Oxygen Consumption by Red Wines under Different Micro-Oxygenation Strategies and Q. Pyrenaica Chips. Effects on Color and Phenolic Characteristics
Abstract
1. Introduction
2. Materials and Methods
2.1. Wood Samples
2.2. Wine
2.3. Wood and Micro-Oxygenation (MOX) Strategies
2.4. Oxygen Determination
2.5. Consumed Oxygen Determination
2.6. Wine Analysis
2.6.1. Phenolic, Anthocyanin, and Tannin Global Parameters Determination
2.6.2. Color Analysis
2.6.3. Copigmentation Parameter Determination
2.6.4. Anthocyanin Individual Determination
2.7. Statistical Analysis
3. Results and Discussion
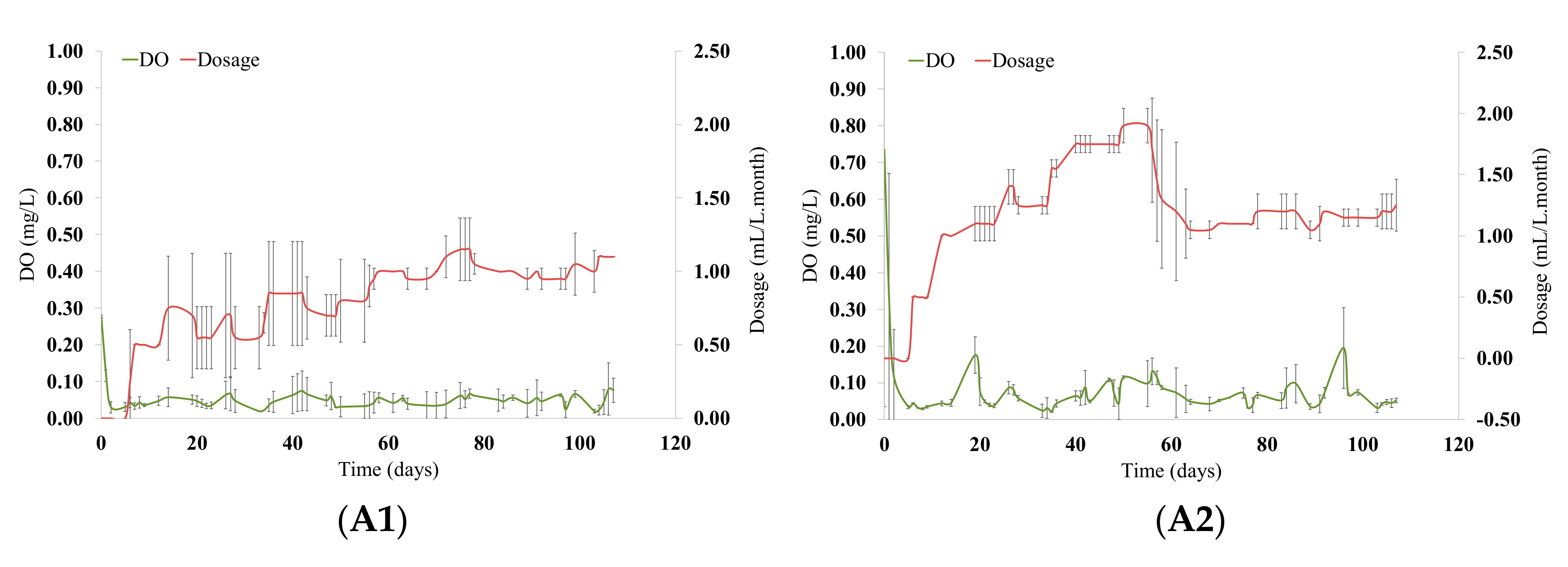
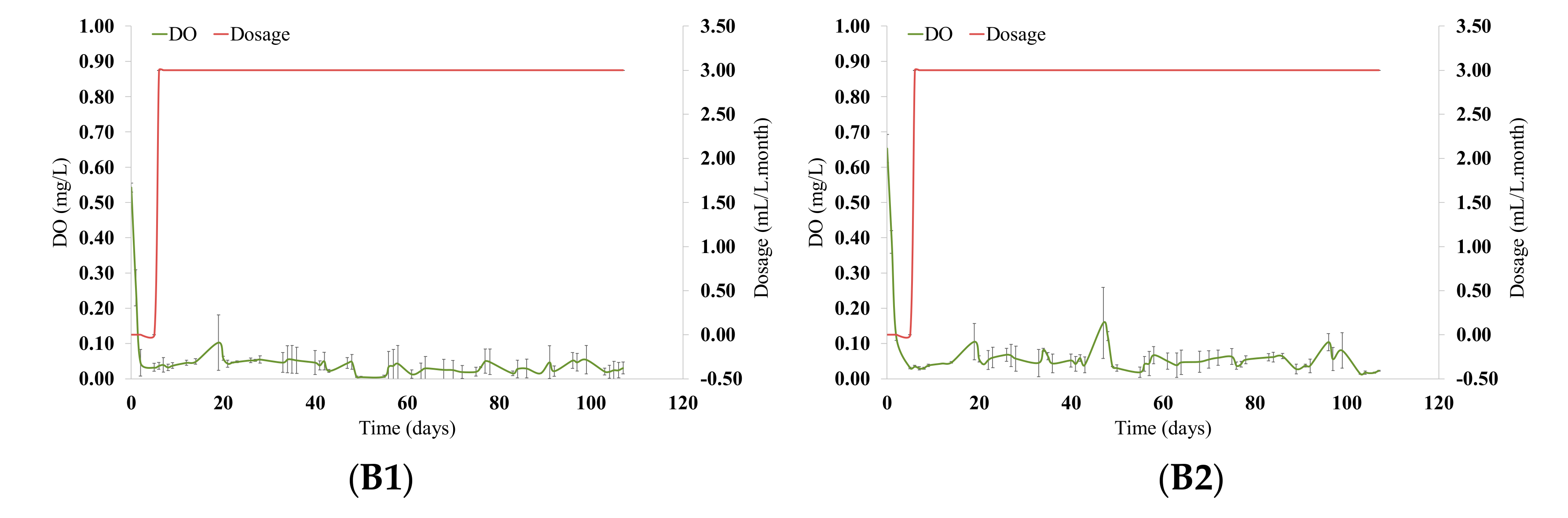
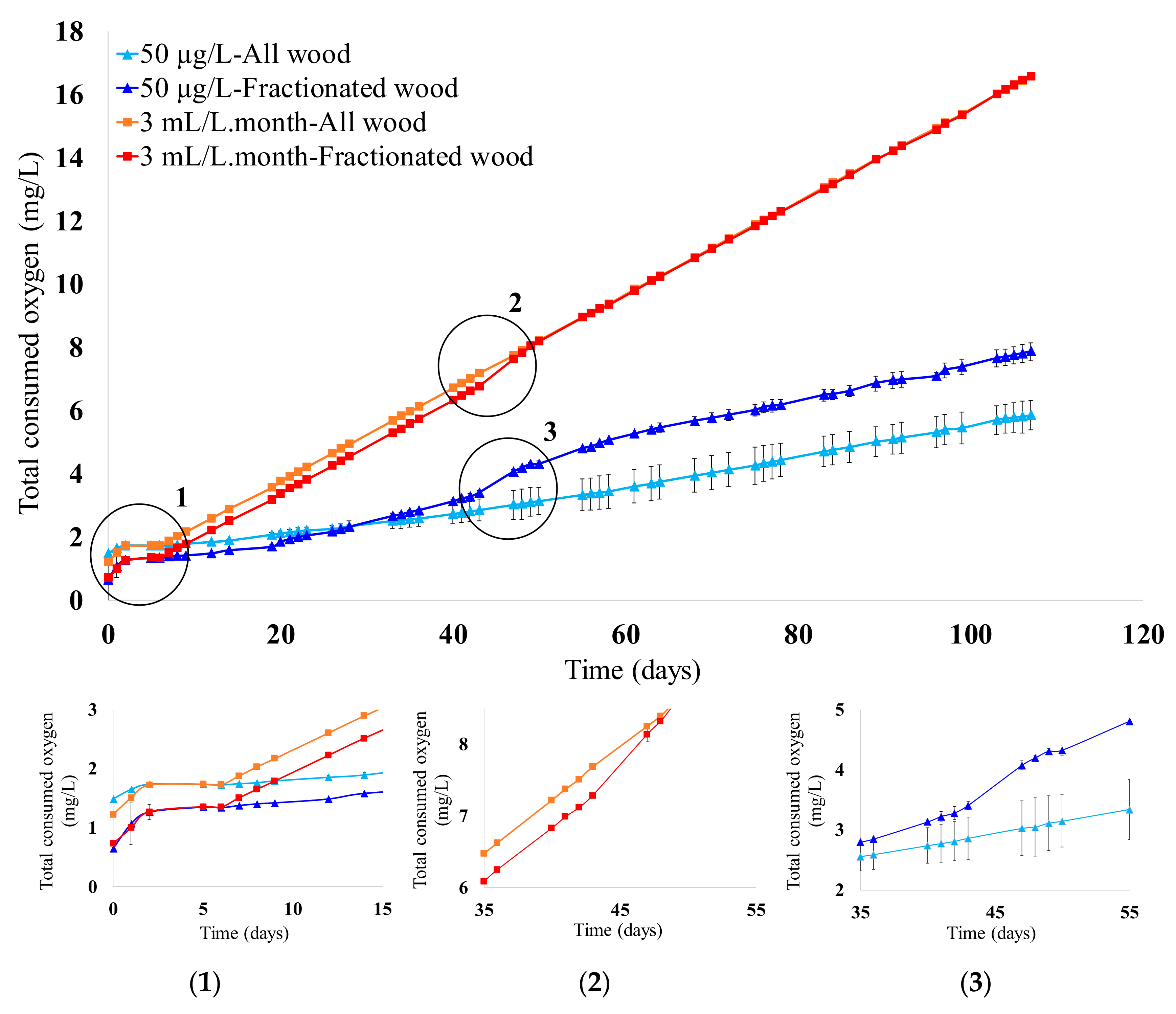
3.1. Micro-Oxygenation Strategy and Evolution of Oxygen Consumption in Wines
3.2. Effect of Micro-Oxygenation in Wines
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Anli, R.E.; C avuldak, Ö.A. A review of microoxygenation application in wine. J. Inst. Brew. 2012, 118, 368–385. [Google Scholar] [CrossRef]
- Chiciuc, I.; Farines, V.; Mietton-Peuchot, M.; Devatine, A. Effect of wine properties and operating mode upon mass transfer in micro-oxygenation. Int. J. Food Eng. 2010, 6, 9. [Google Scholar] [CrossRef]
- Du Toit, W.J.; Marais, J.; Pretorius, I.S.; du Toit, M. Oxygen in must and wine: A review. S. Afr. J. Enol. Vitic. 2006, 27, 76–94. [Google Scholar] [CrossRef]
- Parish, M.; Wollan, D.; Paul, R. Micro-oxygenation—A review. Aust. N.Z. Grapegrow. Winemak. 2000, 438a, 47–50. [Google Scholar]
- Parpinello, G.P.; Versari, A. Micro-oxygenation of red wine: Chemistry and sensory aspects. In Wine: Types, Production and Health; Peeters, A.S., Ed.; Nova Science Publishers: New York, NY, USA, 2012; pp. 93–123. ISBN 978-161470635-9. [Google Scholar]
- Trivellin, N.; Barbisan, D.; Badocco, D.; Pastore, P.; Meneghesso, G.; Meneghini, M.; Zanoni, E.; Belgioioso, G.; Cenedese, A. Study and development of a fluorescence based sensor system for monitoring oxygen in wine production: The WOW project. Sensors 2018, 18, 1130. [Google Scholar] [CrossRef] [PubMed]
- Del Alamo Sanza, M.; Nevares Domínguez, I.; Cárcel Cárcel, L.M.; Navas Gracia, L. Analysis for low molecular weight phenolic compounds in a red wine aged in oak chips. Anal. Chim. Acta 2004, 513, 229–237. [Google Scholar] [CrossRef]
- Del Álamo Sanza, M.; Domínguez, I.N.; Merino, S.G. Influence of different aging systems and oak woods on aged wine colour and anthocyanin composition. Eur. Food Res. Technol. 2004, 219, 124–132. [Google Scholar] [CrossRef]
- Del Alamo-Sanza, M.; Laurie, V.F.; Nevares, I. Wine evolution and spatial distribution of oxygen during storage in high-density polyethylene tanks. J. Sci. Food Agric. 2014, 95, 1313–1320. [Google Scholar] [CrossRef] [PubMed]
- Oberholster, A.; Elmendorf, B.L.; Lerno, L.A.; King, E.S.; Heymann, H.; Brenneman, C.E.; Boulton, R.B. Barrel maturation, oak alternatives and micro-oxygenation: Influence on red wine aging and quality. Food Chem. 2015, 173, 1250–1258. [Google Scholar] [CrossRef] [PubMed]
- Gallego, L.; Del Alamo, M.; Nevares, I.; Fernández, J.A.; De Simón, B.F.; Cadahía, E. Phenolic compounds and sensorial characterization of wines aged with alternative to barrel products made of Spanish oak wood (Quercus pyrenaica Willd.). Food Sci. Technol. Int. 2012, 18, 151–165. [Google Scholar] [CrossRef] [PubMed]
- Nevares, I.; Del Alamo, M.; Cárcel, L.M.; Crespo, R.; Martin, C.; Gallego, L. Measure the dissolved oxygen consumed by red wines in aging tanks. Food Bioproc. Technol. 2009, 2, 328–336. [Google Scholar] [CrossRef]
- Del Álamo, M.; Nevares, I.; Gallego, L.; Fernández de Simón, B.; Cadahía, E. Micro-oxygenation strategy depends on origin and size of oak chips or staves during accelerated red wine aging. Anal. Chim. Acta 2010, 660, 92–101. [Google Scholar] [CrossRef] [PubMed]
- García-Estévez, I.; Alcalde-Eon, C.; Martínez-Gil, A.M.; Rivas-Gonzalo, J.C.; Escribano-Bailón, M.T.; Nevares, I.; del Alamo-Sanza, M. An approach to the study of the interactions between ellagitannins and oxygen during oak wood aging. J. Agric. Food Chem. 2017, 65, 6369–6378. [Google Scholar] [CrossRef] [PubMed]
- Nevares, I.; del Álamo-Sanza, M. Wine aging technologies. In Recent Advances in Wine Stabilization and Conservation Technologies; Jordão, A.M., Cosme, F., Eds.; Nova Science Publishers: New York, NY, USA, 2016; pp. 209–245, ISBN 978-163484899-2; 978-163484883-1. [Google Scholar]
- De Simón, B.F.; Cadahía, E.; Sanz, M.; Poveda, P.; Perez-Magariño, S.; Ortega-Heras, M.; González-Huerta, C. Volatile compounds and sensorial characterization of wines from four Spanish denominations of origin, aged in Spanish Rebollo (Quercus pyrenaica Willd.) oak wood barrels. J. Agric. Food Chem. 2008, 56, 9046–9055. [Google Scholar] [CrossRef] [PubMed]
- Castro-Vázquez, L.; Alañón, M.E.; Ricardo-Da-Silva, J.M.; Pérez-Coello, M.S.; Laureano, O. Study of phenolic potential of seasoned and toasted Portuguese wood species (Quercus pyrenaica and Castanea sativa). J. Int. Sci. Vigne Vin 2013, 47, 311–319. [Google Scholar] [CrossRef]
- De Coninck, G.; Jordão, A.M.; Ricardo-Da-Silva, J.M.; Laureano, O. Evolution of phenolic composition and sensory properties in red wine aged in contact with Portuguese and French oak wood chips. J. Int. Sci. Vigne Vin 2006, 40, 25–34. [Google Scholar] [CrossRef]
- De Simón, B.F.; Sanz, M.; Cadahía, E.; Poveda, P.; Broto, M. Chemical characterization of oak heartwood from Spanish forests of Quercus pyrenaica (Wild.). Ellagitannins, low molecular weight phenolic, and volatile compounds. J. Agric. Food Chem. 2006, 54, 8314–8321. [Google Scholar] [CrossRef] [PubMed]
- De Simon, B.F.; Cadahia, E.; Jalocha, J. Volatile compounds in a Spanish red wine aged in barrels made of Spanish, French, and American oak wood. J. Agric. Food Chem. 2003, 51, 7671–7678. [Google Scholar] [CrossRef] [PubMed]
- De Simón, B.F.; Muiño, I.; Cadahía, E. Characterization of volatile constituents in commercial oak wood chips. J. Agric. Food Chem. 2010, 58, 9587–9596. [Google Scholar] [CrossRef] [PubMed]
- Del Alamo Sanza, M.; Escudero, J.A.F.; De Castro Torío, R. Changes in phenolic compounds and colour parameters of red wine aged with oak chips and in oak barrels. Food Sci. Technol. Int. 2004, 10, 233–241. [Google Scholar] [CrossRef]
- Castellari, M.; Simonato, B.; Tornielli, G.B.; Spinelli, P.; Ferrarini, R. Effects of different enological treatments on dissolved oxygen in wines. Ital. J. Food Sci. 2004, 16, 387–396. [Google Scholar]
- Moutounet, M.; Mazauric, J.-P. L’oxygène dissous dans les vins. Rev. Fran. D’Oenol. 2001, 186, 12–15. Available online: https://prodinra.inra.fr/record/56312 (accessed on 20 June 2018).
- Laurie, V.F.; Law, R.; Joslin, W.S.; Waterhouse, A.L. In situ measurements of dissolved oxygen during low-level oxygenation in red wines. Am. J. Enol. Vitic. 2008, 59, 215–219. [Google Scholar]
- Nevares, I.; del Álamo, M. Measurement of dissolved oxygen during red wines tank aging with chips and micro-oxygenation. Anal. Chim. Acta 2008, 621, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Folin, O.; Ciocalteu, V. On tyrosine and tryptophane determination in proteins. J. Biol. Chem. 1927, 73, 627–650. [Google Scholar]
- Masquelier, J.; Michaud, J.; Triaud, J. Fractionnement des leucoanthocyannes du vin. Bull. Soc. Pharm. Bordx. 1965, 104, 81–85. [Google Scholar]
- Paronetto, L. Polifenoli e Tecnica Enológica; Edagricole: Bologna, Italy, 1977; ISBN 88-206-1704-8. [Google Scholar]
- Ribereau-Gayon, J.; Stonstreet, E. Determination of anthocyanins in red wine. In Bulletin de la Societe de Pharmacie de Bordeaux; Société de Pharmacie (Bordeaux): Bordeaux, France, 1965; Volume 9, pp. 2649–2652. ISSN 00378968. [Google Scholar]
- Swain, T.; Hillis, W.E. The phenolic constituents of Prunus domestica. I.—The quantitative analysis of phenolic constituents. J. Sci. Food Agric. 1959, 10, 63–68. [Google Scholar] [CrossRef]
- Somers, T.C.; Evans, M.E. Spectral evaluation of young red wines: Anthocyanin equilibria, total phenolics, free and molecular SO2, “chemical age”. J. Sci. Food Agric. 1977, 28, 279–287. [Google Scholar] [CrossRef]
- Ribéreau-Gayón, P.; Glories, Y.; Maujean, A.; Dobourdieu, D. Tratado de Enología 2. Química del vino. Estabilización y Tratamientos; Ediciones Mundi prensa: Madrid, España, 2003; ISBN 978950504571. [Google Scholar]
- Glories, Y. La couleur des vins rouges 2. Mesure, origine et interprétation. Connais. Vigne Vin 1984, 18, 253–271. [Google Scholar] [CrossRef]
- Boulton, R.B. The copigmentation of anthocyanins and its role in the colour of red wine: A critical review. Am. J. Enol. Vitic. 2001, 52, 67–87. [Google Scholar]
- Atanasova, V.; Fulcrand, H.; Cheynier, V.; Moutounet, M. Effect of oxygenation on polyphenol changes occurring in the course of wine-making. Anal. Chim. Acta 2002, 458, 15–27. [Google Scholar] [CrossRef]
- Castellari, M.; Matricardi, L.; Arfelli, G.; Galassi, S.; Amati, A. Level of single bioactive phenolics in red wine as a function of the oxygen supplied during storage. Food Chem. 2000, 69, 61–67. [Google Scholar] [CrossRef]
- Morata, A.; Gomez-Cordoves, C.; Colomo, B.; Suarez, J.A. Pyruvic acid and acetaldehyde by different strains of Saccharomyces cerevisiae: Relationship with vitisin A and B formation in red wines. J. Agric. Food Chem. 2003, 51, 7402–7409. [Google Scholar] [CrossRef] [PubMed]
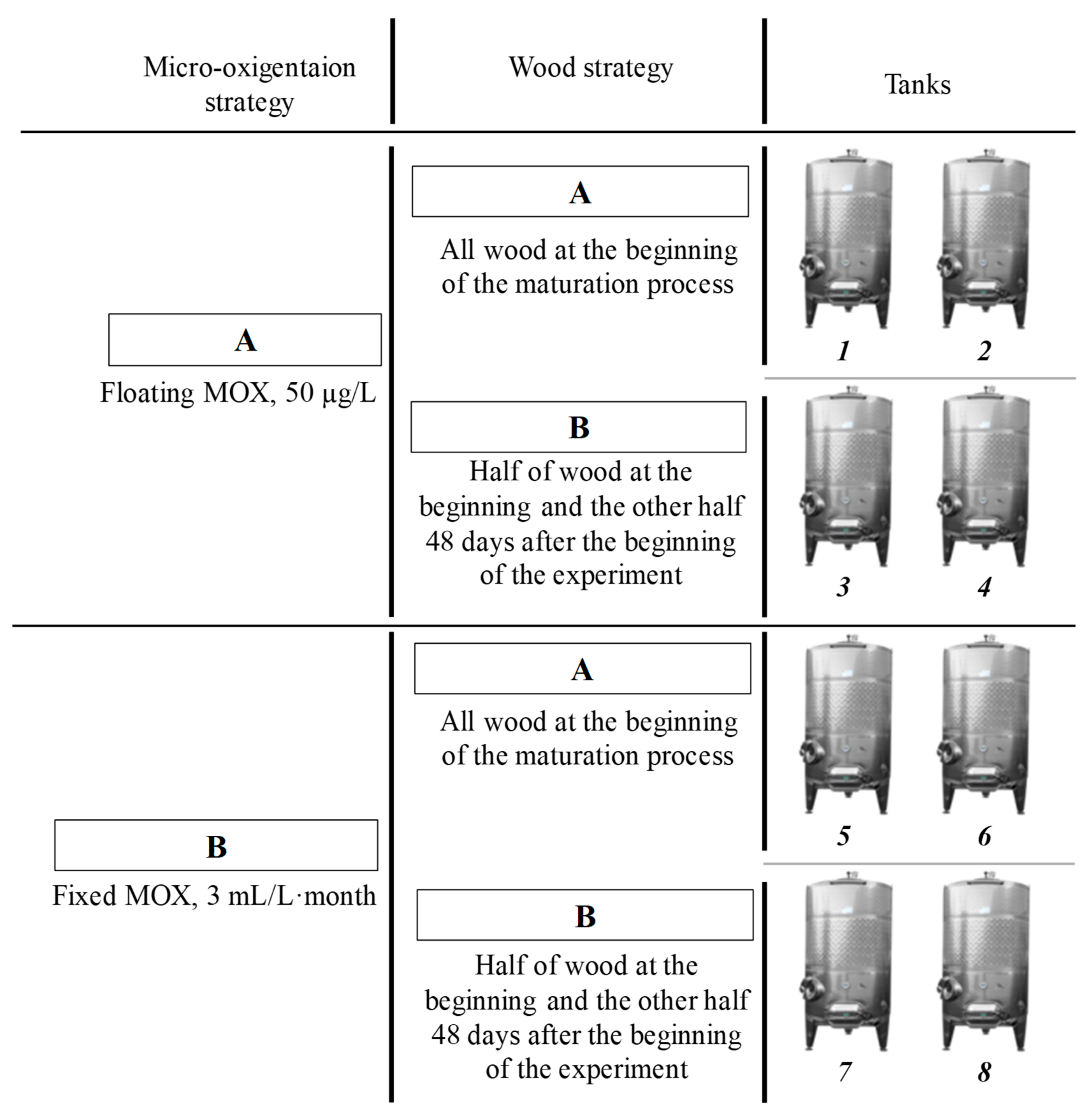
| Micro-Oxygenation | 50 µg/L | 3 mL/L·month | ||
|---|---|---|---|---|
| Time wood was added | At the beginning | Half of the wood at the beginning and the other half at 48 days | At the beginning | Half of the wood at the beginning and the other half at 48 days |
| MOX dosage (mL/L·month) | 0.92 ± 0.17 | 1.22 ± 0.35 | 3.00 ± 0.00 | 3.00 ± 0.00 |
| Total O2 inputs (mL/L) | 6.06 ± 0.27 | 7.60 ± 0.37 | 16.60 ± 0.02 | 16.41 ± 0.25 |
| MOX dosage (mg/month) | 0.62 ± 0.14 | 0.83 ± 0.24 | 2.04 ± 0.00 | 2.04 ± 0.00 |
| Parameter | 50 µg/L | 3 mL/L·month | ||||
|---|---|---|---|---|---|---|
| Means | Std. Dev. | TCO (µg/L) | Means | Std. Dev. | TCO (µg/L) | |
| TCO (µg/L) | 4731.553 | 2027.881 | 1.000000 | 10834.07 | 4902.537 | 1.000000 |
| D-LPP | 15.865 | 92.833 | 0.141855 | 27.19 | 173.186 | 0.439508 |
| D-HPP | 575.850 | 182.728 | 0.492817 | 497.68 | 247.238 | 0.470771 |
| D-CAT | 5.581 | 116.711 | −0.138876 | −12.84 | 70.835 | −0.380093 |
| D-ACY | −106.595 | 62.470 | −0.521031 | −90.65 | 33.655 | −0.621881 |
| D-TAN | 3.026 | 0.285 | 0.118416 | 2.92 | 0.187 | 0.078329 |
| D-CI | −1.789 | 3.240 | −0.561610 | −1.99 | 3.315 | −0.683212 |
| D-T | 0.115 | 0.075 | 0.782966 | 0.12 | 0.077 | 0.909374 |
| D-FC | −0.400 | 1.666 | −0.098521 | −0.61 | 1.681 | −0.261112 |
| D-TP | −11.231 | 14.193 | −0.453367 | −10.55 | 12.323 | −0.321815 |
| D-COP | 0.029 | 0.048 | 0.065126 | 0.04 | 0.056 | 0.097685 |
| D-AL | −0.154 | 0.032 | −0.602250 | −0.16 | 0.065 | −0.286415 |
| D-PP | 0.074 | 0.040 | 0.402556 | 0.07 | 0.034 | 0.381631 |
| D-EtOH-I | 6.613 | 2.660 | −0.431844 | 5.36 | 4.294 | −0.658603 |
| D-HCl-I | 3.529 | 2.668 | −0.464282 | 3.50 | 3.059 | −0.488752 |
| D-GEL-I | −20.827 | 7.877 | −0.095990 | −19.63 | 5.599 | −0.029458 |
| D-ION-I | −8.443 | 8.122 | −0.516031 | −9.93 | 8.022 | −0.775361 |
| D-OD | −182.100 | 60.460 | −0.738996 | −162.80 | 53.456 | −0.756095 |
| D-IPT | 0.103 | 6.066 | 0.009464 | −1.37 | 3.020 | 0.002513 |
| D-%A420 | 0.975 | 3.415 | 0.318594 | 0.92 | 3.513 | 0.379195 |
| D-%A520 | −7.347 | 5.000 | −0.588424 | −7.58 | 5.243 | −0.645097 |
| D-%A620 | 6.372 | 6.934 | 0.267373 | 6.66 | 7.274 | 0.281831 |
| D-Df-3-Gl | −4.957 | 6.689 | −0.359792 | −5.45 | 6.259 | −0.164040 |
| D-Cy-3-Gl | −2.007 | 1.846 | −0.876709 | −2.00 | 1.808 | −0.829679 |
| D-Pt-3-Gl | −2.196 | 3.909 | −0.162831 | −2.37 | 3.643 | 0.055472 |
| D-Pe-3-Gl | −0.883 | 1.095 | −0.407683 | −0.92 | 1.005 | −0.226321 |
| D-Mv-3-Gl | −10.421 | 11.659 | −0.436685 | −10.85 | 10.893 | −0.264131 |
| D-Vitisin A | 0.179 | 0.191 | 0.657446 | 0.18 | 0.193 | 0.659883 |
| D-Mv-3-Gl-Ethyl | 0.108 | 0.205 | 0.540355 | 0.09 | 0.213 | 0.699438 |
| D-Pe-3-Gl-Ac | −0.485 | 0.523 | −0.350600 | −0.59 | 0.440 | −0.323565 |
| D-Df-3-Gl-Cm | −2.470 | 1.986 | −0.813771 | −2.52 | 1.918 | −0.868531 |
| D-Mv-3-Gl-Ac | −0.258 | 0.190 | −0.361746 | −0.30 | 0.203 | −0.107868 |
| D-Cy-3-Gl-Cm | 0.954 | 1.845 | 0.114454 | 1.02 | 1.962 | 0.130637 |
| D-Mv-3-Gl-Cm C | 0.316 | 0.336 | 0.525493 | 0.42 | 0.575 | 0.383817 |
| D-Pt-3-Gl-Cm | −0.152 | 0.214 | −0.467845 | −0.14 | 0.164 | −0.390148 |
| D-Mv-3-Gl-Cm T | −2.396 | 1.985 | −0.875855 | −2.36 | 1.944 | −0.884408 |
| D-Acet | 4.270 | 6.419 | 0.677614 | 3.63 | 5.936 | 0.771172 |
| D-Cum | 5.378 | 8.351 | 0.660566 | 5.44 | 8.005 | 0.810550 |
| D-Total | 32.185 | 76.224 | 0.516223 | 30.13 | 73.811 | 0.678868 |
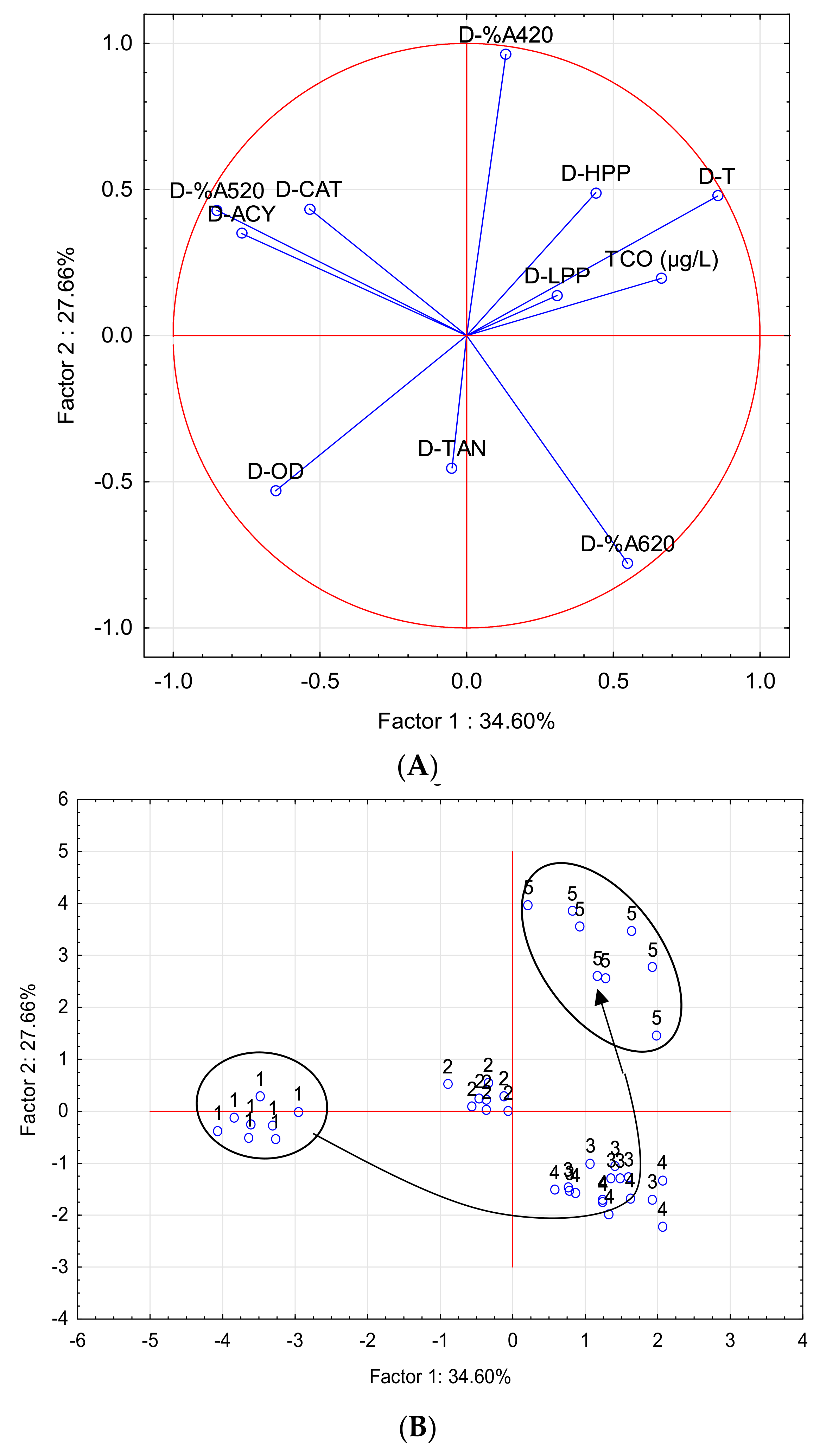
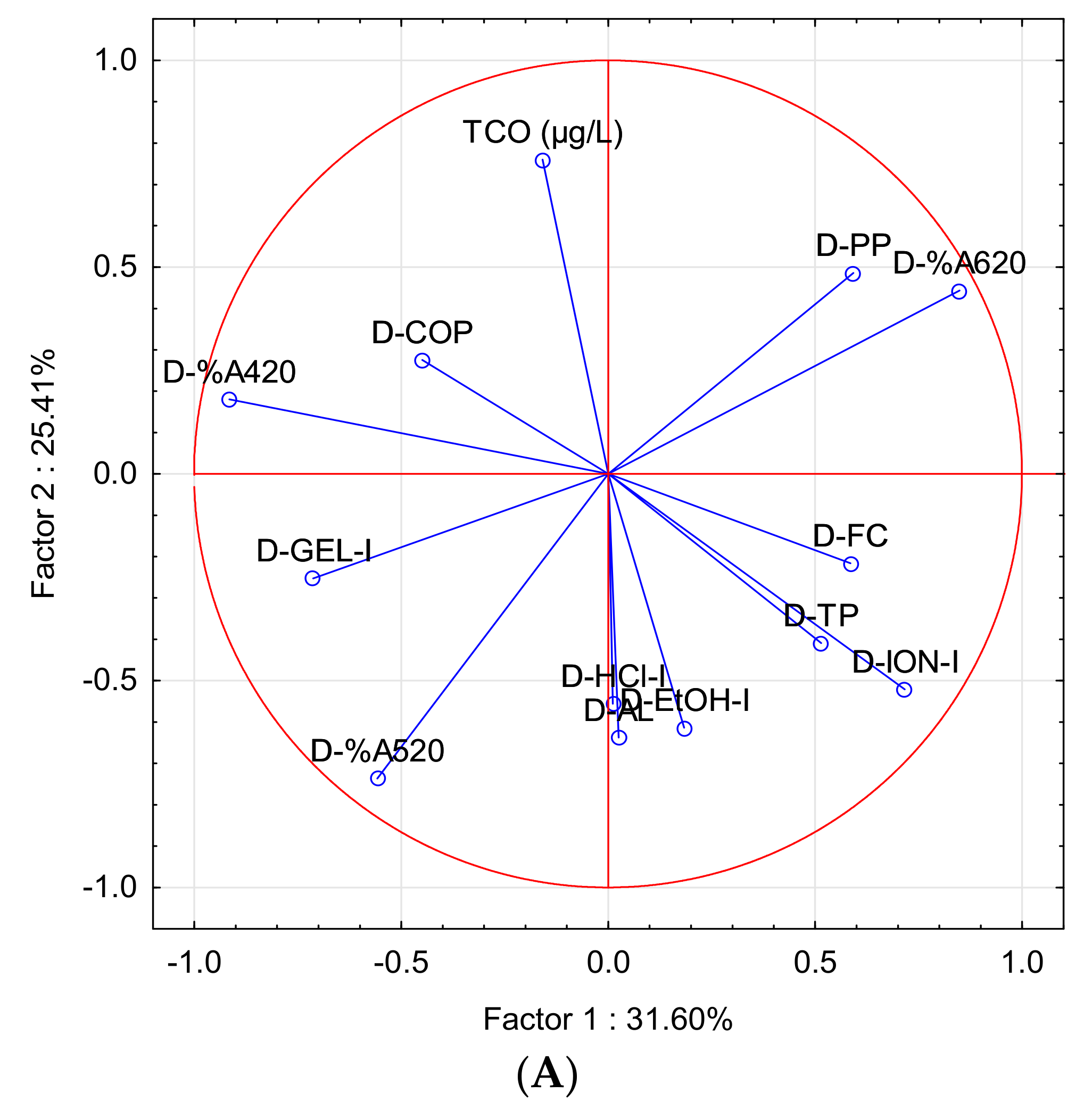
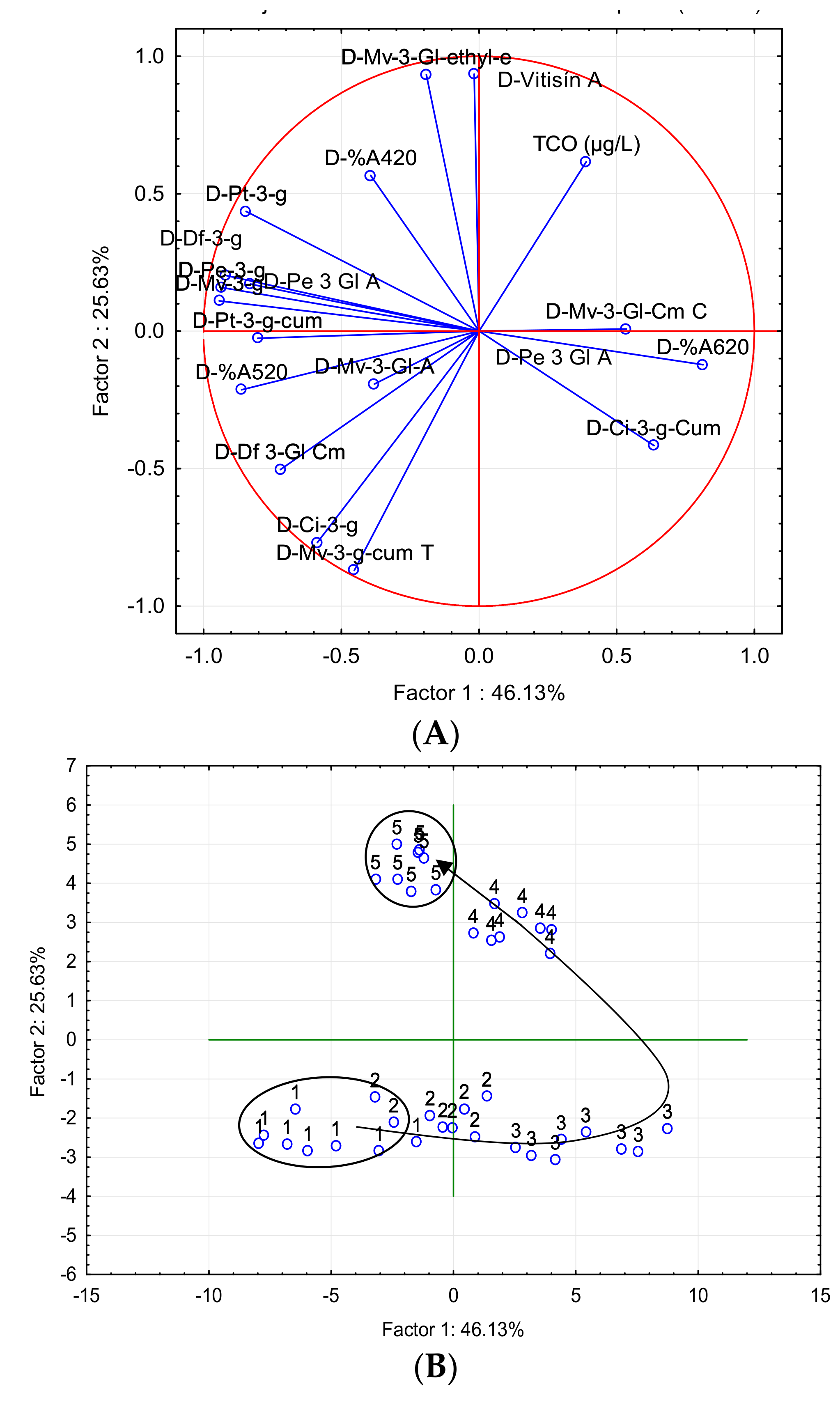
| Parameter | Factor 1 | Factor 2 |
|---|---|---|
| PCA-A | ||
| TCO (µg/L) | 0.6634 | 0.1964 |
| D-LPP | 0.3082 | 0.1378 |
| D-HPP | 0.4402 | 0.4895 |
| D-CAT | −0.5360 | 0.4341 |
| D-ACY | −0.7675 | 0.3499 |
| D-TAN | −0.0502 | −0.4529 |
| D-T | 0.8553 | 0.4782 |
| D-%A420 | 0.1334 | 0.9629 |
| D-%A520 | −0.8512 | 0.4295 |
| D-%A620 | 0.5486 | −0.7790 |
| D-OD | −0.6510 | −0.5311 |
| PCA-B | ||
| TCO (µg/L) | −0.1588 | 0.7599 |
| D-%A420 | −0.9154 | 0.1802 |
| D-%A520 | −0.5577 | −0.7365 |
| D-%A620 | 0.8483 | 0.4431 |
| D-COP | −0.4493 | 0.2756 |
| D-TP | 0.5143 | −0.4087 |
| D-FC | 0.5851 | −0.2164 |
| D-AL | 0.0251 | −0.6377 |
| D-PP | 0.5921 | 0.4860 |
| D-EtOH-I | 0.1834 | −0.6141 |
| D-HCl-I | 0.0113 | −0.5556 |
| D-GEL-I | −0.7125 | −0.2527 |
| D-ION-I | 0.7163 | −0.5213 |
| PCA-C | ||
| TCO (µg/L) | 0.3890 | 0.6159 |
| D-%A420 | −0.3931 | 0.5658 |
| D-%A520 | −0.8618 | −0.2132 |
| D-%A620 | 0.8129 | −0.1221 |
| D-Df-3-Gl | −0.9192 | 0.2025 |
| D-Cy-3-Gl | −0.5871 | −0.7686 |
| D-Pt-3-Gl | −0.8454 | 0.4356 |
| D-Pe-3-Gl | −0.9341 | 0.1583 |
| D-Mv-3-Gl | −0.9427 | 0.1100 |
| D-Vitisin A | −0.0175 | 0.9354 |
| D- Mv-3-Gl-Ethyl | −0.1916 | 0.9326 |
| D-Pe-3-Gl-Ac | −0.8297 | 0.1714 |
| D-Df-3-Gl-Cm | −0.7212 | −0.5033 |
| D-Mv-3-Gl-Ac | −0.3799 | −0.1920 |
| D-Cy-3-Gl-Cm | 0.6353 | −0.4160 |
| D-Mv-3-Gl-Cm C | 0.5347 | 0.0076 |
| D-Pt-3-Gl-Cm | −0.8031 | −0.0258 |
| D-Mv-3-Gl-Cm T | −0.4527 | −0.8697 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sánchez-Gómez, R.; Nevares, I.; Martínez-Gil, A.M.; Del Alamo-Sanza, M. Oxygen Consumption by Red Wines under Different Micro-Oxygenation Strategies and Q. Pyrenaica Chips. Effects on Color and Phenolic Characteristics. Beverages 2018, 4, 69. https://doi.org/10.3390/beverages4030069
Sánchez-Gómez R, Nevares I, Martínez-Gil AM, Del Alamo-Sanza M. Oxygen Consumption by Red Wines under Different Micro-Oxygenation Strategies and Q. Pyrenaica Chips. Effects on Color and Phenolic Characteristics. Beverages. 2018; 4(3):69. https://doi.org/10.3390/beverages4030069
Chicago/Turabian StyleSánchez-Gómez, Rosario, Ignacio Nevares, Ana María Martínez-Gil, and Maria Del Alamo-Sanza. 2018. "Oxygen Consumption by Red Wines under Different Micro-Oxygenation Strategies and Q. Pyrenaica Chips. Effects on Color and Phenolic Characteristics" Beverages 4, no. 3: 69. https://doi.org/10.3390/beverages4030069
APA StyleSánchez-Gómez, R., Nevares, I., Martínez-Gil, A. M., & Del Alamo-Sanza, M. (2018). Oxygen Consumption by Red Wines under Different Micro-Oxygenation Strategies and Q. Pyrenaica Chips. Effects on Color and Phenolic Characteristics. Beverages, 4(3), 69. https://doi.org/10.3390/beverages4030069









