The Potential of Arctic Pseudogymnoascus Fungi in the Biosynthesis of Natural Products
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Calixto, J.B. The role of natural products in modern drug discovery. An. Acad. Bras. Cienc. 2019, 91, e20190105. [Google Scholar] [CrossRef]
- Shankar, A.; Sharma, K.K. Fungal secondary metabolites in food and pharmaceuticals in the era of multi-omics. Appl. Microbiol. Biotechnol. 2022, 106, 3465–3488. [Google Scholar] [CrossRef]
- Keller, N.P. Fungal secondary metabolism: Regulation, function and drug discovery. Nat. Rev. Microbiol. 2019, 17, 167–180. [Google Scholar] [CrossRef]
- Avalos, J.; Limón, M.C. Fungal Secondary Metabolism. Encyclopedia 2022, 2, 1–13. [Google Scholar] [CrossRef]
- Conrado, R.; Gomes, T.C.; Roque, G.S.C.; De Souza, A.O. Overview of bioactive fungal secondary metabolites: Cytotoxic and antimicrobial compounds. Antibiotics 2022, 11, 1604. [Google Scholar] [CrossRef] [PubMed]
- Antipova, T.V.; Zhelifonova, V.; Zaitsev, K.V.; Zherebker, A.; Baskunov, B.; Oprunenko, Y.F. Formation of azaphilone pigments and monasnicotinic acid by the fungus Aspergillus cavernicola. J. Agric. Food Chem. 2022, 70, 7122–7129. [Google Scholar] [CrossRef]
- Antipova, T.V.; Zhelifonova, V.P.; Zaitsev, K.V.; Vainshtein, M.B. Fungal azaphilone pigments as promising natural colorants. Microbiology 2023, 92, 1–10. [Google Scholar] [CrossRef]
- Robey, M.T.; Caesar, L.K.; Drott, M.T.; Keller, N.P.; Kelleher, N.L. An interpreted atlas of biosynthetic gene clusters from 1,000 fungal genomes. Proc. Natl. Acad. Sci. USA 2021, 118, e2020230118. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.A.; Bacha, N.; Ahmad, B.; Lutfullah, G.; Farooq, U.; Cox, R.J. Fungi as chemical industries and genetic engineering for the production of biologically active secondary metabolites. Asian Pac. J. Trop. Biomed. 2014, 4, 859–870. [Google Scholar] [CrossRef]
- Caesar, L.K.; Butun, F.A.; Robey, M.T.; Ayon, N.J.; Gupta, R.; Dainko, D.; Bok, J.W.; Nickles, G.; Stankey, R.J.; Johnson, D.; et al. Correlative metabologenomics of 110 fungi reveals metabolite–gene cluster pairs. Nat. Chem. Biol. 2023, 19, 846–854. [Google Scholar] [CrossRef]
- Marshall, W.A. Aerial transport of keratinaceous substrate and distribution of the fungus Geomyces pannorum in antarctic soils. Microb. Ecol. 1998, 36, 212–219. [Google Scholar] [CrossRef] [PubMed]
- Ozerskaya, S.; Kochkina, G.; Ivanushkina, N.; Gilichinsky, D. Fungi in permafrost. In Permafrost Soils. Soil Biology; Margesin, R., Ed.; Springer: Berlin/Heidelberg, Germany, 2009; Volume 16, pp. 85–95. [Google Scholar] [CrossRef]
- Kochkina, G.; Ivanushkina, N.; Ozerskaya, S.; Chigineva, N.; Vasilenko, O.; Firsov, S.; Spirina, E.; Gilichinsky, D. Ancient fungi in Antarctic permafrost environments. FEMS Microbiol. Ecol. 2012, 82, 501–509. [Google Scholar] [CrossRef] [PubMed]
- Leushkin, E.V.; Logacheva, M.D.; Penin, A.A.; Sutormin, R.A.; Gerasimov, E.S.; Kochkina, G.A.; Ivanushkina, N.E.; Vasilenko, O.V.; Kondrashov, A.S.; Ozerskaya, S.M. Comparative genome analysis of Pseudogymnoascus spp. reveals primarily clonal evolution with small genome fragments exchanged between lineages. BMC Genom. 2015, 16, 400. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.Y.; Zhang, Y.; Xu, X.Y.; Qi, S.H. Diverse deep-sea fungi from the south China sea and their antimicrobial activity. Curr. Microbiol. 2013, 67, 525–530. [Google Scholar] [CrossRef]
- Kochkina, G.A.; Ivanushkina, N.E.; Akimov, V.N.; Gilichinskiĭ, D.A.; Ozerskaya, S.M. Halo- and psychrotolerant Geomyces fungi from arctic cryopegs and marine deposits. Microbiology 2007, 76, 31–38. [Google Scholar] [CrossRef]
- Shcherbakova, V.A.; Kochkina, G.A.; Ivanushkina, N.E.; Laurinavichius, K.S.; Ozerskaya, S.M.; Akimenko, V.K. Growth of the fungus Geomyces pannorum under anaerobiosis. Microbiology 2010, 79, 845–848. [Google Scholar] [CrossRef]
- Farrell, R.L.; Arenz, B.E.; Duncan, S.M.; Held, B.W.; Jurgens, J.A.; Blanchette, R.A. Introduced and indigenous fungi of the Ross Island historic huts and pristine areas of Antarctica. Polar Biol. 2011, 34, 1669. [Google Scholar] [CrossRef]
- Tagawa, M.; Tamaki, H.; Manome, A.; Koyama, O.; Kamagata, Y. Isolation and characterization of antagonistic fungi against potato scab pathogens from potato field soils. FEMS Microbiol. Lett. 2010, 305, 136–142. [Google Scholar] [CrossRef]
- Loperena, L.; Soria, V.; Varela, H.S.; Lupo, S.; Bergalli, A.; Guigou, M.; Pellegrino, A.; Bernardo, A.; Calviño, A.; Rivas, F.; et al. Extracellular enzymes produced by microorganisms isolated from maritime Antarctica. World J. Microbiol. Biotechnol. 2012, 28, 2249–2256. [Google Scholar] [CrossRef] [PubMed]
- Poveda, G.; Gil-Durán, C.; Vaca, I.; Levicán, G.; Chávez, R. Cold-active pectinolytic activity produced by filamentous fungi associated with Antarctic marine sponges. Biol. Res. 2018, 51, 28. [Google Scholar] [CrossRef] [PubMed]
- Furhan, J. Adaptation, production, and biotechnological potential of cold-adapted proteases from psychrophiles and psychrotrophs: Recent overview. J. Genet. Eng. Biotechnol. 2020, 18, 36. [Google Scholar] [CrossRef] [PubMed]
- Xia, Q.; Rufty, T.; Shi, W. Predominant microbial colonizers in the root endosphere and rhizosphere of turfgrass systems: Pseudomonas veronii, Janthinobacterium lividum, and Pseudogymnoascus spp. Front. Microbiol. 2021, 12, 643904. [Google Scholar] [CrossRef]
- Urbina, J.; Chestnut, T.; Allen, J.M.; Levi, T. Pseudogymnoascus destructans growth in wood, soil and guano substrates. Sci. Rep. 2011, 11, 763. [Google Scholar] [CrossRef] [PubMed]
- Figueroa, L.; Jiménez, C.; Rodríguez, J.; Areche, C.; Chávez, R.; Henríquez, M.; de la Cruz, M.; Díaz, C.; Segade, Y.; Vaca, I. 3-Nitroasterric acid derivatives from an Antarctic sponge-derived Pseudogymnoascus sp. fungus. J. Nat. Prod. 2015, 78, 919–923. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.Z.; Wei, Q.; Gao, J.; Liu, B.Y.; Zhang, T.; Hua, H.M.; Hu, Y.C. Metabolites of the psychrophilic fungus Pseudogymnoascus pannorum. Nat. Prod. Res. Dev. 2019, 31, 446–449. [Google Scholar]
- Fujita, K.; Ikuta, M.; Nishimura, S.; Sugiyama, R.; Yoshimura, A.; Kakeya, H. Amphiol, an antifungal fungal pigment from Pseudogymnoascus sp. PF1464. J. Nat. Prod. 2021, 84, 986–992. [Google Scholar] [CrossRef]
- Shi, T.; Yu, Y.Y.; Dai, J.J.; Zhang, Y.T.; Hu, W.P.; Zheng, L.; Shi, D.Y. New polyketides from the Antarctic fungus Pseudogymnoascus sp. HSX2#-11. Mar. Drugs 2021, 19, 168. [Google Scholar] [CrossRef]
- Li, Y.; Sun, B.; Liu, S.; Jiang, L.; Liu, X.; Zhang, H.; Che, Y. Bioactive asterric acid derivatives from the Antarctic ascomycete fungus Geomyces sp. J. Nat. Prod. 2008, 71, 1643–1646. [Google Scholar] [CrossRef]
- Parish, C.A.; de la Cruz, M.; Smith, S.K.; Zink, D.; Baxter, J.; Tucker-Samaras, S.; Collado, J.; Platas, G.; Bills, G.; Díez, M.T.; et al. Antisense-guided isolation and structure elucidation of pannomycin, a substituted cis-decalin from Geomyces pannorum. J. Nat. Prod. 2009, 72, 59–62. [Google Scholar] [CrossRef]
- Purić, J.; Vieira, G.; Cavalca, L.B.; Sette, L.D.; Ferreira, H.; Vieira, M.L.C.; Sass, D.C. Activity of Antarctic fungi extracts against phytopathogenic bacteria. Lett. Appl. Microbiol. 2018, 66, 530–536. [Google Scholar] [CrossRef]
- Vaca, I.; Chávez, R. Bioactive compounds produced by Antarctic filamentous fungi. In Fungi of Antarctica; Rosa, L., Ed.; Springer: Cham, Switzerland, 2019; pp. 265–283. [Google Scholar] [CrossRef]
- Gonçalves, V.N.; Carvalho, C.R.; Johann, S.; Mendes, G.; Alves, T.M.A.; Zani, C.L.; Junior, P.A.S.; Murta, S.M.F.; Romanha, A.J.; Cantrell, C.L.; et al. Antibacterial, antifungal and antiprotozoal activities of fungal communities present in different substrates from Antarctica. Polar Biol. 2015, 38, 1143–1152. [Google Scholar] [CrossRef]
- Shi, T.; Li, X.Q.; Zheng, L.; Zhang, Y.H.; Dai, J.J.; Shang, E.L.; Yu, Y.Y.; Zhang, Y.T.; Hu, W.P.; Shi, D.Y. Sesquiterpenoids from the Antarctic fungus Pseudogymnoascus sp. HSX2#-11. Front. Microbiol. 2021, 12, 688202. [Google Scholar] [CrossRef]
- Camacho, C.; Coulouris, G.; Avagyan, V.; Ma, N.; Papadopoulos, J.; Bealer, K.; Madden, T.L. BLAST+: Architecture and applications. BMC Bioinform. 2009, 10, 421. [Google Scholar] [CrossRef]
- Harvey, C.J.B.; Tang, M.; Schlecht, U.; Horecka, J.; Fischer, C.R.; Lin, H.C.; Li, J.; Naughton, B.; Cherry, J.; Miranda, M.; et al. HEx: A heterologous expression platform for the discovery of fungal natural products. Sci. Adv. 2018, 11, eaar5459. [Google Scholar] [CrossRef]
- Tarlachkov, S.V.; Starodumova, I.P. TaxonDC: Calculating the similarity value of the 16S rRNA gene sequences of prokaryotes or ITS regions of fungi. J. Bioinform. Genom. 2017, 3. [Google Scholar] [CrossRef]
- Takamatsu, S.; Kim, Y.P.; Hayashi, M.; Hiraoka, H.; Natori, M.; Komiyama, K.; Omura, S. Macrosphelide, a novel inhibitor of cell-cell adhesion molecule. II. Physiochemical properties and structural elucidation. J. Antibiot. 1996, 49, 95–98. [Google Scholar] [CrossRef]
- Sunazuka, T.; Hirose, T.; Harigaya, Y.; Takamatsu, S.; Hayashi, M.; Komiyama, K.; Ōmura, S.; Sprengeler, P.A.; Smith, A.B. Relative and absolute stereochemistries and total synthesis of (+)-macrosphelides A and B, potent, orally bioavailable inhibitors of cell−cell adhesion. J. Am. Chem. Soc. 1997, 119, 10247–10248. [Google Scholar] [CrossRef]
- Sunazuka, T.; Hirose, T.; Chikaraishi, N.; Harigaya, Y.; Hayashi, M.; Komiyama, K.; Sprengeler, P.A.; Smith, A.B.; Ōmura, S. Absolute stereochemistries and total synthesis of (+)/(−)-macrosphelides, potent, orally bioavailable inhibitors of cell–cell adhesion. Tetrahedron 2005, 61, 3789–3803. [Google Scholar] [CrossRef]
- Prasad, K.R.; Gutala, P. Enantioselective total synthesis of macrosphelides A and E. Tetrahedron 2011, 67, 4514–4520. [Google Scholar] [CrossRef]
- Kobayashi, Y.; Kumar, G.B.; Kurachi, T.; Acharya, H.P.; Yamazaki, T.; Kitazume, T. Furan ring oxidation strategy for the synthesis of macrosphelides A and B. J. Org. Chem. 2001, 66, 2011–2018. [Google Scholar] [CrossRef] [PubMed]
- Kawaguchi, T.; Funamori, N.; Matsuya, Y.; Nemoto, H. Total synthesis of macrosphelides A, B, and E: first application of ring-closing metathesis for macrosphelide synthesis. J. Org. Chem. 2004, 69, 505–509. [Google Scholar] [CrossRef]
- Matsuya, Y.; Kawaguchi, T.; Nemoto, H. New strategy for the total synthesis of Macrosphelides A and B based on ring-closing metathesis. Org. Lett. 2003, 5, 2939–2941. [Google Scholar] [CrossRef]
- Paek, S.M.; Seo, S.Y.; Kim, S.H.; Jung, J.W.; Lee, Y.S.; Jung, J.K.; Suh, Y.G. Concise syntheses of (+)-macrosphelides A and B. Org. Lett. 2005, 7, 3159–3162. [Google Scholar] [CrossRef] [PubMed]
- Paek, S.M.; Suh, Y.G. Synthetic studies on bioactive natural polyketides: Intramolecular nitrile oxide-olefin cycloaddition approach for construction of a macrolactone skeleton of macrosphelide B. Molecules 2011, 16, 4850–4860. [Google Scholar] [CrossRef]
- Takamatsu, S.; Hiraoka, H.; Kim, Y.P.; Hayashi, M.; Natori, M.; Komiyama, K.; Omura, S. Macrosphelides C and D, novel inhibitors of cell adhesion. J. Antibiot. 1997, 50, 878–880. [Google Scholar] [CrossRef] [PubMed]
- Yamada, T.; Minoura, K.; Tanaka, R.; Numata, A. Cell-adhesion inhibitors produced by a sea hare-derived Periconia sp. J. Antibiot. 2007, 60, 370–375. [Google Scholar] [CrossRef]
- Fukami, A.; Taniguchi, Y.; Nakamura, T.; Rho, M.C.; Kawaguchi, K.; Hayashi, M.; Komiyama, K.; Omura, S. New members of the macrosphelides from Microsphaeropsis sp. FO-050 IV. J. Antibiot. 1999, 52, 501–504. [Google Scholar] [CrossRef] [PubMed]
- Ivanova, V.; Kolarova, M.; Aleksieva, K.; Graefe, U.; Schlegel, B. Diphenylether and macrotriolides occurring in a fungal isolate from the antarctic lichen Neuropogon. Prep. Biochem. Biotechnol. 2007, 37, 39–45. [Google Scholar] [CrossRef]
- McQuilken, M.P.; Gemmell, J.; Hill, R.A.; Whipps, J.M. Production of macrosphelide A by the mycoparasite Coniothyrium minitans. FEMS Microbiol. Lett. 2003, 219, 27–31. [Google Scholar] [CrossRef]
- Wińska, K.; Mączka, W.; Grabarczyk, M.; Sugimoto, K.; Matsuya, Y.; Szumny, A.; Anioł, M. A macrosphelide as the unexpected product of a Pleurotus ostreatus strain-mediated biotransformation of halolactones containing the gem-dimethylcyclohexane ring. Part 1. Molecules 2016, 21, 859. [Google Scholar] [CrossRef]
- Hayashi, M.; Kim, Y.P.; Hiraoka, H.; Natori, M.; Takamatsu, S.; Kawakubo, T.; Masuma, R.; Komiyama, K.; Omura, S. Macrosphelide, a novel inhibitor of cell-cell adhesion molecule. I. Taxonomy, fermentation, isolation and biological activities. J. Antibiot. 1995, 48, 1435–1439. [Google Scholar] [CrossRef] [PubMed]
- Tomprefa, N.; McQuilken, M.P.; Hill, R.A.; Whipps, J.M. Antimicrobial activity of Coniothyrium minitans and its macrolide antibiotic macrosphelide A. J. Appl. Microbiol. 2009, 106, 2048–2056. [Google Scholar] [CrossRef] [PubMed]
- Albert, D.; Dumonceaux, T.; Carisse, O.; Beaulieu, C.; Filion, C. Combining desirable traits for a good biocontrol strategy against Sclerotinia sclerotiorum. Microorganisms 2022, 10, 1189. [Google Scholar] [CrossRef] [PubMed]
- Heo, Y.M.; Lee, H.; Shin, Y.K.; Paek, S.M. Development of an advanced synthetic route to macrosphelides and its application to the discovery of a more potent macrosphelide derivative. Molecules 2014, 19, 15572–15583. [Google Scholar] [CrossRef]
- Ahmed, K.; Matsuya, Y.; Nemoto, H.; Zaidi, S.F.; Sugiyama, T.; Yoshihisa, Y.; Shimizu, T.; Kondo, T. Mechanism of apoptosis induced by a newly synthesized derivative of macrosphelides with a thiazole side chain. Chem. Biol. Interact. 2009, 177, 218–226. [Google Scholar] [CrossRef] [PubMed]
- Song, K.; Rajasekaran, N.; Chelakkot, C.; Lee, H.S.; Paek, S.M.; Yang, H.; Jia, L.; Park, H.G.; Son, W.S.; Kim, Y.J.; et al. Macrosphelide A exhibits a specific anti-cancer effect by simultaneously inactivating ENO1, ALDOA, and FH. Pharmaceuticals 2021, 14, 1060. [Google Scholar] [CrossRef]
- Fukami, A.; Iijima, K.; Hayashi, M.; Komiyama, K.; Omura, S. Macrosphelide B suppressed metastasis through inhibition of adhesion of sLe(x)/E -selectin molecules. Biochem. Biophys. Res. Commun. 2002, 291, 1065–1070. [Google Scholar] [CrossRef]
- Nemoto, H.; Matsuya, Y.-J. Method for Synthesizing Macrosphelides. US Patent US 7265229B2, 9 April 2007. [Google Scholar]
- Paek, S.M. Development of advanced macrosphelides: Potent anticancer agents. Molecules 2015, 20, 4430–4449. [Google Scholar] [CrossRef]
- Keller, M.; Zengler, K. Tapping into microbial diversity. Nat. Rev. Microbiol. 2004, 2, 141–150. [Google Scholar] [CrossRef]
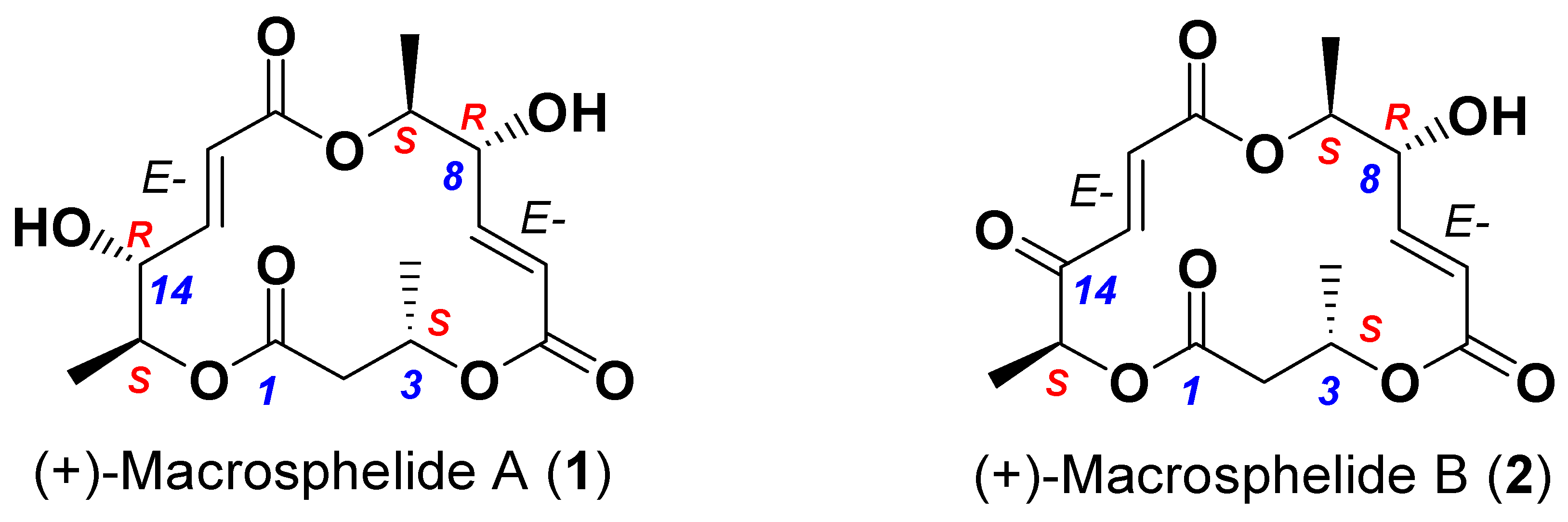

| Paraphaeosphaeria sporulosa AP3s5-JAC2a | Pseudogymnoascus sp. VKM F-4518 | Pseudogymnoascus sp. VKM F-4519 | |||
|---|---|---|---|---|---|
| Gene locus tag (protein accession) | Description | Gene locus tag (protein accession) | Protein identity with P. sporulosa | Gene locus tag (protein accession) | Protein identity with P. sporulosa |
| CC84DRAFT_1205319 (OAG05541.1) | DH | V500_01681 (KFY98390.1) | 75.60% | V501_03182 (KFZ14540.1) | 75.40% |
| CC84DRAFT_1216878 (OAG05542.1) | SDR | V500_01680 (KFY98389.1) | 79.92% | V501_03183 (KFZ14541.1) | 80.72% |
| CC84DRAFT_1195956 (OAG05543.1) | p450 | V500_01679 (KFY98388.1) | 83.91% | V501_03184 (KFZ14542.1) | 83.72% |
| CC84DRAFT_1091541 (OAG05544.1) | SH | V500_01682 (KFY98391.1) | 90.29% | V501_03181 (KFZ14539.1) | 90.29% |
| CC84DRAFT_1091839 (OAG05545.1) | KS-AT-DH-ER-KR-ACP | V500_01683 (KFY98392.1) | 85.45% | V501_03180 (KFZ14538.1) | 85.28% |
| Region | Type | From | To | Most Similar Known Cluster | Similarity, % |
|---|---|---|---|---|---|
| VKM F-4518 | |||||
| 1.1. | T1PKS | 1 | 15,076 | Ustilaginoidins | 23 |
| 53.1 | T1PKS | 6889 | 32,830 | Secalonic acids | 12 |
| 399.1 | T1PKS | 1 | 39,308 | HEx-pks1 polyketide | 100 |
| 496.1 | T1PKS | 4600 | 51,229 | Orsellinic acid | 50 |
| 714.1 | T1PKS | 1 | 27,887 | Monacolin K | 22 |
| 940.1 | T1PKS | 1 | 25,739 | 1,3,6,8-Tetrahydronaphthalene | 100 |
| VKM F-4519 | |||||
| 183.1 | T1PKS | 1 | 43,063 | Azanigerones | 20 |
| 342.1 | T1PKS | 1 | 18,121 | Scytalone | 40 |
| 384.1 | NRPS | 35,666 | 61,986 | Choline | 100 |
| 850.1 | T1PKS | 1 | 20,490 | HEx-pks1 polyketide | 100 |
| 914.1 | NRPS, T1PKS | 1 | 32,243 | Phyllostictines | 20 |
| 960.1 | NRPS, T1PKS | 1 | 23,427 | Phomacins D, C | 22 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Antipova, T.V.; Zaitsev, K.V.; Zhelifonova, V.P.; Tarlachkov, S.V.; Grishin, Y.K.; Kochkina, G.A.; Vainshtein, M.B. The Potential of Arctic Pseudogymnoascus Fungi in the Biosynthesis of Natural Products. Fermentation 2023, 9, 702. https://doi.org/10.3390/fermentation9080702
Antipova TV, Zaitsev KV, Zhelifonova VP, Tarlachkov SV, Grishin YK, Kochkina GA, Vainshtein MB. The Potential of Arctic Pseudogymnoascus Fungi in the Biosynthesis of Natural Products. Fermentation. 2023; 9(8):702. https://doi.org/10.3390/fermentation9080702
Chicago/Turabian StyleAntipova, Tatiana V., Kirill V. Zaitsev, Valentina P. Zhelifonova, Sergey V. Tarlachkov, Yuri K. Grishin, Galina A. Kochkina, and Mikhail B. Vainshtein. 2023. "The Potential of Arctic Pseudogymnoascus Fungi in the Biosynthesis of Natural Products" Fermentation 9, no. 8: 702. https://doi.org/10.3390/fermentation9080702
APA StyleAntipova, T. V., Zaitsev, K. V., Zhelifonova, V. P., Tarlachkov, S. V., Grishin, Y. K., Kochkina, G. A., & Vainshtein, M. B. (2023). The Potential of Arctic Pseudogymnoascus Fungi in the Biosynthesis of Natural Products. Fermentation, 9(8), 702. https://doi.org/10.3390/fermentation9080702







