Bioaugmentation Versus pH Adjustment in High-Load Food Waste Anaerobic Digestion: Divergent Microbial Responses and Methanogenesis Regulation
Abstract
1. Introduction
2. Materials and Methods
2.1. Material Preparation
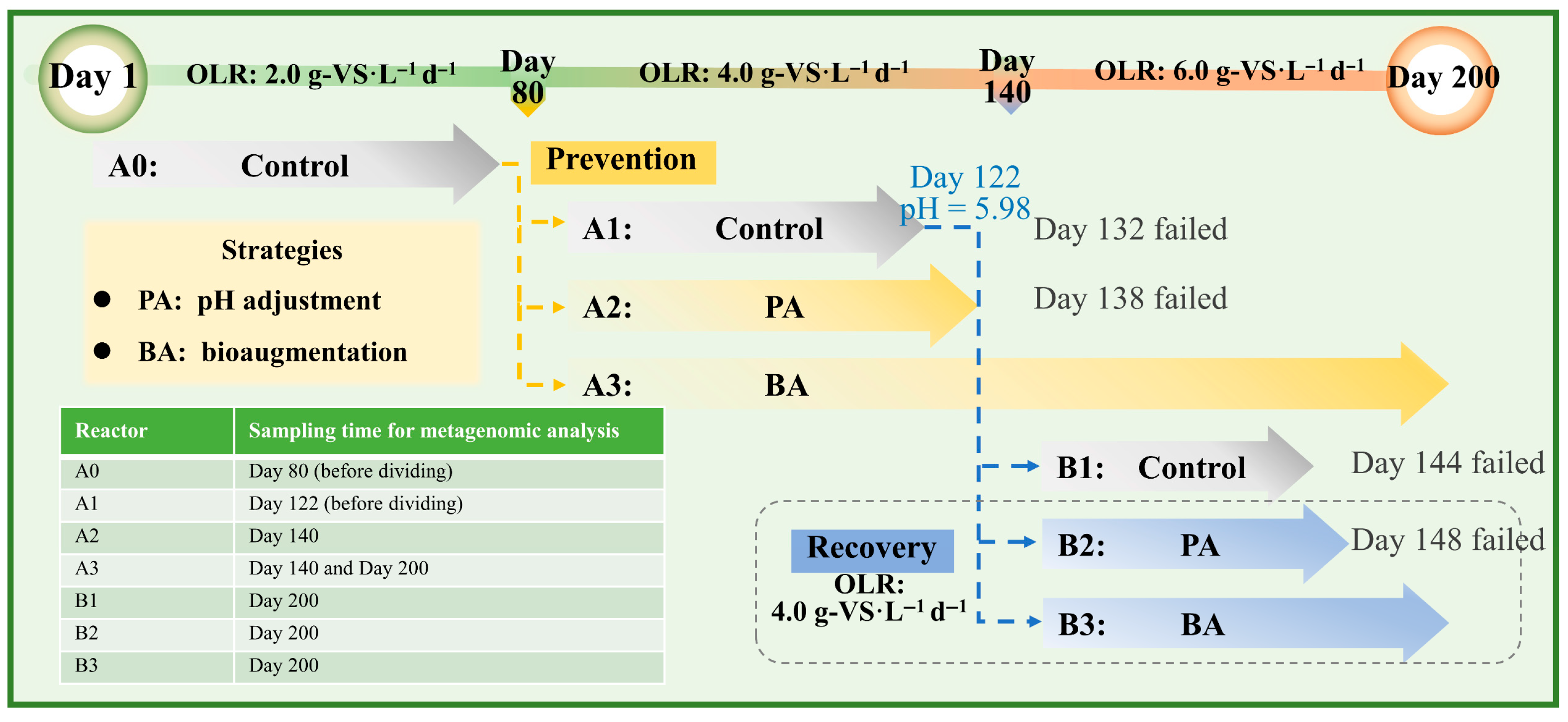
2.2. Experimental Procedures
2.3. Analytical Methods
2.4. Metagenomic Analysis
2.5. Data Analysis
3. Results and Discussion
3.1. Reactor Performance
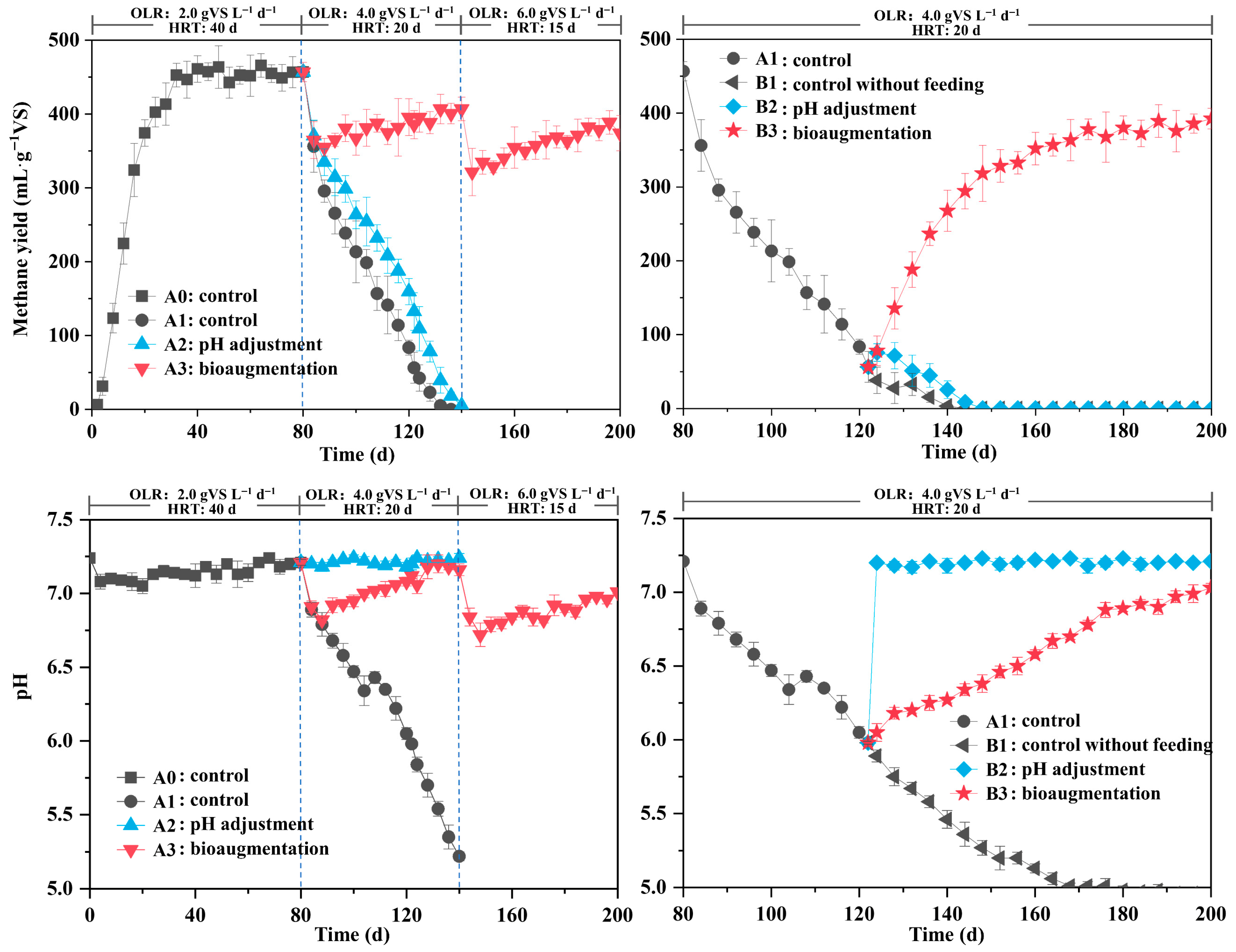
3.1.1. Methane Production
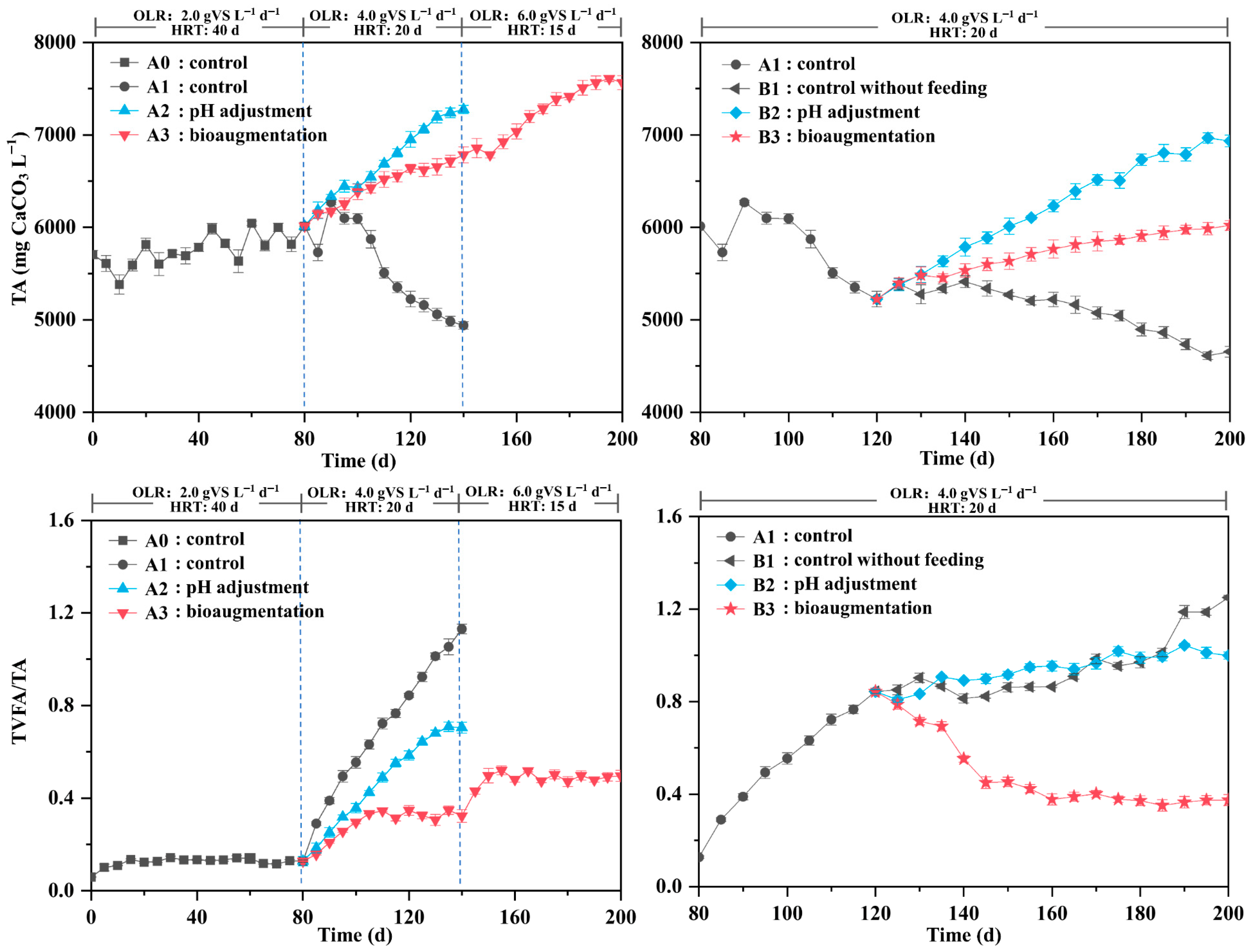
3.1.2. VFA and Other Physicochemical Parameters
3.2. Microbial Community Analysis
3.2.1. Alpha Diversity of Microbial Community
3.2.2. Effect of Bioaugmentation and pH Adjustment on Bacterial Community Structure
3.2.3. Effect of Bioaugmentation and pH Adjustment on Archaeal Community Structure
3.3. Metabolic Analysis
3.3.1. Hydrolysis
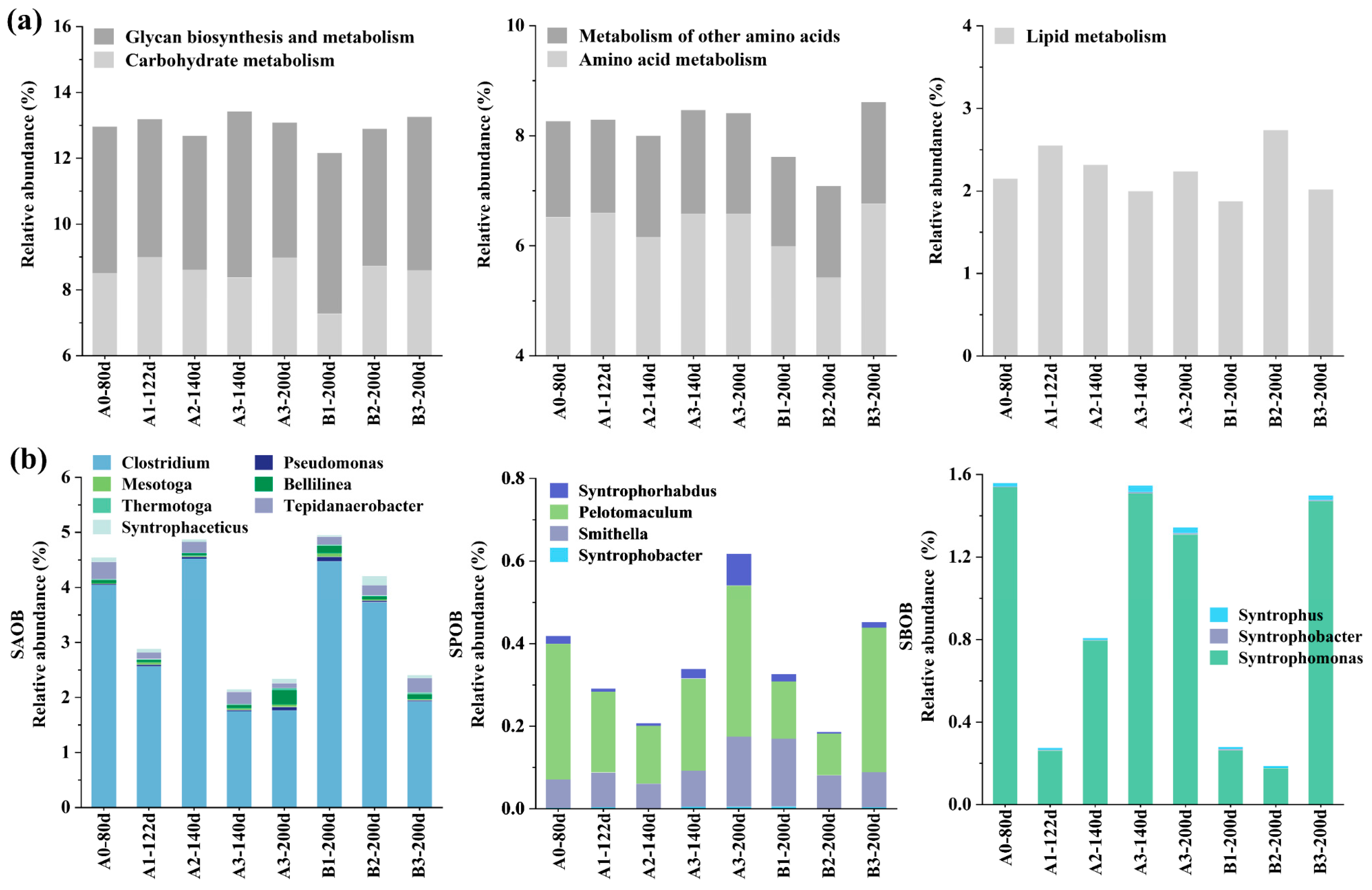
3.3.2. Acid Metabolism
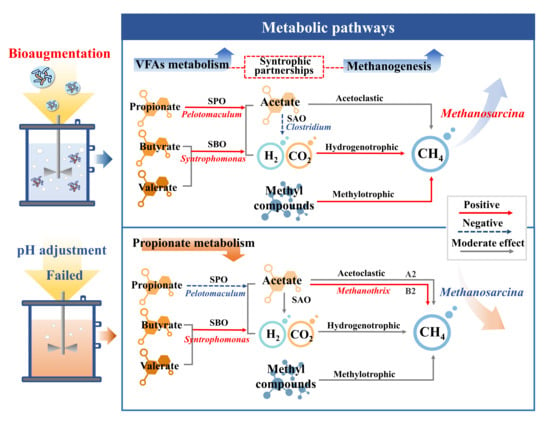
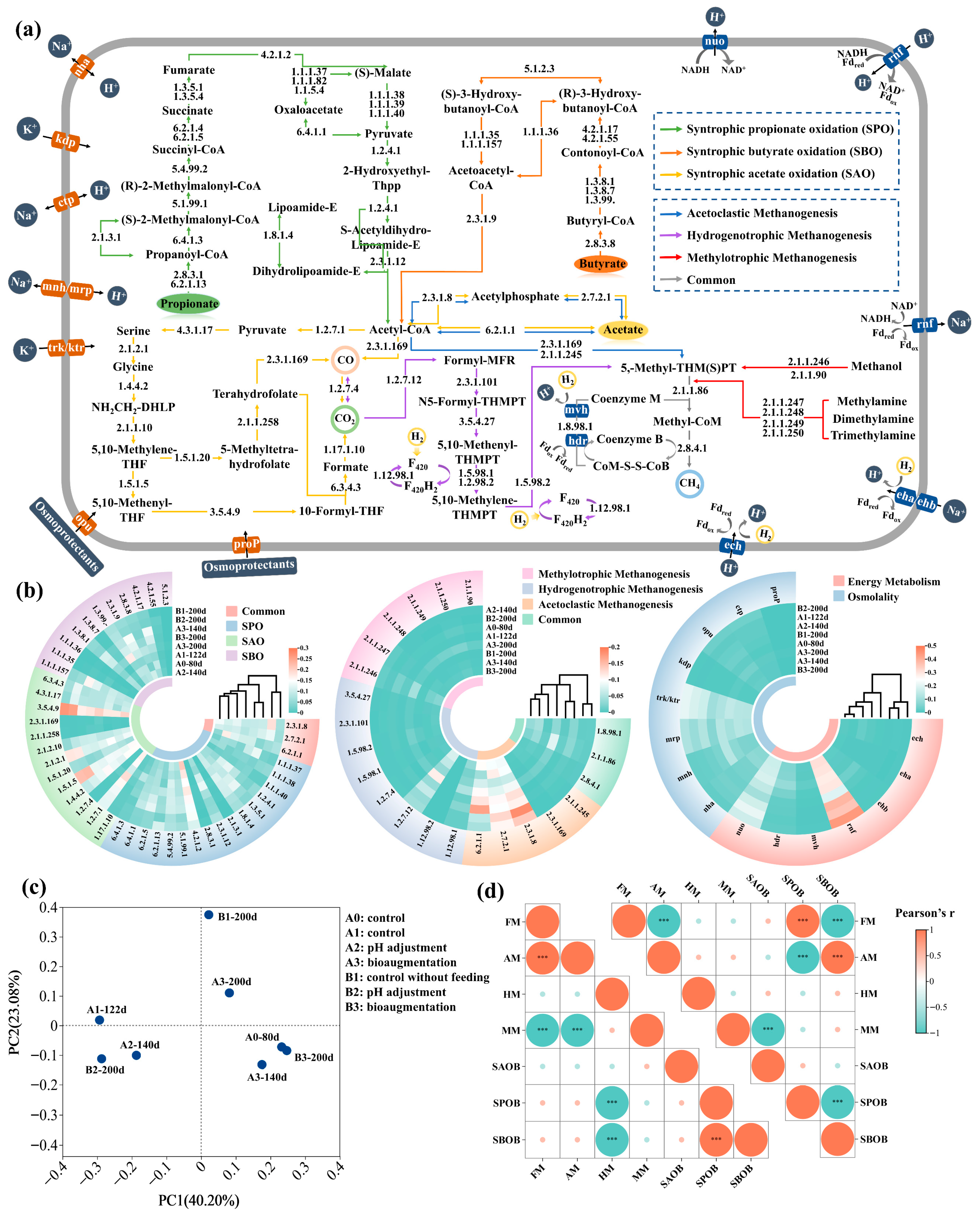
3.3.3. Methanogenesis
3.3.4. Osmolality and Energy Metabolism
3.4. Regulation of AD System by Bioaugmentation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AD | Anaerobic digestion |
| FW | Food waste |
| OLR | Organic loading rate |
| HRT | Hydraulic retention time |
| VFA | Volatile fatty acid |
| TVFA | Total volatile fatty acids |
| BA | Bioaugmentation |
| PA | pH adjustment |
| sCOD | Soluble chemical oxygen demand |
| TS | Total solids |
| VS | Volatile solids |
| TA | Total alkalinity |
| TVFA/TA | Total volatile fatty acids/Total alkalinity |
| TAN | Total ammonia nitrogen |
| FAN | Free ammonia nitrogen |
| SATP | Standard ambient temperature and pressure |
| KEGG | Kyoto encyclopedia of genes and genomes |
| EC | Enzyme Commission |
| ATP | Adenosine triphosphate |
| PCoA | Principal coordinates analysis |
| ANOVA | Analysis of variance |
| Ca. | Candidatus |
| SAO | Syntrophic acetate oxidation |
| SAOB | Syntrophic acetate-oxidizing bacteria |
| SBO | Syntrophic butyrate oxidation |
| SBOB | Syntrophic butyrate-oxidizing bacteria |
| SPO | Syntrophic propionate oxidation |
| SPOB | Syntrophic propionate-oxidizing bacteria |
| AM | Acetoclastic methanogen |
| HM | Hydrogenotrophic methanogen |
| FM | Facultative methanogen |
| MM | Methylotrophic methanogen |
References
- Chen, X.; He, H.; Zhu, N.; Jia, P.; Tian, J.; Song, W.; Cui, Z.; Yuan, X. Food waste impact on dry anaerobic digestion of straw in a novel reactor: Biogas yield, stability, and hydrolysis-methanogenesis processes. Bioresour. Technol. 2024, 406, 131023. [Google Scholar] [CrossRef] [PubMed]
- Regueiro, L.; Lema, J.M.; Carballa, M. Key microbial communities steering the functioning of anaerobic digesters during hydraulic and organic overloading shocks. Bioresour. Technol. 2015, 197, 208–216. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Xia, W.; Du, Y.; Li, D.; You, J.; Zhao, Q.; Wei, L. Enhanced bioavailability of trace elements for improving anaerobic digestion of food waste using sludge extracellular polymeric substances. Chem. Eng. J. 2025, 515, 163589. [Google Scholar] [CrossRef]
- Li, L.; He, Q.; Ma, Y.; Wang, X.; Peng, X. Dynamics of microbial community in a mesophilic anaerobic digester treating food waste: Relationship between community structure and process stability. Bioresour. Technol. 2015, 189, 113–120. [Google Scholar] [CrossRef]
- Li, Y.; Yang, G.; Li, L.; Sun, Y. Bioaugmentation for overloaded anaerobic digestion recovery with acid-tolerant methanogenic enrichment. Waste Manag. 2018, 79, 744–751. [Google Scholar] [CrossRef]
- Voelklein, M.A.; O’ Shea, R.; Jacob, A.; Murphy, J.D. Role of trace elements in single and two-stage digestion of food waste at high organic loading rates. Energy 2017, 121, 185–192. [Google Scholar] [CrossRef]
- Duan, X.; Chen, Y.; Feng, L.; Zhou, Q. Metagenomic analysis reveals nonylphenol-shaped acidification and methanogenesis during sludge anaerobic digestion. Water Res. 2021, 196, 117004. [Google Scholar] [CrossRef]
- Fan, Y.; Yang, X.; Lei, Z.; Adachi, Y.; Kobayashi, M.; Zhang, Z.; Shimizu, K. Novel insight into enhanced recoverability of acidic inhibition to anaerobic digestion with nano-bubble water supplementation. Bioresour. Technol. 2021, 326, 124782. [Google Scholar] [CrossRef]
- Mata-Alvarez, J.; Dosta, J.; Romero-Güiza, M.S.; Fonoll, X.; Peces, M.; Astals, S. A critical review on anaerobic co-digestion achievements between 2010 and 2013. Renew. Sustain. Energy Rev. 2014, 36, 412–427. [Google Scholar] [CrossRef]
- González, R.; Peña, D.C.; Gómez, X. Anaerobic co-digestion of wastes: Reviewing current status and approaches for enhancing biogas production. Appl. Sci. 2022, 12, 8884. [Google Scholar] [CrossRef]
- Hua, B.; Cai, Y.; Cui, Z.; Wang, X. Bioaugmentation with methanogens cultured in a micro-aerobic microbial community for overloaded anaerobic digestion recovery. Anaerobe 2022, 76, 102603. [Google Scholar] [CrossRef] [PubMed]
- Town, J.R.; Dumonceaux, T.J. Laboratory-scale bioaugmentation relieves acetate accumulation and stimulates methane production in stalled anaerobic digesters. Appl. Microbiol. Biotechnol. 2016, 100, 1009–1017. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, Y.; Sun, Y.; Wu, S.; Kong, X.; Yuan, Z.; Dong, R. The performance efficiency of bioaugmentation to prevent anaerobic digestion failure from ammonia and propionate inhibition. Bioresour. Technol. 2017, 231, 94–100. [Google Scholar] [CrossRef]
- Tale, V.P.; Maki, J.S.; Zitomer, D.H. Bioaugmentation of overloaded anaerobic digesters restores function and archaeal community. Water Res. 2015, 70, 138–147. [Google Scholar] [CrossRef] [PubMed]
- Acharya, S.M.; Kundu, K.; Sreekrishnan, T.R. Improved stability of anaerobic digestion through the use of selective acidogenic culture. J. Environ. Eng. 2015, 141, 04015001. [Google Scholar] [CrossRef]
- Zhang, W.; Xing, W.; Li, R. Real-time recovery strategies for volatile fatty acid-inhibited anaerobic digestion of food waste for methane production. Bioresour. Technol. 2018, 265, 82–92. [Google Scholar] [CrossRef]
- APHA. Standard Methods for the Examination of Water and Wastewater; American Public Health Association: Washington, DC, USA, 2005. [Google Scholar]
- Ren, W.; Wu, Q.; Deng, L.; Wang, H.; Zhang, Y.; Guo, W. Insights into chain elongation mechanisms of weak electric-field-stimulated continuous caproate biosynthesis: Key enzymes, specific species functions, and microbial collaboration. ACS EST Eng. 2023, 3, 1649–1660. [Google Scholar] [CrossRef]
- Peng, X.; Zhang, S.; Li, L.; Zhao, X.; Ma, Y.; Shi, D. Long-term high-solids anaerobic digestion of food waste: Effects of ammonia on process performance and microbial community. Bioresour. Technol. 2018, 262, 148–158. [Google Scholar] [CrossRef]
- Ohemeng-Ntiamoah, J.; Datta, T. Perspectives on variabilities in biomethane potential test parameters and outcomes: A review of studies published between 2007 and 2018. Sci. Total Environ. 2019, 664, 1052–1062. [Google Scholar] [CrossRef] [PubMed]
- Schievano, A.; D’Imporzano, G.; Malagutti, L.; Fragali, E.; Ruboni, G.; Adani, F. Evaluating inhibition conditions in high-solids anaerobic digestion of organic fraction of municipal solid waste. Bioresour. Technol. 2010, 101, 5728–5732. [Google Scholar] [CrossRef]
- Zhang, S.; Ma, X.; Sun, H.; Xie, D.; Zhao, P.; Wang, Q.; Wu, C.; Gao, M. Semi-continuous mesophilic-thermophilic two-phase anaerobic co-digestion of food waste and spent mushroom substance: Methanogenic performance, microbial, and metagenomic analysis. Bioresour. Technol. 2022, 360, 127518. [Google Scholar] [CrossRef]
- Zhang, W.; Zhang, F.; Li, Y.-X.; Jianxiong Zeng, R. Inhibitory effects of free propionic and butyric acids on the activities of hydrogenotrophic methanogens in mesophilic mixed culture fermentation. Bioresour. Technol. 2019, 272, 458–464. [Google Scholar] [CrossRef]
- Baek, G.; Kim, J.; Shin, S.G.; Lee, C. Bioaugmentation of anaerobic sludge digestion with iron-reducing bacteria: Process and microbial responses to variations in hydraulic retention time. Appl. Microbiol. Biotechnol. 2016, 100, 927–937. [Google Scholar] [CrossRef]
- Sangeetha, T.; Guo, Z.; Liu, W.; Gao, L.; Wang, L.; Cui, M.; Chen, C.; Wang, A. Energy recovery evaluation in an up flow microbial electrolysis coupled anaerobic digestion (ME-AD) reactor: Role of electrode positions and hydraulic retention times. Appl. Energy 2017, 206, 1214–1224. [Google Scholar] [CrossRef]
- Jo, Y.; Rhee, C.; Choi, H.; Shin, J.; Shin, S.G.; Lee, C. Long-term effectiveness of bioaugmentation with rumen culture in continuous anaerobic digestion of food and vegetable wastes under feed composition fluctuations. Bioresour. Technol. 2021, 338, 125500. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Xue, H.; Yao, Y.; Jing, C.; Liu, R.; Niu, Q.; Lu, H. Overcoming methanogenesis barrier to acid inhibition and enhancing PAHs removal by granular biochar during anaerobic digestion. Chem. Eng. J. 2023, 477, 147229. [Google Scholar] [CrossRef]
- Mu, L.; Wang, Y.; Xu, F.; Li, J.; Tao, J.; Sun, Y.; Song, Y.; Duan, Z.; Li, S.; Chen, G. Emerging strategies for enhancing propionate conversion in anaerobic digestion: A review. Molecules 2023, 28, 3883. [Google Scholar] [CrossRef]
- Shrestha, S.; Xue, S.; Raskin, L. Competitive reactions during ethanol chain elongation were temporarily suppressed by increasing hydrogen partial pressure through methanogenesis inhibition. Environ. Sci. Technol. 2023, 57, 3369–3379. [Google Scholar] [CrossRef]
- Wang, S.; Li, D.; Zhang, K.; Ma, Y.; Liu, F.; Li, Z.; Gao, X.; Gao, W.; Du, L. Effects of initial volatile fatty acid concentrations on process characteristics, microbial communities, and metabolic pathways on solid-state anaerobic digestion. Bioresour. Technol. 2023, 369, 128461. [Google Scholar] [CrossRef]
- Stams, A.J.M.; Plugge, C.M. Electron transfer in syntrophic communities of anaerobic bacteria and archaea. Nat. Rev. Microbiol. 2009, 7, 568–577. [Google Scholar] [CrossRef]
- Müller, B.; Sun, L.; Schnürer, A. First insights into the syntrophic acetate-oxidizing bacteria—A genetic study. Microbiologyopen 2013, 2, 35–53. [Google Scholar] [CrossRef]
- Achinas, S.; Achinas, V.; Euverink, G.J.W. Chapter 2—Microbiology and biochemistry of anaerobic digesters: An overview. In wBioreactors; Elsevier: Amsterdam, The Netherlands, 2020; pp. 17–26. [Google Scholar] [CrossRef]
- Banks, C.J.; Zhang, Y.; Jiang, Y.; Heaven, S. Trace element requirements for stable food waste digestion at elevated ammonia concentrations. Bioresour. Technol. 2012, 104, 127–135. [Google Scholar] [CrossRef]
- Li, L.; He, Q.; Zhao, X.; Wu, D.; Wang, X.; Peng, X. Anaerobic digestion of food waste: Correlation of kinetic parameters with operational conditions and process performance. Biochem. Eng. J. 2018, 130, 1–9. [Google Scholar] [CrossRef]
- Ajayi-Banji, A.; Rahman, S. A review of process parameters influence in solid-state anaerobic digestion: Focus on performance stability thresholds. Renew. Sustain. Energy Rev. 2022, 167, 112756. [Google Scholar] [CrossRef]
- Wu, D.; Li, L.; Peng, Y.; Yang, P.; Peng, X.; Sun, Y.; Wang, X. State indicators of anaerobic digestion: A critical review on process monitoring and diagnosis. Renew. Sustain. Energy Rev. 2021, 148, 111260. [Google Scholar] [CrossRef]
- Tampio, E.; Ervasti, S.; Paavola, T.; Heaven, S.; Banks, C.; Rintala, J. Anaerobic digestion of autoclaved and untreated food waste. Waste Manag. 2014, 34, 370–377. [Google Scholar] [CrossRef]
- Shi, X.; Lin, J.; Zuo, J.; Li, P.; Li, X.; Guo, X. Effects of free ammonia on volatile fatty acid accumulation and process performance in the anaerobic digestion of two typical bio-wastes. J. Environ. Sci. 2017, 55, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Piao, C.; Wang, K.; Zhu, E.; Wang, Z. Microbial response to varying ammonia conditions in mesophilic anaerobic digestion mediated by mixed bioaugmentation consortia. Environ. Res. 2026, 289, 123365. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Li, A.; Zhang, H.; Li, S.; Yang, C.; Lv, H.; Yao, Y. Microbial mechanisms for higher hydrogen production in anaerobic digestion at constant temperature versus gradient heating. Microbiome 2024, 12, 170. [Google Scholar] [CrossRef]
- You, M.; Zhao, Q.; Liu, Y.; Zhang, W.; Shen, Z.; Ren, Z.; Xu, C. Insights into lignocellulose degradation: Comparative genomics of anaerobic and cellulolytic Ruminiclostridium-type species. Front. Microbiol. 2023, 14, 1288286. [Google Scholar] [CrossRef]
- Song, G.; Zhao, S.; Wang, J.; Zhao, K.; Zhao, J.; Liang, H.; Liu, R.; Li, Y.-Y.; Hu, C.; Qu, J. Enzyme-enhanced acidogenic fermentation of waste activated sludge: Insights from sludge structure, interfaces, and functional microflora. Water Res. 2024, 249, 120889. [Google Scholar] [CrossRef]
- Hahnke, S.; Langer, T.; Koeck, D.E.; Klocke, M. Description of Proteiniphilum saccharofermentans sp. nov., Petrimonas mucosa sp. nov. and Fermentimonas caenicola gen. nov., sp. nov., isolated from mesophilic laboratory-scale biogas reactors, and emended description of the genus Proteiniphilum. Int. J. Syst. Evol. Microbiol. 2016, 66, 1466–1475. [Google Scholar] [CrossRef]
- Jiang, X.; Zhao, Z.; Zhang, Y. Towards engineering application: Integrating current strategies of promoting direct interspecies electron transfer to enhance anaerobic digestion. Chem. Eng. J. Adv. 2022, 12, 100405. [Google Scholar] [CrossRef]
- Wang, Y.; Hui, X.; Wang, H.; Chen, H. Boosting volatile fatty acids (VFAs) production in fermentation microorganisms through genes expression control: Unraveling the role of iron homeostasis transcription factors. Water Res. 2024, 259, 121850. [Google Scholar] [CrossRef]
- Zhang, J.; Qi, C.; Wang, Y.; Li, Y.; Han, T.; Gong, X.; Shan, M.; Li, G.; Luo, W. Enhancing biogas production from livestock manure in solid-state anaerobic digestion by sorghum-vinegar residues. Environ. Technol. Innov. 2022, 26, 102276. [Google Scholar] [CrossRef]
- Bovio-Winkler, P.; Guerrero, L.D.; Erijman, L.; Oyarzúa, P.; Suárez-Ojeda, M.E.; Cabezas, A.; Etchebehere, C. Genome-centric metagenomic insights into the role of Chloroflexi in anammox, activated sludge and methanogenic reactors. BMC Microbiol. 2023, 23, 45. [Google Scholar] [CrossRef]
- Mohiuddin, M.M.; Salama, Y.; Schellhorn, H.E.; Golding, G.B. Shotgun metagenomic sequencing reveals freshwater beach sands as reservoir of bacterial pathogens. Water Res. 2017, 115, 360–369. [Google Scholar] [CrossRef]
- Bu, J.; Wang, Y.; Gao, Y.; Zhao, Q.; Luo, Y.; Tiong, Y.W.; Lam, H.T.; Zhang, J.; He, Y.; Wang, C.-H.; et al. Enhancing anaerobic digestion of food waste with chemically vapor-deposited biochar: Effective enrichment of Methanosarcina and hydrogenotrophic methanogens. Bioresour. Technol. 2025, 424, 132225. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.K.; Im, W.T.; Kim, D.H.; Kim, M.H.; Shin, H.S.; Oh, S.E. Dry anaerobic digestion of food waste under mesophilic conditions: Performance and methanogenic community analysis. Bioresour. Technol. 2013, 131, 210–217. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Li, Y.; Euverink, G.J.W. Effect of bioaugmentation combined with activated charcoal on the mitigation of volatile fatty acids inhibition during anaerobic digestion. Chem. Eng. J. 2022, 428, 131015. [Google Scholar] [CrossRef]
- Zhao, D.; Yan, B.; Liu, C.; Yao, B.; Luo, L.; Yang, Y.; Liu, L.; Wu, F.; Zhou, Y. Mitigation of acidogenic product inhibition and elevated mass transfer by biochar during anaerobic digestion of food waste. Bioresour. Technol. 2021, 338, 125531. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Yan, Q.; Zhong, X.; Angelidaki, I.; Fotidis, I.A. Metabolic responses and microbial community changes to long chain fatty acids: Ammonia synergetic co-inhibition effect during biomethanation. Bioresour. Technol. 2023, 386, 129538. [Google Scholar] [CrossRef]
- Chandra, R.; Takeuchi, H.; Hasegawa, T. Methane production from lignocellulosic agricultural crop wastes: A review in context to second generation of biofuel production. Renew. Sustain. Energy Rev. 2012, 16, 1462–1476. [Google Scholar] [CrossRef]
- Wang, J.; Ma, D.; Feng, K.; Lou, Y.; Zhou, H.; Liu, B.; Xie, G.; Ren, N.; Xing, D. Polystyrene nanoplastics shape microbiome and functional metabolism in anaerobic digestion. Water Res. 2022, 219, 118606. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Sun, H.; Zhou, L.; Arhin, S.G.; Papadakis, V.G.; Goula, M.A.; Liu, G.; Zhang, Y.; Wang, W. Bioaugmentation with well-constructed consortia can effectively alleviate ammonia inhibition of practical manure anaerobic digestion. Water Res. 2022, 215, 118244. [Google Scholar] [CrossRef] [PubMed]
- Rajagopal, R.; Massé, D.I.; Singh, G. A critical review on inhibition of anaerobic digestion process by excess ammonia. Bioresour. Technol. 2013, 143, 632–641. [Google Scholar] [CrossRef]
- Wang, G.; Dai, X.; Zhang, D.; He, Q.; Dong, B.; Li, N.; Ye, N. Two-phase high solid anaerobic digestion with dewatered sludge: Improved volatile solid degradation and specific methane generation by temperature and pH regulation. Bioresour. Technol. 2018, 259, 253–258. [Google Scholar] [CrossRef]
- Tian, H.; Yan, M.; Treu, L.; Angelidaki, I.; Fotidis, I.A. Hydrogenotrophic methanogens are the key for a successful bioaugmentation to alleviate ammonia inhibition in thermophilic anaerobic digesters. Bioresour. Technol. 2019, 293, 122070. [Google Scholar] [CrossRef]
- Li, J.; Li, A.; Li, Y.; Cai, M.; Luo, G.; Wu, Y.; Tian, Y.; Xing, L.; Zhang, Q. PICRUSt2 functionally predicts organic compounds degradation and sulfate reduction pathways in an acidogenic bioreactor. Front. Environ. Sci. Eng. 2022, 16, 47. [Google Scholar] [CrossRef]
- Zhang, N.; Peng, H.; Li, Y.; Yang, W.; Zou, Y.; Duan, H. Ammonia determines transcriptional profile of microorganisms in anaerobic digestion. Braz. J. Microbiol. 2018, 49, 770–776. [Google Scholar] [CrossRef]
- Zhang, H.; Yuan, W.; Dong, Q.; Wu, D.; Yang, P.; Peng, Y.; Li, L.; Peng, X. Integrated multi-omics analyses reveal the key microbial phylotypes affecting anaerobic digestion performance under ammonia stress. Water Res. 2022, 213, 118152. [Google Scholar] [CrossRef]
- Annamalai, T.; Venkitanarayanan, K. Role of proP and proU in betaine uptake by Yersinia enterocolitica under cold and osmotic stress conditions. Appl. Environ. Microbiol. 2009, 75, 1471–1477. [Google Scholar] [CrossRef]
- Ito, M.; Guffanti, A.A.; Wang, W.; Krulwich, T.A. Effects of nonpolar mutations in each of the seven Bacillus subtilis mrp genes suggest complex interactions among the gene products in support of Na+ and alkali but not cholate resistance. J. Bacteriol. 2000, 182, 5663–5670. [Google Scholar] [CrossRef]
- Nanatani, K.; Shijuku, T.; Takano, Y.; Zulkifli, L.; Yamazaki, T.; Tominaga, A.; Souma, S.; Onai, K.; Morishita, M.; Ishiura, M.; et al. Comparative analysis of kdp and ktr mutants reveals distinct roles of the potassium transporters in the model Cyanobacterium Synechocystis sp. strain PCC 6803. J. Bacteriol. 2015, 197, 676–687. [Google Scholar] [CrossRef] [PubMed]
- Schubiger, C.B.; Hoang, K.H.T.; Häse, C.C. Sodium antiporters of Pseudomonas aeruginosa in challenging conditions: Effects on growth, biofilm formation, and swarming motility. J. Genet. Eng. Biotechnol. 2020, 18, 4. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; He, P.; Zhang, H.; Lü, F. Response of exogenous and indigenous microorganisms in alleviating acetate–ammonium coinhibition during thermophilic anaerobic digestion. ACS EST Eng. 2025, 5, 655–665. [Google Scholar] [CrossRef]
- Nomura, S.; San Segundo-Acosta, P.; Protasov, E.; Kaneko, M.; Kahnt, J.; Murphy, B.J.; Shima, S. Electron flow in hydrogenotrophic methanogens under nickel limitation. Nature 2025, 644, 490–496. [Google Scholar] [CrossRef]
- Montañés, R.; Pérez, M.; Solera, R. Anaerobic mesophilic co-digestion of sewage sludge and sugar beet pulp lixiviation in batch reactors: Effect of pH control. Chem. Eng. J. 2014, 255, 492–499. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Piao, C.; Wang, Z.; Zhao, K.; Du, M.; Wang, K. Bioaugmentation Versus pH Adjustment in High-Load Food Waste Anaerobic Digestion: Divergent Microbial Responses and Methanogenesis Regulation. Fermentation 2025, 11, 702. https://doi.org/10.3390/fermentation11120702
Piao C, Wang Z, Zhao K, Du M, Wang K. Bioaugmentation Versus pH Adjustment in High-Load Food Waste Anaerobic Digestion: Divergent Microbial Responses and Methanogenesis Regulation. Fermentation. 2025; 11(12):702. https://doi.org/10.3390/fermentation11120702
Chicago/Turabian StylePiao, Chenyu, Zhe Wang, Keqian Zhao, Mengfei Du, and Ke Wang. 2025. "Bioaugmentation Versus pH Adjustment in High-Load Food Waste Anaerobic Digestion: Divergent Microbial Responses and Methanogenesis Regulation" Fermentation 11, no. 12: 702. https://doi.org/10.3390/fermentation11120702
APA StylePiao, C., Wang, Z., Zhao, K., Du, M., & Wang, K. (2025). Bioaugmentation Versus pH Adjustment in High-Load Food Waste Anaerobic Digestion: Divergent Microbial Responses and Methanogenesis Regulation. Fermentation, 11(12), 702. https://doi.org/10.3390/fermentation11120702