Analysis of Disruption of Airflow and Particle Distribution by Surgical Personnel and Lighting Fixture in Operating Rooms
Abstract
1. Introduction
1.1. Air Cleanliness
1.2. Air Distribution Systems of the Operating Room
- Laminar air flow (LAF);
- Turbulent mixed flow;
- Temperature-controlled airflow (TCAF).
1.2.1. Laminar Airflow
1.2.2. Turbulent Mixed Airflow
1.2.3. Temperature-Controlled Airflow
2. Methodology
2.1. CFD Modelling
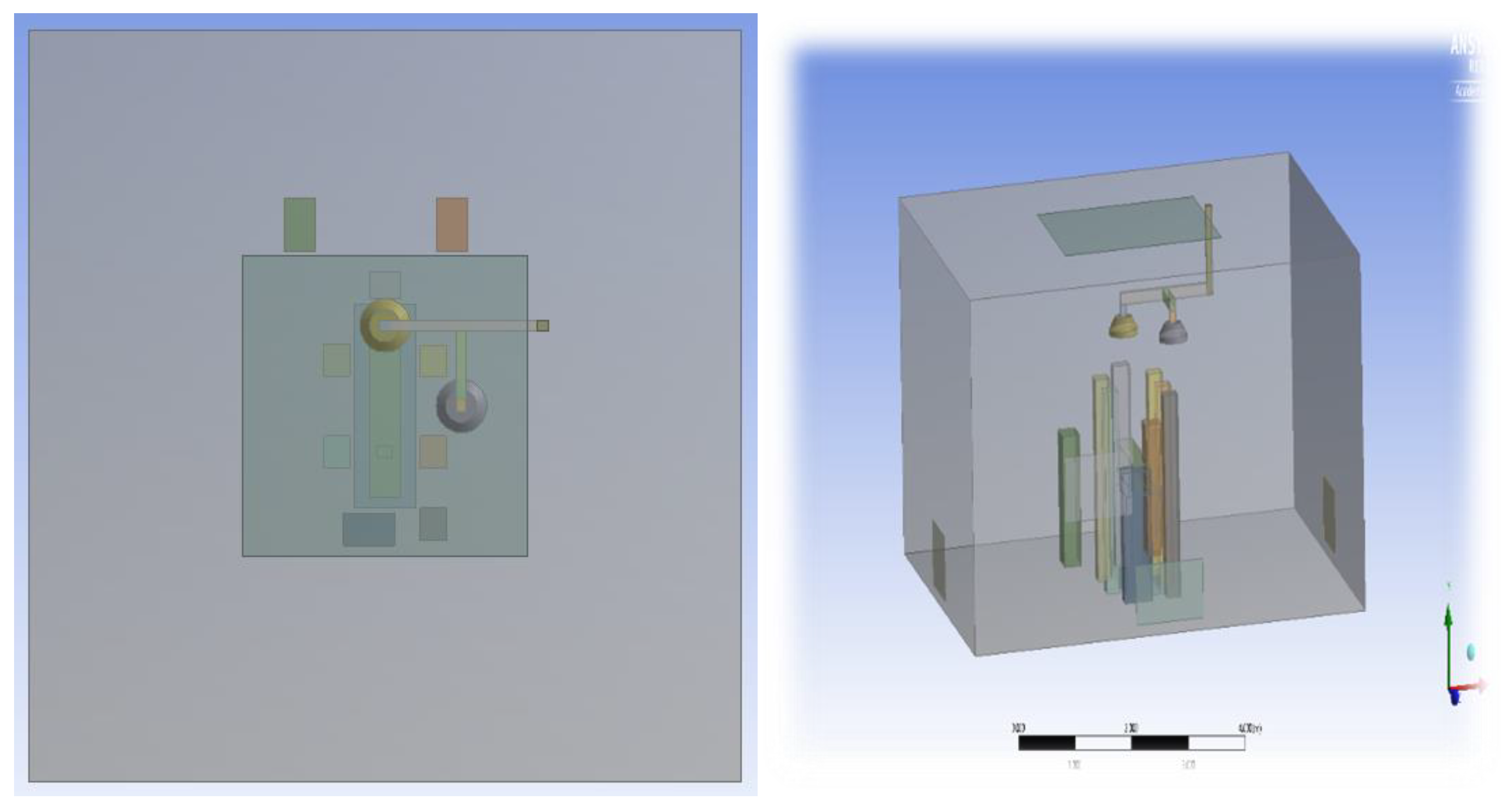
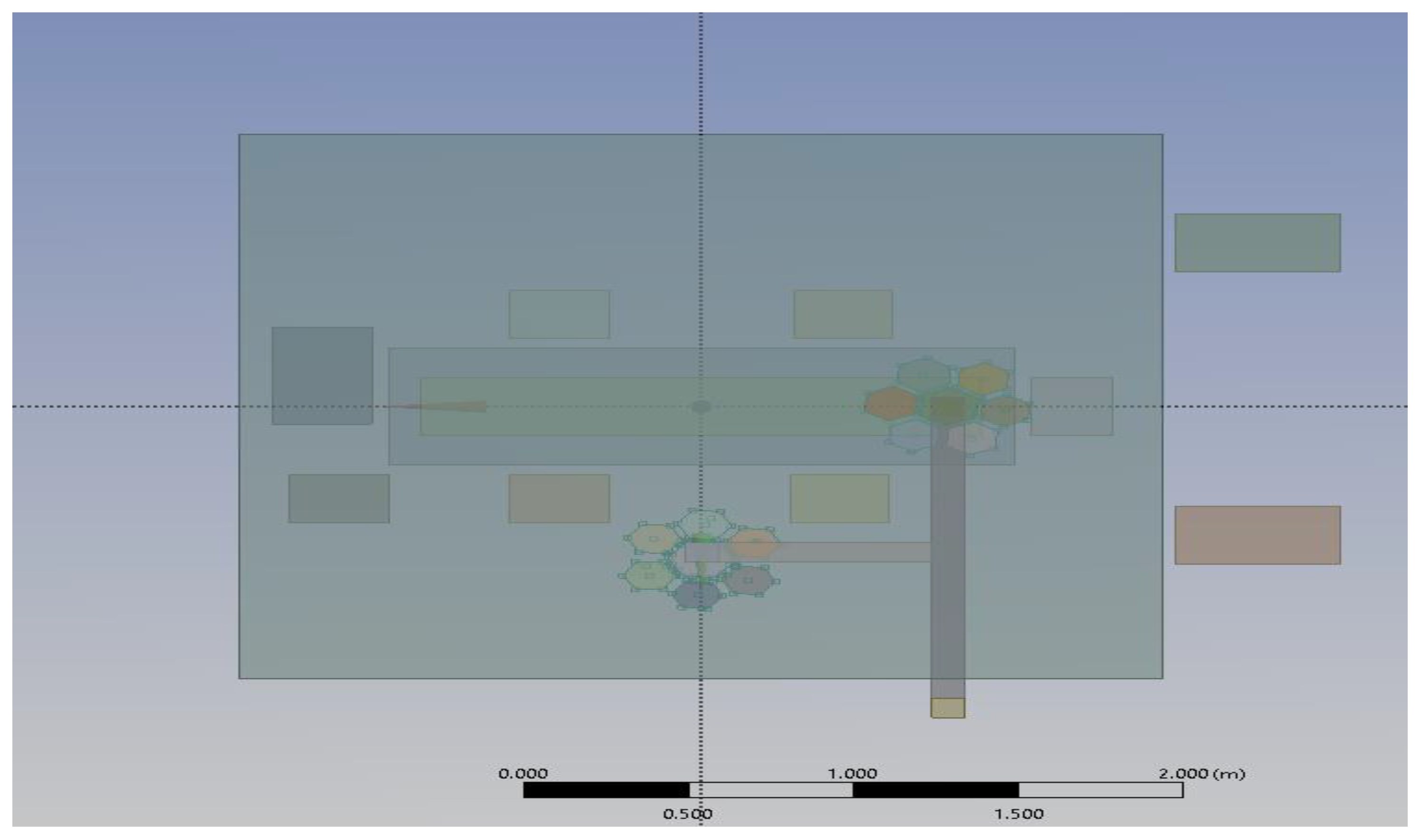
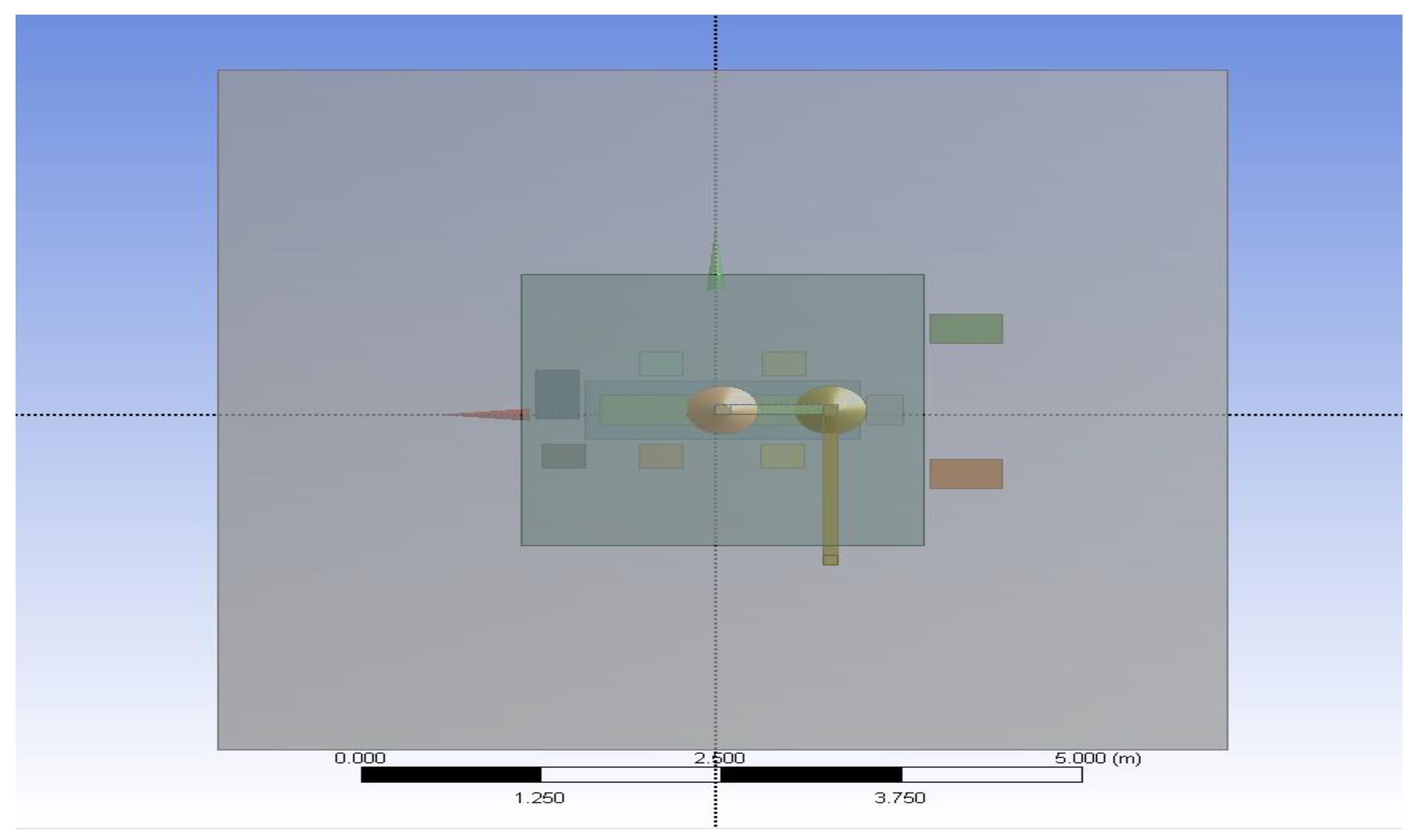
2.1.1. Computational Domain and Modelling Assumptions
2.1.2. CFD Parameters
- Turbulence model: Realisable k–ε model with enhanced wall treatment.
- Discretisation schemes: Second-order upwind for momentum, energy, turbulence quantities; SIMPLE algorithm for pressure-velocity coupling.
- Gradient calculation: Least-squares cell-based.
- Gravity: −9.81 m·s−2 along the z axis.
- Meshing: Tetrahedron, refined at surfaces inside the LAF volume.
- ○
- Supply diffusers: Constant velocity inlet.
- ○
- Return diffusers: Outflow or pressure outlet boundary.
- ○
- Walls and equipment: No-slip adiabatic or prescribed heat flux (for heated components).
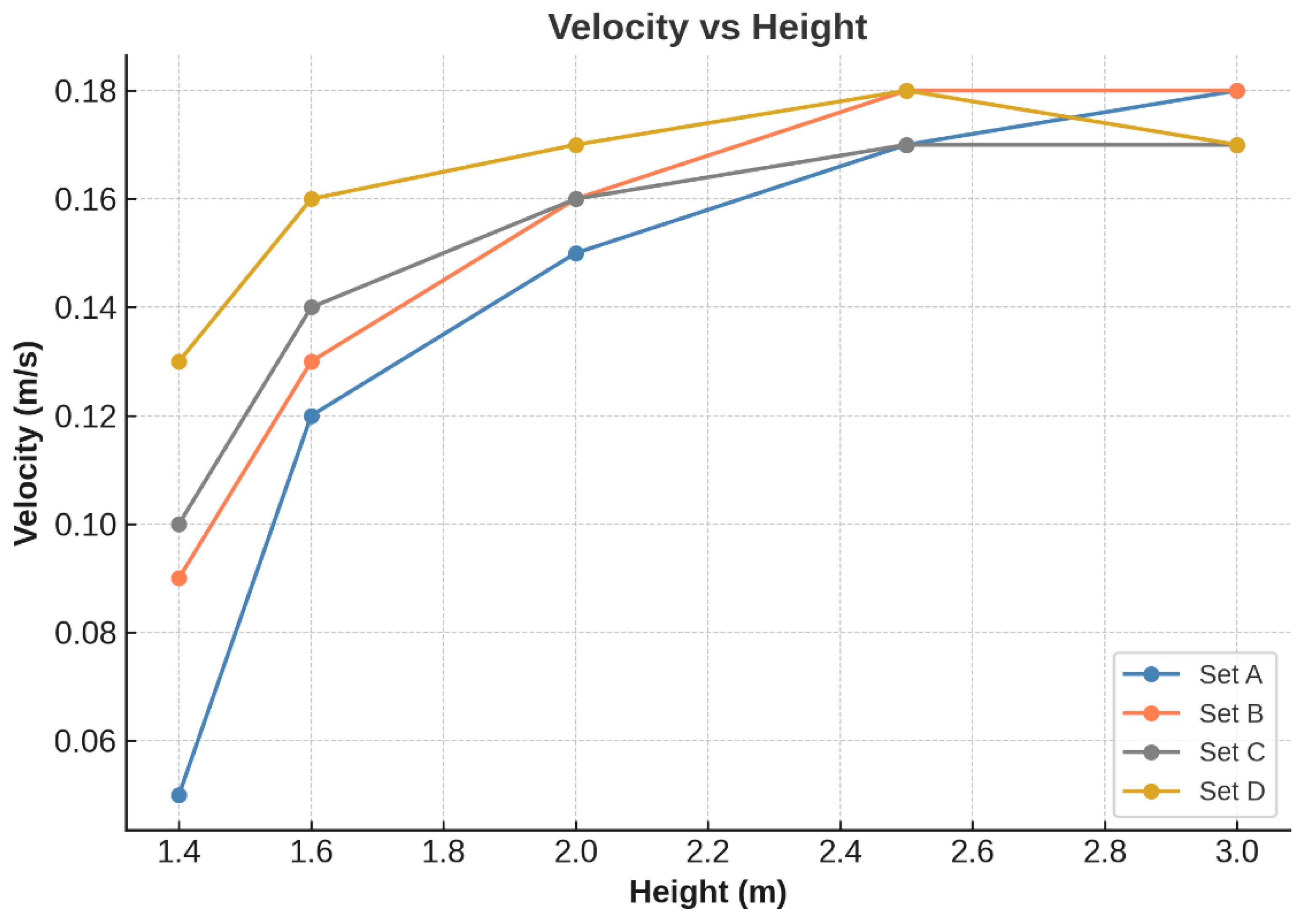
2.1.3. Grid Independence Study
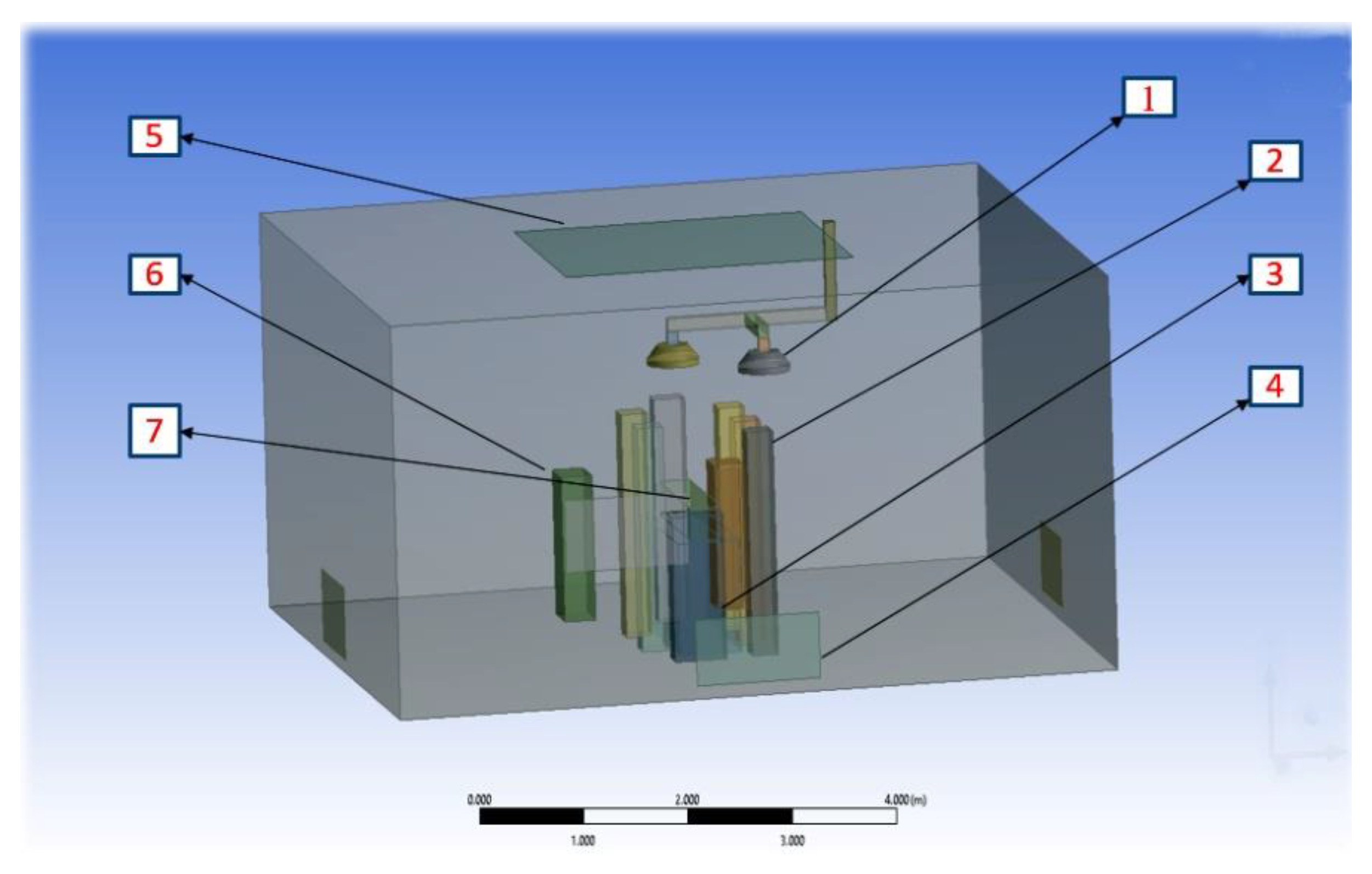
2.1.4. Modelling of Equipment and Occupants
2.1.5. Modelling of Gaseous Contaminants
2.1.6. Modelling of Particle Tracking
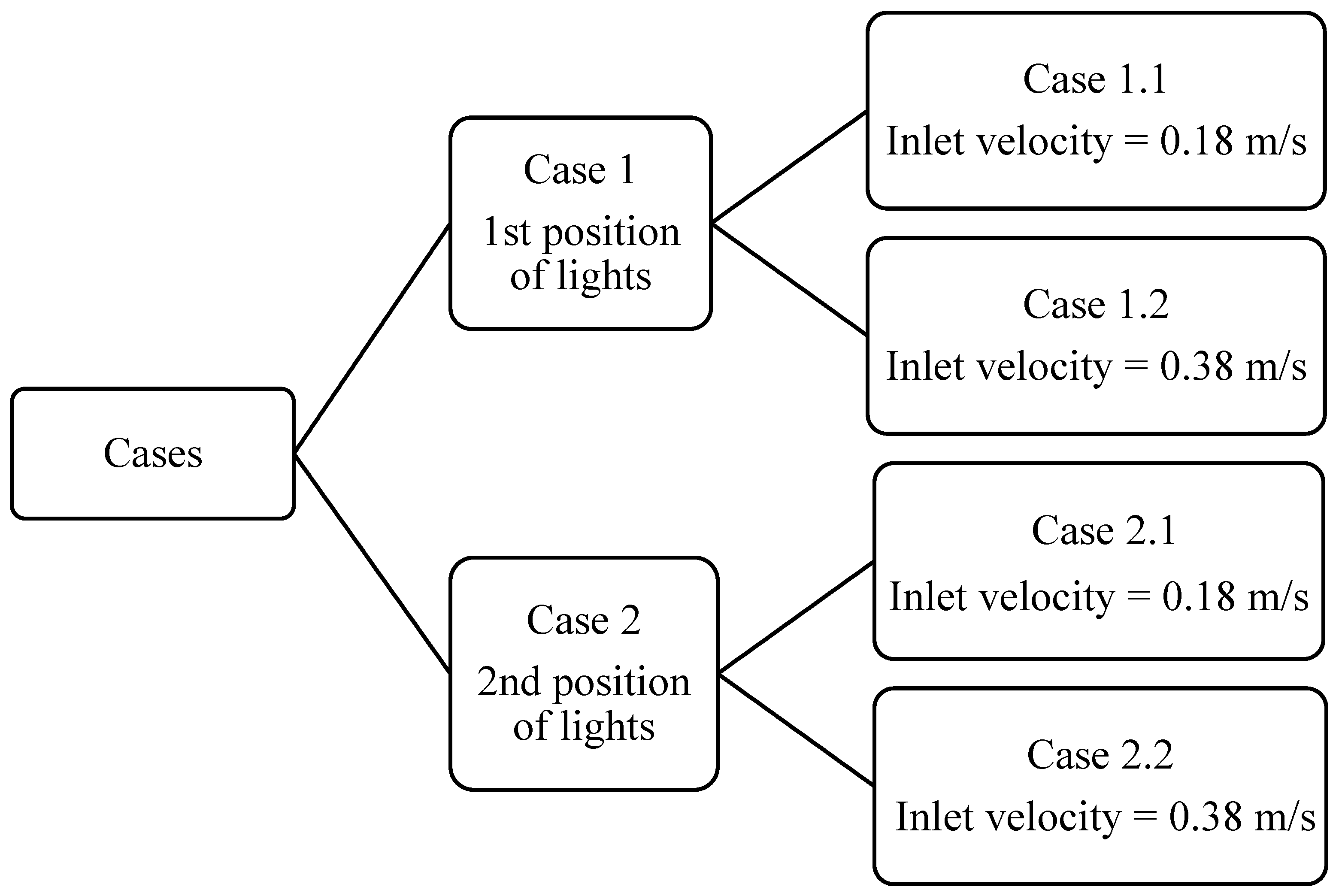
2.1.7. Boundary Conditions
- Inlet air velocity, temperature, and pressure, which were varied across simulation cases to study airflow sensitivity.
- Outlet boundaries were defined using a pressure outlet condition.
- Thermal and metabolic heat loads applied to human and equipment surfaces were specified based on empirical values, as detailed in Table 3.
2.2. Field Measurement
3. Results and Discussion
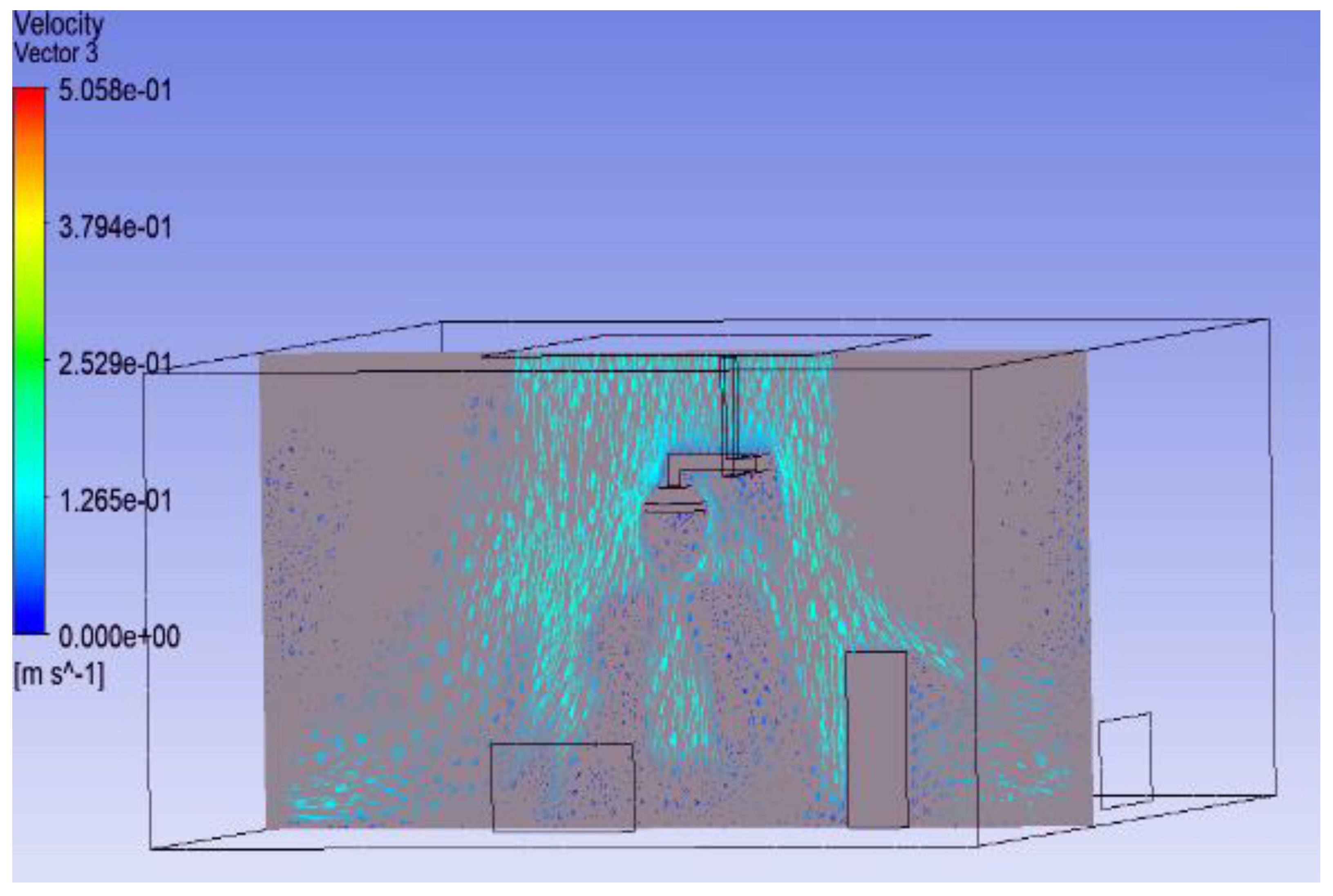
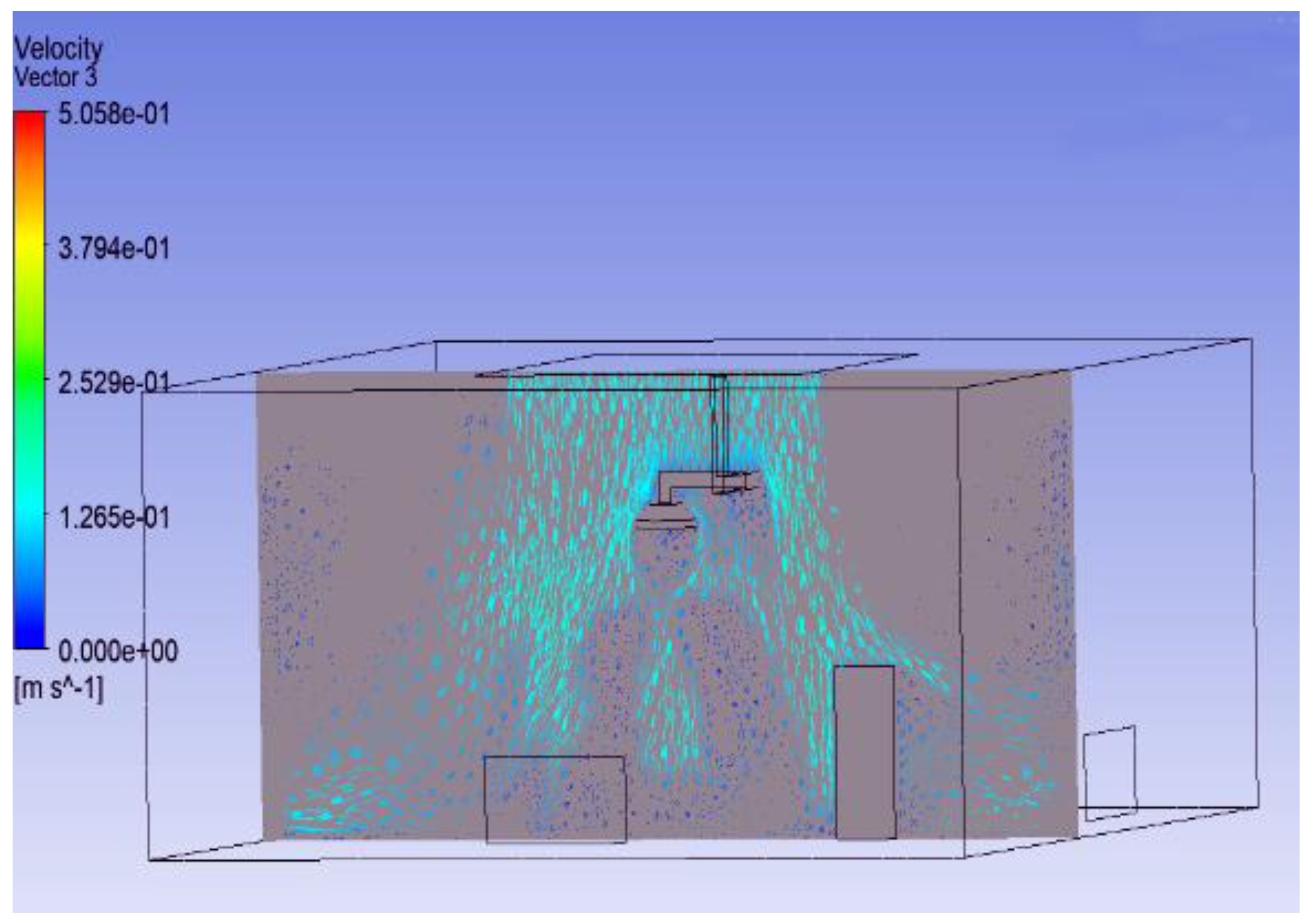
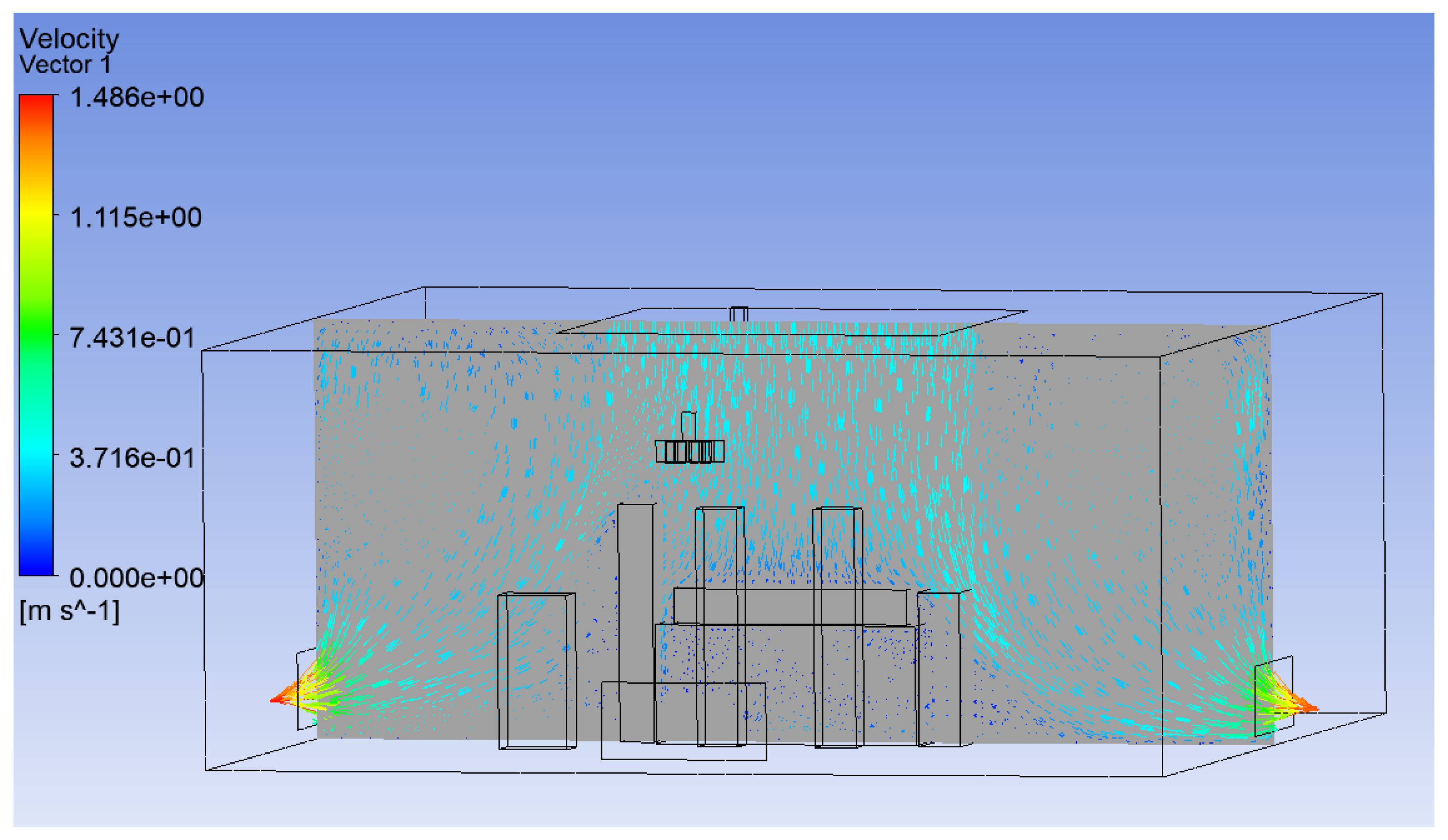
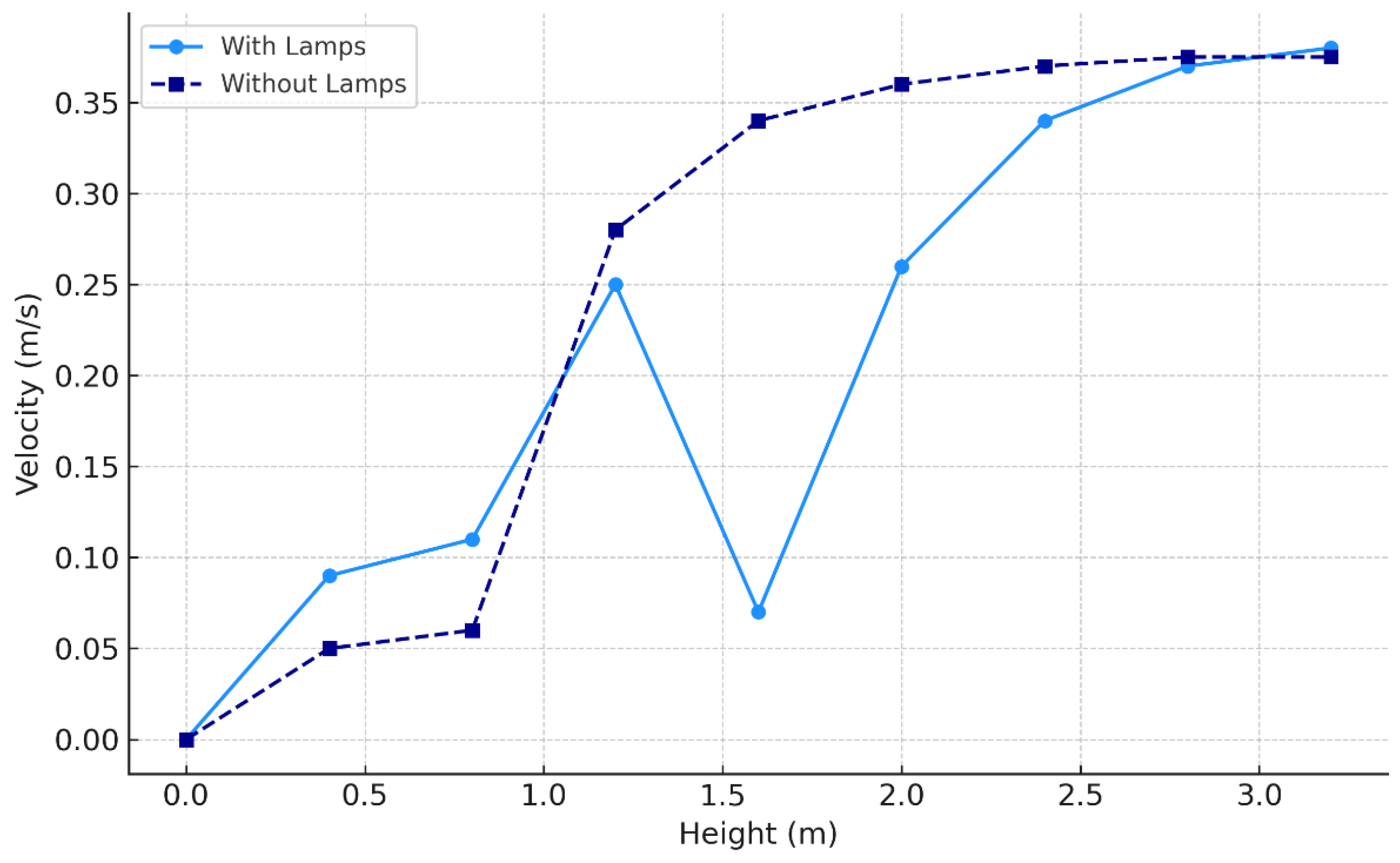
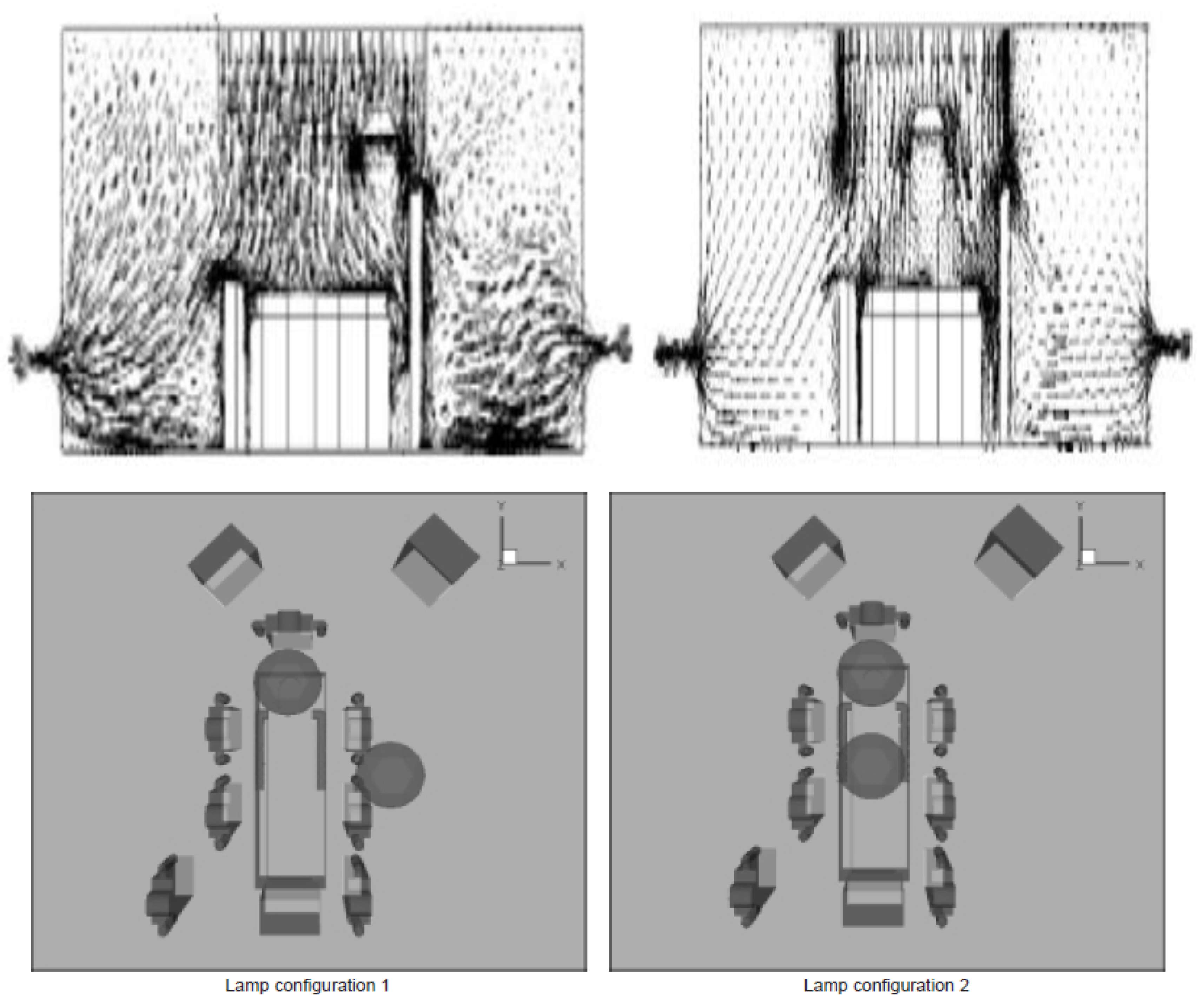
3.1. Effect of Position of Surgical Lamp on Flow Distribution
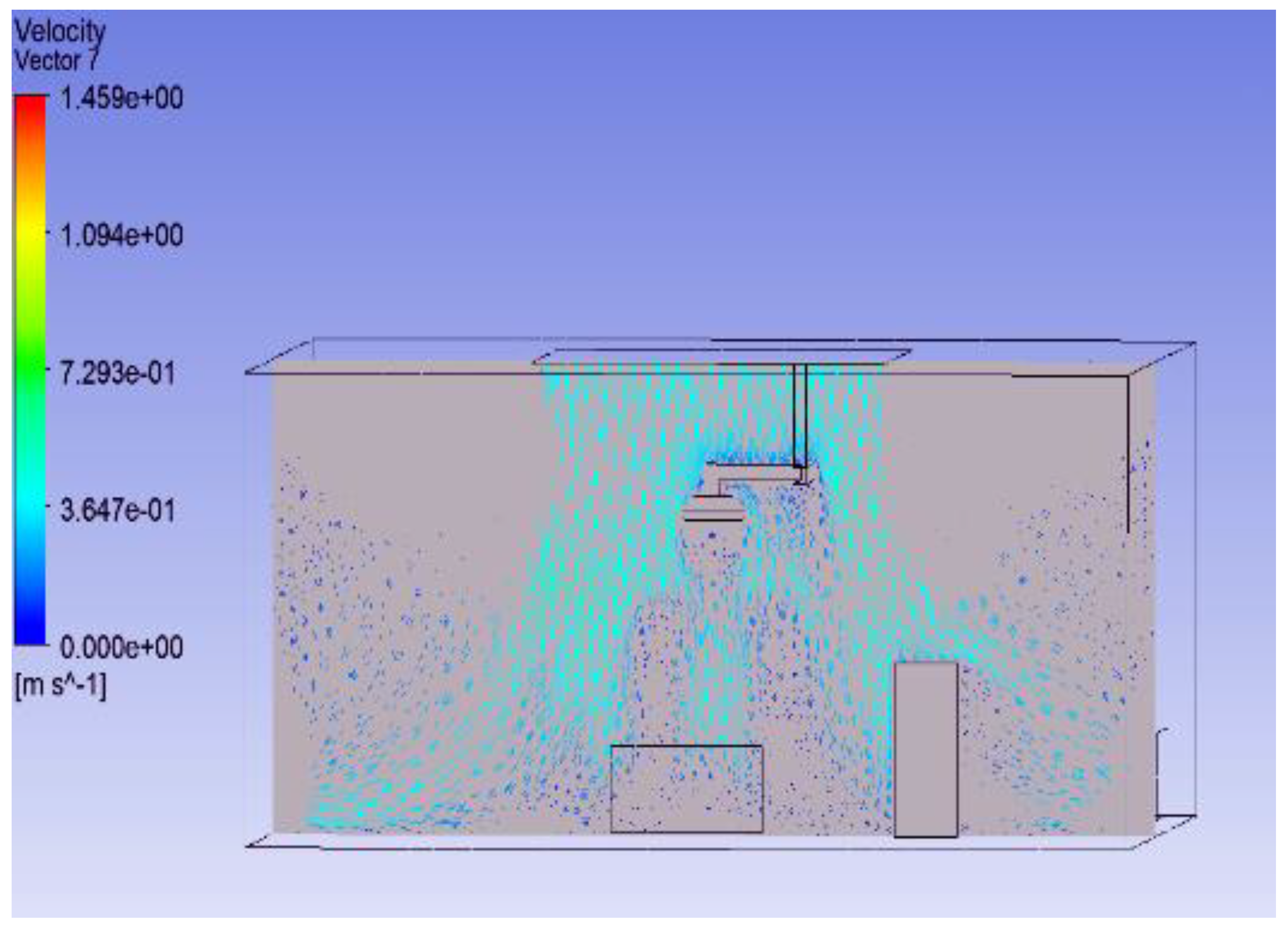
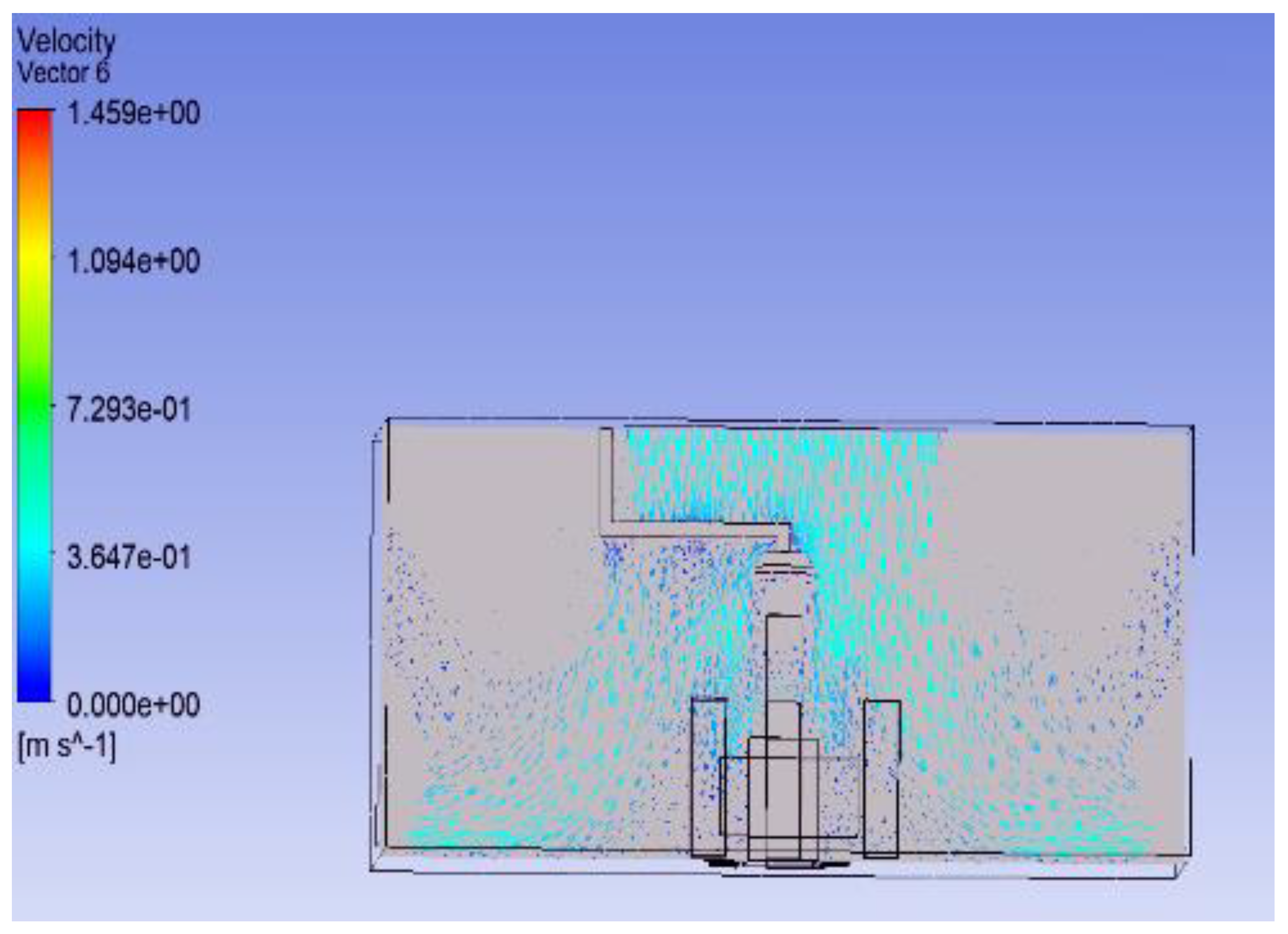
3.2. Shape of Surgical Lamp on Flow Distribution
- A classic closed-shape lamp, and
- A semi-open shape featuring ventilation gaps between its structural elements.
- Figure 15 demonstrates that the airflow is markedly reduced beneath the closed lamp, leading to stagnation zones.
- Figure 16, on the other hand, shows that the semi-open lamp allows airflow to maintain its uniformity, with minimal distortion observed below the fixture.
3.3. Comparison of the Results with Literature
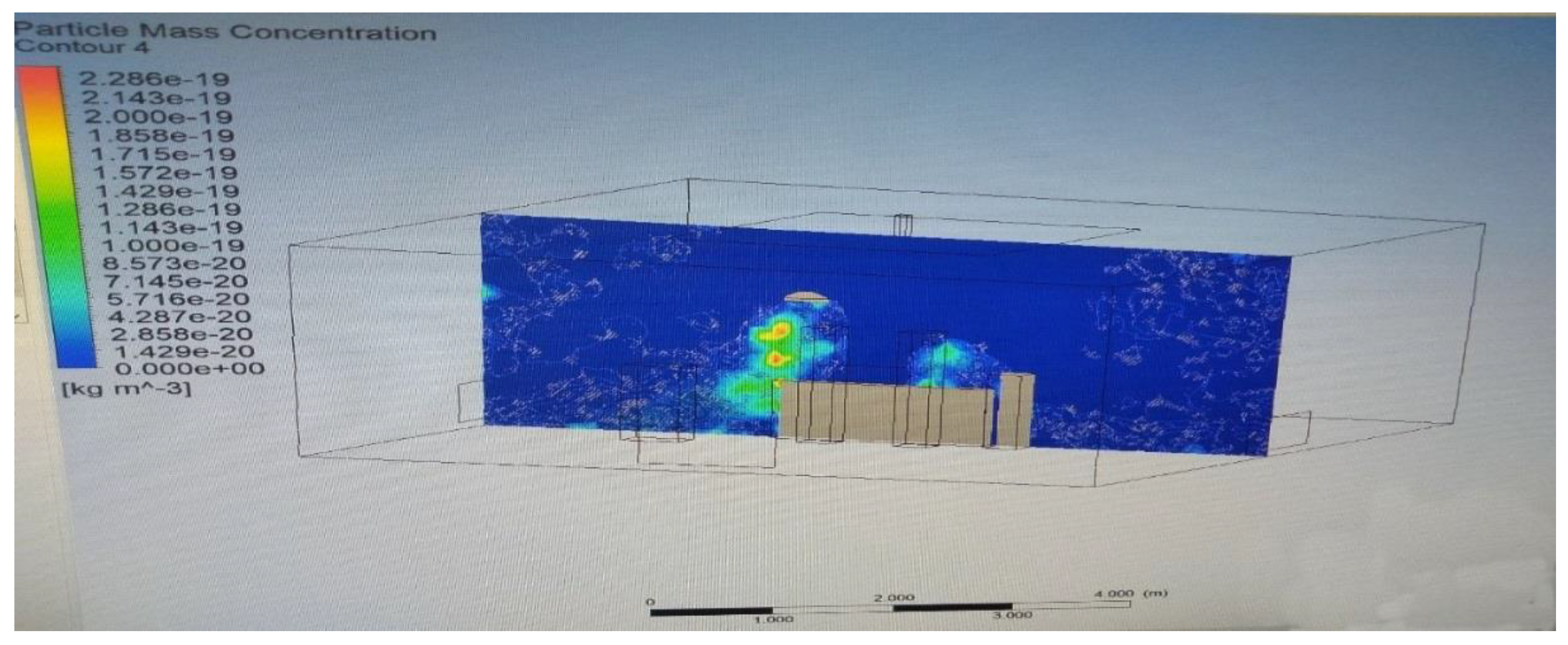
3.4. Modelling of Particle Tracking
3.5. Experimental Results
On-Site Measurement
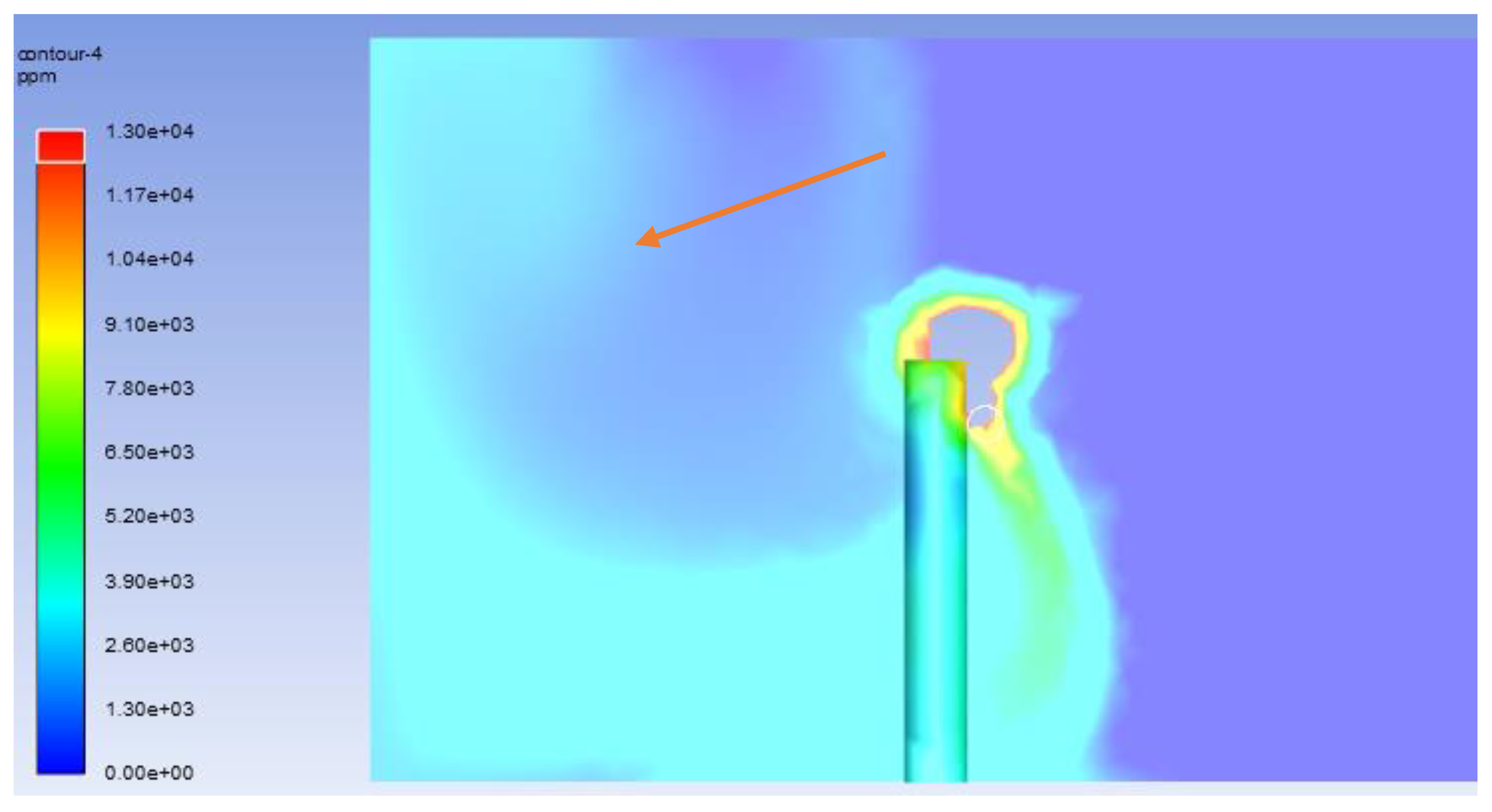
3.6. Modelling of Contaminant Concentration Using Species Transport
- ➢
- Mass fraction of carbon dioxide in the air within the model,
- ➢
- The exhalation velocity of human beings lies in the range of 0.6–1.4 m/s. So, on average, 1 m/s is taken.
4. Conclusions
- Position 1: One lamp positioned above the patient’s bed and the other laterally above the surgical staff.
- Position 2: Both lamps placed directly above the patient, over the head and abdominal region.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- American Society of Heating, Refrigerating and Air-Conditioning Engineers. ASHRAE Handbook—Fundamentals; ASHRAE: Atlanta, GA, USA, 2017. [Google Scholar]
- Geshwiler, M. HVAC Design Manual for Hospitals and Clinics; ASHRAE: Atlanta, GA, USA, 2003. [Google Scholar]
- Alkhalaf, M.; Ilinca, A.; Hayyani, M.Y.; Martini, F. Impact of Diffuser Location on Thermal Comfort Inside a Hospital Isolation Room. Fluids 2024, 8, 19. [Google Scholar] [CrossRef]
- Owens, C.D.; Stoessel, K. Surgical site infections: Epidemiology, microbiology and prevention. J. Hosp. Infect. 2008, 70 (Suppl. 2), 3–10. [Google Scholar] [CrossRef] [PubMed]
- Whitcomb, J.G.; Clapper, W.E. Ultraclean operating room. Am. J. Surg. 1966, 112, 681–685. [Google Scholar] [CrossRef] [PubMed]
- American Society of Heating, Refrigerating and Air-Conditioning Engineers. ASHRAE Handbook—Heating, Ventilating, and Air-Conditioning Systems and Equipment; ASHRAE: Atlanta, GA, USA, 2008. [Google Scholar]
- ISO 14644-1:2015; Cleanrooms and Associated Controlled Environments, Part 1: Classification of Air Cleanliness by Particle Concentration. International Organization for Standardization: Geneva, Switzerland, 2015; Volume 1.
- Ho, S.H.; Rosario, L.; Rahman, M.M. Three-dimensional analysis for hospital operating room thermal comfort and contaminant removal. Appl. Therm. Eng. 2009, 29, 2080–2092. [Google Scholar] [CrossRef]
- Agodi, A.; Auxilia, F.; Barchitta, M.; Cristina, M.L.; D’Alessandro, D.; Mura, I.; Nobile, M.; Pasquarella, C.; Avondo, S.; Bellocchi, P.; et al. Operating theatre ventilation systems and microbial air contamination in total joint replacement surgery: Results of the GISIO-ISChIA study. J. Hosp. Infect. 2015, 90, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Aganovic, A.; Cao, G.; Stenstad, L.-I.; Skogås, J.G. Impact of surgical lights on the velocity distribution and airborne contamination level in an operating room with laminar airflow system. Build. Environ. 2017, 126, 42–53. [Google Scholar] [CrossRef]
- Balocco, C.; Petrone, G.; Cammarata, G. Numerical Investigation of Different Airflow Schemes in a Real Operating Theatre. J. Biomed. Sci. Eng. 2015, 8, 73–89. [Google Scholar] [CrossRef]
- Chow, T.T.; Lin, Z.; Bai, W. The integrated effect of medical lamp position and diffuser discharge velocity on ultra-clean ventilation performance in an operating theatre. Indoor Built Environ. 2006, 15, 315–331. [Google Scholar] [CrossRef]
- Romano, F.; Milani, S.; Ricci, R.; Joppolo, C.M. Operating theatre ventilation systems and their performance in contamination control: ‘at rest’ and ‘in operation’ particle and microbial measurements made in an Italian large and multi-year inspection campaign. Int. J. Environ. Res. Public Health 2020, 17, 7275. [Google Scholar] [CrossRef] [PubMed]
- Kameel, R.; Khalil, E.E. Air flow regimes in operating theatres for energy efficient performance. In Proceedings of the 41st Aerospace Sciences Meeting and Exhibit, Reno, NV, USA, 6–9 January 2003; pp. 1–11. [Google Scholar] [CrossRef]
- Alsved, M.; Civilis, A.; Ekolind, P.; Tammelin, A.; Andersson, A.E.; Jakobsson, J.; Svensson, T.; Ramstorp, M.; Sadrizadeh, S.; Larsson, P.-A.; et al. Temperature-controlled airflow ventilation in operating rooms compared with laminar airflow and turbulent mixed airflow. J. Hosp. Infect. 2018, 98, 181–190. [Google Scholar] [CrossRef] [PubMed]
- James, M.; Khan, W.; Nannaparaju, M.; Bhamra, J.; Morgan-Jones, R. Current Evidence for the Use of Laminar Flow in Reducing Infection Rates in Total Joint Arthroplasty. Open Orthop. J. 2015, 9, 495–498. [Google Scholar] [CrossRef] [PubMed]
- Memarzadeh, F.; Manning, A.P. Comparison of operating room ventilation systems in the protection of the surgical site. ASHRAE Trans. 2002, 108 Pt 2, 3–15. [Google Scholar]
- Stevenson, T.C.; Jeter, S.M. Comparison of CFD simulations of hospital operating room air distribution with experimental PIV results. ASHRAE Trans. 2013, 119, 63–74. [Google Scholar]
- Lin, T.; Zargar, O.A.; Lin, K.-Y.; Juiña, O.; Sabusap, D.L.; Hu, S.-C.; Leggett, G. An experimental study of the flow characteristics and velocity fields in an operating room with laminar airflow ventilation. J. Build. Eng. 2020, 29, 101184. [Google Scholar] [CrossRef]
- Ouyang, X.; Wang, Q.; Li, X.; Zhang, T.; Rastogi, S. Laminar airflow ventilation systems in orthopaedic operating room do not prevent surgical site infections: A systematic review and meta-analysis. J. Orthop. Surg. Res. 2023, 18, 572. [Google Scholar] [CrossRef] [PubMed]









| ISO CLASS | 0.1 μm | 0.2 μm | 0.3 μm | 0.5 μm | 1.0 μm | 5.0 μm |
|---|---|---|---|---|---|---|
| 1 | 10 | 2 | - | - | - | - |
| 2 | 100 | 24 | 10 | 4 | - | - |
| 3 | 1000 | 237 | 102 | 35 | 8 | - |
| 4 | 10,000 | 2370 | 1020 | 352 | 83 | - |
| 5 | 100,000 | 23,700 | 10,200 | 3520 | 832 | 29 |
| 6 | 1,000,000 | 237,000 | 102,000 | 35,200 | 8320 | 293 |
| 7 | - | - | - | 352,000 | 83,200 | 2930 |
| 8 | - | - | - | 3,520,000 | 832,000 | 29,300 |
| 9 | - | - | - | 35,200,000 | 8,320,000 | 293,000 |
| No. | Set | Nodes | Elements |
|---|---|---|---|
| 1 | A | 92,776 | 488,978 |
| 2 | B | 91,138 | 479,564 |
| 3 | C | 89,395 | 470,395 |
| 4 | D | 87,291 | 465,236 |
| Items | Dimensions | Heat Dissipation |
|---|---|---|
| Operating table | 1.9 × 0.6 × 0.85 m | None |
| Surgical lights (1) | 0.5 m in diameter | 150 W each |
| Surgical staff (2) | 0.3 × 0.25 × 1.7 m high | 100 W each |
| Anaesthesia machine | 0.5 × 0.3 × 1.2 m high | 200 W |
| Patient | 0.3 × 0.25 × 1.7 m high | 46 W |
| Back table | 0.5 × 0.3 × 1.1 m high | None |
| No. | Instrument Used | Specifications | |
|---|---|---|---|
| 1 | Vane anemometer Testo-400 | measuring range +0.3 to +35 m/s, (−20 to +70 °C) | |
| 2 | Testo-480 | Accuracy (Temp) | ±0.2 °C (with high-precision probe) |
| Accuracy (Humidity) | ±1.0% RH | ||
| CO2 Measurement Range | 0 to 10,000 ppm | ||
| Flow Velocity | 0 to 100 m/s (with pitot tube/anemometer probe) | ||
| 3 | IAQ Probe | Temperature—NTC | |
| Measuring range—0 to +50 °C | |||
| Accuracy—±0.5 °C | |||
| Resolution—0.1 °C | |||
| Humidity—Capacitive | |||
| Measuring range | |||
| 0 to +100% RH | |||
| Accuracy | |||
| ±(1.8% RH + 0.7% of mv) | |||
| Resolution | |||
| 0.1% RH | |||
| Absolute Pressure | |||
| Measuring range | |||
| +700 to +1100 hPa | |||
| Accuracy ±3.0 hPa | |||
| Resolution | |||
| 0.1 hPa | |||
| Ambient CO2 | |||
| Measuring range | |||
| 0 to +10,000 ppm | |||
| Accuracy | |||
| ±(75 ppm + 3% of mv) | |||
| ±(150 ppm + 5% of mv) | |||
| 0 to +5000 ppm | |||
| 5001 to +10,000 ppm | |||
| Resolution 1 ppm | |||
| 4 | Black globe thermometer | Thermocouple type K | |
| Measuring range: 0 to +120 °C | |||
| 5 | Mini Laser Aerosol Spectrometer (Mini-LAS) 11-R | Count range: | |
| 1 to 2,000,000 particles/L | |||
| Particle mass: | |||
| From 0.1 μg/m3 to 100 mg/m3 | |||
| Size channels: 31 channels | |||
| Time Stamp | Runtime (s) | hPa Abs | hPa Diff | Velocity | Temp (°C) | RH (%) | hPa Abs | CO2 (ppm) | g/m3 |
|---|---|---|---|---|---|---|---|---|---|
| 19:55:11 | 0 | 1005.7 | −0.006 | 1.0 | 22.9 | 80.5 | 1005.3 | 770 | 16.5 |
| 19:55:21 | 10 | 1005.7 | −0.006 | 1.0 | 23.0 | 80.8 | 1005.3 | 774 | 16.6 |
| 19:55:31 | 20 | 1005.7 | −0.006 | 1.0 | 23.0 | 80.4 | 1005.3 | 778 | 16.5 |
| 19:55:41 | 30 | 1005.7 | −0.006 | 1.0 | 23.0 | 80.5 | 1005.3 | 792 | 16.6 |
| 19:55:51 | 40 | 1005.7 | −0.006 | 1.0 | 23.0 | 80.1 | 1005.3 | 798 | 16.5 |
| 19:56:01 | 50 | 1005.7 | −0.006 | 1.0 | 23.0 | 80.1 | 1005.3 | 791 | 16.5 |
| 19:56:11 | 60 | 1005.7 | −0.006 | 1.0 | 23.0 | 80.2 | 1005.3 | 781 | 16.5 |
| 19:56:21 | 70 | 1005.7 | −0.006 | 1.0 | 23.1 | 80.1 | 1005.3 | 783 | 16.6 |
| 19:56:31 | 80 | 1005.8 | −0.006 | 1.0 | 23.1 | 80.2 | 1005.3 | 786 | 16.6 |
| 19:56:41 | 90 | 1005.8 | −0.006 | 1.0 | 23.1 | 80.4 | 1005.3 | 792 | 16.6 |
| 19:56:51 | 100 | 1005.8 | −0.006 | 1.0 | 23.1 | 80.2 | 1005.3 | 785 | 16.6 |
| Time Stamp | Runtime (s) | hPa Abs | hPa Diff | Velocity | Temp (°C) | RH (%) | hPa Abs | CO2 (ppm) | g/m3 |
|---|---|---|---|---|---|---|---|---|---|
| 20:10:23 | −1 | 1006.0 | −0.005 | 0.9 | 23.4 | 78.1 | 1005.5 | 755 | 16.5 |
| 20:10:33 | 9 | 1006.0 | −0.005 | 0.9 | 23.3 | 78.4 | 1005.5 | 756 | 16.5 |
| 20:10:43 | 19 | 1006.0 | −0.005 | 0.9 | 23.4 | 78.8 | 1005.6 | 765 | 16.6 |
| 20:10:53 | 29 | 1006.0 | −0.005 | 0.9 | 23.5 | 78.4 | 1005.6 | 761 | 16.6 |
| 20:11:03 | 39 | 1006.0 | −0.005 | 0.9 | 23.5 | 78.3 | 1005.6 | 770 | 16.5 |
| 20:11:13 | 49 | 1006.0 | −0.005 | 0.9 | 23.5 | 78.2 | 1005.5 | 773 | 16.5 |
| 20:11:23 | 59 | 1006.0 | −0.005 | 0.9 | 23.5 | 78.4 | 1005.6 | 770 | 16.6 |
| 20:11:33 | 69 | 1006.0 | −0.005 | 0.9 | 23.5 | 78.4 | 1005.5 | 770 | 16.6 |
| 20:11:43 | 79 | 1006.0 | −0.005 | 0.9 | 23.5 | 78.3 | 1005.6 | 770 | 16.6 |
| 20:11:53 | 89 | 1006.0 | −0.005 | 0.9 | 23.5 | 78.2 | 1005.6 | 774 | 16.6 |
| 20:12:03 | 99 | 1006.0 | −0.005 | 0.9 | 23.5 | 78.1 | 1005.6 | 768 | 16.6 |
| 0.25 µm | 0.28 µm | 0.30 µm | 0.35 µm | 0.40 µm | 0.45 µm | 0.50 µm |
|---|---|---|---|---|---|---|
| 200 | 100 | 200 | 50 | 50 | 0 | 0 |
| 100 | 200 | 0 | 50 | 0 | 50 | 0 |
| 200 | 0 | 150 | 0 | 0 | 0 | 50 |
| 150 | 0 | 50 | 0 | 0 | 0 | 100 |
| 150 | 150 | 200 | 100 | 50 | 0 | 0 |
| 250 | 200 | 0 | 0 | 100 | 0 | 0 |
| 250 | 300 | 300 | 150 | 50 | 100 | 0 |
| 200 | 0 | 100 | 50 | 0 | 0 | 0 |
| 150 | 50 | 250 | 250 | 50 | 0 | 0 |
| 50 | 0 | 0 | 100 | 0 | 0 | 0 |
| 0.25 µm | 0.28 µm | 0.30 µm | 0.35 µm | 0.40 µm | 0.45 µm | 0.50 µm |
|---|---|---|---|---|---|---|
| 700 | 150 | 300 | 100 | 0 | 0 | 0 |
| 150 | 200 | 100 | 0 | 0 | 0 | 0 |
| 200 | 150 | 100 | 150 | 0 | 0 | 0 |
| 250 | 150 | 250 | 250 | 50 | 0 | 0 |
| 200 | 150 | 200 | 200 | 50 | 0 | 0 |
| 200 | 150 | 150 | 50 | 0 | 0 | 0 |
| 150 | 150 | 100 | 150 | 0 | 50 | 0 |
| 200 | 200 | 100 | 0 | 0 | 50 | 0 |
| 350 | 150 | 200 | 200 | 50 | 0 | 100 |
| 250 | 150 | 200 | 50 | 0 | 50 | 0 |
| 0.25 µm | 0.28 µm | 0.30 µm | 0.35 µm | 0.40 µm | 0.45 µm | 0.50 µm |
|---|---|---|---|---|---|---|
| 2850 | 1550 | 950 | 1000 | 350 | 100 | 50 |
| 2000 | 1650 | 1550 | 1500 | 100 | 50 | 50 |
| 2000 | 1850 | 1600 | 1150 | 250 | 300 | 100 |
| 2050 | 1450 | 1200 | 1050 | 300 | 50 | 50 |
| 2750 | 1950 | 1800 | 1200 | 400 | 150 | 150 |
| 2050 | 1500 | 1450 | 1000 | 300 | 50 | 150 |
| 1750 | 1350 | 950 | 1200 | 200 | 100 | 0 |
| 2050 | 1550 | 2000 | 1250 | 150 | 200 | 250 |
| 1650 | 1050 | 1450 | 1200 | 350 | 300 | 50 |
| 2150 | 1400 | 950 | 1100 | 500 | 100 | 100 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valsala Krishnankutty, V.; Muraleedharan, C.; Palatel, A. Analysis of Disruption of Airflow and Particle Distribution by Surgical Personnel and Lighting Fixture in Operating Rooms. Fluids 2025, 10, 225. https://doi.org/10.3390/fluids10090225
Valsala Krishnankutty V, Muraleedharan C, Palatel A. Analysis of Disruption of Airflow and Particle Distribution by Surgical Personnel and Lighting Fixture in Operating Rooms. Fluids. 2025; 10(9):225. https://doi.org/10.3390/fluids10090225
Chicago/Turabian StyleValsala Krishnankutty, Vikas, Chandrasekharan Muraleedharan, and Arun Palatel. 2025. "Analysis of Disruption of Airflow and Particle Distribution by Surgical Personnel and Lighting Fixture in Operating Rooms" Fluids 10, no. 9: 225. https://doi.org/10.3390/fluids10090225
APA StyleValsala Krishnankutty, V., Muraleedharan, C., & Palatel, A. (2025). Analysis of Disruption of Airflow and Particle Distribution by Surgical Personnel and Lighting Fixture in Operating Rooms. Fluids, 10(9), 225. https://doi.org/10.3390/fluids10090225





