Fermented Cultured Wild Ginseng Roots (Panax ginseng C.A. Meyer) Using Limosilactobacillus fermentum HY7303 Enhances the Intestinal Barrier by Bioconversion of Ginsenosides and Extracellular Vesicle Production
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. Measurement of pH, Total Carbohydrates, and Reducing Sugar Contents
2.3. High-Performance Liquid Chromatography (HPLC) Analysis
2.4. Tangential Flow Filtration (TFF) System and Size Analyze of EVs
2.5. Evaluation of Barrier Improvement Efficacy
2.5.1. Cell Culture
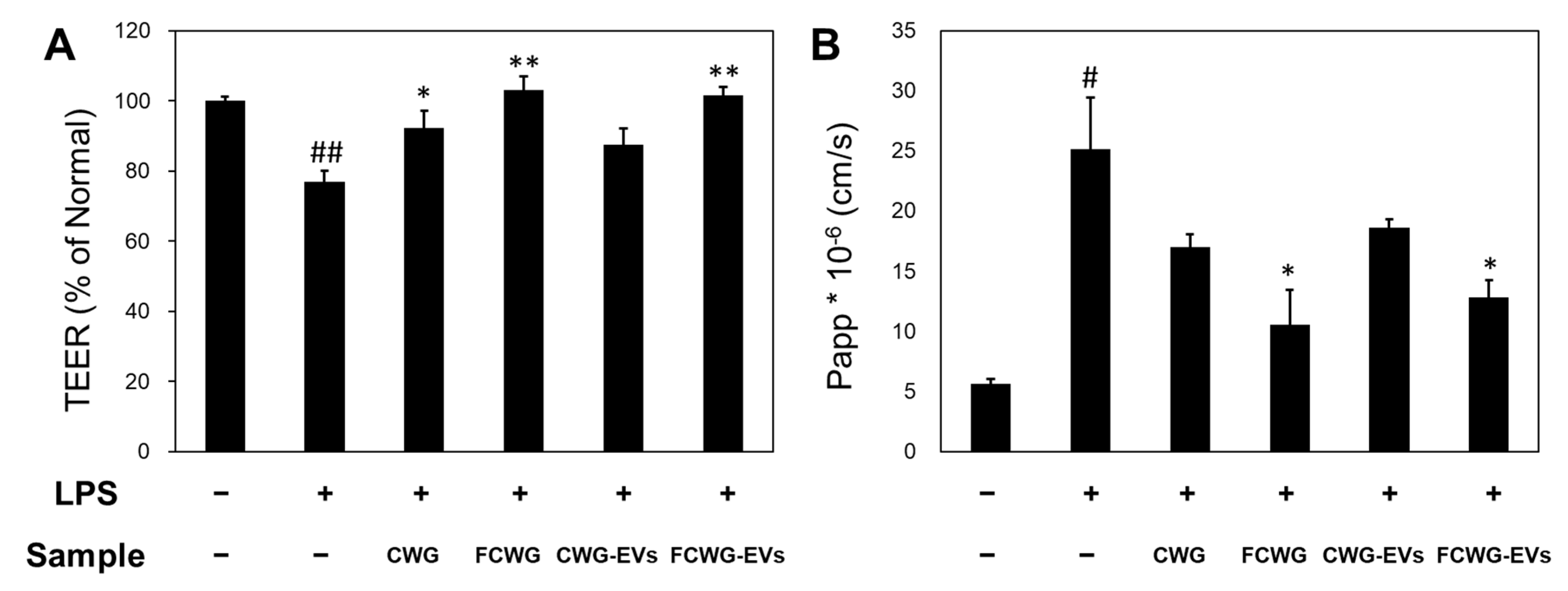
2.5.2. Transepithelial Electrical Resistance (TEER)
2.5.3. Determination of Paracellular Apparent Permeability
- dQ/dt: The amount of product present in the basal (A-B) compartment as a function of time (nmol/s).
- A: Area (cm2).
- C0: Initial concentration (nmol/mL).
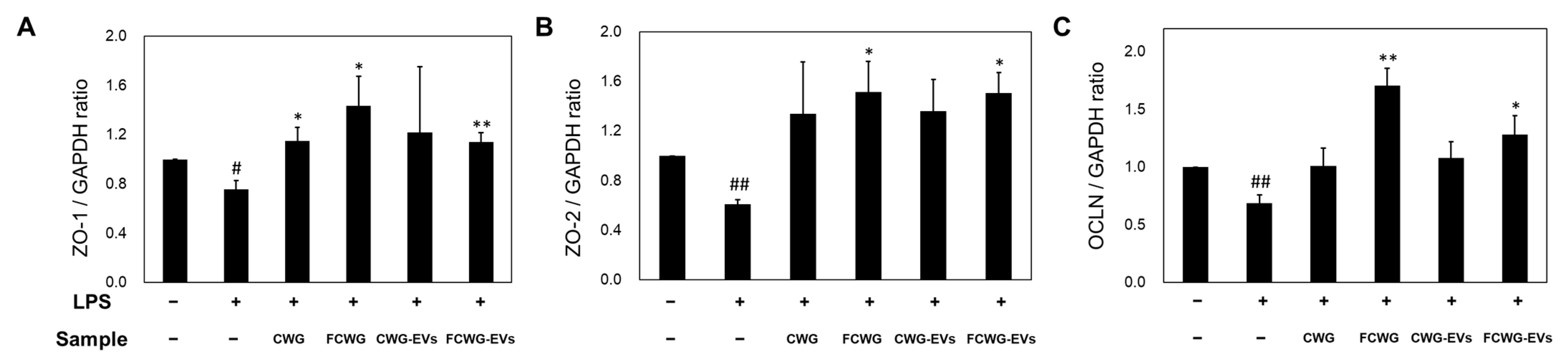
2.5.4. qRT-PCR
2.6. Statistical Analysis
3. Results
3.1. Screening of Bacteria in CWG
3.2. Physiological Changes of CWG by Fermentation Using HY7303
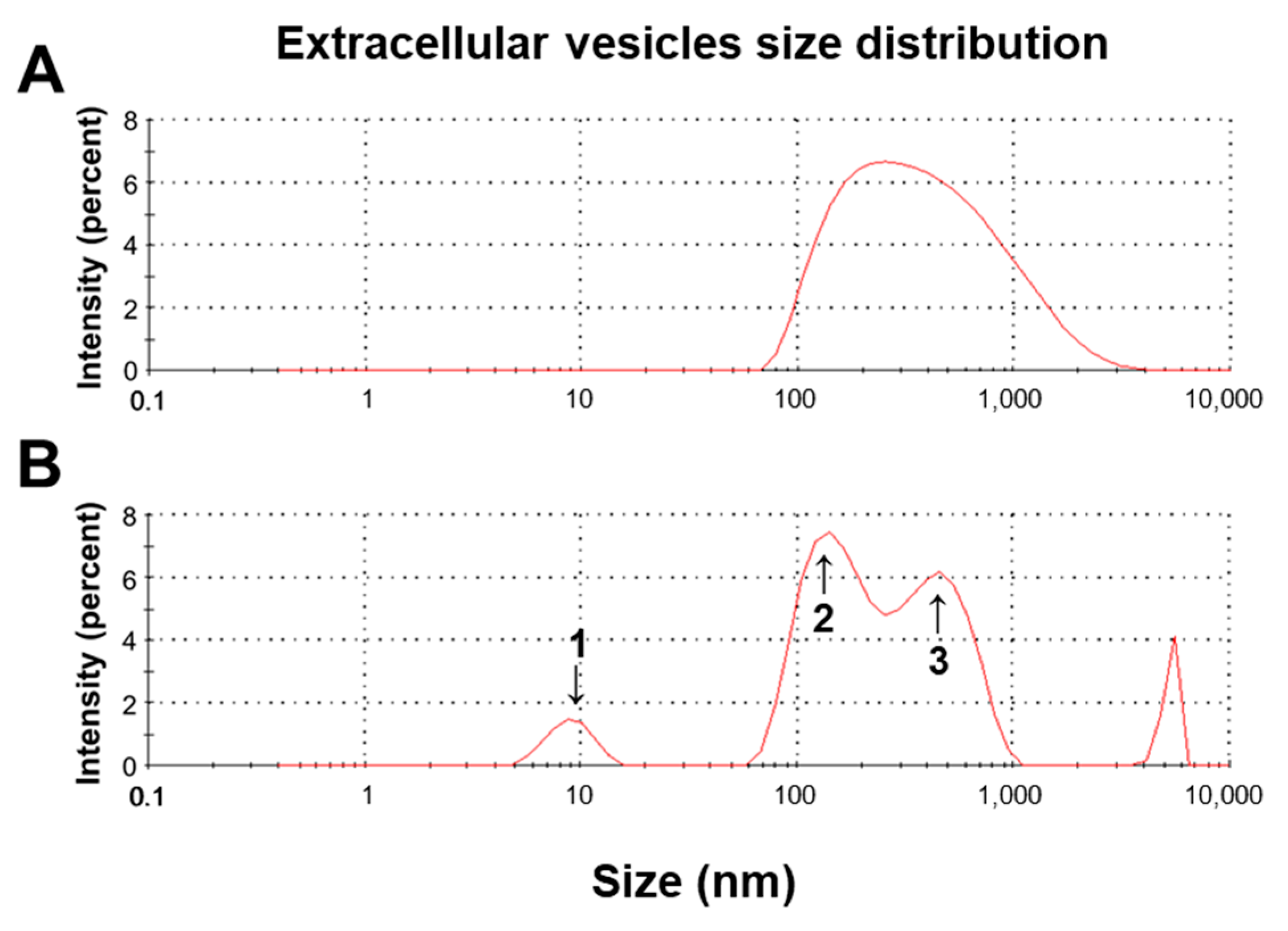
3.3. Size Difference between CWG- and FCWG-Derived EVs
3.4. Effect of Gut Health for Treatment of FCWG and EVs
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Yoo, Y.-G.; Joung, M.-S.; Lee, Y.-H.; Kim, J.-Y.; Joong-Hoi, K.; Kee-Yoeup, P. A Study on the Effect of Mountain Ginseng Adventitious Roots Extract. J. Soc. Cosmet. Scientists Korea 2004, 30, 337–383. [Google Scholar]
- Hong, M.L.; Lim, H.K.; Ji-Eun, P.; Neung, J.J.; Young, J.L.; Moonjae, C.; Somi Kim, C. The Antihypertensive and Vasodilating Effects of Adventitious Root Extracts of Wild Ginseng. J. Korean Soc. Appl. Biol. Chem. 2009, 5, 102–107. [Google Scholar]
- Park, H.P.S.; Sang, H. Production and quality of mountain ginseng. In Proceedings of the 8th International Symposium on Ginseng, Korean Society of Ginseng, Seoul, Republic of Korea, 28–31 October 2002; pp. 456–466. [Google Scholar]
- Kim, E.-L.; Lee, H.Y.; Lee, H.R.; Kim, E.Y.; Yoon, M.C.; Shin, S.S. Mountain cultivated ginseng water boiled extract decreases blood glucose level and improves lipid metabolism in male db/db mice. Korean J. Herbol. 2012, 27, 69–75. [Google Scholar] [CrossRef][Green Version]
- Lee, Y.W. Mass production of wild ginseng (Panax ginseng C. A. Meyer) roots in a bioreactor and enhancement of ginsenoside contents using an elicitor, methyl jasmonate. Ph.D. Thesis, Seoul National University, Seoul, Republic of Korea, 2007; pp. 57–68. [Google Scholar]
- Chang-Sik, S.; Do-Hyun, L.; Sung-Han, K.; Min-Ho, S.; Chang-Ho, J.; Ki-Hwan, S. Ginsenoside Contents and Antioxidative Activities from Red Ginseng Treated with High Hydrostatic Pressure. J. Agric. Life Sci. 2010, 44, 133–140. [Google Scholar]
- Cha, B.-C.; Yoon, H.C.; Dae-Ho, L.; Jae-Seuk, P.; Ki-Rok, K. Component analysis of cultivated ginseng and mountain ginseng to the change of ginsenoside components in the process of heating and fermentation. J. Pharmacopunct. 2010, 13, 33–49. [Google Scholar] [CrossRef]
- Akao, T.; Kanaoka, M.; Kobashi, K. Appearance of compound K, a major metabolite of ginsenoside Rb1 by intestinal bacteria, in rat plasma after oral administration--measurement of compound K by enzyme immunoassay. Biol. Pharm. Bull. 1998, 21, 245–249. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.J.; Cosmetics, H.H.; Seong, E.S.; Yoo, J.H.; Lee, J.G.; Kim, N.J.; Choi, S.K.; Lim, J.D.; Yu, C.Y. Biological Activity of Panax ginseng C. A. Meyer Culture Roots. Fermented with Microorganisms. Korean J. Med. Crop. Sci. 2016, 24, 191–197. [Google Scholar] [CrossRef]
- Kang, S.-W.; Min, H.-Y. Ginseng, the ‘immunity boost’: The effects of panax ginseng on immune system. J. Ginseng Res. 2012, 36, 354. [Google Scholar] [CrossRef]
- Yuan, C.-S.; Wang, C.-Z.; Wicks, S.M.; Qi, L.-W. Chemical and pharmacological studies of saponins with a focus on American ginseng. J. Ginseng Res. 2010, 34, 160–167. [Google Scholar] [CrossRef]
- Huang, X.; Liu, Y.; Zhang, Y.; Li, S.-P.; Yue, H.; Chen, C.-B.; Liu, S.-Y. Multicomponent assessment and ginsenoside conversions of Panax quinquefolium L. roots before and after steaming by HPLC-MSn. J. Ginseng Res. 2010, 43, 27–37. [Google Scholar] [CrossRef]
- Cui, C.-H.; Kim, J.-K.; Kim, S.-C.; Im, W.-T. Characterization of a ginsenoside-transforming β-glucosidase from Paenibacillus mucilaginosus and its application for enhanced production of minor ginsenoside F2. PLoS ONE 2014, 9, e85727. [Google Scholar] [CrossRef] [PubMed]
- Park, E.-H.; Kim, Y.-J.; Yamabe, N.; Park, S.-H.; Kim, H.-K.; Jang, H.-J.; Kim, J.H.; Cheon, G.J.; Ham, J.; Kang, K.S. Stereospecific anticancer effects of ginsenoside Rg3 epimers isolated from heat-processed American ginseng on human gastric cancer cell. J. Ginseng Res. 2014, 38, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Siddiqi, M.Z.; Ahn, S.; Kang, S.; Kim, Y.; Veerappan, K.; Yang, D.; Yang, D. Stimulative effect of ginsenosides Rg5:Rk1 on murine osteoblastic MC3T3-E1 cells. Phytother. Res. 2014, 28, 1447–1455. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Si, M.; Wang, Y.; Liu, L.; Zhang, Y.; Zhou, A.; Wei, W. Ginsenoside metabolite compound K exerts anti-inflammatory and analgesic effects via downregulating COX2. Inflammopharmacology 2019, 27, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Schrezenmeir, J.; de Vrese, M. Probiotics, prebiotics, and synbiotics—Approaching a definition. Am. J. Clin. Nutr. 2001, 73, 361s–364s. [Google Scholar] [CrossRef]
- Figueroa-González, I.; Cruz-Guerrero, A.; Quijano, G. The benefits of probiotics on human health. J. Microbial Biochem. Technol. S 2011, 1, 1948–5948. [Google Scholar]
- Parvez, S.; Malik, K.A.; Ah Kang, S.; Kim, H.-Y. Probiotics and their fermented food products are beneficial for health. J. Appl. Microbiol. 2006, 100, 1171–1185. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Lee, K.; Hsu, M.; Nau, G.; Mylonakis, E.; Ramratnam, B. Lactobacillus-derived extracellular vesicles enhance host immune responses against vancomycin-resistant enterococci. BMC Microbiol. 2017, 17, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Colombo, M.; Raposo, G.; Théry, C. Biogenesis, Secretion, and Intercellular Interactions of Exosomes and Other Extracellular Vesicles. Annu. Rev. Cell Dev. Biol. 2014, 30, 255–289. [Google Scholar] [CrossRef]
- Tkach, M.; Théry, C. Communication by Extracellular Vesicles: Where We Are and Where We Need to Go. Cell 2016, 164, 1226–1232. [Google Scholar] [CrossRef]
- Sel, A.; Mäger, I.; Breakefield, X.O.; Wood, M.J. Extracellular vesicles: Biology and emerging therapeutic opportunities. Nat. Rev. Drug Discov. 2013, 12, 347–357. [Google Scholar]
- Kim, J.-H.; Jeun, E.-J.; Hong, C.-P.; Kim, S.-H.; Jang, M.S.; Lee, E.-J.; Moon, S.J.; Yun, C.H.; Im, S.-H.; Jeong, S.-G.; et al. Extracellular vesicle–derived protein from Bifidobacterium longum alleviates food allergy through mast cell suppression. J. Allergy Clin. Immunol. 2016, 137, 507–516.e8. [Google Scholar] [CrossRef] [PubMed]
- Behzadi, E.; Mahmoodzadeh Hosseini, H.; Imani Fooladi, A.A. The inhibitory impacts of Lactobacillus rhamnosus GG-derived extracellular vesicles on the growth of hepatic cancer cells. Microb. Pathog. 2017, 110, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Defourny, K.A.Y.; Smid, E.J.; Abee, T. Gram-Positive Bacterial Extracellular Vesicles and Their Impact on Health and Disease. Front. Microbiol. 2018, 9, 1502. [Google Scholar] [CrossRef] [PubMed]
- Kang, E.A.; Choi, H.-I.; Hong, S.W.; Kang, S.; Jegal, H.-Y.; Choi, E.W.; Park, B.-S.; Kim, J.S. Extracellular Vesicles Derived from Kefir Grain Lactobacillus Ameliorate Intestinal Inflammation via Regulation of Proinflammatory Pathway and Tight Junction Integrity. Biomedicines 2020, 8, 522. [Google Scholar] [CrossRef] [PubMed]
- Mo, S.-J.; Nam, B.; Bae, C.-H.; Park, S.-D.; Shim, J.-J.; Lee, J.-L. Characterization of novel Lactobacillus paracasei HY7017 capable of improving physiological properties and immune enhancing effects using red ginseng extract. Fermentation 2021, 7, 238. [Google Scholar] [CrossRef]
- Nielsen, S.S. Total carbohydrate by phenol-sulfuric acid method. In Food Analysis Laboratory Manual; Springer: Cham, Switzerland, 2017; pp. 137–141. ISBN 978-1-4419-1462-0. [Google Scholar]
- Miller, G.L. Use of Dinitrosalicylic acid reagent for determination of reducing sugar. Anal. Chem. 1959, 31, 426–428. [Google Scholar] [CrossRef]
- Kim, H.A.; Kim, S.; Chang, S.H.; Hwang, H.J.; Choi, Y.-N. Anti-arthritic effect of ginsenoside Rb1 on collagen induced arthritis in mice. Int. Immunopharmacol. 2007, 7, 1286–1291. [Google Scholar] [CrossRef]
- Yun, T.-K. Experimental and epidemiological evidence on non-organ specific cancer preventive effect of Korean ginseng and identification of active compounds. Mutat. Res. Mol. Mech. Mutagen. 2003, 523–524, 63–74. [Google Scholar] [CrossRef]
- An, K.S.; Choi, Y.O.; Lee, S.M.; Ryu, H.Y.; Kang, S.J.; Yeon, Y.; Kim, Y.R.; Lee, J.G.; Kim, C.J.; Lee, Y.J.; et al. Ginsenosides Rg5 and Rk1 Enriched Cultured Wild Ginseng Root Extract Bioconversion of Pediococcus pentosaceus HLJG0702: Effect on Scopolamine-Induced Memory Dysfunction in Mice. Nutrients 2019, 11, 1120. [Google Scholar] [CrossRef]
- Jang, W.; Jang, Y.; Kim, N.-H.; Waminal, N.E.; Kim, Y.C.; Lee, J.W.; Yang, T.-J. Genetic diversity among cultivated and wild Panax ginseng populations revealed by high-resolution microsatellite markers. J. Ginseng Res. 2020, 44, 637–643. [Google Scholar] [CrossRef] [PubMed]
- Xavier, R.J.; Podolsky, D.K. Unravelling the pathogenesis of inflammatory bowel disease. Nature 2007, 448, 427–434. [Google Scholar] [CrossRef] [PubMed]
- Turner, J.R. Intestinal mucosal barrier function in health and disease. Nat. Rev. Immunol. 2009, 9, 799–809. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Al-Sadi, R.; Said, H.M.; Ma, T.Y. Lipopolysaccharide causes an increase in intestinal tight junction permeability in vitro and in vivo by inducing enterocyte membrane expression and localization of TLR-4 and CD14. Am. J. Pathol. 2013, 182, 375–387. [Google Scholar] [CrossRef] [PubMed]
- Ng, S.C.; Shi, H.Y.; Hamidi, N.; Underwood, F.E.; Tang, W.; Benchimol, E.I.; Panaccione, R.; Ghosh, S.; Wu, J.C.Y.; Chan, F.K.L.; et al. Worldwide incidence and prevalence of inflammatory bowel disease in the 21st century: A systematic review of population-based studies. Lancet 2017, 390, 2769–2778. [Google Scholar] [CrossRef]
- Rahimi, R.; Nikfar, S.; Abdollahi, M. Induction of clinical response and remission of inflammatory bowel disease by use of herbal medicines: A meta-analysis. World J. Gastroenterol. WJG 2013, 19, 5738. [Google Scholar] [CrossRef]
- Nielsen, O.H.; Munck, L.K. Drug Insight: Aminosalicylates for the treatment of IBD. Nat. Clin. Pract. Gastroenterol. Hepatol. 2007, 4, 160–170. [Google Scholar] [CrossRef] [PubMed]
- Magro, F.; Portela, F. Management of inflammatory bowel disease with infliximab and other anti-tumor necrosis factor alpha therapies. BioDrugs 2010, 24, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Pithadia, A.B.; Jain, S. Treatment of inflammatory bowel disease (IBD). Pharmacol. Rep. 2011, 63, 629–642. [Google Scholar] [CrossRef]
- Jakubczyk, D.; Leszczyńska, K.; Górska, S. The effectiveness of probiotics in the treatment of inflammatory bowel disease (IBD)—A critical review. Nutrients 2020, 12, 1973. [Google Scholar] [CrossRef]
- Veerappan, G.R.; Betteridge, J.; Young, P.E. Probiotics for the treatment of inflammatory bowel disease. Curr. Gastroenterol. Rep. 2012, 14, 324–333. [Google Scholar] [CrossRef] [PubMed]
- Marco, M.L.; Heeney, D.; Binda, S.; Cifelli, C.J.; Cotter, P.D.; Foligné, B.; Gänzle, M.; Kort, R.; Pasin, G.; Pihlanto, A.; et al. Health benefits of fermented foods: Microbiota and beyond. Curr. Opin. Biotechnol. 2017, 44, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Michlmayr, H.; Kneifel, W. β-Glucosidase activities of lactic acid bacteria: Mechanisms, impact on fermented food and human health. FEMS Microbiol. Lett. 2014, 352, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Filannino, P.; Di Cagno, R.; Gobbetti, M. Metabolic and functional paths of lactic acid bacteria in plant foods: Get out of the labyrinth. Curr. Opin. Biotechnol. 2018, 49, 64–72. [Google Scholar] [CrossRef] [PubMed]
- Cho, K.M.; Lee, H.Y.; Lee, Y.M.; Seo, E.Y.; Kim, D.H.; Son, K.-H.; Lee, J.; Cho, D.Y.; Lee, J.H. Comparative assessment of compositional constituents and antioxidant effects in ginseng sprouts (Panax ginseng) through aging and fermentation processes. LWT 2022, 164, 113644. [Google Scholar] [CrossRef]
- Singhania, R.R.; Patel, A.K.; Sukumaran, R.K.; Larroche, C.; Pandey, A. Role and significance of beta-glucosidases in the hydrolysis of cellulose for bioethanol production. Bioresour. Technol. 2013, 127, 500–507. [Google Scholar] [CrossRef] [PubMed]
- Faure, D. The Family-3 glycoside hydrolases: From housekeeping functions to host-microbe interactions. Appl. Environ. Microbiol. 2002, 68, 1485–1490. [Google Scholar] [CrossRef] [PubMed]
- Park, B.; Hwang, H.; Lee, J.; Sohn, S.-O.; Lee, S.H.; Jung, M.Y.; Lim, H.I.; Park, H.W.; Lee, J.-H. Evaluation of ginsenoside bioconversion of lactic acid bacteria isolated from kimchi. J. Ginseng Res. 2017, 41, 524–530. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-K.; Cui, C.-H.; Liu, Q.; Yoon, M.-H.; Kim, S.-C.; Im, W.-T. Mass production of the ginsenoside Rg3(S) through the combinative use of two glycoside hydrolases. Food Chem. 2013, 141, 1369–1377. [Google Scholar] [CrossRef]
- Cheng, L.-Q.; Na, J.R.; Bang, M.H.; Kim, M.K.; Yang, D.-C. Conversion of major ginsenoside Rb1 to 20 (S)-ginsenoside Rg3 by Microbacterium sp. GS514. Phytochemistry 2008, 69, 218–224. [Google Scholar] [CrossRef]
- Ke, Y.; Huang, L.; Song, Y.; Liu, Z.; Liang, L.; Wang, L.; Wang, T. Preparation and pharmacological effects of minor ginsenoside nanoparticles: A review. Front. Pharmacol. 2022, 13, 974274. [Google Scholar] [CrossRef] [PubMed]
- Duan, X.; Cai, H.; Hu, T.; Lin, L.; Zeng, L.; Wang, H.; Cao, L.; Li, X. Ginsenoside Rg3 treats acute radiation proctitis through the TLR4/MyD88/NF-κB pathway and regulation of intestinal flora. Front. Cell. Infect. Microbiol. 2022, 12, 1028576. [Google Scholar] [CrossRef] [PubMed]
- Maeng, Y.-S.; Maharjan, S.; Kim, J.-H.; Park, J.-H.; Yu, Y.S.; Kim, Y.-M.; Kwon, Y.-G. Rk1, a Ginsenoside, is a new blocker of vascular leakage acting through actin structure remodeling. PLoS ONE 2013, 8, e68659. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Yang, H.; Zhu, C.; Deng, J.; Fan, D. Hypoglycemic Effect of Ginsenoside Rg5 Mediated Partly by Modulating Gut Microbiota Dysbiosis in Diabetic db/db Mice. J. Agric. Food Chem. 2020, 68, 5107–5117. [Google Scholar] [CrossRef] [PubMed]
- Gill, S.; Catchpole, R.; Forterre, P. Extracellular membrane vesicles in the three domains of life and beyond. FEMS Microbiol. Rev. 2019, 43, 273–303. [Google Scholar] [CrossRef] [PubMed]
- Zomer, A.; Vendrig, T.; Hopmans, E.S.; van Eijndhoven, M.; Middeldorp, J.M.; Pegtel, D.M. Exosomes: Fit to deliver small RNA. Commun. Integr. Biol. 2010, 3, 447–450. [Google Scholar] [CrossRef] [PubMed]
- Martínez, G.P.; Giner-Pérez, L.; Castillo-Romero, K.F. Bacterial extracellular vesicles and associated functional proteins in fermented dairy products with Lacticaseibacillus paracasei. Front. Microbiol. 2023, 14, 1165202. [Google Scholar] [CrossRef] [PubMed]
- Odenwald, M.A.; Turner, J.R. Intestinal permeability defects: Is. it time to treat? Clin. Gastroenterol. Hepatol. 2013, 11, 1075–1083. [Google Scholar] [CrossRef] [PubMed]
- Martel, J.; Chang, S.H.; Ko, Y.F.; Hwang, T.L.; Young, J.D.; Ojcius, D.M. Gut barrier disruption and chronic disease. Trends Endocrinol. Metab. TEM 2022, 33, 247–265. [Google Scholar] [CrossRef]
- Dong, J.; Liang, W.; Wang, T.; Sui, J.; Wang, J.; Deng, Z.; Chen, D. Saponins regulate intestinal inflammation in colon cancer and IBD. Pharmacol. Res. 2019, 144, 66–72. [Google Scholar] [CrossRef]
- Iizuka, M.; Konno, S. Wound healing of intestinal epithelial cells. World J. Gastroenterol. WJG 2011, 17, 2161. [Google Scholar] [CrossRef] [PubMed]
- Tong, L.; Zhang, S.; Liu, Q.; Huang, C.; Hao, H.; Tan, M.S.; Yu, X.; Lou, C.K.L.; Huang, R.; Zhang, Z.; et al. Milk-derived extracellular vesicles protect intestinal barrier integrity in the gut-liver axis. Sci. Adv. 2023, 9, ade5041. [Google Scholar] [CrossRef] [PubMed]
- Chelakkot, C.; Ghim, J.; Ryu, S.H. Mechanisms regulating intestinal barrier integrity and its pathological implications. Exp. Mol. Med. 2018, 50, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Salim, S.Y.; Söderholm, J.D. Importance of disrupted intestinal barrier in inflammatory bowel diseases. Inflamm. Bowel Dis. 2011, 17, 362–381. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Fan, D.; Zhou, Z.; Wang, Y.; Huang, X.; Zhang, L.; Wu, D.; Ren, Y.; Lu, F.; Gao, X. Ginsenoside protects intestinal barrier function and improves epithelium injury in sepsis by regulating the miR-30e-5p/FBXO11 axis. Electron. J. Biotechnol. 2023, 66, 67–74. [Google Scholar] [CrossRef]
- Seong, M.A.; Woo, J.K.; Kang, J.-H.; Jang, Y.S.; Choi, S.; Jang, Y.S.; Lee, T.H.; Jung, K.H.; Kang, D.K.; Hurh, B.S.; et al. Oral administration of fermented wild ginseng ameliorates DSS-induced acute colitis by inhibiting NF-κB signaling and protects intestinal epithelial barrier. BMB Rep. 2015, 48, 419. [Google Scholar] [CrossRef]
- Ratajczak, J.; Wysoczynski, M.; Hayek, F.; Janowskawieczorek, A.; Ratajczak, M.Z. Membrane-derived microvesicles: Important and underappreciated mediators of cell-to-cell communication. Leukemia 2006, 20, 1487–1495. [Google Scholar] [CrossRef]


| Time (min) | A: Water (%) | B: ACN (%) | Flow Rate (mL/min) |
|---|---|---|---|
| 0 | 83 | 17 | 0.8 |
| 25 | 75 | 25 | 0.8 |
| 38 | 58 | 42 | 0.8 |
| 85 | 40 | 60 | 0.8 |
| 95 | 20 | 80 | 0.8 |
| 115 | 83 | 17 | 0.8 |
| Gene Symbol | Forward Sequence | Amplicon Size (bp) |
|---|---|---|
| Reverse Sequence | ||
| TJP1 | GTCCAGAATCTCGGAAAAGTGCC | 148 |
| CTTTCAGCGCACCATACCAACC | ||
| TJP2 | ATTAGTGCGGGAGGATGCCGTT | 92 |
| TCTGCCACAAGCCAGGATGTCT | ||
| OCLN | ATGGCAAAGTGAATGACAAGCGG | 68 |
| CTGTAACGAGGCTGCCTGAAGT | ||
| GAPDH | GTCTCCTCTGACTTCAACAGCG | 122 |
| ACCACCCTGTTGCTGTAGCCAA |
| Measurement | Before Fermentation | After Fermentation | p-Value |
|---|---|---|---|
| pH | 5.27 ± 0.04 1 | 4.24 ± 0.19 1 | 0.015 |
| Total carbohydrates (mg D-glucose/mL) | 150.28 ± 5.98 1 | 126.77 ± 5.93 1 | 0.017 |
| Reducing sugar (mg D-glucose/mL) | 4.35 ± 0.10 1 | 4.86 ± 0.13 1 | 0.016 |
| Ginsenoside | RT (min) | Ginsenoside Contents (mg/g) | |
|---|---|---|---|
| CWG | FCWG | ||
| Rg1 | 37.846 | 2.71 ± 0.02 | 1.79 ± 0.65 |
| Re | 38.395 | 2.41 ± 0.25 | 1.68 ± 0.68 |
| Rf | 54.046 | 1.28 ± 0.20 | 1.33 ± 0.45 |
| Rg2 | 59.658 | 1.76 ± 0.29 | 1.40 ± 0.78 |
| Rb1 | 64.423 | 50.09 ± 1.64 | 11.94 ± 2.81 |
| Rc | 66.589 | 14.41 ± 1.22 | 3.97 ± 0.19 |
| Rb2 | 68.919 | 18.80 ± 1.46 | 6.54 ± 0.85 |
| Rd | 73.738 | 19.37 ± 2.95 | 14.19 ± 4.34 |
| Rg3(s) | 93.603 | 1.13 ± 0.10 | 29.22 ± 1.78 |
| Rk1 | 100.713 | 1.48 ± 0.06 | 33.02 ± 0.80 |
| Rg5 | 101.940 | 2.77 ± 0.27 | 25.29 ± 1.40 |
| Total | - | 116.21 ± 2.58 | 130.37 ± 2.29 |
| Source | Peaks | Size (nm) | Distribution Rate (%) |
|---|---|---|---|
| CWG | - | 493.7 ± 435.9 | 100 |
| FCWG | 1 | 9.065 ± 1.98 | 5.8 |
| 2 | 155.6 ± 51.5 | 47.5 | |
| 3 | 459 ± 161.3 | 41.1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mo, S.-J.; Kim, E.-J.; Lee, Y.-H.; Park, S.-D.; Shim, J.-J.; Lee, J.-L.; Lee, J.-H. Fermented Cultured Wild Ginseng Roots (Panax ginseng C.A. Meyer) Using Limosilactobacillus fermentum HY7303 Enhances the Intestinal Barrier by Bioconversion of Ginsenosides and Extracellular Vesicle Production. Fermentation 2024, 10, 362. https://doi.org/10.3390/fermentation10070362
Mo S-J, Kim E-J, Lee Y-H, Park S-D, Shim J-J, Lee J-L, Lee J-H. Fermented Cultured Wild Ginseng Roots (Panax ginseng C.A. Meyer) Using Limosilactobacillus fermentum HY7303 Enhances the Intestinal Barrier by Bioconversion of Ginsenosides and Extracellular Vesicle Production. Fermentation. 2024; 10(7):362. https://doi.org/10.3390/fermentation10070362
Chicago/Turabian StyleMo, Sung-Joon, Eun-Ji Kim, Yun-Ha Lee, Soo-Dong Park, Jae-Jung Shim, Jung-Lyul Lee, and Jae-Hwan Lee. 2024. "Fermented Cultured Wild Ginseng Roots (Panax ginseng C.A. Meyer) Using Limosilactobacillus fermentum HY7303 Enhances the Intestinal Barrier by Bioconversion of Ginsenosides and Extracellular Vesicle Production" Fermentation 10, no. 7: 362. https://doi.org/10.3390/fermentation10070362
APA StyleMo, S.-J., Kim, E.-J., Lee, Y.-H., Park, S.-D., Shim, J.-J., Lee, J.-L., & Lee, J.-H. (2024). Fermented Cultured Wild Ginseng Roots (Panax ginseng C.A. Meyer) Using Limosilactobacillus fermentum HY7303 Enhances the Intestinal Barrier by Bioconversion of Ginsenosides and Extracellular Vesicle Production. Fermentation, 10(7), 362. https://doi.org/10.3390/fermentation10070362







