Abstract
Cashew apples are by-products in the production of cashew nuts, which are mostly left to rot in the fields. Cashew apple juice (CAJ), a highly nutritious beverage, can be produced from them. It is rich in sugars and ascorbic acid, but its high polyphenol content makes it bitter and astringent, and therefore difficult to commercialize. The kingdom of fungi contains more than 2000 yeast species, of which only a few species have been studied in relation to their potential to produce aroma compounds. The aim of this research was to develop a new low-alcoholic fermented beverage to valorize cashew apples. For this purpose, a screening was carried out employing non-conventional yeast species and some species of the genus Saccharomyces for comparison, followed by a more detailed study with four selected strains cultured at different conditions. The production of volatile aroma compounds as a function of the presence of oxygen, temperature, and yeast species was investigated. The results showed that the more diverse aroma profiles appeared at 25 °C under anaerobic cultivation conditions, where Saccharomyces cerevisiae WUR 102 and Hanseniaspora guilliermondii CBS 2567 excelled in the synthesis of certain aroma compounds, such as β-phenylethanol and its acetate ester (rose aroma). Further studies are needed to test consumer acceptance of these new products.
1. Introduction
Cashew (Anacardium occidentale L.) is a native crop from tropical America, widely available in several countries of Asia, Africa, and Central America [1]. The most important product from Anacardium occidentale L. is the cashew nut. However, the tree also yields a pseudo-fruit called cashew apple to which the nut is attached, which can be either yellow, orange, or bright red [1]. It is a fleshy, fibrous and highly juicy fruit with a soft peel [2], which can be consumed raw or in the form of fresh juice, jam, syrups, candied fruit, jelly, pectin, soft drinks, or other beverages. However, the cashew apple is very bitter and astringent due to its high polyphenol content and therefore, not as palatable as other fruits. For this reason, the cashew apple is almost neglected in commercial terms as compared to the nut. Its industrialization represents not even 10% of the annual Brazilian production [2]. According to official data, 90% of the annual production of about two million tons of cashew apples in Northeast Brazil are lost or underutilized [3]. In 2006, the world production was estimated to be 30 million tons [4,5]. Being considered as an agriculture residue, it leads to a large amount of waste. New products and processes are needed to reduce this high wastage.
Peeling of cashew apples and clarification of cashew apple juice (CAJ) are strategies that can be used to reduce polyphenol content and therefore the bitterness and astringency of the product. The skin of cashew apple contains much higher tannin concentration (516–802 mg/100 g) than the flesh (149–155 mg/100 g) [5] and among several clarification methods, the use of gelatin showed the highest rate of success, being cheap and easily available in Benin [6,7]. However, other innovative strategies, such as the use of microorganisms to decrease polyphenols, could be considered. For instance, yeasts have been demonstrated to be able to metabolize these compounds [8,9,10].
The most attractive property of cashew apple is its extremely high ascorbic acid content, which is about three to six times that of orange juice and about 10-fold the content in pineapple juice [1]. It also contains thiamine, niacin, and riboflavin in addition to significant concentrations of minerals, such as copper, zinc, sodium, potassium, calcium, iron, phosphorous, and magnesium [1]. Furthermore, its capabilityin reducing sugars (glucose and fructose) makes the substrate appropriate for alcoholic fermentation [11]. In this way, cashew apple would be an excellent, highly available, and low-cost substrate for the production of fermented beverages. In addition, cashew apple juice (CAJ) is believed to have several beneficial properties, such as antibacterial, antifungal, antitumoral, antioxidant, and antimutagenic actions [12,13,14].
CAJ has been previously used to produce metabolites of interest such as lactic acid, oxalic acid, dextran, mannitol, oligosaccharides, and a biosurfactant in fermentations mediated by bacteria of the genera Lactobacillus, Leuconostoc, Pseudomonas or Bacillus [3,11,15,16,17,18]. Moreover, yeast species of the genera Saccharomyces and Hanseniaspora have been demonstrated to have the capacity of producing bioethanol when growing in this substrate [19,20,21]. Finally, CAJ has been used to produce probiotic beverages employing lactic acid bacteria [22,23,24,25,26] and alcoholic beverages using the yeasts Saccharomyces cerevisiae and S. bayanus [2,27,28].
Non-conventional yeasts are those that are not used commonly in industrial processes, although some of them present interesting properties, thereby making them an untapped potential for food applications [29]. There is evidence that these species are able to carry out fermentations of sugary substrates producing very diverse aroma profiles [30,31,32,33].
The main objective of the current study is to develop a locally feasible process for the exploitation and valorization of cashew apples employing Saccharomyces and non-conventional yeasts to develop an innovative low-alcoholic fermented beverage rich in vitamin C.
2. Materials and Methods
2.1. Production and Characterization of Cashew Apple Juice
2.1.1. Plant Materials
Twenty kilograms of yellow cashew apples and twenty kilograms of red cashew apples were harvested at the mature stage in Benin, immediately frozen, and sent to the laboratory in Wageningen, The Netherlands. Ten frozen cashew apples of each batch were randomly chosen and rapidly peeled by grating the epidermis with a razor blade. Next, the frozen flesh was quickly cut into bits, immediately dipped in liquid nitrogen to prevent oxidation, and homogenized to a very fine powder with a blender. Part of the flesh powder was freeze-dried at −20 °C, while the rest was kept at room temperature for juice extraction.
2.1.2. Juice Extraction and Clarification
CAJ was obtained through a mechanical process and clarified by adding gelatin to remove polyphenols and suspended solids [3]. The extraction process was modified according to a local method used in Benin, which is based on direct pressing of the fruits [34]. Peeled cashew apples were cut and ground by a mixer. Then, 1% (w/v) gelatin was added to CAJ, stirred for 15 min, and left to settle for 10–15 min at 4 °C. Next, CAJ was filtered through a cheese cloth.
The clarified CAJ was physicochemically characterized and stored frozen (−20 °C) for further analytical studies. Dimethyl dicarbonate (DMDC) was added to CAJ for sterilization and to prevent vitamin loss.
2.1.3. Sugar Determination
Glucose, fructose, and sucrose were determined by HPLC (Thermo Fisher Scientific, Waltham, MA, USA) equipped with a P-2000 pump and ELSD-2100 polymer labs detector. Separations were carried out in a 250 mm × 4.6 mm Alltech prevail carbohydrates column, with an evaporator temperature of 80 °C and nebulizer temperature of 60 °C. The running time was 14 min with a flow rate 1 mL/min on isocratic 75%/25% acetonitrile/water. In brief, 1 mL of sample was mixed with distilled water of about 80 °C, after which the solution was incubated at 80 °C in a water bath for 5 min, then homogenized with an Ultra Turrax T20B for 1 min and centrifuged (ALC PK131R) for 5 min at 2255× g at 20 °C. The extract was diluted up to eighty times with distilled water. For the external standard, sucrose, glucose, and fructose with the range 45–680 μg/mL were used. Subsequently, 2 mL of samples or standards were filtered through 0.45 μm filter and used for HPLC analysis.
2.1.4. Total Polyphenol Determination
Total phenolic content was determined using the Folin–Ciocalteu method as described by Georgé et al. [35], with some modifications. A volume of 0.25 mL clarified CAJ was diluted 1:4 with distilled water. A calibration curve was made, employing tannic acid with a concentration range from 0.03125 mg/mL to 1 mg/mL. The reaction mixture was composed of 1 mL of the sample or standard, 30 mL of distilled water, and 1 mL of Folin–Ciocalteu reagent. After 15 min, the intensity of the blue color that had developed was measured spectrophotometrically (Cary 50-UV visible, Varian, Palo Alto, CA, USA) at 725 nm. The total phenolic content was expressed as Gallic acid equivalents (GAE) per ml of sample.
2.1.5. Ascorbic Acid Determination
The method used for the determination of ascorbic acid (AA) content was described by Hernández et al. [36], with some modifications. An amount of 2.5 mL of sample was mixed with 2.5 mL of the extracting solution containing 3% MPA (metaphosphoric acid) and 1 mM TBHQ (tert-butylhydroquinone) in 10 mL tubes. After homogenizing, the mixture was centrifuged for 5 min at 2255× g at 4 °C. The extract was diluted up to 8 times with distilled water. All extractions were carried out employing ice and under reduced light. For the calibration, commercial L-ascorbic acid was prepared with a concentration range of 1.56–200 μg/mL. Subsequently, 2 mL of the sample or standard were filtered through 0.45 μm filter paper and used for HPLC analysis.
The HPLC system was from Thermo Fisher Scientific P-2000 (USA), equipped with a binary gradient pump and UV 2000 detector. Separations were carried out on a 150 mm × 4.6 mm Varian Polaris C18-A column, with 5.5 min running time and 20 μL injection volumes using an autosampler. The mobile phase employed was 0.2% orthophosphoric acid in distilled water. The flow rate of the mobile phase was 1 mL/min. A UVdetector at a wavelength of 245 nm was employed. The AA peak was identified by comparing its UV-visible spectral characteristics and retention time with the commercial standard of AA.
2.2. Fermentation of Cashew Apple Juice
2.2.1. Yeast Strains
The yeast strains employed in this study are listed in Table 1. The three Saccharomyces cerevisiae strains were from the culture collection of the Laboratory of Food Microbiology of Wageningen University, whereas one S. bayanus and 21 non-saccharomyces yeasts were supplied by Westerdijk Fungal Biodiversity Institute-CBS-KNAW, Utrecht, The Netherlands.

Table 1.
Yeast strains employed in this study.
2.2.2. Yeast Screening
Prior to fermentation, CAJ was treated with dimethyl dicarbonate (DMDC) (1ml/L) and stored overnight at 4 °C to inhibit the growth of undesirable microorganisms. Then, juice was inoculated with the chosen strains that had been precultured overnight in GPY medium (2% glucose, 1% bacteriological peptone, 0.5% yeast extract) at optical density at 600 nm (OD600 nm) of 0.1. The 25 yeast cultures were inoculated in 5 mL of yellow CAJ employing microplates. The fermentations were carried out at 25 °C during 7 days in microaerophilic conditions. Out of the 25, four strains were selected to carry out further research. The selection criteria were the following: Growth kinetics and fermentation performance (ethanol content). The fermentations were repeated with the four selected strains, however not only at 25 °C but also at 30 °C and 37 °C under strictly anaerobic conditions employing anaerobic jars. In addition, experiments were carried out at 25 °C under aerobic conditions shaking the microplates.
The first screening was carried out without taking care of creating an anaerobic environment for the fermentations in order to simplify and make a fast selection. The fermentations with the selected yeasts were carried out employing two extreme conditions (strictly anaerobic and aerobic) in order to compare the effect of the oxygen in fermentation performance and aroma profile.
2.2.3. Growth Kinetics
A spectrophotometer (Bio-screen, Los Angeles, CA, USA) was used to determine the growth kinetics by automatically measuring the OD600 nm at 25 °C during 70 h at 15 min intervals. The data generated were then converted into Microsoft Excel format (Microsoft, Redmond, WA, USA) and processed into growth curves to calculate the specific growth rates of the yeast cultures.
2.2.4. Ethanol Determination
The ethanol content in CAJ was determined by HPLC. Samples were deproteinized using Carrez reagents, which precipitated proteins and colloidal compounds. After 5 min centrifugation, the clear supernatant was diluted 1:1 and transferred to an HPLC vial. The HPLC system used was a Thermo Fisher Scientific (Waltham, MA, USA) equipped with Ultimate 3000 (Dionex), using a 300 mm × 7.8 mm Aminex HPX-87H column (Bio-rad, Hercules, CA, USA) with pre-column. A 5 mM H2SO4 was used as eluent at a flow rate of 0.6 mL/min at 40 °C. Detection was by refractive index (Shodex RI 101). Ethanol in the range 1%–10% was used as external standard. Subsequently about 200 μL of samples or standards were used for HPLC analysis. The ethanol peaks were identified and quantified by comparing retention times with those of the external standards.
2.2.5. Aroma Analysis
Aroma compounds were determined by headspace solid-phase dynamic extraction gas chromatography–mass spectrometry (HS-SPDE-GC-MS) employing a 2.5 mL HS syringe with a polydimethylsiloxane active charcoal (PDMS/AC) coated needle (Chromtech, Bad Camberg, Germany). The incubation of the samples was at 60 °C during 15 min at 500 rpm. Afterwards, the sampling was carried out by taking 1 mL of the headspace at 200 μL/s five times. The needle was then desorbed and the headspace injected into the GC column at 25 μL/s. GC–MS employed was a Finnigan Trace GC ultra (Thermo Fisher Scientific, Waltham, MA, USA) equipped with a 20 cm pre-column (CP-Sil 5CB 0.53 mm; df = 1 μm), a VF-1ms (30 m × 0.25 mm; df = 1 μm) capillary column (Varian) and a Combi PAL autosampler (CTC Analytics, Zwingen, Switzerland) in combination with a split injector in splitless mode (1 min) at 250 °C. The carrier gas was helium at a constant flow rate of 1.5 mL/min. The GC oven was initially set at 40 °C for 2 min, raised to 250 °C (10 °C/min) and then kept at 250 °C for 5 min. The total runtime was 28 min. Mass spectral data werecollected over a range of m/z 35–300 in full scan mode (scan time 0.25 s).
2.2.6. Other Analytical Determinations
Residual sugars, total polyphenols, and ascorbic acid were determined in the fermented products with the four selected strains following the same methodology as described in Section 2.1.3, Section 2.1.4 and Section 2.1.5, respectively.
2.3. Statistical Analysis
One-way ANOVA at a 95% confidence level and a Tukey test were employed to compare the composition of the fermented products, whereas multifactorial ANOVA was carried out to determine which factors were significantly affecting the composition of fermented products and the aroma profiles. Finally, principal component analysis (PCA) was used to group the different CAJ and fermented products at the different conditions. All tests were done using XLSTAT (Microsoft, Redmond, WA, USA).
3. Results and Discussion
3.1. Composition of Clarified Cashew Apple Juice
The yellow juice presented several advantages over the red juice, namely a lower polyphenol content and higher ascorbic acid and sugar concentration (Table 2). Consequently, yellow juice was selected to perform the fermentations.

Table 2.
Composition of cashew apple juice (CAJ) from yellow and red varieties of cashew apples.
3.1.1. Sugar Content
The major sugars in the CAJ were glucose and fructose. The obtained quantities were slightly higher than the values reported by Azevedo and Rodrigues [37], who found around 40 g/L for each of these sugars. Usually, fruits such as orange, apple, and pineapple contain more sucrose, which leads to a sweeter taste. This fruit only contains a small amount of sucrose (around 0.8 g/L), resulting in less masking of bitterness. On the other hand, the fact that monosaccharides are the major sugars in CAJ is an advantage for the fermentation since yeasts can directly consume them.
3.1.2. Total Polyphenol
The total phenolic content of fresh yellow CAJ was 1.6 mg/mL, whereas red CAJ contained 2.3 mg/mL (Table 2). These values were in the range of 1.7 to 2.4 mg/mL reported by Adou et al. [34], who also found that the yellow juice had a lower polyphenol concentration than the red one.
3.1.3. Ascorbic Acid
There was a large difference in the concentration of AA between red and yellow CAJ. The ascorbic acid concentration in yellow juice was 1.1 mg/mL whereas red juice only contained 0.45 mg/mL (Table 2). Assunçao et al. [38] reported slightly higher AA levels in yellow CAJ when compared to red CAJ. However, our results showed large differences. According to the same authors, several environmental factors can account for AA levels, such as geographic location, solar intensity, temperature, and soil type.
3.2. Yeast Screening
Twenty-five yeast strains (Table 1) were used to ferment yellow CAJ at 25 °C during seven days under microaerophilic conditions. Four of them were selected for further studies on the basis of their growth kinetics and sugar conversion to ethanol. The selected strains were Hanseniaspora guilliermondii CBS 2567, Torulaspora microellipsoides CBS 2734 and CBS 6641, and Saccharomyces cerevisiae WUR 102.
3.2.1. Growth Kinetics
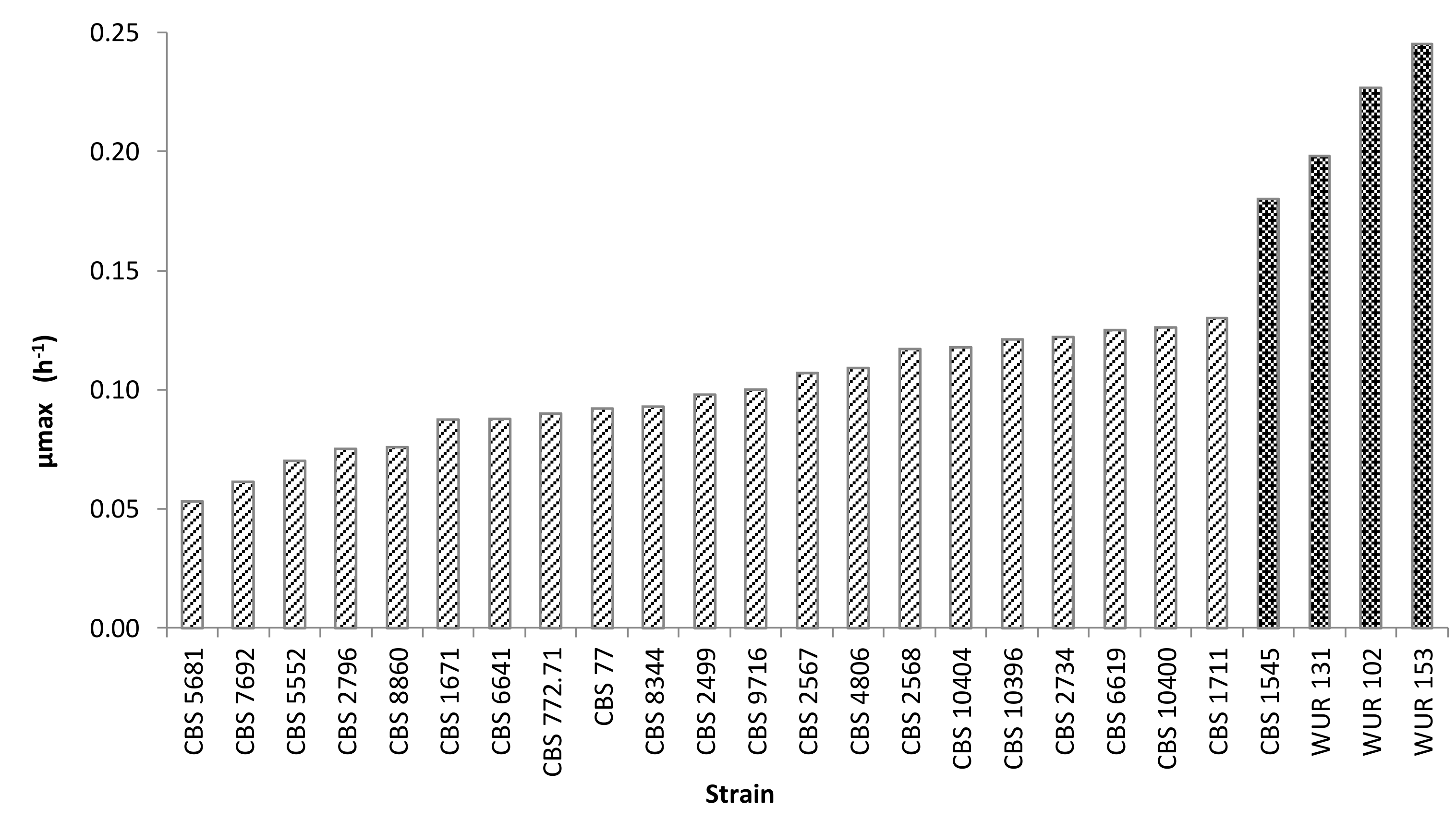
The growth kinetics of cultures of all strains were tracked by OD measurements over time at 25 °C for 70 h. The strains could be separated into two groups based on their maximum OD values. The strains of the group with the highest maximum OD values (over 1.2) were considered as good growers whereas those with lower maximum OD values apparently could not make proper use of the carbon source in CAJ and were therefore not selected for further research. The specific growth rate was another important criterion for selection. Cultures of strains with higher specific growth rates reproduce faster, which could save time in the fermentation process. The specific growth rates of non-Saccharomyces strains ranged from 0.05 to 0.13 h−1, whereas Saccharomyces strains presented values of at least 0.18 h−1. Specific growth rates of all cultures are presented in Figure 1.
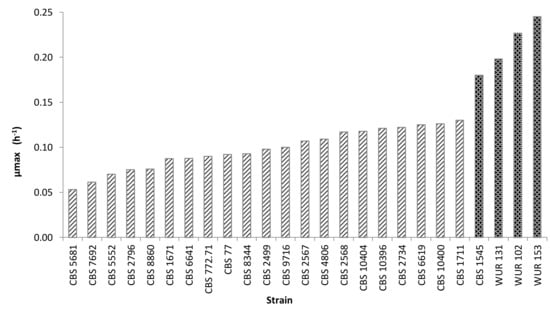
Figure 1.
Growth rates of the strains employed in the screening (yellow CAJ, 25°C). Striped bars indicate non-conventional yeasts; dark bars, Saccharomyces strains.
Interestingly, some strains showed diauxic growth, such as Hanseniaspora guilliermondii CBS 2567. Diauxic growth is observed when an organism is grown in a medium containing two carbon sources and there is a preferential utilization of one carbon source before utilizing the other. This preferred carbon source is consumed first, which leads to rapid growth, followed by a lag phase. After that, the microorganism experiences a slower growth phase, during which the second carbon source is metabolized [39]. The presence of this behavior was taken into account for the yeast selection.
3.2.2. Sugar Conversion to Ethanol
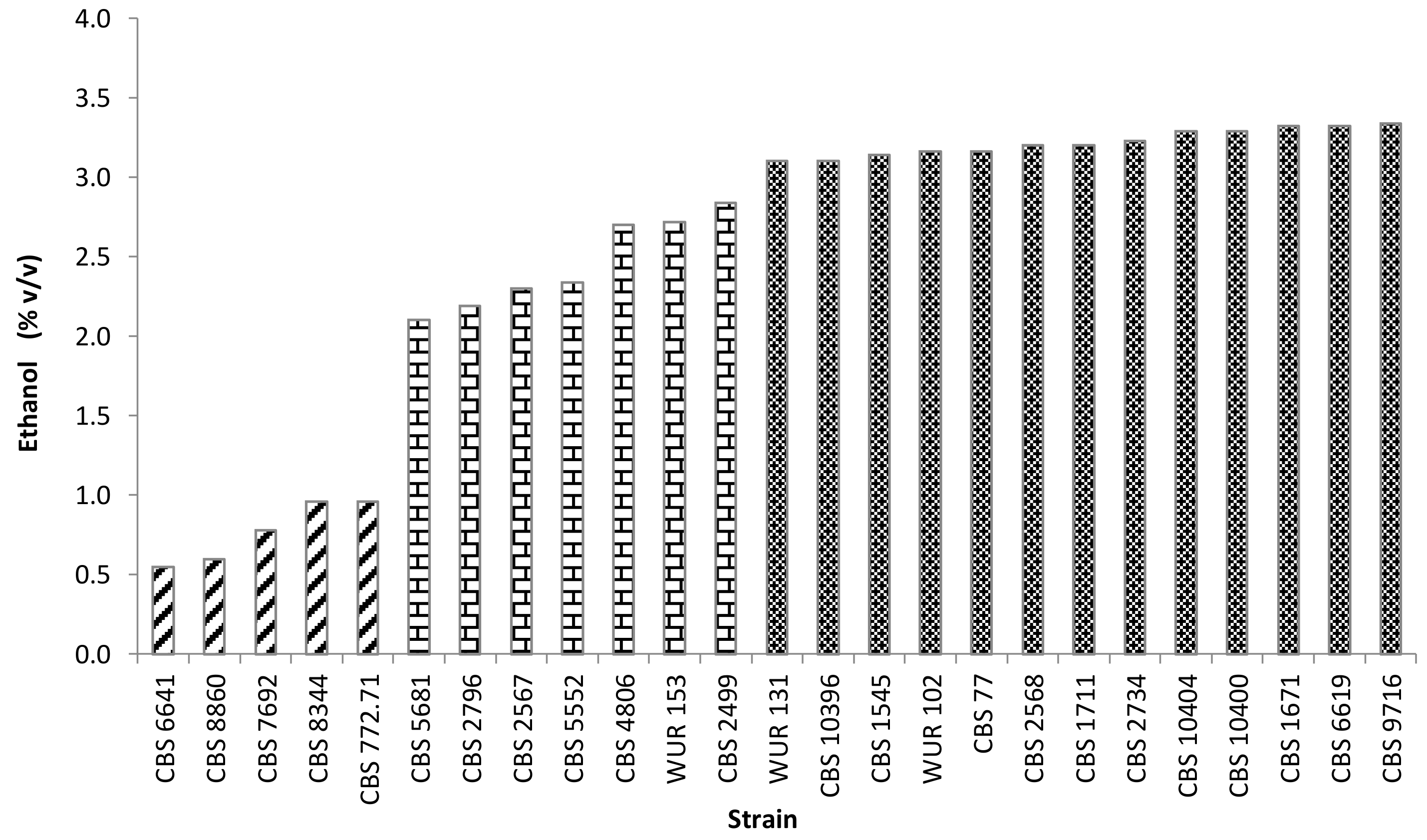
Yellow CAJ contained 112 g/L of sugars (glucose + fructose) prior to fermentation. Most of the strains were able to ferment all the sugars whereas a few strains were not (CBS 772.71, CBS 7692, CBS 8344, and CBS 8860). All of the latter strains belonged to the group with low maximum OD values and low specific growth rates. The conversion of sugars to ethanol reflects the alcoholic fermentation performance of the strains. The strains could be divided into three groups based on their ethanol yield: High, intermediate, and low producers (Figure 2). However, there was an interesting strain, Torulaspora microellipsoides CBS 6641, in the low ethanol producing group. Unlike the other strains in this group, CBS 6641 did grow well and consumed all the sugar in the juice but produced alow ethanol amount during fermentation. There might be two reasons. Firstly, because the conditions were not strictly anaerobic, oxygen might have affected the ethanol production by some yeasts while others produced ethanol even with oxygen in the medium due to the “Crabtree effect” [40]. Secondly, it might be due to the redirection of the metabolic flux towards the production of other metabolites, such as organic acids (acetic acid) or glycerol [41,42,43]. Consequently, CBS 6641 was considered an interesting strain for further research.
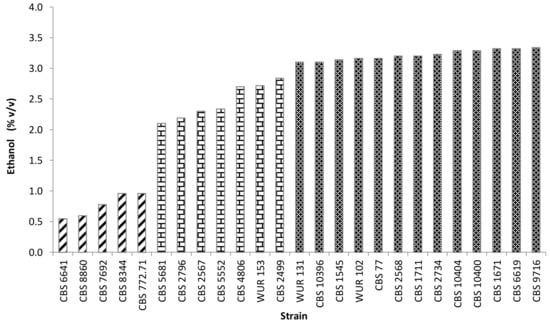
Figure 2.
Production during yeast fermentation in yellow cashew apple juice at 25 °C during 7 days. Striped bars depict low ethanol producers; bricked bars, intermediate ethanol producers; dark bars, high ethanol producers.
3.2.3. Yeast Selection
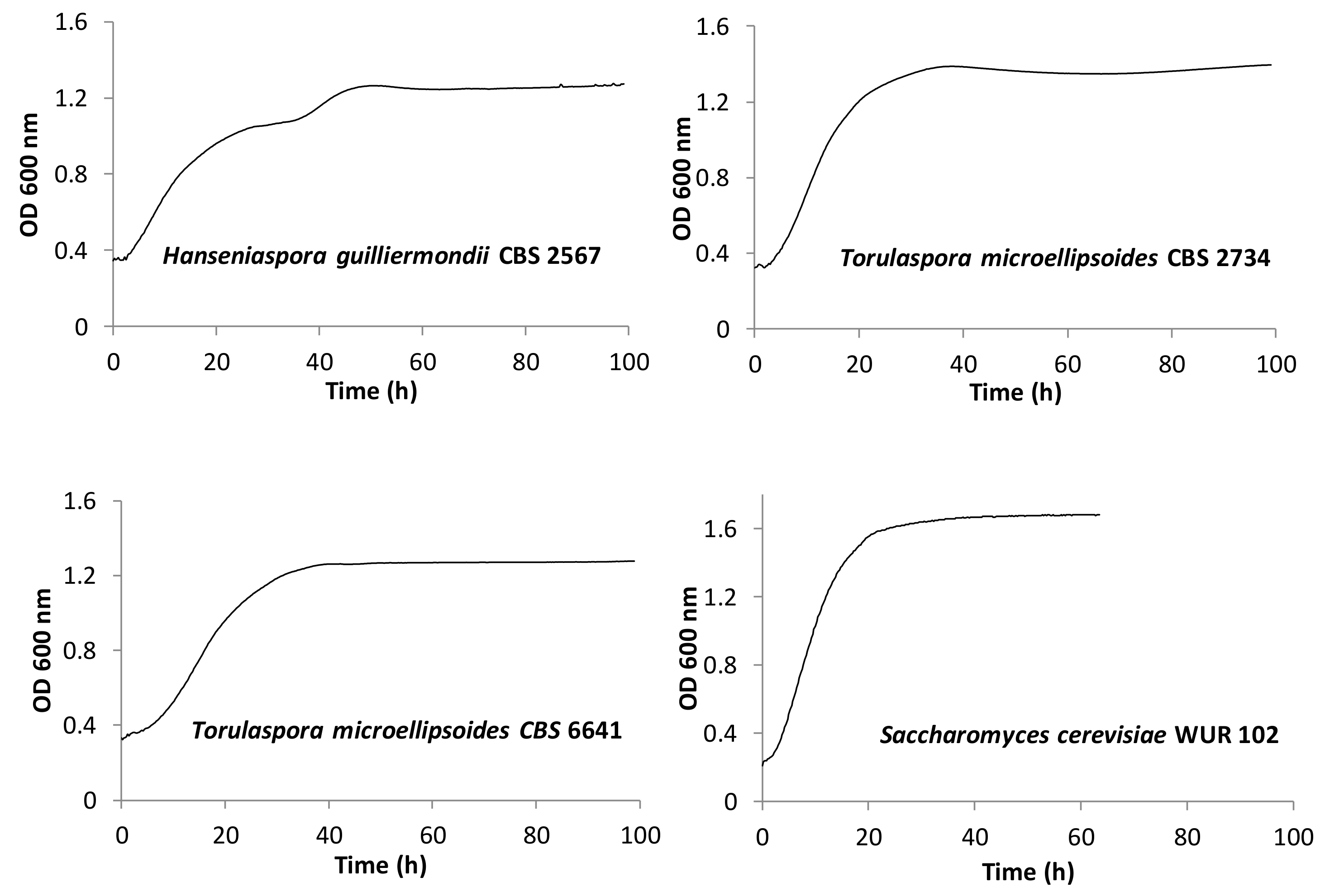
Based on the combined results, four strains were selected for further research. The selected strains were three non-conventional strains and one Saccharomyces strain. Table 3 shows the criteria taken into account for the selection and Figure 3 depicts their growth curves. Among the non-conventional yeast strains, the selected yeasts were Torulaspora microellipsoides CBS 2734 and CBS 6641 and Hanseniaspora guilliermondii CBS 2567, which showed good growth and high, low, and medium ethanol production, respectively. In addition, CBS 2567 was selected for its diauxic growth. Finally, the selected Saccharomyces cerevisiae WUR 102 showed a high growth and fermentation performance, thus representing the high ethanol producers.

Table 3.
Yeast strains from the screening.
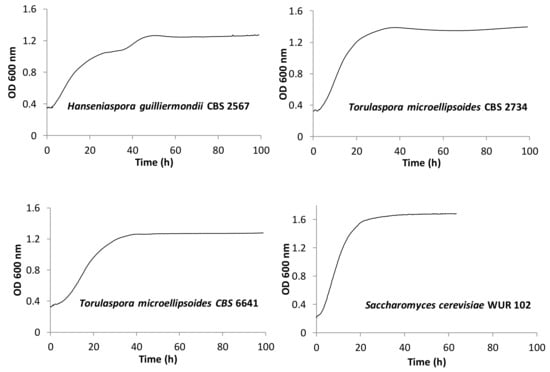
Figure 3.
Kinetics of the selected strains at 25 °C employing yellow cashew apple juice.
3.3. Cashew Apple Juice Fermentation with the Selected Strains
Fermentation of yellow CAJ as substrate using the four selected strains was carried out at different temperatures, namely 25 °C, 30 °C, and 37 °C, under strictly anaerobic conditions. In addition, the four strains were grown in yellow CAJ at 25 °C under aerobic conditions.
3.3.1. Sugar Conversion to Ethanol
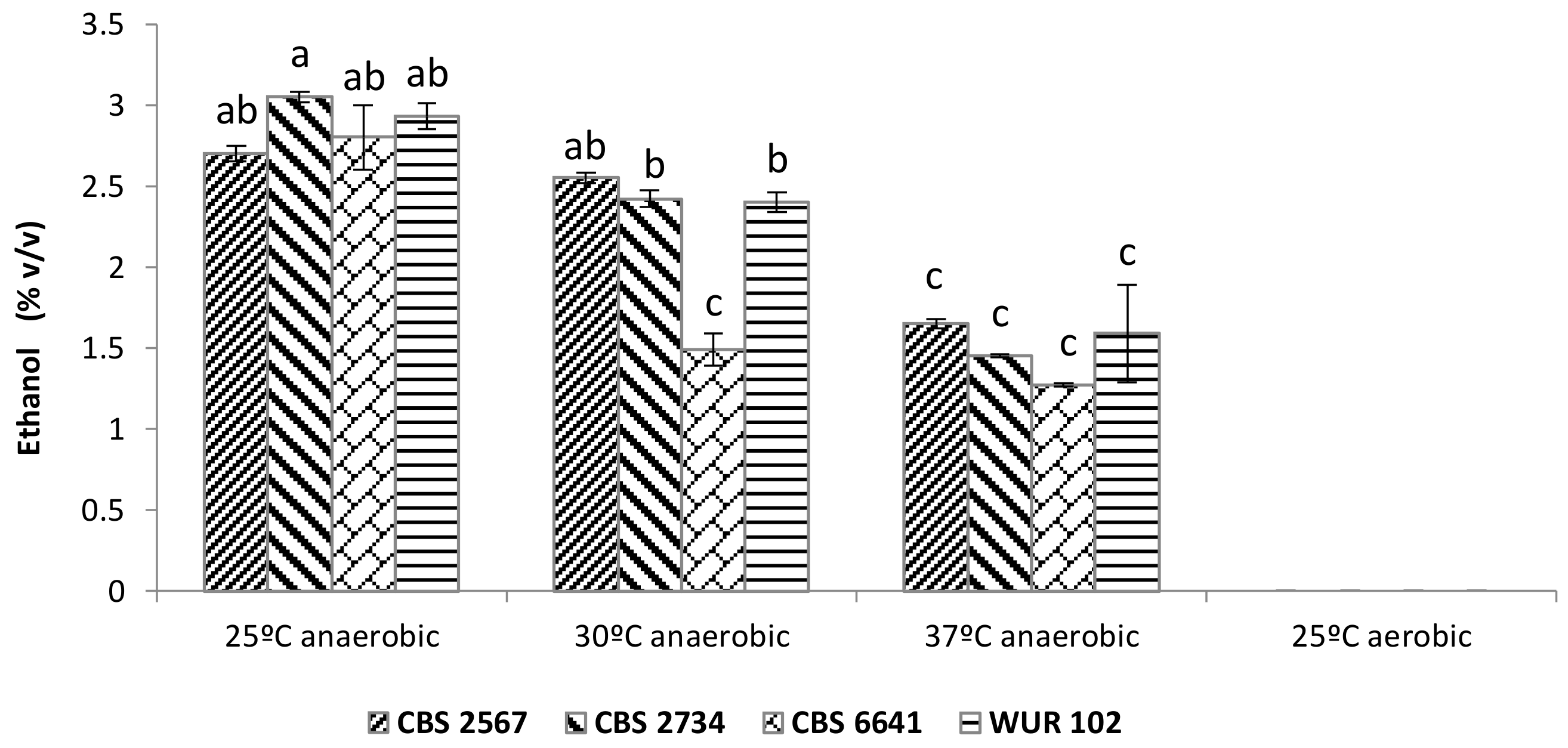
Analysis of the conversion of sugar into ethanol revealed remarkable differences among the strains (Figure 4). According to multifactorial ANOVA, all three factors studied (i.e., strain, oxygen, and temperature) were significantly affecting ethanol yield. Furthermore, we observed an interaction between the “strain” and “temperature” factors.
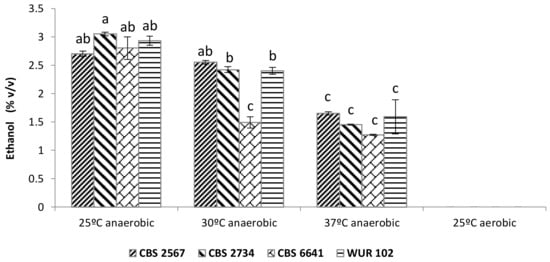
Figure 4.
Ethanol production in yellow cashew apple juice during incubation at diferent temperatures employing the selected strains CBS 2567, CBS 2734, CBS 6641, and WUR 102. Letters above bars indicate grouping according to ANOVA.
On the other hand, one-way ANOVA showed three clear groups among the different conditions: (1) Significantly higher ethanol production at 25 °C and 30 °C in anaerobic conditions; (2) significantly lower ethanol production at 37 °C in anaerobic conditions as well as for the strain CBS 6641 at 30 °C in anaerobic conditions; (3) no ethanol production at 25 °C in aerobic conditions. Consequently, in anaerobic conditions, higher incubation temperatures lead to lower ethanol concentrations. This might be due to a reduction in yeast growth and a lower fermentation performance, which is supported by the presence of residual sugars in the juice after fermentation. All four strains produced 2.7%–3% of ethanol at 25 °C. However, ethanol production was reduced to around 2.5% when the temperature reached 30 °C, except in the case of CBS 6641, whose ethanol yield was reduced to only 1.5%, suggesting a poor fermentation performance of this strain at 30 °C. Finally, at 37 °C, the ethanol production was reduced up to 1.5%–1.7%, except in the case of CBS 6641, where ethanol synthesis was only 1.3%. Therefore, strain CBS 6641 proved to be the least robust strain, not being able to ferment efficiently above 25 °C.
Furthermore, CAJ incubated aerobically showed no ethanol formation despite the fact that no residual sugars could be detected at the end of the incubation period. This observation might be explained by either redirection of the metabolic flux towards organic acids instead of ethanol, pointing to the Crabtree negative nature of those strains or by full oxidation of the produced ethanol into CO2 and H2O by aerobic respiration.
It is worth mentioning that strain CBS 6641 produced much less ethanol at 25 °C during the screening. This could be explained by the presence of residual oxygen that allowed this strain to produce other compounds instead of ethanol, such as organic acids or glycerol [41,42,43]. This would mean that this strain is Crabtree negative, since Crabtree positive strains are able to produce ethanol even in the presence of oxygen and when at the same time high levels of glucose are present in the medium as previously commented [40]. During the fermentations of the selected strains, strictly anaerobic conditions were provided, so CBS 6641 and the other selected strains were forced to ferment the sugars.
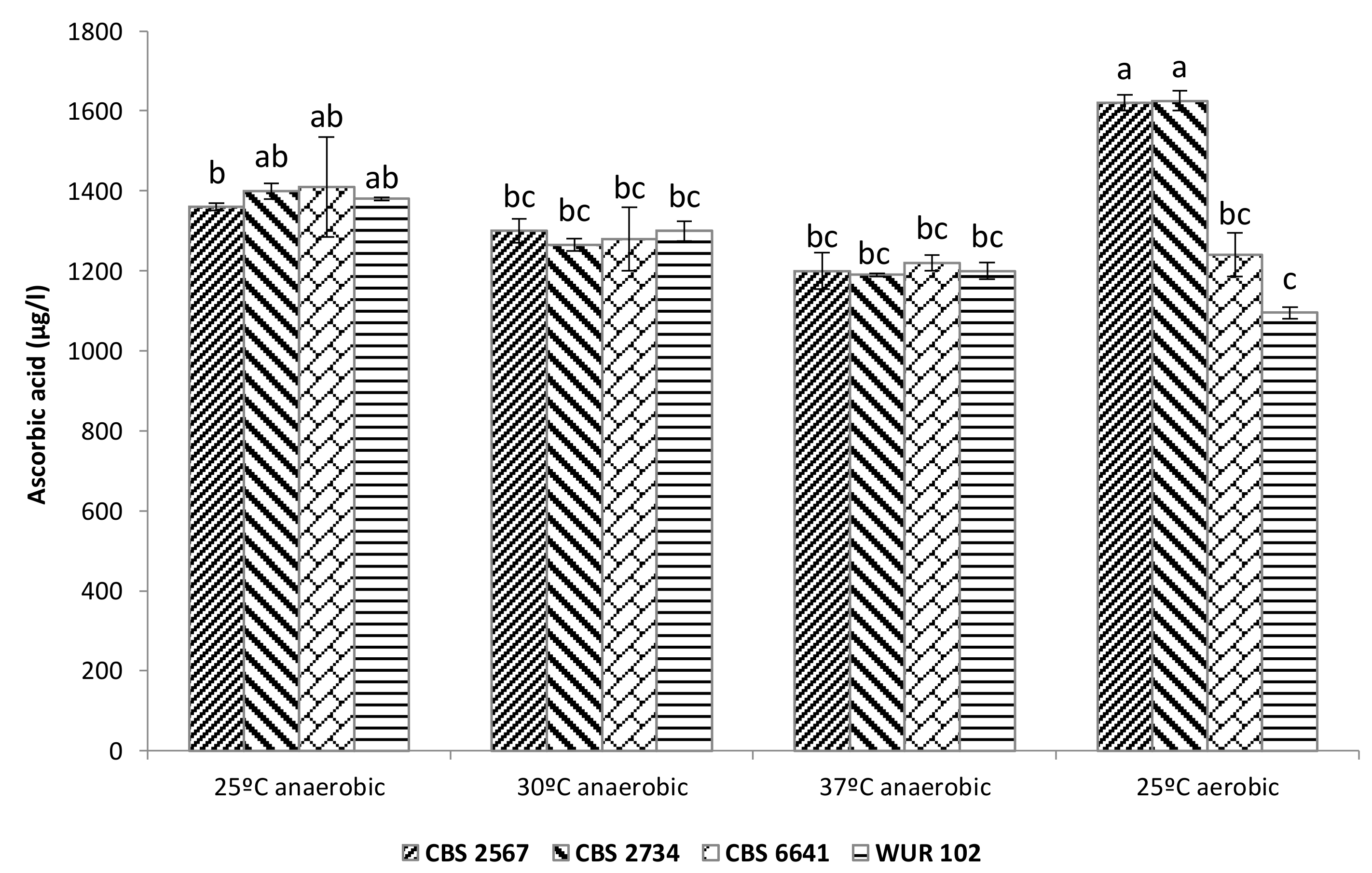
3.3.2. Ascorbic Acid Content in Fermented Cashew Apple Juice
Multifactorial ANOVA showed that temperature and strain were significant factors determining final ascorbic acid content. In the case of the factor “strain”, there was an interaction with the oxygen levels. For instance, under anaerobic cultivation conditions, a reduction in ascorbic acid was observed, which seems to be linked to an increase in temperature (Figure 5).
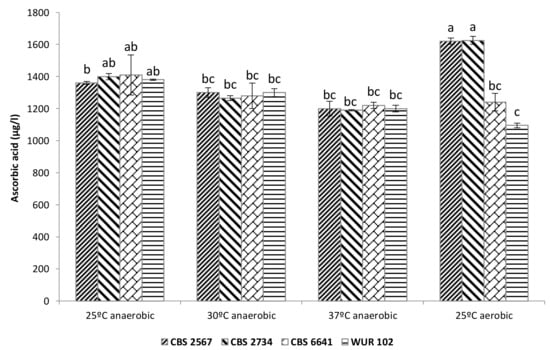
Figure 5.
Ascorbic acid content in fermented cashew apple juice employing the selected strains. Letters above bars indicate grouping according to ANOVA.
Furthermore, the factor “strain” significantly affected the ascorbic acid content in aerobic conditions, since strains CBS 2567 and CBS 2734 were even able to increase the levels of this vitamin with respect to the initial CAJ content (1100 ± 350 μg/L). We hypothesize that these strains are capable of synthesizing ascorbic acid, since it has been demonstrated that some yeasts are able to produce this compound under certain circumstances [44]. This production could compensate for ascorbic acid losses due to oxidation.
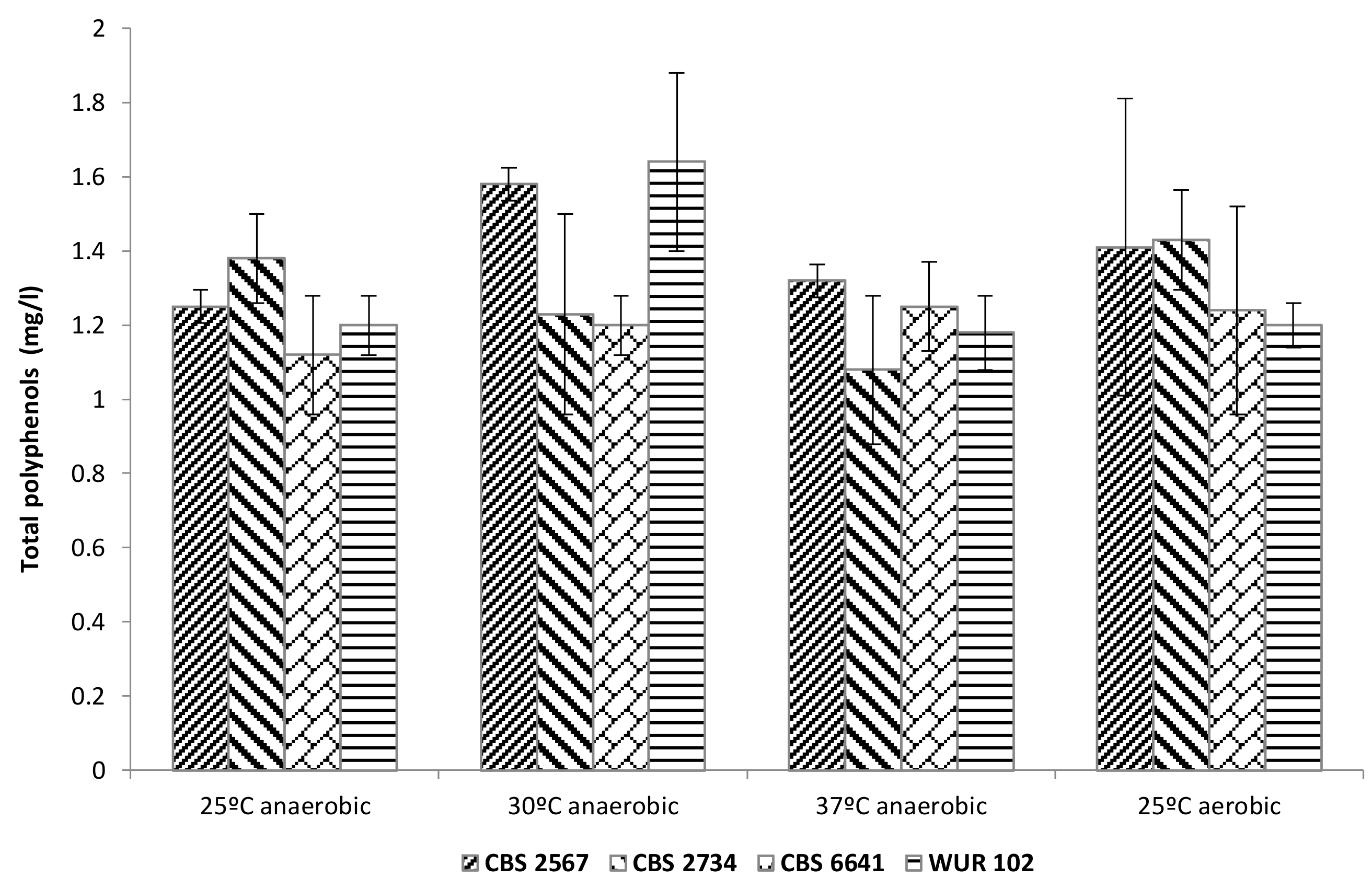
3.3.3. Total Polyphenol Content in the Fermented Cashew Apple Juice
ANOVA showed no significant differences among the strains regarding the total polyphenol content in the fermented products. In this way, polyphenols were not significantly affected by fermentation with any of the selected strains, temperatures, or oxygen levels tested (Figure 6). However, polyphenol levels in the fermented products were, in most cases, lower than the initial content in yellow CAJ (1.6 mg/L), indicating a possible degradation or bioconversion of polyphenols by the tested strains, which could decrease bitterness and astringency of the final products. The changes in polyphenol content caused by yeasts have been already reported by several authors [8,9,10]. Another hypothesis to explain the drop in polyphenols could be that part of them were adsorbed to yeast lees and therefore removed prior analysis, as already seen in other alcoholic fermentations [45,46].
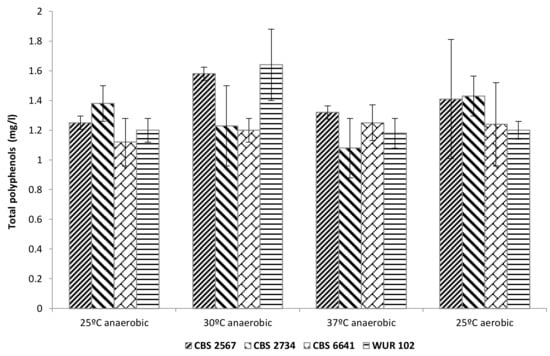
Figure 6.
Total polyphenol content in fermented cashew apple juice employing the selected strains.
3.3.4. Aroma Profiles of the Fermented Cashew Apple Juice
The concentrations of the major aroma compounds in fermented CAJ samples were compared. These compounds and their odor descriptions appear in Table 4 and can be divided into five groups: (1) Aldehydes: Acetaldehyde, benzeneacetaldehyde; 3-hydroxybutanal, 3-methylbutanal; (2) alcohols: 2-butanol, isobutanol, 2-methyl-1-butanol, 3-methyl-1-butanol, 1-pentanol, 4-methyl-1-pentanol, 3-methyl-1-pentanol, β-phenylethyl alcohol; (3) acids: Acetic acid; (4) acetate esters: Isoamyl acetate, n-amyl acetate, β-phenylethyl acetate; and (5) ethyl esters: Ethyl butyrate, ethyl lactate, ethyl caproate. According to Garruti et al. [2], the major compounds of fermented CAJ were 3-methyl butanol, isoamyl acetate, isobutanol, and several ethyl esters—ethyl hexanoate, ethyl decanoate, ethyl 3-methyl butyrate, ethyl lactate, and ethyl butyrate. Most of them were found in our study and they are, among others, related to sweet and fruity aromas [2].

Table 4.
Aroma compounds found in fermented cashew apple juice.
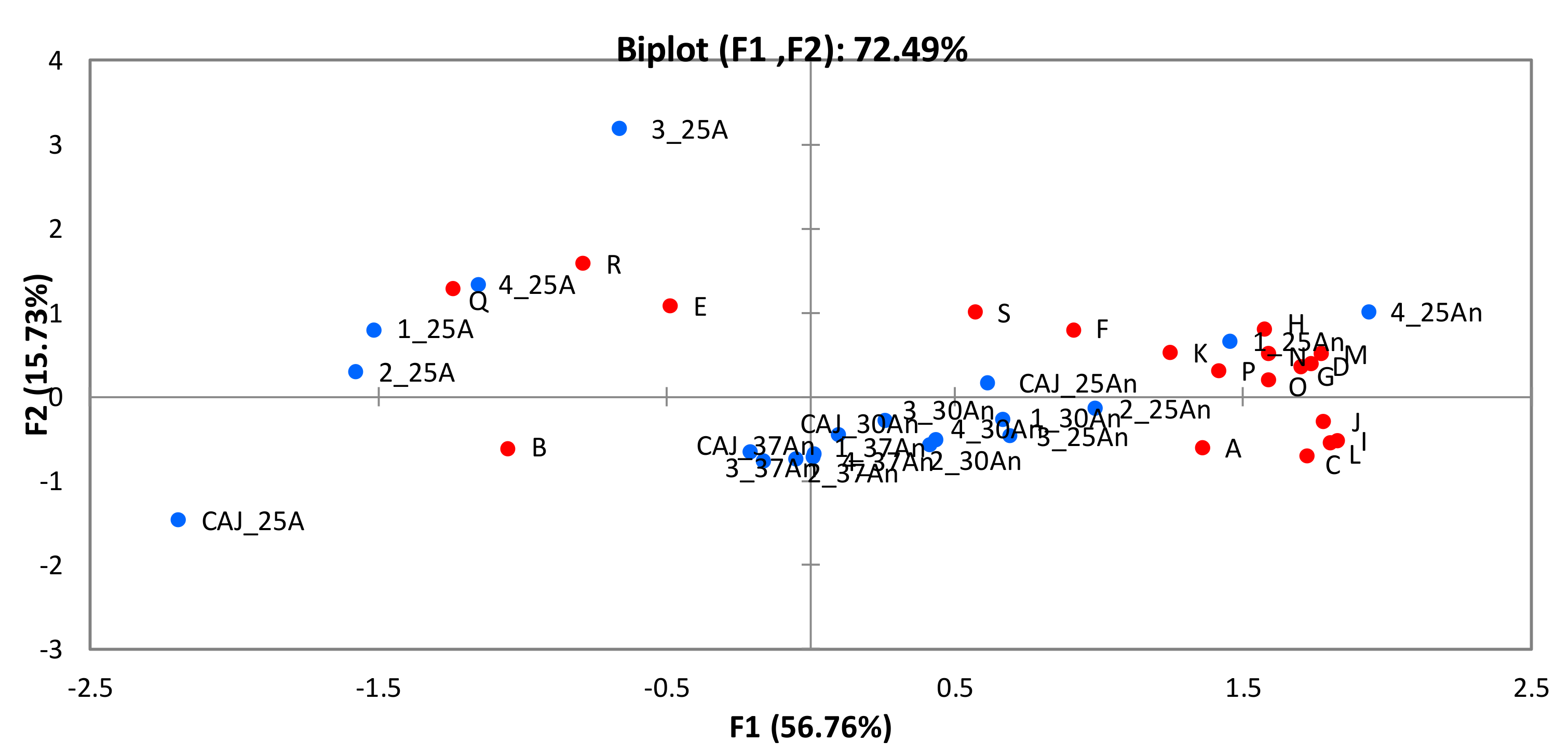
In our study, CAJ was fermented by four different strains (CBS 2567, CBS 2734, CBS 6641, and WUR 102) at three temperatures (25 °C, 30 °C and 37 °C) and employing two levels of oxygen (anaerobic and aerobic) in the case of the fermentations at 25 °C. In order to investigate the differences in the resulting aroma profiles, principal component analysis (PCA) was carried out (Figure 7). The first observation that stood out, was that the condition that yielded less aromatic products was incubation at 25 °C in the presence of oxygen. This was specifically the case of the unfermented CAJ and of the juice fermented with strain CBS 6641. At 25 °C under aerobic conditions, just a few aroma compounds appeared at high concentrations (i.e., benzeneacetaldehyde, phenylethyl alcohol, 3-methylbutanal and 2-butanol). On the contrary, the fermentation condition that yielded more diverse aroma profiles was anaerobiosis at 25 °C, especially for the fermentations carried out by CBS 2567 and WUR 102, followed by the other strains, the unfermented CAJ, and the fermentations at 30 °C. Unfermented CAJ at 30 °C and 37 °C and the fermentations at 37 °C presented intermediate aroma profiles in terms of compound diversity.
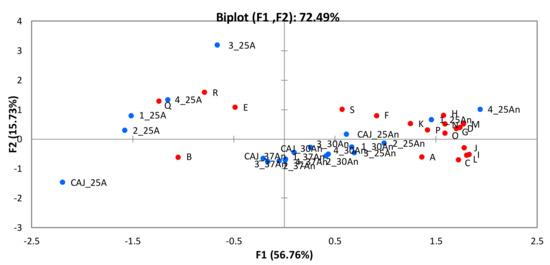
Figure 7.
Principal Component Analysis of the aroma profiles of the different fermentations employing the four selected strains. Blue dots: CAJ: cashew apple juice; 1: CBS 2567; 2: CBS 2734; 3: CBS 6641; 4: WUR 102; 25A: 25 °C, aerobic; 25An: 25 °C anaerobic; 30An: 30 °C, anaerobic; 37An: 37 °C, anaerobic; Red dots: Capital letters from A to S indicate the aroma compounds according to the notation in Table 4.
Moreover, multifactorial ANOVA was carried out in order to assess which factors (oxygen, temperature and/or strain) were affecting the aroma profiles. In the case of unfermented CAJ, just two factors (i.e., oxygen and temperature) were taken into account. The results show that both factors affected most of the aroma compounds studied. Exceptions were: 3-methylbutanal, 2-butanol, β-phenylethyl alcohol, acetic acid, and ethyl lactate. In case of the fermented CAJ, the three factors were investigated. The results indicate that oxygen affected the synthesis of almost all the aromas. In addition, a significant interaction between oxygen and strain factors was observed in β-phenylethyl acetate and acetic acid production. The second factor studied was temperature, which affected the production of approximately half of the aroma compounds. Finally, strain type significantly influenced the synthesis of all the ethyl esters, acetic acid, and almost all the aldehydes, with the exception of 3-hydroxybutanal. In addition, it was possible to find interaction among factors. An interaction between strain and temperature in case of 4-methyl pentanol and several esters, ethyl caproate, n-amyl acetate, and β-phenylethyl acetate; and an interaction between strain and oxygen in case of all the ethyl esters, almost all aldehydes (except acetaldehyde), half of alcohols, acetic acid, and almost all the acetate esters (except isoamyl acetate).
Certain aroma compounds were only synthesized under anaerobic conditions, such as ethyl butyrate, 4-methyl-1-pentanol, and n-amyl acetate, whereas the production of 2-butanol by CBS 2567 and CBS 2734 was exclusively under aerobic conditions. In fact, under aerobic conditions, most of the qualitative differences in the aroma profiles were observed. In particular, strain CBS 2567, which was not able to produce 3-hydroxybutanal and ethyl lactate as the other strains, was the only one able to synthesize 3-methyl-1-pentanol. In the anaerobic group, alcohols and esters were the major components. Acetate esters were derived from the condensation of higher alcohols and acetyl CoA, whereas ethyl esters came from condensation of ethanol and acyl-CoA. Temperature, the secondary factor, imposed restrictions on most aromas, especially at 37 °C, except for 3-methylbutanal and acetaldehyde, which appeared at higher levels at this temperature. Regarding the effect of the strains on the aroma profiles in anaerobic fermentations, a result that stands out is the high production of certain aroma compounds by strains WUR 102 and CBS 2567. Strain WUR 102 synthesized higher quantities of benzeneacetaldehyde, 3-methylbutanol, β-phenylethyl alcohol, amyl acetate, ethyl butyrate, and ethyl caproate at 25 °C. In addition, this strain synthesized high levels of phenylethyl acetate at 37 °C. The levels of all these aroma compounds were on average two to four-fold higher than those found in juices fermented by the other strains. On the other hand, the non-conventional yeast CBS 2567 excelled in the production of β-phenylethyl alcohol at 25 °C and its corresponding acetate ester (phenylethyl acetate) at all three temperatures, showing a huge production of up to 35-fold the level found for the other strains. Finally, the non-conventional yeast CBS 2734 yielded high β-phenylethyl alcohol as well. The so-called “fusel or higher alcohols”, such as β-phenylethyl alcohol, are either derived from the conversion of amino acids via the Ehrlich pathway or synthesized from sugars [27,47]. They contribute positively to the aroma profile by themselves and because they serve as precursors for the formation of acetate esters [27]. CAJ is rich in amino acids such as alanine, serine, leucine, phenylalanine, proline, glutamic acid, tyrosine, and aspartic acid. The aromatic β-phenylethyl alcohol is derived from the amino acid phenylalanine, whereas phenylethyl acetate is synthesized from β-phenylethyl alcohol. The presence of both compounds is remarkable for the global aroma as they are known to impart herbaceous and rose nuances in the fermented beverages [27].
The odour descriptions were obtained from Garruti et al. [2] and Leffingwell and Associates, Flavor-base 10th edition [48].
4. Conclusions
The aim of this study was to investigate the use of CAJ as a low-cost substrate for developing a fermentation process for the production of a novel alcoholic beverage. Although the clarification step of the CAJ did not help to reduce the levels of total polyphenols in the juice, it cleared the juice without affecting other properties, such as the concentration of ascorbic acid.
Yellow CAJ proved to have better properties than the red juice, such as higher ascorbic acid and sugar contents and lower total polyphenols, making it the preferred substrate for the production of a fermented beverage. However, initial quality control of the raw materials should be included as the composition of cashew apples can vary due to the type of soil, climatic conditions, stage of maturation and seasonality besides type of cultivar [1].
Non-conventional yeasts showed, in general, a poor fermentation performance when compared to Saccharomyces strains. However, some of them were able to grow and ferment CAJ efficiently, especially at the lowest temperatures tested (25 °C and 30 °C).
The selected yeast strains were evaluated at different temperature and oxygen conditions. The best condition in terms of aroma production was 25 °C under anaerobic conditions, where the aroma profiles showed the highest diversity. S. cerevisiae WUR 102 and H. guilliermondii CBS 2567 excelled in the synthesis of certain aroma compounds, especially in this condition, giving a high production of interesting aroma compounds such as β-phenylethanol and its corresponding acetate ester (rose aroma). In addition, the strains showed the potential to metabolize polyphenols while maintaining high ascorbic acid levels.
In conclusion, the optimal fermentation conditions for the production of a fermented alcoholic beverage from CAJ proved to be 25 °C in the absence of oxygen. The final products were slightly yellowish, acidic in taste, low in alcohol (around 3% v/v), contained relatively lower total polyphenols, and maintained high levels of ascorbic acid. Further studies are needed to test consumer acceptance of these new products.
Author Contributions
Conceptualization, A.G., Y.L., E.J.S., and A.R.L.; formal analysis, A.G. and X.R.; investigation, X.R.; resources, C.d.J., E.J., and A.R.L.; supervision, A.G., E.J.S., and A.R.L.; visualization, A.G., X.R., and Y.L.; writing—original draft, A.G. and X.R.; writing—review and editing, A.G., Y.L., C.d.J., E.J.S., and A.R.L.
Funding
This research was founded by the European Comission-Marie Curie Initial Training Network CORNUCOPIA (FP7-PEOPLE-2010-ITN, grant agreement nr. 264717).
Acknowledgments
The authors thank the European Comission for the funding; the International Institute of Tropical Agriculture, Benin station (Africa) for providing the cashew apples and Westerdijk Fungal Biodiversity Institute-CBS-KNAW for providing most of the strains.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Das, I.; Arora, A. Post-harvest processing technology for cashew apple—A review. J. Food Eng. 2017, 194, 87–98. [Google Scholar] [CrossRef]
- Garruti, D.S.; Franco, M.R.B.; da Silva, M.A.A.P.; Janzantti, N.S.; Alves, G.L. Assessment of aroma impact compounds in a cashew apple-based alcoholic beverage by GC-MS and GC-olfactometry. LWT Food Sci. Technol. 2006, 39, 373–378. [Google Scholar] [CrossRef]
- Lopes, T.; Rabelo, M.C.; Barros, L.R.; Saavedra, G.A.; Rodrigues, S. Fermentation of cashew apple juice to produce high added value products. World J. Microbiol. Biotechnol. 2007, 23, 1409–1415. [Google Scholar]
- Michodjehoun-Mestres, L.; Souquet, J.M.; Fulcrand, H.; Bouchut, C.; Reynes, M.; Brillouet, J.M. Monomeric phenols of cashew apple (Anacardium occidentale L.). Food Chem. 2009, 112, 851–857. [Google Scholar] [CrossRef]
- Michodjehoun-Mestres, L.; Souquet, J.M.; Fulcrand, H.; Bouchut, C.; Reynes, M.; Brillouet, J.M. Characterisation of highly polymerised prodelphinidins from skin and flesh of four cashew apple (Anacardium occidentale L.) genotypes. Food Chem. 2009, 114, 989–995. [Google Scholar] [CrossRef]
- Cormier, R. Clarification of Cashew Apple Juice and Commercial Applications; Oxfam: Quebec, Benin, 2008; pp. 1–9. [Google Scholar]
- Jayalekshmy, V.G.; John, P.S. ‘Sago’—A natural product for cashew apple juice clarification. J. Trop. Agric. 2004, 42, 67–68. [Google Scholar]
- Jayabalan, R.; Marimuthu, S.; Swaminathan, K. Changes in content of organic acids and tea polyphenols during kombucha tea fermentation. Food Chem. 2007, 102, 392–398. [Google Scholar] [CrossRef]
- Hainal, A.R.; Ignat, I.; Volf, I.; Popa, V.I. Transformation of polyphenols from biomass by some yeast species. Cellul. Chem. Technol. 2011, 45, 211–219. [Google Scholar]
- Ettayebi, K.; Errachidi, F.; Jamai, L.; Tahri-Jouti, M.A.; Sendide, K.; Ettayebi, M. Biodegradation of polyphenols with immobilized Candida tropicalis under metabolic induction. FEMS Microbiol. Lett. 2003, 223, 215–219. [Google Scholar] [CrossRef]
- Silveira, M.S.; Fontes, C.P.M.L.; Guilherme, A.A.; Fernandes, F.A.N.; Rodrigues, S. Cashew apple juice as substrate for lactic acid production. Food Bioproc. 2012, 5, 947–953. [Google Scholar] [CrossRef]
- Kubo, I.; Ochi, M.; Vieira, P.C.; Komatsu, S. Antitumor agents from the cashew (Anacardium occidentale) apple juice. Food Chem. 1993, 41, 1012–1015. [Google Scholar] [CrossRef]
- Melo, A.A.; Rubensam, G.; Picada, J.N.; Gomes, E.; Fonseca, J.C.; Henriques, J.A. Mutagenicity, antioxidant potential, and antimutagenic activity against hydrogen peroxide of cashew (Anacardium occidentale) apple juice and cajuina. Environ. Mol. Mutagen. 2003, 41, 360–369. [Google Scholar] [CrossRef] [PubMed]
- Melo, A.A.; Rübensam, G.; Erdtmann, B.; Brendel, M.; Henriques, J.A.P. Cashew (Anacardium occidentale) apple juice lowers mutagenicity of aflatoxin B1 in S. typhimurium TA102. Genet. Mol. Biol. 2005, 28, 328–333. [Google Scholar]
- Nogueira, A.K.; Martins, J.J.L.; Lima, J.G.; Giroa, M.E.A.; Franco, K.; Maciel, V.M.; Loiola, O.D.; Valderez, M.; Barros, L.R.; de Santiago, R.S. Purification and characterization of a biosurfactant produced by Bacillus subtilis in cashew apple juice and its application in the remediation of oil-contaminated soil. Colloids Surf. B Biointerfaces 2019, 175, 256–263. [Google Scholar] [CrossRef] [PubMed]
- Betiku, E.; Emeko, H.A.; Solomon, B.O. Fermentation parameter optimization of microbial oxalic acid production from cashew apple juice. Heliyon 2016, 2, e00082. [Google Scholar] [CrossRef] [PubMed]
- Chagas, C.M.; Honorato, T.L.; Pinto, G.A.; Maia, G.A.; Rodrigues, S. Dextran sucrase production using cashew apple juice as substrate: Effect of phosphate and yeast extract addition. Bioprocess Biosyst. Eng. 2007, 30, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Rocha, M.V.; Souza, M.C.; Benedicto, S.C.; Bezerra, M.S.; Macedo, G.R.; Pinto, G.A.; Gonçalves, L.R. Production of biosurfactant by Pseudomonas aeruginosa grown on cashew apple juice. Appl. Biochem. Biotechnol. 2007, 137–140, 185–194. [Google Scholar]
- Barros, E.M.; Rodrigues, T.H.S.; Pinheiro, A.D.T.; Angelim, A.L.; Melo, V.M.M.; Rocha, M.V.P.; Gonçalves, L.R.B. A yeast isolated from cashew apple juice and its ability to produce first- and second-generation ethanol. Appl. Biochem. Biotechnol. 2014, 174, 2762–2776. [Google Scholar] [CrossRef]
- Talasila, U.; Vechalapu, R.R. Optimization of medium constituents for the production of bioethanol from cashew apple juice using Doehlert experimental design International. J. Fruit Sci. 2015, 15, 161–172. [Google Scholar] [CrossRef]
- Teles, A.D.; da Silva, A.; Meneses, E.; Ceccato, S.R.; Marques, S.J.; Valderez, M.; Rocha, L. Mathematical modeling of the ethanol fermentation of cashew apple juice by a flocculent yeast: The effect of initial substrate concentration and temperature. Bioprocess Biosyst. Eng. 2017, 40, 1221–1235. [Google Scholar]
- Kaprasob, R.; Kerdchoechuen, O.; Laohakunjit, N.; Thumthanaruk, B.; Shetty, K. Changes in physico-chemical, astringency, volatile compounds and antioxidant activity of fresh and concentrated cashew apple juice fermented with Lactobacillus plantarum. J. Food Sci. Technol. 2018, 55, 3979–3990. [Google Scholar] [CrossRef] [PubMed]
- Kaprasob, R.; Kerdchoechuen, O.; Laohakunjit, N.; Somboonpanyakul, P.B. Vitamins and prebiotic fructooligosaccharides of cashew apple fermented with probiotic strains Lactobacillus spp., Leuconostoc mesenteroides and Bifidobacterium longum. Process. Biochem. 2018, 70, 9–19. [Google Scholar] [CrossRef]
- Kaprasob, R.; Kerdchoechuen, O.; Laohakunjit, N.; Sarkar, D.; Shetty, K. Fermentation-based biotransformation of bioactive phenolics and volatile compounds from cashew apple juice by select lactic acid bacteria. Process. Biochem. 2017, 59, 141–149. [Google Scholar] [CrossRef]
- Dantas, F.N.; Bezerra, J.; da Costa, M.; dos Santos, M.; Bertoldo, M.T.; Estevez, M.M.; de Souza, J.; Leite, E. Potential prebiotic properties of cashew apple (Anacardium occidentale L.) agro-industrial by product on Lactobacillus species. J. Sci. Food Agric. 2017, 97, 3712–3719. [Google Scholar]
- de Godoy, E.; Soares, T.H.; Narciso, F.A.; Fernandes, A.L.; Narain, N.; Sousa, E.; Rodrigues, S. Chemometric evaluation of the volatile profile of probiotic melon and probiotic cashew juice. Food Res. Int. 2017, 99, 461–468. [Google Scholar] [CrossRef] [PubMed]
- Araújo, S.M.; Silva, C.F.; Moreira, J.J.S.; Narain, N.; Souza, R.R. Biotechnological process for obtaining new fermented products from cashew apple fruit by Saccharomyces cerevisiae strains. J. Ind. Microbiol. Biotechnol. 2011, 38, 1161–1169. [Google Scholar] [CrossRef] [PubMed]
- Mohanty, S.; Ray, P.; Swain, M.R.; Ray, R.C. Fermentation of cashew (Anacardium occidentale L.) ‘apple’ into wine. J. Food Process. Pres. 2005, 30, 314–322. [Google Scholar] [CrossRef]
- Gamero, A.; Quintilla, R.; Groenewald, M.; Alkema, W.; Boekhout, T.; Hazelwood, L. High-throughput screening of a large collection of non-conventional yeasts reveals their potential for aroma formation in food fermentation. Food Microbiol. 2016, 60, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Dashko, S.; Zhou, N.; Tinta, T.; Sivilotti, P.; Lemut, M.S.; Trost, K.; Gamero, A.; Boekhout, T.; Butinar, L.; Vhrovsek, U.; et al. Use of non-conventional yeast improves the wine aroma profile of Ribolla Gialla. J. Ind. Microbiol. Biotechnol. 2015, 42, 997–1010. [Google Scholar] [CrossRef]
- Zhou, N.; Schifferdecker, A.J.; Gamero, A.; Compagno, C.; Boekhout, T.; Piskur, J.; Knecht, W. Kazachstania gamospora and Wickerhamomyces subpelliculosus: Two alternative baker´s yeasts in the modern bakery. Int. J. Food Microbiol. 2017, 250, 45–58. [Google Scholar] [CrossRef]
- Gutiérrez, A.; Boekhout, T.; Gojkovic, Z.; Katz, M. Evaluation of non-Saccharomycesyeasts in the fermentation of wine, beer and cider for the development of new beverages. J. Inst. Brew. 2018, 124, 389–402. [Google Scholar] [CrossRef]
- Van Rijswijck, I.M.H.; Wolkers-Rooijackers, J.C.M.; Abee, T.; Smid, E.J. Performance of non-conventional yeasts in co-culture with brewers’ yeast for steering ethanol and aroma production. Microb. Biotechnol. 2017, 10, 1591–1602. [Google Scholar] [CrossRef] [PubMed]
- Adou, M.; Kouassi, D.A.; Tetchi, F.A.; Amani, G. Phenolic profile of cashew apple juice (Anacardium occidentale L.) from Yamoussoukro and Korhogo (Côte d’Ivoire). J. Appl. Biosci. 2012, 49, 3331–3338. [Google Scholar]
- Georgé, S.; Brat, P.; Alter, P.; Amiot, M.J. Rapid determination of polyphenols and vitamin C in plant-derived products. J. Agric. Food Chem. 2005, 53, 1370–1373. [Google Scholar] [CrossRef] [PubMed]
- Hernández, Y.; Lobo, M.G.; González, M. Determination of vitamin C in tropical fruits: A comparative evaluation of methods. Food Chem. 2006, 96, 654–664. [Google Scholar] [CrossRef]
- Azevedo, D.C.S.; Rodrigues, A. Obtainment of high-fructose solutions from cashew (Anacardium occidentale) apple juice by simulated moving-bed chromatography. Sep. Sci. Technol. 2000, 35, 2561–2581. [Google Scholar] [CrossRef]
- Assunção, R.B.; Mercadante, A.Z. Carotenoids and ascorbic acid from cashew apple (Anacardium occidentale L.): Variety and geographic effects. Food Chem. 2003, 81, 495–502. [Google Scholar] [CrossRef]
- George, S.E.; Costenbader, C.J.; Melton, T. Diauxic growth in Azotobacter vinelandii. J. Bacteriol. 1985, 164, 866–871. [Google Scholar]
- De Deken, R.H. The Crabtree effect: A regulatory system in yeast. J. Gen. Microbiol. 1966, 44, 149–156. [Google Scholar] [CrossRef]
- Bely, M.; Stoeckle, P.; Masneuf-Pomarède, I.; Dubourdieu, D. Impact of mixed Torulaspora delbrueckii-Saccharomyces cerevisiae culture on high-sugar fermentation. Int. J. Food Microbiol. 2008, 122, 312–320. [Google Scholar] [CrossRef]
- Erasmus, D.J. Impact of yeast strain on the production of acetic acid, glycerol, and the sensory attributes of icewine. Am. J. Enol. Vitic. 2004, 4, 371–378. [Google Scholar]
- Finogenova, T.V.; Morgunov, I.G.; Kamzolova, S.V.; Chernyavskaya, O.G. Organic acid production by the yeast Yarrowia lipolytica: A review of prospects. Appl. Biochem. Microbiol. 2005, 41, 418–425. [Google Scholar] [CrossRef]
- Petrescu, S.; Hulea, S.A.; Stan, R.; Avram, D.; Herlea, V. A yeast strain that uses D-galacturonic acid as a substrate for L-ascorbic acid biosynthesis. Biotechnol. Lett. 1992, 14, 6. [Google Scholar] [CrossRef]
- Mazauric, J.P.; Salmon, J.M. Interactions between yeast lees and wine polyphenols during simulation of wine aging: II. Analysis of desorbed polyphenol compounds from yeast lees. J. Agric. Food Chem. 2006, 54, 3876–3881. [Google Scholar] [CrossRef] [PubMed]
- Salmon, J.M.; Fornairon-Bonnefond, C.; Mazauric, J.P. Interactions between wine lees and polyphenols: Influence on oxygen consumption capacity during simulation of wine aging. J. Food Sci. 2002, 67, 1604–1609. [Google Scholar] [CrossRef]
- Nisbet, M.A.; Tobias, H.J.; Brenna, J.T.; Sacks, G.L.; Mansfield, A.K. Quantifying the contribution of grape hexoses to wine volatiles by high-precision [U13C]-glucose tracer studies. J. Agric. Food Chem. 2014, 62, 6820–6827. [Google Scholar] [CrossRef] [PubMed]
- Leffingwell & Associates. Flavor-base 10th Edition. Available online: https://www.leffingwell.com/flavbase.htm (accessed on 27 June 2019).
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).