Advancing Microplastic and Nanoplastic Toxicity Assessment: Insights from Human Organoid Models
Abstract
1. Introduction
2. Human Exposure to MNPs

3. Human Organoids for Toxicity Assessment of MNPs
3.1. Human Organoids Applied for Assessing MP Toxicity
3.2. Human Organoids Applied for Assessing NP Toxicity
| Organoids | NP Type & Size | Exposure | Toxic Effects | Toxic Mechanism | References |
|---|---|---|---|---|---|
| Intestinal organoid | polystyrene (50 nm) | 2 days (10 and 100 µg/mL) | induced cellular apoptosis and inflammatory response | elevated NF- NF-κB, p65, IL-8, and ROS | [49] |
| Intestinal organoid | polystyrene, polytetrafluoroethylene, and polymethyl methacrylate (100 nm) | 3 days (50 µg/mL) | reduced mitochondrial membrane potential, intracellular ROS accumulation and oxidative stress | inhibited the AKT/mTOR signaling pathway | [57] |
| Lung organoid | polystyrene (40 nm) | 12, 24, 48, and 72 h (100 µg/mL) | restricted organoid growth and caused oxidative damage | increased expression of Cldn4 and decreased expression of Sftpc and Pdpn | [66] |
| Lung organoid | polystyrene (250 nm) | 14 days (1 and 10 µg/cm2) | the internalization of PS-NPs | increased phosphorylation of both AKT and ERK | [67] |
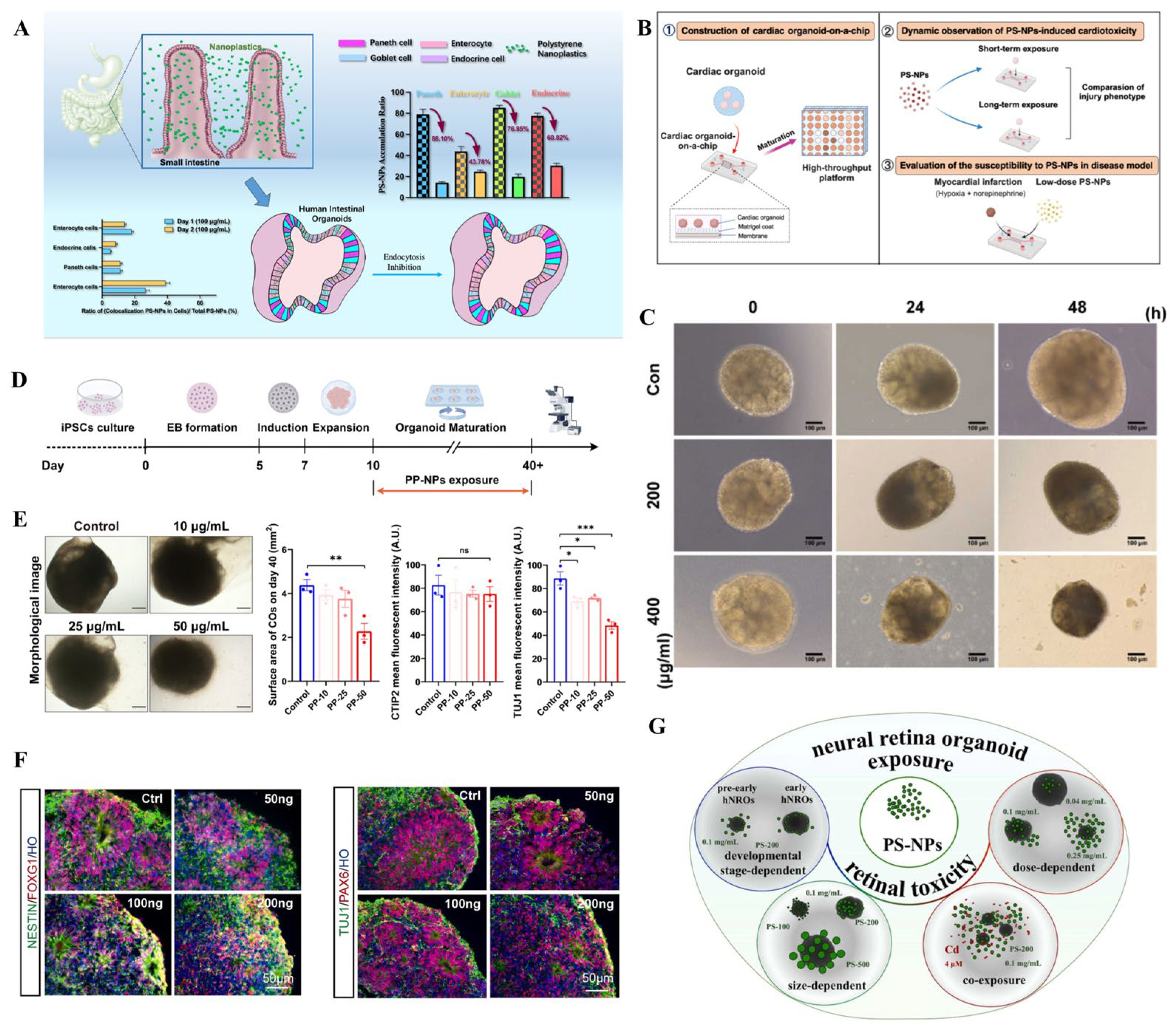
| Cardiac organoid | polystyrene (40 nm) | 10 days (30–150 μg/mL) | induced oxidative stress, inflammation, disruption of calcium ion homeostasis, and mitochondrial dysfunction | activated pathways associated with collagen and extracellular matrix dynamics | [64] |
| Cardiac organoid | polystyrene (20 nm) | 24 h (5 or 20 μg/mL) | disrupted the efficiency of cardiomyocyte differentiation; impaired contractility | involved in autophagy and ROS/p38/Erk MAPK signaling pathways | [65] |
| Cerebral organoid | polypropylene (100 nm) | day 10–40 (10, 25, and 50 µg/mL) | reduced growth and neuronal differentiation; downregulated neuronal markers | disrupted neuroactive ligand- receptor interaction pathway | [68] |
| Cerebral organoids | polystyrene (<50 nm) | day 16 to day 24 (50, 100, 200 ng/mL) | increased cell death; decreased cell differentiation and neuronal activity | induced mitochondrial impairment; disrupted neuronal calcium activity | [69] |
| Brain organoid | polystyrene (50 nm) | 7 days (50 and 100 µg/mL) | inhibited neuronal synaptogenesis | induced endoplasmic reticulum stress; disrupted cholesterol homeostasis | [70] |
| Retinal organoid | polystyrene (100, 200, and 500 nm) | 14 days (0.04, 0.1, and 0.25 mg/mL) | decreased organoid size, reduced cell proliferation, increased apoptosis, and altered gene expression profiles | disrupted axon guidance, anatomical structure development, differentiation, and neurogenesis | [71] |
| Kidney organoid | polystyrene (100 nm) | 48 h (200, 400, and 800 μg/mL) | Inhibited organoid growth; significant cell detachment | disrupted proliferation and differentiation | [72] |
4. Current Limitations and Future Perspectives
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kozlov, M. Landmark study links microplastics to serious health problems. Nature 2024. [Google Scholar]
- Vethaak, A.D.; Legler, J. Microplastics and human health. Science 2021, 371, 672–674. [Google Scholar] [CrossRef] [PubMed]
- van der Laan, L.J.W.; Bosker, T.; Peijnenburg, W. Deciphering potential implications of dietary microplastics for human health. Nat. Rev. Gastroenterol. Hepatol. 2023, 20, 340–341. [Google Scholar] [CrossRef]
- Huang, W.; Xia, X. Element cycling with micro(nano)plastics. Science 2024, 385, 933–935. [Google Scholar] [CrossRef]
- Nasir, M.S.; Tahir, I.; Ali, A.; Ayub, I.; Nasir, A.; Abbas, N.; Sajjad, U.; Hamid, K. Innovative technologies for removal of micro plastic: A review of recent advances. Heliyon 2024, 10, e25883. [Google Scholar] [CrossRef]
- Thompson, R.C.; Courtene-Jones, W.; Boucher, J.; Pahl, S.; Raubenheimer, K.; Koelmans, A.A. Twenty years of microplastics pollution research-what have we learned? Science 2024, 386, eadl2746. [Google Scholar] [CrossRef]
- Seewoo, B.J.; Goodes, L.M.; Thomas, K.V.; Rauert, C.; Elagali, A.; Ponsonby, A.L.; Symeonides, C.; Dunlop, S.A. How do plastics, including microplastics and plastic-associated chemicals, affect human health? Nat. Med. 2024, 30, 3036–3037. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Hou, Z.; Meng, R.; Hao, S.; Wang, B. Unraveling the potential human health risks from used disposable face mask-derived micro/nanoplastics during the COVID-19 pandemic scenario: A critical review. Environ. Int. 2022, 170, 107644. [Google Scholar] [CrossRef] [PubMed]
- Zaini, N.; Kasmuri, N.; Mojiri, A.; Kindaichi, T.; Nayono, S.E. Plastic pollution and degradation pathways: A review on the treatment technologies. Heliyon 2024, 10, e28849. [Google Scholar] [CrossRef] [PubMed]
- Eze, C.G.; Nwankwo, C.E.; Dey, S.; Sundaramurthy, S.; Okeke, E.S. Food chain microplastics contamination and impact on human health: A review. Environ. Chem. Lett. 2024, 22, 1889–1927. [Google Scholar] [CrossRef]
- Abafe, O.A.; Harrad, S.; Abdallah, M.A. Novel Insights into the Dermal Bioaccessibility and Human Exposure to Brominated Flame Retardant Additives in Microplastics. Environ. Sci. Technol. 2023, 57, 10554–10562. [Google Scholar] [CrossRef] [PubMed]
- Song, G.B.; Nam, J.; Ji, S.; Woo, G.; Park, S.; Kim, B.; Hong, J.; Choi, M.G.; Kim, S.; Lee, C.; et al. Deciphering the links: Fragmented polystyrene as a driver of skin inflammation. J. Hazard. Mater. 2024, 480, 135815. [Google Scholar] [CrossRef] [PubMed]
- Akpojevwe Abafe, O.; Harrad, S.; Abou-Elwafa Abdallah, M. Assessment of human dermal absorption of flame retardant additives in polyethylene and polypropylene microplastics using 3D human skin equivalent models. Environ. Int. 2024, 186, 108635. [Google Scholar] [CrossRef]
- Leslie, H.A.; van Velzen, M.J.M.; Brandsma, S.H.; Vethaak, A.D.; Garcia-Vallejo, J.J.; Lamoree, M.H. Discovery and quantification of plastic particle pollution in human blood. Environ. Int. 2022, 163, 107199. [Google Scholar] [CrossRef]
- Schwabl, P.; Köppel, S.; Königshofer, P.; Bucsics, T.; Trauner, M.; Reiberger, T.; Liebmann, B. Detection of Various Microplastics in Human Stool: A Prospective Case Series. Ann. Intern. Med. 2019, 171, 453–457. [Google Scholar] [CrossRef] [PubMed]
- Qiu, L.; Lu, W.; Tu, C.; Li, X.; Zhang, H.; Wang, S.; Chen, M.; Zheng, X.; Wang, Z.; Lin, M.; et al. Evidence of Microplastics in Bronchoalveolar Lavage Fluid among Never-Smokers: A Prospective Case Series. Environ. Sci. Technol. 2023, 57, 2435–2444. [Google Scholar] [CrossRef]
- Li, Y.; Chen, L.; Zhou, N.; Chen, Y.; Ling, Z.; Xiang, P. Microplastics in the human body: A comprehensive review of exposure, distribution, migration mechanisms, and toxicity. Sci. Total Environ. 2024, 946, 174215. [Google Scholar] [CrossRef]
- Zhu, Y.; Che, R.; Zong, X.; Wang, J.; Li, J.; Zhang, C.; Wang, F. A comprehensive review on the source, ingestion route, attachment and toxicity of microplastics/nanoplastics in human systems. J. Environ. Manag. 2024, 352, 120039. [Google Scholar] [CrossRef]
- Nihart, A.J.; Garcia, M.A.; El Hayek, E.; Liu, R.; Olewine, M.; Kingston, J.D.; Castillo, E.F.; Gullapalli, R.R.; Howard, T.; Bleske, B.; et al. Bioaccumulation of microplastics in decedent human brains. Nat. Med. 2025, 31, 1114–1119. [Google Scholar] [CrossRef]
- Zhang, M.; Liu, S.; Wang, Y.; Ge, Y.; Li, X.; Wang, X.; Zhuang, S.; Cui, H. Detection and Characterization of Multiple Microplastics in the Human Retina. Environ. Sci. Technol. Lett. 2025, 12, 1327–1333. [Google Scholar] [CrossRef]
- Yan, Z.; Liu, Y.; Zhang, T.; Zhang, F.; Ren, H.; Zhang, Y. Analysis of Microplastics in Human Feces Reveals a Correlation between Fecal Microplastics and Inflammatory Bowel Disease Status. Environ. Sci. Technol. 2022, 56, 414–421. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Feng, Y.; Wang, R.; Jiang, J.; Guan, Q.; Yang, X.; Wei, H.; Xia, Y.; Luo, Y. Pigment microparticles and microplastics found in human thrombi based on Raman spectral evidence. J. Adv. Res. 2023, 49, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Marfella, R.; Prattichizzo, F.; Sardu, C.; Fulgenzi, G.; Graciotti, L.; Spadoni, T.; D’Onofrio, N.; Scisciola, L.; La Grotta, R.; Frigé, C.; et al. Microplastics and Nanoplastics in Atheromas and Cardiovascular Events. N. Engl. J. Med. 2024, 390, 900–910. [Google Scholar] [CrossRef]
- Feng, Y.; Tu, C.; Li, R.; Wu, D.; Yang, J.; Xia, Y.; Peijnenburg, W.; Luo, Y. A systematic review of the impacts of exposure to micro- and nano-plastics on human tissue accumulation and health. Eco-Environ. Health 2023, 2, 195–207. [Google Scholar] [CrossRef]
- Liang, J.; Ji, F.; Abdullah, A.L.B.; Qin, W.; Zhu, T.; Tay, Y.J.; Li, Y.; Han, M. Micro/nano-plastics impacts in cardiovascular systems across species. Sci. Total Environ. 2024, 942, 173770. [Google Scholar] [CrossRef]
- Zhu, L.; Xie, C.; Chen, L.; Dai, X.; Zhou, Y.; Pan, H.; Tian, K. Transport of microplastics in the body and interaction with biological barriers, and controlling of microplastics pollution. Ecotoxicol. Environ. Saf. 2023, 255, 114818. [Google Scholar] [CrossRef]
- Kögel, T.; Bjorøy, Ø.; Toto, B.; Bienfait, A.M.; Sanden, M. Micro- and nanoplastic toxicity on aquatic life: Determining factors. Sci. Total Environ. 2020, 709, 136050. [Google Scholar] [CrossRef]
- Prüst, M.; Meijer, J.; Westerink, R.H.S. The plastic brain: Neurotoxicity of micro- and nanoplastics. Part. Fibre Toxicol. 2020, 17, 24. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.; Li, Y.; Lee, M.; Andrikopoulos, N.; Lin, S.; Chen, C.; Leong, D.T.; Ding, F.; Song, Y.; Ke, P.C. Anionic nanoplastic exposure induces endothelial leakiness. Nat. Commun. 2022, 13, 4757. [Google Scholar] [CrossRef] [PubMed]
- Bai, J.; Wang, Y.; Deng, S.; Yang, Y.; Chen, S.; Wu, Z. Microplastics caused embryonic growth retardation and placental dysfunction in pregnant mice by activating GRP78/IRE1α/JNK axis induced apoptosis and endoplasmic reticulum stress. Part. Fibre Toxicol. 2024, 21, 36. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.K.; Kumari, U.; Kumar, S. Impact of Microplastics on Pregnancy and Fetal Development: A Systematic Review. Cureus 2024, 16, e60712. [Google Scholar] [CrossRef]
- Kwak, J.I.; Rhee, H.; Kim, L.; An, Y.J. In vivo visualization of environmentally relevant microplastics and evaluation of gut barrier damages in Artemia franciscana. J. Hazard. Mater. 2024, 478, 135596. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhou, X.X.; Jiang, H.; Liu, W.; Chen, F.; Gardea-Torresdey, J.L.; Yan, B. In Vitro Toxicity and Modeling Reveal Nanoplastic Effects on Marine Bivalves. ACS Nano 2024, 18, 17228–17239. [Google Scholar] [CrossRef] [PubMed]
- Hong, W.; Zhang, M.; Wang, F.; Li, Z.; Li, Z.; Wei, J.; Song, M.; Qian, Y.; Pang, S.; Wang, L.; et al. Special Distribution of Nanoplastics in the Central Nervous System of Zebrafish during Early Development. ACS Nano 2024, 18, 17509–17520. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Chen, X.; Li, M.; Cai, Z.; Gong, H.; Yan, M. Microplastics as an aquatic pollutant affect gut microbiota within aquatic animals. J. Hazard. Mater. 2022, 423, 127094. [Google Scholar] [CrossRef]
- Trestrail, C.; Walpitagama, M.; Hedges, C.; Truskewycz, A.; Miranda, A.; Wlodkowic, D.; Shimeta, J.; Nugegoda, D. Foaming at the mouth: Ingestion of floral foam microplastics by aquatic animals. Sci. Total Environ. 2020, 705, 135826. [Google Scholar] [CrossRef]
- Yuan, F.; Chen, H.; Ding, Y.; Wang, Y.; Liao, Q.; Wang, T.; Fan, Q.; Feng, Z.; Zhang, C.; Fu, G.; et al. Effects of microplastics on the toxicity of co-existing pollutants to fish: A meta-analysis. Water Res. 2023, 240, 120113. [Google Scholar] [CrossRef] [PubMed]
- Qiu, X.; Yang, D.; Yu, L.; Song, L.; Yang, L.; Yang, Q. Effect of polyethylene microplastics on tebuconazole bioaccumulation, oxidative stress, and intestinal bacterial community in earthworms. J. Hazard. Mater. 2024, 480, 136056. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Zhang, Y.; Lemos, B.; Ren, H. Tissue accumulation of microplastics in mice and biomarker responses suggest widespread health risks of exposure. Sci. Rep. 2017, 7, 46687. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Zhuan, Q.; Zhang, L.; Meng, L.; Fu, X.; Hou, Y. Polystyrene microplastics induced female reproductive toxicity in mice. J. Hazard. Mater. 2022, 424, 127629. [Google Scholar] [CrossRef]
- Gautam, R.; Jo, J.; Acharya, M.; Maharjan, A.; Lee, D.; Kc, P.; Kim, C.; Kim, K.; Kim, H.; Heo, Y. Evaluation of potential toxicity of polyethylene microplastics on human derived cell lines. Sci. Total Environ. 2022, 838, 156089. [Google Scholar] [CrossRef]
- Chen, Y.C.; Chen, K.F.; Andrew Lin, K.Y.; Su, H.P.; Wu, D.N.; Lin, C.H. Evaluation of toxicity of polystyrene microplastics under realistic exposure levels in human vascular endothelial EA.hy926 cells. Chemosphere 2023, 313, 137582. [Google Scholar] [CrossRef]
- Faiola, F.; Yin, N.; Yao, X.; Jiang, G. The rise of stem cell toxicology. Environ. Sci. Technol. 2015, 49, 5847–5848. [Google Scholar] [CrossRef]
- Hofer, M.; Lutolf, M.P. Engineering organoids. Nat. Rev. Mater. 2021, 6, 402–420. [Google Scholar] [CrossRef] [PubMed]
- Corsini, N.S.; Knoblich, J.A. Human organoids: New strategies and methods for analyzing human development and disease. Cell 2022, 185, 2756–2769. [Google Scholar] [CrossRef]
- Winkler, A.S.; Cherubini, A.; Rusconi, F.; Santo, N.; Madaschi, L.; Pistoni, C.; Moschetti, G.; Sarnicola, M.L.; Crosti, M.; Rosso, L.; et al. Human airway organoids and microplastic fibers: A new exposure model for emerging contaminants. Environ. Int. 2022, 163, 107200. [Google Scholar] [CrossRef]
- Song, S.; van Dijk, F.; Vasse, G.F.; Liu, Q.; Gosselink, I.F.; Weltjens, E.; Remels, A.H.V.; de Jager, M.H.; Bos, S.; Li, C.; et al. Inhalable Textile Microplastic Fibers Impair Airway Epithelial Differentiation. Am. J. Respir. Crit. Care Med. 2024, 209, 427–443. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Nicolas, G.M.; Cai, Y.; Dai, X.; Ma, S. Epidermal and dermal cell-composed organospheres to assess microplastic-induced skin toxicity. Biomaterials 2026, 324, 123513. [Google Scholar] [CrossRef]
- Hou, Z.; Meng, R.; Chen, G.; Lai, T.; Qing, R.; Hao, S.; Deng, J.; Wang, B. Distinct accumulation of nanoplastics in human intestinal organoids. Sci. Total Environ. 2022, 838, 155811. [Google Scholar] [CrossRef]
- Park, S.B.; Jung, W.H.; Choi, K.J.; Koh, B.; Kim, K.Y. A Comparative Systematic Analysis of The Influence of Microplastics on Colon Cells, Mouse and Colon Organoids. Tissue Eng. Regen. Med. 2023, 20, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Wu, Q.; Li, Y.; Feng, Y.; Wang, Y.; Cheng, W. Low-dose of polystyrene microplastics induce cardiotoxicity in mice and human-originated cardiac organoids. Environ. Int. 2023, 179, 108171. [Google Scholar] [CrossRef] [PubMed]
- Cheng, W.; Li, X.; Zhou, Y.; Yu, H.; Xie, Y.; Guo, H.; Wang, H.; Li, Y.; Feng, Y.; Wang, Y. Polystyrene microplastics induce hepatotoxicity and disrupt lipid metabolism in the liver organoids. Sci. Total Environ. 2022, 806, 150328. [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.; Zhang, A.; Wang, Y.; Feng, S.; Xue, Q.; Liu, Z.; Zhao, H.; Jing, Z.; Xie, J. Microplastics induce human kidney development retardation through ATP-mediated glucose metabolism rewiring. J. Hazard. Mater. 2025, 486, 137002. [Google Scholar] [CrossRef]
- Hua, T.; Kiran, S.; Li, Y.; Sang, Q.-X.A. Microplastics exposure affects neural development of human pluripotent stem cell-derived cortical spheroids. J. Hazard. Mater. 2022, 435, 128884. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Gao, X.; Lan, Y.; Pan, Y.; Yuan, Y.; Wu, Z.; Faiola, F.; Zhu, L.; Tang, J.; Gong, J.; et al. Revealing the neurodevelopmental toxicity of face mask-derived microplastics to humans based on neural organoids. J. Hazard. Mater. 2025, 492, 138084. [Google Scholar] [CrossRef]
- Xie, S.; Zhang, R.; Li, Z.; Liu, C.; Chen, Y.; Yu, Q. Microplastics perturb colonic epithelial homeostasis associated with intestinal overproliferation, exacerbating the severity of colitis. Environ. Res. 2023, 217, 114861. [Google Scholar] [CrossRef] [PubMed]
- Xuan, L.; Luo, J.; Qu, C.; Guo, P.; Yi, W.; Yang, J.; Yan, Y.; Guan, H.; Zhou, P.; Huang, R. Predictive metabolomic signatures for safety assessment of three plastic nanoparticles using intestinal organoids. Sci. Total Environ. 2024, 913, 169606. [Google Scholar] [CrossRef]
- Chen, Y.; Williams, A.M.; Gordon, E.B.; Rudolph, S.E.; Longo, B.N.; Li, G.; Kaplan, D.L. Biological effects of polystyrene micro- and nano-plastics on human intestinal organoid-derived epithelial tissue models without and with M cells. Nanomedicine 2023, 50, 102680. [Google Scholar] [CrossRef] [PubMed]
- Cheng, W.; Chen, H.; Zhou, Y.; You, Y.; Lei, D.; Li, Y.; Feng, Y.; Wang, Y. Aged fragmented-polypropylene microplastics induced ageing statues-dependent bioenergetic imbalance and reductive stress: In vivo and liver organoids-based in vitro study. Environ. Int. 2024, 191, 108949. [Google Scholar] [CrossRef] [PubMed]
- McLean, P.; Christopher, E.A.; Sleeuwenhoek, A.; Lofty, M.; Dixon, K.; Galea, K.S. Dermal exposure, review of current knowledge on the uptake of micro-and nano-plastics. Microplastics Nanoplastics 2025, 6, 12. [Google Scholar] [CrossRef]
- Sharma, V.K.; Ma, X.; Lichtfouse, E.; Robert, D. Nanoplastics are potentially more dangerous than microplastics. Environ. Chem. Lett. 2023, 21, 1933–1936. [Google Scholar] [CrossRef]
- Liu, L.; Xu, Y.; Ma, Y.; Duan, F.; Wang, C.; Feng, J.; Yin, H.; Sun, L.; Li, P.; Li, Z.-H. Fate of polystyrene micro- and nanoplastics in zebrafish liver cells: Influence of protein corona on transport, oxidative stress, and glycolipid metabolism. J. Hazard. Mater. 2025, 489, 137596. [Google Scholar] [CrossRef]
- Okkelman, I.A.; Zhou, H.; Borisov, S.M.; Debruyne, A.C.; Lefebvre, A.; Leomil Zoccoler, M.; Chen, L.; Devriendt, B.; Dmitriev, R.I. Visualizing the internalization and biological impact of nanoplastics in live intestinal organoids by Fluorescence Lifetime Imaging Microscopy (FLIM). Light Sci. Appl. 2025, 14, 272. [Google Scholar] [CrossRef]
- Zhang, T.; Yang, S.; Ge, Y.; Yin, L.; Pu, Y.; Gu, Z.; Chen, Z.; Liang, G. Unveiling the Heart’s Hidden Enemy: Dynamic Insights into Polystyrene Nanoplastic-Induced Cardiotoxicity Based on Cardiac Organoid-on-a-Chip. ACS Nano 2024, 18, 31569–31585. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Weng, H.; Liu, S.; Li, F.; Xu, K.; Wen, S.; Chen, X.; Li, C.; Nie, Y.; Liao, B.; et al. Embryonic exposure of polystyrene nanoplastics affects cardiac development. Sci. Total Environ. 2024, 906, 167406. [Google Scholar] [CrossRef]
- Yang, S.; Ge, Y.; Zhang, T.; Yin, L.; Pu, Y.; Chen, Z.; Liang, G. Dynamic non-coding RNA biomarker reveals lung injury and repair induced by polystyrene nanoplastics. Environ. Int. 2025, 195, 109266. [Google Scholar] [CrossRef] [PubMed]
- Ernhofer, B.; Spittler, A.; Ferk, F.; Mišík, M.; Zylka, M.M.; Glatt, L.; Boettiger, K.; Solta, A.; Kirchhofer, D.; Timelthaler, G.; et al. Small Particles, Big Problems: Polystyrene nanoparticles induce DNA damage, oxidative stress, migration, and mitogenic pathways predominantly in non-malignant lung cells. J. Hazard. Mater. 2025, 495, 139129. [Google Scholar] [CrossRef] [PubMed]
- Huang, F.; You, H.; Tang, X.; Su, Y.; Peng, H.; Li, H.; Wei, Z.; Hua, J. Early-life exposure to polypropylene nanoplastics induces neurodevelopmental toxicity in mice and human iPSC-derived cerebral organoids. J. Nanobiotechnol. 2025, 23, 474. [Google Scholar] [CrossRef]
- Tao, M.; Wang, C.; Zheng, Z.; Gao, W.; Chen, Q.; Xu, M.; Zhu, W.; Xu, L.; Han, X.; Guo, X.; et al. Nanoplastics exposure-induced mitochondrial dysfunction contributes to disrupted stem cell differentiation in human cerebral organoids. Ecotoxicol. Environ. Saf. 2024, 285, 117063. [Google Scholar] [CrossRef]
- Tian, L.; Wei, Y.; Ma, J.; Zhao, Y.; Mi, Y.; Zhang, L.; Wang, B.; Chen, J.; Li, K.; Shi, Y.; et al. Disruption of cerebral cholesterol homeostasis by PS-NPs: Astrocytic endoplasmic reticulum stress. J. Nanobiotechnol. 2025, 23, 790. [Google Scholar] [CrossRef]
- Gao, X.; Yuan, Y.; Lan, Y.; Lai, T.; Zhu, L.; Xu, L.; Gong, J.; Ma, N.; Wang, B.; Li, M. Polystyrene nanoplastics induced retinal toxicity: Size-, dose-, and developmental stage-dependent effects on human neural retina organoids. J. Hazard. Mater. 2025, 497, 139573. [Google Scholar] [CrossRef]
- Chen, L.; Han, B.; Yang, S.; Guo, L.; Zhao, L.; Liu, P.; Hong, X.; Zhao, Y.; Peng, Y.; Qi, S.; et al. Toxicological effects and mechanisms of renal injury induced by inhalation exposure to airborne nanoplastics. J. Hazard. Mater. 2025, 488, 137393. [Google Scholar] [CrossRef]
- Hernandez, L.M.; Xu, E.G.; Larsson, H.C.E.; Tahara, R.; Maisuria, V.B.; Tufenkji, N. Plastic Teabags Release Billions of Microparticles and Nanoparticles into Tea. Environ. Sci. Technol. 2019, 53, 12300–12310. [Google Scholar] [CrossRef]
- Zhang, T.; Wang, Z.; Wu, Y.; Zhu, S.; Su, J. Interactions of Micro- and Nanoplastics with Biomolecules: From Public Health to Protein Corona Effect and Beyond. J. Phys. Chem. B 2025, 129, 5355–5374. [Google Scholar] [CrossRef] [PubMed]
- Vilanova, O.; Martinez-Serra, A.; Monopoli, M.P.; Franzese, G. Characterizing the hard and soft nanoparticle-protein corona with multilayer adsorption. Front. Nanotechnol. 2025, 6, 1531039. [Google Scholar] [CrossRef]
- Fadare, O.O.; Wan, B.; Liu, K.; Yang, Y.; Zhao, L.; Guo, L.H. Eco-Corona vs Protein Corona: Effects of Humic Substances on Corona Formation and Nanoplastic Particle Toxicity in Daphnia magna. Environ. Sci. Technol. 2020, 54, 8001–8009. [Google Scholar] [CrossRef]
- Bu, N.; Du, Q.; Xiao, T.; Jiang, Z.; Lin, J.; Chen, W.; Fan, B.; Wang, J.; Xia, H.; Cheng, C.; et al. Mechanism of S-Palmitoylation in Polystyrene Nanoplastics-Induced Macrophage Cuproptosis Contributing to Emphysema through Alveolar Epithelial Cell Pyroptosis. ACS Nano 2025, 19, 18708–18728. [Google Scholar] [CrossRef] [PubMed]
- Du, T.; Yu, X.; Shao, S.; Li, T.; Xu, S.; Wu, L. Aging of Nanoplastics Significantly Affects Protein Corona Composition Thus Enhancing Macrophage Uptake. Environ. Sci. Technol. 2023, 57, 3206–3217. [Google Scholar] [CrossRef]
- Kihara, S.; Köper, I.; Mata, J.P.; McGillivray, D.J. Reviewing nanoplastic toxicology: It’s an interface problem. Adv. Colloid Interface Sci. 2021, 288, 102337. [Google Scholar] [CrossRef] [PubMed]
- Brouwer, H.; Busch, M.; Yang, S.; Venus, T.; Aalderink, G.; Crespo, J.F.F.; Villacorta, A.; Hernández, A.; Estrela-Lopis, I.; Boeren, S.; et al. Toxicity of true-to-life microplastics to human iPSC-derived intestinal epithelia correlates to their protein corona composition. J. Hazard. Mater. 2025, 495, 138908. [Google Scholar] [CrossRef]
- Nikonorova, V.G.; Chrishtop, V.V.; Mironov, V.A.; Prilepskii, A.Y. Advantages and Potential Benefits of Using Organoids in Nanotoxicology. Cells 2023, 12, 610. [Google Scholar] [CrossRef]
- Wang, H.; Li, X.; You, X.; Zhao, G. Harnessing the power of artificial intelligence for human living organoid research. Bioact. Mater. 2024, 42, 140–164. [Google Scholar] [CrossRef]
- Tenreiro, M.F.; Branco, M.A.; Cotovio, J.P.; Cabral, J.M.S.; Fernandes, T.G.; Diogo, M.M. Advancing organoid design through co-emergence, assembly, and bioengineering. Trends Biotechnol. 2023, 41, 923–938. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Gao, L.; Zhao, L.; Zou, T.; Xu, H. Toward the next generation of vascularized human neural organoids. Med. Res. Rev. 2023, 43, 31–54. [Google Scholar] [CrossRef]
- Li, M.; Gong, J.; Gao, L.; Zou, T.; Kang, J.; Xu, H. Advanced human developmental toxicity and teratogenicity assessment using human organoid models. Ecotoxicol. Environ. Saf. 2022, 235, 113429. [Google Scholar] [CrossRef]
- Ching, T.; Toh, Y.-C.; Hashimoto, M.; Zhang, Y.S. Bridging the academia-to-industry gap: Organ-on-a-chip platforms for safety and toxicology assessment. Trends Pharmacol. Sci. 2021, 42, 715–728. [Google Scholar] [CrossRef]
- Bai, L.; Wu, Y.; Li, G.; Zhang, W.; Zhang, H.; Su, J. AI-enabled organoids: Construction, analysis, and application. Bioact. Mater. 2024, 31, 525–548. [Google Scholar] [CrossRef] [PubMed]
- Yao, Q.; Cheng, S.; Pan, Q.; Yu, J.; Cao, G.; Li, L.; Cao, H. Organoids: Development and applications in disease models, drug discovery, precision medicine, and regenerative medicine. MedComm 2024, 5, e735. [Google Scholar] [CrossRef]
- Gerbolés, A.G.; Galetti, M.; Rossi, S.; Lo Muzio, F.P.; Pinelli, S.; Delmonte, N.; Caffarra Malvezzi, C.; Macaluso, C.; Miragoli, M.; Foresti, R. Three-Dimensional Bioprinting of Organoid-Based Scaffolds (OBST) for Long-Term Nanoparticle Toxicology Investigation. Int. J. Mol. Sci. 2023, 24, 6595. [Google Scholar] [CrossRef] [PubMed]
- Bouwmeester, M.C.; Bernal, P.N.; Oosterhoff, L.A.; van Wolferen, M.E.; Lehmann, V.; Vermaas, M.; Buchholz, M.B.; Peiffer, Q.C.; Malda, J.; van der Laan, L.J.W.; et al. Bioprinting of Human Liver-Derived Epithelial Organoids for Toxicity Studies. Macromol. Biosci. 2021, 21, e2100327. [Google Scholar] [CrossRef] [PubMed]
| Organoids | NP Type & Size | Exposure | Toxic Effects | Toxic Mechanism | References |
|---|---|---|---|---|---|
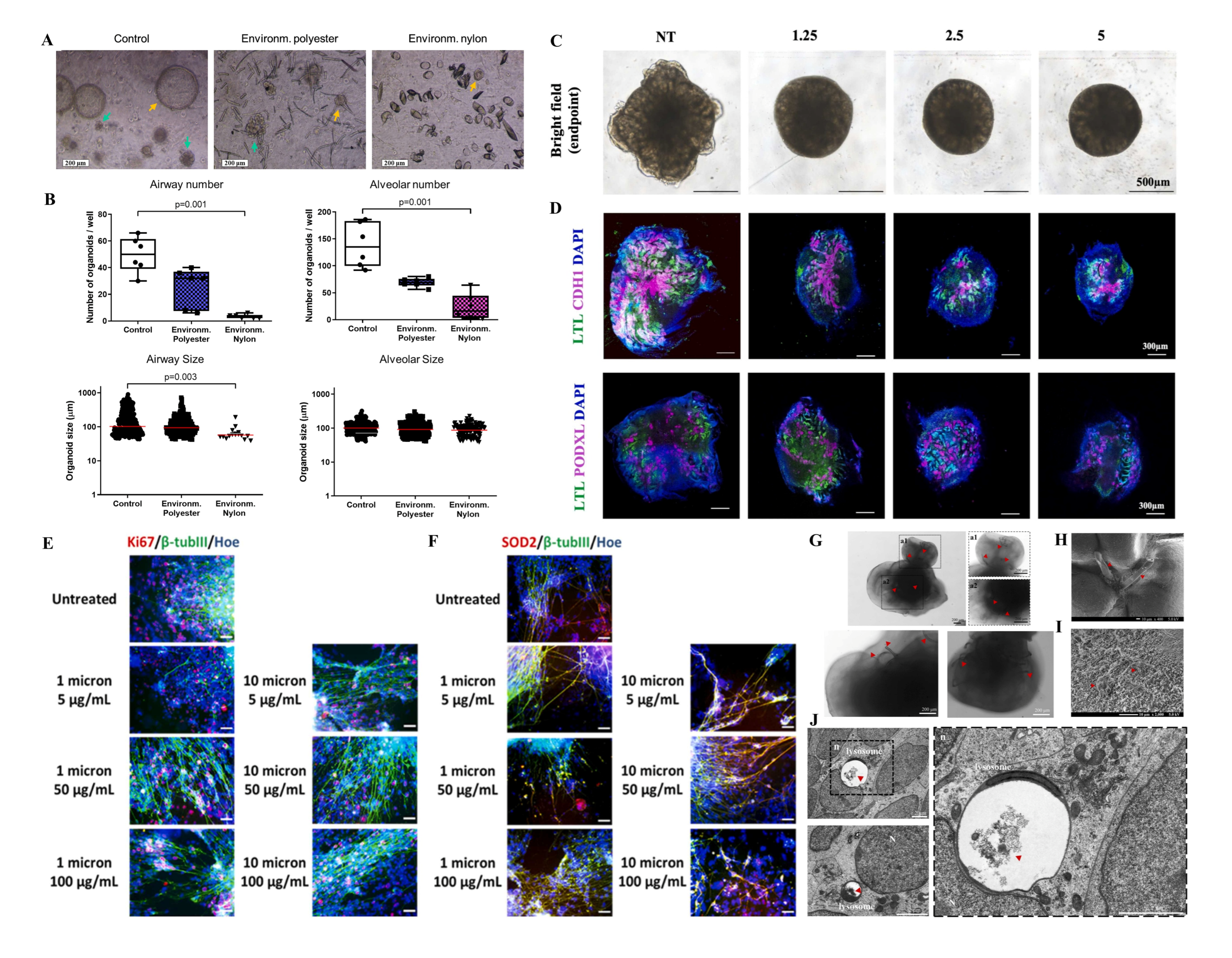
| Airway organoid | polyester fibers from the air filter of a dryer machine | 7 days (1, 10, and 50 µg/mL) | did not inhibit organoid growth | reduction of SCGB1A1 | [46] |
| Lung organoid | Nylon MPs (1–5 μm and 5–10 μm) | 7 days (16–39 μg/mL) | impaired organoid growth | upregulation of Hoxa5 | [47] |
| Cardiac organoid | polystyrene (1 μm) | day 18-day 21 (0.025, 0.25 and 2.5 µg/mL) | increased oxidative stress, inflammatory response, apoptosis, and collagen accumulation | aberrant expression of hypertrophic-related and cardiac-specific genes | [51] |
| Liver organoid | polystyrene (1 μm) | 48 h (0.25, 2.5 and 25 μg/mL) | Inducing ROS generation, oxidative stress, inflammation response, and alteration in lipid metabolism | increase the expression of hepatic HNF4A and CYP2E1 | [52] |
| Kidney organoid | polystyrene (1 μm) | 2 days (1.25, 2.5, and 5 μg/mL) | reduced size and abnormal tubular structures; increased level of mitochondrial ROS and DNA damage | glycolysis inhibition | [53] |
| Cortical spheroid | polystyrene (1 µm and 10 µm) | day 4–30 (100, 50, and 5 µg/mL) | short-term exposure promoted proliferation; log-term exposure reduced cell viability; DNA damage | elevated SOD2 gene expression; downregulated TUBB3 and TBR1/TBR2 expression | [54] |
| Retinal organoid | MPs derived from face masks (0.2 mm filtering) | 21 days (0.01, 0.1, 0.5, and 1 mg/mL) | disrupted the growth and development of retinal organoids | disordered neurogenesis, anatomical structure morphogenesis, and axon guidance | [55] |
| Skin organoid | polystyrene (100 and 500 nm) | 12 h co-culture (100 μg/mL) | size-dependent MP uptake and ROS induction | size-dependent expression of oxidative stress-associated genes (p53 and Bax) | [48] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Ge, L.; Lan, Y.; Gong, J.; Gao, X.; Faiola, F.; Zhang, S.; Li, M. Advancing Microplastic and Nanoplastic Toxicity Assessment: Insights from Human Organoid Models. Bioengineering 2026, 13, 309. https://doi.org/10.3390/bioengineering13030309
Ge L, Lan Y, Gong J, Gao X, Faiola F, Zhang S, Li M. Advancing Microplastic and Nanoplastic Toxicity Assessment: Insights from Human Organoid Models. Bioengineering. 2026; 13(3):309. https://doi.org/10.3390/bioengineering13030309
Chicago/Turabian StyleGe, Lingling, Yingying Lan, Jing Gong, Xue Gao, Francesco Faiola, Shaocheng Zhang, and Minghui Li. 2026. "Advancing Microplastic and Nanoplastic Toxicity Assessment: Insights from Human Organoid Models" Bioengineering 13, no. 3: 309. https://doi.org/10.3390/bioengineering13030309
APA StyleGe, L., Lan, Y., Gong, J., Gao, X., Faiola, F., Zhang, S., & Li, M. (2026). Advancing Microplastic and Nanoplastic Toxicity Assessment: Insights from Human Organoid Models. Bioengineering, 13(3), 309. https://doi.org/10.3390/bioengineering13030309








