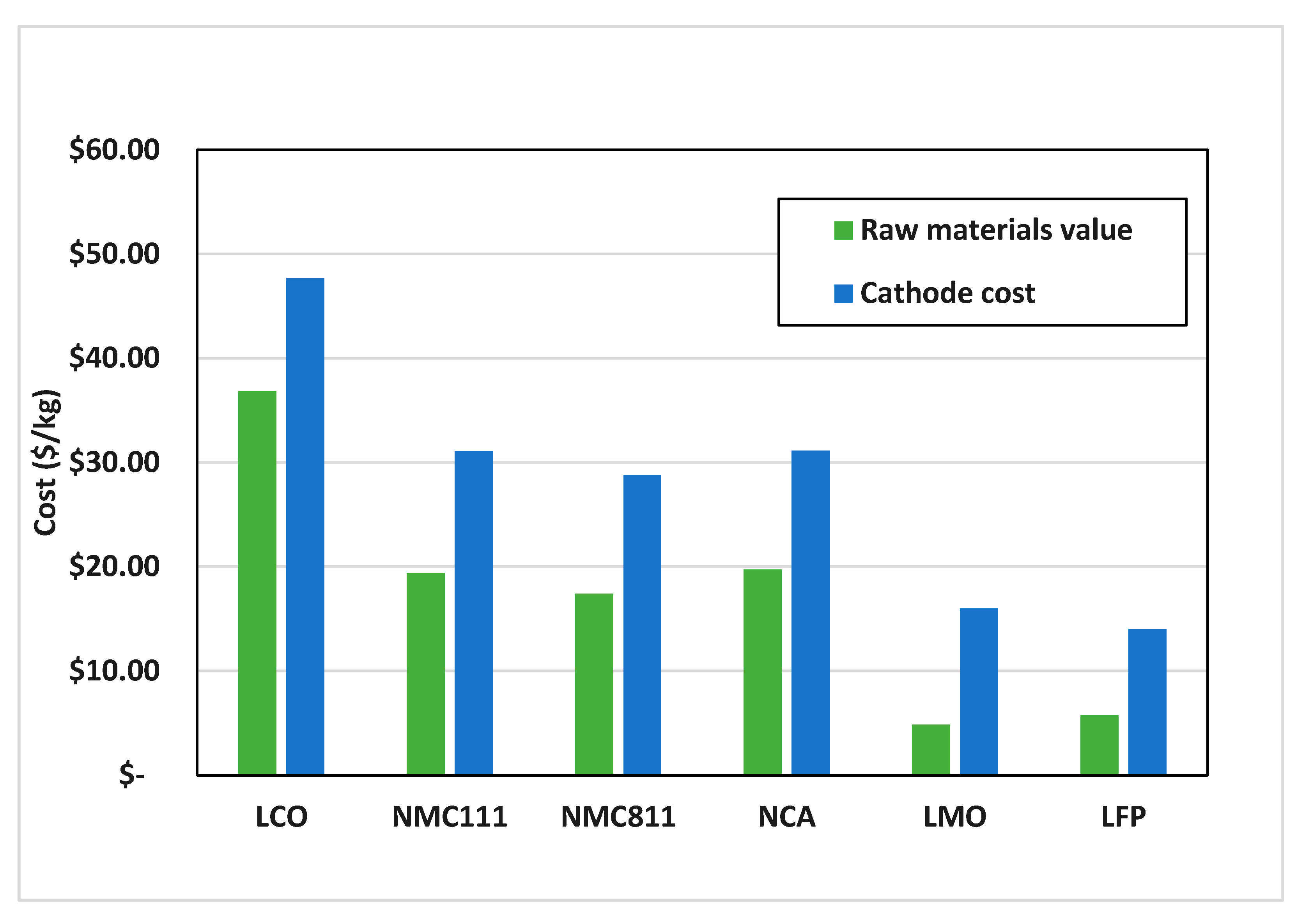
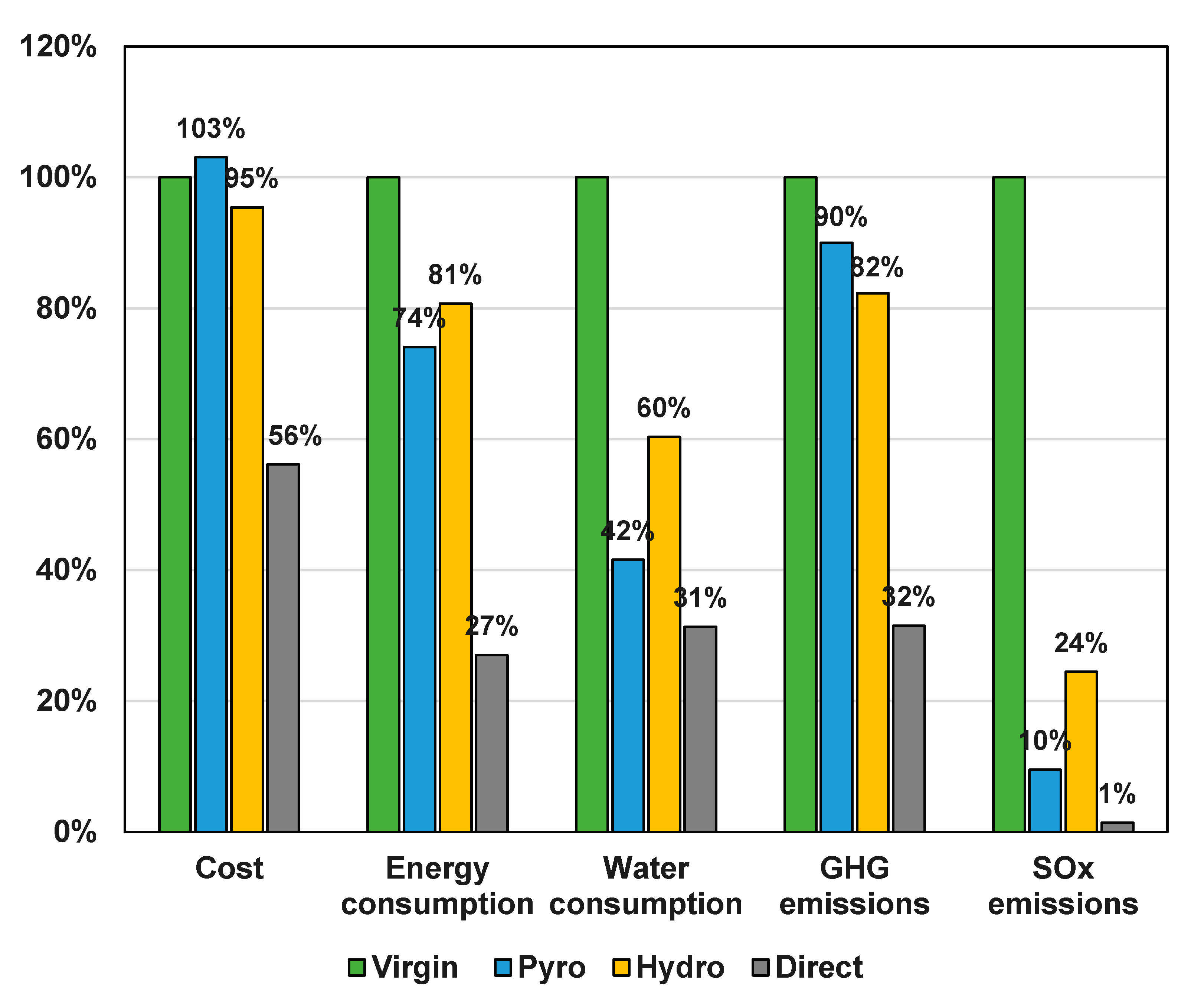
3.1. Recycling Process Comparison
3.2. Recycling Process Comparison and the EverBatt Model
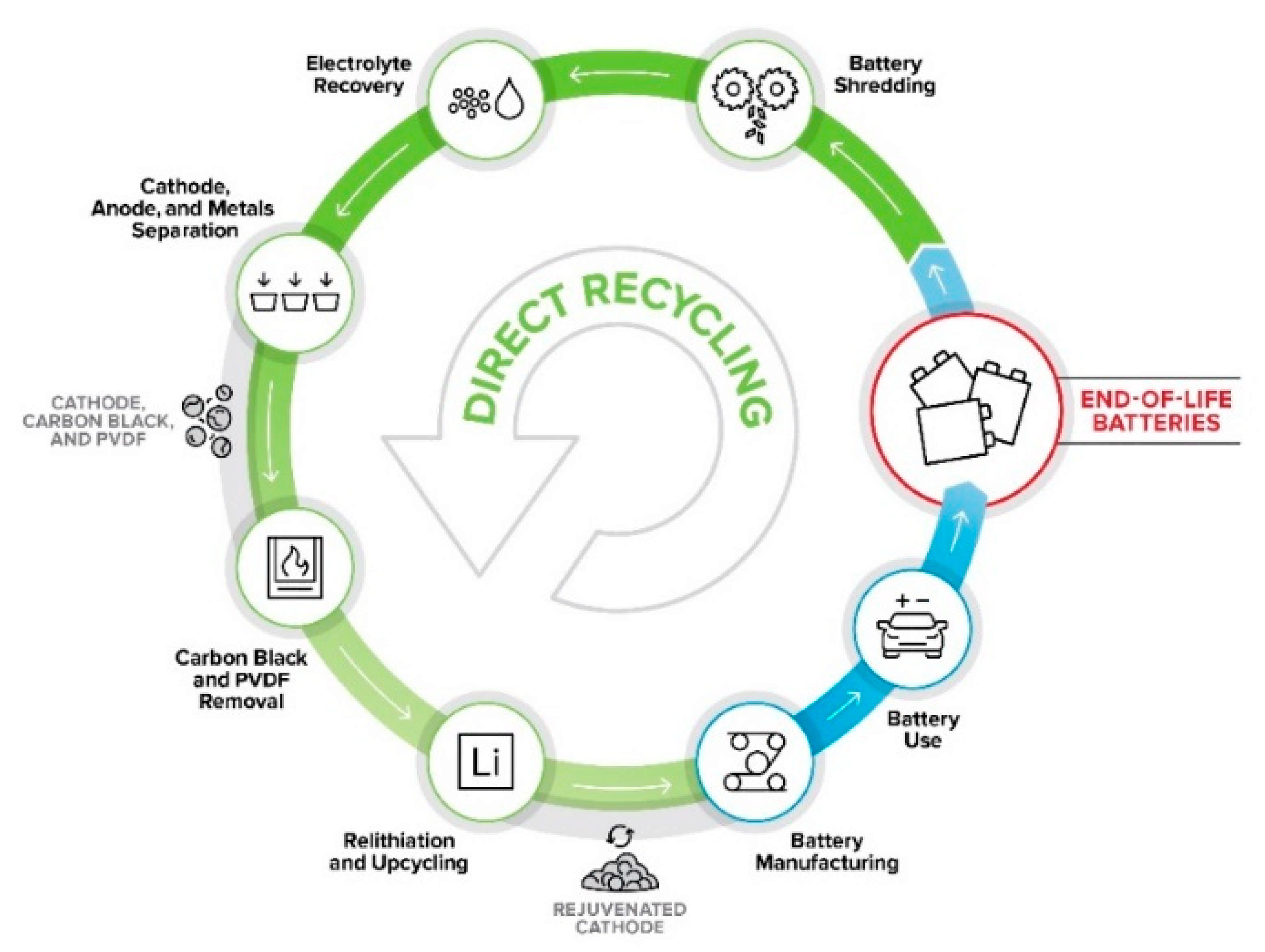
4. Direct Recycling Process Development
4.1. Preprocessing
4.1.1. Collection, Transportation, and Sorting
4.1.2. Disassembly
4.1.3. Size Reduction
4.2. Separation Processes
- Solvent extraction/dissolution;
- Precipitation;
- State or property change with temperature;
- Chemical replacement or addition;
- Gravity separation;
- Distillation;
- Froth flotation;
- Surface modification;
- Electrochemical processing;
- Microwave, ultrasonics, or other irradiation.
4.2.1. Electrolyte Recovery
4.2.2. Delamination: Separation of Active Materials from Current Collectors
4.2.3. Anode–Cathode and Cathode–Cathode Separation by Froth Flotation
4.2.4. Binder Removal and Recovery
4.3. Product Upgrading
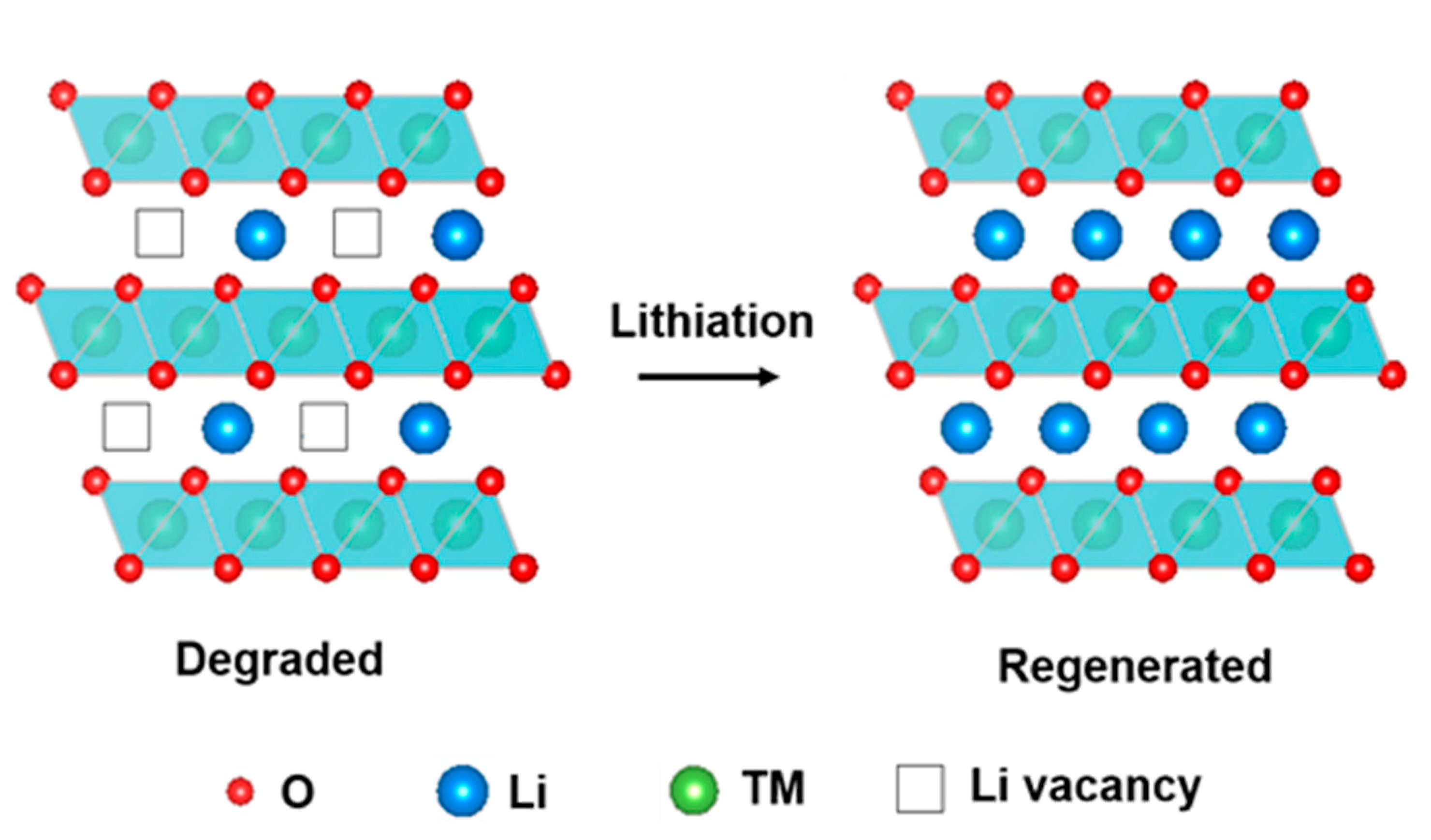
4.3.1. Relithiation
4.3.2. Upcycling
4.4. Requirements for Industry Acceptance
5. Looking Forward
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A. Supporting Facilities Are Crucial
References
- Energy Department Announces Battery Recycling Prize and Battery Recycling R&D Center. Available online: https://www.energy.gov/articles/energy-department-announces-battery-recycling-prize-and-battery-recycling-rd-center (accessed on 7 January 2021).
- Circular Energy Storage Research and Consulting. The Lithium-ion Battery Life Cycle Report 2021; Circular Energy Storage Research and Consulting: London, UK, 2020. [Google Scholar]
- Smith, C. (Call2Recycle, Atlanta, GA, USA). Personal communication, 2020.
- New-Eichlin, D.; Spalding, D. (Battery Solutions, Wixom, MI, USA). Personal communication, 2020.
- Sanders, M. (Avicenne Energy, Landenberg, PA, USA). Personal communication, 2020.
- Tesla Battery Day Preview—Redwood Materials. Available online: https://www.youtube.com/watch?v=EWRvmFR8vKc (accessed on 1 January 2021).
- Wood, C. Amazon, Panasonic, And Recycling Start-Ups Prepare for Battery Wave tsunami. CNBC.com, 12 December 2020. (updated on 14 December 2020). Available online: https://www.cnbc.com/2020/12/12/amazon-panasonic-preparing-for-demand-of-battery-recycling.html (accessed on 1 January 2021).
- Argus. Explosion Hits CATL Battery Precursors Plant in China. Available online: https://www.argusmedia.com/en/news/2175179-explosion-hits-catl-battery-precursors-plant-in-china#:~:text=An%20explosion%20has%20hit%20a,Contemporary%20Amperex%20Technology%20(CATL).&text=The%20explosion%20was%20caused%20after,garbage%20dump%2C%20Hunan%20Brunp%20said (accessed on 8 January 2021).
- Melin, H.E. The Dynamics of the EV Battery End-Of-Life Market (Zoom Tutorial); Circular Energy Storage: London, UK, 22 October 2020. [Google Scholar]
- Herox. Lithium-Ion Battery Recycling Prize. Available online: https://www.herox.com/BatteryRecyclingPrize (accessed on 1 January 2021).
- World Economic Forum, prepared in collaboration with the Ellen MacArthur Foundation and McKinsey & Company. Towards the Circular Economy: Accelerating the Scale-Up across Global Supply Chains; World Economic Forum: Geneva, Switzerland, 2014. [Google Scholar]
- Avicenne Energy for NAATBatt International. Study of Large Format EV Lithium-Ion Battery Recycling in China; Unpublished work; 2018. [Google Scholar]
- Sloop, S.E. Recycling of Battery Electrode Materials. U.S. Patent 12/709,144, 15 March 2016. [Google Scholar]
- Sloop, S.E. Reintroduction of Lithium into Recycled Battery Materials. U.S. Patent 12/390,364, 15 March 2016. [Google Scholar]
- Argonne National Laboratory. EverBatt: Argonne’s Closed-Loop Battery Life-Cycle Model. Available online: https://www.anl.gov/egs/everbatt (accessed on 2 January 2021).
- U.S. Department of Energy. Battery Recycling Prize Announces Phase II Prize Winners. Available online: https://www.energy.gov/eere/articles/Battery-Recycling-Prize-Announces-Phase-Ii-Prize-Winners (accessed on 30 December 2020).
- Li-Cycle. A Unique and Dependable Approach to Solving the Global Battery Recycling Problem. Available online: https://li-cycle.com/technology/ (accessed on 5 January 2021).
- Harper, G.; Sommerville, R.; Kendrick, E.; Driscoll, L.; Slater, P.; Stolkin, R.; Walton, A.; Christensen, P.; Heidrich, O.; Lambert, S.; et al. Recycling lithium-ion batteries from electric vehicles. Nature 2019, 575, 79. [Google Scholar] [CrossRef] [PubMed]
- Thompson, D.; Hartley, J.; Lambert, S.; Shiref, M.; Harper, G.; Kendrick, E.; Anderson, P.; Ryder, K.; Gaines, L.; Abbott, A. The importance of design in lithium ion battery recycling—A critical review. Green Chem. 2020, 22, 7585–7603. [Google Scholar] [CrossRef]
- Groux, O. Kyburz Battery Recycling. In Proceedings of the International WEEE and Battery Recycling Conference, Virtual, 9 December 2020. [Google Scholar]
- Martin, J.; Sherr, I. How Apple’s Daisy iPhone Recycling Robot Works. Cnet.com. Available online: https://www.cnet.com/news/how-apples-daisy-iphone-recycling-robot-works/ (accessed on 3 January 2021).
- Eisert, S. Selective Decomposition of Composites: The Potential of Continuous Shockwave Technology. In Proceedings of the E-Waste World Conference and Expo, Virtual, 18–19 November 2020. [Google Scholar]
- Zhang, R.; Meng, Z.; Ma, X.; Chen, M.; Chen, B.; Zheng, Y.; Yao, Z.; Vanaphuti, P.; Bong, S.; Yang, Z.; et al. Understanding fundamental effects of Cu impurity in different forms for recovered LiNi0.6Co0.2Mn0.2O2 cathode materials. Nano Energy 2020, 78, 105214. [Google Scholar] [CrossRef]
- Zhang, R.; Zheng, Y.; Yao, Z.; Vanaphuti, P.; Ma, X.; Bong, S.; Chen, M.; Liu, Y.; Cheng, F.; Yang, Z.; et al. Systematic Study of Al Impurity for NCM622 Cathode Materials. ACS Sustain. Chem. Eng. 2020, 8, 9875–9884. [Google Scholar] [CrossRef]
- Bai, Y.; Muralidharan, N.; Li, J.; Essehli, R.; Belharouak, I. Sustainable direct recycling of lithium-ion batteries via solvent recovery of electrode materials. ChemSusChem 2020, 13, 5664–5670. [Google Scholar] [CrossRef]
- Zhan, R.; Yang, Z.; Bloom, I.; Pan, L. Significance of a Solid Electrolyte Interphase on Separation of Anode and Cathode Materials from Spent Li-Ion Batteries by Froth Flotation. ACS Sustain. Chem. Eng. 2021, 9, 531–540. [Google Scholar] [CrossRef]
- Ellis, T.W. Magnetic Separation of Electrochemical Cell Materials. U.S. Patent 2013/0256198A1, 3 October 2013. [Google Scholar]
- Bai, Y.; Hawley, W.B.; Jafta, C.J.; Muralidharan, N.; Polzin, B.J.; Belharouak, I. Sustainable recycling of cathode scraps via Cyrene-based separation. Sustain. Mater. Technol. 2020, 25, e00202. [Google Scholar] [CrossRef]
- Pupek, K. Other Materials Separation. Available online: https://www.energy.gov/sites/default/files/2019/06/f64/bat380_%20pupek_2019_o_5.14_3.27pm_jl.pdf (accessed on 6 May 2021).
- Sanders, M. The Rechargeable Battery Market and Main Trends 2019–2030. In Proceedings of the International Battery Seminar and Exhibition, Virtual, 30 July 2020. [Google Scholar]
- Implementation Appraisal: Batteries Directive. European Parliamentary Research Service. Available online: https://www.europarl.europa.eu/RegData/etudes/BRIE/2020/654184/EPRS_BRI(2020)654184_EN.pdf (accessed on 4 January 2021).



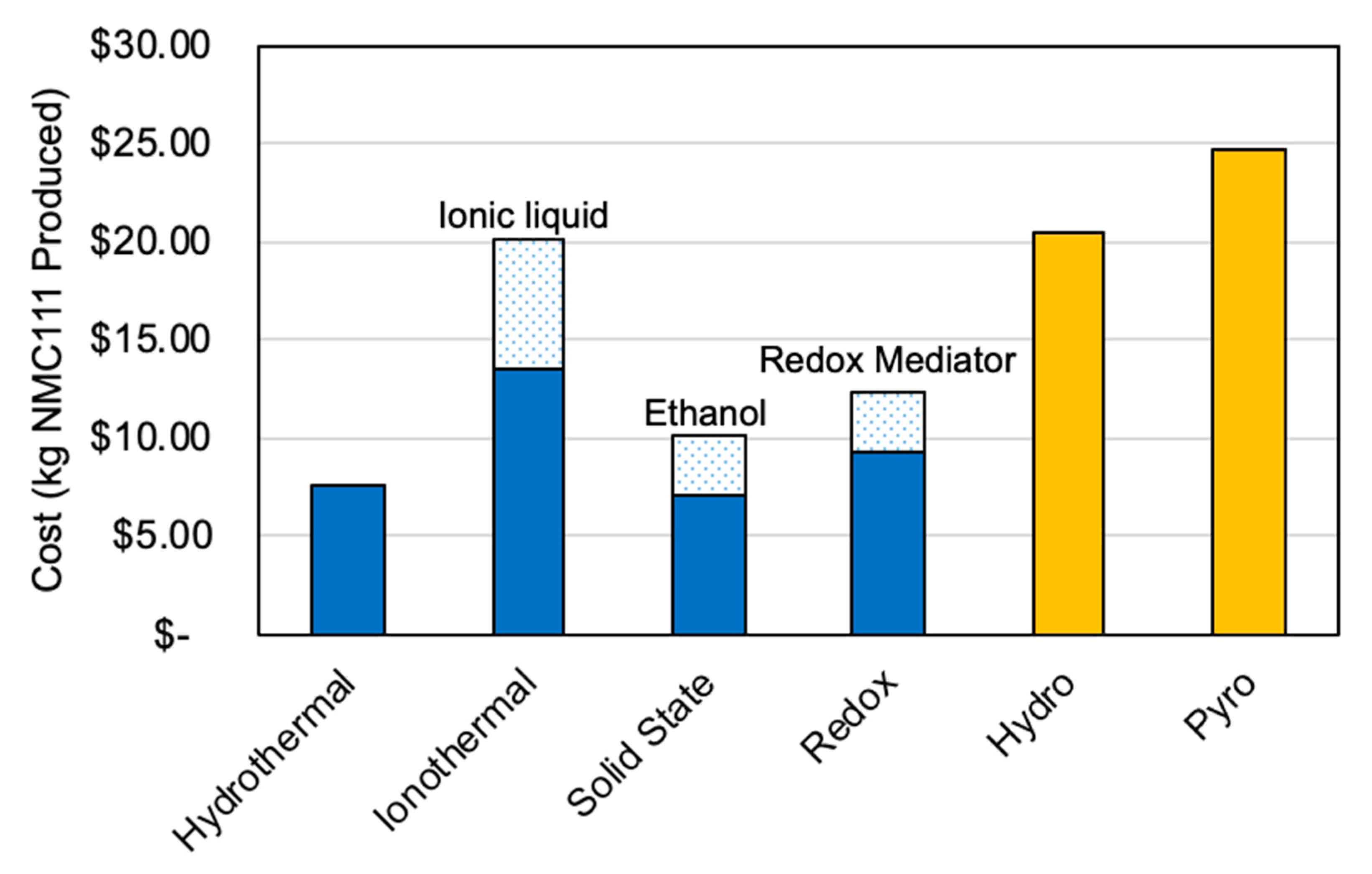
| Process Type | Lithium Source | Conditions | Unique Features |
|---|---|---|---|
| Thermal | LiOH with knowledge of lithium vacancy concentration | Heating in 2 stages | — |
| Hydrothermal | LiOH/KOH solution | Low temperature hydrothermal reaction followed by high temperature anneal | — |
| Redox Mediator | Anode of electrochemical cell | Room temperature | Reaction facilitated by mediator |
| Ionothermal | Li salt in ionic liquid | Low temperature ionothermal reaction followed by high temperature anneal | Takes place in ionic liquid |
| Electrochemical | Anode of electrochemical cell | Room temperature | Roll-to-roll reactor under development |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gaines, L.; Dai, Q.; Vaughey, J.T.; Gillard, S. Direct Recycling R&D at the ReCell Center. Recycling 2021, 6, 31. https://doi.org/10.3390/recycling6020031
Gaines L, Dai Q, Vaughey JT, Gillard S. Direct Recycling R&D at the ReCell Center. Recycling. 2021; 6(2):31. https://doi.org/10.3390/recycling6020031
Chicago/Turabian StyleGaines, Linda, Qiang Dai, John T. Vaughey, and Samuel Gillard. 2021. "Direct Recycling R&D at the ReCell Center" Recycling 6, no. 2: 31. https://doi.org/10.3390/recycling6020031
APA StyleGaines, L., Dai, Q., Vaughey, J. T., & Gillard, S. (2021). Direct Recycling R&D at the ReCell Center. Recycling, 6(2), 31. https://doi.org/10.3390/recycling6020031






