Hydrothermal Carbonization of Brewery’s Spent Grains for the Production of Solid Biofuels
Abstract
1. Introduction
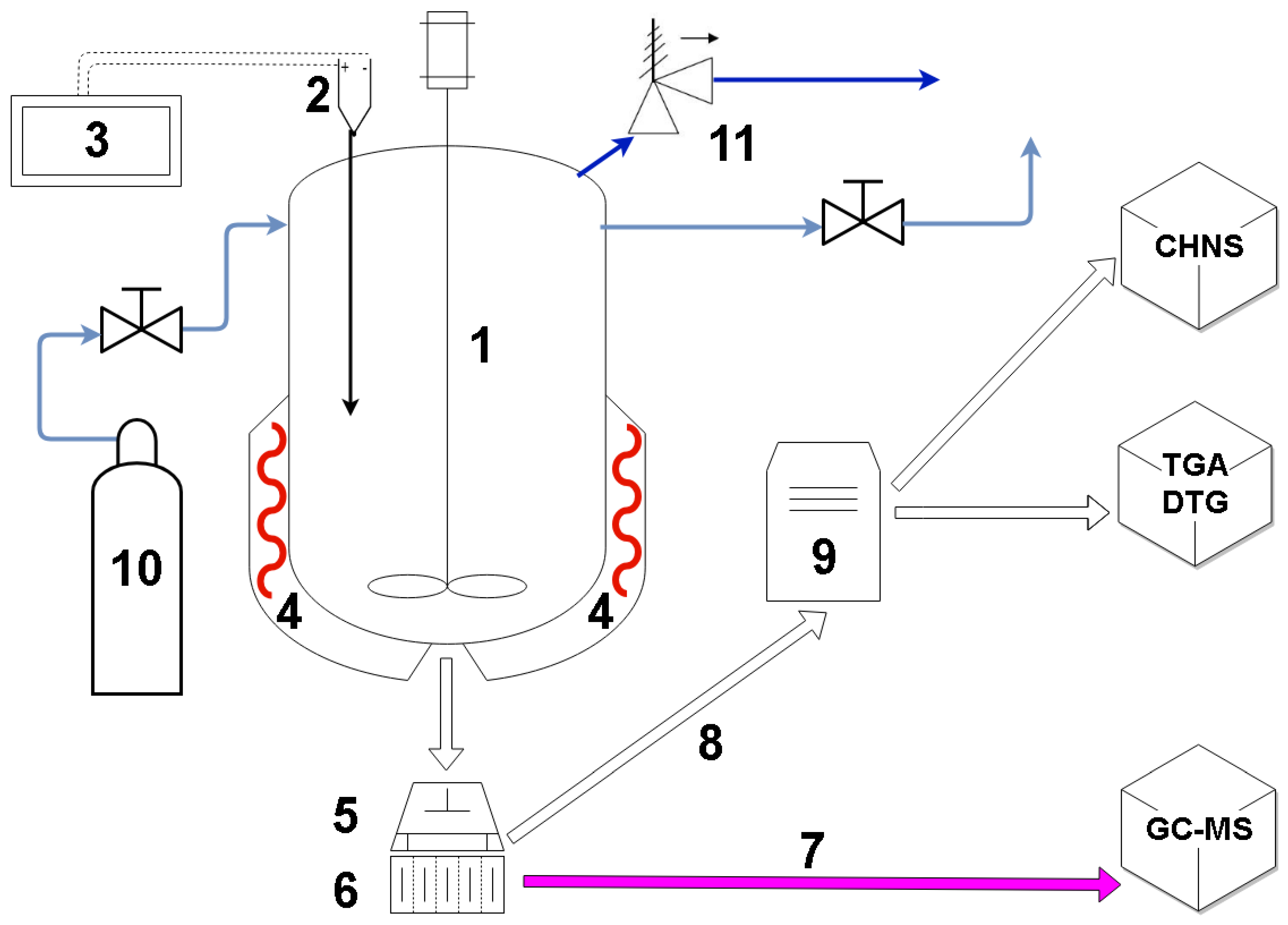
2. Materials and Methods
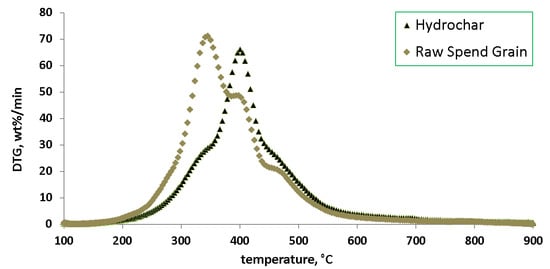
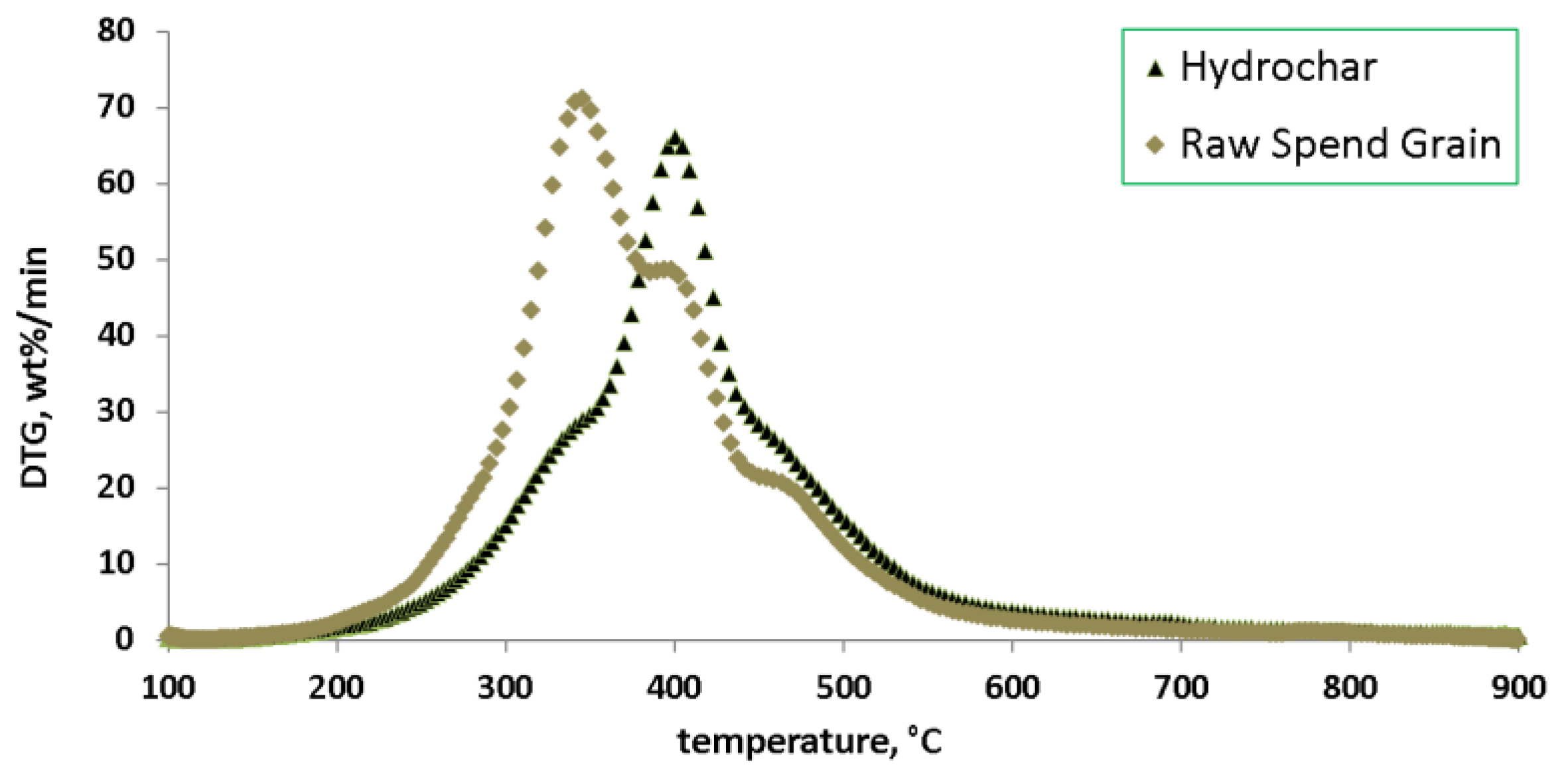
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Wiśniewski, P. Piwa Historie Niezwykłe; Print Shops PREGO: Warszawa, Poland, 1993; ISBN 83-85830-00-6. [Google Scholar]
- Valamoti, S.M. Brewing beer in wine country? First archaeobotanical indications for beer making in Early and Middle Bronze Age Greece. Veg. Hist. Archaeobot. 2018, 27, 611–625. [Google Scholar] [CrossRef]
- Beer Production Worldwide from 1998 to 2017 (in Billion Hectoliters). Available online: https://www.statista.com/statistics/270275/worldwide-beer-production/ (accessed on 21 December 2018).
- Bentzen, J.; Smith, V. Structural Changes in the Consumption of Beer, Wine and Spirits in OECD Countries from 1961 to 2014. Beverages 2018, 4, 8. [Google Scholar] [CrossRef]
- Lynch, K.M.; Steffen, E.J.; Arendt, E.K. Brewers’ spent grain: A review with an emphasis on food and health. J. Inst. Brew. 2016, 122, 553–568. [Google Scholar] [CrossRef]
- Stojceska, V.; Ainsworth, P. The effect of different enzymes on the quality of high-fibre enriched brewer’s spent grain breads. Food Chem. 2008, 110, 865–872. [Google Scholar] [CrossRef] [PubMed]
- Kreisz, S. Malting. In Handbook of Brewing Processes, Technology, Markets; Eßlinger, H.M., Ed.; WILEY-VCH: Weinheim, Germany, 2009; pp. 147–164. ISBN 978-3-527-31674-8. [Google Scholar]
- Dylkowski, W. Technologia Browarnictwa; Wydawnictwo przemysłu lekkiego i spożywczego: Warszawa, Poland, 1963. [Google Scholar]
- Sperandio, G.; Amoriello, T.; Carbone, K.; Fedrizzi, M.; Monteleone, A.; Tarangioli, S.; Pagano, M. Increasing the value of spent grain from craft microbreweries for energy purposes. Chem. Eng. Trans. 2017, 58, 487–492. [Google Scholar] [CrossRef]
- Enweremadu, C.C.; Waheed, M.A.; Adekunle, A.A.; Adeala, A. The Energy Potential of Brewer’s Spent Grain for Breweries in Nigeria. Eng. Appl. Sci. 2008, 3, 175–177. [Google Scholar]
- Funke, A.; Ziegler, F. Hydrothermal carbonisation of biomass: A summary and discussion of chemical mechanisms for process engineering. Biofuels Bioprod. Biorefin. 2010, 4, 160–177. [Google Scholar] [CrossRef]
- Moscicki, K.J.; Niedzwiecki, L.; Owczarek, P.; Wnukowski, M. Commoditization of wet and high ash biomass: Wet torrefaction—A review. J. Power Technol. 2017, 97, 354–369. [Google Scholar]
- Reza, M.T.; Lynam, J.G.; Uddin, M.H.; Coronella, C.J. Hydrothermal carbonization: Fate of inorganics. Biomass Bioenergy 2013, 49, 86–94. [Google Scholar] [CrossRef]
- Reza, M.T.; Andert, J.; Wirth, B.; Busch, D.; Pielert, J.; Lynam, J.G.; Mumme, J. Hydrothermal Carbonization of Biomass for Energy and Crop Production. Appl. Bioenergy 2014, 1, 11–29. [Google Scholar] [CrossRef]
- Reza, M.T.; Yan, W.; Uddin, M.H.; Lynam, J.G.; Hoekman, S.K.; Coronella, C.J.; Vásquez, V.R. Reaction kinetics of hydrothermal carbonization of loblolly pine. Bioresour. Technol. 2013, 139, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Tungal, R.; Shende, R.V. Hydrothermal liquefaction of pinewood (Pinus ponderosa) for H2, biocrude and bio-oil generation. Appl. Energy 2014, 134, 401–412. [Google Scholar] [CrossRef]
- Nan, W.; Shende, A.R.; Shannon, J.; Shende, R.V. Insight into Catalytic Hydrothermal Liquefaction of Cardboard for Biofuels Production. Energy Fuels 2016, 30, 4933–4944. [Google Scholar] [CrossRef]
- Shende, R.; Tungal, R. Subcritical Aqueous Phase Reforming of Wastepaper for Biocrude and H2 Generation. Energy Fuels 2013, 27, 3194–3203. [Google Scholar] [CrossRef]
- Funke, A.; Ziegler, F. Heat of reaction measurements for hydrothermal carbonization of biomass. Bioresour. Technol. 2011, 102, 7595–7598. [Google Scholar] [CrossRef] [PubMed]
- Acharjee, T.C.; Coronella, C.J.; Vasquez, V.R. Effect of thermal pretreatment on equilibrium moisture content of lignocellulosic biomass. Bioresour. Technol. 2011, 102, 4849–4854. [Google Scholar] [CrossRef] [PubMed]
- Favas, G.; Jackson, W.R.; Marshall, M. Hydrothermal dewatering of lower rank coals. 3. High-concentration slurries from hydrothermally treated lower rank coals. Fuel 2003, 82, 71–79. [Google Scholar] [CrossRef]
- Nakagawa, H.; Namba, A.; Böhlmann, M.; Miura, K. Hydrothermal dewatering of brown coal and catalytic hydrothermal gasification of the organic compounds dissolving in the water using a novel Ni/carbon catalyst. Fuel 2004, 83, 719–725. [Google Scholar] [CrossRef]
- Favas, G.; Jackson, W.R. Hydrothermal dewatering of lower rank coals. 1: Effects of process conditions on the properties of dried product. Fuel 2003, 82, 53–57. [Google Scholar] [CrossRef]
- Favas, G.; Jackson, W.R. Hydrothermal dewatering of lower rank coals. 2. Effects of coal characteristics for a range of Australian and international coals. Fuel 2003, 82, 59–69. [Google Scholar] [CrossRef]
- Rao, Z.; Zhao, Y.; Huang, C.; Duan, C.; He, J. Recent developments in drying and dewatering for low rank coals. Prog. Energy Combust. Sci. 2015, 46, 1–11. [Google Scholar] [CrossRef]
- Mursito, A.T.; Hirajima, T.; Sasaki, K. Upgrading and dewatering of raw tropical peat by hydrothermal treatment. Fuel 2010, 89, 635–641. [Google Scholar] [CrossRef]
- Wnukowski, M.; Owczarek, P.; Niedźwiecki, Ł. Wet Torrefaction of Miscanthus – Characterization of Hydrochars in View of Handling, Storage and Combustion Properties. J. Ecol. Eng. 2015, 16, 161–167. [Google Scholar] [CrossRef]
- Liu, Z.; Quek, A.; Balasubramanian, R. Preparation and characterization of fuel pellets from woody biomass, agro-residues and their corresponding hydrochars. Appl. Energy 2014, 113, 1315–1322. [Google Scholar] [CrossRef]
- Volpe, M.; Wüst, D.; Merzari, F.; Lucian, M.; Andreottola, G.; Kruse, A.; Fiori, L. One stage olive mill waste streams valorisation via hydrothermal carbonisation. Waste Manag. 2018, 80, 224–234. [Google Scholar] [CrossRef] [PubMed]
- Koppejan, J.; Sokhansanj, S.; Melin, S.; Madrali, S. Status Overview of Torrefaction Technologies; Bioenergy Task 32 report; International Energy Agency: Paris, France, 2015; ISBN 978-1-910154-23-6. [Google Scholar]
- Ruiz, H.A.; Rodríguez-Jasso, R.M.; Fernandes, B.D.; Vicente, A.A.; Teixeira, J.A. Hydrothermal processing, as an alternative for upgrading agriculture residues and marine biomass according to the biorefinery concept: A review. Renew. Sustain. Energy Rev. 2013, 21, 35–51. [Google Scholar] [CrossRef]
- Svensson, K.; Kjørlaug, O.; Higgins, M.J.; Linjordet, R.; Horn, S.J. Post-anaerobic digestion thermal hydrolysis of sewage sludge and food waste: Effect on methane yields, dewaterability and solids reduction. Water Res. 2018, 132, 158–166. [Google Scholar] [CrossRef]
- Erdogan, E.; Atila, B.; Mumme, J.; Reza, M.T.; Toptas, A.; Elibol, M.; Yanik, J. Characterization of products from hydrothermal carbonization of orange pomace including anaerobic digestibility of process liquor. Bioresour. Technol. 2015, 196, 35–42. [Google Scholar] [CrossRef]
- Funke, A.; Mumme, J.; Koon, M.; Diakité, M. Cascaded production of biogas and hydrochar from wheat straw: Energetic potential and recovery of carbon and plant nutrients. Biomass Bioenergy 2013, 58, 229–237. [Google Scholar] [CrossRef]
- Wirth, B.; Mumme, J. Anaerobic Digestion of Waste Water from Hydrothermal Carbonization of Corn Silage. Appl. Bioenergy 2014, 1, 1–10. [Google Scholar] [CrossRef]
- Luz, F.C.; Cordiner, S.; Manni, A.; Mulone, V.; Rocco, V. Anaerobic digestion of coffee grounds soluble fraction at laboratory scale: Evaluation of the biomethane potential. Appl. Energy 2017, 207, 166–175. [Google Scholar] [CrossRef]
- Fagbohungbe, M.O.; Herbert, B.M.J.; Hurst, L.; Ibeto, C.N.; Li, H.; Usmani, S.Q.; Semple, K.T. The challenges of anaerobic digestion and the role of biochar in optimizing anaerobic digestion. Waste Manag. 2017, 61, 236–249. [Google Scholar] [CrossRef] [PubMed]
- Codignole Luz, F.; Cordiner, S.; Manni, A.; Mulone, V.; Rocco, V. Biochar characteristics and early applications in anaerobic digestion—A review. J. Environ. Chem. Eng. 2018, 6, 2892–2909. [Google Scholar] [CrossRef]
- Zhou, Y.; Engler, N.; Nelles, M. Symbiotic relationship between hydrothermal carbonization technology and anaerobic digestion for food waste in China. Bioresour. Technol. 2018, 260, 404–412. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Mustafa, A.M.; Lin, H.; Choe, U.Y.; Sheng, K. Effect of hydrochar on anaerobic digestion of dead pig carcass after hydrothermal pretreatment. Waste Manag. 2018, 78, 849–856. [Google Scholar] [CrossRef]
- Codignole Luz, F.; Volpe, M.; Fiori, L.; Manni, A.; Cordiner, S.; Mulone, V.; Rocco, V. Spent coffee enhanced biomethane potential via an integrated hydrothermal carbonization-anaerobic digestion process. Bioresour. Technol. 2018, 256, 102–109. [Google Scholar] [CrossRef]
- Wilk, M.; Magdziarz, A. Hydrothermal carbonization, torrefaction and slow pyrolysis of Miscanthus giganteus. Energy 2017, 140, 1292–1304. [Google Scholar] [CrossRef]
- Gao, L.; Volpe, M.; Lucian, M.; Fiori, L.; Goldfarb, J.L. Does Hydrothermal Carbonization as a Biomass Pretreatment Reduce Fuel Segregatuin of Coal-Biomass Blends During Oxidation? Energy Convers. Manag. 2018, 93–104. [Google Scholar] [CrossRef]
- Pearce, I.; Culbert, J.; Cass, D.; Cozzolino, D.; Wilkinson, K. Influence of Sample Storage on the Composition of Carbonated Beverages by MIR Spectroscopy. Beverages 2016, 2, 26. [Google Scholar] [CrossRef]
- Yan, W.; Hastings, J.T.; Acharjee, T.C.; Coronella, C.J.; Vásquez, V.R. Mass and energy balances of wet torrefaction of lignocellulosic biomass. Energy Fuels 2010, 24, 4738–4742. [Google Scholar] [CrossRef]
- Phyliss2 Database. Available online: https://phyllis.nl/Home/Help (accessed on 24 December 2018).
- Pawlak-Kruczek, H.; Krochmalny, K.; Mościcki, K.; Zgóra, J.; Czerep, M.; Ostrycharczyk, M.; Niedźwiecki, Ł. Torrefaction of Various Types of Biomass in Laboratory Scale, Batch-Wise Isothermal Rotary Reactor and Pilot Scale, Continuous Multi-Stage Tape Reactor. Eng. Prot. Environ. 2017, 20, 457–472. [Google Scholar] [CrossRef]
- Weber, K.; Heuer, S.; Quicker, P.; Li, T.; Løvås, T.; Scherer, V. An Alternative Approach for the Estimation of Biochar Yields. Energy Fuels 2018. [Google Scholar] [CrossRef]
- Moscicki, K.J.; Niedzwiecki, L.; Owczarek, P.; Wnukowski, M. Commoditization of biomass: Dry torrefaction and pelletization—A review. J. Power Technol. 2014, 94, 233–249. [Google Scholar]
- Pawlak-Kruczek, H.; Krochmalny, K.K.; Wnukowski, M.; Niedzwiecki, L. Slow pyrolysis of the sewage sludge with additives: Calcium oxide and lignite. J. Energy Resour. Technol. 2018, 140. [Google Scholar] [CrossRef]
- Poudel, J.; Karki, S.; Gu, J.H.; Lim, Y.; Oh, S.C. Effect of Co-Torrefaction on the Properties of Sewage Sludge and Waste Wood to Enhance Solid Fuel Qualities. J. Residuals Sci. Technol. 2017, 14, 23–36. [Google Scholar] [CrossRef]
- Pulka, J.; Wiśniewski, D.; Gołaszewski, J.; Białowiec, A. Is the biochar produced from sewage sludge a good quality solid fuel? Arch. Environ. Prot. 2016, 42, 125–134. [Google Scholar] [CrossRef]
- Royal Society of Chemistry ChemSpider | Search and Share Chemistry. Available online: http://www.chemspider.com/ (accessed on 23 November 2018).
- Bridgwater, A.V. Review of fast pyrolysis of biomass and product upgrading. Biomass Bioenergy 2012, 38, 68–94. [Google Scholar] [CrossRef]
- Louwes, A.C.; Basile, L.; Yukananto, R.; Bhagwandas, J.C.; Bramer, E.A.; Brem, G. Torrefied biomass as feed for fast pyrolysis: An experimental study and chain analysis. Biomass Bioenergy 2017, 105, 116–126. [Google Scholar] [CrossRef]
- Prins, M.J.; Ptasinski, K.J.; Janssen, F.J.J.G. More efficient biomass gasification via torrefaction. Energy 2006, 31, 3458–3470. [Google Scholar] [CrossRef]
- Thompson Witrick, K.; Duncan, S.; Hurley, K.; O’Keefe, S. Acid and Volatiles of Commercially-Available Lambic Beers. Beverages 2017, 3, 51. [Google Scholar] [CrossRef]
- Stemann, J.; Putschew, A.; Ziegler, F. Hydrothermal carbonization: Process water characterization and effects of water recirculation. Bioresour. Technol. 2013, 143, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Reza, M.T.; Rottler, E.; Herklotz, L.; Wirth, B. Hydrothermal carbonization (HTC) of wheat straw: Influence of feedwater pH prepared by acetic acid and potassium hydroxide. Bioresour. Technol. 2015, 182, 336–344. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Bhalla, A.; Shende, R.V.; Sani, R.K. Decentralized thermophilic biohydrogen: A more efficient and cost-effective process. BioResources 2012, 7, 1–2. [Google Scholar]
- Kumar, S.; Gupta, R.B. Biocrude production from switchgrass using subcritical water. Energy Fuels 2009, 23, 5151–5159. [Google Scholar] [CrossRef]
- Braida, D.; Capurro, V.; Zani, A.; Rubino, T.; Viganò, D.; Parolaro, D.; Sala, M. Potential anxiolytic- And antidepressant-like effects of salvinorin A, the main active ingredient of Salvia divinorum, in rodents. Br. J. Pharmacol. 2009, 157, 844–853. [Google Scholar] [CrossRef] [PubMed]
- Hanes, K.R. Antidepressant effects of the herb Salvia divinorum: A case report. J. Clin. Psychopharmacol. 2001, 21, 634–635. [Google Scholar] [CrossRef] [PubMed]
- Hanes, K. Salvia divinorum: Clinical and Research Potential. Maps 2003, XIII, 18–20. [Google Scholar]
| Test | Symbol | Raw | Raw 4 (Data from [10]) | After HTC | Unit | Standard Procedure |
|---|---|---|---|---|---|---|
| Moisture content 1 | MC | 77.27 | - | 64.15 | % | EN ISO 18134-2:2015 |
| Volatile matter content | VM d | 71.20 | - | 64.42 | % | EN 15148:2009 |
| Ash content | A d | 4.30 | 4.46 | 1.91 | % | EN ISO 1822:2015 |
| Higher heating value 2 | HHV | 20,628 | 19,515 | 26,455 | kJ/kg | EN 14918:2009 |
| Lower heating value 3 | LHV | 2412 | - | 7281 | kJ/kg | EN 14918:2009 |
| Carbon content | C d | 47.18 | 48.36 | 58.57 | % | EN ISO 16948:2015 |
| Hydrogen content | H d | 8.20 | 6.02 | 8.29 | % | EN ISO 16948:2015 |
| Nitrogen content | N d | 3.32 | 4.11 | 3.75 | % | EN ISO 16948:2015 |
| Sulfur content | S d | 0.26 | 0.32 | 0.24 | % | EN ISO 16994:2016 |
| Oxygen content | O d | 41.04 | 36.73 | 29.15 | % | EN ISO 16993:2015 |
| Oxygen to carbon ratio | O/C | 0.65 | - | 0.37 | mol/mol | - |
| Hydrogen to carbon ratio | H/C | 2.07 | - | 1.68 | mol/mol | - |
| Oxygen to hydrogen ratio | O/H | 0.32 | - | 0.22 | mol/mol | - |
| Parameter | Symbol | Value | Unit |
|---|---|---|---|
| Temperature | THTC | 200 | °C |
| Residence time | tHTC | 150 | min |
| Water: biomass ratio | W:B | 10:1 | - |
| Mass yield | Ym | 0.809 | - |
| Energy yield | Ye | ~1 | - |
| Ash yield | Ya | 0.360 | - |
| Sample | Moisture Content 1 | Unit |
|---|---|---|
| Raw spent grain | 77.52 | % |
| Spent grain after HTC and dripping | 84.48 | % |
| Carbonized spent grain after mechanical dewatering | 64.15 | % |
| Compound | Form | Avg. Mass | Relative Area of the Peak 1 | |
|---|---|---|---|---|
| Relative Area | SD 2 | |||
| IUPAC name | Da | % | % | |
| Methanol | CH4O | 32.042 | 0.39 | 0.02 |
| Formic acid | CH2O2 | 46.025 | 0.60 | 0.11 |
| Ethanol | C2H6O | 46.068 | 2.73 | 0.12 |
| Acetone | C3H6O | 58.079 | 1.30 | 0.53 |
| Acetic acid | C2H4O2 | 60.052 | 16.00 | 0.24 |
| 1-Hydroxyacetone | C3H6O2 | 74.078 | 4.99 | 0.04 |
| 1,2-Propanediol 3 | C3H8O2 | 76.094 | 0.41 | 0.04 |
| 2-Pyrrolidinone | C4H7NO | 85.104 | 0.23 | 0.01 |
| 3-Hydroxybutan-2-one | C4H8O2 | 88.105 | 1.17 | 0.01 |
| 1-Hydroxy-2-butanone | C4H8O2 | 88.105 | 0.04 | 0.01 |
| (2S)-2-Hydroxypropanoic acid 4 | C3H6O3 | 90.078 | 25.18 | 0.48 |
| 2,3-Butanediol 3 | C4H10O | 90.121 | 17.48 | 0.19 |
| 1,3-Propanediol 5 | C3H8O3 | 92.094 | 4.95 | 0.09 |
| 3-Pyridinol | C5H5NO | 95.099 | 17.90 | 0.15 |
| 2-Furaldehyde | C5H4O2 | 96.084 | 1.05 | 0.02 |
| 6-Methyl-3-pyridinol 3 | C6H7NO | 109.126 | 2.88 | 0.06 |
| 1-(2-Furyl)ethanone | C6H6O2 | 110.111 | 0.34 | 0.03 |
| 1,2-Cyclopentanedione, 3-methyl 3 | C6H8O2 | 112.127 | 0.30 | 0.01 |
| 5-(Hydroxymethyl)-2(3H)-furanone | C5H6O3 | 114.099 | 0.36 | 0.02 |
| 4-Oxopentanoic acid | C5H8O3 | 116.115 | 1.42 | 0.08 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jackowski, M.; Semba, D.; Trusek, A.; Wnukowski, M.; Niedzwiecki, L.; Baranowski, M.; Krochmalny, K.; Pawlak-Kruczek, H. Hydrothermal Carbonization of Brewery’s Spent Grains for the Production of Solid Biofuels. Beverages 2019, 5, 12. https://doi.org/10.3390/beverages5010012
Jackowski M, Semba D, Trusek A, Wnukowski M, Niedzwiecki L, Baranowski M, Krochmalny K, Pawlak-Kruczek H. Hydrothermal Carbonization of Brewery’s Spent Grains for the Production of Solid Biofuels. Beverages. 2019; 5(1):12. https://doi.org/10.3390/beverages5010012
Chicago/Turabian StyleJackowski, Mateusz, Damian Semba, Anna Trusek, Mateusz Wnukowski, Lukasz Niedzwiecki, Marcin Baranowski, Krystian Krochmalny, and Halina Pawlak-Kruczek. 2019. "Hydrothermal Carbonization of Brewery’s Spent Grains for the Production of Solid Biofuels" Beverages 5, no. 1: 12. https://doi.org/10.3390/beverages5010012
APA StyleJackowski, M., Semba, D., Trusek, A., Wnukowski, M., Niedzwiecki, L., Baranowski, M., Krochmalny, K., & Pawlak-Kruczek, H. (2019). Hydrothermal Carbonization of Brewery’s Spent Grains for the Production of Solid Biofuels. Beverages, 5(1), 12. https://doi.org/10.3390/beverages5010012