An Analysis of the Factors Influencing Cadmium Removal in Aquatic Environments by Chlorella vulgaris-Derived Solids
Abstract
1. Introduction
2. Materials and Methods
2.1. CV and Derived Materials
2.2. Materials Characterization
- pH and electrical conductivity (EC): Measured after the samples had been put in deionized water (with a 1/10 mass ratio) for 24 h on a reciprocating shaker at 20 °C. Then, pH value and electrical conductivity at 20 °C were measured according to the ASTM 2866-D standard method, employing an EYELA pH meter and 4320-JENWAY conductivity meter, respectively;
- Ash content: Obtained by keeping the samples at 730 °C for 8 h in a muffle furnace; the obtained ashes were then weighed, and their content was calculated by dividing it by the initial dry mass of the material;
- Volatile matter content: Measured according to ISO 562-2010 [28]. One gram of each sample was put into a porcelain crucible. Samples were heated via a furnace at 950 °C for 10 min in nitrogen flow, and after cooling, the final mass of the samples was weighed. The amount of the volatilized substance was calculated as the relative weight difference before and after the treatment;
- Ultimate analysis: The measurement of carbon (C), hydrogen (H), nitrogen (N), and sulfur (S) mass fractions within the samples was performed using a Thermo Finnigan elemental analyzer (FlashEA 1112 series). Oxygen was instead calculated by the difference in the mass fractions of the other elements and ash. Employing the element mass fractions, the products’ gross calorific value (GCV) was obtained through the equation proposed by Friedl et al. [29]: GCV = 3.55C2 − 232C − 2230H + 51.2C × H + 131N + 20,600 (MJ kg−1);
- Specific surface area (SSA): Measured based on the Brunauer–Emmett–Teller (BET) method using the Belsorp mini II Surface Area Analyzer (Microtrac Bel Corp, Haan, Germany). External or non-microporous area and micro-pore volume (Vp) were calculated based on nitrogen gas adsorption before measuring the samples’ surface areas.
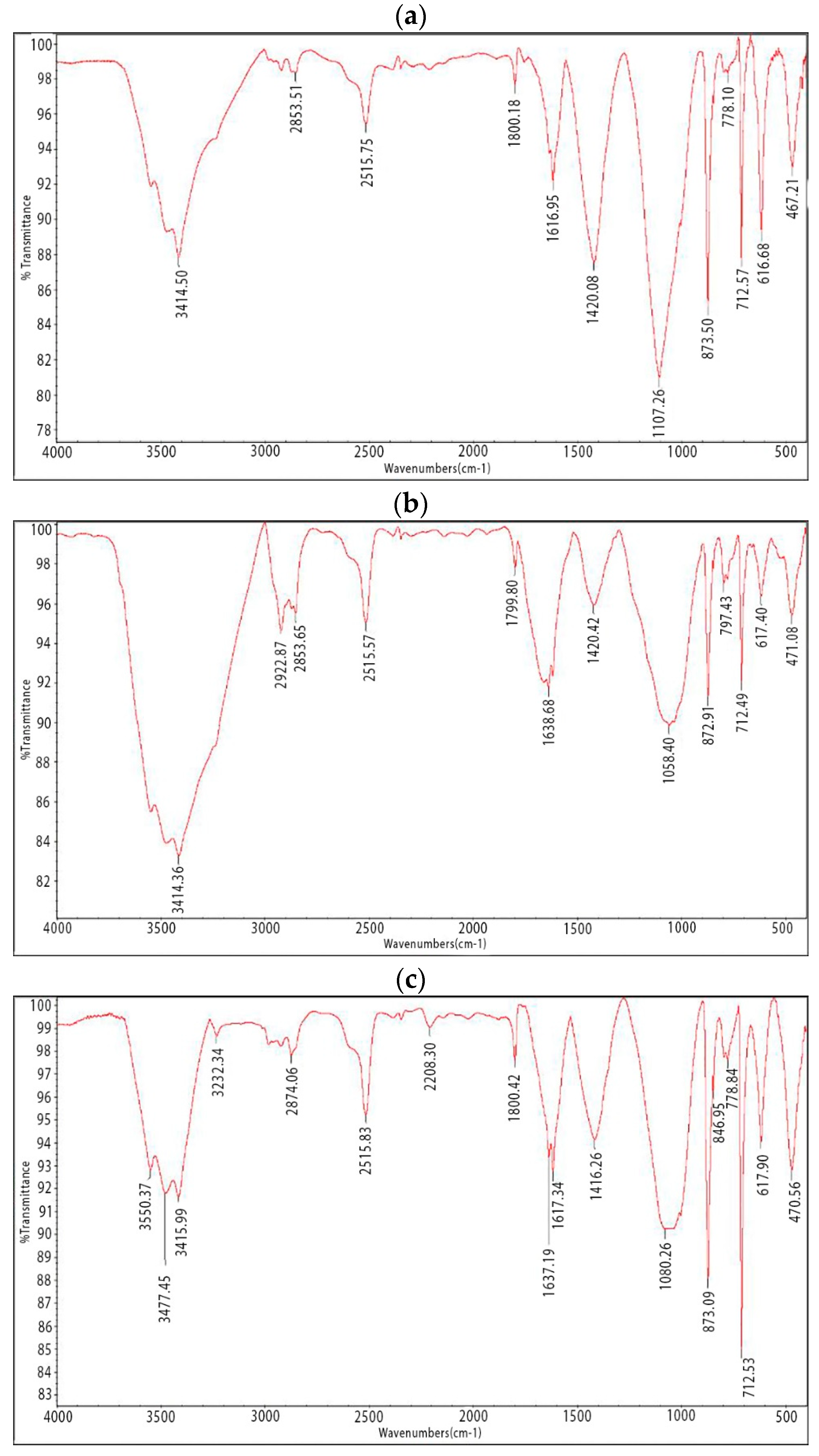
- Surface properties: The morphology of the samples was investigated using the FESEM-EDX Inspect 24 (ZEISS Sigma 300, Oberkochen, Germany). FESEM testing was conducted to determine the surface and pore patterns of the products. The surface functional groups of activated carbon were analyzed using an FTIR spectrophotometer (Nicolet is 10/Thermo Scientific, Waltham, MA, USA).
2.3. Adsorption Experiments
2.3.1. Isotherm Study
2.3.2. Structural Equation Modeling (SEM)
- Latent variables: These are variables that cannot be directly observed but are inferred from a set of observed variables that are related to them. Latent variables are also known as constructs or factors;
- Observed variables: These are variables that are directly measured or observed in the study. They are also referred to as indicators or manifest variables;
- Structural model: This is the part of SEM that represents the hypothesized relationships between the latent variables. It is specified by proposing paths (direct or indirect effects) between the latent variables based on theoretical assumptions;
- Measurement model: This represents the relationships between the latent variables and their observed indicators. It specifies how the latent variables are measured by the observed variables;
- SEM allows researchers to test complex hypotheses, explore the underlying structure of the data, and evaluate the fit of the proposed models to the data. It also provides estimates of the strength and significance of the relationships between variables and allows for the inclusion of measurement errors in the analysis.
3. Results and discussion
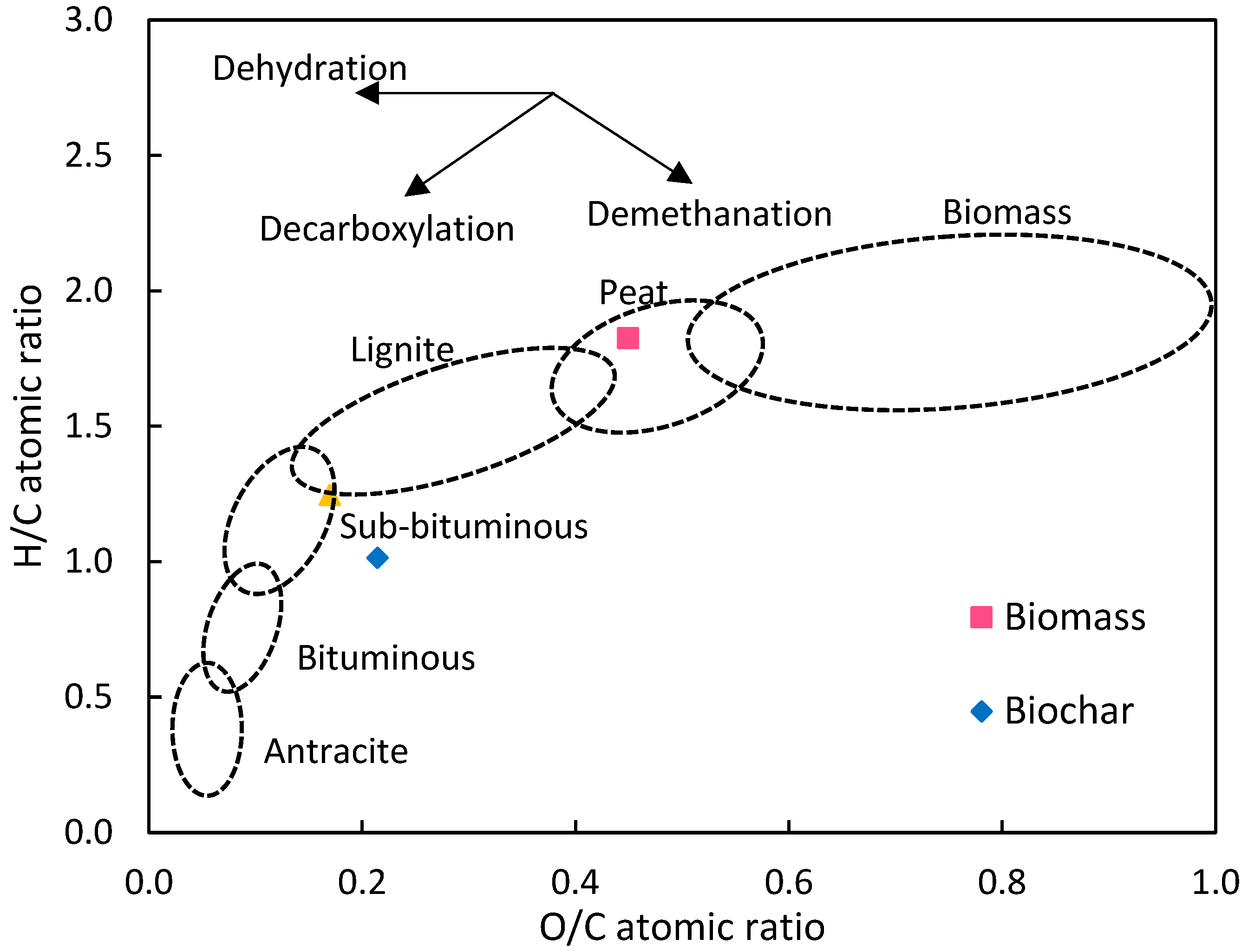
3.1. Properties of CV and Its Biochar and Hydrochar
3.2. Adsorption Tests
3.2.1. Results
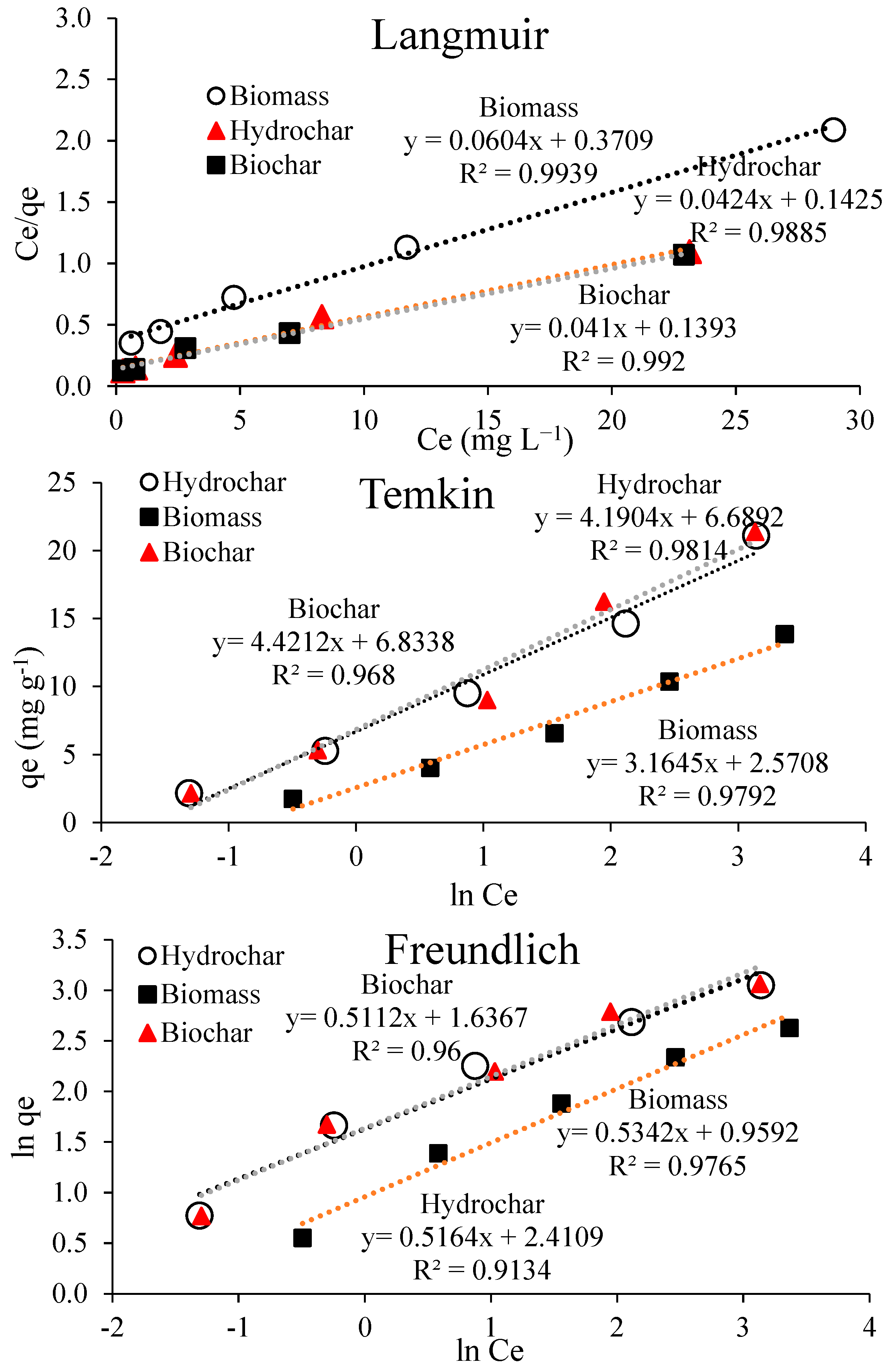
3.2.2. Adsorption Isotherms Modeling
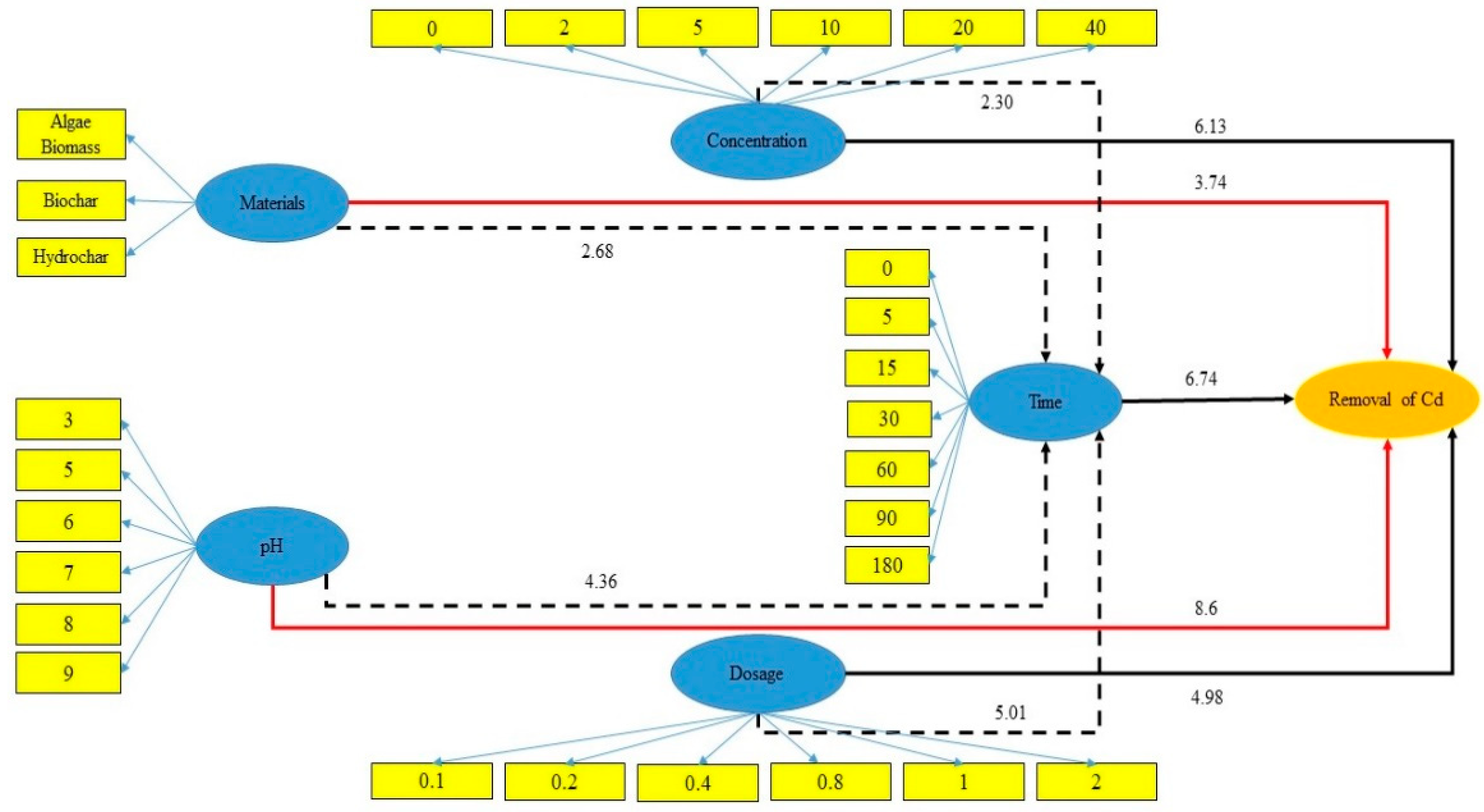
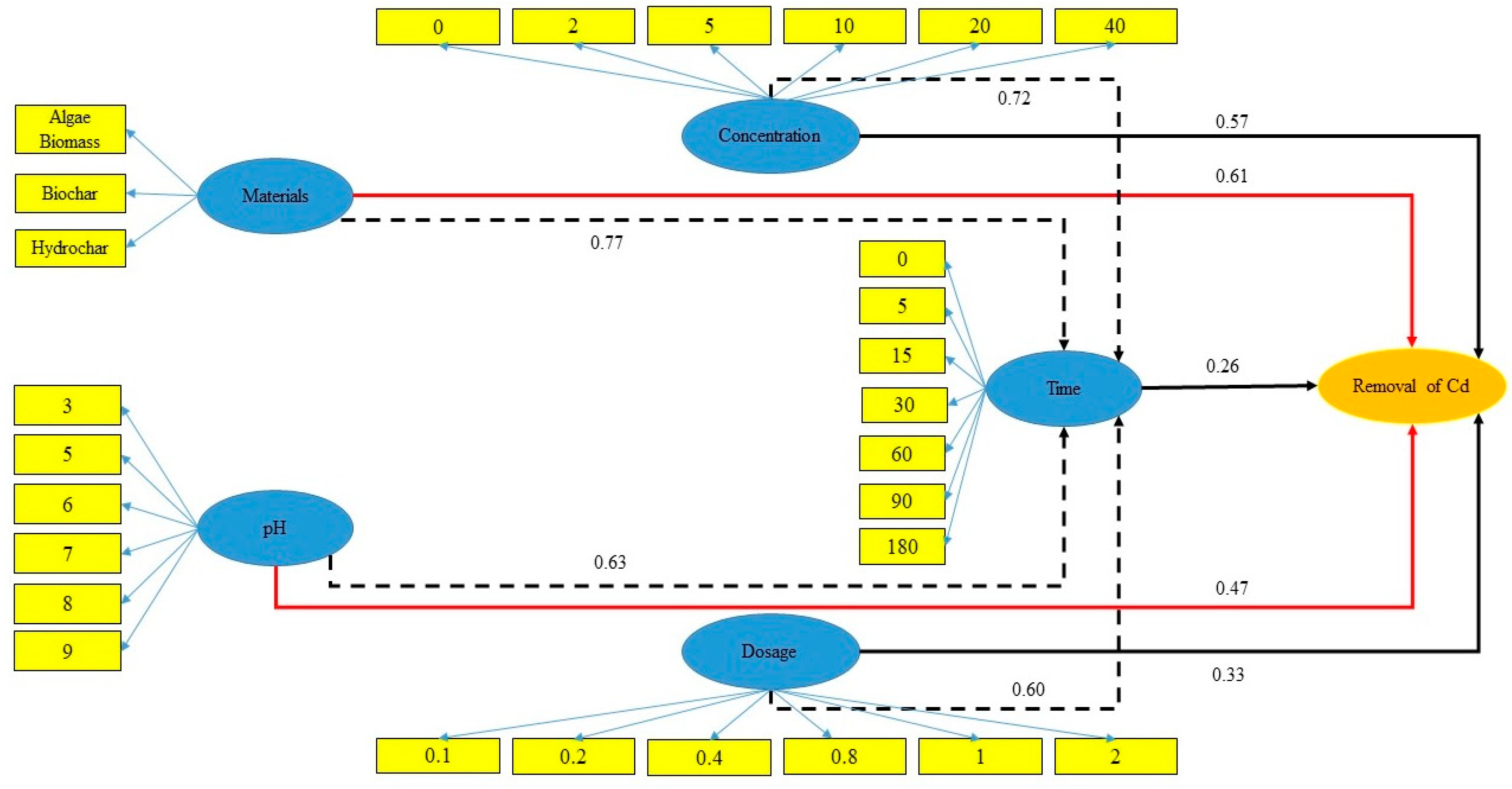
3.2.3. Structural Equation Modeling (SEM)
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- El-Sorogy, A.S.; Youssef, M.; Al-Hashim, M.H. Water Quality Assessment and Environmental Impact of Heavy Metals in the Red Sea Coastal Seawater of Yanbu, Saudi Arabia. Water 2023, 15, 201. [Google Scholar] [CrossRef]
- Shetaia, S.A.; Nasr, R.A.; Lasheen, E.S.R.; Dar, M.A.; Al-Mur, B.A.; Zakaly, H.M.H. Assessment of Heavy Metals Contamination of Sediments and Surface Waters of Bitter Lake, Suez Canal, Egypt: Ecological Risks and Human Health. Mar. Pollut. Bull. 2023, 192, 115096. [Google Scholar] [CrossRef] [PubMed]
- Gautam, R.K.; Sharma, S.K.; Mahiya, S.; Chattopadhyaya, M.C. CHAPTER 1. Contamination of Heavy Metals in Aquatic Media: Transport, Toxicity and Technologies for Remediation. In Heavy Metals In Water; Royal Society of Chemistry: Cambridge, UK, 2014; pp. 1–24. [Google Scholar]
- Liapun, V.; Motola, M. Current Overview and Future Perspective in Fungal Biorecovery of Metals from Secondary Sources. J. Environ. Manag. 2023, 332, 117345. [Google Scholar] [CrossRef] [PubMed]
- Zeng, X.; Xu, X.; Qin, Q.; Ye, K.; Wu, W.; Huo, X. Heavy Metal Exposure Has Adverse Effects on the Growth and Development of Preschool Children. Environ. Geochem. Health 2019, 41, 309–321. [Google Scholar] [CrossRef] [PubMed]
- Usman, A.; Sallam, A.; Zhang, M.; Vithanage, M.; Ahmad, M.; Al-Farraj, A.; Ok, Y.S.; Abduljabbar, A.; Al-Wabel, M. Sorption Process of Date Palm Biochar for Aqueous Cd (II) Removal: Efficiency and Mechanisms. Water Air Soil Pollut. 2016, 227, 449. [Google Scholar] [CrossRef]
- Appenroth, K.J. Definition of “Heavy Metals” and Their Role in Biological Systems. In Soil Heavy Metals; Soil Biology; Springer: Berlin/Heidelberg, Germany, 2010; Volume 19, pp. 19–29. [Google Scholar]
- Mishra, S.; Bharagava, R.N.; More, N.; Yadav, A.; Zainith, S.; Mani, S.; Chowdhary, P. Heavy Metal Contamination: An Alarming Threat to Environment and Human Health. In Environmental Biotechnology: For Sustainable Future; Springer: Singapore, 2019; pp. 103–125. [Google Scholar]
- Bouida, L.; Rafatullah, M.; Kerrouche, A.; Qutob, M.; Alosaimi, A.M.; Alorfi, H.S.; Hussein, M.A. A Review on Cadmium and Lead Contamination: Sources, Fate, Mechanism, Health Effects and Remediation Methods. Water 2022, 14, 3432. [Google Scholar] [CrossRef]
- Rafati Rahimzadeh, M.; Rafati Rahimzadeh, M.; Kazemi, S.; Moghadamnia, A.-A. Cadmium Toxicity and Treatment: An Update. Casp. J. Intern. Med. 2017, 8, 135–145. [Google Scholar] [CrossRef]
- Irfan, M.; Liu, X.; Hussain, K.; Mushtaq, S.; Cabrera, J.; Zhang, P. The Global Research Trend on Cadmium in Freshwater: A Bibliometric Review. Environ. Sci. Pollut. Res. 2021, 30, 71585–71598. [Google Scholar] [CrossRef]
- C, F.C.; Kamalesh, T.; Kumar, P.S.; Rangasamy, G. A Critical Review on the Sustainable Approaches for the Removal of Toxic Heavy Metals from Water Systems. Ind. Eng. Chem. Res. 2023, 62, 8575–8601. [Google Scholar] [CrossRef]
- Dai, Y.; Zhang, N.; Xing, C.; Cui, Q.; Sun, Q. The Adsorption, Regeneration and Engineering Applications of Biochar for Removal Organic Pollutants: A Review. Chemosphere 2019, 223, 12–27. [Google Scholar] [CrossRef]
- Li, S.; Tasnady, D. Biochar for Soil Carbon Sequestration: Current Knowledge, Mechanisms, and Future Perspectives. C 2023, 9, 67. [Google Scholar] [CrossRef]
- O’Connor, D.; Peng, T.; Zhang, J.; Tsang, D.C.W.; Alessi, D.S.; Shen, Z.; Bolan, N.S.; Hou, D. Biochar Application for the Remediation of Heavy Metal Polluted Land: A Review of in Situ Field Trials. Sci. Total Environ. 2018, 619–620, 815–826. [Google Scholar] [CrossRef] [PubMed]
- Fang, J.; Gao, B.; Chen, J.; Zimmerman, A.R. Hydrochars Derived from Plant Biomass under Various Conditions: Characterization and Potential Applications and Impacts. Chem. Eng. J. 2015, 267, 253–259. [Google Scholar] [CrossRef]
- Hu, B.; Wang, K.; Wu, L.; Yu, S.; Antonietti, M.; Titirici, M. Engineering Carbon Materials from the Hydrothermal Carbonization Process of Biomass. Adv. Mater. 2010, 22, 813–828. [Google Scholar] [CrossRef] [PubMed]
- Saha, N.; Volpe, M.; Fiori, L.; Volpe, R.; Messineo, A.; Reza, M.T. Cationic Dye Adsorption on Hydrochars of Winery and Citrus Juice Industries Residues: Performance, Mechanism, and Thermodynamics. Energies 2020, 13, 4686. [Google Scholar] [CrossRef]
- Purnomo, C.; Castello, D.; Fiori, L. Granular Activated Carbon from Grape Seeds Hydrothermal Char. Appl. Sci. 2018, 8, 331. [Google Scholar] [CrossRef]
- Li, G.; Xiao, W.; Yang, T.; Lyu, T. Optimization and Process Effect for Microalgae Carbon Dioxide Fixation Technology Applications Based on Carbon Capture: A Comprehensive Review. C 2023, 9, 35. [Google Scholar] [CrossRef]
- Ru, I.T.K.; Sung, Y.Y.; Jusoh, M.; Wahid, M.E.A.; Nagappan, T. Chlorella Vulgaris: A Perspective on Its Potential for Combining High Biomass with High Value Bioproducts. Appl. Phycol. 2020, 1, 2–11. [Google Scholar] [CrossRef]
- Sekar, M.; Mathimani, T.; Alagumalai, A.; Chi, N.T.L.; Duc, P.A.; Bhatia, S.K.; Brindhadevi, K.; Pugazhendhi, A. A Review on the Pyrolysis of Algal Biomass for Biochar and Bio-Oil—Bottlenecks and Scope. Fuel 2021, 283, 119190. [Google Scholar] [CrossRef]
- Safi, C.; Zebib, B.; Merah, O.; Pontalier, P.-Y.; Vaca-Garcia, C. Morphology, Composition, Production, Processing and Applications of Chlorella Vulgaris: A Review. Renew. Sustain. Energy Rev. 2014, 35, 265–278. [Google Scholar] [CrossRef]
- Chong, J.W.R.; Khoo, K.S.; Yew, G.Y.; Leong, W.H.; Lim, J.W.; Lam, M.K.; Ho, Y.-C.; Ng, H.S.; Munawaroh, H.S.H.; Show, P.L. Advances in Production of Bioplastics by Microalgae Using Food Waste Hydrolysate and Wastewater: A Review. Bioresour. Technol. 2021, 342, 125947. [Google Scholar] [CrossRef] [PubMed]
- Carnovale, G.; Rosa, F.; Shapaval, V.; Dzurendova, S.; Kohler, A.; Wicklund, T.; Horn, S.J.; Barbosa, M.J.; Skjånes, K. Starch Rich Chlorella Vulgaris: High-Throughput Screening and Up-Scale for Tailored Biomass Production. Appl. Sci. 2021, 11, 9025. [Google Scholar] [CrossRef]
- Li, H.; Dong, X.; da Silva, E.B.; de Oliveira, L.M.; Chen, Y.; Ma, L.Q. Mechanisms of Metal Sorption by Biochars: Biochar Characteristics and Modifications. Chemosphere 2017, 178, 466–478. [Google Scholar] [CrossRef] [PubMed]
- Guillard, R.R.L. Culture of Phytoplankton for Feeding Marine Invertebrates. In Culture of Marine Invertebrate Animals; Springer: Boston, MA, USA, 1975; pp. 29–60. [Google Scholar]
- ISO 562:2010; Hard Coal and Coke—Determination of Volatile Matter. International Organization for Standardization: Geneva, Switzerland, 2010.
- Friedl, A.; Padouvas, E.; Rotter, H.; Varmuza, K. Prediction of Heating Values of Biomass Fuel from Elemental Composition. Anal. Chim. Acta 2005, 544, 191–198. [Google Scholar] [CrossRef]
- Isaac, P.J.; Amaravadi, S.; MSM, K.; KK, C.; R, L. Synthesis of Zeolite/Activated Carbon Composite Material for the Removal of Lead (II) and Cadmium (II) Ions. Environ. Prog. Sustain. Energy 2019, 38, e13246. [Google Scholar] [CrossRef]
- N’goran, K.P.D.A.; Diabaté, D.; Yao, K.M.; Kouassi, N.L.B.; Gnonsoro, U.P.; Kinimo, K.C.; Trokourey, A. Lead and Cadmium Removal from Natural Freshwater Using Mixed Activated Carbons from Cashew and Shea Nut Shells. Arab. J. Geosci. 2018, 11, 498. [Google Scholar] [CrossRef]
- Sellaoui, L.; Dotto, G.L.; Lamine, A.B.; Erto, A. Interpretation of Single and Competitive Adsorption of Cadmium and Zinc on Activated Carbon Using Monolayer and Exclusive Extended Monolayer Models. Environ. Sci. Pollut. Res. 2017, 24, 19902–19908. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Saravanan, A.; Kumar, P.S.; Vo, D.-V.N.; Swetha, S.; Ngueagni, P.T.; Karishma, S.; Jeevanantham, S.; Yaashikaa, P.R. Ultrasonic Assisted Agro Waste Biomass for Rapid Removal of Cd(II) Ions from Aquatic Environment: Mechanism and Modelling Analysis. Chemosphere 2021, 271, 129484. [Google Scholar] [CrossRef]
- Langmuir, I. The Adsorption of Gases on Plane Surfaces of Glass, Mica and Platinum. J. Am. Chem. Soc. 1918, 40, 1361–1403. [Google Scholar] [CrossRef]
- Gransee, A.; Merbach, W. Phosphorus Dynamics in a Long-Term P Fertilization Trial on Luvic Phaeozem at Halle. J. Plant Nutr. Soil Sci. 2000, 163, 353–357. [Google Scholar] [CrossRef]
- Freundlich, H. Über Die Adsorption in Lösungen. Z. Für Phys. Chem. 1907, 57U, 385–470. [Google Scholar] [CrossRef]
- Bohn, H.L.; McNeal, B.L.; O’Connor, G.A. Soil Chemistry; Wiley: Hoboken, NJ, USA, 1985; ISBN 0471822175. [Google Scholar]
- Temkin, M.J.; Pyzhev, V. Recent Modifications to Langmuir Isotherms. Acta Physiochim URSS 1940, 12, 217–225. [Google Scholar]
- Binda, G.; Spanu, D.; Bettinetti, R.; Magagnin, L.; Pozzi, A.; Dossi, C. Comprehensive Comparison of Microalgae-Derived Biochar from Different Feedstocks: A Prospective Study for Future Environmental Applications. Algal Res. 2020, 52, 102103. [Google Scholar] [CrossRef]
- Tsarpali, M.; Kuhn, J.N.; Philippidis, G.P. Hydrothermal Carbonization of Residual Algal Biomass for Production of Hydrochar as a Biobased Metal Adsorbent. Sustainability 2022, 14, 455. [Google Scholar] [CrossRef]
- Khoo, C.G.; Lam, M.K.; Mohamed, A.R.; Lee, K.T. Hydrochar Production from High-Ash Low-Lipid Microalgal Biomass via Hydrothermal Carbonization: Effects of Operational Parameters and Products Characterization. Environ. Res. 2020, 188, 109828. [Google Scholar] [CrossRef] [PubMed]
- Spokas, K.A. Review of the Stability of Biochar in Soils: Predictability of O:C Molar Ratios. Carbon Manag. 2010, 1, 289–303. [Google Scholar] [CrossRef]
- Chen, X.; Chen, G.; Chen, L.; Chen, Y.; Lehmann, J.; McBride, M.B.; Hay, A.G. Adsorption of Copper and Zinc by Biochars Produced from Pyrolysis of Hardwood and Corn Straw in Aqueous Solution. Bioresour. Technol. 2011, 102, 8877–8884. [Google Scholar] [CrossRef]
- Peters, K.E.; Xia, X.; Pomerantz, A.E.; Mullins, O.C. Geochemistry Applied to Evaluation of Unconventional Resources. In Unconventional Oil and Gas Resources Handbook; Elsevier: Amsterdam, The Netherlands, 2016; pp. 71–126. [Google Scholar]
- Chen, S.; Yang, M.; Ba, C.; Yu, S.; Jiang, Y.; Zou, H.; Zhang, Y. Preparation and Characterization of Slow-Release Fertilizer Encapsulated by Biochar-Based Waterborne Copolymers. Sci. Total Environ. 2018, 615, 431–437. [Google Scholar] [CrossRef]
- Deng, J.; Li, X.; Wei, X.; Liu, Y.; Liang, J.; Song, B.; Shao, Y.; Huang, W. Hybrid Silicate-Hydrochar Composite for Highly Efficient Removal of Heavy Metal and Antibiotics: Coadsorption and Mechanism. Chem. Eng. J. 2020, 387, 124097. [Google Scholar] [CrossRef]
- Enders, A.; Hanley, K.; Whitman, T.; Joseph, S.; Lehmann, J. Characterization of Biochars to Evaluate Recalcitrance and Agronomic Performance. Bioresour. Technol. 2012, 114, 644–653. [Google Scholar] [CrossRef]
- Chaiwong, K.; Kiatsiriroat, T.; Vorayos, N.; Thararax, C. Study of Bio-Oil and Bio-Char Production from Algae by Slow Pyrolysis. Biomass Bioenergy 2013, 56, 600–606. [Google Scholar] [CrossRef]
- Ghani, W.A.W.A.K.; Mohd, A.; da Silva, G.; Bachmann, R.T.; Taufiq-Yap, Y.H.; Rashid, U.; Al-Muhtaseb, A.H. Biochar Production from Waste Rubber-Wood-Sawdust and Its Potential Use in C Sequestration: Chemical and Physical Characterization. Ind. Crops Prod. 2013, 44, 18–24. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, Y.; Cai, J.; Wilson, K.; Lee, A.F. Bio/Hydrochar Sorbents for Environmental Remediation. Energy Environ. Mater. 2020, 3, 453–468. [Google Scholar] [CrossRef]
- Bird, M.I.; Wurster, C.M.; de Paula Silva, P.H.; Bass, A.M.; de Nys, R. Algal Biochar—Production and Properties. Bioresour. Technol. 2011, 102, 1886–1891. [Google Scholar] [CrossRef] [PubMed]
- Bargmann, I.; Rillig, M.C.; Buss, W.; Kruse, A.; Kuecke, M. Hydrochar and Biochar Effects on Germination of Spring Barley. J. Agron. Crop Sci. 2013, 199, 360–373. [Google Scholar] [CrossRef]
- Rehrah, D.; Reddy, M.R.; Novak, J.M.; Bansode, R.R.; Schimmel, K.A.; Yu, J.; Watts, D.W.; Ahmedna, M. Production and Characterization of Biochars from Agricultural By-Products for Use in Soil Quality Enhancement. J. Anal. Appl. Pyrolysis 2014, 108, 301–309. [Google Scholar] [CrossRef]
- Sun, K.; Tang, J.; Gong, Y.; Zhang, H. Characterization of Potassium Hydroxide (KOH) Modified Hydrochars from Different Feedstocks for Enhanced Removal of Heavy Metals from Water. Environ. Sci. Pollut. Res. 2015, 22, 16640–16651. [Google Scholar] [CrossRef] [PubMed]
- Michalak, I.; Mironiuk, M.; Marycz, K. A Comprehensive Analysis of Biosorption of Metal Ions by Macroalgae Using ICP-OES, SEM-EDX and FTIR Techniques. PLoS ONE 2018, 13, e0205590. [Google Scholar] [CrossRef]
- Kołtowski, M.; Charmas, B.; Skubiszewska-Zięba, J.; Oleszczuk, P. Effect of Biochar Activation by Different Methods on Toxicity of Soil Contaminated by Industrial Activity. Ecotoxicol. Environ. Saf. 2017, 136, 119–125. [Google Scholar] [CrossRef]
- Wang, K.; Brown, R.C.; Homsy, S.; Martinez, L.; Sidhu, S.S. Fast Pyrolysis of Microalgae Remnants in a Fluidized Bed Reactor for Bio-Oil and Biochar Production. Bioresour. Technol. 2013, 127, 494–499. [Google Scholar] [CrossRef]
- Chang, Y.-M.; Tsai, W.-T.; Li, M.-H. Chemical Characterization of Char Derived from Slow Pyrolysis of Microalgal Residue. J. Anal. Appl. Pyrolysis 2015, 111, 88–93. [Google Scholar] [CrossRef]
- Argun, M.E.; Dursun, S.; Ozdemir, C.; Karatas, M. Heavy Metal Adsorption by Modified Oak Sawdust: Thermodynamics and Kinetics. J. Hazard. Mater. 2007, 141, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Chen, H.; Huang, W.; Arocena, J.M.; Ge, S. Sorption of Atrazine, 17α-Estradiol, and Phenanthrene on Wheat Straw and Peanut Shell Biochars. Water Air Soil Pollut. 2016, 227, 7. [Google Scholar] [CrossRef]
- Deng, J.; Liu, Y.; Liu, S.; Zeng, G.; Tan, X.; Huang, B.; Tang, X.; Wang, S.; Hua, Q.; Yan, Z. Competitive Adsorption of Pb(II), Cd(II) and Cu(II) onto Chitosan-Pyromellitic Dianhydride Modified Biochar. J. Colloid Interface Sci. 2017, 506, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Olgun, A.; Atar, N. Equilibrium, Thermodynamic and Kinetic Studies for the Adsorption of Lead (II) and Nickel (II) onto Clay Mixture Containing Boron Impurity. J. Ind. Eng. Chem. 2012, 18, 1751–1757. [Google Scholar] [CrossRef]
- Li, H.; Shi, Y.; Bai, L.; Chi, M.; Xu, X.; Liu, Y. Low Temperature One-Pot Hydrothermal Carbonization of Corn Straw into Hydrochar for Adsorbing Cadmium (II) in Wastewater. Energies 2021, 14, 8503. [Google Scholar] [CrossRef]
- Ma, F.; Zhao, B.; Diao, J. Adsorption of Cadmium by Biochar Produced from Pyrolysis of Corn Stalk in Aqueous Solution. Water Sci. Technol. 2016, 74, 1335–1345. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Zhang, T.C.; Ouyang, L.; Yuan, S. Single-Step Hydrothermal Synthesis of Biochar from H3PO4-Activated Lettuce Waste for Efficient Adsorption of Cd(II) in Aqueous Solution. Molecules 2022, 27, 269. [Google Scholar] [CrossRef]
- Kumar, M.; Singh, A.K.; Sikandar, M. Study of Sorption and Desorption of Cd (II) from Aqueous Solution Using Isolated Green Algae Chlorella Vulgaris. Appl. Water Sci. 2018, 8, 225. [Google Scholar] [CrossRef]
- Edris, G.; Alhamed, Y.; Alzahrani, A. Biosorption of Cadmium and Lead from Aqueous Solutions by Chlorella Vulgaris Biomass: Equilibrium and Kinetic Study. Arab. J. Sci. Eng. 2014, 39, 87–93. [Google Scholar] [CrossRef]
- Biswas, K.; Saha, S.K.; Ghosh, U.C. Adsorption of Fluoride from Aqueous Solution by a Synthetic Iron(III)−Aluminum(III) Mixed Oxide. Ind. Eng. Chem. Res. 2007, 46, 5346–5356. [Google Scholar] [CrossRef]
- Chen, T.; Zhou, Z.; Han, R.; Meng, R.; Wang, H.; Lu, W. Adsorption of Cadmium by Biochar Derived from Municipal Sewage Sludge: Impact Factors and Adsorption Mechanism. Chemosphere 2015, 134, 286–293. [Google Scholar] [CrossRef] [PubMed]
- Hulland, J. Use of Partial Least Squares (PLS) in Strategic Management Research: A Review of Four Recent Studies. Strateg. Manag. J. 1999, 20, 195–204. [Google Scholar] [CrossRef]
- Kline, R.B. Principles and Practice of Structural Equation Modeling, 5th ed.; Guildford Press: New York, NY, USA, 2023; ISBN 9781462552009. [Google Scholar]



| Nutrient | Concentration (g L−1) |
|---|---|
| Solution A: Nitrate and phosphate stock solution | |
| NaNO3 | 84.2 |
| Na2MoO4·2H2O | 6.0 |
| FeCl3·6H2O | 2.9 |
| Na2EDTA·2H2O | 10.0 |
| Solution B: Silicate stock solution | |
| Na2SiO3·9H2O | 33.0 |
| Solution C: Trace element stock solution | |
| CuSO4·5H2O | 2.0 |
| Na2MoO4·2H2O | 4.4 |
| CoCl2·6H2O | 2.0 |
| MnCl2·4H2O | 36.0 |
| Solution D: Vitamins stock solution | |
| Biotin | 1.0 × 10−4 |
| Vitamin B1 | 0.4 |
| Vitamins B12 | 2.0 × 10−6 |
| Property | Material | ||
|---|---|---|---|
| Biomass | Biochar | Hydrochar | |
| Yield (%) | - | 56.2 ± 0.2 | 61.5 ± 0.3 |
| C (%) | 37.20 | 45.92 | 48.55 |
| H (%) | 5.66 | 3.88 | 5.04 |
| N (%) | 5.11 | 4.41 | 4.12 |
| S (%) | 0.84 | 0.57 | 0.51 |
| O (%) | 22.29 | 13.11 | 10.98 |
| Ash (%) | 28.9 ± 0.1 | 32.1 ± 0.2 | 30.8 ± 0.2 |
| Volatile matter (%) | 54.3 ± 0.2 | 43.2 ± 0.4 | 48.8 ± 0.2 |
| GCV (MJ kg−1) | 15.7 ± 0.3 | 18.5 ± 0.1 | 19.5 ± 0.3 |
| pH | 6.9 ± 0.1 | 7.9 ± 0.1 | 6.7 ± 0.1 |
| EC (dS m−1) | 2.6 ± 0.1 | 4.5 ± 0.2 | 3.4 ± 0.1 |
| SSA (m2 g−1) | 6.8 ± 0.3 | 8.5 ± 0.1 | 8.1 ± 0.3 |
| Pore diameter (nm) | 15.0 ± 0.2 | 22.3 ± 0.3 | 12.8 ± 0.3 |
| Class | Functional Group | Wavelength Range (cm−1) | |||
|---|---|---|---|---|---|
| Reference | Biomass | Biochar | Hydrochar | ||
| Alcohol | O-–H stretch | 3400–3500 | 3414.36 | 3414.5 | 3415.99 |
| Carboxylic acids | O–H stretch (s) | 3300–2500 | 2515.57 | 2515.75 | 2515.83 |
| Alkanes | C–H stretch (s) | 3000–2850 | 2922.27 | 2853.21 | 2874.05 |
| Aldehydes and ketones | C=O stretch (s) | 1730–1720 1640–1600 | 1638.68 | 1616.95 | 1617.34 |
| Amides | N–H out of plane | 1470–1350 | 1420.42 | 1420.08 | 1416.26 |
| Alkyl aryl ether | C–O stretch (s) | 1075–1020 | 1058.40 | - | - |
| Aromatics | C–H out of plane (m) | 885–870 | 872.91 | 873.50 | 873.09 |
| Alkenes | C=C plane (s) | 730–665 | 712.49 | 712.57 | 712.53 |
| Halo compound | C–Cl stretch, C–Br stretch, and C–I stretch | 400–600 | 471.08 | 467.21 | 470.56 |
| Materials | Current Study | [63] | [64] | [65] | [66] | [67] |
| Biochar | 24.4 | - | 33.9 | 87.4 | - | - |
| Hydrochar | 23.6 | 11.7–18.1 | - | - | - | - |
| Biomass | 16.6 | - | - | - | 97.4 | 149.9 |
| Materials | Langmuir | Freundlich | Temkin | ||||||
|---|---|---|---|---|---|---|---|---|---|
| qmax | b | R2 | Kf | 1/n | R2 | AT (L g−1) | bT | R2 | |
| Biochar | 24.39 | 0.1393 | 0.99 | 6.77 | 0.563 | 0.96 | 4.69 | 560.66 | 0.968 |
| Hydrochar | 23.58 | 0.142 | 0.988 | 11.13 | 0.515 | 0.913 | 4.94 | 591.60 | 0.981 |
| Biomass | 16.56 | 0.3709 | 0.993 | 2.61 | 0.534 | 0.976 | 2.25 | 783.44 | 0.979 |
| Source | Parameter | Weight Factor | Source | Parameter | Weight Factor |
|---|---|---|---|---|---|
| Dosage | 0.79 | 0.1 | pH | 0.86 | 3 |
| 0.83 | 0.2 | 0.76 | 5 | ||
| 0.80 | 0.4 | 0.85 | 6 | ||
| 0.75 | 0.8 | 0.85 | 7 | ||
| 0.91 | 1.0 | 0.87 | 8 | ||
| 0.87 | 2.0 | 0.83 | 9 | ||
| Concentration | 0.83 | 0 | Time | 0.86 | 0 |
| 0.77 | 5 | 0.82 | 5 | ||
| 0.86 | 10 | 0.81 | 15 | ||
| 0.89 | 20 | 0.67 | 30 | ||
| 0.90 | 40 | 0.75 | 60 | ||
| Materials | 0.73 | Biomass | 0.93 | 90 | |
| 0.88 | Biochar | 0.92 | 180 | ||
| 0.81 | Hydrochar |
| R2 | Source |
|---|---|
| 0.17 | Time—Materials |
| 0.45 | Time—pH |
| 0.36 | Time—Concentration |
| 0.54 | Time—Dosage |
| 0.65 | Removal of Cd |
| Source | Q2 (=1 − SSE/SSO) |
|---|---|
| Materials | 0.839 |
| pH | 0.521 |
| Concentration | 0.458 |
| Dosage | 0.744 |
| Time | 0.388 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sufian, J.; Babaakbari Sari, M.; Marchelli, F.; Fiori, L.; Avanes, A.; Moradi, S. An Analysis of the Factors Influencing Cadmium Removal in Aquatic Environments by Chlorella vulgaris-Derived Solids. C 2024, 10, 2. https://doi.org/10.3390/c10010002
Sufian J, Babaakbari Sari M, Marchelli F, Fiori L, Avanes A, Moradi S. An Analysis of the Factors Influencing Cadmium Removal in Aquatic Environments by Chlorella vulgaris-Derived Solids. C. 2024; 10(1):2. https://doi.org/10.3390/c10010002
Chicago/Turabian StyleSufian, Jafar, Mohammad Babaakbari Sari, Filippo Marchelli, Luca Fiori, Armen Avanes, and Salahedin Moradi. 2024. "An Analysis of the Factors Influencing Cadmium Removal in Aquatic Environments by Chlorella vulgaris-Derived Solids" C 10, no. 1: 2. https://doi.org/10.3390/c10010002
APA StyleSufian, J., Babaakbari Sari, M., Marchelli, F., Fiori, L., Avanes, A., & Moradi, S. (2024). An Analysis of the Factors Influencing Cadmium Removal in Aquatic Environments by Chlorella vulgaris-Derived Solids. C, 10(1), 2. https://doi.org/10.3390/c10010002








