Wine Phenolic Compounds: Antimicrobial Properties against Yeasts, Lactic Acid and Acetic Acid Bacteria
Abstract
:1. Introduction
2. Materials and Methods
2.1. Microorganisms and Culture Conditions
2.2. Chemicals
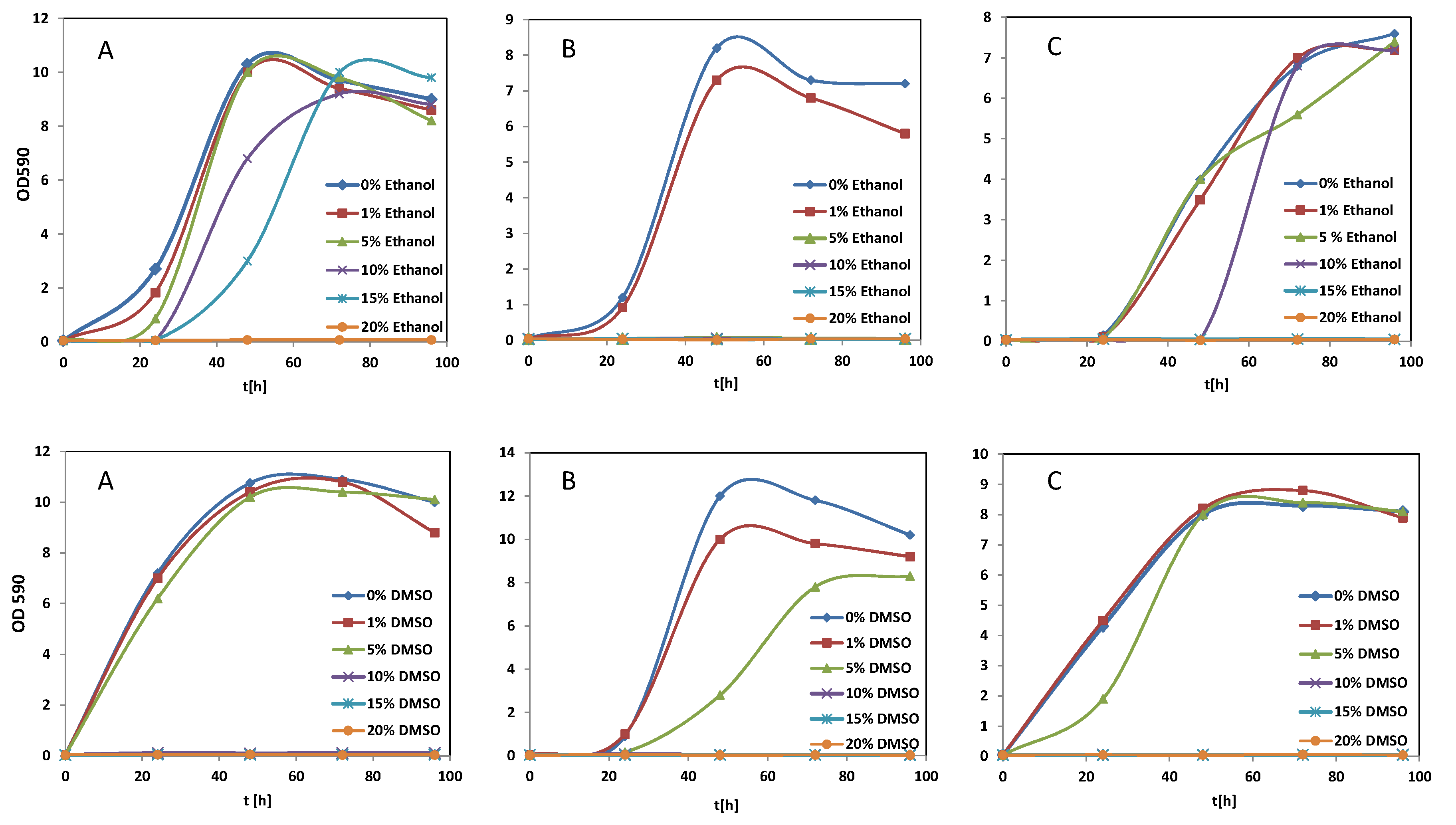
2.3. Influence of Ethanol and DMSO on Yeast Growth
2.4. Influence of Phenols on Growth of Microorganisms
2.4.1. Method 1
2.4.2. Method 2
2.5. Influence of Laccase Oxidation on the Inhibition Effect of Phenolic Compounds
3. Results
3.1. Effect of Solvents
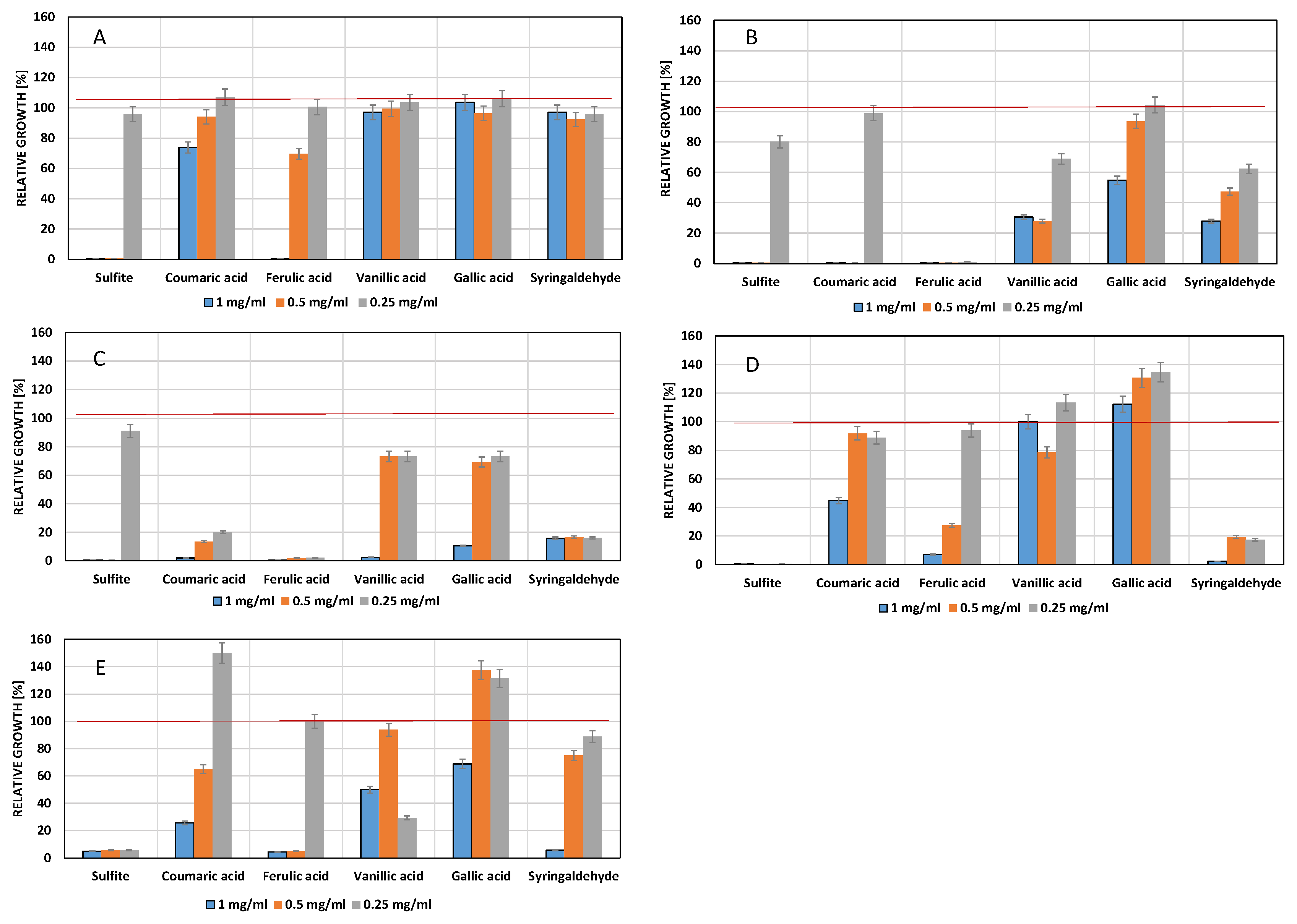
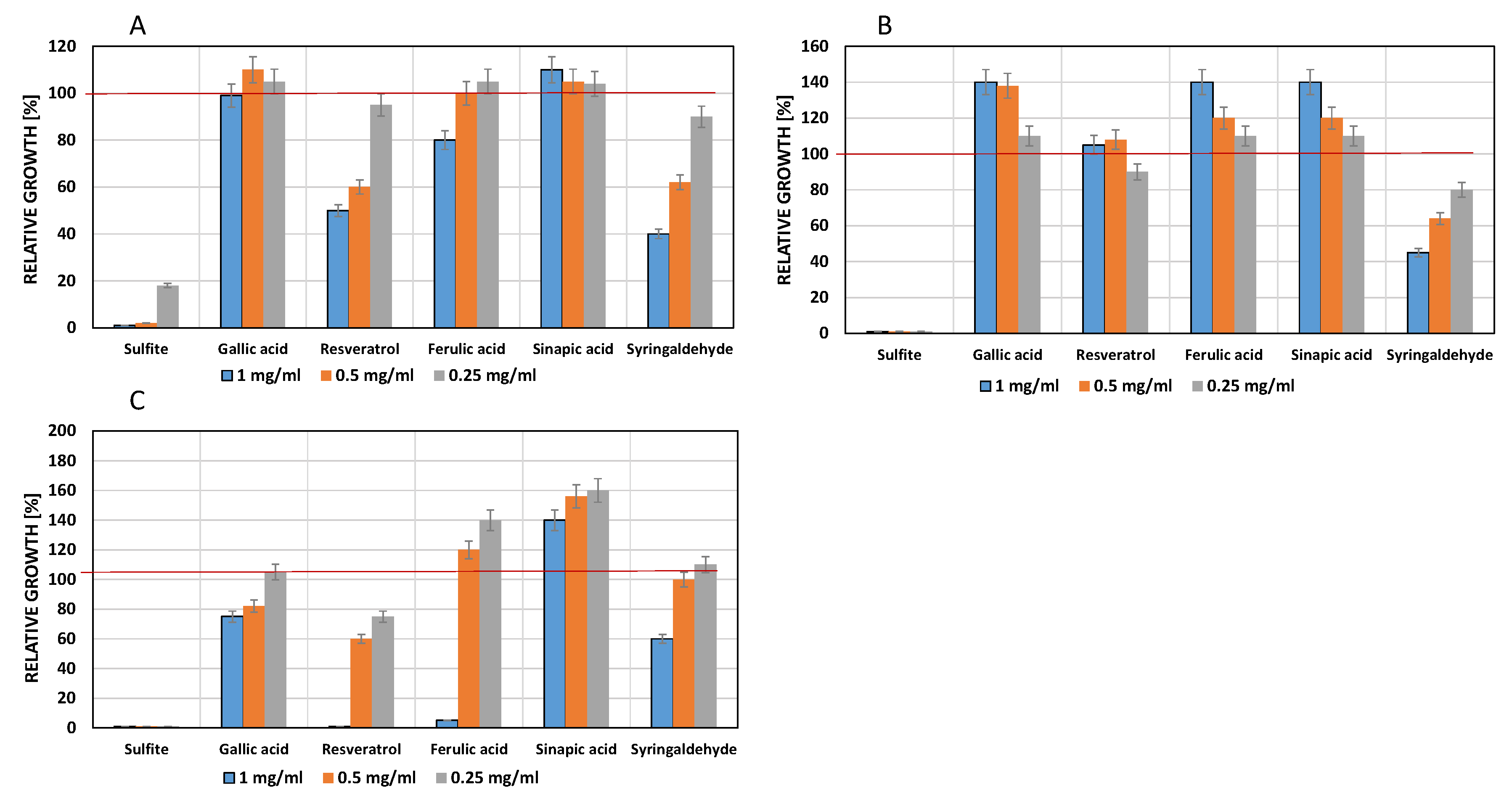
3.2. Influence of Phenols on Yeast Growth
3.3. Influence of Phenols on Growth of Lactic Acid Bacteria
3.4. Influence of Phenols on Growth of Acetic Acid Bacteria
3.5. Effects of Enzymatic Oxidation
4. Discussion
4.1. Influence of Phenolic Compounds on Yeast Growth
4.2. Influence of Phenolic Compounds on the Growth of Lactic Acid Bacteria
4.3. Influence of Phenolic Compounds on the Growth of Acetic Acid Bacteria
4.4. Mode of Antimicrobial Action
4.5. Enyzmatic Oxidations
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- König, H.; Unden, F.; Fröhlich, J. Biology of Microorganisms on Grapes, in Must and in Wine; Springer: Berlin/Heidelberg, Germany, 2009. [Google Scholar]
- Campos, F.M.; Couto, J.A.; Hogg, T. Utilisation of natural and by-products to improve wine safety. In Wine Safety, Consumer Preference, and Human Health; Moreno-Arribas, M.V., Bartolomé Sualdea, B., Eds.; Springer Int. Publ.: Cham, Switzerland, 2016; pp. 27–49. [Google Scholar]
- Pozo-Bayón, M.A.; Monagas, M.; Bartolomé, B.; Moreno-Arribas, M.V. Wine features related to safety and consumer health: An integrated perspective. Crit. Rev. Food Sci. Nutr. 2012, 52, 31–54. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulou, C.; Soulti, K.; Roussis, I.G. Potential antimicrobial activity of red and white wine phenolic extracts against strains of Staphylococcus aureus, Escherichia coli and Candida albicans. Food Technol. Biotechnol. 2005, 43, 41–46. [Google Scholar]
- Cueva, C.; Mingo, S.; Muňoz-González, I.; Bustos, I.; Requena, T.; Del Campo, R.; Martín-Álvarez, P.J.; Bartolomé, B.; Moreno-Arribas, M.V. Antibacterial activity of wine phenolic compounds and oenological extracts against potential respiratory pathogens. Lett. Appl. Microbiol. 2012, 54, 557–563. [Google Scholar] [CrossRef] [PubMed]
- Katalinic, V.; Smole Mozina, S.; Generalic, I.; Skroza, D.; Ljubenkov, I.; Klancik, A. Phenolic profile, antioxidant capacity, and antimicrobial activity of leaf extracts from six Vitis. vinifera L. varieties. Int. J. Food Prop. 2013, 16, 45–60. [Google Scholar] [CrossRef]
- Xu, C.; Yagiz, Y.; Zhao, L.; Simonne, A.; Lu, J.; Marshall, M.R. Fruit quality, nutraceutical and antimicrobial properties of 58 muscadine grape varieties (Vitis. rotindifolia Michx.) grown in United States. Food Chem. 2017, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Landete, J.M. Updated knowledge about polyphenols: Functions, bioavailability, metabolism, and health. Crit. Rev. Food Sci. Nutr. 2012, 52, 936–948. [Google Scholar] [CrossRef] [PubMed]
- Dietrich, H.; Pour-Nikfardjam, M.S. Influence of phenolic compounds and tannins on wine-related microorganisms. In Biology of Microorganisms on Grapes, in Must and in Wine; König, H., Unden, F., Fröhlich, J., Eds.; Springer: Berlin/Heidelberg, Germany, 2009; pp. 307–334. [Google Scholar]
- García-Ruiz, A.; Moreno-Arribas, M.V.; Martín-Álvarez, P.J.; Bartolomé, B. Comparative study of the inhibitory effects of wine polyphenols on the growth of enological lactic acid bacteria. Int. J. Food Microbiol. 2011, 145, 426–431. [Google Scholar] [CrossRef] [PubMed]
- García-Ruiz, A.; Cueva, C.; Gonzales-Rampinelli, E.M.; Yuste, M.; Torres, M.; Martin-Alvarez, P.J.; Bartolomé, B.; Moreno-Arribas, M.V. Antimicrobial phenolic extracts able to inhibit lactic acid bacteria growth. Food Control 2012, 28, 212–219. [Google Scholar] [CrossRef]
- Pastorkowa, E.; Zakova, T.; Landa, P.; Navakova, J.; Vadlejch, J.; Kokoska, L. Growth inhibitory effect of grape phenolics against wine spoilage yeast and acetic acid bacteria. Int. J. Food Microb. 2013, 161, 209–213. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Rompinelli, E.M.; Rodriguez-Bencomo, J.J.; Garcia-Ruiz, A.; Sanchez-Patan, F.; Martin-Alvarez, P.J.; Bartolome, B.; Morena-Arribas, M. A winery-scale trial of the use of antimicrobial plant phenolic extracts as preservatives during wine ageing in barrels. Food Control 2013, 33, 440–447. [Google Scholar] [CrossRef]
- Sabel, A.; Martens, S.; Petri, A.; König, H.; Claus, H. Wickerhamomyces anomalus AS1: A new strain with potential to improve wine aroma. Ann. Microbiol. 2014, 64, 483–491. [Google Scholar] [CrossRef]
- Christ, E.; Kowalczyk, M.; Zuchowska, M.; Claus, H.; Löwenstein, R.; Szopinska-Morawska, A.; Renaut, J.; König, H. An exemplary model study for overcoming stuck fermentation during spontaneous fermentation with the aid of a Saccharomyces triple hybrid. J. Agric. Sci. 2015, 7, 18–34. [Google Scholar] [CrossRef]
- Zuchowska, M.; Jaenicke, E.; König, H.; Claus, H. Allelic variants of hexose transporter Hxt3p and hexokinases Hxk1p/Hxk2p in strains of Saccharomyces cerevisiae and interspecies hybrids. Yeast 2015, 32, 657–669. [Google Scholar] [CrossRef] [PubMed]
- Bäumlisberger, M.; Moellecken, U.; König, H.; Claus, H. The potential of the yeast Debaryomyces hansenii H525 to degrade biogenic amines in food. Microorganisms 2015, 3, 839–850. [Google Scholar] [CrossRef] [PubMed]
- Riebel, M.; Sabel, A.; Claus, H.; Xia, N.; Li, H.; König, H.; Decker, H.; Fronk, P. Antioxidant capacity of phenolic compounds on human cell lines as affected by grape-tyrosinase and Botrytis-laccase oxidation. Food Chem. 2017, 229, 779–789. [Google Scholar] [CrossRef] [PubMed]
- Sadowska-Bartosz, I.; Pączka, A.; Molon, M.; Bartosz, G. Dimethyl sulfoxid induces oxidative stress in the yeast Saccharomyces cerevisiae. FEMS Yeast Res. 2013, 13, 820–830. [Google Scholar] [CrossRef] [PubMed]
- Claus, H.; Sabel, A.; König, H. Wine Phenols and Laccase: An ambivalent relationship. In Wine Phenolic Composition, Classification and Health Benefits; El Rayess, E.Y., Ed.; Nova publishers: New York, NY, USA, 2014; pp. 155–185. [Google Scholar]
- Dittrich, H.H.; Großmann, M. Mikrobiologie des Weines 4. Auflage; Verlag Eugen Ulmer: Stuttgart, Germany, 2011. [Google Scholar]
- Harris, V.; Jiranek, V.; Ford, C.M.; Grbin, P.R. Inhibitory effect of hydroxycinnamic acids on Dekkera spp. Appl. Microbiol. Biotechnol. 2010, 86, 721–729. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, M.N.M.; Sriprasanthi, R.B.; Shamsudeen, S.; Adam, F.; Bhawani, S.A. A concise review of the natural existence, synthesis, properties and applications of syringaldehyde. BioResources 2012, 7, 4377–4399. [Google Scholar]
- Kelly, C.; Jones, O.; Barnhart, C.; Lajoie, C. Effect of furfural, vanillin and syringaldehyde on Candida guilliermondii: Growth and xylitol biosynthesis. Appl. Biochem. Biotechnol. 2008, 148, 97–108. [Google Scholar] [CrossRef] [PubMed]
- Stead, D. The effect of hydroxycinnamic acids on the growth of wine-spoilage lactic acid bacteria. J. Appl. Bact. 1993, 75, 135–141. [Google Scholar] [CrossRef]
- Rozès, N.; Peres, C. Effects of phenolic compounds on the growth and fatty acid composition of Lactobacillus plantarum. Appl. Microbiol. Biotechnol. 1998, 49, 108–111. [Google Scholar] [CrossRef]
- Salih, A.G.; Le Quéré, J.M.; Drilleau, J.F. Effect of hydroxycinnamic acids on the growth of lactic bacteria. Sci. Aliment. 2000, 20, 537–560. [Google Scholar] [CrossRef]
- Campos, F.M.; Couto, J.A.; Hogg, T. Influence of phenolic acids on growth and inactivation of Oenococcus oeni and Lactobacillus hilgardii. J. Appl. Microbiol. 2003, 94, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Stead, D. The effect of chlorogenic, gallic and quinic acids on the growth of spoilage strains of Lactobacillus collinoides and Lactobacillus brevis. Lett. Appl. Microbiol. 1994, 112–114. [Google Scholar] [CrossRef]
- Vivas, N.; Lonvaud-Funel, A.; Glories, Y. Effect of phenolic acids and anthoyanins on growth, viability and malolactic activity of a lactic acid bacterium. Food Microbiol. 1997, 291–299. [Google Scholar] [CrossRef]
- Alberto, M.R.; Farías, M.E.; Manca de Nadra, M.C. Effect of gallic acid and catechin on Lactobacillus hilgardii 5w growth and metabolism of organic compounds. J. Agric. Food Chem. 2001, 49, 4359–4363. [Google Scholar] [CrossRef] [PubMed]
- Alberto, M.R.; Gómez-Cordovés, C.; Manca de Nadra, M.C. Metabolism of gallic acid and catechin on Lactobacillus hilgardii from wine. J. Agric. Food Chem. 2004, 52, 6465–6469. [Google Scholar] [CrossRef] [PubMed]
- Theobald, S.; Pfeiffer, P.; Zuber, U.; König, H. Influence of epigallocatchin gallate and phenolic compounds from green tea on the growth of Oenococcus oeni. J. Appl. Microbiol. 2008, 104, 566–672. [Google Scholar] [CrossRef] [PubMed]
- González de Llano, D.; Gil-Sánchez, I.; Esteban-Fernández, A.; Ramos, A.M.; Cueva, C.; Moreno-Arribas, M.V.; Bartolomé, B. Some contributions to the study of oenological lactic acid bacteria through their interaction with polyphenols. Beverages 2016, 2, 27. [Google Scholar] [CrossRef]
- Landete, J.M.; Rodríguez, H.; de las Rivas, B.; Munoz, R. High-added-value antioxidants obtained from the degradation of wine phenolic by Lactobacillus plantarum. J. Food. Prot. 2007, 70, 2670–2675. [Google Scholar] [CrossRef] [PubMed]
- Guillamón, J.M.; Mas, A. Acetic acid bacteria. In Molecular Wine Microbiology; Carrascosa, A.V., Muñoz, R., González, R., Eds.; Elsevier: London, UK, 2011; pp. 227–255. [Google Scholar]
- Buchert, J.; Niemelä, K. Oxidative detoxification of wood-derived inhibitors by Gluconobacter oxydans. J. Biotechnol. 1991, 18, 1–12. [Google Scholar] [CrossRef]
- Amborabé, B.E.; Fleurat-Lessard, P.; Chollet, J.F.; Roblin, G. Antifungal effects of salicylic acid and other benzoic derivates towards Eutypa lata: Structure-activity relationship. Plant Phys. Biochem. 2002, 40, 1051–1060. [Google Scholar] [CrossRef]
- Rodríguez, H.; Curiel, J.A.; Landete, J.M.; de las Rivas, B.; de Filipe, F.L.; Gómez-Cordovés, C.; Mancheño, J.M.; Muñoz, R. Food phenolics and lactic acid bacteria. Int. J. Food. Microbiol. 2009, 132, 79–90. [Google Scholar] [CrossRef] [PubMed]
- Suart, J.A.; Robb, E.A. Bioactive Polyphenols from Wine Grapes; Springer: New York, NY, USA, 2013. [Google Scholar]
- El Rayess, Y. Wine–Phenolic Composition, Classification and Health Benefits; Nova Publishers: New York, NY, USA, 2014. [Google Scholar]
- Yang, S.C.; Tseng, C.H.; Wang, P.W.; Lu, P.L.; Weng, Y.H.; Yen, F.L.; Fang, J.Y. Pterostilbene, a methoxylated resveratrol derivate, efficiently eradicates planctonic, biofilm, and intracellular MRSA by topical application. Front. Microbiol. 2017, 8, 1103. [Google Scholar] [CrossRef]
- Fernandes, M.S.; Kerkar, S. Microorganisms as a source of tyrosinase inhibitors: A review. Ann. Microbiol. 2017, 67, 343–358. [Google Scholar] [CrossRef]
- Pourcel, L.; Routaboul, J.M.; Cheynier, V.; Lepiniec, L.; Debeaujon, I. Flavanoid oxidation in plants: From biochemical properties to physiological functions. Trends Plant Sci. 2006, 12, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Kumar, N.; Pruthi, V. Potential applications of ferulic acid from natural sources. Biotechnol. Rep. 2014, 4, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Schwentke, J.; Sabel, A.; Petri, A.; König, H.; Claus, H. The yeast Wickerhamomyces. anomalus AS1 secretes a multifunctional exo-β-1,3-glucanase with implications for winemaking. Yeast 2014, 31, 349–359. [Google Scholar] [CrossRef] [PubMed]
- García-Ruiz, A.; Rodríguez-Bencomo, J.J.; Garrido, I.; Martín-Álvarez, M.; Moreno-Arribas, M.V.; Bartolomé, B. Assessment of the impact of the additions of antimicrobial plant extracts to wine: Volatile and phenolic composition. J. Sci. Food Agric. 2013, 93, 2507–2516. [Google Scholar] [CrossRef] [PubMed]


| Chemical Class | Representative Structures | Examples | Mean Contents [µg/mL] 1 in | |
|---|---|---|---|---|
| Red Wine | White Wine | |||
| Hydroxybenzoic acids |  | Gallic acid | 35.9 | 2.2 |
| Vanillic acid | 3.2 | 0.4 | ||
| 4-Hydroxybenzoic acid | 5.5 | 0.2 | ||
| Syringic acid | 2.7 | <0.01 | ||
| Ethylgallate | 15.3 | nd | ||
| Cinnamic acids |  | p-Coumaric acid | 5.5 | 1.5 |
| Caffeic acid | 18.8 | 2.4 | ||
| Ferulic acid | 0.8 | 0.9 | ||
| Sinapic acid | 0.7 | 0.6 | ||
| Stilbenes |  | cis-Resveratrol | 1.3 | 0.2 |
| Trans-Resveratrol | 1.8 | 0.4 | ||
| trans-Resveratrol-3-O-glucoside | 4.1 | 1.7 | ||
| Hydroxybenz-aldehydes |  | Syringaldehyde | 6.6 | <0.01 |
| Species | Caffeic Acid | Gallic Acid | Resveratrol | Polydatin | 3,4-di-HB | Ferulic Acid | Sinapic Acid | Syring-Aldehyde | Ethyl-gallate |
|---|---|---|---|---|---|---|---|---|---|
| S. cerevisiae | +4.29 | −3.98 | +2.30 | −103.00 | +3.94 | +5.51 | −9.37 | +9.63 | +4.36 |
| S. bayanus | +24.23 | +7.77 | ±0 | −106.00 | −3.72 | +3.98 | −7.00 | +2.07 | −5.77 |
| D. hansenii | +7.11 | −15.58 | −4.74 | −14.29 | −19.94 | +1.04 | −4.12 | −8.75 | −32.30 |
| W. anomalus | +9.19 | −0.89 | +6.17 | −88.13 | −9.36 | −4.98 | −7.12 | −1.58 | −5.37 |
| L. hilgardii | +5.01 | +3.71 | −8.37 | +15.57 | +7.99 | +1.49 | +21.93 | +0.49 | +3.29 |
| L. plantarum | +10.00 | +2.36 | −4.41 | −11.17 | −7.94 | +8.77 | +23.12 | −2.14 | +5.06 |
| P. parvulus | +1.51 | +8.16 | ± 0 | +0.68 | +16.01 | +8.34 | +37.18 | −31.65 | +29.22 |
| O. oeni | +19.21 | +115.74 | +9.79 | +40.63 | +47.38 | +12.20 | +25.37 | +3.01 | +30.15 |
| (A) Yeasts 1 | |||||
| Compound | S. cerevisiae | S. bayanus | S. c. × S. k. × S. b. | W. anomalus | D. hansenii |
| Caffeic acid | >1000 | >1000 | nd | >1000 | >1000 |
| p-Coumaric acid | >1000 | 500 | 1000 | 1000 | >1000 |
| Ferulic acid | 1000 | 250–500 | 1000 | 1000 | 500 |
| Sinapic acid | >1000 | >1000 | nd | >1000 | 1000 |
| Gallic acid | >1000 | >1000 | 1000 | >1000 | >1000 |
| Vanillic acid | >1000 | 1000 | 1000 | >1000 | >1000 |
| Hydroxybenzoic acid | >1000 | >1000 | nd | >1000 | 1000 |
| Syringaldehyde | 250–1000 | 250–500 | 250 | 250 | 250–1000 |
| Resveratrol | 250 | 250 | 250 | 250–500 | 250 |
| Polydatin | >1000 | >1000 | nd | >1000 | >1000 |
| Potassium sulfite | 250–500 | 500–1000 | 500 | 250 | 250 |
| (B) Lactic Acid Bacteria | |||||
| Compound | L. hilgardii | L. plantarum | P. parvulus | O. oeni | |
| Caffeic acid | 250 | >1000 | 250 | 250 | |
| Ferulic acid | 250 | 250 | 250 | 250 | |
| Sinapic acid | 250 | 250 | 250 | 250 | |
| Gallic acid | >1000 | >1000 | >1000 | >1000 | |
| Hydroxybenzoic acid | 250 | >1000 | 1000 | 500 | |
| Syringaldehyde | 250 | 250 | 1000 | 250 | |
| Resveratrol | 250 | 250 | 250 | 250 | |
| Polydatin | >1000 | >1000 | >1000 | >1000 | |
| Potassium sulfite | 250 | 250 | 250 | 250 | |
| (C) Acetic Acid Bacteria | |||||
| Compound | G. cerinus | A. acetii | |||
| Ferulic acid | >1000 | 1000 | |||
| Sinapic acid | >1000 | >1000 | |||
| Gallic acid | >1000 | 1000 | |||
| Syringaldehyde | 250 | 1000 | |||
| Resveratrol | >1000 | 250 | |||
| Potassium sulfite | 250 | 250 | |||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sabel, A.; Bredefeld, S.; Schlander, M.; Claus, H. Wine Phenolic Compounds: Antimicrobial Properties against Yeasts, Lactic Acid and Acetic Acid Bacteria. Beverages 2017, 3, 29. https://doi.org/10.3390/beverages3030029
Sabel A, Bredefeld S, Schlander M, Claus H. Wine Phenolic Compounds: Antimicrobial Properties against Yeasts, Lactic Acid and Acetic Acid Bacteria. Beverages. 2017; 3(3):29. https://doi.org/10.3390/beverages3030029
Chicago/Turabian StyleSabel, Andrea, Simone Bredefeld, Martina Schlander, and Harald Claus. 2017. "Wine Phenolic Compounds: Antimicrobial Properties against Yeasts, Lactic Acid and Acetic Acid Bacteria" Beverages 3, no. 3: 29. https://doi.org/10.3390/beverages3030029
APA StyleSabel, A., Bredefeld, S., Schlander, M., & Claus, H. (2017). Wine Phenolic Compounds: Antimicrobial Properties against Yeasts, Lactic Acid and Acetic Acid Bacteria. Beverages, 3(3), 29. https://doi.org/10.3390/beverages3030029





