Simultaneous Sensing of Temperature and Bi-Directional Strain in a Prismatic Li-Ion Battery
Abstract
:1. Introduction
2. Materials and Methods
2.1. FBG Sensors: Mechanism of Operation
2.2. Strain and Temperature Discrimination: Reference FBG Method
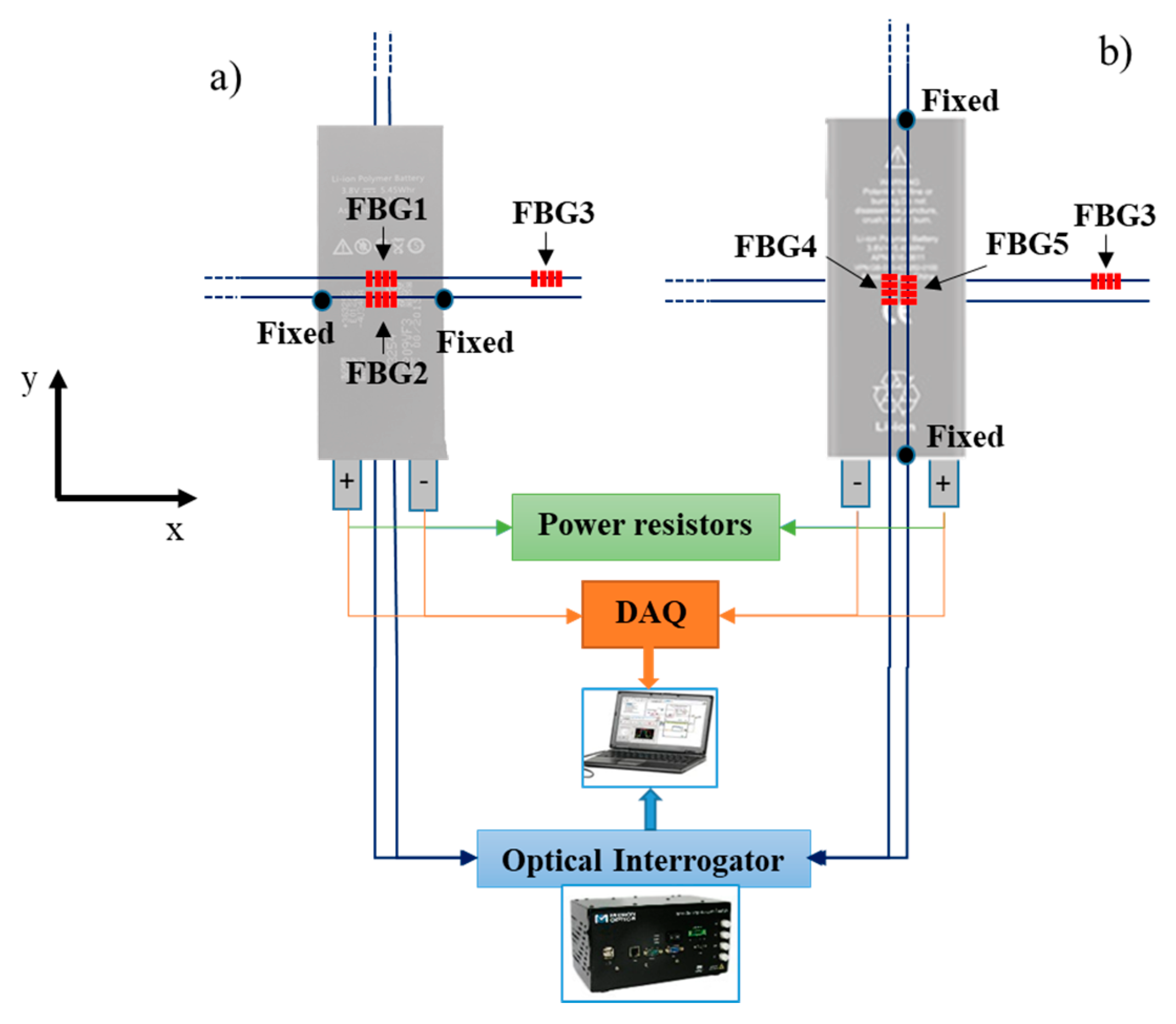
2.3. Experimental Setup
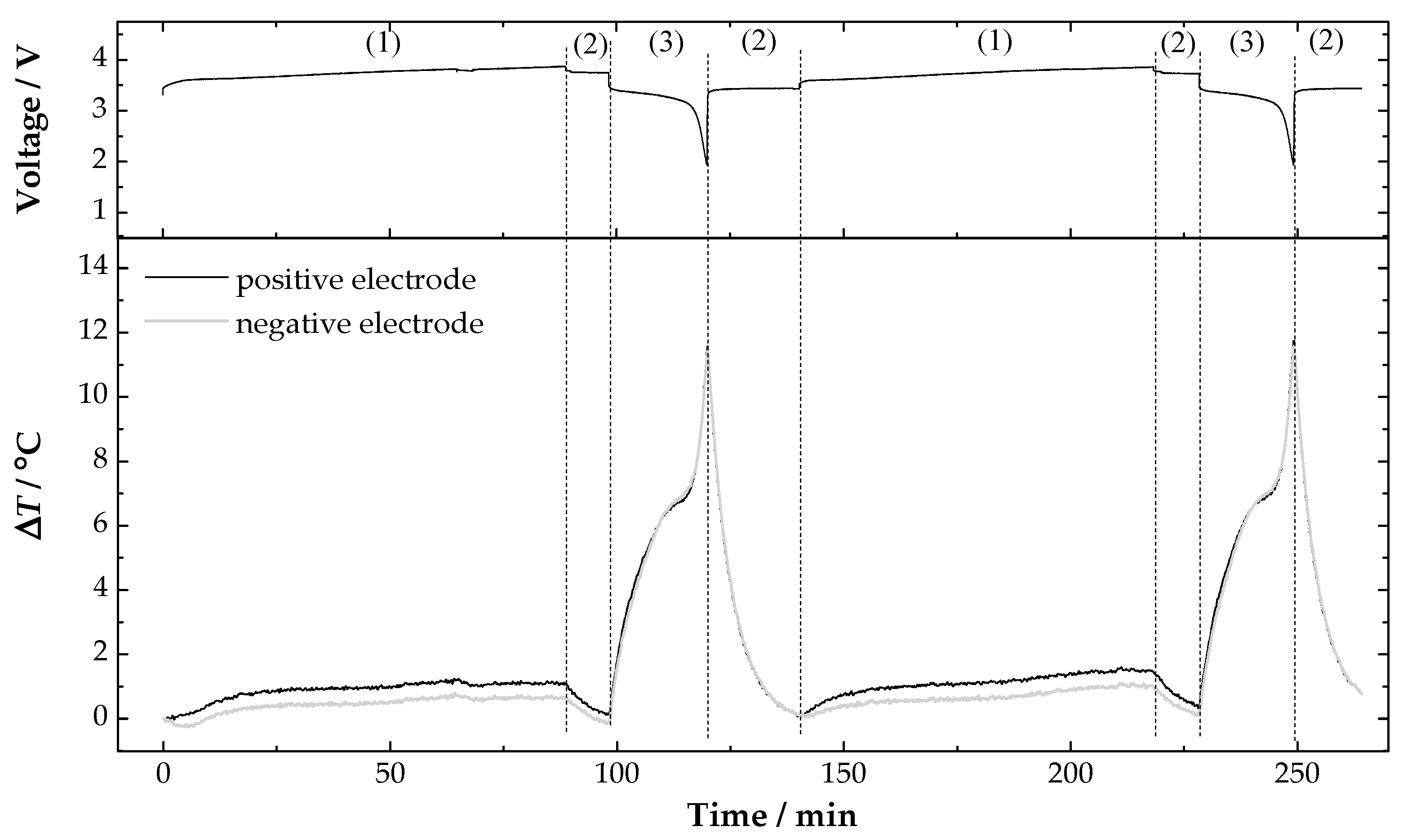
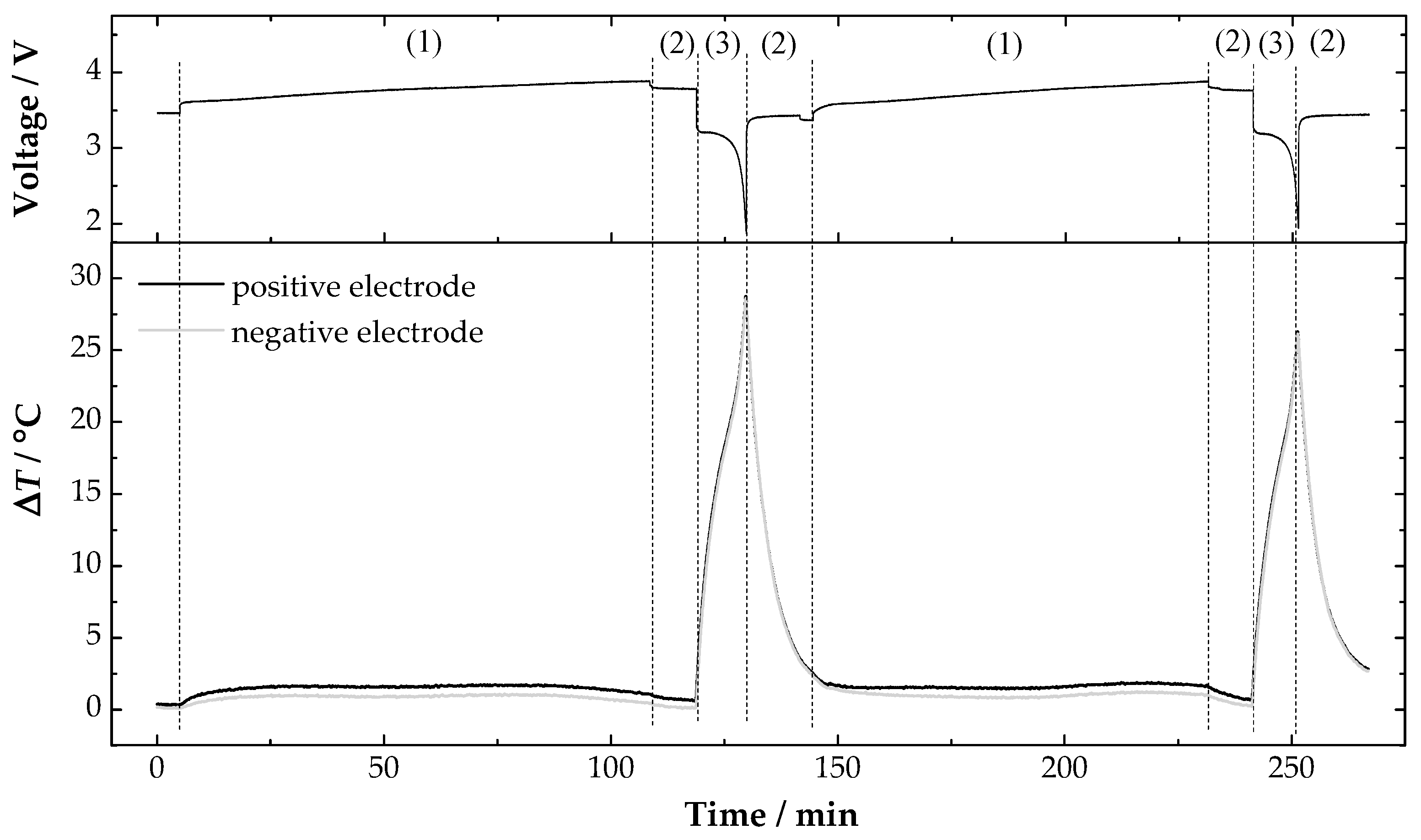
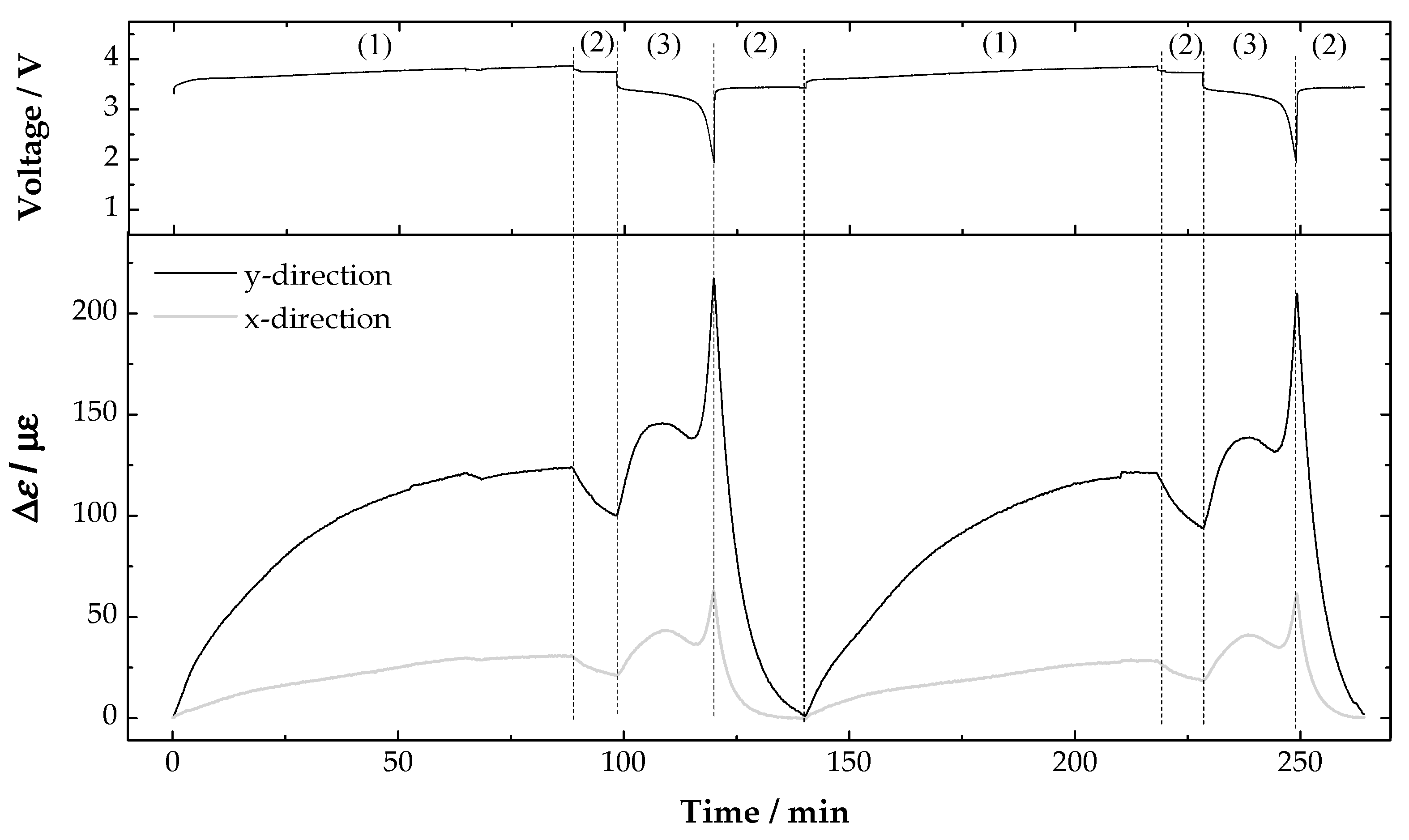
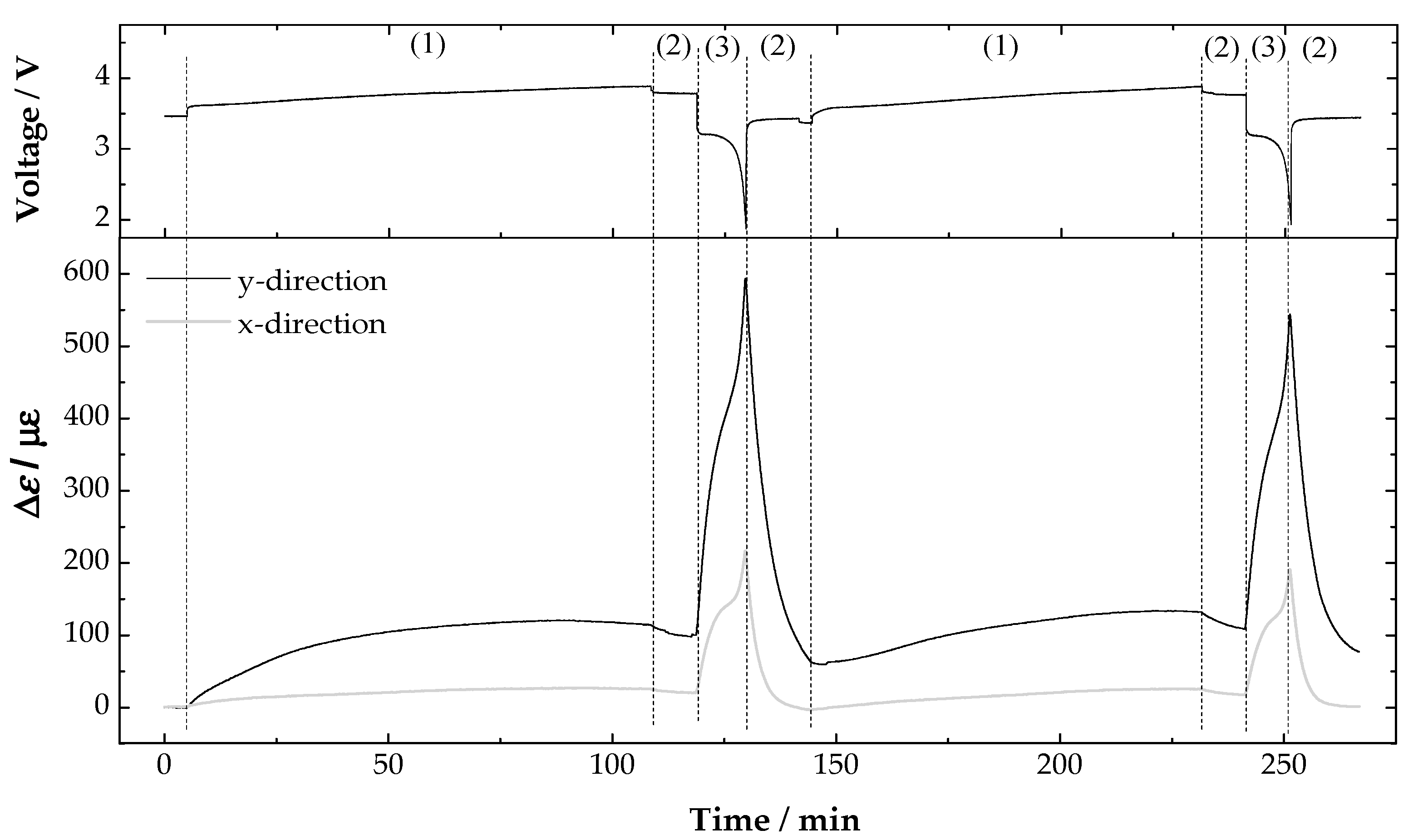
3. Results and Discussion
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Zhang, Z.; Ramadass, P.; Fang, W. Safety of Lithium-ion Batteries. In Lithium-Ion Batteries: Advances and Applications, 1st ed.; Elsevier: Amsterdam, The Netherlands, 2014; Volume 18, pp. 409–435. ISBN 978-0-444-59513-3. [Google Scholar]
- Spotnitz, R.; Franklin, J. Abuse behavior of high-power, lithium-ion cells. J. Power Sources 2003, 113, 81–100. [Google Scholar] [CrossRef]
- Mankowski, P.J.; Kanevski, J.; Bakirtzian, P.; Cugno, S. Cellular phone collateral damage: A review of burns associated with lithium battery powered mobile devices. Burns 2016, 42, e61–e64. [Google Scholar] [CrossRef] [PubMed]
- Duh, Y.S.; Lin, K.H.; Kao, C.S. Experimental investigation and visualization on thermal runaway of hard prismatic lithium-ion batteries used in smart phones. J. Therm. Anal. Calorim. 2018, 1–16. [Google Scholar] [CrossRef]
- Leung, P.K.; Moreno, C.; Masters, I.; Hazra, S.; Conde, B.; Mohamed, M.R.; Dashwood, R.J.; Bhagat, R. Real-time displacement and strain mappings of lithium-ion batteries using three-dimensional digital image correlation. J. Power Sources 2014, 271, 82–86. [Google Scholar] [CrossRef]
- Wang, Q.; Ping, P.; Zhao, X.; Chu, G.; Sun, J.; Chen, C. Thermal runaway caused fire and explosion of lithium ion battery. J. Power Sources 2012, 208, 210–224. [Google Scholar] [CrossRef]
- Mutyala, M.S.K.; Zhao, J.; Li, J.; Pan, H.; Yuan, C.; Li, X. In situ temperature measurement in lithium-ion battery by flexible thin film thermocouples. J. Power Sources 2014, 260, 43–49. [Google Scholar] [CrossRef]
- Fu, Y.; Lu, S.; Li, K.; Liu, C.; Cheng, X.; Zhang, H. An experimental study on burning behaviors of 18650 lithium ion batteries using a cone calorimeter. J. Power Sources 2015, 273, 216–222. [Google Scholar] [CrossRef]
- Panchal, S.; Dincer, I.; Agelin-Chaab, M.; Fraser, R.; Fowler, M. Experimental temperature distributions in a prismatic lithium-ion battery at varying conditions. Int. Commun. Heat Mass Transf. 2016, 71, 35–43. [Google Scholar] [CrossRef]
- Wang, X.; Sone, Y.; Segami, G.; Naito, H.; Yamada, C.; Kibe, K. Understanding volume change in Lithium-ion cells during charging and discharging using in situ measurements. J. Electrochem. Soc. 2007, 154, A14–A21. [Google Scholar] [CrossRef]
- Grattan, K.T.V.; Meggitt, B.T. Optical Fiber Sensor Technology: Applications and Systems; Kluwer Academic: London, UK, 1999; Volume 3, pp. 357–363. ISBN 978-1-4757-6077-4. [Google Scholar]
- Yang, G.; Leitão, C.; Lib, Y.; Pinto, J.L.; Jiang, X. Real-time temperature measurement with fiber Bragg sensors in lithium batteries for safety usage. Measurement 2013, 46, 3166–3172. [Google Scholar] [CrossRef]
- Nascimento, M.; Novais, S.; Leitão, C.; Domingues, M.F.; Alberto, A.; Antunes, P.; Pinto, J.L. Lithium Batteries Temperature and Strain Fiber Monitoring. In Proceedings of the 24th International Conference on Optical Fiber Sensors, Curitiba, Brazil, 28 September–2 October 2015; Volume 9634 96347V-1. [Google Scholar] [CrossRef]
- Sommer, L.W.; Kiesel, P.; Ganguli, A.; Lochbaum, A.; Saha, B.; Schwartz, J.; Bae, C.-J.; Alamgir, M. Fast and slow ion diffusion processes in lithium-ion pouch cells during cycling observed with fiber optic strain sensors. J. Power Sources 2015, 296, 46–52. [Google Scholar] [CrossRef]
- Novais, S.; Nascimento, M.; Grande, L.; Domingues, M.F.; Antunes, P.; Alberto, A.; Leitão, C.; Oliveira, R.; Koch, S.; Kim, G.T.; et al. Internal and external temperature monitoring of a Li-ion battery with fiber Bragg grating sensors. Sensors 2016, 16, 1394. [Google Scholar] [CrossRef] [PubMed]
- Nascimento, M.; Ferreira, M.; Pinto, J.L. Real time thermal monitoring of lithium batteries with fiber sensors and thermocouples: A comparative study. Measurement 2017, 111, 260–263. [Google Scholar] [CrossRef]
- Jin, W. Simultaneous measurement of strain and temperature: Error analysis. Opt. Eng. 1997, 36, 598. [Google Scholar] [CrossRef]
- James, S.W.; Dockney, M.L.; Tatam, R.P. Simultaneous independent temperature and strain measurement using in-fibre Bragg grating sensors. Electron. Lett. 1996, 32, 1133–1334. [Google Scholar] [CrossRef]
- Guan, B.O.; Tam, H.Y.; Tao, X.M.; Dong, X.Y. Simultaneous strain and temperature measurement using a superstructure fiber Bragg grating. IEEE Photonics Technol. Lett. 2000, 12, 675–677. [Google Scholar] [CrossRef]
| FBG | Direction | λB/nm | kT ± 0.13/pm/°C | kε ± 0.01/pm/µε |
|---|---|---|---|---|
| 1 | x | 1553.57 | 7.92 | - |
| 2 | x | 1553.32 | 7.92 | 1.16 |
| 3 | Room temperature sensor | 1540.17 | 8.04 | - |
| 4 | y | 1535.70 | 7.80 | - |
| 5 | y | 1535.42 | 7.80 | 1.20 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nascimento, M.; Ferreira, M.S.; Pinto, J.L. Simultaneous Sensing of Temperature and Bi-Directional Strain in a Prismatic Li-Ion Battery. Batteries 2018, 4, 23. https://doi.org/10.3390/batteries4020023
Nascimento M, Ferreira MS, Pinto JL. Simultaneous Sensing of Temperature and Bi-Directional Strain in a Prismatic Li-Ion Battery. Batteries. 2018; 4(2):23. https://doi.org/10.3390/batteries4020023
Chicago/Turabian StyleNascimento, Micael, Marta S. Ferreira, and João L. Pinto. 2018. "Simultaneous Sensing of Temperature and Bi-Directional Strain in a Prismatic Li-Ion Battery" Batteries 4, no. 2: 23. https://doi.org/10.3390/batteries4020023
APA StyleNascimento, M., Ferreira, M. S., & Pinto, J. L. (2018). Simultaneous Sensing of Temperature and Bi-Directional Strain in a Prismatic Li-Ion Battery. Batteries, 4(2), 23. https://doi.org/10.3390/batteries4020023






