Probiotics in the Treatment of Colorectal Cancer
Abstract
1. Introduction
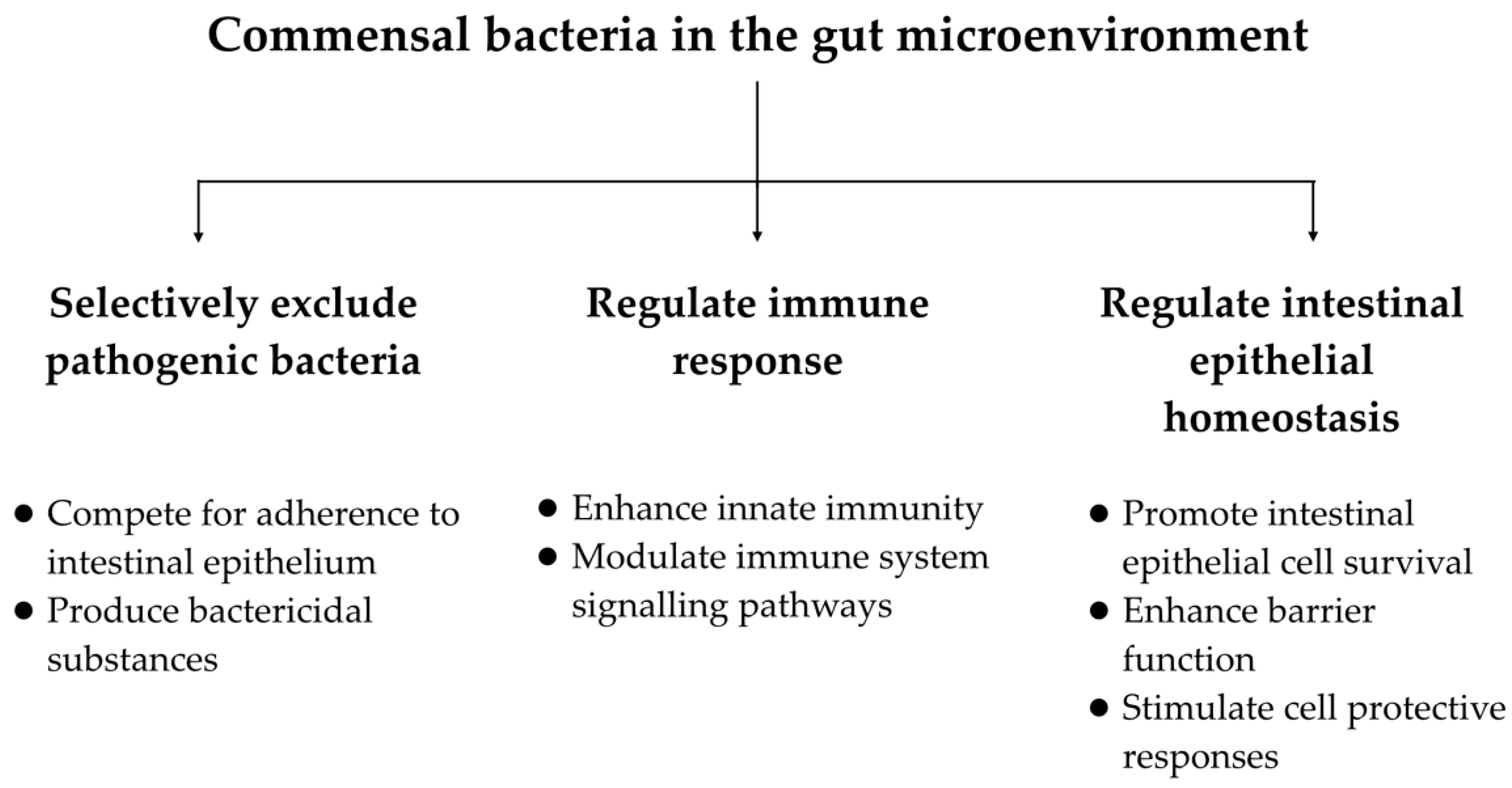
2. Correlation between the Microbiome and Overall Gastrointestinal Health
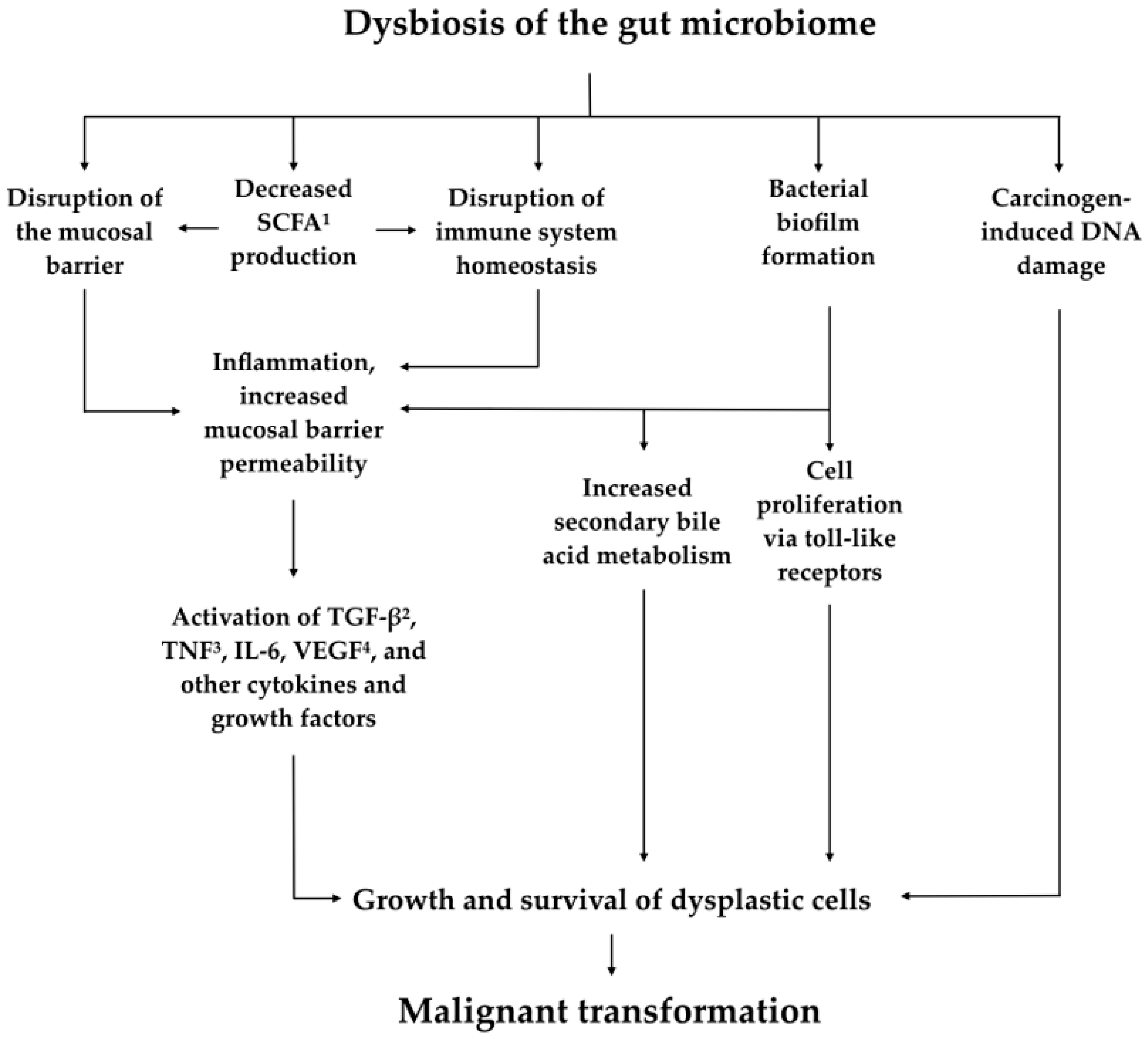
3. Correlation between the Microbiome and the Development of Colorectal Cancer
4. Correlation between the Microbiome and Cancer Treatment Effectiveness
5. Manipulation of the Microbiome as a Component of Cancer Treatment
5.1. Cancer Prevention
5.2. Alleviating Adverse Effects
5.3. Cancer Outcomes
6. Discussion
7. Conclusions
Funding
Conflicts of Interest
References
- Torre, L.A.; Bray, F.; Siegel, R.L.; Ferlay, J.; Lortet-Tieulent, J.; Jemal DVM, A. Global cancer statistics, 2012. Cancer J. Clin. 2015, 65, 87–108. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer Statistics, 2017. Cancer J. Clin. 2017, 67, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Jemal, A.; Ward, E.M.; Johnson, C.J.; Cronin, K.A.; Ma, J.; Ryerson, A.B.; Mariotto, A.; Lake, A.J.; Wilson, R.; Sherman, R.L.; et al. Annual Report to the Nation on the Status of Cancer, 1975–2014, Featuring Survival. J. Natl. Cancer Inst. 2017, 109. [Google Scholar] [CrossRef] [PubMed]
- Edge, S.B.; Byrd, D.R.; Compton, C.C.; Frita, A.G.; Greene, F.L.; Trotti, A. Colon and Rectum. In Cuccurullo, V. AJCC Cancer Staging Handbook: From the AJCC Cancer Staging Manual, 7th ed.; Cuccurullo, V., Mansi, L., Eds.; Springer: New York, NY, USA, 2010; p. 143. [Google Scholar]
- Ursell, L.K.; Metcalf, J.L.; Parfrey, L.W.; Knight, R. Defining the human microbiome. Nutr. Rev. 2012, 70, S38–S44. [Google Scholar] [CrossRef] [PubMed]
- The NIH HMP Working Group; Peterson, J.; Garges, S.; Giovanni, M.; Mclnnes, P.; Wang, L.; Schloss, J.A.; Bonazzi, V.; McEwen, J.E.; Wetterstrand, K.A.; et al. The NIH human microbiome project. Genome Res. 2009, 19, 2317–2323. [Google Scholar] [CrossRef] [PubMed]
- Qin, J.; Li, R.; Raes, J.; Arumugam, M.; Burgdorf, K.S.; Manichanh, C.; Nielsen, T.; Pons, F.; Levenez, F.; Yamada, T.; et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 2010, 464, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Lloyd-Price, J.; Abu-Ali, G.; Huttenhower, C. The healthy human microbiome. Genome Med. 2016, 8, 51. [Google Scholar] [CrossRef] [PubMed]
- Barlow, G.M.; Yu, A.; Mathur, R. Role of the Gut Microbiome in Obesity and Diabetes Mellitus. Nutr. Clin. Pract. 2015, 30, 787–797. [Google Scholar] [CrossRef] [PubMed]
- Ambalam, P.; Raman, M.; Purama, R.K.; Doble, M. Probiotics, prebiotics and colorectal cancer prevention. Best Pract. Res. Clin. Gastroenterol. 2016, 30, 119–131. [Google Scholar] [CrossRef] [PubMed]
- Zackular, J.P.; Baxter, N.T.; Iverson, K.D.; Iverson, K.D.; Sadler, W.D.; Petrosino, J.F.; Chen, G.Y.; Schloss, P.D. The gut microbiome modulates colon tumorigenesis. mBio 2013, 4, e000692-13. [Google Scholar] [CrossRef] [PubMed]
- Raskov, H.; Burcharth, J.; Pommergaard, H.C. Linking Gut Microbiota to Colorectal Cancer. J. Cancer 2017, 8, 3378–3395. [Google Scholar] [CrossRef] [PubMed]
- Aarnoutse, R.; De Vos-Geelen, J.M.P.G.M.; Penders, J.; Boerma, E.G.; Warmerdam, F.A.R.M.; Goorts, B.; Olde Damink, S.W.M.; Soons, Z.; Rensen, S.S.M.; Smidt, M.L. Study protocol on the role of intestinal microbiota in colorectal cancer treatment: A pathway to personalized medicine 2.0. Int. J. Colorectal. Dis. 2017, 32, 1077–1084. [Google Scholar] [CrossRef] [PubMed]
- Iida, N.; Dzutsev, A.; Stewart, C.A.; Smith, L.; Bouladoux, N.; Weingarten, R.A.; Molina, D.A.; Salcedo, R.; Back, T.; Cramer, T.; et al. Commensal bacteria control cancer response to therapy modulating the tumor microenvironment. Science 2013, 342, 967–970. [Google Scholar] [CrossRef] [PubMed]
- Pennisi, E. Biomedicine. Cancer therapies use a little help from microbial friends. Science 2013, 342, 921. [Google Scholar] [CrossRef] [PubMed]
- Vetizou, M.; Pitt, J.M.; Dalliere, R.; Lepage, P.; Waldschmitt, N.; Flament, C.; Rusakiewicz, S.; Routy, B.; Roberti, M.P.; Duong, C.P.M.; et al. Anticancer immunotherapy by CTLA-4 blockade relies on the gut microbiota. Science 2015, 350. [Google Scholar] [CrossRef] [PubMed]
- Sivan, A.; Corrales, L.; Hubert, N. Commensal Bifidobacterium promotes antitumor immunity and facilitates anti-PD-L1 efficacy. Science 2015, 350, 1084–1089. [Google Scholar] [CrossRef] [PubMed]
- Krebs, B. Prebiotic and synbiotic treatment before colorectal surgery–randomised double blind trial. Coll. Antropol. 2016, 40, 35–40. [Google Scholar] [PubMed]
- Della, P.; Sansotta, G.; Donato, V.; Frosina, P.; Messina, G.; De Renzis, C.; Famularo, G. Use of probiotics for prevention of radiation-induced diarrhea. World J. Gastroenterol. 2007, 13, 912–915. [Google Scholar] [CrossRef]
- Mego, M.; Chovanec, J.; Vochyanova-Andrezalova, I.; Konkolovsky, P.; Mikulova, M.; Reckova, M.; Miskovska, V.; Bysticky, B.; Beniak, J.; Medvecova, L.; et al. Prevention of irinotecan induced diarrhea by probiotics: A randomized double blind, placebo controlled pilot study. Complement. Ther. Med. 2015, 23, 356–362. [Google Scholar] [CrossRef] [PubMed]
- Kotzampassi, K.; Stavrou, G.; Damoraki, G.; Georgitsi, M.; Basdanis, G.; Tsaousi, G.; Giamarellos-Bourboulis, E.J. A four-probiotics regimen reduces postoperative complications after colorectal surgery: A randomized, double-blind, placebo-controlled study. World J. Surg. 2015, 39, 2776–2783. [Google Scholar] [CrossRef] [PubMed]
- Turnbaugh, P.J.; Ridaura, V.K.; Faith, J.J.; Turnbaugh, P.J.; Knight, R.; Gorfon, J.I. The effect of diet on the human gut microbiome: A metagenomic analysis in humanized gnotobiotic mice. Sci. Transl. Med. 2009, 11. [Google Scholar] [CrossRef] [PubMed]
- Wasilewski, A.; Zielinska, M.; Storr, M.; Fichna, J. Beneficial effects of probiotics, prebiotics, synbiotics, and psychobiotics in inflammatory bowel disease. Inflamm. Bowel Dis. 2015, 21, 1674–1682. [Google Scholar] [CrossRef] [PubMed]
- Gibson, G.R.; Beatty, E.R.; Wang, X.; Cummings, J.H. Selective stimulation of bifidobacteria in the human colon by oligofructose and inulin. Gastroenterology 1995, 108, 975–982. [Google Scholar] [CrossRef]
- Looijer-van Langen, M.A.C.; Dieleman, L.A. Prebiotics in chronic intestinal inflammation. Inflamm. Bowel Dis. 2009, 15, 454–462. [Google Scholar] [CrossRef] [PubMed]
- Vanderpool, C.; Yan, F.; Polk, D.B. Mechanisms of probiotic action: Implications for therapeutic applications in inflammatory bowel disease. Inflamm. Bowel Dis. 2008, 14, 1585. [Google Scholar] [CrossRef] [PubMed]
- Holubar, S.D.; Cima, R.R.; Sandborn, W.J.; Pardi, D.S. Treatment and prevention of pouchitis after ileal pouch-anal anastomosis for chronic ulcerative colitis. Cochrane Database Syst. Rev. 2010, 6. [Google Scholar] [CrossRef]
- Allen, S.J.; Martinez, E.G.; Gregorio, G.V.; Dans, L.F. Probiotics for treating acute infectious diarrhoea. Cochrane Database Syst. Rev. 2010, 2010. [Google Scholar] [CrossRef] [PubMed]
- Van Niel, C.W.; Feudtner, C.; Garrison, M.M.; Christakis, D.A. Lactobacillus therapy for acute infectious diarrhea in children: A meta-analysis. Pediatrics 2002, 109, 678–684. [Google Scholar] [CrossRef] [PubMed]
- Dimidi, E.; Christodoulides, S.; Fragkos, K.C.; Scott, S.M.; Whelan, K. The effect of probiotics on functional constipation in adults: A systematic review and meta-analysis of randomized control trials. Am. J. Clin. Nutr. 2014, 100, 1075–1084. [Google Scholar] [CrossRef] [PubMed]
- Kasai, C.; Sugimoto, K.; Moritani, I.; Tanaka, J.; Oya, Y.; Inoue, H.; Tamede, M.; Shiraki, K.; Ito, M.; Takei, Y.; et al. Comparison of human gut microbiota in control subjects and patients with colorectal carcinoma in adenoma: Terminal restriction fragment length polymorphism and next-generation sequencing analyses. Oncol. Rep. 2016, 35, 325–333. [Google Scholar] [CrossRef] [PubMed]
- Flemer, B.; Lynch, D.B.; Brown, J.M.R.; Jeffery, I.B.; Ryan, F.J.; Claesson, M.J.; O’Riordain, M.; Shanahan, F.; O’Toole, P.W. Tumour-associated and non-tumour-associated microbiota in colorectal cancer. Gut 2016, 66, 633–643. [Google Scholar] [CrossRef] [PubMed]
- Klampfer, L. Cytokines, inflammation and colon cancer. Curr. Cancer Drug Targets 2011, 11, 451–464. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Konstantinov, S.R.; Smits, R.; Peppelenbosch, M.P. Bacterial biofilms in colorectal cancer initiation and progression. Trends Mol. Med. 2017, 23, 18–30. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Rhee, K.-J.; Albesiano, E.; Rabizadeh, S.; Wu, X.; Yen, H.-R.; Huso, D.L.; Brancati, F.L.; Wick, E.; McAllister, F.; et al. A human colonic commensal promotes tumorigenesis via activation of T helper type 17 T cell responses. Nat. Med. 2009, 15, 1016–1022. [Google Scholar] [CrossRef] [PubMed]
- Kostik, A.D.; Gevers, D.; Pedamallu, C.S.; Michaud, M.; Duke, F.; Earl, A.M.; Ojesina, A.I.; Jung, J.; Bass, A.J.; Tabernero, J.; et al. Genomic analysis identified association of Fusobacterium with colorectal carcinoma. Genome Res. 2012, 22, 292–298. [Google Scholar] [CrossRef] [PubMed]
- Castellarin, M.; Warren, R.L.; Freeman, J.D.; Dreolini, L.; Krzywinki, M.; Strauss, J.; Barnes, R.; Watson, R.; Allen-Vercoe, E.; Moore, R.A.; et al. Fusobacterium nucleatum infection is prevalent in human colorectal carcinoma. Genome Res. 2012, 22, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Shang, F.; Liu, H. Fusobacterium nucleatum and colorectal cancer: A review. World J. Gastrointest. Oncol. 2018, 10, 71–81. [Google Scholar] [CrossRef] [PubMed]
- Ericcson, A.C.; Akter, S.; Hanson, M.M.; Busi, S.B.; Parker, T.W.; Schehr, R.J.; Hankins, M.A.; Ahner, C.E.; Davis, J.W.; Franklin, C.L.; et al. Differential susceptibility to colorectal cancer due to naturally occurring gut microbiota. Oncotarget 2015, 6, 33689–33704. [Google Scholar] [CrossRef] [PubMed]
- An, J.; Ha, E.M. Combination therapy of Lactobacillus plantarum Supernatant and 5-Fluouracil Increases Chemosensitivity in Colorectal Cancer Cells. J. Microbiol. Biotechnol. 2016, 26, 1490–1503. [Google Scholar] [CrossRef] [PubMed]
- Viaud, S.; Saccheri, F.; Mignot, G.; Yamazaki, T.; Daillere, R.; Hannani, D.; Enot, D.P.; Pfirschke, C.; Engblom, C.; Pittet, M.J.; et al. The intestinal microbiota modulates the anticancer immune effects of cyclophosphamide. Science 2013, 342, 971–976. [Google Scholar] [CrossRef] [PubMed]
- Rafter, J.; Bennett, M.; Caderni, G.; Clune, Y.; Hughes, R.; Karlsson, P.C.; Klinder, A.; O’Riordan, M.; O’Sullivan, G.C.; Pool-Zobel, P.; et al. Dietary synbiotics reduce cancer risk factors in polypectomized and colon cancer patients. Am. J. Clin. Nutr. 2007, 85, 488–496. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, H.; Akedo, I.; Otani, T.; Suzuki, T.; Nakamura, T.; Takeyama, I.; Ishiguro, S.; Miyaoka, E.; Sobue, T.; Kakizoe, T. Randomized trial of dietary fiber and Lactobacillus casei administration for prevention of colorectal tumors. Int. J. Cancer 2005, 116, 752–757. [Google Scholar] [CrossRef] [PubMed]
- Pala, V.; Sieri, S.; Berrino, F.; Vineis, P.; Sacerdote, C.; Palli, D.; Masala, G.; Panico, S.; Mattiello, A.; Tumino, R.; et al. Yogurt consumption and risk of colorectal cancer in the Italian European prospective investigation into cancer and nutrition cohort. Int. J. Cancer 2011, 129, 2712–2719. [Google Scholar] [CrossRef] [PubMed]
- Osterlund, P.; Ruotsalainen, T.; Korpela, R.; Ollus, M.; Valta, P.; Kouri, M.; Elomaa, I.; Joensuu, H. Lactobacillus supplementation for diarrhoea related to chemotherapy of colorectal cancer: A randomised study. Br. J. Cancer 2007, 97, 1028–1034. [Google Scholar] [CrossRef] [PubMed]
- Fuccio, L.; Guido, A.; Eusebi, L.H.; Laterza, L.; Grilli, D.; Cennamo, V.; Ceroni, L.; Barbieri, E.; Bazzoil, F. Effects of probiotics for the prevention and treatment of radiation-induced diarrhea. J. Clin. Gastroenterol. 2009, 43, 506–513. [Google Scholar] [CrossRef] [PubMed]
- Aso, Y.; Akaza, H.; Kotake, T.; Tsukamoto, T.; Imai, K.; Naito, S. The BLP Study Group. Preventive effect of a Lactobacillus casei preparation on the recurrence of superficial bladder cancer in a double-blind trial. Eur. Urol. 1995, 27, 104–109. [Google Scholar] [CrossRef] [PubMed]
- Naito, S.; Koga, H.; Yamaguchi, A.; Fujimoto, N.; Hasui, Y.; Kuramoto, H.; Iguch, A. Kyushu University Urological Oncology Group. Prevention of recurrence with epirubicin and Lactobacillus casei after transurethral resection of bladder cancer. J. Urol. 2008, 179, 485–490. [Google Scholar] [CrossRef] [PubMed]
- Chong, E.S.L. A potential role of probiotics in colorectal cancer prevention: Review of possible mechanisms of action. World J. Microbiol. Biotechnol. 2014, 30, 351–374. [Google Scholar] [CrossRef] [PubMed]
- Fooks, L.J.; Gibson, G.R. Probiotics as moderators of gut flora. Br. J. Nutr. 2002, 88, S39–S49. [Google Scholar] [CrossRef] [PubMed]
- Tareb, R.; Bernardeau, M.; Geuguen, M.; Vernoux, J.-P. In vitro characterization of aggregation and adhesion properties of viable and heat-killed forms of two probiotic Lactobacillus strains and interaction with foodborne zoonotic bacteria, especially Campylobacter jejuni. J. Med. Microbiol. 2013, 62, 637–649. [Google Scholar] [CrossRef] [PubMed]
- Verma, A.; Shukla, G. Probiotics Lacobacillus rhamnosus GG, Lactobacillus acidophilus suppressed DMH-induced procarcinogenic fecal enzymes and preneoplastic aberrant crypt foci in early colon carcinogenesis in Sprague Dawley rats. Nutr. Cancer 2013, 65, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Sato, S.; Nagai, H.; Igarashi, Y. Effect of probiotics on serum bile acids in patients with ulcerative colitis. Hepatogastroenterology 2012, 59, 1804–1808. [Google Scholar] [CrossRef] [PubMed]
- Oberreuther-Moschner, D.L.; Jahreis, G.; Rechkemmer, G.; Pool-Zobel, B.L. Dietary intervention with the probiotics Lactobacillus acidophilus 145 and Bifidobacterium longum 913 modulates the potential of human faecal water to induce damage in HT29clone19A cells. Br. J. Nutr. 2004, 91, 925–932. [Google Scholar] [CrossRef] [PubMed]
- Mennigen, R.; Nolte, K.; Rijcken, E.; Utech, M.; Loeffler, B.; Senninger, N.; Bruewer, M. Probiotic mixture VSL#3 protects the epithelial barrier by maintaining tight junction protein expression and preventing apoptosis in a murine model of colitis. Am. J. Physiol. Gastrointest. Liver Physiol. 2009, 296, G1140–G1149. [Google Scholar] [CrossRef] [PubMed]
- Brady, L.J.; Gallaher, D.D.; Busta, F.F. The role of probiotic cultures in the prevention of colon cancer. J. Nutr. 2000, 130, 410S–414S. [Google Scholar] [CrossRef] [PubMed]
- Yeung, C.-Y.; Chan, W.-T.; Jiang, C.-B.; Cheng, M.-L.; Liu, C.-Y.; Chiau, J.-S.C.; Lee, H.-C. Amelioration of chemotherapy-induced intestinal mucositis by orally administered probiotics in a mouse model. PLoS ONE 2015, 10, e0141402. [Google Scholar] [CrossRef] [PubMed]
- Bowen, J.M.; Stringer, A.M.; Gibson, R.J.; Yeoh, A.S.J.; Hannam, S.; Keefe, D.M.K. VSL#3 probiotic treatment reduces chemotherapy-induced diarrhea and weight loss. Cancer Biol. Ther. 2007, 6, 1449–1454. [Google Scholar] [PubMed]
- Salva, S.; Marranzino, G.; Villena, J. Probiotic Lactobacillus strains protect against myelosuppression and immunosuppression in cyclophosphamide-treated mice. Int. Immunopharmacol. 2014, 22, 209–221. [Google Scholar] [CrossRef] [PubMed]
- Packey, C.D.; Ciorba, M.A. Microbial influences on the small intestinal response to radiation injury. Curr. Opin. Gastroenterol. 2010, 26, 88–94. [Google Scholar] [CrossRef] [PubMed]
- Redman, M.G.; Ward, E.J.; Phillips, R.S. The efficacy and safety of probiotics in people with cancer: A systematic review. Ann. Oncol. 2014, 25, 1919–1929. [Google Scholar] [CrossRef] [PubMed]
- Baumler, A.J.; Sperandio, V. Interactions between the microbiota and pathogenic bacteria in the gut. Nature 2016, 535, 85–93. [Google Scholar] [CrossRef] [PubMed]
| Study Name | Study Type | Population | Intervention/Cohort Arms | Summary of Key Results |
|---|---|---|---|---|
| Studies evaluating probiotics and cancer prevention: | ||||
| Rafter 2007 [42] | RCT 1 | Colon cancer (n = 37) & polypectomized (n = 43) patients | SYN1 2 + LGG 3 + BB12 4 vs. placebo | Several CRC 5 biomarkers altered favorably (e.g., decreased genotoxin exposure, IL-2 6, and IFNγ 7) |
| Ishikawa 2005 [43] | RCT | Tumor-free patients with history of ≥2 colorectal tumors removed (n = 398) | Wheat bran vs. Lactobacillus casei vs. both vs. neither | No significant difference in colorectal tumor occurrence rate with wheat bran or L. casei. However, atypia of tumors was lower in the L. casei group. |
| Pala 2011 [44] | Prospective cohort study | EPIC-Italy cohort (n = 45,241) | Yogurt intake by tertile 8 | CRC occurrence was significantly lower in highest vs. lowest tertile of yogurt intake. HR 9 = 0.62 (95% CI 10, 0.46–0.83). |
| Studies evaluating probiotics and alleviating adverse effects of cancer therapy: | ||||
| Mego 2015 [20] | RCT | CRC patients starting treatment with irinotecan-based therapy (n = 46) 11 | Colon Dophilus TM probiotic formula vs. placebo | Reduced incidence in probiotic group of severe diarrhea (0% vs. 17.4%, p = 0.11) and diarrhea overall (39.1% vs. 60.9%, p = 0.11), but not statistically significant. |
| Osterlund 2007 [45] | RCT | Post-resection CRC patients requiring adjuvant chemotherapy (n = 150) | Randomized to 5-FU via Mayo regimen vs. de Gramont regimen, then randomized to LGG vs. no probiotic | Less grade 3–4 diarrhea in patients receiving LGG (22% vs. 37%, p = 0.027) |
| Fuccio 2009 [46] | Meta-analysis | Three RCTs evaluating probiotic supplementation to prevent radiation induced diarrhea (n = 632). One RCT evaluating therapeutic role. | Probiotic supplementation vs. placebo/control | No significant difference in rates of radiation-induced diarrhea between probiotic and control arms in preventative trials (OR 12 0.47, 95% CI 0.13–1.67) or in the single therapeutic trial |
| Kotzampassi 2015 [21] | RCT | Patients undergoing surgery for CRC (n = 168) 13 | Probiotic formulation 14 vs. placebo | Significant decrease in all major post-operative complications in probiotics arm (28.6% vs. 48.8%, p = 0.010, OR 0.42) |
| Krebs 2016 [18] | RCT | Patients undergoing surgery for CRC (n = 73) | Preoperative prebiotics 15 vs. preoperative synbiotics 16 vs. mechanical bowel cleansing | No statistical difference in systemic inflammatory response, postoperative course, or complication rate |
| Studies evaluating probiotics and cancer outcomes: | ||||
| Aso 1995 [47] | RCT | Patients with superficial transitional cell carcinoma of the bladder after transurethral resection (n = 138) | Oral L. casei preparation vs. placebo | Reduced recurrence rate in patients with primary multiple tumors or recurrent single tumors (p = 0.01), but not with recurrent multiple tumors |
| Naito 2008 [48] | RCT | Patients with superficial bladder cancer after transurethral resection and intravesicular epirubicin (n = 202) | Intravesicular epirubicin plus Lactobacillus casei vs. epirubicin alone | Three-year recurrence-free survival rate was significantly higher in the group receiving L. casei (74.6% vs. 59.9%, p = 0.0234), no difference in PFS 17 or OS 18 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hendler, R.; Zhang, Y. Probiotics in the Treatment of Colorectal Cancer. Medicines 2018, 5, 101. https://doi.org/10.3390/medicines5030101
Hendler R, Zhang Y. Probiotics in the Treatment of Colorectal Cancer. Medicines. 2018; 5(3):101. https://doi.org/10.3390/medicines5030101
Chicago/Turabian StyleHendler, Robert, and Yue Zhang. 2018. "Probiotics in the Treatment of Colorectal Cancer" Medicines 5, no. 3: 101. https://doi.org/10.3390/medicines5030101
APA StyleHendler, R., & Zhang, Y. (2018). Probiotics in the Treatment of Colorectal Cancer. Medicines, 5(3), 101. https://doi.org/10.3390/medicines5030101




