Synthesis, Characterization, and Properties of Polyvinyl Alcohol/Jackfruit Peel Carboxymethylcellulose/Graphene Oxide/Kaolin Composite Hydrogels
Abstract
1. Introduction
2. Results and Discussion
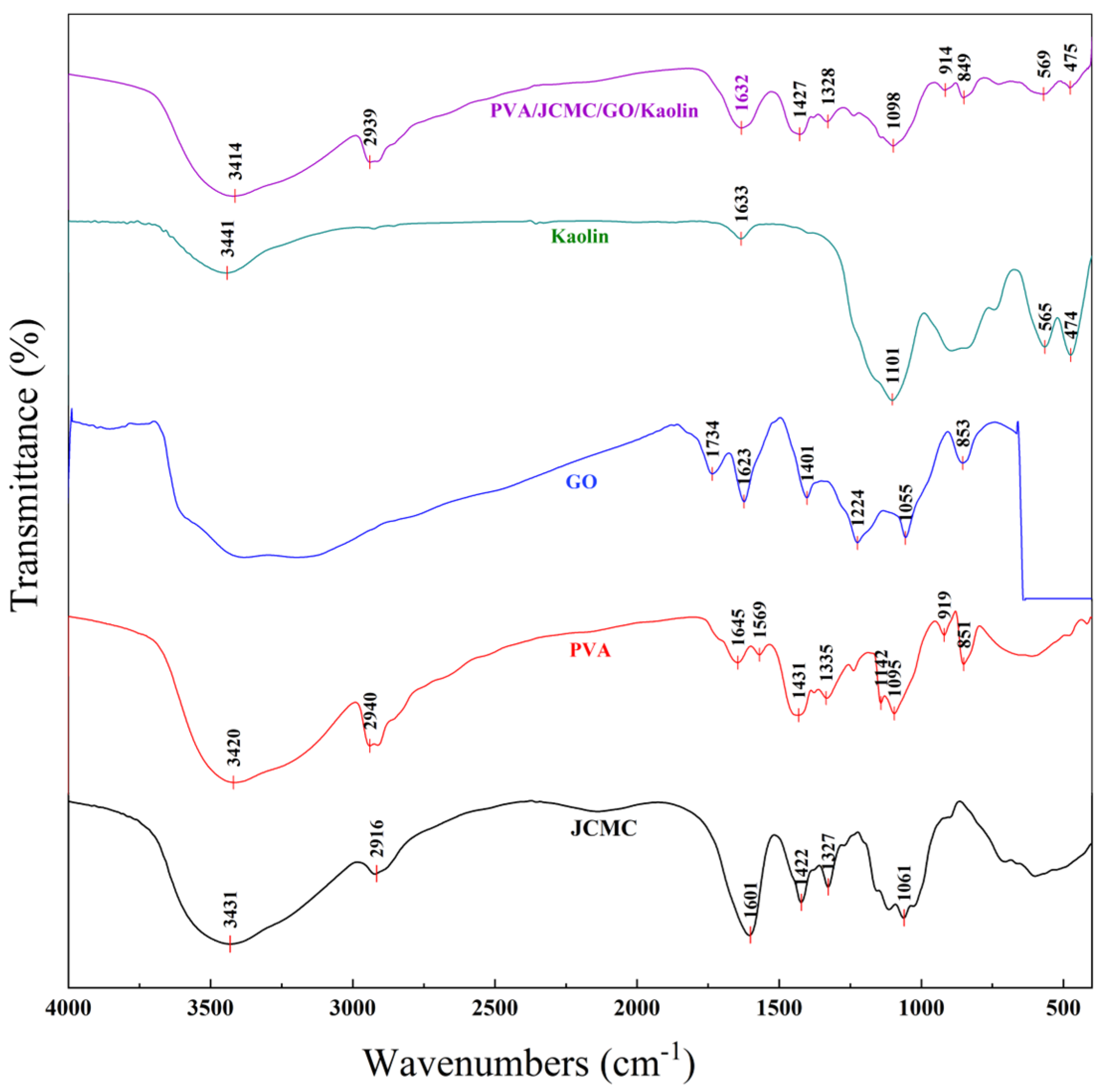
2.1. FTIR Spectral Analysis of PVA/JCMC/GO/Kaolin Composite Hydrogels
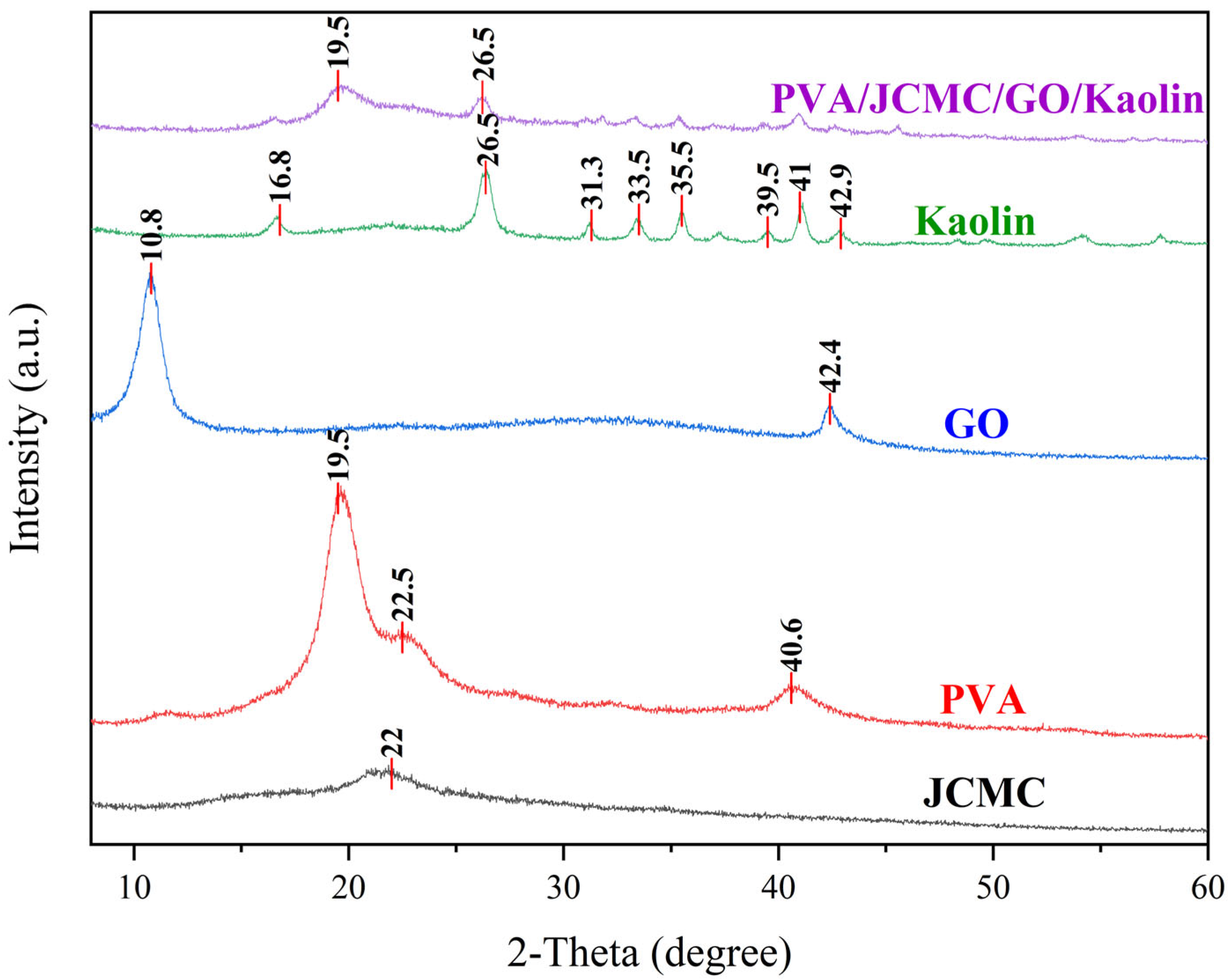
2.2. XRD Analysis of Composite Hydrogel Structure
2.3. Morphological Characterization of Composite Hydrogels
2.4. Adsorption Properties of CR
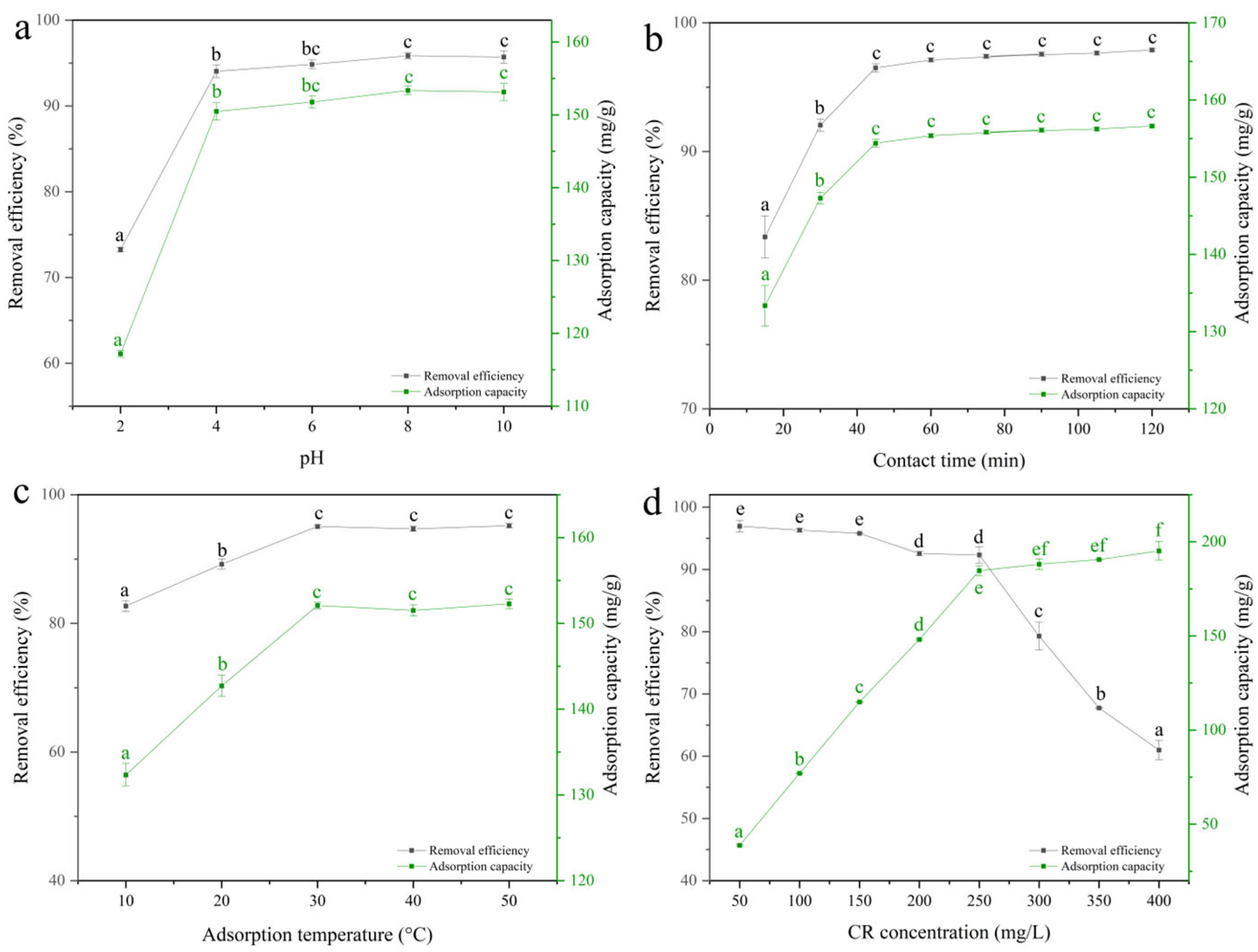
2.4.1. Optimization of Adsorption Parameters
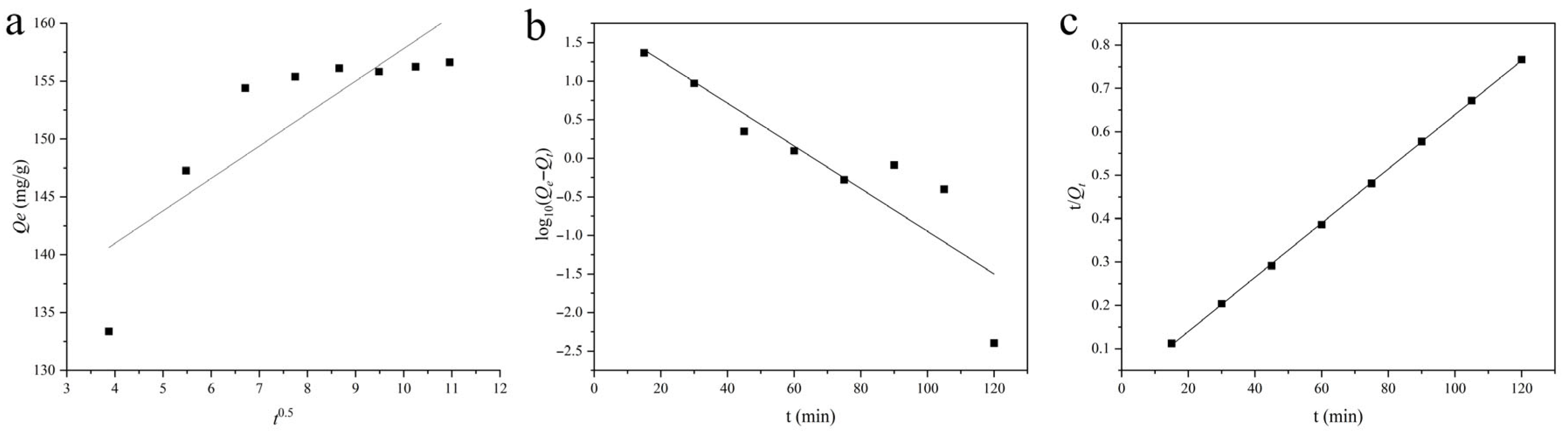
2.4.2. Kinetic Analysis of Adsorption Behavior
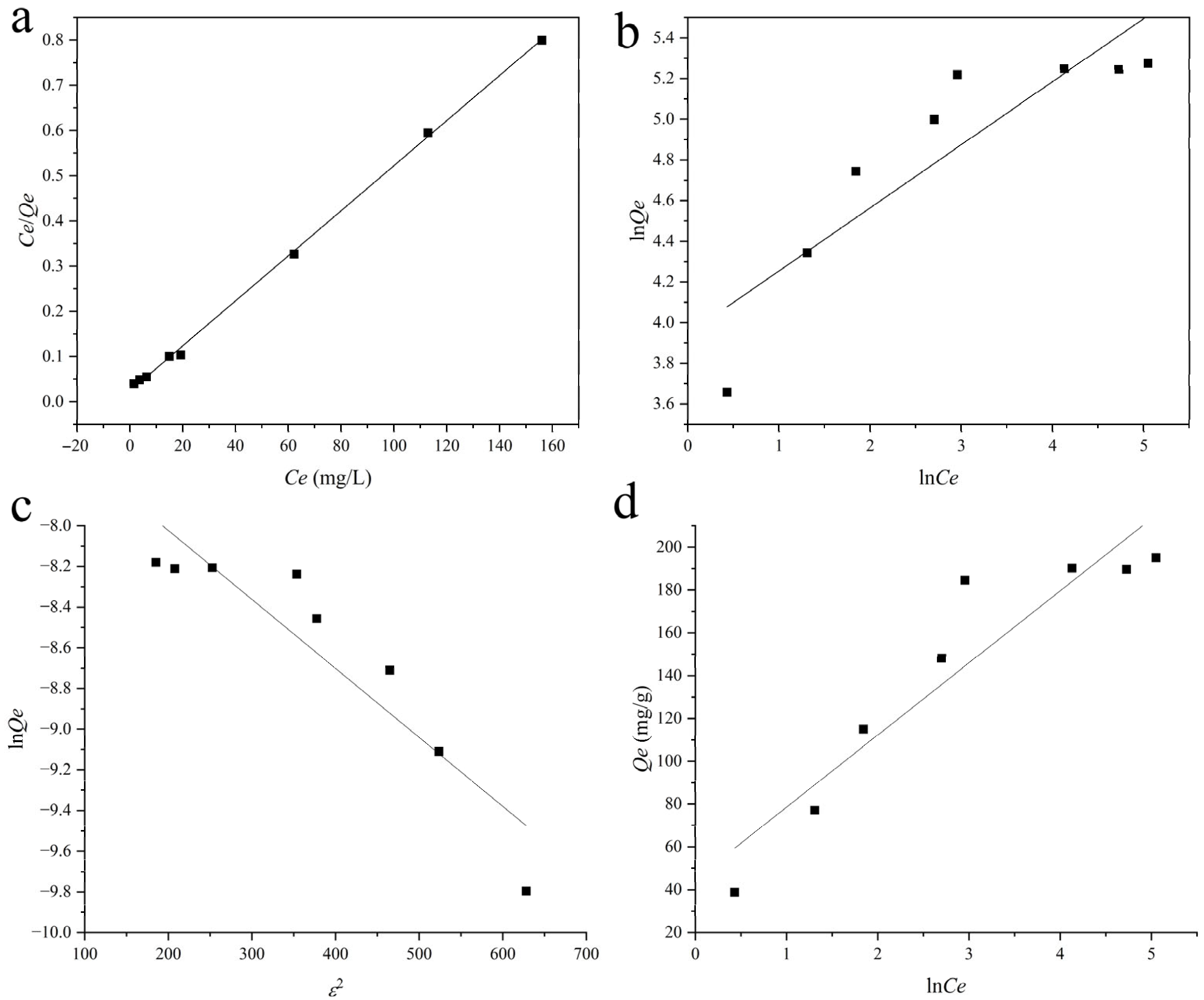
2.4.3. Adsorption Isotherm Analysis
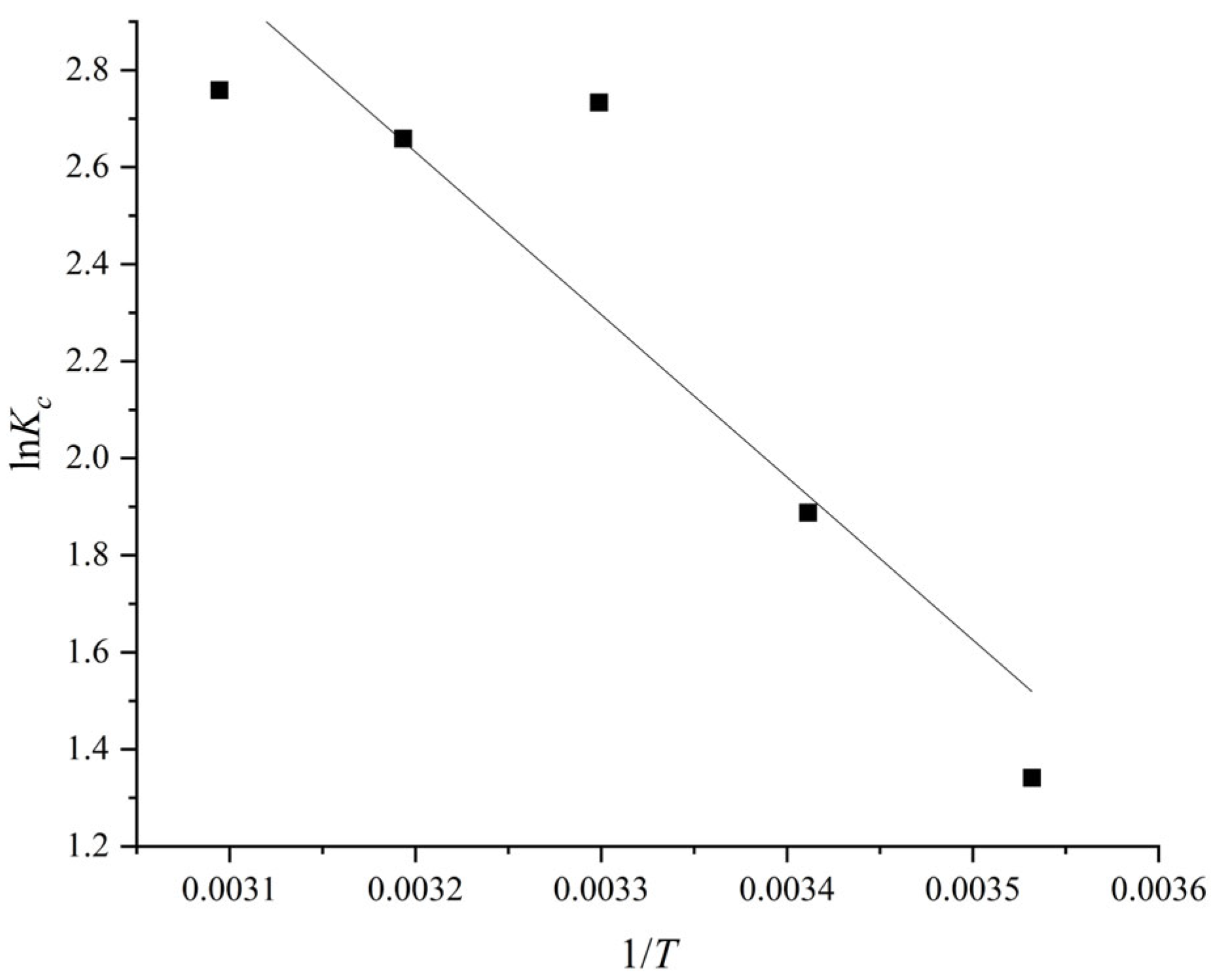
2.4.4. Thermodynamics Analysis
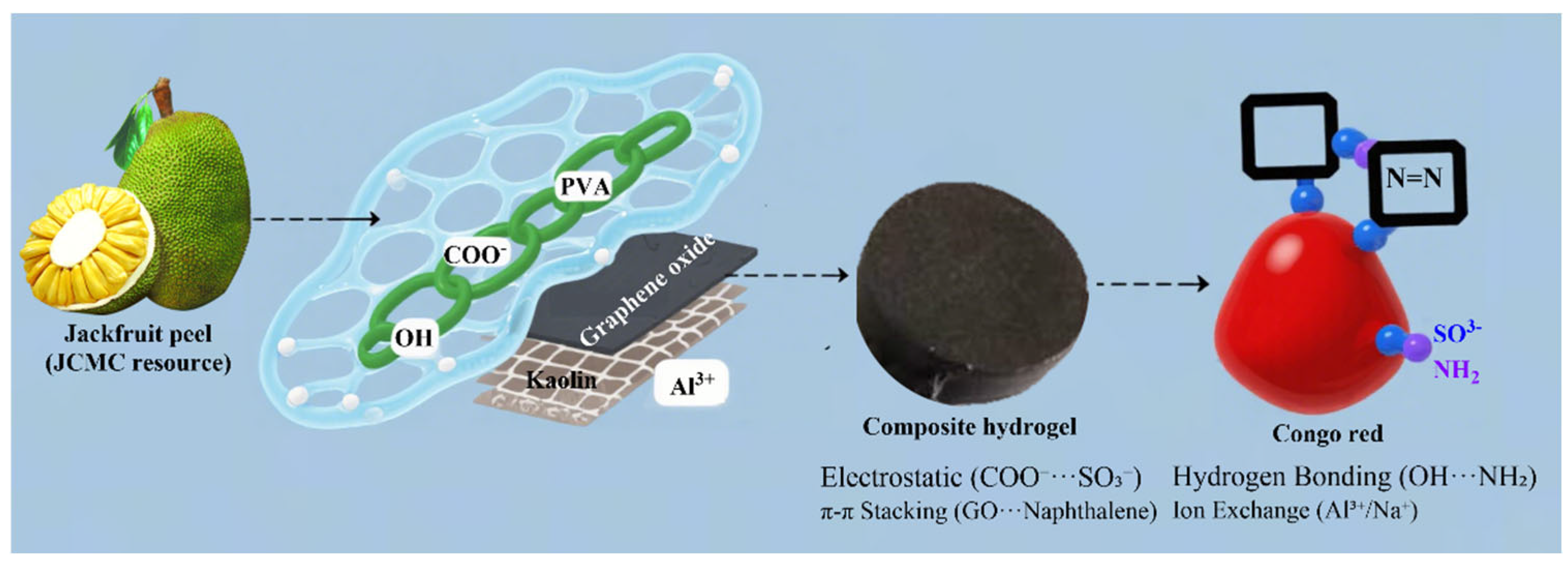
2.5. Adsorption Mechanism
3. Conclusions
4. Materials and Methods
4.1. Materials and Reagents
4.2. Synthesis of Jackfruit Peel Carboxymethyl Cellulose (JCMC)
4.3. Preparation of PVA/JCMC/GO/Kaolin Composite Hydrogels
4.4. Characterization Techniques
4.4.1. Fourier Transform Infrared Spectroscopy (FTIR)
4.4.2. X-Ray Diffraction (XRD)
4.4.3. Scanning Electron Microscopy (SEM)
4.5. Adsorption Performance Evaluation
4.6. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jin, L.; Qian, T.; Cheng, Y.; Qi, D.; Xin, J.H.; Xia, G.; Cui, Z. Synthesis, recovery, and application of environmentally friendly disperse dyes with high light fastness. Text Res. J. 2024, 94, 566–582. [Google Scholar] [CrossRef]
- Uduma, R.C.; Oguzie, K.L.; Chijioke, C.F.; Ogbulie, T.E.; Oguzie, E.E. Bioelectrochemical technologies for simultaneous treatment of dye wastewater and electricity generation: A review. Int. J. Environ. Sci. Technol. 2023, 20, 10415–10434. [Google Scholar] [CrossRef]
- Hu, C.; Zhang, Y.; Zhou, Y.; Liu, Z.F.; Feng, X.S. Unsymmetrical dimethylhydrazine and related compounds in the environment: Recent updates on pretreatment, analysis, and removal techniques. J. Hazard Mater. 2022, 432, 128708. [Google Scholar] [CrossRef] [PubMed]
- Karaday, M.; Güllüce, E.; Ahin, Y.G.; Olak, L.; Karaday, G.K.E.; Aksu, E.; Güllüce, M. Molecular docking assisted toxicity assessment of congo red and detoxification potential of Fraxinus excelsior L. (Oleaceae) biosorbent application. Biomass Convers. Biorefin. 2025, 25, 6842–6849. [Google Scholar] [CrossRef]
- Zinatloo-Ajabshir, S.; Rakhshani, S.; Mehrabadi, Z.; Farsadrooh, M.; Feizi-Dehnayebi, M.; Rakhshani, S.; Duek, M.; Eigner, V.; Rtimi, S.; Aminabhavi, T.M. Novel rod-like [Cu(phen)2(OAc)]·PF6 complex for high-performance visible-light-driven photocatalytic degradation of hazardous organic dyes: DFT approach, Hirshfeld and fingerprint plot analysis. J. Environ. Manag. 2025, 350, 119545. [Google Scholar] [CrossRef]
- Song, Y.; Wang, L.; Qiang, X.; Gu, W.; Ma, Z.; Wang, G. An overview of biological mechanisms and strategies for treating wastewater from printing and dyeing processes. J. Water Process Eng. 2023, 55, 104242. [Google Scholar] [CrossRef]
- Srivastava, A.; Rani, R.M.; Patle, D.S.; Kumar, S. Emerging bioremediation technologies for the treatment of textile wastewater containing synthetic dyes: A comprehensive review. J. Chem. Technol. Biotechnol. 2022, 97, 26–41. [Google Scholar] [CrossRef]
- Dai, H.; Huang, Y.; Zhang, Y.; Zhang, H.; Huang, H. Green and facile fabrication of pineapple peel cellulose/magnetic diatomite hydrogels in ionic liquid for methylene blue adsorption. Cellulose 2019, 26, 3825–3844. [Google Scholar] [CrossRef]
- Eftekhari-Sis, B.; Akbari, A.; Motlagh, P.Y.; Bahrami, Z.; Arsalani, N. Dye adsorption on cubic polyhedral oligomeric silsesquioxane-based poly(acrylamide-co-itaconic acid) hybrid nanocomposites: Kinetic, thermodynamic and isotherms studies. J. Inorg. Organomet. Polym. Mater. 2018, 28, 1728–1738. [Google Scholar] [CrossRef]
- Anastopoulos, I.; Ighalo, J.O.; Adaobi Igwegbe, C.; Giannakoudakis, D.A.; Triantafyllidis, K.S.; Pashalidis, I.; Kalderis, D. Sunflower-biomass derived adsorbents for toxic/heavy metals removal from (waste) water. J. Mol. Liq. 2021, 342, 117540. [Google Scholar] [CrossRef]
- Bobde, P.V.; Sharma, A.K.; Kumar, R.; Pandey, J.K.; Wadhwa, S. Recent advances on sustainable removal of emerging contaminants from water by bio-based adsorbents. New J. Chem. 2023, 47, 17626–17644. [Google Scholar] [CrossRef]
- Sulyman, M. Agricultural by-products/waste as dye and metal ions adsorbents: A review. Int. J. Eng. Sci. 2024, 6, 1–20. [Google Scholar]
- Huang, R.; Xu, Y.; Kuznetsov, B.N.; Sun, M.; Zhou, X.; Luo, J.; Jiang, K. Enhanced hybrid hydrogel based on wheat husk lignin-rich nanocellulose for effective dye removal. Front. Bioeng. Biotechnol. 2023, 11, 1160698. [Google Scholar] [CrossRef] [PubMed]
- Tavakoli, S.; Klar, A.S. Advanced hydrogels as wound dressings. Biomolecules 2020, 10, 1169. [Google Scholar] [CrossRef]
- Xiao, Z.; Li, Q.; Liu, H.; Zhao, Q.; Niu, Y.; Zhao, D. Adhesion mechanism and application progress of hydrogels. Eur. Polym. J. 2022, 173, 111277. [Google Scholar] [CrossRef]
- Ahmaruzzaman, M.; Roy, P.; Bonilla-Petriciolet, A.; Badawi, M.; Ganachari, S.V.; Shetti, N.P.; Aminabhavi, T.M. Polymeric hydrogels-based materials for wastewater treatment. Chemosphere 2023, 331, 138743. [Google Scholar] [CrossRef]
- Bao, Y.; Ganesh, K.; Xiao, H.U.; Zhan, S.; Zhang, H. Experimental design for the preparation of low-cost carboxymethylcellulose hydrogel and its adsorption performance. Exp. Technol. Manag. 2024, 41, 81–89. [Google Scholar] [CrossRef]
- Cao, M.; Peng, Q.; Wang, Y.; Luo, G.; Feng, L.; Zhao, S.; Yuan, Y.; Wang, N. High-efficiency uranium extraction from seawater by low-cost natural protein hydrogel. Int. J. Biol. Macromol. 2023, 242, 124792. [Google Scholar] [CrossRef]
- Huang, H.; Yang, Q.; Huang, C.; Zhang, L. Facile and low-cost fabrication of composite hydrogels to improve adsorption of copper ions. Environ. Technol. Innov. 2022, 27, 102427. [Google Scholar] [CrossRef]
- Ran, S.; Xue, L.; Wei, X.; Huang, J.; Yan, X.; He, T.; Tang, Z.; Zhang, H.; Gu, M. Recent advances in injectable hydrogel therapies for periodontitis. J. Mater. Chem. B 2024, 12, 65–632. [Google Scholar] [CrossRef]
- Yadav, P.; Singh, S.; Jaiswal, S.; Kumar, R. Synthetic and natural polymer hydrogels: A review of 3d spheroids and drug delivery. Int. J. Biol. Macromol. 2024, 280, 136126. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Fu, S.; Jin, K.; Cheng, Y.; Li, Y.; Zhao, Y.; Liu, R.; Tian, Y. Advances in polysaccharide-based conductive hydrogel for flexible electronics. Carbohydr. Polym. 2025, 348, 122836. [Google Scholar] [CrossRef] [PubMed]
- Shukla, A.; Syaifie, P.H.; Rochman, N.T.; Jaya Syaifullah, S.; Jauhar, M.M.; Mardliyati, E. A recent study of natural hydrogels: Improving mechanical properties for biomedical applications. Biomed. Mater. 2025, 20, 22010. [Google Scholar] [CrossRef] [PubMed]
- Jasni, A.H.; Azmi, A.S.; Puad, N.I.P.M.; Ali, F.; Nor, Y.A. Sustainable synthesis of cellulose-derived hydrogels for tissue engineering. In Biomaterials, Bioengineering and Sustainability; Springer: Cham, Switzerland, 2025; pp. 343–372. [Google Scholar] [CrossRef]
- Legrand, G.; Baeza, G.P.; Manneville, S.; Divoux, T. Rheological properties of acid-induced carboxymethylcellulose hydrogels. Cellulose 2025, 32, 903–917. [Google Scholar] [CrossRef]
- Zhou, Y.; Li, T.; Shen, J.; Meng, Y.; Tong, S.; Guan, Q.; Xia, X. Core-shell structured magnetic carboxymethyl cellulose-based hydrogel nanosorbents for effective adsorption of methylene blue from aqueous solution. Polymers 2021, 13, 3054. [Google Scholar] [CrossRef]
- Srivastava, R.; Singh, A. Jackfruit (Artocarpus heterophyllus Lam) biggest fruit with high nutritional and pharmacological values: A review. Int. J. Curr. Microbiol. App. Sci. 2020, 9, 764–774. [Google Scholar] [CrossRef]
- Novrianti; Novriansyah, A.; Khalid, I.; Amani, Z.D.; Riswandi, H.; Igarashi, T.; Tabelin, C.; Pasang, T.; Ugurly, O.F.; Rizkianto, Y.; et al. Rheology analysis of drilling mud with additive carboxy methyl cellulose from jackfruit’s peel: Experimental study. AIP Conf. Proc. 2023, 2598, 030023. [Google Scholar] [CrossRef]
- Dai, H.; Zhang, Y.; Ma, L.; Zhang, H.; Huang, H. Synthesis and response of pineapple peel carboxymethyl cellulose-g-poly (acrylic acid-co-acrylamide)/graphene oxide hydrogels. Carbohydr. Polym. 2019, 215, 366–376. [Google Scholar] [CrossRef]
- Dai, H.; Huang, H. Synthesis, characterization and properties of pineapple peel cellulose-g-acrylic acid hydrogel loaded with kaolin and sepia ink. Cellulose 2017, 24, 69–84. [Google Scholar] [CrossRef]
- Muslim, W.A.; Al-Nasri, S.K.; Albayati, T.M. Evaluation of bentonite, attapulgite, and kaolinite as eco-friendly adsorbents in the treatment of real radioactive wastewater containing cs-137. Prog. Nucl. Energy 2023, 162, 104730. [Google Scholar] [CrossRef]
- Gang, W.; Zheng-ye, G.; Shui-shui, G.; Shan-yi, G.; Fu-you, K.; Hong-yao, X. Determination of the oxidizability on the surface of the graphene oxide layer by infrared spectroscopy. Spectrosc. Spect. Anal. 2020, 40, 1722–1727. [Google Scholar]
- Ogunleye, D.T.; Akpotu, S.O.; Moodley, B. Adsorption of sulfamethoxazole and reactive blue 19 using graphene oxide modified with imidazolium based ionic liquid. Environ. Technol. Innov. 2020, 17, 100616. [Google Scholar] [CrossRef]
- Dai, H.; Huang, Y.; Huang, H. Eco-friendly polyvinyl alcohol/carboxymethyl cellulose hydrogels reinforced with graphene oxide and bentonite for enhanced adsorption of methylene blue. Carbohydr. Polym. 2018, 185, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Dai, H.; Huang, H. Enhanced swelling and responsive properties of pineapple peel carboxymethyl cellulose-g-poly(acrylic acid-co-acrylamide) superabsorbent hydrogel by the introduction of carclazyte. J. Agric. Food Chem. 2017, 65, 565–574. [Google Scholar] [CrossRef] [PubMed]
- Adinugraha, M.P.; Marseno, D.W.; Haryadi. Synthesis and characterization of sodium carboxymethylcellulose from cavendish banana pseudo stem (Musa cavendishii LAMBERT). Carbohydr. Polym. 2005, 62, 164–169. [Google Scholar] [CrossRef]
- Li, X.; Wang, D.; Liu, Q.; Komarneni, S. A comparative study of synthetic tubular kaolinite nanoscrolls and natural halloysite nanotubes. Appl. Clay. Sci. 2019, 168, 421–427. [Google Scholar] [CrossRef]
- Liu, D.; Bian, Q.; Li, Y.; Wang, Y.; Xiang, A.; Tian, H. Effect of oxidation degrees of graphene oxide on the structure and properties of poly(vinyl alcohol) composite films. Compos. Sci. Technol. 2016, 129, 146–152. [Google Scholar] [CrossRef]
- Rasyida, A.; Silaen, Y.M.T.; Wicaksono, S.T.; Ardyananta, H.; Nurdiansah, H.; Halimah, S. Preliminary characterization of hydrogel composite alginate/PVA/r-GO as an injectable materials for medical applications. Mater. Sci. Forum 2019, 964, 161–167. [Google Scholar] [CrossRef]
- Shi, Y.; Xiong, D.; Li, J.; Wang, N. The water-locking and cross-linking effects of graphene oxide on the load-bearing capacity of poly(vinyl alcohol) hydrogel. RSC Adv. 2016, 6, 82467–82477. [Google Scholar] [CrossRef]
- Zheng, C.; Mao, Y.; Markland, T.E.; Boxer, S.G. Beyond the vibrational stark effect: Unraveling the large redshifts of alkyne C-H bond in solvation environments. J. Am. Chem. Soc. 2025, 147, 6227–6235. [Google Scholar] [CrossRef]
- Stan, M.S.; Cinteza, L.O.; Petrescu, L.; Mernea, M.A.; Calborean, O.; Mihailescu, D.F.; Sima, C.; Dinischiotu, A. Dynamic analysis of the interactions between Si/SiO2 quantum dots and biomolecules for improving applications based on nano-bio interfaces. Sci. Rep. 2018, 8, 5289. [Google Scholar] [CrossRef]
- Dai, H.; Ou, S.; Liu, Z.; Huang, H. Pineapple peel carboxymethyl cellulose/polyvinyl alcohol/mesoporous silica SBA-15 hydrogel composites for papain immobilization. Carbohydr. Polym. 2017, 169, 504–514. [Google Scholar] [CrossRef] [PubMed]
- Guan, Y.; Bian, J.; Peng, F.; Zhang, X.; Sun, R. High strength of hemicelluloses based hydrogels by freeze/thaw technique. Carbohydr. Polym. 2014, 101, 272–280. [Google Scholar] [CrossRef] [PubMed]
- Sahraei, R.; Sekhavat Pour, Z.; Ghaemy, M. Novel magnetic bio-sorbent hydrogel beads based on modified gum tragacanth/graphene oxide: Removal of heavy metals and dyes from water. J. Clean. Prod. 2017, 142, 2973–2984. [Google Scholar] [CrossRef]
- Güneyisi, E.; Lu, M.G.; Zturan, T.; Mermerda, K.M. Microstructural properties and pozzolanic activity of calcined kaolins as supplementary cementing materials. Can. J. Civ. Eng. 2012, 39, 1274–1284. [Google Scholar] [CrossRef]
- Wang, X.; Zhao, Z.; Shu, Z.; Chen, Y.; Zhou, J.; Li, T.; Wang, W.; Tan, Y.; Sun, N. One-pot synthesis of metakaolin/g-C3N4 composite for improved visible-light photocatalytic H2 evolution. Appl. Clay. Sci. 2018, 166, 80–87. [Google Scholar] [CrossRef]
- Tian, T.; Yi-le, F.; Li, G.; Yi-yuan, W.; Jun, Z. Preparation of sodium alginate-carboxymethyl cellulose-graphene oxide composite aerogel for adsorption of pb(II) ion. Acta Mater. Compos. Sinica 2023, 40, 5792–5802. [Google Scholar] [CrossRef]
- Yang, M.; Shi, J.; Xia, Y. Effect of SiO2, PVA and glycerol concentrations on chemical and mechanical properties of alginate-based films. Int. J. Biol. Macromol. 2018, 107, 2686–2694. [Google Scholar] [CrossRef]
- Ge, M.; Xi, Z.; Zhu, C.; Liang, G.; Hu, G.; Jamal, L.; SM, J.A. Preparation and characterization of magadiite–magnetite nanocomposite with its sorption performance analyses on removal of methylene blue from aqueous solutions. Polymers 2019, 11, 607. [Google Scholar] [CrossRef]
- Sudarsan, S.; Murugesan, G.; Varadavenkatesan, T.; Vinayagam, R.; Selvaraj, R. Efficient adsorptive removal of congo red dye using activated carbon derived from spathodea campanulata flowers. Sci. Rep. 2025, 15, 1831. [Google Scholar] [CrossRef]
- Gamboa, D.M.P.; Abatal, M.; Lima, E.; Franseschi, F.A.; Ucán, C.A.; Tariq, R.; Elías, M.A.R.; Vargas, J. Sorption behavior of azo dye congo red onto activated biochar from haematoxylum campechianum waste: Gradient boosting machine learning-assisted bayesian optimization for improved adsorption process. Int. J. Mol. Sci. 2024, 25, 4771. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Liang, J.; Wu, Q.; Hu, X. Synthesis, characterization and dye adsorption properties of modified jackfruit peel cellulose adsorbent. J. Yunnan Univ. 2022, 44, 328–337. [Google Scholar] [CrossRef]
- Zhu, F.; Huang, H.; Yang, Z.; Xu, M. Dual-responsive copolymer hydrogel as broad-spectrum adsorbents for metal ions. Polym. Test. 2019, 77, 105887. [Google Scholar] [CrossRef]
- Da Silva, A.C.; Wang, J.; Minev, I.R. Electro-assisted printing of soft hydrogels via controlled electrochemical reactions. Nat. Commun. 2022, 13, 1353. [Google Scholar] [CrossRef] [PubMed]
- Sriram, G.; Uthappa, U.T.; Losic, D.; Kigga, M.; Kurkuri, M.D. Mg–al-layered double hydroxide (LDH) modified diatoms for highly efficient removal of congo red from aqueous solution. Appl. Sci. 2020, 10, 2285. [Google Scholar] [CrossRef]
- Manjuladevi, M.; Anitha, R.; Manonmani, S. Kinetic study on adsorption of Cr(VI), Ni(II), Cd(II) and Pb(II) ions from aqueous solutions using activated carbon prepared from Cucumis melo peel. Appl. Water Sci. 2018, 8, 36. [Google Scholar] [CrossRef]
- Uysal, E.; Dursun, H.N.; Güler, R.; Takmaz, U.; Küt, A.; Çeri, M.; Uysal, E.C.; Gürmen, S. Waste refractory brick material added chitosan/oxidized pullulan complex gel production and removal of heavy metals from waste water. Sci. Rep. 2024, 14, 26216–26229. [Google Scholar] [CrossRef]
- Su, T.; Zhang, X.; Wang, Z.; Guo, Y.; Wei, X.; Xu, B.; Xia, H.; Yang, W.; Xu, H. Cellulose nanocrystal-based polymer hydrogel embedded with iron oxide nanorods for efficient arsenic removal. Carbohydr. Polym. 2024, 331, 121855. [Google Scholar] [CrossRef]
- Sayed, M.A.; Abdelhameed, R.M.; Badr, I.H.A.; Abdel-Aziz, A.M. Efficient adsorptive removal of hazardous congo red dye using ce-BTC@microcrystalline cellulose composite. Sci. Rep. 2025, 15, 19734. [Google Scholar] [CrossRef]
- Hamri, N.; Imessaoudene, A.; Hadadi, A.; Cheikh, S.; Boukerroui, A.; Bollinger, J.C.; Amrane, A.; Tahraoui, H.; Tran, H.N.; Ezzat, A.O. Enhanced adsorption capacity of methylene blue dye onto kaolin through acid treatment: Batch adsorption and machine learning studies. Water 2024, 16, 243. [Google Scholar] [CrossRef]
- Zhao, D.; Shen, X. Preparation of chitosan-diatomite/calcium alginate composite hydrogel beads for the adsorption of congo red dye. Water 2023, 15, 2254. [Google Scholar] [CrossRef]
- Quan, W.; Wang, J.; Huang, J.; Zhang, D. Structure characterization and dye adsorption properties of modified fiber from wheat bran. Molecules 2024, 29, 2581. [Google Scholar] [CrossRef] [PubMed]
- Adawiyah, R.; Hanifah, Y.; Palapa, N.R. Analysis congo red dye adsorption on hydrochar and nanohydrochar from areca catechu: Synthesis, characterization, and comparative study. J. Ecol. Eng. 2025, 26, 239. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Xue, Z.; Wang, L. Adsorption and desorption capacity of congo red on CMC/OMMT nanocomposite. Chin. J. Environ. Eng. 2014, 8, 1001–1006. [Google Scholar]
- Qiu, S.; Zhang, H.; Sang, M.; Liu, F.; Nie, G. Fabrication of a 3D cellulose/MoS2 aerogel for efficient and regenerative adsorption of congo red. Water Air Soil. Pollut. 2023, 234, 401. [Google Scholar] [CrossRef]
- Cui, C.; Qiao, W.; Li, D.; Wang, L. Dual cross-linked magnetic gelatin/carboxymethyl cellulose cryogels for enhanced congo red adsorption: Experimental studies and machine learning modelling. J. Colloid Interface Sci. 2025, 678, 619–635. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Dai, R.; Liu, T. The preparation of carboxymethyl cellulose from banana peel. Sci. Technol. Food Ind. 2018, 39, 201–206. [Google Scholar] [CrossRef]

| Model | Qe,exp (mg/g) | Qe,cal (mg/g) | R2 | k | C |
|---|---|---|---|---|---|
| Intraparticle diffusion | 0.7140 | 2.8067 | 129.75 | ||
| Pseudo-first-order | 156.64 | 66.23 | 0.8127 | 0.02767 | |
| Pseudo-second-order | 156.64 | 160.00 | 0.9998 | 0.00267 |
| Model | Qmax (mg/g) | R2 | K | 1/n | A |
|---|---|---|---|---|---|
| Langmuir | 200.80 | 1.0000 | 0.2086 | ||
| Freundlich | 0.8900 | 51.63 | 0.31 | ||
| Dubinin–Radushkevitch | 451.50 | 0.8552 | 0.0034 | ||
| Temkin | 0.8781 | 3.80 | 33.68 |
| ∆G° (kJ/mol) | ∆H° (kJ/mol) | ∆S° (J/mol·K−1) | ||||
|---|---|---|---|---|---|---|
| 10 °C | 20 °C | 30 °C | 40 °C | 50 °C | ||
| −3.58 | −4.69 | −5.80 | −6.91 | −8.02 | 27.86 | 111.04 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, S.; Ma, J.; Yang, F.; Ye, H.; Liang, Y.; Deng, Y.; Li, J.; Wang, R. Synthesis, Characterization, and Properties of Polyvinyl Alcohol/Jackfruit Peel Carboxymethylcellulose/Graphene Oxide/Kaolin Composite Hydrogels. Gels 2025, 11, 626. https://doi.org/10.3390/gels11080626
Liu S, Ma J, Yang F, Ye H, Liang Y, Deng Y, Li J, Wang R. Synthesis, Characterization, and Properties of Polyvinyl Alcohol/Jackfruit Peel Carboxymethylcellulose/Graphene Oxide/Kaolin Composite Hydrogels. Gels. 2025; 11(8):626. https://doi.org/10.3390/gels11080626
Chicago/Turabian StyleLiu, Shumin, Jing Ma, Fuqi Yang, Hailin Ye, Yu Liang, Yijia Deng, Jianrong Li, and Rundong Wang. 2025. "Synthesis, Characterization, and Properties of Polyvinyl Alcohol/Jackfruit Peel Carboxymethylcellulose/Graphene Oxide/Kaolin Composite Hydrogels" Gels 11, no. 8: 626. https://doi.org/10.3390/gels11080626
APA StyleLiu, S., Ma, J., Yang, F., Ye, H., Liang, Y., Deng, Y., Li, J., & Wang, R. (2025). Synthesis, Characterization, and Properties of Polyvinyl Alcohol/Jackfruit Peel Carboxymethylcellulose/Graphene Oxide/Kaolin Composite Hydrogels. Gels, 11(8), 626. https://doi.org/10.3390/gels11080626






