Assessment of Longitudinal Left Ventricle Deformation by 2-Dimensional Speckle Tracking Echocardiography Obtained from Different Views in Cats
Abstract
1. Introduction
2. Materials and Methods
2.1. Standard Echocardiography
2.2. Speckle Tracking Echocardiography
2.3. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Mor-Avi, V.; Lang, R.M.; Badano, L.P.; Belohlavek, M.; Cardim, N.M.; Derumeaux, G.; Galderisi, M.; Marwick, T.; Nagueh, S.F.; Sengupta, P.P.; et al. Current and evolving echocardiographic techniques for the quantitative evaluation of cardiac mechanics: ASE/EAE consensus statement on methodology and indications endorsed by the Japanese Society of Echocardiography. Eur. J. Echocardiogr. 2011, 12, 167–205. [Google Scholar] [CrossRef] [PubMed]
- Collier, P.; Phelan, D.; Klein, A. A test in context: Myocardial strain measured by speckle-tracking echocardiography. J. Am. Coll. Cardiol. 2017, 69, 1043–1056. [Google Scholar] [CrossRef] [PubMed]
- Fruganti, A.; Cerquetella, M.; Copponi, I.; Spaterna, A.; Tesei, B. Preliminary Evaluation of M-Mode, B-Mode, and X-Strain® Echocardiographic Indices Before and During Therapy in Dogs Affected by Dilated Cardiomyopathy. In Current Aspects in Biology, Animal Pathology, Clinic and Food Hygiene, 1st ed.; Pugliese, A., Gaiti, A., Boiti, C., Eds.; Springer: Berlin/Heidelberg, Germany, 2012; Volume 1, pp. 97–102. [Google Scholar]
- Zois, N.E.; Tidholm, A.; Nägga, K.M.; Moesgaard, S.G.; Rasmussen, C.E.; Falk, T.; Häggström, J.; Pedersen, H.D.; Åblad, B.; Nilsen, H.Y.; et al. Radial and longitudinal strain and strain rate assessed by speckle-tracking echocardiography in dogs with myxomatous mitral valve disease. J. Vet. Intern. Med. 2012, 26, 1309–1319. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, R.; Matsumoto, H.; Teshima, T.; Koyama, H. Clinical assessment of systolic myocardial deformations in dogs with chronic mitral valve insufficiency using two-dimensional speckle-tracking echocardiography. J. Vet. Cardiol. 2013, 15, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Pedro, B.; Stephenson, H.; Linney, C.; Cripps, P.; Dukes-McEwan, J. Assessment of left ventricular function in healthy Great Danes and in Great Danes with dilated cardiomyopathy using speckle tracking echocardiography. J. Vet. Cardiol. 2017, 19, 363–375. [Google Scholar] [CrossRef] [PubMed]
- Spalla, I.; Locatelli, C.; Zanaboni, A.M.; Brambilla, P.; Bussadori, C. Echocardiographic assessment of cardiac function by conventional and speckle-tracking echocardiography in dogs with patent Ductus Arteriosus. J. Vet. Intern. Med. 2016, 30, 706–713. [Google Scholar] [CrossRef]
- Caivano, D.; Rishniw, M.; Birettoni, F.; Petrescu, V.F.; Porciello, F. Transverse Right Ventricle Strain and Strain Rate Assessed by 2-Dimensional Speckle Tracking Echocardiography in Dogs with Pulmonary Hypertension. Vet. Sci. 2020, 7, 19. [Google Scholar] [CrossRef]
- Caivano, D.; Rishniw, M.; Patata, V.; Giorgi, M.E.; Birettoni, F.; Porciello, F. Left atrial deformation and phasic function determined by 2-dimensional speckle tracking echocardiography in healthy dogs. J. Vet. Cardiol. 2016, 18, 146–155. [Google Scholar] [CrossRef] [PubMed]
- Caivano, D.; Rishniw, M.; Birettoni, F.; Patata, V.; Giorgi, M.E.; Porciello, F. Left atrial deformation and phasic function determined by two-dimensional speckle-tracking echocardiography in dogs with myxomatous mitral valve disease. J. Vet. Cardiol. 2018, 20, 102–114. [Google Scholar] [CrossRef] [PubMed]
- Corda, A.; Pinna Parpaglia, M.L.; Sotgiu, G.; Zobba, R.; Gomez Ochoa, P.; Prieto Ramos, J.; French, A. Use of 2-dimensional speckle-tracking echocardiography to assess left ventricular systolic function in dogs with systemic inflammatory response syndrome. J. Vet. Intern. Med. 2019, 33, 423–431. [Google Scholar] [CrossRef]
- Leitman, M.; Lysyansky, P.; Sidenko, S.; Shir, V.; Peleg, E.; Binenbaum, M.; Kaluski, E.; Krakover, R.; Vered, Z. Two-dimensional strain—A novel software for real-time quantitative echocardiographic assessment of myocardial function. J. Am. Soc. Echocardiogr. 2004, 17, 1021–1029. [Google Scholar] [CrossRef] [PubMed]
- Blessberger, H.; Binder, T. Two-dimensional speckle tracking echocardiography: Basic principles. Heart 2010, 96, 716–722. [Google Scholar] [CrossRef] [PubMed]
- Sugimoto, K.; Fuji, Y.; Sunahara, H.; Aoki, T. Assessment of left ventricular longitudinal function in cats with subclinical hypertrophic cardiomyopathy using tissue Doppler imaging and speckle tracking echocardiography. J. Vet. Med. Sci. 2015, 77, 1101–1108. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, R.; Mochizuki, Y.; Yoshimatsu, H. Determination of multidirectional myocardial deformations in cats with hypertrophic cardiomyopathy by using two-dimensional speckle tracking echocardiography. J. Fel. Med. Surg. 2017, 19, 1283–1289. [Google Scholar] [CrossRef]
- Suzuki, R.; Mochizuki, Y.; Yoshimatsu, H. Layer-specific myocardial function in asymptomatic cats with obstructive hypertrophic cardiomyopathy assessed using 2-dimensional speckle-tracking echocardiography. J. Vet. Intern. Med. 2019, 33, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Wess, G.; Sarkar, R.; Hartmann, K. Assessment of left ventricular systolic function by strain imaging echocardiography in various stages of feline hypertrophic cardiomyopathy. J. Vet. Intern. Med. 2010, 24, 1375–1382. [Google Scholar] [CrossRef]
- Santarelli, G.; Talavera López, J.; Fernández Del Palacio, J. Evaluation of the right parasternal four-chamber view for the assessment of left ventricular longitudinal strain and strain rate by two-dimensional speckle tracking echocardiography in dogs. Res. Vet. Sci. 2018, 120, 78–85. [Google Scholar] [CrossRef]
- Thomas, W.P.; Gaber, C.E.; Jacobs, G.J.; Kaplan, P.M.; Lombard, C.W.; Moise, N.S.; Moses, B.L. Recommendations for standards in transthoracic two-dimensional echocardiography in the dog and cat. Echocardiography committee of the specialty of cardiology, American college of veterinary internal medicine. J. Vet. Intern. Med. 1993, 7, 247–252. [Google Scholar] [CrossRef]
- Dickson, D.; Caivano, D.; Patteson, M.; Rishniw, M. The times they are a-changin’: Two-dimensional aortic valve measurements differ throughout diastole. J. Vet. Cardiol. 2016, 18, 15–25. [Google Scholar] [CrossRef]
- Vezzosi, T.; Schober, K.E. Doppler-derived echocardiographic evidence of pulmonary hypertension in cats with left-sided congestive heart failure. J. Vet. Cardiol. 2019, 23, 58–68. [Google Scholar] [CrossRef]
- Santilli, R.A.; Bussadori, C. Doppler echocardiographic study of left ventricular diastole in non-anaesthetized healthy cats. Vet. J. 1998, 156, 203–215. [Google Scholar] [CrossRef]
- Altman, D.G.; Bland, J.M. Measurement in medicine: The analysis of method comparison studies. Statistician 1983, 32, 307–317. [Google Scholar] [CrossRef]
- Patata, V.; Caivano, D.; Porciello, F.; Rishniw, M.; Domenech, O.; Marchesotti, F.; Giorgi, M.E.; Guglielmini, C.; Poser, H.; Spina, F.; et al. Pulmonary vein to pulmonary artery ratio in healthy and cardiomyopathic cats. J. Vet. Cardiol. 2020, 27, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Abduch, M.C.; Assad, R.S.; Mathias, W., Jr.; Aiello, V.D. The echocardiography in the cardiovascular laboratory: A guide to research with animals. Arq. Bras. Cardiol. 2014, 102, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Lang, R.M.; Badano, L.P.; Mor-Avi, V.; Afilalo, J.; Armstrong, A.; Ernande, L.; Flachskampf, F.A.; Foster, E.; Goldstein, S.A.; Kuznetsova, T.; et al. Recommendations for cardiac chamber quantification by echocardiography in adults: An update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J. Am. Soc. Echocardiogr. 2015, 28, 1–39. [Google Scholar] [CrossRef]
- Smets, P.; Daminet, S.; Wess, G. Simpson’s Method of Discs for Measurement of Echocardiographic End-Diastolic and End-Systolic Left Ventricular Volumes: Breed-Specific Reference Ranges in Boxer Dogs. J. Vet. Intern. Med. 2014, 28, 116–122. [Google Scholar] [CrossRef]
- Wess, G.; Mäurer, J.; Simak, J.; Hartmann, K. Use of Simpson’s method of disc to detect early echocardiographic changes in Doberman Pinschers with dilated cardiomyopathy. J. Vet. Intern. Med. 2010, 24, 1069–1076. [Google Scholar] [CrossRef]
- Seckerdieck, M.; Holler, P.; Smets, P.; Wess, G. Simpson’s method of discs in Salukis and Whippets: Echocardiographic reference intervals for end-diastolic and end-systolic left ventricular volumes. J. Vet. Cardiol. 2015, 17, 271–281. [Google Scholar] [CrossRef]
- Kurosawa, T.A.; Sist, M.D.; Sanders, R.A. Echocardiographic variables in healthy North American Salukis. J. Vet. Cardiol. 2018, 20, 301–306. [Google Scholar] [CrossRef]
- Santarelli, G.; Baron Toaldo, M.; Bouvard, J.; Glaus, T.M.; Fernández Del Palacio, J. Variability among strain variables derived from two-dimensional speckle tracking echocardiography in dogs by use of various software. Am. J. Vet. Res. 2019, 80, 347–357. [Google Scholar] [CrossRef]
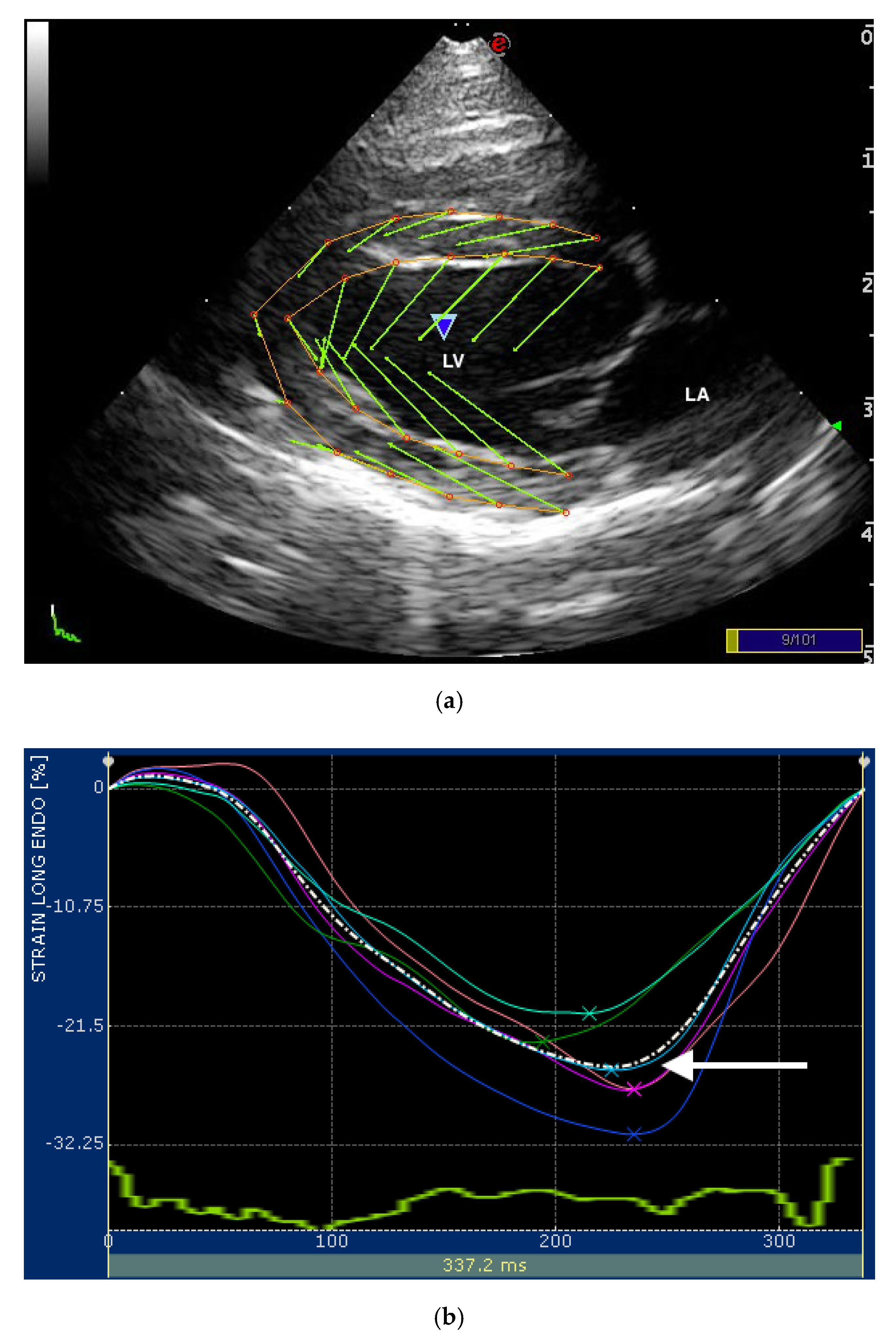
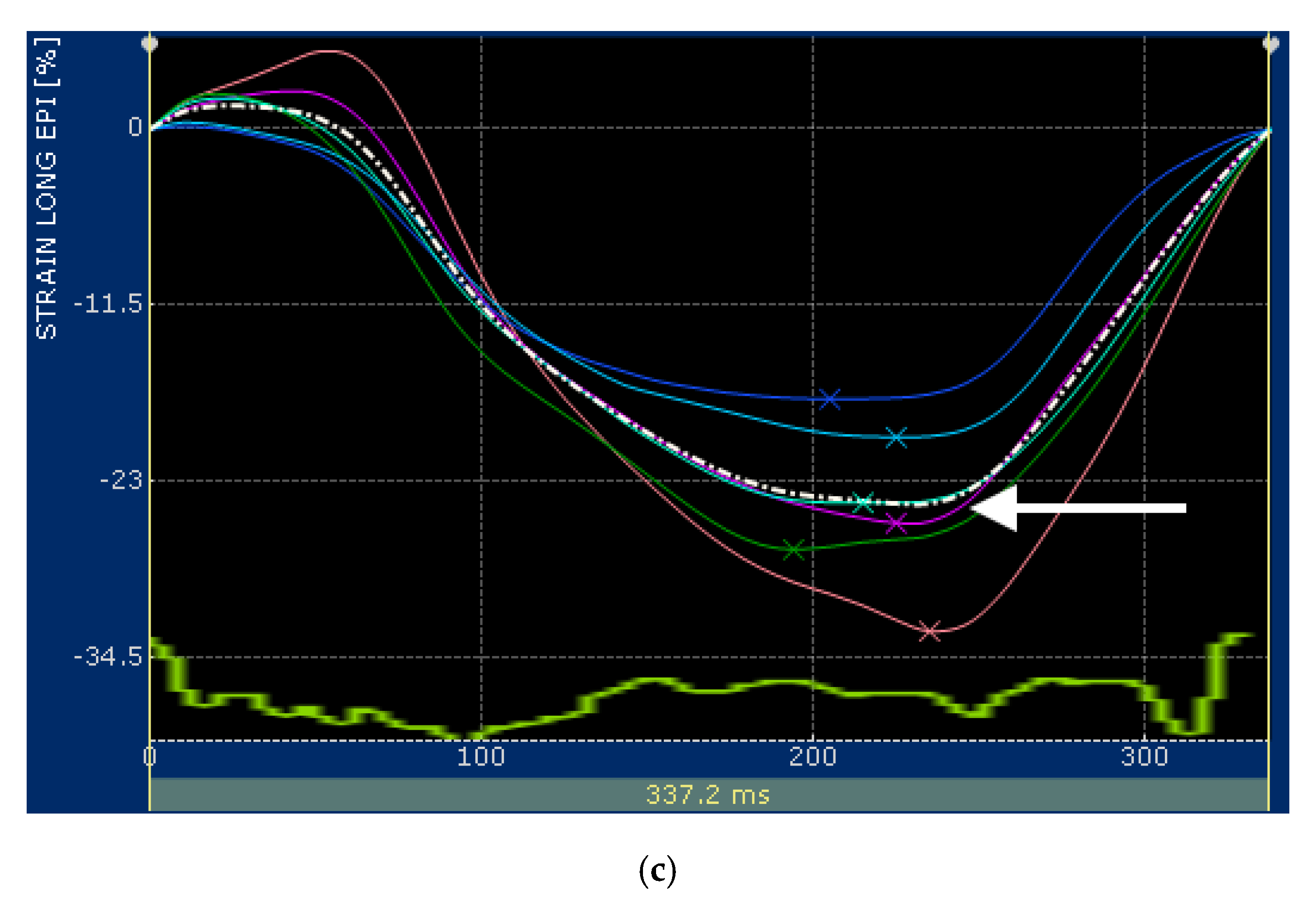
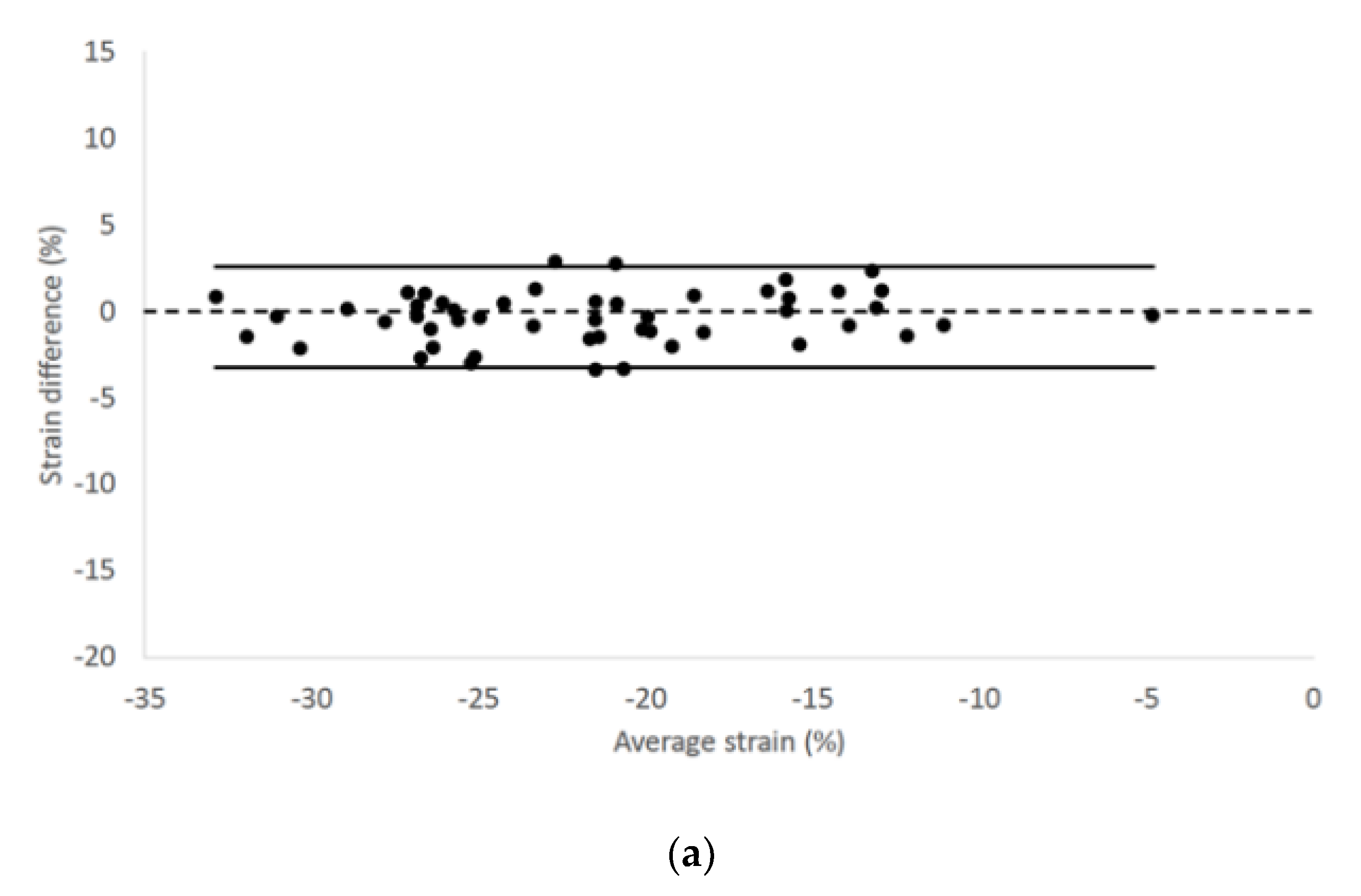
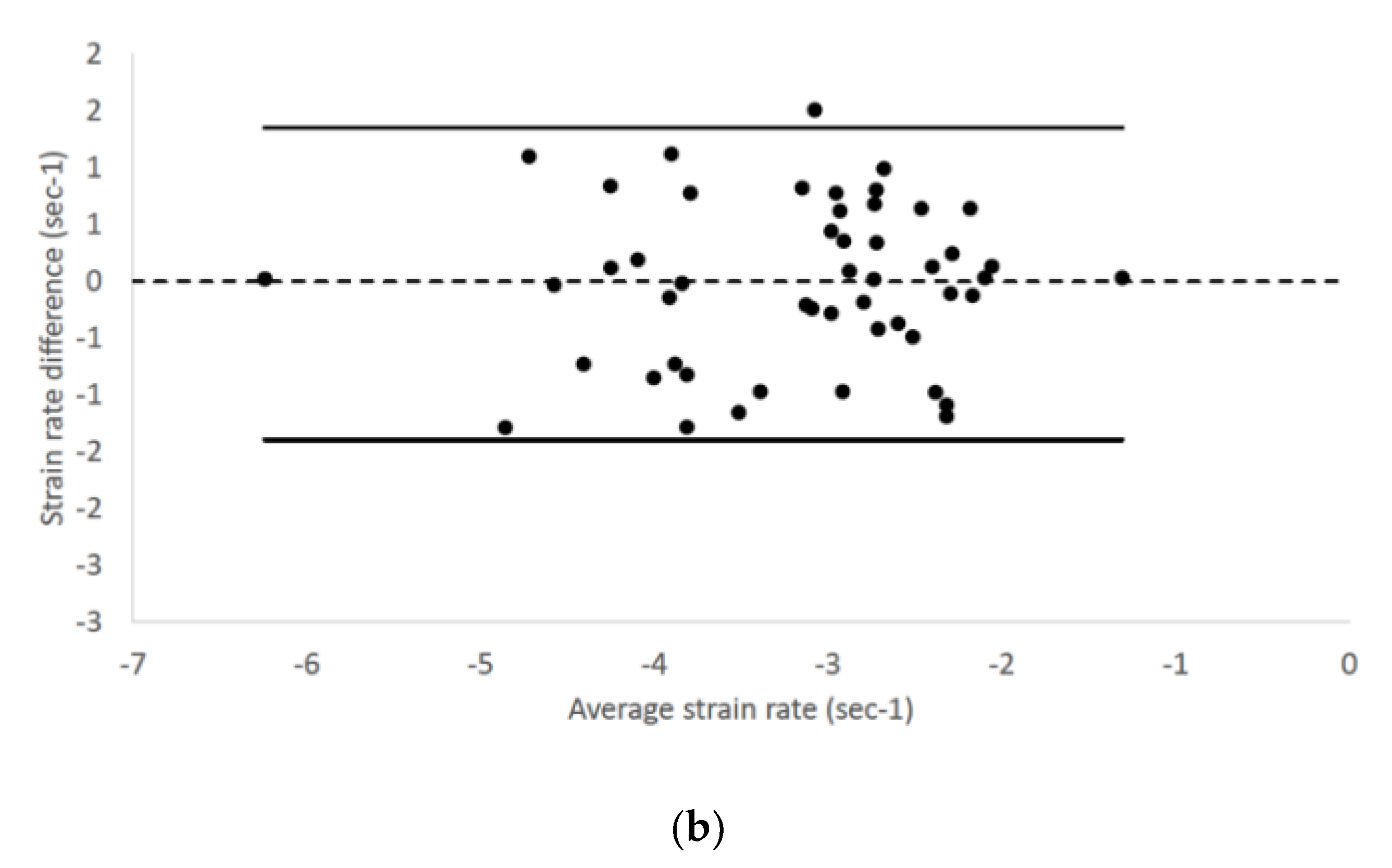
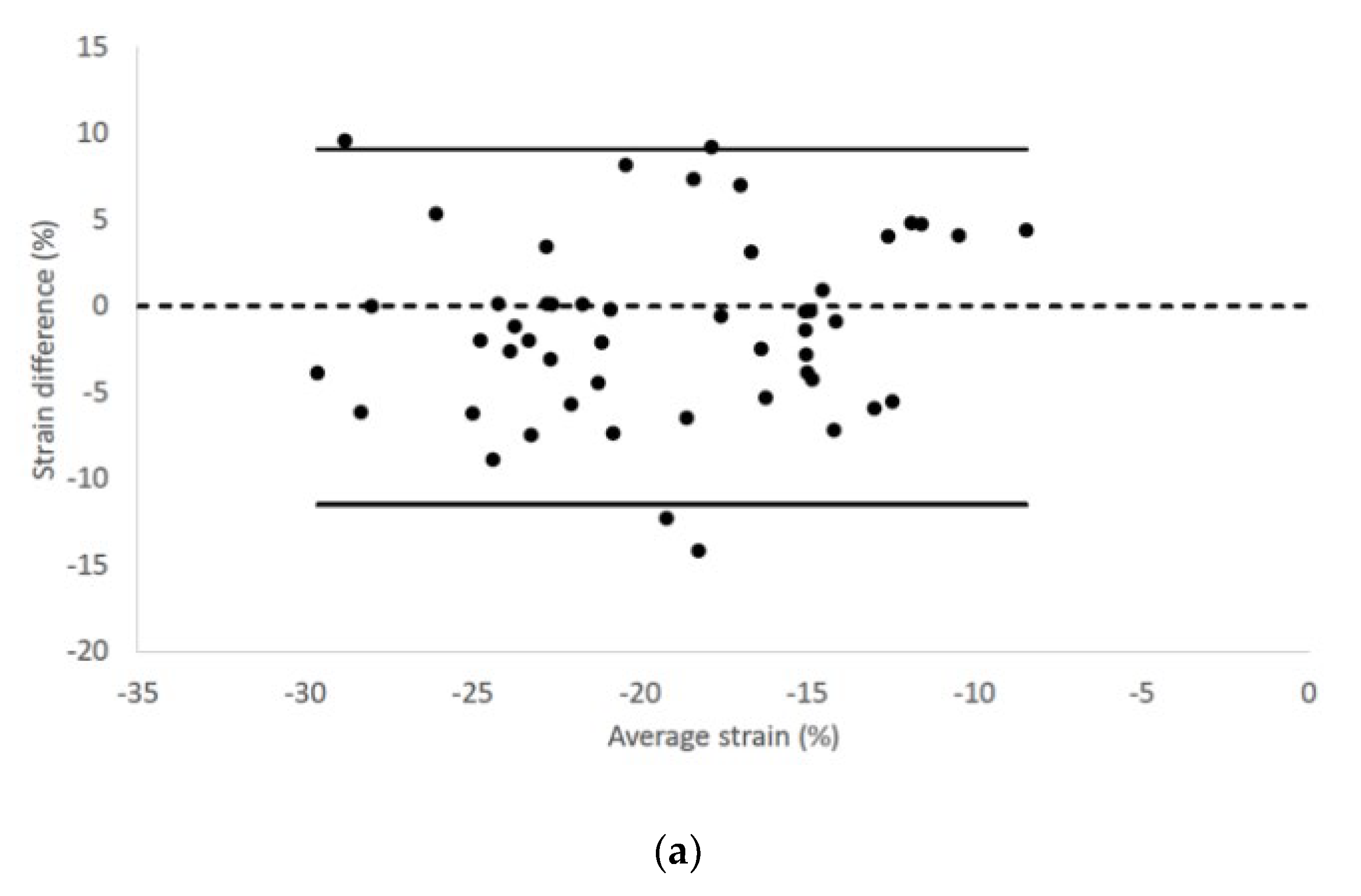
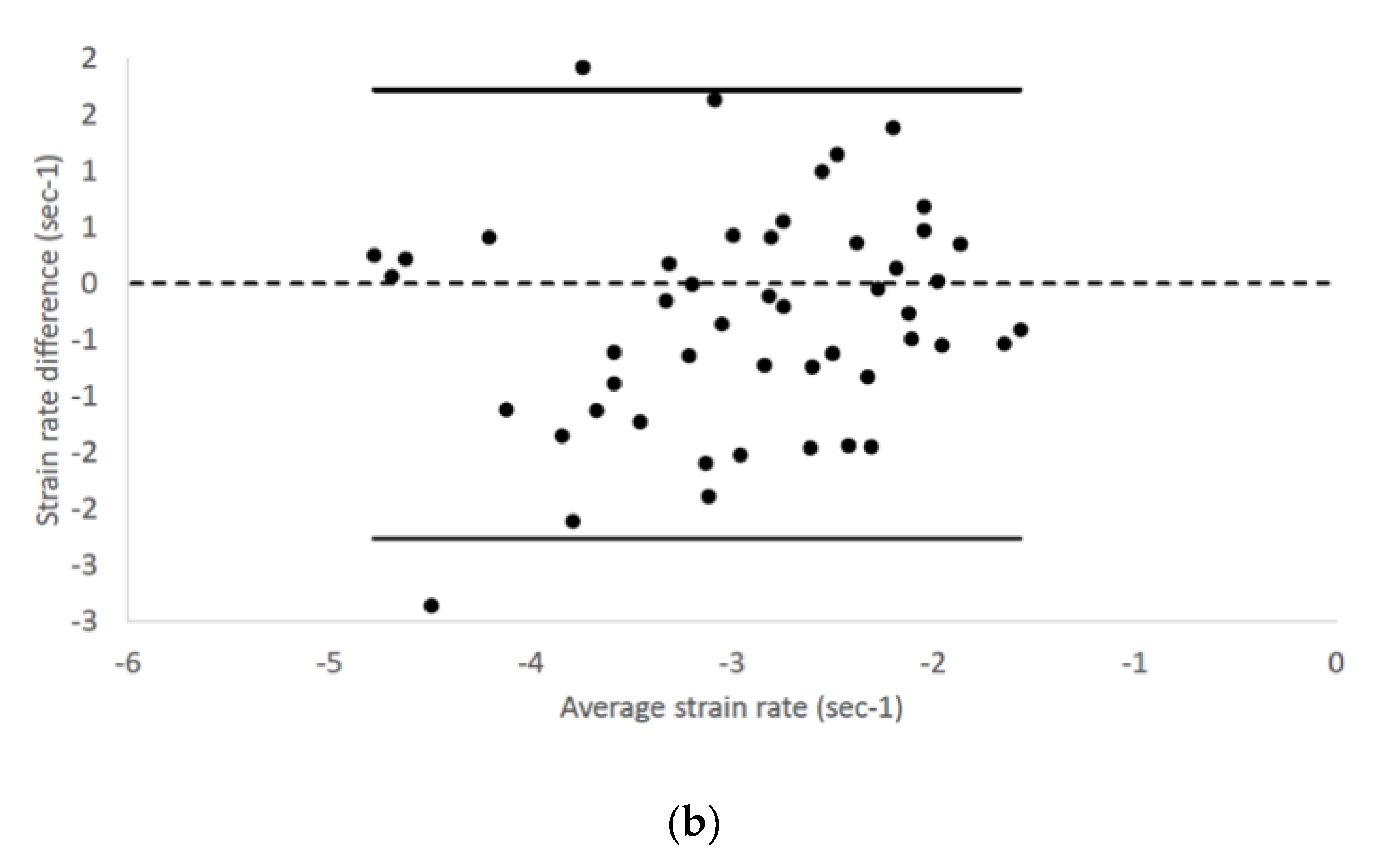
| STE Variables | RP4Ch View | LAP4Ch View |
|---|---|---|
| Endocardial LV longitudinal strain (%) | −21.71 ± 6.22 −22.23 (−32.68–−4.93) | −21.36 ± 5.98 −21.53 (−33.30–−4.70) |
| Epicardial LV longitudinal strain (%) | −19.83 ± 6.11 −21.02 (−31.56–−6.24) | −18.63 ± 5.55 −19.13 (−33.62–−9.68) |
| Endocardial LV longitudinal strain rate (Sec−1) | −3.19 ± 0.99 −2.88 (−6.23–−1.29) | −3.16 ± 0.96 −3.05 (−6.25–−1.32) |
| Epicardial LV longitudinal strain rate (Sec−1) | −3.08 ± 1.02 −2.92 (−5.92–−1.51) | −2.81 ± 0.90 −2.80 (−4.90–−1.36) |
| STE Variables | RP4Ch View | LAP4Ch View |
|---|---|---|
| Endocardial LV longitudinal strain | 5.4 | 7.3 |
| Epicardial LV longitudinal strain | 8.2 | 13 |
| Endocardial LV longitudinal strain rate | 18.7 | 15.3 |
| Epicardial LV longitudinal strain rate | 17.5 | 19.2 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caivano, D.; Rishniw, M.; Baiona, L.; Birettoni, F.; Nisini, N.; Porciello, F. Assessment of Longitudinal Left Ventricle Deformation by 2-Dimensional Speckle Tracking Echocardiography Obtained from Different Views in Cats. Vet. Sci. 2020, 7, 104. https://doi.org/10.3390/vetsci7030104
Caivano D, Rishniw M, Baiona L, Birettoni F, Nisini N, Porciello F. Assessment of Longitudinal Left Ventricle Deformation by 2-Dimensional Speckle Tracking Echocardiography Obtained from Different Views in Cats. Veterinary Sciences. 2020; 7(3):104. https://doi.org/10.3390/vetsci7030104
Chicago/Turabian StyleCaivano, Domenico, Mark Rishniw, Lucia Baiona, Francesco Birettoni, Noemi Nisini, and Francesco Porciello. 2020. "Assessment of Longitudinal Left Ventricle Deformation by 2-Dimensional Speckle Tracking Echocardiography Obtained from Different Views in Cats" Veterinary Sciences 7, no. 3: 104. https://doi.org/10.3390/vetsci7030104
APA StyleCaivano, D., Rishniw, M., Baiona, L., Birettoni, F., Nisini, N., & Porciello, F. (2020). Assessment of Longitudinal Left Ventricle Deformation by 2-Dimensional Speckle Tracking Echocardiography Obtained from Different Views in Cats. Veterinary Sciences, 7(3), 104. https://doi.org/10.3390/vetsci7030104





