QbD Based Media Development for the Production of Fab Fragments in E. coli
Abstract
1. Introduction
2. Materials and Methods
2.1. Strain and Genetic Construct
2.2. Chemicals and Media
2.3. Screening of Growth Media
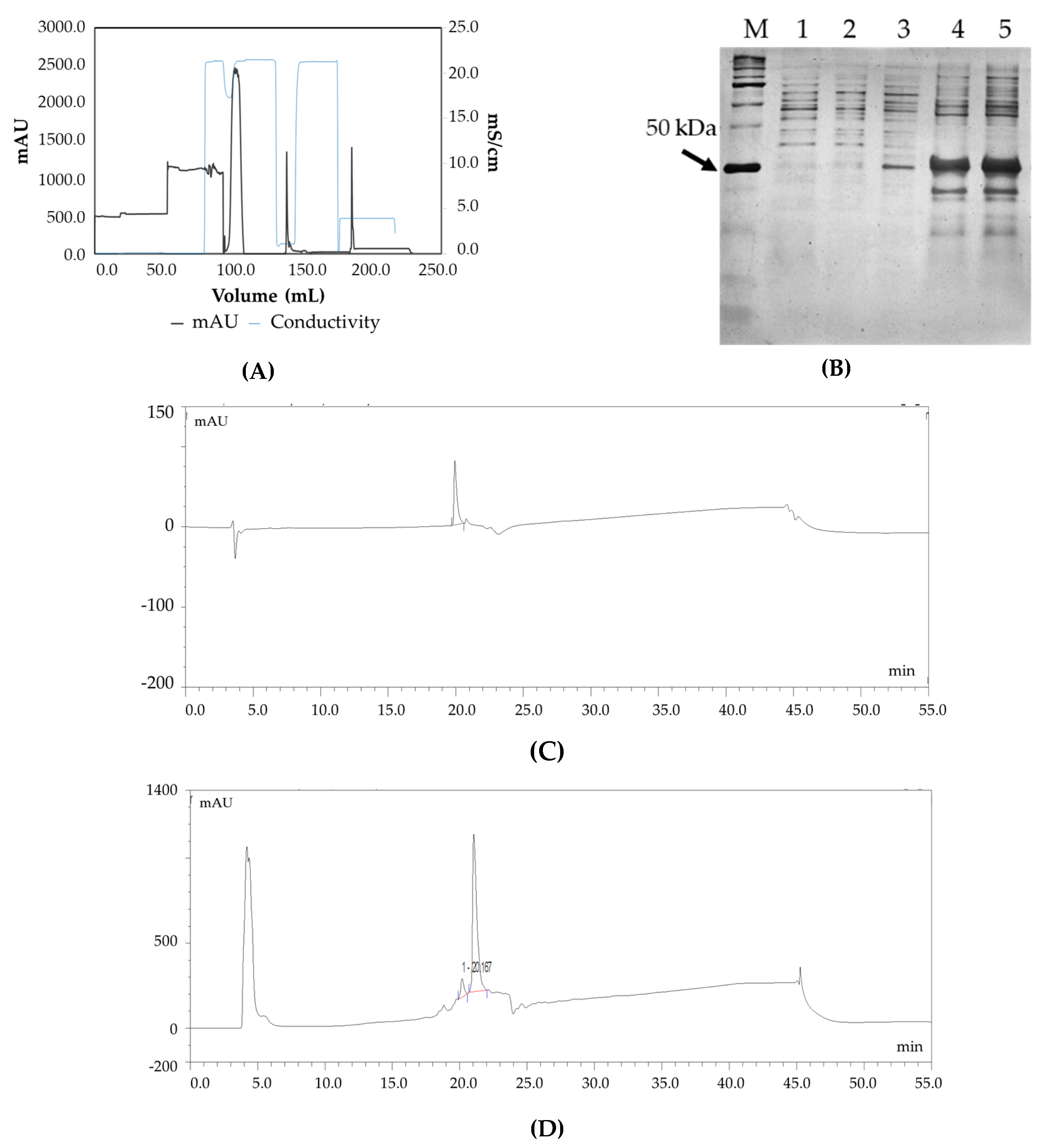
2.4. Purification of Fab Using Capto L Resin
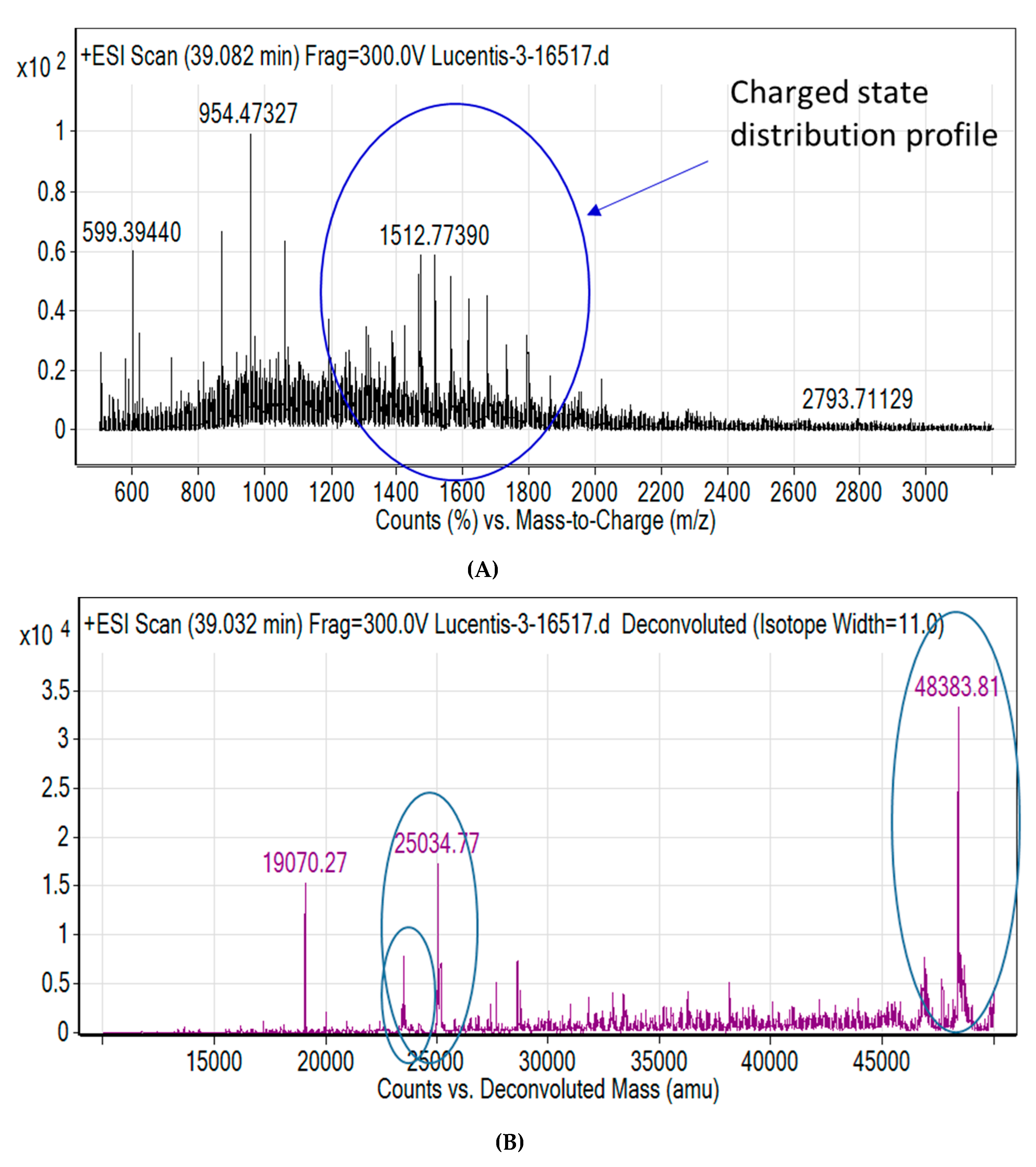
2.5. Liquid Chromatography-Mass Spectrometry (LC-MS) of Intact Fab
2.6. Screening of Process Parameters
2.7. Screening of Various Medium Components Using OFAT Approach
2.8. Fractional Factorial Design Based Two-Level Screening of Selected Medium Components
2.9. Periplasmic Protein Extraction
2.10. Fab Quantification by SDS PAGE
2.11. Fab Quantification by Reversed-Phase High-Performance Liquid Chromatography (RP-HPLC)
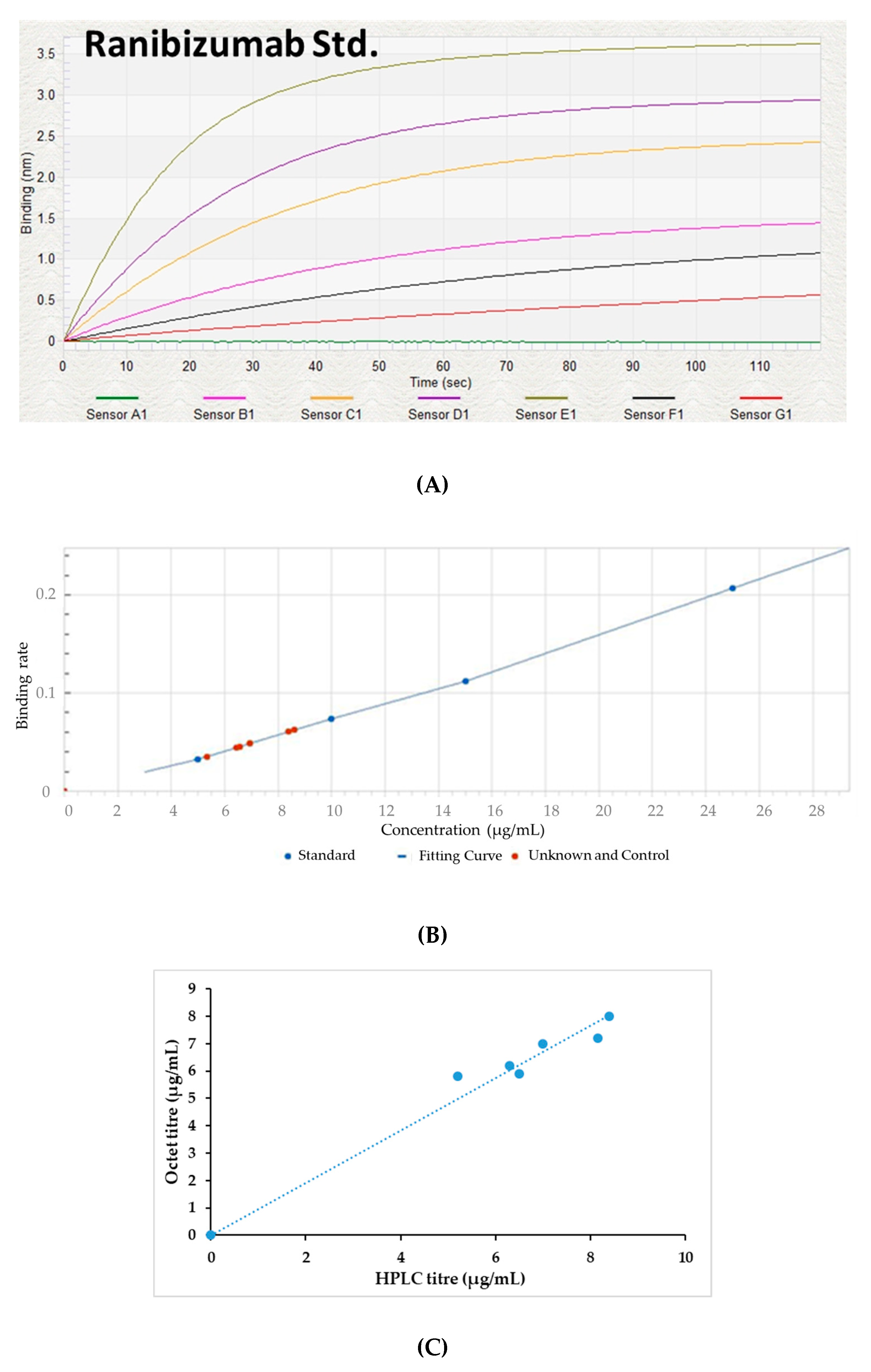
2.12. Fab Quantification by BLI
3. Results
3.1. Screening of Basal Growth Media
3.2. Confirmation of Product Formation
3.3. Fab Quantification Using BLI
3.4. Effect of Process Parameters
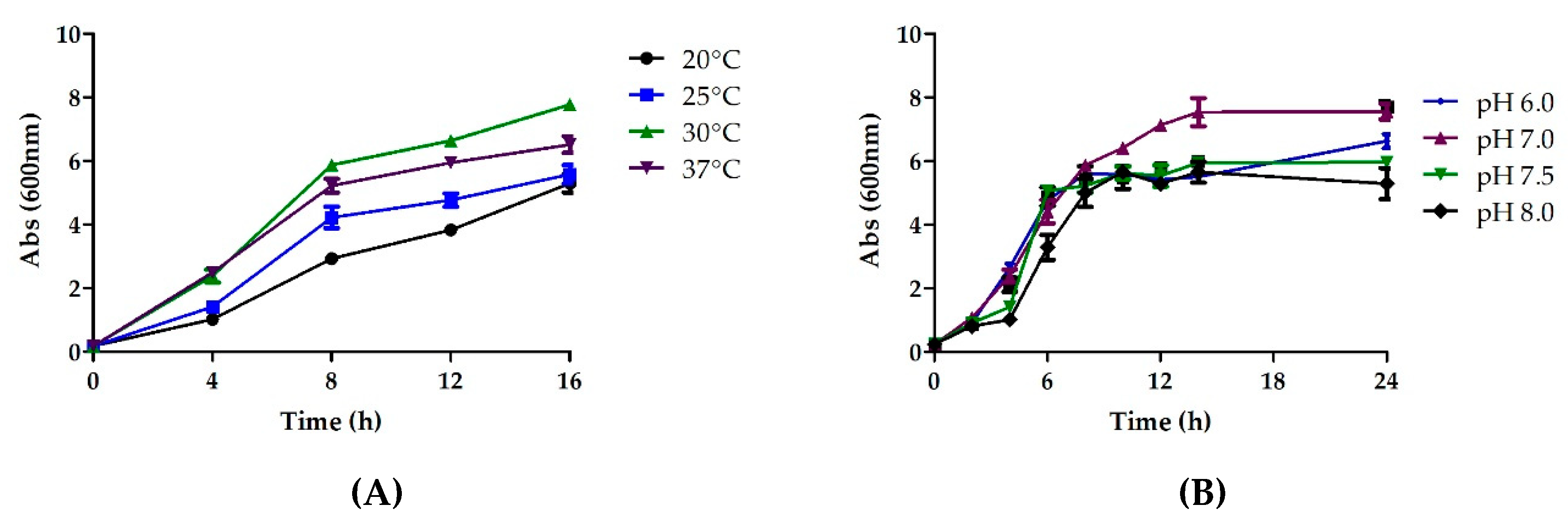
3.4.1. Effect of Temperature
3.4.2. Effect of pH
3.4.3. Effect of Inducer Concentration
3.4.4. Effect of Agitation
3.5. One Factor at a Time (OFAT) Experimentation for Screening of Media Components
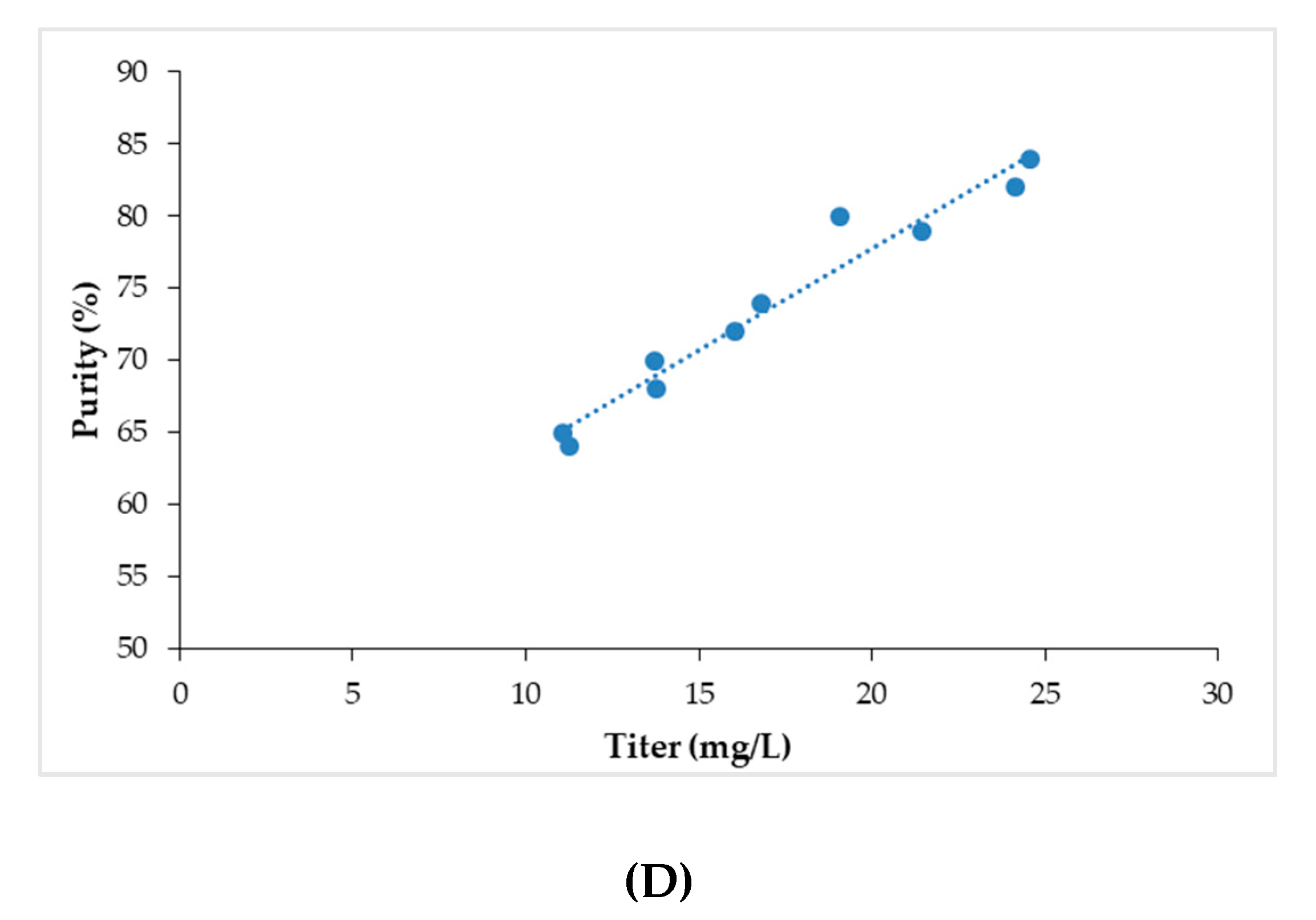
3.6. Screening of Additives using Fractional Factorial DOE
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Walsh, G. Biopharmaceutical benchmarks 2014. Nat. Biotechnol. 2014, 32, 992–1000. [Google Scholar] [CrossRef] [PubMed]
- Rathore, A.S.; Batra, J. Antibody Production in Microbial Hosts. BioPharm Int. 2016, 28, 18–23. [Google Scholar]
- Zhang, K.; Zhang, L.; Weinreb, R.N. Ophthalmic drug discovery: Novel targets and mechanisms for retinal diseases and glaucoma. Nat. Rev. Drug Discov. 2012, 11, 541–559. [Google Scholar] [CrossRef] [PubMed]
- Mullard, A. Can next-generation antibodies offset biosimilar competition? Nat. Rev. Drug Discov. 2012, 11, 426–428. [Google Scholar] [CrossRef] [PubMed]
- Million, R.P. Therapeutic area crossroads: Anti-angiogenesis. Nat. Rev. Drug Discov. 2008, 7, 115–116. [Google Scholar] [CrossRef]
- Rathore, A.S. Follow-on protein products: Scientific issues, developments and challenges. Trends Biotechnol. 2009, 27, 698–705. [Google Scholar] [CrossRef]
- Thiry, M.; Cingolani, D. Optimizing scale-up fermentation processes. Trends Biotechnol. 2002, 20, 103–105. [Google Scholar] [CrossRef]
- Yee, L.; Blanch, H.W. Defined media optimization for growth of recombinant Escherichia coli X90. Biotechnol. Bioeng. 1993, 41, 221–230. [Google Scholar] [CrossRef]
- Ukkonen, K.; Veijola, J.; Vasala, A.; Neubauer, P. Effect of culture medium, host strain and oxygen transfer on recombinant Fab antibody fragment yield and leakage to medium in shaken E. coli cultures. Microb. Cell Fact. 2013, 12, 73. [Google Scholar] [CrossRef]
- Eiteman, M.A.; Altman, E. Overcoming acetate in Escherichia coli recombinant protein fermentations. Trends Biotechnol. 2006, 24, 530–536. [Google Scholar] [CrossRef]
- Kipriyanov, S.M.; Moldenhauer, G.; Little, M. High level production of soluble single chain antibodies in small-scale Escherichia coli cultures. J. Immunol. Methods 1997, 200, 69–77. [Google Scholar] [CrossRef]
- Rathore, A.S. Quality by design (QbD)-based process development for purification of a biotherapeutic. Trends Biotechnol. 2016, 34, 358–370. [Google Scholar] [CrossRef] [PubMed]
- Rathore, A.S. QbD/PAT for bioprocessing: Moving from theory to implementation. Curr. Opin. Chem. Eng. 2014, 6, 1–8. [Google Scholar] [CrossRef]
- Rathore, A.S.; Winkle, H. Quality by design for biopharmaceuticals. Nat. Biotechnol. 2009, 27, 26. [Google Scholar] [CrossRef] [PubMed]
- ICH Harmonised Tripartite Guideline. Pharmaceutical development. Q8 (2R). As Revis, 23 August 2009.
- Rathore, A.S. Roadmap for implementation of quality by design (QbD) for biotechnology products. Trends Biotechnol. 2009, 27, 546–553. [Google Scholar] [CrossRef]
- Saida, F.; Uzan, M.; Odaert, B.; Bontems, F. Expression of highly toxic genes in E. coli: Special strategies and genetic tools. Curr. Protein Pept. Sci. 2006, 7, 47–56. [Google Scholar] [CrossRef]
- King, D.J.; Byron, O.D.; Mountain, A.; Weir, N.; Harvey, A.; Lawson, A.D.G.; Proudfoot, K.A.; Baldock, D.; Harding, S.E.; Yarranton, G.T.; et al. Expression, purification and characterization of B72. 3 Fv fragments. Biochem. J. 1993, 290, 723–729. [Google Scholar] [CrossRef]
- Cossins, A.J.; Harrison, S.; Popplewell, A.G.; Gore, M.G. Recombinant production of a VL single domain antibody in Escherichia coli and analysis of its interaction with peptostreptococcal protein L. Protein Expr. Purif. 2007, 51, 253–259. [Google Scholar] [CrossRef]
- Sletta, H.; Nedal, A.; Aune, T.E.V.; Hellebust, H.; Hakvåg, S.; Aune, R.; Ellingsen, T.E.; Valla, S.; Brautaset, T. Broad-host-range plasmid pJB658 can be used for industrial-level production of a secreted host-toxic single-chain antibody fragment in Escherichia coli. Appl. Environ. Microbiol. 2004, 70, 7033–7039. [Google Scholar] [CrossRef]
- Zhang, C.; Chen, X.; Zou, R.; Zhou, K.; Stephanopoulos, G.; Too, H.-P. Combining genotype improvement and statistical media optimization for isoprenoid production in E. coli. PLoS ONE 2013, 8, e75164. [Google Scholar] [CrossRef]
- Champion, K.M.; Nishihara, J.C.; Joly, J.C.; Arnott, D. Similarity of the Escherichia coli proteome upon completion of different biopharmaceutical fermentation processes. Proteomics 2001, 1, 1133–1148. [Google Scholar] [CrossRef]
- Riesenberg, D.; Schulz, V.; Knorre, W.A.; Pohl, H.-D.; Korz, D.; Sanders, E.A.; Ross, A.; Deckwer, W.-D. High cell density cultivation of Escherichia coli at controlled specific growth rate. J. Biotechnol. 1991, 20, 17–27. [Google Scholar] [CrossRef]
- Kumar, V.; Bhalla, A.; Rathore, A.S. Design of experiments applications in bioprocessing: Concepts and approach. Biotechnol. Prog. 2014, 30, 86–99. [Google Scholar] [CrossRef] [PubMed]
- Erdag, B.; Koray Balcioglu, B.; Ozdemir Bahadir, A.; Serhatli, M.; Kacar, O.; Bahar, A.; Seker, U.O.S.; Akgun, E.; Ozkan, A.; Kilic, T.; et al. Identification of novel neutralizing single-chain antibodies against vascular endothelial growth factor receptor 2. Biotechnol. Appl. Biochem. 2011, 58, 412–422. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Nilson, B.H.K.; Lögdberg, L.; Kastern, W.; Björck, L.; Åkerström, B. Purification of antibodies using protein L-binding framework structures in the light chain variable domain. J. Immunol. Methods 1993, 164, 33–40. [Google Scholar] [CrossRef]
- Rathore, A.S.; Kumar, D.; Batra, J.; Krull, I. Biolayer Interferometry as an Alternative to HPLC for measuring Product Concentration in Fermentation Broth. LC GC N. Am. 2017, 35, 870–877. [Google Scholar]
- Shokri, A.; Sandén, A.; Larsson, G. Cell and process design for targeting of recombinant protein into the culture medium of Escherichia coli. Appl. Microbiol. Biotechnol. 2003, 60, 654–664. [Google Scholar] [CrossRef]
- Sørensen, H.P.; Mortensen, K.K. Advanced genetic strategies for recombinant protein expression in Escherichia coli. J. Biotechnol. 2005, 115, 113–128. [Google Scholar] [CrossRef]
- Huang, C.-J.; Lin, H.; Yang, X. Industrial production of recombinant therapeutics in Escherichia coli and its recent advancements. J. Ind. Microbiol. Biotechnol. 2012, 39, 383–399. [Google Scholar] [CrossRef]
- Carter, P.; Kelley, R.F.; Rodrigues, M.L.; Snedecor, B.; Covarrubias, M.; Velligan, M.D.; Wong, W.L.T.; Rowland, A.M.; Kotts, C.E.; Carver, M.E.; et al. High level Escherichia coli expression and production of a bivalent humanized antibody fragment. Nat. Biotechnol. 1992, 10, 163–167. [Google Scholar] [CrossRef]
- Dragosits, M.; Frascotti, G.; Bernard-Granger, L.; Vázquez, F.; Giuliani, M.; Baumann, K.; Rodríguez-Carmona, E.; Tokkanen, J.; Parrilli, E.; Wiebe, M.G.; et al. Influence of growth temperature on the production of antibody Fab fragments in different microbes: A host comparative analysis. Biotechnol. Prog. 2011, 27, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Cabilly, S. Growth at sub-optimal temperatures allows the production of functional, antigen-binding Fab fragments in Escherichia coli. Gene 1989, 85, 553–557. [Google Scholar] [CrossRef]
- Gasser, B.; Saloheimo, M.; Rinas, U.; Dragosits, M.; Rodríguez-Carmona, E.; Baumann, K.; Giuliani, M.; Parrilli, E.; Branduardi, P.; Lang, C.; et al. Protein folding and conformational stress in microbial cells producing recombinant proteins: A host comparative overview. Microb. Cell Fact. 2008, 7, 11. [Google Scholar] [CrossRef] [PubMed]
- Frenzel, A.; Hust, M.; Schirrmann, T. Expression of recombinant antibodies. Front. Immunol. 2013, 4, 217. [Google Scholar] [CrossRef] [PubMed]
- Wilms, B.; Hauck, A.; Reuss, M.; Syldatk, C.; Mattes, R.; Siemann, M.; Altenbuchner, J. High-cell-density fermentation for production of L-N-carbamoylase using an expression system based on the Escherichia coli rhaBAD promoter. Biotechnol. Bioeng. 2001, 73, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Jeske, M.; Altenbuchner, J. The Escherichia coli rhamnose promoter rhaPBAD is in Pseudomonas putida KT2440 independent of Crp–cAMP activation. Appl. Microbiol. Biotechnol. 2010, 85, 1923–1933. [Google Scholar] [CrossRef]
- Wegerer, A.; Sun, T.; Altenbuchner, J. Optimization of an E. coli L-rhamnose-inducible expression vector: Test of various genetic module combinations. BMC Biotechnol. 2008, 8, 2. [Google Scholar] [CrossRef]
- Singh, S.; Du Preez, J.C.; Pillay, B.; Prior, B.A. The production of hemicellulases by Thermomyces lanuginosus strain SSBP: Influence of agitation and dissolved oxygen tension. Appl. Microbiol. Biotechnol. 2000, 54, 698–704. [Google Scholar] [CrossRef]
- Carlson, H.K.; Stoeva, M.K.; Justice, N.B.; Sczesnak, A.; Mullan, M.R.; Mosqueda, L.A.; Kuehl, J.V.; Deutschbauer, A.M.; Arkin, A.P.; Coates, J.D. Monofluorophosphate is a selective inhibitor of respiratory sulfate-reducing microorganisms. Environ. Sci. Technol. 2015, 49, 3727–3736. [Google Scholar] [CrossRef]
- Rock, C.O.; Calder, R.B.; Karim, M.A.; Jackowski, S. Pantothenate kinase regulation of the intracellular concentration of coenzyme A. J. Biol. Chem. 2000, 275, 1377–1383. [Google Scholar] [CrossRef] [PubMed]
- Gande, R.; Dover, L.G.; Krumbach, K.; Besra, G.S.; Sahm, H.; Oikawa, T.; Eggeling, L. The two carboxylases of Corynebacterium glutamicum essential for fatty acid and mycolic acid synthesis. J. Bacteriol. 2007, 189, 5257–5264. [Google Scholar] [CrossRef] [PubMed]
- Schäffner, J.; Winter, J.; Rudolph, R.; Schwarz, E. Cosecretion of chaperones and low-molecular-size medium additives increases the yield of recombinant disulfide-bridged proteins. Appl. Environ. Microbiol. 2001, 67, 3994–4000. [Google Scholar] [CrossRef] [PubMed]
- Papaneophytou, C.; Kontopidis, G. A Comparison of Statistical Approaches Used for the Optimization of Soluble Protein Expression in Escherichia coli; Elsevier: Amsterdam, The Netherlands, 2016. [Google Scholar]
- Rosano, G.L.; Ceccarelli, E.A. Recombinant protein expression in Escherichia coli: Advances and challenges. Front. Microbiol. 2014, 5, 172. [Google Scholar] [CrossRef] [PubMed]
- Mandenius, C.-F.; Brundin, A. Bioprocess optimization using design-of-experiments methodology. Biotechnol. Prog. 2008, 24, 1191–1203. [Google Scholar] [CrossRef]



| Medium | Components |
|---|---|
| Cossins Medium | (NH4)2SO4 (5.2 g/L), NaH2PO4 (4.15 g/L), KCl (4.025 g/L), Citric Acid (5.2 g/L), Glycerol (93.33 g/L), MgSO4.7H2O (1.04 g/L), Thiamine Hydrochloride (6.67 mM) and Trace Elements * |
| Sletta Medium | KH2PO4 (16.6 g/L), (NH4)2HPO4 (4.0 g/L), Citric Acid (2.1 g/L), Glucose (20 g/L), MgSO4.7H2O (1.04 g/L) and Trace Elements * |
| King Medium | (NH4)2SO4 (5.2 g/L) NaH2PO4 (4.15 g/L) Yeast Extract (5 g/L), Glycerol, MgSO4.7H2O (1.04 g/L) and Trace Elements * |
| Zhang Medium | Glycerol (20 g/L), KH2PO4 (20 g/L), (NH4)2HPO4 (5 g/L), Na (Pyruvate) (5 g/L), MgSO4.7H2O (1.04 g/L), Thiamine Hydrochloride (6.67 mM) and Trace Elements * |
| Champion Medium | (NH4)2SO4 (55.7 mM), NaH2PO4 (13.9 mM), K2HPO4 (21.9 mM), Sodium Citrate (5 mM), KCl (29.6 mM), MgSO4.7H2O (14.7 mM), NZ Amine AS (1.11%), Yeast Extract (1.11%), Glucose (0.11%) and Trace Elements * |
| Riesenberg Medium | Glucose (27.5 g/L), KH2PO4 (13.3 g/L), (NH4)2HPO4 (4.0 g/L), Citric Acid (1.7 g/L), MgSO4.7H2O (1.04 g/L), Thiamine Hydrochloride (4.5 mg/L) and Trace Elements * |
| S. No. | Variables | Lower Level | Higher Level |
|---|---|---|---|
| 1 | K2HPO4 (%) | 0.5 | 1.2 |
| 2 | Biotin (mg/L) | 0.5 | 2 |
| 3 | Glutathione (mM) | 5 | 20 |
| 4 | Sucrose (%) | 0.5 | 2 |
| 5 | Potassium Sulfate (%) | 0.09 | 0.36 |
| 6 | Calcium Sulfate (%) | 0.05 | 0.2 |
| 7 | Panthothenic Acid (mg/L) | 0.5 | 2 |
| 8 | Cyanocobalamin (mg/L) | 0.7 | 2.8 |
| S. No. | Media Additives | Concentration | Protein Titre (mg/L) |
|---|---|---|---|
| 1 | CoSO4 (g/L) | 0.75 | 1.24 |
| 2 | Di-sodium succinate (%) | 1.00 | 2.10 |
| 3 | NaEDTA (mg/L) | 10.50 | 2.21 |
| 4 | Nickel sulphate (%) | 0.25 | 2.35 |
| 5 | Al2(SO4)3 (g/L) | 2.00 | 2.75 |
| 6 | NaCl (%) | 1.00 | 3.34 |
| 7 | Yeast extract (%) | 1.00 | 4.50 |
| 8 | Peptone (%) | 2.00 | 4.79 |
| 9 | Glycine (%) | 0.20 | 4.90 |
| 10 | NH4CI (%) | 0.20 | 5.22 |
| 11 | FeSO4 (%) | 0.10 | 5.73 |
| 12 | (NH4)2SO4 (%) | 0.40 | 5.95 |
| 13 | L-Isoleucine (g/L) | 0.20 | 6.75 |
| 14 | FeCl2 (%) | 2.70 | 6.95 |
| 15 | L-Proline (g/L) | 0.20 | 7.30 |
| 16 | NaHCO3 (%) | 1.00 | 7.65 |
| 17 | ZnCl2 (%) | 0.20 | 7.80 |
| 18 | Panthothenic Acid (mg/L) | 1.00 | 8.30 |
| 19 | Calcium sulfate (%) | 0.10 | 8.86 |
| 20 | Potassium sulfate (%) | 0.18 | 8.88 |
| 21 | Biotin (mg/L) | 1.00 | 10.05 |
| 22 | Sucrose (%) | 1.00 | 10.65 |
| 23 | Cyanocobalamin (mg/L) | 1.40 | 12.54 |
| 24 | K2HPO4 (%) | 0.12 | 13.80 |
| 25 | Glutathione (mM) | 10.00 | 16.80 |
| 26 | Control (no additive) | - | 8.00 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumar, D.; Batra, J.; Komives, C.; Rathore, A.S. QbD Based Media Development for the Production of Fab Fragments in E. coli. Bioengineering 2019, 6, 29. https://doi.org/10.3390/bioengineering6020029
Kumar D, Batra J, Komives C, Rathore AS. QbD Based Media Development for the Production of Fab Fragments in E. coli. Bioengineering. 2019; 6(2):29. https://doi.org/10.3390/bioengineering6020029
Chicago/Turabian StyleKumar, Deepak, Jyoti Batra, Claire Komives, and Anurag S. Rathore. 2019. "QbD Based Media Development for the Production of Fab Fragments in E. coli" Bioengineering 6, no. 2: 29. https://doi.org/10.3390/bioengineering6020029
APA StyleKumar, D., Batra, J., Komives, C., & Rathore, A. S. (2019). QbD Based Media Development for the Production of Fab Fragments in E. coli. Bioengineering, 6(2), 29. https://doi.org/10.3390/bioengineering6020029