Chemical Characterization of Opuntia ficus-indica (L.) Mill. Hydroalcoholic Extract and Its Efficiency against Gastrointestinal Nematodes of Sheep
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material and Preparation of the Hydroalcoholic Extract of the Cladode Peels of O. ficus-indica
2.2. Total Carbohydrate Content
2.3. Total Protein Content
2.4. Total Phenolic Content
2.5. Condensed Ttannins Content
2.6. Liquid Chromatography Coupled to Mass Spectrometry/Electron Spray Ionization (LC-MS/ESI)
2.7. Fecal Sample Collection and Eggs Retrieval
2.8. Egg Hatch Test (EHT)
2.9. Larval Migration Test (LMT) and Extract-Drug Interaction
2.10. Statistical Analysis
3. Results and Discussion
3.1. Colorimetric Dosages of the OFIEOH of Cladode Peels from O. ficus-indica
3.2. Characterization of Phenolic Profile of OFIEOH Extracted by LC-MS/ESI
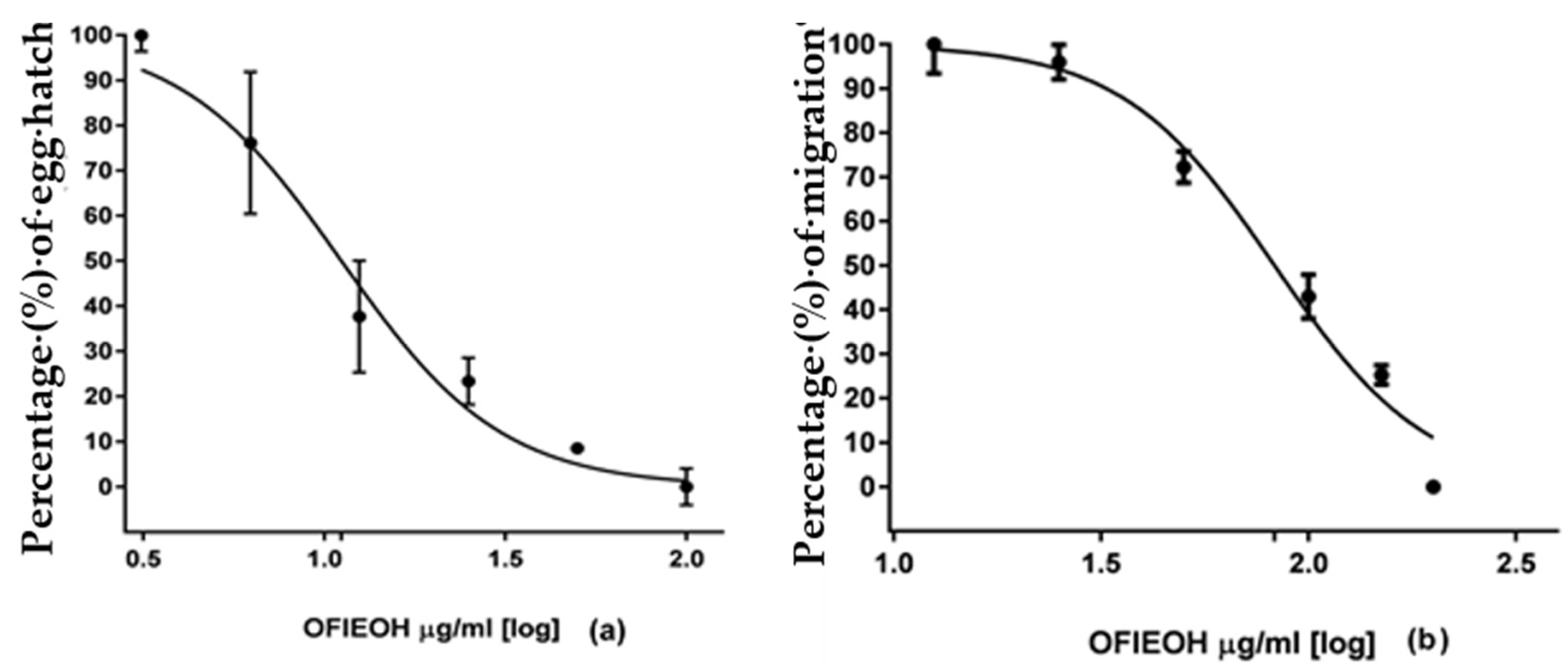
3.3. Egg Hatch Test (EHT) and Larval Migration Test (LMT)
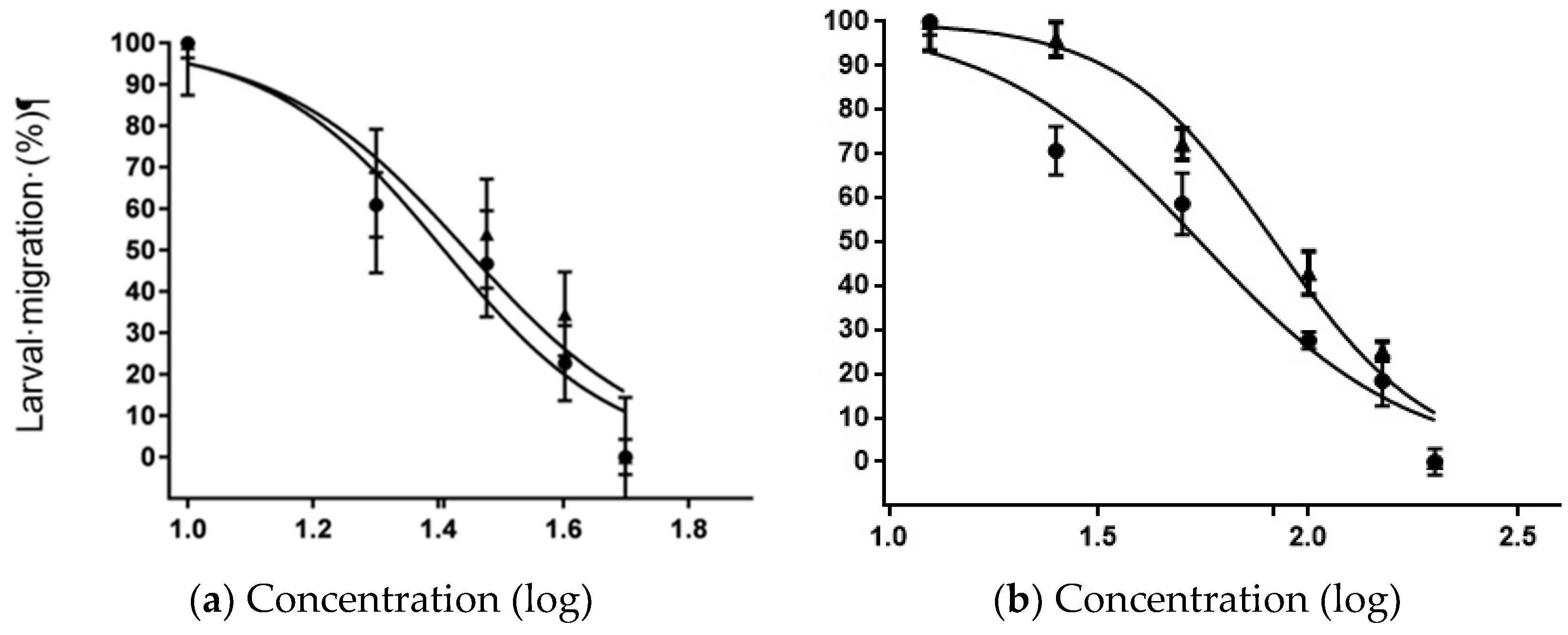
3.4. Larval Migration Test (LMT) and Extract-Drug Interaction
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Marinho, M.L.; Alves, M.S.; Rodrigues, M.L.C.; Rotondano, T.E.F.; Vidal, I.F.; Silva, W.W.; Athayde, A.C.R. A utilização de plantas medicinais em medicina veterinária: Um resgate do saber popular. Rev. Bras. Plant. Med. 2007, 9, 64–69. [Google Scholar]
- Soldera-Silva, A.; Seyfried, M.; Campestrini, L.H.; Minho, A.P.; Zawadzki-Baggio, S.F.; Molento, M.; Maurer, J.B.B. Assessment of anthelmintic activity and bio-guided chemical analysis of Persea americana seed extracts. Vet. Parasitol. 2018, 251, 34–43. [Google Scholar] [CrossRef] [PubMed]
- Mendonça, V.M.; Santos, A.J.; Nascimento, I.R.; Oliveira, M.A.S.; Rocha, S.S.; Cabral, E.S.S. Perspectivas da fitoterapia veterinária: Plantas potenciais na terapia dos animais de produção. Cad. Agroecol. 2014, 9, 1–5. [Google Scholar]
- Sprenger, L.K.; Giese, E.; Santos, J.N.; Molento, M.B. Atividade antimicrobiana do extrato hidroalcóolico de Ficus carica e Polygala spectabilis. Arch. Vet. Sci. 2017, 22, 1–7. [Google Scholar] [CrossRef]
- Stintzing, F.C.; Carle, R. Cactus stems (Opuntia spp.): A review on their chemistry, technology, and uses. Mol. Nutr. Food Res. 2005, 49, 175–194. [Google Scholar] [CrossRef] [PubMed]
- Feugang, J.M.; Konarski, P.; Zou, D.; Stintzing, F.C.; Zou, C. Medicinal use of cactus pear (Opuntia spp.) cladodes and fruits. Front. Biosci. 2006, 11, 2574–2589. [Google Scholar] [CrossRef] [PubMed]
- Griffith, M.P. The origins of an important cactus crop, Opuntia ficus-indica (Cactaceae): New molecular evidence. Am. J. Bot. 2004, 91, 1915–1921. [Google Scholar] [CrossRef] [PubMed]
- Wanderley, W.L.; Ferreira, M.A.; Batista, A.M.V.; Véras, A.S.; Bispo, S.V.; Silva, F.M.; Santos, V.L.F. Consumo, digestibilidade e parâmetros ruminais em ovinos recebendo silagens e fenos em associação à palma forrageira. Rev. Bras. Saúde Prod. Anim. 2012, 13, 444–456. [Google Scholar] [CrossRef]
- Rangel, A.H.N.; Lima Junior, D.M.; Braga, A.P.; Simplício, A.A.; Aguiar, E.M. Suprimento e demanda de nutrientes em sistemas em não equilíbrio. Rev. Verde 2009, 4, 14–30. [Google Scholar]
- Viegi, L.; Pieroni, A.; Guarrera, P.M.; Vangelisti, R. A review of plants used in folk veterinary medicine in Italy as basis for a databank. J. Ethnopharmacol. 2003, 89, 221–244. [Google Scholar] [CrossRef] [PubMed]
- Féboli, A.; Laurentiz, A.C.; Soares, S.C.; Augusto, J.G.; Anjos, L.A.; Magalhães, L.G.; Filardi, R.S.; Laurentiz, R.S. Ovicidal and larvicidal activity of extracts of Opuntia ficus-indica against gastrointestinal nematodes of naturally infected sheep. Vet. Parasitol. 2016, 226, 65–68. [Google Scholar] [CrossRef] [PubMed]
- Shedbalkar, U.U.; Adki, V.S.; Jadhav, J.P.; Bapat, V.A. Opuntia and other cacti: Applications and biotechnological insights. Trop. Plant Biol. 2010, 3, 136–150. [Google Scholar] [CrossRef]
- Ginestra, G.; Parker, M.L.; Bennett, R.N.; Robertson, J.; Mandalari, G.; Narbad, A.; Curto, R.B.L.; Bisignano, G.; Faulds, C.B.; Waldron, K.W. Anatomical, chemical, and biochemical characterization of cladodes from prickly pear [Opuntia ficus-indica (L.) Mill.]. J. Agric. Food Chem. 2009, 57, 10323–10330. [Google Scholar] [CrossRef] [PubMed]
- Cardador-Martínez, A.; Jiménez-Martínez, C.; Sandoval, G. Revalorization of cactus pear (Opuntia spp.) wastes as a source of antioxidants. Food Sci. Technol. 2011, 31, 782–788. [Google Scholar]
- Fox, J.D.; Robyt, J.F. Miniaturization of three carbohydrate analysis using a micro sample plate reader. Anal. Biochem. 1991, 195, 93–96. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Morais, S.A.L.; Aquino, F.J.T.; Nascimento, P.M.; Nascimento, E.A.; Chang, R. Compostos bioativos e atividade antioxidante do café conilon submetido a diferentes graus de torra. Quim. Nova 2009, 32, 327–331. [Google Scholar] [CrossRef]
- Queiroz, C.R.A.A.; Morais, S.A.L.; Nascimento, E.A. Caracterização dos taninos da aroeira-preta. Soc. Invest. Flor. 2002, 26, 485–492. [Google Scholar]
- Yamassaki, F.T.; Campestrini, L.H.; Zawadzki-Baggio, S.F.; Maurer, J.B.B. Avocado leaves: Influence of drying process, thermal incubation, and storage conditions on preservation of polyphenolic compounds and antioxidant activity. Int. J. Food Prop. 2017, 20, 2280–2293. [Google Scholar] [CrossRef]
- Coles, G.C.; Jackson, F.; Pomroy, W.E.; Prichard, K.; Von Samson-Himmelstjerna, G.; Silvestre, A.; Taylor, M.A.; Vercruysse, J. The detection of anthelmintic resistance in nematodes of veterinary importance. Vet. Parasitol. 2006, 136, 167–185. [Google Scholar] [CrossRef] [PubMed]
- D’Assonville, J.A.; Janovsky, E.; Verster, A. In vitro screening of Haemonchus contortus third stage larvae for ivermectin resistance. Vet. Parasitol. 1996, 61, 73–80. [Google Scholar] [CrossRef]
- Molento, M.B.; Prichard, R.K. Effect of multidrug resistance modulators on the activity of ivermectin and moxidectin against selected strains of Haemonchus contortus infective larvae. Pesqui. Vet. Bras. 2001, 21, 117–121. [Google Scholar] [CrossRef]
- Alves, F.A.L.; Andrade, A.P.; Bruno, R.L.A.; Silva, M.G.V.; De Souza, M.F.V.; Santos, D.C. Seasonal variability of phenolic compounds and antioxidant activity in prickly pear cladodes of Opuntia and Nopalea genres. Food Sci. Technol. 2017, 37, 536–543. [Google Scholar] [CrossRef]
- Lee, E.B.; Hyun, J.E.; Li, D.W.; Moon, Y.I. Effects of Opuntia ficus-indica var. saboten stem on gastric damage in rats. Arch. Pharm. Res. 2002, 25, 67–70. [Google Scholar]
- Jorge, A.J.; De La Garza, T.H.; Alejandro, Z.; Ruth, B.C.; Noé, A.C. The optimization of phenolic compounds extraction from cactus pear (Opuntia ficus-indica) skin in a reflux system using response surface methodology. Asian Pac. J. Trop. Biomed. 2013, 3, 436–442. [Google Scholar] [CrossRef]
- Horai, H.; Arita, M.; Kanaya, S.; Nihei, Y.; Ikeda, T.; Suwa, K.; Ojima, Y.; Tanaka, K.; Tanaka, S.; Aoshima, K.; et al. MassBank: A public repository for sharing mass spectral data for life sciences. J. Mass Spectrom. 2010, 45, 703–714. [Google Scholar] [CrossRef] [PubMed]
- Figueroa-Pérez, M.G.; Pérez-Ramírez, I.F.; Paredes-López, O.; Mondragón-Jacobo, C.; Reynoso-Camacho, R. Phytochemical composition and in vitro analysis of nopal (O. ficus-indica) cladodes at different stages of maturity. Int. J. Food Prop. 2018, 21, 1728–1742. [Google Scholar] [CrossRef]
- Kim, J.W.; Kim, T.B.; Yang, H.; Sung, H.S. Phenolic compounds isolated from Opuntia ficus-indica fruits. Nat. Prod. Sci. 2016, 22, 117–121. [Google Scholar] [CrossRef]
- Allai, L.; Karym, E.M.; Amiri, B.E.; Nasser, B.; Essamad, A.; Terzioğlu, P.; Ertas, A.; Öztürk, M. Evaluation of antioxidant activity and phenolic composition of Opuntia ficus-indica cladodes collected from Moroccan settat region. Eur. J. Anal. Chem. 2017, 12, 105–117. [Google Scholar] [CrossRef]
- De Leo, M.; Bruzual de Abreu, M.; Pawlowska, A.M.; Cioni, P.L.; Braca, A. Profiling the chemical content of Opuntia ficus-indica flowers by HPLC–PDA-ESI-MS and GC/EIMS analyses. Phytochem. Lett. 2010, 3, 48–52. [Google Scholar] [CrossRef]
- Naczk, M.; Shahidi, F. Extraction and analysis of phenolics in food. J. Chromatogr. A 2004, 1054, 95–111. [Google Scholar] [CrossRef]
- Russo, M.; Spagnuolo, C.; Tedesco, I.; Bilotto, S.; Russo, G.L. The flavonoid quercetin in disease prevention and therapy: Facts and fancies. Biochem. Pharmacol. 2012, 83, 6–15. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Ortíz-De-Montellano, C.; Vargas-Magaña, J.J.; Canul-Ku, H.L.; Miranda-Soberanis, R.; Capetillo-Leal, C.; Sandoval-Castro, C.A.; Hoste, H.; Torres-Acosta, J.F.J. Effect of a tropical tannin-rich plant Lysiloma latisiliquum on adult populations of Haemonchus contortus in sheep. Vet. Parasitol. 2010, 172, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Russo, G.L.; Russo, M.; Spagnuolo, C. The pleiotropic flavonoid quercetin: From its metabolism to the inhibition of protein kinases in chronic lymphocytic leukemia. Food Funct. 2014, 5, 2393–2401. [Google Scholar] [CrossRef] [PubMed]
- Hoste, H.; Martinez-Ortiz-de-Montellano, C.; Manolaraki, F.; Brunet, S.; Ojeda-Robertos, N.; Fourquaux, I.; Torres-Acosta, J.F.F.; Sandoval-Castro, C.A. Direct and indirect effects of bioactive tannin-rich tropical and temperate legumes against nematode infections. Vet. Parasitol. 2012, 186, 18–27. [Google Scholar] [CrossRef] [PubMed]
- Akkari, H.; Rtibi, K.; B’chir, F.; Rekik, M.; Darghouth, M.A.; Gharbi, M. In vitro evidence that the pastoral Artemisia campestris species exerts an anthelmintic effect on Haemonchus contortus from sheep. Vet. Res. Commun. 2014, 38, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Lasisi, A.A.; Kareem, S.O. Evaluation of anthelmintic activity of the stem bark extract and chemical constituents of Bridelia ferruginae (Benth) Euphorbiaceae. Afr. J. Plant Sci. 2011, 5, 469–474. [Google Scholar]
- Klongsiriwet, C.; Quijada, J.; Williams, A.R.; Mueller-Harvey, I.; Williamson, E.M.; Hoste, H. Synergistic inhibition of Haemonchus contortus exsheathment by flavonoid monomers and condensed tannins. Int. J. Parasitol. Drugs Drug Resist. 2015, 5, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Molento, M.B. Parasite control in the age of drug resistance and changing agricultural practices. Vet. Parasitol. 2009, 163, 229–234. [Google Scholar] [CrossRef] [PubMed]
- Molento, M.B.; Fortes, F.S.; Pondelek, D.A.S.; Borges, F.A.; Chagas, A.C.; Torres-Acosta, J.F.J.; Geldhof, P. Challenges of nematode control in ruminants: Focus on Latin America. Vet. Parasitol. 2011, 180, 126–132. [Google Scholar] [CrossRef] [PubMed]
- Geary, T.G.; Hosking, B.C.; Skuce, P.J.; Samson-Himmelstjerna, G.; Maeder, S.; Holdsworth, P.; Pomroy, W.; Vercruysse, J. World association for the advancement of veterinary parasitology (W.A.A.V.P.) guideline: Anthelmintic combination products targeting nematode infections of ruminants and horses. Vet. Parasitol. 2012, 190, 306–316. [Google Scholar] [CrossRef]
- Canton, C.; Ceballos, L.; Fiel, C.; Moreno, L.; Yagüez, P.D.; Bernat, G.; Lanusse, C.; Alvarez, L. Resistant nematodes in cattle: Pharmaco-therapeutic assessment of the ivermectin-ricobendazole combination. Vet. Parasitol. 2017, 234, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Fugh-Berman, A. Herb-drug interactions. Lancet 2000, 355, 134–138. [Google Scholar] [CrossRef]
- Sunada, N.S.; Orrico Junior, M.P.; Orrico, A.C.A.; Oliveira, A.M.; Centurion, S.R.; Lima, S.R.N.; Fernandes, R.M.; Vargas Junior, F.M. Parasite control using levamisol, ivermectin and dried garlic (Allium sativum) in Santa Inês sheep. Rev. Agric. 2011, 4, 140–145. [Google Scholar]
- Borges, F.A.; Rossini, J.B.; Velludo, P.P.; Buzzulini, C.B.; Costa, G.H.; Molento, M.B.; Costa, A.J. Weak phenotypic reversion of ivermectin resistance in a field resistant isolate of Haemonchus contortus by verapamil. Pesqui. Vet. Bras. 2011, 31, 731–736. [Google Scholar] [CrossRef]
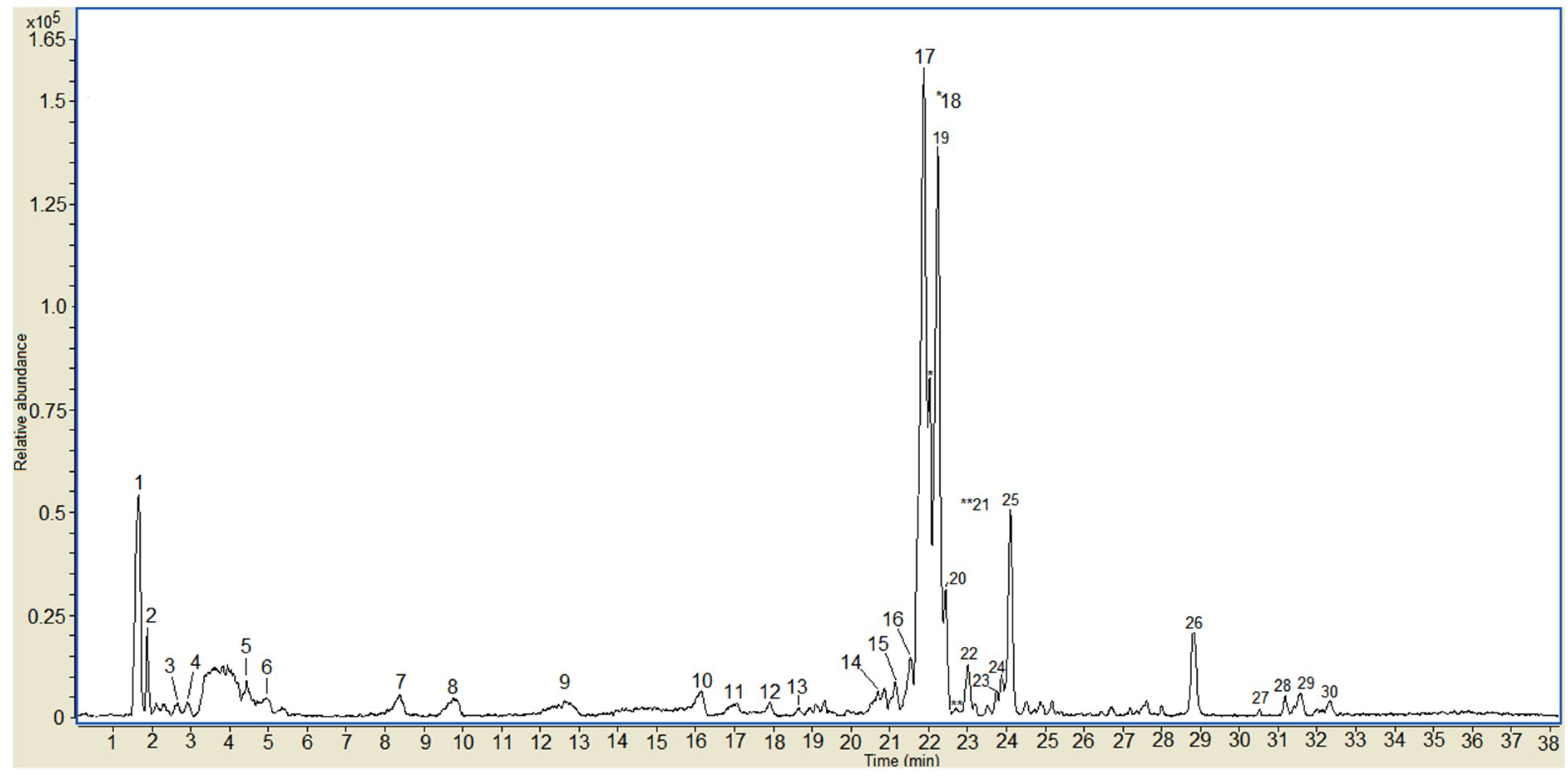
| Peak | Rt (min) | [M − H]− (m/z) | Identified and Suggested Compounds |
|---|---|---|---|
| 5 | 4.4 | 447 | mono-glycosylated kaempferol |
| 14 | 20.1 | 300 | quercetin |
| 16 | 21.5 | 739 | tri-glycoslylated kaempferol |
| 17 | 21.7 | 769 | tri-glycosylated methyl-quercetin derivative I |
| 18 | 21.9 | 769 | tri-glycosylated methyl-quercetin derivative II |
| 19 | 22.0 | 769 | tri-glycosylated methyl-quercetin derivative III |
| 20 | 22.2 | 755 | tri-glycosylated quercetin I |
| 21 | 22.4 | 755 | tri-glycosylated quercetin II |
| 22 | 23.0 | 609 | di-glycosylated quercetin (Rutin) |
| 23 | 23.7 | 593 | di-glycosylated kaempferol |
| 24 | 23.9 | 623 | di-glycosylated methyl-quercetin I |
| 25 | 24.1 | 623 | di-glycosylated methyl-quercetin II |
| 26 | 28.8 | 315 | methyl-quercetin |
| 29 | 31.6 | 299 | kaempferide (Kaempferol 4’-methyl ether) |
| 30 | 32.3 | 297 | mono-glycosylated acetyl phenyl derivative |
| OFIEOH (mg/mL) | Inhibition of Egg Hatch (%) |
|---|---|
| 100 | 90.00 ± 3.04 |
| 50 | 80.67 ± 0.96 |
| 25 | 70.67 ± 3.94 |
| 12.5 | 61.00 ± 9.39 |
| 6.25 | 33.67 ± 11.92 |
| 3.125 | 19.33 ± 2.74 |
| H2O | 5.33 ± 2.98 |
| ivermectin (114.3 µM) | 90.67 ± 3.63 |
| IC50 | 11.15 mg/mL |
| OFIEOH (mg/mL) | Inhibition of Larval Migration |
|---|---|
| 200 | 77.26 ± 0.66 |
| 150 | 59.21 ± 1.77 |
| 100 | 49.57 ± 4.22 |
| 50 | 25.63 ± 2.88 |
| 25 | 8.66 ± 3.11 |
| 12.5 | 5.78 ± 5.33 |
| H2O | 1.08 ± 4.44 |
| ivermectin (114.3 µM) | 87.00 ± 4.66 |
| IC50 | 82.79 mg/mL |
| Concentration of IVM (µM) + OFIEOH (mg/mL) | Inhibition (%) |
|---|---|
| 57.1 + 82.80 | 73.78 ± 6.11 |
| 47.7 + 82.80 | 53.85 ± 4.11 |
| 34.3 + 82.80 | 42.66 ± 5.53 |
| 22.8 + 82.80 | 38.11 ± 6.68 |
| 11.4 + 82.80 | 16.08 ± 1.33 |
| H2O/Control | 0.70 ± 2.22 |
| IC50 IVM | 25.75 µg/mL |
| Concentration of OFIEOH (mg/mL) + IVM (µM) | Inhibition (%) |
|---|---|
| 200 + 25.7 | 62.49 ± 2.11 |
| 150 + 25.7 | 45.82 ± 4.22 |
| 100 + 25.7 | 37.48 ± 1.33 |
| 50 + 25.7 | 9.35 ± 5.33 |
| 25 + 25.7 | 3.09 ± 7.33 |
| 12.5 + 25.7 | 1.53 ± 4.11 |
| H2O/Control | 0.35 ± 3.33 |
| IC50 OFIEOH | 82.79 mg/mL |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santos, C.; Campestrini, L.H.; Vieira, D.L.; Pritsch, I.; Yamassaki, F.T.; Zawadzki-Baggio, S.F.; Maurer, J.B.B.; Molento, M.B. Chemical Characterization of Opuntia ficus-indica (L.) Mill. Hydroalcoholic Extract and Its Efficiency against Gastrointestinal Nematodes of Sheep. Vet. Sci. 2018, 5, 80. https://doi.org/10.3390/vetsci5030080
Santos C, Campestrini LH, Vieira DL, Pritsch I, Yamassaki FT, Zawadzki-Baggio SF, Maurer JBB, Molento MB. Chemical Characterization of Opuntia ficus-indica (L.) Mill. Hydroalcoholic Extract and Its Efficiency against Gastrointestinal Nematodes of Sheep. Veterinary Sciences. 2018; 5(3):80. https://doi.org/10.3390/vetsci5030080
Chicago/Turabian StyleSantos, Carolina, Luciano Henrique Campestrini, Douglas Luis Vieira, Izanara Pritsch, Fábio Tomio Yamassaki, Selma Faria Zawadzki-Baggio, Juliana Bello Baron Maurer, and Marcelo Beltrão Molento. 2018. "Chemical Characterization of Opuntia ficus-indica (L.) Mill. Hydroalcoholic Extract and Its Efficiency against Gastrointestinal Nematodes of Sheep" Veterinary Sciences 5, no. 3: 80. https://doi.org/10.3390/vetsci5030080
APA StyleSantos, C., Campestrini, L. H., Vieira, D. L., Pritsch, I., Yamassaki, F. T., Zawadzki-Baggio, S. F., Maurer, J. B. B., & Molento, M. B. (2018). Chemical Characterization of Opuntia ficus-indica (L.) Mill. Hydroalcoholic Extract and Its Efficiency against Gastrointestinal Nematodes of Sheep. Veterinary Sciences, 5(3), 80. https://doi.org/10.3390/vetsci5030080






