Concentrations of Persistent Organic Pollutants in Women’s Serum in the European Arctic Russia
Abstract
1. Introduction
2. Materials and Methods
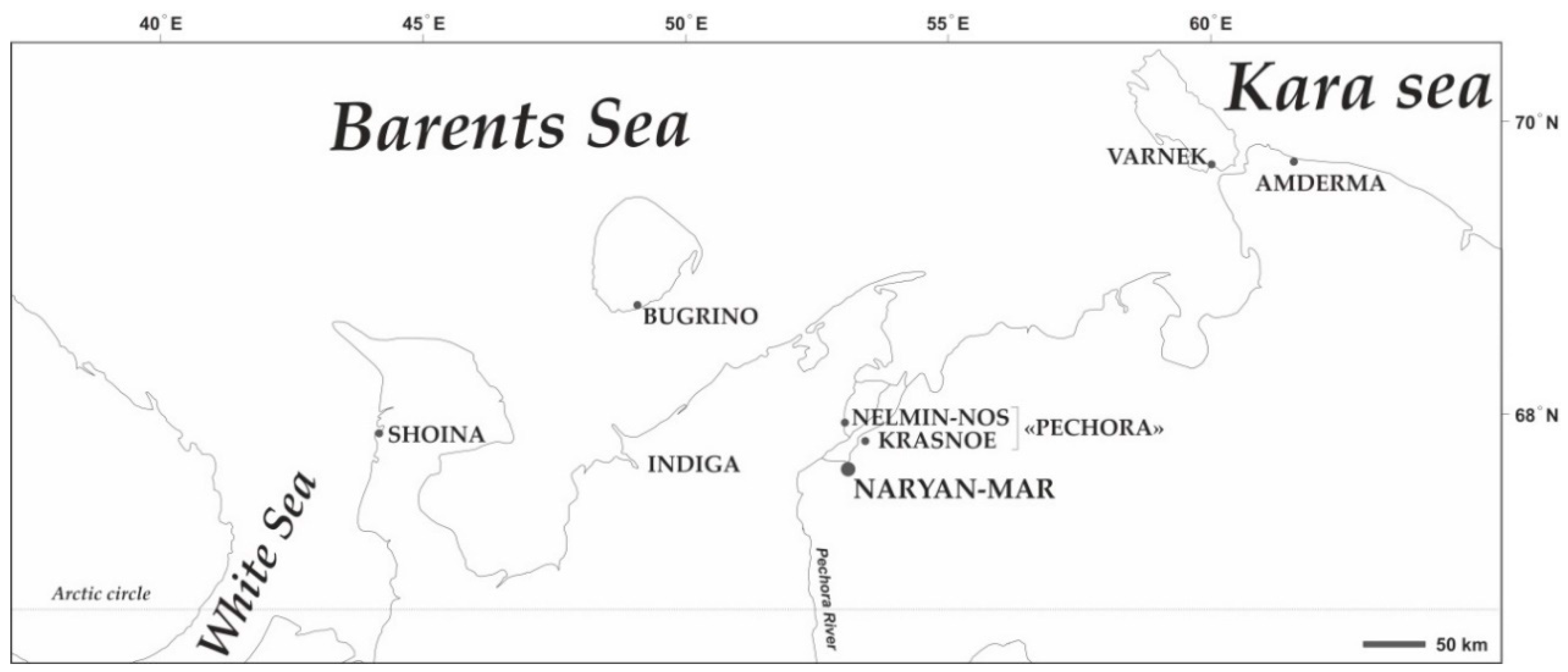
2.1. Study Design and Sampling
2.2. Materials
2.3. Serum Analysis
2.4. Instrumental Analysis
2.5. Quality Assurance, Quality Control and Method Validation
2.6. Determination of Total Serum Lipids
2.7. Statistical Analysis
2.8. Ethical Considerations
3. Results
3.1. Basic Characteristics of the Study Participants
3.2. Serum Concentrations of Lipophilic PCB
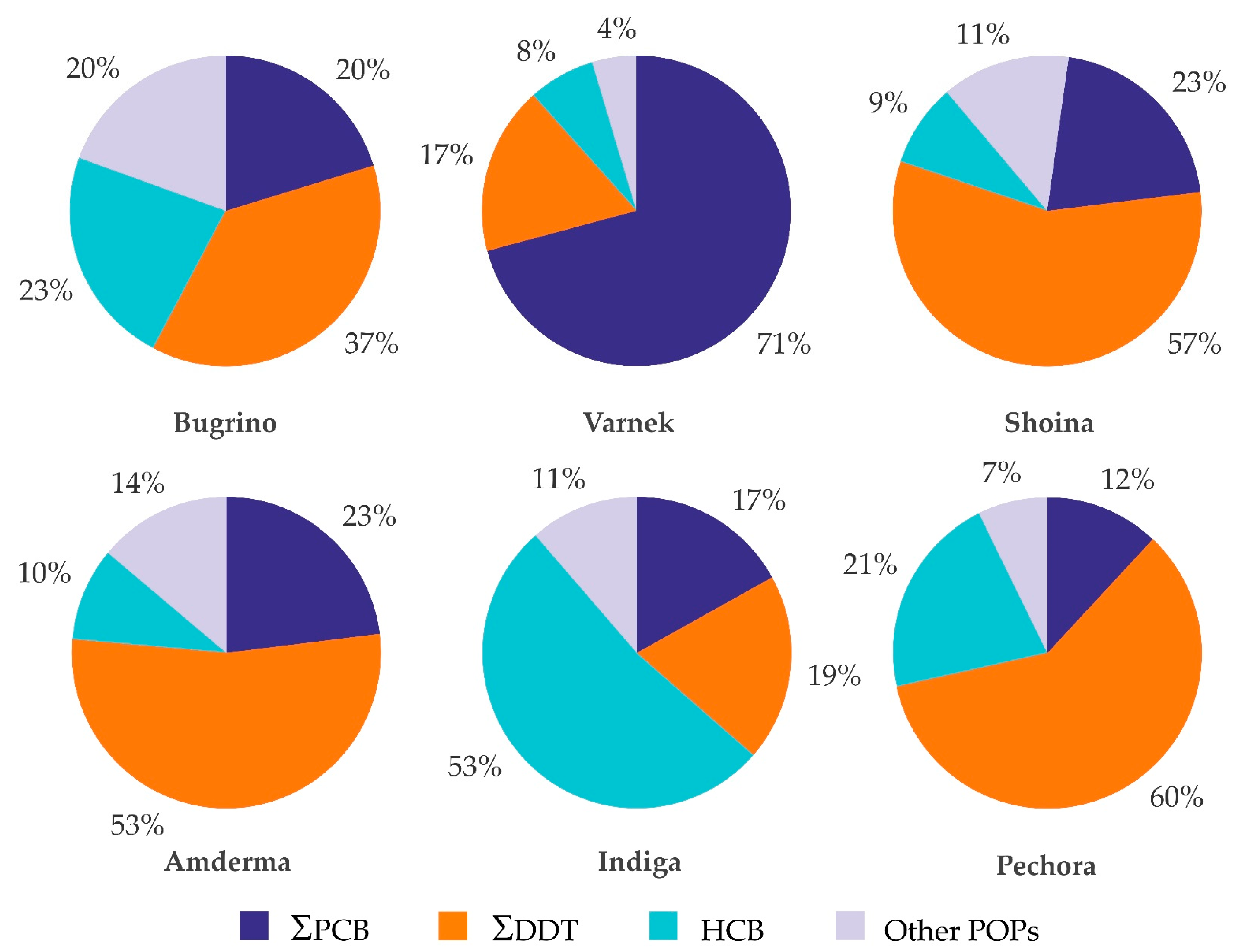
3.2.1. Distribution of POPs across the Study Settlements
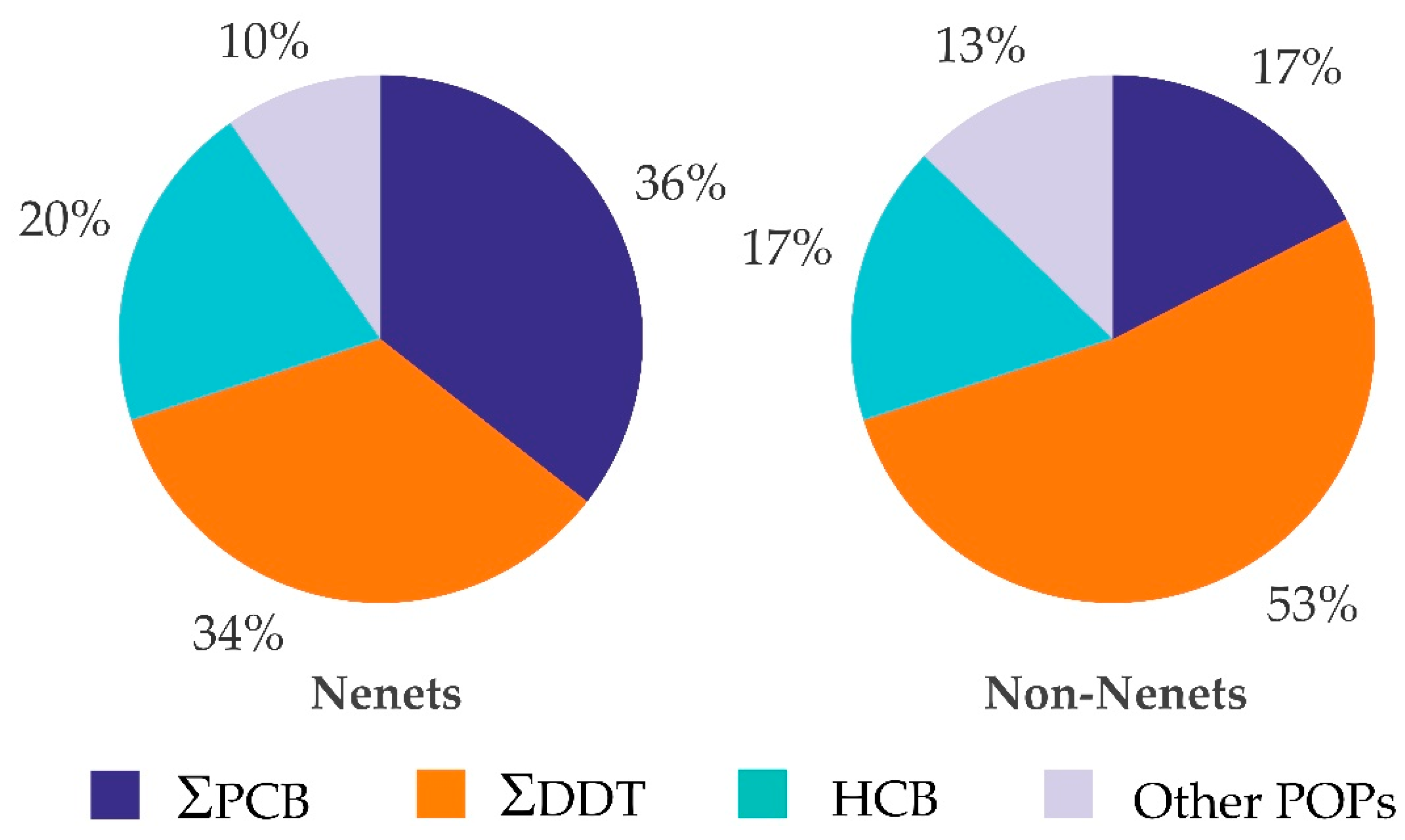
3.2.2. Distribution of POPs between Nenets and Non-Nenets
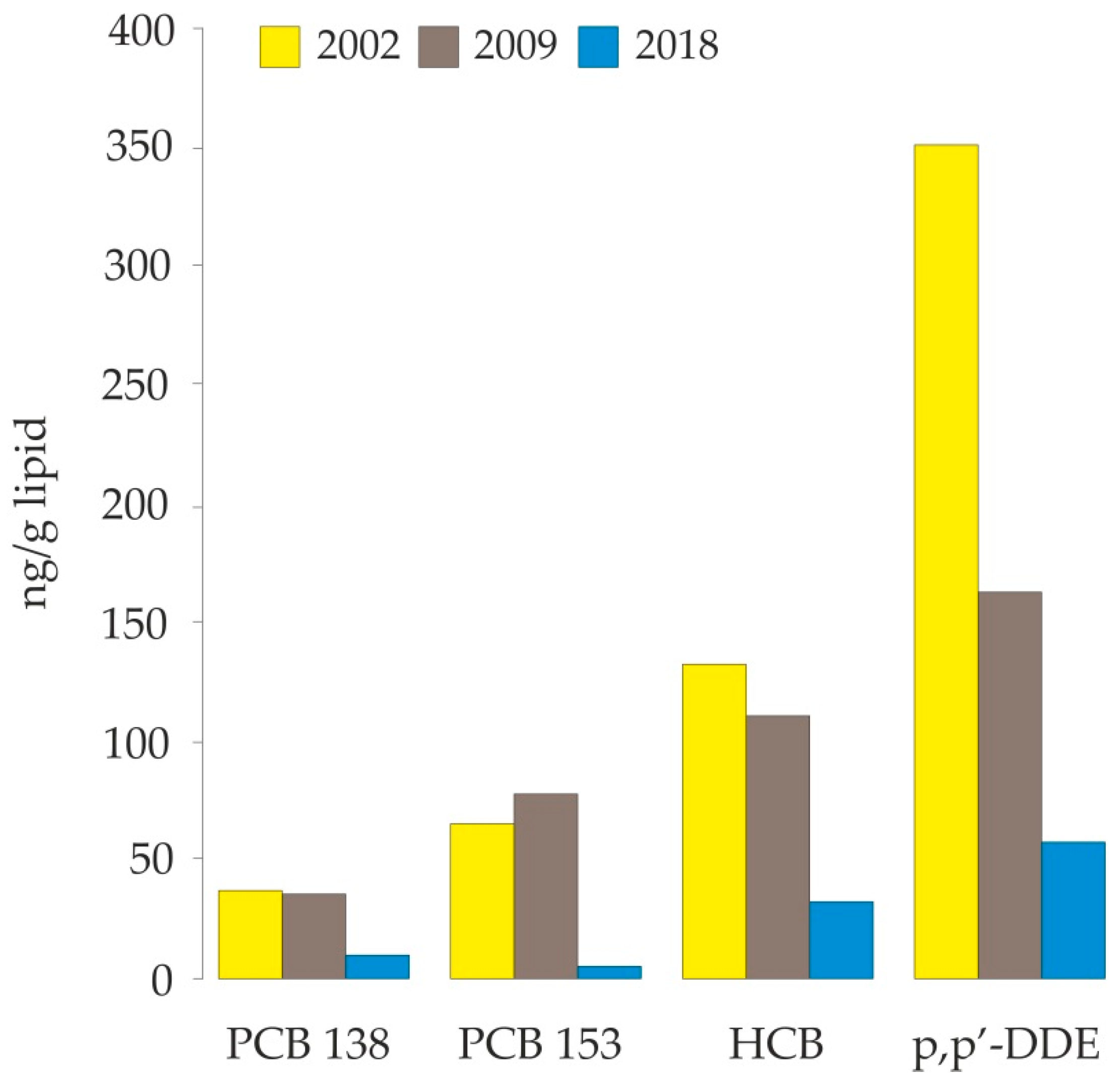
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mouly, T.A.; Toms, L.-M.L. Breast cancer and persistent organic pollutants (excluding DDT): A systematic literature review. Environ. Sci. Pollut. Res. 2016, 23, 22385–22407. [Google Scholar] [CrossRef] [PubMed]
- Stockholm Convention on Persistent Organic Pollutants Was Adopted on 22 May 2001 in Stockholm, Sweden. Available online: http://chm.pops.int/TheConvention/ThePOPs/TheNewPOPs (accessed on 7 December 2020).
- AMAP. Human Health in the Arctic; Arctic Monitoring and Assessment Programme (AMAP): Oslo, Norway, 2015; Available online: https://www.amap.no/documents/doc/amap-assessment-2015-human-health-in-the-arctic/1346 (accessed on 7 December 2020).
- Borlakoglu, J.T.; Haegele, K.D. Comparative aspects on the bioaccumulation, metabolism and toxicity with PCBs. Comp. Biochem. Physiol. Part C Comp. Pharmacol. 1991, 100, 327–338. [Google Scholar] [CrossRef]
- Mrema, E.J.; Rubino, F.M.; Brambilla, G.; Moretto, A.; Tsatsakis, A.M.; Colosio, C. Persistent organochlorinated pesticides and mechanisms of their toxicity. Toxicology 2012, 307, 74–88. [Google Scholar] [CrossRef] [PubMed]
- Gray, J.M.; Rasanayagam, S.; Engel, C.; Rizzo, J. State of the evidence 2017: An update on the connection between breast cancer and the environment. Environ. Health 2017, 16, 94. [Google Scholar] [CrossRef] [PubMed]
- Fiore, M.; Oliveri Conti, G.; Caltabiano, R.; Buffone, A.; Zuccarello, P.; Cormaci, L.; Cannizzaro, M.A.; Ferrante, M. Role of Emerging Environmental Risk Factors in Thyroid Cancer: A Brief Review. Int. J. Environ. Res. Public Health 2019, 16, 1185. [Google Scholar] [CrossRef]
- Lee, D.H.; Porta, M.; Jacobs, D.R.; Vandenberg, L.N. Chlorinated persistent organic pollutants, obesity, and type 2 diabetes. Endocr. Rev. 2014, 35, 557–601. [Google Scholar] [CrossRef]
- Lind, P.M.; Salihovic, S.; Stubleski, J.; Kärrman, A.; Lind, L. Association of Exposure to Persistent Organic Pollutants with Mortality Risk: An Analysis of Data from the Prospective Investigation of Vasculature in Uppsala Seniors (PIVUS) Study. JAMA Netw. Open 2019, 2, 193070. [Google Scholar] [CrossRef]
- Yegambaram, M.; Manivannan, B.; Beach, T.G.; Halden, R.U. Role of environmental contaminants in the etiology of Alzheimer’s disease: A review. Curr. Alzheimer Res. 2015, 12, 116–146. [Google Scholar] [CrossRef]
- Rosen, E.M.; Muñoz, M.I.; McElrath, T.; Cantonwine, D.E.; Ferguson, K.K. Environmental contaminants and preeclampsia: A systematic literature review. J. Toxicol. Environ. Health B Crit. Rev. 2018, 21, 291–319. [Google Scholar] [CrossRef]
- Zheng, T.; Zhang, J.; Sommer, K.; Bassig, B.A.; Zhang, X.; Braun, J.; Xu, S.; Boyle, P.; Zhang, B.; Shi, K.; et al. Effects of Environmental Exposures on Fetal and Childhood Growth Trajectories. Ann. Glob. Health 2016, 82, 41–99. [Google Scholar] [CrossRef]
- Bae, J.; Kim, S.; Barr, D.B.; Buck Louis, G.M. Maternal and paternal serum concentrations of persistent organic pollutants and the secondary sex ratio: A population-based preconception cohort study. Environ. Res. 2018, 161, 9–16. [Google Scholar] [CrossRef] [PubMed]
- AMAP. Adaptation Actions for a Changing Arctic: Perspectives from the Barents Area; Arctic Monitoring and Assessment Programme (AMAP): Oslo, Norway, 2017. [Google Scholar]
- AMAP. Persistent Toxic Substances, Food Security and Indigenous Peoples of the Russian North; Arctic Monitoring and Assessment Programme (AMAP): Oslo, Norway, 2004. [Google Scholar]
- AMAP. Human Health in the Arctic; Arctic Monitoring and Assessment Programme (AMAP): Oslo, Norway, 2009; Available online: https://www.amap.no/documents/doc/amap-assessment-2009-human-health-in-the-arctic/98 (accessed on 7 December 2020).
- Bjerregaard, P.; Dewailly, E.; Ayotte, P.; Pars, T.; Ferron, L.; Mulvad, G. Exposure of Inuit in Greenland to organochlorines through the marine diet. J. Toxicol. Environ. Health 2001, 62, 69–81. [Google Scholar] [CrossRef] [PubMed]
- Bonefeld-Jorgensen, E.C.; Ghisari, M.; Wielsoe, M.; Bjerregaard-Olesen, C.; Kjeldsen, L.S.; Long, M. Biomonitoring and hormone-disrupting effect biomarkers of persistent organic pollutants in vitro and ex vivo. Basic Clin. Pharmacol. Toxicol. 2014, 115, 118–128. [Google Scholar] [CrossRef] [PubMed]
- Abass, K.; Emelyanova, A.; Rautio, A. Temporal trends of contaminants in Arctic human populations. Environ. Sci. Pollut. Res. 2018, 25, 28834–28850. [Google Scholar] [CrossRef]
- Emelyanova, A. Population Projections of the Arctic by Levels of Education; Working Paper WP-17-022; The International Institute for Applied Systems Analysis: Laxenburg, Austria, 2017. [Google Scholar]
- Dudarev, A.A.; Chupakhin, V.; Odland, J.O.; Reiersen, L.; Chashchin, V. A follow-up study of blood levels of persistent toxic substances (PTS) among indigenous peoples of the coastal Chukotka, Russia, 2001–2007. Int. J. Circumpolar Health 2010, 7, 263–268. [Google Scholar]
- Bravo, N.; Grimalt, J.O.; Chashchin, M.; Chashchin, V.P.; Odland, J.-Ø. Drivers of maternal accumulation of organohalogen pollutants in Arctic areas (Chukotka, Russia) and 4,4′-DDT effects on the newborns. Environ. Int. 2019, 124, 541–552. [Google Scholar] [CrossRef]
- Sorokina, T.Y. A national system of biological monitoring in the Russian Arctic as a tool for the implementation of the Stockholm Convention. Int. Environ. Agreem. Politics Law Econ. 2019, 19, 341–355. [Google Scholar] [CrossRef]
- Lakhmanov, D.; Varakina, Y.; Aksenov, A.; Sorokina, T.; Sobolev, N.; Kotsur, D.; Plakhina, E.; Chashchin, V.; Thomassen, Y. Persistent organic pollutants (POPs) in fish consumed by the indigenous peoples from Nenets Autonomous Okrug. Environments 2020, 7, 3. [Google Scholar] [CrossRef]
- Sobolev, N.; Ellingsen Dag, G.; Belova, N.; Aksenov, A.; Sorokina, T.; Trofimova, A.; Varakina, Y.; Kotsur, D.; Grjibovski, A.M.; Chashchin, V.; et al. Essential and non-essential elements in biological samples of inhabitants residing in Nenets Autonomous Okrug of the Russian Arctic. Environ. Int. 2021. submitted. [Google Scholar]
- Grimalt, J.O.; Howsam, M.; Carrizo, D.; Otero, R.; de Marchi, M.R.R.; Vizcaino, E. Integrated analysis of halogenated organic pollutants in sub-millilitre volumes of venous and umbilical cord blood sera. Anal. Bioanal. Chem. 2010, 396, 2265–2272. [Google Scholar] [CrossRef]
- Bernert, J.T.; Turner, W.E.; Patterson, D.G., Jr.; Needham, L.L. Calculation of serum “total lipid” concentrations for the adjustment of persistent organohalogen toxicant measurements in human samples. Chemosphere 2007, 68, 824–831. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Park, J.; Kim, H.-J.; Lee, J.J.; Choi, G.; Choi, S.; Kim, S.; Kim, S.Y.; Moon, H.B.; Kim, S.; et al. Association between Several Persistent Organic Pollutants and Thyroid Hormone Levels in Cord Blood Serum and Bloodspot of the Newborn Infants of Korea. PLoS ONE 2015, 10, e0125213. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bjerregaard, P.; Holm, A.L.; Olesen, I.; Schnor, O.; Niclasen, B. Ivaaq-The Greenland Inuit Child Cohort a Preliminary; National Institute of Public Health: Copenhagen, Denmark, 2007.
- Eik Anda, E.; Nieboer, E.; Dudarev, A.A.; Sandanger, T.M.; Odland, J.O. Intra- and ntercompartmental associations between levels of organochlorines inmaternal plasma, cord plasma and breast milk, and lead and cadmium in whole blood, for indigenous peoples of Chukotka, Russia. J. Environ. Monit. 2007, 9, 884–893. [Google Scholar] [CrossRef] [PubMed]
- Hjermitslev, M.H.; Long, M.; Wielsøe, M.; Bonefeld-Jørgensen, E.C. Persistent organic pollutants in Greenlandic pregnant women and indices of foetal growth: The ACCEPT study. Sci. Total Environ. 2019, 698, 134118. [Google Scholar] [CrossRef]
- Long, M.; Knudsen, A.-K.S.; Pedersen, H.S.; Bonefeld-Jørgensen, E.C. Food intake and serum persistent organic pollutants in the Greenlandic pregnant women: The ACCEPT sub-study. Sci. Total Environ. 2015, 529, 198–212. [Google Scholar] [CrossRef]
- Wielsøe, M.; Tarantini, L.; Bollati, V.; Long, M.; Bonefeld-Jørgensen, E.C. DNA methylation level in blood and relations to breast cancer, risk factors and environmental exposure in Greenlandic Inuit women. Basic Clin. Pharmacol. Toxicol. 2020, 127, 338–350. [Google Scholar] [CrossRef]
- Berg, V.; Nost, T.H.; Huber, S.; Rylander, C.; Hansen, S.; Veyhe, A.S.; Fuskevåg, O.M.; Øyvind Odland, J.; Sandanger, T.M. Maternal serum concentrations of per- and polyfluoroalkyl substances and their predictors in years with reduced production and use. Environ. Int. 2014, 69, 58–66. [Google Scholar] [CrossRef]
- Araujo-León, J.A.; Mena-Rejón, G.J.; Canché-Pool, E.B.; Ruiz-Piña, H.A. Biomonitoring Organochlorine Pesticides in Didelphis virginiana from Yucatan, Mexico by GC-ECD. Bull. Environ. Contam. Toxicol. 2019, 102, 836–842. [Google Scholar] [CrossRef]
- Rylander, C.M.; Sandanger, T.; Petrenya, N.; Konoplev, A.; Bojko, E.; Øyvind Odland, J. Indications of decreasing human PTS concentrations in North West Russia. Glob. Health Action 2011, 4, 8427. [Google Scholar] [CrossRef][Green Version]
- AMAP. Biological Effects of Contaminants on Arctic Wildlife and Fish; Arctic Monitoring and Assessment Programme (AMAP): Tromsø, Norway, 2018. [Google Scholar]
| Analyte | LOD | LOQ | Spiked Level | Accuracy (%) | |||
|---|---|---|---|---|---|---|---|
| 0.2 ng/mL | 1 ng/mL | ||||||
| ng/mL | Recovery (%) | RSD (%) | Recovery (%) | RSD (%) | |||
| 1,2,3,5-Tetrachlorobenzene | 0.01 | 0.04 | 106 | 9 | 102 | 8% | 112 |
| 1,2,4,5-Tetrachlorobenzene | 0.01 | 0.04 | 106 | 9 | 102 | 8% | 112 |
| α-HCH | 0.06 | 0.21 | 126 | 8 | 110 | 7% | 96 |
| HCB | 0.03 | 0.11 | 126 | 12 | 97 | 7% | 102 |
| β-HCH | 0.02 | 0.07 | 118 | 13 | 98 | 8 | 93 |
| γ-HCH | 0.04 | 0.13 | 121 | 5 | 110 | 7 | 101 |
| PCB28 | 0.004 | 0.02 | 119 | 10 | 105 | 5 | 90 |
| Heptachlor | 0.09 | 0.29 | 118 | 12 | 115 | 9 | 120 |
| PCB52 | 0.03 | 0.11 | 105 | 5 | 99 | 6 | 90 |
| Aldrin | 0.09 | 0.24 | 75 | 20 | 86 | 8 | 120 |
| o,p′-DDE | 0.02 | 0.07 | 123 | 11 | 90 | 9 | 112 |
| trans-Chlordane | 0.1 | 0.32 | 117 | 14 | 102 | 7 | 86 |
| PCB101 | 0.05 | 0.15 | 102 | 8 | 94 | 5 | 92 |
| cis-Chlordane | 0.11 | 0.38 | 109 | 17 | 111 | 8 | 120 |
| trans-Nonachlor | 0.09 | 0.29 | 105 | 14 | 86 | 7 | 101 |
| p,p′-DDE | 0.11 | 0.36 | 108 | 10 | 114 | 15 | 98 |
| o,p′-DDD | 0.01 | 0.03 | 129 | 10 | 111 | 5 | 104 |
| PCB123 | 0.05 | 0.16 | 116 | 7 | 98 | 5 | 95 |
| PCB118 | 0.08 | 0.27 | 124 | 11 | 109 | 7 | 103 |
| p,p′-DDD | 0.05 | 0.16 | 113 | 7 | 94 | 8 | 100 |
| cis-Nonachlor | 0.04 | 0.13 | 124 | 5 | 101 | 8 | 111 |
| PCB153 | 0.02 | 0.08 | 126 | 15 | 106 | 6 | 90 |
| PCB105 | 0.07 | 0.22 | 117 | 9 | 97 | 4 | 96 |
| PCB138 | 0.09 | 0.31 | 121 | 13 | 108 | 5 | 90 |
| PCB183 | 0.05 | 0.18 | 89 | 10 | 88 | 7 | 90 |
| PCB128 | 0.05 | 0.16 | 109 | 7 | 105 | 18 | 93 |
| PCB180 | 0.01 | 0.04 | 106 | 11 | 97 | 7 | 103 |
| Mirex | 0.06 | 0.2 | 77 | 13 | 85 | 6 | 104 |
| Characteristic | Measures | Bugrino | Varnek | Amderma | Shoina | Indiga | Pechora | p-Value 1 | Nenets | Non-Nenets |
|---|---|---|---|---|---|---|---|---|---|---|
| Age (years) | n (%) | 24(11.8) | 11(5.4) | 33(16.2) | 32(15.7) | 35(17.1) | 69(33.8) | 113 (55.4) | 91 (44.6) | |
| Mean (SD) | 41.9 (11.3) | 46.6 (15.5) | 49.1(12.5) | 49.7 (16.2) | 59.9 (15.1) | 49.2 (12.8) | <0.001 | 46.6 (13.9) | 54.5 (14.1) | |
| Median Range | 43 20–59 | 52 22–65 | 53 28–69 | 53 24–87 | 61 19–86 | 49 24–71 | 46 20–79 | 55 19–87 | ||
| Proportion of Nenets | % | 22 (91.6) | 11 (100) | 11 (33.3) | 11 (34.4) | 15 (42.8) | 43 (62.3) | |||
| BMI (kg/m2) 2 | Mean (SD) | 27.8 (6.9) | 25.7 (6.2) | 30.0 (7.0) | 27.1 (4.9) | 26.8 (3.5) | 27.5 (5.3) | 0.426 | 27.1 (5.8) | 28.4 (5.4) |
| Median Range | 26.4 17.1–41.4 | 24.4 16.4–39.6 | 29.3 20.9–46.0 | 26.9 16.2–42.7 | 26.7 19.9–35.1 | 26.9 17.0–42.8 | 26.6 16.2–42.9 | 27.3 19.2–46.0 | ||
| Cholesterol (mmol/L) | Mean (SD) | 5.8 (1.1) | 5.0 (1.3) | 5.7 (1.2) | 5.7 (1.4) | 5.3 (0.9) | 5.8 (1.4) | 0.140 | 5.5 (1.1) | 5.9 (1.2) |
| Median Range | 5.4 4.3–8.1 | 5.0 2.8–7.4 | 5.5 2.7–7.7 | 5.6 3.7–10.6 | 5.2 3.1–7.2 | 5.9 3.9–8.0 | 5.4 2.8–8.0 | 5.6 3.8–10.6 | ||
| Triglycerides (mmol/L) | Mean (SD) | 1.4 (0.8) | 2.1 (1.6) | 1.9 (1.1) | 1.5 (0.7) | 1.4 (0.8) | 1.4 (0.7) | 0.267 | 1.5 (0.9) | 1.6 (0.8) |
| Median Range | 1.0 0.4–3.6 | 1.7 0.4–5.5 | 1.8 0.3–4.3 | 1.4 0.2–2.9 | 1.4 0.5–4.1 | 1.4 0.4–2.9 | 1.4 0.2–5.5 | 1.5 0.3–4.3 | ||
| Total Lipid (mg/dL) | Mean (SD) | 618 (116) | 583 (138) | 638 (127) | 623 (141) | 581(106) | 628 (113) | 0.374 | 601 (122) | 637 (119) |
| Median Range | 576 453–854 | 565 371–802 | 647 315–856 | 620 408–1079 | 559 353–801 | 640 425–852 | 588 315–856 | 629 444–1079 | ||
| Smoking | Yes (%) | 4 (16.7) | 3 (27.3) | 10 (30.3) | 11 (34.4) | 4 (11.4) | 19 (27.5) | 28(24.8) | 23(25.3) |
| Analyte | Measures | Bugrino | Varnek | Shoina | Amderma | Indiga | Pechora | Nenets | Non-Nenets |
|---|---|---|---|---|---|---|---|---|---|
| (n = 24) | (n = 11) | (n = 32) | (n = 33) | (n = 35) | (n = 69) | (n = 113) | (n = 91) | ||
| PCBs (ng/g lipid) | |||||||||
| PCB118 | GM (95% CI) | 8.2 (6.7–10.0) | 12.2 (5.5–24.5) | 11.0 (8.2–14.9) | 18.2 (14.9–22.2) | 11.0 (8.2–14.9) | 9.0 (8.2–11.0) | 9.0 (8.2–11.0) | 13.5 (12.2–16.4) |
| Median | 7.3 | 7.3 | 7.9 | 18.3 | 7.5 | 7.8 | 7.5 | 12.0 | |
| Range | <LOD–34.6 | <LOD–183 | <LOD–76.9 | <LOD–48.2 | <LOD–128 | <LOD–57.6 | <LOD–183 | <LOD–118 | |
| PCB138 | GM (95% CI) | 12.2 (8.2–16.4) | 12.2 (8.2–16.4) | 90.0 (30.0–244) | 18.2 (13.5–22.2) | 16.4 (12.2–22.2) | 10.0 (8.2–13.5) | 13.5 (11.0–16.4) | 13.5 (11.0–14.9) |
| Median | 8.6 | 143 | 17.7 | 16.0 | 8.2 | 7.9 | 9.0 | 9.8 | |
| Range | <LOD–116 | <LOD–811 | <LOD–114 | <LOD–67.7 | <LOD–144 | <LOD–96.9 | <LOD–811 | <LOD–114 | |
| PCB153 | GM (95% CI) | 14.9 (8.2–27.1) | 200 (44.7–897) | 18.2 (12.2–30.0) | 24.5 (16.4–36.6) | 8.2 (5.0–13.5) | 8.2 (6.0–12.2) | 12.2 (9.0–18.2) | 16.4 (13.5–22.2) |
| Median | 19.5 | 558 | 23.7 | 31.1 | 10.8 | 9.5 | 13.8 | ||
| Range | <LOD–260 | <LOD–2413 | <LOD–171 | <LOD–196 | <LOD–246 | <LOD–164 | <LOD–2413 | <LOD–171 | |
| PCB180 | GM (95% CI) | 9.0 (6.7–13.5) | 121 (33.1–445) | 7.4 (5.0–11.0) | 11.0 (6.7–16.4) | 2.2 (1.5–3.7) | <LOD | 4.5 (3.0–6.0) | 3.0 (2.5–4.1) |
| Median | 7.3 | 298 | 8.6 | 13.2 | 1.1 | 5.0 | 2.7 | ||
| Range | <LOD–270 | <LOD–1006 | LOD–74.6 | LOD–96.6 | LOD–77.8 | <LOD–1006 | <LOD–61.7 | ||
| PCB183 | GM (95% CI) | <LOD | 13.5 (7.4–27.1) | <LOD | <LOD | <LOD | <LOD | 5.0 (4.5–5.0) | 4.1 (3.7–4.1) |
| Median | 17.7 | 4.3 | 4.0 | ||||||
| Range | <LOD–48.5 | <LOD–48.6 | <LOD–5.6 | ||||||
| OC Pesticides (ng/g lipid) | |||||||||
| o,p′-DDE | GM (95% CI) | 16.4 (11.0–27.1) | 24.5 (16.4–36.6) | 12.2 (9.0–18.2) | 5.0 (3.0–8.2) | 3.3 (2.5–4.5) | 33.1 (24.5–44.7) | 16.4 (12.2–20.1) | 9.0 (7.4–12.2) |
| Median | 22.4 | 29.0 | 15.1 | 2.0 | 2.1 | 41.6 | 23.1 | 14.5 | |
| Range | <LOD–77.0 | 8.7–67.0 | <LOD–243 | <LOD–275 | <LOD–19.7 | <LOD–596 | <LOD–597 | <LOD–144 | |
| p,p′-DDE | GM (95% CI) | 73.7 (49.4–121) | 99.5 (30.0–330) | 110 (66.7–164) | 148 (109–200) | 40.4 (27.1–54.6) | 60.3 (49.4–81.5) | 11.0 (9.0–14.9) | 109.6 (81.5–134) |
| Median | 60.5 | 155 | 121 | 142 | 37.9 | 65.3 | 59.9 | 125.3 | |
| Range | <LOD–733 | <LOD–1239 | <LOD–1318 | 28.0–1313 | LOD–336 | <LOD–2577 | <LOD–1313 | <LOD–2577 | |
| p,p′-DDD | GM (95% CI) | <LOD | <LOD | <LOD | <LOD | <LOD | 54.6 (49.4–66.7) | 54.6 (40.4–66.7) | 9.0 (7.4–12.2) |
| Median | 35.7 | 5.3 | 4.5 | ||||||
| Range | <LOD–194 | <LOD–194 | <LOD–152 | ||||||
| HCB | GM (95% CI) | 49.4 (27.1–90.0) | 73.7 (27.1–181) | 20.1 (13.5–30.0) | 33.1 (24.5–44.7) | 99.5 (54.6–164) | 54.6 (44.7–73.7) | 12.2 (10.0–16.4) | 44.7 (33.1–54.6) |
| Median | 68.7 | 77.9 | 26.2 | 39.3 | 150 | 71.2 | 74.3 | 42.1 | |
| Range | <LOD–255 | <LOD–363 | <LOD–121 | <LOD–114 | <LOD–767 | <LOD–335 | <LOD–767 | <LOD–723 | |
| β-HCH | GM (95% CI) | 10.0 (5.5–16.4) | 16.4 (7.4–36.6) | 22.2 (13.5–33.1) | 33.1 (22.2–44.7) | 20.1 (14.9–30.0) | 14.9 (11.4–20.1) | 9.0 (8.2–11.0) | 27.1 (22.2–36.6) |
| Median | 9.1 | 17.8 | 25.0 | 41.5 | 19.0 | 16.5 | 13.0 | 32.0 | |
| Range | <LOD–136 | <LOD–81.5 | <LOD–275 | 7.3–232 | <LOD–123 | <LOD–313 | <LOD–34.0 | <LOD–55.7 | |
| Aldrin | GM (95% CI) | 20.1 (11.0–36.6) | 12.2 (5.5–27.1) | <LOD | 8.2 (6.7–10.0) | <LOD | <LOD | 5.0 (4.5–5.5) | 8.2 (7.4–9.0) |
| Median | 8.2 | 8.2 | 7.3 | 7.8 | 7.3 | ||||
| Range | <LOD–351 | <LOD–352 | <LOD–50.5 | <LOD–352 | <LOD–103 | ||||
| Mirex | GM (95% CI) | <LOD | 5.5 (4.1–6.7) | <LOD | <LOD | 5.5 (4.1–6.7) | <LOD | 5.0 (4.5–5.5) | 4.5 (4.1–5.0) |
| Median | 5.5 | 5.2 | 5.0 | 4.7 | |||||
| Range | <LOD–10.0 | <LOD–111 | <LOD–111 | <LOD–6.6 | |||||
| Location (year) | p,p′-DDE | PCB 118 | PCB 138 | PCB 153 | HCB | β-HCH | Reference |
|---|---|---|---|---|---|---|---|
| Arctic Russia | |||||||
| NAO (2018) | 73.7 | 11.0 | 13.5 | 14.9 | 49.4 | 18.2 | Present study |
| Nelmin-Nos (2018) | 59.5 | 9.0 | 9.0 | 6.3 | 33.8 | 35.7 | |
| Nelmin-Nos (2002–2003) | 246 | 48.0 | 46.0 | 98.0 | 135 | [32] | |
| Chukotka (2001–2002) | 308 | 97.0 | [33] | ||||
| Chukotka (2015) | 120 | 10.0 | 17.0 | 31.0 | 35.0 | 35.0 | [22] |
| Other Arctic locations | |||||||
| Greenland (2010–2013) | 130 | 9.4 | 29.5 | 60.5 | 24.5 | 3.8 | [29] |
| Greenland (2010–2015) | 120 | 8.4 | 26.0 | 53.0 | 26.0 | 3.6 | [30] |
| Greenland (1999–2005) | 238 | 104 | 152 | 37.8 | [31] | ||
| Norway (2004) | 38.7 | 24.8 | [37] | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Varakina, Y.; Lahmanov, D.; Aksenov, A.; Trofimova, A.; Korobitsyna, R.; Belova, N.; Sobolev, N.; Kotsur, D.; Sorokina, T.; Grjibovski, A.M.; et al. Concentrations of Persistent Organic Pollutants in Women’s Serum in the European Arctic Russia. Toxics 2021, 9, 6. https://doi.org/10.3390/toxics9010006
Varakina Y, Lahmanov D, Aksenov A, Trofimova A, Korobitsyna R, Belova N, Sobolev N, Kotsur D, Sorokina T, Grjibovski AM, et al. Concentrations of Persistent Organic Pollutants in Women’s Serum in the European Arctic Russia. Toxics. 2021; 9(1):6. https://doi.org/10.3390/toxics9010006
Chicago/Turabian StyleVarakina, Yulia, Dmitry Lahmanov, Andrey Aksenov, Anna Trofimova, Rimma Korobitsyna, Natalia Belova, Nikita Sobolev, Dmitry Kotsur, Tatiana Sorokina, Andrej M. Grjibovski, and et al. 2021. "Concentrations of Persistent Organic Pollutants in Women’s Serum in the European Arctic Russia" Toxics 9, no. 1: 6. https://doi.org/10.3390/toxics9010006
APA StyleVarakina, Y., Lahmanov, D., Aksenov, A., Trofimova, A., Korobitsyna, R., Belova, N., Sobolev, N., Kotsur, D., Sorokina, T., Grjibovski, A. M., Chashchin, V., & Thomassen, Y. (2021). Concentrations of Persistent Organic Pollutants in Women’s Serum in the European Arctic Russia. Toxics, 9(1), 6. https://doi.org/10.3390/toxics9010006





