Effect of Marine Basidiomycetes Fulvifomes sp.-Derived Ergosterol Peroxide on Cytotoxicity and Apoptosis Induction in MCF-7 Cell Line
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.2. Sample Collection and Identification
2.3. Extraction and Purification
2.4. Semi-Preparative High Performance Liquid Chromatography (HPLC) Analysis
2.5. Fourier-Transform Infared (FTIR) Spectroscopy Analysis
2.6. Nuclear Magnetic Resonance (NMR) Spectroscopy
2.7. MTT Assay
2.8. Assessment of Reactive Oxygen Species (ROS) by DCFH-DA
2.9. Assessment of Mitochondrial Membrane Potential (MMP)
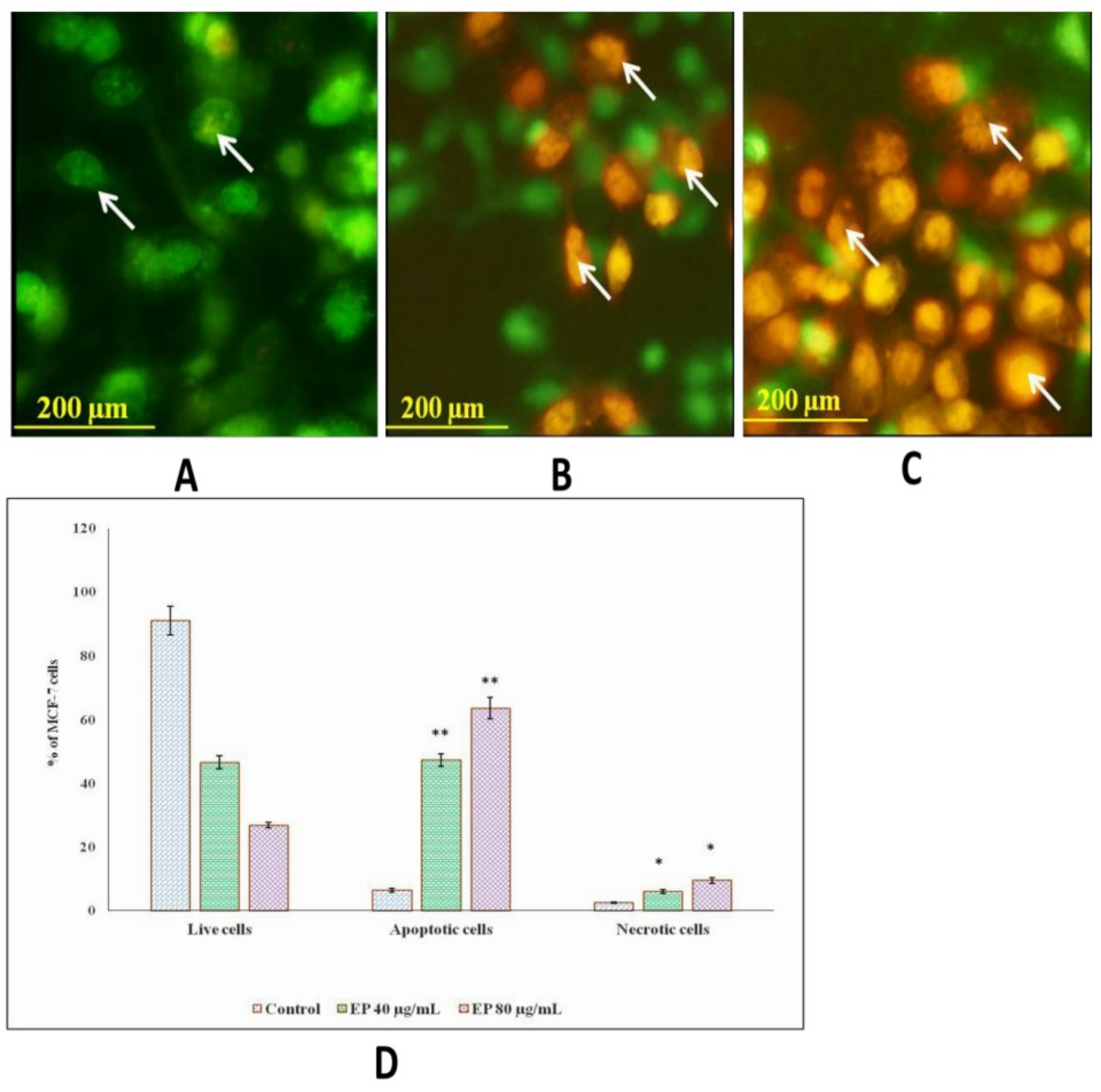
2.10. AO/EtBr Staining
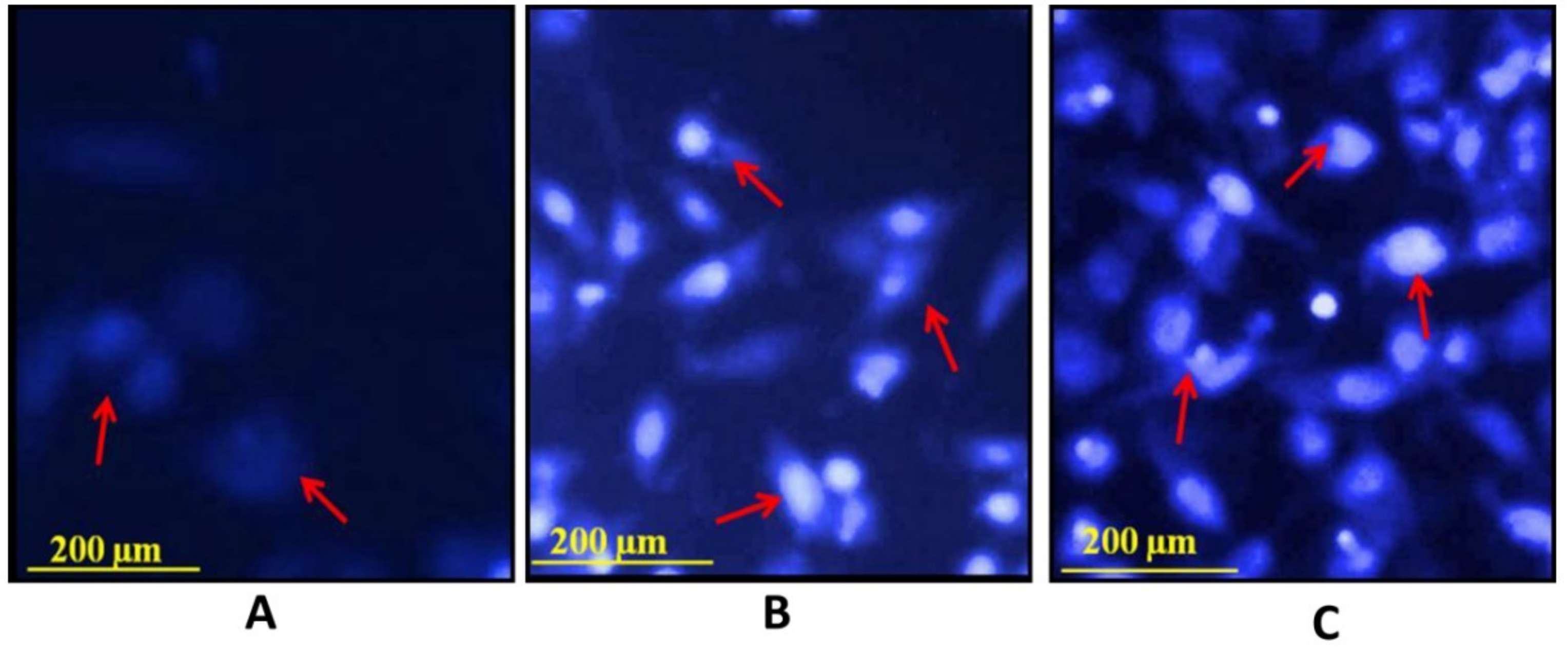
2.11. Nuclear Damage Observed with Hoechst 33258 Staining
2.12. Statistical Analysis
3. Results
3.1. Morphological Characteristics of Marine Basidiomycetes
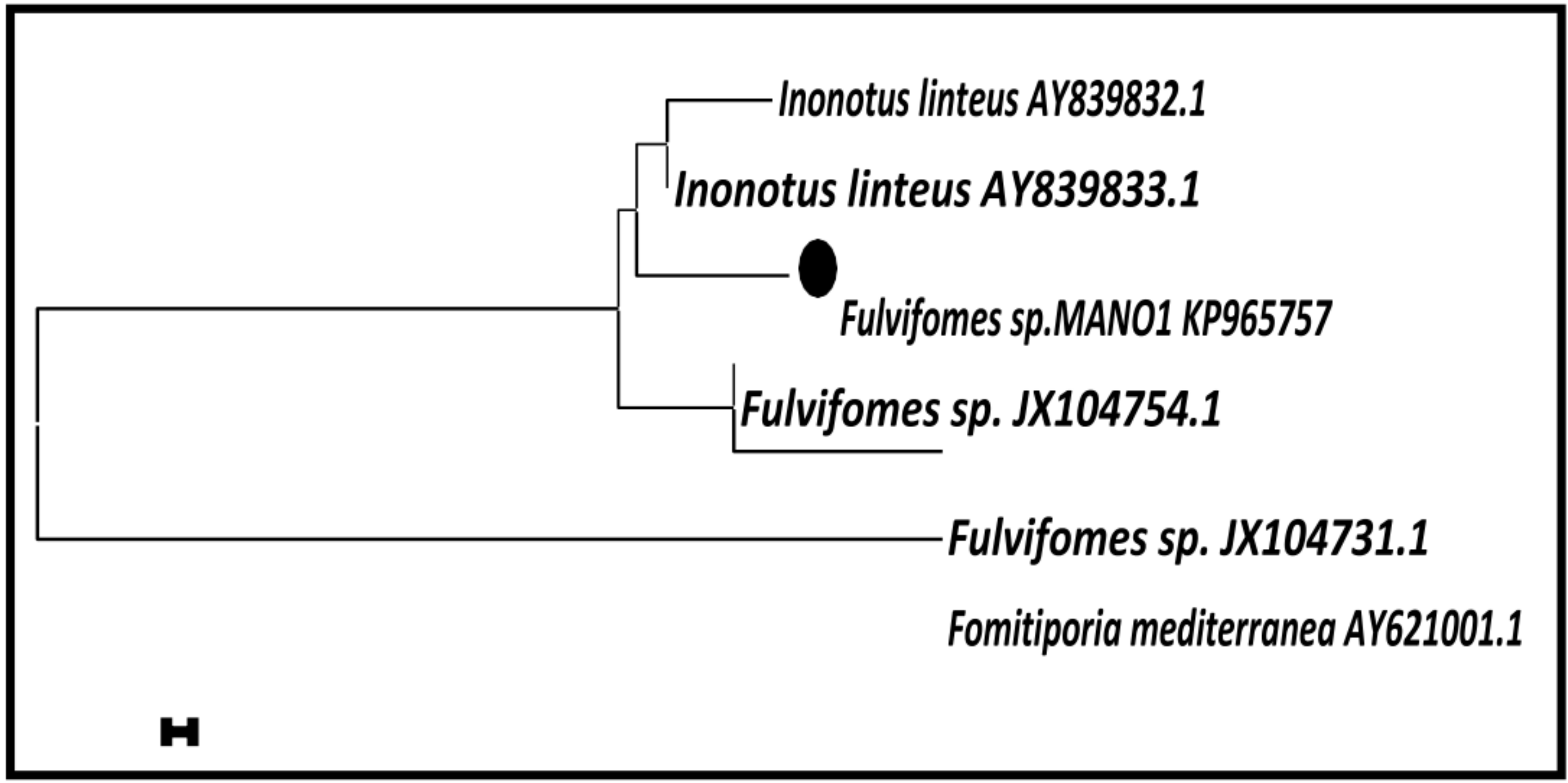
3.2. Molecular Taxonomy of Marine Basidiomycetes
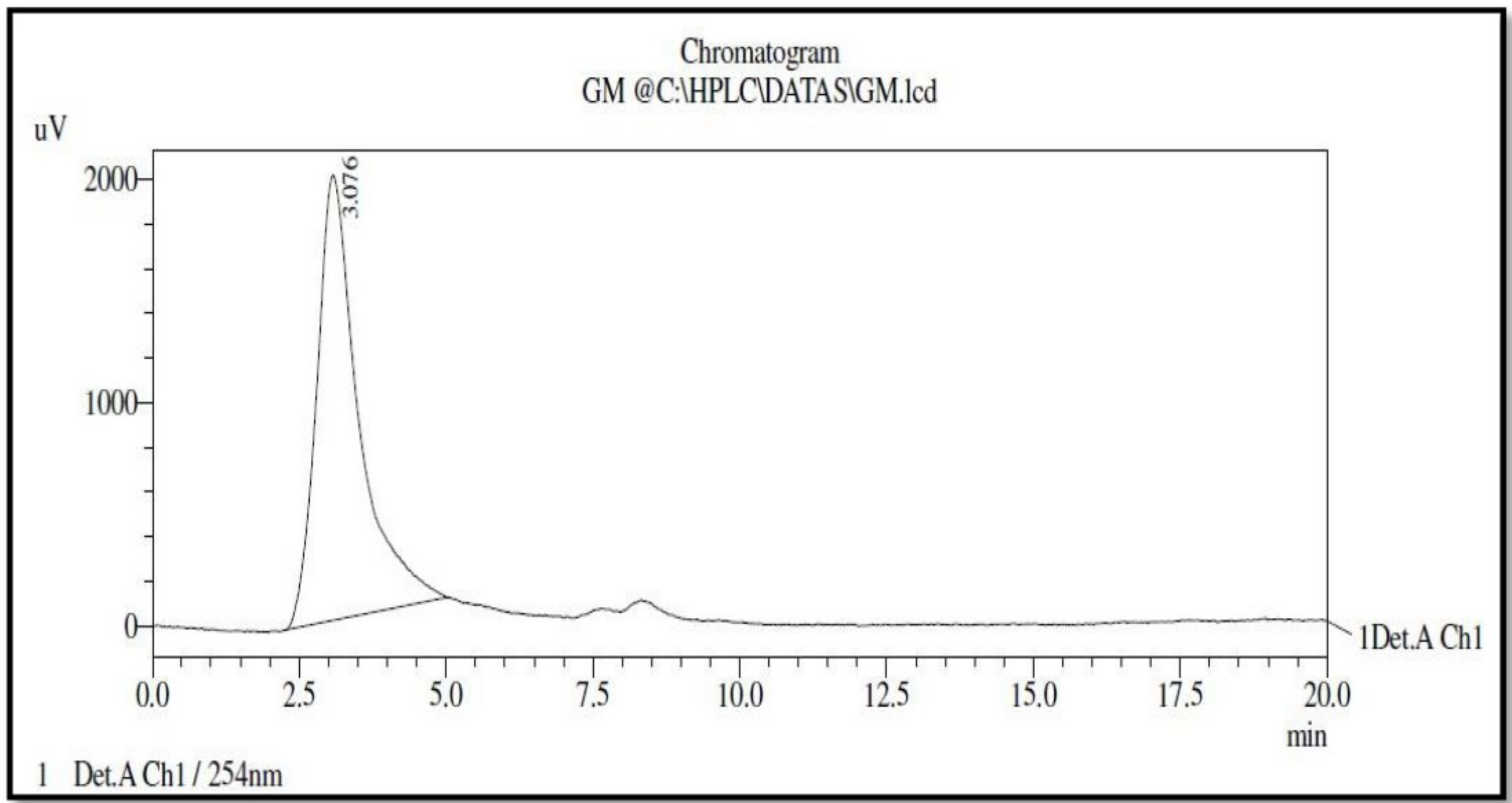
3.3. High Performance Liquid Chromatography (HPLC) Analysis of Bioactive Compound
3.4. Infrared (IR) Spectrometry
3.5. Structural Prediction of Bioactive Compounds
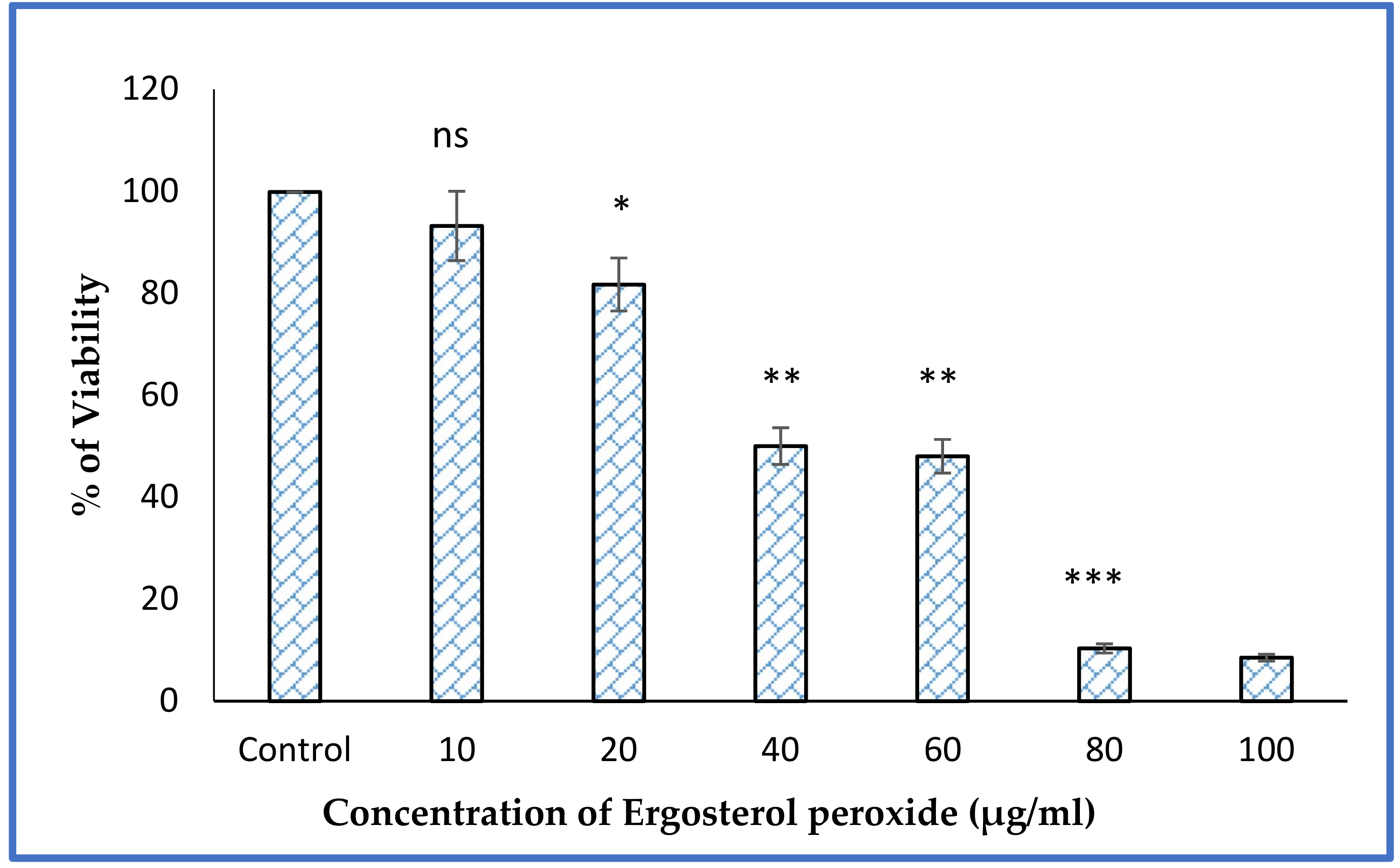
3.6. MTT Assay
3.7. Marine Basidiomycetes-Derived Ergosterol Peroxide (EP) Induces Reactive Oxygen Species (ROS)-Mediated Cell Death
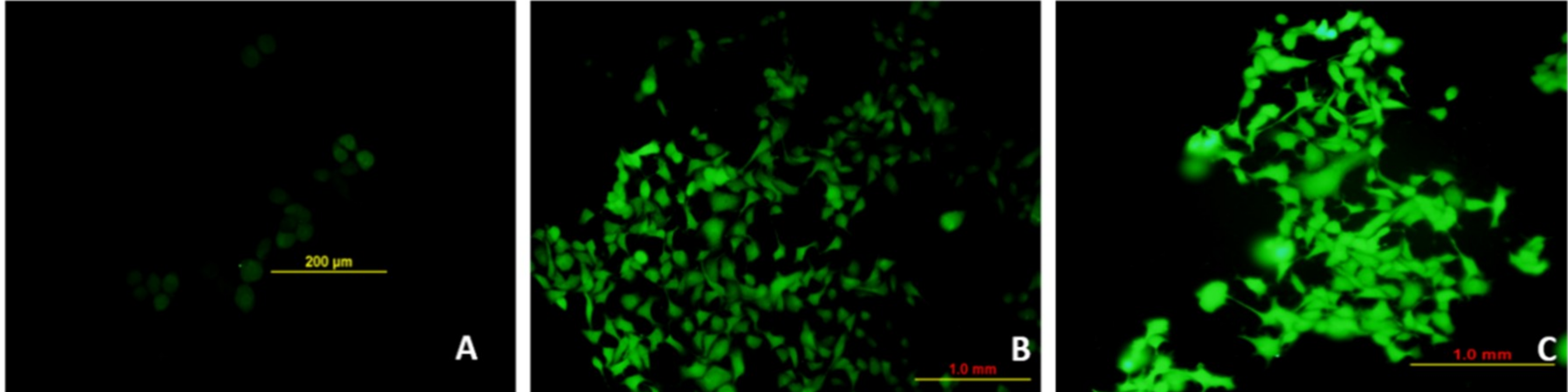
3.8. Et/Br Staining
3.9. Loss of Mitochondrial Membrane Potential
3.10. Hoechst Staining 33342
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Webster, J.; Weber, R. Introduction to Fungi; Cambridge University Press: Cambridge, UK, 2007. [Google Scholar]
- Wasser, S. Medicinal mushrooms as a source of antitumor and immunomodulating polysaccharides. Appl. Microbiol. Biotechnol. 2003, 60, 258–274. [Google Scholar]
- Silva, M.A.B.; Bernini, E.; Carmo, T.M.S. Características estruturais de bosques de mangue do estuário do rio São Mateus, Brasil. Acta Bot. Bras. 2005, 19, 465–471. [Google Scholar] [CrossRef]
- Robledo, G.L.; Renison, D. Wood-decaying polypores in the mountains of central Argentina in relation to Polylepis forest structure and altitude. Fungal Ecol. 2010, 3, 178–184. [Google Scholar] [CrossRef]
- Gilbert, G.S.; Gorospe, J.; Ryvarden, L. Host and habitat preferences of polypore fungi in Micronesian tropical flooded forests. Mycol. Res. 2008, 112, 674–680. [Google Scholar] [CrossRef] [PubMed]
- Hattori, T.; Sakayaroj, J.; Jones, E.B.G.; Suetrong, S.; Preedanon, S.; Klaysuban, A. Three species of Fulvifomes (Basidiomycota, Hymenochaetales) associated with rots on mangrove tree Xylocarpus granatum in Thailand. Mycoscience 2014, 55, 344–354. [Google Scholar] [CrossRef]
- Malvia, S.; Bagadi, S.A.; Dubey, U.S.; Saxena, S. Epidemiology of breast cancer in Indian women. Asia Pac. J. Clin. Oncol. 2017, 13, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Rastogi, R.P.; Singh, S.P.; Häder, D.P.; Sinha, R.P. Detection of reactive oxygen species (ROS) by the oxidant-sensing probe 2’,7’-dichlorodihydrofluorescein diacetate in the cyanobacterium Anabaena variabilis PCC 7937. Biochem. Biophys. Res. Commun. 2010, 397, 603–607. [Google Scholar] [CrossRef]
- Ribble, D.; Goldstein, N.B.; Norris, D.A.; Shellman, Y.G. A simple technique for quantifying apoptosis in 96-well plates. BMC Biotechnol. 2005, 5, 12. [Google Scholar] [CrossRef]
- Emaus, R.K.; Grunwald, R.; Lemasters, J.J. Rhodamine 123 as a probe of transmembrane potential in isolated rat-liver mitochondria. Biochim. Biophys. Acta 1986, 850, 436–448. [Google Scholar] [CrossRef]
- Dash, S.K.; Chattopadhyay, S.; Ghosh, T.; Tripathy, S.; Das, S.; Das, D.; Roy, S. Antileukemic Efficacy of Monomeric Manganese-Based Metal Complex on KG-1A and K562 Cell Lines. ISRN Oncol. 2013, 2013, 709269. [Google Scholar] [CrossRef] [PubMed]
- Sakayaroj, J.; Preedanon, S.; Suetrong, S. Molecular characterization of basidiomycetes associated with the decayed mangrove tree Xylocarpus granatum in Thailand. Fungal Divers. 2012, 145–156. [Google Scholar] [CrossRef]
- Nam, K.S.; Jo, Y.S.; Kim, Y.H.; Hyun, J.W.; Kim, H.W. Cytotoxic activities of acetoxyscirpenediol and ergosterol peroxide from Paecilomyces tenuipes. Life Sci. 2001, 69, 229–237. [Google Scholar] [CrossRef]
- Kuo, Y.C.; Weng, S.C.; Chou, C.J.; Chang, T.T.; Tsai, W.J. Activation and proliferation signals in primary human T lymphocytes inhibited by ergosterol peroxide isolated from Cordyceps cicadae. Br. J. Pharmacol. 2003, 140, 895–906. [Google Scholar] [CrossRef] [PubMed]
- Tan, W.; Pan, M.; Liu, H.; Tian, H.; Ye, Q.; Liu, H. Ergosterol peroxide inhibits ovarian cancer cell growth through multiple pathways. Onco Targets Ther. 2017, 10, 3467–3474. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Sohn, E.J.; Kim, B.; Kim, S.; Won, G.; Yoon, S.; Lee, J.; Kim, M.J.; Lee, H.; Chung, K.; et al. Upregulation of death receptor 5 and activation of caspase 8/3 play a critical role in ergosterol peroxide induced apoptosis in DU 145 prostate cancer cells. Cancer Cell Int. 2014, 14, 1–8. [Google Scholar] [CrossRef]
- Wu, Q.P.; Xie, Y.Z.; Deng, Z.; Li, X.M.; Yang, W.; Jiao, C.W.; Fang, L.; Li, S.Z.; Pan, H.H.; Yee, A.J.; et al. Ergosterol Peroxide Isolated from Ganoderma lucidum Abolishes MicroRNA miR-378-Mediated Tumor Cells on Chemoresistance. PLoS ONE 2012, 7, e44579. [Google Scholar] [CrossRef]
- Takei, T.; Yoshida, M.; Ohnishi-kameyama, M.; Kobori, M. Ergosterol Peroxide, an Apoptosis-Inducing Component Isolated from Sarcodon aspratus (Berk.) S. Ito. Biosci. Biotechnol. Biochem. 2005, 69, 212–215. [Google Scholar] [CrossRef]
- Rhee, Y.H.; Jeong, S.J.; Lee, H.J.; Lee, H.J.; Koh, W.; Jung, J.H.; Kim, S.H.; Sung-Hoon, K. Inhibition of STAT3 signaling and induction of SHP1 mediate antiangiogenic and antitumor activities of ergosterol peroxide in U266 multiple myeloma cells. BMC Cancer 2012, 12, 28. [Google Scholar] [CrossRef]
- Kang, J.; Jang, J.; Mishra, S.K.; Lee, H.; Nho, C.W.; Shin, D.; Jin, M.; Kim, M.K.; Choi, C.; Oh, S.H. Author ’ s Accepted Manuscript. J. Ethnopharmacol. 2015. [Google Scholar]
- Russo, A.; Cardile, V.; Piovano, M.; Caggia, S.; Espinoza, C.L.; Garbarino, J.A. Chemico-Biological Interactions Pro-apoptotic activity of ergosterol peroxide and (22E)-ergosta-7,22-dien-5-hydroxy-3,6-dione in human prostate cancer cells. Chem. Biol. Interact. 2010, 184, 352–358. [Google Scholar] [CrossRef] [PubMed]
- Kobori, M.; Yoshida, M.; Ohnishi-Kameyama, M.; Shinmoto, H. Ergosterol peroxide from an edible mushroom suppresses inflammatory responses in RAW264.7 macrophages and growth of HT29 colon adenocarcinoma cells. Br. J. Pharmacol. 2007, 150, 209–219. [Google Scholar] [PubMed]
- Prompiboon, P.; Bhumiratana, A.; Ruchirawat, S.; Boucias, D.G.; Wiwat, C. Isolation of ergosterol peroxide from Nomuraea rileyi infected larvae of tobacco cutworm. World J. Microbiol. Biotechnol. 2008, 24, 2909–2917. [Google Scholar] [CrossRef]


| Atom Position | δ13C (ppm) | δ1H(ppm) |
|---|---|---|
| 1 | 36.96 | 2.1 |
| 2 | 30.14 | 1.97 |
| 3 | 66.45 | 3.95 |
| 4 | 51.17 | 0 |
| 5 | 79.4 | 5.24 |
| 6 | 132.36 | 6.24 |
| 7 | 135.41 | 6.5 |
| 8 | 82.13 | 5.23 |
| 9 | 34.72 | 0 |
| 10 | 36.96 | 0 |
| 11 | 20.86 | 0 |
| 12 | 39.38 | 0 |
| 13 | 44.57 | 0 |
| 14 | 51.71 | 0 |
| 15 | 28.58 | 0 |
| 16 | 23.4 | 0 |
| 17 | 56.26 | 1.21 |
| 18 | 12.86 | 0.88 |
| 19 | 18.14 | 0.9 |
| 20 | 39.64 | 0 |
| 21 | 28.86 | 0.99 |
| 22 | 132.34 | 5.16 |
| 23 | 135.19 | 5.16 |
| 24 | 42.78 | 1.85 |
| 25 | 33.06 | 1.47 |
| 26 | 19.06 | 0.88 |
| 27 | 19.61 | 0.88 |
| 28 | 17.53 | 1.21 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Govindharaj, M.; Arumugam, S.; Nirmala, G.; Bharadwaj, M.; Murugiyan, K. Effect of Marine Basidiomycetes Fulvifomes sp.-Derived Ergosterol Peroxide on Cytotoxicity and Apoptosis Induction in MCF-7 Cell Line. J. Fungi 2019, 5, 16. https://doi.org/10.3390/jof5010016
Govindharaj M, Arumugam S, Nirmala G, Bharadwaj M, Murugiyan K. Effect of Marine Basidiomycetes Fulvifomes sp.-Derived Ergosterol Peroxide on Cytotoxicity and Apoptosis Induction in MCF-7 Cell Line. Journal of Fungi. 2019; 5(1):16. https://doi.org/10.3390/jof5010016
Chicago/Turabian StyleGovindharaj, Mano, Sathishkumar Arumugam, Grace Nirmala, Mausumi Bharadwaj, and Kalaiselvam Murugiyan. 2019. "Effect of Marine Basidiomycetes Fulvifomes sp.-Derived Ergosterol Peroxide on Cytotoxicity and Apoptosis Induction in MCF-7 Cell Line" Journal of Fungi 5, no. 1: 16. https://doi.org/10.3390/jof5010016
APA StyleGovindharaj, M., Arumugam, S., Nirmala, G., Bharadwaj, M., & Murugiyan, K. (2019). Effect of Marine Basidiomycetes Fulvifomes sp.-Derived Ergosterol Peroxide on Cytotoxicity and Apoptosis Induction in MCF-7 Cell Line. Journal of Fungi, 5(1), 16. https://doi.org/10.3390/jof5010016





