Deletion of ptn1, a PTEN/TEP1 Orthologue, in Ustilago maydis Reduces Pathogenicity and Teliospore Development
Abstract
:1. Introduction
2. Materials and Methods
2.1. Strains and Growth Conditions
2.2. Vector Construction and Nucleic Acid Manipulations
2.3. RNA Isolation and Expression Analysis
2.4. Cell Growth, Mating, and Plant Infection
2.5. Statistical Analysis
3. Results
3.1. In Silico Analysis of ptn1
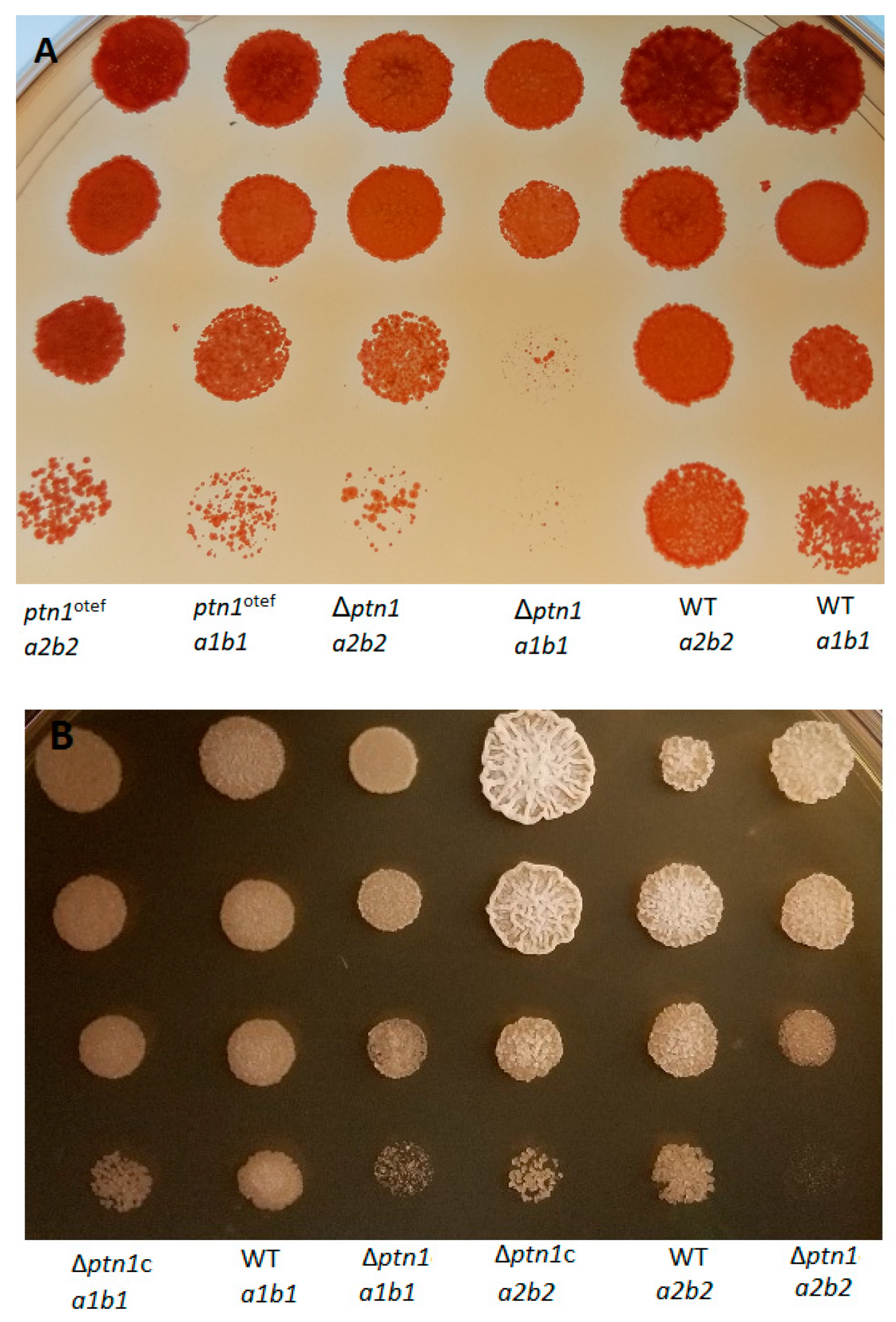
3.2. Deletion of ptn1 Led to the Reduced Production of the Aerial Hyphae Characteristics of the Mating Reaction on Charcoal Media
3.3. prf1 Is Upregulated in Δptn1 under Low Ammonium Conditions
3.4. Cell Growth of ptn1 Mutants Is Largely Unaffected, while Stress Tolerance Is Reduced under Certain Conditions
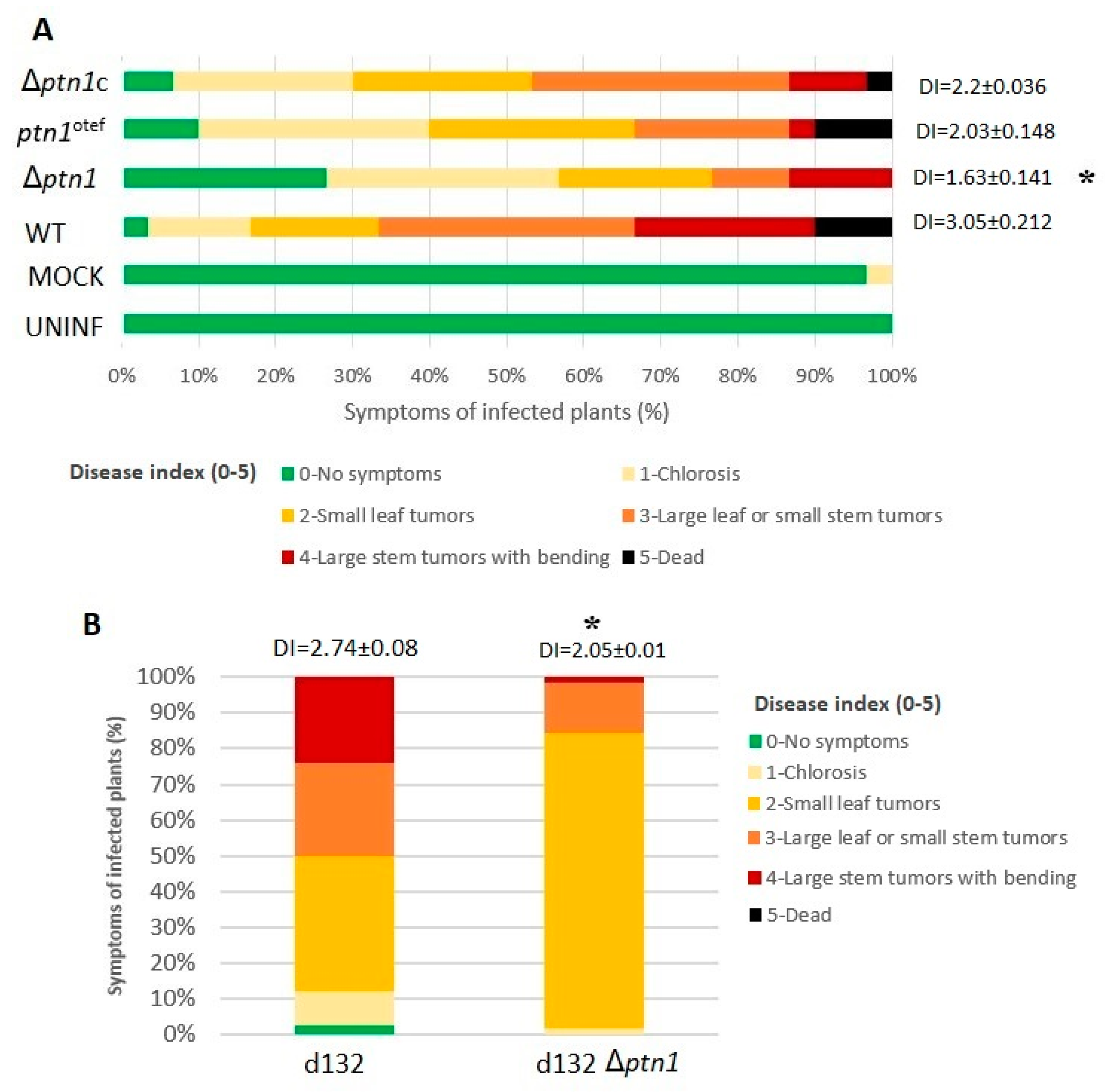
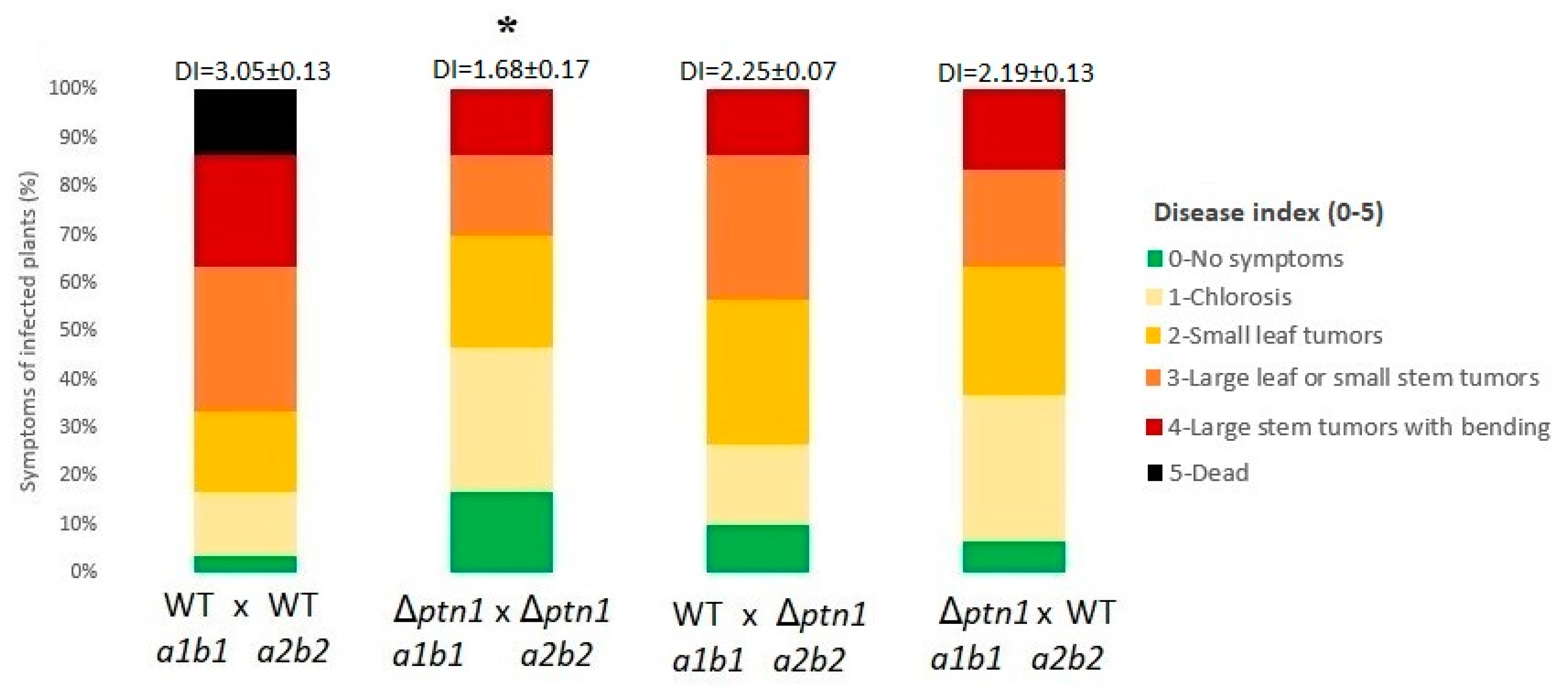
3.5. Pathogenicity Is Reduced by ptn1 Deletion
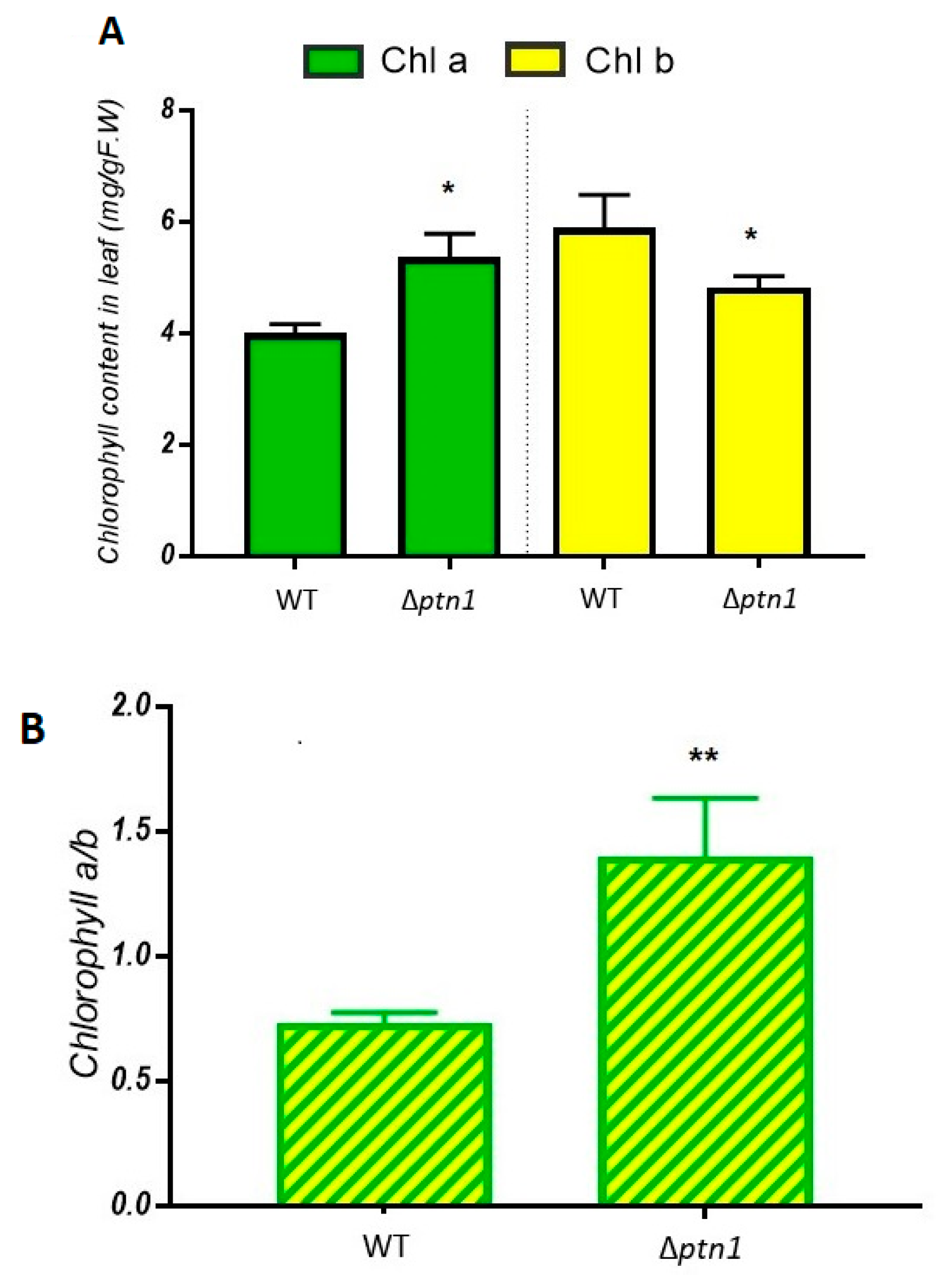
3.6. Pathogenicity Calculated by Additional Plant Growth Indicators
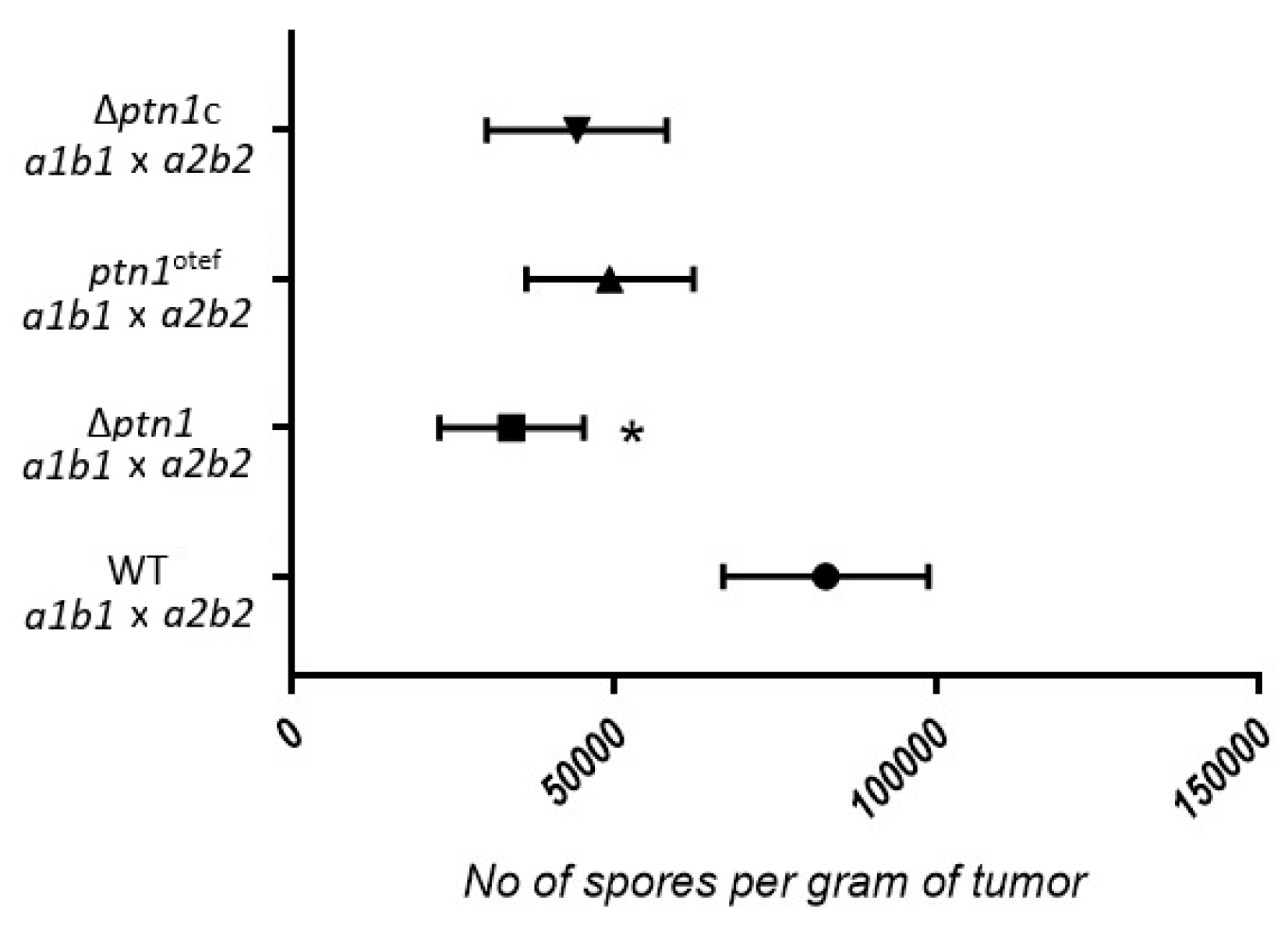
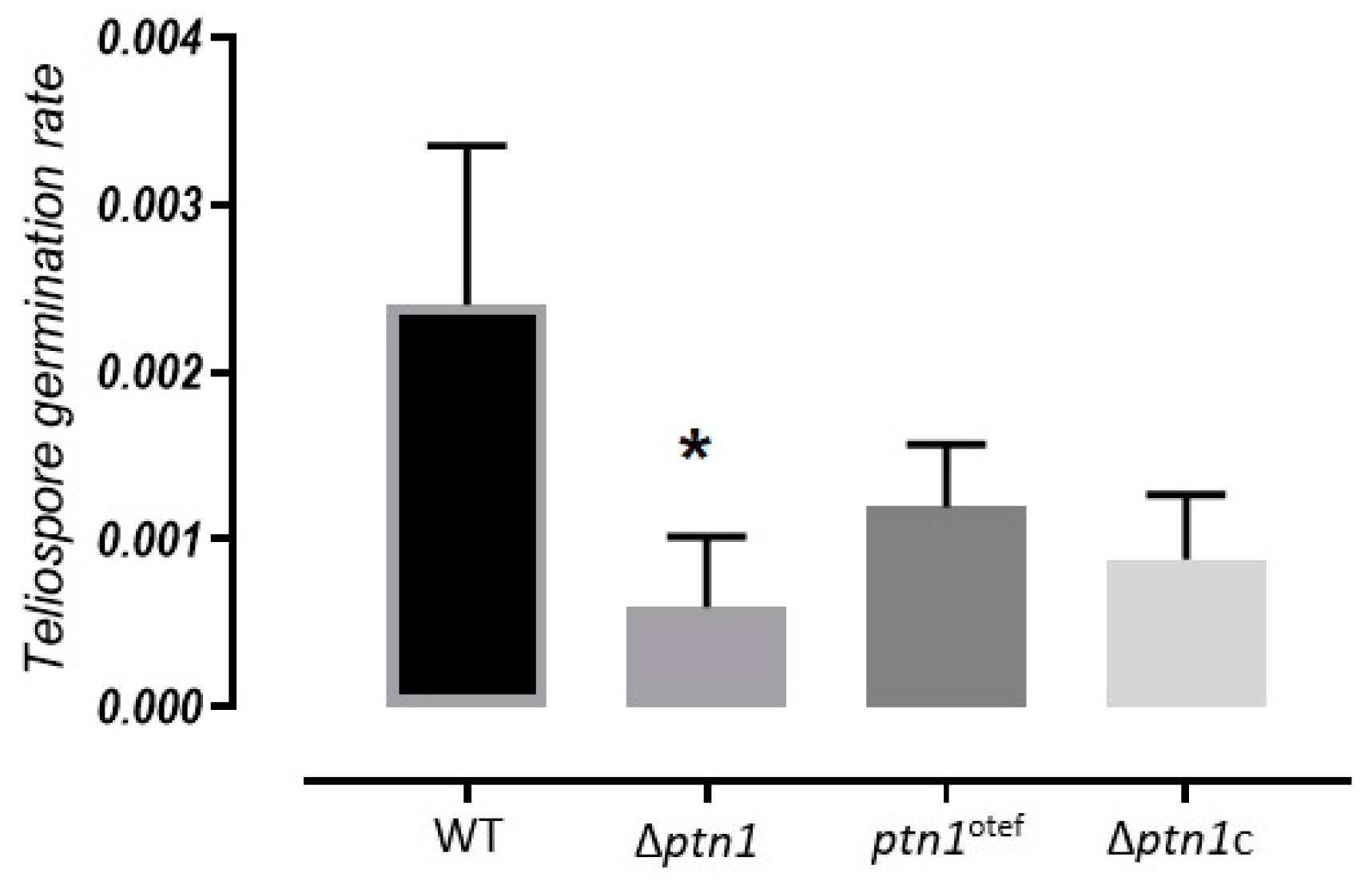
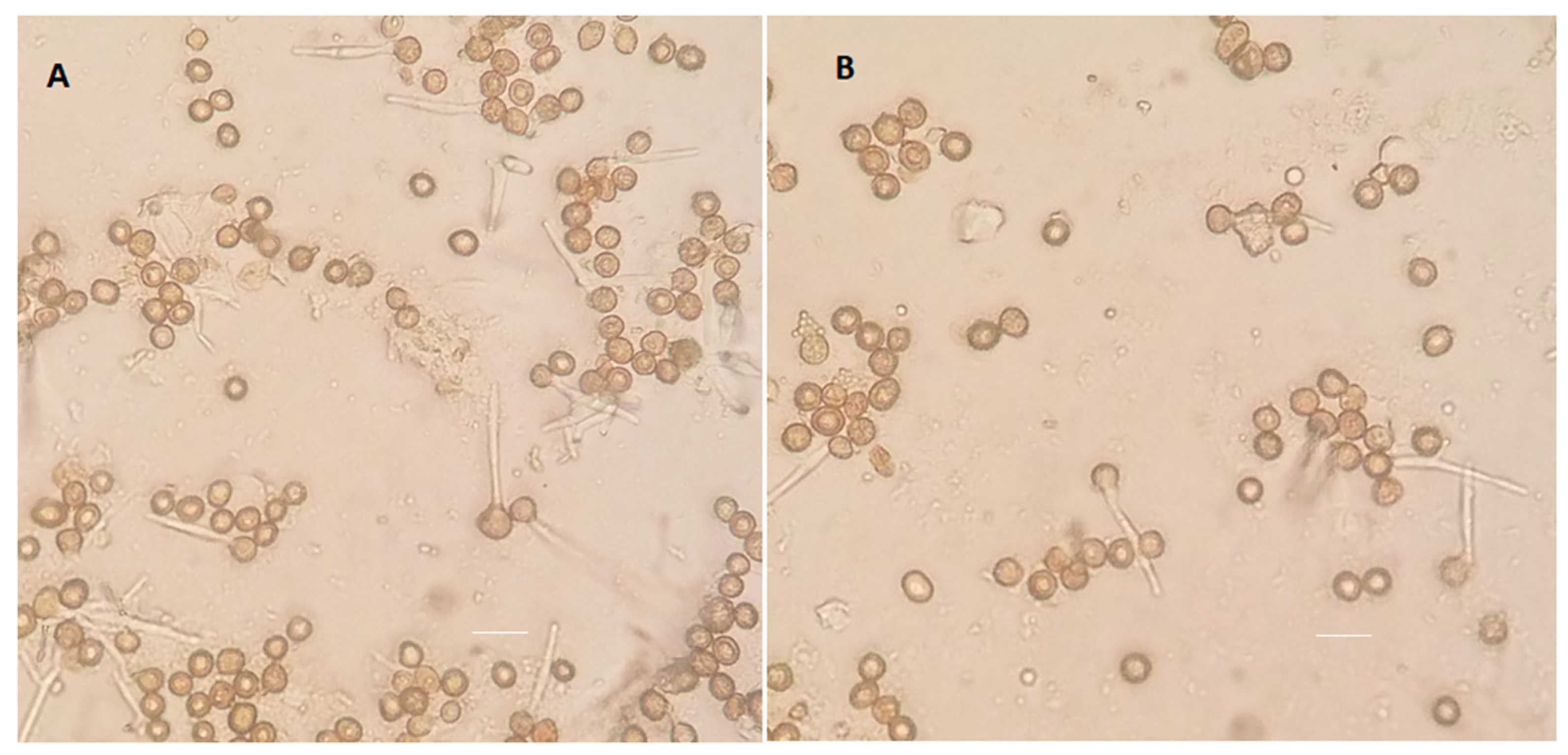
3.7. Impact of ptn1 Mutations on Spore Formation and Germination
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Brefort, T.; Doehlemann, G.; Mendoza-Mendoza, A.; Reissmann, S.; Djamei, A.; Kahmann, R. Ustilago maydis as a Pathogen. Annu. Rev. Phytopathol. 2009, 47, 423–445. [Google Scholar] [CrossRef] [PubMed]
- Steinberg, G.; Perez-Martin, J. Ustilago maydis, a new fungal model system for cell biology. Trends Cell Biol. 2008, 18, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Perez-Nadales, E.; Nogueira, M.F.; Baldin, C.; Castanheira, S.; El Ghalid, M.; Grund, E.; Lengeler, K.; Marchegiani, E.; Mehrotra, P.V.; Moretti, M.; et al. Fungal model systems and the elucidation of pathogenicity determinants. Fungal Genet. Biol. 2014, 70, 42–67. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vollmeister, E.; Schipper, K.; Baumann, S.; Haag, C.; Pohlmann, T.; Stock, J.; Feldbrugge, M. Fungal development of the plant pathogen Ustilago maydis. FEMS Microbiol. Rev. 2012, 36, 59–77. [Google Scholar] [CrossRef] [PubMed]
- Steck, P.A.; Pershouse, M.A.; Jasser, S.A.; Yung, W.K.A.; Lin, H.; Ligon, A.H.; Langford, L.A.; Baumgard, M.L.; Hattier, T.; Davis, T.; et al. Identification of a candidate tumour suppressor gene, MMAC1, at chromosome 10q23.3 that is mutated in multiple advanced cancers. Nat. Genet. 1997, 15, 356. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Yen, C.; Liaw, D.; Podsypanina, K.; Bose, S.; Wang, S.I.; Puc, J.; Miliaresis, C.; Rodgers, L.; McCombie, R.; et al. A Putative Protein Tyrosine Phosphatase Gene Mutated in Human Brain, Breast, and Prostate Cancer. Science 1997, 275, 1943. [Google Scholar] [CrossRef] [PubMed]
- Song, M.S.; Salmena, L.; Pandolfi, P.P. The functions and regulation of the PTEN tumour suppressor. Nat. Rev. Mol. Cell Biol. 2012, 13, 283–296. [Google Scholar] [CrossRef] [PubMed]
- Leslie, N.R.; Downes, C.P. PTEN function: How normal cells control it and tumour cells lose it. Biochem. J. 2004, 382, 1–11. [Google Scholar] [CrossRef]
- Alimonti, A.; Carracedo, A.; Clohessy, J.G.; Trotman, L.C.; Nardella, C.; Egia, A.; Salmena, L.; Sampieri, K.; Haveman, W.J.; Brogi, E.; et al. Subtle variations in Pten dose determine cancer susceptibility. Nat. Genet. 2010, 42, 454. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Chin-Sang, I.D. C. elegans as a model to study PTEN’s regulation and function. Methods 2015, 77–78, 180–190. [Google Scholar] [CrossRef]
- Huang, H.; Potter, C.J.; Tao, W.; Li, D.M.; Brogiolo, W.; Hafen, E.; Sun, H.; Xu, T. PTEN affects cell size, cell proliferation and apoptosis during Drosophila eye development. Development 1999, 126, 5365. [Google Scholar] [PubMed]
- Gil, E.B.; Malone Link, E.; Liu, L.X.; Johnson, C.D.; Lees, J.A. Regulation of the insulin-like developmental pathway of Caenorhabditis elegans by a homolog of the PTEN tumor suppressor gene. Proc. Natl. Acad. Sci. USA 1999, 96, 2925–2930. [Google Scholar] [CrossRef] [Green Version]
- Solari, F.; Bourbon-Piffaut, A.; Masse, I.; Payrastre, B.; Chan, A.M.L.; Billaud, M. The human tumour suppressor PTEN regulates longevity and dauer formation in Caenorhabditis elegans. Oncogene 2005, 24, 20. [Google Scholar] [CrossRef] [PubMed]
- Serezani, C.H.; Kane, S.; Medeiros, A.I.; Cornett, A.M.; Kim, S.H.; Marques, M.M.; Lee, S.P.; Lewis, C.; Bourdonnay, E.; Ballinger, M.N.; et al. PTEN directly activates the actin depolymerization factor cofilin-1 during PGE2-mediated inhibition of phagocytosis of fungi. Sci. Signal 2012, 5, ra12. [Google Scholar] [CrossRef] [PubMed]
- Heymont, J.; Berenfeld, L.; Collins, J.; Kaganovich, A.; Maynes, B.; Moulin, A.; Ratskovskaya, I.; Poon, P.P.; Johnston, G.C.; Kamenetsky, M.; et al. TEP1, the yeast homolog of the human tumor suppressor gene PTEN/MMAC1/TEP1, is linked to the phosphatidylinositol pathway and plays a role in the developmental process of sporulation. Proc. Natl. Acad. Sci. USA 2000, 97, 12672–12677. [Google Scholar] [CrossRef] [Green Version]
- Brachmann, A.; König, J.; Julius, C.; Feldbrügge, M. A reverse genetic approach for generating gene replacement mutants in Ustilago maydis. Mol. Genet. Genom. 2004, 272, 216–226. [Google Scholar] [CrossRef]
- Holliday, R. Ustilago maydis. In Bacteria, Bacteriophages, and Fungi: Volume 1; King, R.C., Ed.; Springer US: Boston, MA, USA, 1974. [Google Scholar]
- Cervantes-Chávez, J.A.; Ali, S.; Bakkeren, G. Response to environmental stresses, cell-wall integrity, and virulence are orchestrated through the calcineurin pathway in Ustilago hordei. Mol. Plant-Microb. Interact. 2011, 24, 219–232. [Google Scholar] [CrossRef]
- Gold, S.E.; Brogdon, S.M.; Mayorga, M.E.; Kronstad, J.W. The Ustilago maydis regulatory subunit of a cAMP-dependent protein kinase is required for gall formation in maize. Plant Cell 1997, 9, 1585–1594. [Google Scholar] [CrossRef]
- Muller, P.; Aichinger, C.; Feldbrugge, M.; Kahmann, R. The MAP kinase kpp2 regulates mating and pathogenic development in Ustilago maydis. Mol. Microbiol. 1999, 34, 1007–1017. [Google Scholar] [CrossRef]
- Kronstad, J.W.; Leong, S.A. Isolation of two alleles of the b locus of Ustilago maydis. Proc. Natl. Acad. Sci. USA 1989, 86, 978. [Google Scholar] [CrossRef]
- Garcia-Pedrajas, M.D.; Nadal, M.; Kapa, L.B.; Perlin, M.H.; Andrews, D.L.; Gold, S.E. DelsGate, a robust and rapid gene deletion construction method. Fungal Genet. Biol. 2008, 45, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Pham, C.D.; Yu, Z.; Sandrock, B.; Bölker, M.; Gold, S.E.; Perlin, M.H. Ustilago maydis Rho1 and 14-3-3 homologues participate in pathways controlling cell separation and cell polarity. Eukaryot Cell 2009, 8, 977–989. [Google Scholar] [CrossRef] [PubMed]
- Harrison, N.; Cavinder, B.; Townsend, J.P.; Trail, F. Optimized primers and other critical conditions for efficient fusion PCR to generate knockout vectors in filamentous fungi. Fungal Genet. Rep. 2013, 60, 1–10. [Google Scholar] [CrossRef]
- Kamper, J. A PCR-based system for highly efficient generation of gene replacement mutants in Ustilago maydis. Mol. Genet. Genom. 2004, 271, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Paul, J.A.; Wallen, R.M.; Zhao, C.; Shi, T.; Perlin, M.H. Coordinate regulation of Ustilago maydis ammonium transporters and genes involved in mating and pathogenicity. Fungal Biol. 2018, 122, 639–650. [Google Scholar] [CrossRef]
- Ho, E.C.H.; Cahill, M.J.; Saville, B.J. Gene discovery and transcript analyses in the corn smut pathogen Ustilago maydis: Expressed sequence tag and genome sequence comparison. BMC Genom. 2007, 8, 334. [Google Scholar] [CrossRef]
- Pham, C.D.; Yu, Z.; Ben Lovely, C.; Agarwal, C.; Myers, D.A.; Paul, J.A.; Cooper, M.; Barati, M.; Perlin, M.H. Haplo-insufficiency for different genes differentially reduces pathogenicity and virulence in a fungal phytopathogen. Fungal Genet. Biol. 2012, 49, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Doyle, C.E.; Kitty Cheung, H.Y.; Spence, K.L.; Saville, B.J. Unh1, an Ustilago maydis Ndt80-like protein, controls completion of tumor maturation, teliospore development, and meiosis. Fungal Genet. Biol. 2016, 94, 54–68. [Google Scholar] [CrossRef]
- Akhtar, T.; Zia-Ur-Rehman, M.; Naeem, A.; Nawaz, R.; Ali, S.; Murtaza, G.; Maqsood, M.A.; Azhar, M.; Khalid, H.; Rizwan, M. Photosynthesis and growth response of maize (Zea mays L.) hybrids exposed to cadmium stress. Environ. Sci. Pollut. Res. Int. 2017, 24, 5521–5529. [Google Scholar] [CrossRef] [PubMed]
- Manolopoulou, E.; Varzakas, T.; Petsalaki, A. Chlorophyll Determination in Green Pepper Using two Different Extraction Methods. Curr. Res. Nutr. Food Sci. J. 2018, 4, 52–60. [Google Scholar] [CrossRef]
- Hiscox, J.D.; Israelstam, G.F. A method for the extraction of chlorophyll from leaf tissue without maceration. Can. J. Bot. 1979, 57, 1332–1334. [Google Scholar] [CrossRef]
- Morrison, E.N.; Emery, R.J.; Saville, B.J. Phytohormone Involvement in the Ustilago maydis-Zea mays Pathosystem: Relationships between Abscisic Acid and Cytokinin Levels and Strain Virulence in Infected Cob Tissue. PLoS ONE 2015, 10, e0130945. [Google Scholar] [CrossRef]
- Conover, W.J. Practical Nonparametric Statistics, 3rd ed.; John Wiley: New York, NY, USA; Chichester, UK, 1999. [Google Scholar]
- Redkar, A.; Hoser, R.; Schilling, L.; Zechmann, B.; Krzymowska, M.; Walbot, V.; Doehlemann, G. A Secreted Effector Protein of Ustilago maydis Guides Maize Leaf Cells to Form Tumors. Plant Cell 2015, 27, 1332–1351. [Google Scholar] [CrossRef] [PubMed]
- Torró, I.; Bretó, P.; García-Yzaguirre, A. The influence of plant spacing in the early stages of selection of rice (Oryza sativa L.) varieties. Span. J. Agric. Res. 2016, 14, e0704. [Google Scholar] [CrossRef]
- Klose, J.; de Sa, M.M.; Kronstad, J.W. Lipid-induced filamentous growth in Ustilago maydis. Mol. Microbiol. 2004, 52, 823–835. [Google Scholar] [CrossRef]
- Smith, D.G.; Garcia-Pedrajas, M.D.; Gold, S.E.; Perlin, M.H. Isolation and characterization from pathogenic fungi of genes encoding ammonium permeases and their roles in dimorphism. Mol. Microbiol. 2003, 50, 259–275. [Google Scholar] [CrossRef] [Green Version]
- Ruiz-Herrera, J.; Leon, C.G.; Guevara-Olvera, L.; Carabez-Trejo, A. Yeast-mycelial dimorphism of haploid and diploid strains of Ustilago maydis. Microbiology 1995, 141, 695–703. [Google Scholar] [CrossRef]
- Kretschmer, M.; Lambie, S.; Croll, D.; Kronstad, J.W. Acetate provokes mitochondrial stress and cell death in Ustilago maydis. Mol. Microbiol. 2018, 107, 488–507. [Google Scholar] [CrossRef]
- Kaffarnik, F.; Muller, P.; Leibundgut, M.; Kahmann, R.; Feldbrugge, M. PKA and MAPK phosphorylation of Prf1 allows promoter discrimination in Ustilago maydis. EMBO J. 2003, 22, 5817–5826. [Google Scholar] [CrossRef]
- Doehlemann, G.; Wahl, R.; Horst, R.J.; Voll, L.M.; Usadel, B.; Poree, F.; Stitt, M.; Pons-Kühnemann, J.; Sonnewald, U.; Kahmann, R.; et al. Reprogramming a maize plant: Transcriptional and metabolic changes induced by the fungal biotroph Ustilago maydis. Plant J. 2008, 56, 181–195. [Google Scholar] [CrossRef]
- Horst, R.J.; Engelsdorf, T.; Sonnewald, U.; Voll, L.M. Infection of maize leaves with Ustilago maydis prevents establishment of C4 photosynthesis. J. Plant Physiol. 2008, 165, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Funayama-Noguchi, S.; Terashima, I. Effects of Eupatorium yellow vein virus infection on photosynthetic rate, chlorophyll content and chloroplast structure in leaves of Eupatorium makinoi during leaf development. J. Funct. Plant Biol. 2006, 33, 165–175. [Google Scholar] [CrossRef]
- Pavlovic, D.; Nikolic, B.; Djurovic, S.; Waisi, H.; Andjelkovic, A.; Marisavljevic, D. Chlorophyll as a measure of plant health: Agroecological aspects. Pestic. Fitomed. 2014, 29, 21–34. [Google Scholar] [CrossRef] [Green Version]
- Boyce, K.J.; Chang, H.; D’Souza, C.A.; Kronstad, J.W. An Ustilago maydis septin is required for filamentous growth in culture and for full symptom development on maize. Eukaryot Cell 2005, 4, 2044–2056. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.; Ting, J.T.L.; Sokolov, L.N.; Johnson, S.A.; Luan, S. A tumor suppressor homolog, AtPTEN1, is essential for pollen development in Arabidopsis. Plant Cell 2002, 14, 2495–2507. [Google Scholar] [CrossRef] [PubMed]
- Mattison, C.P.; Spencer, S.S.; Kresge, K.A.; Lee, J.; Ota, I.M. Differential regulation of the cell wall integrity mitogen-activated protein kinase pathway in budding yeast by the protein tyrosine phosphatases Ptp2 and Ptp3. Mol. Cell. Biol. 1999, 19, 7651–7660. [Google Scholar] [CrossRef]
- Bankoglu, E.E.; Tschopp, O.; Schmitt, J.; Burkard, P.; Jahn, D.; Geier, A.; Stopper, H. Role of PTEN in Oxidative Stress and DNA Damage in the Liver of Whole-Body Pten Haplodeficient Mice. PLoS ONE 2016, 11, e0166956. [Google Scholar] [CrossRef]
- Banuett, F.; Herskowitz, I. Discrete developmental stages during teliospore formation in the corn smut fungus, Ustilago maydis. Development 1996, 122, 2965–2976. [Google Scholar]
- Muller, P.; Katzenberger, J.D.; Loubradou, G.; Kahmann, R. Guanyl nucleotide exchange factor Sql2 and Ras2 regulate filamentous growth in Ustilago maydis. Eukaryot Cell 2003, 2, 609–617. [Google Scholar] [CrossRef]
- Salmena, L.; Carracedo, A.; Pandolfi, P.P. Tenets of PTEN Tumor Suppression. Cell 2008, 133, 403–414. [Google Scholar] [CrossRef]
- Flor-Parra, I.; Vranes, M.; Kamper, J.; Perez-Martin, J. Biz1, a zinc finger protein required for plant invasion by Ustilago maydis, regulates the levels of a mitotic cyclin. Plant Cell 2006, 18, 2369–2387. [Google Scholar] [CrossRef]
- Ma, J.; Jaraba, J.; Kirkpatrick, T.L.; Rothrock, C.S. Effects of Meloidogyne incognita and Thielaviopsis basicola on Cotton Growth and Root Morphology. Phytopathology 2013, 104, 507–512. [Google Scholar] [CrossRef] [PubMed]
- Scarpari, L.M.; Meinhardt, L.W.; Mazzafera, P.; Pomella, A.W.; Schiavinato, M.A.; Cascardo, J.C.; Pereira, G.A. Biochemical changes during the development of witches’ broom: The most important disease of cocoa in Brazil caused by Crinipellis perniciosa. J. Exp. Bot. 2005, 56, 865–877. [Google Scholar] [CrossRef] [PubMed]
- Lobato, A.K.S.; Gonçalves-Vidigal, M.C.; Vidigal Filho, P.S.; Andrade, C.A.B.; Kvitschal, M.V.; Bonato, C.M. Relationships between leaf pigments and photosynthesis in common bean plants infected by anthracnose. N. Z. J. Crop Horticult. Sci. 2010, 38, 29–37. [Google Scholar] [CrossRef] [Green Version]
- Lewandowski, T.J.; Dunfield, K.E.; Antunes, P.M. Isolate Identity Determines Plant Tolerance to Pathogen Attack in Assembled Mycorrhizal Communities. PLoS ONE 2013, 8, e61329. [Google Scholar] [CrossRef] [PubMed]
- Mitra, P.; Zhang, Y.; Rameh, L.E.; Ivshina, M.P.; McCollum, D.; Nunnari, J.J.; Hendricks, G.M.; Kerr, M.L.; Field, S.J.; Cantley, L.C.; et al. A novel phosphatidylinositol(3,4,5)P3 pathway in fission yeast. J. Cell Biol. 2004, 166, 205–211. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, P.; Zhou, Z.; Hu, A.; Ponte de Albuquerque, C.; Zhou, Y.; Hong, L.; Sierecki, E.; Ajiro, M.; Kruhlak, M.; Harris, C.; et al. Both Decreased and Increased SRPK1 Levels Promote Cancer by Interfering with PHLPP-Mediated Dephosphorylation of Akt. Mol. Cell 2014, 54, 378–391. [Google Scholar] [CrossRef]
- Reichmann, M.; Jamnischek, A.; Weinzierl, G.; Ladendorf, O.; Huber, S.; Kahmann, R.; Kamper, J. The histone deacetylase Hda1 from Ustilago maydis is essential for teliospore development. Mol. Microbiol. 2002, 46, 1169–1182. [Google Scholar] [CrossRef] [PubMed]
- Quadbeck-Seeger, C.; Wanner, G.; Huber, S.; Kahmann, R.; Kamper, J. A protein with similarity to the human retinoblastoma binding protein 2 acts specifically as a repressor for genes regulated by the b mating type locus in Ustilago maydis. Mol. Microbiol. 2000, 38, 154–166. [Google Scholar] [CrossRef]
- Flor-Parra, I.; Castillo-Lluva, S.; Pérez-Martín, J. Polar growth in the infectious hyphae of the phytopathogen Ustilago maydis depends on a virulence-specific cyclin. Plant Cell 2007, 19, 3280–3296. [Google Scholar] [CrossRef]
- Durrenberger, F.; Laidlaw, R.D.; Kronstad, J.W. The hgl1 gene is required for dimorphism and teliospore formation in the fungal pathogen Ustilago maydis. Mol. Microbiol. 2001, 41, 337–348. [Google Scholar] [CrossRef]
- Tollot, M.; Assmann, D.; Becker, C.; Altmüller, J.; Dutheil, J.Y.; Wegner, C.E.; Kahmann, R. The WOPR Protein Ros1 Is a Master Regulator of Sporogenesis and Late Effector Gene Expression in the Maize Pathogen Ustilago maydis. PLoS Pathog. 2016, 12, e1005697. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Fan, F.; Yang, J.; Wang, X.; Qiu, D.; Jiang, L. FgTep1p is linked to the phosphatidylinositol-3 kinase signalling pathway and plays a role in the virulence of Fusarium graminearum on wheat. Mol. Plant Pathol. 2010, 11, 495–502. [Google Scholar] [CrossRef] [PubMed]





| Ustilago Strain | Genotype a | References |
|---|---|---|
| 1/2 | a1b1 | [19] |
| 2/9 | a2b2 | [19] |
| 1/2 Δptn1 | a1b1 ptn1::cbxR | This study |
| 1/2 Δptn1J | a1b1 ptn1::hygR | This study |
| 2/9 Δptn1 | a2b2 ptn1::cbxR | This study |
| 2/9 Δptn1J | a2b2 ptn1::hygR | This study |
| FB1 Δptn1J | a1b1 ptn1::hygR | This study |
| 1/2 ptn1otef | a1b1 Potef-ump2, cbxR | This study |
| 2/9 ptn1otef | a2b2 Potef-ump2, cbxR | This study |
| SG200 | a1mfa2 bE1bW2 | [20] |
| SG200 Δptn1 | a1mfa2 bE1bW2 ptn1::hygR | This study |
| d132 | a1a2b1b2 | [21] |
| d132 Δptn1 | a1a2b1b2 ptn1:: hygR | This study |
| 1/2 Δptn1c | a1b1 ptn1::cbxR Potef-ptn1, hygR | This study |
| 2/9 Δptn1c | a2b2 ptn1::cbxR Potef-ptn1, hygR | This study |
| FBD11-7 | a1a1 b1b2 | [16] |
| FBD12-17 | a2a2 b1b2 | [16] |
| Plasmids | Genotype | References |
| DelsGate ptn1 | kanR ptn1::cbxR | [22] |
| Otef-ptn1-cbx | bla(ampR) Potef-ptn1 cbxR | This study |
| Otef-ptn1-hyg | bla(ampR) Potef-ptn1 hygR | This study |
| Primers | Sequence (5’-3’) |
|---|---|
| UPTN-K1 | TAGGGATAACAGGGTAATCAACAAAATGACGCAACCAC |
| UPTN-K3 | GGGACAAGTTTGTACAAAAAAGCAGGCTAAGAACGATGTTTGCGTGTCAG |
| UPTN-K4 | GGGGACCACTTTGTACAAGAAAGCTGGGTACGTTAGACAAAGCGCAATCA |
| UPTN-K2 | GGAAGGTGATCGTGAAGGAA |
| PTENUPLT | GCAACAAAATGACGCAACC |
| PTENUPRT | GTAGTTACCACGTTCGGCCATAAGGTGGTGGGACTGCTTT |
| PTENDNLT | GCTTTGCTCTTTGCTCTTCTG |
| CPDNRT | GCCTTAATTAATATACGCCCGATTGCTTACC |
| HYGPTENLT | AAAGCAGTCCCACCACCTTATGGCCGAACGTGGTAACTAC |
| HYGPTENRT | AGAAGAGCAAAGAGCAAAGCCTCAGGCCTCATGTTTGACA |
| PTEN5 | TGAGGCCTGAGTGGCCTATATACCCCCTGCCCCTGT |
| PTEN3 | CGAATTCTTGCGCTTTGTCTAACGAAC |
| HYGSPH1RTNEW | TTTGCATGCCTCAGGCCTCATGTTTGACA |
| HYGMFE1 | TTTCAATTGTGGCCGAACGTGGTAACTAC |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vijayakrishnapillai, L.M.K.; Desmarais, J.S.; Groeschen, M.N.; Perlin, M.H. Deletion of ptn1, a PTEN/TEP1 Orthologue, in Ustilago maydis Reduces Pathogenicity and Teliospore Development. J. Fungi 2019, 5, 1. https://doi.org/10.3390/jof5010001
Vijayakrishnapillai LMK, Desmarais JS, Groeschen MN, Perlin MH. Deletion of ptn1, a PTEN/TEP1 Orthologue, in Ustilago maydis Reduces Pathogenicity and Teliospore Development. Journal of Fungi. 2019; 5(1):1. https://doi.org/10.3390/jof5010001
Chicago/Turabian StyleVijayakrishnapillai, Lalu M. K., John S. Desmarais, Michael N. Groeschen, and Michael H. Perlin. 2019. "Deletion of ptn1, a PTEN/TEP1 Orthologue, in Ustilago maydis Reduces Pathogenicity and Teliospore Development" Journal of Fungi 5, no. 1: 1. https://doi.org/10.3390/jof5010001







