Design and Development of Rehabi, a mHealth Telerehabilitation Platform with Markerless Motion Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Functional and Non-Functional Requirements Elicitation
2.2. System Design
- -
- Hip angle: computed between the thigh vector (Hip → Knee) and the vertical axis (0, −1). This quantifies the extent to which the thigh deviates from upright posture.
- -
- Knee angle: computed between the thigh vector (Hip → Knee) and the shank vector (Knee → Ankle). This yields the flexion/extension of the knee joint.
- -
- Ankle angle: computed between the shank vector (Knee → Ankle) and the foot vector (Ankle → Toe). This represents dorsiflexion/plantarflexion at the ankle.
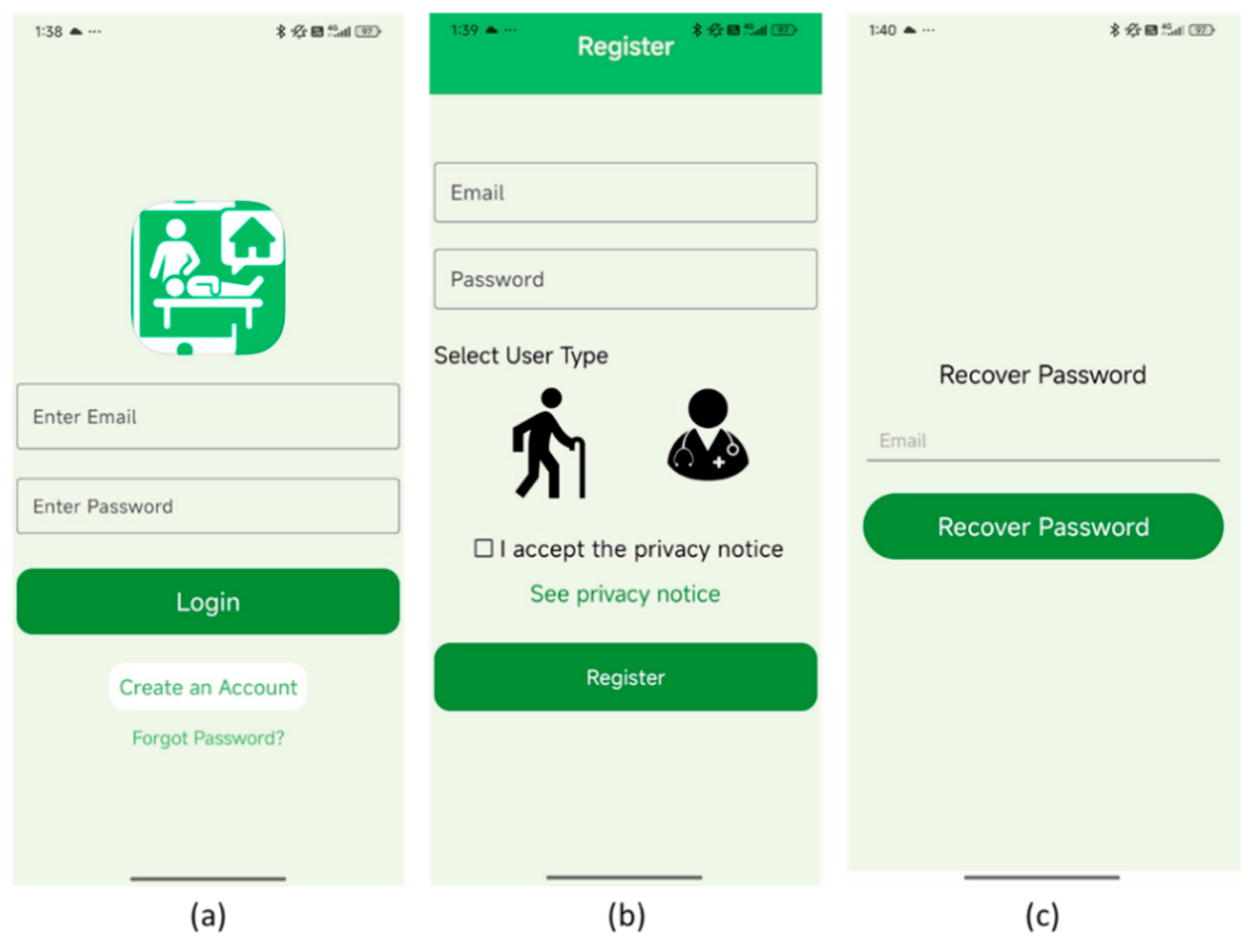
2.3. Interface Design
2.4. Mobile App Video-Based Motion Analysis Testing
3. Results
3.1. Requirements Determination
3.2. Proposed Developed Application
3.3. Mobile App Video-Based Motion Analysis Results

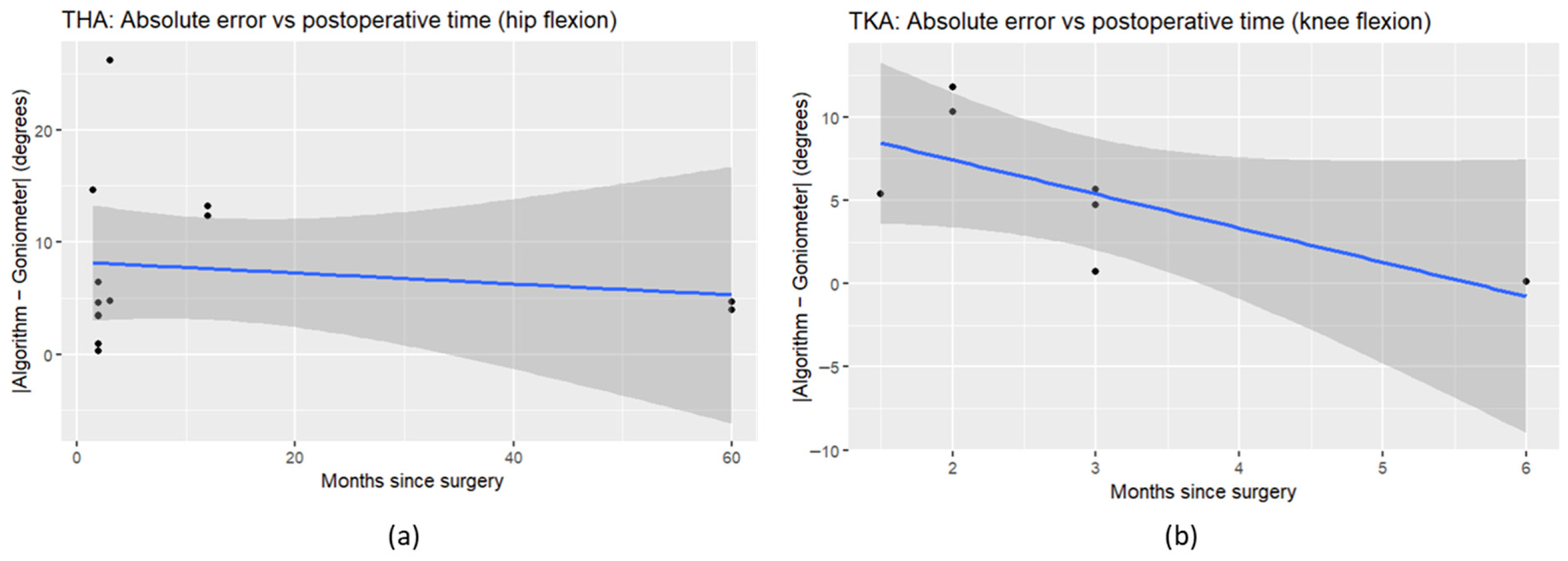
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| THA | Total Hip Arthroplasty |
| TKA | Total Knee Arthroplasty |
| ICC | Intraclass Correlation Coefficient |
| NLP | Natural Language Processing |
| MVIs | Medical Virtual Instruments |
| IoT | Internet of Things |
| IPAQ | International Physical Activity Questionnaire |
| GNSS | Global Navigation Satellite System |
| mHealth | Mobile Health |
| UI/UX | User Interface/User Experience |
| CRUD | Create, Read, Update, Delete |
| API | Application Programming Interface |
| AI | Artificial Intelligence |
| ICTs | Information Communication Technologies |
References
- Mehraeen, E.; SeyedAlinaghi, S.A.; Heydari, M.; Karimi, A.; Mahdavi, A.; Mashoufi, M.; Sarmad, A.; Mirghaderi, P.; Shamsabadi, A.; Qaderi, K.; et al. Telemedicine Technologies and Applications in the Era of COVID-19 Pandemic: A Systematic Review. Health Inform. J. 2023, 29, 14604582231167431. [Google Scholar] [CrossRef]
- Leochico, C.F.D.; Espiritu, A.I.; Ignacio, S.D.; Mojica, J.A.P. Challenges to the Emergence of Telerehabilitation in a Developing Country: A Systematic Review. Front. Neurol. 2020, 11, 1007. [Google Scholar] [CrossRef] [PubMed]
- Krupinski, E.A.; Patterson, T.; Norman, C.D.; Roth, Y.; ElNasser, Z.; Abdeen, Z.; Noyek, A.; Sriharan, A.; Ignatieff, A.; Black, S.; et al. Successful Models for Telehealth. Otolaryngol. Clin. N. Am. 2011, 44, 1275–1288. [Google Scholar] [CrossRef]
- Wells, D.; DeNiro, K.L.; Ramsey, A. Telemedicine in Drug Hypersensitivity. Immunol. Allergy Clin. N. Am. 2022, 42, 323–333. [Google Scholar] [CrossRef]
- Ndwabe, H.; Basu, A.; Mohammed, J. Post Pandemic Analysis on Comprehensive Utilization of Telehealth and Telemedicine. Clin. Ehealth 2024, 7, 5–14. [Google Scholar] [CrossRef]
- Esparza, W.; Aladro-Gonzalvo, A.R.; Baldeon, J.; Ortiz, S. Toward a Design of a Telerehabilitation Program for the Functional Recovery in Post-Hip Arthroplasty Patients. In Assistive and Rehabilitation Engineering; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef][Green Version]
- Kocyigit, B.F.; Assylbek, M.I.; Yessirkepov, M. Telerehabilitation: Lessons from the COVID-19 Pandemic and Future Perspectives. Rheumatol. Int. 2024, 44, 577–582. [Google Scholar] [CrossRef]
- Boccalandro, E.A.; Dallari, G.; Mannucci, P.M. Telemedicine and Telerehabilitation: Current and Forthcoming Applications in Haemophilia. Blood Transfus. 2019, 17, 385. [Google Scholar] [CrossRef] [PubMed]
- Pramuka, M.; van Roosmalen, L. Telerehabilitation Technologies: Accessibility and Usability. Int. J. Telerehabil. 2009, 1, 85. [Google Scholar] [CrossRef]
- Serra, C.M.; Tanarro, A.A. Teleconsultation and Videoconsultation Forever? Med. Clin. 2022, 158, 122. [Google Scholar] [CrossRef]
- Liljeroos, M.; Arkkukangas, M. Implementation of Telemonitoring in Health Care: Facilitators and Barriers for Using EHealth for Older Adults with Chronic Conditions. Risk Manag. Healthc. Policy 2023, 16, 43–53. [Google Scholar] [CrossRef]
- Black, R.J.; Cross, M.; Haile, L.M.; Culbreth, G.T.; Steinmetz, J.D.; Hagins, H.; Kopec, J.A.; Brooks, P.M.; Woolf, A.D.; Ong, K.L.; et al. Global, Regional, and National Burden of Rheumatoid Arthritis, 1990–2020, and Projections to 2050: A Systematic Analysis of the Global Burden of Disease Study 2021. Lancet Rheumatol. 2023, 5, e594–e610. [Google Scholar] [CrossRef]
- Qiao, L.; Li, M.; Deng, F.; Wen, X.; Deng, H.; Xue, Z.; Zhou, J.; Lin, J.; Amhare, A.F.; Xiang, R.; et al. Epidemiological Trends of Osteoarthritis at the Global, Regional, and National Levels from 1990 to 2021 and Projections to 2050. Arthritis Res. Ther. 2025, 27, 199. [Google Scholar] [CrossRef] [PubMed]
- Zou, H.; Liu, M.; Lu, Z.; Wang, J.; Zhao, P. The Relative Efficacy of Different Types of Telerehabilitation for Managing Chronic Musculoskeletal Pain: A Systematic Review with Network Meta-Analysis. J. Orthop. Sports Phys. Ther. 2025, 55, 695–704. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.Y.L.; Wong, A.K.C.; Hung, T.T.M.; Yan, J.; Yang, S. Nurse-Led Telehealth Intervention for Rehabilitation (Telerehabilitation) Among Community-Dwelling Patients with Chronic Diseases: Systematic Review and Meta-Analysis. J. Med. Internet Res. 2022, 24, e40364. [Google Scholar] [CrossRef] [PubMed]
- Tohănean, D.I.; Vulpe, A.M.; Mijaica, R.; Alexe, D.I. Embedding Digital Technologies (AI and ICT) into Physical Education: A Systematic Review of Innovations, Pedagogical Impact, and Challenges. Appl. Sci. 2025, 15, 9826. [Google Scholar] [CrossRef]
- Nussbaum, R.; Kelly, C.; Quinby, E.; Mac, A.; Parmanto, B.; Dicianno, B.E. Systematic Review of Mobile Health Applications in Rehabilitation. Arch. Phys. Med. Rehabil. 2019, 100, 115–127. [Google Scholar] [CrossRef]
- Wildenbos, G.A.; Peute, L.; Jaspers, M. Aging Barriers Influencing Mobile Health Usability for Older Adults: A Literature Based Framework (MOLD-US). Int. J. Med. Inform. 2018, 114, 66–75. [Google Scholar] [CrossRef]
- Muñoz Ñañez, D.; Gomez Sayas, J.; Andrade Díaz, K.-V.; Muñoz Ñañez, D.; Gomez Sayas, J.; Andrade Díaz, K.-V. Salud Móvil (MHealth) En Profundidad: Un Estudio Bibliométrico. Rev. Espac. 2025, 46, 130–152. [Google Scholar] [CrossRef]
- Lin, W.T.M.; Lin, B.S.; Lee, I.J.; Lee, S.H. Development of a Smartphone-Based MHealth Platform for Telerehabilitation. IEEE Trans. Neural Syst. Rehabil. Eng. 2022, 30, 2682–2691. [Google Scholar] [CrossRef]
- Marcolino, M.S.; Oliveira, J.A.Q.; D’Agostino, M.; Ribeiro, A.L.; Alkmim, M.B.M.; Novillo-Ortiz, D. The Impact of MHealth Interventions: Systematic Review of Systematic Reviews. JMIR Mhealth Uhealth 2018, 6, e8873. [Google Scholar] [CrossRef]
- Thomas, C.; Nolte, K.; Schmidt, M.; Jaitner, T. Comparison of Marker-Based and Markerless Motion Capture Systems for Measuring Throwing Kinematics. Biomechanics 2025, 5, 100. [Google Scholar] [CrossRef]
- Wade, L.; Needham, L.; McGuigan, P.; Bilzon, J. Applications and Limitations of Current Markerless Motion Capture Methods for Clinical Gait Biomechanics. PeerJ 2022, 10, e12995. [Google Scholar] [CrossRef]
- Scataglini, S.; Abts, E.; Van Bocxlaer, C.; Van den Bussche, M.; Meletani, S.; Truijen, S. Accuracy, Validity, and Reliability of Markerless Camera-Based 3D Motion Capture Systems versus Marker-Based 3D Motion Capture Systems in Gait Analysis: A Systematic Review and Meta-Analysis. Sensors 2024, 24, 3686. [Google Scholar] [CrossRef]
- Sweetline, B.C.; Vijayakumaran, C.; Samydurai, A. Patient Monitoring for Personalized Mobile Health (PMH) Based on Medical Virtual Instruments. Int. J. Interact. Mob. Technol. (IJIM) 2023, 17, 82–94. [Google Scholar] [CrossRef]
- Corrêa, A.G.D.; Ravaglio, A.d.S.C.; Navikas, F.H.; Do Amaral, J.D.; Filho, P.C.M.; Cunha, D.V.; Silva, M.G.d.L.; Rodrigues, B.d.S. Analysis of the Recognition of Facial Movements of App Designed for Rehabilitation of People with Facial Paralysis. Int. J. Interact. Mob. Technol. (IJIM) 2024, 18, 112–129. [Google Scholar] [CrossRef]
- Mohammed, B.G.; Hasan, D.S. Smart Healthcare Monitoring System Using IoT. Int. J. Interact. Mob. Technol. (IJIM) 2023, 17, 141–152. [Google Scholar] [CrossRef]
- Mauricio, D.; Flores-Cortegana, C.M.; Shuanarias, A.J.; Castañeda, P.; Rojas-Mezarina, L.; Castillo-Sequera, J.L. Sedentary: A Healthy Lifestyle App for Home Office Workers. Int. J. Interact. Mob. Technol. (IJIM) 2025, 19, 188–209. [Google Scholar] [CrossRef]
- Karim, S.; Gide, E. The Use of Interactive Mobile Technology to Improve the Quality of Health Care Services in Private and Public Hospitals in Australia. Int. J. Interact. Mob. Technol. (IJIM) 2018, 12, 4–18. [Google Scholar] [CrossRef][Green Version]
- Zowghi, D.; Coulin, C. Requirements Elicitation: A Survey of Techniques, Approaches, and Tools. In Engineering and Managing Software Requirements; Aurum, A., Wohlin, C., Eds.; Springer: Berlin/Heidelberg, Germany, 2005; pp. 19–46. [Google Scholar] [CrossRef]
- Software Architecture in Practice: Software Architect Practice_c3—Len Bass, Paul Clements, Rick Kazman—Google Libros. Available online: https://books.google.com.mx/books/about/Software_Architecture_in_Practice.html?id=-II73rBDXCYC&redir_esc=y (accessed on 22 January 2026).
- Robert, C.M. Clean Architecture: A Cra Sman’s Guide to So Ware Structure and Design, First Edition Table of Contents; Pearson: Boston, MA, USA, 2017; pp. 1–9. [Google Scholar]
- Fowler, M. Patterns of Enterprise Application Architecture; Addison-Wesley Professional: Boston, MA, USA, 2012. [Google Scholar]
- Guía de Soluciones de MediaPipe | Google AI Edge | Google AI for Developers. Available online: https://ai.google.dev/edge/mediapipe/solutions/guide?hl=es-419 (accessed on 25 January 2026).
- OpenCV on Android—OpenCV. Available online: https://opencv.org/android/ (accessed on 25 January 2026).
- Chaquopy Version 16.1.0—Chaquopy. Available online: https://chaquo.com/chaquopy/chaquopy-version-16-1-0/ (accessed on 25 January 2026).
- Yu, B.; Gabriel, D.; Noble, L.; An, K.-N.; Yu, B.; Gabriel, D.; Noble, L.; An, K.-N. Estimate of the Optimum Cutoff Frequency for the Butterworth Low-Pass Digital Filter. J. Appl. Biomech. 1999, 15, 318–329. [Google Scholar] [CrossRef]
- Firestore | Firebase. Available online: https://firebase.google.com/docs/firestore?hl=es-419 (accessed on 25 January 2026).
- Cloud Storage for Firebase. Available online: https://firebase.google.com/docs/storage?hl=es-419 (accessed on 25 January 2026).
- Firebase Authentication. Available online: https://firebase.google.com/docs/auth?hl=es-419 (accessed on 25 January 2026).
- Elliot, A.J.; Maier, M.A. Color Psychology: Effects of Perceiving Color on Psychological Functioning in Humans. Annu. Rev. Psychol. 2014, 65, 95–120. [Google Scholar] [CrossRef] [PubMed]
- Fisk, A.D.; Czaja, S.J.; Rogers, W.A.; Charness, N.; Czaja, S.J.; Sharit, J. Designing for Older Adults: Principles and Creative Human Factors Approaches, Second Edition. In Designing for Older Adults; CRC Press: Boca Raton, FL, USA, 2020. [Google Scholar] [CrossRef]
- Rothgangel, A.; Braun, S.; Smeets, R.; Beurskens, A. Design and Development of a Telerehabilitation Platform for Patients with Phantom Limb Pain: A User-Centered Approach. JMIR Rehabil. Assist. Technol. 2017, 4, e2. [Google Scholar] [CrossRef] [PubMed]






| Section | Questions | Purpose of Data Collected |
|---|---|---|
| 1. Sociodemographic Information |
| To characterize the respondent population and classify user roles. This ensures that requirements reflect the diverse demographics and perspectives of both patients and clinicians. |
| 2. Needs and Problems |
| To capture user pain points, expectations, and benchmarks against existing solutions. This informs the definition of functional requirements and highlights unmet needs. |
| 3. User Environment and Technology Use |
| To assess digital literacy, usability preferences, and support needs. This section outlines non-functional requirements, including accessibility, onboarding strategies, and help mechanisms, to ensure inclusivity and ease of adoption. |
| 4. Evaluation of Opportunities |
| To understand user acceptance criteria and identify opportunities for innovation. This ensures the application aligns with user expectations and incorporates desirable features. |
| Age (Years) | Weight (kg) | Height (m) | Surgery Type | Months Since Surgery (Months) |
|---|---|---|---|---|
| 61 | 78 | 1.63 | Left THA | 60 |
| 50 | 94 | 1.72 | Left THA | 2 |
| 64 | 61.5 | 1.48 | Right TKA | 2 |
| 73 | 69.8 | 1.56 | Left TKA | 1 |
| 48 | 67 | 1.54 | Left and Right THA | 2 |
| 72 | 72 | 1.89 | Right TKA | 2 |
| 66 | 58 | 1.57 | Left TKA | 3 |
| 68 | 68 | 1.63 | Left THA | 1.5 |
| 77 | 68.3 | 1.37 | Right TKA | 3 |
| 50 | 74 | 1.58 | Left and Right THA | 12 |
| 63 | 47.3 | 1.42 | Left THA | 3 |
| 55 | 78.5 | 1.5 | Left THA | 6 |
| 46 | 90 | 1.72 | Left and Right THA | 1 |
| 53 | 55 | 1.57 | Left THA | 1.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
González-Mendoza, A.; Aguilar-Sierra, H.; Zepeda-Mora, R.; Alessi-Montero, A.; Rodríguez-Reyes, G.; Carrera, L.N.; Quiñones-Uriostegui, I.; Ayala-Cadena, P.; Gomez-Verdad, A. Design and Development of Rehabi, a mHealth Telerehabilitation Platform with Markerless Motion Analysis. Bioengineering 2026, 13, 308. https://doi.org/10.3390/bioengineering13030308
González-Mendoza A, Aguilar-Sierra H, Zepeda-Mora R, Alessi-Montero A, Rodríguez-Reyes G, Carrera LN, Quiñones-Uriostegui I, Ayala-Cadena P, Gomez-Verdad A. Design and Development of Rehabi, a mHealth Telerehabilitation Platform with Markerless Motion Analysis. Bioengineering. 2026; 13(3):308. https://doi.org/10.3390/bioengineering13030308
Chicago/Turabian StyleGonzález-Mendoza, Arturo, Hipólito Aguilar-Sierra, Rafael Zepeda-Mora, Aldo Alessi-Montero, Gerardo Rodríguez-Reyes, Lidia Núñez Carrera, Ivett Quiñones-Uriostegui, Paola Ayala-Cadena, and Adriana Gomez-Verdad. 2026. "Design and Development of Rehabi, a mHealth Telerehabilitation Platform with Markerless Motion Analysis" Bioengineering 13, no. 3: 308. https://doi.org/10.3390/bioengineering13030308
APA StyleGonzález-Mendoza, A., Aguilar-Sierra, H., Zepeda-Mora, R., Alessi-Montero, A., Rodríguez-Reyes, G., Carrera, L. N., Quiñones-Uriostegui, I., Ayala-Cadena, P., & Gomez-Verdad, A. (2026). Design and Development of Rehabi, a mHealth Telerehabilitation Platform with Markerless Motion Analysis. Bioengineering, 13(3), 308. https://doi.org/10.3390/bioengineering13030308







