Long-Term Irrigation with Treated Municipal Wastewater from the Wadi-Musa Region: Soil Heavy Metal Accumulation, Uptake and Partitioning in Olive Trees
Abstract
1. Introduction
2. Materials and Methods
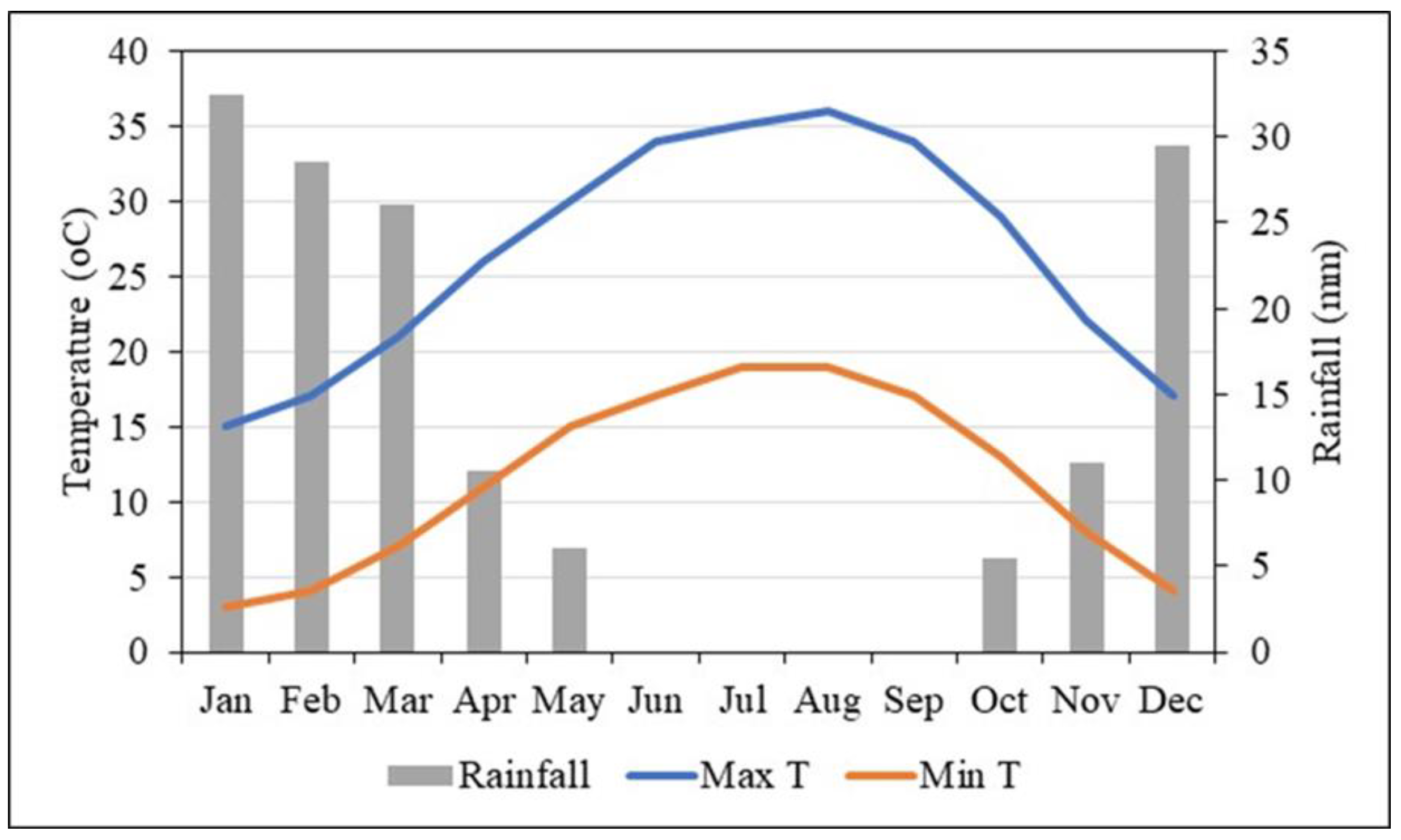
2.1. Study Area, Soil, and Water Characteristics
2.2. Sampling and Analysis
2.2.1. Plant Sampling
2.2.2. Irrigation Water and Soil Sampling
2.2.3. Heavy Metals Analysis of Plant, Soil and Water
2.2.4. Bioconcentration and Translocation Factors
2.3. Statistical Analysis
3. Results and Discussion
3.1. Chemical Analysis of Irrigation Water and Soil
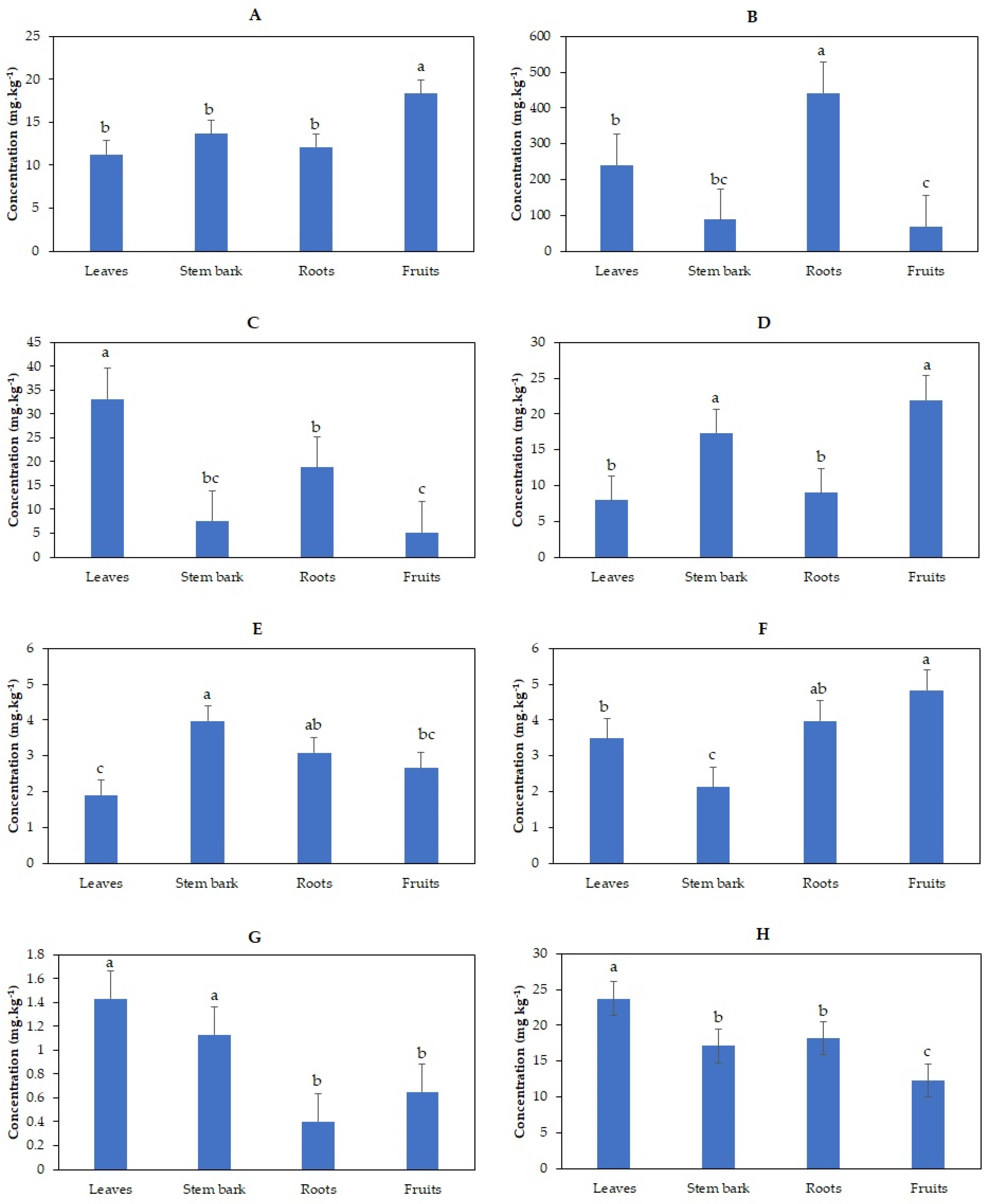
3.2. Effects of Irrigation with TWW on Heavy Metals Content in Olive Trees
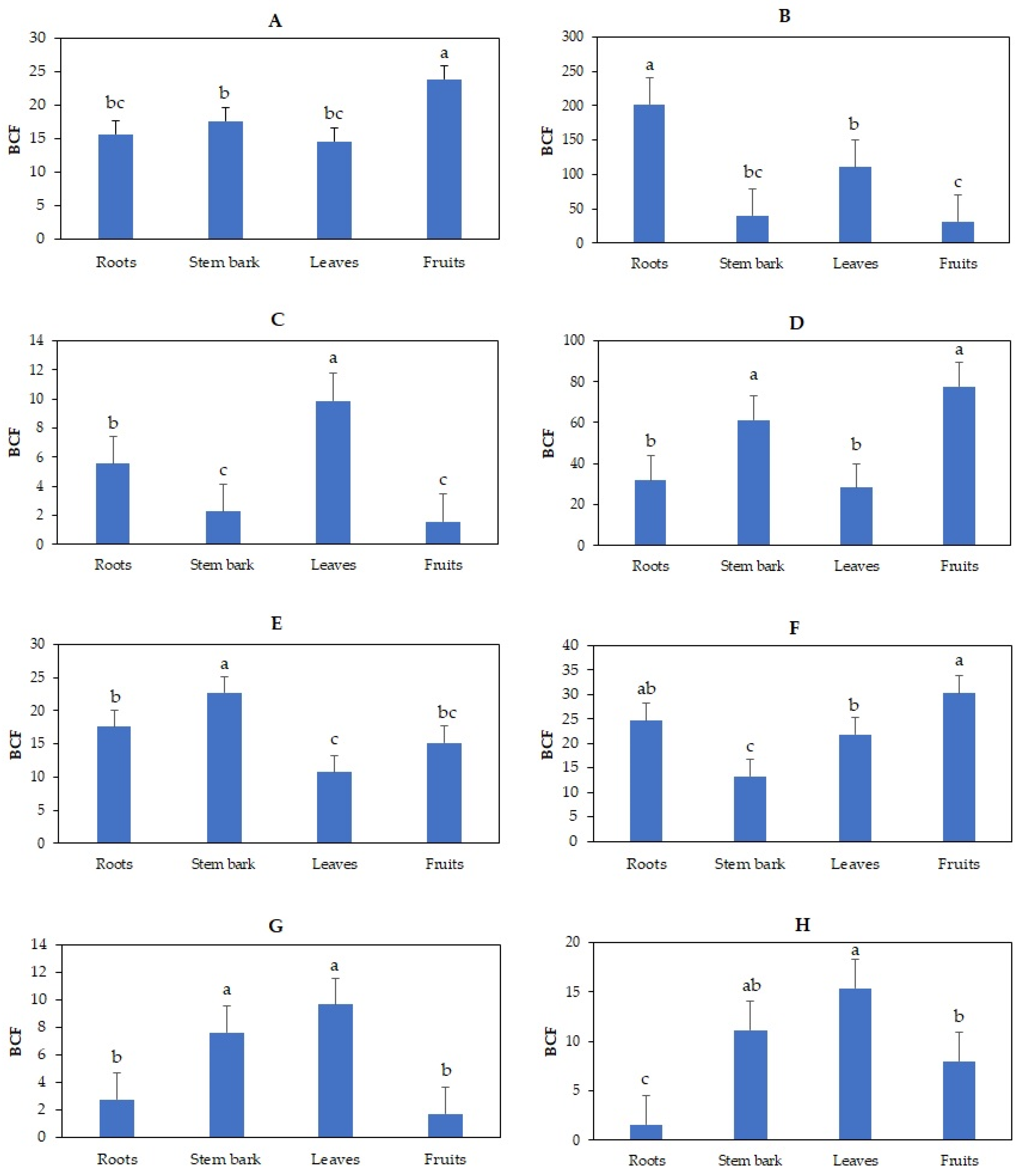
3.3. Effects of Irrigation with TWW on Bioconcentration Factor of Heavy Metals in Olive Trees
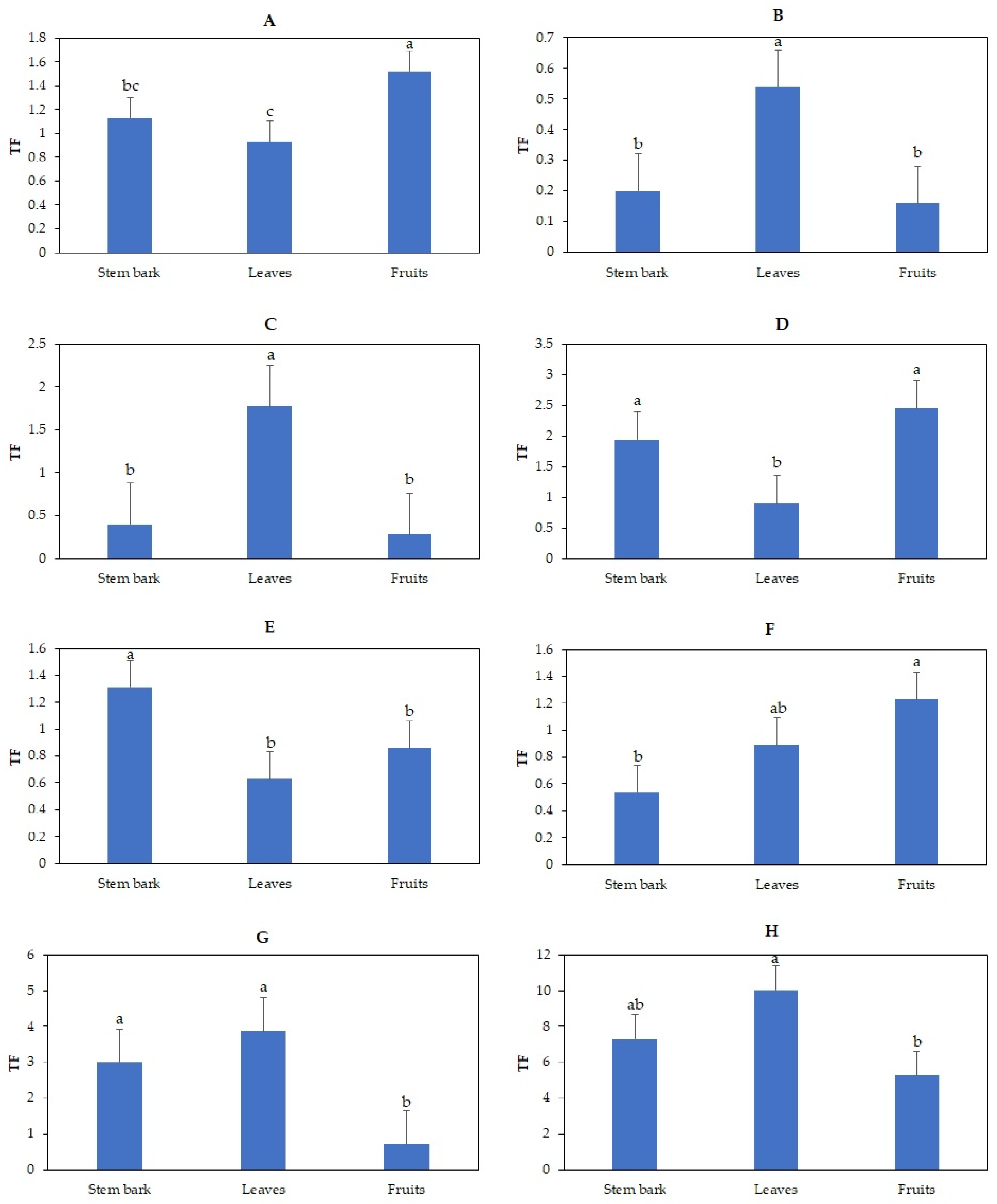
3.4. Effects of Irrigation with TWW on Translocation Factor of Heavy Metal in Olive Trees
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Al-Bakri, J.; Suleiman, A.; Abdulla, F.; Ayad, J. Potential impact of climate change on rainfed agriculture of a semi-arid basin in Jordan. Phys. Chem. Earth Parts A/B/C 2011, 36, 125–134. [Google Scholar] [CrossRef]
- Rad, S.V.; Valadabadi, S.A.R.; Pouryousef, M.; Saifzadeh, S.; Zakrin, H.R.; Mastinu, A. Quantitative and Qualitative Evaluation of Sorghum bicolor L. under Intercropping with Legumes and Different Weed Control Methods. Horticulturae 2020, 6, 78. [Google Scholar] [CrossRef]
- Al-Satari, Y.; Al-Ramamneh, E.A.-D.; Ayad, J.; Abu Dalbouh, M.; Amayreh, I.; Khreisat, Z. Impact of seedling age on the survival and productivity of Atriplex halimus shrubs in drought-affected rangelands of Jordan. Rangel. J. 2018, 40, 287–296. [Google Scholar] [CrossRef]
- United Nations Development Programme; Sustainable development goals; UNDP Jordan: Amman, Jordan, 2019.
- Qtaishat, T. Water Policy in Jordan. In Water Policies in MENA Countries, 1st ed.; Zekri, S., Ed.; Global Issues in Water Policy; Springer: Cham, Germany, 2020; Volume 23. [Google Scholar] [CrossRef]
- Heras, J.D.L.; Mañas, P. Reclaimed Wastewater to Irrigate Olive Groves and Vineyards: Effects on Soil Properties. Agronomy 2020, 10, 649. [Google Scholar] [CrossRef]
- Gharsallaoui, M.; Benincasa, C.; Ayadi, M.; Perri, E.; Khlif, M.; Gabsi, S. Study on the impact of wastewater irrigation on the quality of oils obtained from olives harvested by hand and from the ground and extracted at different times after the harvesting. Sci. Hortic. 2011, 128, 23–29. [Google Scholar] [CrossRef]
- DOS. Department of Statistics Year Book; Jordan Department of Statistics: Amman, Jordan, 2019. Available online: http://www.dos.gov.jo (accessed on 15 March 2021).
- Duqqah, M.M. Treated Sewagewater Use in Irrigated Agriculture: Theoretical Design of Farming Systems in Seil Al Zarqa and the Middle Jordan Valley in Jordan. Ph.D. Thesis, Wageningen University, Wageningen, The Netherlands, 2002. [Google Scholar]
- Stietiya, M.H.; Duqqah, M.; Udeigwe, T.; Zubi, R.; Ammari, T. Fate and Distribution of Heavy Metals in Wastewater Irrigated Calcareous Soils. Sci. World J. 2014, 2014, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Al-Nakshabandi, G.; Saqqar, M.; Shatanawi, M.; Fayyad, M.; Al-Horani, H. Some environmental problems associated with the use of treated wastewater for irrigation in Jordan. Agric. Water Manag. 1997, 34, 81–94. [Google Scholar] [CrossRef]
- Al-Zu’Bi, Y. Effect of irrigation water on agricultural soil in Jordan valley: An example from arid area conditions. J. Arid. Environ. 2007, 70, 63–79. [Google Scholar] [CrossRef]
- Ghrefat, H.A.; Yusuf, N.; Jamarh, A.; Nazzal, J. Fractionation and risk assessment of heavy metals in soil samples collected along Zerqa River, Jordan. Environ. Earth Sci. 2012, 66, 199–208. [Google Scholar] [CrossRef]
- Abderahman, N.; Abu-Rukah, Y.H. An assessment study of heavy metal distribution within soil in upper course of Zarqa River basin/Jordan. Environ. Earth Sci. 2006, 49, 1116–1124. [Google Scholar] [CrossRef]
- Al Farajat, M.; Al Ansari, N.; Mashagbeh, A.; Abou Salah, R. Hydrogeophysical and environmental investigations of groundwater potentials in Al Sokhna alluvium aquifer in Zarqa—Jordan. Hydrogeol. Umw. 2005, 7, 1–15. [Google Scholar]
- Mishra, A.K.; Sahu, A.; Deepika, A.S.; Gauba, P. Phytoremediation of Heavy Metals. J. Pharm. Res. 2014, 8, 1233–1238. [Google Scholar]
- Li, Z.; Ma, Z.; van der Kuijp, T.J.; Yuan, Z.; Huang, L. A review of soil heavy metal pollution from mines in China: Pollution and health risk assessment. Sci. Total. Environ. 2014, 468–469, 843–853. [Google Scholar] [CrossRef] [PubMed]
- Toze, S. Reuse of effluent water—Benefits and risks. Agric. Water Manag. 2006, 80, 147–159. [Google Scholar] [CrossRef]
- Al-Hiary, M.A.H. Assessing Competitiveness of Jordanian Olive Production a Policy Analysis Matrix Pam. J. Stud. Manag. Plan. 2015, 1, 19–29. [Google Scholar]
- Ministry of Agriculture, Annual Reports, Department of Studies and Statistics, Amman-Jordan. 2016. Available online: www.moa.gov.jo (accessed on 15 March 2021).
- Ayoub, S.; Al-Shdiefat, S.; Rawashdeh, H.; Bashabsheh, I. Utilization of reclaimed wastewater for olive irrigation: Effect on soil properties, tree growth, yield and oil content. Agric. Water Manag. 2016, 176, 163–169. [Google Scholar] [CrossRef]
- Filippou, M.; Fasseas, C.; Karabourniotis, G. Photosynthetic characteristics of olive tree (Olea europaea) bark. Tree Physiol. 2007, 27, 977–984. [Google Scholar] [CrossRef]
- Bourazanis, G.; Roussos, P.; Argyrokastritis, I.; Kosmas, C.; Kerkides, P. Evaluation of the use of treated municipal waste water on the yield, oil quality, free fatty acids’ profile and nutrient levels in olive trees cv Koroneiki, in Greece. Agric. Water Manag. 2016, 163, 1–8. [Google Scholar] [CrossRef]
- Freihat, N.M.; Shannag, H.K.; Alkelani, M.A. Effects of supplementary irrigation on performance of ‘Nabali’ and ‘Grossa de Spain’ olives under semi-arid conditions in Jordan. Sci. Hortic. 2021, 275, 109696. [Google Scholar] [CrossRef]
- Al-Mefleh, N.; Othman, Y.; Tadros, M.; Al-Assaf, A.; Talozi, S. An Assessment of Treated Greywater Reuse in Irrigation on Growth and Protein Content of Prosopis and Albizia. Horticulturae 2021, 7, 38. [Google Scholar] [CrossRef]
- Bedbabis, S.; Trigui, D.; Ben Ahmed, C.; Clodoveo, M.L.; Camposeo, S.; Vivaldi, G.A.; Ben Rouina, B. Long-terms effects of irrigation with treated municipal wastewater on soil, yield and olive oil quality. Agric. Water Manag. 2015, 160, 14–21. [Google Scholar] [CrossRef]
- Gizgis, N.; Georgiou, M.; Diamadopoulos, E. Sequential anaerobic/aerobic biological treatment of olive mill wastewater and municipal wastewater. J. Chem. Technol. Biotechnol. 2006, 81, 1563–1569. [Google Scholar] [CrossRef]
- Bozkurt, M.A.; Yarilgaç, T. The effects of sewage sludge applications on the yield, growth, nutrition and heavy metal accu-mulation in apple trees growing in dry conditions. Turk. J. Agric. For. 2003, 27, 285–292. [Google Scholar]
- Guo, J.; Yue, T.; Li, X.; Yuan, Y. Heavy metal levels in kiwifruit orchard soils and trees and its potential health risk assessment in Shaanxi, China. Environ. Sci. Pollut. Res. 2016, 23, 14560–14566. [Google Scholar] [CrossRef]
- Peryea, F.J. Heavy metal contamination in deciduous tree fruit orchards: Implications for mineral nutrient management. Acta Hortic. 2001, 564, 31–39. [Google Scholar] [CrossRef]
- Wilson, B.; Pyatt, F.B. Heavy Metal Bioaccumulation by the Important Food Plant, Olea europaea L., in an Ancient Metalliferous Polluted Area of Cyprus. Bull. Environ. Contam. Toxicol. 2007, 78, 390–394. [Google Scholar] [CrossRef]
- Batarseh, M.I.; Rawajfeh, A.; Ioannis, K.K.; Prodromos, K.H. Treated Municipal Wastewater Irrigation Impact on Olive Trees (Olea Europaea L.) at Al-Tafilah, Jordan. Water Air Soil Pollut. 2010, 217, 185–196. [Google Scholar] [CrossRef]
- Luka, M.F.; Akun, E. Investigation of trace metals in different varieties of olive oils from northern Cyprus and their variation in accumulation using ICP-MS and multivariate techniques. Environ. Earth Sci. 2019, 78, 578. [Google Scholar] [CrossRef]
- López-López, A.; López, R.; Madrid, F.; Garrido-Fernández, A.; López-Núñez, R. Heavy Metals and Mineral Elements Not Included on the Nutritional Labels in Table Olives. J. Agric. Food Chem. 2008, 56, 9475–9483. [Google Scholar] [CrossRef] [PubMed]
- Paithankar, J.G.; Saini, S.; Dwivedi, S.; Sharma, A.; Chowdhuri, D.K. Heavy metal associated health hazards: An interplay of oxidative stress and signal transduction. Chemosphere 2021, 262, 128350. [Google Scholar] [CrossRef] [PubMed]
- Järup, L. Hazards of heavy metal contamination. Br. Med. Bull. 2003, 68, 167–182. [Google Scholar] [CrossRef] [PubMed]
- Addison, E. Political effluent: Implementing wastewater re-use in Wadi Musa, Jordan. Arid. Newsl. University of Arizona: Tucson, AZ, USA, 2005; Volume 57, pp. 1–18. ISSN 1092-5481. Available online: http://ag.arizona.edu/oals/ALN/aln57/addison.html (accessed on 24 March 2021).
- JISM. Water—Reclaimed Grey Water (JS 1776:2013); Jordan Standards and Metrology Organization: Amman, Jordan, 2013; pp. 1–12.
- WHO. Guidelines for the Safe Use of Wastewater, Excreta and Greywater; World Health Organization: Geneva, Switzerland, 2006; Volume 2. [Google Scholar]
- Duncan, H.J.; Flowers, T.H.; Pulford, I.D.; Wilson, W.D. Analytical Procedures for the Analysis of Heavy Metals in Contaminated Soils. In Contaminated Soil’95, 2nd ed.; Van Den Brink, W.J., Bosman, R., Arendt, F., Eds.; Soil & Environment; Springer: Dordrecht, The Netherlands, 1995; Volume 5, pp. 731–732. [Google Scholar]
- Estefan, G.; Sommer, R.; Ryan, J. Methods of Soil, Plant, and Water Analysis. A Manual for the West Asia and North Africa Region, 3rd ed.; ICARDA: Beirut, Lebanon, 2013; pp. 20–199. [Google Scholar]
- Mellem, J.J. Bioaccumulation of Cr, Hg, As, Pb, Cu and Ni with the ability for hyperaccumulation by Amaranthus dubius. Afr. J. Agric. Res. 2012, 7, 591–596. [Google Scholar] [CrossRef]
- Al-Khashman, O.A. Chemical Evaluation of Ma’an Sewage Effluents and its Reuse in Irrigation Purposes. Water Resour. Manag. 2008, 23, 1041–1053. [Google Scholar] [CrossRef]
- Bedbabis, S.; Ben Rouina, B.; Boukhris, M.; Ferrara, G. Effect of irrigation with treated wastewater on soil chemical properties and infiltration rate. J. Environ. Manag. 2014, 133, 45–50. [Google Scholar] [CrossRef]
- Emongor, V.; Ramolemana, G. Treated sewage effluent (water) potential to be used for horticultural production in Botswana. Phys. Chem. Earth Parts A/B/C 2004, 29, 1101–1108. [Google Scholar] [CrossRef]
- Heidarpour, M.; Mostafazadeh-Fard, B.; Koupai, J.A.; Malekian, R. The effects of treated wastewater on soil chemical properties using subsurface and surface irrigation methods. Agric. Water Manag. 2007, 90, 87–94. [Google Scholar] [CrossRef]
- Klay, S.; Charef, A.; Ayed, L.; Houman, B.; Rezgui, F. Effect of irrigation with treated wastewater on geochemical properties (saltiness, C, N and heavy metals) of isohumic soils (Zaouit Sousse perimeter, Oriental Tunisia). Desalination 2010, 253, 180–187. [Google Scholar] [CrossRef]
- Dikinya, O.; Areola, O. Comparative analysis of heavy metal concentration in secondary treated wastewater irrigated soils cultivated by different crops. Int. J. Environ. Sci. Technol. 2010, 7, 337–346. [Google Scholar] [CrossRef]
- Al-Absi, K.; Al-Nasir, F.; Mahadeen, A. Mineral content of three olive cultivars irrigated with treated industrial wastewater. Agric. Water Manag. 2009, 96, 616–626. [Google Scholar] [CrossRef]
- Ernst, W.H.O. Evolution of metal tolerance of plants. For. Snow Landsc. Res. 2006, 80, 251–274. [Google Scholar]
- Al-Lahham, O.; El Assi, N.; Fayyad, M. Translocation of heavy metals to tomato (Solanum lycopersicom L.) fruit irrigated with treated wastewater. Sci. Hortic. 2007, 113, 250–254. [Google Scholar] [CrossRef]
- Petousi, I.; Fountoulakis, M.; Saru, M.; Nikolaidis, N.; Fletcher, L.; Stentiford, E.; Manios, T. Effects of reclaimed wastewater irrigation on olive (Olea europaea L. cv. ‘Koroneiki’) trees. Agric. Water Manag. 2015, 160, 33–40. [Google Scholar] [CrossRef]
- Friedman, H.; Bernstein, N.; Bruner, M.; Rot, I.; Ben-Noon, Z.; Zuriel, A.; Zuriel, R.; Finkelstein, S.; Umiel, N.; Hagiladi, A. Application of secondary-treated effluents for cultivation of sunflower (Helianthus annuus L.) and celosia (Celosia argentea L.) as cut flowers. Sci. Hortic. 2007, 115, 62–69. [Google Scholar] [CrossRef]
- Pulford, I.D.; Dickinson, N.M. Phytoremediation Technologies Using Trees. In Trace Elements in the Environment: Biogeochemistry, Biotechnology, and Bioremediation, 1st ed.; Prasad, M.N.V., Sajwan, K.S., Naidu, R., Eds.; CRC Press: New York, NY, USA, 2005; Volume 383, pp. 283–404. [Google Scholar] [CrossRef]
- Zhao, X.; Liu, J.; Xia, X.; Chu, J.; Wei, Y.; Shi, S.; Chang, E.; Yin, W.; Jiang, Z. The evaluation of heavy metal accumulation and application of a comprehensive bio-concentration index for woody species on contaminated sites in Hunan, China. Environ. Sci. Pollut. Res. 2014, 21, 5076–5085. [Google Scholar] [CrossRef] [PubMed]
- Luo, C.; Liu, C.; Wang, Y.; Liu, X.; Li, F.; Zhang, G.; Li, X. Heavy metal contamination in soils and vegetables near an e-waste processing site, south China. J. Hazard. Mater. 2011, 186, 481–490. [Google Scholar] [CrossRef] [PubMed]
- Mehmood, A.; Mirza, M.A.; Choudhary, M.A.; Kim, K.-H.; Raza, W.; Raza, N.; Lee, S.S.; Zhang, M.; Lee, J.-H.; Sarfraz, M. Spatial distribution of heavy metals in crops in a wastewater irrigated zone and health risk assessment. Environ. Res. 2019, 168, 382–388. [Google Scholar] [CrossRef]
- Lu, Y.; Yao, H.; Shan, D.; Jiang, Y.; Zhang, S.; Yang, J. Heavy Metal Residues in Soil and Accumulation in Maize at Long-Term Wastewater Irrigation Area in Tongliao, China. J. Chem. 2015, 2015, 1–9. [Google Scholar] [CrossRef]
- Wahsha, M.; Bini, C.; Argese, E.; Minello, F.; Fontana, S.; Wahsheh, H. Heavy metals accumulation in willows growing on Spolic Technosols from the abandoned Imperina Valley mine in Italy. J. Geochem. Explor. 2012, 123, 19–24. [Google Scholar] [CrossRef]
| Soil Analysis | Unit | Measurement |
|---|---|---|
| Sand | 20–200 μm | 78.4 ± 6.1 |
| Silt | 2–20 μm | 12.7 ± 0.90 |
| Clay | ˂2 μm | 8.9 ± 0.71 |
| Texture | Loamy-sand | |
| EC | (dS m−1) | |
| pH | 8.2 ± 0.52 | |
| OM | (g kg−1) | 13.5 ± 0.97 |
| CaCO3 | (g kg−1) | 15.0 ± 1.04 |
| Parameter | TWW | JISM 1 | WHO 2 |
|---|---|---|---|
| pH | 7.78 | 6.0–9.0 | 6.5–8.0 |
| EC (dS m−1) | 1.81 | 1.0–3.0 | 0.7–3.0 |
| BOD (mg L−1) | 10 | 60 | 300 |
| COD (mg L−1) | 28.3 | 120 | 500 |
| TDS (mg L−1) | 826.3 | <2000 | 450–2000 |
| SAR (ratio) | 8.3 | 9 | <13 |
| Total Coliforms (MPN/100 mL) | <1.8 | <10 | <9 |
| Element | TWW | JISM 1 | WHO 2 |
|---|---|---|---|
| N (mg L−1) | 11.9 ± 1.49 | 50 | 5–50 |
| PO4 (mg L−1) | 15.2 ± 1.21 | 30 | 30 |
| NO3 (mg L−1) | 38.9 ± 3.74 | 45 | 50 |
| Ca (mg L−1) | 81.2 ± 4.44 | 400 | 230 |
| Mg (mg L−1) | 14.6 ± 1.32 | 60 | 60 |
| K (mg L−1) | 34.2 ± 3.29 | 80 | 80 |
| Na (mg L−1) | 94.2 ± 5.31 | 230 | 69–207 |
| Cl (mg L−1) | 115.4 ± 8.36 | 400 | 140–350 |
| Cr (mg L−1) | 0.01 ± 0.002 | - | 0.02 |
| Cu (mg L−1) | ND | - | 0.2 |
| Mn (mg L−1) | ND | - | 0.2 |
| Ni (mg L−1) | 0.02 ± 0.002 | - | 0.2 |
| Zn (mg L−1) | 0.76 ± 0.14 | 2 | <2.0 |
| Fe (mg L−1) | 3.79 ± 0.18 | 5 | 0.1–1.5 |
| Cd (mg L−1) | 0.002 ± 0.001 | 0.01 | <0.01 |
| Pb (mg L−1) | 3.03 ± 0.2 | 5 | <5.0 |
| Element | Measurement |
|---|---|
| N total (g kg−1) | 13.3 ± 0.3 |
| P (mg kg−1) | 93.7 ± 7.03 |
| K (mg L−1) | 710 ± 44.0 |
| Na (mg L−1) | 345 ± 39.2 |
| Cl (mg L−1) | 749 ± 45.5 |
| Cd (mg L−1) | 0.15 ± 0.05 |
| Cr (mg L−1) | 0.18 ± 0.05 |
| Cu (mg L−1) | 0.28 ± 0.01 |
| Fe (mg L−1) | 2.22 ± 1.20 |
| Mn (mg L−1) | 3.45 ± 1.54 |
| Ni (mg L−1) | 0.16 ± 0.03 |
| Pb (mg L−1) | 1.56 ± 0.19 |
| Zn (mg L−1) | 0.78 ± 0.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Habahbeh, K.A.; Al-Nawaiseh, M.B.; Al-Sayaydeh, R.S.; Al-Hawadi, J.S.; Albdaiwi, R.N.; Al-Debei, H.S.; Ayad, J.Y. Long-Term Irrigation with Treated Municipal Wastewater from the Wadi-Musa Region: Soil Heavy Metal Accumulation, Uptake and Partitioning in Olive Trees. Horticulturae 2021, 7, 152. https://doi.org/10.3390/horticulturae7060152
Al-Habahbeh KA, Al-Nawaiseh MB, Al-Sayaydeh RS, Al-Hawadi JS, Albdaiwi RN, Al-Debei HS, Ayad JY. Long-Term Irrigation with Treated Municipal Wastewater from the Wadi-Musa Region: Soil Heavy Metal Accumulation, Uptake and Partitioning in Olive Trees. Horticulturae. 2021; 7(6):152. https://doi.org/10.3390/horticulturae7060152
Chicago/Turabian StyleAl-Habahbeh, Khaled A., Mohamed B. Al-Nawaiseh, Rabea S. Al-Sayaydeh, Jehad S. Al-Hawadi, Randa N. Albdaiwi, Hmoud S. Al-Debei, and Jamal Y. Ayad. 2021. "Long-Term Irrigation with Treated Municipal Wastewater from the Wadi-Musa Region: Soil Heavy Metal Accumulation, Uptake and Partitioning in Olive Trees" Horticulturae 7, no. 6: 152. https://doi.org/10.3390/horticulturae7060152
APA StyleAl-Habahbeh, K. A., Al-Nawaiseh, M. B., Al-Sayaydeh, R. S., Al-Hawadi, J. S., Albdaiwi, R. N., Al-Debei, H. S., & Ayad, J. Y. (2021). Long-Term Irrigation with Treated Municipal Wastewater from the Wadi-Musa Region: Soil Heavy Metal Accumulation, Uptake and Partitioning in Olive Trees. Horticulturae, 7(6), 152. https://doi.org/10.3390/horticulturae7060152






