Optimization of Oil Recovery from Japonica Luna Rice Bran by Supercritical Carbon Dioxide Applying Design of Experiments: Characterization of the Oil and Mass Transfer Modeling
Abstract
:1. Introduction
2. Materials and Methods
2.1. Raw Material
2.2. Reagents
2.3. Methods and Equipments
2.4. Design of Experiments
2.5. Mathematical Modeling of scCO2 Kinetics Extractions
3. Results and Discussion
3.1. Design of Experiments
3.1.1. Fractional Factorial Design
3.1.2. Central Composite Design
3.2. Extract Compositions—Fatty Acid Methyl Esters (FAMEs) Analysis
3.3. Mathematical Models Based on Differential Mass Balances
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Pandey, R.; Shrivastava, S.L. Comparative evaluation of rice bran oil obtained with two-step microwave assisted extraction and conventional solvent extraction. J. Food Eng. 2018, 218, 106–114. [Google Scholar] [CrossRef]
- Pinto, T.I.; Coelho, J.A.; Pires, B.I.; Neng, N.R.; Nogueira, J.M.; Bordado, J.C.; Sardinha, J.P. Supercritical carbon dioxide extraction, antioxidant activity, and fatty acid composition of bran oil from rice varieties cultivated in Portugal. Separations 2021, 8, 115. [Google Scholar] [CrossRef]
- Okajima, I.; Ito, K.; Aoki, Y.; Kong, C.Y.; Sako, T. Extraction of Rice Bran Oil Using CO2-Expanded Hexane in the Two-Phase Region. Energies 2022, 15, 2594. [Google Scholar] [CrossRef]
- Khan, S.H.; Butt, M.S.; Sharif, M.K.; Sameen, A.; Mumtaz, S.; Sultan, M.T. Functional properties of protein isolates extracted from stabilized rice bran by microwave, dry heat, and parboiling. J. Agric. Food Chem. 2011, 59, 2416–2420. [Google Scholar] [CrossRef] [PubMed]
- El Boulifi, N.; Bouaid, A.; Martinez, M.; Aracil, J. Optimization and oxidative stability of biodiesel production from rice bran oil. Renew. Energy 2013, 53, 141–147. [Google Scholar] [CrossRef]
- Burlando, B.; Cornara, L. Therapeutic properties of rice constituents and derivatives (Oryza sativa L.): A review update. Trends Food Sci. Technol. 2014, 40, 82–98. [Google Scholar] [CrossRef]
- Balachandran, C.; Mayamol, P.N.; Thomas, S.; Sukumar, D.; Sundaresan, A.; Arumughan, C. An ecofriendly approach to process rice bran for high quality rice bran oil using supercritical carbon dioxide for nutraceutical applications. Bioresour. Technol. 2008, 99, 2905–2912. [Google Scholar] [CrossRef]
- Mingyai, S.; Kettawan, A.; Srikaeo, K.; Singanusong, R. Physicochemical and antioxidant properties of rice bran oils produced from colored rice using different extraction methods. J. Oleo Sci. 2017, 66, 565–572. [Google Scholar] [CrossRef] [Green Version]
- Mingyai, S.; Srikaeo, K.; Kettawan, A.; Singanusong, R.; Nakagawa, K.; Kimura, F.; Ito, J. Effects of extraction methods on phytochemicals of rice bran oils produced from colored rice. J. Oleo Sci. 2018, 67, 135–142. [Google Scholar] [CrossRef] [Green Version]
- Tomita, K.; Machmudah, S.; Wahyudiono; Fukuzato, R.; Kanda, H.; Quitain, A.T.; Sasaki, M.; Goto, M. Extraction of rice bran oil by supercritical carbon dioxide and solubility consideration. Sep. Purif. Technol. 2014, 125, 319–325. [Google Scholar] [CrossRef]
- Manosroi, A.; Ruksiriwanich, W.; Abe, M.; Sakai, H.; Manosroi, W.; Manosroi, J. Biological activities of the rice bran extract and physical characteristics of its entrapment in niosomes by supercritical carbon dioxide fluid. J. Supercrit. Fluids 2010, 54, 137–144. [Google Scholar] [CrossRef]
- Soares, J.F.; Dal Prá, V.; De Souza, M.; Lunelli, F.C.; Abaide, E.; Da Silva, J.R.F.; Kuhn, R.C.; Martínez, J.; Mazutti, M.A. Extraction of rice bran oil using supercritical CO2 and compressed liquefied petroleum gas. J. Food Eng. 2016, 170, 58–63. [Google Scholar] [CrossRef] [Green Version]
- Wang, C.-H.; Chen, C.-R.; Wu, J.-J.; Wang, L.-Y.; Chang, C.-M.J.; Ho, W.-J. Designing supercritical carbon dioxide extraction of rice bran oil that contain oryzanols using response surface methodology. J. Sep. Sci. 2008, 31, 1399–1407. [Google Scholar] [CrossRef]
- Sparks, D.; Hernandez, R.; Zappi, M.; Blackwell, D.; Fleming, T. Extraction of rice brain oil using supercritical carbon dioxide and propane. J. Am. Oil Chem. Soc. 2006, 83, 885–891. [Google Scholar] [CrossRef]
- Benito-Román, O.; Varona, S.; Sanz, M.T.; Beltrán, S. Valorization of rice bran: Modified supercritical CO2 extraction of bioactive compounds. J. Ind. Eng. Chem. 2019, 80, 273–282. [Google Scholar] [CrossRef]
- Trevisani Juchen, P.; Nolasco Araujo, M.; Hamerski, F.; Corazza, M.L.; Pedersen Voll, F.A. Extraction of parboiled rice bran oil with supercritical CO2 and ethanol as co-solvent: Kinetics and characterization. Ind. Crops Prod. 2019, 139, 111506. [Google Scholar] [CrossRef]
- Santos, C.P.; Rato, T.J.; Reis, M.S. Design of Experiments: A comparison study from the non-expert user’s perspective. J. Chemom. 2019, 33, e3087. [Google Scholar] [CrossRef] [Green Version]
- Coelho, J.P.; Filipe, R.M.; Paula Robalo, M.; Boyadzhieva, S.; Cholakov, G.S.; Stateva, R.P. Supercritical CO2 extraction of spent coffee grounds. Influence of co-solvents and characterization of the extracts. J. Supercrit. Fluids 2020, 161, 104825. [Google Scholar] [CrossRef]
- Coelho, J.P.; Bernotaityte, K.; Miraldes, M.A.; Mendonca, A.F.; Stateva, R.P. Solubility of ethanamide and 2-propenamide in supercritical carbon dioxide. Measurements and correlation. J. Chem. Eng. Data 2009, 54, 2546–2549. [Google Scholar] [CrossRef]
- Bobo-García, G.; Davidov-Pardo, G.; Arroqui, C.; Marín-Arroyo, M.R.; Vírseda, P.; Marín-Arroyo, M.R.; Navarro, M. Intra-laboratory validation of microplate methods for total phenolic content and antioxidant activity on polyphenolic extracts, and comparison with conventional spectrophotometric methods. J. Sci. Food Agric. 2015, 95, 204–209. [Google Scholar] [CrossRef]
- Reis, F.S.; Pereira, E.; Barros, L.; Sousa, M.J.; Martins, A.; Ferreira, I.C.F.R. Biomolecule profiles in inedible wild mushrooms with antioxidant value. Molecules 2011, 16, 4328–4338. [Google Scholar] [CrossRef] [PubMed]
- Coelho, J.; Veiga, J.; Karmali, A.; Nicolai, M.; Pinto Reis, C.; Nobre, B.; Palavra, A.; Reis, C.P.; Nobre, B.; Palavra, A. Supercritical CO2 Extracts and Volatile Oil of Basil (Ocimum basilicum L.) Comparison with Conventional Methods. Separations 2018, 5, 21. [Google Scholar] [CrossRef] [Green Version]
- De Melo, M.M.R.; Silvestre, A.J.D.; Silva, C.M. Supercritical fluid extraction of vegetable matrices: Applications, trends and future perspectives of a convincing green technology. J. Supercrit. Fluids 2014, 92, 115–176. [Google Scholar] [CrossRef]
- Oliveira, E.L.G.; Silvestre, A.J.D.; Silva, C.M. Review of kinetic models for supercritical fluid extraction. Chem. Eng. Res. Des. 2011, 89, 1104–1117. [Google Scholar] [CrossRef]
- Huang, Z.; Shi, X.-H.; Jiang, W.-J. Theoretical models for supercritical fluid extraction. J. Chromatogr. A 2012, 1250, 2–26. [Google Scholar] [CrossRef]
- Sovovà, H.; Stateva, R.P. Supercritical fluid extraction from vegetable materials. Rev. Chem. Eng. 2011, 27, 79–156. [Google Scholar] [CrossRef]
- Del Valle, J.M. Extraction of natural compounds using supercritical CO2: Going from the laboratory to the industrial application. J. Supercrit. Fluids 2015, 96, 180–199. [Google Scholar] [CrossRef]
- Tan, C.S.; Liou, D.C. Modeling of desorption at super critical conditions. AIChE J. 1989, 35, 1029–1031. [Google Scholar] [CrossRef]
- Grosso, C.; Coelho, J.P.; Pessoa, F.L.P.; Fareleira, J.M.N.A.; Barroso, J.G.; Urieta, J.S.; Palavra, A.A.F.; Sovová, H. Mathematical modelling of supercritical CO2 extraction of volatile oils from aromatic plants. Chem. Eng. Sci. 2010, 65, 3579–3590. [Google Scholar] [CrossRef]
- Sovová, H. Rate of the vegetable oil extraction with supercritical CO2-I. Modelling of extraction curves. Chem. Eng. Sci. 1994, 49, 409–414. [Google Scholar] [CrossRef]
- Sovová, H. Mathematical model for supercritical fluid extraction of natural products and extraction curve evaluation. J. Supercrit. Fluids 2005, 33, 35–52. [Google Scholar] [CrossRef]
- Jesus, S.P.; Grimaldi, R.; Hense, H. Recovery of γ-oryzanol from rice bran oil byproduct using supercritical fluid extraction. J. Supercrit. Fluids 2010, 55, 149–155. [Google Scholar] [CrossRef]
- Chia, S.L.; Boo, H.C.; Muhamad, K.; Sulaiman, R.; Umanan, F.; Chong, G.H. Effect of Subcritical Carbon Dioxide Extraction and Bran Stabilization Methods on Rice Bran Oil. JAOCS J. Am. Oil Chem. Soc. 2015, 92, 393–402. [Google Scholar] [CrossRef]
- Reis, N.; Castanho, A.; Lageiro, M.; Pereira, C.; Brites, C.M.; Vaz-Velho, M. Microwave-Assisted Method and Its Effects on GABA and Gamma-Oryzanol Compounds. Foods 2022, 11, 912. [Google Scholar] [CrossRef]
- Coelho, J.P.; Filipe, R.M.; Robalo, M.P.; Stateva, R.P. Recovering value from organic waste materials: Supercritical fluid extraction of oil from industrial grape seeds. J. Supercrit. Fluids 2018, 141, 68–77. [Google Scholar] [CrossRef]
- Wilke, C.R.; Chang, P. Correlations of diffusion coefficients in dilute solutions. AICHE J. 1955, 1, 264–270. [Google Scholar] [CrossRef]
- Wakao, N.; Kaguei, S. Heat and Mass Transfer in Packed Beds; Gordon and Breach Science Publisher: London, UK, 1982; ISBN 0677058608. [Google Scholar]
- Huang, Z.; Yang, M.; Liu, S.; Ma, Q. Supercritical carbon dioxide extraction of Baizhu: Experiments and modeling. J. Supercrit. Fluids 2011, 58, 31–39. [Google Scholar] [CrossRef]
- Nagy, B. Characterization of packed beds of plant materials processed by supercritical fluid extraction. J. Food Eng. 2008, 88, 104–113. [Google Scholar] [CrossRef]
- Ferreira, S.R.S.; Nikolov, Z.L.; Doraiswamy, L.K.; Meireles, M.A.A.; Petenate, A.J. Supercritical fluid extraction of black pepper (Piper nigrun L.) essential oil. J. Supercrit. Fluids 1999, 14, 235–245. [Google Scholar] [CrossRef]
- del Valle, J.; Rivera, O.; Mattea, M.; Ruetsch, L.; Daghero, J.; Flores, A. Supercritical CO2 processing of pretreated rosehip seeds: Effect of process scale on oil extraction kinetics. J. Supercrit. Fluids 2004, 31, 159–174. [Google Scholar] [CrossRef]
- Ivanovic, J.; Ristic, M.; Skala, D. Supercritical CO2 extraction of Helichrysum italicum: Influence of CO2 density and moisture content of plant material. J. Supercrit. Fluids 2011, 57, 129–136. [Google Scholar] [CrossRef]
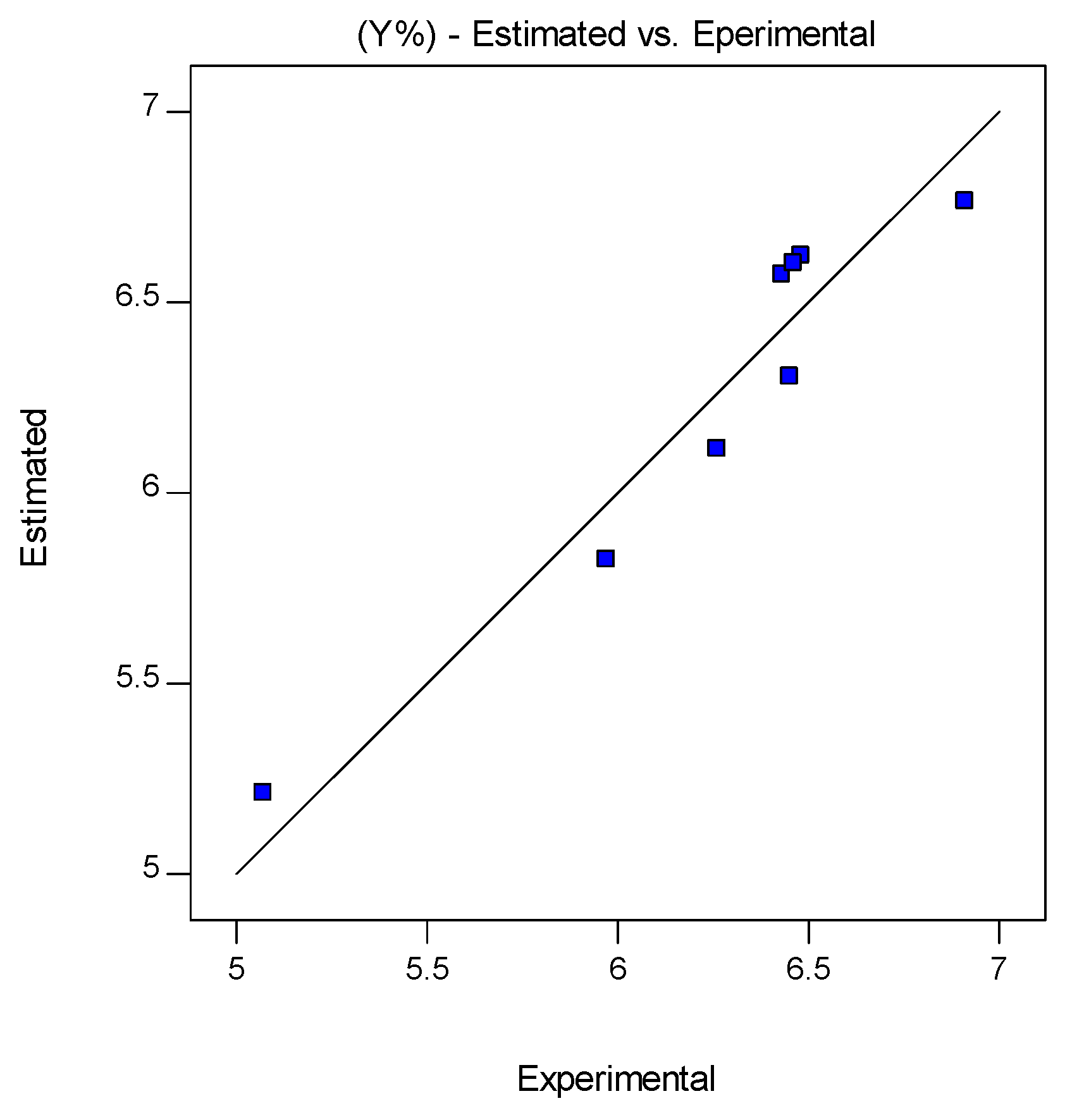
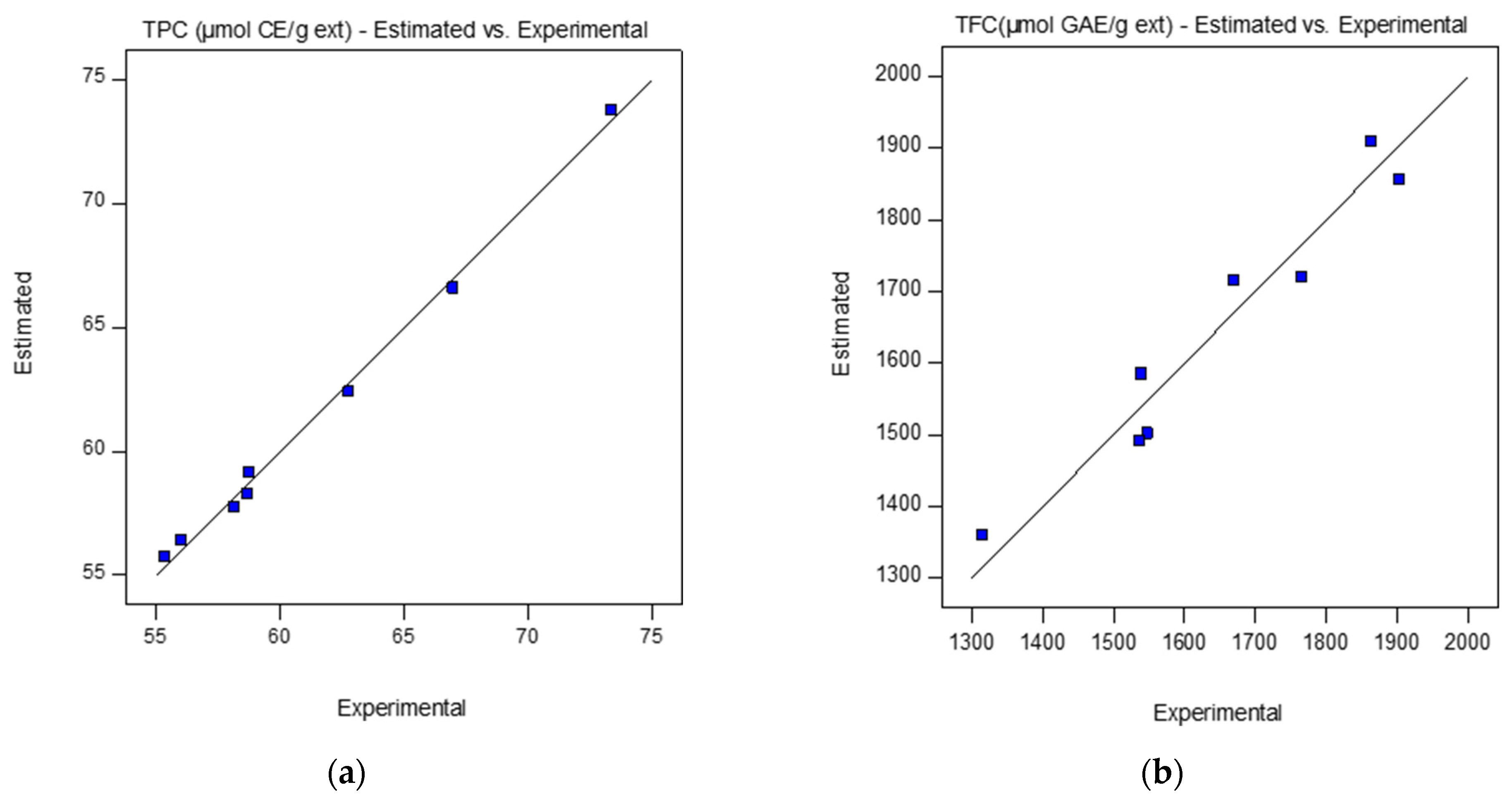
| Run | Pressure (MPa) | Temperature (K) | Flow Rate (kg/min) | Yield (%) | TPC (μmol GAE/g ext) | Flavonoids (μmol CE/g ext) |
|---|---|---|---|---|---|---|
| HE | - | - | - | 8.20 | 60.24 ± 3.91 | 2092.19 ± 78.97 |
| A1 | 25.0 | 313.15 | 2.7 × 10−3 | 6.48 | 58.76 ± 2.67 | 1538.99 ± 97.96 |
| A2 | 40.0 | 353.15 | 0.9 × 10−3 | 6.91 | 58.67 ± 0.52 | 1547.83 ± 78.21 |
| A3 | 25.0 | 313.15 | 0.9 × 10−3 | 6.26 | 58.14 ± 3.31 | 1765.87 ± 29.90 |
| A4 | 40.0 | 313.15 | 0.9 × 10−3 | 6.43 | 73.41 ± 1.55 | 1863.10 ± 124.90 |
| A5 | 40.0 | 313.15 | 2.7 × 10−3 | 5.97 | 66.99 ± 3.55 | 1902.88 ± 105.60 |
| A6 | 25.0 | 353.15 | 0.9 × 10−3 | 5.07 | 55.35 ± 1.12 | 1314.32 ± 79.87 |
| A7 | 25.0 | 353.15 | 2.7 × 10−3 | 6.45 | 62.79 ± 3.70 | 1536.78 ± 174.03 |
| A8 | 40.0 | 353.15 | 2.7 × 10−3 | 6.46 | 56.02 ± 3.50 | 1670.11 ± 81.74 |
| Run | Pressure (MPa) | Temperature (K) | Yield (%) | TPC (μmol GAE/g ext) | Flavonoids (μmol CE/g ext) |
|---|---|---|---|---|---|
| A9 | 30.0 | 323.15 | 5.72 | 59.98 ± 1.99 | 1537.24 ± 129.38 |
| A10 | 40.0 | 323.15 | 6.50 | 62.21 ± 2.58 | 1565.42 ± 73.94 |
| A11 | 30.0 | 353.15 | 6.57 | 53.38 ± 3.03 | 1636.68 ± 77.85 |
| A12 | 42.0 | 338.15 | 6.67 | 60.04 ± 2.12 | 1707.95 ± 24.58 |
| A13 | 35.0 | 338.15 | 6.81 | 68.58 ± 0.84 | 1641.84 ± 194.11 |
| A14 | 35.0 | 338.15 | 6.67 | 65.89 ± 1.95 | 1631.53 ± 116.81 |
| A15 | 35.0 | 317.15 | 6.32 | 63.27 ± 1.99 | 1907.39 ± 4.52 |
| A16 | 35.0 | 338.15 | 7.12 | 60.78 ± 0.96 | 1831.15 ± 254.14 |
| A17 | 40.0 | 353.15 | 6.91 | 54.98 ± 2.37 | 1683.09 ± 155.90 |
| A18 | 35.0 | 359.15 | 6.87 | 63.01 ± 1.71 | 1751.05 ± 121.03 |
| A19 | 27.9 | 338.15 | 5.93 | 61.97 ± 2.04 | 1771.49 ± 128.09 |
| Yield (%) | ||||
|---|---|---|---|---|
| Source | SS a | MS b | F-Value | p-Value |
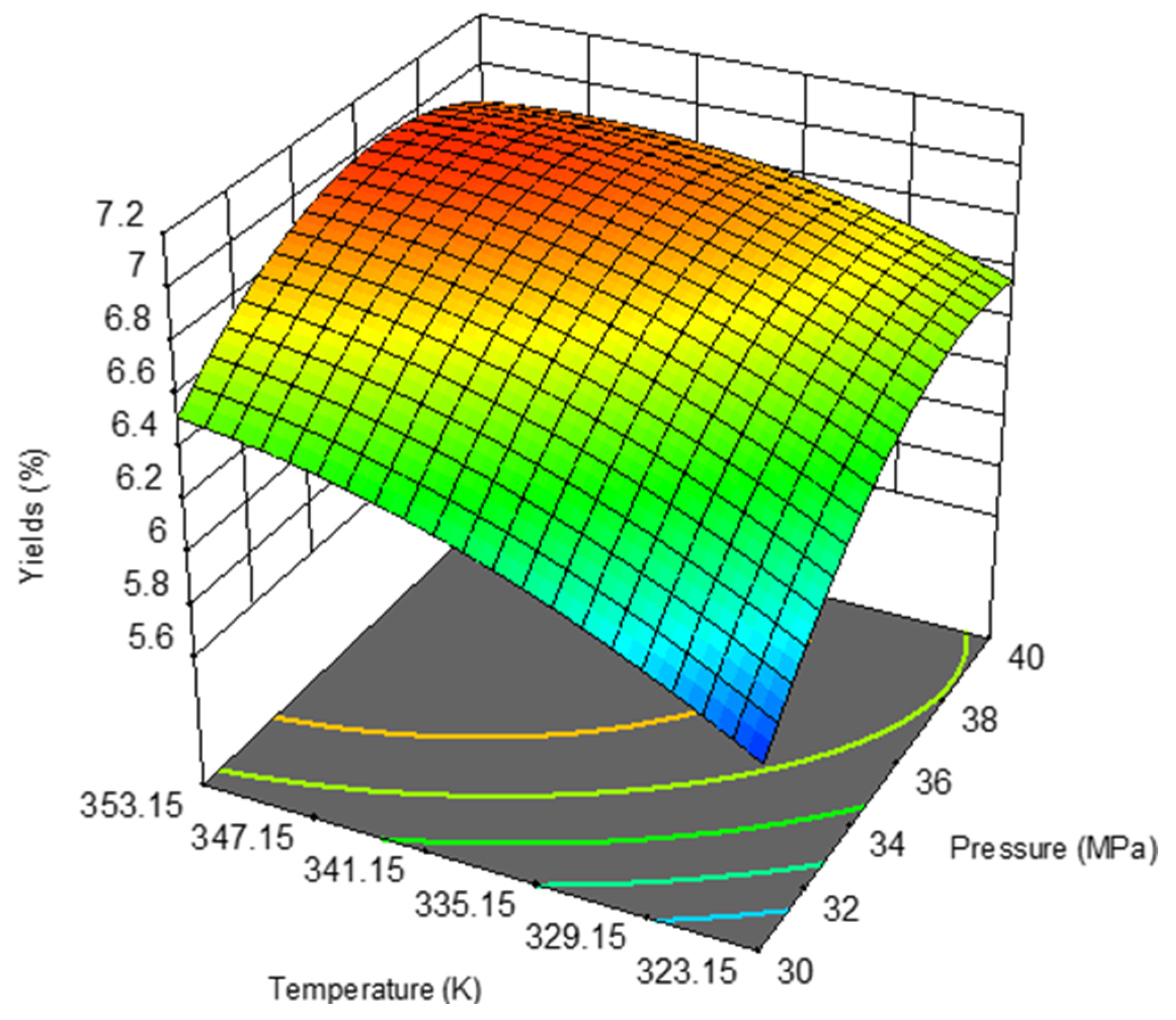
| Model | 1.6000 | 0.3206 | 15.32 | 0.0047 |
| X1-Pressure (MPa) | 0.5867 | 0.5867 | 28.04 | 0.0032 |
| X2-Temperature(K) | 0.5191 | 0.5191 | 24.81 | 0.0042 |
| X1X2 | 0.0484 | 0.0484 | 2.31 | 0.1888 |
| X12 | 0.4342 | 0.4342 | 20.75 | 0.0061 |
| X22 | 0.0951 | 0.0951 | 4.55 | 0.0862 |
| Residual | 0.1046 | 0.0209 | ||
| Lack of Fit | 0.0307 | 0.0102 | 0.2775 | 0.8406 |
| Pure Error | 0.0739 | 0.0369 | ||
| Cor Total | 1.71 | |||
| Pressure (MPa) | Temperature (K) | Yields (%) | TPC (μmol GAE/g ext) | Flavonoids (μmol CE/g ext) | |
|---|---|---|---|---|---|
| Predicted values | 34.3587 | 339.493 | 6.82706 | 61.2809 | 1696.8 |
| Experimental values | 34.5 | 339.5 | 6.79 ± 0.06 | 64.20 ± 5.42 | 1739.1 ± 75.4 |
| FFD | CCD | ||||||
|---|---|---|---|---|---|---|---|
| Run A2 | Run A3 | Run A4 | Run A12 | Run A16 | Run A17 | Run A10 | |
| Fatty Acid | 40/353.15 | 25/313.15 | 40/313.15 | 42/338.15 | 35/338.15 | 40/353.15 | 27.9/338.15 |
| C14:0—Myristic | 0.06 ± 0.02 | 0.08 ± 0.03 | 0.05 ± 0.01 | 0.06 ± 0.02 | 0.06 ± 0.02 | 0.06 ± 0.02 | 0.07 ± 0.03 |
| C16:0—Palmitic | 15.32 ± 0.59 | 15.95 ± 0.61 | 15.35 ± 0.49 | 15.22 ± 0.55 | 15.38 ± 0.63 | 15.12 ± 0.65 | 15.87 ± 0.53 |
| C18:0—Stearic | 1.58 ± 0.08 | 1.84 ± 0.07 | 1.43 ± 0.06 | 1.45 ± 0.07 | 1.41 ± 0.07 | 1.43 ± 0.08 | 1.63 ± 0.07 |
| C18:1—Oleic | 41.37 ± 1.00 | 41.78 ± 0.95 | 41.53 ± 0.89 | 41.65 ± 1.01 | 41.47 ± 1.00 | 41.58 ± 0.96 | 41.31 ± 0.98 |
| C18:2—Linoleic | 39.03 ± 0.97 | 38.69 ± 1.00 | 39.42 ± 1.05 | 39.27 ± 0.94 | 39.65 ± 1.06 | 39.61 ± 1.05 | 39.45 ± 1.01 |
| C18:3—Linolenic | 1.54 ± 0.06 | 1.02 ± 0.07 | 1.34 ± 0.08 | 1.48 ± 0.07 | 1.36 ± 0.06 | 1.46 ± 0.07 | 1.32 ± 0.06 |
| C20:0—Arachidic | 0.25 ± 0.02 | 0.18 ± 0.02 | 0.27± 0.03 | 0.24 ± 0.02 | 0.26 ± 0.02 | 0.12 ± 0.02 | 0.10 ± 0.02 |
| C20:1—Gadoleic | 0.53 ± 0.03 | 0.24 ± 0.02 | 0.38 ± 0.02 | 0.41 ± 0.03 | 0.19 ± 0.02 | 0.34 ± 0.02 | 0.13 ± 0.02 |
| C22:0 —Behenic | 0.11 ± 0.02 | 0.08 ± 0.03 | 0.12 ± 0.02 | 0.09 ± 0.02 | 0.12 ± 0.02 | 0.14 ± 0.02 | 0.07 ± 0.03 |
| C22:1—Erucic | 0.15 ± 0.03 | 0.06 ± 0.02 | 0.07 ± 0.02 | 0.08 ± 0.03 | 0.05 ± 0.02 | 0.12 ± 0.02 | 0.05 ± 0.02 |
| C24:0—Lignoceric | 0.06 ± 0.03 | ----- | 0.07 ± 0.03 | 0.06 ± 0.03 | 0.05 ± 0.04 | 0.07 ± 0.03 | ----- |
| DUFA | 39.03 | 38.69 | 39.42 | 39.27 | 39.65 | 39.61 | 39.45 |
| MUFA | 42.05 | 42.08 | 41.98 | 42.14 | 41.71 | 42.04 | 41.49 |
| SFA | 17.38 | 18.13 | 17.29 | 17.12 | 17.28 | 16.94 | 17.74 |
| UI | 1.201 | 1.195 | 1.208 | 1.207 | 1.210 | 1.213 | 1.204 |
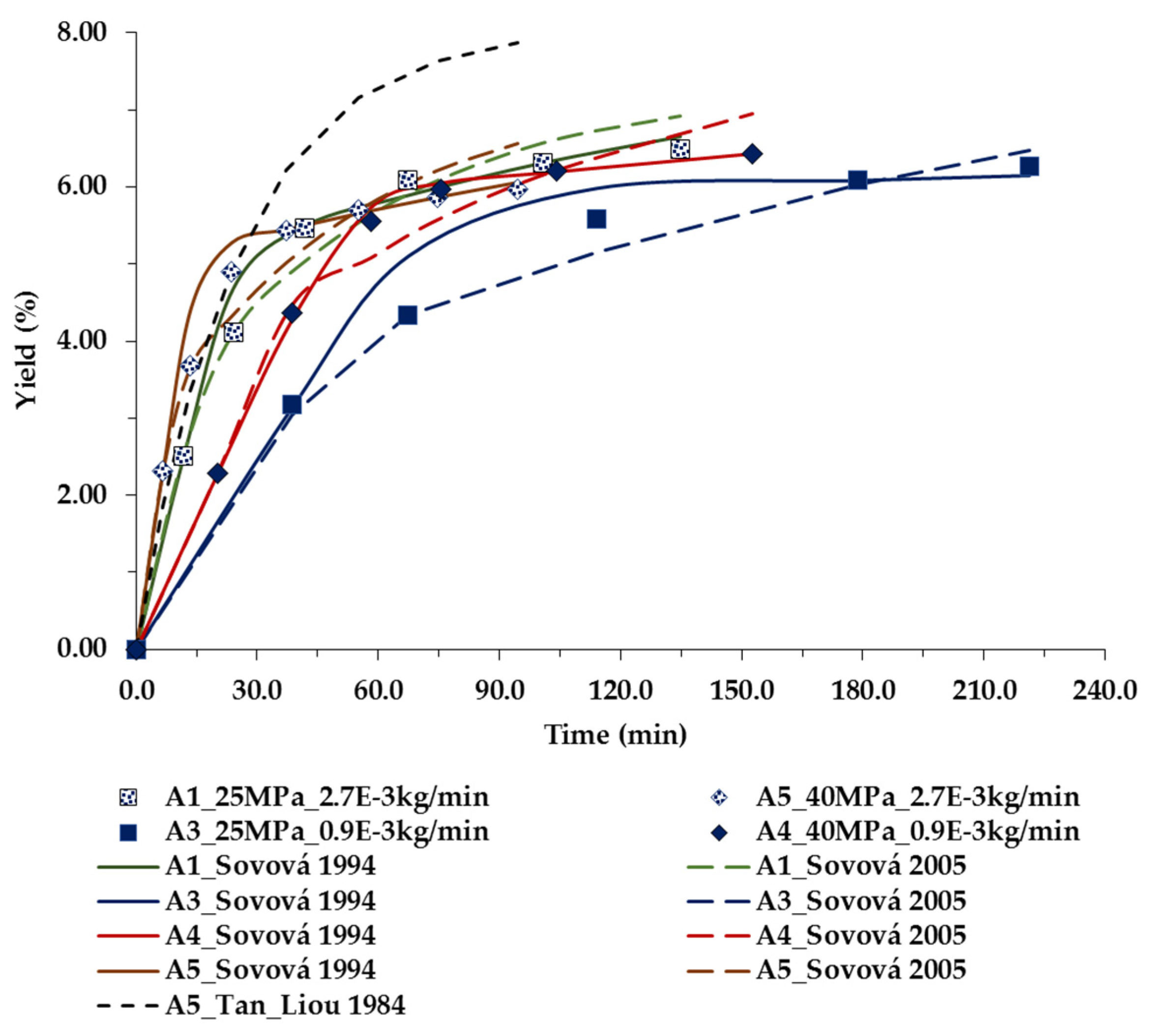
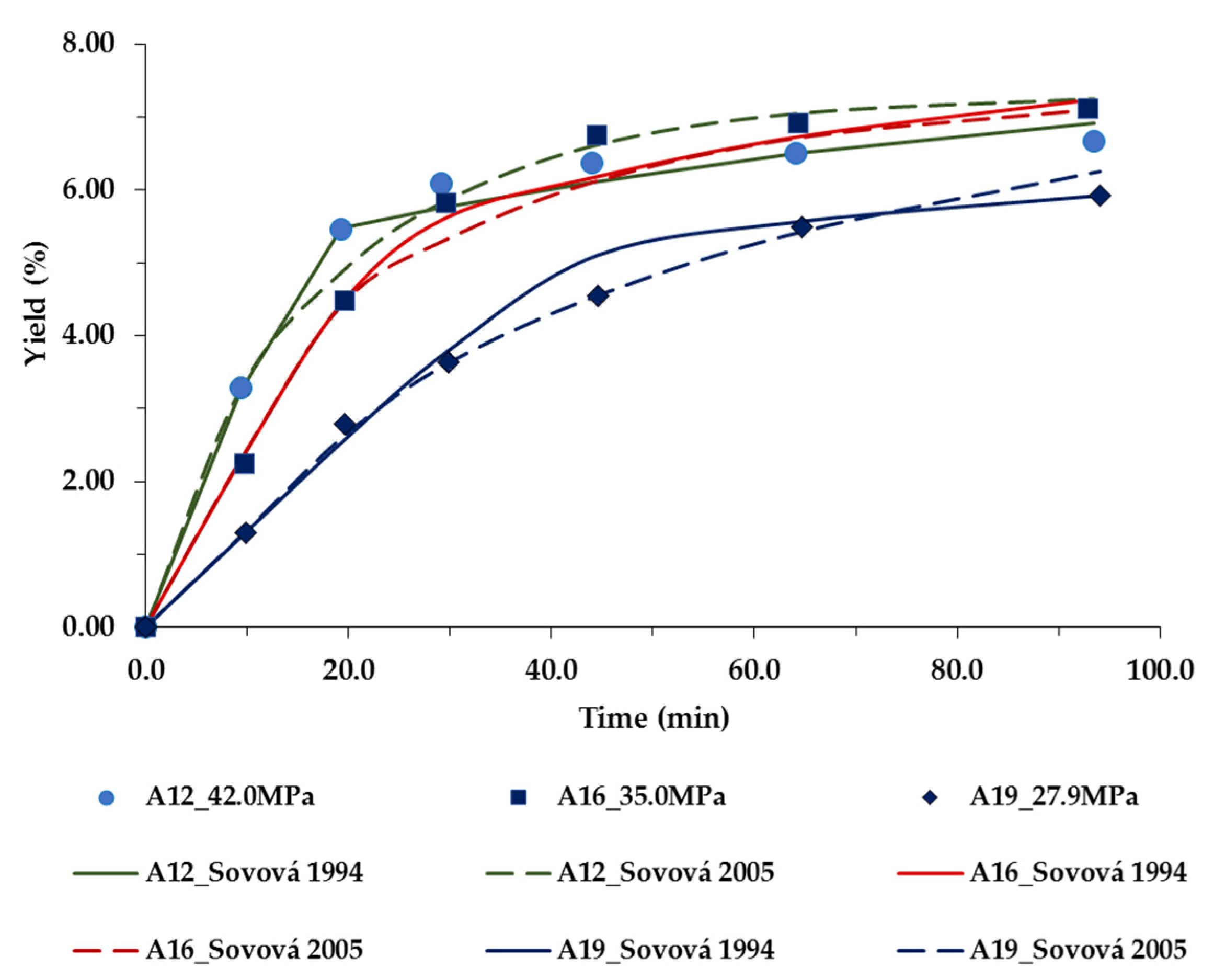
| Run | P (MPa) | T (K) | Flow Rate f (kg/s) × 105 | Superficial Velocity u (m·s−1) × 104 | Co (kg/kg) × 102 | Tan and Liou [28] kd × 104 (s−1) | Sovová [30] ks × 108 (m·s−1)/ kf × 106 (m·s−1) | Sovová [31] ks × 108 (m·s−1) | a D12 × 109 (m2·s−1) | bkf × 106 (m·s−1) |
|---|---|---|---|---|---|---|---|---|---|---|
| A1 (FFD) | 25.0 | 313.15 | 4.49 | 3.32 | 1.30 | 5.056 (8.98%) | 0.609/0.201 (3.42%) | 1.87 (3.86%) | 2.872 | 7.971 |
| A3 (FFD) | 25.0 | 313.15 | 1.62 | 1.19 | 1.30 | 1.431 (10.7%) | 0.0432/0.0876 (5.52%) | 0.444 (3.26%) | 2.872 | 3.401 |
| A6 (FFD) | 25.0 | 353.15 | 1.58 | 1.49 | 0.60 | 0.739 (6.76%) | 0.0376/0.0924 (10.5%) | 0.444 (3.24%) | 5.008 | 6.534 |
| A7 (FFD) | 25.0 | 353.15 | 4.57 | 4.32 | 0.60 | 1.998 (7.97%) | 0.0942/0.230 (7.82%) | 0.790 (4.41%) | 5.008 | 1.581 |
| A19 (CCD) | 27.9 | 338.15 | 3.08 | 2.53 | 1.10 | 3.300 (4.77%) | 0.492/0.188 (4.40%) | 2.85 (2.21%) | 3.826 | 8.120 |
| A9 (CCD) | 30.0 | 323.15 | 3.12 | 2.33 | 1.60 | 4.514 (8.97%) | 0.304/0.155(1.56%) | 1.49 (4.59%) | 3.029 | 6.193 |
| A11 (CCD) | 30.0 | 353.15 | 3.08 | 2.69 | 1.20 | 3.956(6.00%) | 1.099/0.196 (4.87%) | 3.94 (1.24%) | 4.397 | 9.551 |
| A13 (CCD) | 35.0 | 338.15 | 3.12 | 2.40 | 2.20 | 5.872 (5.12%) | 1.572/0.137 (2.87%) | 4.66 (2.63%) | 3.370 | 6.920 |
| A14 (CCD) | 35.0 | 338.15 | 3.09 | 2.38 | 2.20 | 5.884 (7.62%) | 1.627/0.126 (3.13%) | 4.60 (3.69%) | 3.368 | 6.870 |
| A16 (CCD) | 35.0 | 338.15 | 3.11 | 2.40 | 2.20 | 6.337 (6.27%) | 1.711/0.134 (3.77%) | 4.48 (4.77%) | 3.368 | 6.918 |
| A15 (CCD) | 35.0 | 317.15 | 3.15 | 2.22 | 2.00 | 4.964 (8.30%) | 0.948/0.117 (1.90%) | 2.40 (4.13%) | 2.609 | 5.185 |
| A18 (CCD) | 35.0 | 359.15 | 3.10 | 2.62 | 2.10 | 5.663 (4.80%) | 1.769/0.135 (2.13%) | 4.71 (2.81%) | 4.251 | 9.034 |
| A4 (FFD) | 40.0 | 313.15 | 1.61 | 1.09 | 2.10 | 2.335 (8.71%) | 0.148/0.0521 (1.13%) | 0.867 (4.29%) | 2.337 | 2.590 |
| A5 (FFD) | 40.0 | 313.15 | 4.64 | 3.15 | 2.10 | 6.624 (16.3%) | 0.424/0.181 (4.13%) | 1.65 (5.11%) | 2.337 | 6.250 |
| A10 (CCD) | 40.0 | 323.15 | 3.11 | 2.19 | 2.30 | 6.654 (12.7%) | 0.821/3.126 (1.80%) | 3.98 (7.99%) | 2.639 | 5.156 |
| A17 (CCD) | 40.0 | 353.15 | 3.13 | 2.47 | 2.30 | 7.445 (9.22%) | 1.412/1.361 (3.19%) | 7.74 (5.47%) | 3.683 | 7.599 |
| A8 (FFD) | 40.0 | 353.15 | 4.59 | 3.62 | 2.90 | 10.12 (10.0%) | 1.677/0.217 (2.21%) | 7.00 (4.37%) | 3.683 | 1.044 |
| A2 (FFD) | 40.0 | 353.15 | 1.60 | 1.26 | 2.90 | 3.445 (9.91%) | 0.310/0.0691 (1.48%) | 1.97 (4.98%) | 3.683 | 4.346 |
| A12(CCD) | 42.0 | 338.15 | 3.06 | 2.25 | 2.60 | 7.110 (12.6%) | 1.063/2.357 (2.52%) | 8.51 (6.02%) | 3.057 | 5.987 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Coelho, J.P.; Robalo, M.P.; Fernandes, I.S.; Stateva, R.P. Optimization of Oil Recovery from Japonica Luna Rice Bran by Supercritical Carbon Dioxide Applying Design of Experiments: Characterization of the Oil and Mass Transfer Modeling. ChemEngineering 2022, 6, 63. https://doi.org/10.3390/chemengineering6040063
Coelho JP, Robalo MP, Fernandes IS, Stateva RP. Optimization of Oil Recovery from Japonica Luna Rice Bran by Supercritical Carbon Dioxide Applying Design of Experiments: Characterization of the Oil and Mass Transfer Modeling. ChemEngineering. 2022; 6(4):63. https://doi.org/10.3390/chemengineering6040063
Chicago/Turabian StyleCoelho, José P., Maria Paula Robalo, Inês S. Fernandes, and Roumiana P. Stateva. 2022. "Optimization of Oil Recovery from Japonica Luna Rice Bran by Supercritical Carbon Dioxide Applying Design of Experiments: Characterization of the Oil and Mass Transfer Modeling" ChemEngineering 6, no. 4: 63. https://doi.org/10.3390/chemengineering6040063
APA StyleCoelho, J. P., Robalo, M. P., Fernandes, I. S., & Stateva, R. P. (2022). Optimization of Oil Recovery from Japonica Luna Rice Bran by Supercritical Carbon Dioxide Applying Design of Experiments: Characterization of the Oil and Mass Transfer Modeling. ChemEngineering, 6(4), 63. https://doi.org/10.3390/chemengineering6040063








