A Double Histochemical/Immunohistochemical Staining for the Identification of Canine Mast Cells in Light Microscopy
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection
2.2. Immunohistochemistry and Toluidine Blue Counterstain
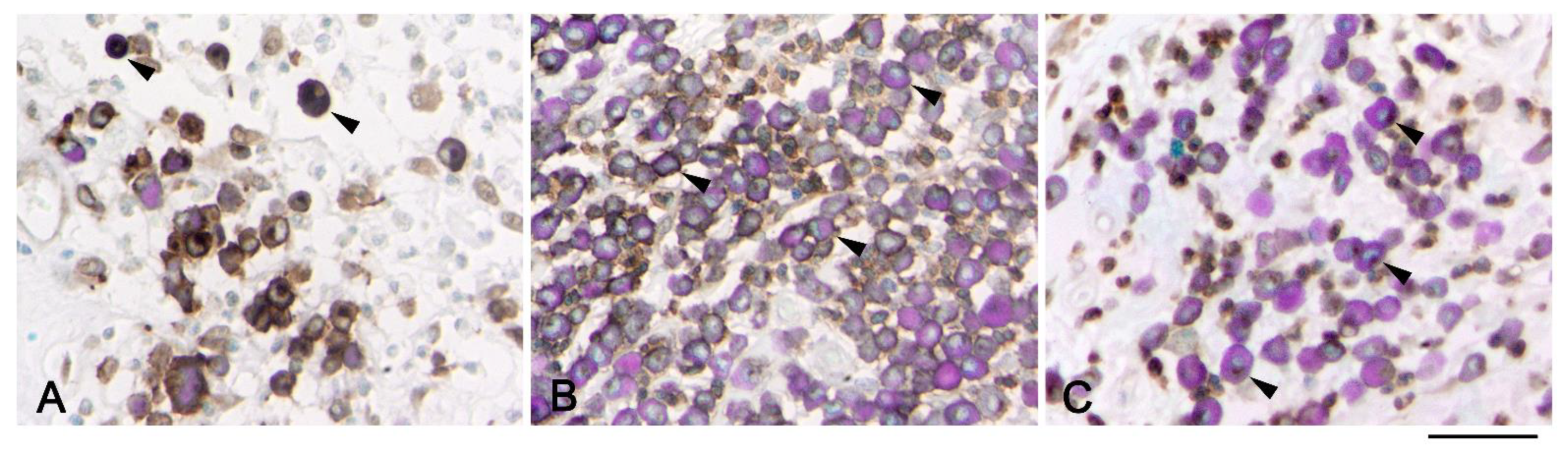
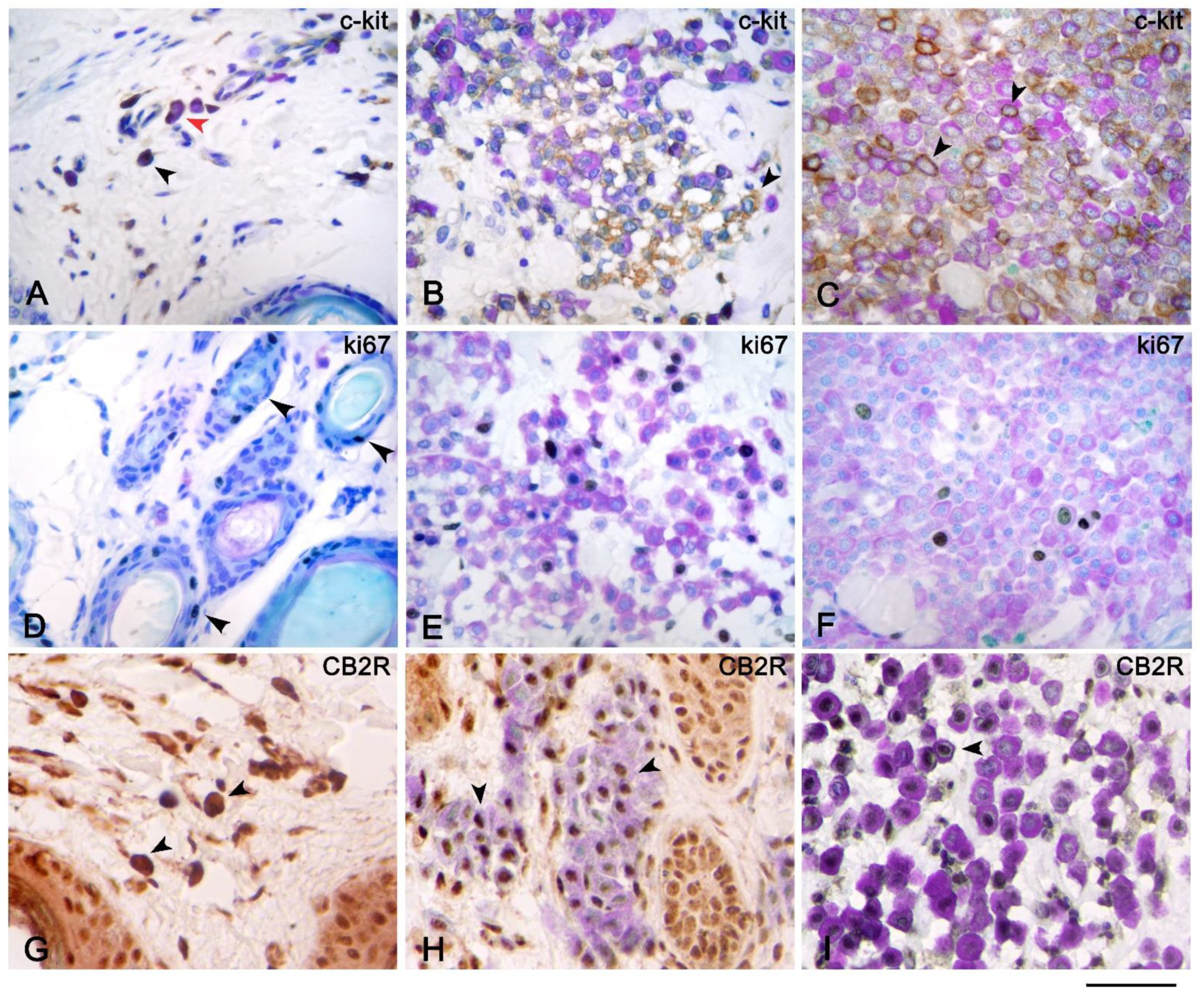
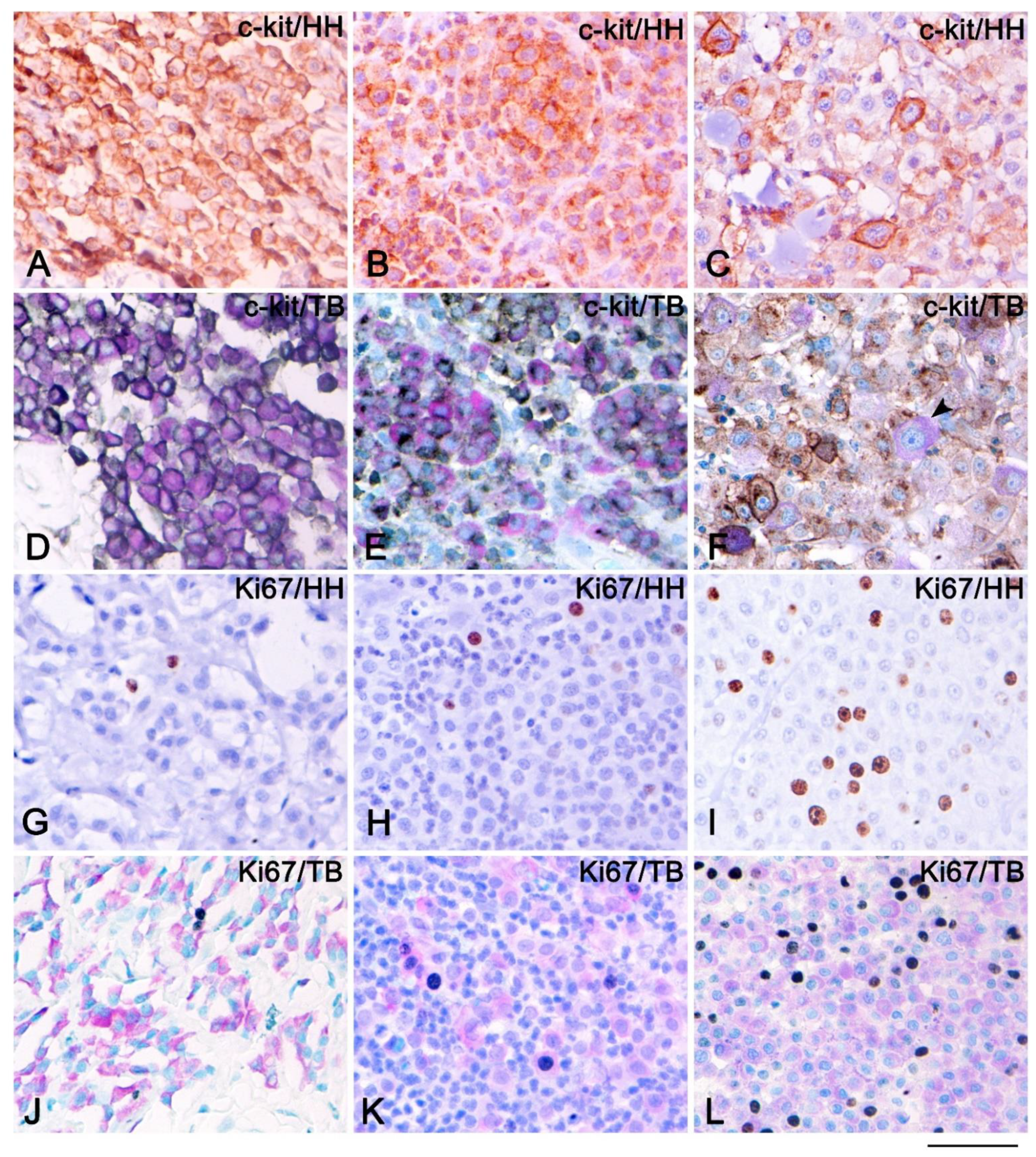
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Smith, N.R.; Womack, C. A Matrix Approach to Guide IHC-Based Tissue Biomarker Development in Oncology Drug Discovery. J. Pathol. 2014, 232, 190–198. [Google Scholar] [CrossRef]
- Misdorp, W. Mast Cells and Canine Mast Cell Tumours. A Review. Vet. Q. 2004, 26, 156–169. [Google Scholar] [CrossRef]
- London, C.A.; Seguin, B. Mast Cell Tumors in the Dog. Vet. Clin. N. Am. Small Anim. Pract. 2003, 33, 473–489. [Google Scholar] [CrossRef]
- Ribatti, D. The Staining of Mast Cells: A Historical Overview. Int. Arch. Allergy Immunol. 2018, 176, 55–60. [Google Scholar] [CrossRef]
- Pedro, J.; Simoes, C.; Schoning, P. Canine Mast Cell Tumors: A Comparison of Staining Techniques. J. Vet. Diagn. Investig. 1994, 465, 458–465. [Google Scholar]
- Schwartz, L.B. Tryptase, a Mediator of Human Mast Cells. J. Allergy Clin. Immunol. 1990, 86, 594–598. [Google Scholar] [CrossRef]
- Lee Gross, T.; Ihrke, P.J.; Walder, E.J.; Affolter, V.K. Mast Cell Tumors. In Skin Diseases of the Dog and Cat; Blackwell Publishing: Ames, IA, USA, 2005; pp. 853–865. ISBN 9780470752487. [Google Scholar]
- Atiakshin, D.; Buchwalow, I.; Tiemann, M. Mast Cell Chymase: Morphofunctional Characteristics. Histochem. Cell Biol. 2019, 152, 253–269. [Google Scholar] [CrossRef] [PubMed]
- Shiromany, A.; Sood, R.; Akifuddin, S.; Sidhu, G.K.; Khan, N.; Singla, K. Comparison of Mast Cells and Inflammatory Cells within Periapical Lesions and Comparison of Degranulated Mast Cells Between Fibrous and Inflamed Area in Radicular Cysts: An Immunohistochemical Study. J. Clin. Diagn. Res. 2014, 8, ZC61-4. [Google Scholar] [CrossRef] [PubMed]
- de Souza Junior, D.A.; Santana, A.C.; da Silva, E.Z.M.; Oliver, C.; Jamur, M.C. The Role of Mast Cell Specific Chymases and Tryptases in Tumor Angiogenesis. Biomed Res. Int. 2015, 2015, 142359. [Google Scholar] [CrossRef]
- Sperr, W.R.; Stehberger, B.; Wimazal, F.; Baghestanian, M.; Schwartz, L.B.; Kundi, M.; Semper, H.; Jordan, J.H.; Chott, A.; Drach, J.; et al. Serum Tryptase Measurements in Patients with Myelodysplastic Syndromes. Leuk. Lymphoma 2002, 43, 1097–1105. [Google Scholar] [CrossRef]
- Valent, P.; Sperr, W.R.; Sotlar, K.; Reiter, A.; Akin, C.; Gotlib, J.; Horny, H.-P.; Arock, M. The Serum Tryptase Test: An Emerging Robust Biomarker in Clinical Hematology. Expert Rev. Hematol. 2014, 7, 683–690. [Google Scholar] [CrossRef]
- Nuttall, T.J.; Marsella, R.; Rosenbaum, M.R.; Gonzales, A.J.; Fadok, V.A. Update on Pathogenesis, Diagnosis, and Treatment of Atopic Dermatitis in Dogs. J. Am. Vet. Med. Assoc. 2019, 254, 1291–1300. [Google Scholar] [CrossRef]
- Favrot, C.; Steffan, J.; Seewald, W.; Picco, F. A Prospective Study on the Clinical Features of Chronic Canine Atopic Dermatitis and Its Diagnosis. Vet. Dermatol. 2010, 21, 23–31. [Google Scholar] [CrossRef]
- Davis, B.J.; Page, R.; Sannes, P.L.; Meuten, D.J. Cutaneous Mastocytosis in a Dog. Vet. Pathol. 1992, 29, 363–365. [Google Scholar] [CrossRef]
- Pariser, M.S.; Gram, D.W. Urticaria Pigmentosa-like Disease in a Dog. Can. Vet. J. 2015, 56, 245–248. [Google Scholar]
- Kato, Y.; Kashiwagi, E.; Masuno, K.; Fujisawa, K.; Tsuchiya, N.; Matsushima, S.; Torii, M.; Takasu, N. Cutaneous Mastocytosis with a Mutation in the Juxtamembrane Domain of C-Kit in a Young Laboratory Beagle Dog. J. Toxicol. Pathol. 2016, 29, 49–52. [Google Scholar] [CrossRef][Green Version]
- Horta, R.S.; Lavalle, G.E.; Monteiro, L.N.; Souza, M.C.C.; Cassali, G.D.; Araújo, R.B. Assessment of Canine Mast Cell Tumor Mortality Risk Based on Clinical, Histologic, Immunohistochemical, and Molecular Features. Vet. Pathol. 2018, 55, 212–223. [Google Scholar] [CrossRef]
- Campora, L.; Miragliotta, V.; Ricci, E.; Cristino, L.; Di Marzo, V.; Albanese, F.; Federica Della Valle, M.; Abramo, F. Cannabinoid Receptor Type 1 and 2 Expression in the Skin of Healthy Dogs and Dogs with Atopic Dermatitis. Am. J. Vet. Res. 2012, 73, 988–995. [Google Scholar] [CrossRef] [PubMed]
- Patnaik, A.K.; Ehler, W.J.; Mac Ewen, E.G. Canine Cutaneous Mast Cell Tumor: Morphologic Grading and Survival Time in 83 Dogs. Vet. Pathol. 1984, 21, 469–474. [Google Scholar] [CrossRef] [PubMed]
- Kiupel, M.; Webster, J.D.; Bailey, K.L.; Best, S.; DeLay, J.; Detrisac, C.J.; Fitzgerald, S.D.; Gamble, D.; Ginn, P.E.; Goldschmidt, M.H.; et al. Proposal of a 2-Tier Histologic Grading System for Canine Cutaneous Mast Cell Tumors to More Accurately Predict Biological Behavior. Vet. Pathol. 2011, 48, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Kiupel, M.; Webster, J.D.; Kaneene, J.B.; Miller, R.; Yuzbasiyan-Gurkan, V. The Use of KIT and Tryptase Expression Patterns as Prognostic Tools for Canine Cutaneous Mast Cell Tumors. Vet. Pathol. 2004, 41, 371–377. [Google Scholar] [CrossRef]
- Gregory-Bryson, E.; Bartlett, E.; Kiupel, M.; Hayes, S.; Yuzbasiyan-Gurkan, V. Canine and Human Gastrointestinal Stromal Tumors Display Similar Mutations in C-KIT Exon 11. BMC Cancer 2010, 10, 559. [Google Scholar] [CrossRef] [PubMed]
- Letard, S.; Yang, Y.; Hanssens, K.; Palmérini, F.; Leventhal, P.S.; Guéry, S.; Moussy, A.; Kinet, J.-P.; Hermine, O.; Dubreuil, P. Gain-of-Function Mutations in the Extracellular Domain of KIT Are Common in Canine Mast Cell Tumors. Mol. Cancer Res. 2008, 6, 1137–1145. [Google Scholar] [CrossRef] [PubMed]
- Maglennon, G.A.; Murphy, S.; Adams, V.; Miller, J.; Smith, K.; Blunden, A.; Scase, T.J. Association of Ki67 Index with Prognosis for Intermediate-Grade Canine Cutaneous Mast Cell Tumours*. Vet. Comp. Oncol. 2008, 6, 268–274. [Google Scholar] [CrossRef] [PubMed]
- Miettinen, M.; Lasota, J. KIT (CD117): A Review on Expression in Normal and Neoplastic Tissues, and Mutations and Their Clinicopathologic Correlation. Appl. Immunohistochem. Mol. Morphol. AIMM 2005, 13, 205–220. [Google Scholar] [CrossRef] [PubMed]
- London, C.A.; Kisseberth, W.C.; Galli, S.J.; Geissler, E.N.; Helfand, S.C. Expression of Stem Cell Factor Receptor (c-Kit) by the Malignant Mast Cells from Spontaneous Canine Mast Cell Tumours. J. Comp. Pathol. 1996, 115, 399–414. [Google Scholar] [CrossRef]
- Reguera, M.J.; Rabanal, R.M.; Puigdemont, A.; Ferrer, L. Canine Mast Cell Tumors Express Stem Cell Factor Receptor. Am. J. Dermatopathol. 2000, 22, 49–54. [Google Scholar] [CrossRef]
- Gil da Costa, R.M.; Matos, E.; Rema, A.; Lopes, C.; Pires, M.A.; Gärtner, F. CD117 Immunoexpression in Canine Mast Cell Tumours: Correlations with Pathological Variables and Proliferation Markers. BMC Vet. Res. 2007, 3, 19. [Google Scholar] [CrossRef]
- Thamm, D.H.; Avery, A.C.; Berlato, D.; Bulman-Fleming, J.; Clifford, C.A.; Hershey, A.E.; Intile, J.L.; Jones, P.D.; Kamstock, D.A.; Liptak, J.M.; et al. Prognostic and Predictive Significance of KIT Protein Expression and C-Kit Gene Mutation in Canine Cutaneous Mast Cell Tumours: A Consensus of the Oncology-Pathology Working Group. Vet. Comp. Oncol. 2019, 17, 451–455. [Google Scholar] [CrossRef]
- Morini, M.; Bettini, G.; Preziosi, R.; Mandrioli, L. C-Kit Gene Product (CD117) Immunoreactivity in Canine and Feline Paraffin Sections. J. Histochem. Cytochem. Off. J. Histochem. Soc. 2004, 52, 705–708. [Google Scholar] [CrossRef]
- Hendrick, M. Mesenchymal Tumors of the Skin and Soft Tissues. In Tumors of Domestic Animals; Meuten, D.J., Ed.; Wiley Blackwell Publishing Blackwell: Ames, IA, USA, 2016; pp. 142–175. [Google Scholar]
- Kiupel, M.; Camus, M. Diagnosis and Prognosis of Canine Cutaneous Mast Cell Tumors. Vet. Clin. N. Am. Small Anim. Pract. 2019, 49, 819–836. [Google Scholar] [CrossRef] [PubMed]
- Scholzen, T.; Gerdes, J. The Ki-67 Protein: From the Known and the Unknown. J. Cell. Physiol. 2000, 182, 311–322. [Google Scholar] [CrossRef]
- Rossi, F.; Bernardo, M.E.; Bellini, G.; Luongo, L.; Conforti, A.; Manzo, I.; Guida, F.; Cristino, L.; Imperatore, R.; Petrosino, S.; et al. The Cannabinoid Receptor Type 2 as Mediator of Mesenchymal Stromal Cell Immunosuppressive Properties. PLoS ONE 2013, 8, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Moreno, E.; Cavic, M.; Krivokuca, A.; Casadó, V.; Canela, E. The Endocannabinoid System as a Target in Cancer Diseases: Are We There Yet? Front. Pharmacol. 2019, 10, 339. [Google Scholar] [CrossRef] [PubMed]
- Galiazzo, G.; Giancola, F.; Stanzani, A.; Fracassi, F.; Bernardini, C.; Forni, M.; Pietra, M.; Chiocchetti, R. Localization of Cannabinoid Receptors CB1, CB2, GPR55, and PPARα in the Canine Gastrointestinal Tract. Histochem. Cell Biol. 2018, 150, 187–205. [Google Scholar] [CrossRef] [PubMed]
- Tan, W.C.C.; Nerurkar, S.N.; Cai, H.Y.; Ng, H.H.M.; Wu, D.; Wee, Y.T.F.; Lim, J.C.T.; Yeong, J.; Lim, T.K.H. Overview of Multiplex Immunohistochemistry/Immunofluorescence Techniques in the Era of Cancer Immunotherapy. Cancer Commun. (Lond. Engl.) 2020, 40, 135–153. [Google Scholar] [CrossRef]
- Im, K.; Mareninov, S.; Diaz, M.F.P.; Yong, W.H. An Introduction to Performing Immunofluorescence Staining. Methods Mol. Biol. 2019, 1897, 299–311. [Google Scholar] [CrossRef] [PubMed]
- Mehnert, A.-K.; Simon, C.S.; Guizetti, J. Immunofluorescence Staining Protocol for STED Nanoscopy of Plasmodium-Infected Red Blood Cells. Mol. Biochem. Parasitol. 2019, 229, 47–52. [Google Scholar] [CrossRef]
- Fonseca-Alves, C.E.; Kobayashi, P.E.; Palmieri, C.; Laufer-Amorim, R. Investigation of C-KIT and Ki67 Expression in Normal, Preneoplastic and Neoplastic Canine Prostate. BMC Vet. Res. 2017, 13, 380. [Google Scholar] [CrossRef]
- Dobson, J.M.; Samuel, S.; Milstein, H.; Rogers, K.; Wood, J.L.N. Canine Neoplasia in the UK: Estimates of Incidence Rates from a Population of Insured Dogs. J. Small Anim. Pract. 2002, 43, 240–246. [Google Scholar] [CrossRef]
- Pazdzior-Czapula, K.; Mikiewicz, M.; Gesek, M.; Zwolinski, C.; Otrocka-Domagala, I. Diagnostic Immunohistochemistry for Canine Cutaneous Round Cell Tumours—Retrospective Analysis of 60 Cases. Folia Histochem. Cytobiol. 2019, 57, 146–154. [Google Scholar] [CrossRef]
- Ramos-Vara, J.A.; Miller, M.A.; Valli, V.E.O. Immunohistochemical Detection of Multiple Myeloma 1/Interferon Regulatory Factor 4 (MUM1/IRF-4) in Canine Plasmacytoma: Comparison with CD79a and CD20. Vet. Pathol. 2007, 44, 875–884. [Google Scholar] [CrossRef]
- Milner, R.J.; Pearson, J.; Nesbit, J.W.; Close, P. Immunophenotypic Classification of Canine Malignant Lymphoma on Formalin-Mixed Paraffin Wax-Embedded Tissue by Means of CD3 and CD79a Cell Markers. Onderstepoort J. Vet. Res. 1996, 63, 309–313. [Google Scholar] [PubMed]
- Cruz, V.S.; Borges, J.C.A.; Nepomuceno, L.L.; Gonçalves, P.A.M.; Prado, Y.C.L.; Bianchi, C.; Fioravanti, M.C.S.; Araújo, E.G. Histological Classification and Expression of Markers of Canine Mast Cell Tumors. Vet. World 2020, 13, 1627–1634. [Google Scholar] [CrossRef] [PubMed]
- London, C.A.; Thamm, D.H. 20—Mast Cell Tumors; In Withrow, S.J., Vail, D.M., Page, R.L.B.T., Eds.; W.B. Saunders: Saint Louis, MO, USA, 2013; pp. 335–355. ISBN 978-1-4377-2362-5. [Google Scholar]
- Wu, H.; Hayashi, T.; Inoue, M. Immunohistochemical Expression of P27 and P21 in Canine Cutaneous Mast Cell Tumors and Histiocytomas. Vet. Pathol. 2004, 41, 296–299. [Google Scholar] [CrossRef] [PubMed]
- Ouban, A.; Dellis, J.; Salup, R.; Morgan, M. Immunohistochemical Expression of Mdm2 and P53 in Penile Verrucous Carcinoma. Ann. Clin. Lab. Sci. 2003, 33, 101–106. [Google Scholar] [PubMed]
- Jaffe, M.H.; Hosgood, G.; Taylor, H.W.; Kerwin, S.C.; Hedlund, C.S.; Lopez, M.K.; Davidson, J.R.; Miller, D.M.; Paranjpe, M. Immunohistochemical and Clinical Evaluation of P53 in Canine Cutaneous Mast Cell Tumors. Vet. Pathol. 2000, 37, 40–46. [Google Scholar] [CrossRef]
| Marker | Type, Clone, Reference | Supplier | Dilution/Incubation | Ag Retrieval |
|---|---|---|---|---|
| c-kit (CD117) | Rabbit polyclonal anti-c-kit (A4502) [41] | Dako, Glostrup, Denmark | 1:300 ON | 10′ Citrate pH6 MW: 750 W |
| Ki67 | Mouse monoclonal anti-Ki67 (MIB1) [41] | Dako, Glostrup, Denmark | 1:600 ON | 20′ Citrate pH6 MW: 750 W |
| CB2R | Rabbit polyclonal anti-Cannabinoid Receptor II (ab45942) [37] | Abcam, Cambridge, UK | 1:500 ON | 10′ EDTA pH8 MW: 750 W |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gobbo, F.; Sarli, G.; De Silva, M.; Galiazzo, G.; Chiocchetti, R.; Morini, M. A Double Histochemical/Immunohistochemical Staining for the Identification of Canine Mast Cells in Light Microscopy. Vet. Sci. 2021, 8, 229. https://doi.org/10.3390/vetsci8100229
Gobbo F, Sarli G, De Silva M, Galiazzo G, Chiocchetti R, Morini M. A Double Histochemical/Immunohistochemical Staining for the Identification of Canine Mast Cells in Light Microscopy. Veterinary Sciences. 2021; 8(10):229. https://doi.org/10.3390/vetsci8100229
Chicago/Turabian StyleGobbo, Francesca, Giuseppe Sarli, Margherita De Silva, Giorgia Galiazzo, Roberto Chiocchetti, and Maria Morini. 2021. "A Double Histochemical/Immunohistochemical Staining for the Identification of Canine Mast Cells in Light Microscopy" Veterinary Sciences 8, no. 10: 229. https://doi.org/10.3390/vetsci8100229
APA StyleGobbo, F., Sarli, G., De Silva, M., Galiazzo, G., Chiocchetti, R., & Morini, M. (2021). A Double Histochemical/Immunohistochemical Staining for the Identification of Canine Mast Cells in Light Microscopy. Veterinary Sciences, 8(10), 229. https://doi.org/10.3390/vetsci8100229






