Deregulation of VEGFR-2 and PDGFR Expression and Microvascular Density in a Triple-Negative Model of Canine Malignant Mammary Tumors with Lymph Node or Lung Metastasis
Abstract
1. Introduction
2. Material and Methods
2.1. Study Design
2.2. Patients
2.3. Clinical Evaluation
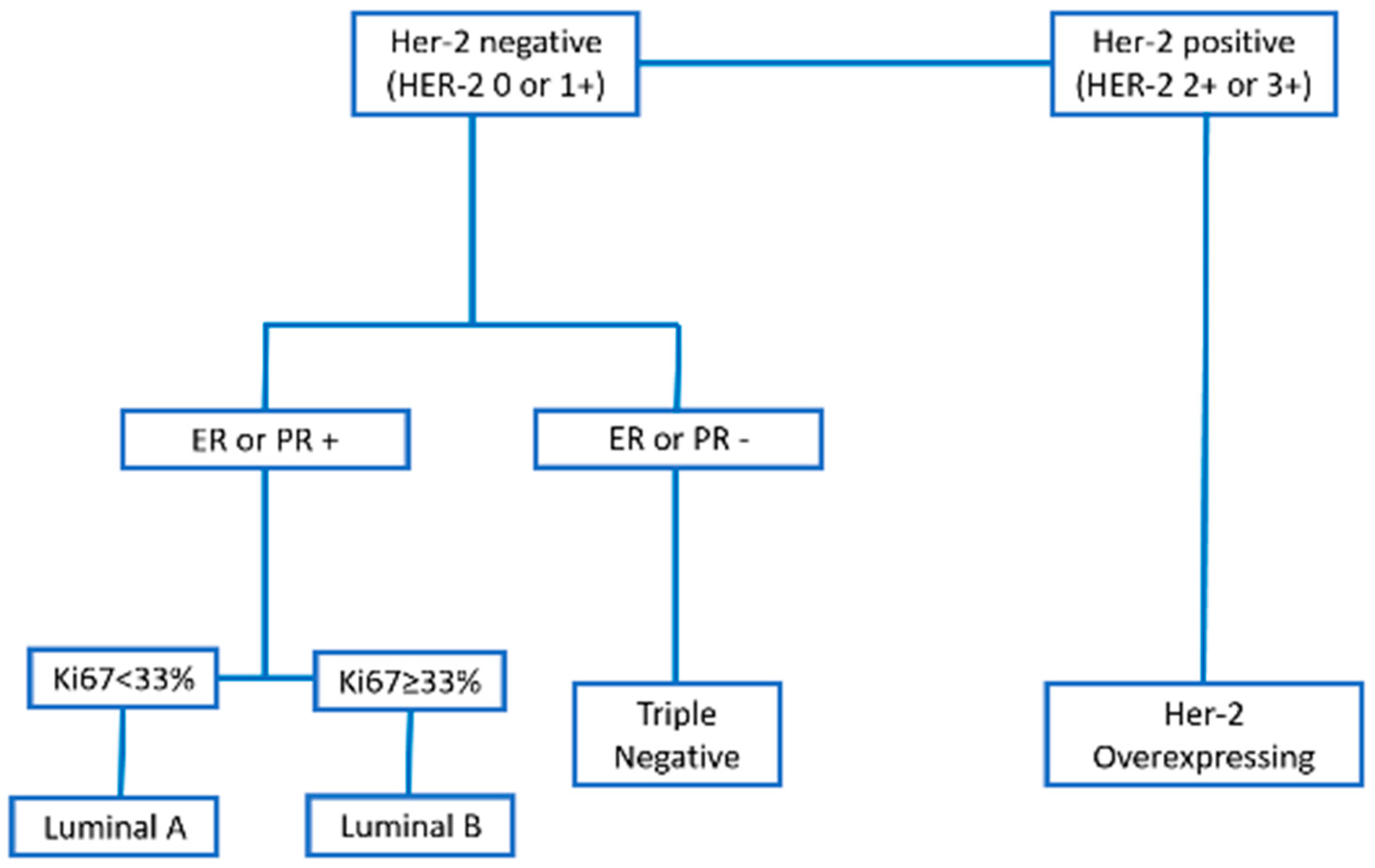
2.4. Molecular Phenotype
2.5. Tumor Groups
2.6. Immunohistochemistry
2.7. Statistical Evaluation
3. Results
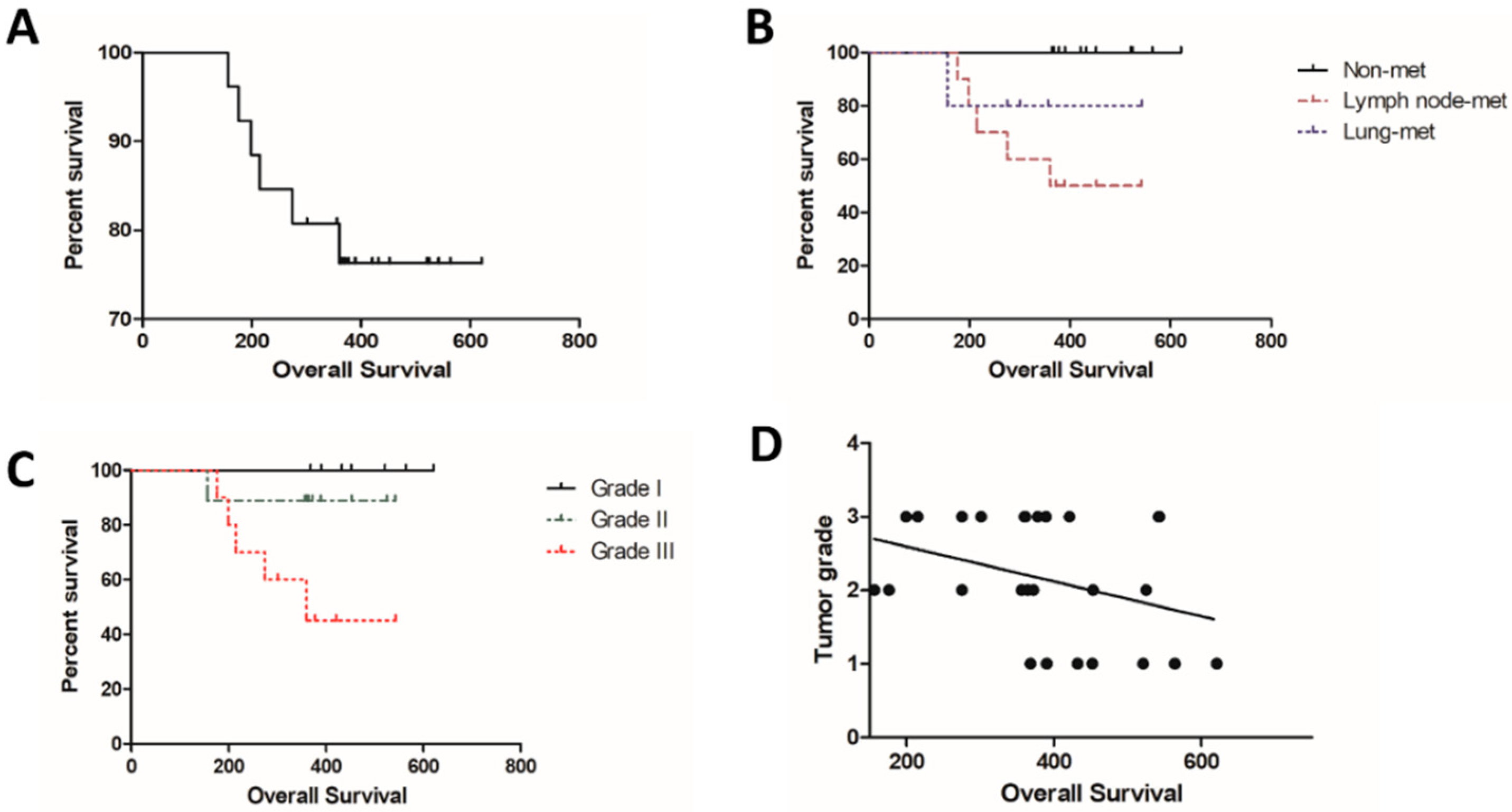
3.1. Clinical and Pathological Evaluation
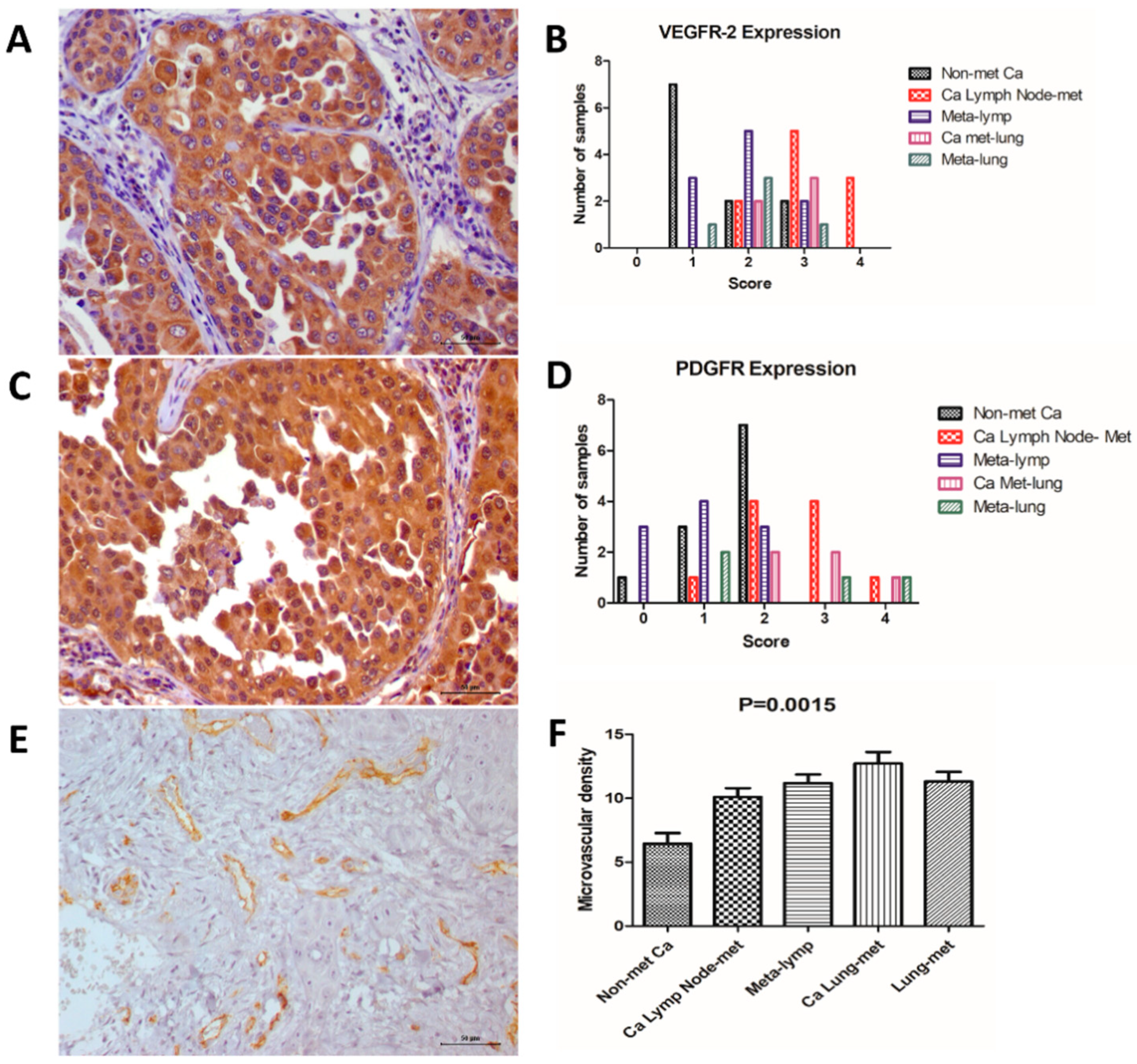
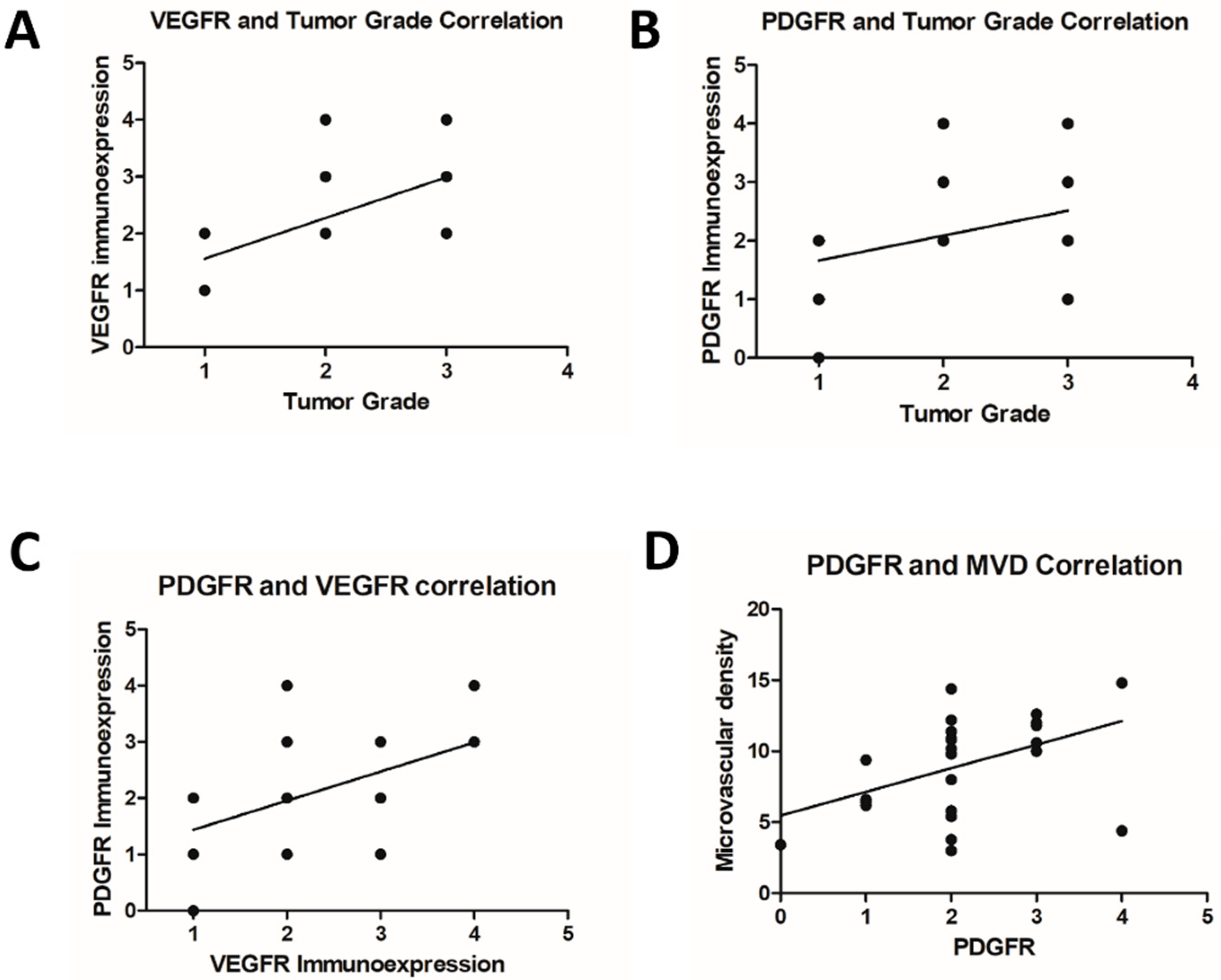
3.2. Immunohistochemistry
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Misdorp, W. Tumors of the mammary gland. In Tumors in Domestic Animals, 4th ed.; Meuten, D.J., Ed.; Iowa State Press: Ames, IA, USA, 2002; pp. 575–606. [Google Scholar]
- Sorenmo, K.U.; Worley, D.R.; Goldschmidt, M.H. Tumors of the Mammary Gland. In Withrow & MacEwen’s Small Animal Clinical Oncology, 5th ed.; Withrow, S.J., Vail, D.M., Page, R.L., Eds.; Elsevier: St Louis, MO, USA, 2013; pp. 538–556. [Google Scholar]
- De Nardi, A.B.; Rodaski, S.; Sousa, R.S.; Costa, T.A.; Macedo, T.R.; Rodigheri, S.M.; Rios, A.; Piekarz, C.H. Prevalência de neoplasias e modalidades de tratamentos em cães, atendidos no hospital veterinário da Universidade Federal do Paraná. Arch. Vet. Sci. 2002, 7, 15–26. [Google Scholar] [CrossRef]
- Santos, I.F.C.; Cardoso, J.M.M.; Oliveira, K.C.; Laisse, C.J.M.; Bessa, S.A.T. Prevalência de neoplasias diagnosticadas em cães no Hospital Veterinário da Universidade Eduardo Mondlane, Moçambique. Arq. Bras. Med. Vet. Zootec. 2013, 65, 773–782. [Google Scholar] [CrossRef]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Cancer Genome Atlas Network. Comprehensive molecular portraits of human breast tumours. Nature 2012, 490, 61–70. [Google Scholar] [CrossRef] [PubMed]
- McCart Reed, A.E.; Kalita-de Croft, P.; Kutasovic, J.; Saunus, J.M.; Lakhani, S.R. Recent advances in breast cancer research impacting clinical diagnostic practice. J. Pathol. 2018. accepted. [Google Scholar] [CrossRef] [PubMed]
- Abadie, J.; Nguyen, F.; Loussouarn, D.; Peña, L.; Gama, A.; Rieder, N.; Belousov, A.; Bemelmans, I.; Jaillardon, L.; Ibisch, C.; et al. Canine invasive mammary carcinomas as models of human breast cancer. Part 2: Immunophenotypes and prognostic significance. Breast Cancer Res. Treat. 2018, 167, 459–468. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Sullivan, C.A.; Zerkowski, M.P.; Molinaro, A.M.; Rimm, D.L.; Camp, R.L.; Chung, G.G. High levels of vascular endothelial growth factor and its receptors (VEGFR-1, VEGFR-2, neuropilin-1) are associated with worse outcome in breast cancer. Hum. Pathol. 2008, 39, 1835–1843. [Google Scholar] [CrossRef]
- Queiroga, F.L.; Pires, I.; Parente, M.; Gregório, H.; Lopes, C.S. COX-2 over-expression correlates with VEGF and tumour angiogenesis in canine mammary cancer. Vet. J. 2011, 189, 77–82. [Google Scholar] [CrossRef]
- Qiu, C.W.; Lin, D.G.; Wang, J.Q.; Li, C.Y.; Deng, G.Z. Expression and significance of PTEN and VEGF in canine mammary gland tumours. Vet. Res. Commun. 2008, 32, 463–472. [Google Scholar] [CrossRef]
- Santos, A.; Lopes, C.; Gärtner, F.; Matos, A.J. VEGFR-2 expression in malignant tumours of the canine mammary gland: A prospective survival study. Vet. Comp. Oncol. 2016, 14, 83–92. [Google Scholar] [CrossRef]
- Cristofanilli, M.; Morandi, P.; Krishnamurthy, S.; Reuben, J.M.; Lee, B.N.; Francis, D.; Booser, D.J.; Green, M.C.; Arun, B.K.; Pusztai, L.; et al. Imatinib mesylate (Gleevec) in advanced breast cancer-expressing C-Kit or PDGFR-beta: Clinical activity and biological correlations. Ann. Oncol. 2008, 19, 1713–1719. [Google Scholar] [CrossRef] [PubMed]
- Modi, S.; Seidman, A.D.; Dickler, M.; Moasser, M.; D’Andrea, G.; Moynahan, M.E.; Menell, J.; Panageas, K.S.; Tan, L.K.; Norton, L.; et al. A phase II trial of imatinib mesylate monotherapy in patients with metastatic breast cancer. Breast Cancer Res. Treat. 2005, 90, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Salvador, R.C.L.; Raposo, T.M.M.; Fonseca-Alves, C.E.; Terra, E.M.; Varallo, G.R.; Laufer-Amorim, R. Evaluation of c-KIT protein expression in canine mammary tumors. In BMC Proceedings; BioMed Central: London, UK, 2013; Volume 7, p. 63. [Google Scholar]
- Brunetti, B.; Beha, G.; Benazzi, C.; Bondin, V.; De Tolla, L.; Sarli, G. CD117 expression influences proliferation but not survival in canine mammary tumours. J. Comp. Pathol. 2014, 151, 202–206. [Google Scholar] [CrossRef] [PubMed]
- Koltai, Z.; Szabó, B.; Jakus, J.; Vajdovich, P. Tyrosine kinase expression analyses in canine mammary gland tumours—A pilot study. Acta Vet. Hung. 2018, 66, 294–308. [Google Scholar] [CrossRef] [PubMed]
- London, C.A.; Hannah, A.L.; Zadovoskaya, R.; Chien, M.B.; Kollias-Baker, C.; Rosenberg, M.; Downing, S.; Post, G.; Boucher, J.; Shenoy, N.; et al. Phase I dose-escalating study of SU11654, a small molecule receptor tyrosine kinase inhibitor, in dogs with spontaneous malignancies. Clin. Cancer Res. 2003, 9, 2755–2768. [Google Scholar] [PubMed]
- Nakano, Y.; Kobayashi, T.; Oshima, F.; Fukazawa, E.; Yamagami, T.; Shiraishi, Y.; Takanosu, M. Imatinib responsiveness in canine mast cell tumors carrying novel mutations of c-KIT exon 11. J. Vet. Med. Sci. 2014, 76, 545–548. [Google Scholar] [CrossRef] [PubMed]
- Foskett, A.; Manley, C.; Naramore, R.; Gordon, I.K.; Stewart, B.M.; Khanna, C. Tolerability of oral sorafenib in pet dogs with a diagnosis of cancer. Vet. Med. 2017, 8, 97–102. [Google Scholar] [CrossRef]
- Cao, R.; Björndahl, M.A.; Religa, P.; Clasper, S.; Garvin, S.; Galter, D.; Meister, B.; Ikomi, F.; Tritsaris, K.; Dissing, S.; et al. PDGF-BB induces intratumoral lymphangiogenesis and promotes lymphatic metastasis. Cancer Cell 2004, 6, 333–345. [Google Scholar] [CrossRef]
- Shibuya, M. Vascular Endothelial Growth Factor (VEGF) and Its Receptor (VEGFR) Signaling in Angiogenesis: A Crucial Target for Anti- and Pro-Angiogenic Therapies. Genes Cancer 2011, 2, 1097–1105. [Google Scholar] [CrossRef]
- Weidner, N.; Folkman, J.; Pozza, F.; Bevilacqua, P.; Allred, E.N.; Moore, D.H.; Meli, S.; Gasparini, G. Tumor angiogenesis: A new significant and independent prognostic indicator in early-stage breast carcinoma. J. Nat. Cancer Inst. 1992, 84, 1875–1887. [Google Scholar] [CrossRef]
- Bosari, S.; Lee, A.K.C.; DeLellis, R.A.; Wiley, B.D.; Heatley, G.J.; Silverman, M.L. Microvessel quantification and prognosis in invasive breast carcinoma. Hum. Pathol. 1992, 23, 755–761. [Google Scholar] [CrossRef]
- Graham, J.C.; Myers, R.K. The prognostic significance of angiogenesis in canine mammary tumors. J. Vet. Intern. Med. 1999, 13, 416–418. [Google Scholar] [CrossRef] [PubMed]
- Beserra, H.E.O.; Cavalcante, R.V.; de Pinho Pessoa, A.W.; Pinheiro, L.G.P. Technical of sentinel lymph detection in canine mammary gland using Patent Blue V and technetium [sup. 99m] Tc/Tecnica de deteccao do linfonodo sentinela da glandula mamaria de cadelas utilizando Azul Patente V e tecnecio [sup. 99m] Tc/Tecnica para la deteccion del ganglio linfatico centinela en glandulas mamarias de perras con Azul Patente y tecnecio [sup. 99m] Tc. Vet. Zootec. 2011, 18, 57–60. [Google Scholar]
- Goldschimidt, M.; Peña, L.; Rasotto, R.; Zappuli, V. Classification and grading of canine mammary tumors. Vet. Pathol. 2011, 48, 117–131. [Google Scholar] [CrossRef] [PubMed]
- Karayannopoulou, M.; Kaldrymidou, E.; Constantinidis, T.C.; Dessiris, A. Histological grading and prognosis in dogs with mammary carcinomas: Application of a humangrading method. J. Comp. Pathol. 2005, 133, 246–252. [Google Scholar] [CrossRef] [PubMed]
- Bonolo De Campos, C.; Lavalle, G.E.; Monteiro, L.N.; Pêgas, G.R.A.; Fialho, S.L.; Balabram, D.; Cassali, G.D. Adjuvant Thalidomide and Metronomic Chemotherapy for the Treatment of Canine Malignant Mammary Gland Neoplasms. In Vivo 2018, 32, 1659–1666. [Google Scholar] [CrossRef] [PubMed]
- Fonseca-Alves, C.E.; Kobayashi, P.E.; Rivera Calderón, L.G.; Felisbino, S.L.; Rinaldi, J.C.; Drigo, S.A.; Rogatto, S.R.; Laufer-Amorim, R. Immunohistochemical panel to characterize canine prostate carcinomas according to aberrant p63 expression. PLoS ONE 2018, 13, 1–16. [Google Scholar] [CrossRef]
- Omidvari, S.; Hamedi, S.H.; Mohammadianpanah, M.; Nasrolahi, H.; Mosalaei, A.; Talei, A.; Ahmadloo, N.; Ansari, M. Very late relapse in breast cancer survivors: A report of 6 cases. Iran. J. Cancer Prev. 2013, 6, 113–117. [Google Scholar]
- Yan, J.D.; Liu, Y.; Zhang, Z.Y.; Liu, G.Y.; Xu, J.H.; Liu, L.Y.; Hu, Y.M. Expression and prognostic significance of VEGFR-2 in breast cancer. Pathol. Res. Pract. 2015, 211, 539–543. [Google Scholar] [CrossRef]
- Sloan, B.; Scheinfeld, N.S. Pazopanib, a VEGF receptor tyrosine kinase inhibitor for cancer therapy. Curr. Opin. Investing. Drug 2008, 9, 1324–1335. [Google Scholar]
- Raposo, T.P.; Pires, I.; Carvalho, M.I.; Prada, J.; Argyle, D.J.; Queiroga, F.L. Tumour-associated macrophages are associated with vascular endothelial growth factor expression in canine mammary tumours. Vet. Comp. Oncol. 2015, 13, 464–474. [Google Scholar] [CrossRef] [PubMed]
- Camacho, L.; Peña, L.; Gil, A.G.; Martín-Ruiz, A.; Dunner, S.; Illera, J.C. Immunohistochemical vascular factor expression in canine inflammatory mammary carcinoma. Vet. Pathol. 2014, 51, 737–748. [Google Scholar] [CrossRef] [PubMed]
| Clinical Parameters | n | % |
|---|---|---|
| Age (years) | ||
| <10.5 | 16 | 61.5 |
| >10.5 | 10 | 38.5 |
| Neutering Status | ||
| Intact | 26 | 100% |
| Neutered | 0 | 0 |
| Tumor size | ||
| <5 cm | 18 | 69.2% |
| >5 cm | 8 | 30.8% |
| Nodal stage * | ||
| N0 | 16 | 61.5% |
| N1 | 10 | 38.5% |
| Histological grade ** | ||
| Grade I | 7 | 26.9% |
| Grade II | 8 | 30.8% |
| Grade III | 11 | 42.3% |
| VEGFR-2 Score | PDGFR Score | Microvascular Density | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 1 | 2 | 3 | 4 | 0 | 1 | 2 | 3 | 4 | Mean (SD *) | |
| Nonmetastatic Carcinomas (n = 11) | 0% (n = 0) | 45.5% (n = 5) | 45.5% (n = 5) | 9% (n = 1) | 0% (n = 0) | 9% (n = 1) | 27.3% (n = 3) | 63.7% (n = 7) | 0% (n = 0) | 0% (n = 0) | 6.4 (±2.7) |
| Carcinomas Lymph Node Metastasis (n = 10) | 0% (n = 0) | 0% (n = 0) | 20% (n = 2) | 50% (n = 5) | 30% (n = 3) | 0% (n = 0) | 10% (n = 1) | 40% (n = 4) | 40% (n = 4) | 10% (n = 1) | 10.1 (±2.2) |
| Lymph Node Metastasis (n = 10) | 0% (n = 0) | 30% (n = 3) | 50% (n = 5) | 20% (n = 2) | 0% (n = 0) | 30% (n = 3) | 40% (n = 4) | 30% (n = 3) | 0% (n = 0) | 0% (n = 0) | 11.2 (±2.0) |
| Carcinomas Lung Metastasis (n = 5) | 0% (n = 0) | 0% (n = 0) | 0% (n = 0) | 60% (n = 3) | 40% (n = 2) | 0% (n = 0) | 0% (n = 0) | 40% (n = 2) | 40% (n = 2) | 20% (n = 1) | 12.7 (±2.0) |
| Lung Metastasis (n = 5) | 0% (n = 0) | 20% (n = 1) | 60% (n = 3) | 20% (n = 1) | 0% (n = 0) | 20% (n = 1) | 40% (n = 2) | 0% (n = 0) | 20% (n = 1) | 20% (n = 1) | 11.3 (±1.7) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dos Anjos, D.S.; Vital, A.F.; Lainetti, P.d.F.; Leis-Filho, A.F.; Dalmolin, F.; Elias, F.; Calazans, S.G.; Fonseca-Alves, C.E. Deregulation of VEGFR-2 and PDGFR Expression and Microvascular Density in a Triple-Negative Model of Canine Malignant Mammary Tumors with Lymph Node or Lung Metastasis. Vet. Sci. 2019, 6, 3. https://doi.org/10.3390/vetsci6010003
Dos Anjos DS, Vital AF, Lainetti PdF, Leis-Filho AF, Dalmolin F, Elias F, Calazans SG, Fonseca-Alves CE. Deregulation of VEGFR-2 and PDGFR Expression and Microvascular Density in a Triple-Negative Model of Canine Malignant Mammary Tumors with Lymph Node or Lung Metastasis. Veterinary Sciences. 2019; 6(1):3. https://doi.org/10.3390/vetsci6010003
Chicago/Turabian StyleDos Anjos, Denner Santos, Aline Fernandes Vital, Patrícia de Faria Lainetti, Antonio Fernando Leis-Filho, Fabiola Dalmolin, Fabiana Elias, Sabryna Gouveia Calazans, and Carlos Eduardo Fonseca-Alves. 2019. "Deregulation of VEGFR-2 and PDGFR Expression and Microvascular Density in a Triple-Negative Model of Canine Malignant Mammary Tumors with Lymph Node or Lung Metastasis" Veterinary Sciences 6, no. 1: 3. https://doi.org/10.3390/vetsci6010003
APA StyleDos Anjos, D. S., Vital, A. F., Lainetti, P. d. F., Leis-Filho, A. F., Dalmolin, F., Elias, F., Calazans, S. G., & Fonseca-Alves, C. E. (2019). Deregulation of VEGFR-2 and PDGFR Expression and Microvascular Density in a Triple-Negative Model of Canine Malignant Mammary Tumors with Lymph Node or Lung Metastasis. Veterinary Sciences, 6(1), 3. https://doi.org/10.3390/vetsci6010003






