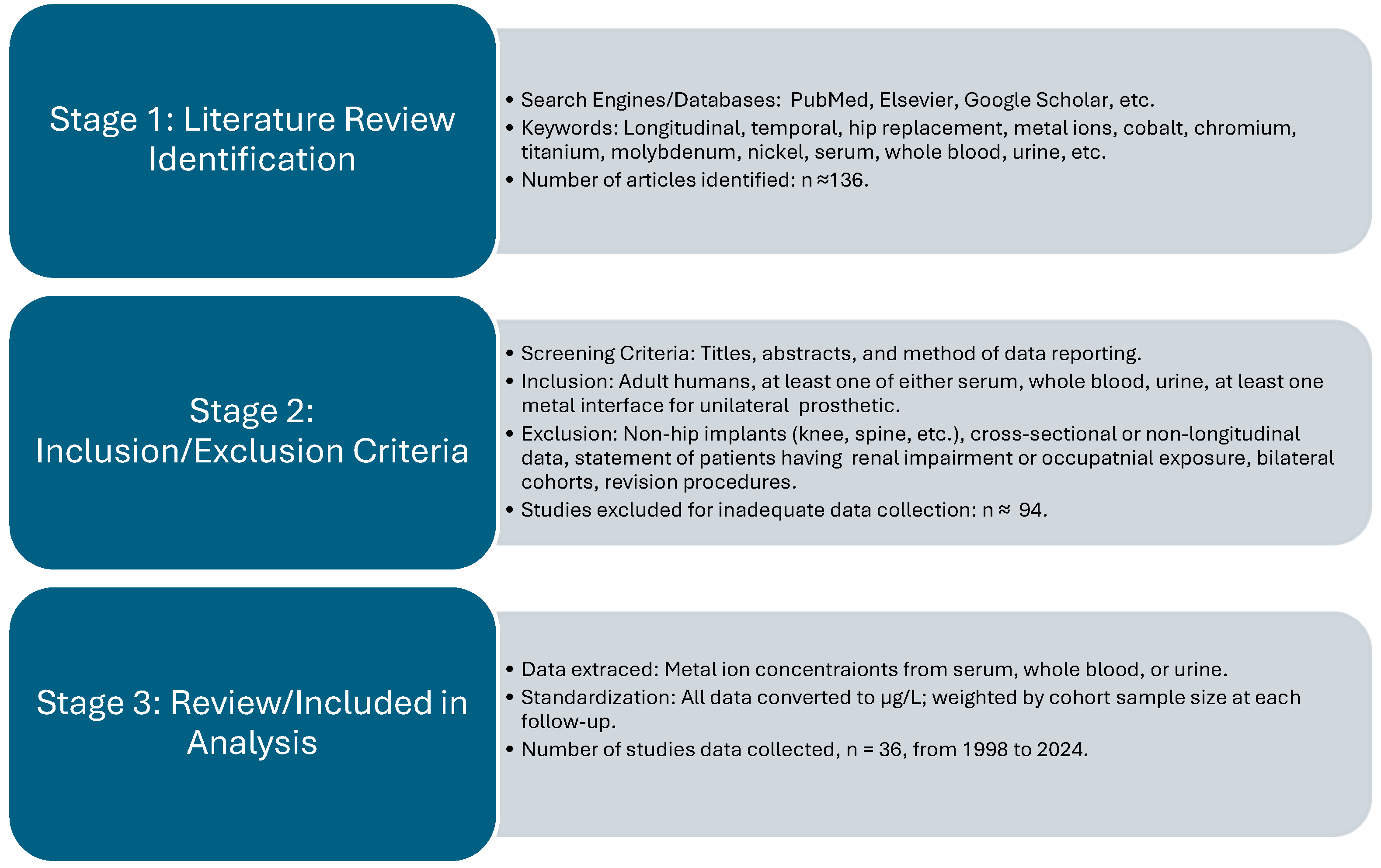
Figure 1.
Schematic summarizing literature search, screening, and inclusion process for studies that report longitudinal metal ion levels in patients with hip prosthetics. The search was performed using different databases and search engines with keywords reflecting longitudinal metal ion concentrations. Thirty-six studies met the inclusion criteria and were utilized for data extraction in the pooled analysis.
Figure 1.
Schematic summarizing literature search, screening, and inclusion process for studies that report longitudinal metal ion levels in patients with hip prosthetics. The search was performed using different databases and search engines with keywords reflecting longitudinal metal ion concentrations. Thirty-six studies met the inclusion criteria and were utilized for data extraction in the pooled analysis.
Figure 2.
Modeled trajectories of serum and whole blood Co concentrations. Pooled and weighted values were modeled using a one-phase exponential association. Serum (blue) rose from 0.18 µg/L to a plateau of 1.96 µg/L with a half-time of 3.9 months , while whole blood (red) rose from 0.45 µg/L to a plateau of 1.96 µg/L with a halftime of 5.7 months . Shaded regions represent the standard deviation (SD) of mean values at each follow-up month.
Figure 2.
Modeled trajectories of serum and whole blood Co concentrations. Pooled and weighted values were modeled using a one-phase exponential association. Serum (blue) rose from 0.18 µg/L to a plateau of 1.96 µg/L with a half-time of 3.9 months , while whole blood (red) rose from 0.45 µg/L to a plateau of 1.96 µg/L with a halftime of 5.7 months . Shaded regions represent the standard deviation (SD) of mean values at each follow-up month.
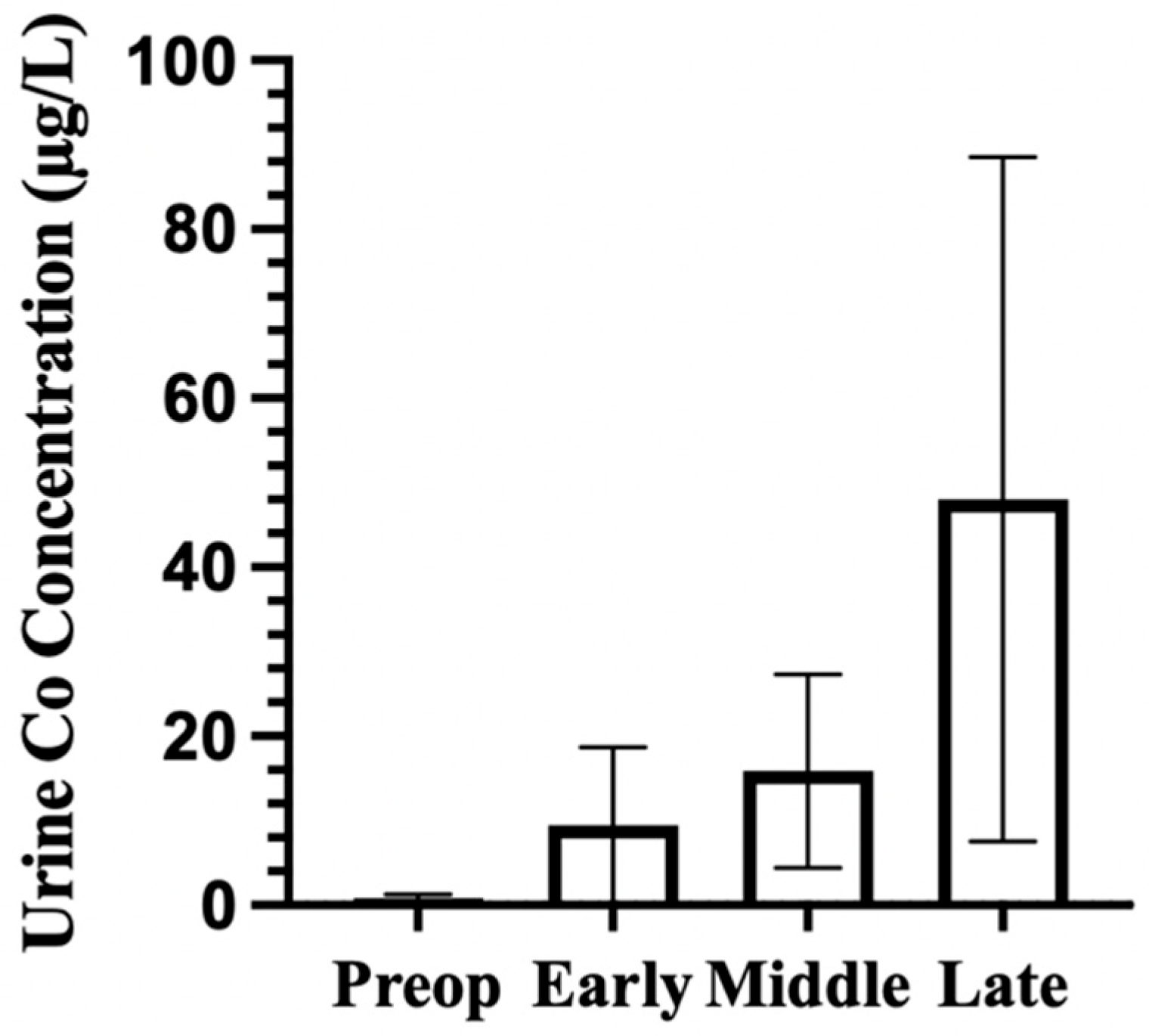
Figure 3.
Urinary cobalt concentrations across time phases. Bars represent mean urinary Co concentrations across each phase. Error bars represent standard deviation (SD). Time phases were defined as preop (before implantation), early (1–23 months), middle (24–47 months), and late (≥48 months). Mean urinary Co tended to increase across time phases, with greater variability in the late phase.
Figure 3.
Urinary cobalt concentrations across time phases. Bars represent mean urinary Co concentrations across each phase. Error bars represent standard deviation (SD). Time phases were defined as preop (before implantation), early (1–23 months), middle (24–47 months), and late (≥48 months). Mean urinary Co tended to increase across time phases, with greater variability in the late phase.
Figure 4.
Modeled trajectories of serum and whole blood Cr concentrations. Pooled and weighted values were modeled using a one-phase exponential association. Serum (blue) rose from 0.28 µg/L to a plateau of 2.02 µg/L with a half-time of 8.0 months . Whole blood (red) rose from 0.3 µg/L to a plateau of 0.99 µg/L with a half-time of 2.4 months . Shaded regions represent the standard deviation (SD) of pooled values at their respective follow-ups.
Figure 4.
Modeled trajectories of serum and whole blood Cr concentrations. Pooled and weighted values were modeled using a one-phase exponential association. Serum (blue) rose from 0.28 µg/L to a plateau of 2.02 µg/L with a half-time of 8.0 months . Whole blood (red) rose from 0.3 µg/L to a plateau of 0.99 µg/L with a half-time of 2.4 months . Shaded regions represent the standard deviation (SD) of pooled values at their respective follow-ups.
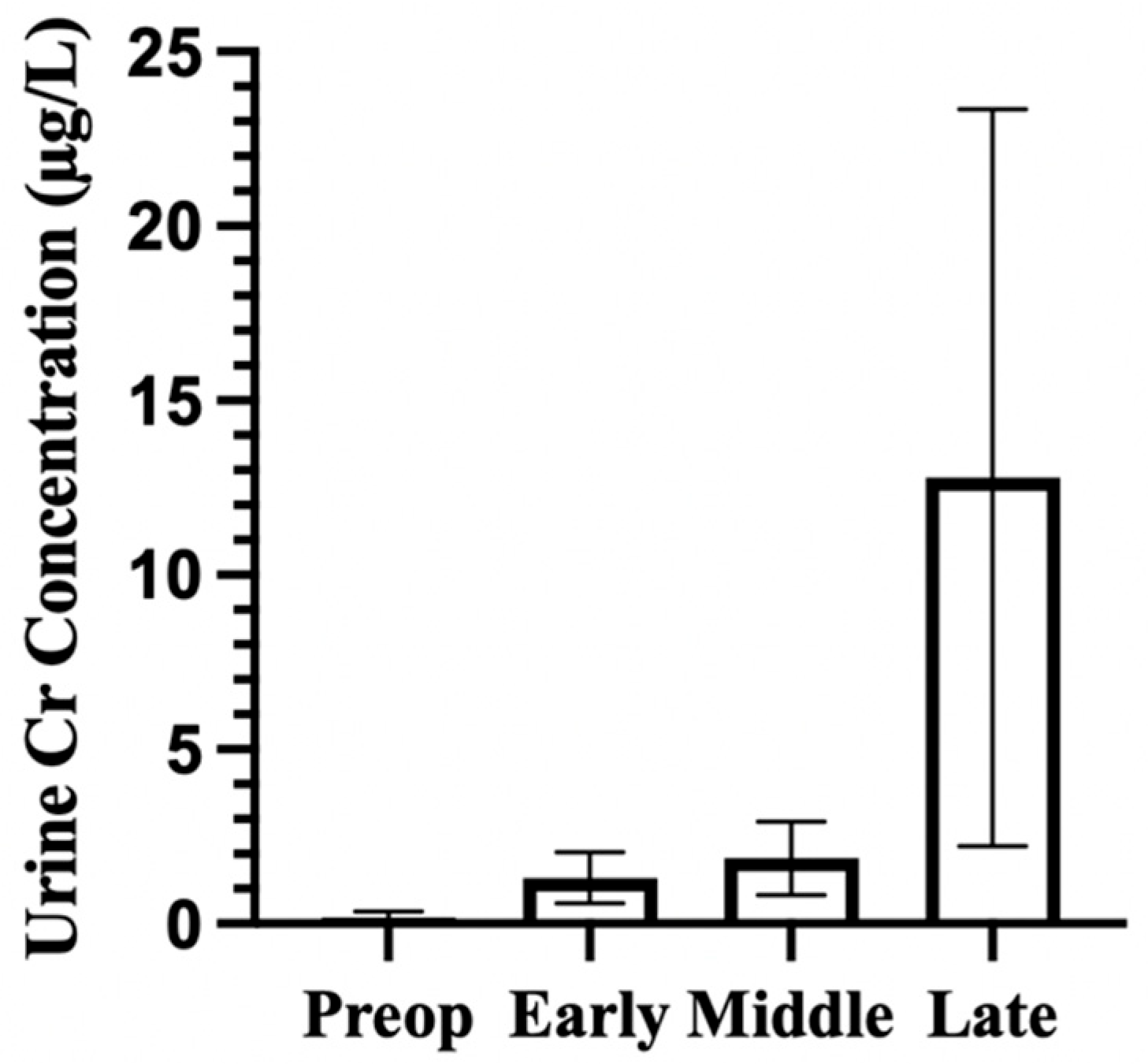
Figure 5.
Urinary chromium concentrations across pooled time phase following hip implantation. Pooled mean urinary chromium concentrations (µg/L) are shown across four time phases: preoperative, early (1–23 months), middle (24–47 months), and late (≥48 months). Bars represent mean values, with error bars indicating standard deviation (SD). Values reflect descriptive pooling across studies that were included.
Figure 5.
Urinary chromium concentrations across pooled time phase following hip implantation. Pooled mean urinary chromium concentrations (µg/L) are shown across four time phases: preoperative, early (1–23 months), middle (24–47 months), and late (≥48 months). Bars represent mean values, with error bars indicating standard deviation (SD). Values reflect descriptive pooling across studies that were included.
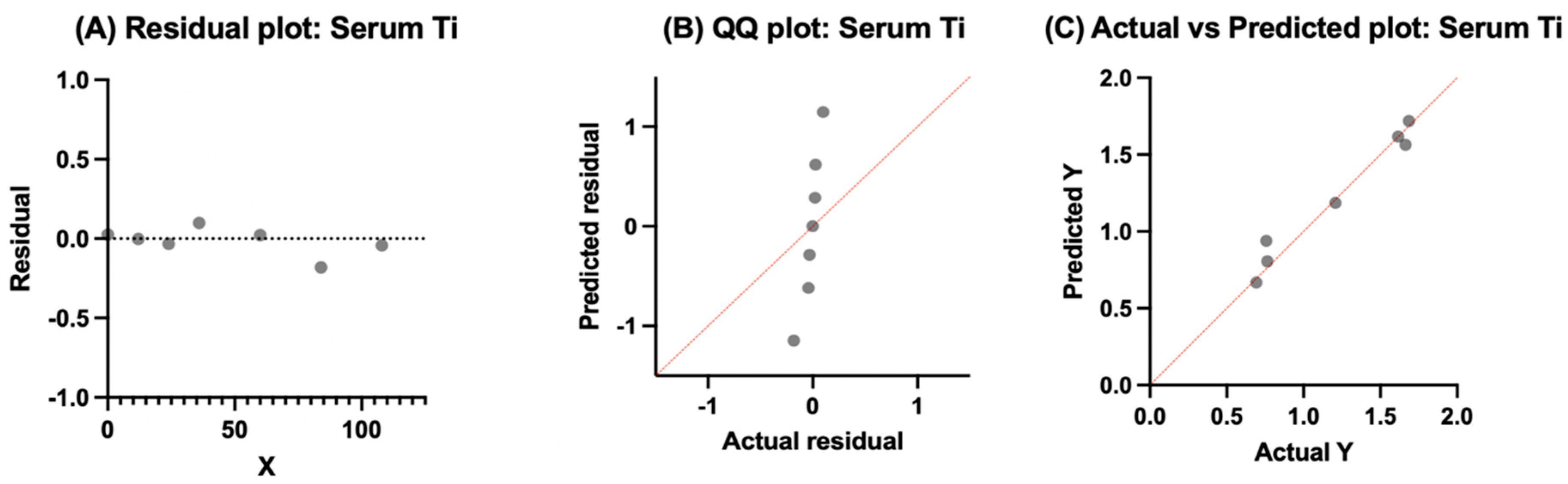
Figure 6.
Modeled trajectory of serum Ti concentrations. Pooled and weighted serum Ti concentrations were captured using an exponential rise-then-decay association. Concentrations rose from a baseline of 0.67 µg/L to a modeled peak around 12–24 months, followed by a gradual decline toward lower levels over time. The fitted function was with best-fit rate constants of k = 0.049 and kd = 0.028. Shaded regions represent the standard deviation at each follow-up month.
Figure 6.
Modeled trajectory of serum Ti concentrations. Pooled and weighted serum Ti concentrations were captured using an exponential rise-then-decay association. Concentrations rose from a baseline of 0.67 µg/L to a modeled peak around 12–24 months, followed by a gradual decline toward lower levels over time. The fitted function was with best-fit rate constants of k = 0.049 and kd = 0.028. Shaded regions represent the standard deviation at each follow-up month.
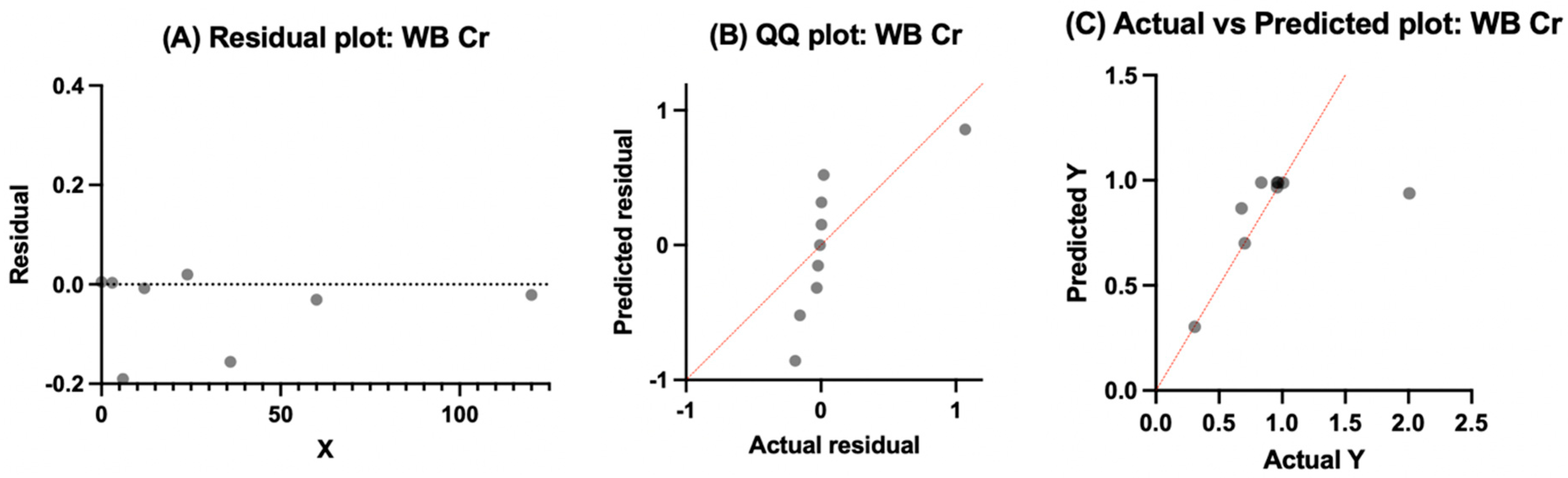
Figure 7.
Model output for serum Co using a one-phase exponential association. (A) Plot of residuals against time does not reveal strong systematic bias. (B) QQ plot showing deviation from normality and curvature away from the angled line. (C) Actual vs. predicted plot values fall close to the identity line with not much scatter, consistent with adequacy of fit in the pooled data.
Figure 7.
Model output for serum Co using a one-phase exponential association. (A) Plot of residuals against time does not reveal strong systematic bias. (B) QQ plot showing deviation from normality and curvature away from the angled line. (C) Actual vs. predicted plot values fall close to the identity line with not much scatter, consistent with adequacy of fit in the pooled data.
Figure 8.
Model output for whole blood Co modeled with a one-phase exponential association. (A) Residuals plotted against time showing no systemic trends. (B) QQ plot demonstrating approximate normality of residuals, with deviations at the tails showing non-normal distribution for residuals. (C) The actual vs. predicted points are clustered near the identity line without great scatter, pointing toward adequacy of model fit in whole blood data.
Figure 8.
Model output for whole blood Co modeled with a one-phase exponential association. (A) Residuals plotted against time showing no systemic trends. (B) QQ plot demonstrating approximate normality of residuals, with deviations at the tails showing non-normal distribution for residuals. (C) The actual vs. predicted points are clustered near the identity line without great scatter, pointing toward adequacy of model fit in whole blood data.
Figure 9.
Model output for serum chromium modeled with a one-phase exponential association. (A) Residuals plotted against time are centered near zero without strong systematic bias, though a few outliers are present. (B) QQ plot showing curvature away from the identity line, indicating deviations from normality. (C) Actual vs. predicted values generally cluster near the identity line with moderate scatter, with one point outside the expected range, consistent with adequacy of the model fit for pooled serum chromium data.
Figure 9.
Model output for serum chromium modeled with a one-phase exponential association. (A) Residuals plotted against time are centered near zero without strong systematic bias, though a few outliers are present. (B) QQ plot showing curvature away from the identity line, indicating deviations from normality. (C) Actual vs. predicted values generally cluster near the identity line with moderate scatter, with one point outside the expected range, consistent with adequacy of the model fit for pooled serum chromium data.
Figure 10.
Model output for whole blood chromium modeled with a one-phase exponential association. (A) Residuals plotted against time are centered around zero without systematic bias. (B) QQ plot showing approximate normality with modest deviations at distribution tails. (C) Actual vs. predicted values closely cluster along the identity line, with one point outside the expected range, consistent with adequacy of model fit for pooled whole blood chromium data.
Figure 10.
Model output for whole blood chromium modeled with a one-phase exponential association. (A) Residuals plotted against time are centered around zero without systematic bias. (B) QQ plot showing approximate normality with modest deviations at distribution tails. (C) Actual vs. predicted values closely cluster along the identity line, with one point outside the expected range, consistent with adequacy of model fit for pooled whole blood chromium data.
Figure 11.
Model output for serum titanium using an exponential rise–decay association. (A) Residuals plotted against time are centered around zero without much evidence of systematic bias. (B) QQ plot showing a near-vertical distribution of points, indicating narrow variance relative to normal expectations. (C) Actual vs. predicted values cluster tightly along the identity line without extreme scatter, supporting an adequate model.
Figure 11.
Model output for serum titanium using an exponential rise–decay association. (A) Residuals plotted against time are centered around zero without much evidence of systematic bias. (B) QQ plot showing a near-vertical distribution of points, indicating narrow variance relative to normal expectations. (C) Actual vs. predicted values cluster tightly along the identity line without extreme scatter, supporting an adequate model.
Figure 12.
Reported serum molybdenum (Mo) concentrations over time following primary hip arthroplasty. Values were extracted directly from individual studies. Circular markers denote reported means, while triangular markers denote reported medians. All concentrations are in micrograms per liter (µg/L). Data include total hip arthroplasty (THA), Birmingham Hip Resurfacing (BHR), and hip resurfacing arthroplasty (HRA), as indicated by the study. Data adapted from [
39,
75].
Figure 12.
Reported serum molybdenum (Mo) concentrations over time following primary hip arthroplasty. Values were extracted directly from individual studies. Circular markers denote reported means, while triangular markers denote reported medians. All concentrations are in micrograms per liter (µg/L). Data include total hip arthroplasty (THA), Birmingham Hip Resurfacing (BHR), and hip resurfacing arthroplasty (HRA), as indicated by the study. Data adapted from [
39,
75].
Figure 13.
Serum nickel (Ni) concentrations following primary total hip arthroplasty as seen from Dahlstrand et al. (2009) [
38]. Data are shown for patients with metal-on-metal (MoM, n = 28) and metal-on-polyethylene (MoP, n = 26) prostheses at preop and 6, 12, and 24 months after surgery. In both groups, Ni concentrations increased over time, with the MoM cohort showing a slightly greater overall rise.
Figure 13.
Serum nickel (Ni) concentrations following primary total hip arthroplasty as seen from Dahlstrand et al. (2009) [
38]. Data are shown for patients with metal-on-metal (MoM, n = 28) and metal-on-polyethylene (MoP, n = 26) prostheses at preop and 6, 12, and 24 months after surgery. In both groups, Ni concentrations increased over time, with the MoM cohort showing a slightly greater overall rise.
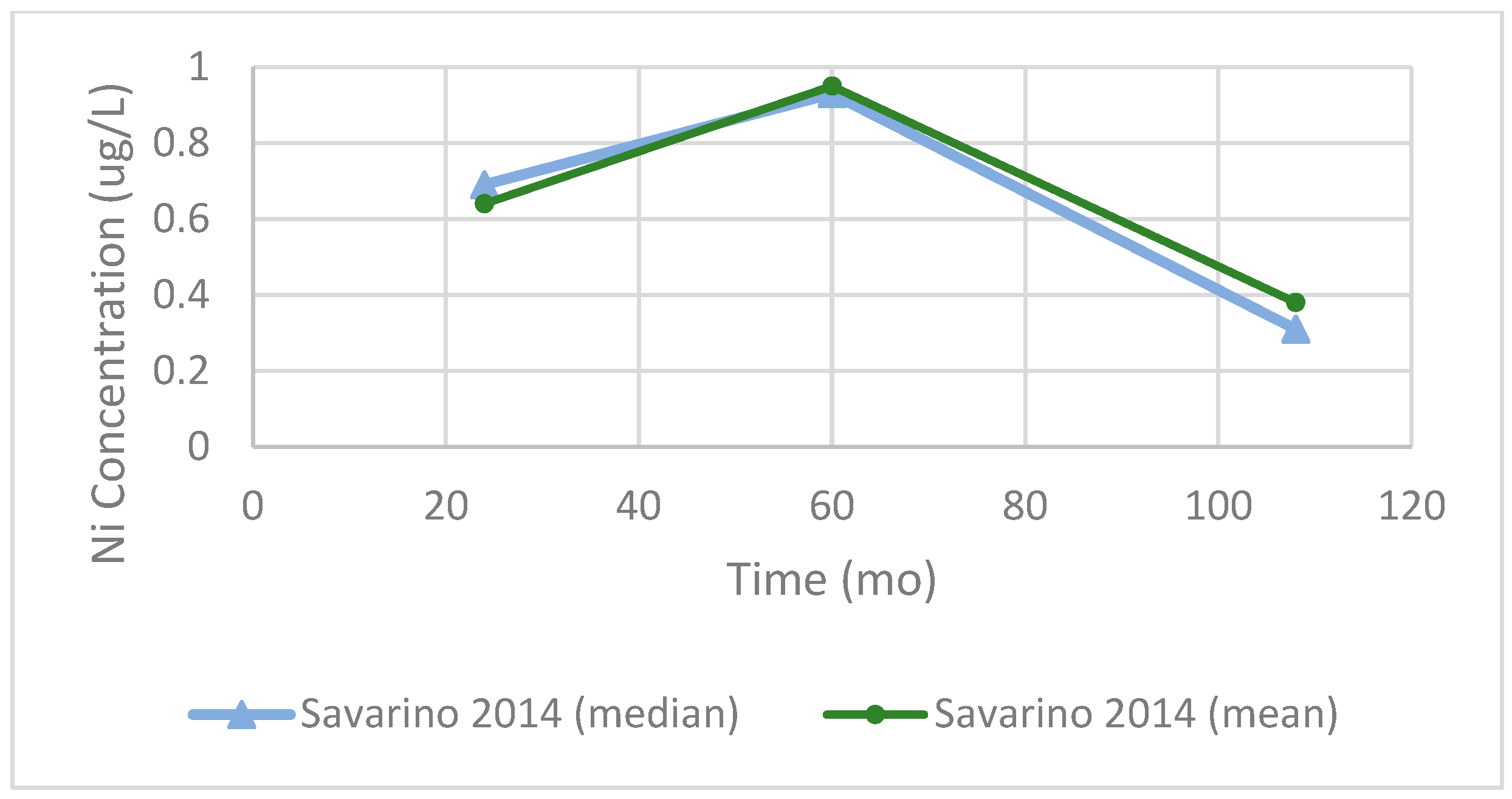
Figure 14.
Serum Ni concentrations after Birmingham Hip Resurfacing (BHR). Data from Savarino et al. (2014) [
77] are shown as both reported means (green circle) and medians (blue triangle) at 24, 60, and 108 months postoperatively. Concentrations peaked at 60 months before declining at longer-term follow-up.
Figure 14.
Serum Ni concentrations after Birmingham Hip Resurfacing (BHR). Data from Savarino et al. (2014) [
77] are shown as both reported means (green circle) and medians (blue triangle) at 24, 60, and 108 months postoperatively. Concentrations peaked at 60 months before declining at longer-term follow-up.
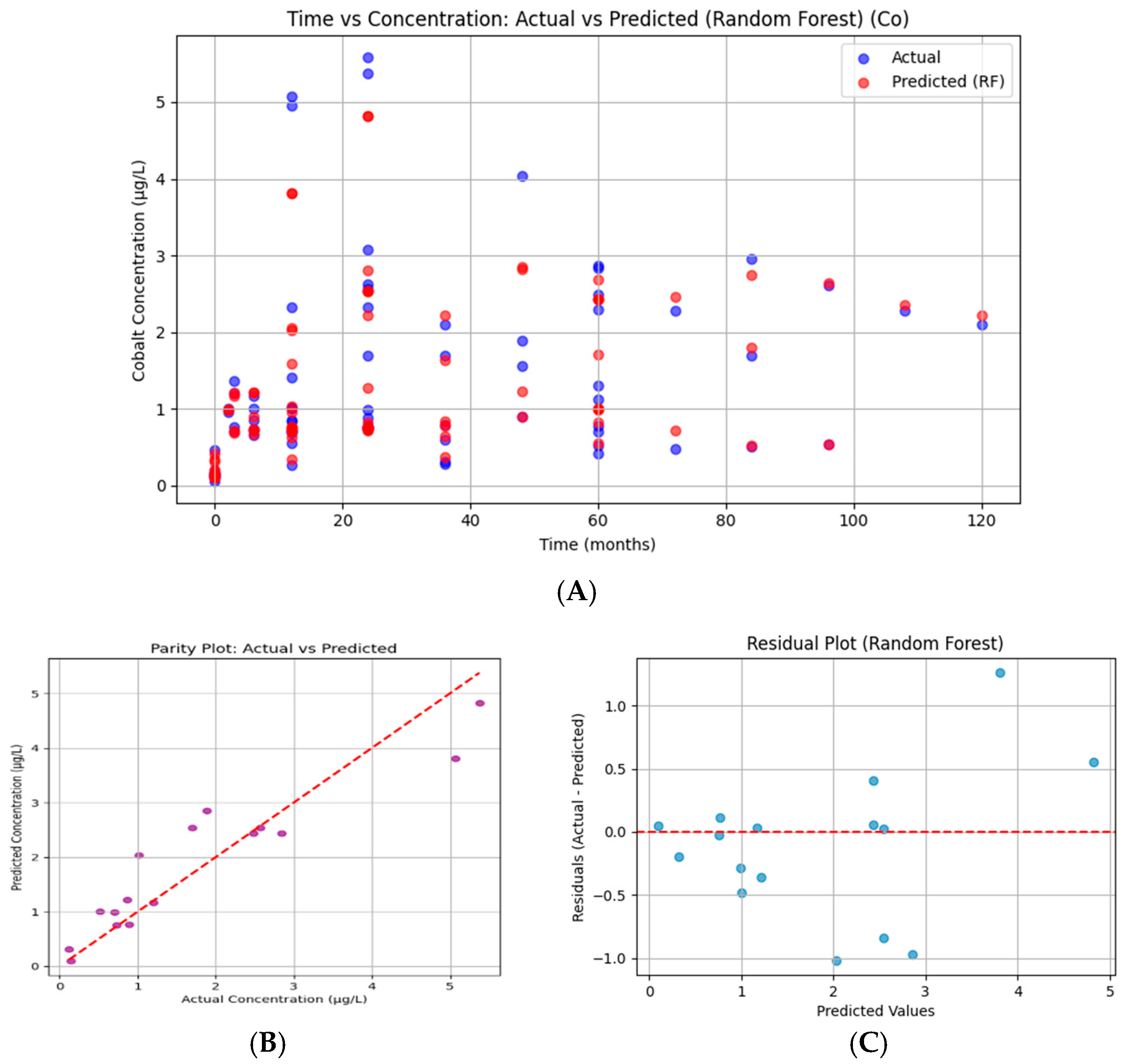
Figure 15.
Random Forest regression for serum cobalt. (A) Time vs. concentration showing actual values (blue) and model predictions (red). (B) Parity plot of predicted versus observed concentrations, with points closely aligned to the 45° diagonal. (C) Residuals plotted against predicted values are distributed symmetrically around zero. Model performance: MAE = 0.417, RMSE = 0.573, R2 = 0.861.
Figure 15.
Random Forest regression for serum cobalt. (A) Time vs. concentration showing actual values (blue) and model predictions (red). (B) Parity plot of predicted versus observed concentrations, with points closely aligned to the 45° diagonal. (C) Residuals plotted against predicted values are distributed symmetrically around zero. Model performance: MAE = 0.417, RMSE = 0.573, R2 = 0.861.
Figure 16.
Random Forest regression for serum chromium. (A) Time versus concentration showing observed values (blue) and Random Forest predictions (red). Predictions captured overall patterns but underestimated high peaks and overestimated mid-range concentrations. (B) Parity plot of predicted versus observed concentrations, showing wider scatter around the 45° line and indicating systematic deviations (R2 = 0.522, RMSE = 0.495). (C) Residual plot demonstrating greater variability and bias at higher predicted values, reflecting moderate predictive accuracy and reduced stability relative to cobalt.
Figure 16.
Random Forest regression for serum chromium. (A) Time versus concentration showing observed values (blue) and Random Forest predictions (red). Predictions captured overall patterns but underestimated high peaks and overestimated mid-range concentrations. (B) Parity plot of predicted versus observed concentrations, showing wider scatter around the 45° line and indicating systematic deviations (R2 = 0.522, RMSE = 0.495). (C) Residual plot demonstrating greater variability and bias at higher predicted values, reflecting moderate predictive accuracy and reduced stability relative to cobalt.
Figure 17.
Random Forest regression for serum titanium. (A) Time versus concentration showing observed values (blue) and Random Forest predictions (red). Predicted values tracked actual concentrations closely, with minor deviations at mid-levels. (B) Parity plot of predicted versus actual concentrations, demonstrating close alignment with the 45° diagonal and confirming robust explanatory power (R2 = 0.707, RMSE = 0.380). (C) Residual plot showing values distributed around zero with slight negative skew at higher predicted levels, supporting stable and reliable model performance.
Figure 17.
Random Forest regression for serum titanium. (A) Time versus concentration showing observed values (blue) and Random Forest predictions (red). Predicted values tracked actual concentrations closely, with minor deviations at mid-levels. (B) Parity plot of predicted versus actual concentrations, demonstrating close alignment with the 45° diagonal and confirming robust explanatory power (R2 = 0.707, RMSE = 0.380). (C) Residual plot showing values distributed around zero with slight negative skew at higher predicted levels, supporting stable and reliable model performance.
Figure 18.
Random Forest regression for serum molybdenum. (A) Time versus concentration showing observed values (blue) and Random Forest predictions (red). Predictions reproduced the temporal trajectory but smoothed concentration extremes, with underestimation at higher values. (B) Parity plot of predicted versus actual concentrations, with most points aligned near the 45° diagonal but showing underprediction at higher actual values (>5 µg/L). Model performance: R2 = 0.718, RMSE = 1.218. (C) Residual plot demonstrating values centered near zero with wider spread at higher predictions, indicating mild heteroscedasticity but overall stable model calibration.
Figure 18.
Random Forest regression for serum molybdenum. (A) Time versus concentration showing observed values (blue) and Random Forest predictions (red). Predictions reproduced the temporal trajectory but smoothed concentration extremes, with underestimation at higher values. (B) Parity plot of predicted versus actual concentrations, with most points aligned near the 45° diagonal but showing underprediction at higher actual values (>5 µg/L). Model performance: R2 = 0.718, RMSE = 1.218. (C) Residual plot demonstrating values centered near zero with wider spread at higher predictions, indicating mild heteroscedasticity but overall stable model calibration.
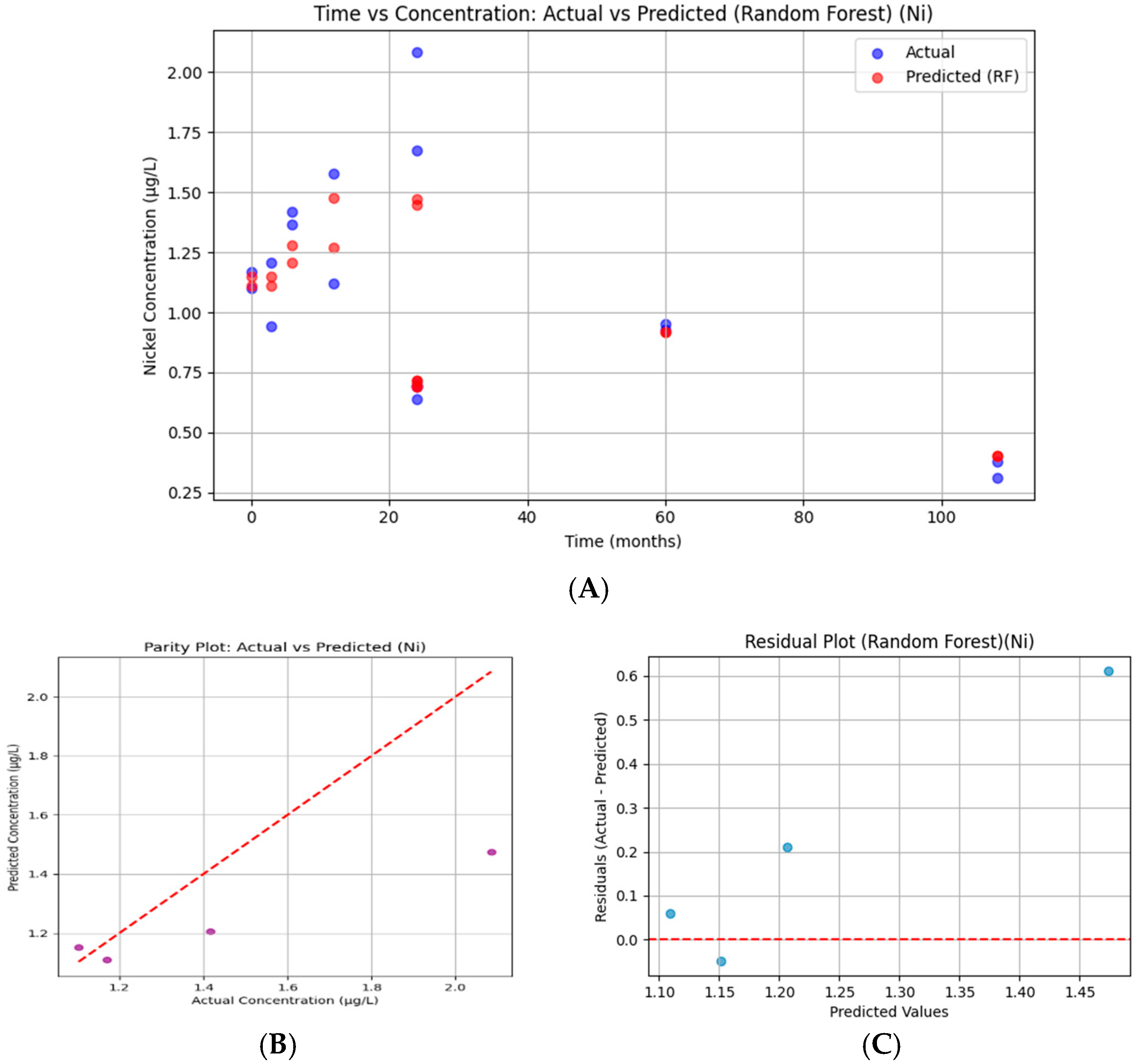
Figure 19.
Random Forest regression for serum nickel. (A) Time versus concentration showing observed values (blue) and Random Forest predictions (red). Predictions approximated overall temporal patterns but systematically underestimated higher concentrations. (B) Parity plot of predicted versus actual values showing deviations from the 45° diagonal, consistent with limited predictive fidelity (R2 = 0.297, RMSE = 0.325). (C) Residual plot indicating consistently positive errors, reflecting systematic underestimation of nickel values. Reduced data density limited the model’s explanatory capacity compared with other ions.
Figure 19.
Random Forest regression for serum nickel. (A) Time versus concentration showing observed values (blue) and Random Forest predictions (red). Predictions approximated overall temporal patterns but systematically underestimated higher concentrations. (B) Parity plot of predicted versus actual values showing deviations from the 45° diagonal, consistent with limited predictive fidelity (R2 = 0.297, RMSE = 0.325). (C) Residual plot indicating consistently positive errors, reflecting systematic underestimation of nickel values. Reduced data density limited the model’s explanatory capacity compared with other ions.
Figure 20.
Proposed systemic distribution, binding, and clearance of metal ions. Excessive wear and corrosion create ions that partition among serum proteins, blood cell components, and filterable fractions, which can be renally excreted. If present at high enough concentrations, downstream effects may occur.
Figure 20.
Proposed systemic distribution, binding, and clearance of metal ions. Excessive wear and corrosion create ions that partition among serum proteins, blood cell components, and filterable fractions, which can be renally excreted. If present at high enough concentrations, downstream effects may occur.
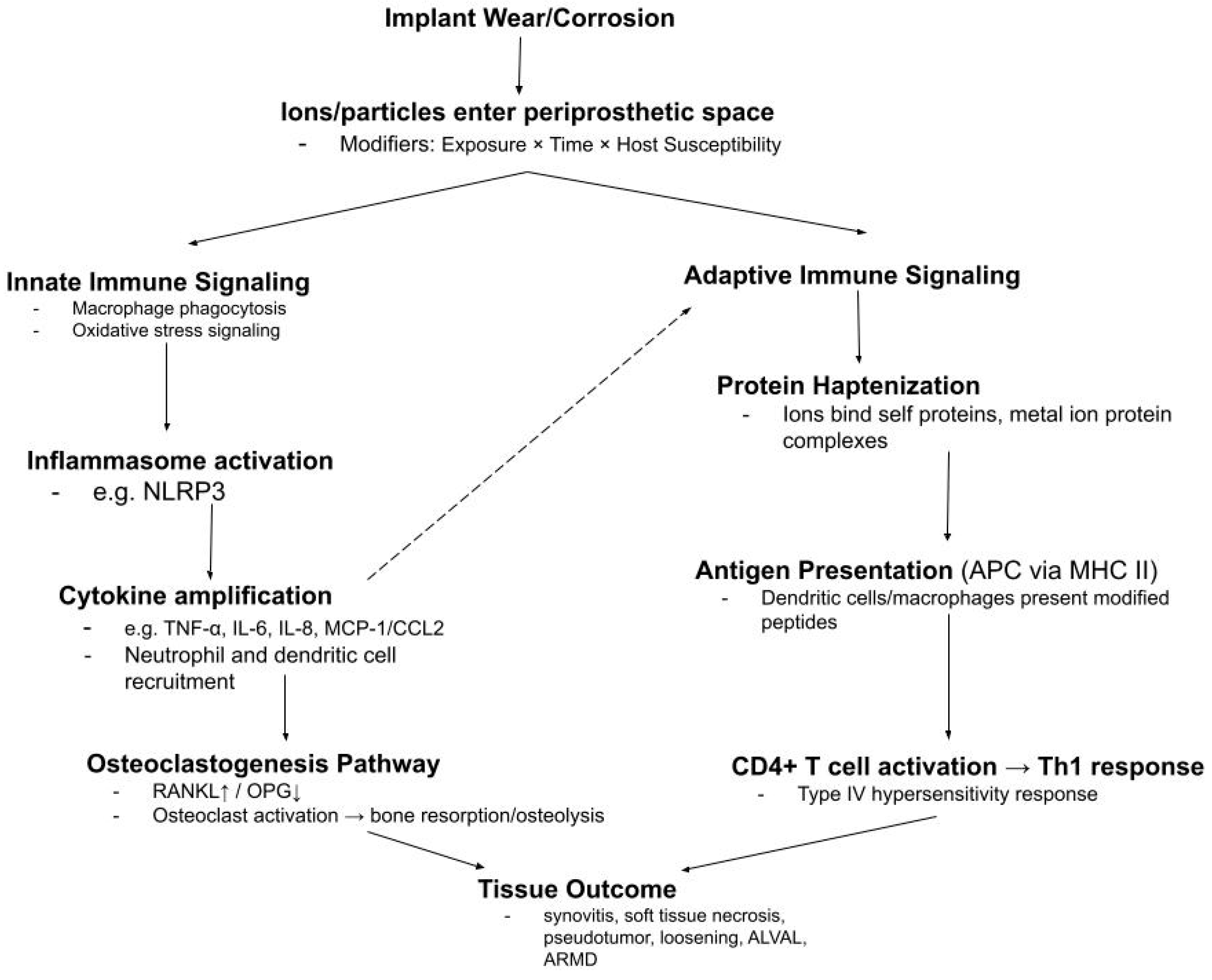
Figure 21.
Conditional implant-to-immune pathway after metal ion exposure. Wear and corrosion create ions and particles that can enter joint space and trigger innate and adaptive responses. Progression varies with exposure, time, and host susceptibility and provides context for subthreshold kinetics observed but also for occasional outlier elevation.
Figure 21.
Conditional implant-to-immune pathway after metal ion exposure. Wear and corrosion create ions and particles that can enter joint space and trigger innate and adaptive responses. Progression varies with exposure, time, and host susceptibility and provides context for subthreshold kinetics observed but also for occasional outlier elevation.
Table 1.
Nominal chemical compositions of common orthopedic implant alloys standardized by ASTM and ISO specifications. Major alloying elements are shown as ranges in weight percent, with trace elements included where applicable. “≤” refers to the maximum amount of corresponding metal. “Bal.” indicates balance, meaning the remainder of the composition is made up of the listed base element to reach 100%.
Table 1.
Nominal chemical compositions of common orthopedic implant alloys standardized by ASTM and ISO specifications. Major alloying elements are shown as ranges in weight percent, with trace elements included where applicable. “≤” refers to the maximum amount of corresponding metal. “Bal.” indicates balance, meaning the remainder of the composition is made up of the listed base element to reach 100%.
| Alloy/Group | ASTM | ISO | UNS | Co | Cr | Mo | Ti | Ni | Al | V | Fe | Si | W | Mn | Nb |
|---|
| Cast CoCrMo | F75 | 5832-4 | R30075 | Bal. | 27–30 | 5–7 | ≤0.1 | ≤0.5 | ≤0.1 | - | ≤0.75 | ≤1 | ≤0.2 | ≤1 | - |
| Wrought CoCrMo | F1537 | 5832-12 | R31538 | Bal. | 26–30 | 5–7 | - | ≤1 | - | - | ≤0.75 | ≤1 | - | ≤1 | - |
| Wrought CoCrWNi | F90 | 5832-5 | R30605 | Bal. | 19–21 | - | - | 9–11 | - | - | ≤3 | ≤0.4 | 14–16 | 1–2 | - |
| Forged CoCrMo | F799 | 5832-12 | R31538 | Bal. | 26–30 | 5–7 | - | ≤1 | 0.3–1 | - | ≤0.75 | ≤1 | - | ≤1 | - |
| Wrought CoNiCr | F562 | 5832-6 | R30035 | Bal. | 19–21 | 9–10.5 | ≤1 | 33–37 | - | - | ≤1 | ≤0.15 | - | ≤0.15 | - |
| Wrought TiAlV | F136 | 5832-3 | R56401 | - | - | - | Bal. | - | 5.5–6.5 | 3.5–4.5 | ≤0.25 | - | - | - | - |
| Wrought TiAlNb | F1295 | 5832-11 | R56700 | - | - | - | Bal. | - | 5.5–6.5 | - | - | ≤0.25 | - | - | 6.5–7.5 |
| Wrought SS 316L | F138 | 5832-1 | S31673 | - | 17–19 | 2.25–3 | - | 13–15 | - | - | Bal. | ≤0.75 | - | ≤2 | - |
| Wrought SS 316L | F139 | 5832-1 | S31673 | <0.1 | 17–19 | 2.25–3 | - | 13–15 | - | - | Bal. | ≤0.75 | - | ≤2 | - |
| Cast TiALV | F1108 | | R56406 | - | - | - | Bal. | - | 5.5–6.75 | 3.5–4.5 | ≤0.3 | - | - | - | - |
| Wrought CrNiMo SS | F1350 | | S31673 | - | 17–19 | 2.25–3 | - | 13–15 | - | - | Bal. | ≤0.75 | - | ≤2 | - |
| Wrought TiAlV | F1472 | 5832-3 | R56400 | - | - | - | Bal. | - | 5.5–6.75 | 3.5–4.5 | ≤0.33 | - | - | - | - |
| Wrought CoCrFeNiMo | F1058 | 5832-7 | R30003 | 39-41 | 19–21 | 6–8 | Bal. | 14–16 | - | - | Bal. | ≤1.2 | - | 1.5–2.5 | - |
| Wrought CoCrFeNiMo | F1058 | 5832-7 | R30008 | 39-42 | 18.5–21.5 | 6.5–7.5 | Bal. | 15–18 | - | - | Bal. | ≤1.2 | - | 1–2 | - |
| cp Ti | F67 | 5832-2 | R50700 | - | - | - | Bal. | - | - | - | ≤0.5 | - | - | - | - |
| Wrought CrNiMnMo SS | F1586 | 5832-9 | S31675 | - | 19.5–22 | 2–3 | - | 9–11 | - | - | Bal. | ≤0.75 | - | 2–4.25 | 0.25–0.8 |
Table 2.
Data coverage for pooled serum (blue) and whole blood (red) cobalt concentrations shown in
Figure 2.
Table 2.
Data coverage for pooled serum (blue) and whole blood (red) cobalt concentrations shown in
Figure 2.
| | Serum | Whole Blood |
|---|
| Time (Months) | Reported Participants | Contributing Cohorts | Reported Participants | Contributing Cohorts |
|---|
| 0 | 428 | 10 | 452 | 13 |
| 2 | 112 | 2 | - | - |
| 3 | 213 | 4 | 95 | 3 |
| 6 | 260 | 6 | 194 | 6 |
| 12 | 636 | 14 | 324 | 11 |
| 24 | 711 | 15 | 344 | 10 |
| 36 | 294 | 6 | 144 | 3 |
| 48 | 190 | 4 | - | - |
| 60 | 471 | 10 | 266 | 8 |
| 72 | 69 | 2 | - | - |
| 84 | 161 | 3 | - | - |
| 120 | - | - | 124 | 2 |
Table 3.
Cohorts and participants contributing to pooled urinary cobalt concentration phase.
Table 3.
Cohorts and participants contributing to pooled urinary cobalt concentration phase.
| Time Phase | Reported Participants | Contributing Cohorts |
|---|
| Preop | 103 | 3 |
| Early | 155 | 5 |
| Middle | 139 | 5 |
| Late | 103 | 3 |
Table 5.
Cohorts and participants contributing to pooled urinary chromium concentration bins.
Table 5.
Cohorts and participants contributing to pooled urinary chromium concentration bins.
| Time Phase | Reported Participants | Contributing Cohorts |
|---|
| Preop | 103 | 3 |
| Early | 155 | 5 |
| Middle | 139 | 5 |
| Late | 103 | 3 |
Table 6.
Data coverage for pooled weighted serum titanium concentrations shown in
Figure 6.
Table 6.
Data coverage for pooled weighted serum titanium concentrations shown in
Figure 6.
| Time (Months) | Reported Participants | Contributing Cohorts |
|---|
| 0 | 388 | 15 |
| 12 | 412 | 16 |
| 24 | 268 | 11 |
| 36 | 161 | 6 |
| 60 | 200 | 7 |
| 84 | 94 | 2 |
| 108 | 66 | 2 |
Table 7.
One-phase association model output for serum and whole blood Co and Cr. Best-fit values, 95% confidence intervals, and goodness-of-fit statistics are shown for serum and whole blood Co and Cr trajectories modeled with a one-phase exponential association. Variables include baseline concentration (Y0), plateau concentration, rate constant (K), time constant (τ = 1/K), half-time (t½ = ln2/K), and span (plateau—Y0). Goodness-of-fit statistics (R2, coefficient of determination; Sy.x, standard deviation of residuals, sum of squares; df, degrees of freedom) are provided.
Table 7.
One-phase association model output for serum and whole blood Co and Cr. Best-fit values, 95% confidence intervals, and goodness-of-fit statistics are shown for serum and whole blood Co and Cr trajectories modeled with a one-phase exponential association. Variables include baseline concentration (Y0), plateau concentration, rate constant (K), time constant (τ = 1/K), half-time (t½ = ln2/K), and span (plateau—Y0). Goodness-of-fit statistics (R2, coefficient of determination; Sy.x, standard deviation of residuals, sum of squares; df, degrees of freedom) are provided.
| One Phase Association | Serum Co | Whole Blood Co | Serum Cr | Whole Blood Cr |
|---|
| Best-fit values | | | | |
| Y0 | 0.175 | 0.454 | 0.285 | 0.304 |
| Plateau | 1.964 | 1.958 | 2.015 | 0.989 |
| K | 0.176 | 0.121 | 0.086 | 0.288 |
| Tau | 5.688 | 8.275 | 11.57 | 3.471 |
| Half-time | 3.943 | 5.736 | 8.021 | 2.406 |
| Span | 1.789 | 1.504 | 1.731 | 0.685 |
| 95% CI (profile likelihood) | | | | |
| Y0 | 0.067 to 0.283 | 0.378 to 0.530 | 0.185 to 0.382 | 0.254 to 0.353 |
| Plateau | 1.91 to 2.02 | 1.885 to 2.038 | 1.961 to 2.071 | 0.957 to 1.021 |
| K | 0.152 to 0.205 | 0.095 to 0.156 | 0.076 to 0.099 | 0.228 to 0.376 |
| Tau | 4.878 to 5.598 | 6.413 to 10.57 | 10.09 to 13.24 | 2.685 to 4.382 |
| Half-time | 3.381 to 4.573 | 4.445 to 7.328 | 6.995 to 9.179 | 1.843 to 3.038 |
| Goodness of Fit | | | | |
| df | 3619 | 1940 | 3688 | 1936 |
| R-squared | 0.197 | 0.364 | 0.218 | 0.213 |
| Sum of Squares | 5423 | 1203 | 4153 | 562.2 |
| Sy.x | 1.224 | 0.788 | 1.061 | 0.539 |
Table 8.
Exponential rise-and-decay output for serum Ti. Best-fit values and goodness-of-fit statistics are shown for serum titanium trajectories modeled with an exponential rise–decay association. Variables include baseline concentration (Y0), scaling parameter (A), and rate constants for the rising (kᵣ) and decaying (kd) phases. Goodness-of-fit statistics (R2, coefficient of determination; Sy.x, standard deviation of residuals; degrees of freedom; sum of squares) are provided.
Table 8.
Exponential rise-and-decay output for serum Ti. Best-fit values and goodness-of-fit statistics are shown for serum titanium trajectories modeled with an exponential rise–decay association. Variables include baseline concentration (Y0), scaling parameter (A), and rate constants for the rising (kᵣ) and decaying (kd) phases. Goodness-of-fit statistics (R2, coefficient of determination; Sy.x, standard deviation of residuals; degrees of freedom; sum of squares) are provided.
| Exponential Rise then Decay | Serum Ti |
|---|
| Best-fit values | |
| Y0 | 0.6668 |
| A | ~3.000 |
| kr | 0.04915 |
| kd | 0.02836 |
| Goodness of Fit | |
| Degrees of Freedom | 1520 |
| R-squared | 0.2215 |
| Sum of Squares | 932.0 |
| Sy.x | 0.7830 |
Table 9.
Summary of cross-sectional reported mean serum molybdenum (Mo) concentrations following primary MoM total hip arthroplasty (THA) or Birmingham Hip Resurfacing (BHR). DL = detection limit.
Table 9.
Summary of cross-sectional reported mean serum molybdenum (Mo) concentrations following primary MoM total hip arthroplasty (THA) or Birmingham Hip Resurfacing (BHR). DL = detection limit.
| Study | Fluid | Average Follow-Up (Months) | Mean Mo Concentration (µg/L) | Hip Device | Number of Patients |
|---|
| Moroni 2010 [76] | Serum | 58 | 0.84 | MoM–BHR | 20 |
| Moroni 2010 [76] | Serum | 24 | <DL (0.83) | MoM–BHR | 15 |
| Moroni 2010 [76] | Serum | 56 | 0.9 | MoM–THA | 35 |
| Moroni 2010 [76] | Serum | 26 | 0.97 | MoM–THA | 25 |
| Savarino 2014 [77] | Serum | 24 | 0.89 | MoM–BHR | 14 |
| Savarino 2014 [77] | Serum | 60 | 0.84 | MoM–BHR | 19 |
| Savarino 2014 [77] | Serum | 108 | 0.85 | MoM–BHR | 22 |
Table 10.
Summary of model performance metrics comparison. Mean absolute error (MAE), mean squared error (MSE), root mean squared error (RMSE), and coefficient of determination (R2) are presented.
Table 10.
Summary of model performance metrics comparison. Mean absolute error (MAE), mean squared error (MSE), root mean squared error (RMSE), and coefficient of determination (R2) are presented.
| | MAE | MSE | RMSE | R2 |
|---|
| Co | 0.417 | 0.329 | 0.573 | 0.861 |
| Cr | 0.374 | 0.245 | 0.495 | 0.522 |
| Ti | 0.233 | 0.144 | 0.380 | 0.707 |
| Mo | 0.857 | 1.483 | 1.218 | 0.718 |
| Ni | 0.233 | 0.106 | 0.325 | 0.297 |
Table 11.
Reported cobalt concentration thresholds and reference limits in the literature. Values are drawn from peer-reviewed studies and regulatory guidance, primarily in the context of monitoring patients with hip devices.
Table 11.
Reported cobalt concentration thresholds and reference limits in the literature. Values are drawn from peer-reviewed studies and regulatory guidance, primarily in the context of monitoring patients with hip devices.
| Biological Fluid | Co Concentration (μg/L) | Source | Threshold/Limit Type | Context |
|---|
| Blood/Serum | 7 | MHRA | Threshold | Regulatory level of concern, prompting closer follow-up. |
| Blood | 4.97 | Hart 2011 [88] | Threshold | Study-derived cutoff for predicting MoM failure. |
| Blood | 4.5 | Sidaginamale 2013 [89] | Threshold | Proposed to indicate abnormal wear in MoM hips. |
| Serum | 4.0 (unilateral)/5.0 (bilateral) | Van Der Straeten 2013 [90] | Acceptable upper limits | Derived in well- vs. poorly functioning hip cohorts. |
| Not specified (most likely Serum or WB) | 3 (upper limit, low risk); 10 (lower limit, high risk) | Kwon 2014 (J Arthroplasty Consensus) [91] | Risk Bands | US consensus for risk stratification; fluid not specified. |
| Blood/Serum | 2–7 | EFORT (European [92] Guidelines) | Concern Range | Values for clinical concern, need for additional imaging even in asymptomatic patients. |
Table 12.
Reference ranges for Co, Cr, Ti, Ni, and Mo in whole blood and serum as reported by two clinical laboratories.
Table 12.
Reference ranges for Co, Cr, Ti, Ni, and Mo in whole blood and serum as reported by two clinical laboratories.
| Element | Fluid | Reference Ranges (µg/L) | Laboratory |
|---|
| Co | Whole Blood | 0–0.5 | London Health Sciences Centre Laboratories |
| Cr | Whole Blood | 0–1.2 | London Health Sciences Centre Laboratories |
| Ti | Whole Blood | 0–3 | London Health Sciences Centre Laboratories |
| Mo | Whole Blood | 0–1.6 | London Health Sciences Centre Laboratories |
| Ni | Whole Blood | 0–1.3 | London Health Sciences Centre Laboratories |
| Co | Serum | 0–0.5 | London Health Sciences Centre Laboratories |
| Cr | Serum | 0–0.65 | London Health Sciences Centre Laboratories |
| Ti | Serum | 0–3 | London Health Sciences Centre Laboratories |
| Co | Serum | <1.0 | Mayo Clinic Laboratory |
| Ti | Serum | <2.0 | Mayo Clinic Laboratory |
| Mo | Serum | 0.3–2.0 | Mayo Clinic Laboratory |
| Ni | Serum | <2.0 | Mayo Clinic Laboratory |
Table 13.
Multiple studies reporting Co and Cr concentrations with at least one outlier, error bar, or range exceeding the 7 μg/L threshold proposed by the MHRA. Follow-up times indicate the specific timepoint at which reported variability crossed the threshold. Superscript letters correspond to the cited source study.
Table 13.
Multiple studies reporting Co and Cr concentrations with at least one outlier, error bar, or range exceeding the 7 μg/L threshold proposed by the MHRA. Follow-up times indicate the specific timepoint at which reported variability crossed the threshold. Superscript letters correspond to the cited source study.
| Metal | Fluid | Follow-Up (Month) with Ranges/Error/Outliers Crossing Threshold | Source Studies |
|---|
| Co | Whole Blood | 48 a, 84 a | Bernstein 2012 [96] a |
| 6 b, 12 b | Bernstein 2012 [96] b |
| 12 c | Høl 2021 [97] c |
| 36 d | Forsthoefel 2017 [98] d |
| 12 e, 24 e | Smolders 2011 [99] e |
| Cr | Whole Blood | 48 a | Bernstein 2012 [96] a |
| 12 b | Bernstein 2012 [96] b |
| 12 c, 60 c | Hol 2021 [97] c |
| 36 d | Forsthoefel 2017 [98] d |
| 24 e | Smolders 2011 [99] e |
| Co | Serum | 12 a | Garbuz 2010 [100] a |
| 72–108 b | Koper 2021 [100] b |
| 6 c | Dahlstrand 2009 [38] c |
| 3 d, 12 d, 24 d | Witzleb 2006 [39] d |
| 56 e | Moroni 2010 [76] e |
| Cr | Serum | 132 a, 168 a | Maezawa 2018 [101] a |
| 24–48 b, 84–108 b | Koper 2021 [102] b |
| 6 c | Dahlstrand 2009 [38] c |
| 3 d, 12 d, 24 d | Witzleb 2006 [39] d |
| 56, e 58 e | Moroni 2010 [76] e |