Fluid Flow Characteristics of Healthy and Calcified Aortic Valves Using Three-Dimensional Lagrangian Coherent Structures Analysis
Abstract
:1. Introduction
2. Materials and Methods
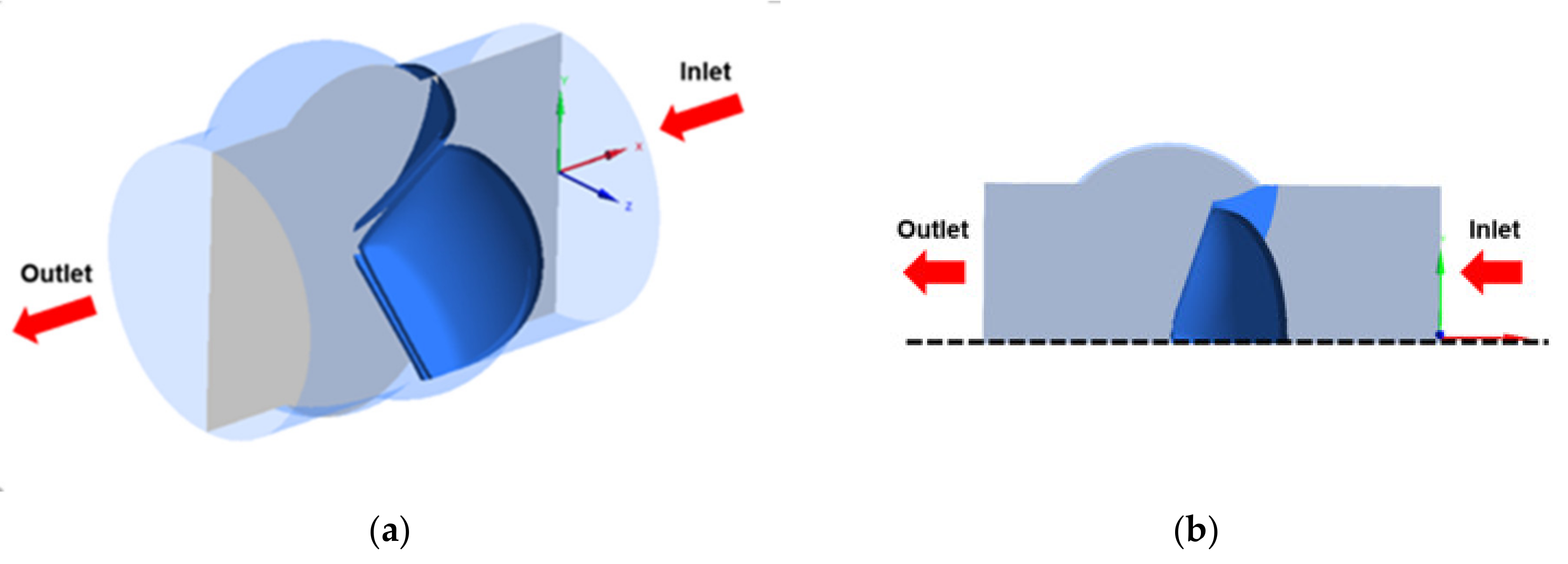
2.1. Model Geometry
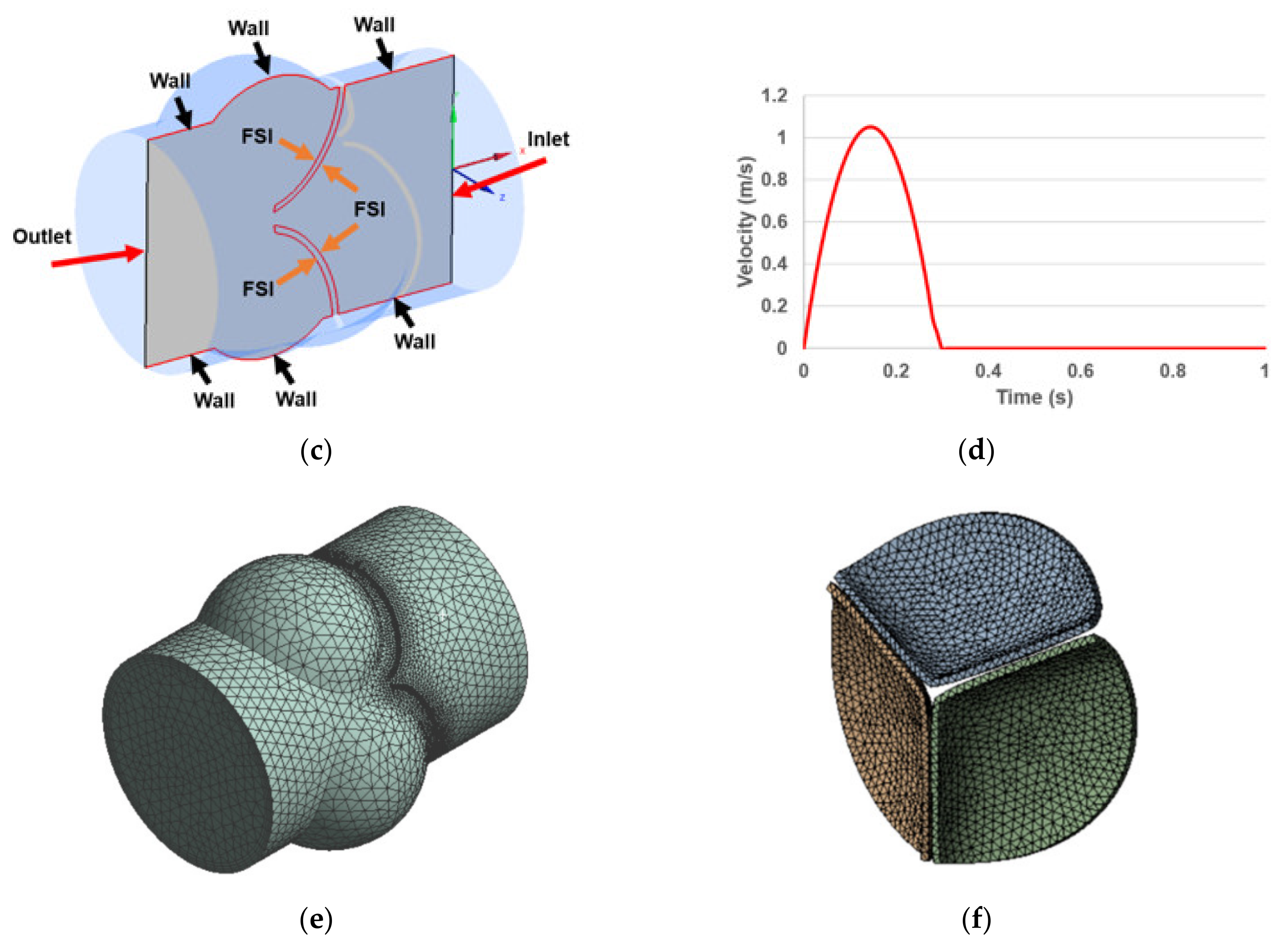
2.2. Boundary Conditions
2.3. Governing Equations in the Fluid and Solid Domains
2.4. FSI Coupling
2.5. Finite-Time Lyapunov Exponent (FTLE) Analysis
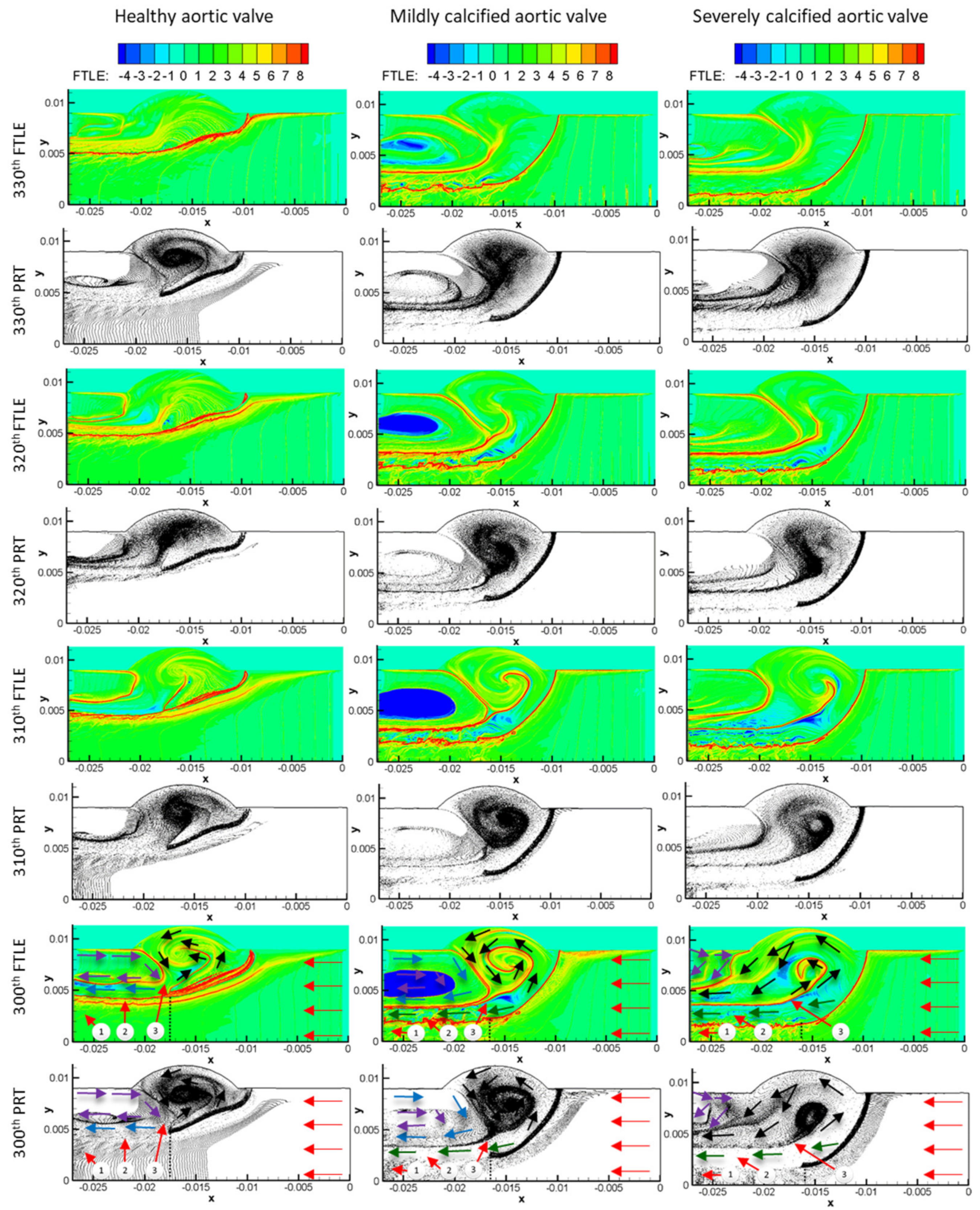
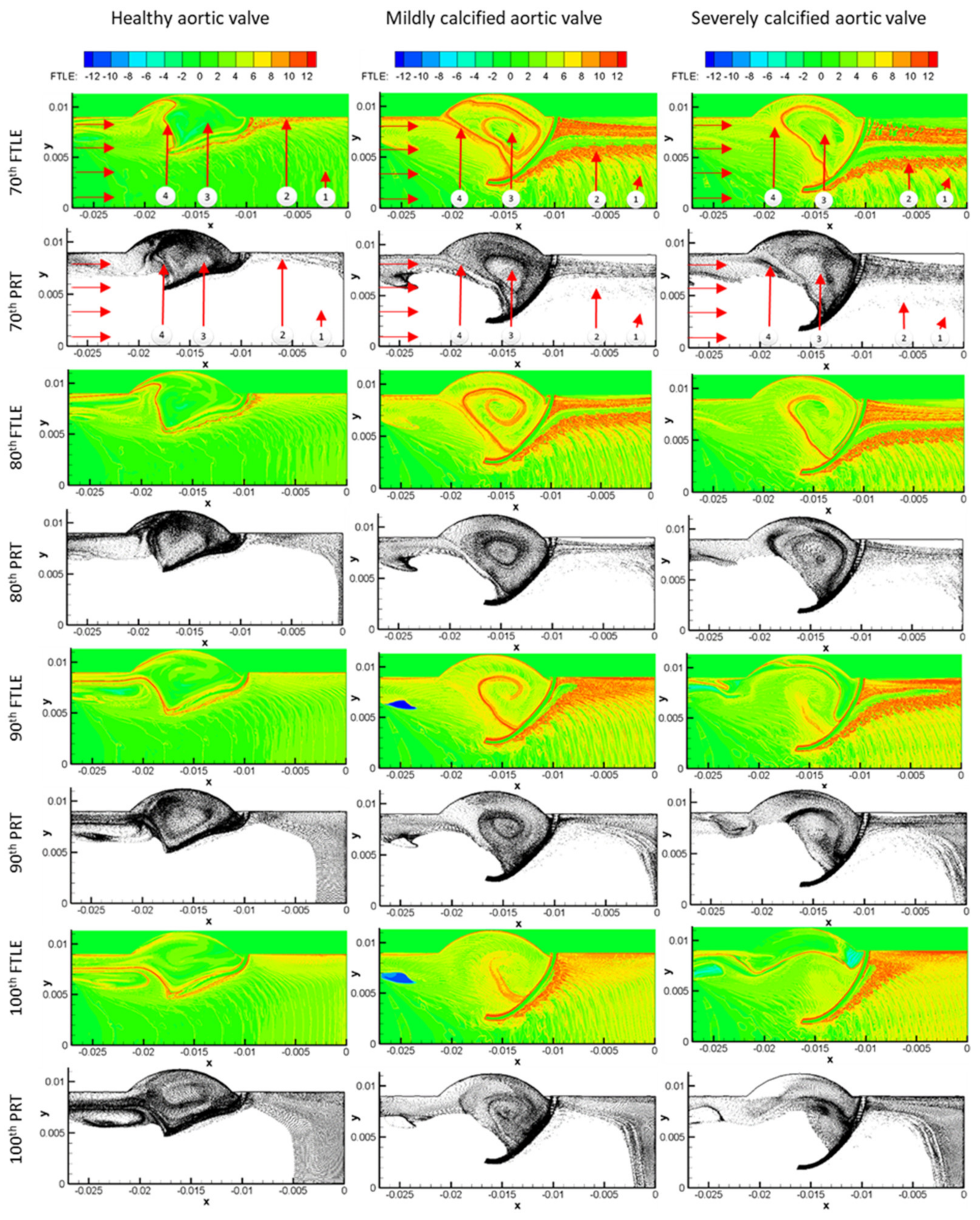
3. Results
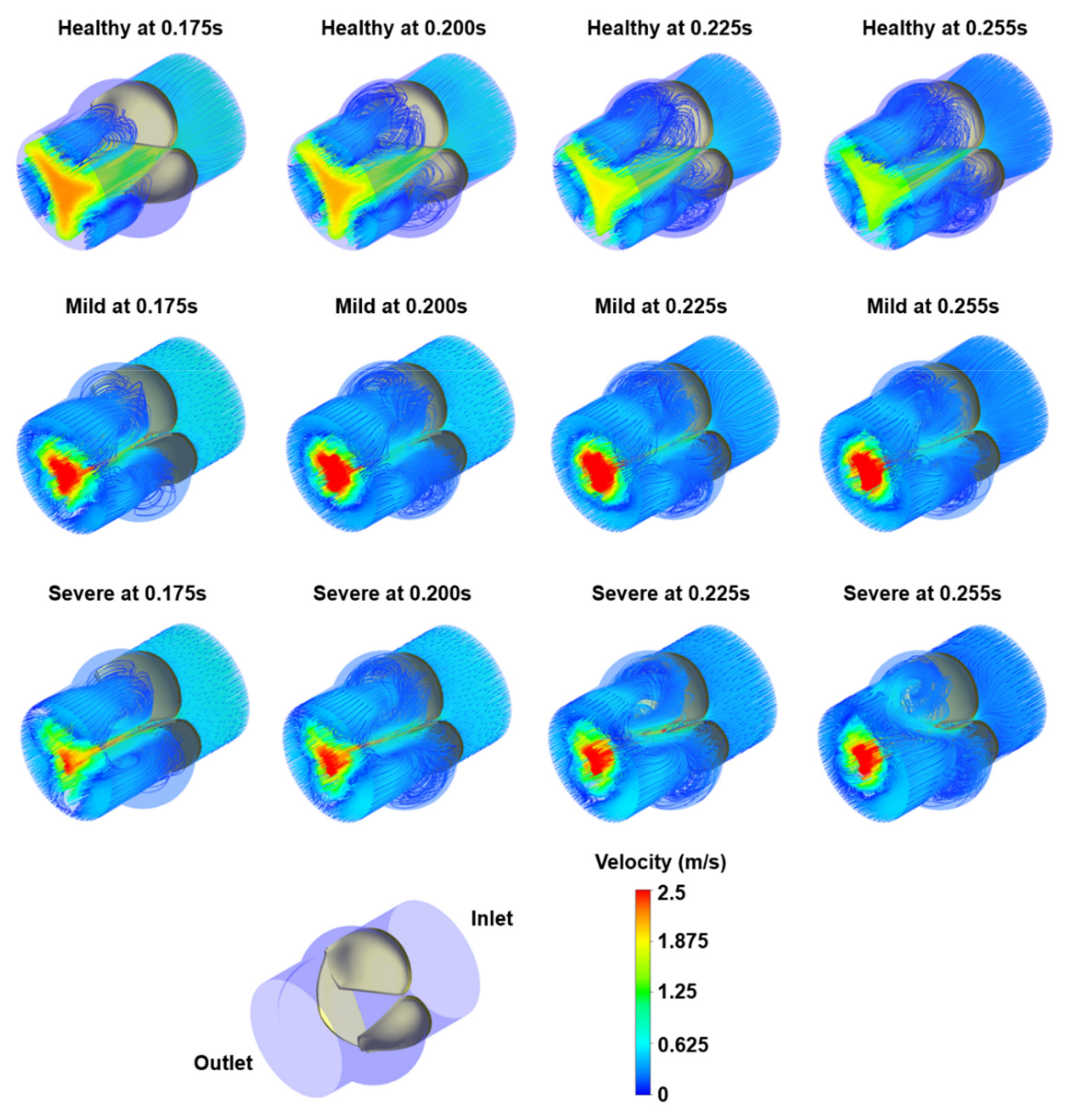
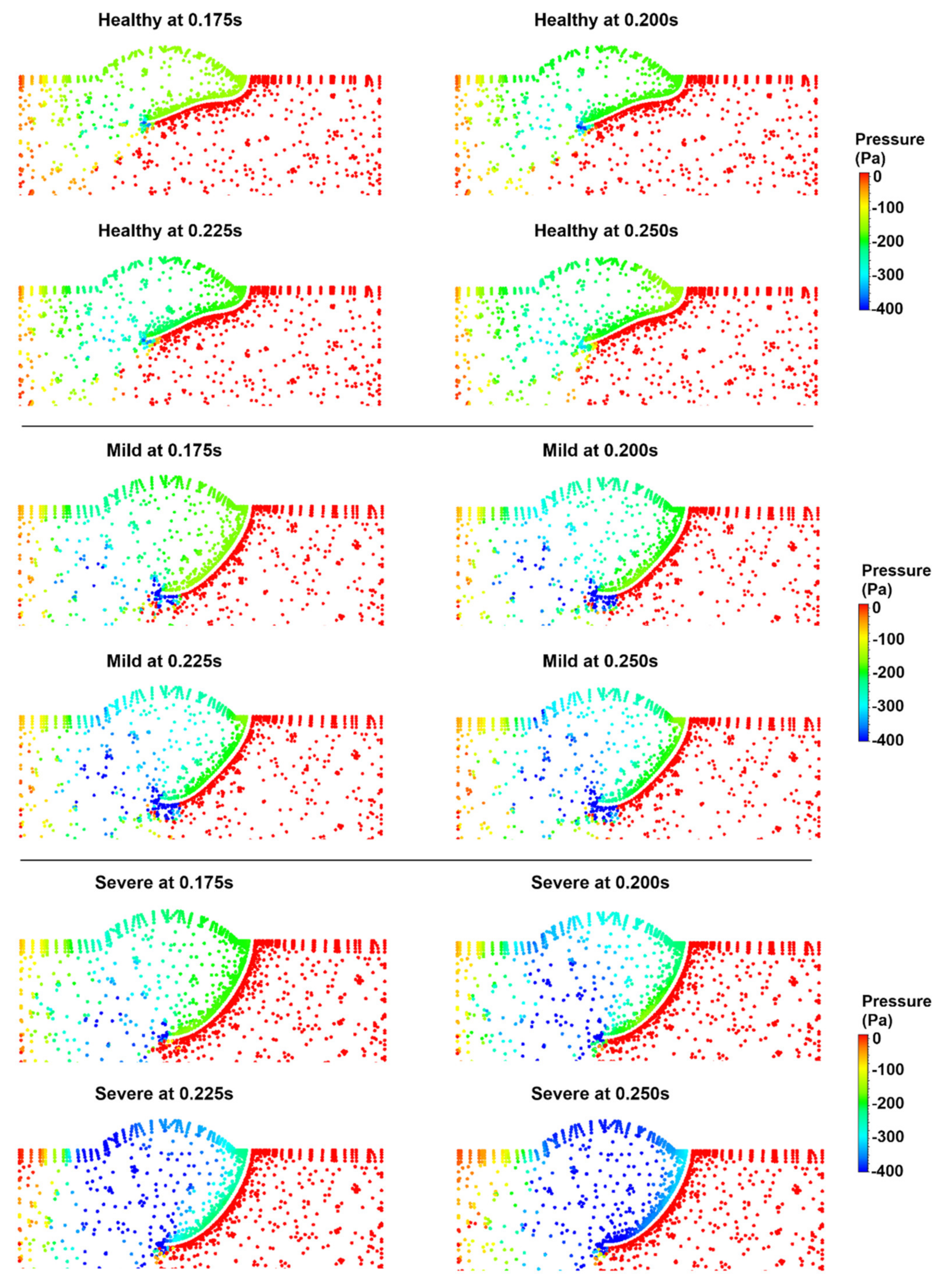
3.1. Flow Streamlines and Pressure Contours
3.2. FTLE Analysis Results
4. Discussion
5. Limitations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gould, S.T.; Srigunapalan, S.; Simmons, C.A.; Anseth, K.S. Hemodynamic and Cellular Response Feedback in Calcific Aortic Valve Disease. Circ. Res. 2013, 113, 186–197. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stewart, B.F.; Siscovick, D.; Lind, B.K.; Gardin, J.M.; Gottdiener, J.S.; Smith, V.E.; Kitzman, D.W.; Otto, C.M. Clinical Factors Associated with Calcific Aortic Valve Disease. J. Am. Coll. Cardiol. 1997, 29, 630–634. [Google Scholar] [CrossRef] [Green Version]
- Halevi, R.; Hamdan, A.; Marom, G.; Mega, M.; Raanani, E.; Haj-Ali, R. Progressive Aortic Valve Calcification: Three-Dimensional Visualization and Biomechanical Analysis. J. Biomech. 2015, 48, 489–497. [Google Scholar] [CrossRef] [PubMed]
- De Hart, J.; Peters, G.W.M.; Schreurs, P.J.G.; Baaijens, F.P.T. A Two-Dimensional Fluid-Structure Interaction Model of the Aortic Value. J. Biomech. 2000, 33, 1079–1088. [Google Scholar] [CrossRef]
- Amindari, A.; Saltik, L.; Kirkkopru, K.; Yacoub, M.; Yalcin, H.C. Assessment of Calcified Aortic Valve Leaflet Deformations and Blood Flow Dynamics Using Fluid-Structure Interaction Modeling. Inform. Med. Unlocked 2017, 9, 191–199. [Google Scholar] [CrossRef]
- De Hart, J.; Peters, G.W.M.; Schreurs, P.J.G.; Baaijens, F.P.T. A Three-Dimensional Computational Analysis of Fluid—Structure Interaction in the Aortic Valve. J. Biomech. 2003, 36, 103–112. [Google Scholar] [CrossRef]
- Spühler, J.H.; Jansson, J.; Jansson, N.; Hoffman, J. 3D Fluid–Structure Interaction Simulation of Aortic Valves Using a Unified Continuum ALE FEM Model. Front. Physiol. 2018, 9, 363. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mao, W.; Caballero, A.; McKay, R.; Primiano, C.; Sun, W. Fully-Coupled Fluid-Structure Interaction Simulation of the Aortic and Mitral Valves in a Realistic 3D Left Ventricle Model. PLoS ONE 2017, 12, e018472. [Google Scholar] [CrossRef] [Green Version]
- Halevi, R.; Hamdan, A.; Marom, G.; Lavon, K.; Ben-Zekry, S.; Raanani, E.; Bluestein, D.; Haj-Ali, R. Fluid–Structure Interaction Modeling of Calcific Aortic Valve Disease Using Patient-Specific Three-Dimensional Calcification Scans. Med. Biol. Eng. Comput. 2016, 54, 1683–1694. [Google Scholar] [CrossRef]
- Haj-Ali, R.; Dasi, L.P.; Kim, H.S.; Choi, J.; Leo, H.W.; Yoganathan, A.P. Structural Simulations of Prosthetic Tri-Leaflet Aortic Heart Valves. J. Biomech. 2008, 41, 1510–1519. [Google Scholar] [CrossRef]
- Dumont, K.; Vierendeels, J.; Kaminsky, R.; Van Nooten, G.; Verdonck, P.; Bluestein, D. Comparison of the Hemodynamic and Thrombogenic Performance of Two Bileaflet Mechanical Heart Valves Using a CFD/FSI Model. J. Biomech. Eng. 2007, 129, 558–565. [Google Scholar] [CrossRef] [Green Version]
- Bavo, A.M.; Rocatello, G.; Iannaccone, F.; Degroote, J.; Vierendeels, J.; Segers, P. Fluid-Structure Interaction Simulation of Prosthetic Aortic Valves: Comparison between Immersed Boundary and Arbitrary Lagrangian-Eulerian Techniques for the Mesh Representation. PLoS ONE 2016, 11, e0154517. [Google Scholar] [CrossRef] [Green Version]
- Borazjani, I. Fluid–Structure Interaction, Immersed Boundary-Finite Element Method Simulations of Bio-Prosthetic Heart Valves. Comput. Methods Appl. Mech. Eng. 2013, 257, 103–116. [Google Scholar] [CrossRef]
- Chandran, K.B.; Vigmostad, S.C. Patient-Specific Bicuspid Valve Dynamics: Overview of Methods and Challenges. J. Biomech. 2013, 46, 208–216. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Conti, C.A.; Votta, E.; Della Corte, A.; Del Viscovo, L.; Bancone, C.; Cotrufo, M.; Redaelli, A. Dynamic Finite Element Analysis of the Aortic Root from MRI-Derived Parameters. Med. Eng. Phys. 2010, 32, 212–221. [Google Scholar] [CrossRef]
- Cao, K.; BukaČ, M.; Sucosky, P. Three-Dimensional Macro-Scale Assessment of Regional and Temporal Wall Shear Stress Characteristics on Aortic Valve Leaflets. Comput. Methods Biomech. Biomed. Eng. 2016, 19, 603–613. [Google Scholar] [CrossRef]
- Cao, K.; Sucosky, P. Computational Comparison of Regional Stress and Deformation Characteristics in Tricuspid and Bicuspid Aortic Valve Leaflets. Int. J. Numer. Method. Biomed. Eng. 2017, 33, e02798. [Google Scholar] [CrossRef]
- Gilmanov, A.; Sotiropoulos, F. Comparative Hemodynamics in an Aorta with Bicuspid and Trileaflet Valves. Theor. Comput. Fluid Dyn. 2016, 30, 67–85. [Google Scholar] [CrossRef]
- Weinberg, E.J.; Kaazempur Mofrad, M.R. A Multiscale Computational Comparison of the Bicuspid and Tricuspid Aortic Valves in Relation to Calcific Aortic Stenosis. J. Biomech. 2008, 41, 3482–3487. [Google Scholar] [CrossRef] [PubMed]
- Chandra, S.; Rajamannan, N.M.; Sucosky, P. Computational Assessment of Bicuspid Aortic Valve Wall-Shear Stress: Implications for Calcific Aortic Valve Disease. Biomech. Model. Mechanobiol. 2012, 11, 1085–1096. [Google Scholar] [CrossRef]
- Kuan, M.Y.S.; Espino, D.M. Systolic Fluid–Structure Interaction Model of the Congenitally Bicuspid Aortic Valve: Assessment of Modelling Requirements. Comput. Methods Biomech. Biomed. Eng. 2015, 18, 1305–1320. [Google Scholar] [CrossRef] [PubMed]
- Marom, G.; Peleg, M.; Halevi, R.; Rosenfeld, M.; Raanani, E.; Hamdan, A.; Haj-Ali, R. Fluid-Structure Interaction Model of Aortic Valve with Porcine-Specific Collagen Fiber Alignment in the Cusps. J. Biomech. Eng. 2013, 135, 101001–101006. [Google Scholar] [CrossRef] [Green Version]
- Grande, K.J.; Cochran, R.P.; Reinhall, P.G.; Kunzelma, K.S. Stress Variations in the Human Aortic Root and Valve: The Role of Anatomic Asymmetry. Ann. Biomed. Eng. 1998, 26, 534–545. [Google Scholar] [CrossRef]
- Votta, E.; Le, T.B.; Stevanella, M.; Fusini, L.; Caiani, E.G.; Redaelli, A.; Sotiropoulos, F. Toward Patient-Specific Simulations of Cardiac Valves: State-of-the-Art and Future Directions. J. Biomech. 2013, 46, 217–228. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Katayama, S.; Umetani, N.; Sugiura, S.; Hisada, T. The Sinus of Valsalva Relieves Abnormal Stress on Aortic Valve Leaflets by Facilitating Smooth Closure. J. Thorac. Cardiovasc. Surg. 2008, 136, 1528–1535.e1. [Google Scholar] [CrossRef] [Green Version]
- Balachandran, K.; Sucosky, P.; Yoganathan, A.P. Hemodynamics and Mechanobiology of Aortic Valve Inflammation and Calcification. Int. J. Inflamm. 2011, 2011, 263870. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hutcheson, J.D.; Goettsch, C.; Rogers, M.A.; Aikawa, E. Revisiting Cardiovascular Calcification: A Multifaceted Disease Requiring a Multidisciplinary Approach. Semin. Cell Dev. Biol. 2015, 46, 68–77. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seaman, C.; McNally, A.; Biddle, S.; Jankowski, L.; Sucosky, P. Generation of Simulated Calcific Lesions in Valve Leaflets for Flow Studies. J. Heart Valve Dis. 2015, 24, 115–125. [Google Scholar] [PubMed]
- Arjunon, S.; Rathan, S.; Jo, H.; Yoganathan, A.P. Aortic Valve: Mechanical Environment and Mechanobiology. Ann. Biomed. Eng. 2013, 41, 1331–1346. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Olcay, A.B.; Amindari, A.; Kirkkopru, K.; Yalcin, H.C. Characterization of Disturbed Hemodynamics Due to Stenosed Aortic Jets with a Lagrangian Coherent Structures Technique. J. Appl. Fluid Mech. 2018, 11, 375–384. [Google Scholar] [CrossRef]
- Shadden, S.C.; Astorino, M.; Gerbeau, J.-F. Computational Analysis of an Aortic Valve Jet with Lagrangian Coherent Structures. Chaos Interdiscip. J. Nonlinear Sci. 2010, 20, 017512. [Google Scholar] [CrossRef] [Green Version]
- May-Newman, K.; Vu, V.; Herold, B. Modeling the Link between Left Ventricular Flow and Thromboembolic Risk Using Lagrangian Coherent Structures. Fluids 2016, 1, 38. [Google Scholar] [CrossRef]
- Green, M.A.; Rowley, C.W.; Smits, A.J. Using Hyperbolic Lagrangian Coherent Structures to Investigate Vortices in Bioinspired Fluid Flows. Chaos 2010, 20, 017510. [Google Scholar] [CrossRef] [Green Version]
- Shadden, S.C.; Arzani, A. Lagrangian Postprocessing of Computational Hemodynamics. Ann. Biomed. Eng. 2015, 43, 41–58. [Google Scholar] [CrossRef]
- Olcay, A.B.; Krueger, P.S. Measurement of Ambient Fluid Entrainment during Laminar Vortex Ring Formation. Exp. Fluids 2008, 44, 235–247. [Google Scholar] [CrossRef]
- Olcay, A.B.; Pottebaum, T.S.; Krueger, P.S. Sensitivity of Lagrangian Coherent Structure Identification to Flow Field Resolution and Random Errors. Chaos 2010, 20, 017506. [Google Scholar] [CrossRef] [PubMed]
- Olcay, A.B.; Krueger, P.S. Momentum Evolution of Ejected and Entrained Fluid during Laminar Vortex Ring Formation. Theor. Comput. Fluid Dyn. 2010, 24, 465–482. [Google Scholar] [CrossRef]
- Olcay, A.B. Investigation of a Wake Formation for Flow over a Cylinder Using Lagrangian Coherent Structures. Prog. Comput. Fluid Dyn. 2016, 16, 126–130. [Google Scholar] [CrossRef]
- Mutlu, O.; Olcay, A.B.; Bilgin, C.; Hakyemez, B. Evaluating the Effectiveness of 2 Different Flow Diverter Stents Based on the Stagnation Region Formation in an Aneurysm Sac Using Lagrangian Coherent Structure. World Neurosurg. 2019, 127, e727–e737. [Google Scholar] [CrossRef] [PubMed]
- Mutlu, O.; Olcay, A.B.; Bilgin, C.; Hakyemez, B. Evaluating the Effect of the Number of Wire of Flow Diverter Stents on the Nonstagnated Region Formation in an Aneurysm Sac Using Lagrangian Coherent Structure and Hyperbolic Time Analysis. World Neurosurg. 2020, 133, e666–e682. [Google Scholar] [CrossRef] [PubMed]
- Mutlu, O.; Olcay, A.B.; Bilgin, C.; Hakyemez, B. Understanding the Effect of Effective Metal Surface Area of Flow Diverter Stent’s on the Patient-Specific Intracranial Aneurysm Numerical Model Using Lagrangian Coherent Structures. J. Clin. Neurosci. 2020, 133, e666–e682. [Google Scholar] [CrossRef]
- Chester, A.H.; El-Hamamsy, I.; Butcher, J.T.; Latif, N.; Bertazzo, S.; Yacoub, M.H. The Living Aortic Valve: From Molecules to Function. Glob. Cardiol. Sci. Pract. 2014, 2014, 11. [Google Scholar] [CrossRef] [Green Version]
- Girfoglio, M.; Quaini, A.; Rozza, G. A Finite Volume Approximation of the Navier-Stokes Equations with Nonlinear Filtering Stabilization. Comput. Fluids 2019, 187, 27–45. [Google Scholar] [CrossRef] [Green Version]
- Frolov, S.V.; Sindeev, S.V.; Lischouk, V.A.; Gazizova, D.S.; Liepsch, D.; Balasso, A. A Lumped Parameter Model of Cardiovascular System with Pulsating Heart for Diagnostic Studies. J. Mech. Med. Biol. 2017, 17, 1750056. [Google Scholar] [CrossRef]
- Young, D.F. Fluid Mechanics of Arterial Stenoses. J. Biomech. Eng. 1979, 101, 157–175. [Google Scholar] [CrossRef]
- Meslem, A.; Bode, F.; Croitoru, C.; Nastase, I. Comparison of Turbulence Models in Simulating Jet Flow from a Cross-Shaped Orifice. Eur. J. Mech. B Fluids 2014, 44, 100–120. [Google Scholar] [CrossRef]
- Benra, F.K.; Dohmen, H.J.; Pei, J.; Schuster, S.; Wan, B. A Comparison of One-Way and Two-Way Coupling Methods for Numerical Analysis of Fluid-Structure Interactions. J. Appl. Math. 2011, 2011, 853560. [Google Scholar] [CrossRef]
- Salman, H.E.; Ramazanli, B.; Yavuz, M.M.; Yalcin, H.C. Biomechanical Investigation of Disturbed Hemodynamics-Induced Tissue Degeneration in Abdominal Aortic Aneurysms Using Computational and Experimental Techniques. Front. Bioeng. Biotechnol. 2019, 7, 111. [Google Scholar] [CrossRef]
- Bathe, K.J.; Zhang, H.; Ji, S. Finite Element Analysis of Fluid Flows Fully Coupled with Structural Interactions. Comput. Struct. 1999, 72, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Haller, G. Distinguished Material Surfaces and Coherent Structures in Three-Dimensional Fluid Flows. Phys. D Nonlinear Phenom. 2001, 149, 248–277. [Google Scholar] [CrossRef] [Green Version]
- Shadden, S.C.; Lekien, F.; Marsden, J.E. Definition and Properties of Lagrangian Coherent Structures from Finite-Time Lyapunov Exponents in Two-Dimensional Aperiodic Flows. Phys. D Nonlinear Phenom. 2005, 212, 271–304. [Google Scholar] [CrossRef]
- Lekien, F.; Shadden, S.C.; Marsden, J.E. Lagrangian Coherent Structures in N-Dimensional Systems. J. Math. Phys. 2007, 48, 065404. [Google Scholar] [CrossRef]
- Shadden, S.C.; Taylor, C.A. Characterization of Coherent Structures in the Cardiovascular System. Ann. Biomed. Eng. 2008, 36, 1152–1162. [Google Scholar] [CrossRef] [PubMed]
- Weinberg, E.J.; Mofrad, M.R.K. Three-Dimensional, Multiscale Simulations of the Human Aortic Valve. Cardiovasc. Eng. 2007, 7, 140–155. [Google Scholar] [CrossRef]
- Weska, R.F.; Aimoli, C.G.; Nogueira, G.M.; Leirner, A.A.; Maizato, M.J.S.; Higa, O.Z.; Polakievicz, B.; Pitombo, R.N.M.; Beppu, M.M. Natural and Prosthetic Heart Valve Calcification: Morphology and Chemical Composition Characterization. Artif. Organs 2010, 34, 311–318. [Google Scholar] [CrossRef] [PubMed]
- Nandy, S.; Tarbell, J.M. Flush Mounted Hot Film Anemometer Measurement of Wall Shear Stress Distal to a Tri-Leaflet Valve for Newtonian and Non-Newtonian Blood Analog Fluids. Biorheology 1987, 24, 483–500. [Google Scholar] [CrossRef]
| Healthy Aortic Valve | Mildly Calcified Aortic Valve | Severely Calcified Aortic Valve | |
|---|---|---|---|
| 400th step (added for the first time) | 104,291 | 104,291 | 104,291 |
| 339th step (Total with readded particles) | 261,418 | 398,350 | 434,337 |
| 330th step (remaining particles) | 153,900 | 285,259 | 325,046 |
| 329th step (total with readded particles) | 254,945 | 382,472 | 421,736 |
| 320th step (remaining particles) | 155,146 | 268,018 | 305,521 |
| 319th step (total with re-added particles) | 255,743 | 367,599 | 403,394 |
| 310th step (remaining particles) | 165,389 | 276,347 | 309,397 |
| 309th step (total with readded particles) | 263,070 | 376,494 | 407977 |
| 300th step (remaining particles) | 185,322 | 295,500 | 323,787 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mutlu, O.; Salman, H.E.; Yalcin, H.C.; Olcay, A.B. Fluid Flow Characteristics of Healthy and Calcified Aortic Valves Using Three-Dimensional Lagrangian Coherent Structures Analysis. Fluids 2021, 6, 203. https://doi.org/10.3390/fluids6060203
Mutlu O, Salman HE, Yalcin HC, Olcay AB. Fluid Flow Characteristics of Healthy and Calcified Aortic Valves Using Three-Dimensional Lagrangian Coherent Structures Analysis. Fluids. 2021; 6(6):203. https://doi.org/10.3390/fluids6060203
Chicago/Turabian StyleMutlu, Onur, Huseyin Enes Salman, Huseyin Cagatay Yalcin, and Ali Bahadir Olcay. 2021. "Fluid Flow Characteristics of Healthy and Calcified Aortic Valves Using Three-Dimensional Lagrangian Coherent Structures Analysis" Fluids 6, no. 6: 203. https://doi.org/10.3390/fluids6060203
APA StyleMutlu, O., Salman, H. E., Yalcin, H. C., & Olcay, A. B. (2021). Fluid Flow Characteristics of Healthy and Calcified Aortic Valves Using Three-Dimensional Lagrangian Coherent Structures Analysis. Fluids, 6(6), 203. https://doi.org/10.3390/fluids6060203








