Repeated Exposition to Mercury (II) Chloride Enhances Susceptibility to S. schenckii sensu stricto Infection in Mice
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Experimental Models of HgCl2 Exposition
2.3. Peritoneal Macrophages and Splenocytes Preparation
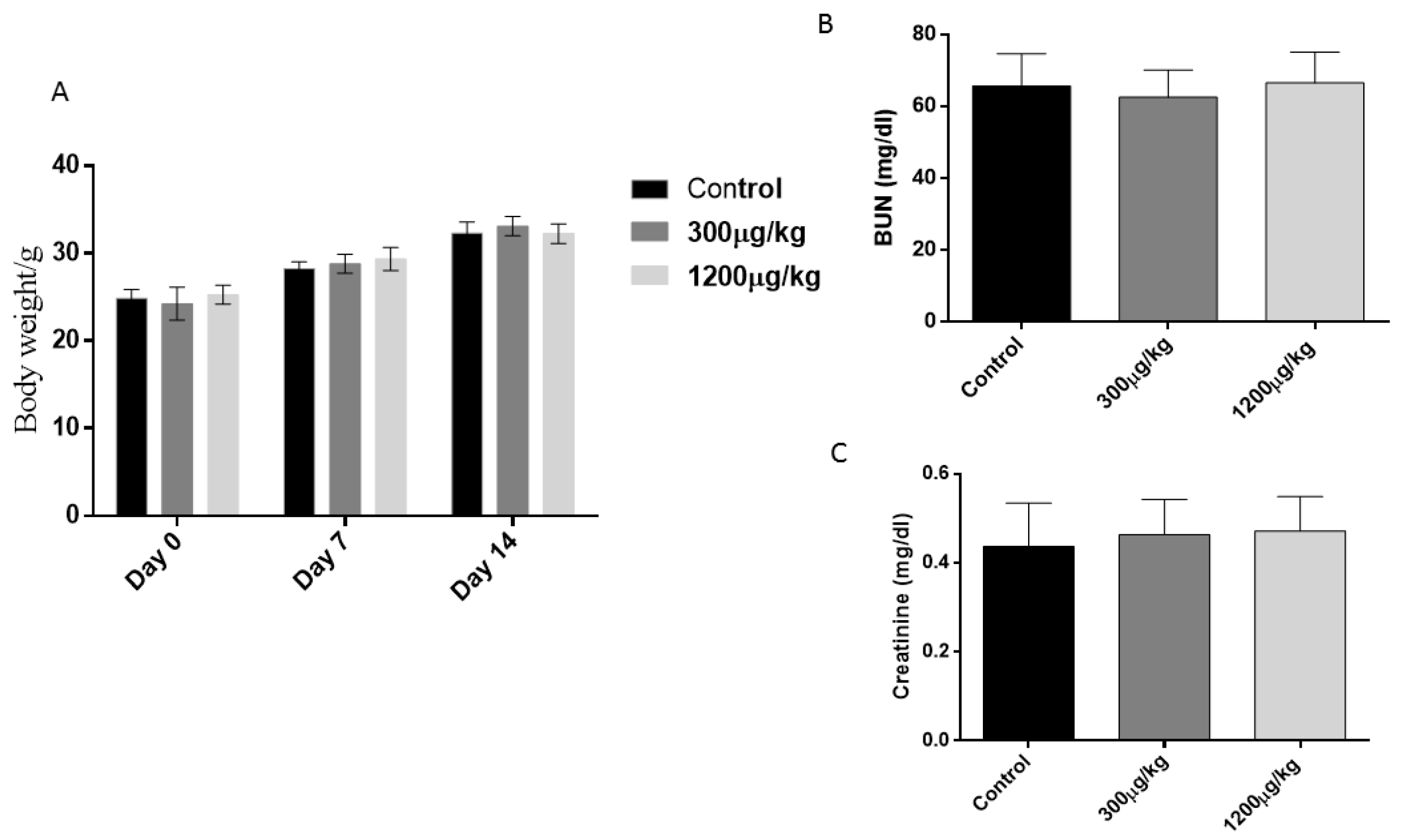
2.4. Blood Urea and Creatinine
2.5. Nitric Oxide (NO) and Cytokine Measurement
2.6. Th1/Th17 Cell Phenotyping and Flow Cytometry Analysis
2.7. Th1/Th2/Th17-Related Cytokines Analysis by Cytometric Bead Array (CBA)
2.8. Microorganism and Growth Conditions
2.9. Experimental Infection
2.10. Quantification of IgG anti Cell Wall Proteins of S. schenckii (ssCWP) by ELISA
2.11. Statistical Analysis
3. Results
3.1. General Toxicity Parameters
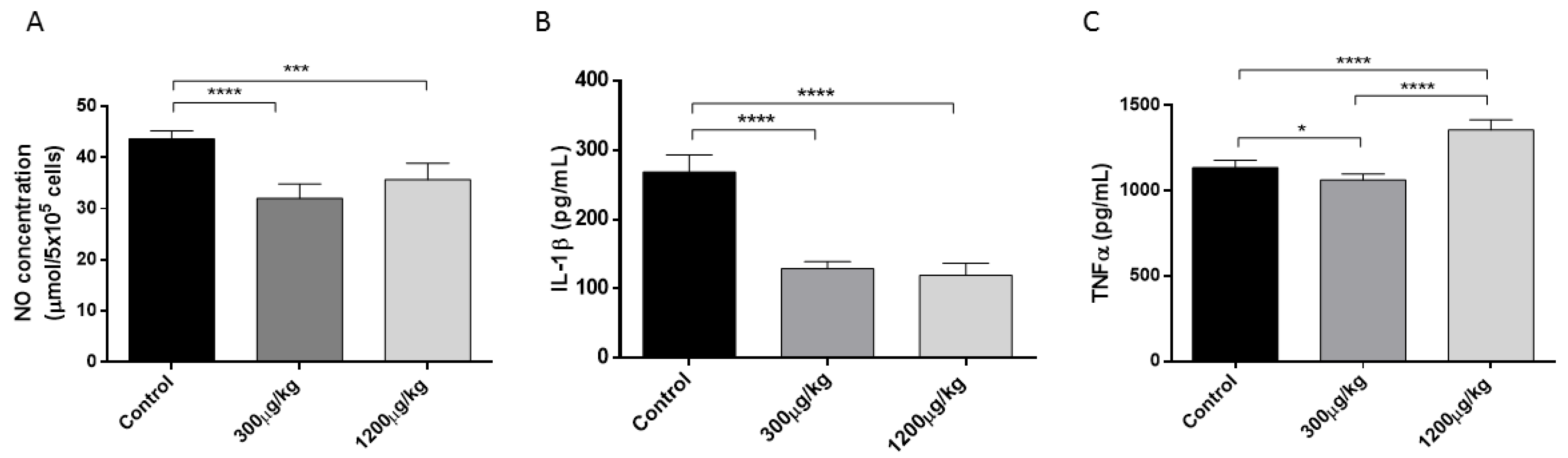
3.2. Mercury Reduced NO and IL-1 While Stimulated TNFα Production by Macrophages
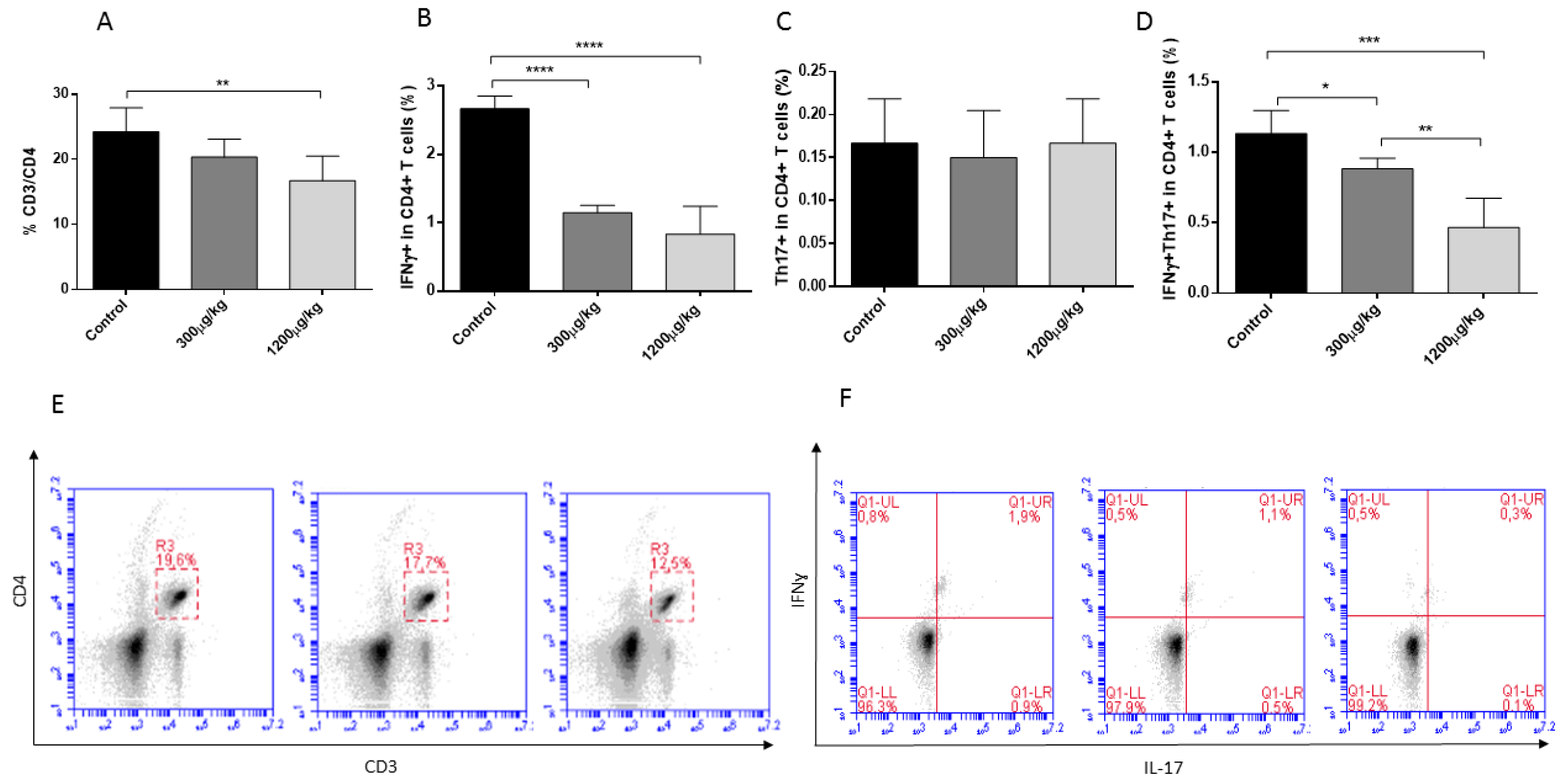
3.3. Mercury Caused Reduction of CD3+CD4+, Th1 and Th17 Lymphocytes
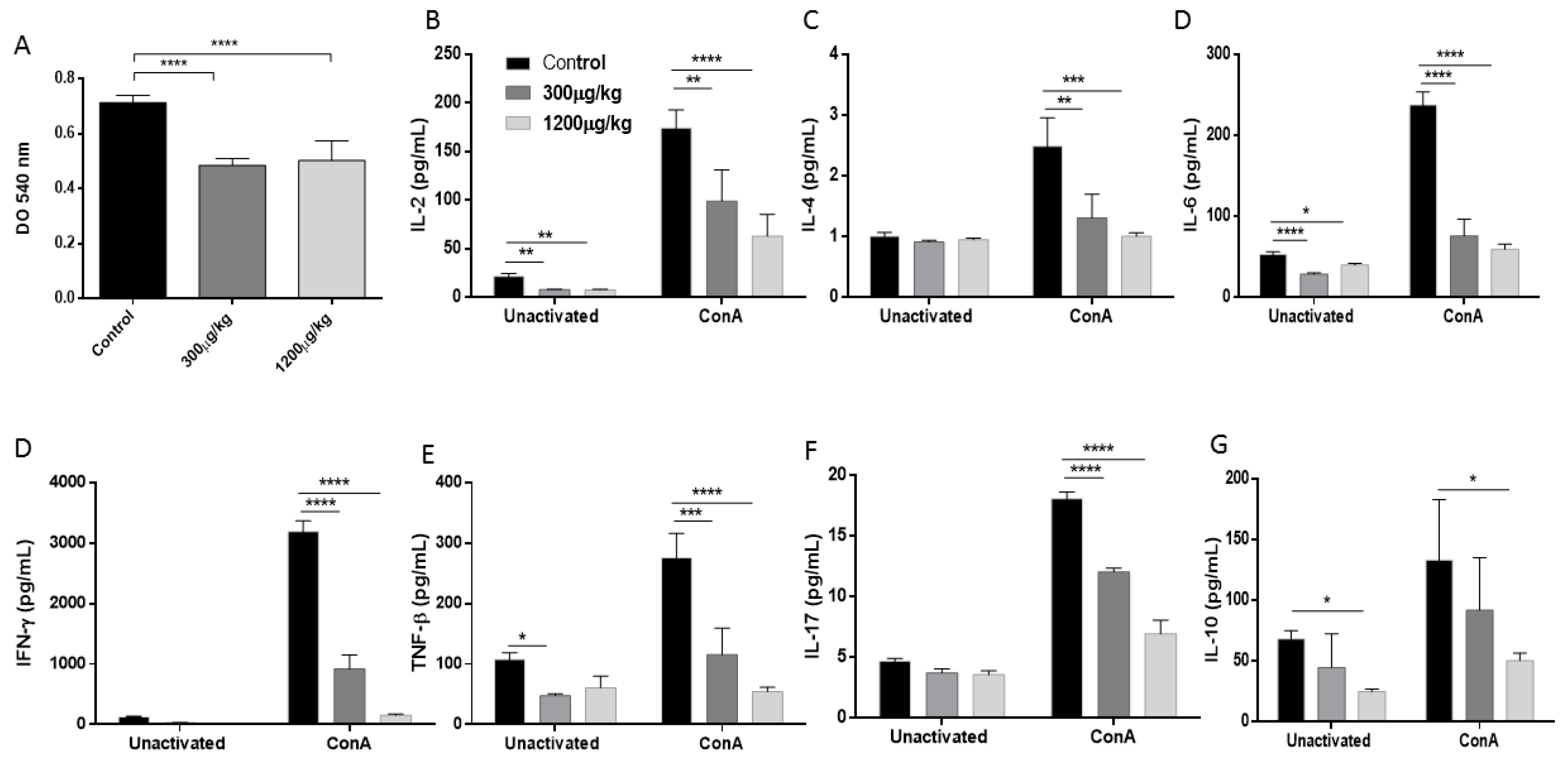
3.4. Mercury Inhibited the Splenocytes Proliferation and Reduced Th1/Th2/Th17 Cytokine Production
3.5. Mercury Enhanced Susceptibility to S. schenckii Infection
4. Discussion
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Gai, K.; Hoelen, T.P.; Hsu-Kim, H.; Lowry, G.V. Mobility of Four Common Mercury Species in Model and Natural Unsaturated Soils. Environ. Sci. Technol. 2016, 50, 3342–3351. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Exposure to Mercury: A Major Public Health Concern WHO, Public Health and Environment; WHO: Geneva, Switzerland, 2007. [Google Scholar]
- Kristensen, A.K.; Thomsen, J.F.; Mikkelsen, S. A review of mercury exposure among artisanal small-scale gold miners in developing countries. Int. Arch. Occup. Environ. Health 2014, 87, 579–590. [Google Scholar] [CrossRef] [PubMed]
- Steckling, N.; Tobollik, M.; Plass, D.; Hornberg, C.; Ericson, B.; Fuller, R.; Bose-O’Reilly, S. Global Burden of Disease of Mercury Used in Artisanal Small-Scale Gold Mining. Ann. Glob. Health 2017, 83, 234–247. [Google Scholar] [CrossRef] [PubMed]
- Esdaile, L.J.; Chalker, J.M. The Mercury Problem in Artisanal and Small-Scale Gold Mining. Chemistry 2018. [Google Scholar] [CrossRef] [PubMed]
- Borosková, Z.; Soltys, J.; Benková, M. Effect of mercury on the immune response and mean intensity of Ascaris suum infection in guinea pigs. J. Helminthol. 1995, 69, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Silbergeld, E.K.; Sacci, J.B., Jr.; Azad, A.F. Mercury exposure and murine response to Plasmodium yoelii infection and immunization. Immunopharmacol. Immunotoxicol. 2000, 22, 685–695. [Google Scholar] [PubMed]
- Bagenstose, L.M.; Mentink-Kane, M.M.; Brittingham, A.; Mosser, D.M.; Monestier, M. Mercury enhances susceptibility to murine leishmaniasis. Parasite Immunol. 2001, 23, 633–640. [Google Scholar] [CrossRef] [PubMed]
- South, P.K.; Morris, V.C.; Levander, O.A.; Smith, A.D. Mortality in mice infected with an amyocarditic coxsackievirus and given a subacute dose of mercuric chloride. J. Toxicol. Environ. Health A 2001, 63, 511–523. [Google Scholar] [CrossRef] [PubMed]
- Ekerfelt, C.; Andersson, M.; Olausson, A.; Bergström, S.; Hultman, P. Mercury exposure as a model for deviation of cytokine responses in experimental Lyme arthritis: HgCl2 treatment decreases T helper cell type 1-like responses and arthritis severity but delays eradication of Borrelia burgdorferi in C3H/HeN mice. Clin. Exp. Immunol. 2007, 150, 189–197. [Google Scholar] [CrossRef] [PubMed]
- Orofino-Costa, R.; Macedo, P.M.; Rodrigues, A.M.; Bernardes-Engemann, A.R. Sporotrichosis: An update on epidemiology, etiopathogenesis, laboratory and clinical therapeutics. An. Bras. Dermatol. 2017, 92, 606–620. [Google Scholar] [CrossRef] [PubMed]
- Carlos, I.Z.; Batista-Duharte, A. Sporotrichosis: An emergent disease. In Sporotrichosis; Springer International Publishing: Cham, Switzerland, 2015; pp. 1–23. [Google Scholar]
- Chakrabarti, A.; Bonifaz, A.; Gutierrez-Galhardo, M.C.; Mochizuki, T.; Li, S. Global epidemiology of sporotrichosis. Med. Mycol. 2015, 53, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Lopes-Bezerra, L.M.; Mora-Montes, H.M.; Zhang, Y.; Nino-Vega, G.; Rodrigues, A.M.; de Camargo, Z.P.; de Hoog, S. Sporotrichosis between 1898 and 2017: The evolution of knowledge on a changeable disease and on emerging etiological agents. Med. Mycol. 2018, 56, 126–143. [Google Scholar] [CrossRef] [PubMed]
- Almeida-Paes, R.; de Oliveira, L.C.; Oliveira, M.M.; Gutierrez-Galhardo, M.C.; Nosanchuk, J.D.; Zancopé-Oliveira, R.M. Phenotypic characteristics associated with virulence of clinical isolates from the Sporothrix complex. Biomed. Res. Int. 2015, 2015, 212308. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.P.; Lau, Y.L. Cellular and Molecular Defects Underlying Invasive Fungal Infections-Revelations from Endemic Mycoses. Front. Immunol. 2017, 8, 735. [Google Scholar] [CrossRef] [PubMed]
- Batista-Duharte, A.; Martínez, D.T.; da Graça Sgarbi, D.B.; Carlos, I.Z. Environmental Conditions and Fungal Pathogenicity. In Sporotrichosis; Zeppone Carlos, I., Ed.; Springer: Cham, Switzarland, 2015. [Google Scholar]
- Téllez, M.D.; Batista-Duharte, A.; Portuondo, D.; Quinello, C.; Bonne-Hernández, R.; Carlos, I.Z. Sporothrix schenckii complex biology: Environment and fungal pathogenicity. Microbiology 2014, 160, 2352–2365. [Google Scholar] [CrossRef] [PubMed]
- Dixon, D.M.; Salkin, I.F.; Duncan, R.A.; Hurd, N.J.; Haines, J.H.; Kemna, M.E.; Coles, F.B. Isolation and characterization of Sporothrix schenckii from clinical and environmental sources associated with the largest U.S. epidemic of sporotrichosis. J. Clin. Microbiol. 1991, 29, 1106–1113. [Google Scholar] [PubMed]
- Ulfig, K. The occurrence of keratinolytic fungi in the polluted environment of the Labedy district in Gliwice. Rocz. Panstw. Zakl. Hig. 1994, 45, 337–346. [Google Scholar] [PubMed]
- Pijper, A.; Pulinger, D.B. An outbreak of sporotrichosis among South African native miners. Lancet 1927, 210, 914–916. [Google Scholar] [CrossRef]
- Dangerfield, L.F.; Gear, J. Sporotrichosis among miners on the Witwatersrand gold mines. S. Afr. Med. J. 1941, 15, 128–131. [Google Scholar]
- Findlay, G.H. Sporotrichosis research in the Transvaal—How it began 60 years ago. S. Afr. Med. J. 1985, 68, 117–118. [Google Scholar] [PubMed]
- Findlay, G.H. The epidemiology of sporotrichosis in the Transvaal. Sabouraudia 1970, 7, 231–236. [Google Scholar] [CrossRef] [PubMed]
- Helm, M.A.F.; Berman, C. The clinical, therapeutic and epidemiological features of the sporotrichosis infection on the mines. In Sporotrichosis Infection on Mines of the Witwatersrand; Proceedings of the Transvaal Mine Medical Officers’ Association, December 1944; The Transvaal Chamber of Mines: Johannesburg, South Africa, 1947; pp. 59–67. [Google Scholar]
- Quintal, D. Sporotrichosis infection on mines of the Witwatersrand. J. Cutan. Med. Surg. 2000, 4, 51–54. [Google Scholar] [CrossRef] [PubMed]
- Govender, N.P.; Maphanga, T.G.; Zulu, T.G.; Patel, J.; Walaza, S.; Jacobs, C.; Ebonwu, J.I.; Ntuli, S.; Naicker, S.D.; Thomas, J. An outbreak of lymphocutaneous sporotrichosis among mine-workers in South Africa. PLoS Negl. Trop. Dis. 2015, 9, e0004096. [Google Scholar] [CrossRef] [PubMed]
- Queiroz-Telles, F.; McGinnis, M.R.; Salkin, I.; Graybill, J.R. Subcutaneous mycoses. Infect. Dis. Clin. N. Am. 2003, 17, 59–85. [Google Scholar] [CrossRef]
- Gonçalves, A.C.; Maia, D.C.; Ferreira, L.S.; Monnazzi, L.G.; Alegranci, P.; Placeres, M.C.; Batista-Duharte, A.; Carlos, I.Z. Involvement of major components from Sporothrix schenckii cell wall in the caspase-1 activation, nitric oxide and cytokines production during experimental sporotrichosis. Mycopathologia 2015, 179, 21–30. [Google Scholar]
- Ferreira, L.S.; Gonçalves, A.C.; Portuondo, D.L.; Maia, D.C.; Placeres, M.C.; Batista-Duharte, A.; Carlos, I.Z. Optimal clearance of Sporothrix schenckii requires an intact Th17 response in a mouse model of systemic infection. Immunobiology 2015, 220, 985–992. [Google Scholar] [CrossRef] [PubMed]
- Cook, J.G.H. Creatinine assay in the presence of protein. Clin. Chim. Acta 1971, 32, 485–486. [Google Scholar] [CrossRef]
- Bernt, E.; Bergmeyer, H.U. Urea, p 401–406. In Methods of Enzymatic Analysis, 2nd ed.; Bergmeyer, H.U., Ed.; Academic Press: New York, NY, USA, 1965. [Google Scholar]
- Castro, R.A.; Kubitschek-Barreira, P.H.; Teixeira, P.A.; Sanches, G.F.; Teixeira, M.M.; Quintella, L.P.; Almeida, S.R.; Costa, R.O.; Camargo, Z.P.; Felipe, M.S.; et al. Differences in cell morphometry, cell wall topography and Gp70 expression correlate with the virulence of Sporothrix brasiliensis clinical isolates. PLoS ONE 2013, 8, e75656. [Google Scholar] [CrossRef] [PubMed]
- Portuondo, D.L.; Batista-Duharte, A.; Ferreira, L.S.; Martínez, D.T.; Polesi, M.C.; Duarte, R.A.; de Paula, E.; Silva, A.C.; Marcos, C.M.; Almeida, A.M.; et al. A cell wall protein-based vaccine candidate induce protective immune response against Sporothrix schenckii infection. Immunobiology 2016, 221, 300–309. [Google Scholar] [CrossRef] [PubMed]
- Silbergeld, E.K.; Silva, I.A.; Nyland, J.F. Mercury and autoimmunity: Implications for occupational and environmental health. Toxicol. Appl. Pharmacol. 2005, 207, S282–S292. [Google Scholar] [CrossRef] [PubMed]
- Silva, I.A.; Nyland, J.F.; Gorman, A.; Perisse, A.; Ventura, A.M.; Santos, E.C.; Souza, J.M.; Burek, C.L.; Rose, N.R.; Silbergeld, E.K. Mercury exposure, malaria, and serum antinuclear/antinucleolar antibodies in Amazon populations in Brazil: A cross-sectional study. Environ. Health 2004, 3, 11. [Google Scholar] [CrossRef] [PubMed]
- Silva, I.A.; Graber, J.; Nyland, J.F.; Silbergeld, E.K. In vitro HgCl2 exposure of immune cells at different stages of maturation: Effects on phenotype and function. Environ. Res. 2005, 98, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Eagles-Smith, C.A.; Silbergeld, E.K.; Basu, N.; Bustamante, P.; Diaz-Barriga, F.; Hopkins, W.A.; Kidd, K.A.; Nyland, J.F. Modulators of mercury risk to wildlife and humans in the context of rapid global change. Ambio 2018, 47, 170–197. [Google Scholar] [CrossRef] [PubMed]
- Gray, J.A.; Kavlock, R.J. Pharmacologic probing of mercuric chloride-induced renal dysfunction in the neonatal rat. J. Pharmacol. Exp. Ther. 1987, 242, 212–216. [Google Scholar] [PubMed]
- Hultman, P.; Eneström, S. Mercury induced antinuclear antibodies in mice: Characterization and correlation with renal immune complex deposits. Clin. Exp. Immunol. 1988, 71, 269–274. [Google Scholar] [PubMed]
- Hultman, P.; Bell, L.J.; Eneström, S.; Pollard, K.M. Murine susceptibility to mercury. II. autoantibody profiles and renal immune deposits in hybrid, backcross, and H-2d congenic mice. Clin. Immunol. Immunopathol. 1993, 68, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Fiuza Tda, L.; Oliveira, C.S.; da Costa, M.; Oliveira, V.A.; Zeni, G.; Pereira, M.E. Effectiveness of (PhSe)2 in protect against the HgCl2 toxicity. J. Trace Elem. Med. Biol. 2015, 29, 255–262. [Google Scholar] [CrossRef] [PubMed]
- Crowe, W.; Allsopp, P.J.; Watson, G.E.; Magee, P.J.; Strain, J.J.; Armstrong, D.J.; Ball, E.; McSorley, E.M. Mercury as an environmental stimulus in the development of autoimmunity—A systematic review. Autoimmun. Rev. 2017, 16, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Dieter, M.; Luster, M.I.; Boorman, G.A. Immunological and biochemical responses in mice treated with mercuric chloride. Toxicol. Appl. Pharmacol. 1983, 68, 218–228. [Google Scholar] [CrossRef]
- Carlos, I.Z.; Zini, M.M.; Sgarbi, D.B.; Angluster, J.; Alviano, C.S.; Silva, C.L. Disturbances in the production of interleukin-1 and tumor necrosis factor in disseminated murine sporotrichosis. Mycopathologia 1994, 127, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Maia, D.C.; Gonçalves, A.C.; Ferreira, L.S.; Manente, F.A.; Portuondo, D.L.; Vellosa, J.C.; Polesi, M.C.; Batista-Duharte, A.; Carlos, I.Z. Response of Cytokines and Hydrogen Peroxide to Sporothrix schenckii Exoantigen in Systemic Experimental Infection. Mycopathologia 2016, 181, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Jellmayer, J.A.; Ferreira, L.S.; Manente, F.A.; Gonçalves, A.C.; Polesi, M.C.; Batista-Duharte, A.; Carlos, I.Z. Dectin-1 expression by macrophages and related antifungal mechanisms in a murine model of Sporothrix schenckii sensu stricto systemic infection. Microb. Pathog. 2017, 110, 78–84. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Johnson, V.J.; Sharma, R.P. Mercury inhibits nitric oxide production but activates proinflammatory cytokine expression in murine macrophage: Differential modulation of NF-kappaB and p38 MAPK signaling pathways. Nitric Oxide 2002, 7, 67–74. [Google Scholar] [CrossRef]
- Iwai-Shimada, M.; Takahashi, T.; Kim, M.S.; Fujimura, M.; Ito, H.; Toyama, T.; Naganuma, A.; Hwang, G.W. Methylmercury induces the expression of TNF-α selectively in the brain of mice. Sci. Rep. 2016, 6, 38294. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Li, Y.; Rao, J.; Liu, Z.; Chen, Q. Effects of inorganic mercury exposure on histological structure, antioxidant status and immune response of immune organs in yellow catfish (Pelteobagrus fulvidraco). J. Appl. Toxicol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Dieguez-Acuña, F.J.; Woods, J.S. Inhibition of NF-κB-DNA binding by mercuric ion: Utility of the non-thiol reductant, tris(2-carboxyethyl)phosphine hydrochloride (TCEP), on detection of impaired NF-kappaB-DNA binding by thiol-directed agents. Toxicol. In Vitro 2000, 14, 7–16. [Google Scholar] [CrossRef]
- Shumilla, J.A.; Wetterhahn, K.E.; Barchowsky, A. Inhibition of NF-κB binding to DNA by chromium, cadmium, mercury, zinc, and arsenite in vitro: Evidence of a thiol mechanism. Arch. Biochem. Biophys. 1998, 349, 356–362. [Google Scholar] [CrossRef] [PubMed]
- Park, H.J.; Youn, H.S. Mercury induces the expression of cyclooxygenase-2 and inducible nitric oxide synthase. Toxicol. Ind. Health 2013, 29, 169–174. [Google Scholar] [CrossRef] [PubMed]
- Fournié, G.J.; Saoudi, A.; Druet, P.; Pelletier, L. Th2-type immunopathological manifestations induced by mercury chloride or gold salts in the rat: Signal transduction pathways, cellular mechanisms and genetic control. Autoimmun. Rev. 2002, 1, 205–212. [Google Scholar] [CrossRef]
- Maia, D.C.G.; Sassá, M.F.; Placeres, M.C.P.; Carlos, I.Z. Influence of Th1/Th2 cytokines and nitric oxide in murine systemic infection induced by Sporothrix schenckii. Mycopathologia 2006, 161, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Uenotsuchi, T.; Takeuchi, S.; Matsuda, T.; Urabe, K.; Koga, T.; Uchi, H.; Nakahara, T.; Fukagawa, S.; Kawasaki, M.; Kajiwara, H.; et al. Differential induction of Th1-prone immunity by human dendritic cells activated with Sporothrix schenckii of cutaneous and visceral origins to determine their different virulence. Int. Immunol. 2006, 18, 1637–1646. [Google Scholar] [CrossRef] [PubMed]
- Flores-García, A.; Velarde-Félix, J.S.; Garibaldi-Becerra, V.; Rangel-Villalobos, H.; Torres-Bugarín, O.; Zepeda-Carrillo, E.A.; Ruíz-Bernés, S.; Ochoa-Ramírez, L.A. Recombinant murine IL-12 promotes a protective Th1/cellular response in Mongolian gerbils infected with Sporothrix schenckii. J. Chemother. 2015, 27, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Manente, F.A.; Quinello, C.; Ferreira, L.S.; de Andrade, C.R.; Jellmayer, J.A.; Portuondo, D.L.; Batista-Duharte, A.; Carlos, I.Z. Experimental sporotrichosis in a cyclophosphamide-induced immunosuppressed mice model. Med. Mycol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Portuondo, D.L.; Batista-Duharte, A.; Ferreira, L.S.; de Andrade, C.R.; Quinello, C.; Téllez-Martínez, D.; de Aguiar Loesch, M.L.; Carlos, I.Z. Comparative efficacy and toxicity of two vaccine candidates against Sporothrix schenckii using either Montanide™ Pet Gel A or aluminum hydroxide adjuvants in mice. Vaccine 2017, 35, 4430–4436. [Google Scholar] [CrossRef] [PubMed]
- Ochel, M.; Vohr, H.W.; Pfeiffer, C.; Gleichmann, E. IL-4 is required for the IgE and IgG1 increase and IgG1 autoantibody formation in mice treated with mercuric chloride. J. Immunol. 1991, 146, 3006–3011. [Google Scholar] [PubMed]
- Arefieva, A.S.; Kamaeva, A.G.; Krasilshchikova, M.S. Low doses of mercuric chloride cause the main features of anti-nucleolar autoimmunity in female outbred CFW mice. Toxicol. Ind. Health 2016, 32, 1663–1674. [Google Scholar] [CrossRef] [PubMed]
- De Vos, G.; Abotaga, S.; Liao, Z.; Jerschow, E.; Rosenstreich, D. Selective effect of mercury on Th2-type cytokine production in humans. Immunopharmacol. Immunotoxicol. 2007, 29, 537–548. [Google Scholar] [CrossRef] [PubMed]
- Germann, T.; Bongartz, M.; Dlugonska, H.; Hess, H.; Schmitt, E.; Kolbe, L.; Kölsch, E.; Podlaski, F.J.; Gately, M.K.; Rüde, E. Interleukin-12 profoundly up-regulates the synthesis of antigen-specific complement-fixing IgG2a, IgG2b and IgG3 antibody subclasses in vivo. Eur. J. Immunol. 1995, 25, 823–829. [Google Scholar] [CrossRef] [PubMed]
- Abedi-Valugerdi, M.; Möller, G. Contribution of H-2 and non-H-2 genes in the control of mercury-induced autoimmunity. Int. Immunol. 2000, 12, 1425–1430. [Google Scholar] [CrossRef] [PubMed]
- Gibb, H.; O’Leary, K.G. Mercury exposure and health impacts among individuals in the artisanal and small-scale gold mining community: A comprehensive review. Environ. Health Perspect. 2014, 122, 667–672. [Google Scholar] [CrossRef] [PubMed]
- Eisler, R. Health risks of gold miners: A synoptic review. Environ. Geochem. Health 2003, 25, 325–345. [Google Scholar] [CrossRef] [PubMed]
- Douine, M.; Mosnier, E.; Le Hingrat, Q.; Charpentier, C.; Corlin, F.; Hureau, L.; Adenis, A.; Lazrek, Y.; Niemetsky, F.; Aucouturier, A.L.; et al. Illegal gold miners in French Guiana: A neglected population with poor health. BMC Public Health 2017, 18, 23. [Google Scholar] [CrossRef] [PubMed]
- Lubick, N. Mercury alters immune system response in artisanal gold miners. Environ. Health Perspect. 2010, 118, A243. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Batista-Duharte, A.; Téllez-Martínez, D.; Aparecida Jellmayer, J.; Leandro Portuondo Fuentes, D.; Campos Polesi, M.; Martins Baviera, A.; Zeppone Carlos, I. Repeated Exposition to Mercury (II) Chloride Enhances Susceptibility to S. schenckii sensu stricto Infection in Mice. J. Fungi 2018, 4, 64. https://doi.org/10.3390/jof4020064
Batista-Duharte A, Téllez-Martínez D, Aparecida Jellmayer J, Leandro Portuondo Fuentes D, Campos Polesi M, Martins Baviera A, Zeppone Carlos I. Repeated Exposition to Mercury (II) Chloride Enhances Susceptibility to S. schenckii sensu stricto Infection in Mice. Journal of Fungi. 2018; 4(2):64. https://doi.org/10.3390/jof4020064
Chicago/Turabian StyleBatista-Duharte, Alexander, Damiana Téllez-Martínez, Juliana Aparecida Jellmayer, Deivys Leandro Portuondo Fuentes, Marisa Campos Polesi, Amanda Martins Baviera, and Iracilda Zeppone Carlos. 2018. "Repeated Exposition to Mercury (II) Chloride Enhances Susceptibility to S. schenckii sensu stricto Infection in Mice" Journal of Fungi 4, no. 2: 64. https://doi.org/10.3390/jof4020064
APA StyleBatista-Duharte, A., Téllez-Martínez, D., Aparecida Jellmayer, J., Leandro Portuondo Fuentes, D., Campos Polesi, M., Martins Baviera, A., & Zeppone Carlos, I. (2018). Repeated Exposition to Mercury (II) Chloride Enhances Susceptibility to S. schenckii sensu stricto Infection in Mice. Journal of Fungi, 4(2), 64. https://doi.org/10.3390/jof4020064






