Abstract
Bacteria have been used in the food industry to produce flavors, dyes, thickeners, and to increase food value, because bacterial fermentations favor the obtention of different metabolites such as tannins and different nutritional compounds in food. Lactiplantibacillus plantarum was one the first species to be studied for industrial purposes, and its efficacy to obtaining tannins using fermentation processes. Bacterial fermentation helps to obtain a product with an added value of better quality and without the need to use strong solvents that can reduce their quality and safety. To release tannins, it is necessary to subject the substrate to different conditions to activate the enzyme tannin acyl hydrolase (tannase). The tannase-released compounds can have beneficial effects on health such as antioxidant, anticancer and cardioprotective properties, among others. Therefore, this review analyzes tannase release and other metabolites by fermentation processes.
1. Introduction
The term fermentation derives from the Latin ‘fervere’, which means to boil, due to the appearance produced by yeast on fruit or malt extracts, as a result of the production of carbon dioxide bubbles caused by the catabolism of the sugars in the extract [1]. The most common concept of fermentation is the conversion of sugar into an organic acid, then into alcohol. It occurs naturally in many food types, and humans have used it since ancient times to improve the preservation and the organoleptic properties of food. This term is only used when referring to microorganisms (bacteria, yeasts, and fungi) to make useful products for humans such as biomasses, enzymes, primary and secondary metabolites, recombinant products, and biotransformed products used in industry [2]. Fermentation is a catabolic process of incomplete oxidation, completely anaerobic, and its final product is an organic compound, where in the absence of oxygen, the final acceptor of the NADH electrons produced in glycolysis is not oxygen, but an organic compound that will be reduced to oxidize NADH to NAD+ [3]. There are different types of fermentation, e.g., alcoholic, acetic, butyric, and lactic. The fermentation processes have some main purposes: (1) the enrichment of the diet by developing flavors, aromas, and textures; (2) food preservation through lactic acid, ethanol, acetic acid, and alkaline fermentations; (3) enrichment of foods with protein, amino acids, lipids, and vitamins; (4) decrease in cooking time and fuel requirements so that the loss of nutrients is less; and (5) it can produce nutrients or eliminate anti-nutrients; being the final product an organic compound that characterizes the types of fermentation [4].
2. Tannins Overview
Tannins are water-soluble polyphenolic compounds distributed in plants mainly in roots, wood, bark, seeds, fruits, and leaves, with molecular masses ranging from 3000 to 20,000 Da [5]. Tannins are secondary metabolites and are the second most abundant group of phenolics in nature [6], with the capacity to form complexes with proteins, carbohydrates, and enzymes, which makes them anti-nutritional factors [7]. They can also bind to metal ions, reducing the availability of iron and the production of enzymes that need metals [8].
Tannins are divided into four major groups: (1) condensed tannins; (2) hydrolysable tannins; (3) gallotannins; and (4) ellagitannins [9]. They are used for tanning skins and coloring foods, and to produce adhesives, lubricants, and cosmetics [10]. Due to the multiple hydroxyl groups that are present in the chemical structure of tannins, they can act as antibiotics [11]. Tannins have the ability to precipitate heavy metals and some alkaloids; these can be used as a possible treatment for poisoning caused by these substances [12]. The tannins can also reduce the chances of suffering a heart attack due to their effect on the oxidation of LDL and cholesterol, can stop the proliferation of viruses, and prevent the generation of cancer cells [13].
2.1. Biological Properties of Tannins
2.1.1. Antioxidant Properties
Reactive oxygen species are produced under oxidative stress, including hydrogen peroxide, hydroxyl, superoxide, and hypochlorite, and are molecules that contain oxygen in the active state, which can cause lipid lipoperoxidation and other complications [14]. Oxidative stress generates free radicals that have an important role in aging, asthma, diabetes, cardiovascular diseases, and neurodegenerative disorders such as Alzheimer. The antioxidant activity of the phenolic compounds is produced through inhibiting the formation of reactive oxygen species and the decrease in their complexation levels. By reducing metal ions, they catalyze the reactions that lead to the formation of ROS, which interrupts the free radical cascade and the protection of natural antioxidants [15]. The antioxidant activity of flavonoids increases with the number of hydroxyls groups and decreases with glycosylation [16].
2.1.2. Anticancer Properties
Procyanidin B2 induces apoptosis against prostate cancer by inhibiting cell growth, decreasing clonogenicity, and inducing the arrest cycle of cancer cells, besides inhibiting the activity of tumor necrosis factor kappa (NF-kB), from activator protein 1 (AP1), and the nuclear translocation of the transducer activator signal transducer 3 (Stat 3), the main regulators of apoptosis, cell survival and inflammation [17]. The chemopreventive efficacy of grape seed extract against the early steps of colon carcinogenesis in rats likely via the targeting of b-catenin and NF-kB signaling suggest its potential usefulness for the prevention of human colorectal cancer [9]. Luteolin promotes cellular apoptosis, and acts on hepatocellular carcinoma in SMMC-7721 cells partially by autophagy [18].
In another study, anticancer activity was reported for resveratrol, stilbenes, and isoflavones on A431 skin cancer cell lines, oral Ca9-22 cancer cells, cells present in U937 leukemia, and 4T1 and HL-60 breast cancer cells. By influencing the cell cycle arrest and death, cell proliferation is reduced, preventing the development of mammary tumors and the incidence of lung cancer metastasis. Resveratrol is a potential antioxidant that reduces lipoperoxidation and prevents DNA damage and suppresses breast and colon carcinogenesis. As for prostate cancer, it regulates metastasis and tumor development, and in renal cancer, it acts on the stimulation of CD8+ by T cells, inhibiting the production of interleukin 6 and 10 [19].
2.1.3. Cardioprotective Properties
Endothelial cells are the protective barriers to the walls of blood vessels and are essential to maintain the physiological homeostasis, and a dysfunction generates a risk factor for cardiovascular diseases involved in the pathogenesis of various diseases like atherosclerosis. ROS dysfunction plays a role in the onset of atheromatous plaque accumulation and mitogen-activated protein kinases and the translocation of the nuclear factor NF-kB from the cytosol [20]. It improves the expression of adhesion molecules and inflammatory cytokines at the translation and transcription level, but an excess could generate an increase in the membrane’s permeability, releasing proteases that affect apoptosis, generating endothelial damage. Therefore, an alternative has been sought with natural products such as luteolin, which is a tannin that protects against the tumor necrosis factor-alpha that produces inflammation in the vascular wall, the adhesion of monocytes which generate ischemia, and luteolin which reduces the effect of oxidative stress and inhibits P38 phosphorylation of mitogen-activated protein kinases (MAPK) in H2O2, and also activates the phosphatidylinositol 3-kinase (PI3K) pathway Akt [7,18,21].
2.1.4. Digestive, Carminative, and Astringent Properties
By acting on inflammatory cytokines, tannins have beneficial effects in diseases in which inflammation is the main condition; for example, tannins have been used to reverse and decrease the effects of intestinal inflammation. Luteolin, a naturally occurring flavonoid, exhibits anti-inflammatory properties. In another study, it has been revealed that luteolin may act against intestinal inflammation, which might support the use of luteolin as a future therapeutic strategy in inflammatory bowel diseases [22].
On the other hand, the interaction of tannins with salivary proteins is involved in astringency and this characteristic was evaluated. The authors concluded that basic proline-rich proteins have a protective effect against the structural alteration induced by dietary tannins [23].
According to the World Health Organization, in 2020, obesity is a worldwide disease, a risk factor for premature death, and obesity development occurs when the size of adipocytes increases up to seven times their normal size and quantity. Its main cause is the imbalance between the calories consumed and the calories expended. Among the treatments against obesity, there is the conversion of white fats to brown fats through the expression of an uncoupling protein (UCP1), induction of the mitochondrial metabolism, and thermogenic and fat-burning properties. Adipocytes participate in energy homeostasis by secreting adipokines such as leptin and adiponectin. Adiponectin regulates insulin sensitivity and metabolism, by improving energy expenditure in tissues, decreasing inflammation and the oxidative stress of adipocytes. It influences the hydrolysis of triglycerides by producing tannase from the exposure of the combined tannins [24].
3. Biodegradation of Tannins
Tannins are resistant to microbial attacks and are recalcitrant substances for biodegradation, making them highly polluting. Although they are substances with an antimicrobial potential, some microorganisms are resistant to tannins and can use them as substrates through the production of enzymes [25]. Enzymes are applied in many industries due to their sensitivity to substrates and their environmentally friendly conditions. Through solid-state fermentation, great enzymatic activity has been obtained at a low cost and with a significant economic impact, because most of the substrates used are agro-industrial waste [26].
The enzyme tannin acid hydrolase releases tannins through their hydrolysis [27] with advantages within the food industry by synthesizing prebiotics, bioactive peptides and antioxidants, modification of functional ingredients, and elimination of anti-nutrients [28]. The tannase family includes two classes of enzymes: (1) tannase and (2) gallic acid decarboxylase. These enzymes are used in the pharmaceutical and food industry, and mainly in beverages because they help to reduce bad odors and the precipitates of certain beverages, and to release the tannins they contain [6]. Tannases could also be used to degrade tannins in industrial effluents, offering an economic benefit [29].
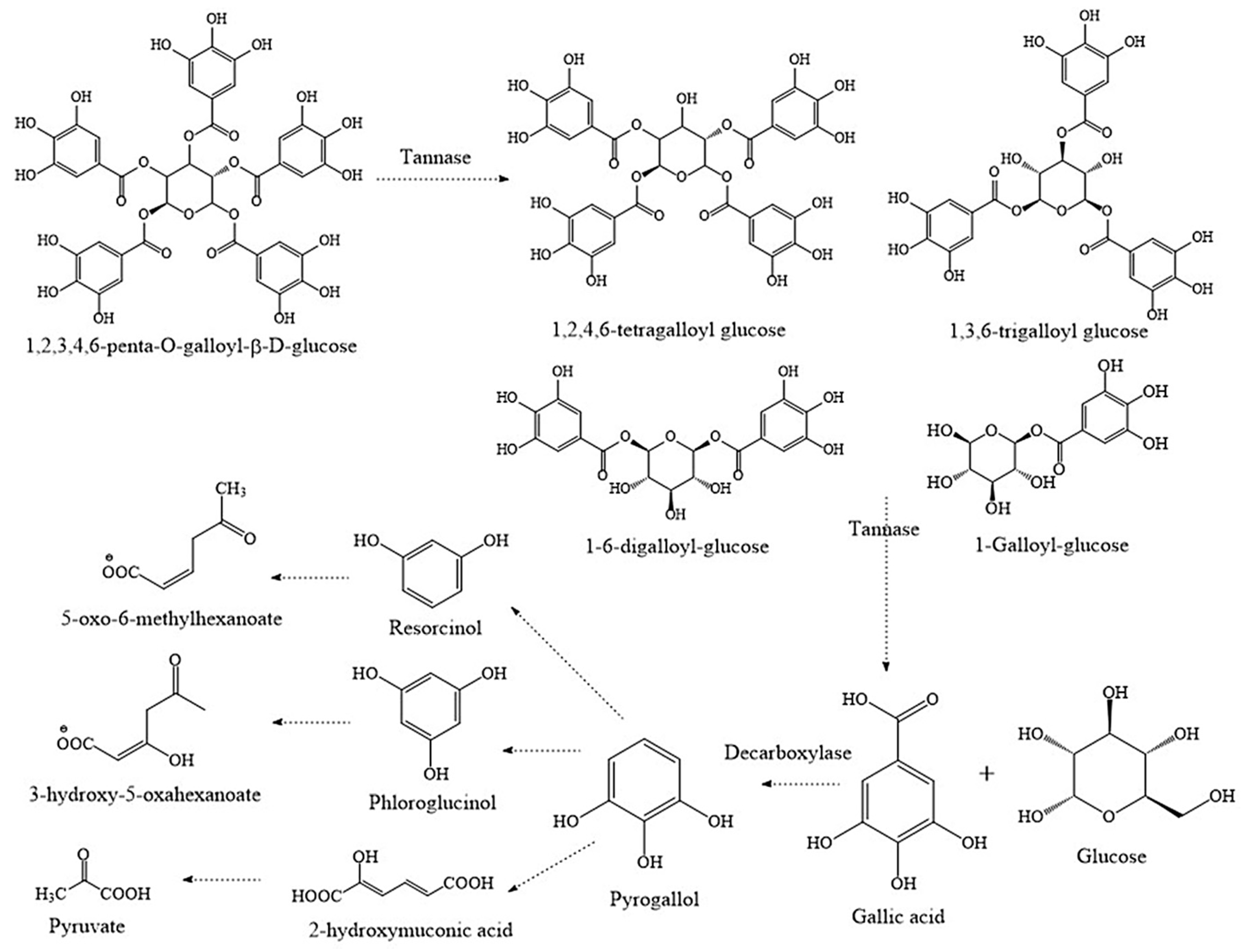
Various microorganisms have been used to produce decarboxylase enzymes to obtain gallic acid, such as Pantoea agglomerans, Enterococcus faecalis, Klebsiella pneumoniae, Lactiplantibacillus plantarum [30]; on the other hand, Aspergillus, Rhizopus, Paecilomyces, Lactiplantibacillus plantarum, and Bacillus licheniformis have been used to obtain tannases [27]. The aerobic catabolism of gallotannins by tannases catalyze the hydrolysis of ester bonds, generating gallic acid and glucose. Anaerobic catabolism produces gallic acid monomers through an oxidative pathway; subsequently, the decarboxylation of gallic acid occurs, generating pyrogallol, which is transformed through the enzyme gallate decarboxylase into pyruvic acid and enters the Krebs cycle [30]. The main biodegradation of gallotannins pathway is shown in Figure 1.
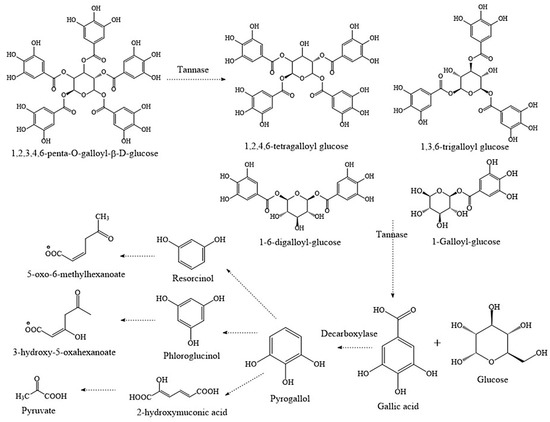
Figure 1.
Biodegradation of tannins. Breakdown of gallotannins by tannase and other enzymes (original source).
Tannic acid can be also hydrolyzed by tannase to obtain gallic acid [29]. This enzyme catalyzes the first step in its biotransformation which is the hydrolysis of tannic acid to release glucose, various galloyl esters of glucose and gallic acid, products that have applications in different areas [12].
To optimize tannase production, one factor to consider should be the substrate, because tannase acts mainly on substrates rich in hydrolysable tannins. For a better tannase performance, different concentrations of tannic acid are required: in submerged fermentation, 0.1 to 10% is required, and in solid-state fermentation, between 10 and 15% is needed [6]; optimal tannase activity is recommended at 30 °C and pH 6 [31]. Bacterial tannases can tolerate temperatures of 30–50 °C, although better results have been observed between 25 °C and 80 °C and pH 9 [13].
4. Production of Tannases with Submerged Fermentation (SF)
Microorganisms require a controlled atmosphere to produce better final quality, ensuring optimal productivity and better benefits [30]. SF has some process characteristics which have slight advantages over control in solid-state fermentation, which are shown in Table 1.

Table 1.
Advantages and disadvantages of submerged and solid-state fermentation.
In submerged fermentation processes, nutrients are used, and some enzymes such as tannase, amylase, and glucoamylases are released, to name a few [37]. It is the most widely used type of fermentation in the industry due to its simplicity, and its final product is easier to recover. The development of microorganisms is typical, which gives rise to a dormancy phase, growth, stationary phase, and death phase. These phases are present in submerged and solid-state fermentation, divided into several types—batch, continuous, and fed—according to the input and output of the substrate and product, and are used for the bioremediation of industrial effluents [32]. In the SF, the initial water content and inoculum and substrate are important; the conditions of the culture medium can produce a wide range of bioactive compounds that have benefits for human health [38]. It is the most widely used in the food industry to obtain enzymes with different substrates and microorganisms.
5. Production of Tannases Using Solid-State Fermentation
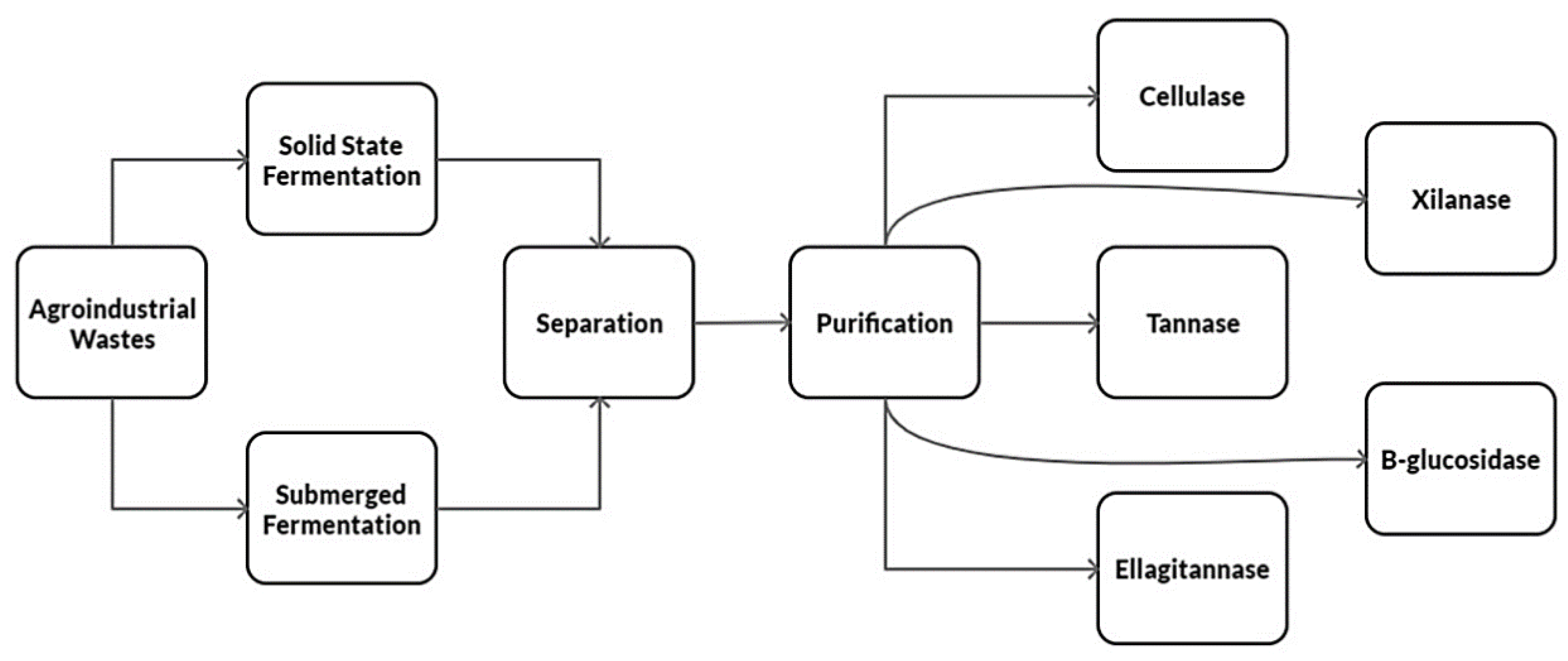
Solid-state fermentation is an effective method to produce microbial enzymes and bioactive compounds [46], allowing the accumulation high-value products. Its main products are enzymes produced using agro-industrial waste. Microorganisms grow by joining solid substrates, imitating their habitat, giving a fruitful result [49]. In this process, the substrate and nutrients can be reused, although this type of fermentation does not apply to bacteria because they require a high-water content [30]. However, it is much more attractive because its chemical and physical processes are low cost, simpler, and have better yields; it allows reproducible conditions similar to the environment in which microorganisms live [45]. Solid-state fermentation has advantages in terms of the final product concentration, has a lower water expenditure, requires less sterile conditions, and has a low repression effect [50]. It is a slightly more laborious process because the growth of the microorganism is slower; there is a low transfer of oxygen and carbon dioxide, heat removal, and it can easily be contaminated [32]. A fundamental stage is the culture media preparation to ensure the fermentation process productivity [51]. The problem that may arise is that the culture substrate is uneven, and changes in temperature, pH, humidity, and substrate concentration lead to making it less reproducible [52]. The water content is an extremely important parameter to produce the desired final product [53]. Growth is carried out on a solid substrate almost without water, but with enough to tolerate growth and metabolism. In solid-state fermentation, the particle size and the humidity level are the most critical factors, as well as the initial pH, substrate, aeration, temperature, inoculation, and incubation [12]. Figure 2 shows the production process of enzymes from agro-industrial waste through solid-state and submerged fermentation.
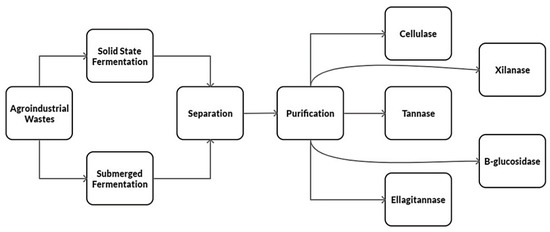
Figure 2.
Diagram showing the main enzyme production processes through solid-state and submerged fermentation (original source).
6. Microorganisms Used in Tannases Production
Tannases can be produced from microorganisms, plants, and animals, even though the most widely used microorganisms are fungi, yeasts, and bacteria. Aspergillus and Penicillium are the most used genera in tannase production; around 27 and 24 species, respectively, have been evaluated for tannase production. Paecilomyces variotii is the best tannase producer using grape pomace as substrate [54]. Four species of Trichoderma and three species of Fusarium have been also evaluated. Regarding bacteria, Lactobacillus is the most studied [35], mainly Lactiplantibacillus plantarum, but Bacillus spp. and Corynebacterium spp. have been also studied [6]. Lactic acid bacteria (LAB) degrade gallotannins to gallic acid, but they live under extreme conditions [25]. Bacteria can change and repair the membrane to reduce the effect of tannins on the digestive tract, increasing the number of unsaturated fatty acids, modifying their isomerization [31].
There are a variety of microorganisms that produce tannases that have been studied for a long time in terms of process, development, and application; the microorganisms for the activation and production of tannases are listed in Table 2.

Table 2.
Microorganisms used to produce tannases.
7. Lactic Acid Bacteria (LAB) That Produce Tannases
Lactic acid bacteria (LAB) have important applications in the food industry and other sectors, as shown in Table 3, for the added value they give to products, influencing the taste, odor, texture, nutritional value, and sensory characteristics. The genus Leuconostoc, Enterococcus, Streptococcus, Pediococcus, and Lactobacillus [42] LAB have been used in submerged fermentation to produce tannases [6]. They are made up of a large group of Gram-positive, non-sporulated bacteria and are the largest producers of lactic acid; they have preservative power because they have antimicrobial properties and have a high content of organic acids and bacteriocins that inhibit the growth of bacteria [58]. LAB are present in groups of microorganisms called starters or lactic cultures, where lactic acid is their main metabolite. LAB are unable to synthesize some vitamins, specific peptides, and amino acids for their growth but can produce antimicrobial substances known as bacteriocins and hydrogen peroxide [59]. They are mesophilic, but some can grow in refrigeration at 4 °C, and others at 45 °C, with a preference for an acidic pH (4–4.5), although sometimes tolerate up to pH 9. They play an important role in biotransformation because they can act on the isolation of new chains of tannases. Among the LAB, the first to be tested to produce tannases was Lactiplantibacillus plantarum [6]. One of the least used microorganisms to obtain tannins through fermentation by LAB [60], there are reports that they degrade and hydrolyze natural tannins and tannic acid efficiently [29]. Their cell wall contains glycerol, teichoic acids, and peptidoglycans [61]. On the other hand, Lactiplantibacillus plantarum was used to produce tannases from olive mill residues using a basic medium, plus 100 mL supplementation with tannic acid as a carbon source and inoculated with 1.38 mg of cells which were incubated at 37 °C for 24 h [13]. It is an optional heterofermentative; it can produce lactic acid and other by-products such as acetate and ethanol, depending on its growing conditions and its carbon source [62]. Lactiplantibacillus plantarum is the most widely used bacteria because it has the greatest yield on plant fermentations where tannins are abundant due to their strains having tannase activity [31], playing an important role in food microbiology and human nutrition for their fermentative power and probiotic functions. It also has effects on cholesterol levels, inhibiting inflammation by acting on interleukins IL-1α, IL-1β, IL-6, and tumor necrosis factor α [59]. The importance of the growth of Lactiplantibacillus plantarum stands out in the technological and physiological characteristics of the probiotic strains, the amino acids valine, arginine, tyrosine, and tryptophan, pantothenic acid and nicotinic acid which are essential, as well as manganese and iron [63]. While the growth during a Lactobacillus fermentation occurs at a pH of 4.5 to 5.8 [59]. Tannase production with Lactiplantibacillus plantarum occurs at 37 °C, with pH 6.0, within 24 h, with tannic acid as a carbon source and the presence of nitrogen ammonium sulfate [64]. Several studies have been carried out obtaining the optimized fermentation process with Lactiplantibacillus plantarum with a maximum tannase activity of 6 U ml-1 [13].

Table 3.
Applications biotechnological of lactic acid bacteria (LAB).
8. Agro-Industrial Wastes to Obtain Tannases
Agro-industrial wastes rich in polyphenolic compounds can be used as a source of carbon and energy so that the microorganism can develop and produce tannases. This section describes some research papers where they used agro-industrial waste to produce tannases with the help of microorganisms. For example, coffee pulp rich in polyphenolic compounds as a source of carbon and energy were evaluated through a solid-state fermentation process for tannase production. The authors concluded that the utilization of this wastes for tannase production has potential applications in the food industry [74]. In another study, solid-state fermentation was used for tannase production, using a medium containing pomegranate peels (10%) and other salts with pH adjusted to 7.0. These production conditions were compared to define the optimal ones, and various carbon sources, including Moringa cake, palm kernel, palm fronds, wheat straw, olive cake, and pomegranate peels were evaluated [75]. Tannase production from solid-state fermentation using coir waste as a substrate was performed. In the reactors, 10 g of the substrate were added and subsequently incubated at 30 °C for 48 h [76]. The tannase-biotransformed grape pomace extracts were evaluated to prove intestinal anti-inflammatory activity using an in vitro colon model. This research mentioned that the biotransformation improved the anti-inflammatory effect in Caco-2 cells, indicating that the application of a bioprocess in grape residues may contribute to the development of new ingredients and nutraceuticals with anti-inflammatory potential [77]. Various agricultural wastes (coffee husk, tamarind seed powder, bagasse, groundnut oil cake, wheat bran and rice bran) and solid substrates (bengal gram powder (Cicer arietinum), black gram flour (Vigna mungo L.), green gram flour (Phasleolus aureus), barley (Hordeum vulgare), millet flour (Peninsetum glaucum), ragi (Eleusine coracana), oats (Avena sativa), corn flour and rice flour) were evaluated to produce tannase by Lactiplantibacillus plantarum [78]. In another study, agro-industrial wastes were shown to be generally rich in tannins and can be used as low-cost substrates for tannase production. Agro-industrial wastes as potential substrates were evaluated for enhanced tannase production using solid-state fermentation. Rice bran showed the highest tannase activity during solid-state fermentation, followed by brewer’s rice, spent coffee grounds, and desiccated coconut waste [48]. The cost-effective substrate Triphala was used as a tannin and energy source for tannase production through solid-state fermentation. In this research, statistical experimental design with Triphala at optimized conditions showed 6.1-fold higher tannase activity than that obtained in submerged fermentation [43]. Tannase production was optimized using pomegranate husk as a solid substrate. Easily available and low-cost substrates such as cow dung, coffee husk, apple peel, and pomegranate peel were initially screened for tannase production [79]. On the other hand, a cocktail of hydrolytic enzymes was performed by solid-state fermentation using as substrate a mixture of grape pomace and wheat bran. They concluded that the use of solid-state fermentation is a less costly alternative to obtain an enzymatic cocktail that can be used in the food industry [80].
9. Conclusions
The fermentation process has many benefits because agro-industrial wastes can be used, it is low in cost, and the recuperation of bioactive compounds is high; this process enhances some biological properties. Fermentation is used as an alternative to other extraction chemical methods of bioactive compounds of high value-added. A great diversity of microorganisms is used through a fermentation process to produce tannases in the food industry. Currently, LAB are used for the production of tannases, because they can take advantage of agro-industrial waste as a source of carbon and energy, and the bioactive compounds they produce can have antioxidant, anticancer, cardioprotective properties, among others.
Author Contributions
Conceptualization and writing—original draft preparation, M.G.G.M.; formal analysis and supervision, T.K.M.M., J.A.A.V. and M.L.C.G.; writing—review and editing and project administration, A.C.F.G. and L.S. All authors have read and agreed to the published version of the manuscript.
Funding
This review was funded by Research Direction and Postgraduate of Autonomous University of Coahuila.
Acknowledgments
The authors appreciate the support of the research and postgraduate management of the Autonomous University of Coahuila. García-Mendez thanks CONACYT for the scholarship for their training in the Master’s program.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Stanbury, P.F.; Whitaker, A.; Hall, S.J. An Introduction to Fermentation Processes. In Principles of Fermentation Technology, 3rd ed.; Stanbury, P.F., Whitaker, A., Hall, S.J., Eds.; Elsevier: London, UK, 2011; pp. 1–20. [Google Scholar]
- Paulová, L.; Patáková, P.; Brányik, T. Advanced Fermentation Processes. In Engineering Aspects of Food Biotechnology, 1st ed.; Teixeira, J.A., Vicente, A.A., Eds.; CRC Press: Boca Raton, FL, USA, 2013; pp. 89–105. [Google Scholar]
- Suárez-Arango, C.; Nieto, I.J. Biotechnological cultivation of edible macro-fungi: An alternative in obtaining nutraceuticals. Rev. Iberoam. Mycol. 2012, 30, 1–8. [Google Scholar]
- Bailon-Neira, R.C. Fermentaciones Industriales. Master’s Thesis, National University of Callao, Bellavista, Peru, April 2012. [Google Scholar]
- Ren, B.; Wu, M.; Wang, Q.; Peng, X.; Wen, H.; McKinstry, W.J.; Chen, Q. Crystal Structure of Tannase from Lactobacillus plantarum. J. Mol. Biol. 2013, 425, 2737–2751. [Google Scholar] [CrossRef] [PubMed]
- Farha, A.K.; Yang, Q.-Q.; Kim, G.; Li, H.-B.; Zhu, F.; Liu, H.-Y.; Gan, R.-Y.; Corke, H. Tannins as an alternative to antibiotics. Food Biosci. 2020, 38, 100751. [Google Scholar] [CrossRef]
- Lin, P.; Tian, X.-H.; Yi, Y.-S.; Jiang, W.-S.; Zhou, Y.-J.; Cheng, W.-J. Luteolin-induced protection of H2O2-induced apoptosis in PC12 cells and the associated pathway. Mol. Med. Rep. 2015, 12, 7699–7704. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Peng, X.; Wen, H.; Wang, Q.; Chen, Q.; McKinstry, W.J.; Ren, B. Expression, purification, crystallization and preliminary X-ray analysis of tannase from Lactobacillus plantarum. Acta Crystallogr. Sect. F Struct. Biol. Cryst. Commun. 2013, 69, 456–459. [Google Scholar] [CrossRef]
- Velmurugan, B.; Singh, R.P.; Agarwal, R.; Agarwal, C. Dietary-feeding of grape seed extract prevents azoxymethane-induced colonic aberrant crypt foci formation in fischer 344 rats. Mol. Carcinog. 2010, 49, 641–652. [Google Scholar] [CrossRef]
- Chávez-González, M.; Guyot, S.; Rodríguez-Herrera, R.; Prado-Barragán, A.; Aguilar, C.N. Production profiles of phenolics from fungal tannic acid biodegradation in submerged and solid-state fermentation. Process Biochem. 2014, 49, 541–546. [Google Scholar] [CrossRef]
- Rani, J.; Indrajeet; Rautela, A.; Kumar, S. Biovalorization of Winery Industry Waste to Produce Value-Added Products. In Biovalorisation of Wastes to Renewable Chemicals and Biofuels; Rathinam, N.K., Sani, R.K., Eds.; Elsevier BV: Amsterdam, The Netherlands, 2020; pp. 63–85. [Google Scholar]
- Rodríguez-Durán, L.V.; Valdivia-Urdiales, B.; Contreras-Esquivel, J.C.; Rodríguez-Herrera, R.; Aguilar, C.N. Química y biotecnología de la tanasa. Acta Química Mex. 2010, 2, 1–10. [Google Scholar]
- Aguilar-Zárate, P.; Cruz-Hernández, M.A.; Montañez, J.C.; Belmares-Cerda, R.E.; Aguilar, C.N. Bacterial tannases: Production, properties and applications. Rev. Mex. Ing. Quim. 2014, 13, 63–74. [Google Scholar]
- Singh, S.K.; Patra, A. Evaluation of phenolic composition, antioxidant, anti-inflammatory and anticancer activities of Polygonatum verticillatum (L.). J. Integr. Med. 2018, 16, 273–282. [Google Scholar] [CrossRef]
- Gouvinhas, I.; Queiroz, M.; Rodrigues, M.; Barros, A.I. Evaluation of the Phytochemistry and Biological Activity of Grape (Vitis vinifera L.) Stems: Toward a Sustainable Winery Industry. In Polyphenols in Plants, 2nd ed.; Watson, R.R., Ed.; Elsevier: London, UK, 2019; pp. 381–394. [Google Scholar]
- Shahidi, F.; Ambigaipalan, P. Phenolics and Polyphenolics in Foods, Beverages and Spices: Antioxidant Activity and Health Effects—A Review. J. Funct. Foods 2015, 18, 820–897. [Google Scholar] [CrossRef]
- Tyagi, A.; Raina, K.; Shrestha, S.P.; Miller, B.; Thompson, J.A.; Wempe, M.F.; Agarwal, R.; Agarwal, C. Procyanidin B2, 3,3¨ di –O- gallate, a biologically active constituent of grape seed extract, induces apoptosis in human prostate cancer cells via target in NK-kB, Stat3 and AP1 transcription factors. Nutr. Cancer 2014, 66, 736–746. [Google Scholar] [CrossRef] [PubMed]
- Cao, Z.; Zhang, H.; Cai, X.; Fang, W.; Chai, D.; Wen, Y.; Chen, H.; Chu, F.; Zhang, Y. Luteolin Promotes Cell Apoptosis by Inducing Autophagy in Hepatocellular Carcinoma. Cell Physiol. Biochem. 2017, 43, 1803–1812. [Google Scholar] [CrossRef] [PubMed]
- Salehi, B.; Vlaisavljevic, S.; Adetunji, C.O.; Adetunji, J.B.; Kregiel, D.; Antolak, H.; Pawlikowska, E.; Uprety, Y.; Mileski, K.S.; Devkota, H.P.; et al. Plants of the genus Vitis: Phenolic compounds, anticancer properties and clinical relevance. Trends Food Sci. Technol. 2019, 91, 362–379. [Google Scholar] [CrossRef]
- Chen, P.-T.; Hong, Z.-S.; Cheng, C.-L.; Ng, I.-S.; Lo, Y.-C.; Nagarajan, D.; Chang, J.-S. Exploring fermentation strategies for enhanced lactic acid production with polyvinyl alcohol-immobilized Lactobacillus plantarum 23 using microalgae as feedstock. Bioresour. Technol. 2020, 308, 123266. [Google Scholar] [CrossRef]
- Bao, X.M.; Wu, C.F.; Lu, G.P. Atorvastatin inhibits homocysteine-induced oxidative stress and apoptosis in endothelial progenitor cells involving Nox4 and p38MAPK. Atherosclerosis 2010, 210, 114–121. [Google Scholar] [CrossRef]
- Nunes, C.; Almeida, L.; Barbosa, R.M.; Laranjinha, J. Luteolin suppresses the JAK/STAT pathway in a cellular model of intestinal inflammation. Food Funct. 2017, 8, 387–396. [Google Scholar] [CrossRef]
- Ployon, S.; Morzel, M.; Belloir, C.; Bonnotte, A.; Bourillot, E.; Briand, L.; Lesniewska, E.; Lherminier, J.; Aybeke, E.; Canon, F. Mechanisms of astringency: Structural alteration of the oral mucosal pellicle by dietary tannins and protective effect of bPRPs. Food Chem. 2018, 253, 79–87. [Google Scholar] [CrossRef]
- Kim, H.-S.; Jeon, D.Y.; Javaid, H.M.A.; Sahar, N.E.; Lee, H.-N.; Hong, S.-J.; Huh, J.Y.; Kim, Y.-M. Bio-transformation of green tea infusion with tannase and its improvement on adipocyte metabolism. Enzym. Microb. Technol. 2020, 135, 109496. [Google Scholar] [CrossRef]
- Govindarajan, R.K.; Revathi, S.; Rameshkumar, N.; Krishnan, M.; Kayalvizhi, N. Microbial tannase: Current perspectives and biotechnological advances. Biocatal. Agric. Biotechnol. 2016, 6, 168–175. [Google Scholar] [CrossRef]
- Kiran, E.U.; Trzcinski, A.P.; Ng, W.J.; Liu, Y. Enzyme Production from Food Wastes Using a Biorefinery Concept. Waste Biomass Valoriz. 2014, 5, 903–917. [Google Scholar] [CrossRef]
- Sharma, K.P.; John, P.J. Purification and characterization of tannase and tannins from Enterobacter sp. Process Biochem. 2011, 46, 240–244. [Google Scholar] [CrossRef]
- Curiel, J.A.; Betancor, L.; Rivas, B.D.L.; Muñoz, R.; Guisán, J.M.; Fernandez-Lorente, G. Hydrolysis of Tannic Acid Catalyzed by Immobilized−Stabilized Derivatives of Tannase from Lactobacillus plantarum. J. Agric. Food Chem. 2010, 58, 6403–6409. [Google Scholar] [CrossRef] [PubMed]
- Chandrasekaran, M.; Beena, P. Tannase: Source, Biocatalytic Characteristics, and Bioprocesses for Production. In Marine Enzymes for Biocatalysis; Trincone, A., Ed.; Elsevier: London, UK, 2013; pp. 259–293. [Google Scholar]
- Jiménez-Martín, N. Metabolismo de Galotaninos en Bacterias Con Actividad Tanasa Presentes en El Tracto Gastrointestinal Humano. Ph.D. Thesis, Autonomous University of Madrid, Madrid, Spain, July 2014. [Google Scholar]
- Jiménez, N.; Esteban-Torres, M.; Mancheño, J.M.; Rivas, B.D.L.; Muñoz, R. Tannin Degradation by a Novel Tannase Enzyme Present in Some Lactobacillus plantarum Strains. Appl. Environ. Microbiol. 2014, 80, 2991–2997. [Google Scholar] [CrossRef] [PubMed]
- Suárez-Arango, C. Utilización de la Fermentación Líquida de Lentinula Edodes (Shiitake), para la Producción de Metabolitos Secundarios Bioactivos y Evaluación de su Potencial Empleo en la Producción de un Alimento Funcional. Master’s Thesis, National University of Colombia, Bogota, Colombia, 2012. [Google Scholar]
- Coradi, G.V.; Da Visitação, V.L.; De Lima, E.A.; Saito, L.Y.T.; Palmieri, D.A.; Takita, M.A.; Neto, P.D.O.; Nascimento, V.M.G. Comparing submerged and solid-state fermentation of agro-industrial residues for the production and characterization of lipase. Ann. Microbiol. 2012, 63, 533–540. [Google Scholar] [CrossRef]
- Pintać, D.; Četojević-Simin, D.; Berežni, S.; Orčić, D.; Mimica-Dukić, N.; Lesjak, M. Investigation of the chemical composition and biological activity of edible grapevine (Vitis vinifera L.) leaf varieties. Food Chem. 2019, 286, 686–695. [Google Scholar] [CrossRef]
- Selwal, M.K.; Selwal, K.K. High-level tannase production by Penicillum atramentosum KM using agro residues under submerged fermentation. Ann. Microbiol. 2012, 62, 139–148. [Google Scholar] [CrossRef]
- Hemansi; Chakraborty, S.; Yadav, G.; Saini, J.K.; Kuhad, R.C. Comparative Study of Cellulase Production Using Submerged and Solid-State Fermentation. In New and Future Developments in Microbial Biotechnology and Bioengineering; Srivastava, N., Srivastava, M., Singh, R.L., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 99–113. [Google Scholar]
- Barragán, L.P.; Figueroa, J.; Durán, L.R.; González, C.A.; Hennigs, C. Fermentative Production Methods. In Biotransformation of Agricultural Waste and By-Products; Poltronieri, P., D’Urso, O.F., Eds.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 189–217. [Google Scholar]
- Fernandes, M.B.A.; Habu, S.; De Lima, M.A.; Soccol, V.T.; Soccol, C.R. Influence of drying methods over in vitro antitumoral effects of exopolysaccharides produced by Agaricus blazei LPB 03 on submerged fermentation. Bioprocess Biosyst. Eng. 2010, 34, 253–261. [Google Scholar] [CrossRef]
- Colla, L.M.; Primaz, A.L.; Benedetti, S.; Loss, R.A.; De Lima, M.; Reinehr, C.O.; Bertolin, T.E.; Costa, J.A.V. Surface response methodology for the optimization of lipase production under submerged fermentation by filamentous fungi. Braz. J. Microbiol. 2016, 47, 461–467. [Google Scholar] [CrossRef]
- Chegwin, C.A.; Nieto, R.I.J. Influence of culture medium in the production of secondary metabolites of edible fungus Pleurotus ostreatus cultivated by liquid state fermentation using grain flour as a carbon source. Rev. Mex. Micol. 2013, 37, 1–9. [Google Scholar]
- Kirsch, L.D.S.; De Macedo, A.J.P.; Teixeira, M.F.S. Production of mycelial biomass by the Amazonian edible mushroom Pleurotus albidus. Braz. J. Microbiol. 2016, 47, 658–664. [Google Scholar] [CrossRef] [PubMed]
- Parras-Huertas, R.A. Review lactic acid bacteria: Functional role in the foods. Rev. Bio. Agro 2010, 8, 93–105. [Google Scholar]
- Lekshmi, R.; Nisha, S.A.; Kaaleeswaran, B.; Alfarhan, A. Pomegranate peel is a low-cost substrate for the production of tannase by Bacillus velezensis TA3 under solid state fermentation. J. King Saud. Univ. Sci. 2020, 32, 1831–1837. [Google Scholar] [CrossRef]
- Liu, J.; Luo, Y.; Guo, T.; Tang, C.; Chai, X.; Zhao, W.; Bai, J.; Lin, Q. Cost-effective pigment production by Monascus purpureus using rice straw hydrolysate as substrate in submerged fermentation. J. Biosci. Bioeng. 2020, 129, 229–236. [Google Scholar] [CrossRef]
- Chebaibi, S.; Grandchamp, M.L.; Burgé, G.; Clément, T.; Allais, F.; Laziri, F. Improvement of protein content and decrease of anti-nutritional factors in olive cake by solid-state fermentation: A way to valorize this industrial by-product in animal feed. J. Biosci. Bioeng. 2019, 128, 384–390. [Google Scholar] [CrossRef]
- Li, Y.; Peng, X.; Chen, H. Comparative characterization of proteins secreted by Neurospora sitophilia in solid-state and submerged fermentation. J. Biosci. Bioeng. 2013, 116, 493–498. [Google Scholar] [CrossRef]
- Joshi, R.; Sharma, V.; Kuila, A. Fermentation Technology: Current Status and Future Prospects. In Principles and Applications of Fermentation Technology, 1st ed.; Kuila, A., Sharma, V., Eds.; Wiley: Hoboken, NJ, USA, 2019; pp. 1–13. [Google Scholar]
- Mansor, A.; Ramli, M.; Rashid, N.A.; Samat, N.; Lani, M.; Sharifudin, S.; Raseetha, S. Evaluation of selected agri-industrial residues as potential substrates for enhanced tannase production via solid-state fermentation. Biocatal. Agric. Biotechnol. 2019, 20, 101216. [Google Scholar] [CrossRef]
- Govindarajan, R.K.; Mathivanan, K.; Rameshkumar, N.; Shyu, D.J.H.; Krishnan, M.; Kayalvizhi, N. Purification, structural characterization and biotechnological potential of tannase enzyme produced by Enterobacter cloacae strain 41. Process Biochem. 2019, 77, 37–47. [Google Scholar] [CrossRef]
- Zhang, B.B.; Xing, H.B.; Jiang, B.J.; Chen, L.; Xu, G.R.; Jiang, Y.; Zhang, D.Y. Using millet as substrate for efficient production of monacolin K by solid-state fermentation of Monascus ruber. J. Biosci. Bioeng. 2018, 125, 333–338. [Google Scholar] [CrossRef]
- Julian, R.M.C.; Ramos-Sánchez, L.B.; Suárez-Rodríguez, Y. Fermentación en estado sólido (II) optimización de medios de cultivos. Tecnol. Química 2008, 28, 5–10. [Google Scholar]
- Ito, K.; Gomi, K.; Kariyama, M.; Miyake, T. Rapid enzyme production and mycelial growth in solid-state fermentation using the non-airflow box. J. Biosci. Bioeng. 2013, 116, 585–590. [Google Scholar] [CrossRef] [PubMed]
- Ito, K.; Gomi, K.; Kariyama, M.; Miyake, T. Change in enzyme production by gradually drying culture substrate during solid-state fermentation. J. Biosci. Bioeng. 2015, 119, 674–677. [Google Scholar] [CrossRef] [PubMed]
- Martins, I.M.; Macedo, G.A.; Macedo, J.A.; Roberto, B.; Chen, Q.; Blumberg, J.B.; Chen, C. Tannase enhances the anti-inflammatory effect of grape pomace in Caco-2 cells treated with IL-1b. J. Funct. Foods 2016, 29, 69–76. [Google Scholar] [CrossRef]
- Ichikawa, K.; Shiono, Y.; Shintani, T.; Watanabe, A.; Kanzaki, H.; Gomi, K.; Koseki, T. Efficient production of recombinant tannase in Aspergillus oryzae using an improved glucoamylase gene promoter. J. Biosci. Bioeng. 2020, 129, 150–154. [Google Scholar] [CrossRef] [PubMed]
- Kanpiengjai, A.; Unban, K.; Nguyen, T.-H.; Haltrich, D.; Khanongnuch, C. Expression and biochemical characterization of a new alkaline tannase from Lactobacillus pentosus. Protein Expr. Purif. 2019, 157, 36–41. [Google Scholar] [CrossRef]
- Sharma, K. Tannin degradation by phytopathogen’s tannase: A Plant’s defense perspective. Biocatal. Agric. Biotechnol. 2019, 21, 101342. [Google Scholar] [CrossRef]
- López, T.; Prado-Barragán, A.; Nevárez-Moorillón, G.V.; Contreras, J.C.; Rodríguez, R.; Aguilar, C.N. Enhancement of antioxidant capacity of coffee pulp extracts by solid-state lactic fermentation. CYTA J. Food 2013, 11, 359–365. [Google Scholar] [CrossRef]
- Jurado-Gámez, H.; Ramírez, C.; Aguirre, D. Fermentation kinetics of Lactobacillus plantarum at a rich culture medium as a potential probiotic. Vet. Anim. Sci. 2013, 7, 37–53. [Google Scholar]
- Bamforth, C.W.; Cook, D.J. The Science Underpinning Food Fermentation. In Food, Fermentation, and Micro-Organisms, 2nd ed.; Bamforth, C.W., Cook, D.J., Eds.; Wiley Blackwell: Hoboken, NJ, USA, 2019; pp. 5–42. [Google Scholar]
- Mayo, B.; Flórez, A.B. Lactic Acid Bacteria: Lactobacillus spp. Lactobacillus plantarum. In Reference Module in Food Science; Elsevier: Amsterdam, The Netherlands, 2020. [Google Scholar]
- Zhang, Z.; Man, C.; Sun, L.; Yang, X.; Li, M.; Jiang, Y.; Zhang, W. Short communication: Complete genome sequence of Lactobacillus plantarum J26, a probiotic strain with immunomodulatory activity. J. Dairy Sci. 2019, 102, 10838–10844. [Google Scholar] [CrossRef]
- Pérez-Leonard, H.; Hernández-Monzón, A. Evaluation of substrates with aloe vera juice for the growth of Lactobacillus plantarum. RTQ 2015, 35, 156–166. [Google Scholar]
- Yao, J.; Guo, G.S.; Ren, G.H.; Liu, Y. Production, characterization and applications of tannase. J. Mol. Catal. B Enzym. 2014, 101, 137–147. [Google Scholar] [CrossRef]
- Sadeghi, A.; Ebrahimi, M.; Mortazavi, S.A.; Abedfar, A. Application of the selected antifungal LAB isolate as a protective starter culture in pan whole-wheat sourdough bread. Food Control 2019, 95, 298–307. [Google Scholar] [CrossRef]
- Arrioja-Bretón, D.; Mani-López, E.; Palou, E.; López-Malo, A. Antimicrobial activity and storage stability of cell-free supernatants from lactic acid bacteria and their applications with fresh beef. Food Control 2020, 115, 107286. [Google Scholar] [CrossRef]
- Moradi, M.; Guimarães, J.T.; Sahin, S. Current applications of exopolysaccharides from lactic acid bacteria in the development of food active edible packaging. Curr. Opin. Food Sci. 2021, 40, 33–39. [Google Scholar] [CrossRef]
- DelCarlo, S.B.; Parada, R.; Schelegueda, L.I.; Vallejo, M.; Marguet, E.R.; Campos, C.A. From the isolation of bacteriocinogenic LAB strains to the application for fish paste biopreservation. LWT Food Sci. Technol. 2019, 110, 239–246. [Google Scholar] [CrossRef]
- Thirabunyanon, M.; Hongwittayakorn, P. Potential Probiotic Lactic Acid Bacteria of Human Origin Induce Antiproliferation of Colon Cancer Cells via Synergic Actions in Adhesion to Cancer Cells and Short-Chain Fatty Acid Bioproduction. Appl. Biochem. Biotechnol. 2013, 169, 511–525. [Google Scholar] [CrossRef]
- Albano, C.; Morandi, S.; Silvetti, T.; Casiraghi, M.; Manini, F.; Brasca, M. Lactic acid bacteria with cholesterol-lowering properties for dairy applications: In vitro and in situ activity. J. Dairy Sci. 2018, 101, 10807–10818. [Google Scholar] [CrossRef]
- Wang, Q.; Lillevang, S.K.; Rydtoft, S.M.; Xiao, H.; Fan, M.; Solem, C.; Liu, J.; Jensen, P.R. No more cleaning up—Efficient lactic acid bacteria cell catalysts as a cost-efficient alternative to purified lactase enzymes. Appl. Microbiol. Biotechnol. 2020, 104, 6315–6323. [Google Scholar] [CrossRef]
- Gregirchak, N.; Stabnikova, O.; Stabnikov, V. Application of Lactic Acid Bacteria for Coating of Wheat Bread to Protect it from Microbial Spoilage. Plant Foods Hum. Nutr. 2020, 75, 223–229. [Google Scholar] [CrossRef]
- Lim, Y.H.; Foo, H.L.; Loh, T.C.; Mohamad, R.; Abdullah, N. Comparative studies of versatile extracellular proteolytic activities of lactic acid bacteria and their potential for extracellular amino acid productions as feed supplements. J. Anim. Sci. Biotechnol. 2019, 10, 1–13. [Google Scholar] [CrossRef]
- Bhoite, R.N.; Murthy, P.S. Biodegradation of coffee pulp tannin by Penicillium verrucosum for production of tannase, statistical optimization and its application. Food Bioprod. Process. 2015, 94, 727–735. [Google Scholar] [CrossRef]
- Azzaz, H.; Kholif, A.; El Tawab, A.A.; Khattab, M.; Murad, H.; Olafadehan, O. A newly developed tannase enzyme from Aspergillus terreus versus commercial tannase in the diet of lactating Damascus goats fed diet containing pomegranate peel. Livest. Sci. 2020, 241, 104228. [Google Scholar] [CrossRef]
- Albuquerque, K.K.S.A.; Albuquerque, W.W.C.; Costa, R.M.P.B.; Batista, J.M.S.; Marques, D.A.V.; Bezerra, R.P.; Herculano, P.N.; Porto, A.L.F. Biotechnological potential of a novel tannase-acyl hydrolase from Aspergillus sydowii using waste coir residue: Aqueous two-phase system and chromatographic techniques. Biocatal. Agric. Biotechnol. 2020, 23, 101453. [Google Scholar] [CrossRef]
- Martins, I.M.; Macedo, G.A.; Macedo, J.A. Biotransformed grape pomace as a potential source of anti-inflammatory polyphenolics: Effects in Caco-2 cells. Food Biosci. 2020, 35, 100607. [Google Scholar] [CrossRef]
- Natarajan, K.; Rajendran, A. Evaluation and optimization of food-grade tannin acyl hydrolase production by a probiotic Lactobacillus plantarum strain in submerged and solid state fermentation. Food Bioprod. Process. 2012, 90, 780–792. [Google Scholar] [CrossRef]
- Selvaraj, S.; Natarajan, K.; Nowak, A.; Murty, V.R. Mathematical modeling and simulation of newly isolated Bacillus cereus M1GT for tannase production through semi-solid state fermentation with agriculture residue triphala. S. Afr. J. Chem. Eng. 2020. [Google Scholar] [CrossRef]
- Teles, A.S.; Chávez, D.W.; Oliveira, R.A.; Bon, E.P.; Terzi, S.C.; Souza, E.F.; Gottschalk, L.M.; Tonon, R.V. Use of grape pomace for the production of hydrolytic enzymes by solid-state fermentation and recovery of its bioactive compounds. Food Res. Int. 2019, 120, 441–448. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).