Abstract
The physiological and biochemical role of the γ-aminobutyric acid (GABA) shunt pathway in green pea seedlings (Pisum sativum L.) was studied in response to soil water holding capacity levels: 80%, 60%, 40%, 20%, and 10% grown under continuous light at 25 °C for 7 days and 14 days, separately. Characterization of seeds germination pattern, seedlings growth (plant height, fresh and dry weight, and chlorophyll contents), GABA shunt metabolite (GABA, glutamate, and alanine) levels, total protein and carbohydrate levels, and oxidative damage (MDA level) were examined. Data showed a significant effect of drought stress on seed germination, plant growth, GABA shunt metabolites level, total protein and carbohydrate contents, and MDA level. A significant decline in seed germination percentage was recorded at a 20% drought level, which indicated that 20% of soil water holding capacity is the threshold value of water availability for normal germination after 14 days. Seedling fresh weight, dry weight, and plant height were significantly reduced with a positive correlation as water availability was decreased. There was a significant decrease with a positive correlation in Chl a and Chl b contents in response to 7 days and 14 days of drought. GABA shunt metabolites were significantly increased with a negative correlation as water availability decreased. Pea seedlings showed a significant increase in protein content as drought stress was increased. Total carbohydrate levels increased significantly when the amount of water availability decreased. MDA content increased slightly but significantly after 7 days and sharply after 14 days under all water stress levels. The maximum increase in MDA content was observed at 20% and 10% water levels. Overall, the significant increases in GABA, protein and carbohydrate contents were to cope with the physiological impact of drought stress on Pisum sativum L. seedlings by maintaining cellular osmotic adjustment, protecting plants from oxidative stress, balancing carbon and nitrogen (C:N) metabolism, and maintaining cell metabolic homeostasis and cell turgor. The results presented in this study indicated that severe (less than 40% water content of the holding capacity) and long-term drought stress should be avoided during the germination stage to ensure proper seedling growth and metabolism in Pisum sativum L.
1. Introduction
Pea (Pisum sativum L.) was used by Mendel to lay the foundation of modern genetics [1]. It is one of the major food legumes that can grow in different regions and rich in proteins, vitamins, minerals, carbohydrates, and seed oil [2]. Pea is predominantly a self-pollinated crop with limited variation in the number of flowers per node [3]. Most garden pea germplasm/varieties lines have either one or two flowers per node [4]. The agro-ecological importance of pea is connected with its capability to form symbiotic nitrogen fixation with rhizobial bacteria that promotes and enhances seed germination, seedling vigor and emergence, root and shoot growth, plant stand and biomass, and seed weight [5].
Drought is a major abiotic stress that affects plant growth and yield [6]. Plant species and genotype adaptation and level of tolerance vary according to the type of drought stress (meteorological, agricultural, hydrological, or socioeconomic drought) [7]. Drought limits the productivity of many crops, especially during the seedling stage [8]. Water scarcity negatively affects vegetative growth by direct influence on its metabolic sink strength and fruit composition [9]. It causes reduced quality and quantity of the crop yield, growth rate, leaf expansion, stem elongation [8,10], stomatal conductance [11], and grain filling) [12]. Increasing the level of CO2 in plants mitigates the impact of drought stress in many legume species [13].
Physiological, biochemical, and morphological processes in plants can occur as a result of drought stress causing changes in the expression of genes that lead to alteration in protein production [8]. In addition, respiration, photosynthesis, enzyme activities, and mineral nutrition, Redox (oxidation/reduction) homeostasis, and chloroplast metabolism are influenced by drought [12,14]. Analysis of proteomics data in plant leaves showed that many drought-responsive proteins that are involved in osmotic regulation, cell structure modulation, ROS scavenging, drought signal transduction, as well as carbohydrate metabolism, were upregulated under drought stress [15].
Drought stress reduces the quality and yield of many crops. Extended periods of water deficit results in a reduction in plant growth, photosynthesis efficiency and initiates a series of actions to maintain plant survival. These actions include regulation of stomatal conductance, osmotic adjustment, cell turgor maintenance, and protection of cellular membranes, enzymes, and macromolecules from oxidative damage [16]. During drought stress, plants close their stomata which leads to plasma membrane damage; consequently, the internal CO2 concentration will be decreased, and excessive generation of reactive oxygen species will be increased, leading to a reduction in the photosynthetic rate and plant growth [17,18]. Water deficiency in maize caused a reduction in seedling survival rate, acceleration of the post-pollination embryo abortion rate, and ultimately, yield loss by postponing silking through the increase in the anthesis to the silking interval that leads to a reduction in ear and kernel number per plant [19,20,21,22]. Furthermore, water scarcity decreased photosynthetic efficiency, chlorophyll content, and CO2 exchange in maize seedlings [21,23].
Drought stress decreased total barley (Hordeum vulgare L.) grain yield through a drop in tillers, spikes, and grains number per individual plant [24]. In soybean, drought stress decreased branch seed and total crop yields [25]. Cell elongation in higher plants can be inhibited through water movement interruption to the surrounding elongating cells from the xylem under severe water deficiency [26].
The development of candidate drought-tolerant associated genes is based on precise screening for germplasm and breeding materials in limited water environments using bioinformatics [27]. Various studies on some plant genes subjected to drought stress described the biochemical pathways that are involved in drought acclimation. Proteins and metabolites that were involved in protective mechanisms against drought conditions were identified. These mechanisms include detoxification enzymes, redox status regulation, signaling pathways, protein folding and degradation, photosynthesis stability, and primary metabolism [18]. In addition, drought and low water content reduction lead to an increase in sugar and amino acid concentrations in plants [28].
GABA is a four-carbon non-protein amino acid that increases in plant tissues under stress [29]. The GABA shunt has a functional role in biotic and abiotic stress in plants through the improvement in the antioxidant activity to restrict ROS species production, plant cell signaling, and metabolism under stress [29,30]. The GABA shunt pathway is composed of three enzymatic reactions. GABA is largely produced through glutamate decarboxylation that is catalyzed by glutamate decarboxylase enzyme, then GABA is catabolized inside the mitochondrial matrix to succinic semialdehyde (SSA) by GABA transaminase enzyme. Then, succinate is produced from SSA oxidization by the mitochondrial succinic semialdehyde dehydrogenase enzyme or reduced to gamma-hydroxybutyrate (GHB) [30].
GABA is made from glutamate by glutamate decarboxylase that is present in symbiotic rhizobia bacteroids [31]. Furthermore, GABA was shown to be the second most plentiful amino acid in detached pea (Pisum sativum L.) nodules through the nuclear magnetic resonance analysis, which, in turn, reflects the vital role of GABA in amino acid cycling during bacteroid metabolism [31]. Data showed that large amounts of GABA is accumulated in root nodules, while GABA that is newly made appeared to be limited to pea nodules that are metabolically active [32]. Bound forms of GABA accumulated in nodules of many legume species, reaching 20% of the total nitrogen content [32].
Drought stress could increase the activation of GABA transporters (ProTs and AAP3) that regulate the entry of GABA across the cell membrane. The entrance of GABA decreased drought leaf wilting and improved membrane stability via the reduction in oxidative damage in plants [33]. Endogenous GABA enhanced drought tolerance through the inhibition of lipid peroxidation and photosynthesis [33]. In addition, GABA stabilized the intracellular pH during drought stress and provided a source of nitrogen and carbon during the Krebs cycle and carbon–nitrogen metabolism in the maturation of green peas [34]. Drought stress directly influences metabolic sink strength during vegetative growth, which triggers an imbalance in redox homeostasis that affects overall plant growth and development.
In this study, the effects of drought stress on the physiological and biochemical characterization of the GABA shunt pathway in green pea (Pisum sativum L.) were investigated. Characterization of seeds germination pattern, seedling growth (plant height, fresh weight, dry weight, and chlorophyll content), oxidative damage (Malondialdehyde level), GABA shunt metabolite levels (GABA, Glutamate and Alanine), total proteins, and total carbohydrate levels were determined.
2. Materials and Methods
2.1. Plant Materials and Growth Conditions
The green pea seeds (Pisum sativum L.) that were used in this study were harvested in 2018 and obtained from local pea growers in Irbid/Jordan. All experiments were performed in the laboratory using a plant growth medium. The growth medium was a mixture of peat moss and perlite growth soil with a ratio of (2:1) wt/wt, later referred to as “soil” in a pot (7 g). Seeds were surface disinfected with 6% (v/v) sodium hypochlorite for 10 min and then washed five times with sterile distilled water. Surface sterilized seeds were planted in soil. Tap water was used to irrigate the seeds at a full water holding capacity level (80–100%) for two weeks until the appearance of seedlings) [35]. The two-week-old seedlings were then subjected to drought treatments according to soil water holding capacity levels: 80%, 60%, 40%, 20%, and 10% separately for 7 days and another set for 14 days. Plants were grown at 25 °C under continuous cool white fluorescent lamp illumination (40 µmol m−2 s −1). For assays and parameters determinations, three replicates of 6 pea (Pisum sativum L.) seeds for each replicate were used.
2.2. Seed Sensitivity to Drought Stress Assay
Three replicates of 15 pea (Pisum sativum L.) surface-sterilized seeds were placed on three filter papers as artificial growth surface in sealed Petri dishes and irrigated with tap water in drought treatments according to full water holding capacity levels: 80%, 60%, 40%, 20%, and 10% separately for 14 days and allowed to grow at 25 °C. Radicle emergence from the seeds was recorded daily for 14 days. Drought treatment effect on seed germination was calculated.
2.3. Seedling Physiological Growth Parameters Assay
After each drought treatment for the 7 and 14 days separately, plant height (cm), fresh weight (g), and dry weight (g) were determined. Shoot fresh weight (g) was determined by directly weighing the collected seedlings’ shoot tissues. Shoot dry weight (g) was determined following weighing the seedlings’ shoot tissues after oven drying at 70 °C for 48 h.
2.4. Chlorophyll Content Determination
After each drought treatment for the 7 and 14 days separately, fresh shoots tissues were harvested. Chlorophyll pigments (Chl a and Chl b) extraction and determination were performed according to Jeffrey et al. protocol [36] with the following adjustments: 300 mg fresh leaves mixed with one ml of ice-cold 90% acetone were ground in an Eppendorf tube using a micro centrifuge tube pestle to prepare the chlorophyll extract. The extracted liquid was placed in a new Eppendorf tube and centrifuged at 11,000× g for 5 min. The supernatant was used for the determination of both chlorophyll pigments. The resulted supernatant absorbance was measured spectrophotometrically at 647 nm, 664 nm, and 750 nm wavelengths. Equation and extension coefficients [36] were used to calculate the concentration of Chl a and Chl b (µg/mL). The average of three replicates for each treatment was calculated.
2.5. Metabolites Extraction
After each drought treatment for the 7 and 14 days separately, fresh shoot tissues were harvested. GABA shunt metabolites were extracted and determined according to Zhang and Bown [37] with the following adjustments: 500 mg of fresh shoot tissues were ground in an Eppendorf tube using a micro centrifuge tube pestle until a fine powder was obtained, and then placed in 1.5 mL Eppendorf tubes and vortexed with 400 µL methanol for 10 min. Methanol from samples was removed by overnight evaporation at room temperature. To each tube, 500 µL of 70 mM lanthanum chloride was added and vortexed for 15 min. Then tubes were centrifuged at 12,400× g for 5 min. One hundred and sixty microliters of 1 M KOH was added to the collected supernatants and vortexed for 10 min. Tubes were centrifuged at 12,400× g for 5 min. The resulted supernatant (metabolites extract) was used for the determination of GABA shunt metabolites (GABA, alanine, and glutamate). The average of three replicates was used for each drought treatment.
2.6. GABA (γ-Aminobutyric Acid) Level Determination
The protocol of Zhang and Bown [37] was used for GABA determination with the following adjustments: 50 µL of metabolites extract, 14 µL of 4 mM NADP+, 19 µL of 0.5 M potassium pyrophosphate, pH (8.6), 10 µL of (2 u/µL) GABASE enzyme (GABASE enzyme (Sigma-Aldrich Corp., St. Louis, MO, USA) was suspended in 0.1 M potassium pyrophosphate, pH (7.2), containing 12.5% Glycerol and 5 mM β-mercaptoethanol), and 10 µL of α-ketoglutarate were mixed in an Eppendorf tube to prepare the reaction mixture. The change in absorbance at 340 nm after the addition of α-ketoglutarate was measured after 90 min incubation at 25 °C using a microplate reader. The GABA level (nmol/mg FW) was determined using the NADPH standard curve. For each drought treatment, the average of three replicates was used.
2.7. Alanine Level Determination
The Bergmeyer protocol [38] was used for alanine determination with the following adjustments: 180 µL of 0.05 M Na-carbonate buffer, pH (10), 7 µL of 30 mM β-NAD+, 10 µL of sample extract, and 1 µL of 0.3 u/µL alanine dehydrogenase (Sigma-Aldrich, St. Louis, MO, USA) enzyme suspension were mixed in an Eppendorf tube to prepare the reaction mixture. The change in absorbance at 340 nm after the addition of alanine dehydrogenase was measured after 60 min incubation at 25 °C using a microplate reader. The alanine level (nmol/mg FW) was determined using the NADH standard curve. For each drought treatment, the average of three replicates was used.
2.8. Glutamate Level Determination
The Bergmeyer protocol [38] was used for glutamate determination with the following adjustments: 180 µL of 0.1 M Tris-HCl pH (8.3), 8 µL of 7.5 mM β-NAD+, 10 µL of sample extract, and 1 µL of 0.8 u/µL glutamate dehydrogenase enzyme suspension (Sigma-Aldrich, St. Louis, MO, USA) were mixed in an Eppendorf tube to prepare the deamination reaction mixture. The change in absorbance at 340 nm after the addition of glutamate dehydrogenase was measured after 60 min incubation at 25 °C using a microplate reader. The glutamate level (nmol/mg FW) was determined using the NADH standard curve. For each drought treatment, the average of three replicates was used.
2.9. Total Protein Content Determination
After each drought treatment for the 7 and 14 days separately, fresh shoot tissues were harvested. Total protein content was determined using a SMART BCA Protein Assay Kit (Intron Biotechnology, Gangnam-gu, Seoul, Korea) according to the manufacturer’s instructions. Five hundred milligrams of fresh shoots tissues mixed with 100 μL of distilled water were ground in an Eppendorf tube using a micro centrifuge tube pestle. Fifty microliters of the extracted liquid was obtained and mixed well with 1 mL of the kit working solution. All tubes were incubated at 37 °C for 30 min and then kept at room temperature for 5 min. The absorbance of the samples was measured at 562 nm using a microplate reader. The BSA standard curve was used to determine the total protein concentration (µg/mL). For each drought treatment, the average of three replicates was used.
2.10. Total Carbohydrates Content Determination
After each drought treatment for the 7 and 14 days separately, fresh shoot tissues were harvested. Total carbohydrates content was determined using the Total Carbohydrate Quantification Assay Kit (catalog number: ab155891, Abcam, Cambridge, MA, USA) according to the manufacturer’s instructions. Fifty milligrams of fresh shoot tissues mixed with 200 μL ice-cold kit assay buffer was ground in an Eppendorf tube using a micro centrifuge tube pestle. Tubes were then centrifuged at 12,400× g for 5 min. The resulted supernatant was collected. In a 96-well microplate, 30 μL of collected supernatant from each sample and the kit standard solution wells were mixed with 150 μL of concentrated H2SO4. The microplate was mixed well for 5 min on a shaker and then incubated at 90 °C for 15 min. The microplate was cooled at room temperature for 15 min. Thirty microliters of the kit developer solution was added to all treated samples and kit standard solution wells, mixed on a shaker at room temperature for 15 min, and measured OD at 490 nm. A glucose standard curve was used to determine the total carbohydrate concentration (µg/mg FW). For each drought treatment, the average of three replicates was used.
2.11. Oxidative Damage Assay
After each drought treatment for the 7 and 14 days separately, fresh shoot tissues were harvested. Lipid peroxidation as malondialdehyde (MDA) equivalent was determined by estimating the TBA reactive substances (TBARS) as described by Heath and Packer [39] with some adjustments: 20 mg of fresh shoots tissues was mixed with 0.25 mL of 0.5% (w/v) thiobarbituric acid in 20% (w/v) trichloroacetic acid and 0.25 mL of 175 mM NaCl in 50 mM Tris-HCl (pH 8) and ground in an Eppendorf tube using a micro centrifuge tube pestle. Tubes were placed in a water bath at 90 °C for 25 min. Tubes were then centrifuged at 12,400× g for 20 min and the supernatant was collected. A microplate reader was used to measure the absorbance of the supernatant at 532 nm. A standard curve of MDA was used to calculate the MDA level (nmol/mg FW). For each drought treatment, the average of three replicates was used.
2.12. Experimental Design and Data Analysis
A completely randomized design (CRD) was used for all experiments in this study. All treatments and assays were replicated three times. For all assays parameters, mean and standard deviation (SD) values were determined. Results were expressed as mean. Data analysis was performed by one-way analysis of variance (ANOVA) using the Least Significant Difference (LSD) multiple comparison tests on the means using the SPSS version 16.0 software. For all data analyses, p-value ≤ 0.05–0.01 was considered significant, and p-value < 0.01 was considered highly significant.
3. Results and Discussion
3.1. Seed Sensitivity to Drought Stress
Germination percentage of Pisum sativum L. was affected significantly (p < 0.05) by drought treatments to all saturation levels (80%, 60%, 40%, 20%, and 10%) in pea seeds for 14 days. In general, the germination percentage significantly decreased with a positive correlation as water content decreased (Table 1). The highest seed germination percentage was observed at the 80% saturation level (p < 0.05). A significant decline was recorded at 40% water saturation level. After 14 days, the germination percentage was 100, 80, and 30 at 60, 40, and 20% soil water saturation levels, respectively. No germination was observed at 10% drought treatment point (Table 1). These results indicated that the 40% water saturation level is the threshold value for normal pea seed germination after 14 days (p = 0.0001, r = 0.929).

Table 1.
Germination percentage of pea (Pisum sativum L.) in response to soil water holding capacity levels: 80%, 60%, 40%, 20%, and 10% grown under continuous light at 25 °C. Means followed by different letters are significantly different (p ≤ 0.05) by LSD; r = correlation coefficient.
Seed germination is a critical stage for plant survival. Drought stress greatly affects seed germination, but the response intensity and harmful effects of stress depend on the plant species [40]. A decrease in the germination of Eremosparton songoricum was observed under drought stress [40]. The results of the current study indicated that as water saturation decreased, a significant drop in the germination percentage in pea (Pisum sativum L.) was observed. Similar to our finding, Liu et al.’s [41] study revealed that seed germination of two maize cultivars (Liansheng15 cultivar and Zhengdan 958 cultivar) was reduced under drought stress. Increased water deficit minimized absorption of water by seeds, therefore preventing their germination [42]. However, it has been shown that failure of the emergence of the root was due to a reduction in water level between the soil and the seeds [43]. In addition, a reduced water gradient affected enzymatic reactions, which caused a delay in seed germination [44].
Muscolo et al. [45] found significant differences between two lentil cultivars, Ustica and Pantelleria, collected from Sicilian islands subjected to drought stress with a remarkable decrease and delay in seed germination. Inhibition of seed germination was directly related to energy production through respiration, enzyme and hormonal activity, reserve mobilization, and dilution of the protoplasm to increase metabolism for successful embryonic growth [45]. Water availability is a limiting factor for non-dormant seed germination. It also affected the speed, uniformity, and percentage of seed emergence [46]. Dornbos et al. [47] showed that the germination percentage was decreased during the seed filling stage in soybean (Glycine max L.c.v Merr.) under severe drought stress. Inhibition of starch catabolism under drought and salt stress decreased seed germination [48,49]. AL-Quraan et al. [50] showed a significant decrease in seed germination of five wheat cultivars (Triticum aestivum L.) under salt and osmotic stress. Furthermore, AL-Quraan et al. [51] reported that cold, heat, salt, and osmotic stresses significantly reduced seed germination in two lentil (Lens culinaris Medik) cultivars. The results of this study showed that reduction in water content had a severe impact on seed germination and early seedling growth in pea (Pisum sativum L) that might be caused by a reduction in water absorption by seeds during metabolic processes and enzymatic activity in germinating pea seeds.
3.2. Seedling Physiological Growth in Response to Drought Stress
Seedling fresh weight, dry weight, and seedling height of Pisum sativum L. were measured in response to drought treatments according to soil water holding capacity levels of 80%, 60%, 40%, 20%, and 10% for 7 and 14 days, separately. Seedling fresh weight and seedlings height were significantly reduced with a positive correlation as water availability decreased (Table 2). The seedling fresh weight (p = 0.001, 0.05, r = 0.957, 0.979) and seedling height (p = 0.001, 0.001, r = 0.867, 0.992) were reduced significantly after 7 and 14 days, respectively. Dry weight reduction was not significant under all drought treatment after 7 (p = 0.096) and 14 days (p = 0.228), respectively.

Table 2.
Seedling height (cm), fresh weight, and dry weight (gm), and water content (%) of pea (Pisum sativum L.) in response to soil water holding capacity levels: 80%, 60%, 40%, 20%, and 10% grown under continuous light at 25 °C for 7 days and 14 days, separately. Means followed by different letters are significantly different (p ≤ 0.05) by LSD; r = correlation coefficient.
In agreement with our study, Embiale et al. [52] reported that unavailability of water resulted in a significant decline in pea’s basal diameter increment, area, width, and expansion length of leaf, number of branches and leaves, and total seedlings length. Plant cells save water by avoiding active growth. Stomatal closure, diminished leaf water potential, turgor loss, reduction in cell enlargement and growth [53], and inhibition of shoot and root growth [54] are common plant responses due to water stress. In general, reduction in plant biomass is positively linked with prolonged water deficiency and inhibition of cell expansion per cell growth due to low turgor pressure [55]. Reduction in plant dry weight under water stress could be due to imbalanced stomatal conductance that leads to a reduction in carbon assimilation per unit leaf area and low biomass production [56,57]. Drought stress was associated with reduced cellular division and elongation during germination, causing a reduction in root length [58].
Khorasaninejad et al. [59] reported a reduction in shoot fresh and dry weight, root dry weight, internodes, and internodes length of peppermint (Mentha piperita L.) under drought stress. A decrease in root and shoot fresh and dry weights and shoot length was observed in sensitive bean genotypes under water stress [60]. Zhang et al. [61] reported that the length and width of maize leaves on seedlings were shorter under water stress due to a reduction in chlorophyll content. A reduction in water content decreased plant height, stem diameter, stem and root biomass, and total biomass of Populus nigra (poli) [62]. In agreement with our study, Riad et al. [63] reported that decreasing water content resulted in a reduction in plant growth parameters (root length, root, and leaves fresh and dry weights, and plant length), biomass accumulation, and vegetative growth of green peas as compared with 100% water availability.
3.3. The Effect of Drought on Chlorophyll Content
Table 3 shows that the chlorophyll content were significantly decreased after 7 and 14 days as water content decreased. Pisum sativum L. seedlings possessed the highest total Chl a and Chl b contents at 80% water saturation level (p = 0.020, 0.003) with positive correlation (r = 0.938, 0.986) after 7 and 14 days, respectively. There was a significant decrease with a positive correlation in Chl a and Chl b contents in response to 7 days and 14 days of drought treatments.

Table 3.
Chlorophyll content (Chl a, Chl b, total chlorophyll) (µg/mL) of pea (Pisum sativum L.) seedlings in response to soil water holding capacity levels: 80%, 60%, 40%, 20%, and 10% grown under continuous light at 25 °C for 7 days and 14 days, separately. Means followed by different letters are significantly different (p ≤ 0.05) by LSD; r = correlation coefficient.
Chlorophyll is a major chloroplast component and has a positive relationship with photosynthetic rate. Our results agreed with Nyachiro et al.’s [64] study, which found a significant reduction in Chl a and Chl b caused by drought stress in six wheat (Triticum aestivum L.) cultivars. Shinde and Thakur [65] reported that drought stress significantly reduced Chl a and Chl b contents in three varieties of chickpeas during vegetative growth or anthesis. Our data indicated that decreased water availability caused a significant decrease in Chl a and Chl b contents in Pisum sativum L. after 7 and 14 days. This reduction in total chlorophyll content leads to a reduction in photosynthesis and plant growth which was correlated with a significant reduction in seedling growth under all water stress levels. Depending on the duration and severity of drought stress, plant genotypes, and environmental conditions, a reduction in Chl a and b contents has been reported in other plant species [66,67]. Previous studies on Triticum aestivum and Zea mays [68], Gossypium hirsutum [69], and Tectona grandis [70] found that the content of photosynthetic pigments reduced as water availability decreased. It has been reported that metabolic imbalance occurred as a result of a decrease in chlorophyll synthesis and an increase in chlorophyll degradation under drought stress [71,72,73].
Nitrogen is a component of the chlorophyll structure in plant tissues. Under stress conditions, a decline in chlorophyll content was associated with changes in nitrogen metabolism as a result of proline synthesis to maintain osmotic adjustment [74]. Low nitrogen levels reduced photosynthetic rates due to a reduction in chlorophyll synthesis [75]. Severe water stress decreased stomatal conductance, transpiration, and photosynthetic rate [56,76]. As a drought response mechanism, chloroplasts reduced light absorption by decreasing the chlorophyll content [77]. Studies revealed that the inhibition of photosynthesis efficiency as a result of an imbalance between light capture and usage under water shortage enhances oxidative stress. Furthermore, under water stress, stomatal closure and variation in photosynthetic metabolism lead to a reduction in CO2 availability and directly impacted the photosynthetic rate [78]. Under severe drought conditions, chlorophyll content in wheat seedlings was reduced as a result of the increased activity of chlorophillase and peroxidase enzymes [79]. In response to drought stress, chlorophyll content was reduced in the vegetative and flowering stages of the Avena species [80]. Alberte et al. [81] reported a reduction in Chl a/b contents occurred in lamellar content in response to water deficit. Patro et al. [82] and Ghotbi-Ravandi et al. [83] reported that stomatal closure due to water deficit is a major factor inhibiting photosynthesis in Arabidopsis thaliana and barley (Hordeum vulgare L.) seedlings, respectively. Moreover, drought stress significantly decreased the chlorophyll content in Pisum sativum L. seedlings [84] and three wheat varieties (Triticum aestivum L.) [85]. The results of our study indicated that a water imbalance due to drought stress caused a reduction in chlorophyll content that might be attributed to the photosynthetic apparatus activity disturbance.
3.4. GABA Shunt Activation in Response to Drought Stress
The GABA shunt metabolites increased significantly as a result of drought stress caused by different water saturation levels (Table 4). There were significant differences in GABA, alanine, and glutamate content at 80%, 60%, 40%, 20%, and 10% soil water content between the 7- and 14-day treatments. In general, there was a significant increase in GABA, alanine, and glutamate (p = 0.001, 0.0001, 0.002, respectively) with a negative correlation (r = −0.987, −0.968, −0.978, respectively) with water availability after the 7-day treatment. A significant increase was also found in GABA, alanine, and glutamate (p = 0.0001, 0.003, 0.870, respectively) with a negative correlation (r = −0.906, −0.989, −0.939, respectively) after the 14-day treatment. The increase in GABA content was about 4 and 5 times higher at the 20% and 10% water levels, respectively, after the 14-day treatment when compared with GABA content at the same water levels after the 7 day treatment. The increase in GABA levels was accompanied by a slight but significant (p ≤ 0.01–0.05) increase in the levels of alanine and glutamate after 7- and 14-day treatments under all water saturation levels.

Table 4.
Levels of GABA shunt metabolites (GABA, alanine, and glutamate) of pea (Pisum sativum L.) seedlings in response to soil water holding capacity levels: 80%, 60%, 40%, 20%, and 10% grown under continuous light at 25 °C for 7 days and 14 days, separately. Metabolite levels were calculated as nmol/mgFW. GABA (γ-Aminobutyric acid), Ala (alanine), Glu (glutamate). Means followed by different letters are significantly different (p ≤ 0.05) by LSD; r = correlation coefficient.
The GABA shunt has been associated with physiological responses, such as cytosolic pH regulation [86], nitrogen metabolism, and carbon fluxes into the Krebs cycle [87], protection against ROS production [88], and osmoregulation and signaling [89]. The GABA shunt is also involved in carbon and nitrogen metabolism, maintenance of cell membrane integrity [90], and minimizing the negative effects of abiotic stresses on plant metabolism [91].
Many studies reported that various stresses caused an increase in endogenous GABA accumulation in plants [92]. The current study showed a significant increase in GABA content as a result of a reduction in water availability to ease the metabolic damage in pea seedlings during vegetative growth. Studies showed that GABA effectively decreased leaf wilting and improved membrane stability induced by drought stress in perennial ryegrass (Lolium perenne) [93], black pepper [94], and white clover (Trifolium repens) [33], which confirmed the beneficial effect of GABA in protecting plants from oxidative stress. Insufficient availability of oxygen resulted in significant suppression of melon seedling growth. However, seedling growth was significantly improved when exogenous GABA was applied [95]. Several amino acid syntheses are regulated by the TCA cycle [96]. Glutamate, as a precursor amino acid of GABA production, is produced from the α-ketoglutaric acid (an intermediate of the TCA cycle) transamination [90]. Glutamate metabolic pathway activation via the TCA cycle was confirmed by GABA production and defense of creeping bentgrass against drought stress [97]. An increase in endogenous GABA levels due to an increase in glutamate content occurred in white clover in response to drought stress [33]. Increased content of glutamic acid and GABA were found in six winter wheat genotypes due to drought stress [98]. In addition, alanine was increased at the beginning of drought stress due to an increase in glutamate levels [99]. In this study, the increase in GABA content as water availability decreased mitigated the damaging effects of drought stress on pea (Pisum sativum L.) seedlings by supplying enough carbon/nitrogen source to the TCA cycle and amino acid synthesis.
3.5. The Effect of Drought on Seedling Proteins Level
In general, the total protein level in Pisum sativum L. seedlings increased with increasing water deficit. A significant difference was found in the total protein level of pea seedlings between the 7-day (p = 0.0001, r = −0.984) and 14-day (p = 0.0001, r = −0.982) treatments (Figure 1). The total protein level was 2 to 3 times higher after 7-day treatments when compared with the 14-day treatments under all water saturation levels. The significant increase in protein content after 7 days indicated that Pisum sativum L. seedlings could accumulate protein to lower the osmotic potential to maintain cellular structures and metabolic stability under drought stress. However, the steady-state level of protein content after 14 days indicated the ability of pea seedlings to tolerate prolonged water deficit by keeping stable protein metabolism to adjust cellular osmolarity and provide protection from further dehydration damage.
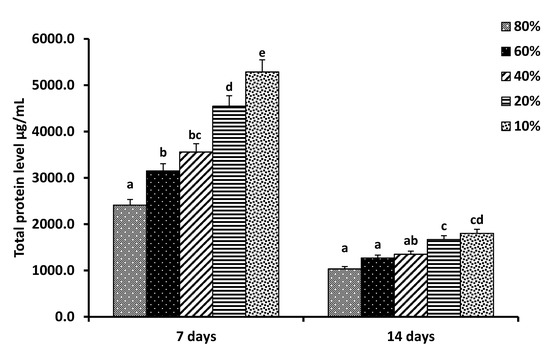
Figure 1.
Total proteins level of pea (Pisum sativum L.) seedlings in response to soil water holding capacity levels: 80%, 60%, 40%, 20%, and 10% grown under continuous light at 25 °C for 7 days and 14 days, separately. Protein levels were calculated as μg/mL. Means (columns) with different letter scripts are significantly different (p ≤ 0.05) by LSD.
Under environmental stress factors, plants stimulated specific changes in protein synthesis [100]. In agreement with our study, Guttieri et al. [101] reported increased protein concentration under water deficit due to higher rates of nitrogen accumulation in hard wheat (Triticum aestivum L.) grains. Ozturk and Aydin [102] also reported an increase in protein and gluten content in winter wheat cultivars grain in response to drought stress when compared with the fully irrigated treatment. During a combination of heat and drought stress, glutamine, ornithine, tyrosine, valine, and tryptophan were accumulated in Arabidopsis thaliana and purslane plants to maintain cellular osmotic adjustment and keep leaf turgor in response to such stress combination [103,104]. Drought stress induced the accumulation of protein by enhancing the protein biosynthesis in wheat and barley [105,106]. However, a reserve of available substrates for protein synthesis could be associated with amino acid accumulation to facilitate quick retrieval of osmotic adjustment and plant metabolism in response to water deficiency [107]. In a study that used three Australian bread wheat (Triticum aestivum L.) cultivars, Ford et al. [108] suggested that the cultivar RAC875 had the highest capacity to withstand drought stress by increasing the cellular protein synthesis. Although there was an increase in total protein levels which were involved in ROS scavenging, they observed a decrease in proteins involved in the Calvin cycle and photosynthesis. In contrast to our results, Gołębiowska et al. [109] observed a decrease in the number of differential proteins in leaves of winter barley subjected to drought stress for three weeks. Mohammadkhani and Heidar [110] reported an initial increase followed by a decrease in protein concentration in maize varieties (Zea mays L.) subjected to drought stress. The initial increase in total soluble proteins might be due to the expression of new proteins involved in stress adaptation. However, the decrease in protein concentration might be due to a severe reduction in photosynthesis. Under water stress, proline amino acid accumulated in plant cells prevents cellular oxidation through scavenge ROS [111] and regulates plant osmotic pressure for efficient water absorption [112]. Free proline level was reported to be increased under water deficit in wheat [113,114]. The amount of proline was increased significantly in ginger (Zingiber officinale) in response to a reduction in water availability and prolonged duration of drought stress [112].
3.6. The Effect of Drought on Seedling Carbohydrates Content
In the current study, data showed that the total carbohydrate level in Pisum sativum L. seedlings increased as water availability decreased (Figure 2)., the total carbohydrate level increased significantly (p = 0.0001) with a negative correlation (r = −0.970) in response to drought stress for 7 days. Similar results were obtained under drought stress for 14 days (p = 0.0001, r = −0.0980).
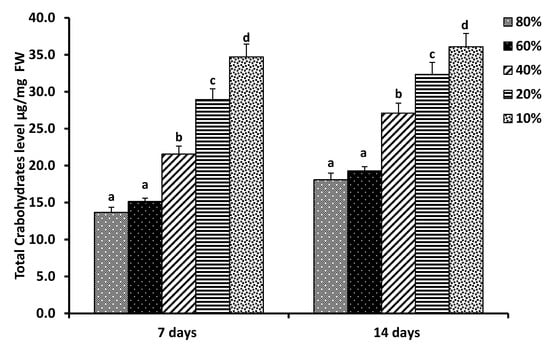
Figure 2.
Total carbohydrates level of pea (Pisum sativum L.) seedlings in response to soil water holding capacity levels: 80%, 60%, 40%, 20%, and 10% grown under continuous light at 25 °C for 7 days and 14 days, separately. Total carbohydrate level was calculated as μg/mg FW. Means (columns) with different letter scripts are significantly different (p ≤ 0.05) by LSD.
Water stress induces carbohydrate accumulation and osmolytes synthesis to maintain the water potential of plants [115,116]. Soluble sugar levels in plants also increased in response to drought stress. Plants respond to water shortage by balancing their potential osmotic proportion with the external environment by increasing their soluble sugars at the cellular level, reducing activities in roots, reducing the metabolism of carbohydrates as a result of severe pressure, and reducing the transfer of sugars in rinsing vessels [117]. In agreement with our study, an increase in total soluble sugars in durum (Triticum durum L.) wheat [118] and oligosaccharides in two sesame cultivars [119] were reported. In contrast, Akinci and Losel [120] reported a decrease in total sugar in cucumber cultivars under water stress. However, a major reserve of carbohydrates was detected in the leaves of cucumber seedlings. Under drought stress, sugar accumulation might be due to the fact that sucrose content increased because the enzyme activities involved in sucrose breakdown were diminished [121]. Furthermore, sugars protect the cells during drought by two physiological mechanisms. The first mechanism involves the hydroxyl group of sugars, which substitute the water to maintain hydrophilic interactions and hydrogen bonding in membranes and proteins during dehydration to prevent protein denaturation [122]. The second mechanism involves sugar vitrification in dehydrated cells through the formation of a biological glass in the cytoplasm to reduce water permeability [123]. Mohammadkhani and Heidari [110] reported an increase in soluble sugar concentrations as a result of starch degradation in two maize (Zea mays L.) cultivars. Soluble sugars accumulated only in roots of eucalyptus trees under drought stress [124]. Regier et al. [62] reported an increase in soluble sugar concentrations in the roots of two divergent clones of Populus nigra to maintain osmotic adjustment under water deficit. Soluble sugars were also reported to be increased and participated in plant metabolic regulation and stress signaling in response to osmotic stress [125]. Additionally, increased soluble sugars may act as osmoprotectants to stabilize the membranes and sustain cell turgor in response to water deficit [126]. Li and Li [127] showed an increase in the glucose, fructose, and sucrose content in micro-propagated apple plants (Malus domestica Borkh) in response to water stress. It is concluded from these previous studies that fluctuations of sugar concentrations in different plants under drought stress might be due to variations in CO2 assimilation, partitioning of carbon source-sink, activity of enzymes involved in sucrose–starch partitioning, and inhibition of Calvin cycle enzymes.
3.7. Oxidative Damage in Response to Drought Stress
The accumulation of MDA, which is a byproduct of membrane lipids’ oxidative damage, can be used as a marker for oxidative stress [128]. In this study, the MDA levels in the pea seedlings were determined to evaluate the lipid peroxidation in response to drought stress. Figure 3 shows that the malondialdehyde (MDA) level of pea (Pisum sativum L.) seedlings was slightly but significantly increased after 7 days (p = 0.0001, r = −0.967). However, the MDA level was increased sharply and significantly under all water stress levels reaching the highest level (140 nmol/mgFW at 10% water saturation level) (p = 0.0001, r = −0.975) after 14 days. Our results showed that the pea seedlings suffered minor damage under mild (60% water saturation level) drought stress, but severe lipid peroxidation and oxidative damage of the cell membrane occurred with increased (40%, 20%, and 10% water saturation levels) water deficit (Figure 3).
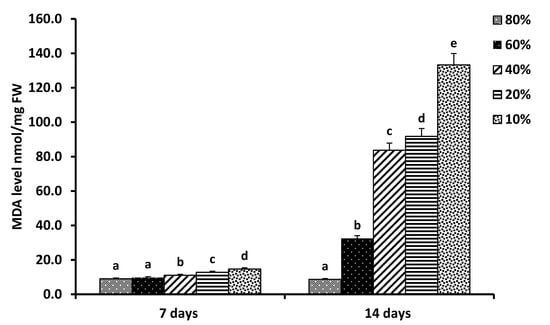
Figure 3.
Malondialdehyde (MDA) level of pea (Pisum sativum L.) seedlings in response to soil water holding capacity levels: 80%, 60%, 40%, 20%, and 10% grown under continuous light at 25 °C for 7 days and 14 days, separately. The MDA level was calculated as nmol/mg FW. Means (columns) with different letter scripts are significantly different (p ≤ 0.05) by LSD.
Many abiotic stresses lead to the accumulation of MDA in plant tissues due to lipid peroxidation of cellular membranes and overproduction of ROS [39]. Severe water stress caused agitation and instability in the metabolic processes in the mitochondria and chloroplasts, leading to high ROS production [129]. ROS resulting from oxidative stress are toxic and highly reactive molecules that can damage cellular macromolecules and subcellular structures [130,131]. The levels of MDA and H2O2 are indicators of the free radical reactions occurring in plant-stressed tissues [132]. Similar to our findings, Pandey et al. [133] reported that the MDA level was increased in Avena species leaves under drought stress, increasing membrane leakage. In addition, Zlatev et al. [134] reported an increase in membrane damage and ROS production in three bean (Phaseolus vulgaris L.) cultivars as a result of water deficit. Similarly, Tatar and Gevrek [135] reported an increase in the level of MDA in wheat as the duration of drought stress increased. Leaves of growing carrots (Daucus carota L.) accumulated high levels of MDA content under water deficit stress [91]. Morabito and Guerrier [136] reported that oxidative stress occurred 12 h after the application of drought stress. However, after a longer period of drought stress, an antioxidants defense system was initiated. Abid et al. [137] showed that a higher MDA concentration in wheat (Triticum aestivum L.) was associated with higher H2O2 content and greater rate of O2•− generation. Hernandez et al. [138] reported that salinity stress in plant tissues could lead to cell membrane rupture due to the accumulation of ROS and lipid peroxidation.
3.8. Correlation between GABA Level and All Physiological and Metabolic Parameters
Under all water treatment levels used in this study, the GABA level was negatively correlated with seed germination, seedlings height, fresh and dry weight, and chlorophyll content (Table 5). On the other hand, GABA level was positively correlated with protein and carbohydrate contents and MDA level under all water treatments. The elevated level of GABA metabolites (GABA, alanine, and glutamate), total proteins, and total carbohydrates content might be involved in cellular osmotic adjustment, protecting plants from oxidative stress, balancing of carbon and nitrogen (C:N) metabolism, and maintaining cell metabolic homeostasis and cell turgor under water stress [9,22].

Table 5.
Correlation analysis between the GABA level and all physiological and metabolic parameters that were measured in this study under drought stress treatments after 7 and14 days, separately of pea (Pisum sativum L.) seedlings. G% = germination percentage; r = correlation coefficient.
4. Conclusions
Our study showed that water deficit had a suppressive effect on Pisum sativum L. growth and metabolism. Drought stress significantly inhibited seed germination, decreased fresh and dry weight and plant height due to a reduction in water absorption by seeds during metabolic processes and enzymatic activities in germinating pea seeds and early seedling growth. In addition, water deficit caused a significant decrease in chlorophyll pigments that might be attributed to disturbances in the activity of the photosynthetic apparatus. Pea seedlings suffered minor cellular damage under mild drought stress. However, the oxidative damage and ROS production were more severe with increased duration and levels of water deficit. The increase in GABA content as water availability decreased mitigated the damaging effects of drought stress on pea (Pisum sativum L.) seedlings by supplying enough carbon/nitrogen sources to the TCA cycle and amino acid synthesis. The significant increase in protein content might lower the osmotic potential to maintain cellular structures and metabolic stability under drought stress. Pea seedlings tolerate prolonged water deficit by keeping stable protein metabolism to adjust cellular osmolarity and to provide protection from further dehydration damage. Water deficit induced carbohydrate accumulation to maintain water potential and osmotic adjustment via the activation of osmolytes synthesis. Overall, the significant increases in GABA, protein and carbohydrate contents were to cope with the physiological impact of drought stress on Pisum sativum L. seedlings by maintaining cellular osmotic adjustment, protecting plants from oxidative stress, balancing carbon and nitrogen (C:N) metabolism and maintaining cell metabolic homeostasis and cell turgor. According to data presented in the current study, a sufficient water supply is vital for normal growth and metabolism in pea seedlings. Severe (less than 40% water content of the holding capacity) and long-term drought stress should be avoided during the germination stage to ensure proper seedling growth.
Author Contributions
N.A.A.-Q.: Study design, statistical analysis, result interpretation, writing and revision of the manuscript. Z.I.A.-A.: Methodology assistance, data analysis, and result discussion. N.F.Q.: Performing the experimental work, data collection and writing the first draft of the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This research was financially supported by the Deanship of Research, Jordan University of Science and Technology, Jordan, grant number [118/2019].
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Raw data is available on request.
Conflicts of Interest
No conflict of interest to declare.
References
- Yang, T.; Fang, L.; Zhang, X.; Hu, J.; Bao, S.; Hao, J.; Li, L.; He, Y.; Jiang, J.; Wang, F.; et al. High-throughput development of SSR markers from pea (Pisum sativum L.) based on next generation sequencing of a purified Chinese commercial variety. PLoS ONE 2015, 10, e0139775. [Google Scholar] [CrossRef]
- Rungruangmaitree, R.; Jiraungkoorskul, W. Pea, Pisum sativum, and its anticancer activity. Pharmacogn. Rev. 2017, 11, 39. [Google Scholar]
- El-Esawi, M.; Al-Ghamdi, A.; Ali, H.; Alayafi, A.; Witczak, J.; Ahmad, M. Analysis of genetic variation and enhancement of salt tolerance in French pea (Pisum Sativum L.). Int. J. Mol. Sci. 2018, 19, 2433. [Google Scholar] [CrossRef]
- Devi, J.; Mishra, G.P.; Sanwal, S.K.; Dubey, R.K.; Singh, P.M.; Singh, B. Development and characterization of penta-flowering and triple-flowering genotypes in garden pea (Pisum sativum L. var. hortense). PLoS ONE 2018, 13, e0201235. [Google Scholar] [CrossRef] [PubMed]
- Ranjbar Sistani, N.; Kaul, H.P.; Desalegn, G.; Wienkoop, S. Rhizobium impacts on seed productivity, quality, and protection of Pisum sativum upon disease stress caused by didymella pinodes: Phenotypic, proteomic, and metabolomic traits. Front. Plant Sci. 2017, 8, 1961. [Google Scholar] [CrossRef]
- Idrissi, O.; Udupa, S.M.; De Keyser, E.; McGee, R.J.; Coyne, C.J.; Saha, G.C.; Muehlbauer, F.J.; Van Damme, P.; De Riek, J. Identification of quantitative trait loci controlling root and shoot traits associated with drought tolerance in a lentil (Lens culinaris Medik.) recombinant inbred line population. Front. Plant Sci. 2016, 7, 1174. [Google Scholar] [CrossRef] [PubMed]
- Muscolo, A.; Junker, A.; Klukas, C.; Weigelt-Fischer, K.; Riewe, D.; Altmann, T. Phenotypic and metabolic responses to drought and salinity of four contrasting lentil accessions. J. Exp. Bot. 2015, 66, 5467–5480. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Zhang, Y.; Wu, X.; Liu, Y. Drought stress impact on leaf proteome variations of faba bean (Vicia faba L.) in the Qinghai–Tibet Plateau of China. 3 Biotech 2018, 8, 110. [Google Scholar] [CrossRef] [PubMed]
- Rahmati, M.; Mirás-Avalos, J.M.; Valsesia, P.; Lescourret, F.; Génard, M.; Davarynejad, G.H.; Bannayan, M.; Azizi, M.; Vercambre, G. Disentangling the effects of water stress on carbon acquisition, vegetative growth, and fruit quality of peach trees by means of the QualiTree model. Front. Plant Sci. 2018, 9, 3. [Google Scholar] [CrossRef]
- Alexieva, V.; Sergiev, I.; Mapelli, S.; Karanov, E. The effect of drought and ultraviolet radiation on growth and stress markers in pea and wheat. Plant Cell Environ. 2001, 24, 1337–1344. [Google Scholar] [CrossRef]
- Biswas, D.K.; Jiang, G.M. Differential drought-induced modulation of ozone tolerance in winter wheat species. J. Exp. Bot. 2011, 62, 4153–4162. [Google Scholar] [CrossRef]
- Maiti, R.K.; Satya, P. Research advances in major cereal crops for adaptation to abiotic stresses. GM Crop. Food 2014, 5, 259–279. [Google Scholar] [CrossRef]
- Jin, J.; Lauricella, D.; Armstrong, R.; Sale, P.; Tang, C. Phosphorus application and elevated CO2 enhance drought tolerance in field pea grown in a phosphorus-deficient vertisol. Ann. Bot. 2014, 116, 975–985. [Google Scholar] [CrossRef] [PubMed]
- Szalonek, M.; Sierpien, B.; Rymaszewski, W.; Gieczewska, K.; Garstka, M.; Lichocka, M.; Sass, L.; Paul, K.; Vass, I.; Vankova, R.; et al. Potato annexin STANN1 promotes drought tolerance and mitigates light stress in transgenic Solanum tuberosum L. plants. PLoS ONE 2015, 10, e0132683. [Google Scholar] [CrossRef]
- Wang, X.; Cai, X.; Xu, C.; Wang, Q.; Dai, S. Drought-responsive mechanisms in plant leaves revealed by proteomics. Int. J. Mol. Sci. 2016, 17, 1706. [Google Scholar] [CrossRef] [PubMed]
- Charlton, A.J.; Donarski, J.A.; Harrison, M.; Jones, S.A.; Godward, J.; Oehlschlager, S.; Arques, J.L.; Ambrose, M.; Chinoy, C.; Mullineaux, P.M.; et al. Responses of the pea (Pisum sativum L.) leaf metabolome to drought stress assessed by nuclear magnetic resonance spectroscopy. Metabolomics 2008, 4, 312. [Google Scholar] [CrossRef]
- Mo, Y.; Wang, Y.; Yang, R.; Zheng, J.; Liu, C.; Li, H.; Ma, J.; Zhang, Y.; Wei, C.; Zhang, X. Regulation of plant growth, photosynthesis, antioxidation and osmosis by an arbuscular mycorrhizal fungus in watermelon seedlings under well-watered and drought conditions. Front. Plant Sci. 2016, 7, 644. [Google Scholar] [CrossRef]
- Chmielnicka, A.; Żabka, A.; Winnicki, K.; Maszewski, J.; Polit, J.T. Endoreplication and its consequences in the suspensor of Pisum sativum. Plant Cell Rep. 2018, 37, 1639–1651. [Google Scholar] [CrossRef] [PubMed]
- Cattivelli, L.; Rizza, F.; Badeck, F.W.; Mazzucotelli, E.; Mastrangelo, A.M.; Francia, E.; Marè, C.; Tondelli, A.; Stanca, A.M. Drought tolerance improvement in crop plants: An integrated view from breeding to genomics. Field Crop. Res. 2008, 105, 1–14. [Google Scholar] [CrossRef]
- Kakumanu, A.; Ambavaram, M.M.; Klumas, C.; Krishnan, A.; Batlang, U.; Myers, E.; Grene, R.; Pereira, A. Effects of drought on gene expression in maize reproductive and leaf meristem tissue revealed by RNA-Seq. Plant Physiol. 2012, 160, 846–867. [Google Scholar] [CrossRef]
- Mao, H.; Wang, H.; Liu, S.; Li, Z.; Yang, X.; Yan, J.; Li, J.; Tran, L.S.P.; Qin, F. A transposable element in a NAC gene is associated with drought tolerance in maize seedlings. Nat. Commun. 2015, 6, 8326. [Google Scholar] [CrossRef]
- Chen, D.; Wang, S.; Cao, B.; Cao, D.; Leng, G.; Li, H.; Yin, L.; Shan, L.; Deng, X. Genotypic variation in growth and physiological response to drought stress and re-watering reveals the critical role of recovery in drought adaptation in maize seedlings. Front. Plant Sci. 2016, 6, 1241. [Google Scholar] [CrossRef]
- Bunce, J.A. Leaf transpiration efficiency of some drought-resistant maize lines. Crop Sci. 2010, 50, 1409–1413. [Google Scholar] [CrossRef]
- Alghabari, F.; Ihsan, M.Z. Effects of drought stress on growth, grain filling duration, yield and quality attributes of barley (Hordeum vulgare L.). Bangladesh J. Bot. 2018, 47, 421–428. [Google Scholar] [CrossRef]
- Hussain, M.; Farooq, S.; Hasan, W.; Ul-Allah, S.; Tanveer, M.; Farooq, M.; Nawaz, A. Drought stress in sunflower: Physiological effects and its management through breeding and agronomic alternatives. Agric. Water Manag. 2018, 201, 152–166. [Google Scholar] [CrossRef]
- Nonami, H. Plant water relations and control of cell elongation at low water potentials. J. Plant Res. 1998, 111, 373–382. [Google Scholar] [CrossRef]
- Zhang, J.; Mason, A.S.; Wu, J.; Liu, S.; Zhang, X.; Luo, T.; Redden, R.; Batley, J.; Hu, L.; Yan, G. Identification of putative candidate genes for water stress tolerance in canola (Brassica napus). Front. Plant Sci. 2015, 6, 1058. [Google Scholar] [CrossRef]
- Sorensen, J.N.; Edelenbos, M.; Wienberg, L. Drought effects on green pea texture and related physical-chemical properties at comparable maturity. J. Am. Soc. Hortic. Sci. 2003, 128, 128–135. [Google Scholar] [CrossRef]
- Kumar, N.; Dubey, A.K.; Upadhyay, A.K.; Gautam, A.; Ranjan, R.; Srikishna, S.; Sahu, N.; Behera, S.K.; Mallick, S. GABA accretion reduces Lsi-1 and Lsi-2 gene expressions and modulates physiological responses in Oryza sativa to provide tolerance towards arsenic. Sci. Rep. 2017, 7, 8786. [Google Scholar] [CrossRef]
- Carillo, P. GABA shunt in durum wheat. Front. Plant Sci. 2018, 9, 100. [Google Scholar] [CrossRef] [PubMed]
- White, J.P.; Prell, J.; Ramachandran, V.K.; Poole, P.S. Characterization of a γ-aminobutyric acid transport system of Rhizobium leguminosarum bv. viciae 3841. J. Bacteriol. 2009, 191, 1547–1555. [Google Scholar] [CrossRef]
- Scharff, A.M.; Egsgaard, H.; Hansen, P.E.; Rosendahl, L. Exploring symbiotic nitrogen fixation and assimilation in pea root nodules by in vivo 15N nuclear magnetic resonance spectroscopy and liquid chromatography-mass spectrometry. Plant Physiol. 2003, 131, 367–378. [Google Scholar] [CrossRef][Green Version]
- Yong, B.; Xie, H.; Li, Z.; Li, Y.P.; Zhang, Y.; Nie, G.; Zhang, X.Q.; Ma, X.; Huang, L.K.; Yan, Y.H.; et al. Exogenous application of GABA improves PEG-induced drought tolerance positively associated with GABA-shunt, polyamines, and proline metabolism in white clover. Front. Physiol. 2017, 8, 1107. [Google Scholar] [CrossRef]
- Xiang, L.; Hu, L.; Xu, W.; Zhen, A.; Zhang, L.; Hu, X. Exogenous γ-aminobutyric acid improves the structure and function of photosystem II in muskmelon seedlings exposed to salinity-alkalinity stress. PLoS ONE 2016, 11, e0164847. [Google Scholar] [CrossRef] [PubMed]
- Samarah, N.H.; Mullen, R.E.; Cianzio, S.R.; Scott, P. Dehydrin-like proteins in soybean seeds in response to drought stress during seed filling. Crop Sci. 2006, 46, 2141–2150. [Google Scholar] [CrossRef]
- Jeffrey, S.T.; Humphrey, G.F. New spectrophotometric equations for determining chlorophylls a, b, c1 and c2 in higher plants, algae and natural phytoplankton. Biochem. Und Physiol. Pflanz. 1975, 167, 191–194. [Google Scholar] [CrossRef]
- Zhang, G.; Bown, A.W. The rapid determination of γ-aminobutyric acid. Phytochemistry 1997, 44, 1007–1009. [Google Scholar] [CrossRef]
- Bergmeyer, H.U. Methods of Enzymatic Analysis, 2nd ed.; Verlag Chemie: Weinheim, Germany, 1983; Volume I, p. 42. [Google Scholar]
- Heath, R.L.; Packer, L. Photoperoxidation in isolated chloroplasts: I. Kinetics and stoichiometry of fatty acid peroxidation. Arch. Biochem. Biophys. 1968, 125, 189–198. [Google Scholar] [CrossRef]
- Li, H.; Li, X.; Zhang, D.; Liu, H.; Guan, K. Effects of drought stress on the seed germination and early seedling growth of the endemic desert plant Eremosparton songoricum (Fabaceae). Excli J. 2013, 12, 89. [Google Scholar]
- Liu, M.; Li, M.; Liu, K.; Sui, N. Effects of drought stress on seed germination and seedling growth of different maize varieties. J. Agric. Sci. 2015, 7, 231. [Google Scholar] [CrossRef]
- Pirdashti, H.; Sarvestani, Z.T.; Nematzadeh, G.H.; Ismail, A. Effect of water stress on seed germination and seedling growth of rice (Oryza sativa L.) genotypes. Pak. J. Agron. 2003, 2, 217–222. [Google Scholar] [CrossRef]
- Murillo-Amador, B.; López-Aguilar, R.; Kaya, C.; Larrinaga-Mayoral, J.; Flores-Hernández, A. Comparative effects of NaCl and polyethylene glycol on germination, emergence and seedling growth of cowpea. J. Agron. Crop Sci. 2002, 188, 235–247. [Google Scholar] [CrossRef]
- Hadas, A. Water uptake and germination of leguniinous seeds under changing external water potential in osmotic solutions. J. Exp. Bot. 1976, 27, 480–489. [Google Scholar] [CrossRef]
- Muscolo, A.; Sidari, M.; Anastasi, U.; Santonoceto, C.; Maggio, A. Effect of PEG-induced drought stress on seed germination of four lentil genotypes. J. Plant Interact. 2014, 9, 354–363. [Google Scholar] [CrossRef]
- Bentsink, L.; Koornneef, M. Seed dormancy and germination. In The Arabidopsis Book/American Society of Plant Biologists; American Society of Plant Biologists: Rockville, MD, USA, 2008; Volume 6. [Google Scholar]
- Dornbos, D.L.; Mullen, R.E.; Shibles, R.E. Drought stress effects during seed fill on soybean seed germination and vigor. Crop Sci. 1989, 29, 476–480. [Google Scholar] [CrossRef]
- Kim, S.K.; Son, T.K.; Park, S.Y.; Lee, I.J.; Lee, B.H.; Kim, H.Y.; Lee, S.C. Influences of gibberellin and auxin on endogenous plant hormone and starch mobilization during rice seed germination under salt stress. J. Environ. Biol. 2006, 27, 181. [Google Scholar]
- Li, Z.; Peng, Y.; Zhang, X.Q.; Ma, X.; Huang, L.K.; Yan, Y.H. Exogenous spermidine improves seed germination of white clover under water stress via involvement in starch metabolism, antioxidant defenses and relevant gene expression. Molecules 2014, 19, 18003–18024. [Google Scholar] [CrossRef] [PubMed]
- AL-Quraan, N.A.; Sartawe, F.A.B.; Qaryouti, M.M. Characterization of γ-aminobutyric acid metabolism and oxidative damage in wheat (Triticum aestivum L.) seedlings under salt and osmotic stress. J. Plant Physiol. 2013, 170, 1003–1009. [Google Scholar] [CrossRef]
- AL-Quraan, N.A.; Al-Sharbati, M.; Dababneh, Y.; Al-Olabi, M. Effect of temperature, salt and osmotic stresses on seed germination and chlorophyll contents in lentil (Lens culinaris Medik). Acta Hortic. 2014, 1054, 47–54. [Google Scholar] [CrossRef]
- Embiale, A.; Hussein, M.; Husen, A.; Sahile, S.; Mohammed, K. Differential sensitivity of Pisum sativum L. cultivars to water-deficit stress: Changes in growth, water status, chlorophyll fluorescence and gas exchange attributes. J. Agron. 2016, 15, 45–57. [Google Scholar] [CrossRef]
- Morgan, J.M. Osmoregulation and water stress in higher plants. Annu. Rev. Plant Physiol. 1984, 35, 299–319. [Google Scholar] [CrossRef]
- Jaleel, C.A.; Manivannan, P.A.; Wahid, A.; Farooq, M.; Al-Juburi, H.J.; Somasundaram, R.A.; Panneerselvam, R. Drought stress in plants: A review on morphological characteristics and pigments composition. Int. J. Agric. Biol. 2009, 11, 100–105. [Google Scholar]
- Jaleel, C.A.; Manivannan, P.; Lakshmanan, G.M.A.; Gomathinayagam, M.; Panneerselvam, R. Alterations in morphological parameters and photosynthetic pigment responses of Catharanthus roseus under soil water deficits. Colloids Surf. B Biointerfaces 2008, 61, 298–303. [Google Scholar] [CrossRef] [PubMed]
- Medrano, H.; Escalona, J.M.; Bota, J.; Gulías, J.; Flexas, J. Regulation of photosynthesis of C3 plants in response to progressive drought: Stomatal conductance as a reference parameter. Ann. Bot. 2002, 89, 895–905. [Google Scholar] [CrossRef]
- Fahad, S.; Bajwa, A.A.; Nazir, U.; Anjum, S.A.; Farooq, A.; Zohaib, A.; Sadia, S.; Nasim, W.; Adkins, S.; Saud, S.; et al. Crop production under drought and heat stress: Plant responses and management options. Front. Plant Sci. 2017, 8, 1147. [Google Scholar] [CrossRef] [PubMed]
- Fraser, T.E.; Silk, W.K.; Rost, T.L. Effects of low water potential on cortical cell length in growing regions of maize roots. Plant Physiol. 1990, 93, 648–651. [Google Scholar] [CrossRef] [PubMed]
- Khorasaninejad, S.; Mousavi, A.; Soltanloo, H.; Hemmati, K.; Khalighi, A. The effect of salinity stress on growth parameters, essential oil yield and constituent of peppermint (Mentha piperita L.). World Appl. Sci. J. 2010, 11, 1403–1407. [Google Scholar]
- Kabay, T.; Erdinc, C.; Sensoy, S. Effects of drought stress on plant growth parameters, membrane damage index and nutrient content in common bean genotypes. J. Anim. Plant Sci. 2017, 27, 940–952. [Google Scholar]
- Zhang, X.; Lei, L.; Lai, J.; Zhao, H.; Song, W. Effects of drought stress and water recovery on physiological responses and gene expression in maize seedlings. Biomedcentral Plant Biol. 2018, 18, 68. [Google Scholar] [CrossRef]
- Regier, N.; Streb, S.; Cocozza, C.; Schaub, M.; Cherubini, P.; Zeeman, S.C.; Frey, B. Drought tolerance of two black poplar (Populus nigra L.) clones: Contribution of carbohydrates and oxidative stress defense. Plant Cell Environ. 2009, 32, 1724–1736. [Google Scholar] [CrossRef]
- Riad, G.S.; Youssef, S.M.; El-Azm, N.A.A.; Ahmed, E.M. Amending Sandy Soil with Biochar or/and Superabsorbent Polymer Mitigates the Adverse Effects of Drought Stress on Green Pea. Egypt. J. Hortic. 2018, 45, 169–183. [Google Scholar]
- Nyachiro, J.M.; Briggs, K.G.; Hoddinott, J.; Johnson-Flanagan, A.M. Chlorophyll content, chlorophyll fluorescence and water deficit in spring wheat. Cereal Res. Commun. 2001, 29, 135–142. [Google Scholar] [CrossRef]
- Shinde, B.P.; Thakur, J. Influence of Arbuscular mycorrhizal fungi on chlorophyll, proteins, proline and total carbohydrates content of the pea plant under water stress condition. Int. J. Curr. Microbiol. Appl. Sci. 2015, 4, 809–821. [Google Scholar]
- Kyparissis, A.; Petropoulou, Y.; Manetas, Y. Summer survival of leaves in a soft-leaved shrub (Phlomis fruticosa L., Labiatae) under Mediterranean field conditions: Avoidance of photoinhibitory damage through decreased chlorophyll contents. J. Exp. Bot. 1995, 46, 1825–1831. [Google Scholar] [CrossRef]
- Sepehri, A.; Golparvar, A.R. The effect of drought stress on water relations, chlorophyll content and leaf area in canola cultivars (Brassica napus L.). Electron. J. Biol. 2011, 7, 49–53. [Google Scholar]
- Nayyar, H.; Gupta, D. Differential sensitivity of C3 and C4 plants to water deficit stress: Association with oxidative stress and antioxidants. Environ. Exp. Bot. 2006, 58, 106–113. [Google Scholar] [CrossRef]
- Massacci, A.; Nabiev, S.M.; Pietrosanti, L.; Nematov, S.K.; Chernikova, T.N.; Thor, K.; Leipner, J. Response of the photosynthetic apparatus of cotton (Gossypium hirsutum) to the onset of drought stress under field conditions studied by gas-exchange analysis and chlorophyll fluorescence imaging. Plant Physiol. Biochem. 2008, 46, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Husen, A. Growth characteristics, physiological and metabolic responses of teak (Tectona grandis Linn. f.) clones differing in rejuvenation capacity subjected to drought stress. Silvae Genet. 2010, 59, 124–136. [Google Scholar] [CrossRef]
- Ashraf, M.Y.; Azmi, A.R.; Khan, A.H.; Ala, S.A. Effect of water stress on total phenols, peroxidase activity and chlorophyll content in wheat (Triticum aestivum L.). Acta Physiol. Plant. 1994, 16, 3. [Google Scholar]
- Santos, M.G.D.; Ribeiro, R.V.; Oliveira, R.F.D.; Pimentel, C. Gas exchange and yield response to foliar phosphorus application in Phaseolus vulgaris L. under drought. Braz. J. Plant Physiol. 2004, 16, 171–179. [Google Scholar] [CrossRef]
- Srivastava, A.K.; Lokhande, V.H.; Patade, V.Y.; Suprasanna, P.; Sjahril, R.; D’Souza, S.F. Comparative evaluation of hydro-, chemo-, and hormonal-priming methods for imparting salt and PEG stress tolerance in Indian mustard (Brassicajuncea L.). Acta Physiol. Plant. 2010, 32, 1135–1144. [Google Scholar] [CrossRef]
- Afshari, M.; Shekari, F.; Azimkhani, R.; Habibi, H.; Fotokian, M.H. Effects of foliar application of salicylic acid on growth and physiological attributes of cowpea under water stress conditions. Iran Agric. Res. 2013, 32, 55–70. [Google Scholar]
- Tóth, V.R.; Mészáros, I.; Veres, S.; Nagy, J. Effects of the available nitrogen on the photosynthetic activity and xanthophyll cycle pool of maize in field. J. Plant Physiol. 2002, 159, 627–634. [Google Scholar] [CrossRef]
- Fariduddin, Q.; Khanam, S.; Hasan, S.A.; Ali, B.; Hayat, S.; Ahmad, A. Effect of 28-homobrassinolide on the drought stress-induced changes in photosynthesis and antioxidant system of Brassica juncea L. Acta Physiol. Plant. 2009, 31, 889–897. [Google Scholar] [CrossRef]
- Pastenes, C.; Pimentel, P.; Lillo, J. Leaf movements and photoinhibition in relation to water stress in field-grown beans. J. Exp. Bot. 2004, 56, 425–433. [Google Scholar] [CrossRef]
- Lawlor, D.W. Limitation to photosynthesis in water-stressed leaves: Stomata vs. metabolism and the role of ATP. Ann. Bot. 2002, 89, 871–885. [Google Scholar] [CrossRef] [PubMed]
- Shahriari, R.; Karimi, L. Evaluation of cold tolerance in wheat germplasm by measuring chlorophyll content and leaf color. In Abstracts Seventh Crop Science Congress of Iran; Seventh Iranian Congress of Plant Breeding and Plants (Persian); Shahid Bahonar University of Kerman, Pazhuhesh Square, Emam Khomeini Blvd.: Kerman, Iran, 2001; p. 507. [Google Scholar]
- Pandey, H.C.; Baig, M.J.; Bhatt, R.K. Effect of moisture stress on chlorophyll accumulation and nitrate reductase activity at vegetative and flowering stage in Avena species. Agric. Sci. Res. J. 2012, 2, 111–118. [Google Scholar]
- Alberte, R.S.; Thornber, J.P.; Fiscus, E.L. Water stress effects on the content and organization of chlorophyll in mesophyll and bundle sheath chloroplasts of maize. Plant Physiol. 1977, 59, 351–353. [Google Scholar] [CrossRef]
- Patro, L.; Mohapatra, P.K.; Biswal, U.C.; Biswal, B. Dehydration induced loss of photosynthesis in Arabidopsis leaves during senescence is accompanied by the reversible enhancement in the activity of cell wall β-glucosidase. J. Photochem. Photobiol. B Biol. 2014, 137, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Ghotbi-Ravandi, A.A.; Shahbazi, M.; Shariati, M.; Mulo, P. Effects of mild and severe drought stress on photosynthetic efficiency in tolerant and susceptible barley (Hordeum vulgare L.) genotypes. J. Agron. Crop Sci. 2014, 200, 403–415. [Google Scholar] [CrossRef]
- Karataş, İ.; Öztürk, L.; Demir, Y.; Ünlükara, A.; Kurunç, A.; Düzdemir, O. Alterations in antioxidant enzyme activities and proline content in pea leaves under long-term drought stress. Toxicol. Ind. Health 2014, 30, 693–700. [Google Scholar] [CrossRef]
- Nikolaeva, M.K.; Maevskaya, S.N.; Shugaev, A.G.; Bukhov, N.G. Effect of drought on chlorophyll content and antioxidant enzyme activities in leaves of three wheat cultivars varying in productivity. Russ. J. Plant Physiol. 2010, 57, 87–95. [Google Scholar] [CrossRef]
- Bown, A.W.; Shelp, B.J. The metabolism and functions of [gamma]-aminobutyric acid. Plant Physiol. 1997, 115, 1. [Google Scholar] [CrossRef]
- Li, W.; Liu, J.; Ashraf, U.; Li, G.; Li, Y.; Lu, W.; Gao, L.; Han, F.; Hu, J. Exogenous γ-aminobutyric acid (GABA) application improved early growth, net photosynthesis, and associated physio-biochemical events in maize. Front. Plant Sci. 2016, 7, 919. [Google Scholar] [CrossRef]
- Zhu, Z.; Shi, Z.; Xie, C.; Gong, W.; Hu, Z.; Peng, Y. A novel mechanism of Gamma-aminobutyric acid (GABA) protecting human umbilical vein endothelial cells (HUVECs) against H2O2-induced oxidative injury. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2019, 217, 68–75. [Google Scholar] [CrossRef]
- Kaplan, F.; Kopka, J.; Haskell, D.W.; Zhao, W.; Schiller, K.C.; Gatzke, N.; Sung, D.Y.; Guy, C.L. Exploring the temperature-stress metabolome of Arabidopsis. Plant Physiol. 2004, 136, 4159–4168. [Google Scholar] [CrossRef]
- Fait, A.; Fromm, H.; Walter, D.; Galili, G.; Fernie, A.R. Highway or byway: The metabolic role of the GABA shunt in plants. Trends Plant Sci. 2008, 13, 14–19. [Google Scholar] [CrossRef]
- Bashir, R.; Riaz, H.N.; Shafiq, M.; Parveen, N.; Alenazi, M.M.; Anwar, S.; Alebidi, A.I. Foliar application of Y-aminobutyric acid (gaba) improves vegetative growth, and the physiological and antioxidative potential of Daucus Carota L. under water deficit conditions. Preprints 2019, 2019030227. [Google Scholar] [CrossRef]
- Kinnersley, A.M.; Turano, F.J. Gamma aminobutyric acid (GABA) and plant responses to stress. Crit. Rev. Plant Sci. 2000, 19, 479–509. [Google Scholar] [CrossRef]
- Krishnan, S.; Laskowski, K.; Shukla, V.; Merewitz, E.B. Mitigation of drought stress damage by exogenous application of a non-protein amino acid γ–aminobutyric acid on perennial ryegrass. J. Am. Soc. Hortic. Sci. 2013, 138, 358–366. [Google Scholar] [CrossRef]
- Vijayakumari, K.; Puthur, J.T. γ-Aminobutyric acid (GABA) priming enhances the osmotic stress tolerance in Piper nigrum Linn. plants subjected to PEG-induced stress. Plant Growth Regul. 2016, 78, 57–67. [Google Scholar] [CrossRef]
- Wang, C.; Fan, L.; Gao, H.; Wu, X.; Li, J.; Lv, G.; Gong, B. Polyamine biosynthesis and degradation are modulated by exogenous gamma-aminobutyric acid in root-zone hypoxia-stressed melon roots. Plant Physiol. Biochem. 2014, 82, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Mibei, E.K.; Owino, W.O.; Ambuko, J.; Giovannoni, J.J.; Onyango, A.N. Metabolomic analyses to evaluate the effect of drought stress on selected African Eggplant accessions. J. Sci. Food Agric. 2018, 98, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Merewitz, E.B.; Du, H.; Yu, W.; Liu, Y.; Gianfagna, T.; Huang, B. Elevated cytokinin content in ipt transgenic creeping bentgrass promotes drought tolerance through regulating metabolite accumulation. J. Exp. Bot. 2011, 63, 1315–1328. [Google Scholar] [CrossRef] [PubMed]
- Marček, T.; Hamow, K.Á.; Végh, B.; Janda, T.; Darko, E. Metabolic response to drought in six winter wheat genotypes. PLoS ONE 2019, 14, e0212411. [Google Scholar]
- Michaletti, A.; Naghavi, M.R.; Toorchi, M.; Zolla, L.; Rinalducci, S. Metabolomics and proteomics reveal drought-stress responses of leaf tissues from spring-wheat. Sci. Rep. 2018, 8, 5710. [Google Scholar] [CrossRef]
- Salama, H.M.; Al Watban, A.A.; Al-Fughom, A.T. Effect of ultraviolet radiation on chlorophyll, carotenoid, protein and proline contents of some annual desert plants. Saudi J. Biol. Sci. 2011, 18, 79–86. [Google Scholar] [CrossRef]
- Guttieri, M.J.; McLean, R.; Stark, J.C.; Souza, E. Managing irrigation and nitrogen fertility of hard spring wheats for optimum bread and noodle quality. Crop Sci. 2005, 45, 2049–2059. [Google Scholar] [CrossRef]
- Ozturk, A.; Aydin, F. Effect of water stress at various growth stages on some quality characteristics of winter wheat. J. Agron. Crop Sci. 2004, 190, 93–99. [Google Scholar] [CrossRef]
- Rizhsky, L.; Liang, H.; Mittler, R. The combined effect of drought stress and heat shock on gene expression in tobacco. Plant Physiol. 2002, 130, 1143–1151. [Google Scholar] [CrossRef]
- Jin, R.; Wang, Y.; Liu, R.; Gou, J.; Chan, Z. Physiological and metabolic changes of purslane (Portulaca oleracea L.) in response to drought, heat, and combined stresses. Front. Plant Sci. 2016, 6, 1123. [Google Scholar] [CrossRef] [PubMed]
- Ashoub, A.; Beckhaus, T.; Berberich, T.; Karas, M.; Brüggemann, W. Comparative analysis of barley leaf proteome as affected by drought stress. Planta 2013, 237, 771–781. [Google Scholar] [CrossRef]
- Alvarez, S.; Roy Choudhury, S.; Pandey, S. Comparative quantitative proteomics analysis of the ABA response of roots of drought-sensitive and drought-tolerant wheat varieties identifies proteomic signatures of drought adaptability. J. Proteome Res. 2014, 13, 1688–1701. [Google Scholar] [CrossRef] [PubMed]
- Suguiyama, V.F.; da Silva, E.A.; Meirelles, S.T.; Centeno, D.D.C.; Braga, M.R. Leaf metabolite profile of the Brazilian resurrection plant Barbacenia purpurea Hook. (Velloziaceae) shows two time-dependent responses during desiccation and recovering. Front. Plant Sci. 2014, 5, 96. [Google Scholar] [CrossRef] [PubMed]
- Ford, K.L.; Cassin, A.; Bacic, A.F. Quantitative proteomic analysis of wheat cultivars with differing drought stress tolerance. Front. Plant Sci. 2011, 2, 44. [Google Scholar] [CrossRef]
- Gołębiowska-Pikania, G.; Kopeć, P.; Surówka, E.; Janowiak, F.; Krzewska, M.; Dubas, E.; Nowicka, A.; Kasprzyk, J.; Ostrowska, A.; Malaga, S.; et al. Changes in protein abundance and activity induced by drought during generative development of winter barley (Hordeum vulgare L.). J. Proteom. 2017, 169, 73–86. [Google Scholar] [CrossRef] [PubMed]
- Mohammadkhani, N.; Heidari, R. Effects of drought stress on soluble proteins in two maize varieties. Turk. J. Biol. 2008, 32, 23–30. [Google Scholar]
- Ashraf, M.; Foolad, M.R. Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ. Exp. Bot. 2007, 59, 206–216. [Google Scholar] [CrossRef]
- Bhosale, K.S.; Shinde, B. Influence of arbuscular mycorrhizal fungi on proline and chlorophyll content in Zingiber officinale Rosc grown under water stress. Indian J. Fundam. Appl. Life Sci. 2011, 1, 172–176. [Google Scholar]
- Nayyar, H. Accumulation of osmolytes and osmotic adjustment in water-stressed wheat (Triticum aestivum L.) and maize (Zea mays) affected by calcium and its antagonists. Environ. Exp. Bot. 2003, 50, 253–264. [Google Scholar] [CrossRef]
- Zhu, X.; Gong, H.; Chen, G.; Wang, S.; Zhang, C. Different solute levels in two spring wheat cultivars induced by progressive field water stress at different developmental stages. J. Arid Environ. 2005, 62, 1–14. [Google Scholar] [CrossRef]
- Singh, M.; Kumar, J.; Singh, S.; Singh, V.P.; Prasad, S.M. Roles of osmoprotectants in improving salinity and drought tolerance in plants: A review. Rev. Environ. Sci. Bio Technol. 2015, 14, 407–426. [Google Scholar] [CrossRef]
- Shahryar, N.; Maali-Amiri, R. Metabolic acclimation of tetraploid and hexaploid wheats by cold stress-induced carbohydrate accumulation. J. Plant Physiol. 2016, 204, 44–53. [Google Scholar] [CrossRef]
- Arabzadeh, N. H aloxylon persicum and H aloxylon aphyllum. Asian J. Plant Sci. 2012, 11, 44–51. [Google Scholar] [CrossRef]
- Kameli, A.; Lösel, D.M. Growth and sugar accumulation in durum wheat plants under water stress. New Phytol. 1996, 132, 57–62. [Google Scholar] [CrossRef]
- Fazeli, F.; Ghorbanli, M.; Niknam, V. Effect of drought on water relations, growth and solute accumulation in two sesame cultivars. Pak. J. Biol. Sci. 2006, 9, 1829–1835. [Google Scholar] [CrossRef]
- Akinci, S.; Losel, D.M. Effects of water stress and recovery periods on soluble sugars and starch content in cucumber cultivars. Fresenius Environ. Bull. 2010, 19, 164–171. [Google Scholar]
- Lemoine, R.; La Camera, S.; Atanassova, R.; Dédaldéchamp, F.; Allario, T.; Pourtau, N.; Bonnemain, J.L.; Laloi, M.; Coutos-Thévenot, P.; Maurousset, L.; et al. Source-to-sink transport of sugar and regulation by environmental factors. Front. Plant Sci. 2013, 4, 272. [Google Scholar] [CrossRef] [PubMed]
- Jain, N.K.; Roy, I. Effect of trehalose on protein structure. Protein Sci. 2009, 18, 24–36. [Google Scholar] [CrossRef]
- Buitink, J.; Leprince, O. Glass formation in plant anhydrobiotes: Survival in the dry state. Cryobiology 2004, 48, 215–228. [Google Scholar] [CrossRef]
- Shvaleva, A.L.; Silva, F.C.E.; Breia, E.; Jouve, J.; Hausman, J.F.; Almeida, M.H.; Maroco, J.P.; Rodrigues, M.L.; Pereira, J.S.; Chaves, M.M. Metabolic responses to water deficit in two Eucalyptus globulus clones with contrasting drought sensitivity. Tree Physiol. 2006, 26, 239–248. [Google Scholar] [CrossRef]
- Gibson, S.I. Control of plant development and gene expression by sugar signaling. Curr. Opin. Plant Biol. 2005, 8, 93–102. [Google Scholar] [CrossRef] [PubMed]
- Van den Ende, W.; Peshev, D. Sugars as antioxidants in plants. In Crop Improvement under Adverse Conditions; Springer: New York, NY, USA, 2013; pp. 285–307. [Google Scholar]
- Li, T.H.; Li, S.H. Leaf responses of micropropagated apple plants to water stress: Nonstructural carbohydrate composition and regulatory role of metabolic enzymes. Tree Physiol. 2005, 25, 495–504. [Google Scholar] [CrossRef] [PubMed]
- Moore, K.; Roberts, L.J. Measurement of lipid peroxidation. Free Radic. Res. 1998, 28, 659–671. [Google Scholar] [CrossRef]
- Apel, K.; Hirt, H. Reactive oxygen species: Metabolism, oxidative stress, and signal transduction. Annu. Rev. Plant Biol. 2004, 55, 373–399. [Google Scholar] [CrossRef] [PubMed]
- Berjak, P. The challenge of recalcitrant germplasm cryopreservation. J. Hortic. Sci. Biotechnol. 2006, 81, 781–782. [Google Scholar] [CrossRef]
- Gill, S.S.; Tuteja, N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol. Biochem. 2010, 48, 909–930. [Google Scholar] [CrossRef]
- Rahal, A.; Kumar, A.; Singh, V.; Yadav, B.; Tiwari, R.; Chakraborty, S.; Dhama, K. Oxidative stress, prooxidants, and antioxidants: The interplay. Biomed Res. Int. 2014, 2014, 761264. [Google Scholar] [CrossRef]
- Pandey, H.C.; Baig, M.J.; Chandra, A.; Bhatt, R.K. Drought stress induced changes in lipid peroxidation and antioxidant system in genus Avena. J. Environ. Biol. 2010, 31, 435–440. [Google Scholar]
- Zlatev, Z.S.; Lidon, F.C.; Ramalho, J.C.; Yordanov, I.T. Comparison of resistance to drought of three bean cultivars. Biol. Plant. 2006, 50, 389–394. [Google Scholar] [CrossRef]
- Tatar, O.; Gevrek, I. Lipid peroxidation and water content of wheat. Asian J. Plant Sci. 2008, 7, 409–412. [Google Scholar] [CrossRef]
- Morabito, D.; Guerrier, G. The free oxygen radical scavenging enzymes and redox status in roots and leaves of Populus x Euramericana in response to osmotic stress, desiccation and rehydration. J. Plant Physiol. 2000, 157, 74–80. [Google Scholar] [CrossRef]
- Abid, M.; Ali, S.; Qi, L.K.; Zahoor, R.; Tian, Z.; Jiang, D.; Snider, J.L.; Dai, T. Physiological and biochemical changes during drought and recovery periods at tillering and jointing stages in wheat (Triticum aestivum L.). Sci. Rep. 2018, 8, 4615. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, J.A.; Jimenez, A.; Mullineaux, P.; Sevilia, F. Tolerance of pea (Pisum sativum L.) to long-term salt stress is associated with induction of antioxidant defences. Plant Cell Environ. 2000, 23, 853–862. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).