The Health of Vineyard Soils: Towards a Sustainable Viticulture
Abstract
1. Introduction
2. Soil Health, Soil Quality, and Soil Fertility
3. The Soil Biological Fertility of Vineyards
3.1. The Impact of Soil Organisms on Chemical Fertility
3.2. The Impact of Soil Organisms on Physical Fertility
3.3. Key Interactions in the Rhizosphere on Soil Biological Fertility
3.4. The Impact of Mycorrhizae on Soil Health
4. Soil Organic Matter as a Key Property of Soil Health in Vineyards
4.1. Soil Organic Matter and Soil-Health Attributes
4.2. Dynamics of Winegrowing Systems and Levels of Soil Organic Matter
5. Soil Physical Fertility in Vineyards
5.1. Soil Compaction, Water Infiltration, and Aeration
5.2. Soil Erosion
6. Tillage and Soil Health in Vineyards
6.1. How Agricultural Traffic and Tillage Affect Soil Compaction
6.2. Cultivation Techniques to Avoid Soil Compaction
6.3. Effects of Cover Crops and Mulching on Soil Health
7. Impacts of Viticultural Phytosanitary Products on Soil Health
7.1. Copper-Based Phytosanitary Products
7.2. Organically Based Phytosanitary Products
7.3. Interrelation of Soil Health and Vine Health: Pest and Disease Pressure
8. Fertilizers and Soil Amendments for Soil Health in Viticulture
8.1. Mineral Nutrients Effects on Vines and Soil Health
8.2. Organic Approaches for Soil Health in Viticulture
8.3. Soil Acidity and Liming Effects on Soil Health in Vineyards
9. The Expression of Terroir and Soil Health
9.1. The Factors That Determine the Terroir Expression
9.2. The Soil Environment and the Terroir Expression
9.3. Terroir Expression and Soil Health in Zoning Procedures
10. Chemical, Physical, and Biological Indicators of Soil Health
10.1. Soil Organic Carbon
10.2. Soil Aggregate Stability
10.3. Soil Bulk Density and Infiltration Capacity
10.4. Soil Chemistry: pH, Salinity, Sodicity, Nutrients, and Pollutants
10.5. Soil Biology
11. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- International Organisation of Vine and Wine. Annual Assessment of the World Vine and Wine Sector in 2022; International Organisation of Vine and Wine: Dijon, France, 2023. [Google Scholar]
- Poore, J.; Nemecek, T. Reducing Food’s Environmental Impacts through Producers and Consumers. Science 2018, 360, 987–992. [Google Scholar] [CrossRef]
- Cataldo, E.; Fucile, M.; Mattii, G.B. A Review: Soil Management, Sustainable Strategies and Approaches to Improve the Quality of Modern Viticulture. Agronomy 2021, 11, 2359. [Google Scholar] [CrossRef]
- International Health Conference. Constitution of the World Health Organization. 1946. Bull. World Health Organ. 2002, 80, 983–984. [Google Scholar]
- Minami, K. Soil Is a Living Substance. Soil Sci. Plant Nutr. 2021, 67, 26–30. [Google Scholar] [CrossRef]
- Doran, J.W.; Zeiss, M.R. Soil Health and Sustainability: Managing the Biotic Component of Soil Quality. Appl. Soil Ecol. 2000, 15, 3–11. [Google Scholar] [CrossRef]
- Yang, T.; Siddique, K.H.M.; Liu, K. Cropping Systems in Agriculture and Their Impact on Soil Health—A Review. Glob. Ecol. Conserv. 2020, 23, e01118. [Google Scholar] [CrossRef]
- Doran, J.W.; Parkin, T.B. Defining and Assessing Soil Quality. In Defining Soil Quality for a Sustainable Environment; Doran, J.W., Coleman, D.C., Bezdicek, D.F., Stewart, B.A., Eds.; SSSA: Madison, WI, USA, 2015; pp. 3–21. ISBN 9780891189305. [Google Scholar]
- Blum, W.E.H. Functions of Soil for Society and the Environment. Rev. Environ. Sci. Biotechnol. 2005, 4, 75–79. [Google Scholar] [CrossRef]
- Riesenfeld, C.S.; Goodman, R.M.; Handelsman, J. Uncultured Soil Bacteria Are a Reservoir of New Antibiotic Resistance Genes. Environ. Microbiol. 2004, 6, 981–989. [Google Scholar] [CrossRef] [PubMed]
- Van Goethem, M.W.; Pierneef, R.; Bezuidt, O.K.I.; Van De Peer, Y.; Cowan, D.A.; Makhalanyane, T.P. A Reservoir of “historical” Antibiotic Resistance Genes in Remote Pristine Antarctic Soils. Microbiome 2018, 6, 40. [Google Scholar] [CrossRef] [PubMed]
- van de Meent, D.; de Bruijn, J.H.M. A Modeling Procedure to Evaluate the Coherence of Independently Derived Environmental Quality Objectives for Air, Water and Soil. Environ. Toxicol. Chem. 1995, 14, 177–186. [Google Scholar] [CrossRef]
- Karlen, D.L.; Mausbach, M.J.; Doran, J.W.; Cline, R.G.; Harris, R.F.; Schuman, G.E. Soil Quality: A Concept, Definition, and Framework for Evaluation (A Guest Editorial). Soil Sci. Soc. Am. J. 1997, 61, 4–10. [Google Scholar] [CrossRef]
- Calistri, P.; Iannetti, S.; Danzetta, M.L.; Narcisi, V.; Cito, F.; Di Sabatino, D.; Bruno, R.; Sauro, F.; Atzeni, M.; Carvelli, A.; et al. The Components of ‘One World-One Health’ Approach. Transbound. Emerg. Dis. 2013, 60, 4–13. [Google Scholar] [CrossRef]
- United Nations. FAOSTAT. Available online: https://www.fao.org/faostat/en/#home (accessed on 6 December 2023).
- Ohno, T.; Hettiarachchi, G.M. Soil Chemistry and the One Health Initiative: Introduction to the Special Section. J. Environ. Qual. 2018, 47, 1305–1309. [Google Scholar] [CrossRef] [PubMed]
- Van Bruggen, A.H.C.; Goss, E.M.; Havelaar, A.; van Diepeningen, A.D.; Finckh, M.R.; Morris, J.G. One Health-Cycling of Diverse Microbial Communities as a Connecting Force for Soil, Plant, Animal, Human and Ecosystem Health. Sci. Total Environ. 2019, 664, 927–937. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.; van der Heijden, M.G.A. Soil Microbiomes and One Health. Nat. Rev. Microbiol. 2022, 21, 6–20. [Google Scholar] [CrossRef] [PubMed]
- Bonilla-Aldana, D.K.; Dhama, K.; Rodriguez-Morales, A.J. Revisiting the One Health Approach in the Context of COVID-19: A Look into the Ecology of This Emerging Disease. Adv. Anim. Vet. Sci. 2020, 8, 234–237. [Google Scholar] [CrossRef]
- Abbott, L.; Murphy, D. Soil Biological Fertility: A Key to Sustainable Land Use in Agriculture; Springer: Berlin/Heidelberg, Germany, 2007. [Google Scholar]
- Stirling, G.; Hayden, H.; Pattison, T.; Stirling, M. Soil Health, Soil Biology, Soilborne Diseases and Sustainable Agriculture; CSIRO: Canberra, Australia, 2016. [Google Scholar]
- Hatfield, J.L.; Walthall, C.L. Soil Biological Fertility: Foundation for the Next Revolution in Agriculture? Commun. Soil Sci. Plant Anal. 2015, 46, 753–762. [Google Scholar] [CrossRef]
- Blanco-Pérez, R.; Vicente-Díez, I.; Ramos-Sáez de Ojer, J.L.; Marco-Mancebón, V.S.; Pérez-Moreno, I.; Campos-Herrera, R. Organic Viticulture Enhanced the Activity of Native Entomopathogenic Nematodes in DOCa Rioja Soils (North of Spain). Agric. Ecosyst. Environ. 2022, 332, 107931. [Google Scholar] [CrossRef]
- Rodrigo-Comino, J. Five Decades of Soil Erosion Research in “Terroir”. The State-of-the-Art. Earth Sci. Rev. 2018, 179, 436–447. [Google Scholar] [CrossRef]
- Karimi, B.; Cahurel, J.Y.; Gontier, L.; Charlier, L.; Chovelon, M.; Mahé, H.; Ranjard, L. A Meta-Analysis of the Ecotoxicological Impact of Viticultural Practices on Soil Biodiversity. Environ. Chem. Lett. 2020, 18, 1947–1966. [Google Scholar] [CrossRef]
- Giffard, B.; Winter, S.; Guidoni, S.; Nicolai, A.; Castaldini, M.; Cluzeau, D.; Coll, P.; Cortet, J.; Le Cadre, E.; d’Errico, G.; et al. Vineyard Management and Its Impacts on Soil Biodiversity, Functions, and Ecosystem Services. Front. Ecol. Evol. 2022, 10, 850272. [Google Scholar] [CrossRef]
- Marques, M.J.; Bienes, R.; Cuadrado, J.; Ruiz-Colmenero, M.; Barbero-Sierra, C.; Velasco, A. Analysing Perceptions Attitudes and Responses of Winegrowers about Sustainable Land Management in Central Spain. Land Degrad. Dev. 2015, 26, 458–467. [Google Scholar] [CrossRef]
- Khmelevtsova, L.E.; Sazykin, I.S.; Azhogina, T.N.; Sazykina, M.A. Influence of Agricultural Practices on Bacterial Community of Cultivated Soils. Agriculture 2022, 12, 371. [Google Scholar] [CrossRef]
- Wu, T.; Milner, H.; Díaz-Pérez, J.C.; Ji, P. Effects of Soil Management Practices on Soil Microbial Communities and Development of Southern Blight in Vegetable Production. Appl. Soil Ecol. 2015, 91, 58–67. [Google Scholar] [CrossRef]
- García-Orenes, F.; Guerrero, C.; Roldán, A.; Mataix-Solera, J.; Cerdà, A.; Campoy, M.; Zornoza, R.; Bárcenas, G.; Caravaca, F. Soil Microbial Biomass and Activity under Different Agricultural Management Systems in a Semiarid Mediterranean Agroecosystem. Soil Tillage Res. 2010, 109, 110–115. [Google Scholar] [CrossRef]
- Pampulha, M.E.; Oliveira, A. Impact of an Herbicide Combination of Bromoxynil and Prosulfuron on Soil Microorganisms. Curr. Microbiol. 2006, 53, 238–243. [Google Scholar] [CrossRef]
- Young, I.M.; Crawford, J.W. Interactions and Self-Organization in the Soil-Microbe Complex. Science 2004, 304, 1634–1637. [Google Scholar] [CrossRef] [PubMed]
- Marín, D.; Armengol, J.; Carbonell-Bejerano, P.; Escalona, J.M.; Gramaje, D.; Hernández-Montes, E.; Intrigliolo, D.S.; Martínez-Zapater, J.M.; Medrano, H.; Mirás-Avalos, J.M.; et al. Challenges of Viticulture Adaptation to Global Change: Tackling the Issue from the Roots. Aust. J. Grape Wine Res. 2020, 27, 8–25. [Google Scholar] [CrossRef]
- Darriaut, R.; Lailheugue, V.; Masneuf-Pomarède, I.; Marguerit, E.; Martins, G.; Compant, S.; Ballestra, P.; Upton, S.; Ollat, N.; Lauvergeat, V. Grapevine Rootstock and Soil Microbiome Interactions: Keys for a Resilient Viticulture. Hortic. Res. 2022, 9, uhac019. [Google Scholar] [CrossRef] [PubMed]
- Agnelli, A.; Bol, R.; Trumbore, S.E.; Dixon, L.; Cocco, S.; Corti, G. Carbon and Nitrogen in Soil and Vine Roots in Harrowed and Grass-Covered Vineyards. Agric. Ecosyst. Environ. 2014, 193, 70–82. [Google Scholar] [CrossRef]
- Ritz, K.; Harris, J.; Murray, P. The Role of Soil Biota in Soil Fertility and Quality, and Approaches to Influencing Soil Communities to Enhance Delivery of These Functions; Department for Environment, Food and Rural Affairs: London, UK, 2010. [Google Scholar]
- Bettenfeld, P.; Cadena i Canals, J.; Jacquens, L.; Fernandez, O.; Fontaine, F.; van Schaik, E.; Courty, P.E.; Trouvelot, S. The Microbiota of the Grapevine Holobiont: A Key Component of Plant Health. J. Adv. Res. 2022, 40, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Zarraonaindia, I.; Owens, S.M.; Weisenhorn, P.; West, K.; Hampton-Marcell, J.; Lax, S.; Bokulich, N.A.; Mills, D.A.; Martin, G.; Taghavi, S.; et al. The Soil Microbiome Influences Grapevine-Associated Microbiota. mBio 2015, 6, e02527-14. [Google Scholar] [CrossRef] [PubMed]
- Custódio, V.; Gonin, M.; Stabl, G.; Bakhoum, N.; Oliveira, M.M.; Gutjahr, C.; Castrillo, G. Sculpting the Soil Microbiota. Plant J. 2022, 109, 508–522. [Google Scholar] [CrossRef]
- Pingel, M.; Reineke, A.; Leyer, I. A 30-Years Vineyard Trial: Plant Communities, Soil Microbial Communities and Litter Decomposition Respond More to Soil Treatment than to N Fertilization. Agric. Ecosyst. Environ. 2019, 272, 114–125. [Google Scholar] [CrossRef]
- Trouvelot, S.; Bonneau, L.; Redecker, D.; van Tuinen, D.; Adrian, M.; Wipf, D. Arbuscular Mycorrhiza Symbiosis in Viticulture: A Review. Agron. Sustain. Dev. 2015, 35, 1449–1467. [Google Scholar] [CrossRef]
- Lemtiri, A.; Colinet, G.; Alabi, T.; Cluzeau, D.; Zirbes, L.; Haubruge, É.; Francis, F. Impacts of Earthworms on Soil Components and Dynamics. A Review. Biotechnol. Agron. Soc. Environ. 2014, 18, 121–133. [Google Scholar]
- Neemisha. Role of Soil Organisms in Maintaining Soil Health, Ecosystem Functioning, and Sustaining Agricultural Production. In Soil Health; Springer: Berlin/Heidelberg, Germany, 2020; pp. 313–335. [Google Scholar]
- Tian, T.; Reverdy, A.; She, Q.; Sun, B.; Chai, Y. The Role of Rhizodeposits in Shaping Rhizomicrobiome. Environ. Microbiol. Rep. 2019, 12, 160–172. [Google Scholar] [CrossRef] [PubMed]
- Raghavendra, M.P.; Santhoshkannada, A.N. Role of Rhizomicrobiome in Maintaining Soil Fertility and Crop Production. In Soil Health; Springer: Berlin/Heidelberg, Germany, 2020; pp. 373–401. [Google Scholar]
- Vaudour, E.; Costantini, E.; Jones, G.V.; Mocali, S. An Overview of the Recent Approaches to Terroir Functional Modelling, Footprinting and Zoning. Soil 2015, 1, 287–312. [Google Scholar] [CrossRef]
- Mcnear, D., Jr. The Rhizosphere-Roots, Soil and Everything in Between. Nat. Educ. Knowl. 2013, 4, 1. [Google Scholar]
- Whalley, W.R.; Riseley, B.; Leeds-Harrison, P.B.; Bird, N.R.A.; Leech, P.K.; Adderley, W.P. Structural Differences between Bulk and Rhizosphere Soil. Eur. J. Soil Sci. 2005, 56, 353–360. [Google Scholar] [CrossRef]
- Youssef, R.A.; Chino, M. Root-Induced Changes in the Rhizosphere of Plants. II. Distribution of Heavy Metals across the Rhizosphere in Soils. Soil Sci. Plant Nutr. 1989, 35, 609–621. [Google Scholar] [CrossRef]
- Youssef, R.A.; Chino, M. Root-Induced Changes in the Rhizosphere of Plants. I. PH Changes in Relation to the Bulk Soil. Soil Sci. Plant Nutr. 1989, 35, 461–468. [Google Scholar] [CrossRef]
- Wang, L.; Rengel, Z.; Zhang, K.; Jin, K.; Lyu, Y.; Zhang, L.; Cheng, L.; Zhang, F.; Shen, J. Ensuring Future Food Security and Resource Sustainability: Insights into the Rhizosphere. iScience 2022, 25, 104168. [Google Scholar] [CrossRef] [PubMed]
- Gianfreda, L. Enzymes of Importance to Rhizosphere Processes. J. Soil Sci. Plant Nutr. 2015, 15, 283–306. [Google Scholar] [CrossRef]
- Custos, J.-M.; Moyne, C.; Sterckeman, T. How Root Nutrient Uptake Affects Rhizosphere PH: A Modelling Study. Geoderma 2020, 369, 114314. [Google Scholar] [CrossRef]
- Afridi, M.S.; Fakhar, A.; Kumar, A.; Ali, S.; Medeiros, F.H.V.; Muneer, M.A.; Ali, H.; Saleem, M. Harnessing Microbial Multitrophic Interactions for Rhizosphere Microbiome Engineering. Microbiol. Res. 2022, 265, 127199. [Google Scholar] [CrossRef]
- Bao, L.; Sun, B.; Wei, Y.; Xu, N.; Zhang, S.; Gu, L.; Bai, Z. Grape Cultivar Features Differentiate the Grape Rhizosphere Microbiota. Plants 2022, 11, 1111. [Google Scholar] [CrossRef]
- Dries, L.; Bussotti, S.; Pozzi, C.; Kunz, R.; Schnell, S.; Löhnertz, O.; Vortkamp, A. Rootstocks Shape Their Microbiome—Bacterial Communities in the Rhizosphere of Different Grapevine Rootstocks. Microorganisms 2021, 9, 822. [Google Scholar] [CrossRef]
- Dries, L.; Hendgen, M.; Schnell, S.; Löhnertz, O.; Vortkamp, A. Rhizosphere Engineering: Leading towards a Sustainable Viticulture? OENO One 2021, 55, 353–363. [Google Scholar] [CrossRef]
- Baweja, P.; Kumar, S.; Kumar, G. Fertilizers and Pesticides: Their Impact on Soil Health and Environment. In Soil Health; Springer: Berlin/Heidelberg, Germany, 2020; pp. 265–285. [Google Scholar]
- Schreiner, R.P. Spatial and Temporal Variation of Roots, Arbuscular Mycorrhizal Fungi, and Plant and Soil Nutrients in a Mature Pinot Noir (Vitis vinifera L.) Vineyard in Oregon, USA. Plant Soil 2005, 276, 219–234. [Google Scholar] [CrossRef]
- Aguilera, P.; Ortiz, N.; Becerra, N.; Turrini, A.; Gaínza-Cortés, F.; Silva-Flores, P.; Aguilar-Paredes, A.; Romero, J.K.; Jorquera-Fontena, E.; Mora, M.d.L.L.; et al. Application of Arbuscular Mycorrhizal Fungi in Vineyards: Water and Biotic Stress Under a Climate Change Scenario: New Challenge for Chilean Grapevine Crop. Front. Microbiol. 2022, 13, 826571. [Google Scholar] [CrossRef]
- Gazioglu Sensoy, R. Effects of Arbuscular Mycorrhizal Fungus (AMF) and Whey Applications on the Grapevine (Vitis Vinifera L.) Cutting Exposed to Salt Stress. J. Elem. 2022, 27, 507–519. [Google Scholar] [CrossRef]
- Cardinale, M.; Minervini, F.; De Angelis, M.; Papadia, P.; Migoni, D.; Dimaglie, M.; Dinu, D.G.; Quarta, C.; Selleri, F.; Caccioppola, A.; et al. Vineyard Establishment under Exacerbated Summer Stress: Effects of Mycorrhization on Rootstock Agronomical Parameters, Leaf Element Composition and Root-Associated Bacterial Microbiota. Plant Soil 2022, 478, 613–634. [Google Scholar] [CrossRef]
- Lumini, E.; Orgiazzi, A.; Borriello, R.; Bonfante, P.; Bianciotto, V. Disclosing Arbuscular Mycorrhizal Fungal Biodiversity in Soil through a Land-use Gradient Using a Pyrosequencing Approach. Environ. Microbiol. 2010, 12, 2165–2179. [Google Scholar] [CrossRef]
- Lehmann, J.; Kleber, M. The Contentious Nature of Soil Organic Matter. Nature 2015, 528, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Kögel-Knabner, I. The Macromolecular Organic Composition of Plant and Microbial Residues as Inputs to Soil Organic Matter: Fourteen Years On. Soil Biol. Biochem. 2017, 105, A3–A8. [Google Scholar] [CrossRef]
- Stockmann, U.; Padarian, J.; McBratney, A.; Minasny, B.; de Brogniez, D.; Montanarella, L.; Hong, S.Y.; Rawlins, B.G.; Field, D.J. Global Soil Organic Carbon Assessment. Glob. Food Secur. 2015, 6, 9–16. [Google Scholar] [CrossRef]
- Hengl, T.; Mendes de Jesus, J.; Heuvelink, G.B.M.; Ruiperez Gonzalez, M.; Kilibarda, M.; Blagotić, A.; Shangguan, W.; Wright, M.N.; Geng, X.; Bauer-Marschallinger, B.; et al. SoilGrids250m: Global Gridded Soil Information Based on Machine Learning. PLoS ONE 2017, 12, e0169748. [Google Scholar] [CrossRef] [PubMed]
- Loveland, P.; Webb, J. Is There a Critical Level of Organic Matter in the Agricultural Soils of Temperate Regions: A Review. Soil Tillage Res. 2003, 70, 1–18. [Google Scholar] [CrossRef]
- Murphy, B.W. Impact of Soil Organic Matter on Soil Properties—A Review with Emphasis on Australian Soils. Soil Res. 2015, 53, 605. [Google Scholar] [CrossRef]
- Foley, J.A. The Sensitivity of the Terrestrial Biosphere to Climatic Change: A Simulation of the Middle Holocene. Global Biogeochem. Cycles 1994, 8, 505–525. [Google Scholar] [CrossRef]
- Ruddiman, W.F. The Anthropogenic Greenhouse Era Began Thousands of Years Ago. Clim. Change 2003, 61, 261–293. [Google Scholar] [CrossRef]
- Lal, R. Soil Carbon Sequestration Impacts on Global Climate Change and Food Security. Science 2004, 304, 1623–1627. [Google Scholar] [CrossRef] [PubMed]
- Feller, C.; Manlay, R.J.; Swift, M.J.; Bernoux, M. Functions, Services and Value of Soil Organic Matter for Human Societies and the Environment: A Historical Perspective. Geol. Soc. Lond. Spec. Publ. 2006, 266, 9–22. [Google Scholar] [CrossRef]
- Tisdall, J.M.; Oades, J.M. Organic Matter and Water-stable Aggregates in Soils. J. Soil Sci. 1982, 33, 141–163. [Google Scholar] [CrossRef]
- Kong, A.Y.Y.; Six, J.; Bryant, D.C.; Denison, R.F.; van Kessel, C. The Relationship between Carbon Input, Aggregation, and Soil Organic Carbon Stabilization in Sustainable Cropping Systems. Soil Sci. Soc. Am. J. 2005, 69, 1078–1085. [Google Scholar] [CrossRef]
- Chen, H.; Hou, R.; Gong, Y.; Li, H.; Fan, M.; Kuzyakov, Y. Effects of 11 Years of Conservation Tillage on Soil Organic Matter Fractions in Wheat Monoculture in Loess Plateau of China. Soil Tillage Res. 2009, 106, 85–94. [Google Scholar] [CrossRef]
- Mustafa, A.; Minggang, X.; Ali Shah, S.A.; Abrar, M.M.; Nan, S.; Baoren, W.; Zejiang, C.; Saeed, Q.; Naveed, M.; Mehmood, K.; et al. Soil Aggregation and Soil Aggregate Stability Regulate Organic Carbon and Nitrogen Storage in a Red Soil of Southern China. J. Environ. Manag. 2020, 270, 110894. [Google Scholar] [CrossRef]
- Xu, N.; Amgain, N.R.; Rabbany, A.; Capasso, J.; Korus, K.; Swanson, S.; Bhadha, J.H. Interaction of Soil Health Indicators to Different Regenerative Farming Practices on Mineral Soils. Agrosystems Geosci. Environ. 2022, 5, e20243. [Google Scholar] [CrossRef]
- Cresswell, H.P.; Kirkegaard, J.A. Subsoil Amelioration by Plant-Roots—The Process and the Evidence. Aust. J. Soil Res. 1995, 33, 221. [Google Scholar] [CrossRef]
- Yunusa, I.A.M.; Newton, P.J. Plants for Amelioration of Subsoil Constraints and Hydrological Control: The Primer-Plant Concept. Plant Soil 2003, 257, 261–281. [Google Scholar] [CrossRef]
- Lavelle, P.; Spain, A.; Blouin, M.; Brown, G.; Decaëns, T.; Grimaldi, M.; Jiménez, J.J.; McKey, D.; Mathieu, J.; Velasquez, E.; et al. Ecosystem Engineers in a Self-Organized Soil: A Review of Concepts and Future Research Questions. Soil Sci. 2016, 181, 91–109. [Google Scholar] [CrossRef]
- Lehmann, A.; Leifheit, E.F.; Rillig, M.C. Mycorrhizas and Soil Aggregation. In Mycorrhizal Mediation of Soil; Elsevier: Amsterdam, The Netherlands, 2017; pp. 241–262. [Google Scholar]
- Wilpiszeski, R.L.; Aufrecht, J.A.; Retterer, S.T.; Sullivan, M.B.; Graham, D.E.; Pierce, E.M.; Zablocki, O.D.; Palumbo, A.V.; Elias, D.A. Soil Aggregate Microbial Communities: Towards Understanding Microbiome Interactions at Biologically Relevant Scales. Appl. Environ. Microbiol. 2019, 85, e00324-19. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Zhang, J.; Zhang, C.; Xin, X.; Li, H. The Coupling Effects of Soil Organic Matter and Particle Interaction Forces on Soil Aggregate Stability. Soil Tillage Res. 2017, 174, 251–260. [Google Scholar] [CrossRef]
- Li, S.; Wang, B.; Zhang, X.; Wang, H.; Yi, Y.; Huang, X.; Gao, X.; Zhu, P.; Han, W. Soil Particle Aggregation and Aggregate Stability Associated with Ion Specificity and Organic Matter Content. Geoderma 2023, 429, 116285. [Google Scholar] [CrossRef]
- Li, Z.; Zhang, X.; Liu, Y. Pore-Scale Simulation of Gas Diffusion in Unsaturated Soil Aggregates: Accuracy of the Dusty-Gas Model and the Impact of Saturation. Geoderma 2017, 303, 196–203. [Google Scholar] [CrossRef]
- Hu, W.; Jiang, Y.; Chen, D.; Lin, Y.; Han, Q.; Cui, Y. Impact of Pore Geometry and Water Saturation on Gas Effective Diffusion Coefficient in Soil. Appl. Sci. 2018, 8, 2097. [Google Scholar] [CrossRef]
- Prifling, B.; Weber, M.; Ray, N.; Prechtel, A.; Phalempin, M.; Schlüter, S.; Vetterlein, D.; Schmidt, V. Quantifying the Impact of 3D Pore Space Morphology on Soil Gas Diffusion in Loam and Sand. Transp. Porous Media 2023, 149, 501–527. [Google Scholar] [CrossRef]
- Ehlers, W. Observations on Earthworm Channels and Infiltration on Tilled and Untilled Loess Soil. Soil Sci. 1975, 119, 242–249. [Google Scholar] [CrossRef]
- Franzluebbers, A.J. Water Infiltration and Soil Structure Related to Organic Matter and Its Stratification with Depth. Soil Tillage Res. 2002, 66, 197–205. [Google Scholar] [CrossRef]
- Lipiec, J.; Kuś, J.; Słowińska-Jurkiewicz, A.; Nosalewicz, A. Soil Porosity and Water Infiltration as Influenced by Tillage Methods. Soil Tillage Res. 2006, 89, 210–220. [Google Scholar] [CrossRef]
- Kirkby, C.A.; Kirkegaard, J.A.; Richardson, A.E.; Wade, L.J.; Blanchard, C.; Batten, G. Stable Soil Organic Matter: A Comparison of C:N:P:S Ratios in Australian and Other World Soils. Geoderma 2011, 163, 197–208. [Google Scholar] [CrossRef]
- Tipping, E.; Somerville, C.J.; Luster, J. The C:N:P:S Stoichiometry of Soil Organic Matter. Biogeochemistry 2016, 130, 117–131. [Google Scholar] [CrossRef] [PubMed]
- Coonan, E.C.; Kirkby, C.A.; Kirkegaard, J.A.; Amidy, M.R.; Strong, C.L.; Richardson, A.E. Microorganisms and Nutrient Stoichiometry as Mediators of Soil Organic Matter Dynamics. Nutr. Cycl. Agroecosyst. 2020, 117, 273–298. [Google Scholar] [CrossRef]
- Schulten, H.-R.; Schnitzer, M. The Chemistry of Soil Organic Nitrogen: A Review. Biol. Fertil. Soils 1997, 26, 1–15. [Google Scholar] [CrossRef]
- Schroth, A.W.; Bostick, B.C.; Graham, M.; Kaste, J.M.; Mitchell, M.J.; Friedland, A.J. Sulfur Species Behavior in Soil Organic Matter during Decomposition. J. Geophys. Res. Biogeosci. 2007, 112, G04011. [Google Scholar] [CrossRef]
- Spohn, M. Increasing the Organic Carbon Stocks in Mineral Soils Sequesters Large Amounts of Phosphorus. Glob. Change Biol. 2020, 26, 4169–4177. [Google Scholar] [CrossRef] [PubMed]
- Parfitt, R.L.; Giltrap, D.J.; Whitton, J.S. Contribution of Organic Matter and Clay Minerals to the Cation Exchange Capacity of Soils. Commun. Soil Sci. Plant Anal. 1995, 26, 1343–1355. [Google Scholar] [CrossRef]
- Oorts, K.; Vanlauwe, B.; Merckx, R. Cation Exchange Capacities of Soil Organic Matter Fractions in a Ferric Lixisol with Different Organic Matter Inputs. Agric. Ecosyst. Environ. 2003, 100, 161–171. [Google Scholar] [CrossRef]
- Fageria, N.; Baligar, V.; Clark, R. Micronutrients in Crop Production. Adv. Agron. 2002, 77, 185–268. [Google Scholar]
- Lindsay, W.L. Iron Oxide Solubilization by Organic Matter and Its Effect on Iron Availability. Plant Soil 1991, 130, 27–34. [Google Scholar] [CrossRef]
- Dhaliwal, S.S.; Naresh, R.K.; Mandal, A.; Singh, R.; Dhaliwal, M.K. Dynamics and Transformations of Micronutrients in Agricultural Soils as Influenced by Organic Matter Build-up: A Review. Environ. Sustain. Indic. 2019, 1–2, 100007. [Google Scholar] [CrossRef]
- Merino, C.; Kuzyakov, Y.; Godoy, K.; Jofré, I.; Nájera, F.; Matus, F. Iron-Reducing Bacteria Decompose Lignin by Electron Transfer from Soil Organic Matter. Sci. Total Environ. 2021, 761, 143194. [Google Scholar] [CrossRef]
- Jones, D.L. Organic Acids in the Rhizosphere-A Critical Review. Plant Soil 1998, 205, 25–44. [Google Scholar] [CrossRef]
- Goldberg, S. Reactions of Boron with Soils. Plant Soil 1997, 193, 35–48. [Google Scholar] [CrossRef]
- Sauvé, S.; Hendershot, W.; Allen, H.E. Solid-Solution Partitioning of Metals in Contaminated Soils: Dependence on PH, Total Metal Burden, and Organic Matter. Environ. Sci. Technol. 2000, 34, 1125–1131. [Google Scholar] [CrossRef]
- Wichard, T.; Mishra, B.; Myneni, S.C.B.; Bellenger, J.-P.; Kraepiel, A.M.L. Storage and Bioavailability of Molybdenum in Soils Increased by Organic Matter Complexation. Nat. Geosci. 2009, 2, 625–629. [Google Scholar] [CrossRef]
- Tolls, J. Sorption of Veterinary Pharmaceuticals in Soils: A Review. Environ. Sci. Technol. 2001, 35, 3397–3406. [Google Scholar] [CrossRef] [PubMed]
- Nam, J.J.; Gustafsson, O.; Kurt-Karakus, P.; Breivik, K.; Steinnes, E.; Jones, K.C. Relationships between Organic Matter, Black Carbon and Persistent Organic Pollutants in European Background Soils: Implications for Sources and Environmental Fate. Environ. Pollut. 2008, 156, 809–817. [Google Scholar] [CrossRef] [PubMed]
- Chien, S.-W.C.; Chen, S.-H.; Li, C.-J. Effect of Soil PH and Organic Matter on the Adsorption and Desorption of Pentachlorophenol. Environ. Sci. Pollut. Res. 2017, 25, 5269–5279. [Google Scholar] [CrossRef]
- Cui, S.; Qi, Y.; Zhu, Q.; Wang, C.; Sun, H. A Review of the Influence of Soil Minerals and Organic Matter on the Migration and Transformation of Sulfonamides. Sci. Total Environ. 2023, 861, 160584. [Google Scholar] [CrossRef]
- Lo, K.S.L.; Yang, W.F.; Lin, Y.C. Effects of Organic Matter on the Specific Adsorption of Heavy Metals by Soil. Toxicol. Environ. Chem. 1992, 34, 139–153. [Google Scholar] [CrossRef]
- Salati, S.; Quadri, G.; Tambone, F.; Adani, F. Fresh Organic Matter of Municipal Solid Waste Enhances Phytoextraction of Heavy Metals from Contaminated Soil. Environ. Pollut. 2010, 158, 1899–1906. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Li, Y.; Yang, X.; Zhang, J.; Lin, Z.; Zhao, B. Microbial Community Structure and Functional Metabolic Diversity Are Associated with Organic Carbon Availability in an Agricultural Soil. J. Integr. Agric. 2015, 14, 2500–2511. [Google Scholar] [CrossRef]
- Zhang, L.; Yan, H.; Ren, T.; Li, S.; Yang, Y.; Peng, G.; Yu, J.; Liu, G. Effects of Organic Matter on Nutrient, Enzyme Activity and Functional Diversity of Microbial Community in Tobacco Planting Soil. Acta Tabacaria Sin. 2019, 25, 55–62. [Google Scholar]
- Johnson, D.C.; Teague, R.; Apfelbaum, S.; Thompson, R.; Byck, P. Adaptive Multi-Paddock Grazing Management’s Influence on Soil Food Web Community Structure for: Increasing Pasture Forage Production, Soil Organic Carbon, and Reducing Soil Respiration Rates in Southeastern USA Ranches. PeerJ 2022, 10, e13750. [Google Scholar] [CrossRef] [PubMed]
- Sokol, N.W.; Kuebbing, S.E.; Karlsen-Ayala, E.; Bradford, M.A. Evidence for the Primacy of Living Root Inputs, Not Root or Shoot Litter, in Forming Soil Organic Carbon. New Phytol. 2018, 221, 233–246. [Google Scholar] [CrossRef] [PubMed]
- Chaney, K.; Swift, R.S. The Influence of Organic Matter on Aggregate Stability in Some British Soils. J. Soil Sci. 1984, 35, 223–230. [Google Scholar] [CrossRef]
- Chenu, C.; Le Bissonnais, Y.; Arrouays, D. Organic Matter Influence on Clay Wettability and Soil Aggregate Stability. Soil Sci. Soc. Am. J. 2000, 64, 1479–1486. [Google Scholar] [CrossRef]
- Le Bissonnais, Y.; Blavet, D.; De Noni, G.; Laurent, J.-Y.; Asseline, J.; Chenu, C. Erodibility of Mediterranean Vineyard Soils: Relevant Aggregate Stability Methods and Significant Soil Variables. Eur. J. Soil Sci. 2007, 58, 188–195. [Google Scholar] [CrossRef]
- Burras, C.; Nyasimi, M.; Butler, L. Soils, Human Health, and Wealth: A Complicated Relationship. In Soils and Human Health; CRC Press: Boca Raton, FL, USA, 2013; pp. 215–226. [Google Scholar]
- Gu, B.; Chen, D.; Yang, Y.; Vitousek, P.; Zhu, Y.-G. Soil-Food-Environment-Health Nexus for Sustainable Development. Research 2021, 2021, 9804807. [Google Scholar] [CrossRef]
- Parras-Alcántara, L.; Martín-Carrillo, M.; Lozano-García, B. Impacts of Land Use Change in Soil Carbon and Nitrogen in a Mediterranean Agricultural Area (Southern Spain). Solid Earth 2013, 4, 167–177. [Google Scholar] [CrossRef]
- Jakšić, S.; Ninkov, J.; Milić, S.; Vasin, J.; Banjac, D.; Jakšić, D.; Živanov, M. The State of Soil Organic Carbon in Vineyards as Affected by Soil Types and Fertilization Strategies (Tri Morave Region, Serbia). Agronomy 2020, 11, 9. [Google Scholar] [CrossRef]
- Maetens, W.; Vanmaercke, M.; Poesen, J.; Jankauskas, B.; Jankauskiene, G.; Ionita, I. Effects of Land Use on Annual Runoff and Soil Loss in Europe and the Mediterranean: A Meta-Analysis of Plot Data. Prog. Phys. Geogr. Earth Environ. 2012, 36, 599–653. [Google Scholar] [CrossRef]
- Prosdocimi, M.; Cerdà, A.; Tarolli, P. Soil Water Erosion on Mediterranean Vineyards: A Review. Catena 2016, 141, 1–21. [Google Scholar] [CrossRef]
- Rodrigo Comino, J.; Senciales, J.M.; Ramos, M.C.; Martínez-Casasnovas, J.A.; Lasanta, T.; Brevik, E.C.; Ries, J.B.; Ruiz Sinoga, J.D. Understanding Soil Erosion Processes in Mediterranean Sloping Vineyards (Montes de Málaga, Spain). Geoderma 2017, 296, 47–59. [Google Scholar] [CrossRef]
- Gristina, L.; Novara, A.; Minacapilli, M. Rethinking Vineyard Ground Management to Counter Soil Tillage Erosion. Soil Tillage Res. 2022, 217, 105275. [Google Scholar] [CrossRef]
- Kibblewhite, M.G.; Ritz, K.; Swift, M.J. Soil Health in Agricultural Systems. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2008, 363, 685–701. [Google Scholar] [CrossRef] [PubMed]
- García-Ruiz, J.M. The Effects of Land Uses on Soil Erosion in Spain: A Review. Catena 2010, 81, 1–11. [Google Scholar] [CrossRef]
- Lozano-Parra, J.; Maldonado-Clavelle, C.F.; Caballero-Calvo, A.; Pulido-Fernández, M.; Gyasi-Agyei, Y.; Rodrigo-Comino, J. Estimation of Soil Mobilization Rates by a Rainy Period and Intense Tillage Practices in Vineyards—A Case Study in the Maule Region (Chile). Hydrol. Process. 2023, 37, e14829. [Google Scholar] [CrossRef]
- Martínez-Casasnovas, J.A.; Ramos, M.C. Soil Alteration Due to Erosion, Ploughing and Levelling of Vineyards in North East Spain. Soil Use Manag. 2009, 25, 183–192. [Google Scholar] [CrossRef]
- Mishra, U.; Lal, R.; Slater, B.; Calhoun, F.; Liu, D.; Van Meirvenne, M. Predicting Soil Organic Carbon Stock Using Profile Depth Distribution Functions and Ordinary Kriging. Soil Sci. Soc. Am. J. 2009, 73, 614–621. [Google Scholar] [CrossRef]
- Bai, J.; Zhang, G.; Zhao, Q.; Lu, Q.; Jia, J.; Cui, B.; Liu, X. Depth-Distribution Patterns and Control of Soil Organic Carbon in Coastal Salt Marshes with Different Plant Covers. Sci. Rep. 2016, 6, 34835. [Google Scholar] [CrossRef]
- Murphy, B.W.; Wilson, B.R.; Koen, T. Mathematical Functions to Model the Depth Distribution of Soil Organic Carbon in a Range of Soils from New South Wales, Australia under Different Land Uses. Soil Syst. 2019, 3, 46. [Google Scholar] [CrossRef]
- Visconti, F.; Intrigliolo, D.S.; Mirás-Avalos, J.M. Effects of the Annual Nitrogen Fertilization Rate on Vine Performance and Grape Quality for Winemaking: Insights from a Meta-Analysis. Aust. J. Grape Wine Res. 2023, 2023, 7989254. [Google Scholar] [CrossRef]
- Baldock, J.; Skjemstad, J. Soil Organic Carbon/Soil Organic Matter. In Soil Analysis: An Interpretation Manual; Peverill, K., Sparrow, L., Reuter, D., Eds.; CSIRO Publishing: Melbourne, Australia, 1999; pp. 159–170. [Google Scholar]
- Lazcano, C.; Decock, C.; Wilson, S.G. Defining and Managing for Healthy Vineyard Soils, Intersections With the Concept of Terroir. Front. Environ. Sci. 2020, 8, 68. [Google Scholar] [CrossRef]
- Müller-Nedebock, D.; Chaplot, V. Soil Carbon Losses by Sheet Erosion: A Potentially Critical Contribution to the Global Carbon Cycle. Earth Surf. Process Landf. 2015, 40, 1803–1813. [Google Scholar] [CrossRef]
- Li, H.; Liu, G.; Gu, J.; Chen, H.; Shi, H.; Abd Elbasit, M.A.M.; Hu, F. Response of Soil Aggregate Disintegration to the Different Content of Organic Carbon and Its Fractions during Splash Erosion. Hydrol. Process. 2021, 35, e14060. [Google Scholar] [CrossRef]
- Vicente-Vicente, J.L.; García-Ruiz, R.; Francaviglia, R.; Aguilera, E.; Smith, P. Soil Carbon Sequestration Rates under Mediterranean Woody Crops Using Recommended Management Practices: A Meta-Analysis. Agric. Ecosyst. Environ. 2016, 235, 204–214. [Google Scholar] [CrossRef]
- Gómez de Barreda, D.; Bautista, I.; Castell, V.; Lidón, A. Rice Straw Mulch Installation in a Vineyard Improves Weed Control and Modifies Soil Characteristics. Agronomy 2023, 13, 3068. [Google Scholar] [CrossRef]
- Qin, T.; Wang, L.; Zhao, J.; Zhou, G.; Li, C.; Guo, L.; Jiang, G. Effects of Straw Mulching Thickness on the Soil Health in a Temperate Organic Vineyard. Agriculture 2022, 12, 1751. [Google Scholar] [CrossRef]
- Morlat, R.; Chaussod, R. Long-Term Additions of Organic Amendments in a Loire Valley Vineyard. I. Effects on Properties of a Calcareous Sandy Soil. Am. J. Enol. Vitic. 2008, 59, 353–363. [Google Scholar] [CrossRef]
- Gaiotti, F.; Marcuzzo, P.; Belfiore, N.; Lovat, L.; Fornasier, F.; Tomasi, D. Influence of Compost Addition on Soil Properties, Root Growth and Vine Performances of Vitis Vinifera Cv Cabernet Sauvignon. Sci. Hortic. 2017, 225, 88–95. [Google Scholar] [CrossRef]
- Torres, N.; Yu, R.; Kurtural, S.K. Inoculation with Mycorrhizal Fungi and Irrigation Management Shape the Bacterial and Fungal Communities and Networks in Vineyard Soils. Microorganisms 2021, 9, 1273. [Google Scholar] [CrossRef] [PubMed]
- Betancur-Agudelo, M.; Meyer, E.; Lovato, P.E. Growth, Heavy Metal Uptake, and Photosynthesis in “Paulsen 1103” (Vitis Berlandieri x Rupestris) Grapevine Rootstocks Inoculated with Arbuscular Mycorrhizal Fungi from Vineyard Soils with High Copper Contents. VITIS—J. Grapevine Res. 2020, 59, 169–180. [Google Scholar] [CrossRef]
- Zhou, J.; Zang, H.; Loeppmann, S.; Gube, M.; Kuzyakov, Y.; Pausch, J. Arbuscular Mycorrhiza Enhances Rhizodeposition and Reduces the Rhizosphere Priming Effect on the Decomposition of Soil Organic Matter. Soil Biol. Biochem. 2020, 140, 107641. [Google Scholar] [CrossRef]
- Guzmán, G.; Cabezas, J.M.; Sánchez-Cuesta, R.; Lora, Á.; Bauer, T.; Strauss, P.; Winter, S.; Zaller, J.G.; Gómez, J.A. A Field Evaluation of the Impact of Temporary Cover Crops on Soil Properties and Vegetation Communities in Southern Spain Vineyards. Agric. Ecosyst. Environ. 2019, 272, 135–145. [Google Scholar] [CrossRef]
- Steenwerth, K.; Belina, K.M. Cover Crops Enhance Soil Organic Matter, Carbon Dynamics and Microbiological Function in a Vineyard Agroecosystem. Appl. Soil Ecol. 2008, 40, 359–369. [Google Scholar] [CrossRef]
- Ruiz-Colmenero, M.; Bienes, R.; Marques, M.J. Soil and Water Conservation Dilemmas Associated with the Use of Green Cover in Steep Vineyards. Soil Tillage Res. 2011, 117, 211–223. [Google Scholar] [CrossRef]
- Fleishman, S.M.; Bock, H.W.; Eissenstat, D.M.; Centinari, M. Undervine Groundcover Substantially Increases Shallow but Not Deep Soil Carbon in a Temperate Vineyard. Agric. Ecosyst. Environ. 2021, 313, 107362. [Google Scholar] [CrossRef]
- Ortega, P.; Sánchez, E.; Gil, E.; Matamoros, V. Use of Cover Crops in Vineyards to Prevent Groundwater Pollution by Copper and Organic Fungicides. Soil Column Studies. Chemosphere 2022, 303, 134975. [Google Scholar] [CrossRef]
- Eon, P.; Robert, T.; Goutouly, J.-P.; Aurelle, V.; Cornu, J.-Y. Cover Crop Response to Increased Concentrations of Copper in Vineyard Soils: Implications for Copper Phytoextraction. Chemosphere 2023, 329, 138604. [Google Scholar] [CrossRef] [PubMed]
- Austin, E.E.; Wickings, K.; McDaniel, M.D.; Robertson, G.P.; Grandy, A.S. Cover Crop Root Contributions to Soil Carbon in a No-till Corn Bioenergy Cropping System. GCB Bioenergy 2017, 9, 1252–1263. [Google Scholar] [CrossRef]
- Mortensen, E.Ø.; De Notaris, C.; Peixoto, L.; Olesen, J.E.; Rasmussen, J. Short-Term Cover Crop Carbon Inputs to Soil as Affected by Long-Term Cropping System Management and Soil Fertility. Agric. Ecosyst. Environ. 2021, 311, 107339. [Google Scholar] [CrossRef]
- Novara, A.; Cerda, A.; Barone, E.; Gristina, L. Cover Crop Management and Water Conservation in Vineyard and Olive Orchards. Soil Tillage Res. 2021, 208, 104896. [Google Scholar] [CrossRef]
- Buesa, I.; Mirás-Avalos, J.M.; De Paz, J.M.; Visconti, F.; Sanz, F.; Yeves, A.; Guerra, D.; Intrigliolo, D.S. Soil Management in Semi-Arid Vineyards: Combined Effects of Organic Mulching and No-Tillage under Different Water Regimes. Eur. J. Agron. 2021, 123, 126198. [Google Scholar] [CrossRef]
- Beslic, Z.; Pantelic, M.; Dabic, D.; Todic, S.; Natic, M.; Tesic, Z. Effect of Vineyard Floor Management on Water Regime, Growth Response, Yield and Fruit Quality in Cabernet Sauvignon. Sci. Hortic. 2015, 197, 650–656. [Google Scholar] [CrossRef]
- Reynolds, W.D.; Bowman, B.T.; Drury, C.F.; Tan, C.S.; Lu, X. Indicators of Good Soil Physical Quality: Density and Storage Parameters. Geoderma 2002, 110, 131–146. [Google Scholar] [CrossRef]
- Kumar, K.S.A.; Karthika, K.S. Abiotic and Biotic Factors Influencing Soil Health and/or Soil Degradation. In Soil Health; Springer: Berlin/Heidelberg, Germany, 2020; pp. 145–161. [Google Scholar]
- Manda, R.R.; Addanki, V.A.; Giabardo, A.; Benjamin, J.; Hossain, M.J.; Khanna, S.; Gaddam, M.; Kumar, R.; Srivastava, S. Soil Health Management and Microorganisms: Recent Development. In Detection, Diagnosis and Management of Soil-Borne Phytopathogens; Springer Nature Singapore: Singapore, 2023; pp. 437–493. [Google Scholar]
- Oliver, D.P.; Bramley, R.G.V.; Riches, D.; Porter, I.; Edwards, J. Review: Soil Physical and Chemical Properties as Indicators of Soil Quality in Australian Viticulture. Aust. J. Grape Wine Res. 2013, 19, 129–139. [Google Scholar] [CrossRef]
- Saljnikov, E.; Mueller, L.; Lavrishchev, A.; Eulenstein, F. Advances in Understanding Soil Degradation Innovations in Landscape Research; Springer: Müncheberg, Germany, 2022. [Google Scholar]
- Schjønning, P. Subsoil Compaction—A Threat to Sustainable Food Production and Soil Ecosystem Services; RECARE Policy Brief; RECARE: Berlin, Germany, 2018. [Google Scholar]
- Batey, T. Soil Compaction and Soil Management—A Review. Soil Use Manag. 2009, 25, 335–345. [Google Scholar] [CrossRef]
- Keller, M. The Science of Grapevines, 3rd ed.; Maragioglio, N., Ed.; Elsevier: Nueva Delhi, India, 2020. [Google Scholar]
- Kirschbaum, M.U.F. The Temperature Dependence of Soil Organic Matter Decomposition, and the Effect of Global Warming on Soil Organic C Storage. Soil Biol. Biochem. 1995, 27, 753–760. [Google Scholar] [CrossRef]
- Keller, T.; Sandin, M.; Colombi, T.; Horn, R.; Or, D. Historical Increase in Agricultural Machinery Weights Enhanced Soil Stress Levels and Adversely Affected Soil Functioning. Soil Tillage Res. 2019, 194, 104293. [Google Scholar] [CrossRef]
- Horn, R.; Schrader, S.; Mordhorst, A.; Fleige, H.; Schroeder, R. Soil Health and Biodiversity: Interactions with Physical Processes and Functions. In Sustainable Soil Management as a Key to Preserve Soil Biodiversity and Stop Its Degradation; Reyes-Sánchez, L., Horn, R., Costantini, E., Eds.; International Union of Soil Sciences (IUSS): Vienna, Austria, 2022; pp. 314–330. [Google Scholar]
- Bogunovic, I.; Bilandzija, D.; Andabaka, Z.; Stupic, D.; Rodrigo Comino, J.; Cacic, M.; Brezinscak, L.; Maletic, E.; Pereira, P. Soil Compaction under Different Management Practices in a Croatian Vineyard. Arab. J. Geosci. 2017, 10, 340. [Google Scholar] [CrossRef]
- Inglett, P.; Reddy, K.; Corstanje, R. Anaerobic Soils. In Encyclopedia of Soils in the Environment; Elsevier: Amsterdam, The Netherlands, 1998; pp. 72–78. [Google Scholar]
- Tan, X.; Chang, S.X. Soil Compaction and Forest Litter Amendment Affect Carbon and Net Nitrogen Mineralization in a Boreal Forest Soil. Soil Tillage Res. 2007, 93, 77–86. [Google Scholar] [CrossRef]
- Tropeano, D. Rate of Soil Erosion Processes on Vineyards in Central Piedmont (NW Italy). Earth Surf. Process Landf. 1984, 9, 253–266. [Google Scholar] [CrossRef]
- Vršič, S. Soil Erosion and Earthworm Population Responses to Soil Management Systems in Steep-Slope Vineyards. Plant Soil Environ. 2011, 57, 258–263. [Google Scholar] [CrossRef]
- Prosdocimi, M.; Jordán, A.; Tarolli, P.; Keesstra, S.; Novara, A.; Cerdà, A. The Immediate Effectiveness of Barley Straw Mulch in Reducing Soil Erodibility and Surface Runoff Generation in Mediterranean Vineyards. Sci. Total Environ. 2016, 547, 323–330. [Google Scholar] [CrossRef]
- Cerdà, A.; Keesstra, S.D.; Rodrigo-Comino, J.; Novara, A.; Pereira, P.; Brevik, E.; Giménez-Morera, A.; Fernández-Raga, M.; Pulido, M.; di Prima, S.; et al. Runoff Initiation, Soil Detachment and Connectivity Are Enhanced as a Consequence of Vineyards Plantations. J. Environ. Manag. 2017, 202, 268–275. [Google Scholar] [CrossRef]
- Pappalardo, S.E.; Gislimberti, L.; Ferrarese, F.; De Marchi, M.; Mozzi, P. Estimation of Potential Soil Erosion in the Prosecco DOCG Area (NE Italy), toward a Soil Footprint of Bottled Sparkling Wine Production in Different Land-Management Scenarios. PLoS ONE 2019, 14, e0210922. [Google Scholar] [CrossRef]
- Stanchi, S.; Zecca, O.; Hudek, C.; Pintaldi, E.; Viglietti, D.; D’Amico, M.E.; Colombo, N.; Goslino, D.; Letey, M.; Freppaz, M. Effect of Soil Management on Erosion in Mountain Vineyards (N-W Italy). Sustainability 2021, 13, 1991. [Google Scholar] [CrossRef]
- Alewell, C.; Ringeval, B.; Ballabio, C.; Robinson, D.A.; Panagos, P.; Borrelli, P. Global Phosphorus Shortage Will Be Aggravated by Soil Erosion. Nat. Commun. 2020, 11, 4546. [Google Scholar] [CrossRef]
- Novara, A.; Pisciotta, A.; Minacapilli, M.; Maltese, A.; Capodici, F.; Cerdà, A.; Gristina, L. The Impact of Soil Erosion on Soil Fertility and Vine Vigor. A Multidisciplinary Approach Based on Field, Laboratory and Remote Sensing Approaches. Sci. Total Environ. 2018, 622–623, 474–480. [Google Scholar] [CrossRef]
- Costantini, E.A.C.; Castaldini, M.; Diago, M.P.; Giffard, B.; Lagomarsino, A.; Schroers, H.-J.; Priori, S.; Valboa, G.; Agnelli, A.E.; Akça, E.; et al. Effects of Soil Erosion on Agro-Ecosystem Services and Soil Functions: A Multidisciplinary Study in Nineteen Organically Farmed European and Turkish Vineyards. J. Environ. Manag. 2018, 223, 614–624. [Google Scholar] [CrossRef]
- Pijl, A.; Wang, W.; Straffelini, E.; Tarolli, P. Soil and Water Conservation in Terraced and Non-Terraced Cultivations: An Extensive Comparison of 50 Vineyards. Land Degrad. Dev. 2022, 33, 596–610. [Google Scholar] [CrossRef]
- Biddoccu, M.; Ferraris, S.; Opsi, F.; Cavallo, E. Long-Term Monitoring of Soil Management Effects on Runoff and Soil Erosion in Sloping Vineyards in Alto Monferrato (North–West Italy). Soil Tillage Res. 2016, 155, 176–189. [Google Scholar] [CrossRef]
- Dezső, J.; Lóczy, D.; Rezsek, M.; Hüppi, R.; Werner, J.; Horváth, L. Crop Growth, Carbon Sequestration and Soil Erosion in an Organic Vineyard of the Villány Wine District, Southwest Hungary. Hung. Geogr. Bull. 2020, 69, 281–298. [Google Scholar] [CrossRef]
- Ruiz-Colmenero, M.; Bienes, R.; Eldridge, D.J.; Marques, M.J. Vegetation Cover Reduces Erosion and Enhances Soil Organic Carbon in a Vineyard in the Central Spain. Catena 2013, 104, 153–160. [Google Scholar] [CrossRef]
- Winter, S.; Bauer, T.; Strauss, P.; Kratschmer, S.; Paredes, D.; Popescu, D.; Landa, B.; Guzmán, G.; Gómez, J.A.; Guernion, M.; et al. Effects of Vegetation Management Intensity on Biodiversity and Ecosystem Services in Vineyards: A Meta-Analysis. J. Appl. Ecol. 2018, 55, 2484–2495. [Google Scholar] [CrossRef] [PubMed]
- Niu, Y.H.; Wang, L.; Wan, X.G.; Peng, Q.Z.; Huang, Q.; Shi, Z.H. A Systematic Review of Soil Erosion in Citrus Orchards Worldwide. Catena 2021, 206, 105558. [Google Scholar] [CrossRef]
- Pou, A.; Gulías, J.; Moreno, M.; Tomàs, M.; Medrano, H.; Cifre, J. Cover Cropping in Vitis vinifera L. Cv. Manto Negro Vineyards under Mediterranean Conditions: Effects on Plant Vigour, Yield and Grape Quality. OENO One 2011, 45, 223. [Google Scholar] [CrossRef]
- Mirás-Avalos, J.M.; Ramírez-Cuesta, J.M.; Fandiño, M.; Cancela, J.J.; Intrigliolo, D.S. Agronomic Practices for Reducing Soil Erosion in Hillside Vineyards under Atlantic Climatic Conditions (Galicia, Spain). Soil Syst. 2020, 4, 19. [Google Scholar] [CrossRef]
- Triplett, G.B.; Dick, W.A. No-Tillage Crop Production: A Revolution in Agriculture! Agron. J. 2008, 100, S-153. [Google Scholar] [CrossRef]
- Osterholz, W.R.; Culman, S.W.; Herms, C.; Joaquim de Oliveira, F.; Robinson, A.; Doohan, D. Knowledge Gaps in Organic Research: Understanding Interactions of Cover Crops and Tillage for Weed Control and Soil Health. Org. Agric. 2021, 11, 13–25. [Google Scholar] [CrossRef]
- Laudicina, V.A.; Palazzolo, E.; Catania, P.; Vallone, M.; García, A.D.; Badalucco, L. Soil Quality Indicators as Affected by Shallow Tillage in a Vineyard Grown in a Semiarid Mediterranean Environment. Land Degrad. Dev. 2017, 28, 1038–1046. [Google Scholar] [CrossRef]
- Shaheb, M.R.; Venkatesh, R.; Shearer, S.A. A Review on the Effect of Soil Compaction and Its Management for Sustainable Crop Production. J. Biosyst. Eng. 2021, 46, 417–439. [Google Scholar] [CrossRef]
- Schwartz, R.C.; Baumhardt, R.L.; Evett, S.R. Tillage Effects on Soil Water Redistribution and Bare Soil Evaporation throughout a Season. Soil Tillage Res. 2010, 110, 221–229. [Google Scholar] [CrossRef]
- Nunes, M.R.; Karlen, D.L.; Moorman, T.B.; Cambardella, C.A. How Does Tillage Intensity Affect Chemical Soil Health Indicators? A United States Meta-Analysis. Agrosyst. Geosci. Environ. 2020, 3, e20083. [Google Scholar] [CrossRef]
- Linares, R.; de la Fuente, M.; Junquera, P.; Lissarrague, J.R.; Baeza, P. Effects of Soil Management in Vineyard on Soil Physical and Chemical Characteristics. BIO Web Conf. 2014, 3, 01008. [Google Scholar] [CrossRef]
- Nunes, M.R.; Karlen, D.L.; Veum, K.S.; Moorman, T.B.; Cambardella, C.A. Biological Soil Health Indicators Respond to Tillage Intensity: A US Meta-Analysis. Geoderma 2020, 369, 114335. [Google Scholar] [CrossRef]
- Badalíková, B. Influence of Soil Tillage on Soil Compaction. In Soil Biology; Dedousis, A., Bartzanas, T., Eds.; Springer: Volos, Greece, 2010; pp. 19–30. [Google Scholar]
- Saffih-Hdadi, K.; Défossez, P.; Richard, G.; Cui, Y.-J.; Tang, A.-M.; Chaplain, V. A Method for Predicting Soil Susceptibility to the Compaction of Surface Layers as a Function of Water Content and Bulk Density. Soil Tillage Res. 2009, 105, 96–103. [Google Scholar] [CrossRef]
- Ahmadi, I.; Ghaur, H. Effects of Soil Moisture Content and Tractor Wheeling Intensity on Traffic-Induced Soil Compaction. J. Cent. Eur. Agric. 2015, 16, 489–502. [Google Scholar] [CrossRef]
- Schjønning, P.; Lamandé, M.; Tøgersen, F.A.; Arvidsson, J.; Keller, T. Modelling Effects of Tyre Inflation Pressure on the Stress Distribution near the Soil–Tyre Interface. Biosyst. Eng. 2008, 99, 119–133. [Google Scholar] [CrossRef]
- Hamza, M.A.; Anderson, W.K. Soil Compaction in Cropping Systems: A Review of the Nature, Causes and Possible Solutions. Soil Tillage Res. 2005, 82, 121–145. [Google Scholar] [CrossRef]
- Van Dijck, S.J.E.; van Asch, T.W.J. Compaction of Loamy Soils Due to Tractor Traffic in Vineyards and Orchards and Its Effect on Infiltration in Southern France. Soil Tillage Res. 2002, 63, 141–153. [Google Scholar] [CrossRef]
- Pessina, D.; Galli, L.E.; Santoro, S.; Facchinetti, D. Sustainability of Machinery Traffic in Vineyard. Sustainability 2021, 13, 24745. [Google Scholar] [CrossRef]
- Elaoud, A.; Chehaibi, S. Soil Compaction Due to Tractor Traffic. J. Fail. Anal. Prev. 2011, 11, 539–545. [Google Scholar] [CrossRef]
- Lagacherie, P.; Coulouma, G.; Ariagno, P.; Virat, P.; Boizard, H.; Richard, G. Spatial Variability of Soil Compaction over a Vineyard Region in Relation with Soils and Cultivation Operations. Geoderma 2006, 134, 207–216. [Google Scholar] [CrossRef]
- Hunter, J.J.; Archer, E.; Van Schalkwyk, D.; Strever, A.E.; Volschenk, C.G. Grapevine Roots: Interaction with Natural Factors and Agronomic Practices. In Acta Horticulturae, Proceedings of the I International Symposium on Grapevine Roots, Chania, Greece, 30 July 2016; International Society for Horticultural Science: Leuven, Belgium, 2016; Volume 1136, pp. 63–80. [Google Scholar]
- Marinello, F.; Pezzuolo, A.; Cillis, D.; Chiumenti, A.; Sartori, L. Traffic Effects on Soil Compaction and Sugar Beet (Beta Vulgaris L.) Taproot Quality Parameters. Span. J. Agric. Res. 2017, 15, e0201. [Google Scholar] [CrossRef]
- Döring, J.; Friedel, M.; Hendgen, M.; Stoll, M.; Kauer, R. Soil Management in Sustainable Viticultural Systems. In Improving Sustainable Viticulture and Winemaking Practices; Elsevier: Amsterdam, The Netherlands, 2022; pp. 85–103. [Google Scholar]
- Burg, P.; Masan, V.; Zemanek, P. Evaluation of Effect of Vegetation Cover on Soil Compaction. In Proceedings of the Engineering for Rural Development, Jelgava, Latvia, 23–25 May 2017; Latvia University of Agriculture: Jelgava, Latvia, 2017; Volume 16, pp. 1346–1353. [Google Scholar]
- Visconti, F.; Jiménez, M.G.; de Paz, J.M. How Do the Chemical Characteristics of Organic Matter Explain Differences among Its Determinations in Calcareous Soils? Geoderma 2022, 406, 115454. [Google Scholar] [CrossRef]
- Hartwig, N.L.; Ammon, H.U. Cover Crops and Living Mulches. Weed Sci. 2002, 50, 688–699. [Google Scholar] [CrossRef]
- Chrysargyris, A.; Xylia, P.; Litskas, V.; Stavrinides, M.; Heyman, L.; Demeestere, K.; Höfte, M.; Tzortzakis, N. Assessing the Impact of Drought Stress and Soil Cultivation in Chardonnay and Xynisteri Grape Cultivars. Agronomy 2020, 10, 670. [Google Scholar] [CrossRef]
- Capri, C.; Gatti, M.; Fiorini, A.; Ardenti, F.; Tabaglio, V.; Poni, S. A Comparative Study of Fifteen Cover Crop Species for Orchard Soil Management: Water Uptake, Root Density Traits and Soil Aggregate Stability. Sci. Rep. 2023, 13, 721. [Google Scholar] [CrossRef]
- Zengin, H.; Sabir, A. Physiological and Growth Responses of Grapevine Rootstocks (Vitis Spp.) to Organic and Synthetic Mulch Application in Arid Ecology under the Effect of Climate Change. J. Cent. Eur. Agric. 2022, 23, 655–664. [Google Scholar] [CrossRef]
- Cabrera-Pérez, C.; Llorens, J.; Escolà, A.; Royo-Esnal, A.; Recasens, J. Organic Mulches as an Alternative for Under-Vine Weed Management in Mediterranean Irrigated Vineyards: Impact on Agronomic Performance. Eur. J. Agron. 2023, 145, 126798. [Google Scholar] [CrossRef]
- Marks, J.N.J.; Lines, T.E.P.; Penfold, C.; Cavagnaro, T.R. Cover Crops and Carbon Stocks: How under-Vine Management Influences SOC Inputs and Turnover in Two Vineyards. Sci. Total Environ. 2022, 831, 154800. [Google Scholar] [CrossRef] [PubMed]
- Gattullo, C.E.; Mezzapesa, G.N.; Stellacci, A.M.; Ferrara, G.; Occhiogrosso, G.; Petrelli, G.; Castellini, M.; Spagnuolo, M. Cover Crop for a Sustainable Viticulture: Effects on Soil Properties and Table Grape Production. Agronomy 2020, 10, 1334. [Google Scholar] [CrossRef]
- Salomé, C.; Coll, P.; Lardo, E.; Metay, A.; Villenave, C.; Marsden, C.; Blanchart, E.; Hinsinger, P.; Le Cadre, E. The Soil Quality Concept as a Framework to Assess Management Practices in Vulnerable Agroecosystems: A Case Study in Mediterranean Vineyards. Ecol. Indic. 2016, 61, 456–465. [Google Scholar] [CrossRef]
- Ferrara, G.; Nigro, D.; Torres, R.; Gadaleta, A.; Fidelibus, M.W.; Mazzeo, A. Cover Crops in the Inter-Row of a Table Grape Vineyard Managed with Irrigation Sensors: Effects on Yield, Quality and Glutamine Synthetase Activity in Leaves. Sci. Hortic. 2021, 281, 109963. [Google Scholar] [CrossRef]
- DeVetter, L.W.; Dilley, C.A.; Nonnecke, G.R. Mulches Reduce Weeds, Maintain Yield, and Promote Soil Quality in a Continental-Climate Vineyard. Am. J. Enol. Vitic. 2015, 66, 54–64. [Google Scholar] [CrossRef]
- Mania, E.; Isocrono, D.; Pedullà, M.L.; Guidoni, S. Plant Diversity in an Intensively Cultivated Vineyard Agro-Ecosystem (Langhe, North-West Italy). S. Afr. J. Enol. Vitic. 2015, 36, 378–388. [Google Scholar] [CrossRef]
- Eisenhauer, N. Plant Diversity Effects on Soil Microorganisms: Spatial and Temporal Heterogeneity of Plant Inputs Increase Soil Biodiversity. Pedobiologia 2016, 59, 175–177. [Google Scholar] [CrossRef]
- Chen, C.; Chen, H.Y.H.; Chen, X.; Huang, Z. Meta-Analysis Shows Positive Effects of Plant Diversity on Microbial Biomass and Respiration. Nat. Commun. 2019, 10, 1332. [Google Scholar] [CrossRef] [PubMed]
- Niles, M.T.; Garrett, R.D.; Walsh, D. Ecological and Economic Benefits of Integrating Sheep into Viticulture Production. Agron. Sustain. Dev. 2018, 38, 1. [Google Scholar] [CrossRef]
- Fermaud, M.; Smits, N.; Merot, A.; Roudet, J.; Thiéry, D.; Wery, J.; Delbac, L. New Multipest Damage Indicator to Assess Protection Strategies in Grapevine Cropping Systems. Aust. J. Grape Wine Res. 2016, 22, 450–461. [Google Scholar] [CrossRef]
- Daane, K.M.; Cooper, M.L.; Triapitsyn, S.V.; Walton, V.M.; Yokota, G.Y.; Haviland, D.R.; Bentley, W.J.; Godfrey, K.E.; Wunderlich, L.R. Vineyard Managers and Researchers Seek Sustainable Solutions for Mealybugs, a Changing Pest Complex. Calif. Agric. 2008, 62, 167–176. [Google Scholar] [CrossRef]
- Reineke, A.; Thiéry, D. Grapevine Insect Pests and Their Natural Enemies in the Age of Global Warming. J. Pest Sci. 2016, 89, 313–328. [Google Scholar] [CrossRef]
- Imperatore, G.; Ghirardelli, A.; Strinna, L.; Baldoin, C.; Pozzebon, A.; Zanin, G.; Otto, S. Evaluation of a Fixed Spraying System for Phytosanitary Treatments in Heroic Viticulture in North-Eastern Italy. Agriculture 2021, 11, 833. [Google Scholar] [CrossRef]
- European Commission Commission Regulation (EC). No 473/2002 of 15 March 2002 Amending Annexes I, II and VI to Council Regulation (EEC) No 2092/91 on Organic Production of Agricultural Products and Indications Referring Thereto on Agricultural Products and Foodstuffs, and Laying Down Detailed Rules as Regards the Transmission of Information on the Use of Copper Compounds. Available online: https://eur-lex.europa.eu/legal-content/EN/ALL/?uri=CELEX%3A32002R0473 (accessed on 6 December 2023).
- Unsworth, J. History of Pesticide Use; International Union of Pure and Applied Chemistry: Durham, NC, USA, 2010. [Google Scholar]
- Vocke, G. The Green Revolution for Wheat in Developing Countries; US Department of Agriculture: New York, NY, USA, 1986.
- Kogan, M. Integrated Pest Management: Historical Perspectives and Contemporary Developments. Annu. Rev. Entomol. 1998, 43, 243–270. [Google Scholar] [CrossRef]
- Barzman, M.; Bàrberi, P.; Birch, A.N.E.; Boonekamp, P.; Dachbrodt-Saaydeh, S.; Graf, B.; Hommel, B.; Jensen, J.E.; Kiss, J.; Kudsk, P.; et al. Eight Principles of Integrated Pest Management. Agron. Sustain. Dev. 2015, 35, 1199–1215. [Google Scholar] [CrossRef]
- Gregoire, C.; Payraudeau, S.; Domange, N. Use and Fate of 17 Pesticides Applied on a Vineyard Catchment. Int. J. Environ. Anal. Chem. 2010, 90, 406–420. [Google Scholar] [CrossRef]
- Cabras, P.; Meloni, M.; Pirisi, F.M. Pesticide Fate from Vine to Wine. Rev. Environ. Contam. Toxicol. 1987, 99, 83–117. [Google Scholar] [CrossRef] [PubMed]
- Pertot, I.; Caffi, T.; Rossi, V.; Mugnai, L.; Hoffmann, C.; Grando, M.S.; Gary, C.; Lafond, D.; Duso, C.; Thiery, D.; et al. A Critical Review of Plant Protection Tools for Reducing Pesticide Use on Grapevine and New Perspectives for the Implementation of IPM in Viticulture. Crop Prot. 2017, 97, 70–84. [Google Scholar] [CrossRef]
- Kullaj, E.; Shahini, S.; Varaku, S.; Çakalli, M. Evaluation of the Efficacy for Reducing Copper Use against Downy Mildew Control in Organic Mediterranean Viticulture. Int. J. Pest. Manag. 2016, 63, 3–9. [Google Scholar] [CrossRef]
- Kraus, C.; Abou-Ammar, R.; Schubert, A.; Fischer, M. Warburgia Ugandensis Leaf and Bark Extracts: An Alternative to Copper as Fungicide against Downy Mildew in Organic Viticulture? Plants 2021, 10, 2765. [Google Scholar] [CrossRef] [PubMed]
- Vargas-Bernal, R.; Rodríguez-Miranda, E.; Herrera-Pérez, G.; Vargas-Bernal, R.; Rodríguez-Miranda, E.; Herrera-Pérez, G. Evolution and Expectations of Enzymatic Biosensors for Pesticides. In Pesticides-Advances in Chemical and Botanical Pesticides; IntechOpen: London, UK, 2012; ISBN 978-953-51-0680-7. [Google Scholar]
- Urruty, N.; Deveaud, T.; Guyomard, H.; Boiffin, J. Impacts of Agricultural Land Use Changes on Pesticide Use in French Agriculture. Eur. J. Agron. 2016, 80, 113–123. [Google Scholar] [CrossRef]
- Droz, B.; Payraudeau, S.; Rodríguez Martín, J.A.; Tóth, G.; Panagos, P.; Montanarella, L.; Borrelli, P.; Imfeld, G. Copper Content and Export in European Vineyard Soils Influenced by Climate and Soil Properties. Environ. Sci. Technol. 2021, 55, 7327–7334. [Google Scholar] [CrossRef] [PubMed]
- Dagostin, S.; Schärer, H.-J.; Pertot, I.; Tamm, L. Are There Alternatives to Copper for Controlling Grapevine Downy Mildew in Organic Viticulture? Crop Prot. 2011, 30, 776–788. [Google Scholar] [CrossRef]
- Lo Scalzo, R.; Fibiani, M.; Pietromarchi, P.; Mandalà, C.; La Torre, A. Effects of Different Fungicide Treatments on Grape, Must and Wine Quality. Commun. Agric. Appl. Biol. Sci. 2012, 77, 151–161. [Google Scholar]
- Thuerig, B.; James, E.E.; Schärer, H.; Langat, M.K.; Mulholland, D.A.; Treutwein, J.; Kleeberg, I.; Ludwig, M.; Jayarajah, P.; Giovannini, O.; et al. Reducing Copper Use in the Environment: The Use of Larixol and Larixyl Acetate to Treat Downy Mildew Caused by Plasmopara Viticola in Viticulture. Pest Manag. Sci. 2017, 74, 477–488. [Google Scholar] [CrossRef]
- Lamichhane, J.R.; Osdaghi, E.; Behlau, F.; Köhl, J.; Jones, J.B.; Aubertot, J.-N. Thirteen Decades of Antimicrobial Copper Compounds Applied in Agriculture. A Review. Agron. Sustain. Dev. 2018, 38, 28. [Google Scholar] [CrossRef]
- Widmer, J.; Norgrove, L. Identifying Candidates for the Phytoremediation of Copper in Viticultural Soils: A Systematic Review. Environ. Res. 2023, 216, 114518. [Google Scholar] [CrossRef]
- Ballabio, C.; Panagos, P.; Lugato, E.; Huang, J.-H.; Orgiazzi, A.; Jones, A.; Fernández-Ugalde, O.; Borrelli, P.; Montanarella, L. Copper Distribution in European Topsoils: An Assessment Based on LUCAS Soil Survey. Sci. Total Environ. 2018, 636, 282–298. [Google Scholar] [CrossRef] [PubMed]
- New South Wales Environment Protection Agency. Environmental Management Guidelines—Use and Disposal of Biosolids Products; New South Wales Environment Protection Agency: Parramatta, Australia, 1997.
- Voluntary Risk Assessment Reports-Copper and Copper Compounds-ECHA. Available online: https://echa.europa.eu/es/copper-voluntary-risk-assessment-reports (accessed on 6 December 2023).
- Sandmann, G.; Böger, P. The Enzymatological Function of Heavy Metals and Their Role in Electron Transfer Processes of Plants. In Encyclopedia of Plant Physiology; Lauchli, A., Bieleski, R.L., Eds.; Springer: Berlin/Heidelberg, Germany, 1983; pp. 563–596. [Google Scholar]
- Solomon, E.I.; Heppner, D.E.; Johnston, E.M.; Ginsbach, J.W.; Cirera, J.; Qayyum, M.; Kieber-Emmons, M.T.; Kjaergaard, C.H.; Hadt, R.G.; Tian, L. Copper Active Sites in Biology. Chem. Rev. 2014, 114, 3659–3853. [Google Scholar] [CrossRef]
- Yruela, I. Copper in Plants. Braz. J. Plant Physiol. 2005, 17, 145–156. [Google Scholar] [CrossRef]
- Broadley, M.; Brown, P.; Cakmak, I.; Rengel, Z.; Zhao, F. Function of Nutrients: Micronutrients. In Marschner’s Nutrition of Higher Plants, 3rd ed.; Academic Press: Cambridge, MA, USA, 2012; pp. 191–248. [Google Scholar] [CrossRef]
- Gaetke, L. Copper Toxicity, Oxidative Stress, and Antioxidant Nutrients. Toxicology 2003, 189, 147–163. [Google Scholar] [CrossRef] [PubMed]
- Borkow, G.; Gabbay, J. Copper, An Ancient Remedy Returning to Fight Microbial, Fungal and Viral Infections. Curr. Chem. Biol. 2009, 3, 272–278. [Google Scholar] [CrossRef]
- Wairich, A.; De Conti, L.; Lamb, T.I.; Keil, R.; Neves, L.O.; Brunetto, G.; Sperotto, R.A.; Ricachenevsky, F.K. Throwing Copper Around: How Plants Control Uptake, Distribution, and Accumulation of Copper. Agronomy 2022, 12, 994. [Google Scholar] [CrossRef]
- Yang, X.; Lu, M.; Wang, Y.; Wang, Y.; Liu, Z.; Chen, S. Response Mechanism of Plants to Drought Stress. Horticulturae 2021, 7, 50. [Google Scholar] [CrossRef]
- Fernandes, J.C.; Henriques, F.S. Biochemical, Physiological, and Structural Effects of Excess Copper in Plants. Bot. Rev. 1991, 57, 246–273. [Google Scholar] [CrossRef]
- Babu, T.S.; Akhtar, T.A.; Lampi, M.A.; Tripuranthakam, S.; Dixon, D.G.; Greenberg, B.M. Similar Stress Responses Are Elicited by Copper and Ultraviolet Radiation in the Aquatic Plant Lemna Gibba: Implication of Reactive Oxygen Species as Common Signals. Plant Cell Physiol. 2003, 44, 1320–1329. [Google Scholar] [CrossRef]
- Sheldon, A.R.; Menzies, N.W. The Effect of Copper Toxicity on the Growth and Root Morphology of Rhodes Grass (Chloris gayana Knuth.) in Resin Buffered Solution Culture. Plant Soil 2005, 278, 341–349. [Google Scholar] [CrossRef]
- Lequeux, H.; Hermans, C.; Lutts, S.; Verbruggen, N. Response to Copper Excess in Arabidopsis thaliana: Impact on the Root System Architecture, Hormone Distribution, Lignin Accumulation and Mineral Profile. Plant Physiol. Biochem. 2010, 48, 673–682. [Google Scholar] [CrossRef]
- Pérez-Rodríguez, P.; Soto-Gómez, D.; De La Calle, I.; López-Periago, J.E.; Paradelo, M. Rainfall-Induced Removal of Copper-Based Spray Residues from Vines. Ecotoxicol. Environ. Saf. 2016, 132, 304–310. [Google Scholar] [CrossRef]
- Hummes, A.P.; Bortoluzzi, E.C.; Tonini, V.; da Silva, L.P.; Petry, C. Transfer of Copper and Zinc from Soil to Grapevine-Derived Products in Young and Centenarian Vineyards. Water Air Soil Pollut. 2019, 230, 150. [Google Scholar] [CrossRef]
- Zhao, F.; Rooney, C.P.; Zhang, H.; McGrath, S.P. Comparison of Soil Solution Speciation and Diffusive Gradients in Thin-films Measurement as an Indicator of Copper Bioavailability to Plants. Environ. Toxicol. Chem. 2006, 25, 733–742. [Google Scholar] [CrossRef]
- Li, L.; Wu, H.; van Gestel, C.A.M.; Peijnenburg, W.J.G.M.; Allen, H.E. Soil Acidification Increases Metal Extractability and Bioavailability in Old Orchard Soils of Northeast Jiaodong Peninsula in China. Environ. Pollut. 2014, 188, 144–152. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.Y.; Zuo, Y.B.; Wang, B.R.; Li, J.M.; Ma, Y.B. Toxicity and Accumulation of Copper and Nickel in Maize Plants Cropped on Calcareous and Acidic Field Soils. Plant Soil 2010, 333, 365–373. [Google Scholar] [CrossRef]
- Huang, J.-S.; Wei, D.-P.; Guo, X.-Y.; Ma, Y.-B. Toxicity and Accumulation of Copper and Nickel in Wheat Plants Cropped on Alkaline and Acidic Field Soils. Huan Jing Ke Xue/Environ. Sci. 2012, 33, 1369–1375. [Google Scholar]
- Trentin, E.; Cesco, S.; Pii, Y.; Valentinuzzi, F.; Celletti, S.; Feil, S.B.; Zuluaga, M.Y.A.; Ferreira, P.A.A.; Ricachenevsky, F.K.; Stefanello, L.O.; et al. Plant Species and PH Dependent Responses to Copper Toxicity. Environ. Exp. Bot. 2022, 196, 104791. [Google Scholar] [CrossRef]
- Miotto, A.; Ceretta, C.A.; Brunetto, G.; Nicoloso, F.T.; Girotto, E.; Farias, J.G.; Tiecher, T.L.; De Conti, L.; Trentin, G. Copper Uptake, Accumulation and Physiological Changes in Adult Grapevines in Response to Excess Copper in Soil. Plant Soil 2013, 374, 593–610. [Google Scholar] [CrossRef]
- Ambrosini, V.G.; Rosa, D.J.; Bastos de Melo, G.W.; Zalamena, J.; Cella, C.; Simão, D.G.; Souza da Silva, L.; Pessoa dos Santos, H.; Toselli, M.; Tiecher, T.L.; et al. High Copper Content in Vineyard Soils Promotes Modifications in Photosynthetic Parameters and Morphological Changes in the Root System of ‘Red Niagara’ Plantlets. Plant Physiol. Biochem. 2018, 128, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Feil, S.B.; Zuluaga, M.Y.A.; Cesco, S.; Pii, Y. Copper Toxicity Compromises Root Acquisition of Nitrate in the High Affinity Range. Front. Plant Sci. 2023, 13, 1034425. [Google Scholar] [CrossRef] [PubMed]
- Rosa, D.J.; Ambrosini, V.G.; Kokkoris, V.; Brunetto, G.; Hart, M.; Ricachenevsky, F.; Pescador, R. Lime Protection for Young Vines Exposed to Copper Toxicity. Water Air Soil Pollut. 2020, 231, 296. [Google Scholar] [CrossRef]
- Ma, Y.; Lombi, E.; Oliver, I.W.; Nolan, A.L.; McLaughlin, M.J. Long-Term Aging of Copper Added to Soils. Environ. Sci. Technol. 2006, 40, 6310–6317. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Hua, L.; Ma, Y. A Biotic Ligand Model Predicting Acute Copper Toxicity for Barley (Hordeum Vulgare): Influence of Calcium, Magnesium, Sodium, Potassium and PH. Chemosphere 2012, 89, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Stevenson, F.J.; Fitch, A. Reactions with Organic Matter. In Copper in Soils and Plants; Loneragan, J.F., Robson, A.D., Graham, R.D., Eds.; Academic Press: Sydney, Australia, 1981; pp. 265–285. [Google Scholar]
- Fernández-Calviño, D.; Nóvoa-Muñoz, J.C.; López-Periago, E.; Arias-Estévez, M. Changes in Copper Content and Distribution in Young, Old and Abandoned Vineyard Acid Soils Due to Land Use Changes. Land. Degrad. Dev. 2007, 19, 165–177. [Google Scholar] [CrossRef]
- Lagomarsino, A.; Marabottini, S.; Grego, S.; Stazi, S.R. Copper Distribution among Physical and Chemical Fractions in a Former Vineyard Soil. Agrochimica 2010, LIV, 1–12. [Google Scholar]
- Eijsackers, H.; Beneke, P.; Maboeta, M.; Louw, J.P.E.; Reinecke, A.J. The Implications of Copper Fungicide Usage in Vineyards for Earthworm Activity and Resulting Sustainable Soil Quality. Ecotoxicol. Environ. Saf. 2005, 62, 99–111. [Google Scholar] [CrossRef]
- Duan, X.; Xu, M.; Zhou, Y.; Yan, Z.; Du, Y.; Zhang, L.; Zhang, C.; Bai, L.; Nie, J.; Chen, G.; et al. Effects of Soil Properties on Copper Toxicity to Earthworm Eisenia Fetida in 15 Chinese Soils. Chemosphere 2016, 145, 185–192. [Google Scholar] [CrossRef]
- Qiu, H.; Vijver, M.G.; He, E.; Peijnenburg, W.J.G.M. Predicting Copper Toxicity to Different Earthworm Species Using a Multicomponent Freundlich Model. Environ. Sci. Technol. 2013, 47, 4796–4803. [Google Scholar] [CrossRef] [PubMed]
- Tiecher, T.L.; Tiecher, T.; Ceretta, C.A.; Ferreira, P.A.A.; Nicoloso, F.T.; Soriani, H.H.; De Conti, L.; Kulmann, M.S.S.; Schneider, R.O.; Brunetto, G. Tolerance and Translocation of Heavy Metals in Young Grapevine (Vitis Vinifera) Grown in Sandy Acidic Soil with Interaction of High Doses of Copper and Zinc. Sci. Hortic. 2017, 222, 203–212. [Google Scholar] [CrossRef]
- Shaw, J.L.A.; Ernakovich, J.G.; Judy, J.D.; Farrell, M.; Whatmuff, M.; Kirby, J. Long-Term Effects of Copper Exposure to Agricultural Soil Function and Microbial Community Structure at a Controlled and Experimental Field Site. Environ. Pollut. 2020, 263, 114411. [Google Scholar] [CrossRef] [PubMed]
- Ben Massoud, M.; Kharbech, O.; Sakouhi, L.; Ben Hassine, S.; Zhu, Y.; Chaoui, A.; Sheehan, D.; Djebali, W. Calcium and Citrate Protect Pisum Sativum Roots against Copper Toxicity by Regulating the Cellular Redox Status. J. Soil Sci. Plant Nutr. 2021, 22, 345–358. [Google Scholar] [CrossRef]
- Tomizawa, M.; Casida, J.E. Neonicotinoid Insecticides: Highlights of a Symposium on Strategic Molecular Designs. J. Agric. Food Chem. 2010, 59, 2883–2886. [Google Scholar] [CrossRef]
- Zaller, J.G.; Cantelmo, C.; Dos Santos, G.; Muther, S.; Gruber, E.; Pallua, P.; Mandl, K.; Friedrich, B.; Hofstetter, I.; Schmuckenschlager, B.; et al. Herbicides in Vineyards Reduce Grapevine Root Mycorrhization and Alter Soil Microorganisms and the Nutrient Composition in Grapevine Roots, Leaves, Xylem Sap and Grape Juice. Environ. Sci. Pollut. Res. Int. 2018, 25, 23215–23226. [Google Scholar] [CrossRef] [PubMed]
- Stellin, F.; Gavinelli, F.; Stevanato, P.; Concheri, G.; Squartini, A.; Paoletti, M.G. Effects of Different Concentrations of Glyphosate (Roundup 360®) on Earthworms (Octodrilus Complanatus, Lumbricus Terrestris and Aporrectodea Caliginosa) in Vineyards in the North-East of Italy. Appl. Soil Ecol. 2018, 123, 802–808. [Google Scholar] [CrossRef]
- Albers, C.N.; Banta, G.T.; Hansen, P.E.; Jacobsen, O.S. The Influence of Organic Matter on Sorption and Fate of Glyphosate in Soil–Comparing Different Soils and Humic Substances. Environ. Pollut. 2009, 157, 2865–2870. [Google Scholar] [CrossRef]
- Gros, P.; Ahmed, A.; Kühn, O.; Leinweber, P. Glyphosate Binding in Soil as Revealed by Sorption Experiments and Quantum-Chemical Modeling. Sci. Total Environ. 2017, 586, 527–535. [Google Scholar] [CrossRef] [PubMed]
- Massaccesi, L.; Cartoni Mancinelli, A.; Mattioli, S.; De Feudis, M.; Castellini, C.; Dal Bosco, A.; Marongiu, M.L.; Agnelli, A. Geese Reared in Vineyard: Soil, Grass and Animals Interaction. Animals 2019, 9, 179. [Google Scholar] [CrossRef]
- Gonçalves, P.; Nóbrega, L.; Monteiro, A.; Pedreiras, P.; Rodrigues, P.; Esteves, F. SheepIT, an E-Shepherd System for Weed Control in Vineyards: Experimental Results and Lessons Learned. Animals 2021, 11, 2625. [Google Scholar] [CrossRef]
- Trouillard, M.; Lèbre, A.; Heckendorn, F. Grazing Sheep in Organic Vineyards: An On-Farm Study about Risk of Chronic Copper Poisoning. Sustainability 2021, 13, 12860. [Google Scholar] [CrossRef]
- O’Brien, R.D. Toxic Phosphorous Esters: Chemistry, Metabolism, and Biological Effects; Elsevier: New York, NY, USA, 2016. [Google Scholar]
- Kushwaha, M.; Verma, S.; Chatterjee, S. Profenofos, an Acetylcholinesterase-Inhibiting Organophosphorus Pesticide: A Short Review of Its Usage, Toxicity, and Biodegradation. J. Environ. Qual. 2016, 45, 1478–1489. [Google Scholar] [CrossRef]
- Booth, L.H.; Hodge, S.; O’Halloran, K. Use of Biomarkers in Earthworms to Detect Use and Abuse of Field Applications of a Model Organophosphate Pesticide. Bull. Environ. Contam. Toxicol. 2001, 67, 633–640. [Google Scholar] [CrossRef]
- Reinecke, S.A.; Reinecke, A.J. The Impact of Organophosphate Pesticides in Orchards on Earthworms in the Western Cape, South Africa. Ecotoxicol. Environ. Saf. 2007, 66, 244–251. [Google Scholar] [CrossRef] [PubMed]
- Pelosi, C.; Barot, S.; Capowiez, Y.; Hedde, M.; Vandenbulcke, F. Pesticides and Earthworms. A Review. Agron. Sustain. Dev. 2013, 34, 199–228. [Google Scholar] [CrossRef]
- Mansour, R.; Belzunces, L.P.; Suma, P.; Zappalà, L.; Mazzeo, G.; Grissa-Lebdi, K.; Russo, A.; Biondi, A. Vine and Citrus Mealybug Pest Control Based on Synthetic Chemicals. A Review. Agron. Sustain. Dev. 2018, 38, 37. [Google Scholar] [CrossRef]
- Brück, E.; Elbert, A.; Fischer, R.; Krueger, S.; Kühnhold, J.; Klueken, A.M.; Nauen, R.; Niebes, J.-F.; Reckmann, U.; Schnorbach, H.-J.; et al. Movento®, an Innovative Ambimobile Insecticide for Sucking Insect Pest Control in Agriculture: Biological Profile and Field Performance. Crop Prot. 2009, 28, 838–844. [Google Scholar] [CrossRef]
- Pandey, S.; Singh, D.K. Total Bacterial and Fungal Population after Chlorpyrifos and Quinalphos Treatments in Groundnut (Arachis Hypogaea L.) Soil. Chemosphere 2004, 55, 197–205. [Google Scholar] [CrossRef]
- Chu, X.; Fang, H.; Pan, X.; Wang, X.; Shan, M.; Feng, B.; Yu, Y. Degradation of Chlorpyrifos Alone and in Combination with Chlorothalonil and Their Effects on Soil Microbial Populations. J. Environ. Sci. 2008, 20, 464–469. [Google Scholar] [CrossRef]
- Deutsch, C.A.; Tewksbury, J.J.; Tigchelaar, M.; Battisti, D.S.; Merrill, S.C.; Huey, R.B.; Naylor, R.L. Increase in Crop Losses to Insect Pests in a Warming Climate. Science 2018, 361, 916–919. [Google Scholar] [CrossRef]
- Bois, B.; Zito, S.; Calonnec, A.; Ollat, N. Climate vs Grapevine Pests and Diseases Worldwide: The First Results of a Global Survey. J. Int. Sci. Vigne Vin 2017, 51, 133–139. [Google Scholar]
- Daane, K.M.; Vincent, C.; Isaacs, R.; Ioriatti, C. Entomological Opportunities and Challenges for Sustainable Viticulture in a Global Market. Annu. Rev. Entomol. 2018, 63, 193–214. [Google Scholar] [CrossRef]
- Lodovica Gullino, M.; Garibaldi, A.; Gamliel, A.; Katan, J. Soil Disinfestation: From Soil Treatment to Soil and Plant Health. Plant Dis. 2022, 106, 1541–1554. [Google Scholar] [CrossRef]
- Husson, O.; Sarthou, J.-P.; Bousset, L.; Ratnadass, A.; Schmidt, H.-P.; Kempf, J.; Husson, B.; Tingry, S.; Aubertot, J.-N.; Deguine, J.-P.; et al. Soil and Plant Health in Relation to Dynamic Sustainment of Eh and PH Homeostasis: A Review. Plant Soil 2021, 466, 391–447. [Google Scholar] [CrossRef]
- Akanmu, A.O.; Babalola, O.O.; Venturi, V.; Ayilara, M.S.; Adeleke, B.S.; Amoo, A.E.; Sobowale, A.A.; Fadiji, A.E.; Glick, B.R. Plant Disease Management: Leveraging on the Plant-Microbe-Soil Interface in the Biorational Use of Organic Amendments. Front. Plant Sci. 2021, 12, 700507. [Google Scholar] [CrossRef] [PubMed]
- Davis, L. Soil Health, Plant Health and Pests. In Building Soils for Better Crops; Magdoff, F., Es, H., Eds.; Sustainable Agriculture Research and Education (SARE): New York, NY, USA, 2021; pp. 103–116. [Google Scholar]
- Mpelasoka, B.; Schachtman, D.; Treeby, M.; Thomas, M. A Review of Potassium Nutrition in Grapevines with Special Emphasis on Berry Accumulation. Aust. J. Grape Wine Res. 2003, 9, 154–168. [Google Scholar] [CrossRef]
- James, A.; Mahinda, A.; Mwamahonje, A.; Rweyemamu, E.W.; Mrema, E.; Aloys, K.; Swai, E.; Mpore, F.J.; Massawe, C. A Review on the Influence of Fertilizers Application on Grape Yield and Quality in the Tropics. J. Plant Nutr. 2022, 46, 2936–2957. [Google Scholar] [CrossRef]
- Skinner, P.W.; Ishii, R.; O’Mahony, M.; Matthews, M. Sensory Attributes of Wines Made from Vines of Differing Phosphorus Status. OENO One 2019, 53, 347–361. [Google Scholar] [CrossRef]
- Spayd, S.E.; Stevens, R.G.; Wample, R.L.; Evans, R.G.; Edwards, C.G.; Webster, D. Impact of Nitrogen Fertilization on Vine Performance and Juice and Wine Composition of “Riesling” Grapes (Vitis Vinifera L.) in Washington State. Acta Hortic. 2000, 512, 65–76. [Google Scholar] [CrossRef]
- Bell, S.-J.; Henschke, P.A. Implications of Nitrogen Nutrition for Grapes, Fermentation and Wine. Aust. J. Grape Wine Res. 2005, 11, 242–295. [Google Scholar] [CrossRef]
- Monder, H.; Maillard, M.; Chérel, I.; Zimmermann, S.D.; Paris, N.; Cuéllar, T.; Gaillard, I. Adjustment of K(+) Fluxes and Grapevine Defense in the Face of Climate Change. Int. J. Mol. Sci. 2021, 22, 10398. [Google Scholar] [CrossRef] [PubMed]
- Krasilnikov, P.; Taboada, M.A. Amanullah Fertilizer Use, Soil Health and Agricultural Sustainability. Agriculture 2022, 12, 462. [Google Scholar] [CrossRef]
- Montoroi, J.-P. Soil Degradation by Salinization. In Ecology and Environmental Science: Reduction of Water Stress and Adaptation to Aridity: Proceedings of the Third Ecoscience Workshop; Gawronski, K., Ziernicka-Wojtaszek, A., Ksiazek, L., Nizinski, G., Eds.; Wydawnictwo Uniwersytetu Rolniczego im. H. Kołłątaja: Cracovie, Poland, 2017; pp. 104–106. [Google Scholar]
- Machado, R.M.A.; Serralheiro, R.P. Soil Salinity: Effect on Vegetable Crop Growth. Management Practices to Prevent and Mitigate Soil Salinization. Horticulturae 2017, 3, 30. [Google Scholar] [CrossRef]
- Arnó, J.; Rosell, J.R.; Blanco, R.; Ramos, M.C.; Martínez-Casasnovas, J.A. Spatial Variability in Grape Yield and Quality Influenced by Soil and Crop Nutrition Characteristics. Precis. Agric. 2011, 13, 393–410. [Google Scholar] [CrossRef]
- Lacombe, B. Increasing Our Knowledge on Grapevines Physiology to Increase Yield, Quality and Sustainably. Physiol. Plant 2022, 174, e13664. [Google Scholar] [CrossRef] [PubMed]
- Lambert, J.-J.; Anderson, M.M.; Wolpert, J.A. Vineyard Nutrient Needs Vary with Rootstocks and Soils. Calif. Agric. 2008, 62, 202–207. [Google Scholar] [CrossRef]
- Maeder, P.; Fliessbach, A.; Dubois, D.; Gunst, L.; Fried, P.; Niggli, U. Soil Fertility and Biodiversity in Organic Farming. Science 2002, 296, 1694–1697. [Google Scholar] [CrossRef]
- Tully, K.L.; McAskill, C. Promoting Soil Health in Organically Managed Systems: A Review. Org. Agric. 2020, 10, 339–358. [Google Scholar] [CrossRef]
- Provost, C.; Pedneault, K. The Organic Vineyard as a Balanced Ecosystem: Improved Organic Grape Management and Impacts on Wine Quality. Sci. Hortic. 2016, 208, 43–56. [Google Scholar] [CrossRef]
- Borsato, E.; Zucchinelli, M.; D’Ammaro, D.; Giubilato, E.; Zabeo, A.; Criscione, P.; Pizzol, L.; Cohen, Y.; Tarolli, P.; Lamastra, L.; et al. Use of Multiple Indicators to Compare Sustainability Performance of Organic vs Conventional Vineyard Management. Sci. Total Environ. 2020, 711, 135081. [Google Scholar] [CrossRef]
- Tangolar, S.; Tangolar, S.; Torun, A.A.; Ada, M.; Göçmez, S. Influence of Supplementation of Vineyard Soil with Organic Substances on Nutritional Status, Yield and Quality of “Black Magic” Grape (Vitis Vinifera L.) and Soil Microbiological and Biochemical Characteristics. OENO One 2020, 54, 1143–1157. [Google Scholar] [CrossRef]
- Di Giacinto, S.; Friedel, M.; Poll, C.; Döring, J.; Kunz, R.; Kauer, R. Vineyard Management System Affects Soil Microbiological Properties. OENO One 2020, 54, 131–143. [Google Scholar] [CrossRef]
- Cernansky, R. Agriculture: State-of-the-Art Soil. Nature 2015, 517, 258–260. [Google Scholar] [CrossRef]
- Omondi, M.O.; Xia, X.; Nahayo, A.; Liu, X.; Korai, P.K.; Pan, G. Quantification of Biochar Effects on Soil Hydrological Properties Using Meta-Analysis of Literature Data. Geoderma 2016, 274, 28–34. [Google Scholar] [CrossRef]
- Razzaghi, F.; Obour, P.B.; Arthur, E. Does Biochar Improve Soil Water Retention? A Systematic Review and Meta-Analysis. Geoderma 2020, 361, 114055. [Google Scholar] [CrossRef]
- Shaheen, S.M.; Mosa, A.; Natasha; Arockiam Jeyasundar, P.G.S.; Hassan, N.E.E.; Yang, X.; Antoniadis, V.; Li, R.; Wang, J.; Zhang, T.; et al. Pros and Cons of Biochar to Soil Potentially Toxic Element Mobilization and Phytoavailability: Environmental Implications. Earth Syst. Environ. 2023, 7, 321–345. [Google Scholar] [CrossRef]
- Baronti, S.; Magno, R.; Maienza, A.; Montagnoli, A.; Ungaro, F.; Vaccari, F.P. Long Term Effect of Biochar on Soil Plant Water Relation and Fine Roots: Results after 10 Years of Vineyard Experiment. Sci. Total Environ. 2022, 851, 158225. [Google Scholar] [CrossRef]
- Genesio, L.; Miglietta, F.; Baronti, S.; Vaccari, F.P. Biochar Increases Vineyard Productivity without Affecting Grape Quality: Results from a Four Years Field Experiment in Tuscany. Agric. Ecosyst. Environ. 2015, 201, 20–25. [Google Scholar] [CrossRef]
- Mortvedt, J.J. Heavy Metal Contaminants in Inorganic and Organic Fertilizers; Springer: Cham, Switzerland, 1996; Volume 43. [Google Scholar]
- Roberts, D.; Nachtegaal, M.; Sparks, D.L. Speciation of Metals in Soils. In Chemical Processes in Soils; Tabatabai, M.A., Sparks, D.L., Eds.; Soil Science Society of America: Madison, WI, USA, 2005; pp. 619–654. [Google Scholar]
- Yao, L.-X.; Li, G.-L.; Tu, S.-H.; Gavin, S.; He, Z.-H. Salinity of Animal Manure and Potential Risk of Secondary Soil Salinization through Successive Manure Application. Sci. Total Environ. 2007, 383, 106–114. [Google Scholar] [CrossRef]
- Olego, M.A.; Visconti, F.; Quiroga, M.J.; de Paz, J.M.; Garzón-Jimeno, E. Assessing the Effects of Soil Liming with Dolomitic Limestone and Sugar Foam on Soil Acidity, Leaf Nutrient Contents, Grape Yield and Must Quality in a Mediterranean Vineyard. Span. J. Agric. Res. 2016, 14, e1102. [Google Scholar] [CrossRef]
- Foy, C.D. Physiological Effects of Hydrogen, Aluminum, and Manganese Toxicities in Acid Soil. Soil Acidity Liming 2015, 12, 57–97. [Google Scholar] [CrossRef]
- Hue, N. Soil Acidity: Development, Impacts, and Management. In Structure and Functions of Pedosphere; Springer Nature: Cham, Switzerland, 2022; pp. 103–131. ISBN 9789811687709. [Google Scholar]
- Skinner, P.W.; Matthews, M.A. Reproductive Development in Grape (Vitis Vinifera L.) under Phosphorus-Limited Conditions. Sci. Hortic. 1989, 38, 49–60. [Google Scholar] [CrossRef]
- Čoga, L.; Slunjski, S.; Herak Ćustić, M.; Maslać, J.; Petek, M.; Ćosić, T.; Pavlović, I. Influence of Soil Reaction on Phosphorus, Potassium, Calcium and Magnesium Dynamics in Grapevine (Vitis Vinifera L.). Agric. Conspec. Sci. 2009, 74, 39–43. [Google Scholar]
- Quiroga, M.J.; Olego, M.Á.; Sánchez-García, M.; Medina, J.E.; Visconti, F.; Coque, J.J.R.; Garzón Jimeno, J.E. Effects of Liming on Soil Properties, Leaf Tissue Cation Composition and Grape Yield in a Moderately Acid Vineyard Soil. Influence on Must and Wine Quality. OENO One 2017, 51, 343. [Google Scholar] [CrossRef]
- Tang, C. Causes and Management of Subsoil Acidity. In Proceedings of the SuperSoil 2004: 3rd Australian New Zealand Soils Conference, Camperdown, NSW, Australia, 5 December 2004; University of Sydney: Camperdown, Sydney, NSW, Australia, 2004; pp. 1–7. [Google Scholar]
- Grapevine Rootstock Selector Tool—Rootstock Selector. Available online: https://grapevinerootstock.com/ (accessed on 31 January 2024).
- Van Leeuwen, C.; de Rességuier, L. Major Soil-Related Factors in Terroir Expression and Vineyard Siting. Elements 2018, 14, 159–165. [Google Scholar] [CrossRef]
- Ferretti, C.G. Relationship between the Geology, Soil Assessment, and Terroir of Gewürtztraminer Vineyards: A Case Study in the Dolomites of Northern Italy. Catena 2019, 179, 74–84. [Google Scholar] [CrossRef]
- Jones, G.V. Grapevines in a Changing Environment. In Grapevine in a Changing Environment; Gerós, H., Chaves, M., Medrano Gil, H., Delrot, S., Eds.; Wiley: Hoboken, NJ, USA, 2015; pp. 1–17. [Google Scholar]
- Gobbi, A.; Acedo, A.; Imam, N.; Santini, R.G.; Ortiz-Álvarez, R.; Ellegaard-Jensen, L.; Belda, I.; Hansen, L.H. A Global Microbiome Survey of Vineyard Soils Highlights the Microbial Dimension of Viticultural Terroirs. Commun. Biol. 2022, 5, 241. [Google Scholar] [CrossRef]
- Bonfante, A.; Brillante, L. Terroir Analysis and Its Complexity. OENO One 2022, 56, 375–388. [Google Scholar] [CrossRef]
- Reynard, J.-S.; Zufferey, V.; Nicol, G.-C.; Murisier, F. Vine Water Status as a Parameter of the « terroir » Effect under the Non-Irrigated Conditions of the Vaud Viticultural Area (Switzerland). OENO One 2011, 45, 139. [Google Scholar] [CrossRef]
- Jones, G.V. The Climate Component of Terroir. Elements 2018, 14, 167–172. [Google Scholar] [CrossRef]
- Van Leeuwen, C. Soils and Terroir Expression in Wines. In Soil and Culture; Springer Netherlands: Dordrecht, The Netherlands, 2010; pp. 453–465. [Google Scholar]
- Bohmrich, R. Terroir: Competing Perspectives on the Roles of Soil, Climate and People. J. Wine Res. 1996, 7, 33–46. [Google Scholar] [CrossRef]
- Certini, G.; Scalenghe, R. The Crucial Interactions between Climate and Soil. Sci. Total Environ. 2023, 856, 159169. [Google Scholar] [CrossRef]
- Coli, M.S.; Rangel, A.G.P.; Souza, E.S.; Oliveira, M.F.; Chiaradia, A.C.N. Chloride Concentration in Red Wines: Influence of Terroir and Grape Type. Food Sci. Technol. 2015, 35, 95–99. [Google Scholar] [CrossRef]
- Zhou-Tsang, A.; Wu, Y.; Henderson, S.W.; Walker, A.R.; Borneman, A.R.; Walker, R.R.; Gilliham, M. Grapevine Salt Tolerance. Aust. J. Grape Wine Res. 2021, 27, 149–168. [Google Scholar] [CrossRef]
- Wheeler, S.; Pickering, G. Effects of Soil Management Techniques on Grape and Wine Quality. In Fruits: Growth, Nutrition and Quality; Dris, R., Ed.; Wfl: Helsinki, Finland, 2005; pp. 195–208. [Google Scholar]
- Tomasi, D.; Marcuzzo, P.; Nardi, T.; Lonardi, A.; Lovat, L.; Flamini, R.; Mian, G. Influence of Soil Chemical Features on Aromatic Profile of V. vinifera Cv. Corvina Grapes and Wines: A Study-Case in Valpolicella Area (Italy) in a Calcareous and Non-Calcareous Soil. Agriculture 2022, 12, 1980. [Google Scholar] [CrossRef]
- Coller, E.; Cestaro, A.; Zanzotti, R.; Bertoldi, D.; Pindo, M.; Larger, S.; Albanese, D.; Mescalchin, E.; Donati, C. Microbiome of Vineyard Soils Is Shaped by Geography and Management. Microbiome 2019, 7, 140. [Google Scholar] [CrossRef] [PubMed]
- Nanetti, E.; Palladino, G.; Scicchitano, D.; Trapella, G.; Cinti, N.; Fabbrini, M.; Cozzi, A.; Accetta, G.; Tassini, C.; Iannaccone, L.; et al. Composition and Biodiversity of Soil and Root-Associated Microbiome in Vitis vinifera Cultivar Lambrusco Distinguish the Microbial Terroir of the Lambrusco DOC Protected Designation of Origin Area on a Local Scale. Front. Microbiol. 2023, 14, 1108036. [Google Scholar] [CrossRef]
- Gilbert, J.A.; van der Lelie, D.; Zarraonaindia, I. Microbial Terroir for Wine Grapes. Proc. Natl. Acad. Sci. USA 2014, 111, 5–6. [Google Scholar] [CrossRef]
- Capozzi, V.; Garofalo, C.; Chiriatti, M.A.; Grieco, F.; Spano, G. Microbial Terroir and Food Innovation: The Case of Yeast Biodiversity in Wine. Microbiol. Res. 2015, 181, 75–83. [Google Scholar] [CrossRef]
- Vink, S.N.; Dini-Andreote, F.; Höfle, R.; Kicherer, A.; Salles, J.F. Interactive Effects of Scion and Rootstock Genotypes on the Root Microbiome of Grapevines (Vitis Spp. L.). Appl. Sci. 2021, 11, 1615. [Google Scholar] [CrossRef]
- Qi, Y.; Wang, R.; Qin, Q.; Sun, Q. Soil Affected the Variations in Grape and Wine Properties along the Eastern Foot of Helan Mountain, China. Acta Agric. Scand. B Soil Plant Sci. 2019, 69, 494–502. [Google Scholar] [CrossRef]
- Vaudour, E.; Shaw, A. A Worldwide Perspective on Viticultural Zoning. S. Afr. J. Enol. Vitic. 2005, 26, 106–116. [Google Scholar] [CrossRef][Green Version]
- Villat, J. The Roots of a Better Future: Regenerative Viticulture. Available online: https://portoprotocol.com/the-roots-of-a-better-future-regenerative-viticulture/ (accessed on 11 December 2023).
- Bezuidenhout, C.N.; van Antwerpen, R.; Berry, S.D. An Application of Principal Component Analyses and Correlation Graphs to Assess Multivariate Soil Health Properties. Soil Sci. 2012, 177, 498–505. [Google Scholar] [CrossRef]
- Smith, J.L.; Halvorson, J.J.; Papendick, R.I. Using Multiple-Variable Indicator Kriging for Evaluating Soil Quality. Soil Sci. Soc. Am. J. 1993, 57, 743–749. [Google Scholar] [CrossRef]
- Andrews, S.S.; Karlen, D.L.; Cambardella, C.A. The Soil Management Assessment Framework. Soil Sci. Soc. Am. J. 2004, 68, 1945–1962. [Google Scholar] [CrossRef]
- Zornoza, R.; Acosta, J.A.; Bastida, F.; Domínguez, S.G.; Toledo, D.M.; Faz, A. Identification of Sensitive Indicators to Assess the Interrelationship between Soil Quality, Management Practices and Human Health. Soil 2015, 1, 173–185. [Google Scholar] [CrossRef]
- Moebius-Clune, B.N.; Moebius-Clune, D.J.; Gugino, B.K.; Idowu, O.J.; Schindelbeck, R.R.; Ristow, A.J.; Van Es, H.M.; Thies, J.E.; Shayler, H.A.; Mcbride, M.B.; et al. Comprehensive Assessment of Soil Health—The Cornell Framework; 3.2; Cornell University: Geneva, NY, USA, 2016; ISBN 0967650763. [Google Scholar]
- Alkorta, I.; Aizpurua, A.; Riga, P.; Albizu, I.; Amézaga, I.; Garbisu, C. Soil Enzyme Activities as Biological Indicators of Soil Health. Rev. Environ. Health 2003, 18, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Rochfort, S.J.; Ezernieks, V.; Yen, A.L. NMR-Based Metabolomics Using Earthworms as Potential Indicators for Soil Health. Metabolomics 2008, 5, 95–107. [Google Scholar] [CrossRef]
- Chae, Y.; Cui, R.; Woong Kim, S.; An, G.; Jeong, S.-W.; An, Y.-J. Exoenzyme Activity in Contaminated Soils before and after Soil Washing: SS-Glucosidase Activity as a Biological Indicator of Soil Health. Ecotoxicol. Environ. Saf. 2017, 135, 368–374. [Google Scholar] [CrossRef]
- Madejon, E.; Moreno, F.; Murillo, J.; Pelegrin, F. Soil Biochemical Response to Long-Term Conservation Tillage under Semi-Arid Mediterranean Conditions. Soil Tillage Res. 2007, 94, 346–352. [Google Scholar] [CrossRef]
- Gong, W.; Yan, X.; Wang, J.; Hu, T.; Gong, Y. Long-Term Manure and Fertilizer Effects on Soil Organic Matter Fractions and Microbes under a Wheat–Maize Cropping System in Northern China. Geoderma 2009, 149, 318–324. [Google Scholar] [CrossRef]
- Niemeyer, J.C.; Lolata, G.B.; de Carvalho, G.M.; Da Silva, E.M.; Sousa, J.P.; Nogueira, M.A. Microbial Indicators of Soil Health as Tools for Ecological Risk Assessment of a Metal Contaminated Site in Brazil. Appl. Soil Ecol. 2012, 59, 96–105. [Google Scholar] [CrossRef]
- Norkaew, S.; Miles, R.J.; Brandt, D.K.; Anderson, S.H. Effects of 130 Years of Selected Cropping Management Systems on Soil Health Properties for Sanborn Field. Soil Sci. Soc. Am. J. 2019, 83, 1479–1490. [Google Scholar] [CrossRef]
- Caudle, C.; Osmond, D.; Heitman, J.; Ricker, M.; Miller, G.; Wills, S. Comparison of Soil Health Metrics for a Cecil Soil in the North Carolina Piedmont. Soil Sci. Soc. Am. J. 2020, 84, 978–993. [Google Scholar] [CrossRef]
- Ye, R.; Parajuli, B.; Szogi, A.A.; Sigua, G.C.; Ducey, T.F. Soil Health Assessment after 40 Years of Conservation and Conventional Tillage Management in Southeastern Coastal Plain Soils. Soil Sci. Soc. Am. J. 2021, 85, 1214–1225. [Google Scholar] [CrossRef]
- Fine, A.K.; van Es, H.M.; Schindelbeck, R.R. Statistics, Scoring Functions, and Regional Analysis of a Comprehensive Soil Health Database. Soil Sci. Soc. Am. J. 2017, 81, 589–601. [Google Scholar] [CrossRef]
- Liptzin, D.; Norris, C.E.; Cappellazzi, S.B.; Bean, G.M.; Cope, M.; Greub, K.L.H.; Rieke, E.L.; Tracy, P.W.; Aberle, E.; Ashworth, A.; et al. An Evaluation of Carbon Indicators of Soil Health in Long-Term Agricultural Experiments. Soil Biol. Biochem. 2022, 172, 108708. [Google Scholar] [CrossRef]
- Katayama, A.; Bhula, R.; Burns, G.R.; Carazo, E.; Felsot, A.; Hamilton, D.; Harris, C.; Kim, Y.-H.; Kleter, G.; Koedel, W.; et al. Bioavailability of Xenobiotics in the Soil Environment. In Reviews of Environmental Contamination and Toxicology; Springer: Berlin/Heidelberg, Germany, 2010; pp. 1–86. [Google Scholar]
- Sojka, R.E.; Upchurch, D.R. Reservations Regarding the Soil Quality Concept. Soil Sci. Soc. Am. J. 1999, 63, 1039–1054. [Google Scholar] [CrossRef]
- Chamberlain, L.A.; Aguayo, T.; Zerega, N.J.C.; Dybzinski, R.; Egerton-Warburton, L.M. Rapid Improvement in Soil Health Following the Conversion of Abandoned Farm Fields to Annual or Perennial Agroecosystems. Front. Sustain. Food Syst. 2022, 6, 1010298. [Google Scholar] [CrossRef]
- Barthès, B.; Roose, E. Aggregate Stability as an Indicator of Soil Susceptibility to Runoff and Erosion; Validation at Several Levels. Catena 2002, 47, 133–149. [Google Scholar] [CrossRef]
- Kemper, W.D.; Rosenau, R.C. Aggregate Stability and Size Distribution. Methods of Soil Analysis, Part 1. In Physical and Mineralogical Methods; Klute, A., Ed.; SSSA: Madison, WI, USA, 2018; pp. 425–442. [Google Scholar]
- Amézketa, E. Soil Aggregate Stability: A Review. J. Sustain. Agric. 1999, 14, 83–151. [Google Scholar] [CrossRef]
- Rieke, E.L.; Bagnall, D.K.; Morgan, C.L.S.; Flynn, K.D.; Howe, J.A.; Greub, K.L.H.; Mac Bean, G.; Cappellazzi, S.B.; Cope, M.; Liptzin, D.; et al. Evaluation of Aggregate Stability Methods for Soil Health. Geoderma 2022, 428, 116156. [Google Scholar] [CrossRef]
- Suleiman, A.A.; Ritchie, J.T. Estimating Saturated Hydraulic Conductivity from Soil Porosity. Trans. ASAE 2001, 44, 235. [Google Scholar] [CrossRef]
- Li, Z.; Wu, P.; Feng, H.; Zhao, X.; Huang, J.; Zhuang, W. Simulated Experiment on Effect of Soil Bulk Density on Soil Infiltration Capacity. Nongye Gongcheng Xuebao/Trans. Chin. Soc. Agric. Eng. 2009, 25, 40–45. [Google Scholar]
- Burns, K.N.; Bokulich, N.A.; Cantu, D.; Greenhut, R.F.; Kluepfel, D.A.; O’Geen, A.T.; Strauss, S.L.; Steenwerth, K.L. Vineyard Soil Bacterial Diversity and Composition Revealed by 16S RRNA Genes: Differentiation by Vineyard Management. Soil Biol. Biochem. 2016, 103, 337–348. [Google Scholar] [CrossRef]
- Viers, J.H.; Williams, J.N.; Nicholas, K.A.; Barbosa, O.; Kotzé, I.; Spence, L.; Webb, L.B.; Merenlender, A.; Reynolds, M. Vinecology: Pairing Wine with Nature. Conserv. Lett. 2013, 6, 287–299. [Google Scholar] [CrossRef]
- Stoleru, V.; Liliana, R.; Mihai, M. Fertilization with Green Manure on Chasselas Dore Grape Vine as an Alternative for Sustainable Agriculture. J. Food Agric. Environ. 2011, 9, 8. [Google Scholar]
- Cesco, S.; Pii, Y.; Borruso, L.; Orzes, G.; Lugli, P.; Mazzetto, F.; Genova, G.; Signorini, M.; Brunetto, G.; Terzano, R.; et al. A Smart and Sustainable Future for Viticulture Is Rooted in Soil: How to Face Cu Toxicity. Appl. Sci. 2021, 11, 907. [Google Scholar] [CrossRef]
- MacKie, K.A.; Müller, T.; Kandeler, E. Remediation of Copper in Vineyards—A Mini Review. Environ. Pollut. 2012, 167, 16–26. [Google Scholar] [CrossRef]
- Komárek, M.; Čadková, E.; Chrastný, V.; Bordas, F.; Bollinger, J.C. Contamination of Vineyard Soils with Fungicides: A Review of Environmental and Toxicological Aspects. Environ. Int. 2010, 36, 138–151. [Google Scholar] [CrossRef]
- Casson, A.; Ortuani, B.; Giovenzana, V.; Brancadoro, L.; Corsi, S.; Gharsallah, O.; Guidetti, R.; Facchi, A. A Multidisciplinary Approach to Assess Environmental and Economic Impact of Conventional and Innovative Vineyards Management Systems in Northern Italy. Sci. Total Environ. 2022, 838, 156181. [Google Scholar] [CrossRef]
- Jones, G.V.; White, M.A.; Cooper, O.R.; Storchmann, K. Climate Change and Global Wine Quality. Clim. Change 2005, 73, 319–343. [Google Scholar] [CrossRef]
- Webb, L.B.; Whetton, P.H.; Barlow, E.W.R. Climate Change and Winegrape Quality in Australia. Clim. Res. 2008, 36, 99–111. [Google Scholar] [CrossRef]
- Hall, A.; Mathews, A.J.; Holzapfel, B.P. Potential Effect of Atmospheric Warming on Grapevine Phenology and Post-Harvest Heat Accumulation across a Range of Climates. Int. J. Biometeorol. 2016, 60, 1405–1422. [Google Scholar] [CrossRef]
- Lehmann, J.; Bossio, D.A.; Kögel-Knabner, I.; Rillig, M.C. The Concept and Future Prospects of Soil Health. Nat. Rev. Earth Environ. 2020, 1, 544–553. [Google Scholar] [CrossRef]
- Karlen, D.L.; Veum, K.S.; Sudduth, K.A.; Obrycki, J.F.; Nunes, M.R. Soil Health Assessment: Past Accomplishments, Current Activities, and Future Opportunities. Soil Tillage Res. 2019, 195, 104365. [Google Scholar] [CrossRef]
- Montalvo-Falcón, J.V.; Sánchez-García, E.; Marco-Lajara, B.; Martínez-Falcó, J. Sustainability Research in the Wine Industry: A Bibliometric Approach. Agronomy 2023, 13, 871. [Google Scholar] [CrossRef]
- Villat, J. Down to Earth: Identifying and Promoting Regenerative Viticulture Practices for Soil and Human Health. Ph.D. Thesis, Harvard University, Cambridge, MA, USA, 2021. [Google Scholar]
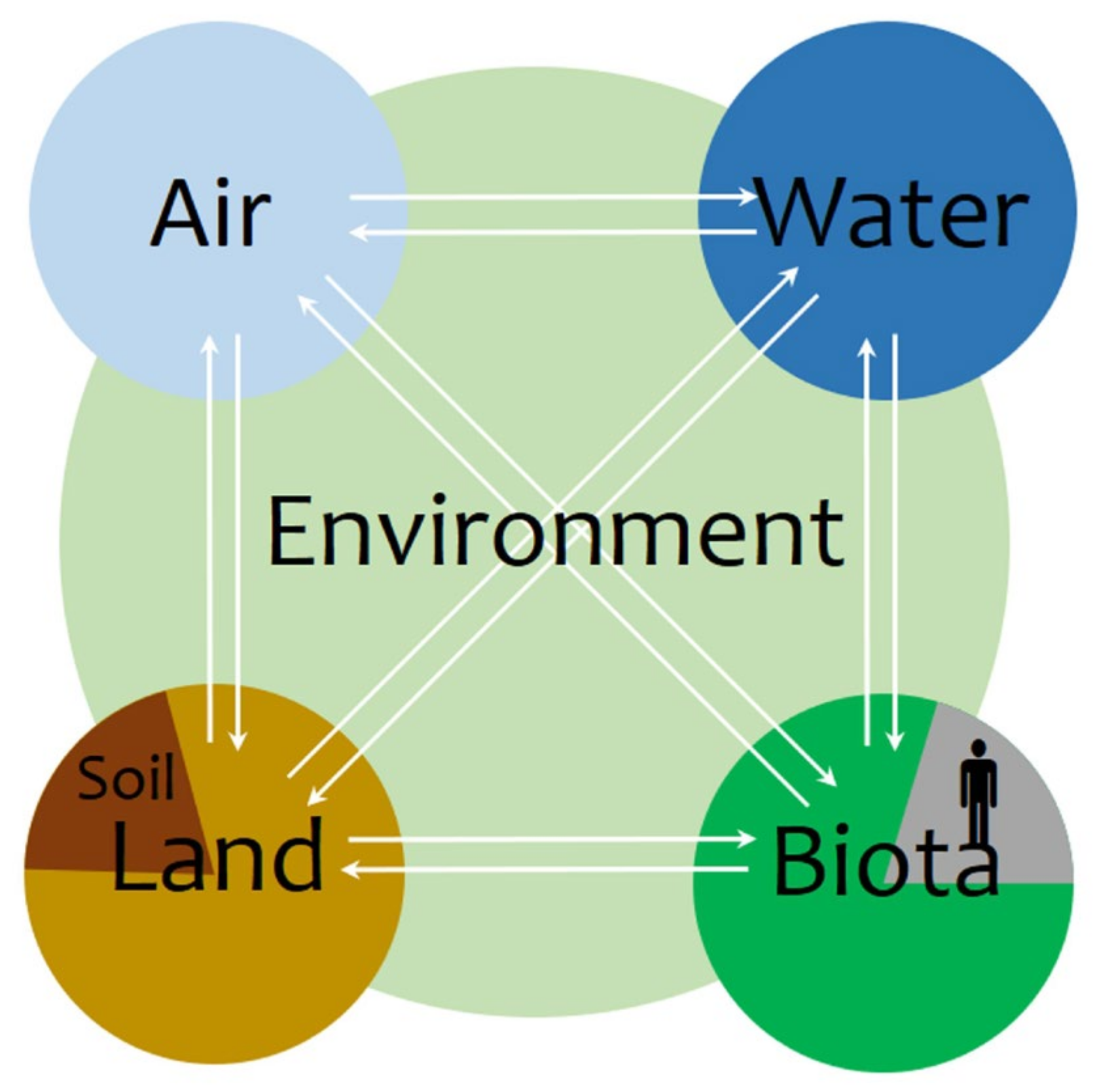

| Soil Practice | Effect on Soil Microbial Population |
|---|---|
| Retention of crop residues | ↑ |
| Application of organic amendments to soil | ↑↓ |
| Minimum tillage practices or absence of ploughing | ↑↓ |
| Irrigation of crops in rain-limited ecosystems | ↑ |
| Application of fungicides to soil | ↓ |
| Application of herbicides to soil | ↑↓ |
| Liming of soil to raise pH on acidic soils | ↑ |
| Unbalanced fertilization | ↓ |
| Characteristic | Rhizosphere | Bulk Soil |
|---|---|---|
| Concentrations of easily degradable substances (sugars, organic acids, and amino acids) | ↑ | ↓ |
| Diversity of microorganisms | ↓ | ↑ |
| Abundance of specific microbial taxa | ↑ | ↓ |
| Phytotoxicity of heavy metals | ↓ | ↑ |
| Enzyme activities | ↑ | ↓ |
| Nutrient availability | ↑ | ↓ |
| Soil pH | ↑ (alkalinisation) | ↓ |
| Water retention because of changes in soil structure | ↑ | ↓ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Visconti, F.; López, R.; Olego, M.Á. The Health of Vineyard Soils: Towards a Sustainable Viticulture. Horticulturae 2024, 10, 154. https://doi.org/10.3390/horticulturae10020154
Visconti F, López R, Olego MÁ. The Health of Vineyard Soils: Towards a Sustainable Viticulture. Horticulturae. 2024; 10(2):154. https://doi.org/10.3390/horticulturae10020154
Chicago/Turabian StyleVisconti, Fernando, Roberto López, and Miguel Ángel Olego. 2024. "The Health of Vineyard Soils: Towards a Sustainable Viticulture" Horticulturae 10, no. 2: 154. https://doi.org/10.3390/horticulturae10020154
APA StyleVisconti, F., López, R., & Olego, M. Á. (2024). The Health of Vineyard Soils: Towards a Sustainable Viticulture. Horticulturae, 10(2), 154. https://doi.org/10.3390/horticulturae10020154






