Magnetotactic Bacteria and Magnetosomes: Basic Properties and Applications
Abstract
1. Introduction
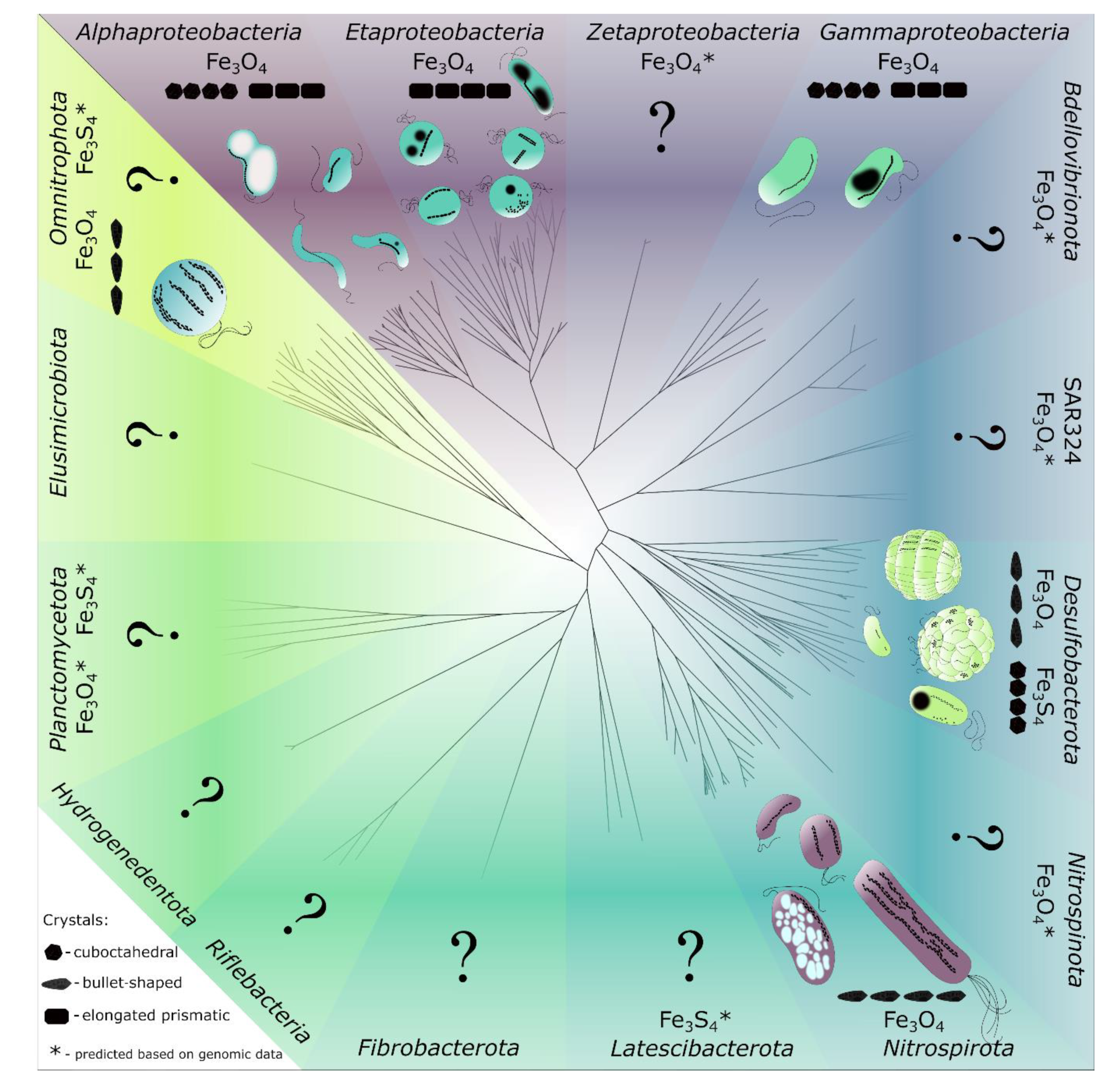
2. MTB and Magnetosomes
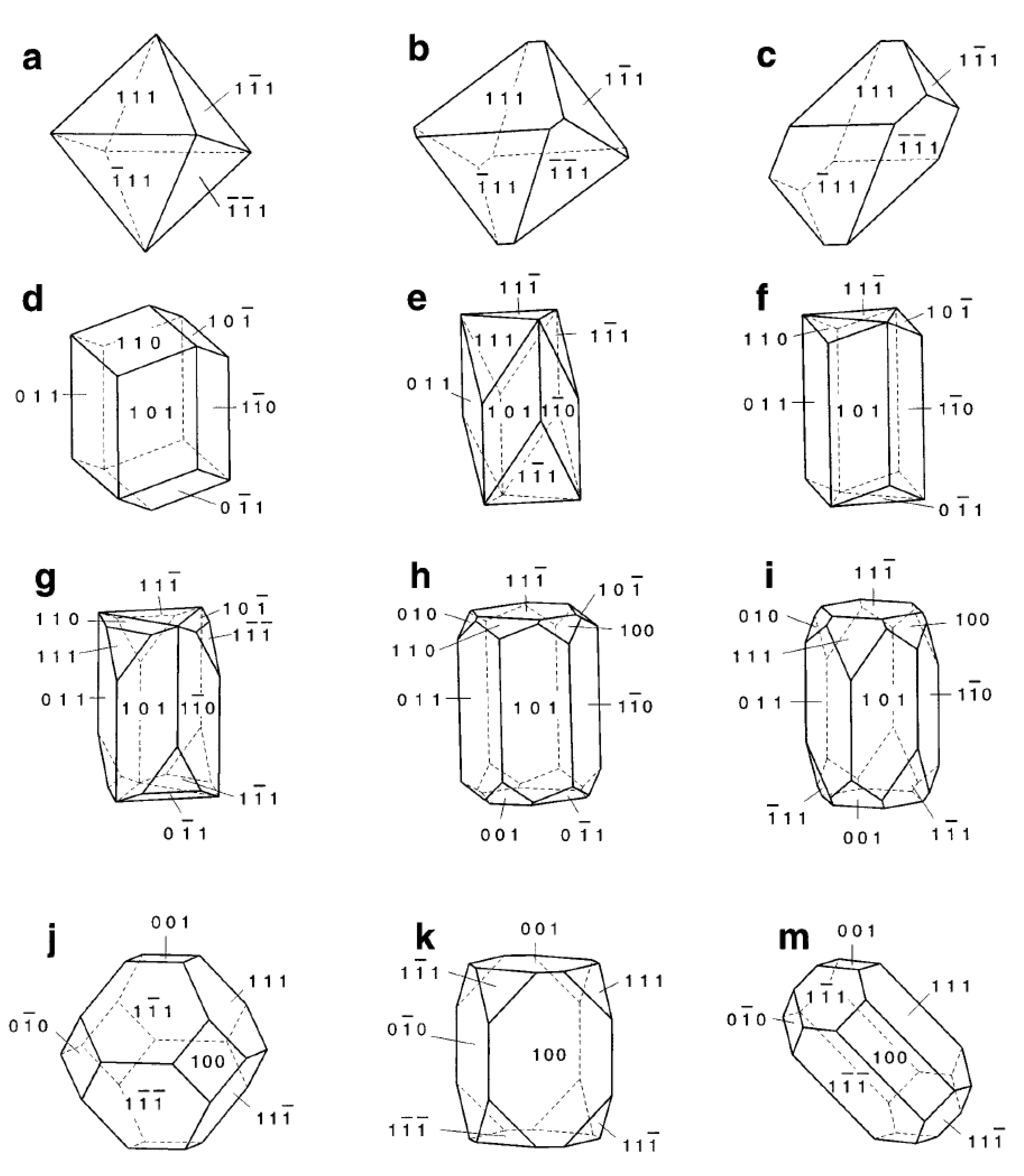
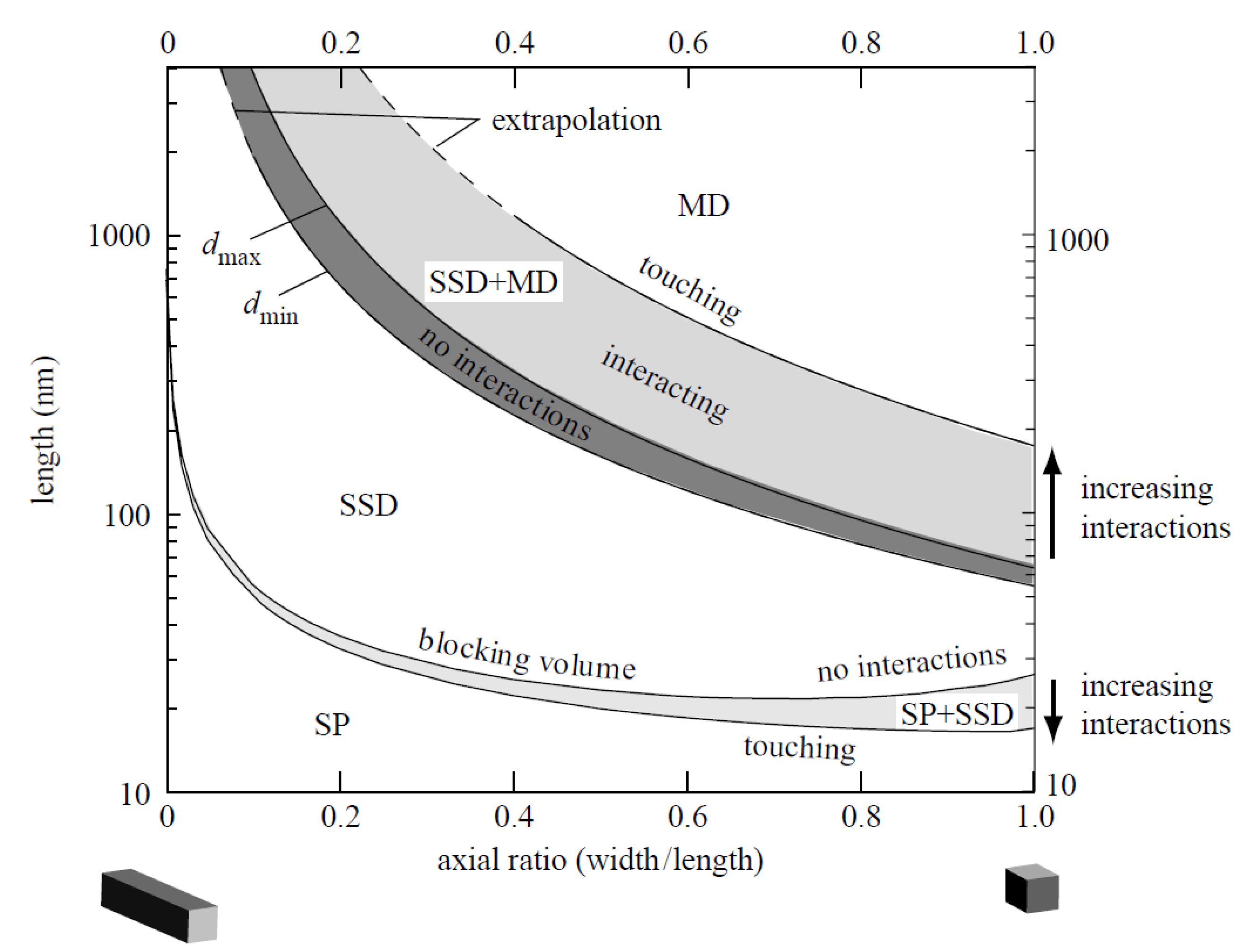
3. Physicochemical Properties and Isolation of Magnetosomes
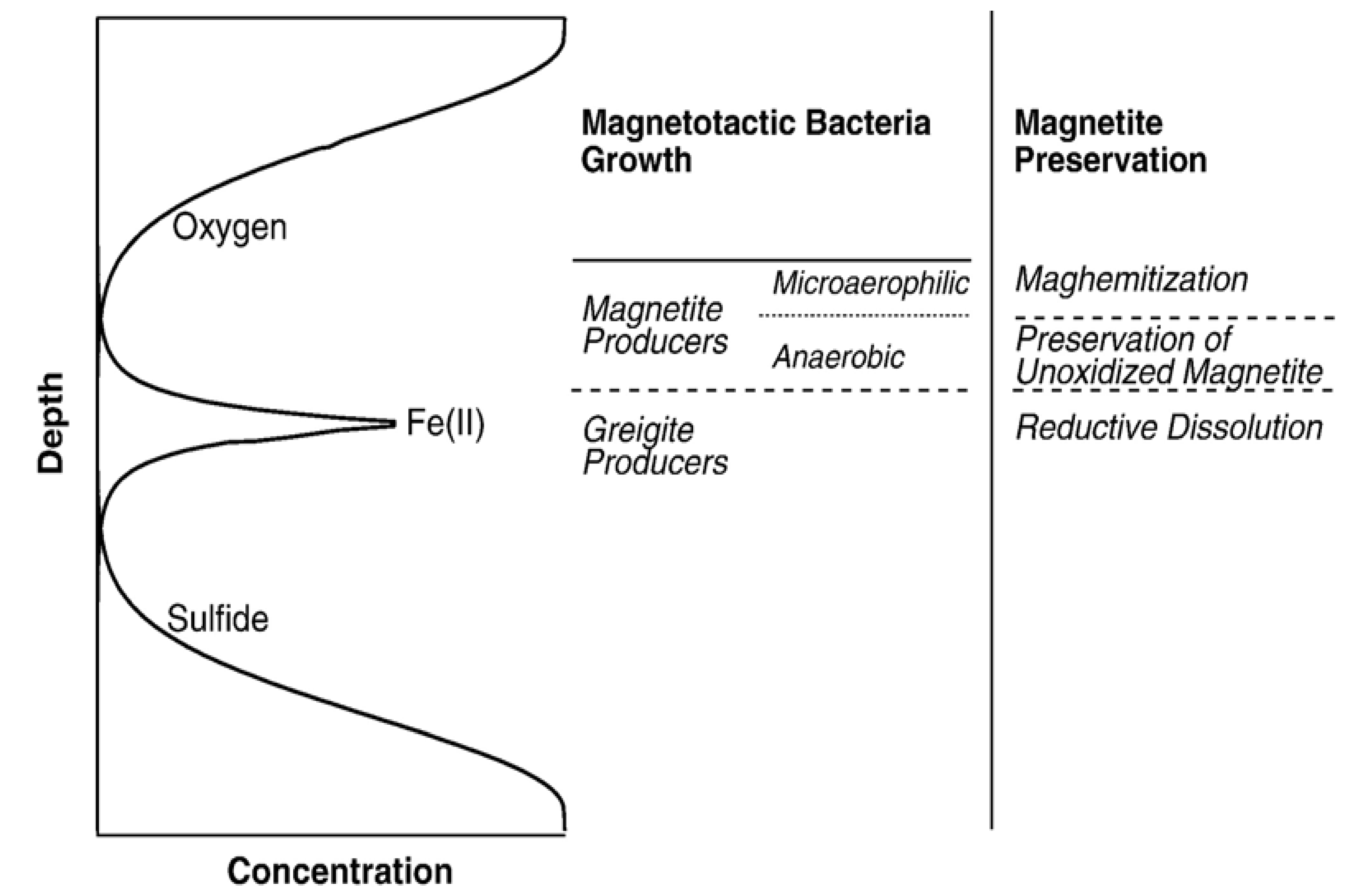
4. Magnetofossils and Their Properties
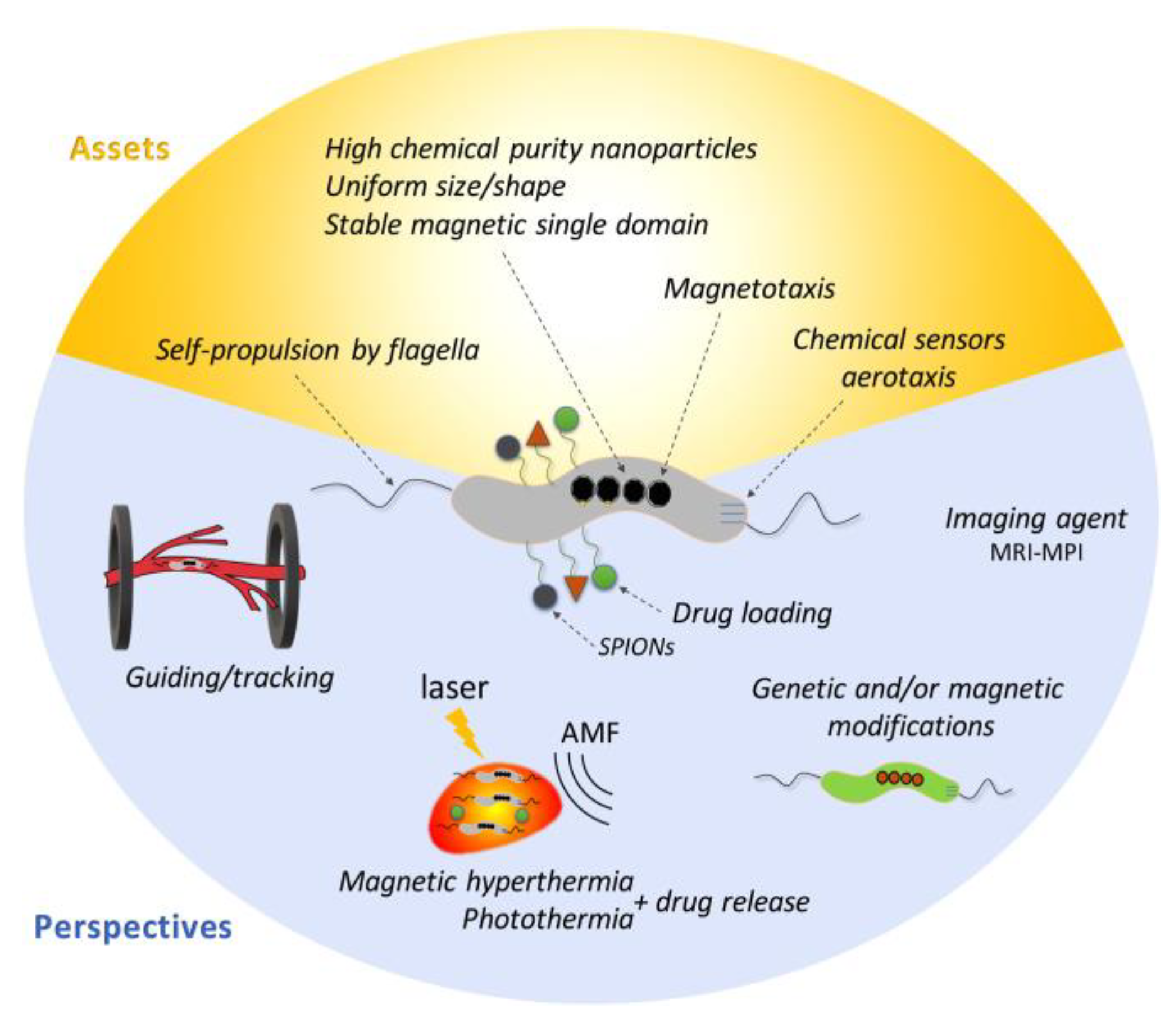
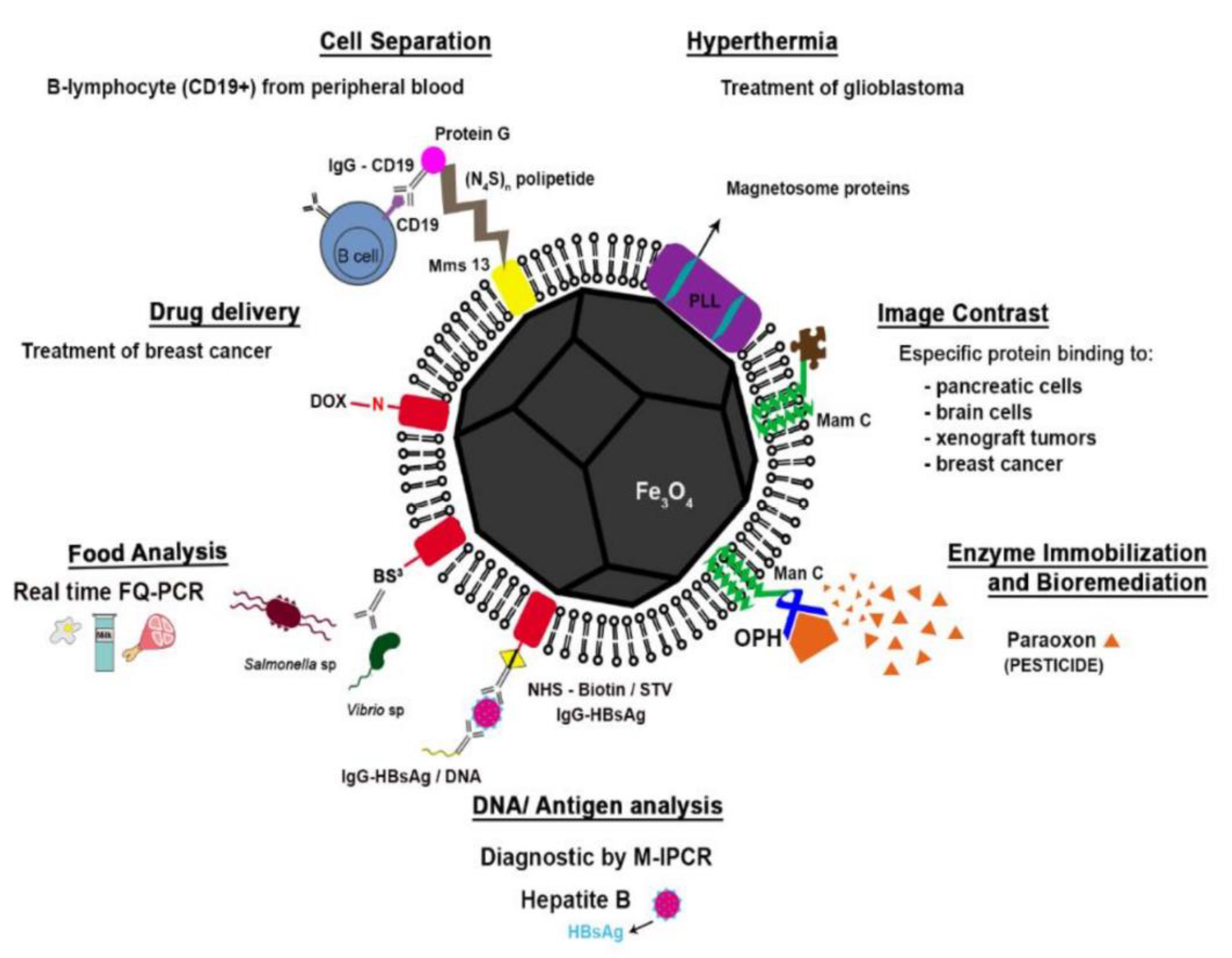
5. Application of Magnetosomes in Biomedicine and Biotechnology
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kirshvink, J.L. (Ed.) Magnetite Biomineralization and Magnetoreception in Organisms. A New Biomagnetism; Plenum Press: New York, NY, USA, 1985; pp. 16–17. [Google Scholar]
- Goldhawk, D.E.; Gelman, N.; Thompson, R.T.; Prato, F.S. Forming magnetosome-like nanoparticles in mammalian cells for molecular MRI. In Design and Applications of Nanoparticles in Biomedical Imaging; Springer: Cham, Switzerland, 2016. [Google Scholar]
- Konhauser, K.O. Bacterial iron biomineralisation in nature. FEMS Microbiol. Rev. 1997, 20, 315–326. [Google Scholar] [CrossRef]
- Revati, K.; Pandey, B.D. Microbial synthesis of iron-based nanomaterials—A review. Bull. Mater. Sci. 2011, 34, 191–198. [Google Scholar] [CrossRef]
- Faivre, D. Iron Oxides. From Nature to Applications; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2016; p. 106. [Google Scholar]
- Joudeikis, R. Engineering Membranes in Escherichia coli: The Magnetosome, LemA Protein Family and Outer Membrane Vesicles. Ph.D. Thesis, University of Kent, Canterbury, UK, 2016; p. 8. [Google Scholar]
- Uebe, R.; Schuler, D. Magnetosome biogenesis in magnetotactic bacteria. Nat. Rev. Microbiol. 2016, 14, 621–637. [Google Scholar] [CrossRef]
- Blakemore, R. Magnetotactic bacteria. Science 1975, 190, 377–379. [Google Scholar] [CrossRef]
- Blakemore, R.P. Magnetotactic bacteria. Ann. Rev. Microbiol. 1982, 36, 217–238. [Google Scholar] [CrossRef]
- Faivre, D.; Schuler, D. Magnetotactic bacteria and magnetosomes. Chem. Rev. 2008, 108, 4875–4898. [Google Scholar] [CrossRef]
- Correa, T.N.; Taveira, I.N.; de Souza Filho, R.P.; de Avila Abreu, F. Biomineralization of magnetosomes: Billion-year evolution shaping modern nanotools [online first]. In Biomineralization; IntechOpen: London, UK, 2020; p. 2. [Google Scholar] [CrossRef]
- Bazylinski, D.; Heywood, B.R.; Mann, S.; Frankel, R.B. Fe3O4 and Fe3S4 in a bacterium. Nature 1993, 366, 218. [Google Scholar] [CrossRef]
- Schuler, D. (Ed.) Magnetoreception and Magnetosomes in Bacteria; Springer: Berlin, Germany, 2007; p. 80. [Google Scholar]
- Kopp, R.E.; Kirschvink, J.L. The identification and biogeochemical interpretation of fossil magnetotactic bacteria. Earth-Sci. Rev. 2008, 86, 42–61. [Google Scholar] [CrossRef]
- Li, J.; Benzerara, K.; Bernard, B.; Beyssac, O. The link between biomineralization and fossilization of bacteria: Insights from field and experimental studies. Chem. Geol. 2013, 359, 49–69. [Google Scholar] [CrossRef]
- Frankel, R.B.; Blakemore, R.P. (Eds.) Iron Biominerals; Springer: Boston, MA, USA, 1991; p. 79. [Google Scholar]
- Yan, L.; Zhang, S.; Chen, P.; Liu, H.; Yin, H.; Li, H. Magnetotactic bacteria, magnetosomes and their application. Microbiol. Res. 2012, 167, 507–519. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, E. (Ed.) The Prokaryotes; Springer: Berlin, Germany, 2013; p. 473. [Google Scholar]
- Lefèvre, C.T.; Bazylinski, D.A. Ecology, diversity, and evolution of magnetotactic bacteria. Microbiol. Mol. Biol. Rev. 2013, 77, 497–526. [Google Scholar] [CrossRef] [PubMed]
- Uzun, M.; Alekseeva, L.; Krutkina, M.; Koziaeva, V.; Grouzdev, D. Unravelling the diversity of magnetotactic bacteria through analysis of open genomic databases. Sci. Data 2020, 7, 252. [Google Scholar] [CrossRef]
- Lin, W.; Zhang, W.; Paterson, G.A.; Zhu, Q.; Zhao, X.; Knight, R.; Bazylinski, D.A.; Roberts, A.P.; Pan, Y. Expanding magnetic organelle biogenesis in the domain Bacteria. Microbiome 2020, 8, 152. [Google Scholar] [CrossRef]
- Koziaeva, V.V.; Rusakova, S.A.; Slobodova, N.V.; Uzun, M.; Kolganova, T.V.; Skryabin, K.G.; Grouzdev, D.S. Magnetospirillum kuznetsovii sp. nov., a novel magnetotactic bacterium isolated from a lake in the Moscow region. Int. J. Syst. Evol. Microbiol. 2019, 69, 1953–1959. [Google Scholar] [CrossRef]
- Xie, J.; Chen, K.; Chen, X. Production, modification and bio-applications of magnetic nanoparticles gestated by magnetotactic bacteria. Nano Res. 2009, 2, 261–278. [Google Scholar] [CrossRef]
- Vargas, G.; Cypriano, J.; Correa, T.; Leão, P.; Bazylinski, D.A.; Abreu, F. Applications of magnetotactic bacteria, magnetosomes and magnetosome crystals in biotechnology and nanotechnology: Mini-review. Molecules 2018, 23, 2438. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.; Da, H.; Zhang, S.; López, V.M.; Wang, W. Bacterial magnetosome and its potential application. Microbiol. Res. 2017, 203, 19–28. [Google Scholar] [CrossRef]
- Nanda, T.; Rathore, A.; Sharma, D. Biomineralized and chemically synthesized magnetic nanoparticles: A contrast. Front. Mater. Sci. 2020, 14, 387–401. [Google Scholar] [CrossRef]
- Prozorov, T.; Bazylinski, D.A.; Mallapragada, S.K.; Prozorov, R. Novel magnetic nanomaterials inspired by magnetotactic bacteria: Topical review. Mater. Sci. Eng. R 2013, 74, 133–172. [Google Scholar] [CrossRef]
- Majidi, S.; Sehrig, F.Z.; Farkhani, S.M.; Goloujeh, M.S.; Akbarzadeh, A. Current methods for synthesis of magnetic nanoparticles. Artif. Cells Nanomed. Biotechnol. 2016, 44, 722–734. [Google Scholar] [CrossRef]
- Joshi, N.; Filip, J.; Coker, V.S.; Sadhukhan, J.; Safarik, I.; Bagshaw, H.; Lloyd, J.R. Microbial reduction of natural Fe(III) Minerals; Toward the sustainable production of functional magnetic nanoparticles front. Environ. Sci. 2018, 6, 127. [Google Scholar] [CrossRef]
- Bain, J.; Legge, C.J.; Beattie, D.L.; Sahota, A.; Dirks, C.; Lovett, J.R.; Staniland, S.S. A biomimetic magnetosome: Formation of iron oxide within carboxylic acid terminated polymersomes. Nanoscale 2019, 11, 11617–11625. [Google Scholar] [CrossRef]
- de Walle, A.V.; Sangnier, A.P.; Abou-Hassan, A.; Curcio, A.; Hémadi, M.; Menguy, N.; Lalatonne, Y.; Luciani, N.; Wilhelm, C. Biosynthesis of magnetic nanoparticles from nanodegradation products revealed in human stem cells. Proc. Natl. Acad. Sci. USA 2019, 116, 4044–4053. [Google Scholar] [CrossRef]
- Komeili, A. Molecular mechanisms of magnetosome formation. Annu. Rev. Biochem. 2007, 76, 351–366. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lefèvre, C.T.; Bennet, M.; Landau, L.; Vach, P.; Pignol, D.; Bazylinski, D.A.; Frankel, R.B.; Klumpp, S.; Faivre, D. Diversity of magneto-aerotactic behaviors and oxygen sensing mechanisms in cultured magnetotactic bacteria. Biophys. J. 2014, 107, 527–538. [Google Scholar] [CrossRef]
- Sharma, M.; Naresh, M.; Mittal, A. Morphological changes in magnetotactic bacteria in presence of magnetic fields. J. Biomed. Nanotechnol. 2007, 3, 1–6. [Google Scholar] [CrossRef]
- Dziuba, M.V.; Kolganova, T.V.; Gorlenko, V.M.; Kuznetsov, B.B. Species diversity of magnetotactic bacteria from the ol’khovka river, Russia. Microbiology 2013, 82, 335–340. [Google Scholar] [CrossRef]
- McCausland, H.C.; Komeili, A. Magnetic genes: Studying the genetics of biomineralization in magnetotactic bacteria. PLoS Genet. 2020, 16, e1008499. [Google Scholar] [CrossRef]
- Amor, M.; Busigny, V.; Louvat, P.; Tharaud, M.; Gelabert, A.; Cartigny, P.; Carlut, J.; Isambert, A.; Durand-Dubief, M.; Ona-Nguema, G.; et al. Iron uptake and magnetite biomineralization in the magnetotactic bacterium Magnetospirillum magneticum strain AMB-1: An iron isotope study. Geochim. Cosmochim. Acta 2018, 232, 225–243. [Google Scholar] [CrossRef]
- Greene, S.E.; Komeili, A. Biogenesis and subcellular organization of the magnetosome organelles of magnetotactic bacteria. Curr. Opin. Cell Biol. 2012, 24, 490–495. [Google Scholar] [CrossRef] [PubMed]
- Amor, M.; Ceballos, A.; Wan, J.; Simon, C.P.; Aron, A.T.; Chang, C.J.; Hellman, F.; Komeili, A. Magnetotactic bacteria accumulate a large pool of iron distinct from their magnetite crystals. Appl. Environ. Microbiol. 2020, 86, e01278-20. [Google Scholar] [CrossRef] [PubMed]
- Niu, W.; Zhang, Y.; Liu, J.; Wen, T.; Miao, T.; Basit, A.; Jiang, W. OxyR controls magnetosome formation by regulating magnetosome island (MAI) genes, iron metabolism, and redox state. Free Radic. Biol. Med. 2020, 161, 272–282. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Li, K.; Cai, Y.; Wang, P.; Gong, W.; Wu, L.-F.; Song, T. Light regulation of resistance to oxidative damage and magnetic crystal biogenesis in Magnetospirillum magneticum mediated by a Cys-less LOV-like protein. Appl. Microbiol. Biotechnol. 2020, 104, 7927–7941. [Google Scholar] [CrossRef]
- Guo, F.F.; Yang, W.; Jiang, W.; Geng, S.; Peng, T.; Li, J.L. Magnetosomes eliminate intracellular reactive oxygen species in Magnetospirillum gryphiswaldense MSR-1. Environ. Microbiol. 2012, 14, 1722–1729. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, D.; Marcano, L.; Martín-Rodríguez, R.; Simonelli, L.; Serrano, A.; García-Prieto, A.; Fdez-Gubieda, M.L.; Muela, A. Magnetosomes could be protective shields against metal stress in magnetotactic bacteria. Sci. Rep. 2020, 10, 11430. [Google Scholar] [CrossRef] [PubMed]
- Lower, B.H.; Bazylinski, D.A. The bacterial magnetosome: A unique prokaryotic organelle. J. Mol. Microbiol. Biotechnol. 2013, 23, 63–80. [Google Scholar] [CrossRef] [PubMed]
- Schübbe, S.; Williams, T.J.; Xie, G.; Kiss, H.E.; Brettin, T.S.; Martinez, D.; Ross, C.A.; Schüler, D.; Cox, D.B.; Nealson, K.H.; et al. Complete genome sequence of the chemolithoautotrophic marine magnetotactic coccus strain MC-1. Appl. Environ. Microbiol. 2009, 75, 4835–4852. [Google Scholar] [CrossRef] [PubMed]
- Nakazawa, H.; Arakaki, A.; Narita-Yamada, S.; Yashiro, I.; Jinno, K.; Aoki, N.; Tsuruyama, A.; Okamura, Y.; Tanikawa, S.; Fujita, N.; et al. Whole genome sequence of Desulfovibrio magneticus strain RS-1 revealed common gene clusters in magnetotactic bacteria. Genome Res. 2009, 19, 1801–1808. [Google Scholar] [CrossRef]
- Abreu, F.; Morillo, V.; Nascimento, F.; Werneck, C.; Cantão, M.E.; Ciapina, L.P.; de Almeida, L.C.P.; Lefèvre, C.T.; Bazylinski, D.A.; de Vasconcelos, A.T.R.; et al. Deciphering unusual uncultured magnetotactic multicellular prokaryotes through genomics. ISME J. 2014, 8, 1055–1068. [Google Scholar] [CrossRef]
- Kolinko, S.; Jogler, C.; Katzmann, E.; Wanner, G.; Peplies, J.; Schüler, D. Single-cell analysis reveals a novel uncultivated magnetotactic bacterium within the candidate division OP3. Environ. Microbiol. 2012, 14, 1709–1721. [Google Scholar] [CrossRef]
- Koziaeva, V.; Dziuba, M.; Leão, P.; Uzun, M.; Krutkina, M.; Grouzdev, D. Genome-Based Metabolic Reconstruction of a Novel Uncultivated Freshwater Magnetotactic coccus “Ca. Magnetaquicoccus inordinatus” UR-1, and Proposal of a Candidate Family “Ca. Magnetaquicoccaceae. Front. Microbiol. 2019, 10, 2290. [Google Scholar] [CrossRef]
- Zhang, W.; Ji, R.; Liu, J.; Pan, Y.; Wu, L.-F.; Lin, W. Two Metagenome-Assembled Genome Sequences of Magnetotactic Bacteria in the Order Magnetococcales. Microbiol. Resour. Announc. 2020, 9, e00363-20. [Google Scholar] [CrossRef]
- Monteil, C.L.; Grouzdev, D.S.; Perriere, G.; Alonso, B.; Rouy, Z.; Cruveiller, S.; Ginet, N.; Pignol, D.; Lefèvre, C.T. Repeated horizontal gene transfers triggered parallel evolution of magnetotaxis in two evolutionary divergent lineages of magnetotactic bacteria. ISME J. 2020, 14, 1783–1794. [Google Scholar] [CrossRef]
- Liu, P.; Liu, Y.; Zhao, X.; Roberts, A.P.; Zhang, H.; Zheng, Y.; Wang, F.; Wang, L.; Menguy, N.; Pan, Y.; et al. Diverse phylogeny and morphology of magnetite biomineralized by magnetotactic cocci. Environ. Microbiol. 2021, 23, 1115–1129. [Google Scholar] [CrossRef]
- Lefèvre, C.T.; Menguy, N.; Abreu, F.; Lins, U.; Pósfai, M.; Prozorov, T.; Pignol, D.; Frankel, R.B.; Bazylinski, D.A. A cultured greigite-producing magnetotactic bacterium in a novel group of sulfate-reducing bacteria. Science 2011, 334, 1720–1723. [Google Scholar] [CrossRef]
- Koziaeva, V.V.; Dziuba, M.V.; Ivanov, T.M.; Kuznetsov, B.B.; Skryabin, K.G.; Grouzdev, D.S. Draft genome sequences of two magnetotactic bacteria, Magnetospirillum moscoviense BB-1 and Magnetospirillum marisnigri SP-1. Genome Announc. 2016, 4, e00814-16. [Google Scholar] [CrossRef] [PubMed]
- Trubitsyn, D.; Abreu, F.; Ward, F.B.; Taylor, T.; Hattori, M.; Kondo, S.; Trivedi, U.; Staniland, S.; Lins, U.; Bazylinski, D.A. Draft genome sequence of Magnetovibrio blakemorei strain MV-1, a marine vibrioid magnetotactic bacterium. Genome Announc. 2016, 4, e01330-16. [Google Scholar] [CrossRef]
- Grouzdev, D.S.; Dziuba, M.V.; Sukhacheva, M.S.; Mardanov, A.V.; Beletskiy, A.V.; Kuznetsov, B.B.; Skryabin, K.G. Draft genome sequence of Magnetospirillum sp. strain SO-1, a freshwater magnetotactic bacterium isolated from the Ol’khovka River, Russia. Genome Announc. 2014, 2, e00235-14. [Google Scholar] [CrossRef] [PubMed]
- Monteil, C.L.; Benzerara, K.; Menguy, N.; Bidaud, C.C.; Michot-Achdjian, E.; Bolzoni, R.; Mathon, F.P.; Coutaud, M.; Alonso, B.; Garau, C.; et al. Intracellular amorphous Ca-carbonate and magnetite biomineralization by a magnetotactic bacterium affiliated to the Alphaproteobacteria. ISME J. 2021, 15, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Ke, L.; Liu, P.; Liu, S.; Gao, M. Complete genome sequence of Magnetospirillum sp. ME-1, a novel magnetotactic bacterium isolated from East Lake, Wuhan, China. Genome Announc. 2017, 5, e00485-17. [Google Scholar] [CrossRef]
- Uebe, R.; Schüler, D.; Jogler, C.; Wiegand, S. Reevaluation of the complete genome sequence of Magnetospirillum gryphiswaldense MSR-1 with single-molecule real-time sequencing data. Genome Announc. 2018, 6, e00309-18. [Google Scholar] [CrossRef] [PubMed]
- Smalley, M.D.; Marinov, G.K.; Bertani, L.E.; DeSalvo, G. Genome sequence of Magnetospirillum magnetotacticum strain MS-1. Genome Announc. 2015, 3, e00233-15. [Google Scholar] [CrossRef] [PubMed]
- Lefèvre, C.T.; Wu, L.-F. Evolution of the bacterial organelle responsible for magnetotaxis. Trends Microbiol. 2013, 21, 534–543. [Google Scholar] [CrossRef]
- Nudelman, H.; Zarivach, R. Structure prediction of magnetosome-associated proteins. Front. Microbiol. 2014, 5, 9. [Google Scholar] [CrossRef]
- Lohße, A.; Ullrich, S.; Katzmann, E.; Borg, S.; Wanner, G.; Richter, M.; Voigt, B.; Schweder, T.; Schüler, D. Functional analysis of the magnetosome island in magnetospirillum gryphiswaldense: The mamAB operon is sufficient for magnetite biomineralization. PLoS ONE 2011, 6, e25561. [Google Scholar] [CrossRef]
- Murat, D.J. Magnetosomes: How do they stay in shape? Mol. Microbiol. Biotechnol. 2013, 23, 81–94. [Google Scholar] [CrossRef]
- Rahn-Lee, L.; Komeili, A. The magnetosome model: Insights into the mechanisms of bacterial biomineralization. Front. Microbiol. 2013, 4, 352. [Google Scholar] [CrossRef]
- Barber-Zucker, S.; Zarivach, R. A look into the biochemistry of magnetosome biosynthesis in magnetotactic bacteria. ACS Chem. Biol. 2017, 12, 13–22. [Google Scholar] [CrossRef]
- Raschdorf, O.; Forstner, Y.; Kolinko, I.; Uebe, R.; Plitzko, J.M.; Schüler, D. Genetic and ultrastructural analysis reveals the key players and initial steps of bacterial magnetosome membrane biogenesis. PLoS Genet. 2016, 12, e1006101. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Li, S.; Huang, X.; Li, J.; Li, L.; Pan, Y.; Li, Y. MamX encoded by the mamXY operon is involved in control of magnetosome maturation in Magnetospirillum gryphiswaldense MSR-1. BMC Microbiol. 2013, 13, 203. [Google Scholar] [CrossRef] [PubMed]
- Toro-Nahuelpan, M.; Giacomelli, G.; Raschdorf, O.; Borg, S.; Plitzko, J.M.; Bramkamp, M.; Schüler, D.; Müller, F.D. MamY is a membrane-bound protein that aligns magnetosomes and the motility axis of helical magnetotactic bacteria. Nat. Microbiol. 2019, 4, 1978–1989. [Google Scholar] [CrossRef] [PubMed]
- Devouard, B.; Pósfai, M.; Hua, X.; Bazylinski, D.A.; Frankel, R.B.; Buseck, P.R. Magnetite from magnetotactic bacteria: Size distributions and twinning. Am. Mineral. 1998, 83, 1387–1398. [Google Scholar] [CrossRef]
- Schuler, D. Genetics and cell biology of magnetosome formation in magnetotactic bacteria. FEMS Microbiol. Rev. 2008, 32, 654–672. [Google Scholar] [CrossRef] [PubMed]
- Abraçado, L.G.; Abreu, F.; Keim, C.N.; Campos, A.P.C.; Lins, U.; Farina, M. Magnetosome chain superstructure in uncultured magnetotactic bacteria. Phys. Biol. 2010, 7, 046016. [Google Scholar] [CrossRef]
- Komeili, A.; Li, Z.; Newman, D.K.; Jensen, G.J. Magnetosomes are cell membrane invaginations organized by the actin-like protein MamK. Science 2006, 311, 242–245. [Google Scholar] [CrossRef] [PubMed]
- Abreu, F.; Sousa, A.A.; Aronova, M.A.; Kim, Y.; Cox, D.; Leapman, R.D.; Andrade, L.R.; Kachar, B.; Bazylinski, D.A.; Lins, U. Cryo-electron tomography of the magnetotactic vibrio Magnetovibrio blakemorei: Insights into the biomineralization of prismatic magnetosomes. J. Struct. Biol. 2013, 181, 162–168. [Google Scholar] [CrossRef]
- Stephens, C. Bacterial cell biology: Managing magnetosomes. Curr. Biol. 2006, 16, R363–R365. [Google Scholar] [CrossRef]
- Le Nagard, L.; Morillo-López, V.; Fradin, C.; Bazylinski, D.A. Growing magnetotactic bacteria of the genus magnetospirillum: Strains MSR-1, AMB-1 and MS-1. J. Vis. Exp. 2018, 140, e58536. [Google Scholar] [CrossRef] [PubMed]
- Paterson, G.A.; Wang, Y.; Pan, Y. The fidelity of paleomagnetic records carried by magnetosome chains. Earth Planet. Sci. Lett. 2013, 383, 82–91. [Google Scholar] [CrossRef]
- Li, J.; Cai, F.; Lv, H.; Sun, J. Selective Competitive Biosorption of Au(III) and Cu(II) in Binary Systems by Magnetospirillum gryphiswaldense. Sep. Sci. Technol. 2013, 48, 960–967. [Google Scholar] [CrossRef]
- Vali, H.; Förster, O.; Amarantidis, G.; Petersen, N. Magnetotactic bacteria and their magnetofossils in sediments. Earth Planet. Sci. Lett. 1987, 86, 389–400. [Google Scholar] [CrossRef]
- Lins, U.; McCartney, M.R.; Farina, M.; Frankel, R.B.; Buseck, P.R. Crystal habits and magnetic microstructures of magnetosomes in coccoid magnetotactic bacteria. An. Acad. Bras. Ciências 2006, 78, 463–474. [Google Scholar] [CrossRef] [PubMed]
- Lins, U.; McCartney, M.R.; Farina, M.; Frankel, R.B.; Buseck, P.R. Habits of magnetosome crystals in coccoid magnetotactic bacteria. Appl. Environ. Microbiol. 2005, 71, 4902–4905. [Google Scholar] [CrossRef]
- Moskowitz, B.M.; Frankel, R.B.; Bazylinski, D.A. Rock magnetic criteria for the detection of biogenic magnetite. Earth Planet. Sci. Lett. 1993, 120, 283–300. [Google Scholar] [CrossRef]
- Scheffel, A.; Gärdes, A.; Grünberg, K.; Wanner, G.; Schüler, D. The major magnetosome proteins MamGFDC are not essential for magnetite biomineralization in magnetospirillum gryphiswaldense but regulate the size of magnetosome crystals. J. Bacteriol. 2008, 190, 377–386. [Google Scholar] [CrossRef]
- Uebe, R.; Keren-Khadmy, N.; Zeytuni, N.; Katzmann, E.; Navon, Y.; Davidov, G.; Bitton, R.; Plitzko, J.M.; Schüler, D.; Zarivach, R. The dual role of MamB in magnetosome membrane assembly and magnetite biomineralization. Mol. Microbiol. 2018, 107, 542–557. [Google Scholar] [CrossRef]
- Bender, P.; Marcano, L.; Orue, I.; Venero, D.A.; Honecker, D.; Barquín, L.F.; Muela, A.; Fdez-Gubieda, M.L. Probing the stability and magnetic properties of magnetosome chains in freeze-dried magnetotactic bacteria. Nanoscale Adv. 2020, 2, 1115–1121. [Google Scholar] [CrossRef]
- Chariaou, M.; Rahn-Lee, L.; Kind, J.; García-Rubio, I.; Komeili, A.; Gehring, A.U. Anisotropy of bullet-shaped magnetite nanoparticles in the magnetotactic bacteria desulfovibrio magneticus sp. Strain RS-1. Biophys. J. 2015, 108, 1268–1274. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Menguy, N.; Gatel, C.; Boureau, V.; Snoeck, E.; Patriarche, G.; Leroy, E.; Pan, Y. Crystal growth of bullet-shaped magnetite in magnetotactic bacteria of the Nitrospirae phylum. J. R. Soc. Interface 2014, 12, 20141288. [Google Scholar] [CrossRef]
- Pan, Y.; Petersen, N.; Davila, A.F.; Zhang, L.; Winklhofer, M.; Liu, Q.; Hanzlik, M.; Zhu, R. The detection of bacterial magnetite in recent sediments of Lake Chiemsee (southern Germany). Earth Planet. Sci. Lett. 2005, 232, 109–123. [Google Scholar] [CrossRef]
- Pan, Y.; Petersen, N.; Winklhofer, M.; Davila, A.F.; Liu, Q.; Frederichs, T.; Hanzlik, M.; Zhu, R. Rock magnetic properties of uncultured magnetotactic bacteria. Earth Planet. Sci. Lett. 2005, 237, 311–325. [Google Scholar] [CrossRef]
- Nguyen, H.V.; Suzuki, E.; Oestreicher, Z.; Minamide, H.; Endoh, H.; Fukumori, Y.; Taoka, A. A protein-protein interaction in magnetosomes: TPR protein MamA interacts with an Mms6 protein. Biochem. Biophys. Rep. 2016, 7, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Nudelman, H.; Valverde-Tercedor, C.; Kolusheva, S.; Gonzalez, T.P.; Widdrat, M.; Grimberg, N.; Levi, H.; Nelkenbaum, O.; Davidov, G.; Faivre, D.; et al. Structure–function studies of the magnetite-biomineralizing magnetosome-associated protein MamC. J. Struct. Biol. 2016, 194, 244–252. [Google Scholar] [CrossRef] [PubMed]
- Cornejo-Warner, E.J. Magnetosome Biogenesis in Magnetospirillum Magneticum AMB-1. Ph.D. Thesis, University of California, Berkeley, CA, USA, 2016; p. 17. [Google Scholar]
- Pfeiffer, D.; Toro-Nahuelpan, M.; Bramkamp, M.; Plitzko, J.M.; Schüler, D. The polar organizing protein PopZ is fundamental for proper cell division and segregation of cellular content in magnetospirillum gryphiswaldense. mBio 2019, 10, e02716-18. [Google Scholar] [CrossRef] [PubMed]
- Dziuba, M.V.; Zwiener, T.; Uebe, R.; Schüler, D. Single-step transfer of biosynthetic operons endows a non-magnetotactic Magnetospirillum strain from wetland with magnetosome biosynthesis. Environ. Microbiol. 2020, 22, 1603–1618. [Google Scholar] [CrossRef]
- Kolinko, I.; Lohße, A.; Borg, S.; Raschdorf, O.; Jogler, C.; Tu, Q.; Pósfai, M.; Tompa, E.; Plitzko, J.M.; Brachmann, A.; et al. Biosynthesis of magnetic nanostructures in a foreign organism by transfer of bacterial magnetosome gene clusters. Nat. Nanotechnol. 2014, 9, 193–197. [Google Scholar] [CrossRef]
- Mickoleit, F.; Borkner, C.B.; Toro-Nahuelpan, M.; Herold, H.M.; Maier, D.S.; Plitzko, J.M.; Scheibel, T.; Schüler, D. In vivo coating of bacterial magnetic nanoparticles by magnetosome expression of spider silk-inspired peptides. Biomacromolecules 2018, 19, 962–972. [Google Scholar] [CrossRef]
- Peigneux, A.; Jabalera, Y.; Fernández Vivas, M.A.; Casares, S.; Azuaga, A.I.; Jimenez-Lopez, C. Tuning properties of biomimetic magnetic nanoparticles by combining magnetosome associated proteins. Sci. Rep. 2019, 9, 8804. [Google Scholar] [CrossRef]
- Staniland, S.S.; Rawlings, A.E. Crystallizing the function of the magnetosome membrane mineralization protein Mms6. Biochem. Soc. Trans. 2016, 44, 883–890. [Google Scholar] [CrossRef]
- Prabhu, N.N.; Kowshik, M. Techniques for the isolation of magnetotactic bacteria. J. Microb. Biochem. Technol. 2016, 8, 188–194. [Google Scholar] [CrossRef]
- Basit, A.; Wang, J.; Guo, F.; Niu, W.; Jiang, W. Improved methods for mass production of magnetosomes and applications: A review. Microb. Cell Fact. 2020, 19, 197. [Google Scholar] [CrossRef]
- Byrne, J.M.; Muhamadali, H.; Coker, V.S.; Cooper, J.; Lloyd, J.R. Scale-up of the production of highly reactive biogenic magnetite nanoparticles using Geobacter sulfurreducens. J. R. Soc. Interface 2015, 12, 20150240. [Google Scholar] [CrossRef] [PubMed]
- Gorby, Y.A. Regulation of Magnetosome Biogenesis by Oxygen and Nitrogen. Ph.D. Thesis, University of New Hampshire, Durham, NH, USA, 1989; p. 12. [Google Scholar]
- Fernández-Castané, A.; Lia, H.; Thomas, O.R.T.; Overton, T.W. Development of a simple intensified fermentation strategy for growth of Magnetospirillum gryphiswaldense MSR-1: Physiological responses to changing environmental conditions. New Biotechnol. 2018, 46, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Castané, A.; Li, H.; Thomas, O.R.T.; Overton, T.W. Flow cytometry as a rapid analytical tool to determine physiological responses to changing O2 and iron concentration by Magnetospirillum. Sci. Rep. 2017, 7, 13118. [Google Scholar] [CrossRef]
- Moskowitz, B.M.; Frankel, R.B.; Flanders, P.J.; Blakemore, R.P.; Schwartz, B.B. Magnetic properties of magnetotactic bacteria. J. Magn. Magn. Mater. 1988, 73, 273–288. [Google Scholar] [CrossRef]
- Kopp, R.E.; Nash, C.Z.; Kobayashi, A.; Weiss, B.P.; Bazylinski, D.A.; Kirschvink, J.L. Ferromagnetic resonance spectroscopy for assessment of magnetic anisotropy and magnetostatic interactions: A case study of mutant magnetotactic bacteria. J. Geophys. Res. 2006, 111, B12S25. [Google Scholar] [CrossRef]
- Kopp, R.E.; Weiss, B.P.; Maloof, A.C.; Vali, H.; Nash, C.Z.; Kirschvink, J.L. Chains, clumps, and strings: Magnetofossil taphonomy with ferromagnetic resonance spectroscopy. Earth Planet. Sci. Lett. 2006, 247, 10–25. [Google Scholar] [CrossRef]
- Ionescu, A.; Darton, N.J.; Vyas, K.; Llandro, J. Detection of endogenous magnetic nanoparticles with a tunnelling magnetoresistance sensor. Philos. Trans. Math. Phys. Eng. Sci. 2010, 368, 4371–4387. [Google Scholar] [CrossRef]
- Bazylinski, D.A.; Garratt-Reed, A.J.; Frankel, R.B. Electron microscopic studies of magnetosomes in magnetotactic bacteria. Microsc. Res. Tech. 1994, 27, 389–401. [Google Scholar] [CrossRef]
- Blondeau, M.; Guyodo, Y.; Guyot, F.; Gatel, C.; Menguy, N.; Chebbi, I.; Haye1, B.; Durand-Dubief, M.; Alphandery, E.; Brayner, R.; et al. Magnetic-field induced rotation of magnetosome chains in silicified magnetotactic bacteria. Sci. Rep. 2018, 8, 7699. [Google Scholar] [CrossRef]
- Li, J.; Menguy, N.; Arrio, M.-A.; Sainctavit, P.; Juhin, A.; Wang, Y.; Chen, H.; Bunau, O.; Otero, E.; Ohresser, P.; et al. Controlled cobalt doping in the spinel structure of magnetosome magnetite: New evidences from element- and site-specific X-ray magnetic circular dichroism analyses. J. R. Soc. Interface 2016, 13, 20160355. [Google Scholar] [CrossRef]
- Fischer, H.; Mastrogiacomo, G.; Löffler, J.F.; Warthmann, R.J.; Weidler, P.G.; Gehring, A.U. Ferromagnetic resonance and magnetic characteristics of intact magnetosome chains in Magnetospirillum gryphiswaldense. Earth Planet. Sci. Lett. 2008, 270, 200–208. [Google Scholar] [CrossRef]
- Gehring, A.U.; Charilaou, M.; García-Rubio, I. Oxidized magnetosomes in magnetotactic bacteria. J. Magn. Magn. Mater. 2012, 324, 1281–1284. [Google Scholar] [CrossRef]
- Gehring, A.U.; Fischer, H.; Charilaou, M.; García-Rubio, I. Magnetic anisotropy and Verwey transition of magnetosome chains in Magnetospirillum gryphiswaldense. Geophys. J. Int. 2011, 187, 1215–1221. [Google Scholar] [CrossRef]
- Gehring, A.U.; Kind, J.; Charilaou, M.; García-Rubio, I. The detection of magnetotactic bacteria and magnetofossils by means of magnetic anisotropy. Earth Planet. Sci. Lett. 2011, 309, 113–117. [Google Scholar] [CrossRef]
- Zhang, T.; Pan, Y. Constraining the magnetic properties of ultrafine- and fine-grained biogenic magnetite. Earth Planets Space 2018, 70, 206. [Google Scholar] [CrossRef]
- Ding, Y.; Li, J.; Liu, J.; Yang, J.; Jiang, W.; Tian, J.; Li, Y.; Pan, Y.; Li, J. Deletion of the ftsZ-like gene results in the production of superparamagnetic magnetite magnetosomes in magnetospirillum gryphiswaldense. J. Bacteriol. 2010, 192, 1097–1105. [Google Scholar] [CrossRef]
- Prozorov, R.; Prozorov, T.; Mallapragada, S.K.; Narasimhan, B. Magnetic irreversibility and the Verwey transition in nanocrystalline bacterial magnetite. Phys. Rev. B 2007, 76, 054406. [Google Scholar] [CrossRef]
- Muxworthy, A.R.; Williams, W. Critical superparamagnetic/single-domain grain sizes in interacting magnetite particles: Implications for magnetosome crystals. J. R. Soc. Interface 2009, 6, 1207–1212. [Google Scholar] [CrossRef]
- Newell, A.J. Transition to superparamagnetism in chains of magnetosome crystals. Geochem. Geophys. Geosyst. 2009, 10, Q11Z08. [Google Scholar] [CrossRef]
- Firlar, E.; Perez-Gonzalez, T.; Olszewska, A.; Faivre, D. Following iron speciation in the early stages of magnetite magnetosome biomineralisation. J. Mater. Res. 2016, 31, 547–555. [Google Scholar] [CrossRef]
- Wang, X.K.; Ma, Q.F.; Jiang, W.; Lv, J.; Pan, W.D.; Song, T.; Wu, L.-F. Effects of Hypomagnetic Field on Magnetosome Formation of Magnetospirillum Magneticum AMB-1. Geomicrobiol. J. 2008, 25, 296–303. [Google Scholar] [CrossRef]
- Lins, U.; Freitas, F.; Keim, C.N.; Farina, M. Electron Spectroscopic Imaging of Magnetotactic Bacteria: Magnetosome Morphology and Diversity. Microsc. Microanal. 2000, 6, 463–470. [Google Scholar] [CrossRef] [PubMed]
- Schuler, D.; Frankel, R.B. Bacterial magnetosomes: Microbiology, biomineralisation and biotechnological applications. Appl. Microbiol. Biotechnol. 1999, 52, 464–473. [Google Scholar] [CrossRef] [PubMed]
- Kalirai, S.S.; Lam, K.P.; Bazylinski, D.A.; Lins, U.; Hitchcock, A.P. Examining the chemistry and magnetism of magnetotactic bacterium Candidatus Magnetovibrio blakemorei strain MV-1 using scanning transmission X-ray microscopy. Chem. Geol. 2012, 300–301, 14–23. [Google Scholar] [CrossRef]
- Firlar, E.; Ouy, M.; Bogdanowicz, A.; Covnot, L.; Song, B.; Nadkarni, Y.; Shahbazian-Yassar, R.; Shokuhfar, T. Investigation of the magnetosome biomineralization in magnetotactic bacteria using graphene liquid cell—Transmission electron microscopy. Nanoscale 2019, 11, 698–705. [Google Scholar] [CrossRef]
- Bogachev, Y.V.; Chernenco, J.S.; Gareev, K.G.; Kononova, I.E.; Matyushkin, L.B.; Moshnikov, V.A.; Nalimova, S.S. The study of aggregation processes in colloidal solutions of magnetite–silica nanoparticles by NMR Relaxometry, AFM, and UV–vis-spectroscopy. Appl. Magn. Reson. 2014, 45, 329–337. [Google Scholar] [CrossRef]
- Yamamoto, D.; Taoka, A.; Uchihashi, T.; Sasaki, H.; Watanabe, H.; Ando, T.; Fukumori, Y. Visualization and structural analysis of the bacterial magnetic organelle magnetosome using atomic force microscopy. Proc. Natl. Acad. Sci. USA 2010, 107, 9382–9387. [Google Scholar] [CrossRef]
- Grouzdev, D.S.; Dziuba, M.V.; Kurek, D.V.; Ovchinnikov, A.I.; Zhigalova, N.A.; Kuznetsov, B.B.; Skryabin, K.G. Optimized method for preparation of IgG-binding bacterial magnetic nanoparticles. PLoS ONE 2014, 9, e109914. [Google Scholar] [CrossRef]
- Kornig, A.; Hartmann, M.A.; Teichert, C.; Fratzl, P.; Faivre, D. Magnetic force imaging of a chain of biogenic magnetite and Monte Carlo analysis of tip–particle interaction. J. Phys. D Appl. Phys. 2014, 47, 235403. [Google Scholar] [CrossRef]
- Marcuello, C.; Chambel, L.; Rodrigues, M.S.; Ferreira, L.P.; Cruz, M.M. Magnetotactic bacteria: Magnetism beyond magnetosomes. IEEE Trans. Nanobioscience 2018, 17, 555–559. [Google Scholar] [CrossRef] [PubMed]
- Józefczak, A.; Leszczyński, B.; Skumiel, A.; Hornowski, T. A comparison between acoustic properties and heat effects in biogenic (magnetosomes) and abiotic magnetite nanoparticles suspensions. J. Magn. Magn. Mater. 2016, 407, 92–100. [Google Scholar] [CrossRef]
- Józefczak, A.; Molčan, M.; Rozynek, Z.; Hornowskia, T.; Skumiel, A.; Timko, M.; Tóthová, J.; Kopčanský, P.; Leszczyński, B. Properties of magnetosome suspension under the influence of magnetic field. Acta Phys. Pol. A 2015, 127, 629–631. [Google Scholar] [CrossRef]
- Kiani, B.; Faivre, D.; Klumpp, S. Elastic properties of magnetosome chains. New J. Phys. 2015, 17, 043007. [Google Scholar] [CrossRef]
- Klumpp, S.; Kiani, B.; Vach, P.; Faivre, D. Navigation with magnetic nanoparticles: Magnetotactic bacteria and magnetic microrobots. Phys. Scr. 2015, T165, 014044. [Google Scholar] [CrossRef]
- Lefèvre, C.T.; Pósfai, M.; Abreu, F.; Lins, U.; Frankel, R.B.; Bazylinski, D.A. Morphological features of elongated-anisotropic magnetosome crystals in magnetotactic bacteria of the Nitrospirae phylum and the Deltaproteobacteria class. Earth Planet. Sci. Lett. 2011, 312, 194–200. [Google Scholar] [CrossRef]
- Moisescu, C.; Ardelean, I.I.; Benning, L.G. The effect and role of environmental conditions on magnetosome synthesis. Front. Microbiol. 2014, 5, 49. [Google Scholar] [CrossRef] [PubMed]
- Wen, T.; Zhang, Y.; Geng, Y.; Liu, J.; Basit, A.; Tian, J.; Li, Y.; Li, J.; Ju, J.; Jiang, W. Epsilon-Fe2O3 is a novel intermediate for magnetite biosynthesis in magnetotactic bacteria. Biomater. Res. 2019, 23, 13. [Google Scholar] [CrossRef]
- Prozorov, T.; Perez-Gonzalez, T.; Valverde-Tercedor, C.; Jimenez-Lopez, C.; Yebra-Rodriguez, A.; Körnig, A.; Faivre, D.; Mallapragada, S.K.; Howse, P.A.; Bazylinski, D.A.; et al. Manganese incorporation into the magnetosome magnetite: Magnetic signature of doping. Eur. J. Mineral. 2014, 26, 457–471. [Google Scholar] [CrossRef]
- Grunberg, K.; Müller, E.-C.; Otto, A.; Reszka, R.; Linder, D.; Kube, M.; Reinhardt, R.; Schüler, D. Biochemical and proteomic analysis of the magnetosome membrane in magnetospirillum gryphiswaldense. Appl. Environ. Microbiol. 2004, 70, 1040–1050. [Google Scholar] [CrossRef]
- Lang, C.; Schüler, D. Expression of green fluorescent protein fused to magnetosome proteins in microaerophilic magnetotactic bacteria. Appl. Environ. Microbiol. 2008, 74, 4944–4953. [Google Scholar] [CrossRef] [PubMed]
- Curcio, A.; de Walle, A.V.; Serrano, A.; Preveral, S.; Péchoux, C.; Pignol, D.; Menguy, N.; Lefèvre, C.T.; Espinosa, A.; Wilhelm, C. Transformation cycle of magnetosomes in human stem cells: From degradation to biosynthesis of magnetic nanoparticles anew. ACS Nano 2020, 14, 1406–1417. [Google Scholar] [CrossRef]
- Cypriano, J.; Bahri, M.; Dembelé, K.; Baaziz, W.; Leão, P.; Bazylinski, D.A.; Abreu, F.; Ersen, O.; Farina, M.; Werckmann, J. Insight on thermal stability of magnetite magnetosomes: Implications for the fossil record and biotechnology. Sci. Rep. 2020, 10, 6706. [Google Scholar] [CrossRef]
- Riese, C.N.; Uebe, R.; Rosenfeldt, S.; Schenk, A.S.; Jérôme, V.; Freitag, R.; Schüler, D. An automated oxystat fermentation regime for microoxic cultivation of Magnetospirillum gryphiswaldense. Microb. Cell Fact. 2020, 19, 206. [Google Scholar] [CrossRef] [PubMed]
- Matsunaga, T.; Kamiya, S. Use of magnetic particles isolated from magnetotactic bacteria for enzyme immobilization. Appl. Microbiol. Biotechnol. 1987, 26, 328–332. [Google Scholar] [CrossRef]
- Alphandéry, E.; Chebbi, I.; Guyot, F.; Durand-Dubief, M. Use of bacterial magnetosomes in the magnetic hyperthermia treatment of tumours: A review. Int. J. Hyperth. 2013, 29, 801–809. [Google Scholar] [CrossRef] [PubMed]
- Kirshvink, J.L. Paleomagnetic evidence for fossil biogenic magnetite in western Crete. Earth Planet. Sci. Lett. 1982, 59, 388–392. [Google Scholar] [CrossRef]
- Yamazaki, T.; Fu, W.; Shimono, T.; Usui, Y. Unmixing biogenic and terrigenous magnetic mineral components in red clay of the Pacific Ocean using principal component analyses of first-order reversal curve diagrams and paleoenvironmental implications. Earth Planets Space 2020, 72, 120. [Google Scholar] [CrossRef]
- Petersen, N.; von Dobeneck, T.; Vali, H. Fossil bacterial magnetite in deep-sea sediments from the South Atlantic Ocean. Nature 1986, 320, 611–615. [Google Scholar] [CrossRef]
- Vali, H.; von Dobeneck, T.; Amarantidis, G.; Förster, O.; Morteani, G.; Bachmann, L.; Petersen, N. Biogenic and lithogenic magnetic minerals in Atlantic and Pacific deep sea sediments and their paleomagnetic significance. Geol. Rundsch. 1989, 78, 753–764. [Google Scholar] [CrossRef]
- Yamazaki, T.; Shimono, T. Abundant bacterial magnetite occurrence in oxic red clay. Geology 2013, 41, 1191–1194. [Google Scholar] [CrossRef]
- Chang, L.; Vasiliev, I.; van Baak, C.; Krijgsman, W.; Dekkers, M.J.; Roberts, A.P.; Fitz Gerald, J.D.; van Hoesel, A.; Winklhofer, M. Identification and environmental interpretation of diagenetic and biogenic greigite in sediments: A lesson from the Messinian Black Sea. Geochem. Geophys. Geosyst. 2014, 15, 3612–3627. [Google Scholar] [CrossRef]
- Kurz, J.; Simon, K.; Heim, C.; Reitner, J.; Queric, N.V.; Volker, T. Trace element and biomarker signatures in iron-precipitating microbial mats from the tunnel of Äspö (Sweden). In Advances in Stromatolite Geobiology, Lecture Notes in Earth Sciences; Reitner, J., Quéric, N.-V., Arp, G., Eds.; Springer: Berlin, Germany, 2011; Volume 131, pp. 221–231. [Google Scholar] [CrossRef]
- Reinholdsson, M.; Snowball, I. Magnetic quantification of Fe and S bound as magnetosomal greigite in laminated sapropels in deeper basins of the Baltic Sea. Biogeosciences Discuss. 2014, 11, 729–752. [Google Scholar] [CrossRef]
- Stolz, J.F.; Chang, S.-B.R.; Kirschvink, J.L. Biogenic magnetite in stromatolites. I. Occurrence in modern sedimentary environments. Precambrian Res. 1989, 43, 295–304. [Google Scholar] [CrossRef]
- Koziaeva, V.V.; Alekseeva, L.M.; Uzun, M.M.; Leão, P.; Sukhacheva, M.V.; Patutina, E.O.; Kolganova, T.V.; Grouzdev, D.S. Biodiversity of magnetotactic bacteria in the freshwater lake Beloe Bordukovskoe, Russia. Microbiology 2020, 89, 348–358. [Google Scholar] [CrossRef]
- Petersen, N.; Weiss, D.G.; Vali, H. Magnetic bacteria in lake sediments. In Geomagnetism and Palaeomagnetism; Lowes, F.J., Collinson, D.W., Parry, J.H., Runcorn, S.K., Tozer, D.C., Soward, A., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1989; pp. 231–241. [Google Scholar] [CrossRef]
- Yamazaki, T. Reductive dissolution of biogenic magnetite. Earth Planets Space 2020, 72, 150. [Google Scholar] [CrossRef]
- Blattmann, T.M.; Lesniak, B.; García-Rubio, I.; Charilaou, M.; Wessels, M.; Eglinton, T.I.; Gehring, A.U. Ferromagnetic resonance of magnetite biominerals traces redox changes. Earth Planet. Sci. Lett. 2020, 545, 116400. [Google Scholar] [CrossRef]
- Egli, R. Characterization of individual rock magnetic components by analysis of remanence curves. 3. Bacterial magnetite and natural processes in lakes. Phys. Chem. Earth 2004, 29, 869–884. [Google Scholar] [CrossRef]
- Yamazaki, T.; Kawahata, H. Organic carbon flux controls the morphology of magnetofossils in marine sediments. Geology 1998, 26, 1064–1066. [Google Scholar] [CrossRef]
- Kopp, R.E.; Schumann, D.; Raub, T.D.; Powars, D.S.; Godfrey, L.V.; Swanson-Hysell, N.L.; Maloof, A.C.; Vali, H. An Appalachian Amazon? Magnetofossil evidence for the development of a tropical river-like system in the mid-Atlantic United States during the Paleocene-Eocene thermal maximum. Paleoceanography 2009, 24, PA4211. [Google Scholar] [CrossRef]
- Chang, S.-B.R.; Kirschvink, J.L. Biogenic magnetite as a primary remanence carrier in limestone deposits. Phys. Earth Planet. Inter. 1987, 46, 289–303. [Google Scholar] [CrossRef]
- Vali, H.; Kirschvink, J.L. Magnetofossil dissolution in a palaeomagnetically unstable deep-sea sediment. Nature 1989, 339, 203–206. [Google Scholar] [CrossRef]
- Weiss, B.P.; Kim, S.S.; Kirschvink, J.L.; Kopp, R.E.; Sankaran, M.; Kobayashi, A.; Komeili, A. Magnetic tests for magnetosome chains in Martian meteorite ALH84001. Proc. Natl. Acad. Sci. USA 2004, 101, 8281–8284. [Google Scholar] [CrossRef] [PubMed]
- Gandia, D.; Gandarias, L.; Marcano, L.; Orue, I.; Gil-Cartón, D.; Alonso, J.; García-Arribas, A.; Muela, A.; Fdez-Gubieda, M.L. Elucidating the role of shape anisotropy in faceted magnetic nanoparticles using biogenic magnetosomes as a model. Nanoscale 2020, 12, 16081–16090. [Google Scholar] [CrossRef] [PubMed]
- Kopp, R.E.; Raub, T.D.; Schumann, D.; Vali, H.; Smirnov, A.V.; Kirschvink, J.L. Magnetofossil spike during the Paleocene-Eocene thermal maximum: Ferromagnetic resonance, rock magnetic, and electron microscopy evidence from Ancora, New Jersey, United States. Paleoceanography 2007, 22, PA4103. [Google Scholar] [CrossRef]
- Kharitonskii, P.; Kamzin, A.; Gareev, K.; Valiullin, A.; Vezo, O.; Sergienko, E.; Korolev, D.; Kosterov, A.; Lebedev, S.; Gurylev, A.; et al. Magnetic granulometry and Mössbauer spectroscopy of FemOn–SiO2 colloidal nanoparticles. J. Magn. Magn. Mater. 2018, 461, 30–36. [Google Scholar] [CrossRef]
- Kharitonskii, P.; Kirillova, S.; Gareev, K.; Kamzin, A.; Gurylev, A.; Kosterov, A.; Sergienko, E.; Valiullin, A.; Shevchenko, E. Magnetic granulometry and Mössbauer spectroscopy of synthetic FemOn–TiO2 composites. IEEE Trans. Magn. 2020, 56, 7200209. [Google Scholar] [CrossRef]
- Kirschvink, J.L.; Chang, S.-B.R. Ultrafine-grained magnetite in deep-sea sediments: Possible bacterial magnetofossils. Geology 1984, 12, 559–562. [Google Scholar] [CrossRef]
- Chang, S.-B.R.; Stolz, J.F.; Kirschvink, J.L.; Awramik, S.M. Biogenic magnetite in stromatolites. II. Occurrence in ancient sedimentary environments. Precambrian Res. 1989, 43, 305–315. [Google Scholar] [CrossRef]
- Pan, Y.; Lin, W.; Tian, L.; Zhu, R.; Petersen, N. Combined approaches for characterization of an uncultivated magnetotactic coccus from Lake Miyun near Beijing. Geomicrobiol. J. 2009, 26, 313–320. [Google Scholar] [CrossRef]
- Chang, S.-B.R.; Kirschvink, J.L. Magnetofossils, the magnetization of sediments, and the evolution of magnetite biomineralization. Annu. Rev. Earth Planet. Sci. 1989, 17, 169–195. [Google Scholar] [CrossRef]
- Zhu, X.; Hitchcock, A.P.; Le Nagard, L.; Bazylinski, D.A.; Morillo, V.; Abreu, F.; Leão, P.; Lins, U. X-ray absorption spectroscopy and magnetism of synthetic greigite and greigite magnetosomes in magnetotactic bacteria. Geomicrobiol. J. 2018, 35, 215–226. [Google Scholar] [CrossRef]
- Reinholdsson, M.; Snowball, I.; Zillén, L.; Lenz, C.; Conley, D.J. Magnetic enhancement of Baltic Sea sapropels by greigite magnetofossils. Earth Planet. Sci. Lett. 2013, 366, 137–150. [Google Scholar] [CrossRef]
- Yan, L.; Yue, X.; Zhang, S.; Chen, P.; Xu, Z.; Li, Y.; Li, H. Biocompatibility evaluation of magnetosomes formed by Acidithiobacillus ferrooxidans. Mater. Sci. Eng. C 2012, 32, 1802–1807. [Google Scholar] [CrossRef] [PubMed]
- Cypriano, J.; Werckmann, J.; Vargas, G.; dos Santos, A.L.; Silva, K.T.; Leão, P.; Almeida, F.P.; Bazylinski, D.A.; Farina, M.; Lins, U.; et al. Uptake and persistence of bacterial magnetite magnetosomes in a mammalian cell line: Implications for medical and biotechnological applications. PLoS ONE 2019, 14, e0215657. [Google Scholar] [CrossRef]
- Jacob, J.J. Suthindhiran. K. Magnetotactic bacteria and magnetosomes—Scope and challenges. Mater. Sci. Eng. C 2016, 68, 919–928. [Google Scholar] [CrossRef] [PubMed]
- Prabhu, N.N.; Kowshik, M. Magnetosomes: The bionanomagnets and its potential use in biomedical applications. J. Nanomed. Res. 2016, 3, 00057. [Google Scholar] [CrossRef]
- Kuzajewska, D.; Wszołek, A.; Żwierełło, W.; Kirczuk, L.; Maruszewska, A. Magnetotactic bacteria and magnetosomes as smart drug delivery systems: A newweapon on the battlefield with cancer? Biology 2020, 9, 102. [Google Scholar] [CrossRef]
- Fdez-Gubieda, M.L.; Alonso, J.; García-Prieto, A.; García-Arribas, A.; Fernández Barquín, L.; Muela, A. Magnetotactic bacteria for cancer therapy. J. Appl. Phys. 2020, 128, 070902. [Google Scholar] [CrossRef]
- Felfoul, O.; Mokrani, N.; Mohammadi, M.; Martel, S. Effect of the chain of magnetosomes embedded in magnetotactic bacteria and their motility on magnetic resonance imaging. In Proceedings of the 32nd Annual International Conference of the IEEE EMBS, Buenos Aires, Argentina, 31 August–4 September 2010. [Google Scholar] [CrossRef]
- Herborn, C.U.; Papanikolaou, N.; Reszka, R.; Grünberg, K.; Schüler, D.; Debatin, J.F. Magnetosomes as biological model for iron binding: Relaxivity determination with MRI. Fortschr. Röntgenstr. 2003, 175, 830–834. [Google Scholar] [CrossRef]
- Martel, S.; Mohammadi, M.; Felfoul, O.; Lu, Z.; Pouponneau, P. Flagellated magnetotactic bacteria as controlled mri-trackable propulsion and steering systems for medical nanorobots operating in the human microvasculature. Int. J. Robot. Res. 2009, 28, 571–582. [Google Scholar] [CrossRef] [PubMed]
- Gandia, D.; Gandarias, L.; Rodrigo, I.; Robles-García, J.; Das, R.; Garaio, E.; García, J.A.; Phan, M.-H.; Srikanth, H.; Orue, I.; et al. Unlocking the potential of magnetotactic bacteria as magnetic hyperthermia agents. Small 2019, 15, 1902626. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Chen, L.; Wang, P.; Wu, L.-F.; Song, T. Steering of magnetotactic bacterial microrobots by focusing magnetic field for targeted pathogen killing. J. Magn. Magn. Mater. 2019, 479, 74–83. [Google Scholar] [CrossRef]
- Boucher, M.; Geffroy, F.; Prévéral, S.; Bellanger, L.; Selingue, E.; Adryanczyk-Perrier, G.; Péan, M.; Lefévre, C.T.; Pignol, D.; Ginet, N.; et al. Genetically tailored magnetosomes used as MRI probe for molecular imaging of brain tumor. Biomaterials 2017, 121, 167–178. [Google Scholar] [CrossRef] [PubMed]
- Xiang, Z.; Yang, X.; Xu, J.; Lai, W.; Wang, Z.; Hu, Z.; Tian, J.; Geng, L.; Fang, Q. Tumor detection using magnetosome nanoparticles functionalized with a newly screened EGFR/HER2 targeting peptide. Biomaterials 2017, 115, 53e64. [Google Scholar] [CrossRef]
- Sun, Q.; Prato, F.S.; Goldhawk, D.E. Optimizing reporter gene expression for molecular magnetic resonance imaging. In Bioimaging; CRC Press: London, UK, 2020. [Google Scholar]
- Yoshino, T.; Hirabe, H.; Takahashi, M.; Kuhara, M.; Takeyama, H.; Matsunaga, T. Magnetic cell separation using nano-sized bacterial magnetic particles with reconstructed magnetosome membrane. Biotechnol. Bioeng. 2008, 101, 470–477. [Google Scholar] [CrossRef]
- Wacker, R.; Ceyhan, B.; Alhorn, P.; Schueler, D.; Lang, C.; Niemeyer, C.M. Magneto Immuno-PCR: A novel immunoassay based on biogenic magnetosome nanoparticles. Biochem. Biophys. Res. Commun. 2007, 357, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Gao, B.; Zhang, F.; Sun, X.; Zhang, Y.; Li, Z. A novel electrochemical immunosensor based on magnetosomes for detection of staphylococcal enterotoxin B in milk. Talanta 2013, 106, 360–366. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Tian, J.; Xu, J.; Wang, K.; Li, J.; Gee, S.J.; Hammock, B.D.; Li, Q.X.; Xu, T. Strong and oriented conjugation of nanobodies onto magnetosomes for the development of a rapid immunomagnetic assay for the environmental detection of tetrabromobisphenol-A. Anal. Bioanal. Chem. 2018, 410, 6633–6642. [Google Scholar] [CrossRef]
- Nakagawa, T.; Hashimoto, R.; Maruyama, K.; Tanaka, T.; Takeyama, H.; Matsunaga, T. Capture and release of DNA using aminosilane-modified bacterial magnetic particles for automated detection system of single nucleotide polymorphisms. Biotechnol. Bioeng. 2006, 94, 862–868. [Google Scholar] [CrossRef]
- Yoza, B.; Matsumoto, M.; Matsunaga, T. DNA extraction using modified bacterial magnetic particles in the presence of amino silane compound. J. Biotechnol. 2002, 94, 217–224. [Google Scholar] [CrossRef]
- Mickoleit, F.; Schüler, D. Generation of multifunctional magnetic nanoparticles with amplified catalytic activities by genetic expression of enzyme arrays on bacterial magnetosomes. Adv. Biosys. 2018, 2, 1700109. [Google Scholar] [CrossRef]
- Mickoleit, F.; Schüler, D. Generation of nanomagnetic biocomposites by genetic engineering of bacterial magnetosomes. Bioinspired Biomim. Nanobiomater. 2019, 8, 86–98. [Google Scholar] [CrossRef]
- Ren, E.; Lei, Z.; Wang, J.; Zhang, Y.; Liu, G. Magnetosome modification: From bio-nano engineering toward nanomedicine. Adv. Therap. 2018, 1, 1800080. [Google Scholar] [CrossRef]
- Sun, J.; Li, Y.; Liang, X.-J.; Wang, P.C. Bacterial magnetosome: A novel biogenetic magnetic targeted drug carrier with potential multifunctions. J. Nanomater. 2011, 2011, 469031. [Google Scholar] [CrossRef] [PubMed]
- Alphandéry, E. Applications of magnetosomes synthesized by magnetotactic bacteria in medicine. Front. Bioeng. Biotechnol. 2014, 2, 5. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Liu, J.; Tong, J.; Tang, T.; Kong, W.-C.; Wang, X.; Li, Y.; Tang, J. Heating effect and biocompatibility of bacterial magnetosomes as potential materials used in magnetic fluid hyperthermia. Prog. Nat. Sci. Mater. Int. 2012, 22, 31–39. [Google Scholar] [CrossRef]
- Toropova, Y.G.; Golovkin, A.S.; Malashicheva, A.B.; Korolev, D.V.; Gorshkov, A.N.; Gareev, K.G.; Afonin, M.V.; Galagudza, M.M. In vitro toxicity of FemOn, FemOn-SiO2 composite, and SiO2-FemOn core-shell magnetic nanoparticles. Int. J. Nanomed. 2017, 12, 593–603. [Google Scholar] [CrossRef]
- Le Fèvre, R.; Durand-Dubief, M.; Chebbi, I.; Mandawala, C.; Lagroix, F.; Valet, J.-P.; Idbaih, A.; Adam, C.; Delattre, J.-Y.; Schmitt, C.; et al. Enhanced antitumor efficacy of biocompatible magnetosomes for the magnetic hyperthermia treatment of glioblastoma. Theranostics 2017, 7, 4618–4631. [Google Scholar] [CrossRef]
- Wang, P.P.; Chen, C.; Chen, C.; Li, Y.; Pan, W.; Song, T. The interaction of bacterial magnetosomes and human liver cancer cells in vitro. J. Magn. Magn. Mater. 2017, 427, 105–110. [Google Scholar] [CrossRef]
- Geng, Y.; Wang, J.; Wang, X.; Liu, J.; Zhang, Y.; Niu, W.; Basit, A.; Liu, W.; Jiang, W. Growth-inhibitory effects of anthracycline-loaded bacterial magnetosomes against hepatic cancer in vitro and in vivo. Nanomedicine 2019, 14, 1663–1680. [Google Scholar] [CrossRef]
- Wang, X.; Wang, J.; Geng, Y.; Wang, J.; Zhang, X.; Yang, S.; Jiang, W.; Liu, W. An enhanced anti-tumor effect of apoptin-cecropin B on human hepatoma cells by using bacterial magnetic particle gene delivery system. Biochem. Biophys. Res. Commun. 2018, 496, 719–725. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Geng, Y.; Zhang, Y.; Wang, X.; Liu, J.; Basit, A.; Miao, T.; Liu, W.; Jiang, W. Bacterial magnetosomes loaded with doxorubicin and transferrin improve targeted therapy of hepatocellular carcinoma. Nanotheranostics 2019, 3, 284–298. [Google Scholar] [CrossRef]
- Macías-Martínez, B.I.; Cortés-Hernández, D.A.; Zugasti-Cruz, A.; Cruz-Ortíz, A.; Múzquiz-Ramos, E.M. Original. Heating ability and hemolysis test of magnetite nanoparticles obtained by a simple co-precipitation method. J. Appl. Res. Technol. 2016, 14, 239–244. [Google Scholar] [CrossRef]
- Mannucci, S.; Tambalo, S.; Conti, G.; Ghin, L.; Milanese, A.; Carboncino, A.; Nicolato, E.; Marinozzi, M.R.; Benati, D.; Bassi, R.; et al. Magnetosomes extracted from magnetospirillum gryphiswaldense as theranostic agents in an experimental model of glioblastoma. Contrast Media Mol. Imaging 2018, 2018, 2198703. [Google Scholar] [CrossRef]
- Briceño, S.; Hernandez, A.C.; Sojo, J.; Lascano, L.; Gonzalez, G. Degradation of magnetite nanoparticles in biomimetic media. J. Nanopart. Res. 2017, 19, 140. [Google Scholar] [CrossRef]
- Chen, C.; Chen, L.; Wang, P.; Wu, L.-F.; Song, T. Magnetically-induced elimination of Staphylococcus aureus by magnetotactic bacteria under a swing magnetic field. Nanomed. Nanotechnol. Biol. Med. 2017, 13, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Cai, F.; Li, J.; Sun, J.; Ji, Y. Biosynthesis of gold nanoparticles by biosorption using Magnetospirillum gryphiswaldense MSR-1. Chem. Eng. J. 2011, 175, 70–75. [Google Scholar] [CrossRef]
- Alphandéry, E.; Guyot, F.; Chebbi, I. Preparation of chains of magnetosomes, isolated from Magnetospirillum magneticum strain AMB-1 magnetotactic bacteria, yielding efficient treatment of tumors using magnetic hyperthermia. Int. J. Pharm. 2012, 434, 444–452. [Google Scholar] [CrossRef]
- Alphandéry, E.; Idbaih, A.; Adame, C.; Delattre, J.-Y.; Schmitt, C.; Guyot, F.; Chebbi, I. Chains of magnetosomes with controlled endotoxin release and partial tumor occupation induce full destruction of intracranial U87-Luc glioma in mice under the application of an alternating magnetic field. J. Control. Release 2017, 262, 259–272. [Google Scholar] [CrossRef]
- Alphandéry, E.; Idbaih, A.; Adam, C.; Delattre, J.-Y.; Schmitt, C.; Gazeau, F.; Guyot, F.; Chebbi, I. Biodegraded magnetosomes with reduced size and heating power maintain a persistent activity against intracranial U87-Luc mouse GBM tumors. J. Nanobiotechnol. 2019, 17, 126. [Google Scholar] [CrossRef] [PubMed]
- Mathuriya, A.S. Magnetotactic bacteria for cancer therapy. Biotechnol. Lett. 2015, 37, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Felfoul, O.; Mohammadi, M.; Taherkhani, S.; De Lanauze, D.; Zhong Xu, Y.; Loghin, D.; Essa, S.; Jancik, S.; Houle, D.; Lafleur, M.; et al. Magneto-aerotactic bacteria deliver drug-containing nanoliposomes to tumour hypoxic regions. Nat. Nanotechnol. 2016, 11, 941–947. [Google Scholar] [CrossRef] [PubMed]

| MTB Species | Crystal Shape | Average Crystal Length (nm) | Average Crystal Width (nm) | Average Magnetosomes Quantity | Refs. |
|---|---|---|---|---|---|
| Magnetospirillum magneticum AMB-1 | prismatic, cuboctahedral | 30–50 | 15–20 | [32,35,73,75,76,77,78] | |
| Magnetococcales spp. | prismatic, cuboctahedral | 70–250 | 40–200 | 10–200 | [49,52,72,79,80,81] |
| Magnetovibrio blakemorei MV-1 | prismatic | 50–60 | 35–40 | 2–20 | [74,82] |
| Magnetospirillum gryphiswaldense MSR-1 | cuboctahedral | 30–50 | 15–40 | [43,63,68,83,84,85] | |
| Desulfovibrio magneticus RS-1 | bullet-shaped | 40–70 | 20–40 | <10 | [86] |
| Nitrospirota bacterium MYR-1 | bullet-shaped | 40–45 | 80–180 | up to 1000 | [87] |
| Ca. Magnetobacterium bavaricum TM-1 | bullet-shaped | 35–40 | 100–120 | [88,89] | |
| MTB Species | Ms300K, 10–3 emu 1 | Hcr300K, Oe 2 | Mrs/Ms300K3 | TV, K 4 | Refs. |
|---|---|---|---|---|---|
| Magnetospirillum magnetotacticum AMB-1 | 1.6–4.5 | 128–380 | 0.43–0.50 | 100–118 | [39,77,106,107,108,109,110,111] |
| Magnetospirillum gryphiswaldense MSR-1 | 0.2–1.5 | 96–234 | 0.38–0.50 | [68,85,112,113,114,115,116,117] | |
| Magnetovibrio blakemorei MV-1 | 3.0 | 250–350 | 0.46–0.50 | 100–117 | [82,118] |
| Ca. Magnetobacterium bavaricum TM-1 | 1.5–8.0 | 400–460 | 0.32–0.51 | 100–110 | [88,89] |
| Magnetococcus marinus MC-1 | – | – | – | 102 | [118] |
| Characteristic | Synthetic Particles 1 | Magnetosomes | Refs. |
|---|---|---|---|
| Acute toxicity (LD50) in rats, mg/kg | 135–180 | 480 | [146,201] |
| Cytotoxicity (HUVEC 2 MTT assay), mg/mL | 0.07–1.0 3 | 0.1–5.1 | [176,177,178,188,189,200,202] |
| Maximum non-hemolytic concentration, mg/mL | 3.0 | 1.6–4.0 | [176,189,208] |
| Magnetic resonance relaxivity r2, mM−1 s−1 | 130–170 | 150–600 | [127,182,187,189,200] |
| Specific absorption rate, kW/g | 0.6–0.8 | 0.4–1.4 | [132,183,185,209] |
| Minimal biodegradation period, days | 30 | 28–42 | [177,200,201,210] |
| Magneto Immuno-PCR limit of detection, ng/mL | 8 | 0.32 | [191] |
| DNA extraction release, μg/mg | 1.0–2.2 | 3.0–19.2 | [195] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gareev, K.G.; Grouzdev, D.S.; Kharitonskii, P.V.; Kosterov, A.; Koziaeva, V.V.; Sergienko, E.S.; Shevtsov, M.A. Magnetotactic Bacteria and Magnetosomes: Basic Properties and Applications. Magnetochemistry 2021, 7, 86. https://doi.org/10.3390/magnetochemistry7060086
Gareev KG, Grouzdev DS, Kharitonskii PV, Kosterov A, Koziaeva VV, Sergienko ES, Shevtsov MA. Magnetotactic Bacteria and Magnetosomes: Basic Properties and Applications. Magnetochemistry. 2021; 7(6):86. https://doi.org/10.3390/magnetochemistry7060086
Chicago/Turabian StyleGareev, Kamil G., Denis S. Grouzdev, Petr V. Kharitonskii, Andrei Kosterov, Veronika V. Koziaeva, Elena S. Sergienko, and Maxim A. Shevtsov. 2021. "Magnetotactic Bacteria and Magnetosomes: Basic Properties and Applications" Magnetochemistry 7, no. 6: 86. https://doi.org/10.3390/magnetochemistry7060086
APA StyleGareev, K. G., Grouzdev, D. S., Kharitonskii, P. V., Kosterov, A., Koziaeva, V. V., Sergienko, E. S., & Shevtsov, M. A. (2021). Magnetotactic Bacteria and Magnetosomes: Basic Properties and Applications. Magnetochemistry, 7(6), 86. https://doi.org/10.3390/magnetochemistry7060086








