Androgen and Oestrogen Affect the Expression of Long Non-Coding RNAs During Phallus Development in a Marsupial
Abstract
:1. Introduction
2. Results
2.1. Principal Component Analysis
2.2. Predicted Hormone-Responsive long non-coding RNAs
3. Discussion
3.1. A Global View of Differentially Expressed Genes and Phallus Phenotype
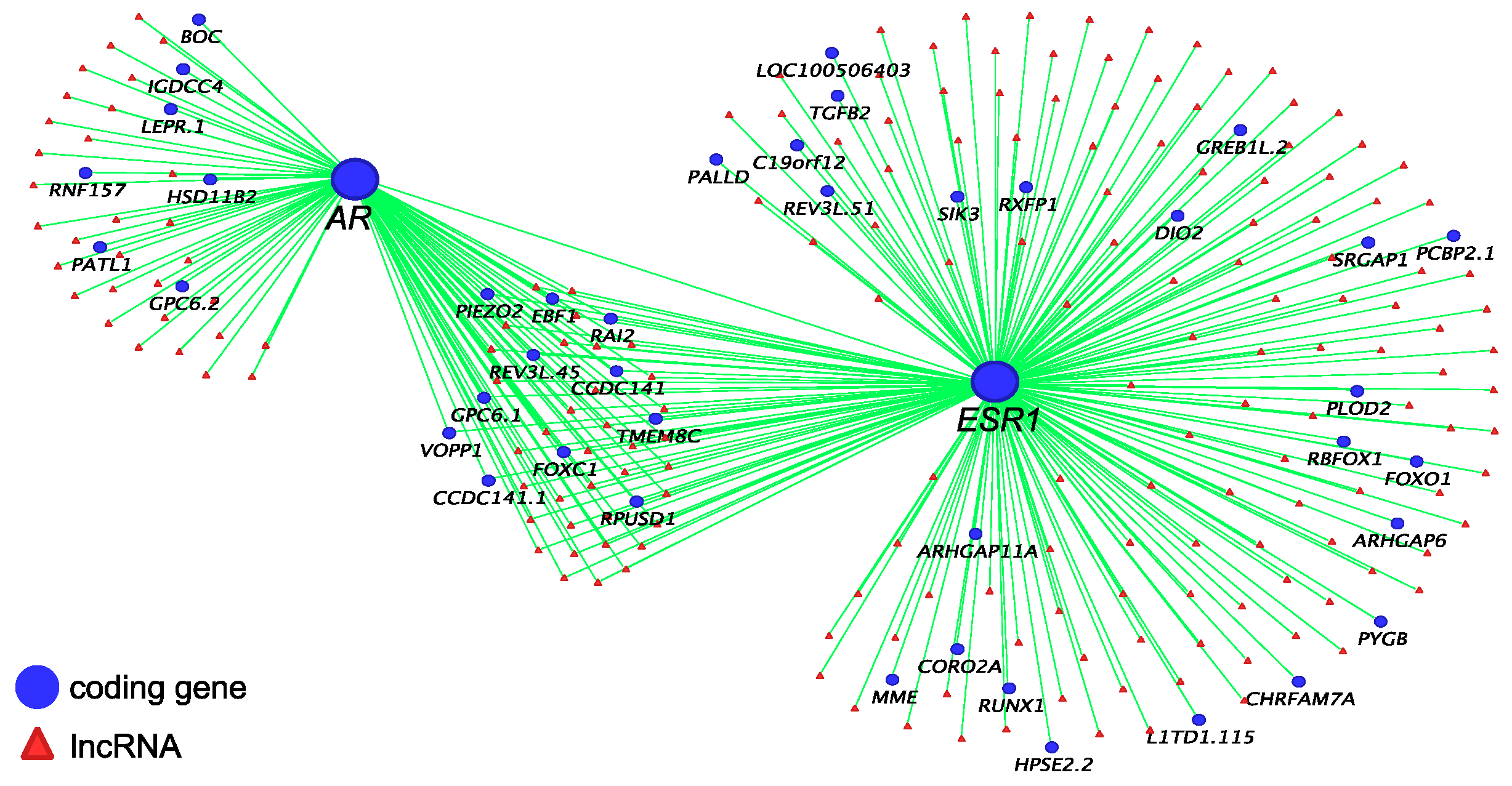
3.2. Co-Regulatory lncRNAs of IGF1, AR and ESR1
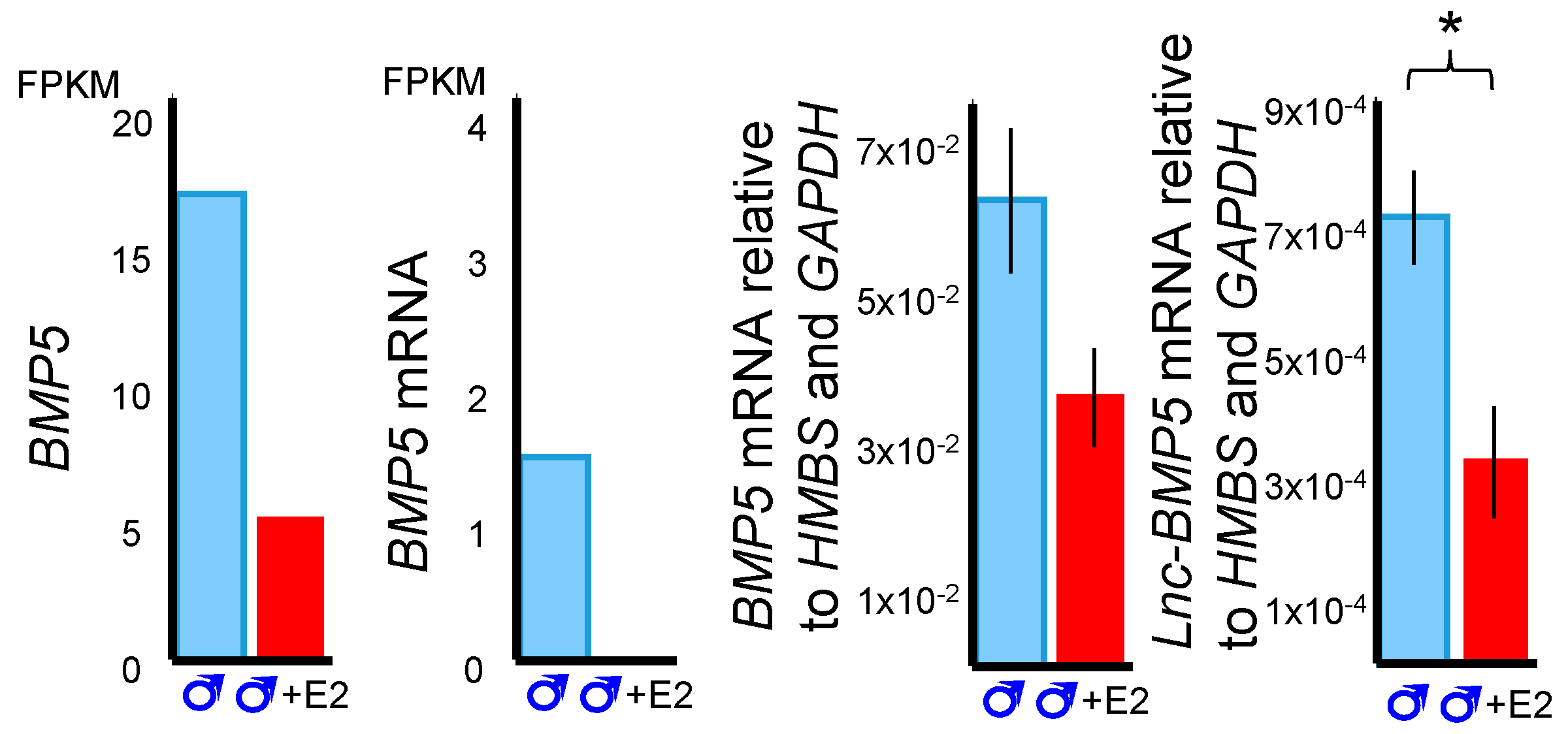
3.3. BMP5 and Lnc-BMP5 in Phallus Development
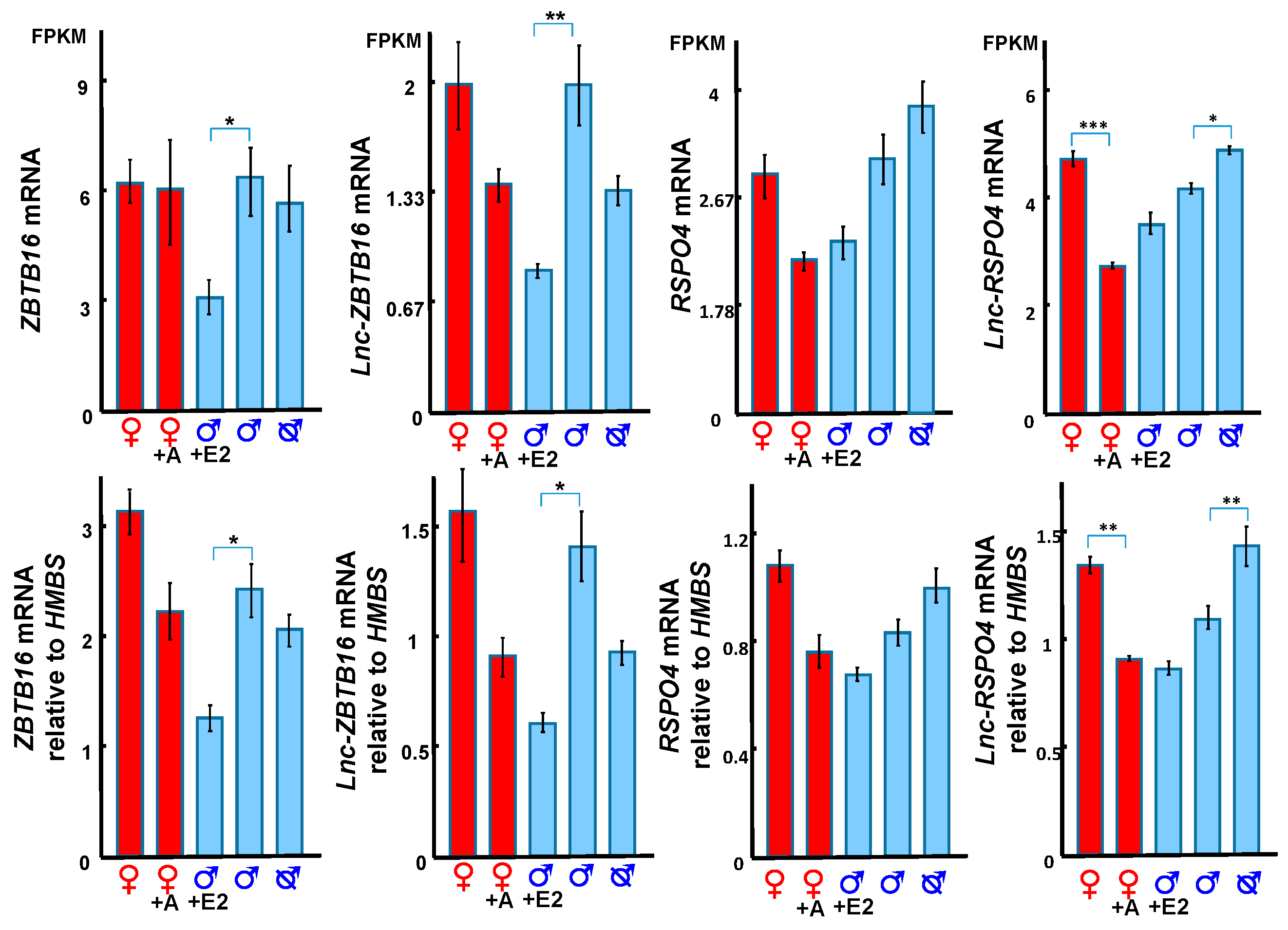
3.4. Hormonal Regulation of ZBTB16 and its Correlated lncRNA
3.5. RSPO4 and lnc-RSPO4 May Regulate Phallus Development Through Activating WNT5A
4. Materials & Methods
4.1. Animals
4.2. Transcriptome Analysis
4.3. Weighted Gene Co-Expression Network Analysis
4.4. RNA Extraction and cDNA Preparation
4.5. Quantitative PCR
4.6. Section in situ Hybridization
4.7. Statistics
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of interest
References
- Apweiler, R.; Bairoch, A.; Wu, C.H.; Barker, W.C.; Boeckmann, B.; Ferro, S.; Gasteiger, E.; Huang, H.; Lopez, R.; Magrane, M.; et al. UniProt: The universal protein knowledgebase. Nucleic Acids Res. 2004, 32, 115–119. [Google Scholar] [CrossRef] [PubMed]
- Birney, E.; Stamatoyannopoulos, J.A.; Dutta, A.; Guigó, R.; Gingeras, T.R.; Margulies, E.H.; Weng, Z.; Snyder, M.; Dermitzakis, E.T.; Thurman, R.E.; et al. Identification and analysis of functional elements in 1% of the human genome by the encode pilot project. Nature 2007, 447, 799–816. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.; Kapranov, P.; Drenkow, J.; Dike, S.; Brubaker, S.; Patel, S.; Long, J.; Stern, D.; Tammana, H.; Helt, G. Transcriptional maps of 10 human chromosomes at 5-nucleotide resolution. Science 2005, 308, 1149–1154. [Google Scholar] [CrossRef] [PubMed]
- Iyer, M.K.; Niknafs, Y.S.; Malik, R.; Singhal, U.; Sahu, A.; Hosono, Y.; Barrette, T.R.; Prensner, J.R.; Evans, J.R.; Zhao, S.; et al. The landscape of long noncoding RNAs in the human transcriptome. Nat. Genet. 2015, 47, 199–208. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eddy, S.R. Non–coding RNA genes and the modern RNA world. Nat. Rev. Genet. 2001, 2, 919–929. [Google Scholar] [CrossRef] [PubMed]
- Managadze, D.; Lobkovsky, A.E.; Wolf, Y.I.; Shabalina, S.A.; Rogozin, I.B.; Koonin, E.V. The vast, conserved mammalian lincRNome. PLoS Comput. Boil. 2013, 9, e1002917. [Google Scholar] [CrossRef] [PubMed]
- Rinn, J.L.; Chang, H.Y. Genome regulation by long noncoding RNAs. Annu. Rev. Biochem. 2012, 81, 145–166. [Google Scholar] [CrossRef]
- Engreitz, J.M.; Ollikainen, N.; Guttman, M. Long non-coding RNAs: Spatial amplifiers that control nuclear structure and gene expression. Nat. Rev. Mol. Cell Boil. 2016, 17, 756–770. [Google Scholar] [CrossRef]
- Rastetter, R.H.; Smith, C.A.; Wilhelm, D. The role of non-coding RNA in male sex determination and differentiation. Reproduction 2015, 150, R93–R107. [Google Scholar] [CrossRef]
- Wilhelm, D.; Bernard, P. Non-Coding RNA and the Reproductive System; Springer: New York, NY, USA, 2016. [Google Scholar]
- Carninci, P.; Kasukawa, T.; Katayama, S.; Gough, J.; Frith, M.; Maeda, N.; Oyama, R.; Ravasi, T.; Lenhard, B.; Wells, C.; et al. The transcriptional landscape of the mammalian genome. Science 2005, 309, 1559–1563. [Google Scholar] [CrossRef]
- Kapranov, P.; Cheng, J.; Dike, S.; Nix, D.A.; Duttagupta, R.; Willingham, A.T.; Stadler, P.F.; Hertel, J.; Hackermüller, J.; Hofacker, I.L.; et al. RNA maps reveal new RNA classes and a possible function for pervasive transcription. Science 2007, 316, 1484–1488. [Google Scholar] [CrossRef] [PubMed]
- Carninci, P. Constructing the landscape of the mammalian transcriptome. J. Exp. Boil. 2007, 210, 1497–1506. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Okazaki, Y.; Furuno, M.; Kasukawa, T.; Adachi, J.; Bono, H.; Kondo, S.; Nikaido, I.; Osato, N.; Saito, R.; Suzuki, H.; et al. Analysis of the mouse transcriptome based on functional annotation of 60,770 full-length cDNAs. Nature 2002, 420, 563–573. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taylor, D.H.; Chu, E.T.J.; Spektor, R.; Soloway, P.D. Long non-coding RNA regulation of reproduction and development. Mol. Reprod. Dev. 2015, 82, 932–956. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kherdjemil, Y.; Lalonde, R.L.; Sheth, R.; Dumouchel, A.; de Martino, G.; Pineault, K.M.; Wellik, D.M.; Stadler, H.S.; Akimenko, M.A.; Kmita, M. Evolution of Hoxa11 regulation in vertebrates is linked to the pentadactyl state. Nature 2016, 539, 89–92. [Google Scholar] [CrossRef] [PubMed]
- Mori, A.; Lehmann, S.; O’Kelly, J.; Kumagai, T.; Desmond, J.C.; Pervan, M.; McBride, W.H.; Kizaki, M.; Koeffler, H.P. Capsaicin, a component of red peppers, inhibits the growth of androgen-independent, p53 mutant prostate cancer cells. Cancer Res. 2006, 66, 3222–3229. [Google Scholar] [CrossRef] [PubMed]
- Chakravarty, D.; Sboner, A.; Nair, S.S.; Giannopoulou, E.; Li, R.; Hennig, S.; Mosquera, J.M.; Pauwels, J.; Park, K.; Kossai, M.; et al. The oestrogen receptor alpha-regulated lncRNA NEAT1 is a critical modulator of prostate cancer. Nat. Commun. 2014, 5, 5383. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kornienko, A.E.; Guenzl, P.M.; Barlow, D.P.; Pauler, F.M. Gene regulation by the act of long non-coding RNA transcription. BMC Boil. 2013, 11, 59. [Google Scholar] [CrossRef] [PubMed]
- Renfree, M.B.; Short, R.; Shaw, G. Sexual differentiation of the urogenital system of the fetal and neonatal tammar wallaby, Macropus eugenii. Anat. Embryol. 1996, 194, 111–134. [Google Scholar] [CrossRef]
- Leihy, M.; Shaw, G.; Wilson, J.; Renfree, M. Development of the penile urethra in the tammar wallaby. Sex. Dev. 2011, 5, 241–249. [Google Scholar] [CrossRef]
- Butler, C.; Shaw, G.; Renfree, M. Development of the penis and clitoris in the tammar wallaby, Macropus Eugenii. Anat. Embryol. 1999, 199, 451–457. [Google Scholar] [CrossRef] [PubMed]
- Leihy, M.W.; Shaw, G.; Wilson, J.D.; Renfree, M.B. Penile development is initiated in the tammar wallaby pouch young during the period when 5α-androstane-3α, 17β-diol is secreted by the testes. Endocrinology 2004, 145, 3346–3352. [Google Scholar] [CrossRef] [PubMed]
- Renfree, M.B.; Chew, K.Y.; Shaw, G. Inducing sex reversal of the urogenital system of marsupials. Differentiation 2014, 87, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Chew, K.; Pask, A.; Hickford, D.; Shaw, G.; Renfree, M. A dual role for SHH during phallus development in a marsupial. Sex. Dev. 2014, 8, 166–177. [Google Scholar] [CrossRef]
- Wilson, J.D.; Auchus, R.J.; Leihy, M.W.; Guryev, O.L.; Estabrook, R.W.; Osborn, S.M.; Shaw, G.; Renfree, M.B. 5α-androstane-3α, 17β-diol is formed in tammar wallaby pouch young testes by a pathway involving 5α-pregnane-3α, 17α-diol-20-one as a key intermediate. Endocrinology 2003, 144, 575–580. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.D.; George, F.W.; Shaw, G.; Renfree, M.B. Virilization of the male pouch young of the tammar wallaby does not appear to be mediated by plasma testosterone or dihydrotestosterone. Boil. Reprod. 1999, 61, 471–475. [Google Scholar] [CrossRef]
- Shaw, G.; Renfree, M.B.; Leihy, M.W.; Shackleton, C.H.; Roitman, E.; Wilson, J.D. Prostate formation in a marsupial is mediated by the testicular androgen 5α-androstane-3α, 17β-diol. Proc. Natl. Acad. Sci. USA 2000, 97, 12256–12259. [Google Scholar] [CrossRef]
- Wilson, J.D.; Shaw, G.; Leihy, M.L.; Renfree, M.B. The marsupial model for male phenotypic development. Trends Endocrinol. Metab. 2002, 13, 78–83. [Google Scholar] [CrossRef]
- Chen, Y.; Yu, H.; Pask, A.J.; Fujiyama, A.; Suzuki, Y.; Sugano, S.; Shaw, G.; Renfree, M.B. Hormone-responsive genes in the SHH and WNT/β-catenin signaling pathways influence urethral closure and phallus growth. Boil. Reprod. 2018, 99, 806–816. [Google Scholar] [CrossRef]
- Chen, Y.; Yu, H.; Pask, A.J.; Fujiyama, A.; Suzuki, Y.; Sugano, S.; Shaw, G.; Renfree, M. Effects of androgen and oestrogen on the IGF pathways controlling phallus growth. Reproduction 2019, 157, 1–12. [Google Scholar] [CrossRef]
- Zheng, Z.; Armfield, B.A.; Cohn, M.J. Timing of androgen receptor disruption and estrogen exposure underlies a spectrum of congenital penile anomalies. Proc. Natl. Acad. Sci. USA 2015, 112, E7194–E7203. [Google Scholar] [CrossRef] [PubMed]
- Laron, Z.; Pertzelan, A.; Mannheimer, S. Genetic pituitary dwarfism with high serum concentation of growth hormone—A new inborn error of metabolism? Isr. J. Med. Sci. 1966, 2, 152–155. [Google Scholar] [PubMed]
- Laron, Z.; Klinger, B. Effect of insulin-like growth factor-1treatment on serum androgens and testicular and penile size in males with laron syndrome (primary growth hormone resistance). Eur. J. Endocrinol. 1998, 138, 176–180. [Google Scholar] [CrossRef] [PubMed]
- Duan, C.; Xu, Q. Roles of insulin-like growth factor (IGF) binding proteins in regulating IGF actions. Gen. Comp. Endocrinol. 2005, 142, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Clemmons, D.R. Role of insulin-like growth factor binding proteins in controlling IGF actions. Mol. Cell. Endocrinol. 1998, 140, 19–24. [Google Scholar] [CrossRef]
- Satoh, Y.; Haraguchi, R.; Wright, T.J.; Mansour, S.L.; Partanen, J.; Hajihosseini, M.K.; Eswarakumar, V.P.; Lonai, P.; Yamada, G. Regulation of external genitalia development by concerted actions of FGF ligands and FGF receptors. Anat. Embryol. 2004, 208, 479–486. [Google Scholar] [CrossRef]
- Harada, M.; Omori, A.; Nakahara, C.; Nakagata, N.; Akita, K.; Yamada, G. Tissue-specific roles of FGF signaling in external genitalia development. Dev. Dyn. 2015, 244, 759–773. [Google Scholar] [CrossRef] [Green Version]
- Gredler, M.L.; Seifert, A.W.; Cohn, M.J. Tissue-specific roles of Fgfr2 in development of the external genitalia. Development 2015, 142, 2203–2212. [Google Scholar] [CrossRef]
- Petiot, A.; Perriton, C.L.; Dickson, C.; Cohn, M.J. Development of the mammalian urethra is controlled by Fgfr2-IIIb. Development 2005, 132, 2441–2450. [Google Scholar] [CrossRef] [Green Version]
- Wu, X.; Ferrara, C.; Shapiro, E.; Grishina, I. Bmp7 expression and null phenotype in the urogenital system suggest a role in re-organization of the urethral epithelium. Gene Expr. Patterns 2009, 9, 224–230. [Google Scholar] [CrossRef]
- Beleza-Meireles, A.; Lundberg, F.; Lagerstedt, K.; Zhou, X.; Omrani, D.; Frisén, L.; Nordenskjöld, A. FGFR2, FGF8, FGF10 and BMP7 as candidate genes for hypospadias. Eur. J. Hum. Genet. 2007, 15, 405–410. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morgan, E.A.; Nguyen, S.B.; Scott, V.; Stadler, H.S. Loss of Bmp7 and Fgf8 signaling in Hoxa13-mutant mice causes hypospadias. Development 2003, 130, 3095–3109. [Google Scholar] [CrossRef] [PubMed]
- Bouty, A.; Ayers, K.L.; Pask, A.; Heloury, Y.; Sinclair, A.H. The genetic and environmental factors underlying hypospadias. Sex. Dev. 2015, 9, 239–259. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, K.; Bachiller, D.; Chen, Y.P.; Kamikawa, M.; Ogi, H.; Haraguchi, R.; Ogino, Y.; Minami, Y.; Mishina, Y.; Ahn, K.; et al. Regulation of outgrowth and apoptosis for the terminal appendage: External genitalia development by concerted actions of BMP signaling. Development 2003, 130, 6209–6220. [Google Scholar] [CrossRef] [PubMed]
- Wieczorek, D.; Köster, B.; Gillessen-Kaesbach, G. Absence of thumbs, A/hypoplasia of radius, hypoplasia of ulnae, retarded bone age, short stature, microcephaly, hypoplastic genitalia, and mental retardation. Am. J. Med. Genet. Part A 2002, 108, 209–213. [Google Scholar] [CrossRef]
- Fischer, S.; Kohlhase, J.; Böhm, D.; Schweiger, B.; Hoffmann, D.; Heitmann, M.; Horsthemke, B.; Wieczorek, D. Biallelic loss of function of the promyelocytic leukaemia zinc finger (PLZF) gene causes severe skeletal defects and genital hypoplasia. J. Med. Genet. 2008, 45, 731–737. [Google Scholar] [CrossRef] [PubMed]
- Carmon, K.S.; Gong, X.; Lin, Q.; Thomas, A.; Liu, Q. R-spondins function as ligands of the orphan receptors LGR4 and LGR5 to regulate Wnt/β-catenin signaling. Proc. Natl. Acad. Sci. USA 2011, 108, 11452–11457. [Google Scholar] [CrossRef]
- Carmon, K.S.; Gong, X.; Yi, J.; Thomas, A.; Liu, Q. RSPO–LGR4 functions via IQGAP1 to potentiate Wnt signaling. Proc. Natl. Acad. Sci. USA 2014, 111, 1221–1229. [Google Scholar] [CrossRef]
- Chen, P.-H.; Chen, X.; Lin, Z.; Fang, D.; He, X. The structural basis of R-spondin recognition by LGR5 and RNF43. Genes Dev. 2013, 27, 1345–1350. [Google Scholar] [CrossRef]
- Kim, K.-A.; Zhao, J.; Andarmani, S.; Kakitani, M.; Oshima, T.; Binnerts, M.E.; Abo, A.; Tomizuka, K.; Funk, W.D. R-Spondin proteins: A novel link to β-catenin activation. Cell Cycle 2006, 5, 23–26. [Google Scholar] [CrossRef]
- Miyagawa, S.; Moon, A.; Haraguchi, R.; Inoue, C.; Harada, M.; Nakahara, C.; Suzuki, K.; Matsumaru, D.; Kaneko, T.; Matsuo, I.; et al. Dosage-dependent hedgehog signals integrated with Wnt/β-catenin signaling regulate external genitalia formation as an appendicular program. Development 2009, 136, 3969–3978. [Google Scholar] [CrossRef] [PubMed]
- Miyagawa, S.; Satoh, Y.; Haraguchi, R.; Suzuki, K.; Iguchi, T.; Taketo, M.M.; Nakagata, N.; Matsumoto, T.; Takeyama, K.-I.; Kato, S.; et al. Genetic interactions of the androgen and Wnt/β-catenin pathways for the masculinization of external genitalia. Mol. Endocrinol. 2009, 23, 871–880. [Google Scholar] [CrossRef] [PubMed]
- Ng, R.C.; Matsumaru, D.; Ho, A.S.; Garcia-Barceló, M.; Yuan, Z.; Smith, D.; Kodjabachian, L.; Tam, P.K.; Yamada, G.; Lui, V.C. Dysregulation of wnt inhibitory factor 1 (Wif1) expression resulted in aberrant Wnt-β-catenin signaling and cell death of the cloaca endoderm, and anorectal malformations. Cell Death Differ. 2014, 21, 978–989. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.; Yin, Y.; Long, F.; Ma, L. Tissue-specific requirements of β-catenin in external genitalia development. Development 2008, 135, 2815–2825. [Google Scholar] [CrossRef] [PubMed]
- Seifert, A.W.; Zheng, Z.; Ormerod, B.K.; Cohn, M.J. Sonic hedgehog controls growth of external genitalia by regulating cell cycle kinetics. Nat. Commun. 2010, 1, 23. [Google Scholar] [CrossRef]
- Perriton, C.L.; Powles, N.; Chiang, C.; Maconochie, M.K.; Cohn, M.J. Sonic hedgehog signaling from the urethral epithelium controls external genital development. Dev. Boil. 2002, 247, 26–46. [Google Scholar] [CrossRef] [PubMed]
- Wai-Sum, O.; Short, R.V.; Renfree, M.B.; Shaw, G. Primary genetic control of somatic sexual differentiation in a mammal. Nature 1988, 331, 716–717. [Google Scholar] [CrossRef]
- Poole, W.; Simms, N.; Wood, J.; Lubulwa, M. Tables for Age Determination of the Kangaroo Island Wallaby (Tammar), Macropus Eugenii, from Body Measurements; Wildlife and Ecology Technical Memorandum #32; CSIRO: Lyneham, ACT, Australia, 1991; ISBN 0-643-05235-6. [Google Scholar]
- Renfree, M.B.; Fenelon, J.; Wijiyanti, G.; Wilson, J.D.; Shaw, G. Wolffian duct differentiation by physiological concentrations of androgen delivered systemically. Dev. Boil. 2009, 334, 429–436. [Google Scholar] [CrossRef] [PubMed]
- Coveney, D.; Shaw, G.; Renfree, M.B. Estrogen-induced gonadal sex reversal in the tammar wallaby. Boil. Reprod. 2001, 65, 613–621. [Google Scholar] [CrossRef]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef]
- Trapnell, C.; Williams, B.A.; Pertea, G.; Mortazavi, A.; Kwan, G.; Van Baren, M.J.; Salzberg, S.L.; Wold, B.J.; Pachter, L. Transcript assembly and quantification by RNA-seq reveals unannotated transcripts and isoform switching during cell differentiation. Nat. Biotechnol. 2010, 28, 511–515. [Google Scholar] [CrossRef] [PubMed]
- Goff, L.; Trapnell, C.; Kelley, D. cummeRbund: Analysis, exploration, manipulation, and visualization of Cufflinks high-throughput sequencing data. R Packag. Version 2018, 2. [Google Scholar] [CrossRef]
- O’Neill, R.; University of Connecticut, Storrs-Mansfield, Connecticut, USA; Heider, T.N.; University of Connecticut, Storrs-Mansfield, Connecticut, USA; Fujiyama, A.; Advanced Genomics Center, National Institute of Genetics, Mishima, Shizuoka, Japan; Shaw, G.; University of Melbourne, Melbourne, Victoria, Australia; Renfree, M.B.; University of Melbourne, Melbourne, Victoria, Australia; Pask, A.J.; University of Melbourne, Melbourne, Victoria, Australia. Personal communication, 2018.
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2014. [Google Scholar] [CrossRef]
- Adler, D.; Murdoch, D.; Nenadic, O.; Urbanek, S. rgl: 3D Visualization Device System (OpenGL). R Package Version 0.75. 2007. Available online: https://CRAN.R-project.org/package=rgl (accessed on 31 May 2018).
- Kong, L.; Zhang, Y.; Ye, Z.-Q.; Liu, X.-Q.; Zhao, S.-Q.; Wei, L.; Gao, G. CPC: Assess the protein-coding potential of transcripts using sequence features and support vector machine. Nucleic Acids Res. 2007, 35, 345–349. [Google Scholar] [CrossRef] [PubMed]
- Langfelder, P.; Horvath, S. Wgcna: An R package for weighted correlation network analysis. BMC Bioinform. 2008, 9, 559. [Google Scholar] [CrossRef] [PubMed]
- Parikshak, N.N.; Luo, R.; Zhang, A.; Won, H.; Lowe, J.K.; Chandran, V.; Horvath, S.; Geschwind, D.H. Integrative functional genomic analyses implicate specific molecular pathways and circuits in autism. Cell 2013, 155, 1008–1021. [Google Scholar] [CrossRef]
- Hu, Z.; Chang, Y.-C.; Wang, Y.; Huang, C.-L.; Liu, Y.; Tian, F.; Granger, B.; DeLisi, C. Visant 4.0: Integrative network platform to connect genes, drugs, diseases and therapies. Nucleic Acids Res. 2013, 41, 225–231. [Google Scholar] [CrossRef]
- Bustin, S.A.; Benes, V.; Garson, J.A.; Hellemans, J.; Huggett, J.; Kubista, M.; Mueller, R.; Nolan, T.; Pfaffl, M.W.; Shipley, G.L.; Vandesompele, J. The MIQE guidelines: Minimum information for publication of quantitative real-time PCR experiments. Clin. Chem. 2009, 55, 611–622. [Google Scholar] [CrossRef]
- Hellemans, J.; Mortier, G.; De Paepe, A.; Speleman, F.; Vandesompele, J. qBase relative quantification framework and software for management and automated analysis of real-time quantitative PCR data. Genome Biol. 2007, 8, R19. [Google Scholar] [CrossRef]



© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, Y.; Kuroki, Y.; Shaw, G.; Pask, A.J.; Yu, H.; Toyoda, A.; Fujiyama, A.; Renfree, M.B. Androgen and Oestrogen Affect the Expression of Long Non-Coding RNAs During Phallus Development in a Marsupial. Non-Coding RNA 2019, 5, 3. https://doi.org/10.3390/ncrna5010003
Chen Y, Kuroki Y, Shaw G, Pask AJ, Yu H, Toyoda A, Fujiyama A, Renfree MB. Androgen and Oestrogen Affect the Expression of Long Non-Coding RNAs During Phallus Development in a Marsupial. Non-Coding RNA. 2019; 5(1):3. https://doi.org/10.3390/ncrna5010003
Chicago/Turabian StyleChen, Yu, Yoko Kuroki, Geoff Shaw, Andrew J. Pask, Hongshi Yu, Atsushi Toyoda, Asao Fujiyama, and Marilyn B. Renfree. 2019. "Androgen and Oestrogen Affect the Expression of Long Non-Coding RNAs During Phallus Development in a Marsupial" Non-Coding RNA 5, no. 1: 3. https://doi.org/10.3390/ncrna5010003
APA StyleChen, Y., Kuroki, Y., Shaw, G., Pask, A. J., Yu, H., Toyoda, A., Fujiyama, A., & Renfree, M. B. (2019). Androgen and Oestrogen Affect the Expression of Long Non-Coding RNAs During Phallus Development in a Marsupial. Non-Coding RNA, 5(1), 3. https://doi.org/10.3390/ncrna5010003




