A Review of Ethnoveterinary Knowledge, Biological Activities and Secondary Metabolites of Medicinal Woody Plants Used for Managing Animal Health in South Africa
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Overview of Eligible Literature and Ethnoveterinary Studies
3.2. Inventory of Woody Plants with Ethnoveterinary Uses
3.3. Overview of Animals/Livestock and Diseases
3.4. In Vitro Biological Screening of Woody Plants
3.4.1. Antibacterial Activity
3.4.2. Anthelmintic Activity
3.4.3. Antioxidant Activity
3.4.4. Cytotoxicity
3.5. Phytochemical Analysis of Plants Used for Ethnoveterinary Purposes
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Comaroff, J.L.; Comaroff, J. Goodly beasts, beastly goods: Cattle and commodities in a South African context. Am. Ethnol. 1990, 17, 195–216. [Google Scholar] [CrossRef]
- Mapiye, O.; Chikwanha, O.C.; Makombe, G.; Dzama, K.; Mapiye, C. Livelihood, food and nutrition security in Southern Africa: What role do indigenous cattle genetic resources play? Diversity 2020, 12, 74. [Google Scholar] [CrossRef]
- Suroowan, S.; Javeed, F.; Ahmad, M.; Zafar, M.; Noor, M.J.; Kayani, S.; Javed, A.; Mahomoodally, M.F. Ethnoveterinary health management practices using medicinal plants in South Asia—A review. Vet. Res. Commun. 2017, 41, 147–168. [Google Scholar] [CrossRef]
- Lans, C.; Sant, C.; Georges, K. Ethnoremedies used for horses in British Columbia and Trinidad and Tobago. In Ethnoveterinary Medicine: Present and Future Concepts; McGaw, L.J., Abdalla, M.A., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 57–71. [Google Scholar] [CrossRef]
- Borges, A.K.M.; Barboza, R.R.D.; Souto, W.M.S.; Alves, R.R.N. Natural remedies for animal health in Latin America. In Ethnoveterinary Medicine: Present and Future Concepts; McGaw, L.J., Abdalla, M.A., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 311–344. [Google Scholar] [CrossRef]
- Mayer, M.; Zbinden, M.; Vogl, C.R.; Ivemeyer, S.; Meier, B.; Amorena, M.; Maeschli, A.; Hamburger, M.; Walkenhorst, M. Swiss ethnoveterinary knowledge on medicinal plants—A within-country comparison of Italian speaking regions with north-western German speaking regions. J. Ethnobiol. Ethnomed. 2017, 13, 1. [Google Scholar] [CrossRef]
- Galaty, J.G. Being “Maasai”; being “People-of-cattle”: Ethnic shifters in East Africa. Am. Ethnol. 1982, 9, 1–20. [Google Scholar] [CrossRef]
- Kazancı, C.; Oruç, S.; Mosulishvili, M. Ethnoveterinary and fodder plants used among transhumant communities around Georgia-Turkey border, in the Western Lesser Caucasus. Ethnobot. Res. Appl. 2021, 21, 28. [Google Scholar] [CrossRef]
- McCorkle, C.M. Back to the future: Lessons from ethnoveterinary RD&E for studying and applying local knowledge. Agric. Hum. Values 1995, 12, 52–80. [Google Scholar] [CrossRef]
- Khan, K.; Rahman, I.U.; Calixto, E.S.; Ali, N.; Ijaz, F. Ethnoveterinary therapeutic practices and conservation status of the medicinal flora of Chamla valley, Khyber Pakhtunkhwa, Pakistan. Front. Vet. Sci. 2019, 6, 122. [Google Scholar] [CrossRef] [PubMed]
- Shrubok, A. Belarusian ethnoveterinary medicine: Ritual practices and traditional remedies. In Ethnoveterinary Medicine: Present and Future Concepts; McGaw, L.J., Abdalla, M.A., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 375–390. [Google Scholar] [CrossRef]
- Zorloni, A. Toward a better understanding of African ethnoveterinary medicine and husbandry. In Ethnoveterinary Medicine: Present and Future Concepts; McGaw, L.J., Abdalla, M.A., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 151–172. [Google Scholar] [CrossRef]
- McCorkle, C.M. An introduction to ethnoveterinary research and development. J. Ethnobiol. 1986, 6, 129–149. [Google Scholar]
- Aziz, M.A.; Khan, A.H.; Pieroni, A. Ethnoveterinary plants of Pakistan: A review. J. Ethnobiol. Ethnomed. 2020, 16, 25. [Google Scholar] [CrossRef] [PubMed]
- McGaw, L.J.; Famuyide, I.M.; Khunoana, E.T.; Aremu, A.O. Ethnoveterinary botanical medicine in South Africa: A review of research from the last decade (2009 to 2019). J. Ethnopharmacol. 2020, 257, 112864. [Google Scholar] [CrossRef] [PubMed]
- Hoveka, L.N.; van der Bank, M.; Davies, T.J. Evaluating the performance of a protected area network in South Africa and its implications for megadiverse countries. Biol. Conserv. 2020, 248, 108577. [Google Scholar] [CrossRef]
- Germishuizen, G.; Meyer, N. Plants of Southern Africa: An Annotated Checklist; National Botanical Institute Pretoria: Pretoria, South Africa, 2003. [Google Scholar]
- Van Staden, J. Ethnobotany in South Africa. J. Ethnopharmacol. 2008, 119, 329–330. [Google Scholar] [CrossRef]
- Da Silva, T.C.; da Silva, J.M.; Ramos, M.A. What factors guide the selection of medicinal plants in a local pharmacopoeia? A case study in a rural community from a historically transformed atlantic forest landscape. Evid.-Based Complement. Altern. Med. 2018, 2018, 2519212. [Google Scholar] [CrossRef]
- Muleba, I.; Yessoufou, K.; Rampedi, I.T. Testing the non-random hypothesis of medicinal plant selection using the woody flora of the Mpumalanga Province, South Africa. Environ. Dev. Sustain. 2021, 23, 4162–4173. [Google Scholar] [CrossRef]
- De Albuquerque, U.P.; Farias Paiva de Lucena, R. Can apparency affect the use of plants by local people in tropical forests? Interciencia 2005, 30, 506–510. [Google Scholar]
- Douwes, E.; Crouch, N.R.; Edwards, T.J.; Mulholland, D.A. Regression analyses of southern African ethnomedicinal plants: Informing the targeted selection of bioprospecting and pharmacological screening subjects. J. Ethnopharmacol. 2008, 119, 356–364. [Google Scholar] [CrossRef]
- Daru, B.H.; Berger, D.K.; Wyk, A.E. Opportunities for unlocking the potential of genomics for African trees. New Phytol. 2016, 210, 772–778. [Google Scholar] [CrossRef]
- Venter, F.; Venter, J.-A. Making the Most of Indigenous Trees; Briza Publications: Pretoria, South Africa, 2002. [Google Scholar]
- Van Wyk, A.S.; Prinsloo, G. A review of the ethnobotanical uses, pharmacology, toxicology, management and cultivation of selected South African protected multi-purpose tree species. S. Afr. J. Bot. 2019, 124, 258–269. [Google Scholar] [CrossRef]
- Moyo, M.; Aremu, A.O.; Van Staden, J. Medicinal plants: An invaluable, dwindling resource in sub-Saharan Africa. J. Ethnopharmacol. 2015, 174, 595–606. [Google Scholar] [CrossRef] [PubMed]
- Yaoitcha, A.S.; Houehanou, T.D.; Fandohan, A.B.; Houinato, M.R.B. Prioritization of useful medicinal tree species for conservation in Wari-Maro Forest Reserve in Benin: A multivariate analysis approach. For. Policy Econ. 2015, 61, 135–146. [Google Scholar] [CrossRef]
- Yirgu, A.; Mohammed, K.; Geldenhuys, C.J. Useful medicinal tree species of Ethiopia: Comprehensive review. S. Afr. J. Bot. 2019, 122, 291–300. [Google Scholar] [CrossRef]
- Ahoyo, C.C.; Houéhanou, T.D.; Yaoïtcha, A.S.; Prinz, K.; Glèlè Kakaï, R.; Sinsin, B.A.; Houinato, M.R.B. Traditional medicinal knowledge of woody species across climatic zones in Benin (West Africa). J. Ethnopharmacol. 2021, 265, 113417. [Google Scholar] [CrossRef] [PubMed]
- Van Andel, T.; Myren, B.; van Onselen, S. Ghana’s herbal market. J. Ethnopharmacol. 2012, 140, 368–378. [Google Scholar] [CrossRef]
- Rivera, D.; Allkin, R.; Obón, C.; Alcaraz, F.; Verpoorte, R.; Heinrich, M. What is in a name? The need for accurate scientific nomenclature for plants. J. Ethnopharmacol. 2014, 152, 393–402. [Google Scholar] [CrossRef]
- Weckerle, C.S.; de Boer, H.J.; Puri, R.K.; van Andel, T.; Bussmann, R.W.; Leonti, M. Recommended standards for conducting and reporting ethnopharmacological field studies. J. Ethnopharmacol. 2018, 210, 125–132. [Google Scholar] [CrossRef]
- Chitura, T.; Muvhali, P.; Shai, K.; Mushonga, B.; Kandiwa, E. Use of medicinal plants by livestock farmers in a local municipality in Vhembe district, South Africa. Appl. Ecol. Environ. Res. 2018, 16, 6589–6605. [Google Scholar] [CrossRef]
- Dold, A.P.; Cocks, M.L. Traditional veterinary medicine in the Alice district of the Eastern Cape Province, South Africa. S. Afr. J. Sci. 2001, 97, 375–379. [Google Scholar]
- Kambizi, L. Indigenous plants for ethnoveterinary uses in the Pondoland, South Africa. In Proceedings of the XXIX International Horticultural Congress on Horticulture: Sustaining Lives, Livelihoods and Landscapes: V World Congress on Medicinal and Aromatic Plants and International Symposium on Plants, as Factories of Natural Substances, Edible and Essential Oils, Brisbane, QLD, Australia, 17 August 2014; pp. 309–314. [Google Scholar]
- Khunoana, E.T.; Madikizela, B.; Erhabor, J.O.; Nkadimeng, S.M.; Arnot, L.F.; Van Wyk, I.; McGaw, L.J. A survey of plants used to treat livestock diseases in the Mnisi community, Mpumalanga, South Africa, and investigation of their antimicrobial activity. S. Afr. J. Bot. 2019, 126, 21–29. [Google Scholar] [CrossRef]
- Luseba, D.; Tshisikhawe, M. Medicinal plants used in the treatment of livestock diseases in Vhembe region, Limpopo province, South Africa. J. Med. Plants Res. 2013, 7, 593–601. [Google Scholar]
- Luseba, D.; Van der Merwe, D. Ethnoveterinary medicine practices among Tsonga speaking people of South Africa. Onderstepoort J. Vet. Res. 2006, 73, 115–122. [Google Scholar] [CrossRef]
- Magwede, K.; Tshisikhawe, M.P.; Luseba, D.; Bhat, R.B. Ethnobotanical survey of medicinal plants used in treatment of ticks. Int. J. Exp. Bot. 2014, 83, 155–165. [Google Scholar]
- Mahlo, S.M. Antibacterial Activity of Selected Plants Used in Ethnoveterinary Medicine. Master’s Thesis, University of Limpopo (Turfloop Campus), Turfloop, South Africa, 2006. [Google Scholar]
- Maphosa, V.; Masika, P.J. Ethnoveterinary uses of medicinal plants: A survey of plants used in the ethnoveterinary control of gastro-intestinal parasites of goats in the Eastern Cape Province, South Africa. Pharm. Biol. 2010, 48, 697–702. [Google Scholar] [CrossRef] [PubMed]
- Mkwanazi, M.V.; Ndlela, S.Z.; Chimonyo, M. Utilisation of indigenous knowledge to control ticks in goats: A case of KwaZulu-Natal Province, South Africa. Trop. Anim. Health Prod. 2020, 52, 1375–1383. [Google Scholar] [CrossRef] [PubMed]
- Moichwanetse, B.I.; Ndhlovu, P.T.; Sedupane, G.; Aremu, A.O. Ethno-veterinary plants used for the treatment of retained placenta and associated diseases in cattle among Dinokana communities, North West Province, South Africa. S. Afr. J. Bot. 2020, 132, 108–116. [Google Scholar] [CrossRef]
- Moyo, B.; Masika, P.J. Tick control methods used by resource-limited farmers and the effect of ticks on cattle in rural areas of the Eastern Cape Province, South Africa. Trop. Anim. Health Prod. 2009, 41, 517–523. [Google Scholar] [CrossRef] [PubMed]
- Mthi, S.; Rust, J.; Morgenthal, T.; Moyo, B. An ethno-veterinary survey of medicinal plants used to treat bacterial diseases of livestock in three geographical areas of the Eastern Cape Province, South Africa. J. Med. Plants Res. 2018, 12, 240–247. [Google Scholar]
- Mwale, M.; Masika, P.J. Ethno-veterinary control of parasites, management and role of village chickens in rural households of Centane district in the Eastern Cape, South Africa. Trop. Anim. Health Prod. 2009, 41, 1685–1693. [Google Scholar] [CrossRef] [PubMed]
- Ndou, R.V. A Study of Ethnoveterinary medicIne in the North West Province, South Africa. Master’s Thesis, North-West University, Mmabatho, South Africa, 2018. [Google Scholar]
- Ramovha, L.I.; Van Wyk, A.E. Ethnoveterinary practices of the Vhavenḓa, South Africa, in the treatment of redwater (mali) in cattle. Indilinga Afr. J. Indig. Knowl. Syst. 2016, 15, 314–327. [Google Scholar]
- Rwodzi, M. Alternative Remedies Used by Resource-Limited Farmers in the Treatment and Manipulation of the Reproductive System of Non-Descript Goats in the Eastern Cape Province, South Africa. Master’s Thesis, University of Fort Hare, Alice, South Africa, 2014. [Google Scholar]
- Sanhokwe, M.; Mupangwa, J.; Masika, P.J.; Maphosa, V.; Muchenje, V.; Masika, P.J. Medicinal plants used to control internal and external parasites in goats. Onderstepoort J. Vet. Res. 2016, 83, a1016. [Google Scholar] [CrossRef]
- Soyelu, O.T.; Masika, P.J. Traditional remedies used for the treatment of cattle wounds and myiasis in Amatola Basin, Eastern Cape Province, South Africa. Onderstepoort J. Vet. Res. 2009, 76, 393–397. [Google Scholar] [CrossRef] [PubMed]
- Van der Merwe, D.; Swan, G.E.; Botha, C.J. Use of ethnoveterinary medicinal plants in cattle by Setswana-speaking people in the Madikwe area of the North West Province of South Africa. J. S. Afr. Vet. Assoc. 2001, 72, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Gaoue, O.G.; Coe, M.A.; Bond, M.; Hart, G.; Seyler, B.C.; McMillen, H. Theories and major hypotheses in ethnobotany. Econ. Bot. 2017, 71, 269–287. [Google Scholar] [CrossRef]
- Hyvärinen, O.; Timm Hoffman, M.; Reynolds, C. Vegetation dynamics in the face of a major land-use change: A 30-year case study from semi-arid South Africa. Afr. J. Range Forage Sci. 2019, 36, 141–150. [Google Scholar] [CrossRef]
- Yirga, G.; Teferi, M.; Gidey, G.; Zerabruk, S. An ethnoveterinary survey of medicinal plants used to treat livestock diseases in Seharti-Samre district, Northern Ethiopia. Afr. J. Plant Sci. 2012, 6, 113–119. [Google Scholar]
- McCorkle, C.M.; Mathias-Mundy, E. Ethnoveterinary Medicine in Africa. Afr. J. Int. Afr. Inst. 1992, 62, 59–93. [Google Scholar] [CrossRef]
- Nchu, F.; Nana, P.; Msalya, G.; Magano, S.R. Ethnoveterinary practices for control of ticks in Africa. In Ethnoveterinary Medicine: Present and Future Concepts; McGaw, L.J., Abdalla, M.A., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 99–122. [Google Scholar] [CrossRef]
- Van Wyk, B.E. A family-level floristic inventory and analysis of medicinal plants used in Traditional African Medicine. J. Ethnopharmacol. 2020, 249, 112351. [Google Scholar] [CrossRef]
- Chakale, M.V.; Mwanza, M.; Aremu, A.O. Ethnoveterinary knowledge and biological evaluation of plants used for mitigating cattle diseases: A critical insight into the trends and patterns in South Africa. Front. Vet. Sci. 2021, 8, 891. [Google Scholar] [CrossRef] [PubMed]
- Eloff, J.N.; McGaw, L.J. Application of plant extracts and products in veterinary infections. In New Strategies Combating Bacterial Infection; Ahmad, I., Aqil, F., Eds.; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2009; pp. 205–228. [Google Scholar] [CrossRef]
- Abdalla, M.A.; McGaw, L.J. Introduction. In Ethnoveterinary Medicine: Present and Future Concepts; McGaw, L.J., Abdalla, M.A., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 1–3. [Google Scholar] [CrossRef]
- Eloff, J.N.; McGaw, L.J. Plant extracts used to manage bacterial, fungal, and parasitic infections in Southern Africa. In Modern Phytomedicine: Turning Medicinal Plants into Drugs; Ahmad, I., Aqil, F., Owais, M., Eds.; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2006; pp. 97–121. [Google Scholar] [CrossRef]
- Eloff, J.N. Avoiding pitfalls in determining antimicrobial activity of plant extracts and publishing the results. BMC Complementary Altern. Med. 2019, 19, 106. [Google Scholar] [CrossRef] [PubMed]
- Van Vuuren, S.; Holl, D. Antimicrobial natural product research: A review from a South African perspective for the years 2009–2016. J. Ethnopharmacol. 2017, 208, 236–252. [Google Scholar] [CrossRef]
- Heinrich, M.; Appendino, G.; Efferth, T.; Fürst, R.; Izzo, A.A.; Kayser, O.; Pezzuto, J.M.; Viljoen, A. Best practice in research—Overcoming common challenges in phytopharmacological research. J. Ethnopharmacol. 2020, 246, 112230. [Google Scholar] [CrossRef] [PubMed]
- Elisha, I.L.; Dzoyem, J.-P.; Botha, F.S.; Eloff, J.N. The efficacy and safety of nine South African medicinal plants in controlling Bacillus anthracis Sterne vaccine strain. BMC Complementary Altern. Med. 2016, 16, 5. [Google Scholar] [CrossRef] [PubMed]
- Dzoyem, J.P.; McGaw, L.J.; Eloff, J.N. In vitro antibacterial, antioxidant and cytotoxic activity of acetone leaf extracts of nine under-investigated Fabaceae tree species leads to potentially useful extracts in animal health and productivity. BMC Complementary Altern. Med. 2014, 14, 147. [Google Scholar] [CrossRef] [PubMed]
- Adamu, M.; Naidoo, V.; Eloff, J.N. The antibacterial activity, antioxidant activity and selectivity index of leaf extracts of thirteen South African tree species used in ethnoveterinary medicine to treat helminth infections. BMC Vet. Res. 2014, 10, 52. [Google Scholar] [CrossRef] [PubMed]
- Aremu, A.O.; Fawole, O.A.; Chukwujekwu, J.C.; Light, M.E.; Finnie, J.F.; Van Staden, J. In vitro antimicrobial, anthelmintic and cyclooxygenase-inhibitory activities and phytochemical analysis of Leucosidea sericea. J. Ethnopharmacol. 2010, 131, 22–27. [Google Scholar] [CrossRef] [PubMed]
- McGaw, L.J.; Van der Merwe, D.; Eloff, J.N. In vitro anthelmintic, antibacterial and cytotoxic effects of extracts from plants used in South African ethnoveterinary medicine. Vet. J. 2007, 173, 366–372. [Google Scholar] [CrossRef]
- Aremu, A.O.; Ndhlala, A.R.; Fawole, O.A.; Light, M.E.; Finnie, J.F.; Van Staden, J. In vitro pharmacological evaluation and phenolic content of ten South African medicinal plants used as anthelmintics. S. Afr. J. Bot. 2010, 76, 558–566. [Google Scholar] [CrossRef]
- Masika, P.J.; Afolayan, A.J. Antimicrobial activity of some plants used for the treatment of livestock disease in the Eastern Cape, South Africa. J. Ethnopharmacol. 2002, 83, 129–134. [Google Scholar] [CrossRef]
- Luseba, D.; Elgorashi, E.; Ntloedibe, D.; Van Staden, J. Antibacterial, anti-inflammatory and mutagenic effects of some medicinal plants used in South Africa for the treatment of wounds and retained placenta in livestock. S. Afr. J. Bot. 2007, 73, 378–383. [Google Scholar] [CrossRef]
- Aremu, A.O.; Finnie, J.F.; Van Staden, J. Potential of South African medicinal plants used as anthelmintics—Their efficacy, safety concerns and reappraisal of current screening methods. S. Afr. J. Bot. 2012, 82, 134–150. [Google Scholar] [CrossRef]
- Fouche, G.; Sakong, B.M.; Adenubi, O.T.; Pauw, E.; Leboho, T.; Wellington, K.W.; Eloff, J.N. Anthelmintic activity of acetone extracts from South African plants used on egg hatching of Haemonchus contortus. Onderstepoort J. Vet. Res. 2016, 83, a1164. [Google Scholar] [CrossRef][Green Version]
- Adamu, M.; Naidoo, V.; Eloff, J.N. Efficacy and toxicity of thirteen plant leaf acetone extracts used in ethnoveterinary medicine in South Africa on egg hatching and larval development of Haemonchus contortus. BMC Vet. Res. 2013, 9, 38. [Google Scholar] [CrossRef]
- Githiori, J.B.; Athanasiadou, S.; Thamsborg, S.M. Use of plants in novel approaches for control of gastrointestinal helminths in livestock with emphasis on small ruminants. Vet. Parasitol. 2006, 139, 308–320. [Google Scholar] [CrossRef]
- Bürglin, T.R.; Lobos, E.; Blaxter, M.L. Caenorhabditis elegans as a model for parasitic nematodes. Int. J. Parasitol. 1998, 28, 395–411. [Google Scholar] [CrossRef]
- Geary, T.G.; Thompson, D.P.; Klein, R.D. Mechanism-based screening: Discovery of the next generation of anthelmintics depends upon more basic research. Int. J. Parasitol. 1999, 29, 105–112. [Google Scholar] [CrossRef]
- McGaw, L.J.; Rabe, T.; Sparg, S.G.; Jäger, A.K.; Eloff, J.N.; Van Staden, J. An investigation on the biological activity of Combretum species. J. Ethnopharmacol. 2001, 75, 45–50. [Google Scholar] [CrossRef]
- Shai, L.J.; Bizimenyera, E.S.; Bagla, V.; McGaw, L.J.; Eloff, J.N. Curtisia dentata (Cornaceae) leaf extracts and isolated compounds inhibit motility of parasitic and free-living nematodes. Onderstepoort J. Vet. Res. 2009, 76, 249–256. [Google Scholar] [CrossRef]
- Gülçin, İ. Antioxidant activity of food constituents: An overview. Arch. Toxicol. 2012, 86, 345–391. [Google Scholar] [CrossRef]
- Granato, D.; Shahidi, F.; Wrolstad, R.; Kilmartin, P.; Melton, L.D.; Hidalgo, F.J.; Miyashita, K.; Camp, J.v.; Alasalvar, C.; Ismail, A.B.; et al. Antioxidant activity, total phenolics and flavonoids contents: Should we ban in vitro screening methods? Food Chem. 2018, 264, 471–475. [Google Scholar] [CrossRef]
- Houghton, P.J.; Howes, M.J.; Lee, C.C.; Steventon, G. Uses and abuses of in vitro tests in ethnopharmacology: Visualizing an elephant. J. Ethnopharmacol. 2007, 110, 391–400. [Google Scholar] [CrossRef]
- Dzoyem, J.P.; Eloff, J.N. Anti-inflammatory, anticholinesterase and antioxidant activity of leaf extracts of twelve plants used traditionally to alleviate pain and inflammation in South Africa. J. Ethnopharmacol. 2015, 160, 194–201. [Google Scholar] [CrossRef] [PubMed]
- Sserunkuma, P.; McGaw, L.J.; Nsahlai, I.V.; Van Staden, J. Selected southern African medicinal plants with low cytotoxicity and good activity against bovine mastitis pathogens. S. Afr. J. Bot. 2017, 111, 242–247. [Google Scholar] [CrossRef]
- Altemimi, A.; Lakhssassi, N.; Baharlouei, A.; Watson, D.G.; Lightfoot, D.A. Phytochemicals: Extraction, isolation, and identification of bioactive compounds from plant extracts. Plants 2017, 6, 42. [Google Scholar] [CrossRef] [PubMed]
- Gálvez, M.; Martín-Cordero, C.; Ayuso, M.J. Pharmacological activities of iridoids biosynthesized by route II. In Studies in Natural Products Chemistry; Attaur, R., Ed.; Elsevier: Amsterdam, The Netherlands, 2005; Volume 32. [Google Scholar]
- Brielmann, H.L.; Setzer, W.N.; Kaufman, P.B.; Kirakosyan, A.; Cseke, L.J. Phytochemicals: The chemical components of plants. In Natural Products from Plants, 2nd ed.; Cseke, L.J., Kirakosyan, A., Kaufman, P.B., Warber, S.L., Duke, J.A., Brielmann, H.L., Eds.; Taylor and Francis Group: Boca Raton, FL, USA, 2006; pp. 1–49. [Google Scholar]
- Olaokun, O.O.; Mkolo, N.M.; Mogale, M.A.; King, P.H. Phytochemical screening, antioxidant, anti-inflammatory and glucose utilization activities of three South African plants used traditionally to treat diseases. Biol. Med. 2017, 9, 1000412. [Google Scholar] [CrossRef]
- Hajam, Y.A.; Rai, S.; Kumar, R.; Bashir, M.; Malik, J.A. Phenolic compounds from medicinal herbs: Their role in animal health and diseases—A new approach for sustainable welfare and development. In Plant Phenolics in Sustainable Agriculture; Lone, R., Shuab, R., Kamili, A.N., Eds.; Springer: Singapore, 2020; Volume 1, pp. 221–239. [Google Scholar] [CrossRef]
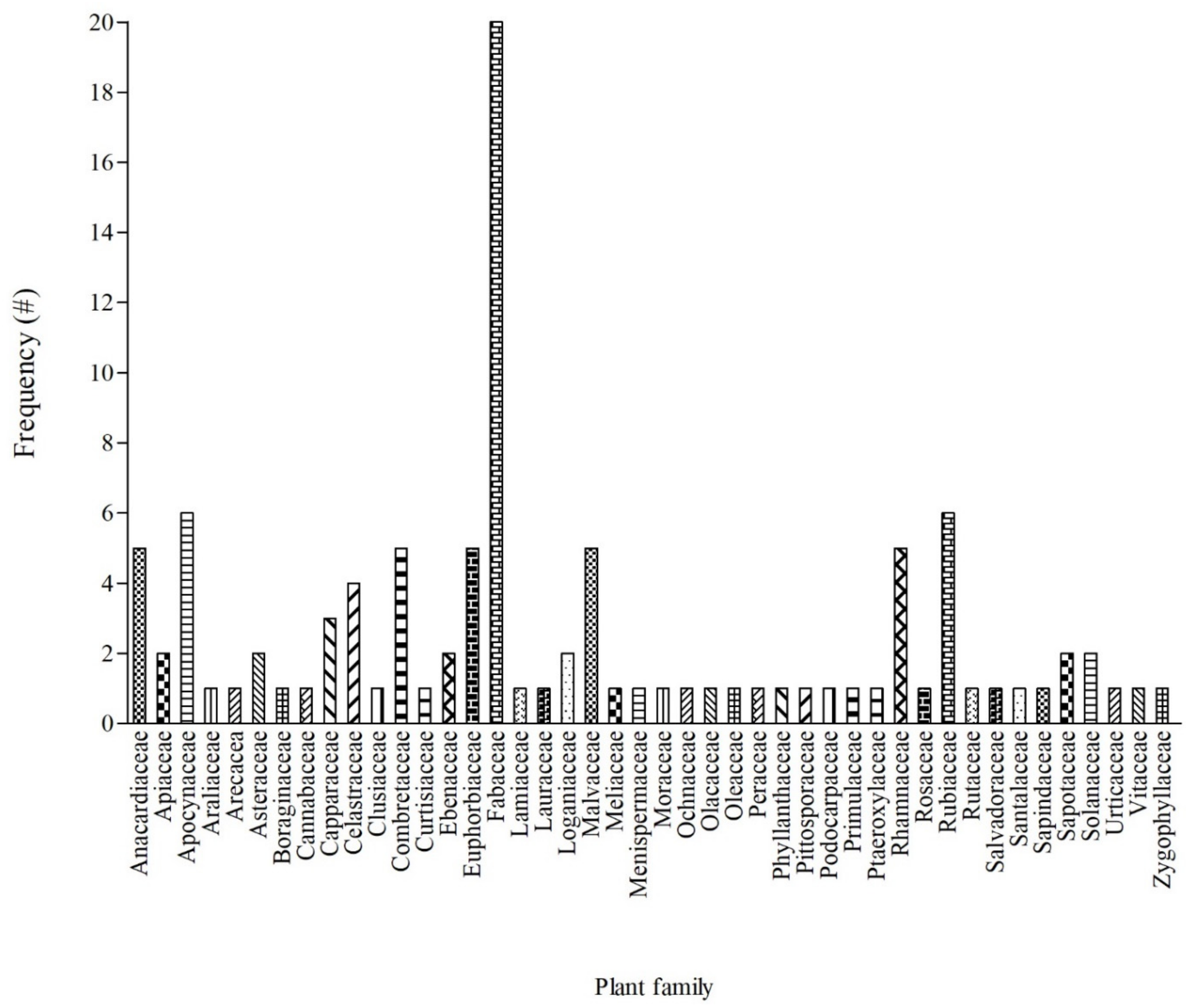
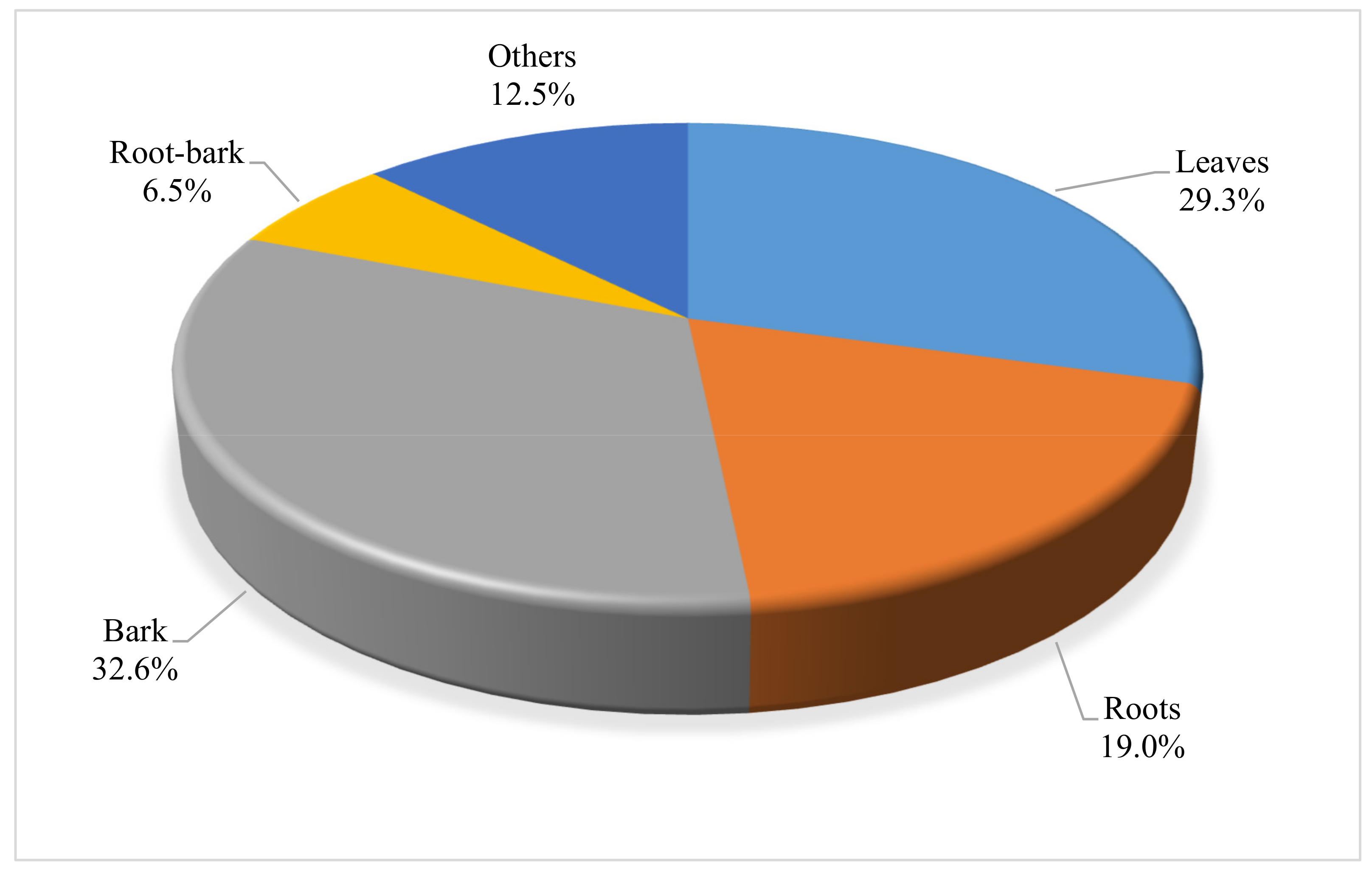
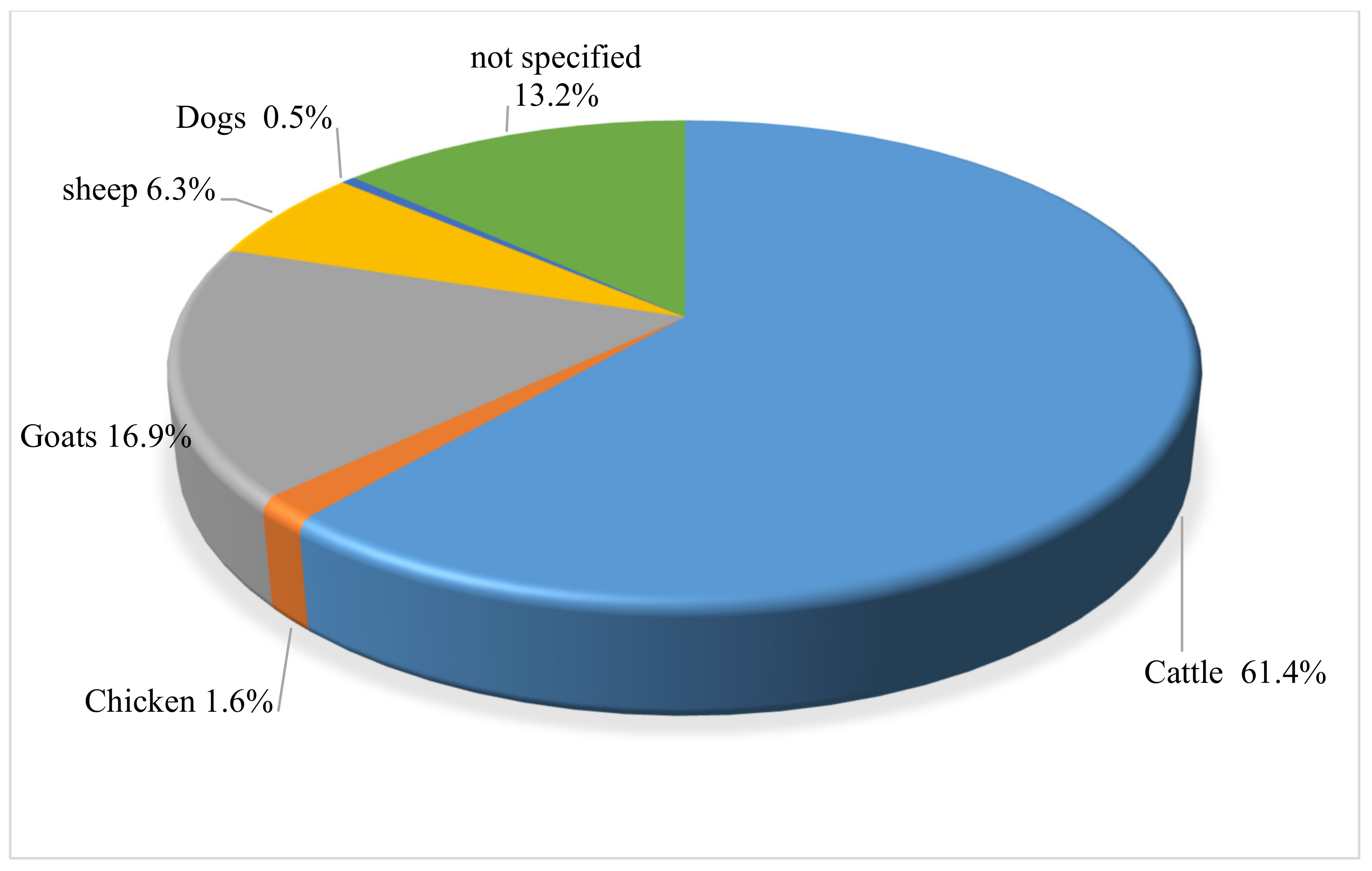
| Reference | Province | Documented Plants | Documented Woody Plants | Method of Survey/Interview | Number of Participants | ||
|---|---|---|---|---|---|---|---|
| Number of Plants | Number of Families | Number of Plants | Number of Families | ||||
| Chitura et al., [33] | Limpopo | 11 | 10 | 7 | 4 | Structured questionnaire | 180 |
| Dold and Cocks [34] | Eastern Cape | 53 | 42 | 21 | 18 | Questionnaire and field interview | Not specified |
| Kambizi [35] | Eastern Cape | 22 | 19 | 8 | 7 | Semi-structured interview | Not specified |
| Khunoana et al., [36] | Mpumalanga | 11 | 9 | 6 | 4 | Semi-structured interview | 50 |
| Luseba and Tshisikhawe [37] | Limpopo | 34 | 22 | 21 | 13 | Focus group discussion | 37 |
| Luseba and Van der Merwe [38] | Limpopo | 19 | 12 | 9 | 6 | Individual and group interview | Not specified |
| Magwede et al., [39] | Limpopo | 27 | 14 | 15 | 9 | Open-ended questions and semi-structured questionnaire | 42 |
| Mahlo [40] | Limpopo | 5 | 4 | 5 | 4 | Not specified | Not specified |
| Maphosa and Masika [41] | Eastern Cape | 28 | 20 | 12 | 9 | Structured questionnaire and general conversation | 30 |
| Mkwanazi et al., [42] | KwaZulu-Natal | 5 | 4 | 1 | 1 | Structured questionnaire | Not specified |
| Moichwanetse et al., [43] | North West | 25 | 18 | 8 | 5 | Face-to-face, semi-structured interview | 15 |
| Moyo and Masika [44] | Eastern Cape | 2 | 2 | 1 | 1 | Structured questionnaire | 59 |
| Mthi et al., [45] | Eastern Cape | 6 | 6 | 4 | 3 | Semi-structured questionnaire, observations and guided field trip | 48 |
| Mwale and Masika [46] | Eastern Cape | 9 | 7 | 1 | 1 | Structured questionnaire | 54 |
| Ndou [47] | North West | 31 | 17 | 8 | 7 | Semi structured face-to-face interview | 21 |
| Ramovha and Van Wyk [48] | Limpopo | 20 | 10 | 14 | 8 | Semi-structured interview | Not specified |
| Rwodzi [49] | Eastern Cape | 10 | 8 | 2 | 2 | Questionnaire | 60 |
| Sanhokwe et al., [50] | Eastern Cape | 9 | 8 | 3 | 3 | Structured questionnaire | 53 |
| Soyelu and Masika [51] | Eastern Cape | 13 | 11 | 7 | 6 | Structured questionnaire | 53 |
| Van der Merwe et al., [52] | North West | 45 | 24 | 19 | 12 | Detailed interview | 28 |
| Plant Species | Family | Common Name | Method of Preparation and Administration | Plant Part Used | Animal Treated | Disease/Health Condition | Reference |
|---|---|---|---|---|---|---|---|
| Acokanthera oppositifolia (Lam.) Codd | Apocynaceae | Bushman’s Poison | Leaves are boiled for 10 min, strained, and left to stand overnight | Leaves | Sheep, goats | Heartwater | Dold and Cocks [34] |
| Acokanthera oppositifolia (Lam.) Codd | Apocynaceae | Bushman’s Poison | Decoction | Leaves | Goats | Gastrointestinal parasites | Maphosa and Masika [41] |
| Acokanthera oppositifolia (Lam.) Codd | Apocynaceae | Bushman’s Poison | Leaves crushed with water and administered orally | Leaves | Cattle | Paratyphoid (Goso) | Mthi et al., [45] |
| Acokanthera oppositifolia (Lam.) Codd | Apocynaceae | Bushman’s Poison | Decoction; ground leaves are boiled, cooled, and administered by drenching the animals. Dose with 1 L bottle for adults and a 300 mL bottle for kids | Leaves | Goats | Helminths, ticks | Sanhokwe et al., [50] |
| Afrocarpus falcatus (Thunb.) C.N.Page | Podocarpaceae | Outeniqua yellowwood | Decoction | Leaves | Dogs | Distemper | Dold and Cocks [34] |
| Albizia sp. | Fabaceae | Xisitana | Root skin is infused in water and is left overnight | Roots | Cattle | Swollen stomach | Khunoana et al., [36] |
| Azima tetracantha Lam. | Salvadoraceae | Beehanger | Dried and ground root is bottled in cold water | Root | Cattle | Dystocia | Dold and Cocks [34] |
| Balanites maughamii Sprague | Zygophyllaceae | Torchwood | Decoction | Leaves | Cattle | Diarrhoea | Mahlo [40] |
| Balanites maughamii Sprague | Zygophyllaceae | Torchwood | Ground leaves are mixed with cold water | Leaves | Cattle | Diarrhoea | Van der Merwe et al., [52] |
| Bauhinia thonningii Schum. (Syn: Piliostigma thonningii (Schumach.) Milne-Redh.) | Fabaceae | Camel’s foot | Decoction | Leaves | Cattle | Diarrhoea | Mahlo [40] |
| Bolusanthus speciosus (Bolus) Harms | Fabaceae | Tree wisteria | Pounded roots are immersed in water | Roots | Cattle | Retained placenta | Luseba and Tshisikhawe [37] |
| Brachylaena ilicifolia (Lam.) E. Phillips & Schweick. | Asteraceae | Bitterblaar | Leaves are mixed with leaves of Leucas capensis (Benth.) Engl. and sap of Aloe ferox Mill and boiled | Leaves | Lambs | Diarrhoea | Dold and Cocks [34] |
| Breonadia salicina (Vahl) Hepper & J.R.I.Wood | Rubiaceae | Transvaal teak | Maceration | Bark | Cattle | General intestinal diseases and retained placenta | Mahlo [40] |
| Burchellia bubaline (L.f.) Sims | Rubiaceae | Wild pomegranate | ns | Leaves | ns | Heartwater | Kambizi [35] |
| Cadaba aphylla (Thunb.) Wild | Capparaceae | leafless cadaba, leafless wormbush, black storm | Root decoction: Combined with roots of Ziziphus zeyheriana, Senna italica, and Dicoma galpinii | Root | ns | Blood cleansing and pains (sores, fractures) | Ndou [47] |
| Calpurnia aurea (Aiton) Benth. | Fabaceae | Common calpurnia | Infusion | Leaves | Cattle | Maggot-infested wounds | Soyelu and Masika [51] |
| Capparis sepiaria L. | Capparaceae | Cape Capers | Infusion | Roots | Goats | Gastro-intestinal parasites | Maphosa and Masika [41] |
| Carissa bispinosa (L.) Desf. ex Brenan | Apocynaceae | Forest num-num | Bulb is ground and mixed with water | Roots, bulb | Cattle | Calving difficulties | Luseba and Tshisikhawe [37] |
| Cassia abbreviata Oliv. | Fabaceae | Sjambok pod | Bark infusion | Bark | Cattle | Retained placenta | Chitura et al., [33] |
| Cassia abbreviata Oliv. | Fabaceae | Sjambok pod | Ground bark is soaked in water overnight or boiled | Bark | ns | Worm infestation | Luseba and Van der Merwe [38] |
| Cassia abbreviata Oliv. | Fabaceae | Sjambok pod | Ground bark is mixed with water | Bark | Cattle | Wounds | Magwede et al., [39] |
| Cassia abbreviata Oliv. | Fabaceae | Sjambok pod | Bark infusion or decoction | Bark, root bark | Cattle | Redwater | Ramovha and Van Wyk [48] |
| Cassine aethiopica Thunb. (Syn: Mystroxylon aethiopicum (Thunb.) Loes.) | Celastraceae | Kooboo-berry | Bark is grated and boiled for 20 min | Bark | Cattle | Heartwater | Dold and Cocks [34] |
| Centella asiatica (L.) Urb. | Apiaceae | Varkoortjie | Decoction | Bark | Goats | Helminths | Sanhokwe et al., [50] |
| Cephalanthus natalensis Oliv. | Rubiaceae | Strawberry bush | Infusion | Leaves | Cattle | Eye problem | Luseba and Tshisikhawe [37] |
| Cissampelos capensis L.f. | Menispermaceae | Davidjies | ns | Roots | ns | Skin problems, wounds | Kambizi [35] |
| Clutia pulchella L. | Peraceae | Lightning bush | Decoction | Roots | Cattle | Gall | Khunoana et al., [36] |
| Coddia rudis (E.Mey. ex. Harv.) Verdc. | Rubiaceae | Small bone apple | ns | Leaves | Leaves | Skin problems (eliminates ticks) | Kambizi [35] |
| Combretum collinum Fresen | Combretaceae | Bicoloured bushwillow | ns | Bark | Cattle | Constipation | Chitura et al., [33] |
| Combretum microphyllum Klotzsch. | Combretaceae | Flame creeper | Infusion or decoction | Roots | Cattle | Redwater | Ramovha and Van Wyk [48] |
| Combretum molle R.Br ex G.Don | Combretaceae | Velvet bushwillow | Infusion | Leaves | Cattle | Gut conditions—diarrhoea. Worm infestation. Breeding problems, difficult calving | Luseba and Tshisikhawe [37] |
| Combretum paniculatum Vent. | Combretaceae | Burning bush | Decoction | Root bark | Cattle | For fertility problems | Luseba and Van der Merwe [38] |
| Croton gratissimus Burch. (Syn: Croton gratissimus Burch. var gratissimus) | Euphorbiaceae | lavender croton, lavender fever berry | Dried leaves are crushed and mixed with supplement feed | Leaves | ns | Fertility enhancement in livestock | Ndou [47] |
| Croton gratissimus Burch. (Syn: Croton gratissimus Burch. var gratissimus) | Euphorbiaceae | Lavender fever berry | ns | Leaves | Cattle | Pneumonia | Van der Merwe et al., [52] |
| Curtisia dentata (Brum. f.) C.A.Smith | Curtisiaceae | Assegai | Bark, together with the bark of Rapanea melanophloeos (L.) Mez, is boiled for 30 min | Bark | Cattle | Heartwater | Dold and Cocks [34] |
| Cussonia spicata Thunb. | Araliaceae | Cabbage-tree | Mixed with leaves of Olea europaea L. subsp. africana (Mill.) P.S.Green to produce concoction or decoction | Leaves | Cattle | Bloody urine after calving (endometritus and/or vaginitis) | Dold and Cocks [34] |
| Cussonia spicata Thunb. | Araliaceae | Cabbage-tree | ns | Bark | ns | Heartwater | Kambizi [35] |
| Cussonia spicata Thunb. | Araliaceae | Cabbage-tree | Infusion | Bark | Goats | Gastro-intestinal parasites | Maphosa and Masika [41] |
| Cussonia spicata Thunb. | Araliaceae | Cabbage-tree | Ground bark is soaked overnight and dose at 300 mL | Bark | Goats | Helmenthis | Sanhokwe et al., [50] |
| Cussonia spicata Thunb. | Araliaceae | Cabbage-tree | ns | Bark | Cattle | Treat retained afterbirth. | Van der Merwe et al., [52] |
| Dalbergia obovata E.Mey. | Fabaceae | Climbing flat bean | Leaves and bark crushed and mixed with water | Leaves, bark | Cattle | Paratyphoid (Goso) | Mthi et al., [45] |
| Dichrostachys cinerea (L.) Wight & Arn. | Fabaceae | Sicklebush | Dried fruit is made into powder | Fruit | Sheep, goats | Wounds | Chitura et al., [33] |
| Diospyros lycioides Desf. (Syn: Diospyros lycioides Desf. subsp. lyciodes) | Ebenaceae | Bluebush, Karoo blue bush | Ground leaves are mixed with water and apply on the affected area | Leaves | Cattle | Ticks | Luseba and Tshisikhawe [37] |
| Diospyros lycioides Desf. (Syn: Diospyros lycioides Desf. subsp. lyciodes) | Ebenaceae | Bluebush, Karoo blue bush | Leaves are crushed and mixed with water | Leaves | Cattle | Wounds | Magwede et al., [39] |
| Diospyros mespiliformis Hochst. ex A.DC. | Ebeneceae | African ebony | Ground bark is mixed with hippopotamus fat; dosed and also rubbed into vagina | Bark | ns | For milk production | Luseba and Van der Merwe [38] |
| Diospyros mespiliformis Hochst. ex A.DC. | Ebeneceae | African ebony | Ground roots are mixed with warm but not boiling water to yield an infusion | Roots | Cattle | Redwater | Ramovha and Van Wyk [48] |
| Dombeya rotundifolia (Hochst.) Planch. | Malvaceae | Wild pear | Ground leaves/flowers are mixed with chicken feed | Leaves, flowers | Chicken | Newcastle disease | Luseba and Van der Merwe [38] |
| Dombeya rotundifolia (Hochst.) Planch. | Malvaceae | Wild pear | Decoction | Leaves | Cattle | Diarrhoea | Mahlo [40] |
| Ehretia rigida (Thunb.) Druce | Boraginaceae | Puzzle bush | Decoction | Roots | Cattle | Eating problems | Luseba and Tshisikhawe [37] |
| Ehretia rigida (Thunb.) Druce | Boraginaceae | Puzzle bush | ns | Roots | Cattle | Fractures | Van der Merwe et al., [52] |
| Elaeodendron transvaalense (Burtt Davy) R.H.Archer | Celastraceae | Bushveld saffron | Ground fruits are mixed with water | Fruit | Cattle | Worms | Luseba and Tshisikhawe [37] |
| Elaeodendron transvaalense (Burtt Davy) R.H.Archer | Celastraceae | Spike-Thorn | ns | Bark | Cattle | Diarrhoea | Van der Merwe et al., [52] |
| Elephantorrhiza burkei Benth. | Fabaceae | Elephant-root | Ground bulb (or bark) is mixed with water | Bark, roots | Cattle | Diarrhoea | Luseba and Tshisikhawe [37] |
| Englerophytum magalismontanum (Sond.) T.D.Penn | Sapotaceae | Transvaal milkplum | ns | Roots | Cattle | Fertility enhancement | Van der Merwe et al., [52] |
| Erythrina caffra Thunb. | Fabaceae | Coast coral tree | ns | Bark | ns | Heartwater | Kambizi [35] |
| Erythrina lysistemon Hutch. | Fabaceae | Common coral tree | Fresh bark is crushed into pulp and juice is applied | Bark | Cattle | Wounds | Magwede et al., [39] |
| Euphorbia cupularis Boiss. | Euphorbiaceae | Crying tree | Milky latex is applied on third eyelid and on the skin of the limping leg | Milky latex | ns | Eye infection and blackquarter | Luseba and Van der Merwe [38] |
| Euphorbia umbellata (Pax) Bruyns | Euphorbiaceae | African milk bush | Milky sap applied directly on the area between the eye and ear | Stem | Cattle | Eye problem | Khunoana et al., [36] |
| Ficus sp. | Moraceae | ns | ns | Bark | ns | Wounds | Kambizi [35] |
| Garcinia livingstonei T. Anderson | Clusiaceae | African mangosteen | Juice from fresh leaves is squeezed | Leaves | Cattle | Eye problems | Luseba and Tshisikhawe [37] |
| Grewia damine Gaertn. (Syn: Grewia bicolor Juss.) | Malvaceae | White raisin | Stem branches are cut into sticks used as lashes | Sticks | Cattle | Redwater | Ramovha and Van Wyk [48] |
| Grewia flava DC. | Malvaceae | Brandybush, wild currant | Root decoction combined with root of Ziziphus zeyheriana and given orally | root | cattle | Diarrhoea | Ndou [47] |
| Grewia flava DC. | Malvaceae | Brandybush, wild currant | ns | Roots | Cattle | Fertility enhancement | Van der Merwe et al., [52] |
| Grewia occidentalis L. | Malvaceae | Crossberry | Infusion is prepared with the leaves of Olea europaea subsp. africana and Zanthoxylum capense and sap of Aloe ferox | Leaves | ns | Gallsickness | Dold and Cocks [34] |
| Grewia occidentalis L. | Malvaceae | Crossberry | Decoction | Bark | Goats | Gastro-intestinal parasites | Maphosa and Masika [41] |
| Grewia occidentalis L. | Malvaceae | Crossberry | Infusion | Leaves twigs | Cattle | Wounds | Soyelu and Masika [51] |
| Gymnosporia sp. | Celastraceae | Xihlangwa | Root skin infused in water and left overnight | Roots | Cattle | Black quarter and diarrhoea | Khunoana et al., [36] |
| Harpephyllum caffrum Bernh. | Anacardiaceae | Wild plum | ns | Bark | ns | Skin problems | Kambizi [35] |
| Harpephyllum caffrum Bernh. | Anacardiaceae | Wild plum | Decoction | Bark | Goats | Gastro-intestinal parasites | Maphosa and Masika [41] |
| Heteromorpha arborescens (Spreng.) Cham. & Schltdl. | Apiaceae | Parsley tree | Ground root powder is mixed with cold or warm water to yield an infusion | Root | Cattle | Redwater | Ramovha and Van Wyk [48] |
| Hippobromus pauciflorus (L.f) Radlk. | Sapindaceae | Bastard horsewood | Bark is mixed with the bark of Protorhus longifolia and is grated and boiled for 10 min | Bark | Cattle | Heartwater and diarrhoea | Dold and Cocks [34] |
| Hippobromus pauciflorus (L.f) Radlk. | Sapindaceae | Bastard horsewood | Infusion | Leaves | Cattle | Wounds | Soyelu and Masika [51] |
| Holarrhena pubescens Wall. ex G.Don | Apocynaceae | Conessi | Crushed roots are mixed with hot water to yield an infusion or are cooked to produce a decoction | Root | Cattle | Redwater | Ramovha and Van Wyk [48] |
| Hyperacanthus amoenus (Sims) Bridson | Rubiaceae | Thorny gardenia | Maceration | Bark | Cattle | Relieving pain, loss of appetite, and general ailments | Mahlo [40] |
| Jatropha curcas L. | Euphorbiaceae | Physic nut | Crushed (1–2) seeds are mixed with water for drenching | Seeds | Cattle, goats | Constipation | Luseba and Van der Merwe [38] |
| Jatropha curcas L. | Euphorbiaceae | Physic nut | Sliced root is cooked to produce a decoction | Root, tuber | Cattle | Redwater | Ramovha and Van Wyk [48] |
| Maerua angolensis DC. | Capparaceae | Bead-bean | Ground leaves are mixed with water. Fresh leaves are squeezed to extract juice | Leaves | Cattle | Eating disorder, drought tonic, eye problems, wounds | Luseba and Tshisikhawe [37] |
| Maytenus peduncularis (Sond.) Loes. | Celastraceae | Blackwood | Root-bark is made into a paste | Root-bark | Cattle | Fractures | Chitura et al., [33] |
| Maytenus peduncularis (Sond.) Loes. | Celastraceae | Blackwood | ns | Leaves | Goats | Ticks | Mkwanazi et al., [42] |
| Millettia grandis (E.Mey) Skeels | Fabaceae | Umzimbeet | Soak the leaves in cold water | Leaves | Chicken | Internal parasites | Mwale and Masika [46] |
| Noltea africana (L.) Rchb. f. | Rhamnaceae | Soap bush | Ground into powder | Roots | Goats | Womb cleansing; fertility | Rwodzi [49] |
| Ochna holstii Engl. | Ochnaceae | Common forest ochna | Leaves and branches boiled for 2 h, 1 litre is given once daily for 3 days | Leaves | Goats, sheep, cattle | Wounds | Luseba and Tshisikhawe [37] |
| Ochna holstii Engl. | Ochnaceae | Common forest ochna | Leaves are ground and boiled | Leaves, twigs, bark | Cattle | Wounds | Magwede et al., [39] |
| Ocotea bullata (Burch.) E. Meyer in Drege | Lauraceae | Stinkwood | Decoction | Bark | Goats | Gastrointestinal parasites | Maphosa and Masika [41] |
| Olea europaea subsp. cuspidata (Wall. & G.Don) Cif. (Syn: Olea europaea L. subsp. africana (Mill.) P.S.Green) | Oleaceae | Wild olive | Bark infusion. Leaves together with Cussonia spicata root. Mixture of Zanthoxylum capense leaves, Grewia occidentalis leaves, and Aloe ferox sap | Bark, leaves | Goats, cattle | Diarrhoea in goats. Bloody urine after calving (endomitritis and vaginitis in cattle). Treating gallsickness in cattle | Dold and Cocks [34] |
| Olea europaea subsp. cuspidata (Wall. & G.Don) Cif. (Syn: Olea europaea L. subsp. africana (Mill.) P.S.Green) | Oleaceae | Wild olive | Crushed bark is soaked in warm water | Bark | Cattle | Black quarter (Ciko) | Mthi et al., [45] |
| Osyris lanceolata Hoscht. & Steud | Santalaceae | Rock tannin-bush | Maceration | Bulb | Cattle | Retained placenta, alleviation of pain and internal bleeding | Moichwanetse et al., [43] |
| Ozoroa paniculosa (Sond.) R.Fern. & A.Fern. (Syn: Ozoroa paniculosa (Sond.) R.Fern. & A.Fern. var. paniculosa) | Anacardiaceae | Common resin tree | ns | Bark, root bark | Cattle | Diarrhoea, redwater, sweating sickness | Van der Merwe et al., [52] |
| Peltophorum africanum Sond. | Fabaceae | Weeping wattle | Bark is ground into powder | Bark | Cattle | Wounds | Magwede et al., [39] |
| Peltophorum africanum Sond. | Fabaceae | Weeping wattle | Poultice | Leaves and bulb | Cattle | Retained placenta, diarrhoea, and removal of blood clots from the skin | Moichwanetse et al., [43] |
| Peltophorum africanum Sond. | Fabaceae | Weeping wattle | ns | Bark, root bark | Cattle | Tonic, diarrhoea | Van der Merwe et al., [52] |
| Philenoptera violacea (Klotzsch) Schrire | Fabaceae | Apple-leaf | Bark is ground and infused in water overnight | Bark | Cattle | Gall, diarrhoea, and general ailments | Khunoana et al., [36] |
| Philenoptera violacea (Klotzsch) Schrire | Fabaceae | Apple-leaf | Bark is boiled in water | Bark | Cattle | Wounds | Magwede et al., [39] |
| Philenoptera violacea (Klotzsch) Schrire | Fabaceae | Apple-leaf | Bark is cooked or soaked in cold water to produce a red decoction/infusion | Stem and root-bark | Cattle | Redwater | Ramovha and Van Wyk [48] |
| Phoenix reclinata Jacq. | Arecacea | Wild date palm | Roots are mixed with Arctotis arctotoides leaves and boiled, warm liquid is used | Roots | Sheep, goats | Footrot | Dold and Cocks [34] |
| Pittosporum viridiflorum Sims | Pittosporaceae | Cheesewood | Infusion | Bark | Goats | Gastrointestinal parasites | Maphosa and Masika [41] |
| Pittosporum viridiflorum Sims | Pittosporaceae | Cheesewood | Decoction | Roots | Chicken | Wounds | Soyelu and Masika [51] |
| Pouzolzia mixta Solms | Urticaceae | Soap-nettle | Poultice | Roots | Cattle | Retained placenta and uterus cleansing | Moichwanetse et al., [43] |
| Protorhus longifolia (Bernh.) Engl. | Anacardiaceae | Red beech | Mixed with bark of Hippobromus pauciflorus and boiled for 20 min | Bark | Cattle | Heartwater and diarrhoea | Dold and Cocks [34] |
| Prunus persica (L.) Batsch | Rosaceae | Peach tree | Decoction | Leaves | Lamb, goats | Diarrhoea | Dold and Cocks [34] |
| Prunus persica (L.) Batsch | Rosaceae | Peach tree | Ground to pulp and mixed with hot paper and liquid | Leaves | ns | Wounds | Magwede et al., [39] |
| Prunus persica (L.) Batsch | Rosaceae | Peach tree | Infusion | Leaves | Cattle | Maggot-infested wounds | Soyelu and Masika [51] |
| Pseudolachnostylis maprouneifoloia Pax | Phyllanthaceae | Kudu berry | Ground bark is mixed with water | Bark | Cattle | Drought tonic | Luseba and Tshisikhawe [37] |
| Ptaeroxylon obliquum (Thunb.) Radlk. | Ptaeroxylaceae | Sneezewood | Decoction | Leaves | Goats | Gastro-intestinal parasites | Maphosa and Masika [41] |
| Ptaeroxylon obliquum (Thunb.) Radlk. | Ptaeroxylaceae | Sneezewood | Crushed and soaked in cold water overnight (infusion) | Bark | Cattle | Ticks | Moyo and Masika [44] |
| Ptaeroxylon obliquum (Thunb.) Radlk. | Ptaeroxylaceae | Sneezewood | Crush bark is mixed with used oil to form paste. Leaf decoction | Bark, leaves | Cattle | Wounds and myiasis | Soyelu and Masika [51] |
| Pterocarpus angolensis DC. | Fabaceae | Paddle-wood | Stem bark infusion | Bark | Cattle | Constipation | Chitura et al., [33] |
| Pterocarpus angolensis DC. | Fabaceae | Paddle-wood | Soak the bark in water | Bark | Cattle | Mali and not eating | Luseba and Tshisikhawe [37] |
| Pterocarpus angolensis DC. | Fabaceae | Paddle-wood | Chopped bark is soaked in cold water after the water has changed to reddish boil for 30–60 min | Bark | ns | General illness, unthriftiness, gallsickness, intestinal worms, blackquarter | Luseba and Van der Merwe [38] |
| Pterocarpus angolensis DC. | Fabaceae | Paddle-wood | Bark is ground to pulp | Bark | Cattle | Wounds | Magwede et al., [39] |
| Pterocarpus angolensis DC. | Fabaceae | Bark is cooked or imbibed in cold water to produce a red decoction/infusion | Bark, root bark | Cattle | Redwater | Ramovha and Van Wyk [48] | |
| Rapanea melanophloeos (L.) Mez (Syn: Myrsine melanophloeos (L.) R.Br. ex Sweet) | Primulaceae | Cape Beech | Mixed with bark of Curtisia dentata and boiled for 30 min | Bark | Cattle | Heartwater | Dold and Cocks [34] |
| Rauvolfia caffra Sond. | Apocynaceae | Quinine tree | Applied as powder on wounds | Bark | Cattle | Wounds | Magwede et al., [39] |
| Rhamnus prinoides L’Hér. | Rhamnaceae | Dogwood | Infusion | Roots | Goats | Twin or triplets production | Rwodzi [49] |
| Rhoicissus tridentata (L.f.) Wild & R.B.Drumm. | Vitaceae | Northern bushman’s grape | Tuber is boiled in water for 15 min | Tubers | Goats, sheep | Diarrhoea | Dold and Cocks [34] |
| Rhoicissus tridentata (L.f.) Wild & R.B.Drumm. | Vitaceae | Northern bushman’s grape | Leaves are boiled | Leaves | Cattle | Lumpy skin disease | Luseba and Tshisikhawe [37] |
| Rhoicissus tridentata (L.f.) Wild & R.B.Drumm. | Vitaceae | Northern bushman’s grape | ns | Tubers | Cattle | Heartwater, redwater internal parasites | Van der Merwe et al., [52] |
| Rothmannia capensis Thunb | Rubiaceae | Wild gardenia | Decoction | Roots | Cattle | Eating problem | Luseba and Tshisikhawe [37] |
| Rothmannia capensis Thunb | Rubiaceae | Wild gardenia | Fresh fruits are grounded to pulp | Fruit | ns | Wounds | Magwede et al., [39] |
| Schotia brachypetala Sond. | Fabaceae | African walnut | Ground bark is boiled in water | Bark | Cattle | Foot and mouth diseases, black quarter, and general ailments | Khunoana et al., [36] |
| Schotia brachypetala Sond. | Fabaceae | African walnut | Bark, preferably from the root, is cooked to make a decoction | Bark, root bark | Cattle | Redwater | Ramovha and Van Wyk [48] |
| Schotia latifolia Jacq. | Fabaceae | Bush Boerbean | Decoction | Bark | Goats | Gastro-intestinal parasites | Maphosa and Masika [41] |
| Sclerocarya birrea (A.Rich.) Hochst. | Anacardiaceae | Marula | Bark is soaked in cold water to yield an infusion or is cooked to produce a decoction | Bark | Cattle | Redwater | Ramovha and Van Wyk [48] |
| Sclerocarya birrea (A.Rich.) Hochst. | Anacardiaceae | Marula | ns | Bark | Cattle | Diarrhoea and fractures | Van der Merwe et al., [52] |
| Searsia lancea (L.f.) F.A.Barkley (Syn: Rhus lancea L.f.) | Anacardiaceae | Karee | ns | Roots | Cattle | Diarrhoea gallsickness | Van der Merwe et al., [52] |
| Secamone filiformis (L.f) J.H.Ross | Apocynaceae | ns | Stem is ground and mixed with cold water | Stem | Cattle | Diarrhoea | Dold and Cocks [34] |
| Senna petersiana (Bolle) Lock | Fabaceae | Monkey pod | Leaves are soaked | Leaves | Goats | General illnesses | Luseba and Tshisikhawe [37] |
| Senna petersiana (Bolle) Lock | Fabaceae | Monkey pod | Ground root powder is mixed with warm water to yield an infusion | Root | Cattle | Redwater | Ramovha and Van Wyk [48] |
| Sideroxylon inerme L. | Sapotaceae | White milkwood | Bark is crushed and boiled for 20 min | Bark | Cattle | Redwater | Dold and Cocks [34] |
| Solanum aculeastrum Dunal | Solanaceae | Goat bitter-apple | Fresh fruits are ground to pulp | Fruit | ns | Wounds | Magwede et al., [39] |
| Spirostachys africana Sond. | Euphorbiaceae | Tamboti | Bark is ground to pulp | Bark | Cattle | Wounds | Magwede et al., [39] |
| Spirostachys africana Sond. | Euphorbiaceae | Tamboti | ns | Wood | Cattle | Sweating sickness | Van der Merwe et al., [52] |
| Strychnos decussata (Pappe) Gilg. | Loganiaceae | Cape teak | Bark is crushed and soaked in water for 20 min, after which the infusion is strained | Bark | Cattle | Roundworms | Dold and Cocks [34] |
| Strychnos henningsii Gilg | Loganiaceae | Red bitter berry | Resin | ns | Cattle, sheep, goats | Arthritis | Chitura et al., [33] |
| Strychnos henningsii Gilg | Loganiaceae | Red bitter berry | Bark is soaked for 20 min and strained | Bark | Cattle | Heartwater and diarrhoea | Dold and Cocks [34] |
| Strychnos henningsii Gilg | Loganiaceae | Red bitter berry | Decoction | Bark | Goats | Gastro-intestinal parasites | Maphosa and Masika [41] |
| Strychnos henningsii Gilg | Loganiaceae | Red bitter berry | ns | Bark | Cattle | Paratyphoid (Goso) | Mthi et al., [45] |
| Tabernaemontana elegans Stapf | Apocynaceae | Toad tree | Crushed or in tact roots are soaked in water to yield an infusion or are cooked to produce a decoction | Roots | Cattle | Redwater | Ramovha and Van Wyk [48] |
| Tarchonanthus camphoratus L. | Asteraceae | Camphor bush | Maceration | Leaves | Cattle | Retained placenta and pain alleviation | Moichwanetse et al., [43] |
| Tarchonanthus camphoratus L. | Asteraceae | Camphor bush | Leaf infusion, oral route: The leaves of the plant are put in drinking water | Leaves | ns | To prevent cold | Ndou [47] |
| Terminalia sericea Burch. ex DC. | Combretaceae | Silver cluster-leaf | Ground roots are mixed with water to apply on the ticks and wounds. Roots are boiled and given to the animal | Roots | Cattle | Ticks and wounds, diarrhoea | Luseba and Tshisikhawe [37] |
| Terminalia sericea Burch. ex DC. | Combretaceae | Silver cluster-leaf | Ground leaves are mixed with water and applied on the wound and are covered with cattle dung | Leaves | Cattle | Wounds | Luseba and Van der Merwe [38] |
| Terminalia sericea Burch. ex DC. | Combretaceae | Silver cluster-leaf | Roots are ground to pulp and mixed with water | Roots | Cattle | Wounds and ticks | Magwede et al., [39] |
| Terminalia sericea Burch. ex DC. | Combretaceae | Silver cluster-leaf | Poultice | Leaves | Cattle | Retained placenta and uterus cleansing | Moichwanetse et al., [43] |
| Terminalia sericea Burch. ex DC. | Combretaceae | Silver cluster-leaf | Root-bark is soaked in cold water to yield an infusion, or dried bark is ground to a powder and is mixed with water | Roots, bark | Cattle | Redwater | Ramovha and Van Wyk [48] |
| Terminalia sericea Burch. ex DC. | Combretaceae | Silver cluster-leaf | ns | Roots | Cattle | Diarrhoea | Van der Merwe et al., [52] |
| Trema orientalis (L.) Blume | Cannabaceae | Pigeonwood | Ground leaves are mixed with water | Leaves | Cattle, goats, sheep | Eye problems and gallsickness | Luseba and Tshisikhawe [37] |
| Triumfetta sonderi Ficalho & Hiern | Malvaceae | Sonder’s truimfetta | ns | Root-bark | Cattle | Retained placenta | Van der Merwe et al., [52] |
| Turraea obtusifolia Hochst. | Meliaceae | Small honeysuckle tree | Crushed leaves are applied directly on the wounds | Leaves | Goats, sheep, cattle | Wounds | Luseba and Tshisikhawe [37] |
| Vachellia karroo (Hayne) Banfi & Galasso (Syn: Acacia karroo) | Fabaceae | Sweet thorn | Bark is chopped into small pieces and boiled | Bark | Goats, sheep | Diarrhoea and intestinal parasites | Dold and Cocks [34] |
| Vachellia karroo (Hayne) Banfi & Galasso (Syn: Acacia karroo) | Fabaceae | Sweet thorn | Maceration | Bulb | Cattle | Retained placenta and bacterial infections | Moichwanetse et al., [43] |
| Vachellia karroo (Hayne) Banfi & Galasso (Syn: Acacia karroo) | Fabaceae | Sweet thorn | Leaves are crushed and mixed with Madubula | Leaves | Cattle | Wounds and myiasis | Soyelu and Masika [51] |
| Vachellia karroo (Hayne) Banfi & Galasso (Syn: Acacia karroo) | Fabaceae | Sweet thorn | ns | Bark | Cattle | Fractures and diarrhoea | Van der Merwe et al., [52] |
| Vachellia karroo (Hayne) Banfi & Gallaso (Syn: Acacia karroo) | Fabaceae | Sweet thorn | For external coaptation of simple bone fractures (thobega) | Thorn, bark | ns | Fracture repair and splints for fracture repair | Ndou [47] |
| Vachellia tortilis (Forssk.) Galasso & Banfi (Syn: Acacia tortilis) | Fabaceae | Umbrella thorn | ns | Branch tips | Cattle | Diarrhoea | Van der Merwe et al., [52] |
| Volkameria glabra (E.Mey.) Mabb. & Y.W.Yuan (Syn: Clerodendrum capense D.Don ex Steud.) | Lamiaceae | Tinderwood | ns | Leaves | ns | Worms | Kambizi [35] |
| Withania somnifera (L.) Dunal | Solanaceae | winter cherry | Tuber infusion combined with roots of Solanum lichtensteinii and Bulbine abyssinica, oral route | Tubers | ns | Internal sores | Ndou [47] |
| Xanthocercis zambesiaca (Baker) Dumaz-le-Grand | Fabaceae | Nyala tree | Ground bark is given to cattle for eating disorders. Ground bark is mixed with salt or leaves are soaked for 12 h | Bark, leaves | Cattle | Eating problem and diarrhoea | Luseba and Tshisikhawe [37] |
| Xanthocercis zambesiaca (Baker) Dumaz-le-Grand | Fabaceae | Nyala tree | Ground bark is applied topically | Bark | ns | Wounds | Magwede et al., [39] |
| Ximenia americana L. var. microphylla Welw. ex Oliv. | Olacaceae | Tallowwood | Root-bark is powdered | Root bark | Cattle sheep, goats | Wounds | Luseba and Tshisikhawe [37] |
| Ximenia americana L. var. microphylla Welw. ex Oliv. | Olacaceae | Tallowwood | ns | Roots | Cattle | Internal parasites | Van der Merwe et al., [52] |
| Zanthoxylum capense (Thunb.) Harv. | Rutaceae | Small knobwood | Infusion prepared Grewia occidentalis, Olea europaea subsp. africana leaves and Aloe ferox sap | Leaves | ns | Gallsickess | Dold and Cocks [34] |
| Zanthoxylum capense (Thunb.) Harv. | Rutaceae | Small knobwood | Decoction | Roots | Goats | Gastro-intestinal parasites | Maphosa and Masika [41] |
| Ziziphus mucronata Willd. | Rhamnaceae | Buffalo thorn | Leaf paste | Leaves | Cattle | Mastitis | Chitura et al., [33] |
| Ziziphus mucronata Willd. | Rhamnaceae | Buffalo thorn | Bark is soaked in water while leaves are ground into pulp | Bark and leaves | ns | Wound | Magwede et al., [39] |
| Ziziphus mucronata Willd. | Rhamnaceae | Buffalo thorn | Infusion | Roots | Goats | Gastro-intestinal parasites | Maphosa and Masika [41] |
| Ziziphus mucronata Willd. | Rhamnaceae | Buffalo thorn | Poultice | Roots | Cattle | Retained placenta | Moichwanetse et al., (2020) |
| Ziziphus mucronata Willd. | Rhamnaceae | Buffalo thorn | Crushed leaves and soft branches poultice: crushed and placed on a hard abscess | Leaves, branches | ns | Abscess ripening | Ndou [47] |
| Ziziphus mucronata Willd. | Rhamnaceae | Buffalo thorn | ns | Leaves, roots | Cattle | Fertility enhancement, sores and burns | Van der Merwe et al., [52] |
| Ziziphus oxyphylla Edgew (Syn: Ziziphus acuminata Royle) | Rhamnaceae | Pointed-leaf jujube | Poultice | Roots | Cattle | Retained placenta and increase stimulation for separating retained placenta | Moichwanetse et al., [43] |
| Ziziphus zeyheriana Sond. | Rhamnaceae | Dwarf buffalo-thorn | ns | Root-stock | ns | Diarrhoea internal parasites. General ailments | Luseba and Van der Merwe [38] |
| Ziziphus zeyheriana Sond. | Rhamnaceae | Dwarf buffalo-thorn | Root decoction: Combined with roots of Cadaba aphylla, Senna italica and Dicoma galpinii. Root decoction combined with root of Helichrysum caespititium. Root decoction combined with root of Grewia flava, oral route. The sick calf is given about half a litre of the decoction orally | Roots | Cattle | Blood cleansing, pains (from sores, fractures), calf diarrhoea | Ndou [47] |
| # Plant Species | Plant Part | Solvent | Test System | Test Organism | Positive Control | Findings | Reference |
|---|---|---|---|---|---|---|---|
| Acokanthera oppositifolia | Leaves | Petroleum ether, dichloromethane, ethanol, and water | Serial microplate dilution | Bacillus subtilis, Staphylococcus aureus, Escherichia coli and Klebsiella pneumoniae | Neomycin (0.39–1.56 µg/mL) | All extract had no noteworthy (MIC > 1 mg/mL) antibacterial effect | Aremu et al., [71] |
| Alsophila dregei (Kunze) R.M.Tryon (Syn: Cyathea dregei) | Leaves | Acetone | Serial microplate dilution | Escherichia coli, Enterococcus faecalis, Pseudomonas aeruginosa, and Staphylococcus aureus | Gentamicin ≤ 0.02 mg/mL | Moderate antibacterial activity with MIC = 0.63 mg/mL | Adamu et al., [68] |
| Alsophila dregei (Kunze) R.M.Tryon (Syn: Cyathea dregei) | Leaves, roots | Petroleum ether, dichloromethane, ethanol, and water | Serial microplate dilution | Bacillus subtilis, Staphylococcus aureus, Escherichia coli and Klebsiella pneumoniae | Neomycin (0.39–1.56 µg/mL) | Petroleum ether and ethanol root extracts had noteworthy antibacterial activity (MIC < 1 mg/mL) against Gram-positive bacteria | Aremu et al., [71] |
| Apodytes dimidiata E.Mey. ex Arn. | Leaves | Acetone | Serial microplate dilution | Escherichia coli, Enterococcus faecalis, Pseudomonas aeruginosa, and Staphylococcus aureus | Gentamicin ≤ 0.02 mg/mL | Moderate antibacterial activity with MIC = 0.31 mg/mL against Staphylococcus aureus and Pseudomonas aeruginosa | Adamu et al., [68] |
| Baphia racemosa (Hochst.) Baker | Leaves | Acetone | Serial microplate dilution | Staphylococcus aureus, Enterococcus faecalis, Bacillus cereus, Escherichia coli, Pseudomonas aeruginosa, and Salmonella typhimurium | Gentamicin = 0.2–1.56 μg/mL | Noteworthy effect against Enterococcus faecalis (MIC = 160 μg/mL) and Staphylococcus aureus (MIC = 310 μg/mL) | Dzoyem et al., [67] |
| Berchemia zeyheri (Sond.) Grubov | Bark | Hexane, methanol, and water | Serial microplate dilution method | Escherichia coli, Enterococcus faecalis, Pseudomonas aeruginosa, and Staphylococcus aureus | Neomycin (0.78–25 μM) | Staphylococcus aureus was susceptible (MIC < 1 mg/mL) to hexane and methanol extracts | McGaw et al., [70] |
| # Bolusanthus speciosus (Bolus) Harms | Leaves | Acetone | Serial microplate dilution | Bacillus anthracis | Gentamicin = 0.0002 mg/mL | MIC = 0.04 mg/mL | Elisha et al., [66] |
| # Calpurnia aurea (Aiton) Benth. | Leaves | Acetone | Serial microplate dilution | Bacillus anthracis | Gentamicin = 0.0002 mg/mL | MIC = 0.31 mg/mL | Elisha et al., [66] |
| Clausena anisata (Willd.) Hook.f. ex. Benth. | Leaves | Acetone | Serial microplate dilution | Escherichia coli, Enterococcus faecalis, Pseudomonas aeruginosa, and Staphylococcus aureus | Gentamicin ≤ 0.02 mg/mL | Noteworthy antibacterial activity (MIC = 0.16–0.31 mg/mL) | Adamu et al., [68] |
| Combretum caffrum Eckl. & Zeyh.) Kuntze | Bark | Acetone, methanol, and water | Agar plate | Escherichia coli, Pseudomonas aeruginosa, Staphylococcus aureus, Bacillus cereus, Bacillus pumilus, Bacillus subtilis, Micrococcus kristinae, Klebsiella pneumonia, Serratia marcescens, and Enterobacter cloacae | ns | Methanol extract inhibited both Gram-positive and Gram-negative bacteria ranging from 0.5–5 mg/mL. Acetone extract mainly inhibited (MIC = 0.5 mg/mL) Gram-positive bacterial strains. Water extract showed activity against five Gram-positive and one Gram-negative bacteria | Masika and Afolayan [72] |
| Cremaspora triflora (Thonn.) K.Schum. | Leaves | Acetone | Serial microplate dilution | Bacillus anthracis | Gentamicin = 0.0002 mg/mL | MIC = 0.16 mg/mL | Elisha et al., [66] |
| Crotalaria capensis Jacq. | Leaves | Acetone | Serial microplate dilution | Staphylococcus aureus, Enterococcus faecalis, Bacillus cereus, Escherichia coli, Pseudomonas aeruginosa, and Salmonella typhimurium | Gentamicin = 0.2–1.56 μg/mL | Noteworthy effect against Enterococcus faecalis (MIC = 80 μg/mL) and Salmonella typhimurium (MIC = 20 μg/mL) | Dzoyem et al., [67] |
| # Cussonia spicata Thunb. | Bark | Methanol and dichloromethane | Serial microplate dilution | Escherichia coli, Pseudomonas aeruginosa, and Staphylococcus aureus | Neomycin = 0.l mg/mL | No noteworthy antibacterial activity | Luseba et al., [73] |
| # Cussonia spicata Thunb. | Root | Hexane, methanol, and water | Serial microplate dilution | Escherichia coli, Enterococcus faecalis, Pseudomonas aeruginosa, and Staphylococcus aureus | Neomycin (0.78–25 μM) | No noteworthy (MIC > 1 mg/mL) antibacterial effects for all of the tested extracts | McGaw et al., [70] |
| Dalbergia nitidula Baker | Leaves | Acetone | Serial microplate dilution | Staphylococcus aureus, Enterococcus faecalis, Bacillus cereus, Escherichia coli, Pseudomonas aeruginosa, and Salmonella typhimurium | Gentamicin = 0.2–1.56 μg/mL | Noteworthy effect against Bacillus cereus (MIC = 80 μg/mL) | Dzoyem et al., [67] |
| # Dombeya rotundifolia (Hochst.) Planch. | Aerial part | Hexane, methanol, and water | Serial microplate dilution | Escherichia coli, Enterococcus faecalis, Pseudomonas aeruginosa, and Staphylococcus aureus | Neomycin (0.78–25 μM) | Methanol extract had noteworthy (MIC = 0.4 mg/mL) antibacterial effect against Gram-positive bacteria | McGaw et al., [70] |
| Elaeodendron croceum (Thunb.) DC. | Leaves | Acetone | Serial microplate dilution | Bacillus anthracis | Gentamicin = 0.0002 mg/mL | MIC = 0.31 mg/mL | Elisha et al., [66] |
| Erythrina caffra Thunb. | Leaves | Acetone | Serial microplate dilution | Staphylococcus aureus, Enterococcus faecalis, Bacillus cereus, Escherichia coli, Pseudomonas aeruginosa, and Salmonella typhimurium | Gentamicin = 0.2–1.56 μg/mL | Noteworthy effect against Enterococcus faecalis (MIC = 80 μg/mL) | Dzoyem et al., [67] |
| # Euphorbia cupularis Boiss. Synadenium cuplare) | Stem/leaves | Hexane | Serial microplate dilution | Escherichia coli, Enterococcus faecalis, Pseudomonas aeruginosa, and Staphylococcus aureus | Neomycin (0.78–25 μM) | Hexane extract showed a weak inhibition against two Gram- positive | McGaw et al., [70] |
| # Heteromorpha arborescens (Spreng.) Cham. & Schltdl. | Leaves | Acetone | Serial microplate dilution | Bacillus anthracis | Gentamicin = 0.0002 mg/mL | MIC = 0.16 mg/mL | Elisha et al., [66] |
| Heteromorpha trifoliata (H.L.Wendl.) Eckl. & Zeyh | Leaves | Acetone | Serial microplate dilution | Escherichia coli, Enterococcus faecalis, Pseudomonas aeruginosa, and Staphylococcus aureus | Gentamicin ≤ 0.02 mg/mL | Moderate antibacterial activity with MIC = 0.63 against two Gram-negative bacteria | Adamu et al., [68] |
| # Hippobromus pauciflorus (L.f.) Radlk. | Aerial part | Hexane, methanol, and water | Serial microplate dilution | Escherichia coli, Enterococcus faecalis, Pseudomonas aeruginosa, and Staphylococcus aureus | Neomycin (0.78–25 μM) | Methanol extracts had noteworthy antibacterial effect (MIC = 0.2 mg/mL) against Staphylococcus aureus | McGaw et al., [70] |
| Indigofera frutescens L.f. (Syn: Indigofera cylindrica DC.) | Leaves | Acetone | Serial microplate dilution | Escherichia coli, Enterococcus faecalis, Pseudomonas aeruginosa, and Staphylococcus aureus | Gentamicin ≤ 0.02 mg/mL | Noteworthy antibacterial effect (MIC = 0.08–0.31 mg/mL) against the four bacterial strains | Adamu et al., [68] |
| Indigofera frutescens L.f. (Syn: Indigofera cylindrica DC.) | Leaves | Acetone | Serial microplate dilution | Staphylococcus aureus, Enterococcus faecalis, Bacillus cereus, Escherichia coli, Pseudomonas aeruginosa, and Salmonella typhimurium | Gentamicin = 0.2–1.56 μg/mL | Noteworthy effect against Salmonella typhimurium (MIC = 40 μg/mL) | Dzoyem et al., [67] |
| Leucosidea sericea Eckl. & Zeyh. | Leaves | Acetone | Serial microplate dilution | Escherichia coli, Enterococcus faecalis, Pseudomonas aeruginosa, and Staphylococcus aureus | Gentamicin ≤ 0.02 mg/mL | Noteworthy antibacterial effect (MIC = 0.02–0.08 mg/mL) against the four bacterial strains | Adamu et al., [68] |
| Leucosidea sericea Eckl. & Zeyh. | Leaves, stem | Petroleum ether, dichloromethane, ethanol, water | Serial microplate dilution | Bacillus subtilis, Staphylococcus aureus, Escherichia coli, and Klebsiella pneumonia | Neomycin (0.39–1.56 μg/mL) | Majority of the solvent extracts from the leaves had noteworthy antibacterial effect (MIC = 0.025–0.78 mg/mL) against all four bacterial strains. Stem organic solvent extracts had remarkable MIC (0.39–0.78 mg/mL) against Gram-positive bacteria | [69] |
| Lonchocarpus nelsii (Schinz) Heering & Grimme | Leaves | Acetone | Serial microplate dilution | Staphylococcus aureus, Enterococcus faecalis, Bacillus cereus, Escherichia coli, Pseudomonas aeruginosa, and Salmonella typhimurium | Gentamicin = 0.2–1.56 μg/mL | Noteworthy effect against Enterococcus faecalis and Salmonella typhimurium (MIC = 80 μg/mL) | Dzoyem et al., [67] |
| Maesa lanceolata Forssk. | Leaves | Acetone | Serial microplate dilution | Escherichia coli, Enterococcus faecalis, Pseudomonas aeruginosa, and Staphylococcus aureus | Gentamicin ≤ 0.02 mg/mL | Noteworthy antibacterial effect (MIC = 0.02–0.08 mg/mL) against the four bacterial strains | Adamu et al., [68] |
| Maesa lanceolata Forssk. | Leaves | Acetone | Serial microplate dilution | Bacillus anthracis | Gentamicin = 0.0002 mg/mL | MIC = 0.02 mg/mL | Elisha et al., [66] |
| Melia azedarach L. | Leaves | Acetone | Serial microplate dilution | Escherichia coli, Enterococcus faecalis, Pseudomonas aeruginosa, and Staphylococcus aureus | Gentamicin ≤ 0.02 mg/mL | Noteworthy antibacterial effect (MIC = 0.16–0.63 mg/mL) against the four bacterial strains | Adamu et al., [68] |
| # Millettia grandis (E.Mey.) Skeels | Leaves | Acetone | Serial microplate dilution | Escherichia coli, Enterococcus faecalis, Pseudomonas aeruginosa, and Staphylococcus aureus | Gentamicin ≤ 0.02 mg/mL | Moderate antibacterial activity with MIC = 0.31 mg/mL against four bacteria | Adamu et al., [68] |
| Morus mesozygia Stapf | Leaves | Acetone | Serial microplate dilution | Bacillus anthracis | Gentamicin = 0.0002 mg/mL | MIC = 0.04 mg/mL | Elisha et al., [66] |
| # Pittosporum viridiflorum Sims | Leaves | Acetone | Serial microplate dilution | Bacillus anthracis | Gentamicin = 0.0002 mg/mL | MIC = 0.08 mg/mL | Elisha et al., [66] |
| Podalyria calyptrata (Retz.) Willd. | Leaves | Acetone | Serial microplate dilution | Staphylococcus aureus, Enterococcus faecalis, Bacillus cereus, Escherichia coli, Pseudomonas aeruginosa, and Salmonella typhimurium | Gentamicin = 0.2–1.56 μg/mL | Noteworthy effect against Salmonella typhimurium (MIC = 160 μg/mL) | Dzoyem et al., [67] |
| # Pterocarpus angolensis DC. | Bark | Dichloromethane and 90% methanol | Serial microplate dilution | Escherichia coli, Pseudomonas aeruginosa, and Staphylococcus aureus | Neomycin = 0.l mg/mL | Moderate antibacterial activity with MIC = 0.31 mg/mL against Staphylococcus aureus | Luseba et al., [73] |
| Salix mucronata subsp. capensis (Thunb.) Immelman (Salix capensis) | Bark | Acetone, methanol, and water | Agar plate | Escherichia coli, Pseudomonas aeruginosa, Staphylococcus aureus, Bacillus cereus, Bacillus pumilus, Bacillus subtilis, Micrococcus kristinae, Klebsiella pneumonia, Serratia marcescens, and Enterobacter cloacae | ns | Acetone and methanol extracts inhibited both Gram-positive and Gram-negative bacteria ranging from 0.5 to 5 mg/mL | Masika and Afolayan [72] |
| # Schotia brachypetala Sond. | Bark, leavse | Hexane, methanol, and water | Serial microplate dilution | Escherichia coli, Enterococcus faecalis, Pseudomonas aeruginosa, and Staphylococcus aureus | Neomycin (0.78–25 μM) | Bark methanol extract had noteworthy antibacterial effect (MIC = 0.1–0.2 mg/mL) against two Gram-positive bacterial strains. Leaf methanol extract had noteworthy antibacterial effect (MIC = 0.2–0.4 mg/mL) against two Gram-positive bacterial strains | McGaw et al., [70] |
| # Sclerocarya birrea (A. Rich.) Hochst. | Leaves | Hexane, methanol, and water | Serial microplate dilution | Escherichia coli, Enterococcus faecalis, Pseudomonas aeruginosa, and Staphylococcus aureus | Neomycin (0.78–25 μM) | Methanol extract had noteworthy antibacterial effect (MIC = 0.1–0.4 mg/mL) against two Gram- positive bacterial strains | McGaw et al., [70] |
| # Searsia lancea (L.f.) F.A.Barkley (Rhus lanceas) | Bark, leaves | Hexane, methanol, and water | Serial microplate dilution | Escherichia coli, Enterococcus faecalis, Pseudomonas aeruginosa, and Staphylococcus aureus | Neomycin (0.78–25 μM) | Bark methanol extract had noteworthy antibacterial effect (MIC = 0.2 mg/mL) against two Gram-positive bacterial strains. Leaf methanol extract had noteworthy MIC (0.2 mg/mL) against Staphylococcus aureus | McGaw et al., [70] |
| Virgilia divaricata Adamson | Leaves | Acetone | Serial microplate dilution | Staphylococcus aureus, Enterococcus faecalis, Bacillus cereus, Escherichia coli, Pseudomonas aeruginosa, and Salmonella typhimurium | Gentamicin = 0.2–1.56 μg/mL | Noteworthy effect against Bacillus cereus and Salmonella typhimurium (MIC = 80 μg/mL) | Dzoyem et al., [67] |
| # Volkameria glabra (E. Mey.) Mabb. & Y. W. Yuan (Syn: Clerodendrum glabrum) | Leaves | Acetone | Serial microplate dilution | Escherichia coli, Enterococcus faecalis, Pseudomonas aeruginosa, and Staphylococcus aureus | Gentamicin ≤ 0.02 mg/mL | Noteworthy antibacterial effect (MIC = 0.31–0.63 mg/mL) against two Gram-negative bacterial strains | Adamu et al., [68] |
| Xylia torreana Brenan | Leaves | Acetone | Serial microplate dilution | Staphylococcus aureus, Enterococcus faecalis, Bacillus cereus, Escherichia coli, Pseudomonas aeruginosa, and Salmonella typhimurium | Gentamicin = 0.2–1.56 μg/mL | Noteworthy effect against Bacillus cereus and Salmonella typhimurium (MIC = 160 μg/mL) | Dzoyem et al., [67] |
| # Zanthoxylum capense (Thunb.) Harv. | Leaves | Acetone | Serial microplate dilution | Escherichia coli, Enterococcus faecalis, Pseudomonas aeruginosa, and Staphylococcus aureus | Gentamicin ≤ 0.02 mg/mL | Noteworthy antibacterial effect (MIC = 0.31 mg/mL) against Enterococcus faecalis and Pseudomonas aeruginosa | Adamu et al., [68] |
| # Ziziphus mucronata Willd. | Bark, leaves | Hexane, methanol, and water | Serial microplate dilution | Escherichia coli, Enterococcus faecalis, Pseudomonas aeruginosa, and Staphylococcus aureus | Neomycin (0.78–25 μM) | Bark extracts had no noteworthy antibacterial effect (MIC > 1 mg/mL) against all of the tested bacterial strains. Leaf methanol extracts had noteworthy antibacterial effect (MIC = 0.2 mg/mL) against Staphylococcus aureus | McGaw et al., [70] |
| # Plant Species | Solvent | $ Assay Type | Plant-Part | Parasite | Positive Control | * Findings | Reference |
|---|---|---|---|---|---|---|---|
| # Acokanthera oppositifolia (Lam.) Codd | Petroleum ether, dichloromethane, ethanol, and water | CA | Leaves, twigs | Caenorhabditis elegans | Levamisole (40 μg/mL) | Petroleum ether and ethanol leaf extracts had noteworthy MLC (0.52 mg/mL) | Aremu et al., [71] |
| Alsophila dregei (Kunze) R.M.Tryon (Cyathea dregei) | Petroleum ether, dichloromethane, ethanol, and water | CA | Leaves, roots | Caenorhabditis elegans | Levamisole (40 μg/mL) | Dichloromethane and ethanol leaf extracts had noteworthy MLC (0.52 mg/mL) | Aremu et al., [71] |
| Alsophila dregei (Kunze) R.M.Tryon (Cyathea dregei) | Acetone | DBA | Leaves | Haemonchus contortus | Albendazole (0.008–25 μg/mL) | EC50 = 17.64 mg/mL (EHA), 17.93 mg/mL (LDT) | Adamu et al., [76] |
| Alsophila dregei (Kunze) R.M.Tryon (Cyathea dregei) | Petroleum ether, dichloromethane, ethanol, and water | CA | Leaves, roots | Caenorhabditis elegans | Levamisole (40 μg/mL) | Dichloromethane and ethanol extracts had noteworthy MLC (0.52 mg/mL) | Aremu et al., [71] |
| Apodytes dimidiata | Acetone | DBA | Leaves | Haemonchus contortus | Albendazole (0.008–25 μg/mL) | EC50 = 5.7 mg/mL (EHA), 4.13 mg/mL (LDT) | Adamu et al., [76] |
| Berchemia zeyheri | Hexane, methanol, and water | DBA | Bark | Caenorhabditis elegans | Levamisole (10 μg/mL) | Methanol extract had moderate (30%) lethality at 2 mg/mL | McGaw et al., [70] |
| Brachylaena discolor | Acetone | DBA | Leaves | Haemonchus contortus | Albendazole (0.008–25 μg/mL) | EC50 = 3.55 mg/mL (EHA), 17.23 mg/mL (LDT) | Adamu et al., [76] |
| # Calpurnia aurea | Acetone | DBA | Leaves/flowers, stem | Haemonchus contortus | Albendazole (100% at 0.008–25 μg/mL) | EHA inhibition = 27% (leaves/flowers), 32% (stem) | Fouche et al., [75] |
| Clausena anisata | Acetone | DBA | Leaves | Haemonchus contortus | Albendazole (0.008–25 μg/mL) | EC50 = 1.8 mg/mL (EHA), 2.07 mg/mL (LDT) | Adamu et al., [76] |
| Combretum apiculatum Sond. Subsp. Apiculatum | Ethyl acetate, acetone, and water | DBA | Leaves | Caenorhabditis elegans | Levamisole | Ethyl acetate extract had 70–80% lethality at 1 mg/mL | McGaw et al., [80] |
| Combretum bracteosum (Hochst.) Engl. & Diels | Ethyl acetate, acetone, and water | DBA | Leaves | Caenorhabditis elegans | Levamisole | No activity observed | McGaw et al., [80] |
| Combretumcelastroides Welw ex Laws subsp. Celastroides | Ethyl acetate, acetone, and water | DBA | Leaves | Caenorhabditis elegans | Levamisole | No activity observed | McGaw et al., [80] |
| Combretumcollinum Fresen | Ethyl acetate, acetone, and water | DBA | Leaves | Caenorhabditis elegans | Levamisole | Acetone extract had 10–20% lethality at 0.5 and 1 mg/mL | McGaw et al., [80] |
| Combretum edwardsii Exell | Ethyl acetate, acetone, and water | DBA | Leaves | Caenorhabditis elegans | Levamisole | Acetone and ethyl acetate extracts had 10–20% lethality at 1 mg/mL | McGaw et al., [80] |
| Combretumerythrophyllum (Burch.) Sond. | Ethyl acetate, acetone, and water | DBA | Leaves | Caenorhabditis elegans | Levamisole | Acetone and ethyl acetate extracts had 10–20% lethality at 1 mg/mL | McGaw et al., [80] |
| Combretumhereroense Schinz | Ethyl acetate, acetone, and water | DBA | Leaves | Caenorhabditis elegans | Levamisole | Acetone extract had 20–30% lethality at 1 mg/mL | McGaw et al., [80] |
| Combretumimberbe Wawra | Ethyl acetate, acetone, and water | DBA | Leaves | Caenorhabditis elegans | Levamisole | Acetone extract had 20–30% lethality at 1 mg/mL | McGaw et al., [80] |
| Combretumkraussii Hochst. (Syn: Combretum nelsonii) | Ethyl acetate, acetone, and water | DBA | Leaves | Caenorhabditis elegans | Levamisole | Acetone extract had 20–30% lethality at 0.5 mg/mL | McGaw et al., [80] |
| # Combretum microphyllum | Ethyl acetate, acetone, and water | DBA | Leaves | Caenorhabditis elegans | Levamisole | Acetone and ethyl acetate extracts had 10–20% lethality at 0.5 and 1 mg/mL | McGaw et al., [80] |
| Combretum mkuzense J.D.Carr & Retief | Ethyl acetate, acetone, and water | DBA | Leaves | Caenorhabditis elegans | Levamisole | Acetone extract had 20–30% lethality at 1 mg/mL | McGaw et al., [80] |
| Combretum moggi Exell | Ethyl acetate, acetone, and water | DBA | Leaves | Caenorhabditis elegans | Levamisole | No activity observed | McGaw et al., [80] |
| # Combretum molle R.Br. ex. G.Don | Ethyl acetate, acetone, and water | DBA | Leaves | Caenorhabditis elegans | Levamisole | Acetone extract had 20–30% lethality at 1 mg/mL | McGaw et al., [80] |
| Combretum mossambicense (Klotzsch) Engl. | Ethyl acetate, acetone, and water | DBA | Leaves | Caenorhabditis elegans | Levamisole | Acetone extract had 20–30% lethality at 1 mg/mL | McGaw et al., [80] |
| Combretum padoides Engl. | Ethyl acetate, acetone, and water | DBA | Leaves | Caenorhabditis elegans | Levamisole | No activity observed | McGaw et al., [80] |
| # Combretum paniculatum Vent. | Ethyl acetate, acetone, and water | DBA | Leaves | Caenorhabditis elegans | Levamisole | Acetone extract had 10–20% lethality at 0.5 mg/mL | McGaw et al., [80] |
| Combretum petrophilum Retief | Ethyl acetate, acetone, and water | DBA | Leaves | Caenorhabditis elegans | Levamisole | Acetone extract had 10–20% lethality at 0.5 mg/mL | McGaw et al., [80] |
| Combretum woodii | Ethyl acetate, acetone, and water | DBA | Leaves | Caenorhabditis elegans | Levamisole | No activity observed | McGaw et al., [80] |
| Combretum zeyheri | Ethyl acetate, acetone, and water | DBA | Leaves | Caenorhabditis elegans | Levamisole | No activity observed | McGaw et al., [80] |
| # Curtisia dentata | Dichloromethane and acetone | DBA | Leaves | Caenorhabditis elegans, Haemonchus contortus, Trichostrongylus colubriformis | Levamisole (10 μg/mL) | Acetone extracts had the highest inhibition at 2.5 mg/mL after 2 h and 7 days of incubation. Both extracts inhibited the highest motility at 1.25–2.5 (Haemonchus contortus) and 0.63–2.5 mg/mL (Trichostrongylus colubriformis) | Shai et al., [81] |
| # Cussonia spicata | Hexane, methanol, and water | DBA | Roots | Caenorhabditis elegans | Levamisole (10 μg/mL) | No noteworthy activity | McGaw et al., [70] |
| # Dombeya rotundifolia | Hexane, methanol, and water | DBA | Aerial parts | Caenorhabditis elegans | Levamisole (10 μg/mL) | Water extract had 20% lethality at 1 and 2 mg/mL | McGaw et al., [70] |
| # Euphorbia cupularis (Syn: Synadenium cupulare (Boiss.) | Hexane, methanol, and water | DBA | Stem/leaves | Caenorhabditis elegans | Levamisole (10 μg/mL) | No noteworthy activity | McGaw et al., [70] |
| Ficus sycomorus | Acetone | DBA | Bark/stem, stem | Haemonchus contortus | Albendazole (100% at 0.008–25 μg/mL) | EHA inhibition = 25% (bark/stem), 21% (stem) | Fouche et al., [75] |
| Heteromorpha trifoliata | Acetone | DBA | Leaves | Haemonchus contortus | Albendazole (0.008–25 μg/mL) | EC50 = 0.62 mg/mL (EHA), 0.64 mg/mL (LDT) | Adamu et al., [76] |
| # Hippobromus pauciflorus | Hexane, methanol, and water | DBA | Aerial parts | Caenorhabditis elegans | Levamisole (10 μg/mL) | Hexane extract had 50% lethality at 2 mg/mL | McGaw et al., [70] |
| Indigofera frutescens | Acetone | DBA | Leaves | Haemonchus contortus | Albendazole (0.008–25 μg/mL) | EC50 = 7.11 mg/mL (EHA), 7.58 mg/mL (LDT) | Adamu et al., [76] |
| Leucosidea sericea | Acetone | DBA | Leaves | Haemonchus contortus | Albendazole (0.008–25 μg/mL) | EC50 = 1.08 mg/mL (EHA), 1.27 mg/mL (LDT) | Adamu et al., [76] |
| Leucosidea sericea | Petroleum ether, dichloromethane, ethanol, and water | CA | Leaves, stem | Caenorhabditis elegans | Levamisole (40 μg/mL) | Petroleum ether, dichloromethane, and ethanol leaf extracts had noteworthy anthelmintic effect (MLC = 0.26–0.52 mg/mL) | Aremu et al., [69] |
| # Maerua angolensis | Acetone | DBA | Stem, leaves | Haemonchus contortus | Albendazole (100% at 0.008–25 μg/mL) | EHA inhibition = 65% (stem), 25% (leaves) | Fouche et al., [75] |
| Maesa lanceolata | Acetone | DBA | Leaves | Haemonchus contortus | Albendazole (0.008–25 μg/mL) | EC50 = 0.72 mg/mL (EHA), 1.68 mg/mL (LDT) | Adamu et al., [76] |
| Melia azedarach | Acetone | DBA | Leaves | Haemonchus contortus | Albendazole (0.008–25 μg/mL) | EC50= 6.24 mg/mL (EHA), 10.96 mg/mL (LDT) | Adamu et al., [76] |
| # Milletia grandis | Acetone | DBA | Leaves | Haemonchus contortus | Albendazole (0.008–25 μg/m) | EC50= 5.57 mg/mL (EHA), 6.11 mg/mL (LDT) | Adamu et al., [76] |
| # Schotia brachypetala Sond | Hexane, methanol, and water | DBA | Leaves, bark | Caenorhabditis elegans | Levamisole (10 μg/mL) | All solvent extracts from, bark had 10% at 2 mg/mL. Hexane extract from leaves had 10% lethality at 2 mg/mL | McGaw et al., [70] |
| # Sclerocarya birrea | Acetone | DBA | Fruit | Haemonchus contortus | Albendazole (100% at 0.008–25 μg/mL) | EHA inhibition = 28% | Fouche et al., [75] |
| # Sclerocarya birrea | Hexane, methanol, and water | DBA | Bark | Caenorhabditis elegans | Levamisole (10 μg/mL) | Methanol extract had 40% lethality at 2 mg/mL | McGaw et al., [70] |
| # Searsia lancea (Syn: Rhus lancea) | Hexane, methanol, and water | DBA | Leaves, bark | Caenorhabditis elegans | Levamisole (10 μg/mL) | Hexane extracts had 50% (leaves) and 40% (bark) lethality at 2 mg/mL | McGaw et al., [70] |
| # Senna petersiana | Petroleum ether, dichloromethane, ethanol, and water | CA | Leaves | Caenorhabditis elegans | Levamisole (40 μg/mL) | Ethanol extract = 0.52 mg/mL | Aremu et al., [71] |
| Strychnos mitis | Acetone | DBA | Leaves | Haemonchus contortus | Albendazole (0.008–25 μg/mL) | EC50 = 16.56 mg/mL (EHA), 16.94 mg/mL (LDT) | Adamu et al., [76] |
| # Tabernaemontana elegans | Acetone | DBA | Leaves | Haemonchus contortus | Albendazole (100% at 0.008–25 μg/mL) | EHA inhibition = 47% | Fouche et al., [75] |
| # Volkameria glabra (Clerodendrum glabrum) | Acetone | DBA | Leaves | Haemonchus contortus | Albendazole (0.008–25 μg/mL) | EC50 = 1.48 mg/mL (EHA), 12.97 mg/mL (LDT) | Adamu et al., [76] |
| # Zanthoxylum capense | Acetone | DBA | Leaves | Haemonchus contortus | Albendazole (0.008–25 μg/mL) | EC50 = 13.26 mg/mL (EHA), 13.64 mg/mL (LDT) | Adamu et al., [76] |
| # Ziziphus mucronata | Hexane, methanol, and water | DBA | Bark, leaves | Caenorhabditis elegans | Levamisole (10 μg/mL) | No noteworthy activity | McGaw et al., [70] |
| # Plant Species | Assay Type | Plant Part | Findings | Reference |
|---|---|---|---|---|
| Alsophila dregei (Kunze) R.M.Tryon (Syn: Cyathea dregei) | DPPH | Leaves | EC50 = 3 µg/mL | Adamu et al., [68] |
| Alsophila dregei (Kunze) R.M.Tryon (Syn: Cyathea dregei) | ABTS | Leaves | 0.4 TEAC | Adamu et al., [68] |
| Apodytes dimidiata E.Mey. ex. Arn. | DPPH | Leaves | EC50 = 3.5 µg/mL | Adamu et al., [68] |
| Apodytes dimidiata E.Mey. ex. Arn. | ABTS | Leaves | 0.3 TEAC | Adamu et al., [68] |
| Brachylaena discolor DC. | DPPH | Leaves | EC50 = 2.6 µg/mL | Adamu et al., [68] |
| Brachylaena discolor DC. | ABTS | Leaves | 0.2 TEAC | Adamu et al., [68] |
| Burkea africana Hook. | DPPH | Leaves | IC50 = 3.55 µg/mL | Dzoyem and Eloff [85] |
| Burkea africana Hook. | ABTS | Leaves | IC50 = 3.21 µg/mL | Dzoyem and Eloff [85] |
| Burkea africana Hook. | FRAP | Leaves | IC50 = 231.07 µg Fe (II)/g | Dzoyem and Eloff [85] |
| Clausena anisata (Willd.) Hook.f. ex. Benth. | DPPH | Leaves | EC50 = 2.5 µg/mL | Adamu et al., [68] |
| Clausena anisata (Willd.) Hook.f. ex. Benth. | ABTS | Leaves | 0.2 TEAC | Adamu et al., [68] |
| Combretum zeyheri Sond. | DPPH | Leaves | IC50 = 3.52 µg/mL | Dzoyem and Eloff [85] |
| Combretum zeyheri Sond. | ABTS | Leaves | IC50 = 4.64 µg/mL | Dzoyem and Eloff [85] |
| Combretum zeyheri Sond. | FRAP | Leaves | IC50 = 95.98 µg Fe (II)/g | Dzoyem and Eloff [85] |
| Dalbergia nitidula Welw. ex. Baker | DPPH | Leaves | IC50 = 9.31 μg/mL | Dzoyem et al., [67] |
| Dalbergia nitidula Welw. ex. Baker | ABTS | Leaves | IC50 = 21.3 μg/mL | Dzoyem et al., [67] |
| # Englerophytum magalismontanum (Sond.) T.D.Penn | DPPH | Leaves | IC50 = 10.8 µg/mL | Dzoyem and Eloff [85] |
| # Englerophytum magalismontanum (Sond.) T.D.Penn | ABTS | Leaves | IC50 = 12.22 µg/mL | Dzoyem and Eloff [85] |
| # Englerophytum magalismontanum (Sond.) T.D.Penn | FRAP | Leaves | IC50 = 76 µg Fe (II)/g | Dzoyem and Eloff [85] |
| #Erythrina caffra Thunb. | DPPH | Leaves | IC50 = 268.6 μg/mL | Dzoyem et al., [67] |
| #Erythrina caffra Thunb. | ABTS | Leaves | IC50 = 173.28 μg/mL | Dzoyem et al., [67] |
| Euclea undulata Thunb. | DPPH | Leaves | 31.66 µg/mL | Dzoyem and Eloff [85] |
| Euclea undulata Thunb. | ABTS | Leaves | 32.67 µg/mL | Dzoyem and Eloff [85] |
| Euclea undulata Thunb. | FRAP | Leaves | 274.19 µg Fe (II)/g | Dzoyem and Eloff [85] |
| Heteromorpha trifoliata (H.L.Wendl.) Eckl. & Zeyh. | DPPH | Leaves | EC50 = 4.36 µg/mL | Adamu et al., [68] |
| Heteromorpha trifoliata (H.L.Wendl.) Eckl. & Zeyh. | ABTS | Leaves | 0.2 TEAC | Adamu et al., [68] |
| Indigofera frutescens L.f. | DPPH | Leaves | EC50 = 0 µg/mL | Adamu et al., [68] |
| Indigofera frutescens L.f. | ABTS | Leaves | 0.5 TEAC | Adamu et al., [68] |
| Indigofera frutescens L.f. | DPPH | Leaves | IC50 = 22.31 μg/mL | Dzoyem et al., [67] |
| Indigofera frutescens L.f. | ABTS | Leaves | IC50 = 134.64 μg/mL | Dzoyem et al., [67] |
| # Jatropha curcas L. | DPPH | Leaves | IC50 = 137.08 µg/mL | Dzoyem and Eloff [85] |
| # Jatropha curcas L. | ABTS | Leaves | IC50 = 115.23 µg/mL | Dzoyem and Eloff [85] |
| # Jatropha curcas L. | FRAP | Leaves | IC50 = 68.17 µg Fe (II)/g | Dzoyem and Eloff [85] |
| Leucaena leucocephala (Lam.) de Wit | DPPH | Leaves | IC50 = 9.86 µg/mL | Dzoyem and Eloff [85] |
| Leucaena leucocephala (Lam.) de Wit | ABTS | Leaves | IC50 = 9.85 µg/mL | Dzoyem and Eloff [85] |
| Leucaena leucocephala (Lam.) de Wit | FRAP | Leaves | IC50 = 289.27 µg Fe (II)/g | Dzoyem and Eloff [85] |
| Leucosidea sericea | DPPH | Leaves | EC50 = 0.0 µg/mL | Adamu et al., [68] |
| Leucosidea sericea | ABTS | Leaves | 0.7 TEAC | Adamu et al., [68] |
| Maesa lanceolata Forssk. | DPPH | Leaves | EC50 = 1.4 µg/mL | Adamu et al., [68] |
| Maesa lanceolata Forssk. | ABTS | Leaves | 1.2 TEAC | Adamu et al., [68] |
| Melia azedarach L. | DPPH | Leaves | EC50 = 3.3 µg/mL | Adamu et al., [68] |
| Melia azedarach L. | ABTS | Leaves | 0.8 TEAC | Adamu et al., [68] |
| # Millettia grandis (E.Mey.) Skeels | DPPH | Leaves | EC50 = 4.6 µg/mL | Adamu et al., [68] |
| # Millettia grandis (E.Mey.) Skeels | ABTS | Leaves | 0.6 TEAC | Adamu et al., [68] |
| Morus mesozygia Stapf | DPPH | Leaves | IC50 = 15.85 µg/mL | Dzoyem and Eloff [85] |
| Morus mesozygia Stapf | ABTS | Leaves | IC50 = 271.86 µg/mL | Dzoyem and Eloff [85] |
| Morus mesozygia Stapf | FRAP | Leaves | IC50 = 127.34 µg Fe (II)/g | Dzoyem and Eloff [85] |
| Philenoptera nelsii (Schinz) Schrire (Lonchocarpus nelsii) | DPPH | Leaves | IC50 = 247.7 μg/mL | Adamu et al., [68] |
| Philenoptera nelsii (Schinz) Schrire (Lonchocarpus nelsii) | ABTS | Leaves | IC50 = 41.39 μg/mL | Adamu et al., [68] |
| Strychnos mitis S.Moore | DPPH | Leaves | EC50 = 3.5 µg/mL | Adamu et al., [68] |
| Strychnos mitis S.Moore | ABTS | Leaves | 0.3 TEAC | Adamu et al., [68] |
| Uapaca nitida Müll.Arg. | DPPH | Leaves | IC50 = 125.86 µg/mL | Dzoyem and Eloff [85] |
| Uapaca nitida Müll.Arg. | ABTS | Leaves | IC50 = 28.81 µg/mL | Dzoyem and Eloff [85] |
| Uapaca nitida Müll.Arg. | FRAP | Leaves | IC50 = 177.32 µg Fe (II)/g | Dzoyem and Eloff [85] |
| # Volkameria glabra (E. Mey.) Mabb. & Y. W. Yuan (Syn: Clerodendrum glabrum) | DPPH | Leaves | EC50 = 3.5 µg/mL | Adamu et al., [68] |
| # Volkameria glabra (E. Mey.) Mabb. & Y. W. Yuan (Syn: Clerodendrum glabrum) | ABTS | Leaves | 0.5 TEAC | Adamu et al., [68] |
| # Zanthoxylum capense (Thunb.) Harv. | DPPH | Leaves | EC50 = 4 µg/mL | Adamu et al., [68] |
| # Zanthoxylum capense (Thunb.) Harv. | ABTS | Leaves | 0.4 TEAC | Adamu et al., [68] |
| # Plant Species | Solvent | Plant Part | * Test System | Positive Control | Findings | Reference |
|---|---|---|---|---|---|---|
| Alsophila dregei (Syn: Cyathea dregei) | Acetone | Leaves | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Berberine chloride | LC50 = 0.00332 mg/mL | Adamu et al., [76] |
| Apodytes dimidiata | Acetone | Leaves | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Berberine chloride | LC50 = 0.00396 mg/mL | Adamu et al., [76] |
| Berchemia zeyheri | Hexane, methanol, and water | Bark | Brine shrimp lethality/toxicity using Artemia salina | Podophyllotoxin (7 μg/mL) | Water extract had the highest lethal effect (LC50 = 3.9 mg/mL) | McGaw et al., [70] |
| # Bolusanthus speciosus | Acetone | Leaves | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Doxorubicin = 1.76 µg/mL | LC50 = 52.8 μg/mL | Elisha et al., [66] |
| Brachylaena discolor | Acetone | Leaves | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Berberine chloride | LC50 = 0.00752 mg/mL | Adamu et al., [76] |
| # Calpurnia aurea | Acetone | Leaves | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Doxorubicin = 1.76 µg/mL | LC50 = 13.6 μg/mL | Elisha et al., [66] |
| # Calpurnia aurea | Acetone | Leaves/flowers, stem | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Doxorubicin (2.97 μg/mL) | Leaves/flowers, LC50 = 166.63 μg/mL, Stem LC50 = 223.97 μg/mL | Fouche et al., [75] |
| Clausena anisata | Acetone | Leaves | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Berberine chloride | LC50 = 0.17186 mg/mL | Adamu et al., [76] |
| Cremaspora triflora | Acetone | Leaves | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Doxorubicin = 1.76 µg/mL | LC50 = 57.4 μg/mL | Elisha et al., [66] |
| # Cussonia spicata | Hexane, methanol, and water | Roots | Brine shrimp lethality/toxicity using Artemia salina | Podophyllotoxin (7 μg/mL) | Water extract had the highest lethal effect (LC50 = 2.6 mg/mL) | McGaw et al., [70] |
| Dalbergia nitidula | Acetone | Leaves | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Doxorubicin (2.29 μg/mL) | LC50 = 51.28 μg/mL | Dzoyem et al., [67] |
| # Dombeya rotundifolia | Hexane, methanol, and water | Aerial part | Brine shrimp lethality/toxicity using Artemia salina | Podophyllotoxin (7 μg/mL) | All extracts had no lethal effect | McGaw et al., [70] |
| Elaeodendron croceum (Thunb.) DC. | Acetone | Leaves | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Doxorubicin = 1.76 µg/mL | LC50 = 5.2 μg/mL | Elisha et al., [66] |
| # Erythrina caffra | Acetone | Leaves | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Doxorubicin (2.29 μg/mL) | LC50 = 19.93 μg/mL | Dzoyem et al., [67] |
| # Euphorbia cupularis (Syn: Synadenium cupulare) | Hexane, methanol, and water | Aerial part | Brine shrimp lethality/toxicity using Artemia salina | Podophyllotoxin (7 μg/mL) | All extracts had no lethal effect | McGaw et al., [70] |
| Ficus sycomorus | Acetone | Bark/stem, stem | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Doxorubicin (2.97 μg/mL) | LC50 = 172.94 μg/mL (bark/stem), LC50 = 48.74 μg/mL (stem) | Fouche et al., [75] |
| # Heteromorpha arborescens | Acetone | Leaves | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Doxorubicin = 1.76 µg/mL | LC50 = 81.0 μg/mL | Elisha et al., [66] |
| Heteromorpha trifoliata | Acetone | Leaves | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Berberine chloride | LC50 = 0.04252 mg/mL | Adamu et al., [76] |
| # Hippobromus pauciflorus | Hexane, methanol, and water | Aerial part | Brine shrimp lethality/toxicity using Artemia salina | Podophyllotoxin (7 μg/mL) | All extract had no lethal effect | McGaw et al., [70] |
| Indigofera cylindrica | Acetone | Leaves | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Doxorubicin (2.29 μg/mL) | LC50 = 77.59 μg/mL | Dzoyem et al., [67] |
| Indigofera frutescens | Acetone | Leaves | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Berberine chloride | LC50 = 0.1044 mg/mL | Adamu et al., [76] |
| Leucosidea sericea | Acetone | Leaves | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Berberine chloride | LC50 = 0.0515 mg/mL | Adamu et al., [76] |
| Lonchocarpus nelsii | Acetone | Leaves | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Doxorubicin (2.29 μg/mL) | LC50 = 81.09 μg/mL | Dzoyem et al., [67] |
| # Maerua angolensis | Acetone | Stem, leaves | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Doxorubicin (2.97 μg/mL) | LC50 = 180.64 μg/mL (stem), LC50 = 73.76 μg/mL (leaves) | Fouche et al., [75] |
| Maesa lanceolata | Acetone | Leaves | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Berberine chloride | LC50 = 0.01577 mg/mL | Adamu et al., [76] |
| Maesa lanceolata | Acetone | Leaves | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Doxorubicin = 1.76 µg/mL | LC50 = 0.38 μg/mL | Elisha et al., [66] |
| Melia azedarach | Acetone | Leaves | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Berberine chloride | LC50 = 0.14466 mg/mL | Adamu et al., [76] |
| # Milletia grandis | Acetone | Leaves | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Berberine chloride | LC50 = 0.05336 mg/mL | Adamu et al., [76] |
| Morus mesozygia | Acetone | Leaves | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Doxorubicin = 1.76 µg/mL | LC50 = 40.7 μg/mL | Elisha et al., [66] |
| # Pittosporum viridiflorum | Acetone | Leaves | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Doxorubicin = 1.76 µg/mL | LC50 = 54.6 μg/mL | Elisha et al., [66] |
| # Pterocarpus angolensis | Hexane, methanol, and water | Bark, leaves | Brine shrimp lethality/toxicity using Artemia salina | Podophyllotoxin (7 μg/mL) | All extracts from the bark had no lethal effect. Hexane and methanol extracts from the leaves had the highest lethal effect (LC50 = 3.6–3.8 mg/mL) | McGaw et al., [70] |
| # Sclerocarya birrea | Acetone | Fruit | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Doxorubicin (2.97 μg/mL) | LC50 = 214.79 μg/mL | Fouche et al., [75] |
| # Sclerocarya birrea | Hexane, methanol, and water | Bark | Brine shrimp lethality/toxicity using Artemia salina | Podophyllotoxin (7 μg/mL) | All extracts had no lethal effect | McGaw et al., [70] |
| # Searsia lancea (Rhus lancea) | Hexane, methanol, and water | Bark, leaves | Brine shrimp lethality/toxicity using Artemia salina | Podophyllotoxin (7 μg/mL) | Water extract from the bark (LC50 = 3.9 mg/mL) and leaves (LC50 = 0.6 mg/mL) had the highest toxic effect | McGaw et al., [70] |
| Strychnos mitis | Acetone | Leaves | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Berberine chloride | LC50 = 0.01721 mg/mL | Adamu et al., [76] |
| # Tabernaemontana elegans | Acetone | Leaves | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Doxorubicin (2.97 μg/mL) | LC50 = 32.35 μg/mL | Fouche et al., [75] |
| # Tetradenia riparia | Acetone and water | Flowers, leaves | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Doxorubicin (5.4326 µM) | LC50, flowers = 0.0823 mg/mL (acetone), 0.1784 mg/mL (water); leaves = 0.0513 mg/mL (acetone), 0.2738 mg/mL (water) | Sserunkuma et al., [86] |
| # Vachellia nilotica (Acacia nilotica) | Acetone and water | Bark, leaves | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Doxorubicin (5.4326 µM) | LC50, bark = 0.0332 mg/mL (acetone), 0.0278 mg/mL (water); leaves = 0.2187 mg/mL (acetone), 0.0688 mg/mL (water) | Sserunkuma et al., [86] |
| Virgilia divaricata Adamson | Acetone | Leaves | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Doxorubicin (2.29 μg/mL) | LC50 = 30.08 μg/mL | Dzoyem et al., [67] |
| # Volkameria glabra (Clerodendrum glabrum) | Acetone | Leaves | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Berberine chloride | LC50 = 0.04251 mg/mL | Adamu et al., [76] |
| # Zanthoxylum capense | Acetone | Leaves | Tetrazolium-based colorimetric MTT assay using Vero monkey kidney cells | Berberine chloride | LC50 = 0.02095 mg/mL | Adamu et al., [76] |
| # Ziziphus mucronata | Hexane, methanol and water | Bark, leaves | Brine shrimp lethality/toxicity using Artemia salina | Podophyllotoxin (7 μg/mL) | Bark extracts had no lethal effect. Hexane extract of leaves had LC50 = 0.9 mg/mL | McGaw et al., [70] |
| # Plant Species | Plant Part | Extract | Findings | Reference |
|---|---|---|---|---|
| # Acokanthera oppositifolia | Leaves, twigs | 50% Methanol | TPC = 2.5 and 7.2 mg GAE/g GC = 2 µg and 5.2 µg GAE/g CT = 0.005% and 0.12% LCE/g TFC = 0.002 and 0.001 mg CTE/g | Aremu et al., [71] |
| Burkea africana | Leaves | Acetone | TPC = 14.39 mg GAE/g | Dzoyem and Eloff [85] |
| Combretum zeyheri | Leaves | Acetone | TPC = 3.29 mg GAE/g | Dzoyem and Eloff [85] |
| # Curtisia dentata | Stem bark | Acetone | TPC = 8.94 mg GAE/g | Olaokun et al., [90] |
| Dalbergia nitidula | Leaves | Acetone | TPC = 1.51 mg GAE/g | Dzoyem et al., [67] |
| # Englerophytum magalismontanum | Leaves | Acetone | TPC = 0.86 mg GAE/g | Dzoyem and Eloff [85] |
| # Erythrina caffra | Leaves | Acetone | TPC = 150.82 mg/g GAE TFC = 72.8 mg QE/g | Dzoyem et al., [67] |
| Euclea undulata | Leaves | Acetone | TPC = 234.56 mg/g GAE TFC = 64.36 mg QE/g | Dzoyem and Eloff [85] |
| Indigofera frutescens (Indigofera cylindrical) | Leaves | Acetone | TPC = 125.12 mg/g GAE TFC = 27.69 mg/g QE | Dzoyem et al., [67] |
| # Jatropha curcas | Leaves | Acetone | TPC = 100.89 mg/g GAE TFC = 68.43 mg QE/g | Dzoyem and Eloff [85] |
| Leucaena leucocephala | Leaves | Acetone | TPC = 129.78 mg/g GAE TFC = 35.16 mg QE/g | Dzoyem and Eloff [85] |
| Leucosidea sericea | Leaves, stem | 50% Methanol | TPC = 36.66 and 6.4 mg GAE/g GC = 29.32 and 5.12 µg GAE/g CT = 0.46 and 0.47% LCE/g TFC = 0.66 and 0.26 mg CTE/g | Aremu et al., [69] |
| Lippia javanica (Burm.f) Spreng | Leaves | Acetone | TPC = 130.12 mg GAE/g | Dzoyem and Eloff [85] |
| Morus mesozygia | Leaves | Acetone | TPC = 427.53 mg/g GAE TFC = 80.72 mg QE/g | Dzoyem and Eloff [85] |
| Philenoptera nelsii (Lonchocarpus nelsii) | Leaves | Acetone | TPC = 258.4 mg/g GAE TFC = 159.61 mg QE/g | Dzoyem et al., [67] |
| # Pittosporum viridiflorum Sims | Stem bark | Acetone | TPC = 181.49 mg/g GAE TFC = 13.75 mg QE/g | Olaokun et al., [90] |
| # Senna petersiana | Leaves | 50% Methanol | TPC = 5 mg GAE/g GC = 4 µg GAE/g CT = 0.18% LCE/g TFC = 0.1 mg CTE/g | Aremu et al., [71] |
| Uapaca nitida | Leaves | Acetone | TPC = 26.08 mg/g GAE TFC = 20.31 mg/g QE | Dzoyem and Eloff [85] |
| Virgilia divaricata Adamson | Leaves | Acetone | TPC = 137.3 mg/g GAE TFC = 15.3 mg QE/g | Dzoyem et al., [67] |
| Ziziphus rivularis Codd | Leaves | Acetone | TPC = 182.79 mg/g GAE TFC = 46.88 mg QE/g | Dzoyem and Eloff [85] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Selogatwe, K.M.; Asong, J.A.; Struwig, M.; Ndou, R.V.; Aremu, A.O. A Review of Ethnoveterinary Knowledge, Biological Activities and Secondary Metabolites of Medicinal Woody Plants Used for Managing Animal Health in South Africa. Vet. Sci. 2021, 8, 228. https://doi.org/10.3390/vetsci8100228
Selogatwe KM, Asong JA, Struwig M, Ndou RV, Aremu AO. A Review of Ethnoveterinary Knowledge, Biological Activities and Secondary Metabolites of Medicinal Woody Plants Used for Managing Animal Health in South Africa. Veterinary Sciences. 2021; 8(10):228. https://doi.org/10.3390/vetsci8100228
Chicago/Turabian StyleSelogatwe, Kelebogile Martha, John Awungnjia Asong, Madeleen Struwig, Rendani Victress Ndou, and Adeyemi Oladapo Aremu. 2021. "A Review of Ethnoveterinary Knowledge, Biological Activities and Secondary Metabolites of Medicinal Woody Plants Used for Managing Animal Health in South Africa" Veterinary Sciences 8, no. 10: 228. https://doi.org/10.3390/vetsci8100228
APA StyleSelogatwe, K. M., Asong, J. A., Struwig, M., Ndou, R. V., & Aremu, A. O. (2021). A Review of Ethnoveterinary Knowledge, Biological Activities and Secondary Metabolites of Medicinal Woody Plants Used for Managing Animal Health in South Africa. Veterinary Sciences, 8(10), 228. https://doi.org/10.3390/vetsci8100228







