Oleic Acid Nanovesicles of Minoxidil for Enhanced Follicular Delivery
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Vesicles
2.3. Vesicle Physical Characterization
2.4. Differential Scanning Calorimetry (DSC)
2.5. Thermogravimetric Analysis (TGA)
2.6. X-Ray Diffraction (XRD) Study
2.7. Transmission Electron Microscopy (TEM)
2.8. Preparation of Vesicular Gel
2.9. In Vitro Drug Release
2.10. Ex Vivo Skin Study
2.10.1. Skin Permeation
2.10.2. Drug Deposition
2.11. HPLC Method
2.12. Confocal Laser Scanning Microscopy
2.13. Statistical Analysis of Data
3. Results and Discussion
3.1. Preparation and Characterization of Vesicles
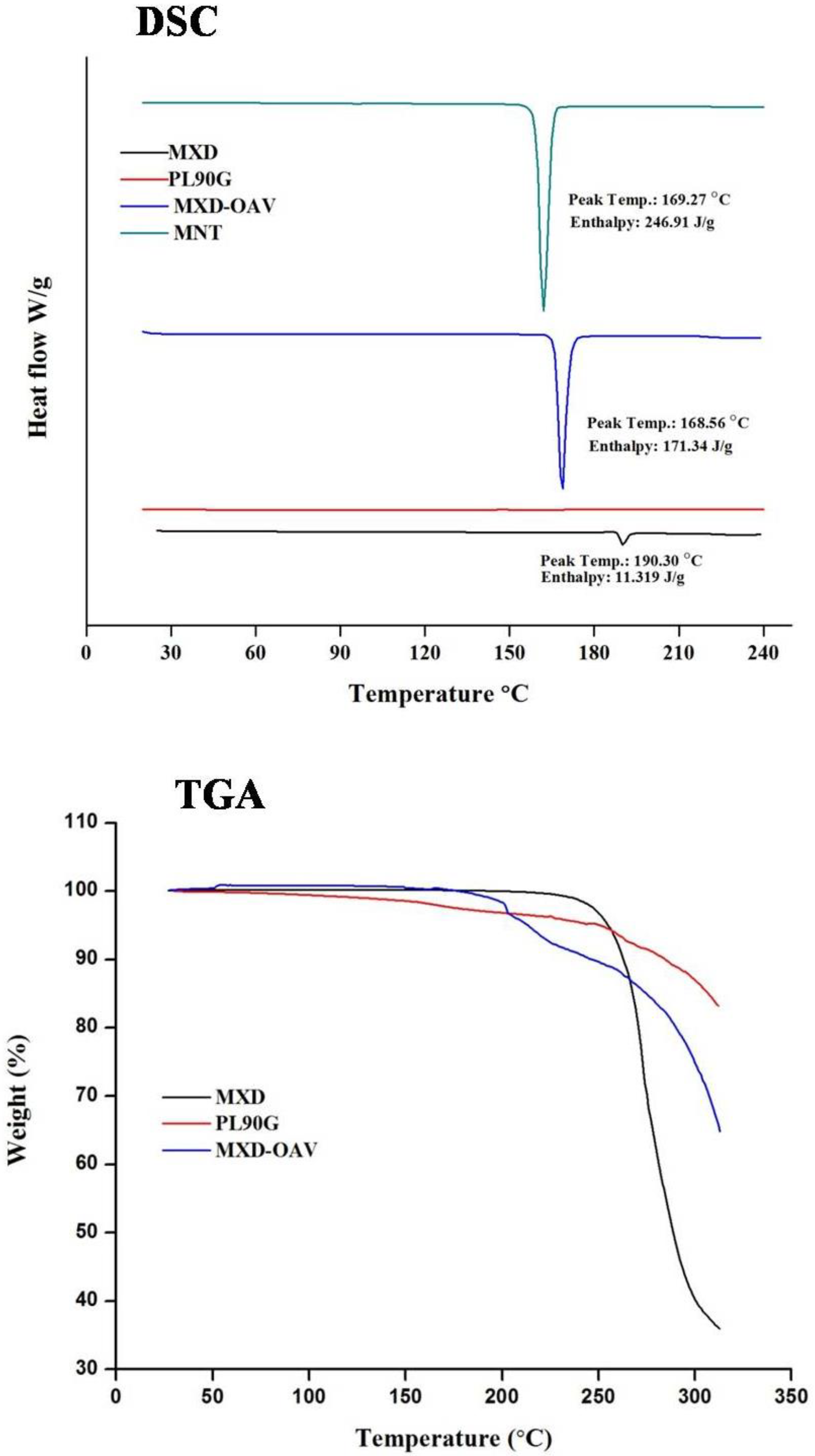
3.2. DSC Analysis
3.3. Thermogravimetric Analysis
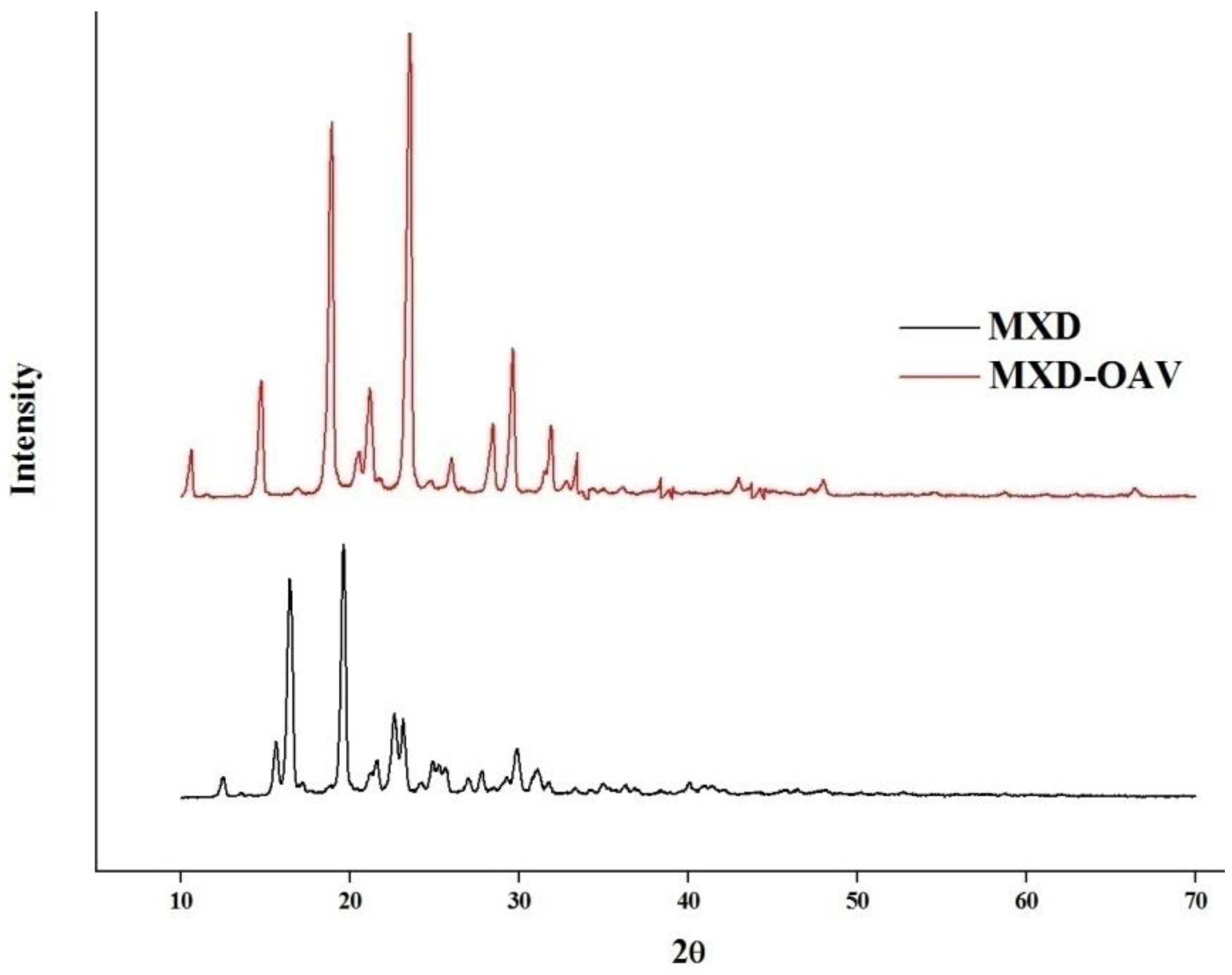
3.4. XRD Analysis
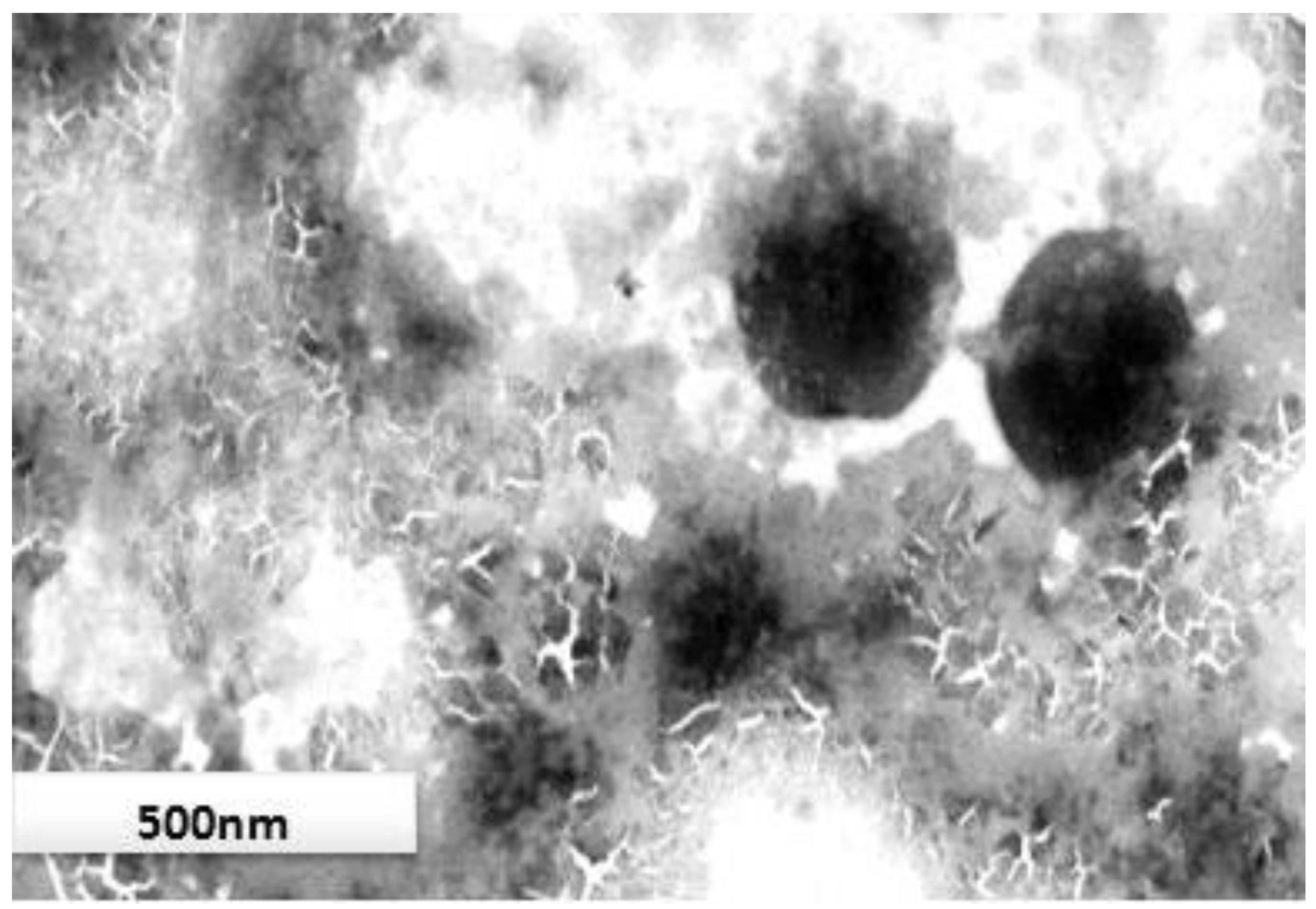
3.5. Morphological Study
3.6. Formulation and Characterization of Gel
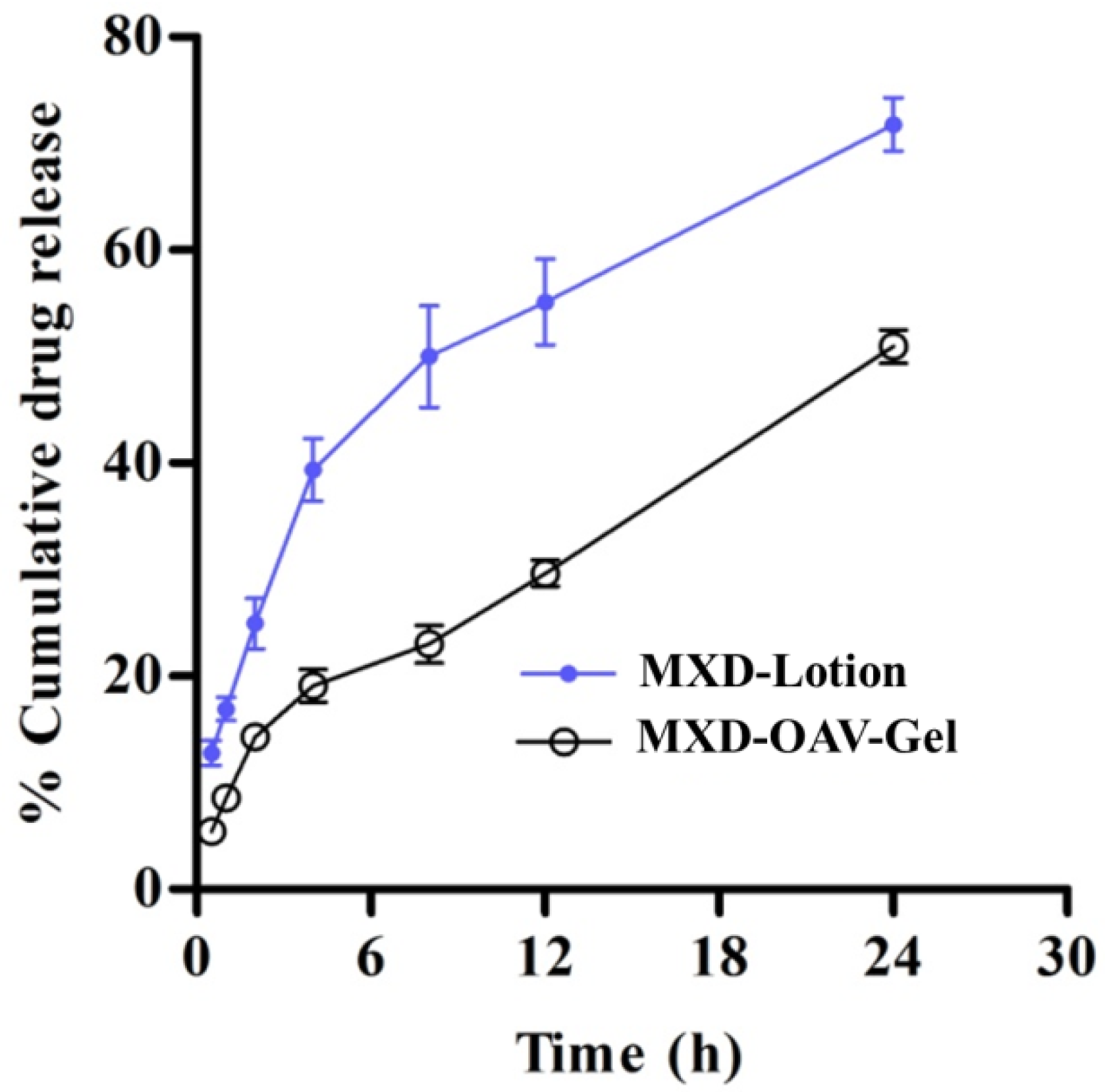
3.7. In Vitro Drug Release
3.8. Ex Vivo Skin Study
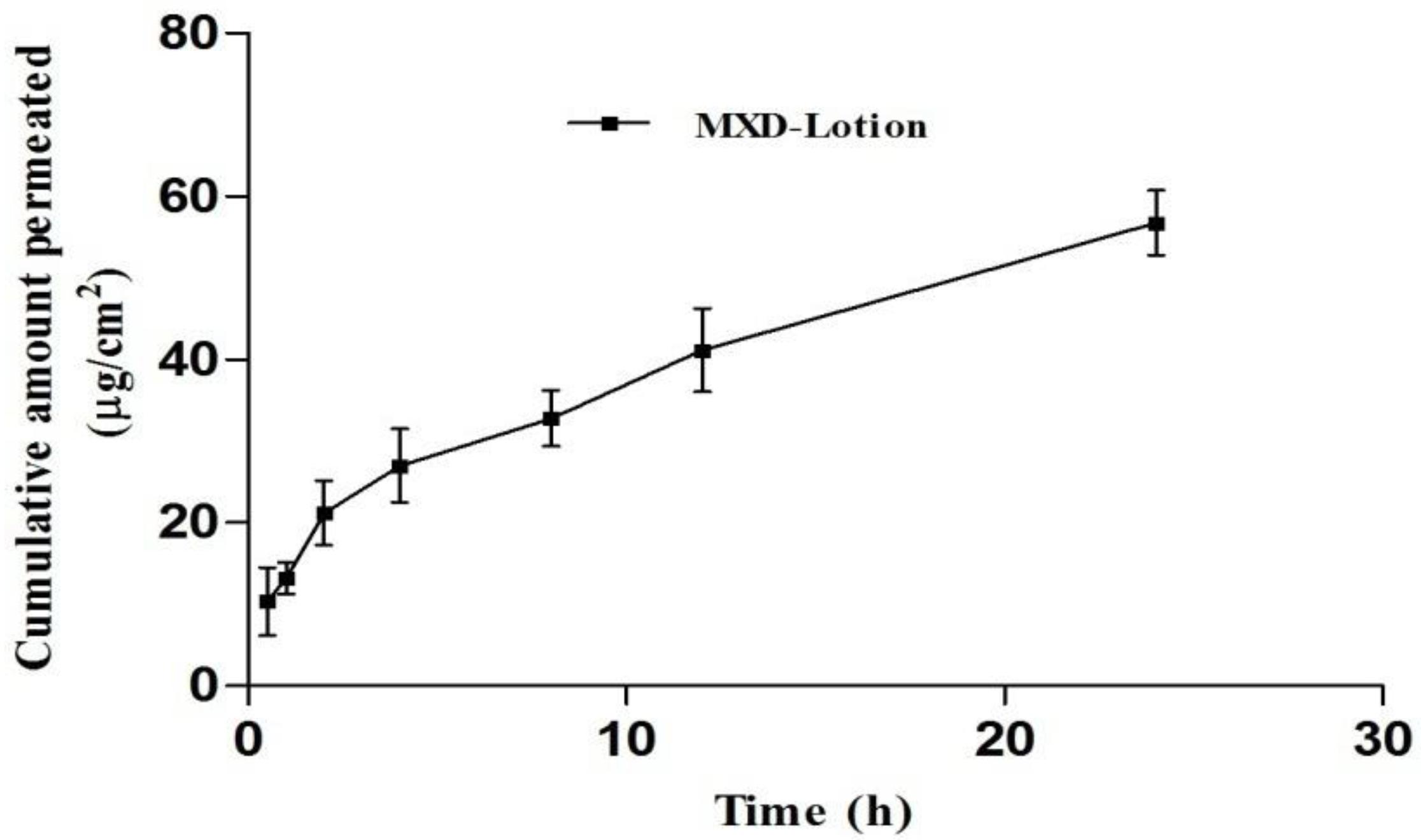
3.8.1. Drug Permeation
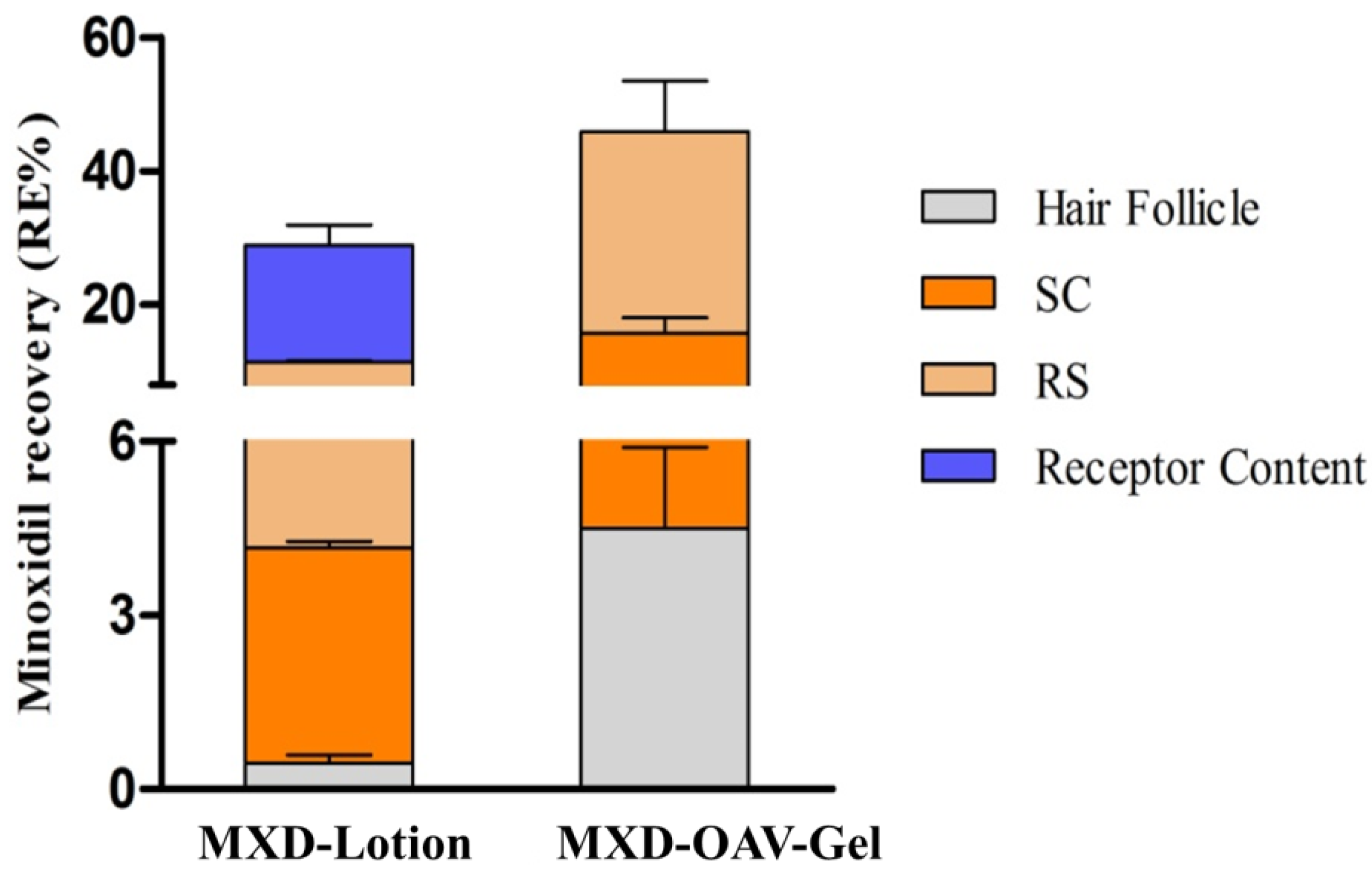
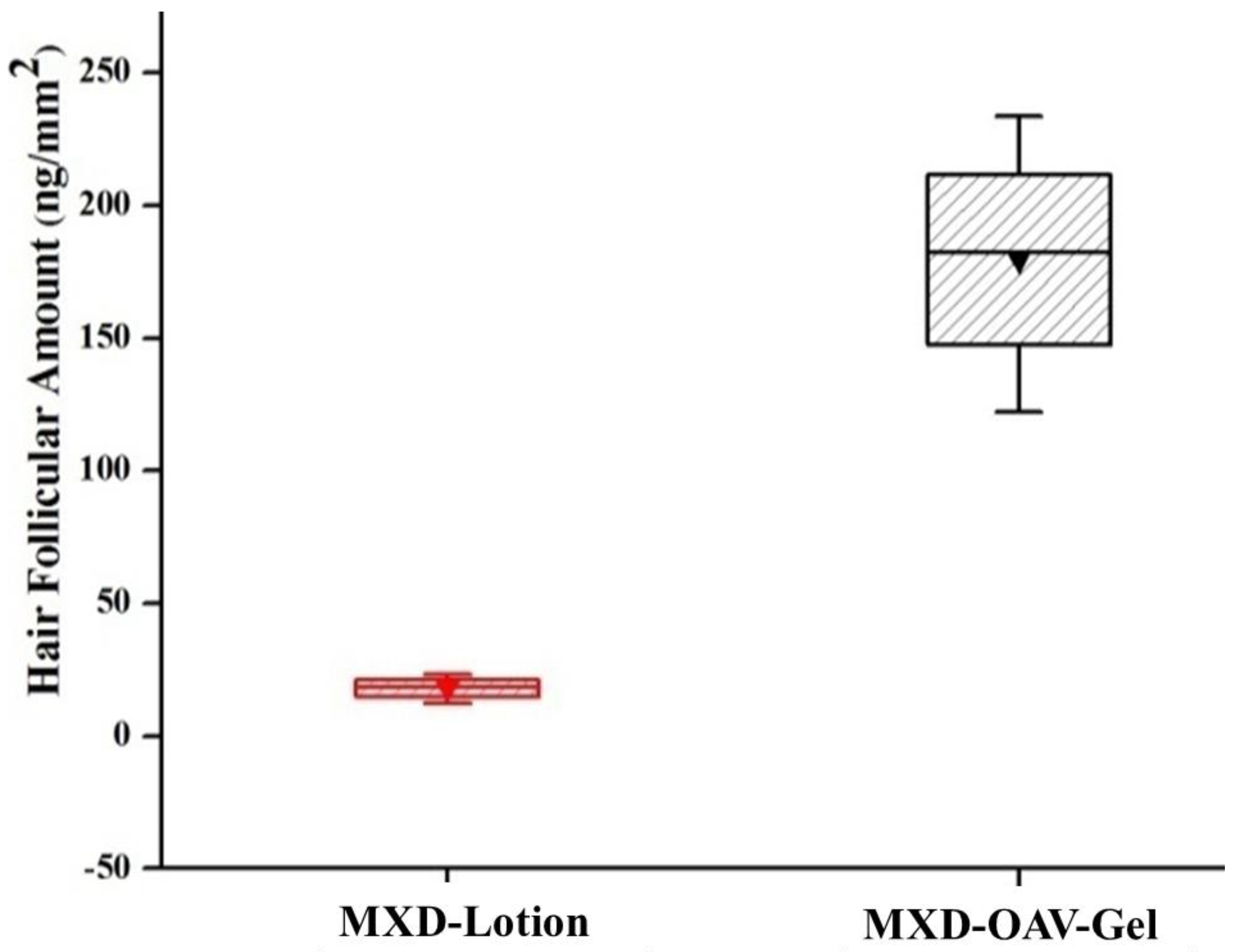
3.8.2. Drug Deposition
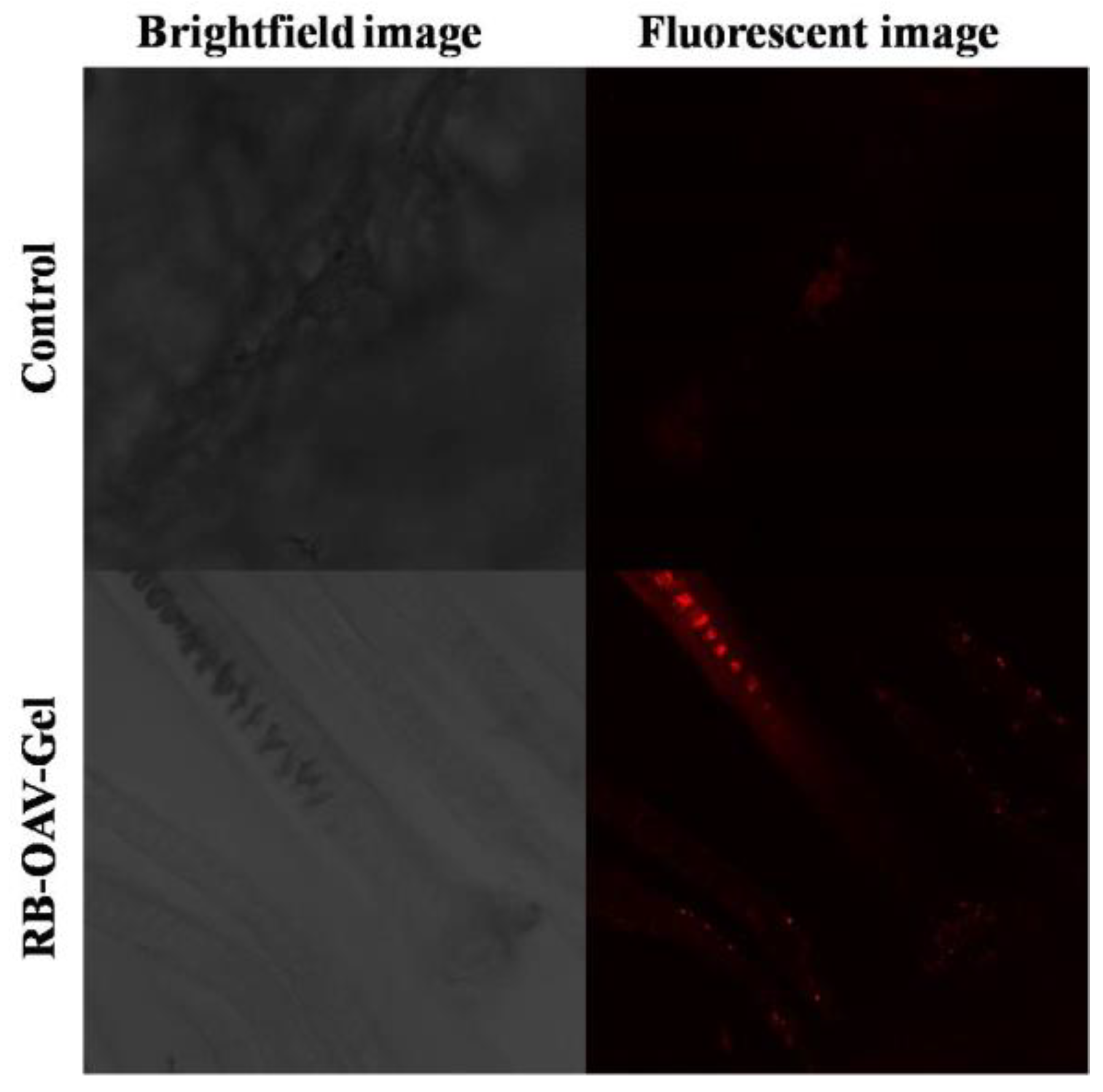
3.9. Confocal Laser Scanning Microscopy
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tricarico, D.; Maqoud, F.; Curci, A.; Camerino, G.; Zizzo, N.; Denora, N.; Cutrignelli, A.; Laquintana, V.; Lopalco, A.; la Forgia, F.; et al. Characterization of minoxidil/hydroxypropyl-beta-cyclodextrin inclusion complex in aqueous alginate gel useful for alopecia management: Efficacy evaluation in male rat. Eur. J. Pharm. Biopharm. 2018, 122, 146–157. [Google Scholar] [CrossRef] [PubMed]
- Lopedota, A.; Denora, N.; Laquintana, V.; Cutrignelli, A.; Lopalco, A.; Tricarico, D.; Maqoud, F.; Curci, A.; Mastrodonato, M.; la Forgia, F.; et al. Alginate-based hydrogel containing minoxidil/hydroxypropyl-beta-cyclodextrin inclusion complex for topical alopecia treatment. J. Pharm. Sci. 2018, 107, 1046–1054. [Google Scholar] [CrossRef] [PubMed]
- Aljuffali, I.A.; Pan, T.L.; Sung, C.T.; Chang, S.H.; Fang, J.Y. Anti-pdgf receptor beta antibody-conjugated squarticles loaded with minoxidil for alopecia treatment by targeting hair follicles and dermal papilla cells. Nanomed. Nanotechnol. Biol. Med. 2015, 11, 1321–1330. [Google Scholar] [CrossRef] [PubMed]
- Mura, S.; Manconi, M.; Sinico, C.; Valenti, D.; Fadda, A.M. Penetration enhancer-containing vesicles (pevs) as carriers for cutaneous delivery of minoxidil. Int. J. Pharm. 2009, 380, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Messenger, A.G.; Rundegren, J. Minoxidil: Mechanisms of action on hair growth. Br. J. Dermatol. 2004, 150, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Goren, A.; Naccarato, T.; Situm, M.; Kovacevic, M.; Lotti, T.; McCoy, J. Mechanism of action of minoxidil in the treatment of androgenetic alopecia is likely mediated by mitochondrial adenosine triphosphate synthase-induced stem cell differentiation. J. Biol. Regul. Homeost. Agents 2017, 31, 1049–1053. [Google Scholar] [PubMed]
- Tata, S.; Flynn, G.L.; Weiner, N.D. Penetration of minoxidil from ethanol/propylene glycol solutions: Effect of application volume and occlusion. J. Pharm. Sci. 1995, 84, 688–691. [Google Scholar] [CrossRef] [PubMed]
- Farkas, A.; Kemeny, L.; Szell, M.; Dobozy, A.; Bata-Csorgo, Z. Ethanol and acetone stimulate the proliferation of hacat keratinocytes: The possible role of alcohol in exacerbating psoriasis. Arch. Dermatol. Res. 2003, 295, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Sakazaki, F.; Ogino, H.; Arakawa, T.; Okuno, T.; Ueno, H. Low-dose ethanol aggravates allergic dermatitis in mice. Alcohol 2014, 48, 501–508. [Google Scholar] [CrossRef] [PubMed]
- Friedman, E.S.; Friedman, P.M.; Cohen, D.E.; Washenik, K. Allergic contact dermatitis to topical minoxidil solution: Etiology and treatment. J. Am. Acad. Dermatol. 2002, 46, 309–312. [Google Scholar] [CrossRef] [PubMed]
- Hagemann, T.; Schlutter-Bohmer, B.; Allam, J.-P.; Bieber, T.; Novak, N. Positive lymphocyte transformation test in a patient with allergic contact dermatitis of the scalp after short-term use of topical minoxidil solution. Contact Dermatitis 2005, 53, 53–55. [Google Scholar] [CrossRef] [PubMed]
- Balakrishnan, P.; Shanmugam, S.; Lee, W.S.; Lee, W.M.; Kim, J.O.; Oh, D.H.; Kim, D.D.; Kim, J.S.; Yoo, B.K.; Choi, H.G.; et al. Formulation and in vitro assessment of minoxidil niosomes for enhanced skin delivery. Int. J. Pharm. 2009, 377, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Mura, S.; Manconi, M.; Valenti, D.; Sinico, C.; Vila, A.O.; Fadda, A.M. Transcutol containing vesicles for topical delivery of minoxidil. J. Drug Target. 2011, 19, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Brown, M.B.; Jones, S.A. The effects of particle properties on nanoparticle drug retention and release in dynamic minoxidil foams. Int. J. Pharm. 2010, 383, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Uprit, S.; Kumar Sahu, R.; Roy, A.; Pare, A. Preparation and characterization of minoxidil loaded nanostructured lipid carrier gel for effective treatment of alopecia. Saudi Pharm. J. 2013, 21, 379–385. [Google Scholar] [CrossRef] [PubMed]
- Padois, K.; Cantieni, C.; Bertholle, V.; Bardel, C.; Pirot, F.; Falson, F. Solid lipid nanoparticles suspension versus commercial solutions for dermal delivery of minoxidil. Int. J. Pharm. 2011, 416, 300–304. [Google Scholar] [CrossRef] [PubMed]
- Aljuffali, I.A.; Sung, C.T.; Shen, F.M.; Huang, C.T.; Fang, J.Y. Squarticles as a lipid nanocarrier for delivering diphencyprone and minoxidil to hair follicles and human dermal papilla cells. AAPS J. 2014, 16, 140–150. [Google Scholar] [CrossRef] [PubMed]
- Blume-Peytavi, U.; Massoudy, L.; Patzelt, A.; Lademann, J.; Dietz, E.; Rasulev, U.; Garcia Bartels, N. Follicular and percutaneous penetration pathways of topically applied minoxidil foam. Eur. J. Pharm. Biopharm. 2010, 76, 450–453. [Google Scholar] [CrossRef] [PubMed]
- Liao, A.H.; Lu, Y.J.; Lin, Y.C.; Chen, H.K.; Sytwu, H.K.; Wang, C.H. Effectiveness of a layer-by-layer microbubbles-based delivery system for applying minoxidil to enhance hair growth. Theranostics 2016, 6, 817–827. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.H.; Kathuria, H.; Tan, J.J.Y.; Kang, L. 3d printed drug delivery and testing systems - a passing fad or the future? Adv. Drug Deliv. Rev. 2018. [Google Scholar] [CrossRef] [PubMed]
- Kathuria, H.; Li, H.; Pan, J.; Lim, S.H.; Kochhar, J.S.; Wu, C.; Kang, L. Large size microneedle patch to deliver lidocaine through skin. Pharm. Res. 2016, 33, 2653–2667. [Google Scholar] [CrossRef] [PubMed]
- Kathuria, H.; Fong, M.H.; Kang, L. Fabrication of photomasks consisting microlenses for the production of polymeric microneedle array. Drug Deliv. Transl. Res. 2015, 5, 438–450. [Google Scholar] [CrossRef] [PubMed]
- Kathuria, H.; Kochhar, J.S.; Kang, L. Micro and nanoneedles for drug delivery and biosensing. Ther. Deliv. 2018, 9, 489–492. [Google Scholar] [CrossRef] [PubMed]
- Kwon, T.K.; Kim, J.C. In vitro skin permeation of monoolein nanoparticles containing hydroxypropyl beta-cyclodextrin/minoxidil complex. Int. J. Pharm. 2010, 392, 268–273. [Google Scholar] [CrossRef] [PubMed]
- Lopedota, A.; Cutrignelli, A.; Denora, N.; Laquintana, V.; Lopalco, A.; Selva, S.; Ragni, L.; Tongiani, S.; Franco, M. New ethanol and propylene glycol free gel formulations containing a minoxidil-methyl-beta-cyclodextrin complex as promising tools for alopecia treatment. Drug Dev. Ind. Pharm. 2015, 41, 728–736. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Oh, D.W.; Na, M.J.; Kim, D.W.; Yuk, D.Y.; Choi, H.C.; Lee, Y.B.; Han, K.; Park, C.W. Preparation and in vivo evaluation of lecithin-based microparticles for topical delivery of minoxidil. Arch. Pharm. Res. 2017, 40, 943–951. [Google Scholar] [CrossRef] [PubMed]
- Pereira, M.N.; Schulte, H.L.; Duarte, N.; Lima, E.M.; Sa-Barreto, L.L.; Gratieri, T.; Gelfuso, G.M.; Cunha-Filho, M.S.S. Solid effervescent formulations as new approach for topical minoxidil delivery. Eur. J. Pharm. Sci. 2017, 96, 411–419. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Fang, Y.; Ma, L. The self-crosslinked ufasome of conjugated linoleic acid: Investigation of morphology, bilayer membrane and stability. Colloids Surf. B 2014, 123, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Gebicki, J.M.; Hicks, M. Ufasomes are stable particles surrounded by unsaturated fatty acid membranes. Nature 1973, 243, 232–234. [Google Scholar] [CrossRef] [PubMed]
- Han, S. Molecular dynamics simulation of oleic acid/oleate bilayers: An atomistic model for a ufasome membrane. Chem. Phys. Lipids 2013, 175–176, 79–83. [Google Scholar] [CrossRef] [PubMed]
- Naik, P.V.; Dixit, S.G. Ufasomes as plausible carriers for horizontal gene transfer. J. Dis. Sci. Technol. 2008, 29, 804–808. [Google Scholar] [CrossRef]
- Verma, S.; Bhardwaj, A.; Vij, M.; Bajpai, P.; Goutam, N.; Kumar, L. Oleic acid vesicles: A new approach for topical delivery of antifungal agent. Artif. Cells Nanomed. Biotechnol. 2014, 42, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Zakir, F.; Vaidya, B.; Goyal, A.K.; Malik, B.; Vyas, S.P. Development and characterization of oleic acid vesicles for the topical delivery of fluconazole. Drug Deliv. 2010, 17, 238–248. [Google Scholar] [CrossRef] [PubMed]
- Al Shuwaili, A.H.; Rasool, B.K.; Abdulrasool, A.A. Optimization of elastic transfersomes formulations for transdermal delivery of pentoxifylline. Eur. J. Pharm. Biopharm. 2016, 102, 101–114. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Kumar, P.; Kumar Jindal, D.; Handa, V.; Bilonia, J. Formulation and evaluation of minoxidil gel using acrylamide/sodium acryloyldimethyl taurate copolymer for alopecia areata. Int. J. Pharm. Sci. Drug Res. 2018, 10, 1–6. [Google Scholar] [CrossRef]
- Zheng, W.S.; Fang, X.Q.; Wang, L.L.; Zhang, Y.J. Preparation and quality assessment of itraconazole transfersomes. Int. J. Pharm. 2012, 436, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Grice, J.E.; Ciotti, S.; Weiner, N.; Lockwood, P.; Cross, S.E.; Roberts, M.S. Relative uptake of minoxidil into appendages and stratum corneum and permeation through human skin in vitro. J. Pharm. Sci. 2010, 99, 712–718. [Google Scholar] [CrossRef] [PubMed]
- Teichmann, A.; Jacobi, U.; Ossadnik, M.; Richter, H.; Koch, S.; Sterry, W.; Lademann, J. Differential stripping: Determination of the amount of topically applied substances penetrated into the hair follicles. J. Investig. Dermatol. 2005, 125, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Cistola, D.P.; Hamilton, J.A.; Jackson, D.; Small, D.M. Ionization and phase behavior of fatty acids in water: Application of the gibbs phase rule. Biochemistry 1988, 27, 1881–1888. [Google Scholar] [CrossRef] [PubMed]
- Brisaert, M.; Gabriels, M.; Matthijs, V.; Plaizier-Vercammen, J. Liposomes with tretinoin: A physical and chemical evaluation. J. Pharm. Biomed. Anal. 2001, 26, 909–917. [Google Scholar] [CrossRef]
- Mohammed, A.R.; Weston, N.; Coombes, A.G.A.; Fitzgerald, M.; Perrie, Y. Liposome formulation of poorly water soluble drugs: Optimisation of drug loading and esem analysis of stability. Int. J. Pharm. 2004, 285, 23–34. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Banga, A.K. Intradermal and follicular delivery of adapalene liposomes. Drug Dev. Ind. Pharm. 2016, 42, 871–879. [Google Scholar] [CrossRef] [PubMed]
- Koynova, R.; Caffrey, M. Phases and phase transitions of the phosphatidylcholines. Biochim. Biophys. Acta 1998, 1376, 91–145. [Google Scholar] [CrossRef]
- Elsayed, I.; Abdelbary, A.A.; Elshafeey, A.H. Nanosizing of a poorly soluble drug: Technique optimization, factorial analysis, and pharmacokinetic study in healthy human volunteers. Int. J. Nanomed. 2014, 9, 2943–2953. [Google Scholar] [CrossRef]
- Patel, K.; Munjal, B.; Bansal, A.K. Effect of cyclophosphamide on the solid form of mannitol during lyophilization. Eur. J. Pharm. Sci. 2017, 101, 251–257. [Google Scholar] [CrossRef] [PubMed]
- Ravi, P.R.; Aditya, N.; Kathuria, H.; Malekar, S.; Vats, R. Lipid nanoparticles for oral delivery of raloxifene: Optimization, stability, in vivo evaluation and uptake mechanism. Eur. J. Pharm. Biopharm. 2014, 87, 114–124. [Google Scholar] [CrossRef] [PubMed]
- Fazil, M.; Md, S.; Haque, S.; Kumar, M.; Baboota, S.; Sahni, J.K.; Ali, J. Development and evaluation of rivastigmine loaded chitosan nanoparticles for brain targeting. Eur. J. Pharm. Sci. 2012, 47, 6–15. [Google Scholar] [CrossRef] [PubMed]
- Salama, A.H.; Aburahma, M.H. Ufasomes nano-vesicles-based lyophilized platforms for intranasal delivery of cinnarizine: Preparation, optimization, ex-vivo histopathological safety assessment and mucosal confocal imaging. Pharm. Dev. Technol. 2016, 21, 706–715. [Google Scholar] [CrossRef] [PubMed]
- Mourtas, S.; Fotopoulou, S.; Duraj, S.; Sfika, V.; Tsakiroglou, C.; Antimisiaris, S.G. Liposomal drugs dispersed in hydrogels. Effect of liposome, drug and gel properties on drug release kinetics. Colloids Surf. B Biointerfaces 2007, 55, 212–221. [Google Scholar] [CrossRef] [PubMed]
- Hussain, A.; Samad, A.; Ramzan, M.; Ahsan, M.N.; Ur Rehman, Z.; Ahmad, F.J. Elastic liposome-based gel for topical delivery of 5-fluorouracil: In vitro and in vivo investigation. Drug Deliv. 2016, 23, 1115–1129. [Google Scholar] [CrossRef] [PubMed]
- Williams, A.C.; Barry, B.W. Penetration enhancers. Adv. Drug Deliv. Rev. 2004, 56, 603–618. [Google Scholar] [CrossRef] [PubMed]
- Morimoto, Y.; Wada, Y.; Seki, T.; Sugibayashi, K. In vitro skin permeation of morphine hydrochloride during the finite application of penetration-enhancing system containing water, ethanol and l-menthol. Biol. Pharm. Bull. 2002, 25, 134–136. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Arora, S. Formulation and in vitro evaluation of ufasomes for dermal administration of methotrexate. ISRN Pharm. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Trotta, M.; Peira, E.; Carlotti, M.E.; Gallarate, M. Deformable liposomes for dermal administration of methotrexate. Int. J. Pharm. 2004, 270, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Vanaja, K.; Shobha Rani, R.H.; Sacchidananda, S. Formulation and clinical evaluation of ultradeformable liposomes in the topical treatment of psoriasis. Clin. Res. Regul. Aff. 2008, 25, 41–52. [Google Scholar] [CrossRef]
- Nilsson, H.G. Model of discharge patterns of units in the cochlear nucleus in response to steady state and time-varying sounds. Biol. Cybern. 1975, 20, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Hoppel, M.; Juric, S.; Ettl, H.; Valenta, C. Effect of monoacyl phosphatidylcholine content on the formation of microemulsions and the dermal delivery of flufenamic acid. Int. J. Pharm. 2015, 479, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, K.; Gribbon, P.N.; Jones, M.N. The application of confocal microscopy to the study of liposome adsorption onto bacterial biofilms. J. Liposome Res. 2002, 12, 285–300. [Google Scholar] [CrossRef] [PubMed]
| Code | MXD (mg) | Ratio of (OA:PL90G) | Vesicle size (nm) | PDI | Zeta Potential (mV) | %EE |
|---|---|---|---|---|---|---|
| OAV1 | 20 | 3.0:1.0 | 317 ± 4 | 0.203 ± 0.016 | −13.97 ± 0.451 | 69.08 ± 3.07 |
| OAV2 | 20 | 3.5:1.0 | 377 ± 5 | 0.293 ± 0.088 | −14.70 ± 0.579 | 58.85 ± 3.00 |
| OAV3 | 20 | 3.5:2.0 | 589 ± 6 | 0.331 ± 0.110 | −13.64 ± 0.526 | 71.75 ± 3.11 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumar, P.; Singh, S.K.; Handa, V.; Kathuria, H. Oleic Acid Nanovesicles of Minoxidil for Enhanced Follicular Delivery. Medicines 2018, 5, 103. https://doi.org/10.3390/medicines5030103
Kumar P, Singh SK, Handa V, Kathuria H. Oleic Acid Nanovesicles of Minoxidil for Enhanced Follicular Delivery. Medicines. 2018; 5(3):103. https://doi.org/10.3390/medicines5030103
Chicago/Turabian StyleKumar, Pawan, Shailendra Kumar Singh, Vandana Handa, and Himanshu Kathuria. 2018. "Oleic Acid Nanovesicles of Minoxidil for Enhanced Follicular Delivery" Medicines 5, no. 3: 103. https://doi.org/10.3390/medicines5030103
APA StyleKumar, P., Singh, S. K., Handa, V., & Kathuria, H. (2018). Oleic Acid Nanovesicles of Minoxidil for Enhanced Follicular Delivery. Medicines, 5(3), 103. https://doi.org/10.3390/medicines5030103





