Multisensory Gas Chromatography for Field Analysis of Complex Gaseous Mixtures
Abstract
1. Introduction
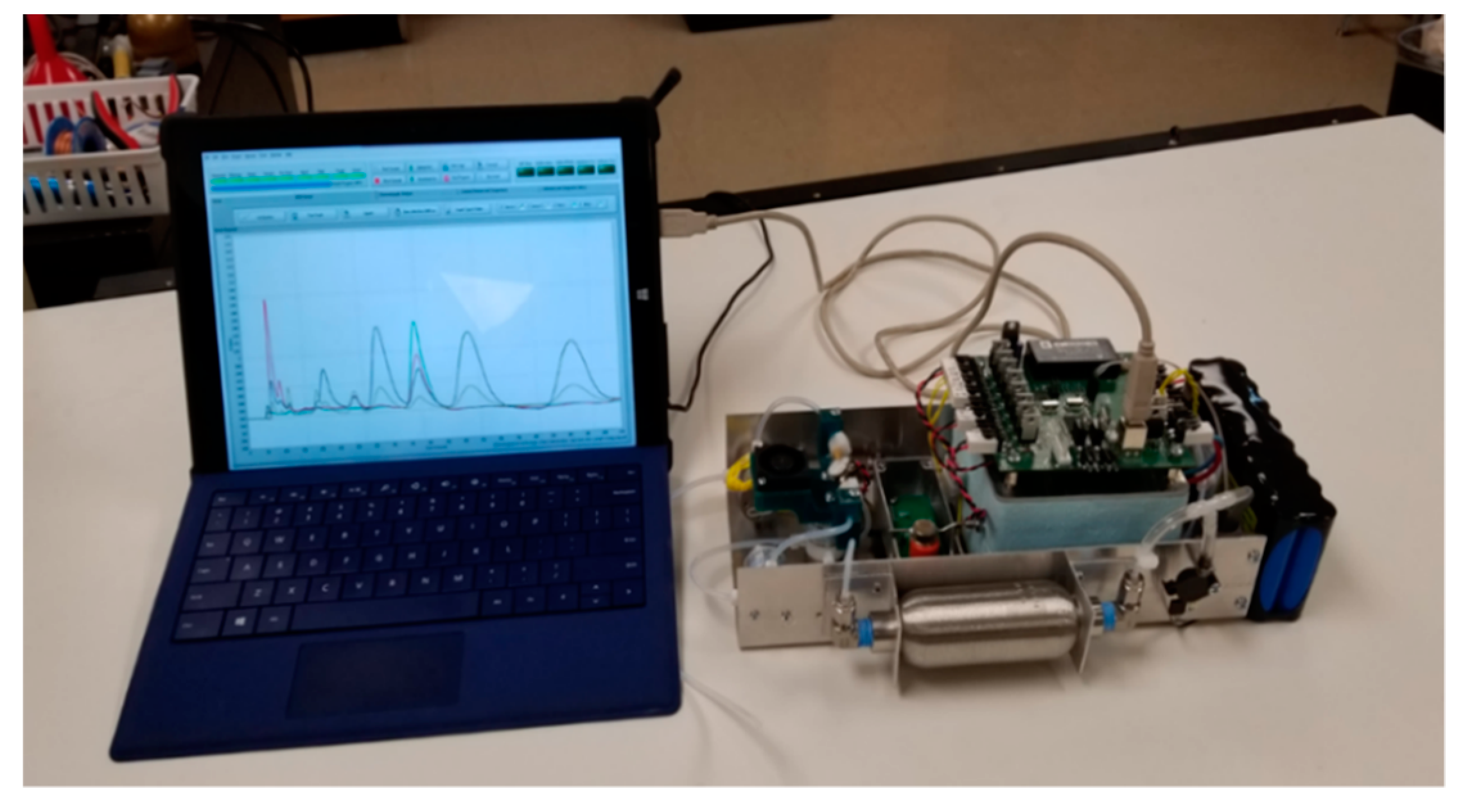
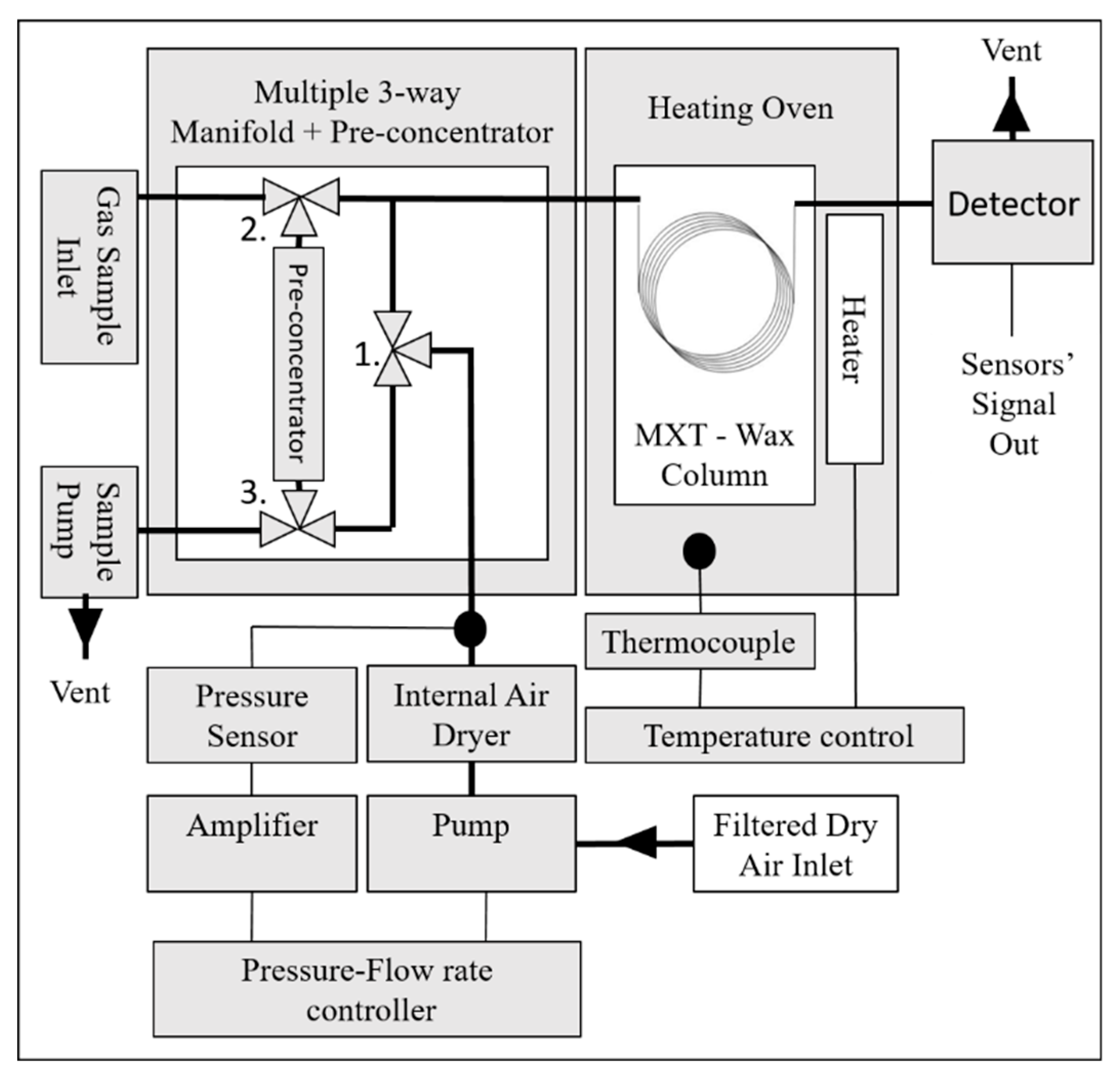
2. Experiment
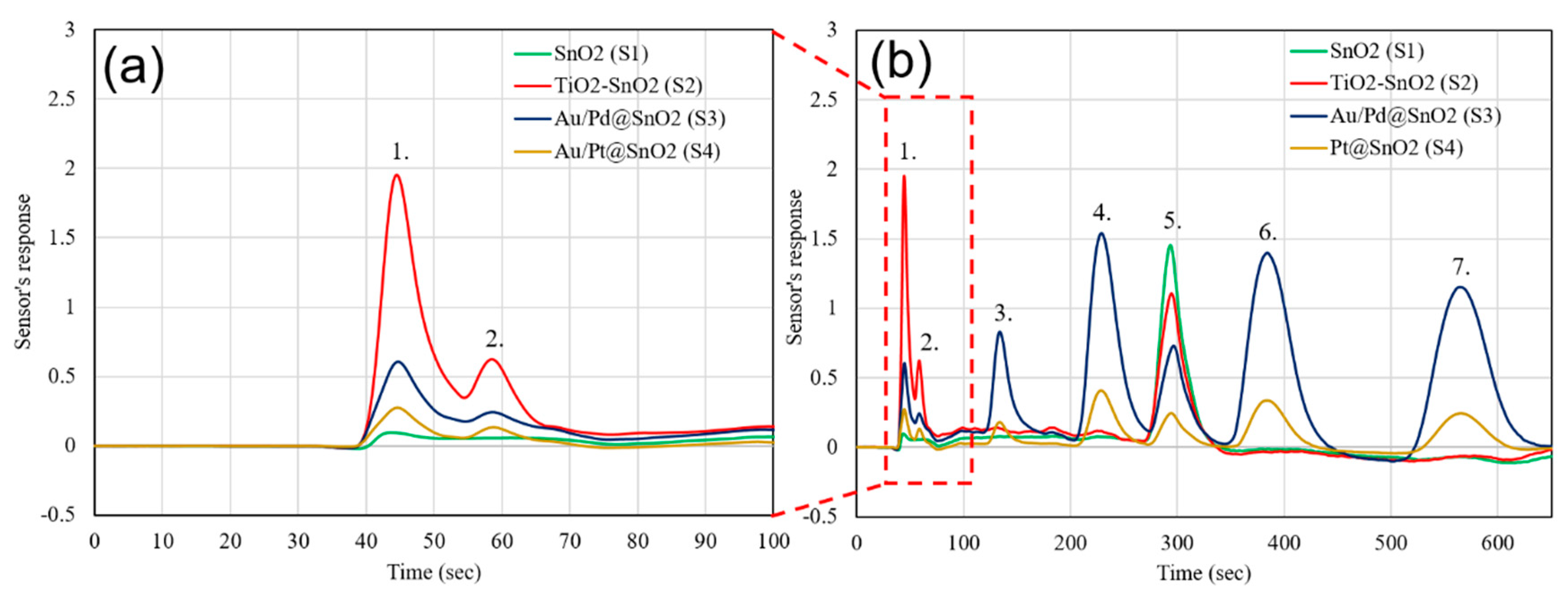
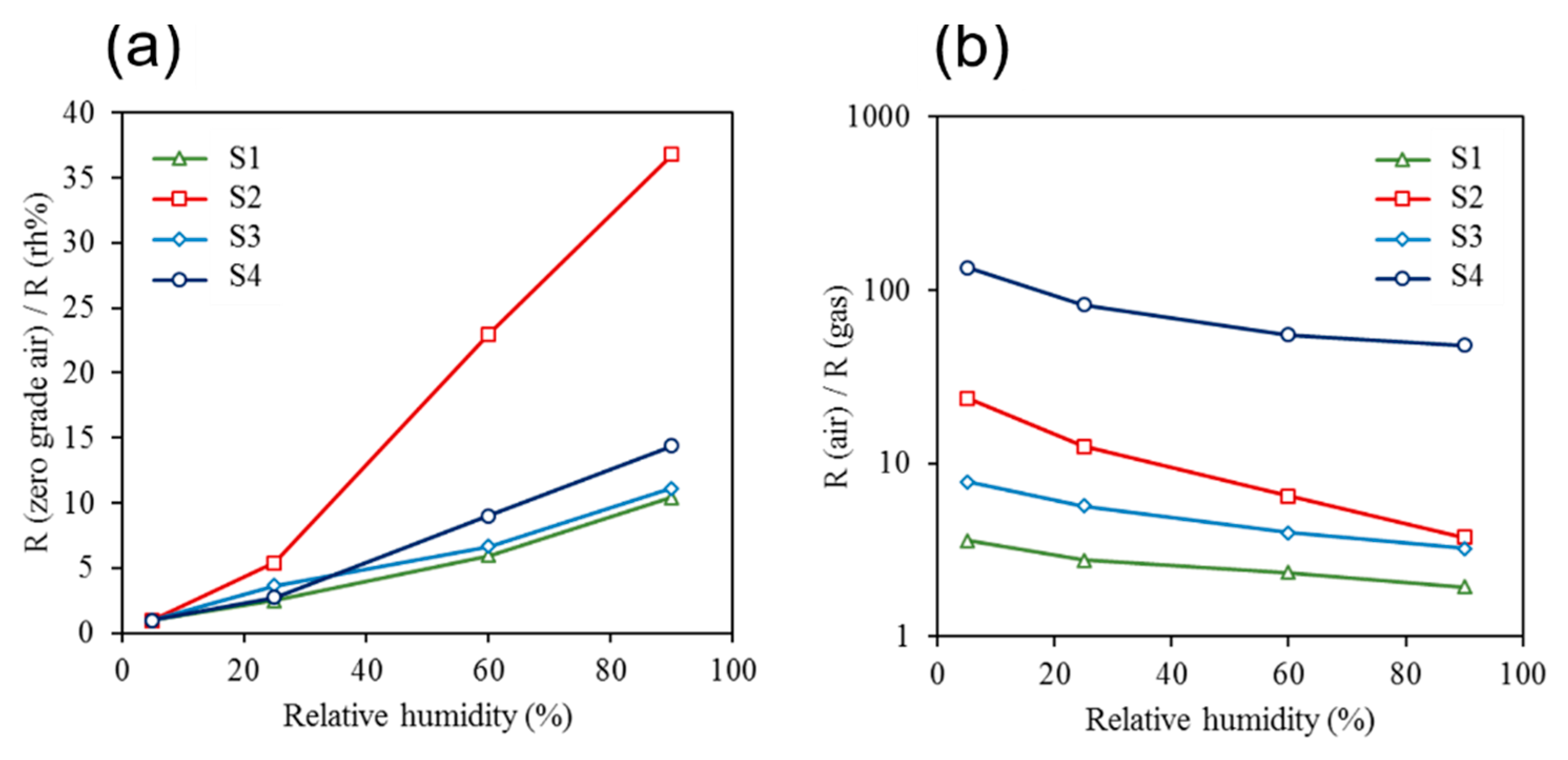
3. Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Mullen, P.W. Modern Gas Analysis. Interscience Manuals, Volume 6; Interscience Publishers: New York, NY, USA, 1955. [Google Scholar]
- Mandayo, G.G.; Gonzalez-Chavarri, J.; Hammes, E.; Newton, H.; Castro-Hurtado, I.; Ayerdi, I.; Knapp, H.; Sweetman, A.; Hewitt, C.N.; Castaño, E. System to control indoor air quality in energy efficient buildings. Urban Clim. 2015, 14, 475–485. [Google Scholar] [CrossRef]
- The National Institute for Occupational Safety and Health (NIOSH); United States Public Health Service, Division of Occupational. The Industrial Environment—Its Evaluation & Control, 3rd ed.; for sale by the Supt. of Docs.; U.S. Govt. Print. Off.: Washington, DC, USA, 1973.
- Bielecki, Z.; Janucki, J.; Kawalec, A.; Mikołajczyk, J.; Pałka, N.; Pasternak, M.; Pustelny, T.; Stacewicz, T.; Wojtas, J. Sensors and Systems for the Detection of Explosive Devices—An Overview. Metrol. Meas. Syst. 2012, 19, 3–28. [Google Scholar] [CrossRef]
- Miekisch, W.; Schubert, J.K.; Noeldge-Schomburg, G.F.E. Diagnostic potential of breath analysis—Focus on volatile organic compounds. Clin. Chim. Acta 2004, 347, 25–39. [Google Scholar] [CrossRef]
- Megson, D.; Reiner, E.J.; Jobst, K.J.; Dorman, F.L.; Robson, M.; Focant, J.F. A review of the determination of persistent organic pollutants for environmental forensics investigations. Anal. Chim. Acta 2016, 941, 10–25. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.S.; Milton, M.J.T.; Cowper, C.J.; Squire, G.D.; Bremser, W.; Branch, R.W. Analysis of natural gas by gas chromatography: Reduction of correlated uncertainties by normalisation. J. Chromatogr. A 2004, 1040, 215–225. [Google Scholar] [CrossRef] [PubMed]
- Patnaik, P. Handbook of Environmental Analysis: Chemical Pollutants in Air, Water, Soil, and Solid Wastes, 1st ed.; CRC Press: Boca Raton, FL, USA, 1997; pp. 285–435. [Google Scholar]
- Brattoli, M.; Cisternino, E.; Dambruoso, P.; De Gennaro, G.; Giungato, P.; Mazzone, A.; Palmisani, J.; Tutino, M. Gas Chromatography Analysis with Olfactometric Detection (GC-O) as a Useful Methodology for Chemical Characterization of Odorous Compounds. Sensors 2013, 13, 16759–16800. [Google Scholar] [CrossRef]
- De Blas, M.; Navazo, M.; Alonso, L.; Durana, N.; Iza, J. Automatic on-line monitoring of atmospheric volatile organic compounds: Gas chromatography-mass spectrometry and gas chromatography-flame ionization detection as complementary systems. Sci. Total Environ. 2011, 409, 5459–5469. [Google Scholar] [CrossRef] [PubMed]
- Cui, D.F.; Chen, X.; Zhang, L.L.; Cai, H.Y.; Li, H. A micro gas chromatography column with a micro thermal conductivity detector for volatile organic compound analysis. Rev. Sci. Instrum. 2013, 84, 025001. [Google Scholar] [CrossRef]
- Skov, H.; Lindskog, A.; Palmgren, F.; Christensen, C.S. An overview of commonly used methods for measuring benzene in ambient air. Atmos. Environ. 2001, 35, 141–148. [Google Scholar] [CrossRef]
- Liaud, C.; Nguyen, N.T.; Nasreddine, R.; Le Calvé, S. Experimental performances study of a transportable GC-PID and two thermo-desorption based methods coupled to FID and MS detection to assess BTEX exposure at sub-ppb level in air. Talanta 2014, 127, 33–42. [Google Scholar] [CrossRef]
- Gallego, E.; Roca, F.J.; Perales, J.F.; Sánchez, G.; Esplugas, P. Characterization and determination of the odorous charge in the indoor air of a waste treatment facility through the evaluation of volatile organic compounds (VOCs) using TD–GC/MS. Waste Manag. 2012, 32, 2469–2481. [Google Scholar] [CrossRef]
- De Blas, M.; Navazo, M.; Alonso, L.; Durana, N.; Gomez, M.; Iza, J. Simultaneous indoor and outdoor on-line hourly monitoring of atmospheric volatile organic compounds in an urban building. The role of inside and outside sources. Sci. Total Environ. 2012, 426, 327–335. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, S.; Wischhusen, S.; Müller, J. Micro flame ionization detector and micro flame spectrometer. Sens. Actuators B Chem. 2000, 63, 159–166. [Google Scholar] [CrossRef]
- Nasreddine, R.; Person, V.; Serra, C.A.; Le Calve, S. Development of a novel portable miniaturized GC for near real-time low level detection of BTEX. Sens. Actuators B Chem. 2016, 224, 159–169. [Google Scholar] [CrossRef]
- Garg, A.; Akbar, M.; Vejerano, E.; Narayanan, S.; Nazhandali, L.; Marrb, L.C.; Agaha, M. Zebra GC: A mini gas chromatography system for trace-level determination of hazardous air pollutants. Sens. Actuators B Chem. 2015, 212, 145–154. [Google Scholar] [CrossRef]
- Alinoori, A.; Masoum, S. Multicapillary Gas Chromatography-Temperature Modulated Metal Oxide Semiconductor Sensors Array Detector for Monitoring of Volatile Organic Compounds in Closed Atmosphere Using Gaussian Apodization Factor Analysis. Anal. Chem. 2018, 90, 6635–6642. [Google Scholar] [CrossRef] [PubMed]
- Dual-Chemiresistor, G.C. Detector Employing Monolayer-Protected Metal Nanocluster Interfaces Cai, Qing-Yun; Zellers, Edward T. Anal. Chem. 2002, 74, 3533–3539. [Google Scholar]
- Dobrokhotov, V.; Larin, A. Doped, Metal Oxide-Based Chemical Sensors. U.S. Patent 10,132,769, 20 November 2018. [Google Scholar]
- Larin, A.; Womble, P.C.; Dobrokhotov, V. Novel highly-integrated MEMS based solid state detectors for analytical gas chromatography. Sens. Actuators B (Chem.) 2018, 256, 1057–1068. [Google Scholar] [CrossRef]
- Larin, A.; Womble, P.C.; Dobrokhotov, V. Hybrid SnO2/TiO2 Nanocomposites for Selective Detection of Ultra-Low Hydrogen Sulfide Concentrations in Complex Backgrounds. Sensors 2016, 16, 1373. [Google Scholar] [CrossRef]
- Stranger, M.; Potgieter-Vermaak, S.; van Grieken, R. Characterization of indoor air quality in primary schools in Antwerp, Belgium. Indoor Air 2008, 18, 454–463. [Google Scholar] [CrossRef]
- Hulin, M.; Caillaud, D.; Annesi-Maesano, I. Indoor air pollution and childhood asthma: Variations between urban and rural areas. Indoor Air 2010, 20, 502–514. [Google Scholar] [CrossRef] [PubMed]
- Darlington, A.B.; Dat, J.F.; Dixon, M.A. The biofiltration of indoor air: Air flux and temperature influences the removal of toluene, ethylbenzene, and xylene. Environ. Sci. Technol. 2001, 35, 240–246. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Lee, S.C.; Li, W.M.; Cao, J.J. Source characterization of BTEX in indoor microenvironments in Hong Kong. Atmos. Environ. 2003, 37, 73–82. [Google Scholar] [CrossRef]
- Reynolds, S.J.; Donham, K.J.; Stookesberry, J.; Thorne, P.S.; Subramanian, P.; Thu, K.; Whitten, P. Air quality assessments in the vicinity of swine production facilities. J. Agromed. 1997, 4, 37–45. [Google Scholar] [CrossRef]
- Godish, T.; Davis, W.T.; Fu, J.S. Air Quality; CRC Press: Boca Raton, FL, USA, 2014. [Google Scholar]
- Kidnay, A.J.; Parrish, W.R.; McCartney, D.G. Fundamentals of Natural Gas Processing; CRC Press: Boca Raton, FL, USA, 2011; Volume 218. [Google Scholar]
- Al-Juaied, M.; Koros, W. Performance of natural gas membranes in the presence of heavy hydrocarbons. J. Membr. Sci. 2006, 274, 227–243. [Google Scholar] [CrossRef]
- Papadias, D.D.; Ahmed, S.; Kumar, R. Fuel quality issues with biogas energy–An economic analysis for a stationary fuel cell system. Energy 2012, 44, 257–277. [Google Scholar] [CrossRef]
- Hill, D.; Binions, R. Breath Analysis for Medical Diagnosis. Int. J. Smart Sens. Intell. Syst. 2012, 5. [Google Scholar] [CrossRef]
- Manolis, A. The diagnostic potential of breath analysis. Clin. Chem. 1983, 29, 5–15. [Google Scholar]
- Dweik, R.A.; Amann, A. Exhaled breath analysis: The new frontier in medical testing. J. Breath Res. 2008, 2, 030301. [Google Scholar] [CrossRef]
- Peng, G.; Tisch, U.; Adams, O.; Hakim, M.; Shehada, N.; Broza, Y.Y.; Billan, S.; Abdah-Bortnyak, R.; Kuten, A.; Haick, H. Diagnosing lung cancer in exhaled breath using gold nanoparticles. Nat. Nanotechnol. 2009, 4, 669–673. [Google Scholar] [CrossRef]
- Phillips, M.; Basa-Dalay, V.; Bothamley, G.; Cataneo, R.N.; Lam, P.K.; Natividad, M.P.R.; Schmitt, P.; Wai, J. Breath biomarkers of active pulmonary tuberculosis. Tuberculosis 2010, 90, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Amorim, L.C.A.; Cardeal, Z.D.L. Breath air analysis and its use as a biomarker in biological monitoring of occupational and environmental exposure to chemical agents. J. Chromatogr. B 2007, 853, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Siedl, N.; Baumann, S.O.; Elser, M.J.; Diwald, O. Particle networks from powder mixtures: Generation of TiO2–SnO2 heterojunctions via surface charge-induced heteroaggregation. J. Phys. Chem. C 2012, 116, 22967–22973. [Google Scholar] [CrossRef] [PubMed]
- Varela, J.A.; Perazolli, L.A.; Cerri, J.A.; Leite, E.R.; Longo, E. Sintering of tin oxide and its applications in electronics and processing of high purity optical glasses. Cerâmica 2001, 47, 117–123. [Google Scholar] [CrossRef]
- De Mendonça, V.R.; Dalmaschio, C.J.; Leite, E.R.; Niederberger, M.; Ribeiro, C. Heterostructure formation from hydrothermal annealing of preformed nanocrystals. J. Mater. Chem. A 2015, 3, 2216–2225. [Google Scholar] [CrossRef]
- Scanlon, D.O.; Dunnill, C.W.; Buckeridge, J.; Shevlin, S.A.; Logsdail, A.J.; Woodley, S.M.; Catlow, C.R.A.; Powell, M.J.; Palgrave, R.G.; Parkin, I.P.; et al. Band alignment of rutile and anatase TiO2. Nat. Mater. 2013, 12, 798–801. [Google Scholar] [CrossRef] [PubMed]
- Dang, V.S.; Parala, H.; Kim, J.H.; Xu, K.; Srinivasan, N.B.; Edengeiser, E.; Havenith, M.; Wieck, A.D.; de los Arcos, T.; Fischer, R.A.; et al. Electrical and optical properties of TiO2 thin films prepared by plasma-enhanced atomic layer deposition. Phys. Status Solidi (a) 2014, 211, 416–424. [Google Scholar] [CrossRef]
- Bedja, I.; Kamat, P.V. Capped semiconductor colloids. Synthesis and photoelectrochemical behavior of TiO2 capped SnO2 nanocrystallites. J. Phys. Chem. 1995, 99, 9182–9188. [Google Scholar] [CrossRef]
- Liu, G.; Jaegermann, W.; He, J.; Sundström, V.; Sun, L. XPS and UPS characterization of the TiO2/ZnPcGly heterointerface: Alignment of energy levels. J. Phys. Chem. B 2002, 106, 5814–5819. [Google Scholar] [CrossRef]
- Kwoka, M.; Ottaviano, L.; Szuber, J. Photoemission studies of the surface electronic properties of L-CVD SnO2 ultra thin films. Appl. Surf. Sci. 2012, 258, 8425–8429. [Google Scholar] [CrossRef]
- Imanishi, A.; Tsuji, E.; Nakato, Y. Dependence of the work function of TiO2 (Rutile) on crystal faces, studied by a scanning auger microprobe. J. Phys. Chem. C 2007, 111, 2128–2132. [Google Scholar] [CrossRef]
- Xiong, G.; Shao, R.; Droubay, T.C.; Joly, A.G.; Beck, K.M.; Chambers, S.A.; Hess, W.P. Photoemission electron microscopy of TiO2 anatase films embedded with rutile nanocrystals. Adv. Funct. Mater. 2007, 17, 2133–2138. [Google Scholar] [CrossRef]
- Li, H.; Xie, C.; Liao, Y.; Liu, Y.; Zou, Z.; Wu, J. Characterization of Incidental Photon-to-electron Conversion Efficiency (IPCE) of porous TiO2/SnO2 composite film. J. Alloy. Compd. 2013, 569, 88–94. [Google Scholar] [CrossRef]
- Cahen, D.; Hodes, G.; Graetzel, M.; Guillemoles, J.F.; Riess, I. Nature of photovoltaic action in dye-sensitized solar cells. J. Phys. Chem. B 2000, 104, 2053–2059. [Google Scholar] [CrossRef]
- Trakhtenberg, L.; Gerasimov, G.N.; Gromov, V.F.; Belysheva, T.V.; Ilegbusi, O.J. Gas semiconducting sensors based on metal oxide nanocomposites. J. Mater. Sci. Res. 2012, 1, 56. [Google Scholar] [CrossRef]
- Suchorska-Woźniak, P.; Rac, O.; Fiedot, M.; Teterycz, H. Analysis of SnO2 | WO3 heterocontact properties during the detection of hydrogen sulphide. Sensors 2014, 14, 20480–20499. [Google Scholar] [CrossRef] [PubMed]
- Mei, L.; Chen, Y.; Ma, J. Gas sensing of SnO2 nanocrystals revisited: Developing ultra-sensitive sensors for detecting the H2S leakage of biogas. Sci. Rep. 2014, 4, 6028. [Google Scholar] [CrossRef] [PubMed]
- Chun, S.W.; Jang, J.Y.; Park, D.W.; Woo, H.C.; Chung, J.S. Selective oxidation of H2S to elemental sulfur over TiO2/SiO2 catalysts. Appl. Catal. B Environ. 1998, 16, 235–243. [Google Scholar] [CrossRef]
- Korotcenkov, G.; Brinzari, V.; Gulina, L.B.; Cho, B.K. The influence of gold nanoparticles on the conductivity response of SnO2-based thin film gas sensors. Appl. Surf. Sci. 2015, 353, 793–803. [Google Scholar] [CrossRef]
- Chlala, D.; Labaki, M.; Giraudon, J.M.; Gardoll, O.; Denicourt-Nowicki, A.; Roucoux, A.; Lamonier, J.F. Toluene total oxidation over Pd and Au nanoparticles supported on hydroxyapatite. C. R. Chim. 2016, 19, 525–537. [Google Scholar] [CrossRef]
- Tabakova, T.; Ilieva, L.; Petrova, P.; Venezia, A.M.; Avdeev, G.; Zanella, R.; Karakirova, Y. Complete benzene oxidation over mono and bimetallic Au–Pd catalysts supported on Fe-modified ceria. Chem. Eng. J. 2015, 260, 133–141. [Google Scholar] [CrossRef]
- Silva, T.A.; Teixeira-Neto, E.; López, N.; Rossi, L.M. Volcano-like behavior of Au-Pd core-shell nanoparticles in the selective oxidation of alcohols. Sci. Rep. 2014, 4, 5766. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Villa, A.; Porta, F.; Prati, L.; Su, D. Bimetallic gold/palladium catalysts: Correlation between nanostructure and synergistic effects. J. Phys. Chem. C 2008, 112, 8617–8622. [Google Scholar] [CrossRef]

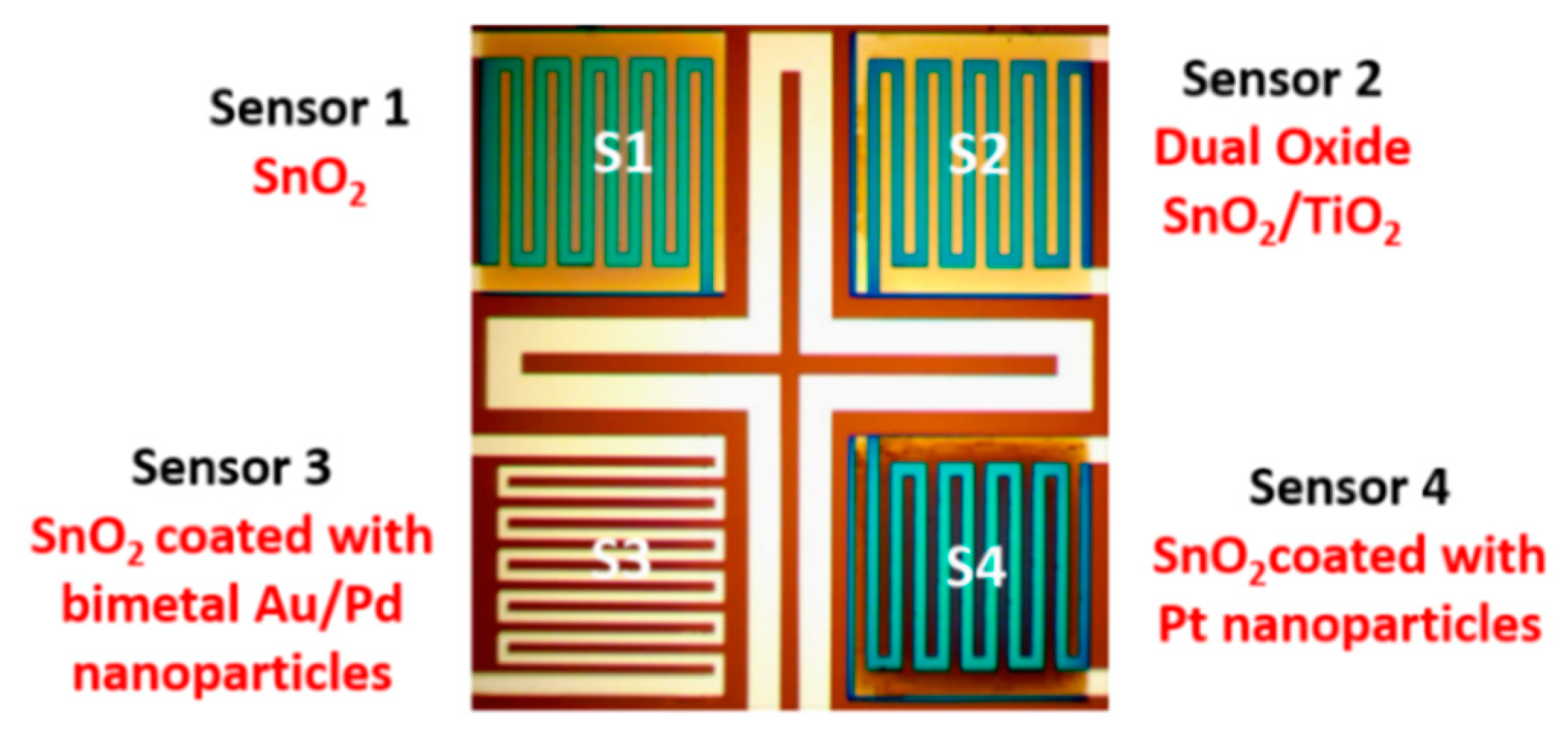
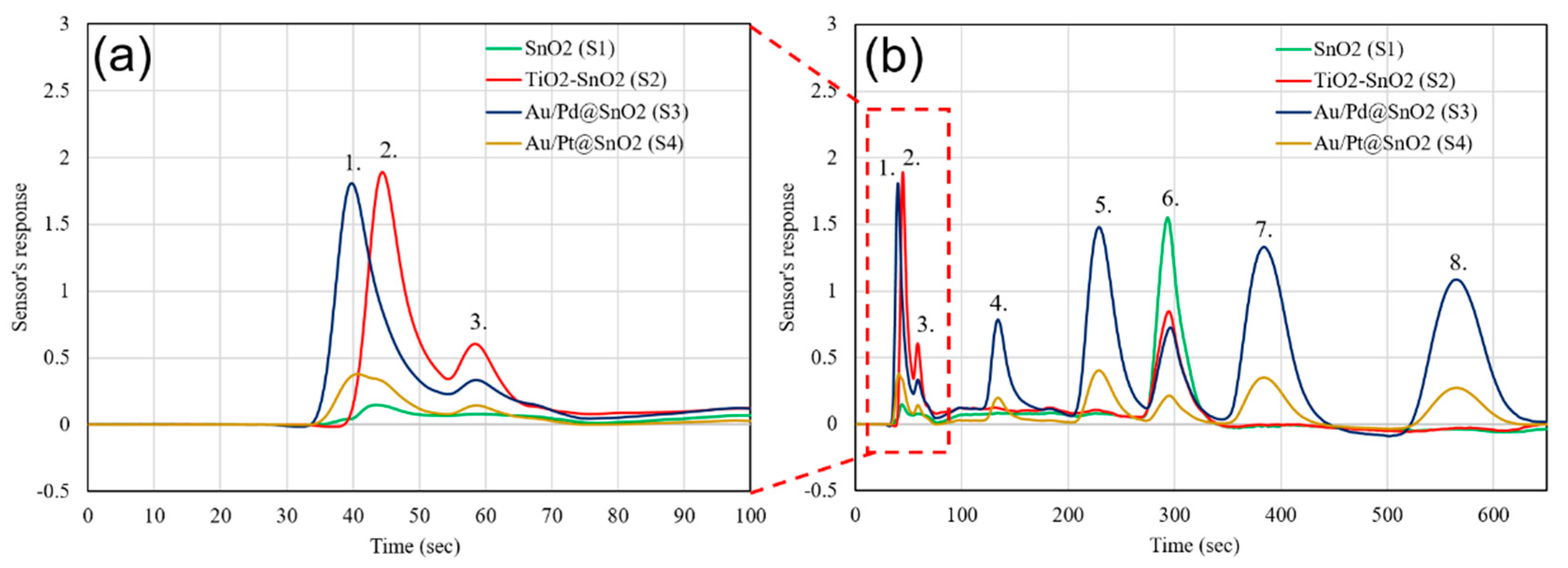
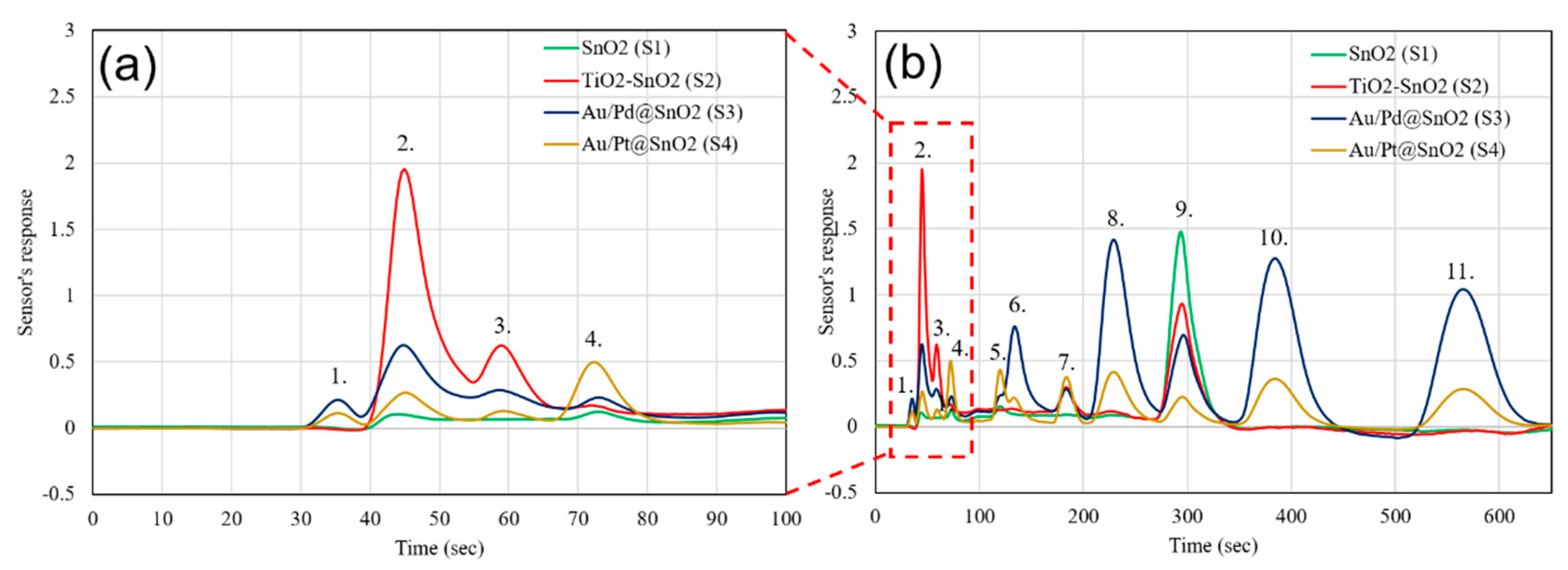
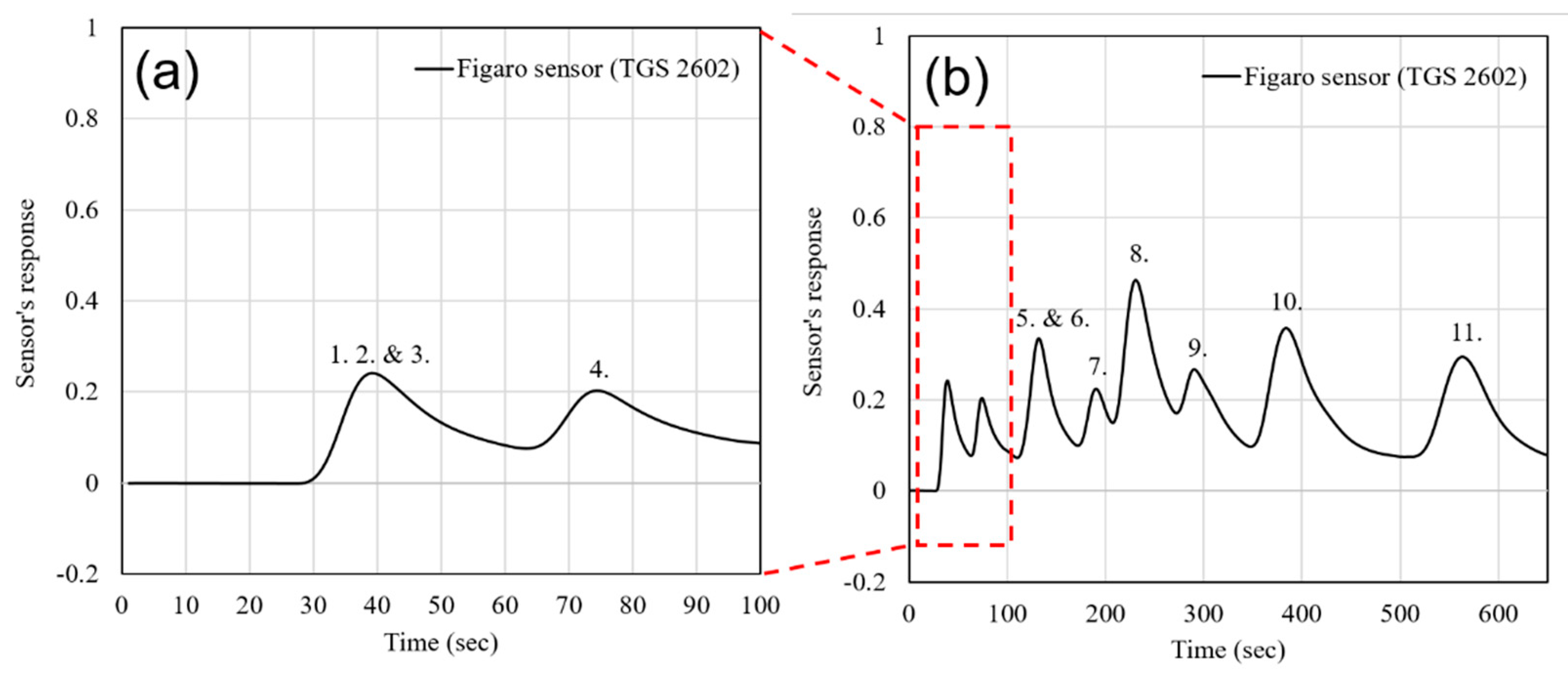
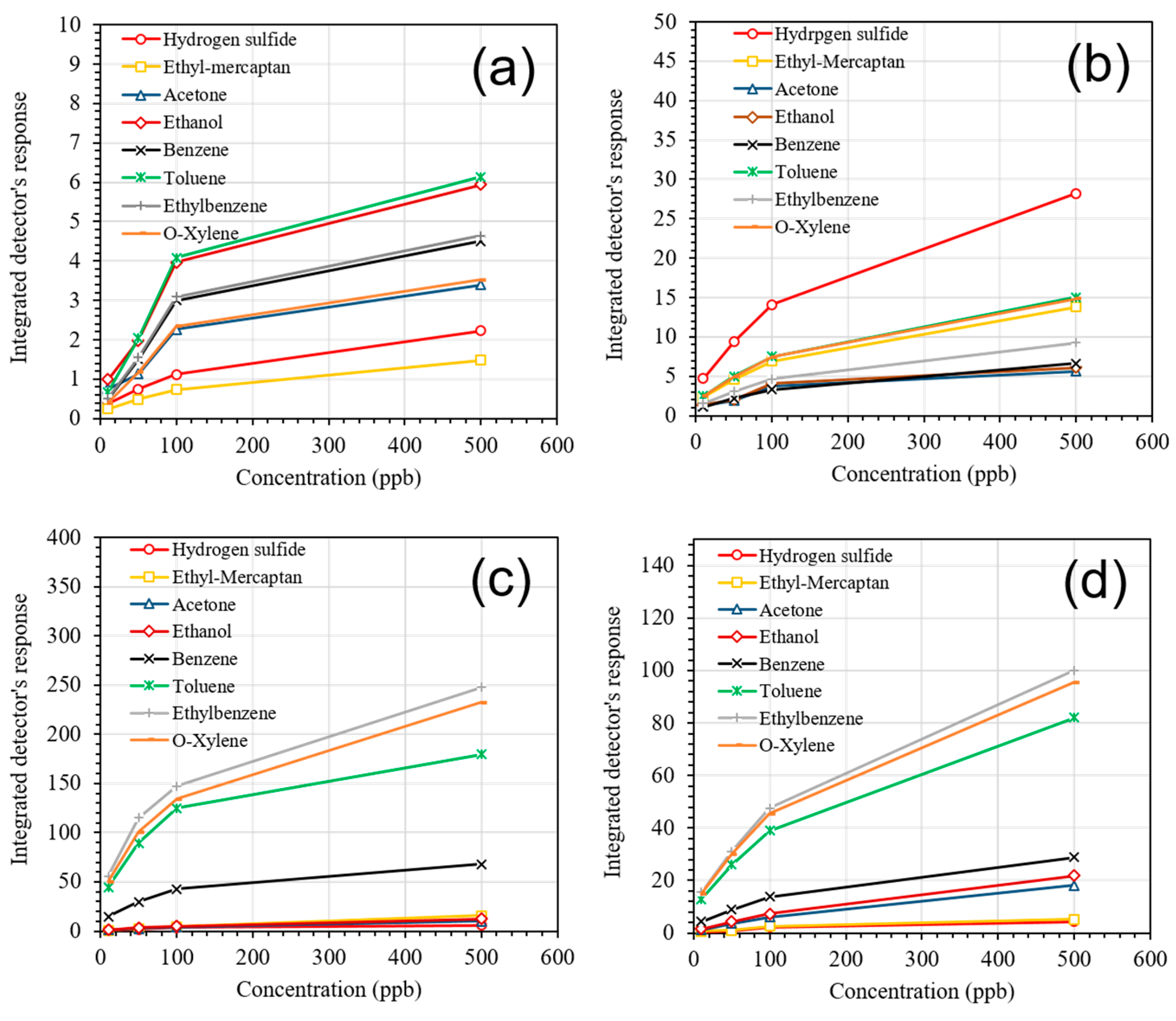
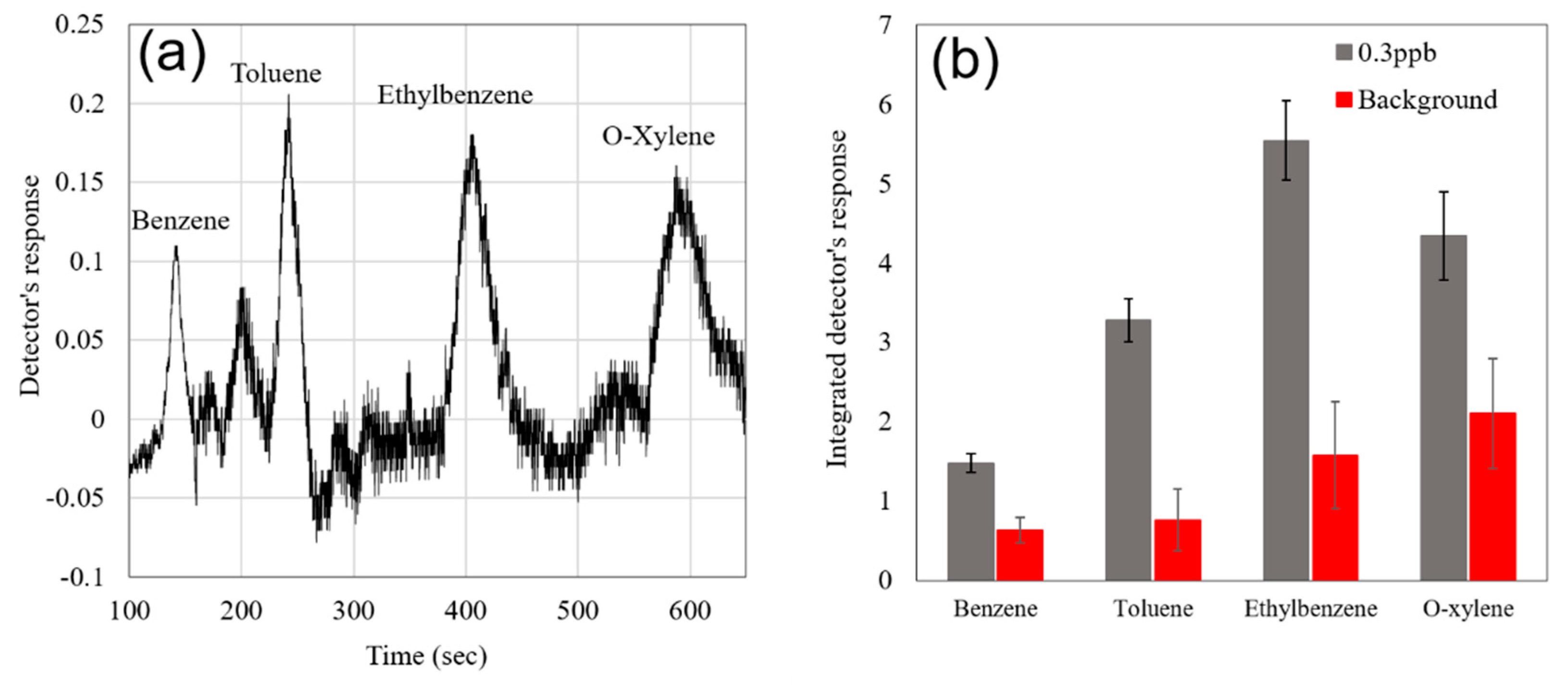
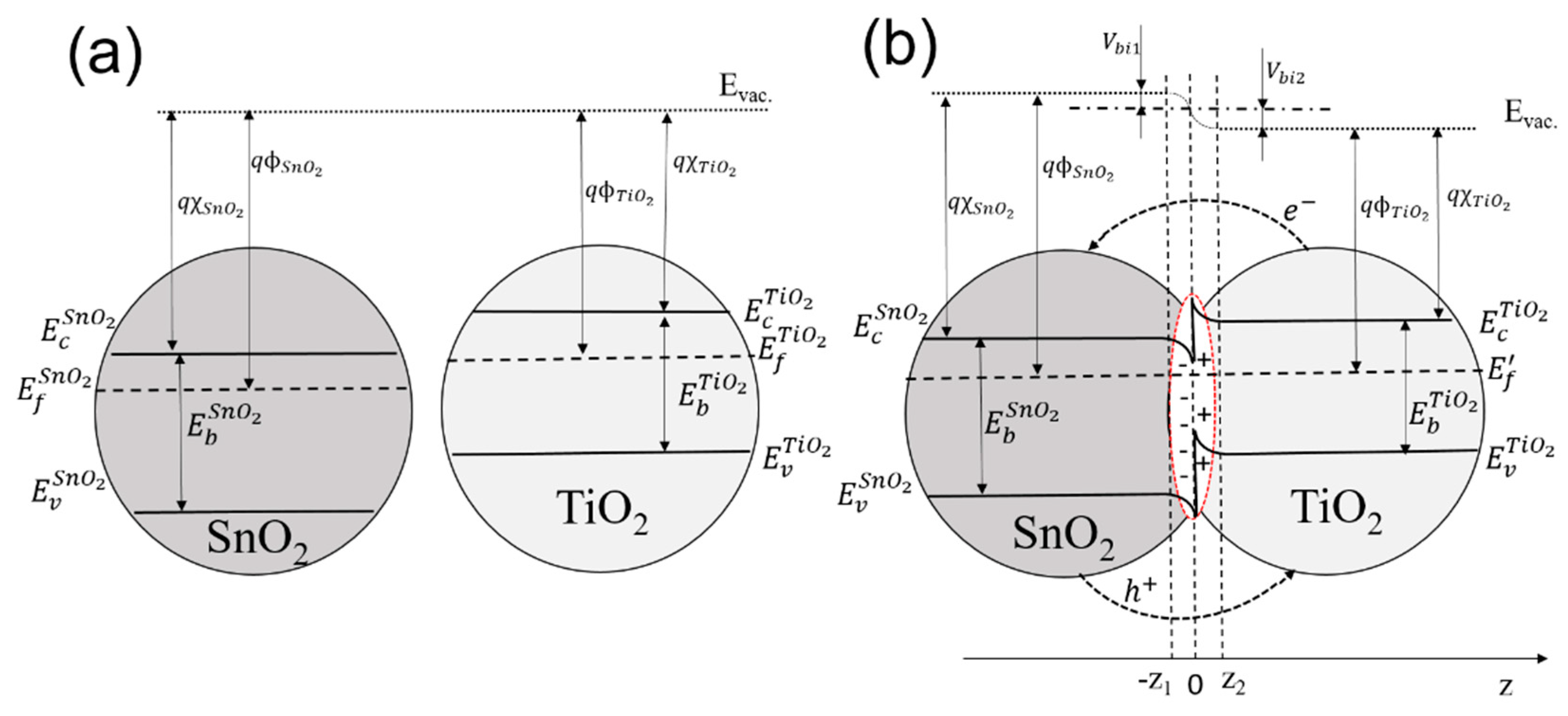
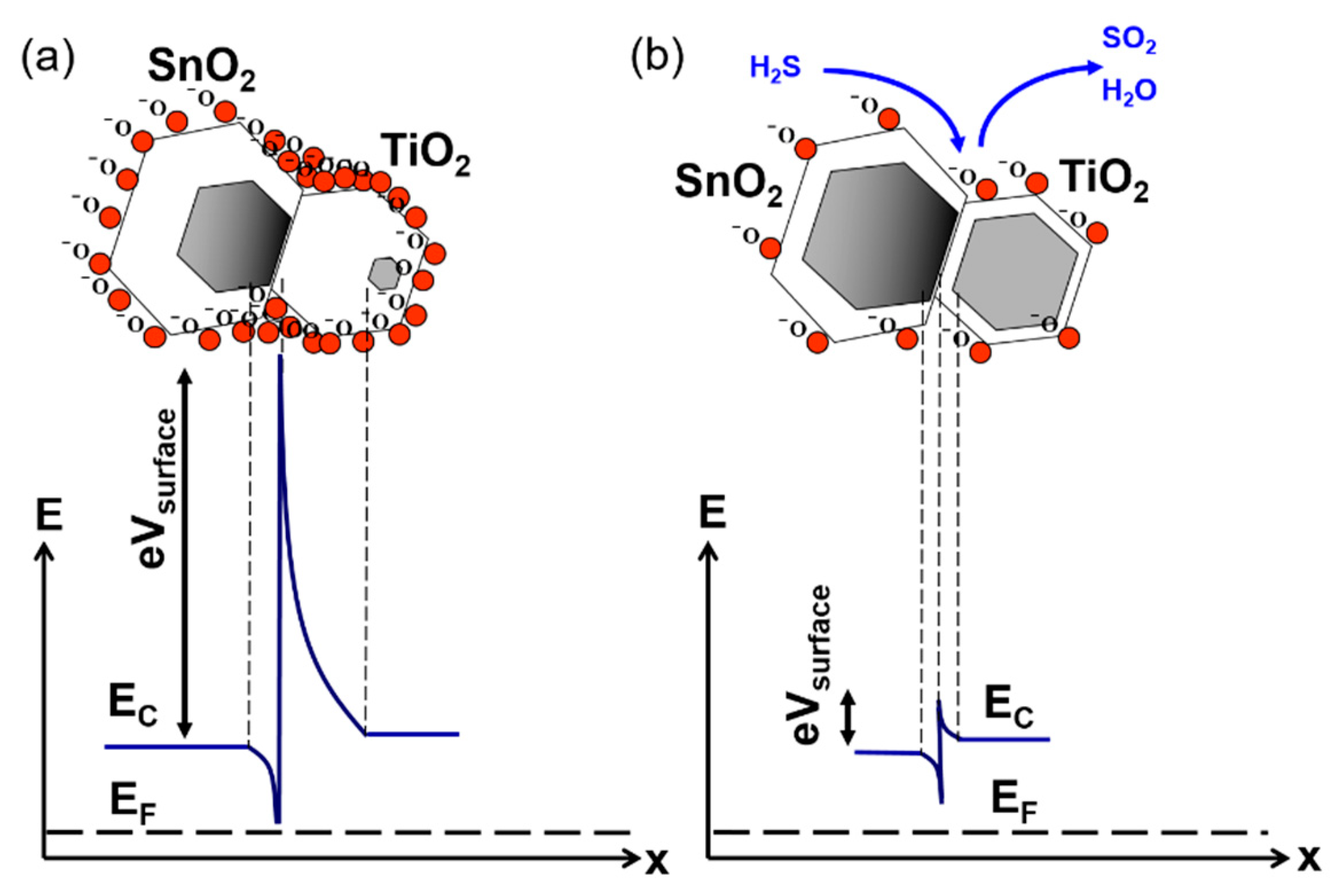
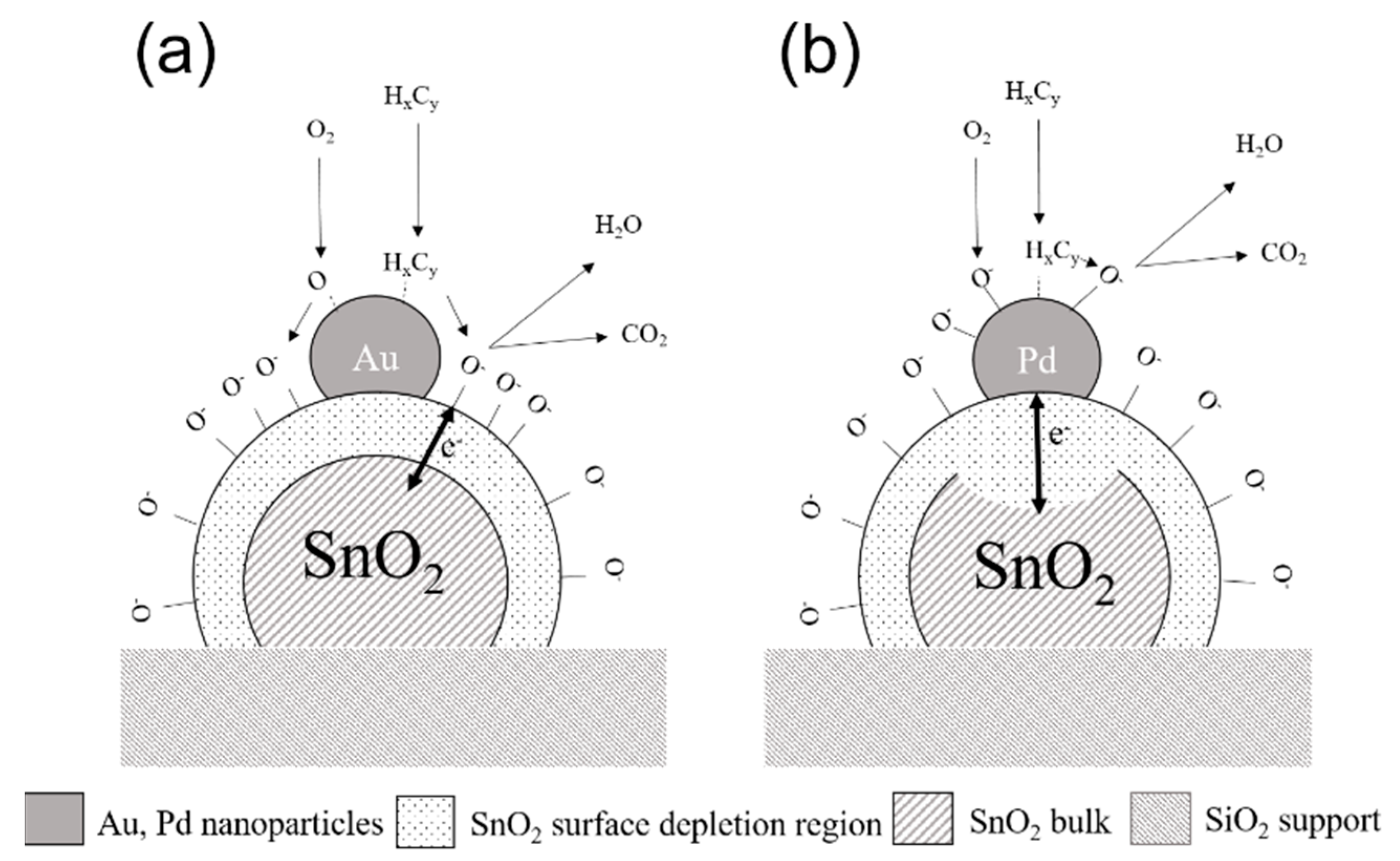
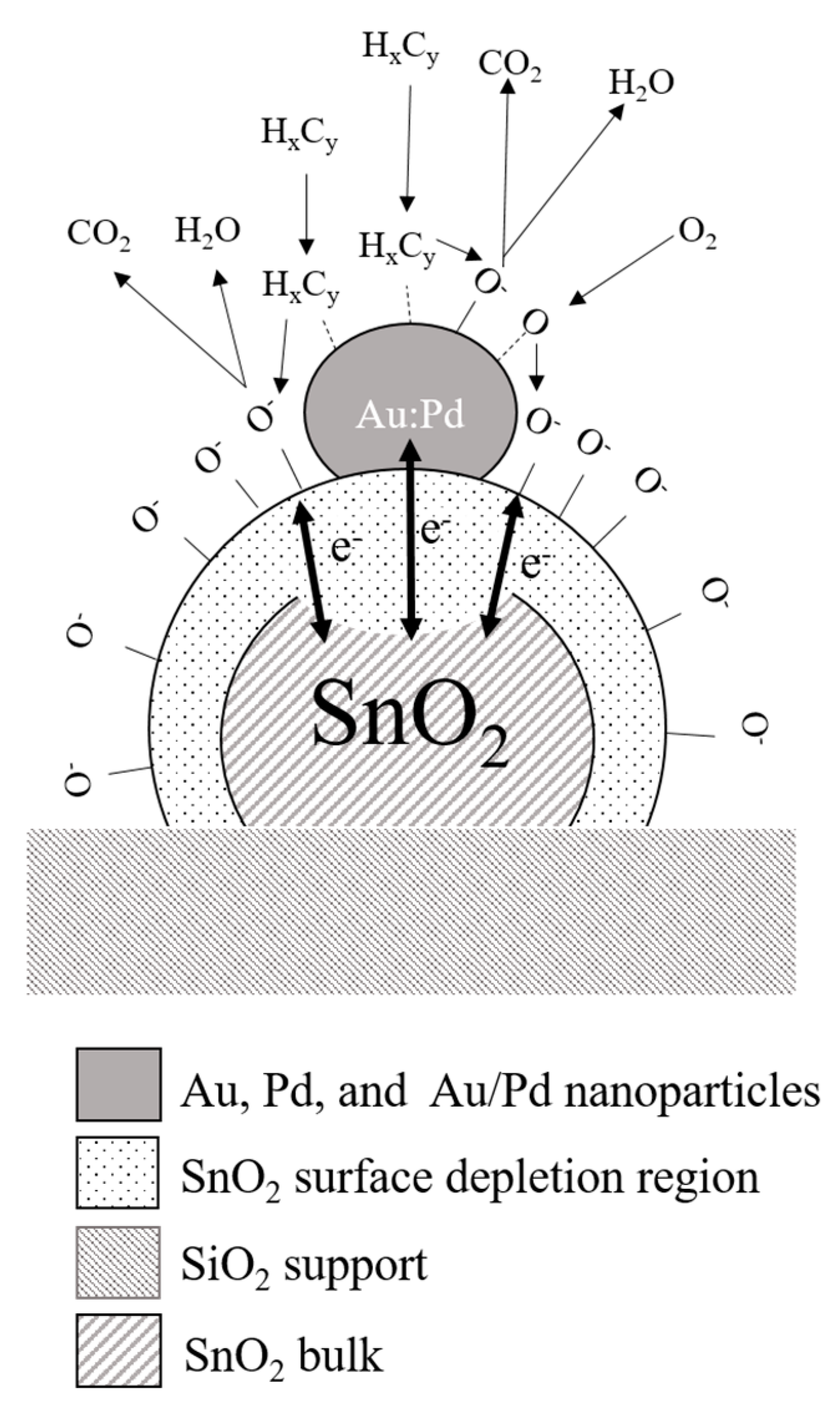
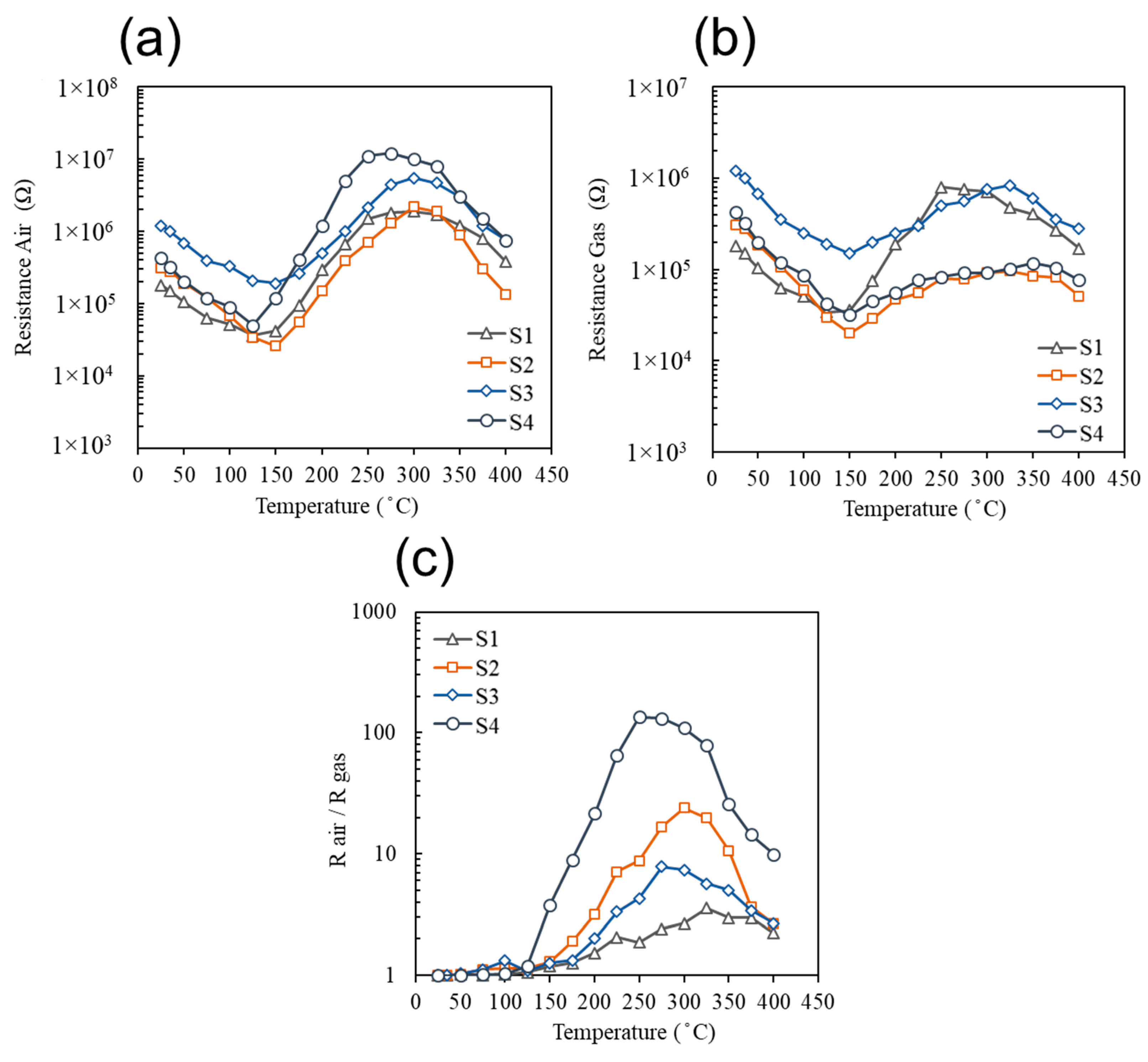
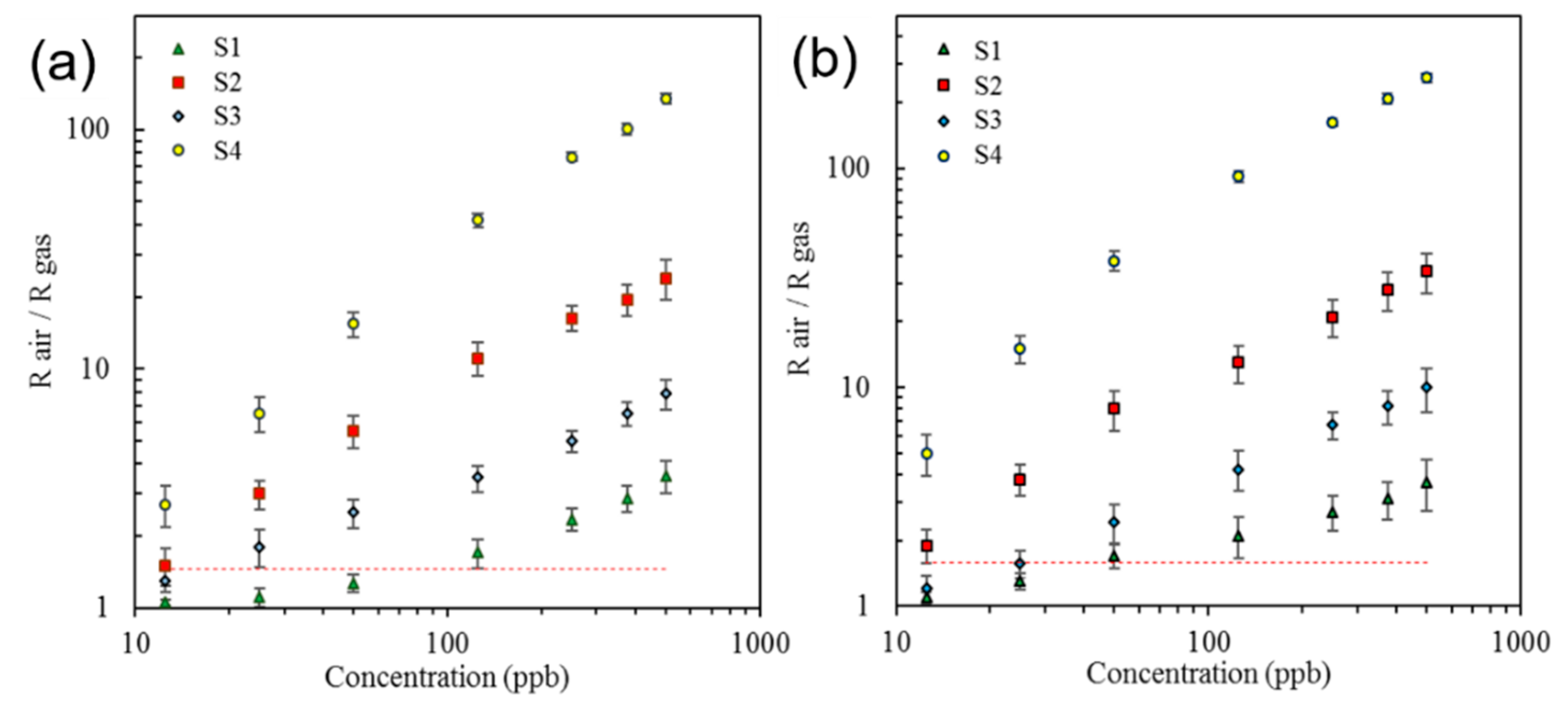
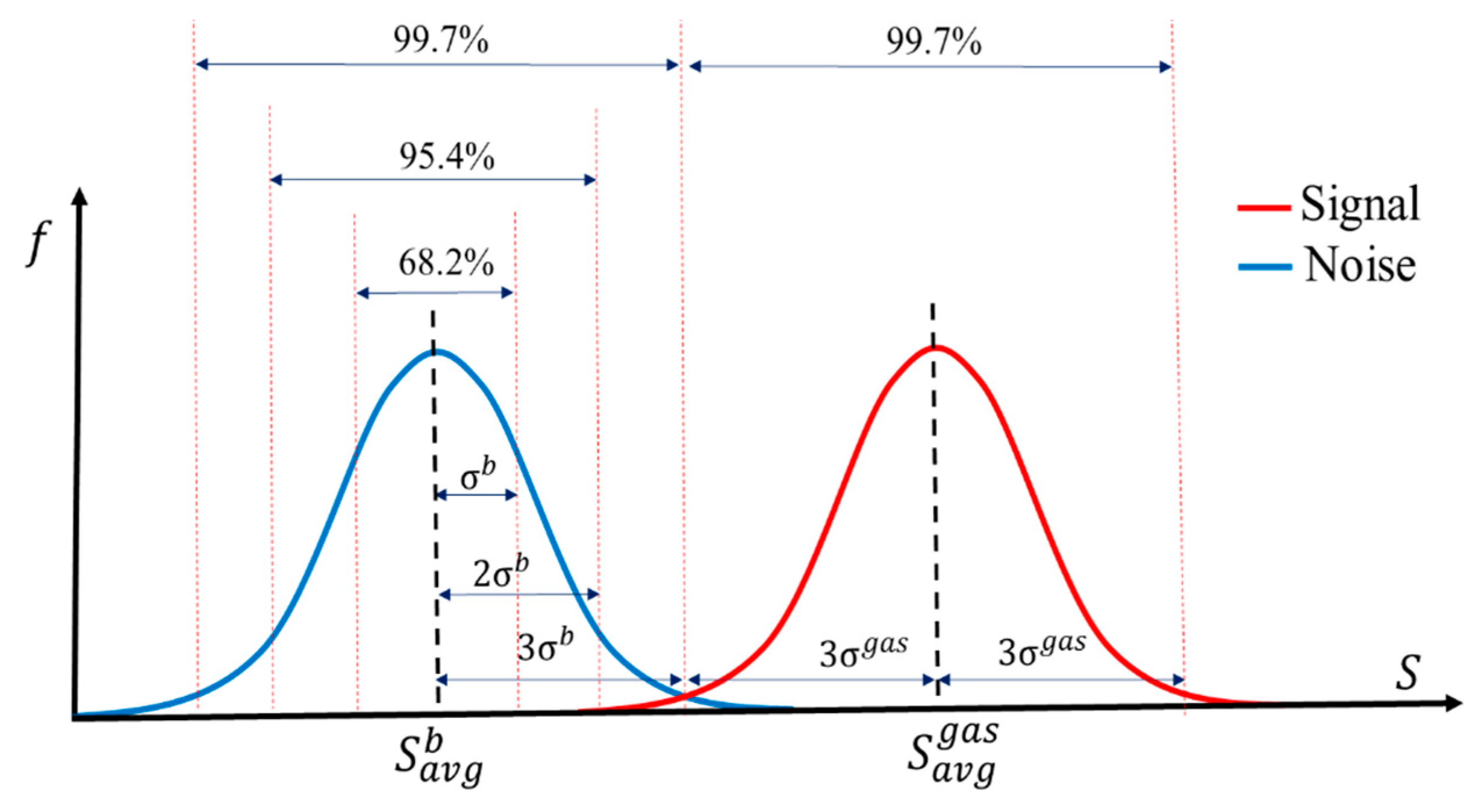
| Sensor # | Sensor’s Bulk Material | Sensor’s Surface Modification |
|---|---|---|
| S1 | SnO2 (30 ± 2 nm) | - |
| S2 | SnO2 (30 ± 2 nm) | TiO2 (20 ± 2 nm) |
| S3 | SnO2 (30 ± 2 nm) | Au/Pd (1.1 ± 0.2 nm) |
| S4 | SnO2 (30 ± 2 nm) | Pt (1.5 ± 0.2 nm) |
| Mix. # | Gases and Concentrations | Possible applications | Ref. |
|---|---|---|---|
| 1 |
| Indoor air quality control | [4,5,6,7,8,9] |
| 2 |
| Natural gas analysis | [10,11,12] |
| 3 |
| Human breath analysis | [13,14,15,16,17,18] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dobrokhotov, V.; Larin, A. Multisensory Gas Chromatography for Field Analysis of Complex Gaseous Mixtures. ChemEngineering 2019, 3, 13. https://doi.org/10.3390/chemengineering3010013
Dobrokhotov V, Larin A. Multisensory Gas Chromatography for Field Analysis of Complex Gaseous Mixtures. ChemEngineering. 2019; 3(1):13. https://doi.org/10.3390/chemengineering3010013
Chicago/Turabian StyleDobrokhotov, Vladimir, and Alexander Larin. 2019. "Multisensory Gas Chromatography for Field Analysis of Complex Gaseous Mixtures" ChemEngineering 3, no. 1: 13. https://doi.org/10.3390/chemengineering3010013
APA StyleDobrokhotov, V., & Larin, A. (2019). Multisensory Gas Chromatography for Field Analysis of Complex Gaseous Mixtures. ChemEngineering, 3(1), 13. https://doi.org/10.3390/chemengineering3010013




