The Role of a SiC Sublayer in Modulating the Electrochemical Behavior of CoxSy/SiC Heterostructure Supercapacitor Electrodes
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Materials
2.2. Characterization
2.3. Electrode Preparation
2.4. Electrochemical Characterization
3. Results and Discussion
3.1. Electrochemical Deposition
3.2. Morphological and Compositional Study
3.3. X-Ray Photoelectron Spectroscopy (XPS)
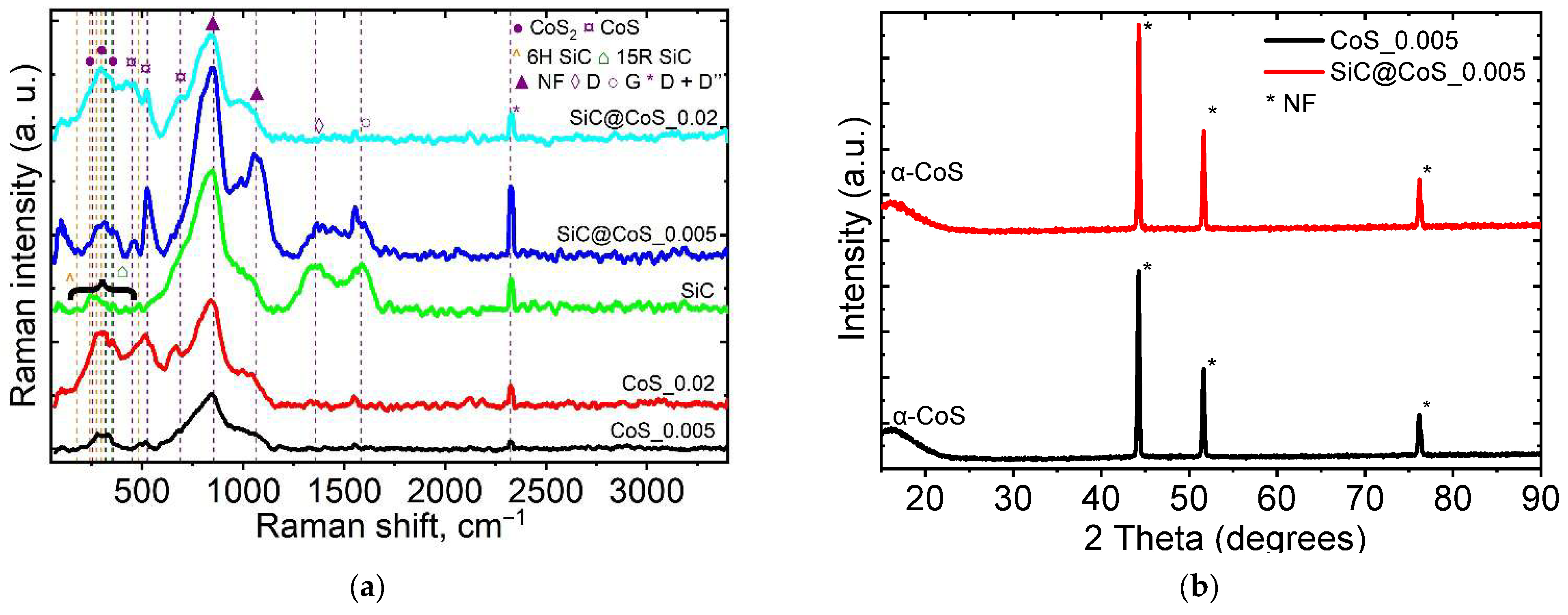
3.4. Raman and XRD Analysis
3.5. Electrochemical Study
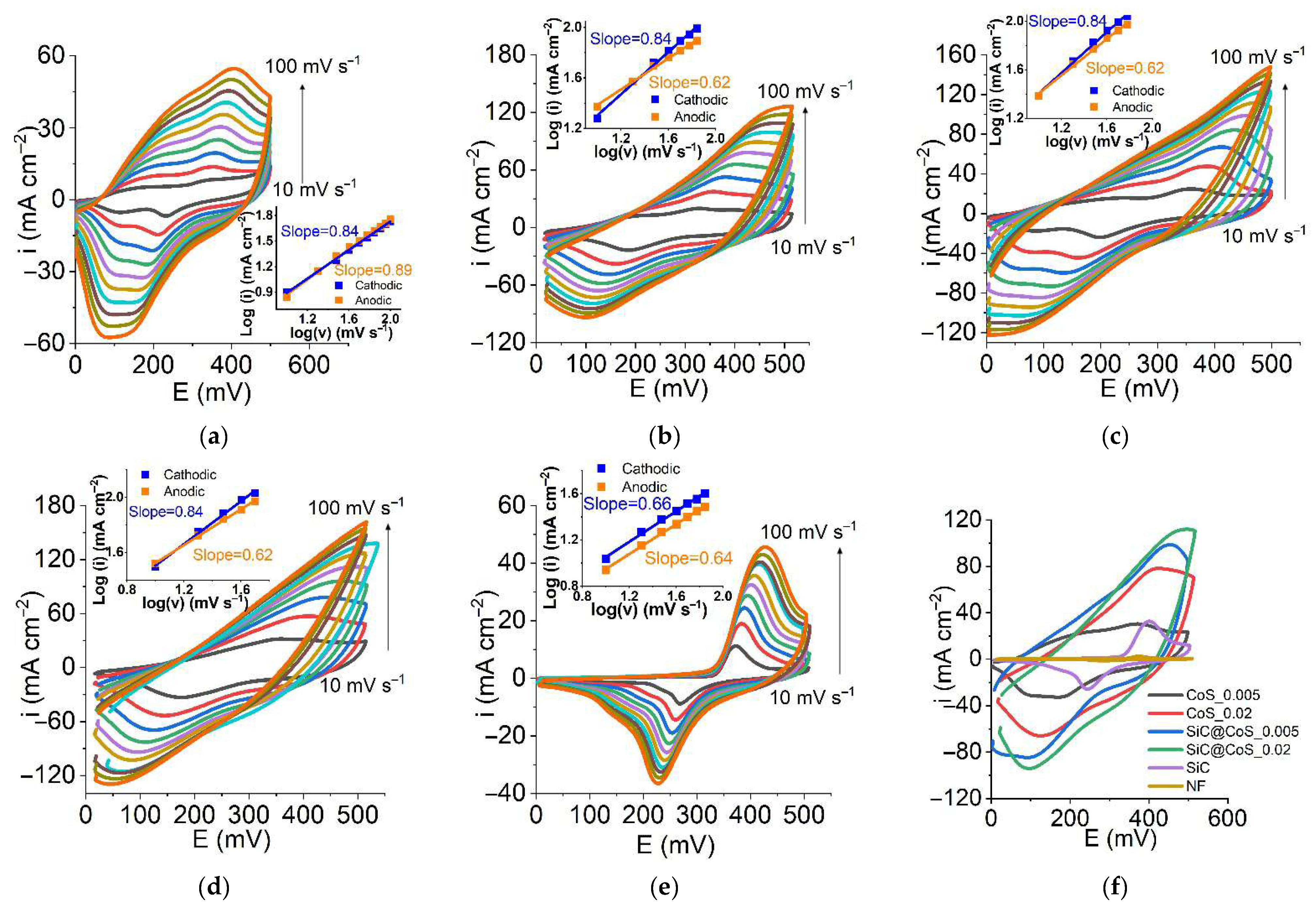
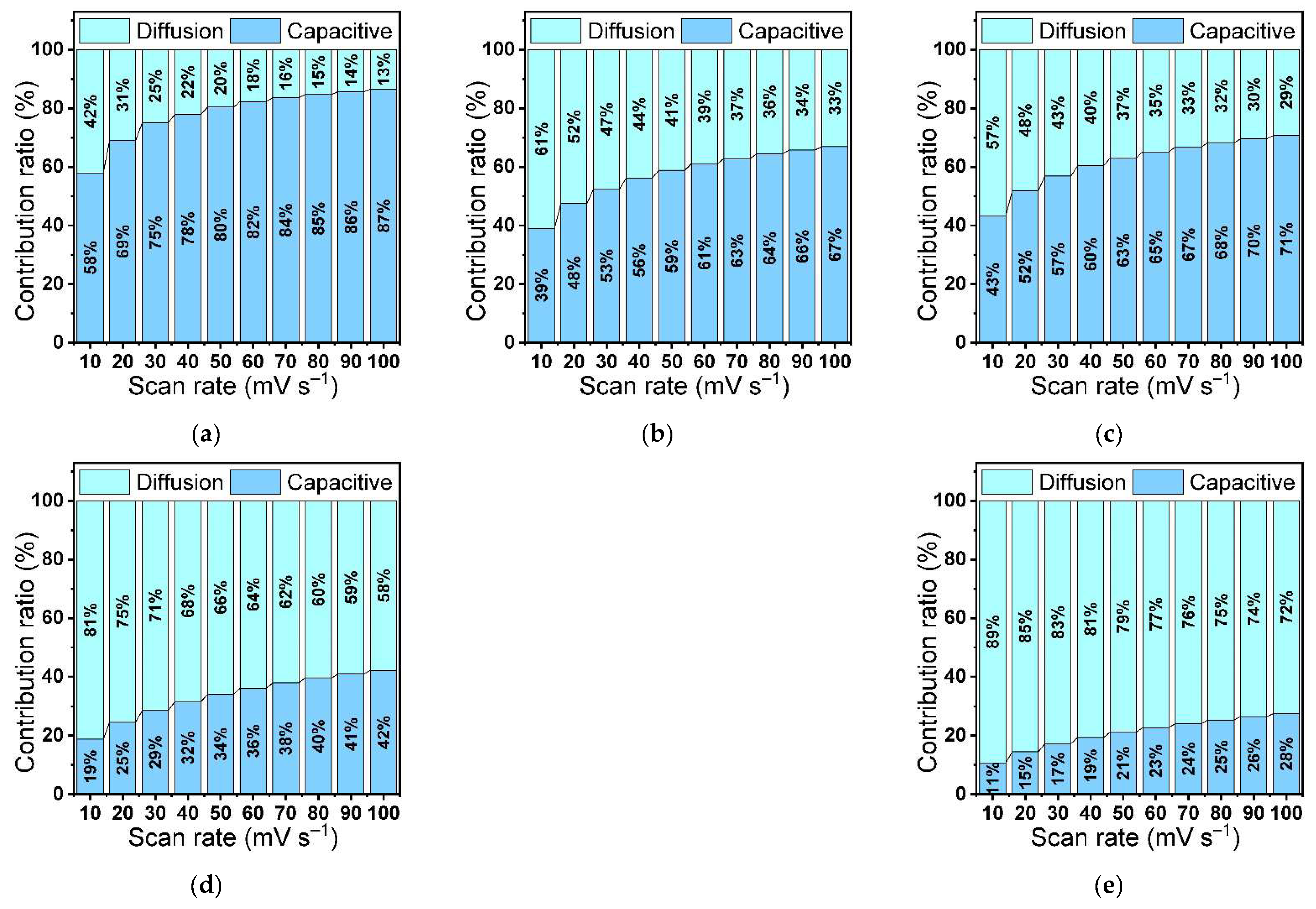
3.5.1. Cyclic Voltammetry (CV)
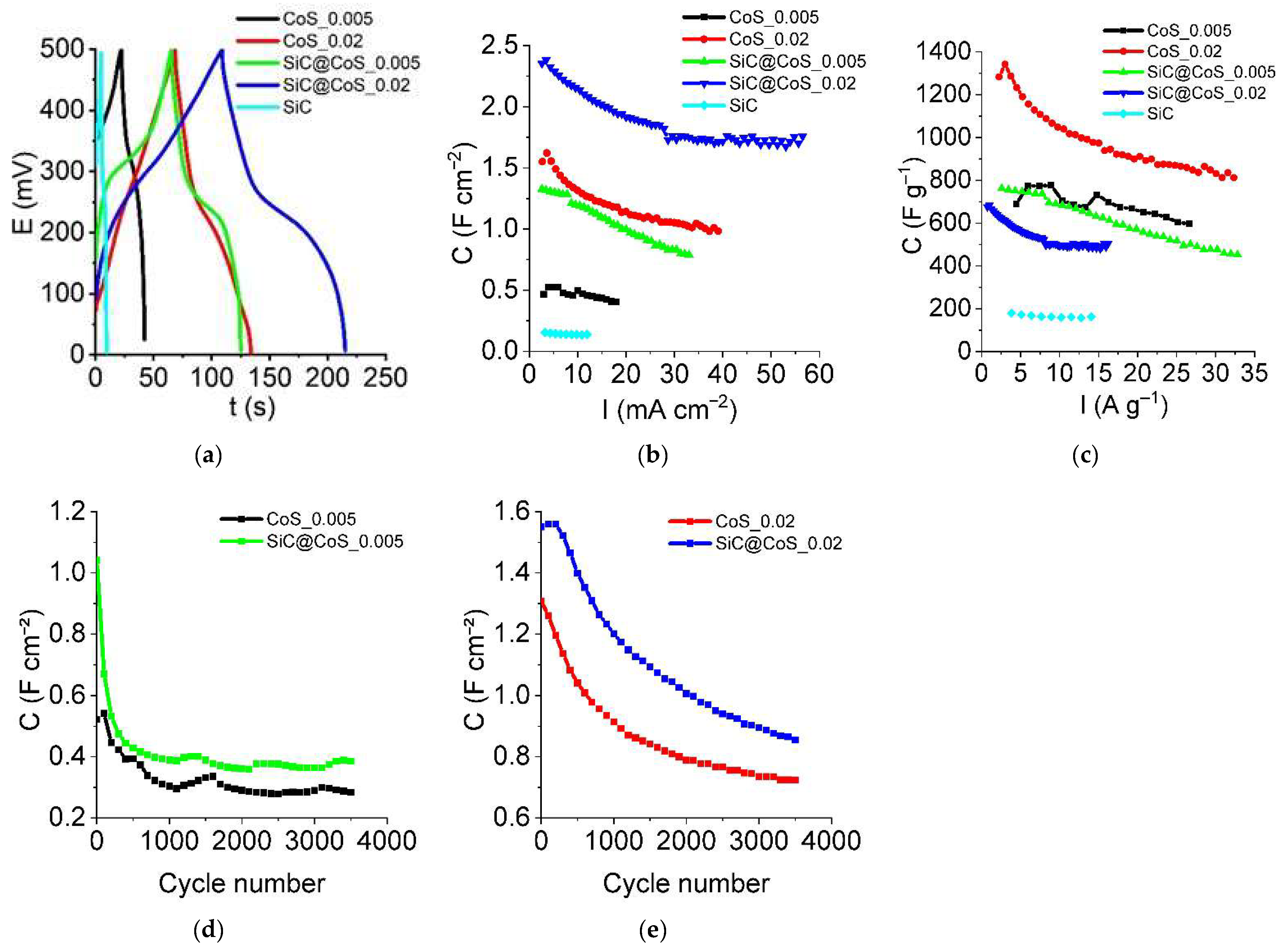
3.5.2. Galvanostatic Charging Discharging (GCD) and Stability Tests
3.6. Electrochemical Impedance Spectroscopy (EIS)
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Li, N.; Lv, T.; Yao, Y.; Li, H.; Liu, K.; Chen, T. Compact graphene/MoS2 composite films for highly flexible and stretchable all-solid-state supercapacitors. J. Mater. Chem. A. 2017, 5, 3267–3273. [Google Scholar] [CrossRef]
- Yang, X.; Zhao, L.; Lian, J. Arrays of hierarchical nickel sulfides/MoS2 nanosheets supported on carbon nanotubes backbone as advanced anode materials for asymmetric supercapacitor. Power Sources 2017, 343, 373–382. [Google Scholar] [CrossRef]
- Poimenidis, I.; Papakosta, N.; Loukakos, P.A.; Marnellos, G.E.; Konsolakis, M. Highly efficient cobalt sulfide heterostructures fabricated on nickel foam electrodes for oxygen evolution reaction in alkaline water electrolysis cells. Surfaces 2023, 6, 493–508. [Google Scholar] [CrossRef]
- Gulen, M. Lithium perchlorate-assisted electrodeposition of CoS catalyst surpassing the performance of platinum in dye sensitized solar cell. J. Mater. Sci. 2022, 57, 3513–3524. [Google Scholar] [CrossRef]
- Prakash, J.; Jasrotia, R.; Himanshi; Singh, J.; Kandwal, A.; Sharm, P. Wastewater Treatment: The Emergence of Cobalt Ferrite and Its Composites in Sulfate Radical-Based Advanced Oxidation Processes. Emergent Mater. 2024, 7, 2237–2259. [Google Scholar] [CrossRef]
- Halder, A.; Aman, M.; Lichchhavi; Jha, S.K. Enhancing supercapacitor performance with binder-free cobalt sulfide pseudo-capacitive electrodes: A path to sustainable energy storage. J. Elect. Chem. 2024, 972, 118631. [Google Scholar] [CrossRef]
- Chen, C.-Y.; Shih, Z.-Y.; Yang, Z.; Chang, H.-T. Carbon nanotubes/cobalt sulfide composites as potential high-rate and high-efficiency supercapacitors. J. Power Sources 2012, 215, 43–47. [Google Scholar] [CrossRef]
- Ojha, G.P.; Kang, G.W.; Kuk, Y.-S.; Hwang, Y.E.; Kwon, O.H.; Pant, B.; Acharya, J.; Park, Y.W.; Park, M. Silicon Carbide Nanostructures as Potential Carbide Material for Electrochemical Supercapacitors: A Review. Nanomaterials 2023, 13, 150. [Google Scholar] [CrossRef]
- Khan, J.; Ahmed, A.; Al-Kahtan, A.A. Strategic design of binary transition metal sulfides for superior asymmetric supercapacitors. Mater. Adv. 2025, 6, 3344–3354. [Google Scholar] [CrossRef]
- Li, X.; Wu, H.; Guan, C.; M Elshahawy, A.; Dong, Y.; J Pennycook, S.; Wang, J. (Ni, Co) Se2/NiCo-LDH core/shell structural electrode with the cactus-like (Ni, Co) Se2 core for asymmetric supercapacitors. Small 2018, 15, 1803895. [Google Scholar] [CrossRef]
- Wang, H. Facile synthesis of silver fungus-like CoS for high-performance supercapacitors. J. Energy Storage 2021, 40, 102764. [Google Scholar] [CrossRef]
- Liu, H.; Hu, R.; Qi, J.; Sui, Y.; He, Y.; Meng, Q.; Wei, F.; Ren, Y.; Yulong, Z.; Wei, W. One-Step Synthesis of Nanostructured CoS2 Grown on Titanium Carbide MXene for High-Performance Asymmetrical Supercapacitors. Adv. Mater. Interfaces 2020, 7, 1901659. [Google Scholar] [CrossRef]
- Luo, F.; Li, J.; Yuan, H.; Xiao, D. Rapid synthesis of three dimensional flower-like cobalt sulfide hierarchitectures by microwave assisted heating method for high-performance supercapacitors. Electrochim. Acta 2014, 123, 183–189. [Google Scholar] [CrossRef]
- Fei, Z.; Song, Y.; Wu, M.; Wu, Y.; Chen, Y.; Kang, D.J.; Bian, C.; Qian, Y. MOF-derived metal sulfides and their composites: Synthesis and their electrochemical water splitting. Catalysts 2025, 15, 928. [Google Scholar] [CrossRef]
- Kalusulingam, R.S.; Ravi, K.; Mathi, S.; Mikhailova, T.S.; Srinivasan, K.; Biradar, A.V.; Myasoedova, T.N. Bagasse derived n-doped graphitic carbon encapsulated cobalt nanoparticles as an efficient bifunctional catalyst for water splitting reaction. Colloids Surf. A Physicochem. Eng. Asp. 2024, 692, 133959. [Google Scholar] [CrossRef]
- Chen, F.; Jia, D.; Jin, X.; Cao, Y.; Liu, A. A general method for the synthesis of graphene oxide-metal sulfide composites with improved photocatalytic activities. Dye. Pigment. 2015, 125, 142–150. [Google Scholar] [CrossRef]
- Fan, J.; Hu, L.; Qi, Z.; Wan, T.; Huang, S.; Zhang, X.; Han, Z.; Chu, D. Electrodeposited cobalt sulfide on a vertical graphene nanocomposite for high-performance supercapacitors. New J. Chem. 2021, 45, 20249. [Google Scholar] [CrossRef]
- Sarma, A.; Dippel, A.-C.; Gutowski, O.; Etter, M.; Lippmann, M.; Seeck, O.; Manna, G.; Sanyal, M.K.; Keller, T.F.; Kulkarni, S.; et al. Electrodeposition of nanowires of a high copper content thiourea precursor of copper sulfide. RSC Adv. 2019, 9, 31900–31910. [Google Scholar] [CrossRef]
- Edwards, P.H.; Bairan Espano, J.R.; Macdonald, J.E. Rational Phase Control in the Synthesis of Cobalt Sulfides. Chem. Mater. 2024, 36, 7186–7196. [Google Scholar] [CrossRef]
- Plugotarenko, N.K.; Myasoedova, T.N.; Mikhailova, T.S.; Grigoryev, M.N. Electrochemical Deposition of Silicon-Carbon Films: A Study on the Nucleation and Growth Mechanism. Nanomaterials 2019, 9, 1754. [Google Scholar] [CrossRef]
- Grigoryev, M.N.; Myasoedova, T.N.; Mikhailova, T.S.; Plugotarenko, N.K. The electrochemical deposition of silicon—Carbon thin films from organic solution. In Saint Petersburg OPEN 2018. Book of Abstracts; Academic University Publishing: Saint Petersburg, Russia, 2018; pp. 563–564. [Google Scholar]
- Myasoedova, T.N.; Mikhailova, T.S.; Grigoryev, M.N. Effect of nickel and manganese doping on the structure, morphology and the electrochemical performance of the silicon-carbon films. J. Alloys Compd. 2021, 855, 157504. [Google Scholar] [CrossRef]
- Skoog, D.; Holler, F.; Crouch, S. Principles of Instrumental Analysis; Brooks/Cole, Cengage Learning: Belmont, CA, USA, 2007. [Google Scholar]
- Grigoryev, M.N.; Myasoedova, T.N.; Mikhailova, T.S. The electrochemical deposition of silicon—Carbon thin films from organic solution. J. Phys. Conf. Ser. 2018, 1124, 081043. [Google Scholar] [CrossRef]
- Li, Z.B.; Liu, P.; Yang, G.W. Efficient All-2D Amorphous Cobalt Sulfide Nanosheets/Multilayered Molybdenum Disulfide Hybrid Heterojunction Catalyst for Electrochemical Hydrogen Evolution. Glob. Chall. 2019, 4, 1900066. [Google Scholar] [CrossRef] [PubMed]
- Ait-Karra, A.; Zakir, O.; Mourak, A.; Lasri, M.; Idouhli, R.; Abouelfida, A.; Mohy Eddine Khadiri, M.E.; Benzakour, J. A Comprehensive Structural, Morphological, and Electrochemical Analysis of Electrodeposited Cobalt Sulfide Thin Films. J. Electrochem. Soc. 2025, 172, 052503. [Google Scholar] [CrossRef]
- Lin, J.-Y.; Liao, J.-H.; Chou, S.-W. Cathodic electrodeposition of highly porous cobalt sulfide counter electrodes for dye-sensitized solar cells. Electrochim. Acta 2011, 56, 8818–8826. [Google Scholar] [CrossRef]
- Ghahremaninezhad, A.; Asselin, E.; Dixon, D.G. Electrodeposition and Growth Mechanism of Copper Sulfide Nanowires. J. Phys. Chem. C 2011, 115, 9320–9334. [Google Scholar] [CrossRef]
- Ait-karra, A.; Zakir, O.; Ait baha, A.; Lasri, M.; Idouhli, R.; Elyaagoubi, M.; Abouelfida, A.; Khadiri, M.; Benzakour, J. Electrodeposition and characterization of copper sulfide (CuS) thin film: Towards an understanding of the growth mechanism. J. Solid. State Electrochem. 2023, 27, 2051–2065. [Google Scholar] [CrossRef]
- Kinner, T.; Bhandari, K.; Bastola, E.; Monahan, B.M.; Haugen, N.; Roland, P.J.; Bigioni, T.; Ellingson, R. Majority carrier type control of cobalt iron sulfide (CoxFe1–xS2) pyrite nanocrystals. J. Phys. Chem. C 2016, 120, 5706–5713. [Google Scholar] [CrossRef]
- Peng, S.; Han, X.; Li, L.; Zhu, Z.; Cheng, F.; Srinivansa, M.; Adams, S.; Ramakrishna, S. Unique cobalt sulfide/reduced graphene oxide composite as an anode for sodium-ion batteries with superior rate capability and long cycling stability. Small 2016, 12, 1359–1368. [Google Scholar] [CrossRef]
- Bokobza, L.; Bruneel, J.-L.; Couzi, M. Raman Spectra of Carbon-Based Materials (from Graphite to Carbon Black) and of Some Silicone Composites. C J. Carbon. Res. 2015, 1, 77–94. [Google Scholar] [CrossRef]
- Merlen, A.; Buijnsters, J.G.; Pardanaud, C. A Guide to and Review of the Use of Multiwavelength Raman Spectroscopy for Characterizing Defective Aromatic Carbon Solids: From Graphene to Amorphous Carbons. Coatings 2017, 7, 153. [Google Scholar] [CrossRef]
- Aksyanov, G.; Kompan, M.E.; Kul’kova, I.V. Raman Scattering in Mosaic Silicon Carbide Films. Fiz. Tverd. Tela 2010, 52, 1724–1728. [Google Scholar] [CrossRef]
- Krasser, W.; Renouprez, A.J. Raman Scattering of Hydrogen Chemisorbed on Silica—Supported Nickel. J. Raman Spectrosc. 1979, 8, 92–94. [Google Scholar] [CrossRef]
- Qiu, Z.; Tai, C.W.; Niklasson, G.A.; Edvinsson, T. Direct observation of active catalyst surface phases and the effect of dynamic self-optimization in NiFe-layered double hydroxides for alkaline water splitting. Energy Environ. Sci. 2019, 12, 572–581. [Google Scholar] [CrossRef]
- Faid, Y.A.; Barnett, A.O.; Seland, F.; Svein Sunde, S. Ni/NiO nanosheets for alkaline hydrogen evolution reaction: In situ electrochemical-Raman study. Electrochim. Acta 2020, 361, 137040. [Google Scholar] [CrossRef]
- Usha, K.S.; Sivakumar, R.; Sanjeeviraja, C. Preparation of pure NiO thin film by radio frequency magnetron sputtering technique and investigation on its properties. J. Mater. Sci. Mater. Electron. 2022, 33, 16136–16143. [Google Scholar] [CrossRef]
- Zang, S.; Hou, Y.; Chang, J.; Xu, F.; Wu, D.; Jiang, K.; Gao, Z. Amorphous-crystalline heterostructures enable energy-level matching of cobalt sulfide/nickel iron layered double hydroxide for efficient oxygen evolution reaction. J. Colloid. Interface Sci. 2024, 656, 485–494. [Google Scholar] [CrossRef]
- Toparli, C. Synthesis and characterization of high entropy oxy-hydroxides for electrocatalytic oxygen evolution and reduction reaction. J. ESOGU Eng. Arch. Fac. 2023, 31, 698–704. [Google Scholar] [CrossRef]
- Bard, A.J.; Faulkner, L.R. Electrochemical Methods: Fundamentals and Applications, 2nd ed; John Wiley & Sons: Hoboken, NJ, USA, 2001. [Google Scholar]
- Wang, C.-M.; Wen, C.-Y.; Chen, Y.-C.; Chang, J.-Y.; Ho, C.-W.; Kao, K.-S.; Shih, W.-C.; Chiu, C.-M.; Shen, Y.-A. The Influence of Specific Surface Area on the Capacitance of the Carbon Electrodes Supercapacitor. In Proceedings of the International Conference on Industrial Application Engineering, Fukuoka, Japan, 28–31 March 2015. [Google Scholar] [CrossRef]
- Junlin, L.; Xu, W.; Li, S.; Javed, M.S.; Liu, G.; Hu, C. Rational design of CuO nanostructures grown on carbon fiber fabrics with enhanced electrochemical performance for flexible supercapacitor. J. Mater. Sci. 2018, 53, 739–748. [Google Scholar] [CrossRef]
- Yang, T.; Liu, J.; Zhang, M.; Yang, D.; Zheng, J.; Ju, Z.; Cheng, J.; Zhuang, J.; Liu, Y.; Zhong, J.; et al. Encapsulating MnSe nanoparticles inside 3D hierarchical carbon frameworks with lithium storage boosted by in situ electrochemical phase transformation. ACS Appl. Mater. Interfaces 2019, 11, 33022–33032. [Google Scholar] [CrossRef]
- Wang, Y.-K.; Liu, M.-C.; Cao, J.; Zhang, H.-J.; Kong, L.-B.; Trudgeon, D.P.; Li, X.; Walsh, F.C. 3D hierarchically structured CoS nanosheets: Li+ storage mechanism and application of the high-performance lithium-ion capacitors. ACS Appl. Mater. Interfaces 2019, 12, 3709–3718. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.M.; Yao, Z.X.; Liu, Y.S.; Diao, Y.X.; Hu, G.X.; Zhang, Q.F.; Li, Z. In situ synthesis of nitrogen site activated cobalt sulfide@N, S dual-doped carbon composite for a high-performance asymmetric supercapacitor. J. Colloid. Interface Sci. 2021, 585, 30–42. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Ma, Y.; Sun, R.; Zhang, W.; Liu, H.; Liu, H.; Liao, L. Enhanced Electrochemical Performance of Metallic CoS-Based Supercapacitor by Cathodic Exfoliation. Nanomaterials 2023, 13, 1411. [Google Scholar] [CrossRef] [PubMed]
- Plugotarenko, N.K.; Myasoedova, T.N.; Bogush, I.Y. The effect of the electrolyte concentration on the charge transfer at the electrolyte/silicon-carbon interface: Electrochemical impedance spectrometry study. Mater. Sci. Semicond. Process. 2021, 135, 106121. [Google Scholar] [CrossRef]
- Jiang, D.; Xu, Q.; Meng, S.; Xia, C.; Chen, M. Construction of cobalt sulfide/graphitic carbon nitride hybrid nanosheet composites for high performance supercapacitor electrodes. J. Alloys Compd. 2017, 706, 41–47. [Google Scholar] [CrossRef]
- Ahmad, R.; Iqbal, N.; Noor, T. Development of ZIF-Derived Nanoporous Carbon and Cobalt Sulfide-Based Electrode Material for Supercapacitor. Materials 2019, 12, 2940. [Google Scholar] [CrossRef]
- Miya, L.A.; Ghosh, S.K.; Kumari, P.; Mallick, K.; Morema, C.N.M. Eco-friendly and sustainable supercapacitor design: Cobalt sulfide nanoparticles embedded on carbon cloth as an electrode material for asymmetric devices. Chem. Pap. 2025, 79, 7617–7631. [Google Scholar] [CrossRef]
- Deng, Y.; Wang, X.; Wang, Z.; Wang, X.; Li, Z.; Wang, L.; Zhou, C.; Chen, D.; Luo, Y. Yolk–Shell Structured Nickel Cobalt Sulfide and Carbon Nanotube Composite for High-Performance Hybrid Supercapacitors. Energy Fuels 2021, 35, 5342–5351. [Google Scholar] [CrossRef]
- Kochuparampil, A.P.; Joshi, J.H.; Rathod, K.R.; Jethva, H.O.; Joshi, M.J. The effect of adding zinc sulphide nano-particles on structural, functional, thermal, dielectric, impedance and nonlinear optical properties of ammonium pentaborate crystals. J. Phys. Chem. Solids 2019, 129, 424–433. [Google Scholar] [CrossRef]
- Ghosh, S.; Paul, A.; Samanta, P.; Landge, B.; Mandal, S.K.; Sinha, S.; Dutta, G.G.; Murmu, N.C.; Kuila, T. Influence of Transition Metals (Cu and Co) on the Carbon-Coated Nickel Sulfide Used as Positive Electrode Material in Hybrid Supercapacitor Device. J. Compos. Sci. 2021, 5, 180. [Google Scholar] [CrossRef]
- Vadhva, P.; Hu, J.; Johnson, J.M.; Stocker, R.; Braglia, M.; Brett, D.J.L.; Rettie, A. Electrochemical Impedance Spectroscopy for All-Solid-State Batteries: Theory, Methods and Future Outlook. ChemElectroChem 2021, 8, 1930–1947. [Google Scholar] [CrossRef]
- Li, H.; Zhao, Y.; Wang, C.A. MoS2/CoS2 composites composed of CoS2 octahedrons and MoS2 nano-flowers for supercapacitor electrode materials. Front. Mater. Sci. 2018, 12, 354–360. [Google Scholar] [CrossRef]
- Li, J.; Li, J.; Shao, M.; Yan, Y.; Li, R. MOF-Derived Ultrathin NiCo-S Nanosheet Hybrid Array Electrodes Prepared on Nickel Foam for High-Performance Supercapacitors. Nanomaterials 2023, 13, 1229. [Google Scholar] [CrossRef]
- Kim, H.-J.; Kim, C.-W.; Punnoose, D.; Gopi, C.V.V.M.; Kim, S.-K.; Prabakar, K.; Rao, S.S. Nickel doped cobalt sulfide as a high performance counter electrode for dye-sensitized solar cells. Appl. Surf. Sci. 2015, 328, 78–85. [Google Scholar] [CrossRef]
- Karmakar, S. Impedance Spectroscopy for Electroceramics and Electrochemical System. Appl. Phys. 2024, 6, 10–56. [Google Scholar] [CrossRef]
- Cesiulis, H.; Tsyntsaru, N.; Ramanavicius, A.; Ragoisha, G. The Study of Thin Films by Electrochemical Impedance Spectroscopy. In Nanostructures and Thin Films for Multifunctional Applications: Technology, Properties and Devices; NanoScience and Technology; Springer: Cham, Switzerland, 2016; pp. 3–42. [Google Scholar] [CrossRef]




| Sample | CoS_0.005 | CoS_0.02 | SiC@CoS_0.005 | SiC@CoS_0.02 | SiC |
|---|---|---|---|---|---|
| Mass of the deposited material (mg·cm−2) | 0.5 | 1.2 | 2.0 | 3.5 | 0.8 |
| Material | Electrolyte | Specific Capacitance | Current Density/Scan Rate | Cycle Stability (Capacitance Retention) | Ref. |
|---|---|---|---|---|---|
| Carbon nanotubes(CNT)/CoS | 1 M KOH | 2000 F·g−1 1000 F·g−1 | 10 mV s−1 100 mV s−1 | 1500 cycles (91%) | [7] |
| Zeolitic Imidazolate Framework -Derived Nanoporous Carbon and Cobalt Sulfide | 2 M KOH | 677 F·g−1 | 100 mV s−1 | 1600 cycles (74%) | [50] |
| Cobalt sulfide nanoparticles on carbon cloth | 2 M KOH | 382.3 F·g−1 | 5 mV s−1 | 5000 (97%) | [51] |
| Yolk−Shell-Structured Nickel Cobalt Sulfide and Carbon Nanotube Composite | 2 M KOH | 464.8 F·g−1 | 1 A·g−1 | 8000 cycles (91.3%) | [52] |
| Cobalt sulfide/graphen | 2 M KOH | 2603 F·g−1 | 5 A·g−1 | 3000 (150%) | [17] |
| SiC@CoS_0.02 | 1 M KOH | 1.55 F·cm−2 (426 F·g−1) | 21 mA·cm−2 (6 A·g−1) | 3500 (66%) | This work |
| SiC@CoS_0.005 | 1 M KOH | 1.04 F·cm−2 (598 F·g−1) | 7 mA·cm−2 (3.5 A·g−1) | 3500 (40%) | This work |
| Rs | CPE-T1 | CPE-P1 | Rf1 | CPE-T2 | CPE-P2 | Rf2 | W-R | W-T | W-P | |
|---|---|---|---|---|---|---|---|---|---|---|
| SiC | 0.94165 (1.6%) | 0.00311 (2.9%) * | 0.81055 (0.8%) | 1607 (2.9%) | 0.003414 (2.2%) | 0.72706 (2.3%) | - | - | - | - |
| SiC@CoS_0.02 | 1.109 (0.6%) | 0.17803 (8.2%) | 0.42947 (3.4%) | 1.514 (6.5%) | - | - | - | 7.41 (5.9%) | 6.916 (6.7%) | 0.62578 (2%) |
| CoS_0.005 | 1.133 (0.8%) | 0.0073565 (5.8%) | 0.770074 (1.3%) | 14.18 (6.6%) | - | - | - | 39.29 (11.7%) | 2.788 (12.8%) | 0.35671 (4.66%) |
| CoS_0.02 | 1.535 (1.2%) | 0.0155 (11.5%) | 0.685 (3.3%) | 2.447 (4.24%) | - | - | - | 8.645 (3.2%) | 15.31 (1.7%) | 0.35725 (12.5%) |
| SiC@CoS_0.005 | 0.87615 (1.5%) | 0.07478 (1.9%) | 0.51386 (5.7%) | 72 (4.6%) | 0.068861 (9.9%) | 0.53236 (4.5%) | 365 (10.3%) | 76 (6.9%) | 0.167 (7.2%) | 0.76 (5%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Moiseeva, T.A.; Bogush, I.Y.; Il’in, O.I.; Yatsenko, A.N.; Kalusulingam, R.; Myasoedova, T.N. The Role of a SiC Sublayer in Modulating the Electrochemical Behavior of CoxSy/SiC Heterostructure Supercapacitor Electrodes. ChemEngineering 2026, 10, 29. https://doi.org/10.3390/chemengineering10020029
Moiseeva TA, Bogush IY, Il’in OI, Yatsenko AN, Kalusulingam R, Myasoedova TN. The Role of a SiC Sublayer in Modulating the Electrochemical Behavior of CoxSy/SiC Heterostructure Supercapacitor Electrodes. ChemEngineering. 2026; 10(2):29. https://doi.org/10.3390/chemengineering10020029
Chicago/Turabian StyleMoiseeva, Tatiana A., Inna Yu. Bogush, Oleg I. Il’in, Alexey N. Yatsenko, Rajathsing Kalusulingam, and Tatiana N. Myasoedova. 2026. "The Role of a SiC Sublayer in Modulating the Electrochemical Behavior of CoxSy/SiC Heterostructure Supercapacitor Electrodes" ChemEngineering 10, no. 2: 29. https://doi.org/10.3390/chemengineering10020029
APA StyleMoiseeva, T. A., Bogush, I. Y., Il’in, O. I., Yatsenko, A. N., Kalusulingam, R., & Myasoedova, T. N. (2026). The Role of a SiC Sublayer in Modulating the Electrochemical Behavior of CoxSy/SiC Heterostructure Supercapacitor Electrodes. ChemEngineering, 10(2), 29. https://doi.org/10.3390/chemengineering10020029








