Sorghum Coffee–Lactose Stout Production and Its Physico-Chemical Characterization
Abstract
:1. Introduction
2. Materials and Methods
2.1. Acquisition of Materials
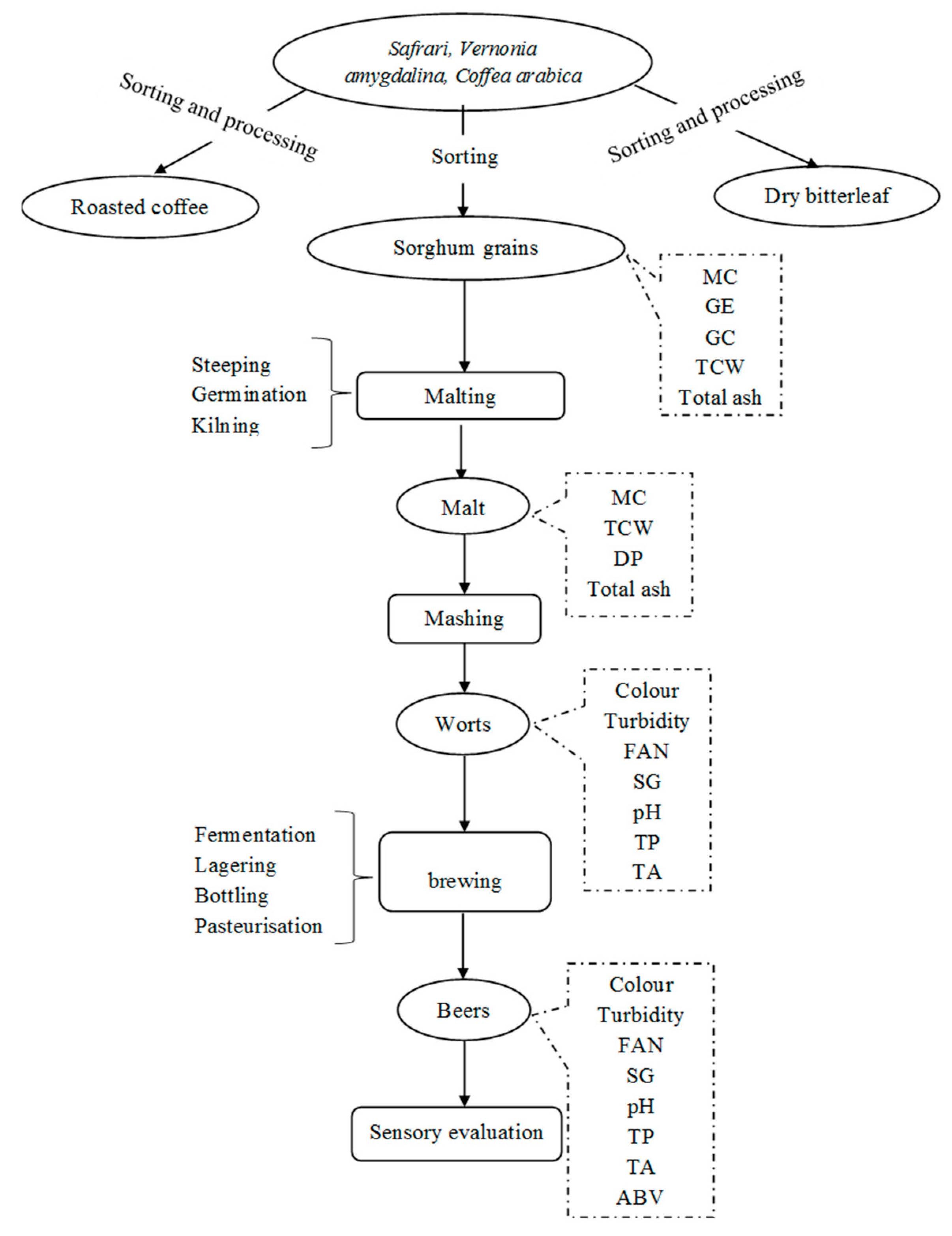
2.2. General Work Overview
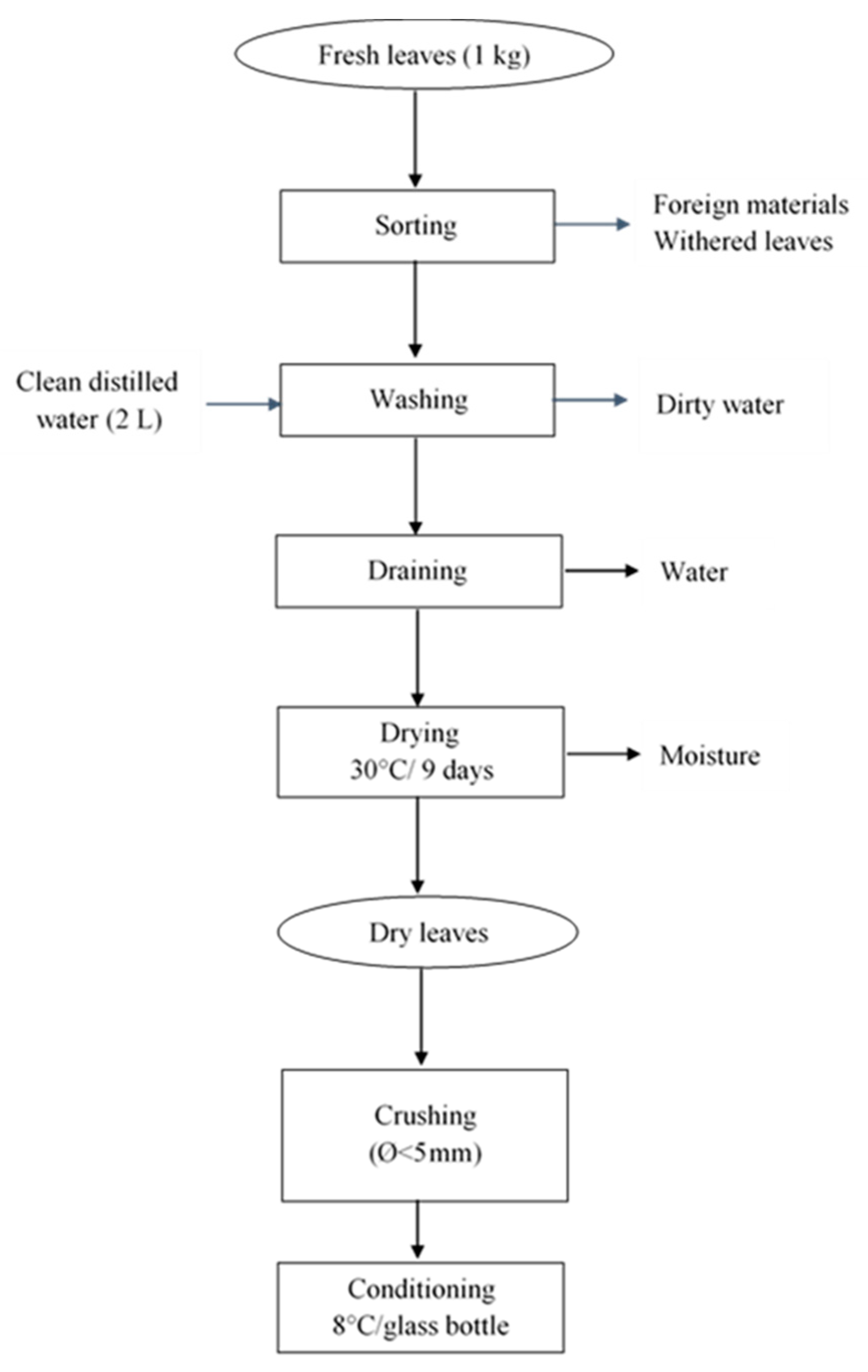
2.3. Processing of Raw Bitter Leaves (Vernonia Amygdalina)
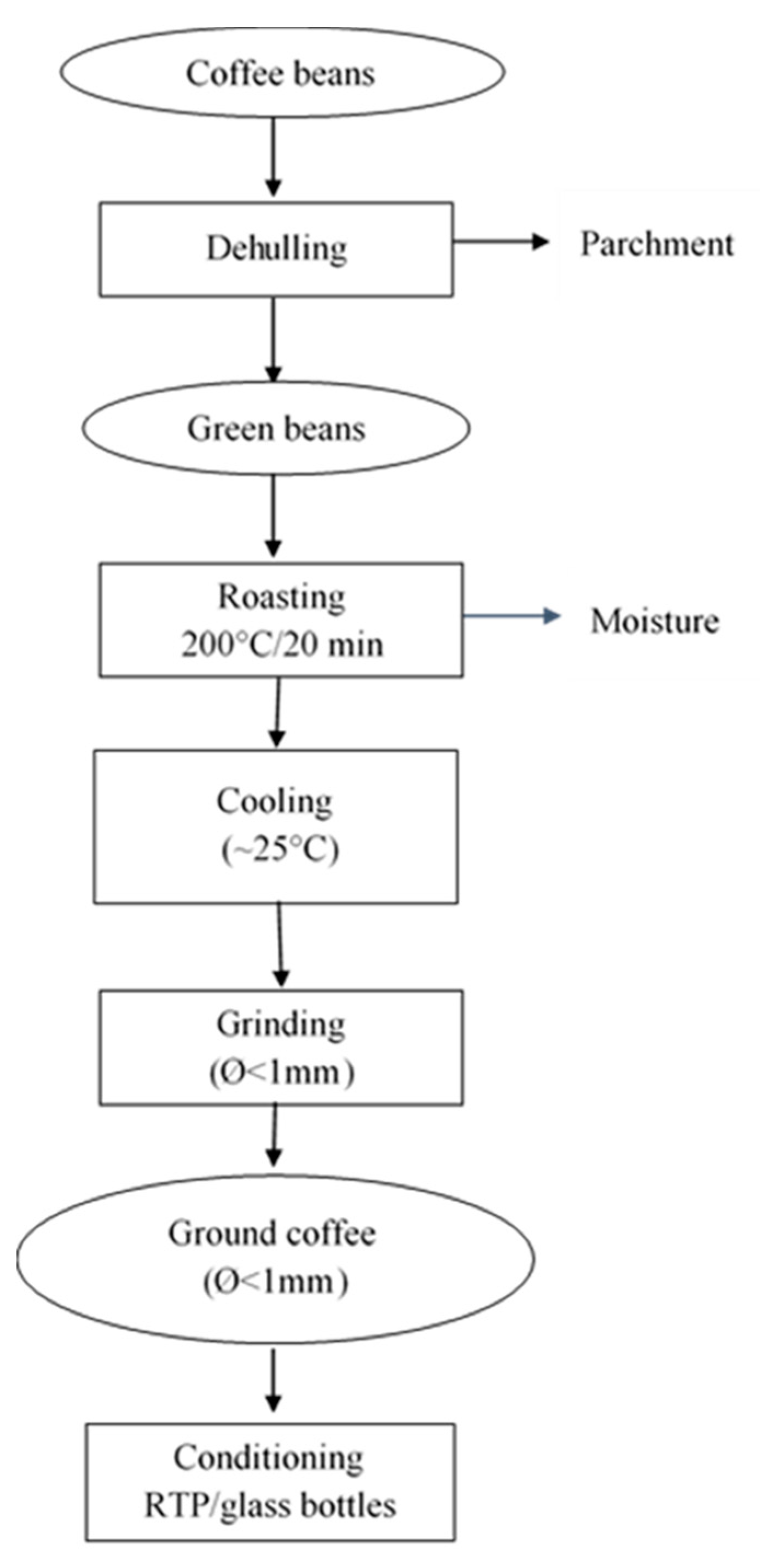
2.4. Processing of Coffee Beans
2.5. Preparation of Sorghum Grains
2.6. Analysis of Sorghum Grain
2.6.1. Determination of the Germinative Energy of Sorghum
2.6.2. Determination of the Germinative Capacity of Sorghum
2.6.3. Determination of the Thousand-Corn Weight of Sorghum
2.6.4. Determination of the Moisture Content
2.6.5. Determination of Total Ash
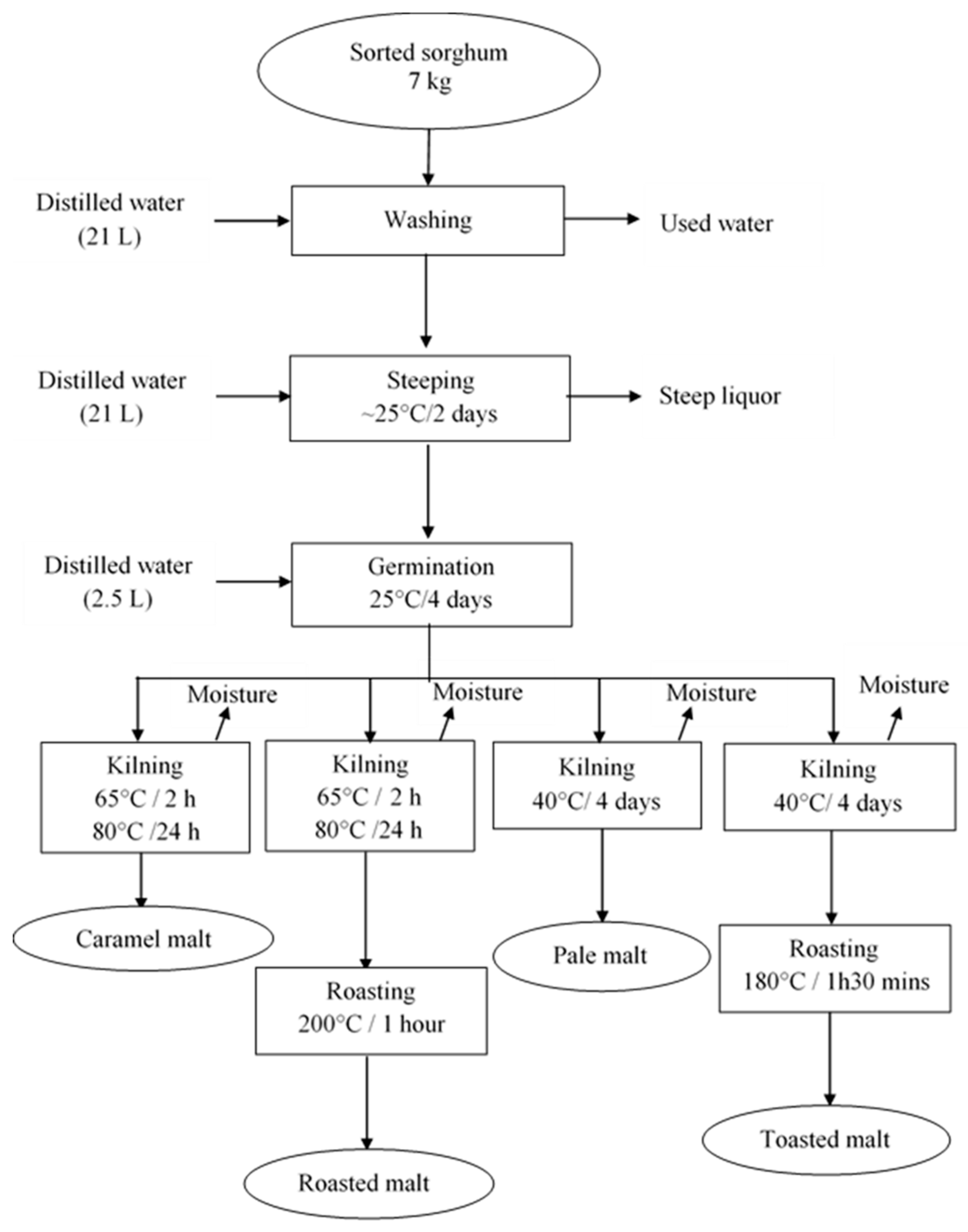
2.7. Experimental Procedure for Malting Sorghum
2.8. Determination of the Diastatic Power of Pale Malt
2.9. Experimental Procedure for Mashing Sorghum
2.10. Wort Boiling
2.11. Clarification and Fermentation
2.12. Physicochemical Analyses of Worts and Beers
2.12.1. Determination of Specific Gravity (Analytica-EBC, 1998)
2.12.2. Determination of Color Using Spectrophotometric Method
2.12.3. Determination of pH
2.12.4. Determination of Turbidity
2.12.5. Determination of Free Amino Nitrogen (FAN) Content
2.12.6. Determination of Titratable Acidity
2.12.7. Determination of Total Polyphenols
2.12.8. Alcohol Determination Using Specific Gravity
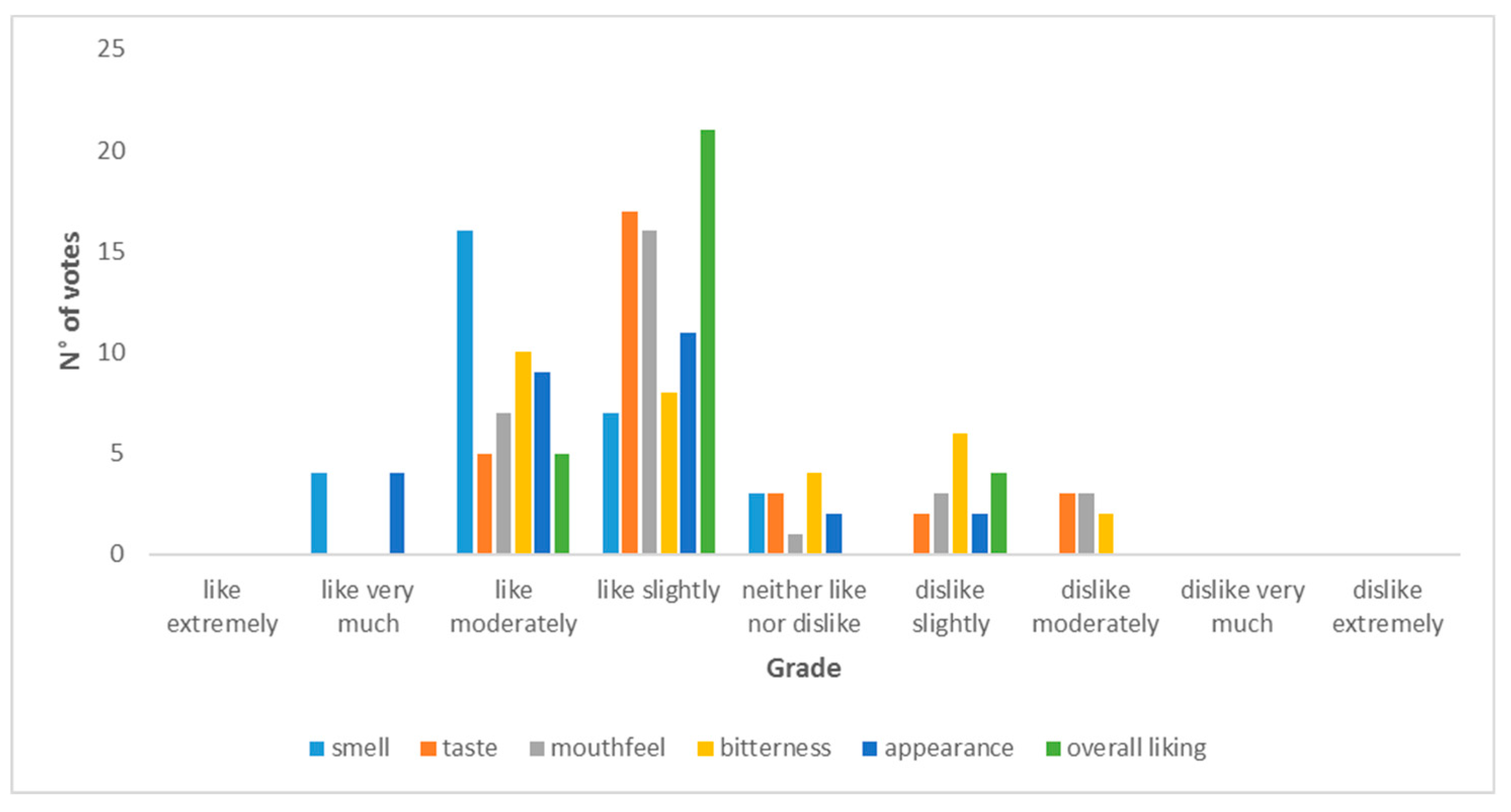
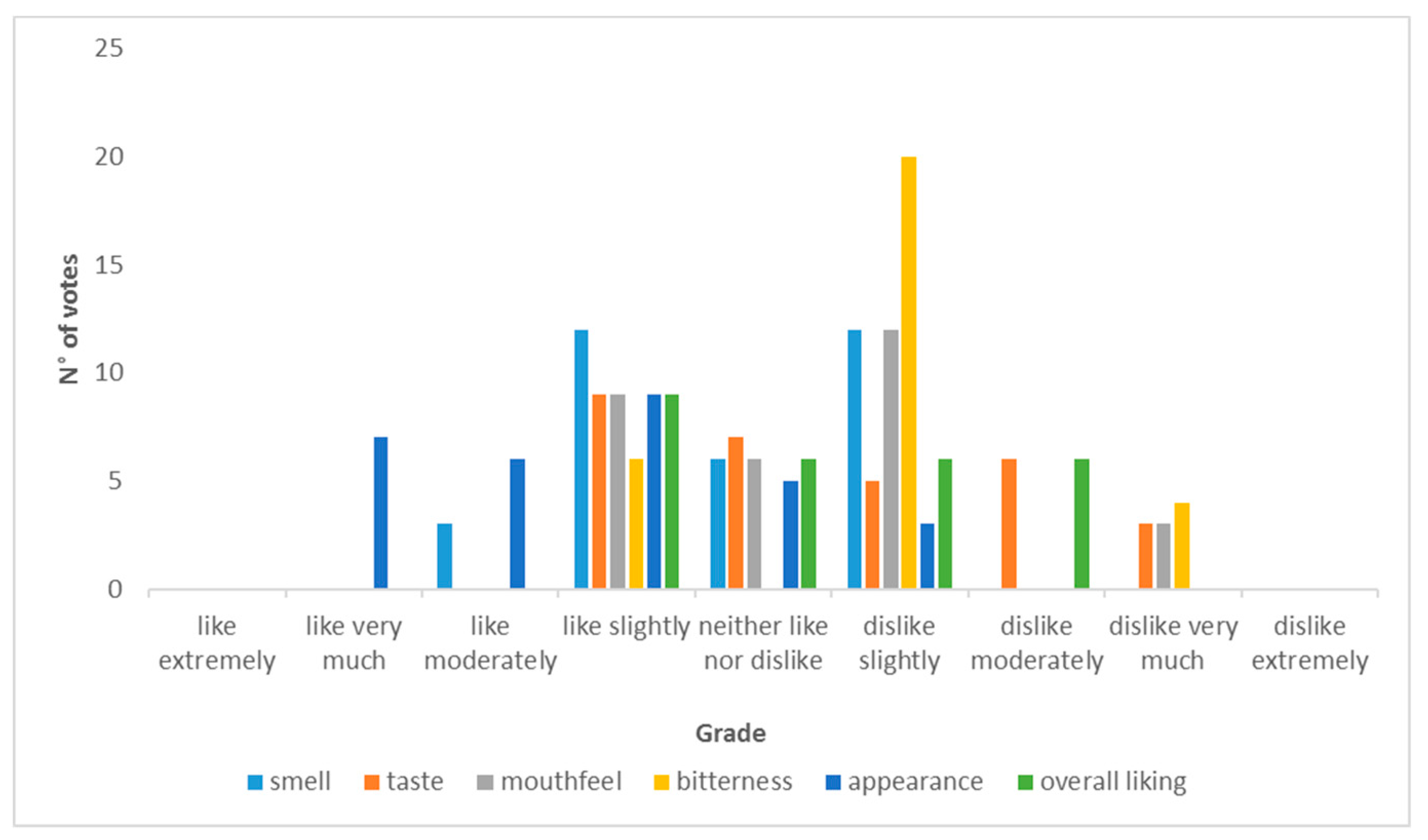
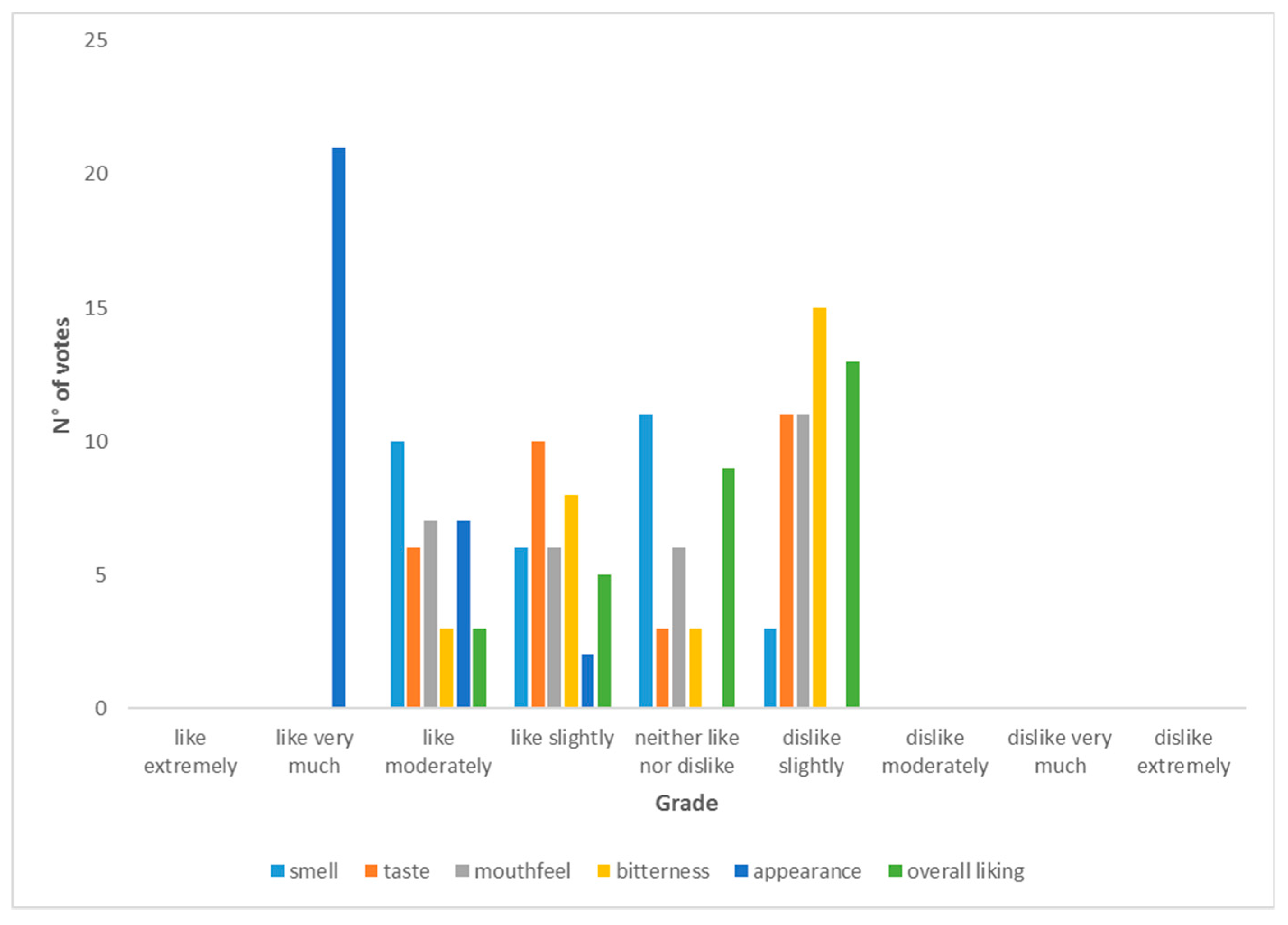
2.12.9. Sensory Evaluation of Beers
3. Results
3.1. Physicochemical Characteristics of Unmalted and Malted Sorghum
3.1.1. Water Content
3.1.2. Germinative Capacity and Energy
3.1.3. Thousand Corn Weight
3.1.4. Diastatic Power
3.1.5. Ash Content
3.2. Analysis of Worts and Beers
3.2.1. Specific Gravities
3.2.2. pH
3.2.3. Turbidity
3.2.4. Total Polyphenols
3.2.5. Free Amino Nitrogen (FAN)
3.2.6. Color
3.2.7. Titratable Acidity (TA)
3.2.8. Alcohol by Volume (ABV) Content of Beer
3.3. Sensory Evaluation Results
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Food and Agriculture Organization of the United Nations (FAO). Faostat Database Collections; Food and Agriculture Organization of the United Nations: Rome, Italy, 2014. [Google Scholar]
- Agu, R.C.; Palmer, G.H. Enzymic modification of endosperm of barley and sorghum of similar total nitrogen. Brew. Dig. 1998, 73, 30–35. [Google Scholar]
- Bajomo, M.F.; Young, T.W. The properties, composition and fermentabilities of worts made from 100% raw sorghum and commercial enzymes. J. Inst. Brew. 1993, 99, 153–158. [Google Scholar] [CrossRef]
- Bajomo, M.F.; Young, T.W. Fermentation of worts made form 100% raw sorghum and enzymes. J. Inst. Brew. 1994, 100, 79–84. [Google Scholar] [CrossRef]
- Taylor, J.R.N.; Robbins, D. Factors affecting beta-amylase activity in sorghum malt. J. Inst. Brew. 1993, 99, 413–416. [Google Scholar] [CrossRef]
- Nso, E.J.; Nanadoum, M.; Palmer, G.H. The effect of formaldehyde on enzyme development in sorghum malts. Tech. Q. Master Brew. Assoc. Am. 2006, 43, 177–182. [Google Scholar]
- Desobgo, Z.S.C.; Nso, E.J.; Tenin, D.; Kayem, G.J. Modelling and optimizing of mashing enzymes-effect on yield of filtrate of unmalted sorghum by use of response surface methodology. J. Inst. Brew. 2010, 116, 62–69. [Google Scholar] [CrossRef]
- Desobgo, Z.S.C. Modélisation de L’action des Hydrolases sur Quelques Caractéristiques Physico-Chimiques des Moûts de Deux Cultivars de Sorgho; Université de Ngaoundéré Ngaoundéré: Ngaoundéré, Cameroon, 2012. [Google Scholar]
- Bubacz, M.; Philip, T.; McCreanor, P.T.; Jenkins, H.E. Engineering of beer: Hard work or too much fun? In 2013 American Society for Engineering Education Southeast Conference; American Society for Engineering Education: Cookeville, TN, USA, 2013; pp. 1–10. [Google Scholar]
- Owuama, C.I. Brewing beer with sorghum. J. Inst. Brew. 1999, 105, 23–34. [Google Scholar] [CrossRef]
- Desobgo, Z.S.C.; Nso, E.J.; Tenin, D. The response surface methodology as a reliable tool for evaluating the need of commercial mashing enzymes for alleviating the levels of reducing sugars of worts of malted sorghum: Case of the Safrari cultivar. J. Brew. Distill. 2011, 2, 5–15. [Google Scholar]
- Desobgo, Z.S.C.; Nso, E.J.; Tenin, D. Use of response surface methodology for optimizing the action of mashing enzymes on wort reducing sugars of the madjeru sorghum cultivar. Af. J. Food Sci. 2011, 5, 91–99. [Google Scholar]
- Desobgo, Z.S.C.; Nso, E.J.; Tenin, D. Modeling the action of technical mashing enzymes on extracts and free-amino nitrogen yields of the madjeru sorghum cultivar. J. Brew. Distill. 2011, 2, 29–44. [Google Scholar]
- Desobgo, Z.S.C.; Nso, E.J.; Tenin, D. Optimisation of the action of commercial mashing enzymes on wort extracts and free amino nitrogen of the Safrari sorghum cultivar. Tech. Q. Master Brew. Assoc. Am. 2011, 48, 77–86. [Google Scholar]
- Nso, E.J.; Ajebesone, P.E.; Mbofung, C.M.; Palmer, G.H. Properties of three sorghum cultivars used for the production of bili-bili beverage in northern cameroon. J. Inst. Brew. 2003, 109, 245–250. [Google Scholar] [CrossRef]
- Wolgang, K. Technology Brewing and Malting, 5th ed.; VLB: Berlin, Germany, 2014; p. 960. [Google Scholar]
- Mackintosh, I.; Higgins, B. The development of a sorghum-based lager beer in uganda: A model of co-operating between industry and government in the development of local ingredients for the production of quality lager beer and consequential benefits for the parties involved. Asp. Appl. Biol. 2004, 72, 235–245. [Google Scholar]
- Taylor, J.R.N.; Schober, J.T.; Bean, S.R. Novel food and non-food uses for sorghum and millets. J. Cereal Sci. 2006, 44, 252–271. [Google Scholar] [CrossRef]
- Veith, K.N. Evaluation of four Sorghum Hybrids through the Development of Glutenfree Beer; Master of Science; Kansas State University: Manhattan, AR, USA, 2007. [Google Scholar]
- Ajebesone, P.E.; Aina, J.O. Potential african substitutes for hops in tropical beer brewing. J. Food Technol. Afr. 2004, 9, 13–16. [Google Scholar]
- Desobgo, Z.S.C.; Naponni, F.Y.; Nso, E.J. Characterization of Safrari sorghum worts and beers hopped with Vernonia amygdalina and nauclea diderrichii. Int. J. Innov. Appl. Stud. 2013, 2, 83–91. [Google Scholar]
- Okafor, N. Prospects and challenges of the use of local cereals and starchy subtrates in brewing. In National Symposium on the Brewing Industry, Now and the 21st Century; Owerri, Nigeria, 1985; pp. 22–25. [Google Scholar]
- Okafor, N.; Aniche, G.N. West african hop substitute for sorghum lager. Brew. Distill. Int. 1983, 13, 200–204. [Google Scholar]
- Owuama, C.I.; Okafor, N. Use of unmalted sorghum as a brewing adjunct. World J. Microbiol. Biotechnol. 1990, 6, 318–322. [Google Scholar] [CrossRef] [PubMed]
- Analytica-EBC. European Brewery Convention; Fachverlag Hans Carl: Nürnberg, Germany, 1998. [Google Scholar]
- AFNOR. Association française de normalisation. In Recueil des Normes Françaises des Produits Dérivés des Fruits et Légumes. Jus de Fruits, 1st ed.; AFNOR: Paris, France, 1982. [Google Scholar]
- Briggs, D.E. Malts and Malting, 1st ed.; Blackie Academic & Professional: London, UK, 1998. [Google Scholar]
- Marigo, G. Méthode de fractionnement et d’estimation des composés phénoliques chez les végétaux. Analysis 1973, 2, 106–110. [Google Scholar]
- Ogu, E.; Odibo, F.; Agu, R.C.; Palmer, G.H. Malting studies of some selected brewing sorghum varieties. MBAA TQ 2004, 41, 386–389. [Google Scholar]
- Okon, E.U.; Uwaifo, A.O. Evaluation of malting sorghum i: The malting potentials of nigerian varieties of sorghum. Brew. Dig. 1985, 60, 24–27. [Google Scholar]
- Agu, R.C.; Palmer, G.H. A reassessment of sorghum for lager-beer brewing. Bioresour. Technol. 1998, 66, 253–261. [Google Scholar] [CrossRef]
- Agu, R.C.; Palmer, G.H. Effect of mashing with commercial enzymes on the properties of sorghum worts. World J. Micro-Biol. Biotechnol. 1998, 14, 43–48. [Google Scholar] [CrossRef]
- Briggs, D.E.; Boulton, C.A.; Brookes, P.A.; Stevens, R. Brewing Science and Practice; Woodhead Publishing Limited: Cambridge, UK; CRC Press LLC: Boca Raton, FL, USA, 2004. [Google Scholar]
- Gupta, M.; Abu-Ghannam, N.; Gallaghar, E. Barley for brewing: Characteristic changes during malting, brewing and applications of its by-products. Compr. Rev. Food Sci. Food Saf. 2010, 9, 318–328. [Google Scholar] [CrossRef]
- Pontieri, P.; Di-Maro, A.; Tamburino, R.; De-Stefano, M.; Tilley, M.; Bean, S.R.; Roemer, E.; De-Vita, P.; Alifano, P.; Del-Giudice, L.; et al. Chemical composition of selected food-grade sorghum varieties grown under typical mediterranean conditions. Maydica 2010, 55, 139–143. [Google Scholar]
- Pontieri, P.; De-Vita, P.; Boffa, A.; Tuinstra, M.R.; Bean, S.R.; Krishnamoorthy, G.; Miller, C.; Roemer, E.; Alifano, P.; Pignone, D.; et al. Yield and morpho-agronomical evaluation of food-grade white sorghum hybrids grown in southern italy. J. Plant Interact. 2012, 7, 341–347. [Google Scholar] [CrossRef]
- Bamforth, C.W. Brewing: New Technologies; Woodhead Publishing: Cambridge, UK, 2006; p. 500. [Google Scholar]
- Bajomo, M.F.; Young, T.W. Development of a mashing profile for the use of microbial enzymes in brewing with raw sorghum (80%) and malted barley or sorghum malt (20%). J. Inst. Brew. 1992, 98, 515–523. [Google Scholar] [CrossRef]
- Walstra, P.; Wouters, J.; Geurts, T. Dairy Science and Technology; CRC Press: Boca Raton, FL, USA, 2006. [Google Scholar]
- Briggs, D.E.; Hough, J.S. Malting and Brewing Science, 2nd ed.; Chapman and Hall: London, UK, 1981. [Google Scholar]
- Fox, P.F.; Uniacke-Lowe, T.; McSweeney, P.L.H.; O’Mahony, J.A. Dairy Chemistry and Biochemistry, 2nd ed.; Springer International Publishing: Basel, Switzerland, 2015; p. 584. [Google Scholar]
- Wang, Y.; Ho, C.-T. Polyphenolic chemistry of tea and coffee: A century of progress. J. Agric. Food Chem. 2009, 57, 8109–8114. [Google Scholar] [CrossRef] [PubMed]
- Kondo, K.; Kurihara, M.; Miyata, N.; Suzuki, T.; Toyoda, M. Scavenging mechanisms of (-)-epigallocatechin gallate and (-)-epicatechin gallate on peroxyl radicals and formation of superoxide during the inhibitory action. Free Radic. Biol. Med. 1999, 27, 855–863. [Google Scholar] [CrossRef]
- Liu, Z.; Ma, L.P.; Zhou, B.; Yang, L.; Liu, Z.L. Antioxidative effects of green tea polyphenols on free radical initiated and photosensitized peroxidation of human low density lipoprotein. Chem. Phys. Lipids 2000, 106, 53–63. [Google Scholar] [CrossRef]
- Yilmaz, Y.; Toledo, R.T. Major flavonoids in grape seeds and skins: Antioxidant capacity of catechin, epicatechin, and gallic acid. J. Agric. Food Chem. 2004, 52, 255–260. [Google Scholar] [CrossRef] [PubMed]
- Beckerich, R.P.; Denault, L.J. Enzymes in the preparation of beer and fuel alcohol. In Enzymes and Their Role in Cereal Technology; Kruger, J.E., Lineback, D., Stauffer, C.E., Eds.; American Association of Cereal Chemists: St. Paul, MN, USA, 1987; pp. 335–354. [Google Scholar]
- Lekkas, C.; Hill, A.; Taidi, B.; Hodgson, J.; Stewart, G. The role of small wort peptides in brewing fermentations. J. Inst. Brew. 2009, 115, 134–139. [Google Scholar] [CrossRef]
- Lekkas, C.; Stewart, G.; Hill, A.; Taidi, B.; Hodgson, J. The importance of free amino nitrogen in wort and beer. Tech. Q.-Master Brew. Assoc. Am. 2005, 42, 113. [Google Scholar]
- Shellhammer, T.H. Beer color. In Beer: A Quality Perspective; Bamforth, C.W., Russell, I., Stewart, G.G., Eds.; Academic Press: London, UK, 2008; pp. 221–226. [Google Scholar]
- Bamforth, C.W. Beer, carbohydrates and diet. J. Inst. Brew. 2005, 111, 259–264. [Google Scholar] [CrossRef]
- Bamforth, C.W. Chemistry of Brewing; Elsevier Science Ltd.: Amsterdam, The Netherlands, 2003. [Google Scholar]



| Commercial Mashing Enzyme | ||
|---|---|---|
| Hitempase 2XL | Bioglucanase TX | |
| Organism of origin | Bacillus licheniformis | Trichoderma reesei |
| Activity | 4416.29 ± 19.34 U/mL | 750 BGU/mL |
| Description | α-amylase | β-glucanase |
| Optimum temperature (°C) | 60–95 | 60 |
| Optimum pH | 4–8 | 4.5–6.5 |
| Recommended application level in adjuncts | 60 U/g | 0.01 and 0.025% (v/w) |
| Form | Solution | Solution |
| Sorghum Malt Type | Quantity/kg |
|---|---|
| Pale malt | 3.1 |
| Caramel malt | 1.2 |
| Toasted malt | 0.3 |
| Roasted malt | 0.4 |
| Std | Run | Block | Proportion | Quantity (g) | ||
|---|---|---|---|---|---|---|
| : Lactose | : Coffee | : Lactose | : Coffee | |||
| 4 | 1 | Block 1 | 1.000 | 0.000 | 500 | 0 |
| 1 | 2 | Block 1 | 0.750 | 0.250 | 375 | 125 |
| 3 | 3 | Block 1 | 0.500 | 0.500 | 250 | 250 |
| 2 | 4 | Block 1 | 0.250 | 0.750 | 125 | 375 |
| 5 | 5 | Block 1 | 0.000 | 1.000 | 0 | 500 |
| Characteristics | Unmalted Safrari | Safrari Malt |
|---|---|---|
| Water content (%) | 8.50 ± 0.01 | 4.80 ± 0.56 |
| Germinative capacity (%) | 99.30 ± 0.58 | N.D. |
| Germinative energy (4 mL) (%) | 98.60 ± 1.79 | N.D. |
| Germinative energy (8 mL) (%) | 97.00 ± 1.21 | N.D. |
| Thousand corn weight (g) | 48.10 ± 0.02 | 38.00 ± 1.35 |
| Diastatic power (WK) | N.D. | 187.40 ± 7.89 |
| Total ash (%) | 1.30 ± 0.10 | 0.90 ± 0.22 |
| Factors | Responses | |||||||
|---|---|---|---|---|---|---|---|---|
| X1 | X2 | SG | pH | Turbidity (NTU) | TP (mgGAE/L) | FAN (mg/L) | Colour (EBC) | TA (g/L H2T) |
| 1.000 | 0.000 | 1.0748 | 5.79 ± 0.01 | 606.0 ± 0.8 | 416.0 ± 0.1 | 308.0 ± 0.2 | 186.40 ± 0.12 | 2.58 ± 0.04 |
| 0.750 | 0.250 | 1.0728 | 5.73 ± 0.00 | 375.0 ± 1.2 | 487.0 ± 0.1 | 313.0 ± 0.2 | 242.50 ± 2.04 | 2.78 ± 0.10 |
| 0.500 | 0.500 | 1.0704 | 5.68 ± 0.01 | 281.0 ± 0.5 | 615.0 ± 0.1 | 317.0 ± 0.5 | 192.50 ± 0.20 | 2.84 ± 0.01 |
| 0.250 | 0.750 | 1.0632 | 5.63 ± 0.00 | 149.0 ± 0.5 | 441.0 ± 0.1 | 339.0 ± 0.9 | 146.70 ± 3.00 | 2.83 ± 0.00 |
| 0.000 | 1.000 | 1.0592 | 5.41 ± 0.01 | 91.0 ± 0.0 | 442.0 ± 0.0 | 286.0 ± 0.2 | 151.30 ± 0.12 | 2.84 ± 0.00 |
| Factors | Responses | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| X1 | X2 | SG | pH | Turbidity (NTU) | TP (mg GAE/L) | FAN (mg/L) | Colour (EBC) | TA (g/L H2T) | ABV (%) |
| 1.000 | 0.000 | 1.0040 | 4.64 ± 0.00 | 120.0 ± 0.5 | 108.0 ± 0.6 | 140.0 ± 0.1 | 155.00 ± 0.00 | 4.38 ± 0.02 | 9.30 ± 0.00 |
| 0.750 | 0.250 | 1.0027 | 4.62±0.01 | 299.0 ± 0.5 | 127.0 ± 0.7 | 131.0 ± 0.4 | 189.70 ± 0.20 | 4.37 ± 0.02 | 9.20 ± 0.09 |
| 0.500 | 0.500 | 0.9987 | 4.68 ± 0.03 | 141.0 ± 0.0 | 135.0 ± 0.4 | 120.0 ± 0.5 | 183.90 ± 0.24 | 4.18 ± 0.04 | 9.40 ± 0.01 |
| 0.250 | 0.750 | 0.9960 | 4.57 ± 0.00 | 11.0 ± 0.1 | 139.0 ± 0.7 | 112.0 ± 0.1 | 109.20 ± 1.18 | 3.97 ± 0.05 | 8.80 ± 0.02 |
| 0.000 | 1.000 | 0.9923 | 4.47 ± 0.02 | 6.0 ± 0.0 | 146.0 ± 0.6 | 110.0 ± 0.2 | 112.50 ± 0.00 | 3.92 ± 0.08 | 8.80 ± 0.02 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mbeh. Harry, F.; Zangué Steve Carly, D.; Emmanuel Jong, N. Sorghum Coffee–Lactose Stout Production and Its Physico-Chemical Characterization. Beverages 2019, 5, 20. https://doi.org/10.3390/beverages5010020
Mbeh. Harry F, Zangué Steve Carly D, Emmanuel Jong N. Sorghum Coffee–Lactose Stout Production and Its Physico-Chemical Characterization. Beverages. 2019; 5(1):20. https://doi.org/10.3390/beverages5010020
Chicago/Turabian StyleMbeh. Harry, Fali, Desobgo Zangué Steve Carly, and Nso Emmanuel Jong. 2019. "Sorghum Coffee–Lactose Stout Production and Its Physico-Chemical Characterization" Beverages 5, no. 1: 20. https://doi.org/10.3390/beverages5010020
APA StyleMbeh. Harry, F., Zangué Steve Carly, D., & Emmanuel Jong, N. (2019). Sorghum Coffee–Lactose Stout Production and Its Physico-Chemical Characterization. Beverages, 5(1), 20. https://doi.org/10.3390/beverages5010020






