High-Resolution Mass Spectrometry Identification of Secondary Metabolites in Four Red Grape Varieties Potentially Useful as Traceability Markers of Wines
Abstract
:1. Introduction
2. Materials and Methods
2.1. Samples and Standards
2.2. Sample Preparation
2.3. UHPLC-Q/TOF Analysis
2.4. Statistical Analysis
3. Results and Discussion
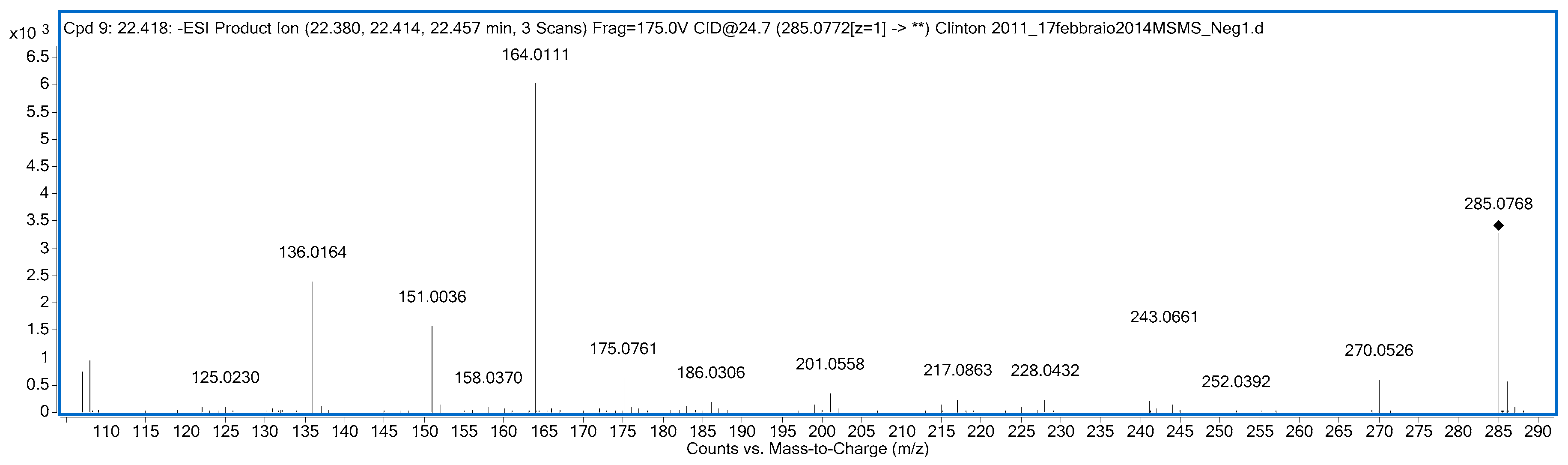
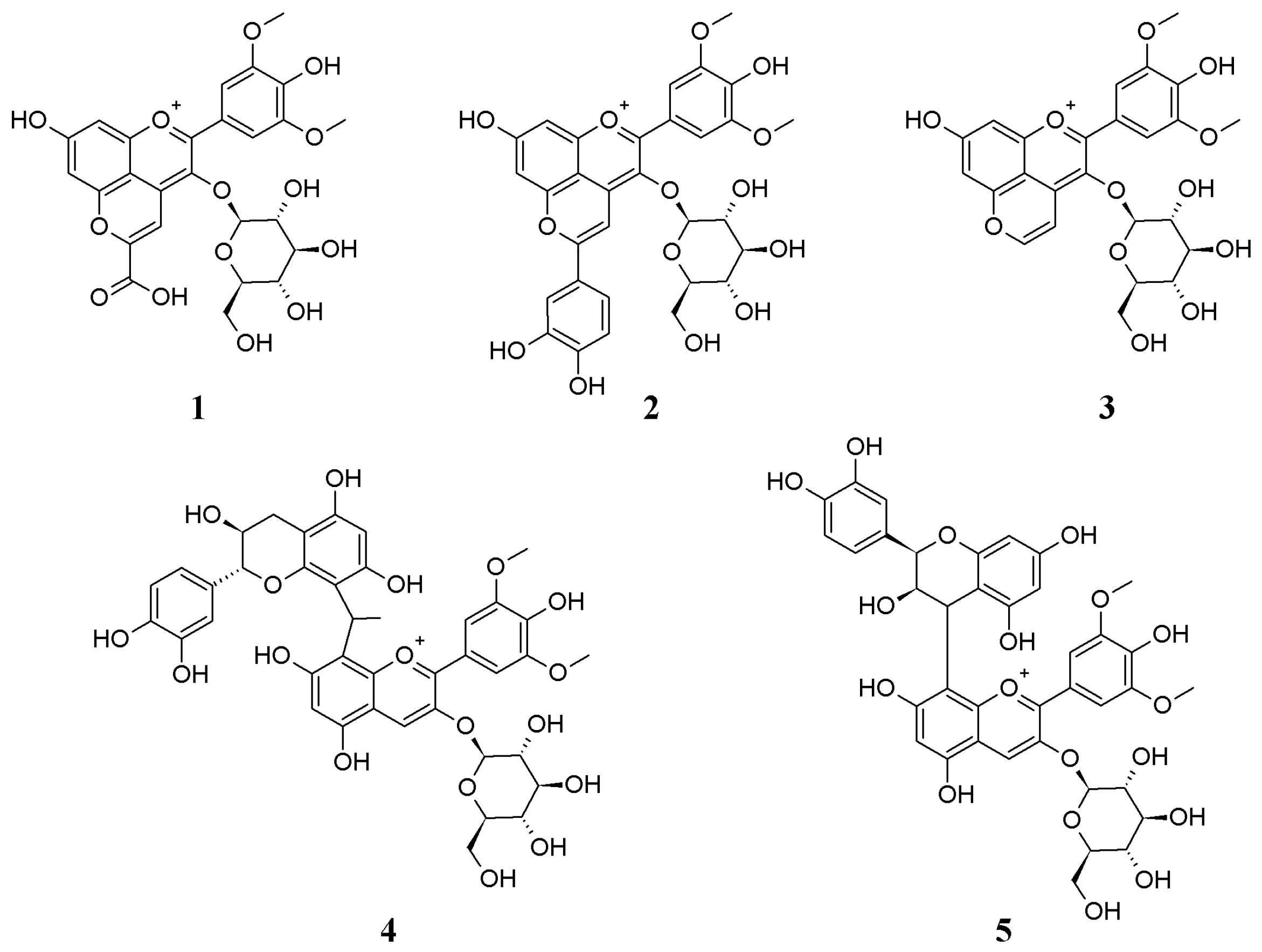
3.1. Identification of the Metabolites
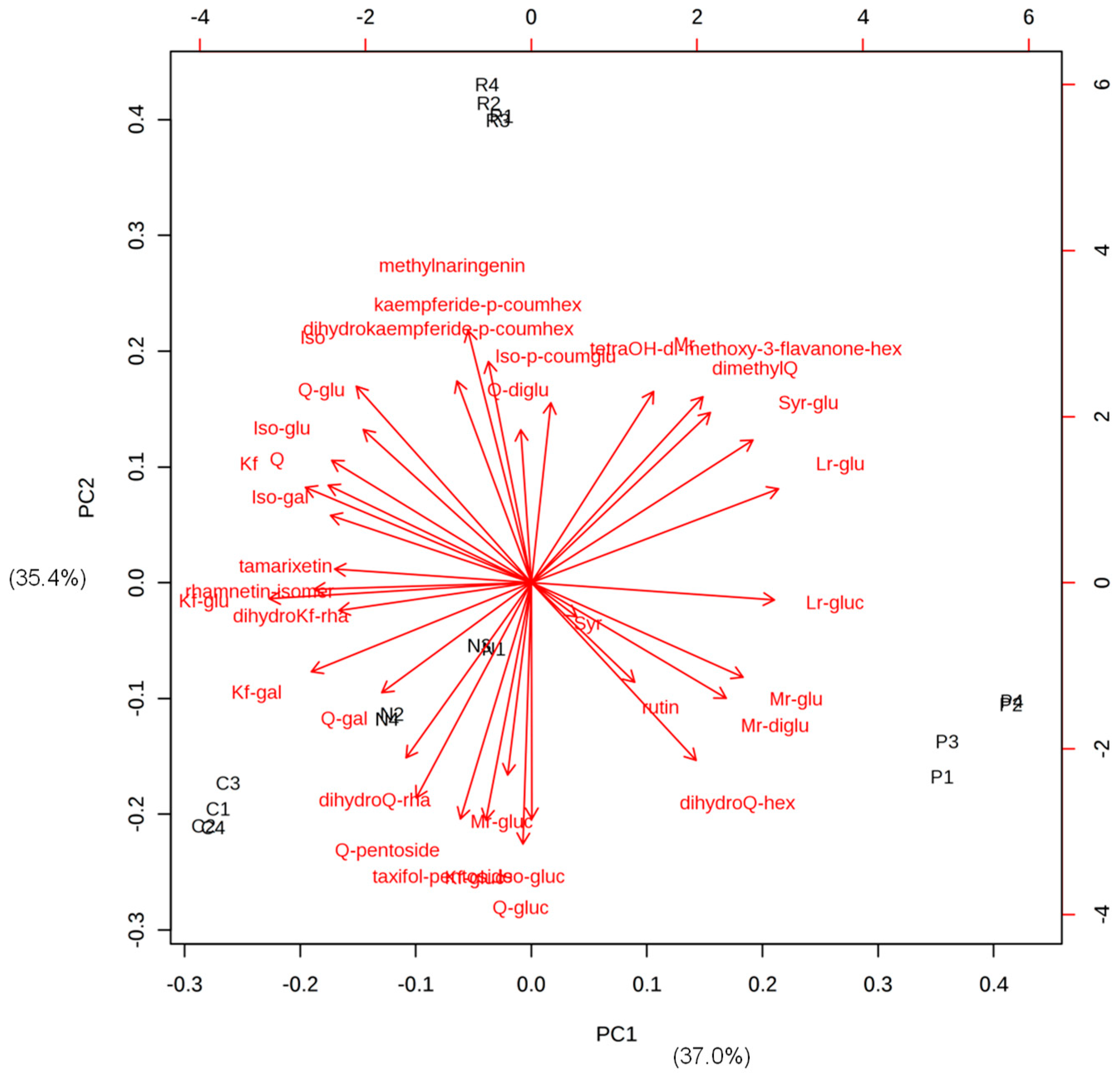
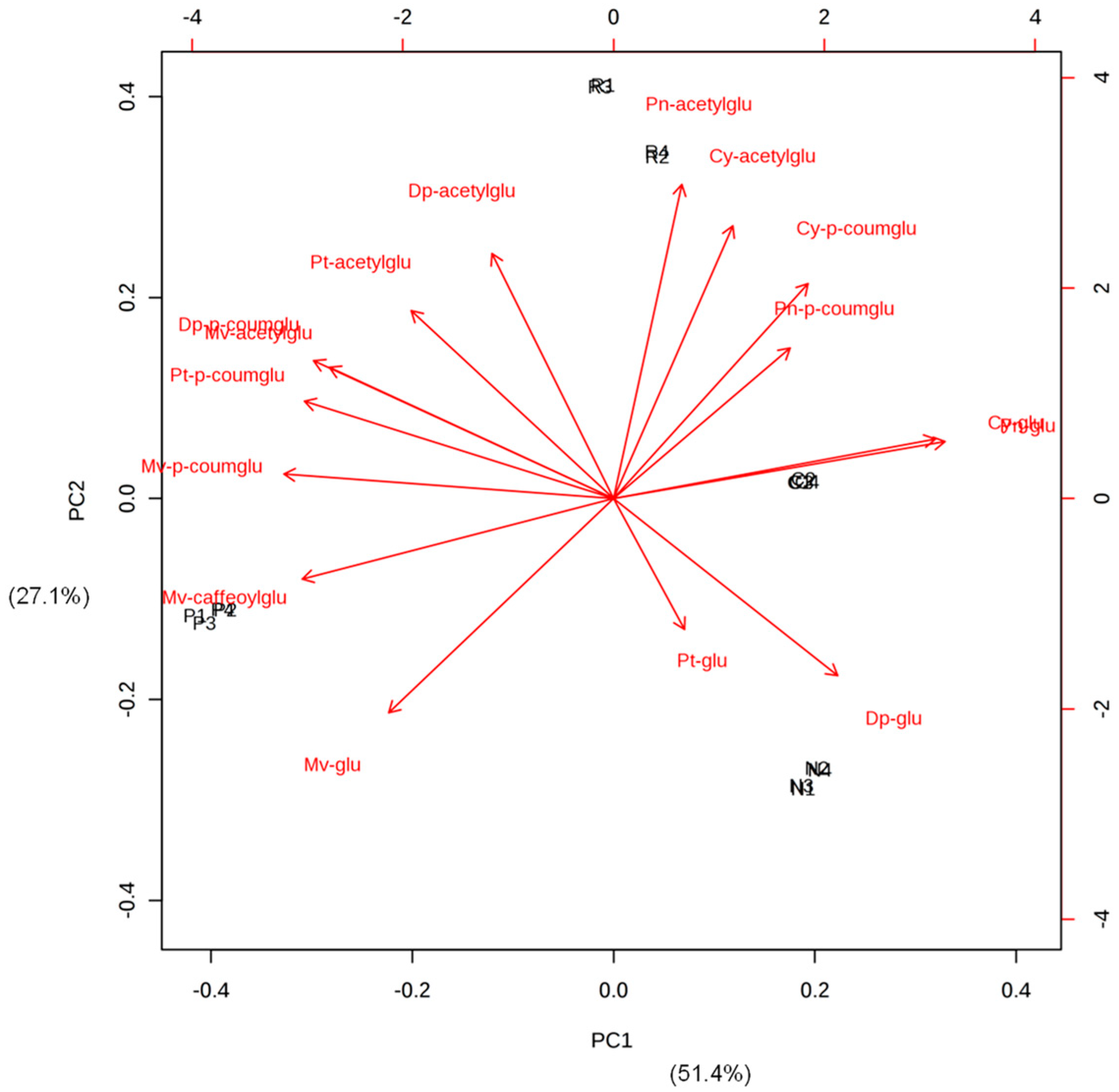
3.2. Potential Flavonoid Markers of the Wine Varieties
3.3. Monoterpene Glycosides (Aroma Precursors)
3.4. Other Metabolites
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- De Rosso, M.; Panighel, A.; Carraro, R.; Padoan, E.; Favaro, A.; Della Vedova, A.; Flamini, R. Chemical characterization and enological potential of raboso varieties by study of secondary grape metabolites. J. Agric. Food Chem. 2010, 2005, 11364–11371. [Google Scholar] [CrossRef] [PubMed]
- Del Gaudio, S.; Nico, G. Primitivo. In Principali Vitigni da vino Coltivati in Italia; Ministero dell’Agricoltura e delle Foreste: Roma, Italy, 1960. [Google Scholar]
- Del Gaudio, S.; Panzera, C. Negro Amaro. In Principali Vitigni da vino Coltivati in Italia; Ministero dell’Agricoltura e delle Foreste: Roma, Italy, 1960. [Google Scholar]
- Ragusa, A.; Centonze, C.; Grasso, M.E.; Latronico, M.F.; Mastrangelo, P.F.; Sparascio, F.; Fanizzi, F.P.; Maffia, M. A comparative study of phenols in apulian Italian wines. Foods 2017, 6, 24. [Google Scholar] [CrossRef] [PubMed]
- Holmberg, L. Wine fraud. Int. J. Wine Res. 2010, 2, 105–113. [Google Scholar] [CrossRef]
- Versari, A.; Laurie, V.F.; Ricci, A.; Laghi, L.; Parpinello, G.P. Progress in authentication, typification and traceability of grapes and wines by chemometric approaches. Food Res. Int. 2014, 60, 2–18. [Google Scholar] [CrossRef]
- Villano, C.; Lisanti, M.T.; Gambuti, A.; Vecchio, R.; Moio, L.; Frusciante, L.; Aversano, R.; Carputo, D. Wine varietal authentication based on phenolics, volatiles and DNA markers: State of the art, perspectives and drawbacks Clizia. Food Control 2017, 80, 1–10. [Google Scholar] [CrossRef]
- Mattivi, F.; Guzzon, R.; Vrhovsek, U.; Stefanini, M.; Velasco, R. Metabolite profiling of grape: Flavonols and anthocyanins. J. Agric. Food Chem. 2006, 54, 7692–7702. [Google Scholar] [CrossRef] [PubMed]
- Figueiredo-González, M.; Martínez-Carballo, E.; Cancho-Grande, B.; Santiago, J.L.; Martínez, M.C.; Simal-Gándara, J. Pattern recognition of three Vitis vinifera L. red grapes varieties based on anthocyanin and flavonol profiles, with correlations between their biosynthesis pathways. Food Chem. 2012, 130, 9–19. [Google Scholar] [CrossRef]
- Ghaste, M.; Narduzzi, L.; Carlin, S.; Vrhovsek, U.; Shulaev, V.; Mattivi, F. Chemical composition of volatile aroma metabolites and their glycosylated precursors that can uniquely differentiate individual grape cultivars. Food Chem. 2015, 188, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Nasi, A.; Ferranti, P.; Amato, S.; Chianese, L. Identification of free and bound volatile compounds as typicalness and authenticity markers of non-aromatic grapes and wines through a combined use of mass spectrometric techniques. Food Chem. 2008, 110, 762–768. [Google Scholar] [CrossRef]
- Favretto, D.; Flamini, R. Application of electrospray ionization mass spectrometry to the study of grape anthocyanins. Am. J. Enol. Vitic. 2000, 51, 55–64. [Google Scholar]
- Flamini, R.; Della Vedova, A.; Calo, A. Study of the monoterpene contents of 23 accessions of Muscat grape: Correlation between aroma profile and variety. Riv. Vitic. Enol. 2001, 54, 35–50. [Google Scholar]
- De Rosso, M.; Tonidandel, L.; Larcher, R.; Nicolini, G.; Ruggeri, V.; Dalla Vedova, A.; De Marchi, F.; Gardiman, M.; Flamini, R. Study of anthocyanic profiles of twenty-one hybrid grape varieties by liquid chromatography and precursor-ion mass spectrometry. Anal. Chim. Acta 2012, 732, 120–129. [Google Scholar] [CrossRef] [PubMed]
- Rubert, J.; Lacina, O.; Fauhl-hassek, C.; Hajslova, J. Metabolic fingerprinting based on high-resolution tandem mass spectrometry: A reliable tool for wine authentication? Anal. Bioanal. Chem. 2014, 406, 6791–6803. [Google Scholar] [CrossRef] [PubMed]
- Vaclavik, L.; Lacina, O.; Hajslova, J.; Zweigenbaum, J. The use of high performance liquid chromatography-quadrupole time-of-flight mass spectrometry coupled to advanced data mining and chemometric tools for discrimination and classification of red wines according to their variety. Anal. Chim. Acta 2011, 685, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Arapitsas, P.; Ugliano, M.; Perenzoni, D.; Angeli, A.; Pangrazzi, P.; Mattivi, F. Wine metabolomics reveals new sulfonated products in bottled white wines, promoted by small amounts of oxygen. J. Chromatogr. A 2016, 1429, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Arbulu, M.; Sampedro, M.C.; Gómez-caballero, A.; Goicolea, M.A.; Barrio, R.J. Untargeted metabolomic analysis using liquid chromatography quadrupole time-of-flight mass spectrometry for non-volatile profiling of wines. Anal. Chim. Acta 2015, 858, 32–41. [Google Scholar] [CrossRef] [PubMed]
- Arapitsas, P.; Della Corte, A.; Gika, H.; Narduzzi, L.; Mattivi, F. Studying the effect of storage conditions on the metabolite content of red wine using HILIC LC–MS based metabolomics. Food Chem. 2016, 197, 1331–1340. [Google Scholar] [CrossRef] [PubMed]
- Flamini, R.; De Rosso, M.; Bavaresco, L. Study of grape polyphenols by liquid chromatography-high-resolution mass spectrometry (UHPLC/QTOF) and suspect screening analysis. J. Anal. Methods Chem. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Flamini, R.; De Rosso, M.; De Marchi, F.; Dalla Vedova, A.; Panighel, A.; Gardiman, M.; Maoz, I.; Bavaresco, L. An innovative approach to grape metabolomics: Stilbene profiling by suspect screening analysis. Metabolomics 2013, 9, 1243–1253. [Google Scholar] [CrossRef]
- Flamini, R.; De Rosso, M.; Panighel, A.; Dalla Vedova, A.; De Marchi, F.; Bavaresco, L. Profiling of grape monoterpene glycosides (aroma precursors) by ultra-high performanceliquid chromatography-high resolution mass spectrometry (UHPLC/QTOF). J. Mass Spectrom. 2014, 49, 1214–1222. [Google Scholar] [CrossRef] [PubMed]
- Di Stefano, R.; Flamini, R. High Performance Liquid Chromotagraphy Analysis of Grape and Wine Polyphenols; John Wiley & Sons: Hoboken, NJ, USA, 2008; pp. 33–79. [Google Scholar]
- Pezet, R.; Perret, C.; Jean-Denis, J.B.; Tabacchi, R.; Gindro, K.; Viret, O. δ-Viniferin, a resveratrol dehydrodimer: One of the major stilbenes synthesized by stressed grapevine leaves. J. Agric. Food Chem. 2003, 51, 5488–5492. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.; Wishart, D.S. Using MetaboAnalyst3.0 for comprehensive metabolomics data analysis. Curr. Protoc. Bioinform. 2016, 55, 14.10.1–14.10.91. [Google Scholar] [CrossRef] [PubMed]
- Fernández-López, J.A.; Almela, L.; Muñoz, J.A.; Hidalgo, V.; Carreño, J. Dependence between colour and individual anthocyanin content in ripening grapes. Food Res. Int. 1998, 31, 667–672. [Google Scholar] [CrossRef]
- Figueiredo-Gonzalez, M.; Cancho-Grande, B.; Simal-Gandara, J. Evolution of colour and phenolic compounds during Garnacha Tintorera grape raisining. Food Chem. 2013, 141, 3230–3240. [Google Scholar] [CrossRef] [PubMed]
- Ortega-Regules, A.; Romero-Cascales, I.; López-Roca, J.M.; Ros-García, J.M.; Gómez-Plaza, E. Anthocyanin fingerprint of grapes: Environmental and genetic variations. J. Sci. Food Agric. 2006, 86, 1460–1467. [Google Scholar] [CrossRef]
- Liang, N.-N.; Zhu, B.-Q.; Han, S.; Wang, J.-H.; Pan, Q.-H.; Reeves, M.J.; Duan, C.-Q.; He, F. Regional characteristics of anthocyanin and flavonol compounds from grapes of four Vitis vinifera varieties in five wine regions of China. Food Res. Int. 2014, 64, 264–274. [Google Scholar] [CrossRef] [PubMed]
- Squadrito, M.; Corona, O.; Ansaldi, G.; Di Stefano, R. Possible relations between biosynthetic pathways of HCTA, flavonols and anthocyanins in grape berry skin. Riv. Vitic. Enol. 2007, 60, 59–70. [Google Scholar]
- Cacho, J.; Fernández, P.; Ferreira, V.; Castells, J.E. Evolution of five anthocyanidin-3-glucosides in the skin of the Tempranillo, Moristel, and Garnacha grape varieties and influence of climatological variables. Am. J. Enol. Vitic. 1992, 43, 244–248. [Google Scholar]
- Carreño, J.; Almela, L.; Martínez, A.; Fernández-López, J.A. Chemotaxonomical classification of red table grapes based on anthocyanin profile and external colour. LWT Food Sci. Technol. 1997, 30, 259–265. [Google Scholar] [CrossRef]
- Makris, D.P.; Kallithraka, S.; Mamalos, A. Differentiation of young red wines based on cultivar and geographical origin with application of chemometrics of principal polyphenolic constituents. Talanta 2006, 70, 1143–1152. [Google Scholar] [CrossRef] [PubMed]
- Heller, W.; Forkmann, G. Biosynthesis. In The Flavonoids: Advances in Research since 1980; Chapman & Hall: London, UK, 1980; pp. 399–425. [Google Scholar]
- Jeong, S.T.; Goto-Yamamoto, N.; Hashizume, K.; Esaka, M. Expression of the flavonoid 3′-hydroxylase and flavonoid 3′,5′-hydroxylase genes and flavonoid composition in grape (Vitis vinifera). Plant Sci. 2006, 170, 61–69. [Google Scholar] [CrossRef]
- Arapitsas, P.; Perenzoni, D.; Nicolini, G.; Mattivi, F. Study of Sangiovese Wines pigment profile by UHPLC-MS/MS. J. Agric. Food Chem. 2012, 60, 10461–10471. [Google Scholar] [CrossRef] [PubMed]
- Fulcrand, H.; Dueñas, M.; Salas, E.; Cheynier, V. Phenolic reactions during winemaking and aging. Am. J. Enol. Vitic. 2006, 57, 289–297. [Google Scholar]
- Rentzsch, M.; Schwarz, M.; Winterhalter, P.; Hermosín-Gutiérrez, I. Formation of hydroxyphenyl-pyranoanthocyanins in Grenache wines: Precursor levels and evolution during aging. J. Agric. Food Chem. 2007, 55, 4883–4888. [Google Scholar] [CrossRef] [PubMed]
- Dipalmo, T.; Crupi, P.; Pati, S.; Lisa, M.; Di, A. Studying the evolution of anthocyanin-derived pigments in a typical red wine of Southern Italy to assess its resistance to aging. LWT Food Sci. Technol. 2016, 71, 1–9. [Google Scholar] [CrossRef]
- Castillo-Muñoz, N.; Gómez-Alonso, S.; García-Romero, E.; Hermosín-Gutiérrez, I. Flavonol profiles of Vitis vinifera red grapes and their single-cultivar wines. J. Agric. Food Chem. 2007, 55, 992–1002. [Google Scholar] [CrossRef] [PubMed]
- Monagas, M.; Gómez-Cordovés, C.; Bartolomé, B. Evolution of polyphenols in red wines from Vitis vinifera L. during aging in the bottle I. Anthocyanins and pyranoanthocyanins. Eur. Food Res. Technol. 2005, 220, 607–614. [Google Scholar] [CrossRef]
- Lingua, M.S.; Fabani, M.P.; Wunderlin, D.A.; Baroni, M.V. From grape to wine: Changes in phenolic composition and its influence on antioxidant activity. Food Chem. 2016, 208, 228–238. [Google Scholar] [CrossRef] [PubMed]
- Bimpilas, A.; Tsimogiannis, D.; Balta-brouma, K.; Lymperopoulou, T.; Oreopoulou, V. Evolution of phenolic compounds and metal content of wine during alcoholic fermentation and storage. Food Chem. 2015, 178, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Von Baer, D.; Mardones, C.; Gutierrez, L.; Hofmann, G.; Becerra, J.; Hitschfeld, A.; Vergara, C. Varietal authenticity verification of Cabernet sauvignon, Merlot and Carmenere wines produced in Chile by their anthocyanin, flavonol and shikimic acid profiles. Bull. OIV 2005, 887, 45–57. [Google Scholar]
- Cordente, A.G.; Curtin, C.D.; Varela, C.; Pretorius, I.S. Flavour-active wine yeasts. Appl. Microbiol. Biotechnol. 2012, 96, 601–618. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ebeler, S.E.; Thorngate, J.H. Wine chemistry and flavor: Looking into the crystal glass. J. Agric. Food Chem. 2009, 57, 8098–8108. [Google Scholar] [CrossRef] [PubMed]
- Styger, G.; Prior, B.; Bauer, F.F. Wine flavor and aroma. J. Ind. Microbiol. Biotechnol. 2011, 1145–1159. [Google Scholar] [CrossRef] [PubMed]
- Flamini, R.; Menicatti, M.; De Rosso, M.; Gardiman, M.; Mayr, C.; Pallecchi, M.; Danza, G.; Bartolucci, G. Combining liquid chromatography and tandem mass spectrometry approaches to the study of monoterpene glycosides (aroma precursors) in wine grape. J. Mass Spectrom. 2018, 53, 1–35. [Google Scholar] [CrossRef] [PubMed]
- Welke, J.E.; Manfroi, V.; Zanus, M.; Lazzarotto, M.; Alcaraz Zini, C. Differentiation of wines according to grape variety using multivariate analysis of comprehensive two-dimensional gas chromatography with time-of-flight mass spectrometric detection data. Food Chem. 2013, 141, 3897–3905. [Google Scholar] [CrossRef] [PubMed]
- Tamborra, P.; Esti, M. Authenticity markers in Aglianico, Uva di Troia, Negroamaro and Primitivo grapes. Anal. Chim. Acta 2010, 660, 221–226. [Google Scholar] [CrossRef] [PubMed]
- Goetz, G.; Fkyerat, A.; Metais, N.; Kunz, M.; Tabacchi, R.; Pezet, R.; Pont, V. Resistance factors to grey mould in grape berries: Identification of some phenolics inhibitors of Botrytis cinerea stilbene oxidase. Phytochemistry 1999, 52, 759–767. [Google Scholar] [CrossRef]
- Bavaresco, L.; Mattivi, F.; De Rosso, M.; Flamini, R. Effects of elicitors, viticultural factors, and enological practices on resveratrol and stilbenes in grapevine and wine. Mini Rev. Med. Chem. 2012, 12, 1366–1381. [Google Scholar] [PubMed]

| Flavonols/Flavanones | p < 0.01 | |||
|---|---|---|---|---|
| Corvina | Primitivo | Negro Amaro | Raboso | |
| dihydrokaempferol-rhamnoside | b | a | a | c |
| dihydroquercetin-hexoside | a | c | ab | b |
| dihydroquercetin-rhamnoside | b | a | a | a |
| dimethylquercetin | a | b | a | c |
| isorhamnetin | a | n.f. | a | b |
| isorhamnetin-galactoside | a | b | c | a |
| isorhamnetin-glucoside | b | c | a | a |
| isorhamnetin-glucuronide | ab | ab | a | b |
| kaempferol | ab | a | ab | b |
| kaempferol-galactoside | a | b | ab | ab |
| kaempferol-glucoside | a | b | a | a |
| kaempferol-glucuronide | a | ab | ab | b |
| laricitrin-glucoside | a | b | a | c |
| laricitrin-glucuronide | a | c | b | ab |
| methylnaringenin | a | a | a | b |
| myricetin | a | b | c | d |
| myricetin-diglucoside | a | b | b | a |
| myricetin-glucoside | a | b | b | a |
| myricetin-glucuronide | a | a | b | a |
| quercetin | ab | b | c | ac |
| quercetin-diglucoside | a | a | b | b |
| quercetin-galactoside | a | b | c | ab |
| quercetin-glucoside | ab | a | bc | c |
| quercetin-glucuronide | a | b | a | c |
| quercetin-pentoside | a | a | a | a |
| rhamnetin-isomer | a | b | a | a |
| rutin | a | a | a | a |
| syringetin | a | ab | b | ab |
| syringetin-glucoside | b | a | c | a |
| tamarixetin | a | b | c | a |
| taxifolin-pentoside | b | a | a | a |
| tetrahydroxy-dimethoxyflavanone hexoside | b | a | a | c |
| kaempferide-p-coumaroylhexoside | a | a | a | b |
| isorhamnetin-p-coumaroylglucoside | a | a | b | c |
| dihydrokaempferide-p-coumaroylhexoside | a | a | a | b |
| Anthocyanins | p < 0.01 | |||
|---|---|---|---|---|
| Corvina | Primitivo | Negro Amaro | Raboso | |
| Cy-acetylglucoside | a | a | a | b |
| Cy-p-coumaroylglucoside | b | a | a | c |
| Cy-glucoside | b | c | a | a |
| Dp-acetylglucoside | a | a | a | b |
| Dp-p-coumaroylglucoside | a | b | a | c |
| Dp-glucoside | a | a | b | ab |
| Mv-acetylglucoside | a | b | a | b |
| Mv-caffeoylglucoside | a | b | a | a |
| Mv-p-coumaroylglucoside | a | b | a | c |
| Mv-glucoside | b | c | a | a |
| Pn-acetylglucoside | a | a | a | b |
| Pn-p-coumaroylglucoside | b | a | a | b |
| Pn-glucoside | a | c | ab | b |
| Pt-acetylglucoside | a | b | a | c |
| Pt-p-coumaroylglucoside | a | b | a | c |
| Pt-glucoside | a | ab | b | b |
| Procyanidins | Normalized [M − H]− Signal Area | |||||||
|---|---|---|---|---|---|---|---|---|
| Corvina | Primitivo | Negro Amaro | Raboso | |||||
| Mean | CV% | Mean | CV% | Mean | CV% | Mean | CV% | |
| (−)-epicatechin | 1,687,850 | 19 | 851,612 | 12 | 1,784,264 | 17 | 1,294,547 | 25 |
| (+)-catechin | 5,299,475 | 16 | 2,007,948 | 55 | 2,028,302 | 10 | 1,571,897 | 8 |
| (−)-epigallocatechin | 446,924 | 1 | 653,619 | 17 | 442,091 | 4 | 448,730 | 14 |
| (−)-epicatechin gallate | 984,163 | 24 | 445,224 | 25 | 729,303 | 21 | 51,256 | 14 |
| procyanidin (B3/B4/B5) | 1,314,453 | 15 | 477,508 | 8 | 882,147 | 18 | 289,062 | 2 |
| procyanidin B1 | 6,813,192 | 4 | 2,778,443 | 7 | 2,501,480 | 8 | 839,493 | 6 |
| procyanidin B2 | 111,031 | 15 | n.f. | 55,704 | 26 | 95,472 | 22 | |
| procyanidin T2/T3(T4)/C1 | 927,837 | 6 | 256,004 | 6 | 261,175 | 13 | 58,481 | 18 |
| procyanidin T2/T3(T4)/C1 | 280,969 | 6 | 67,896 | 39 | 104,278 | 14 | 28,399 | 12 |
| procyanidin T2/T3(T4)/C1 | 312,539 | 15 | 120,654 | 6 | 264,665 | 20 | 45,451 | 18 |
| prodelphinidin T2/T3 | 134,478 | 10 | 51,808 | 26 | 89,307 | 9 | 41,249 | 17 |
| Sum | 18,312,911 | (384%) | 7,710,716 | (162%) | 9,142,716 | (192%) | 4,764,039 | (100%) |
| Monoterpene Glycosides | p < 0.01 | |||
|---|---|---|---|---|
| Corvina | Primitivo | Negro Amaro | Raboso | |
| α-terpineol pentosyl-hexoside | n.f. | n.f. | a | b |
| linalool pentosyl-hexoside | n.f. | n.f. | a | b |
| geraniol pentosyl-hexoside | b | c | a | a |
| Ho-diendiol I pentosyl-hexoside | b | c | a | a |
| Ho-diendiol I rhamnosyl-hexoside | b | c | a | a |
| trans/cis 8-hydroxylinalool pentosyl-hexoside | a | a | b | c |
| trans/cis furan/pyran linalool oxide pentosyl-hexoside | a | a | b | n.f. |
| 3,7-dimethyl-1-octen-6-one-3,7-diol pentosyl-hexoside 1 | a | n.f. | a | b |
| 3,7-dimethyl-1-octen-6-one-3,7-diol pentosyl-hexoside 2 | a | a | a | b |
| 3,7-dimethyl-1-octen-6-one-3,7-diol rhamnosyl-hexoside 1 | a | a | b | c |
| 3,7-dimethyl-1-octen-6-one-3,7-diol rhamnosyl-hexoside 2 | b | a | c | a |
| Stilbenes | Normalized [M − H]− Signal Area | |||||||
|---|---|---|---|---|---|---|---|---|
| Corvina | Primitivo | Negro Amaro | Raboso | |||||
| Mean | CV% | Mean | CV% | Mean | CV% | Mean | CV% | |
| trans-resveratrol | 283,900 | 53 | 22,549 | 17 | 59,096 | 10 | 807,651 | 15 |
| piceatannol | 269,846 | 45 | 106,655 | 49 | 205,075 | 30 | 1,400,281 | 4 |
| cis-piceid | 1,270,082 | 10 | 429,127 | 9 | 1,026,866 | 19 | 1,691,503 | 8 |
| trans-piceid | 177,040 | 17 | 99,308 | 14 | 96,404 | 30 | 313,008 | 15 |
| E-astringin | 67,013 | 15 | 47,798 | 10 | 68,551 | 9 | 44,347 | 10 |
| Z-astringin | 34,428 | 15 | 29,354 | 41 | 38,666 | 25 | 45,557 | 15 |
| pallidol | 246,754 | 8 | 71,397 | 30 | 42,236 | 26 | 172,312 | 18 |
| resveratrol dimer 2 | 60,835 | 12 | 26,747 | 21 | 13,466 | 30 | 265,081 | 24 |
| Z-ε-viniferin | 169,259 | 2 | 63,423 | 15 | 72,224 | 24 | 1,771,218 | 18 |
| E-ε-viniferin | 187,388 | 10 | 110,183 | 13 | 68,958 | 4 | 990,456 | 14 |
| Z-ω-viniferin | 66,891 | 3 | 40,938 | 14 | 28,140 | 18 | 703,006 | 29 |
| δ-viniferin | 70,332 | 5 | 10,490 | 17 | 23,925 | 14 | 137,580 | 41 |
| caraphenol | 22,200 | 25 | 11,504 | 23 | 4690 | 10 | 126,287 | 43 |
| pallidol-3-O-glucoside | 33,095 | 12 | 29,203 | 9 | 14,455 | 12 | 91,436 | 11 |
| α-viniferin | 7699 | 53 | 59,992 | 37 | 16,784 | 83 | 113,833 | 57 |
| Z-miyabenol C | 25,288 | 4 | 18,977 | 17 | 11,579 | 21 | 342,147 | 23 |
| E-miyabenol C | 62,101 | 25 | 118,412 | 24 | 39,757 | 24 | 1,256,874 | 35 |
| tetramer resveratrol 1 | 60,484 | 65 | 11,364 | 50 | 8958 | 14 | 152,028 | 49 |
| tetramer resveratrol 2 | 31,491 | 27 | 12,509 | 48 | 8728 | 25 | 1,129,196 | 18 |
| Sum | 3,146,127 | (27%) | 1,319,930 | (11%) | 1,848,558 | (16%) | 11,553,801 | (100%) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mayr, C.M.; De Rosso, M.; Dalla Vedova, A.; Flamini, R. High-Resolution Mass Spectrometry Identification of Secondary Metabolites in Four Red Grape Varieties Potentially Useful as Traceability Markers of Wines. Beverages 2018, 4, 74. https://doi.org/10.3390/beverages4040074
Mayr CM, De Rosso M, Dalla Vedova A, Flamini R. High-Resolution Mass Spectrometry Identification of Secondary Metabolites in Four Red Grape Varieties Potentially Useful as Traceability Markers of Wines. Beverages. 2018; 4(4):74. https://doi.org/10.3390/beverages4040074
Chicago/Turabian StyleMayr, Christine M., Mirko De Rosso, Antonio Dalla Vedova, and Riccardo Flamini. 2018. "High-Resolution Mass Spectrometry Identification of Secondary Metabolites in Four Red Grape Varieties Potentially Useful as Traceability Markers of Wines" Beverages 4, no. 4: 74. https://doi.org/10.3390/beverages4040074





