Cadmium and Lead Exposure, Nephrotoxicity, and Mortality
Abstract
1. Introduction
2. Health Risk Assessment of Chronic Exposure to Cadmium and Lead
2.1. The Critical Target of Toxicity
2.2. Tolerable Intake Levels
2.3. Urinary Cd Threshold Level
3. Exposure Sources and Dietary Intake Estimates
3.1. Environmental Sources of Cadmium and Lead
3.2. Total Diet Studies and Dietary Intake Estimates
3.2.1. Estimated Cadmium Intake Levels in Various Populations
3.2.2. Estimated Lead Intake Levels in Various Populations
3.3. Absorption of Cadmium and Lead: An Overview
3.4. The Kinetics of Cadmium and Lead in the Human Body
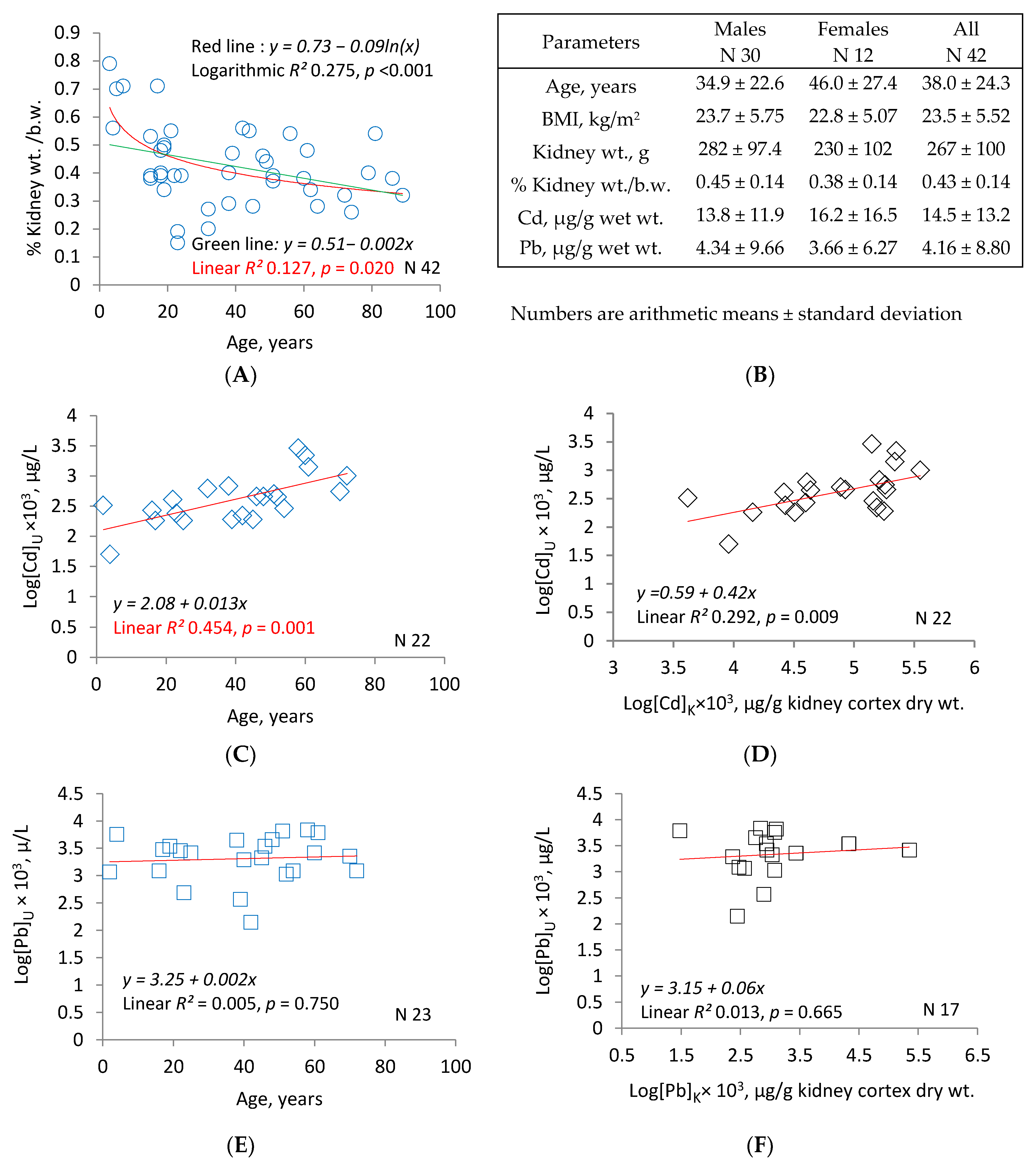
3.5. Cadmium and Lead Accumulation in Kidneys and Urinary Excretion: Australian Experience
4. Dosimetry and Nephrotoxicity Assessment
4.1. Cadmium Dosimetry
4.1.1. Blood Cadmium Versus Urinary Cadmium
4.1.2. Cadmium in Erythrocytes Versus Blood Plasma (Serum)
4.2. Lead Dosimetry
4.2.1. Blood Lead Versus Bone Lead
4.2.2. Plasma (Serum) Lead Versus Urinary Lead
4.3. Assessment of Cadmium Nephrotoxicity
4.3.1. Release of Intracellular Proteins into the Filtrate
N-Acetyl-β-D-Glucosaminidase
Kidney Injury Molecule 1
4.3.2. Excretion of Cadmium
4.3.3. Cadmium Toxicity and the Glomerular Filtration Rate (GFR)
4.3.4. Impaired Reabsorption of Small Filterable Proteins
β2-Microglobulin
Retinol-Binding Protein 4
4.3.5. Normalization of Excretion Rates to Creatinine Excretion or Creatinine Clearance
4.3.6. A Pathophysiologic Synopsis of Cadmium Nephropathy
4.3.7. Assessment of Cadmium Nephrotoxicity: Summary
5. Environmental Exposure to Cd and Pb, Toxic Kidney Burden, CKD, and Other Common Ailments
5.1. The Increased Risk of CKD Associated with Cadmium and Lead Exposure
5.1.1. U.S. Population
5.1.2. Swedish Population
5.1.3. Thai Population
5.1.4. Chinese Population
5.1.5. Korean and Belgian Populations
5.2. Environmental Exposures and Mortality from All Causes
5.2.1. Cadmium and Mortality in the U.S.
5.2.2. Cadmium and Mortality in Sweden and Australia
5.2.3. Cadmium and Mortality in Japan
5.2.4. Lead and Mortality in the U.S., Korea and China
6. Summary and Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| Ca | Calcium |
| Cd | Cadmium |
| Cu | Copper |
| Pb | Lead |
| Zn | Zn |
| MT | Metallothionein |
| PC | Phytochelatin |
| CdMT | Cadmium-metallothionein complex |
| CdPC | Cadmium-phytochelatin complex |
| GSH | Glutathione |
| δ-ALAD | Delta-aminolevulinic acid dehydratase |
| δ-ALA | Delta-aminolevulinic acid |
| AST | Aspartate aminotransferase |
| ALT | Alanine aminotransferase |
| β2MG | Beta2-microglobulin |
| KIM1 | Kidney injury molecule 1 |
| NAG | N-acetyl-β-D-glucosaminidase |
| RBP | Retinol-binding protein |
| GFR | Glomerular filtration rate, units of volume/time |
| eGFR | Estimated glomerular filtration rate, units of mL/min/1.73 m2 |
| CKD-EPI | Chronic kidney disease epidemiology collaboration |
| JECFA | The Joint Expert Committee on Food Additives and Contaminants of the Food and Agriculture Organization and the World Health Organization of the United Nations |
| PTWI | Provisional tolerable weekly intake |
| TMI | Tolerable monthly intake |
| TDS | Total diet study |
| [x]u | Urinary concentration of x. |
| [x]p | Plasma concentration of x. |
| [x]b | Blood concentration of x. |
| [x]k | Kidney content of x. |
| Ccr | Creatinine clearance, units of volume/time |
| Vu | Urine flow rate, units of volume/time |
| Ex/Ccr | Excretion rate of x per volume of filtrate, units of mass/volume, where x = Cd, NAG, or β2MG |
| FEβ2MG | Fractional excretion of β2MG, % |
| FRβ2MG | Fractional reabsorption of β2MG, % |
References
- Satarug, S.; Vesey, D.A.; Gobe, G.C. Health risk assessment of dietary cadmium intake: Do current guidelines indicate how much is safe? Environ. Health Perspect. 2017, 125, 284–288. [Google Scholar] [CrossRef]
- Satarug, S.; Vesey, D.A.; Gobe, G.C. Current health risk assessment practice for dietary cadmium: Data from different countries. Food Chem. Toxicol. 2017, 106, 430–445. [Google Scholar] [CrossRef] [PubMed]
- Shefa, S.T.; Héroux, P. Both physiology and epidemiology support zero tolerable blood lead levels. Toxicol. Lett. 2017, 280, 232–237. [Google Scholar] [CrossRef] [PubMed]
- Daley, G.M.; Pretorius, C.J.; Ungerer, J.P. Lead toxicity: An Australian perspective. Clin. Biochem. Rev. 2018, 39, 61–98. [Google Scholar] [PubMed]
- World Health Organization (WHO). Preventing Disease through Healthy Environments: Ten Chemicals of Major Public Health Concern; Public Environment WHO: Geneva, Switzerland. Available online: https://www.who.int/ipcs/features/10chemicals_en.pdf?ua=1 (accessed on 12 August 2020).
- Satarug, S.; Haswell-Elkins, M.R.; Moore, M.R. Safe levels of cadmium intake to prevent renal toxicity in human subjects. Br. J. Nutr. 2000, 84, 791–802. [Google Scholar] [CrossRef] [PubMed]
- Satarug, S.; Baker, J.R.; Urbenjapol, S.; Haswell-Elkins, M.; Reilly, P.E.; Williams, D.J.; Moore, M.R. A global perspective on cadmium pollution and toxicity in non-occupationally exposed population. Toxicol. Lett. 2003, 137, 65–83. [Google Scholar] [CrossRef]
- Satarug, S. Dietary cadmium intake and its effects on kidneys. Toxics 2018, 6, 15. [Google Scholar] [CrossRef]
- Satarug, S. Long-term exposure to cadmium in food and cigarette smoke, liver effects and hepatocellular carcinoma. Curr. Drug Metab. 2012, 13, 257–271. [Google Scholar] [CrossRef]
- Satarug, S.; Moore, M.R. Emerging roles of cadmium and heme oxygenase in type-2 diabetes and cancer susceptibility. Tohoku J. Exp. Med. 2012, 228, 267–288. [Google Scholar] [CrossRef]
- Gibb, H.J.; Barchowsky, A.; Bellinger, D.; Bolger, P.M.; Carrington, C.; Havelaar, A.H.; Oberoi, S.; Zang, Y.; O’Leary, K.; Devleesschauwer, B. Estimates of the 2015 global and regional disease burden from four foodborne metals-arsenic, cadmium, lead and methylmercury. Environ. Res. 2019, 174, 188–194. [Google Scholar] [CrossRef]
- Satarug, S.; Gobe, G.C.; Ujjin, P.; Vesey, D.A. A comparison of the nephrotoxicity of low doses of cadmium and lead. Toxics 2020, 8, 18. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Ding, N.; Tucker, K.L.; Weisskopf, M.G.; Sparrow, D.; Hu, H.; Park, S.K. A Western diet pattern is associated with higher concentrations of blood and bone lead among middle-aged and elderly men. J. Nutr. 2017, 147, 1374–1383. [Google Scholar] [CrossRef] [PubMed]
- Ding, N.; Wang, X.; Tucker, K.L.; Weisskopf, M.G.; Sparrow, D.; Hu, H.; Park, S.K. Dietary patterns, bone lead and incident coronary heart disease among middle-aged to elderly men. Environ. Res. 2019, 168, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Shi, Z.; Taylor, A.W.; Riley., M.; Byles., J.; Liu, J.; Noakes, M. Association between dietary patterns, cadmium intake and chronic kidney disease among adults. Clin. Nutr. 2018, 37, 276–284. [Google Scholar] [CrossRef]
- Shi, Z.; Zhen, S.; Orsini, N.; Zhou, Y.; Zhou, Y.; Liu, J.; Taylor, A.W. Association between dietary lead intake and 10-year mortality among Chinese adults. Environ. Sci. Pollut. Res. 2017, 24, 12273–12280. [Google Scholar] [CrossRef]
- Gobe, G.; Crane, D. Mitochondria, reactive oxygen species and cadmium toxicity in the kidney. Toxicol. Lett. 2010, 198, 49–55. [Google Scholar] [CrossRef]
- Nair, A.R.; Lee, W.K.; Smeets, K.; Swennen, Q.; Sanchez, A.; Thévenod, F.; Cuypers, A. Glutathione and mitochondria determine acute defense responses and adaptive processes in cadmium-induced oxidative stress and toxicity of the kidney. Arch. Toxicol. 2015, 89, 2273–2289. [Google Scholar] [CrossRef]
- Matović, V.; Buha, A.; Ðukić-Ćosić, D.; Bulat, Z. Insight into the oxidative stress induced by lead and/or cadmium in blood, liver and kidneys. Food Chem. Toxicol. 2015, 78, 130–140. [Google Scholar] [CrossRef]
- Satarug, S.; Vesey, D.A.; Gobe, G.C. Kidney cadmium toxicity, diabetes and high blood pressure: The perfect storm. Tohoku J. Exp. Med. 2017, 241, 65–87. [Google Scholar] [CrossRef]
- Garza-Lombó, C.; Posadas, Y.; Quintanar, L.; Gonsebatt, M.E.; Franco, R. Neurotoxicity linked to dysfunctional metal ion homeostasis and xenobiotic metal exposure: Redox signaling and oxidative stress. Antioxid. Redox Signal. 2018, 28, 1669–1703. [Google Scholar] [CrossRef]
- Valko, M.; Jomova, K.; Rhodes, C.J.; Kuča, K.; Musílek, K. Redox- and non-redox-metal-induced formation of free radicals and their role in human disease. Arch. Toxicol. 2016, 90, 1–37. [Google Scholar] [CrossRef] [PubMed]
- Moulis, J.M.; Bourguinon, J.; Catty, P. Chapter 23 Cadmium. In RSC Metallobiology Series No. 2, Binding, Transport. and Storage of Metal. Ions in Biological Cells; Wolfgang, M., Anthony, W., Eds.; The Royal Society of Chemistry: London, UK, 2014; pp. 695–746. [Google Scholar]
- Cangelosi, V.; Pecoraro, V. Chapter 28 Lead. In RSC Metallobiology Series No. 2, Binding, Transport. and Storage of Metal. Ions in Biological Cells; Wolfgang, M., Anthony, W., Eds.; The Royal Society of Chemistry: London, UK, 2014; pp. 843–882. [Google Scholar]
- Sanders, T.; Liu, Y.; Buchner, V.; Tchounwou, P.B. Neurotoxic effects and biomarkers of lead exposure: A review. Rev. Environ. Health 2009, 24, 15–45. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, M.C.; Shami Shah, A.; DeSilva, S.; Gleaton, A.; Su, A.; Goundie, B.; Croteau, M.L.; Stevenson, M.J.; Wilcox, D.E.; Austin, R.N. Thermodynamics of Pb(ii) and Zn(ii) binding to MT-3, a neurologically important metallothionein. Metallomics 2016, 8, 605–617. [Google Scholar] [CrossRef] [PubMed]
- Satarug, S.; Baker, J.R.; Reilly, P.E.; Esumi, H.; Moore, M.R. Evidence for a synergistic interaction between cadmium and endotoxin toxicity and for nitric oxide and cadmium displacement of metals in the kidney. Nitric Oxide 2000, 4, 431–440. [Google Scholar] [CrossRef]
- Satarug, S.; Baker, J.R.; Reilly, P.E.; Moore, M.R.; Williams, D.J. Changes in zinc and copper homeostasis in human livers and kidneys associated with exposure to environmental cadmium. Hum. Exp. Toxicol. 2001, 20, 205–213. [Google Scholar] [CrossRef]
- Satarug, S.; Nishijo, M.; Ujjin, P.; Moore, M.R. Chronic exposure to low-level cadmium induced zinc-copper dysregulation. J. Trace Elem. Med. Biol. 2018, 46, 32–38. [Google Scholar] [CrossRef]
- Prozialeck, W.C.; Lamar, P.C.; Edwards, J.R. Effects of sub-chronic Cd exposure on levels of copper, selenium, zinc, iron and other essential metals in rat renal cortex. Toxicol. Rep. 2016, 3, 740–746. [Google Scholar] [CrossRef][Green Version]
- Thevenod, F. Nephrotoxicity and the proximal tubule. Insights from cadmium. Nephron Physiol. 2003, 93, 87–93. [Google Scholar] [CrossRef]
- Moulis, J.M. Cellular mechanisms of cadmium toxicity related to the homeostasis of essential metals. Biometals 2010, 23, 877–896. [Google Scholar] [CrossRef]
- Nzengue, Y.; Candéias, S.M.; Sauvaigo, S.; Douki, T.; Favier, A.; Rachidi, W.; Guiraud, P. The toxicity redox mechanisms of cadmium alone or together with copper and zinc homeostasis alteration: Its redox biomarkers. J. Trace Elem. Med. Biol. 2011, 25, 171–180. [Google Scholar] [CrossRef]
- Nzengue, Y.; Steiman, R.; Rachidi, W.; Favier, A.; Guiraud, P. Oxidative stress induced by cadmium in the C6 cell line: Role of copper and zinc. Biol. Trace Elem. Res. 2012, 146, 410–419. [Google Scholar] [CrossRef] [PubMed]
- Eom, S.Y.; Yim, D.H.; Huang, M.; Park, C.H.; Kim, G.B.; Yu, S.D.; Choi, B.S.; Park, J.D.; Kim, Y.D.; Kim, H. Copper-zinc imbalance induces kidney tubule damage and oxidative stress in a population exposed to chronic environmental cadmium. Int. Arch. Occup. Environ. Health 2020, 93, 337–344. [Google Scholar] [CrossRef] [PubMed]
- Rubino, F.M. Toxicity of glutathione-binding metals: A review of targets and mechanisms. Toxics 2015, 3, 20–62. [Google Scholar] [CrossRef] [PubMed]
- Phillips, J.D. Heme biosynthesis and the porphyrias. Mol. Genet. Metab. 2019, 128, 164–177. [Google Scholar] [CrossRef] [PubMed]
- Tobwala, S.; Wang, H.-J.; Carey, J.W.; Banks, W.A.; Ercal, N. Effects of lead and cadmium on brain endothelial cell survival, monolayer permeability, and crucial oxidative stress markers in an in vitro model of the blood-brain barrier. Toxics 2014, 2, 258–275. [Google Scholar] [CrossRef]
- Wang, W.; Duan, B.; Xu, H.; Xu, L.; Xu, T.L. Calcium-permeable acid-sensing ion channel is a molecular target of the neurotoxic metal ion lead. J. Biol. Chem. 2006, 281, 2497–2505. [Google Scholar] [CrossRef] [PubMed]
- FAO/WHO. Evaluation of Certain Food Additives and Contaminants (Forty-First Report of the Joint FAO/WHO Expert Committee on Food Additives); WHO Technical Report Series No. 837; World Health Organization: Geneva, Switzerland, 1993. [Google Scholar]
- Food and Agriculture Organization of the United Nations (FAO); World Health Organization (WHO). Summary and Conclusions. In Proceedings of the Joint FAO/WHO Expert Committee on Food Additives Seventy-Third Meeting, Geneva, Switzerland, 8–17 June 2010; Available online: http://www.who.int/foodsafety/publications/chem/summary73.pdf (accessed on 12 August 2020).
- Flannery, B.M.; Dolan, L.C.; Hoffman-Pennesi, D.; Gavelek, A.; Jones, O.E.; Kanwal, R.; Wolpert, B.; Gensheimer, K.; Dennis, S.; Fitzpatrick, S.U.S. Food and Drug Administration’s interim reference levels for dietary lead exposure in children and women of childbearing age. Regul. Toxicol. Pharmacol. 2020, 110, 104516. [Google Scholar] [CrossRef]
- Dolan, L.C.; Flannery, B.M.; Hoffman-Pennesi, D.; Gavelek, A.; Jones, O.E.; Kanwal, R.; Wolpert, B.; Gensheimer, K.; Dennis, S.; Fitzpatrick, S. A review of the evidence to support interim reference level for dietary lead exposure in adults. Regul. Toxicol. Pharmacol. 2020, 111, 104579. [Google Scholar] [CrossRef] [PubMed]
- Ferraro, P.M.; Costanzi, S.; Naticchia, A.; Sturniolo, A.; Gambaro, G. Low level exposure to cadmium increases the risk of chronic kidney disease: Analysis of the NHANES 1999–2006. BMC Public Health 2010, 10, 304. [Google Scholar] [CrossRef]
- Lin, Y.S.; Ho, W.C.; Caffrey, J.L.; Sonawane, B. Low serum zinc is associated with elevated risk of cadmium nephrotoxicity. Environ. Res. 2014, 134, 33–38. [Google Scholar] [CrossRef]
- Madrigal, J.M.; Ricardo, A.C.; Persky, V.; Turyk, M. Associations between blood cadmium concentration and kidney function in the U.S. population: Impact of sex, diabetes and hypertension. Environ. Res. 2018, 169, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Crinnion, W.J. The CDC fourth national report on human exposure to environmental chemicals: What it tells us about our toxic burden and how it assists environmental medicine physicians. Altern. Med. Rev. 2010, 15, 101–108. [Google Scholar] [PubMed]
- Levey, A.S.; Stevens, L.A.; Schmid, C.H.; Zhang, Y.; Castro, A.F., III; Feldman, H.I.; Kusek, J.W.; Eggers, P.; Van Lente, F.; Greene, T.; et al. A new equation to estimate glomerular filtration rate. Ann. Intern. Med. 2009, 150, 604–612. [Google Scholar] [CrossRef] [PubMed]
- Levey, A.S.; Inker, L.A.; Coresh, J. GFR estimation: From physiology to public health. Am. J. Kidney Dis. 2014, 63, 820–834. [Google Scholar] [CrossRef] [PubMed]
- Levey, A.S.; Becker, C.; Inker, L.A. Glomerular filtration rate and albuminuria for detection and staging of acute and chronic kidney disease in adults: A systematic review. JAMA 2015, 313, 837–846. [Google Scholar] [CrossRef] [PubMed]
- Satarug, S.; Ruangyuttikarn, W.; Nishijo, M.; Ruiz, P. Urinary cadmium threshold to prevent kidney disease development. Toxics 2018, 6, 26. [Google Scholar]
- Satarug, S.; Boonprasert, K.; Gobe, G.C.; Ruenweerayut, R.; Johnson, D.W.; Na-Bangchang, K.; Vesey, D.A. Chronic exposure to cadmium is associated with a marked reduction in glomerular filtration rate. Clin. Kidney J. 2018, 12, 468–475. [Google Scholar] [CrossRef]
- Satarug, S.; Vesey, D.A.; Nishijo, M.; Ruangyuttikarnm, W.; Gobe, G.C. The inverse association of glomerular function and urinary β2-MG excretion and its implications for cadmium health risk assessment. Environ. Res. 2019, 173, 40–47. [Google Scholar] [CrossRef]
- Satarug, S.; Vesey, D.A.; Ruangyuttikarn, W.; Nishijo, M.; Gobe, G.C.; Phelps, K.R. The source and pathophysiologic significance of excreted cadmium. Toxics 2019, 7, 55. [Google Scholar] [CrossRef]
- Järup, L. Hazards of heavy metal contamination. Br. Med. Bull. 2003, 68, 167–182. [Google Scholar] [CrossRef]
- Wu, S.; Deng, F.; Hao, Y.; Shima, M.; Wang, X.; Zheng, C.; Wei, H.; Lv, H.; Lu, X.; Huang, J.; et al. Chemical constituents of fine particulate air pollution and pulmonary function in healthy adults: The Healthy Volunteer Natural Relocation study. J. Hazard. Mater. 2013, 260, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Jung, M.S.; Kim, J.Y.; Lee, H.S.; Lee, C.G.; Song, H.S. Air pollution and urinary N-acetyl-β-glucosaminidase levels in residents living near a cement plant. Ann. Occup. Environ. Med. 2016, 28, 52. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Lu, Y.; Li, Y.; Zhao, H.; Wang, X.; Shen, Y.; Kuang, X. Correlation between environmental low-dose cadmium exposure and early kidney damage: A comparative study in an industrial zone vs. a living quarter in Shanghai, China. Environ.Toxicol. Pharmacol. 2020, 79, 103381. [Google Scholar] [CrossRef] [PubMed]
- Repić, A.; Bulat, P.; Antonijević, B.; Antunović, M.; Džudović, J.; Buha, A.; Bulat, Z. The influence of smoking habits on cadmium and lead blood levels in the Serbian adult people. Environ. Sci. Pollut. Res. Int. 2020, 27, 751–760. [Google Scholar] [CrossRef]
- Dumkova, J.; Vrlikova, L.; Vecera, Z.; Putnova, B.; Docekal, B.; Mikuska, P.; Fictum, P.; Hampl, A.; Buchtova, M. Inhaled cadmium oxide nanoparticles: Their in vivo fate and effect on target organs. Int. J. Mol. Sci. 2016, 17, 874. [Google Scholar] [CrossRef]
- Dumková, J.; Smutná, T.; Vrlíková, L.; Le Coustumer, P.; Večeřa, Z.; Dočekal, B.; Mikuška, P.; Čapka, L.; Fictum, P.; Hampl, A.; et al. Sub-chronic inhalation of lead oxide nanoparticles revealed their broad distribution and tissue-specific subcellular localization in target organs. Part. Fibre Toxicol. 2017, 14, 55. [Google Scholar] [CrossRef]
- Tulinska, J.; Masanova, V.; Liskova, A.; Mikusova, M.L.; Rollerova, E.; Krivosikova, Z.; Stefikova, K.; Uhnakova, I.; Ursinyova, M.; Babickova, J.; et al. Six-week inhalation of CdO nanoparticles in mice: The effects on immune response, oxidative stress, antioxidative defense, fibrotic response, and bones. Food Chem. Toxicol. 2020, 136, 110954. [Google Scholar] [CrossRef]
- Sutunkova, M.P.; Solovyeva, S.N.; Chernyshov, I.N.; Klinova, S.V.; Gurvich, V.B.; Shur, V.Y.; Shishkina, E.V.; Zubarev, I.V.; Privalova, L.I.; Katsnelson, B.A. Manifestation of systemic toxicity in rats after a short-time inhalation of lead oxide nanoparticles. Int. J. Mol. Sci. 2020, 21, 690. [Google Scholar] [CrossRef]
- Zahran, S.; McElmurry, S.P.; Sadler, R.C. Four phases of the Flint qater crisis: Evidence from blood lead levels in children. Environ. Res. 2017, 157, 160–172. [Google Scholar] [CrossRef]
- Roy, S.; Tang, M.; Edwards, M.A. Lead release to potable water during the Flint, Michigan water crisis as revealed by routine biosolids monitoring data. Water Res. 2019, 160, 475–483. [Google Scholar] [CrossRef]
- Bandara, J.M.; Wijewardena, H.V.; Liyanege, J.; Upul, M.A.; Bandara, J.M. Chronic renal failure in Sri Lanka caused by elevated dietary cadmium: Trojan horse of the green revolution. Toxicol. Lett. 2010, 198, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Kader, M.; Lamb, D.T.; Mahbub, K.R.; Megharaj, M.; Naidu, R. Predicting plant uptake and toxicity of lead (Pb) in long-term contaminated soils from derived transfer functions. Environ. Sci. Pollut. Res. Int. 2016, 23, 15460–15470. [Google Scholar] [CrossRef] [PubMed]
- Lamb, D.T.; Kader, M.; Ming, H.; Wang, L.; Abbasi, S.; Megharaj, M.; Naidu, R. Predicting plant uptake of cadmium: Validated with long-term contaminated soils. Ecotoxicology 2016, 25, 1563–1574. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, J.M.; Hill, J.; Phillips, C.J. The accumulation of potentially-toxic metals by grazing ruminants. Proc. Nutr. Soc. 2003, 62, 267–277. [Google Scholar] [CrossRef]
- Bischoff, K.; Hillebrandt, J.; Erb, H.N.; Thompson, B.; Johns, S. Comparison of blood and tissue lead concentrations from cattle with known lead exposure. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2016, 33, 1563–1569. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. CDC Response to Advisory Committee on Childhood Lead Poisoning Prevention Recommendations in “Low Level Lead Exposure Harms Children: A Renewed Call of Primary Prevention”. 2012. Available online: http://www.cdc.gov/nceh/lead/ACCLPP/CDC_Response_Lead_Exposure_Recs.pdf (accessed on 12 August 2020).
- Feng, C.X.; Cao., J.; Bendell, L. Exploring spatial and temporal variations of cadmium concentrations in pacific oysters from British Columbia. Biometrics 2011, 67, 1142–1152. [Google Scholar] [CrossRef]
- Losasso, C.; Bille, L.; Patuzzi, I.; Lorenzetto, M.; Binato, G.; Pozza, M.D.; Ferrè, N.; Ricci, N. Possible influence of natural events on heavy metals exposure from shellfish consumption: A case study in the north-east of Italy. Front. Public Health 2015, 3, 21. [Google Scholar] [CrossRef]
- Guéguen, M.; Amiard, J.-C.; Arnich, N.; Badot, P.-M.; Claisse, D.; Guérin, T.; Vernoux, J.-P. Shellfish and residual chemical contaminants: Hazards, monitoring, and health risk assessment along French coasts. Rev. Environ. Contam. Toxicol. 2011, 213, 55–111. [Google Scholar]
- Burioli, E.A.V.; Squadrone, S.; Stella, C.; Foglini, C.; Abete, M.C.; Prearo, M. Trace element occurrence in the Pacific oyster Crassostrea gigas from coastal marine ecosystems in Italy. Chemosphere 2017, 187, 248–260. [Google Scholar] [CrossRef]
- Renieri, E.A.; Alegakis, A.K.; Kiriakakis, M.; Vinceti, M.; Ozcagli, E.; Wilks, M.F.; Tsatsakis, A.M. Cd, Pb and Hg biomonitoring in fish of the Mediterranean region and risk estimations on fish consumption. Toxics 2014, 2, 417–442. [Google Scholar] [CrossRef]
- Cobbett, C.S. Phytochelatins and their roles in heavy metal detoxification. Plant. Physiol. 2000, 123, 825–832. [Google Scholar] [CrossRef] [PubMed]
- Cobbett, C.; Goldsbrough, P. Phytochelatins and metallothioneins: Roles in heavy metal detoxification and homeostasis. Annu. Rev. Plant Biol. 2002, 53, 159–182. [Google Scholar] [CrossRef] [PubMed]
- Pivato, M.; Fabrega-Prats, M.; Masi, A. Low-molecular-weight thiols in plants: Functional and analytical implications. Arch. Biochem. Biophys. 2014, 560, 83–99. [Google Scholar] [CrossRef] [PubMed]
- Klaassen, C.D.; Liu, J.; Diwan, B.A. Metallothionein protection of cadmium toxicity. Toxicol. Appl. Pharmacol. 2009, 238, 215–220. [Google Scholar] [CrossRef]
- Scott, S.R.; Smith, K.E.; Dahman, C.; Gorski, P.R.; Adams, S.V.; Shafer, M.M. Cd isotope fractionation during tobacco combustion produces isotopic variation outside the range measured in dietary sources. Sci. Total Environ. 2019, 688, 600–608. [Google Scholar] [CrossRef]
- Aoshima, K. Epidemiology and tubular dysfunction in the inhabitants of a cadmium-polluted area in the Jinzu River basin in Toyama Prefecture. Tohoku J. Exp. Med. 1987, 152, 151–172. [Google Scholar] [CrossRef]
- Spungen, J.H. Children’s exposures to lead and cadmium: FDA total diet study 2014–2016. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2019, 36, 893–903. [Google Scholar] [CrossRef]
- Gavelek, A.; Spungen, J.; Hoffman-Pennesi, D.; Flannery, B.; Dolan, L.; Dennis, S.; Fitzpatrick, S. Lead exposures in older children (males and females 7–17 years), women of childbearing age (females 16–49 years) and adults (males and females 18+ years): FDA total diet study 2014-16. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2020, 37, 104–109. [Google Scholar] [CrossRef]
- European Food Safety Agency (EFSA). Statement on tolerable weekly intake for cadmium. EFSA J. 2011, 9, 1975. [Google Scholar]
- European Food Safety Agency (EFSA). Cadmium dietary exposure in the European population. EFSA J. 2012, 10, 2551. [Google Scholar] [CrossRef]
- Callan, A.; Hinwood, A.; Devine, A. Metals in commonly eaten groceries in Western Australia: A market basket survey and dietary assessment. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2014, 31, 1968–1981. [Google Scholar] [CrossRef] [PubMed]
- Sand, S.; Becker, W. Assessment of dietary cadmium exposure in Sweden and population health concern including scenario analysis. Food Chem. Toxicol. 2012, 50, 536–544. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.; Gao, J.; Cen, K. Levels of eight heavy metals and health risk assessment considering food consumption by China’s residents based on the 5th China total diet study. Sci. Total Environ. 2019, 689, 1141–1148. [Google Scholar] [CrossRef] [PubMed]
- Xiao, G.; Liu, Y.; Dong, K.F.; Lu, J. Regional characteristics of cadmium intake in adult residents from the 4th and 5th Chinese total diet study. Environ. Sci. Pollut. Res. Int. 2020, 27, 3850–3857. [Google Scholar] [CrossRef]
- Jin, Y.; Liu, P.; Sun, J.; Wang, C.; Min, J.; Zhang, Y.; Wang, S.; Wu, Y. Dietary exposure and risk assessment to lead of the population of Jiangsu province, China. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2014, 31, 1187–1195. [Google Scholar]
- Lim, J.A.; Kwon, H.J.; Ha, M.; Kim, H.; Oh, S.Y.; Kim, J.S.; Lee, S.A.; Park, J.D.; Hong, Y.S.; Sohn, S.J.; et al. Korean research project on the integrated exposure assessment of hazardous substances for food safety. Environ. Health Toxicol. 2015, 30, e2015004. [Google Scholar] [CrossRef]
- Kim, H.; Lee, J.; Woo, H.D.; Kim, D.W.; Choi, I.J.; Kim, Y.I.; Kim, J. Association between dietary cadmium intake and early gastric cancer risk in a Korean population: A case-control study. Eur. J. Nutr. 2019, 58, 3255–3266. [Google Scholar] [CrossRef]
- Schwarz, M.A.; Lindtner, O.; Blume, K.; Heinemeyer, G.; Schneider, K. Cadmium exposure from food: The German LExUKon project. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2014, 31, 1038–1051. [Google Scholar] [CrossRef]
- Marín, S.; Pardo, O.; Báguena, R.; Font, G.; Yusà, V. Dietary exposure to trace elements and health risk assessment in the region of Valencia, Spain: A total diet study. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2017, 34, 228–240. [Google Scholar] [CrossRef]
- Puerto-Parejo, L.M.; Aliaga, I.; Canal-Macias, M.L.; Leal-Hernandez, O.; Roncero-Martín, R.; Rico-Martín, S.; Moran, J.M. Evaluation of the dietary intake of cadmium, lead and mercury and its relationship with bone health among postmenopausal women in Spain. Int. J. Environ. Res. Public Health 2017, 14, 564. [Google Scholar] [CrossRef]
- Kim, K.; Melough, M.M.; Vance, T.M.; Noh, H.; Koo, S.I.; Chun, O.K. Dietary cadmium intake and sources in the US. Nutrients 2018, 11, 2. [Google Scholar] [CrossRef] [PubMed]
- Adams, S.V.; Quraishi, S.M.; Shafer, M.M.; Passarelli, M.N.; Freney, E.P.; Chlebowski, R.T.; Luo, J.; Meliker, J.R.; Mu, L.; Neuhouser, M.L.; et al. Dietary cadmium exposure and risk of breast, endometrial, and ovarian cancer in the Women’s Health Initiative. Environ. Health Perspect. 2014, 122, 594–600. [Google Scholar] [CrossRef] [PubMed]
- Filippini, T.; Cilloni, S.; Malavolti, M.; Violi, F.; Malagoli, C.; Tesauro, M.; Bottecchi, I.; Ferrari, A.; Vescovi, L.; Vinceti, M. Dietary intake of cadmium, chromium, copper, manganese, selenium and zinc in a Northern Italy community. J. Trace Elem. Med. Biol. 2018, 50, 508–517. [Google Scholar] [CrossRef] [PubMed]
- Schneider, K.; Schwarz, M.A.; Lindtner, O.; Blume, K.; Heinemeyer, G. Lead exposure from food: The German LExUKon. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2014, 31, 1052–1063. [Google Scholar] [CrossRef]
- Arnich, N.; Sirot, V.; Rivière, G.; Jean, J.; Noël, L.; Guérin, T.; Leblanc, J.-C. Dietary exposure to trace elements and health risk assessment in the 2nd French Total Diet Study. Food Chem. Toxicol. 2012, 50, 2432–2449. [Google Scholar] [CrossRef]
- Vromman, V.; Waegeneers, N.; Cornelis, C.; De Boosere, I.; Van Holderbeke, M.; Vinkx, C.; Smolders, E.; Huyghebaert, A.; Pussemier, L. Dietary cadmium intake by the Belgian adult population. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2010, 27, 1665–1673. [Google Scholar] [CrossRef]
- Horiguchi, H.; Oguma, E.; Sasaki, S.; Miyamoto, K.; Hosoi, Y.; Ono, A.; Kayama, F. Exposure assessment of cadmium in female farmers in cadmium-polluted areas in Northern Japan. Toxics 2020, 8, 44. [Google Scholar] [CrossRef]
- Nishito, Y.; Kambe, T. Absorption mechanisms of iron, copper, and zinc: An overview. J. Nutr. Sci. Vitaminol. 2018, 64, 1–7. [Google Scholar] [CrossRef]
- Vesey, D.A. Transport pathways for cadmium in the intestine and kidney proximal tubule: Focus on the interaction with essential metals. Toxicol. Lett. 2010, 198, 13–19. [Google Scholar] [CrossRef]
- Thévenod, F.; Lee, W.-K.; Garrick, M.D. Iron and cadmium entry into renal mitochondria: Physiological and toxicological implications. Front. Cell Develop. Biol. 2020, 8, 848. [Google Scholar] [CrossRef]
- Kovacs, G.; Danko, T.; Bergeron, M.J.; Balazs, B.; Suzuki, Y.; Zsembery, A.; Hediger, M.A. Heavy metal cations permeate the TRPV6 epithelial cation channel. Cell Calcium. 2011, 49, 43–55. [Google Scholar] [CrossRef] [PubMed]
- Kovacs, G.; Montalbetti, N.; Franz, M.C.; Graeter, S.; Simonin, A.; Hediger, M.A. Human TRPV5 and TRPV6: Key players in cadmium and zinc toxicity. Cell Calcium. 2013, 54, 276–286. [Google Scholar] [CrossRef] [PubMed]
- Fujishiro, H.; Hamao, S.; Tanaka, R.; Kambe, T.; Himeno, S. Concentration-dependent roles of DMT1 and ZIP14 in cadmium absorption in Caco-2 cells. J. Toxicol. Sci. 2017, 42, 559–567. [Google Scholar] [CrossRef] [PubMed]
- Thevenod, F.; Fels, J.; Lee, W.-K.; Zarbock, R. Channels, transporters and receptors for cadmium and cadmium complexes in eukaryotic cells: Myths and facts. Biometals 2019, 32, 469–489. [Google Scholar] [CrossRef]
- Mackenzie, B.; Takanaga, H.; Hubert, N.; Rolfs, A.; Hediger, M.A. Functional properties of multiple isoforms of human divalent metal-ion transporter 1 (DMT1). Biochem. J. 2007, 403, 59–69. [Google Scholar] [CrossRef]
- Illing, A.C.; Shawki, A.; Cunningham, C.L.; Mackenzie, B. Substrate profile and metal-ion selectivity of human divalent metal-ion transporter-1. J. Biol. Chem. 2012, 287, 30485–30496. [Google Scholar] [CrossRef]
- Bannon, D.I.; Abounader, R.; Lees, P.S.; Bressler, J.P. Effect of DMT1 knockdown on iron, cadmium, and lead uptake in Caco-2 cells. Am. J. Physiol. Cell Physiol. 2003, 284, C44–C50. [Google Scholar] [CrossRef]
- Aduayom, I.; Jumarie, C. Reciprocal inhibition of Cd and Pb sulfocomplexes for uptake in Caco-2 cells. J. Biochem. Mol. Toxicol. 2005, 19, 256–265. [Google Scholar] [CrossRef]
- Mitchell, C.J.; Shawki, A.; Ganz, T.; Nemeth, E.; Mackenzie, B. Functional properties of human ferroportin, a cellular iron exporter reactive also with cobalt and zinc. Am. J. Physiol. Cell Physiol. 2014, 306, C450–C459. [Google Scholar] [CrossRef]
- Jeon, H.-K.; Jin, H.-S.; Lee, D.-H.; Choi, W.-S.; Moon, C.-K.; Oh, Y.J.; Lee, T.H. Proteome analysis associated with cadmium adaptation in U937 cells: Identification of calbindin-D28k as a secondary cadmium-responsive protein that confers resistance to cadmium-induced apoptosis. J. Biol. Chem. 2004, 279, 31575–31583. [Google Scholar] [CrossRef]
- Fujita, Y.; ElBelbasi, H.I.; Min, K.-S.; Onosaka, S.; Okada, Y.; Matsumoto, Y.; Mutoh, N.; Tanaka, K. Fate of cadmium bound to phytochelatin in rats. Res. Commun. Chem. Pathol. Pharmacol. 1993, 82, 357–365. [Google Scholar] [PubMed]
- Langelueddecke, C.; Roussa, E.; Fenton, R.A.; Thévenod, F. Expression and function of the lipocalin-2 (24p3/NGAL) receptor in rodent and human intestinal epithelia. PLoS ONE 2013, 8, e71586. [Google Scholar] [CrossRef] [PubMed]
- Langelueddecke, C.; Lee, W.-K.; Thevenod, F. Differential transcytosis and toxicity of the hNGAL receptor ligands cadmium-metallothionein and cadmium-phytochelatin in colon-like Caco-2 cells: Implications for cadmium toxicity. Toxicol. Lett. 2014, 226, 228–235. [Google Scholar] [CrossRef] [PubMed]
- Jorge-Nebert, L.F.; Gálvez-Peralta, M.; Figueroa, J.L.; Somarathna, M.; Hojyo, S.; Fukada, T.; Nebert, D.W. Comparing gene expression during cadmium uptake and distribution: Untreated versus oral Cd-treated wild-type and ZIP14 knockout mice. Toxicol. Sci. 2015, 143, 26–35. [Google Scholar] [CrossRef]
- McKenna, I.M.; Gordon, T.; Chen, L.C.; Anver, M.R.; Waalkes, M.P. Expression of metallothionein protein in the lungs of Wistar rats and C57 and DBA mice exposed to cadmium oxide fumes. Toxicol. Appl. Pharmacol. 1998, 153, 169–178. [Google Scholar] [CrossRef]
- Takeda, K.; Fujita, H.; Shibahara, S. Differential control of the metal-mediated activation of the human heme oxygenase-1 and metallothionein IIA genes. Biochem. Biophys. Res. Commun. 1995, 207, 160–167. [Google Scholar] [CrossRef]
- Hart, B.A. Cellular and biochemical response of the rat lung to repeated inhalation of cadmium. Toxicol. Appl. Pharmacol. 1986, 82, 281–291. [Google Scholar] [CrossRef]
- Hart, B.A.; Gong, Q.; Eneman, J.D. Pulmonary metallothionein expression in rats following single and repeated exposure to cadmium aerosols. Toxicology 1996, 112, 205–218. [Google Scholar] [CrossRef]
- Chandler, J.D.; Wongtrakool, C.; Banton, S.A.; Li, S.; Orr, M.L.; Barr, D.B.; Neujahr, D.C.; Sutliff, R.L.; Go, Y.M.; Jones, D.P. Low-dose oral cadmium increases airway reactivity and lung neuronal gene expression in mice. Physiol. Rep. 2016, 4, e12821. [Google Scholar] [CrossRef]
- Sabolić, I.; Breljak, D.; Skarica, M.; Herak-Kramberger, C.M. Role of metallothionein in cadmium traffic and toxicity in kidneys and other mammalian organs. Biometals 2010, 23, 897–926. [Google Scholar] [CrossRef]
- Yu, J.; Fujishiro, H.; Miyataka, H.; Oyama, T.M.; Hasegawa, T.; Seko, Y.; Miura, N.; Himeno, S. Dichotomous effects of lead acetate on the expression of metallothionein in the liver and kidney of mice. Biol. Pharm. Bull. 2009, 32, 1037–1042. [Google Scholar] [PubMed]
- Dai, S.; Yin, Z.; Yuan, G.; Lu, H.; Jia, R.; Xu, J.; Song, X.; Li, L.; Shu, Y.; Liang, X.; et al. Quantification of metallothionein on the liver and kidney of rats by subchronic lead and cadmium in combination. Environ. Toxicol. Pharmacol. 2013, 36, 1207–1216. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi, Y.; Nomiyama, T.; Kumagai, N.; Dekio, F.; Uemura, T.; Takebayashi, T.; Nishiwaki, Y.; Matsumoto, Y.; Sano, Y.; Hosoda, K.; et al. Uptake of cadmium in meals from the digestive tract of young non-smoking Japanese female volunteers. J. Occup. Health 2003, 45, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Kim, D.; Tucker, K.L.; Weisskopf, M.G.; Sparrow, D.; Hu, H.; Park, S.K. Effect of dietary sodium and potassium on the mobilization of bone lead among middle-aged and older men: The Veterans Affairs Normative Aging Study. Nutrients 2019, 11, 2750. [Google Scholar]
- Nielsen, R.; Christensen, E.I.; Birn, H. Megalin and cubilin in proximal tubule protein reabsorption: From experimental models to human disease. Kidney Int. 2016, 89, 58–67. [Google Scholar]
- Onodera, A.; Tani, M.; Michigami, T.; Yamagata, M.; Min, K.S.; Tanaka, K.; Nakanishi, T.; Kimura, T.; Itoh, N. Role of megalin and the soluble form of its ligand RAP in Cd-metallothionein endocytosis and Cd-metallothionein-induced nephrotoxicity in vivo. Toxicol. Lett. 2012, 212, 91–96. [Google Scholar]
- Langelueddecke, C.; Roussa, E.; Fenton, R.A.; Wolff, N.A.; Lee, W.K.; Thévenod, F. Lipocalin-2 (24p3/neutrophil gelatinase-associated lipocalin (NGAL)) receptor is expressed in distal nephron and mediates protein endocytosis. J. Biol. Chem. 2012, 287, 159–169. [Google Scholar]
- Fels, J.; Scharner, B.; Zarbock, R.; Zavala Guevara, I.P.; Lee, W.K.; Barbier, O.C.; Thévenod, F. Cadmium complexed with β2-microglubulin, albumin and lipocalin-2 rather than metallothionein cause megalin:cubilin dependent toxicity of the renal proximal tubule. Int. J. Mol. Sci. 2019, 20, 2379. [Google Scholar]
- Nascimento, C.R.B.; Risso, W.E.; Martinez, C.B.D.R. Lead accumulation and metallothionein content in female rats of different ages and generations after daily intake of Pb-contaminated food. Environ. Toxicol. Pharmacol. 2016, 48, 272–277. [Google Scholar]
- Satarug, S.; Baker, J.R.; Reilly, P.E.; Moore, M.R.; Williams, D.J. Cadmium levels in the lung, liver, kidney cortex, and urine samples from Australians without occupational exposure to metals. Arch. Environ. Health 2002, 57, 69–77. [Google Scholar]
- Baker, J.R.; Edwards, R.J.; Lasker, J.M.; Moore, M.R.; Satarug, S. Renal and hepatic accumulation of cadmium and lead in the expression of CYP4F2 and CYP2E1. Toxicol. Lett. 2005, 159, 182–191. [Google Scholar] [PubMed]
- Barregard, L.; Fabricius-Lagging, E.; Lundh, T.; Mölne, J.; Wallin, M.; Olausson, M.; Modigh, C.; Sallstenm, G. Cadmium, mercury, and lead in kidney cortex of living kidney donors: Impact of different exposure sources. Environ. Res. 2010, 110, 47–54. [Google Scholar] [PubMed]
- Järup, L.; Rogenfelt, A.; Elinder, C.G.; Nogawa, K.; Kjellström, T. Biological half-time of cadmium in the blood of workers after cessation of exposure. Scand. J. Work Environ. Health 1983, 9, 327–331. [Google Scholar] [PubMed]
- Börjesson, J.; Bellander, T.; Järup, L.; Elinder, C.G.; Mattsson, S. In vivo analysis of cadmium in battery workers versus measurements of blood, urine, and workplace air. Occup. Environ. Med. 1997, 54, 424–531. [Google Scholar]
- Suwazono, Y.; Kido, T.; Nakagawa, H.; Nishijo, M.; Honda, R.; Kobayashi, E.; Dochi, M.; Nogawa, K. Biological half-life of cadmium in the urine of inhabitants after cessation of cadmium exposure. Biomarkers 2009, 14, 77–81. [Google Scholar] [CrossRef]
- Ishizaki, M.; Suwazono, Y.; Kido, T.; Nishijo, M.; Honda, R.; Kobayashi, E.; Nogawa, K.; Nakagawa, H. Estimation of biological half-life of urinary cadmium in inhabitants after cessation of environmental cadmium pollution using a mixed linear model. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2015, 32, 1273–1276. [Google Scholar]
- Fransson, M.N.; Barregard, L.; Sallsten, G.; Akerstrom, M.; Johanson, G. Physiologically-based toxicokinetic model for cadmium using Markov-chain Monte Carlo analysis of concentrations in blood, urine, and kidney cortex from living kidney donors. Toxicol. Sci. 2014, 141, 365–376. [Google Scholar]
- Specht, A.J.; Lin, Y.; Weisskopf, M.; Yan, C.; Hu, H.; Xu, J.; Nie, L.H. XRF-measured bone lead (Pb) as a biomarker for Pb exposure and toxicity among children diagnosed with Pb poisoning. Biomarkers 2016, 21, 347–352. [Google Scholar]
- Orlowski, C.; Piotrowski, J.K.; Subdys, J.K.; Gross, A. Urinary cadmium as indicator of renal cadmium in humans: An autopsy study. Hum. Exp. Toxicol. 1998, 17, 302–306. [Google Scholar]
- Akerstrom, M.; Barregard, L.; Lundh, T.; Sallsten, G. The relationship between cadmium in kidney and cadmium in urine and blood in an environmentally exposed population. Toxicol. Appl. Pharmacol. 2013, 268, 286–293. [Google Scholar]
- Wallin, M.; Sallsten, G.; Lundh, T.; Barregard, L. Low-level cadmium exposure and effects on kidney function. Occup. Environ. Med. 2014, 71, 848–854. [Google Scholar] [CrossRef] [PubMed]
- Gerhardsson, L.; Englyst, V.; Lundström, N.G.; Sandberg, S.; Nordberg, G. Cadmium, copper and zinc in tissues of deceased copper smelter workers. J. Trace Elem. Med. Biol. 2002, 16, 261–266. [Google Scholar] [CrossRef]
- Lou, M.; Garay, R.; Alda., J.O. Cadmium uptake through the anion exchanger in human red blood cells. J. Physiol. 1991, 443, 123–136. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.; Satchwell, T.J.; Toye, A.M. Anion exchanger 1 in red blood cells and kidney: Band 3’s in a pod. Biochem. Cell Biol. 2011, 89, 106–114. [Google Scholar] [PubMed]
- Parker, M.D.; Boron, W.F. The divergence, actions, roles, and relatives of sodium-coupled bicarbonate transporters. Physiol. Rev. 2013, 93, 803–959. [Google Scholar] [CrossRef]
- Savigni, D.L.; Morgan, E.H. Transport mechanisms for iron and other transition metals in rat and rabbit erythroid cells. J. Physiol. 1998, 508, 837–850. [Google Scholar] [CrossRef] [PubMed]
- Simons, T.J. The role of anion transport in the passive movement of lead across the human red cell membrane. J. Physiol. 1986, 378, 287–312. [Google Scholar] [CrossRef]
- Simons, T.J. Lead transport and binding by human erythrocytes in vitro. Pflugers Arch. 1993, 423, 307–313. [Google Scholar] [CrossRef]
- Lang, F.; Abed, M.; Lang, E.; Föller, M. Oxidative stress and suicidal erythrocyte death. Antioxid. Redox Signal. 2014, 21, 138–153. [Google Scholar] [CrossRef]
- Lang, E.; Lang, F. Mechanisms and pathophysiological significance of eryptosis, the suicidal erythrocyte death. Semin. Cell Dev. Biol. 2015, 39, 35–42. [Google Scholar] [CrossRef]
- Attanzio, A.; Frazzitta, A.; Vasto, S.; Tesoriere, L.; Pintaudi, A.M.; Livrea, M.A.; Cilla, A.; Allegra, M. Increased eryptosis in smokers is associated with the antioxidant status and C-reactive protein levels. Toxicology 2019, 411, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Scott, B.J.; Bradwell, A.R. Identification of the serum binding proteins for iron, zinc, cadmium, nickel, and calcium. Clin. Chem. 1983, 29, 629–633. [Google Scholar] [CrossRef] [PubMed]
- Horn, N.M.; Thomas, A.L. Interactions between the histidine stimulation of cadmium and zinc influx into human erythrocytes. J. Physiol. 1996, 496, 711–718. [Google Scholar] [CrossRef] [PubMed]
- Turell, L.; Radi, R.; Alvarez, B. The thiol pool in human plasma: The central contribution of albumin to redox processes. Free Radic. Biol. Med. 2013, 65, 244–253. [Google Scholar] [CrossRef] [PubMed]
- Morris, T.T.; Keir, J.L.; Boshart, S.J.; Lobanov, V.P.; Ruhland, A.M.; Bahl, N.; Gailer, J. Mobilization of Cd from human serum albumin by small molecular weight thiols. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2014, 958, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Sagmeister, P.; Gibson, M.A.; McDade, K.H.; Gailer, J. Physiologically relevant plasma d,l-homocysteine concentrations mobilize Cd from human serum albumin. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2016, 1027, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Gaudet, M.M.; Deubler, E.L.; Kelly, R.S.; Diver, W.R.; Teras, L.R.; Hodge, J.M.; Levine, K.E.; Haines, L.G.; Lundh, T.; Lenner, P.; et al. Blood levels of cadmium and lead in relation to breast cancer risk in three prospective cohorts. Int. J. Cancer 2019, 144, 1010–1016. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Zhang, F.; Lei, Y. Dietary intake and urinary level of cadmium and breast cancer risk: A meta-analysis. Cancer Epidemiol. 2016, 42, 101–107. [Google Scholar] [CrossRef]
- Rokadia, H.K.; Agarwal, S. Serum heavy metals and obstructive lung disease: Results from the National Health and Nutrition Examination Survey. Chest 2013, 143, 388–397. [Google Scholar] [CrossRef]
- Yang, G.; Sun, T.; Han, Y.Y.; Rosser, F.; Forno, E.; Chen, W.; Celedón, J.C. Serum cadmium and lead, current wheeze, and lung function in a nationwide study of adults in the United States. J. Allergy Clin. Immunol. Pract. 2019, 7, 2653–2660.e3. [Google Scholar] [CrossRef]
- Bergdahl, I.A.; Schütz, A.; Gerhardsson, L.; Jensen, A.; Skerfving, S. Lead concentrations in human plasma, urine and whole blood. Scand. J. Work Environ. Health 1997, 23, 359–363. [Google Scholar] [CrossRef] [PubMed]
- Manton, W.I.; Rothenberg, S.J.; Manalo, M. The lead content of blood serum. Environ. Res. 2001, 86, 263–273. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Smith, D.; Hernandez-Avila, M.; Téllez-Rojo, M.M.; Mercado, A.; Hu, H. The relationship between lead in plasma and whole blood in women. Environ. Health Perspect. 2002, 110, 263–268. [Google Scholar] [CrossRef]
- Barbosa, F., Jr.; Tanus-Santos, J.E.; Gerlach, R.F.; Parsons, P.J. A critical review of biomarkers used for monitoring human exposure to lead: Advantages, limitations, and future needs. Environ. Health Perspect. 2005, 113, 1669–1674. [Google Scholar] [CrossRef]
- Gulson, B.L.; Mizon, K.J.; Korsch, M.J.; Horwarth, D.; Phillips, A.; Hall, J. Impact on blood lead in children and adults following relocation from their source of exposure and contribution of skeletal tissue to blood lead. Bull. Environ. Contam. Toxicol. 1996, 56, 543–550. [Google Scholar]
- Gulson, B.L.; Mahaffey, K.R.; Mizon, K.F.; Korsch, M.J.; Cameron, M.A.; Vimpani, G. Contribution of tissue lead to bone lead in adult female subjects based on stable lead-isotope methods. J. Lab. Clin. Med. 1995, 125, 703–712. [Google Scholar] [PubMed]
- Gwiazda, R.; Campbell, C.; Smith, D. A noninvasive isotopic approach to estimate the bone lead contribution to blood in children: Implications for assessing the efficacy of lead abatement. Environ. Health Perspect. 2005, 113, 104–110. [Google Scholar] [CrossRef] [PubMed]
- Manton, W.I.; Angle, C.R.; Stanek, K.L.; Reese, Y.R.; Kuehnemann, T.J. Acquisition and retention of lead by young children. Environ. Res. 2000, 82, 60–80. [Google Scholar] [CrossRef]
- Roberts, J.R.; Reigart., J.R.; Ebeling., M.; Hulsey, T.C. Time required for blood lead levels to decline in nonchelated children. Clin. Toxicol. 2001, 39, 153–160. [Google Scholar] [CrossRef]
- Landrigan, P.J.; Todd, A.C. Direct measurement of lead in bone-a promising biomarker. JAMA 1994, 271, 239–240. [Google Scholar] [CrossRef]
- O’Flaherty, E.J. Physiologically based models for bone-seeking elements V: Lead absorption and disposition in childhood. Toxicol. Appl. Pharmacol. 1995, 131, 297–308. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Rabinowitz, M.; Smith., D. Bone lead as a biological marker in epidemiologic studies of chronic toxicity: Conceptual paradigms. Environ. Health Perspect. 1998, 106, 1–8. [Google Scholar] [CrossRef]
- Nilsson, U.; Attewell, R.; Christoffersson, J.O.; Schütz, A.; Ahlgren, L.; Skerfving, S.; Mattsson, S. Kinetics of lead in bone and blood after end of occupational exposure. Pharmacol. Toxicol. 1991, 68, 477–484. [Google Scholar] [CrossRef] [PubMed]
- Price, J.; Grudzinski, A.W.; Craswell, P.W.; Thomas, B.J. Repeated bone lead levels in Queensland, Australia—Previously a high lead environment. Arch. Environ. Health 1992, 47, 256–262. [Google Scholar] [CrossRef] [PubMed]
- Brito, J.A.; McNeill, F.E.; Stronach, I.; Webber, C.E.; Wells, S.; Richard, N.; Chettle, D.R. Longitudinal changes in bone lead concentration: Implications for modelling of human bone lead metabolism. J. Environ. Monit. 2001, 3, 343–351. [Google Scholar] [CrossRef] [PubMed]
- Wilker, E.; Korrick, S.; Nie, L.H.; Sparrow, D.; Vokonas, P.; Coull, B.; Wright, R.O.; Schwartz., J.; Hu, H. Longitudinal changes in bone lead levels: The VA normative aging study. J. Occup. Environ. Med. 2011, 53, 850–855. [Google Scholar] [CrossRef] [PubMed]
- Gerhardsson, L.; Akantis, A.; Lundström, N.G.; Nordberg, G.F.; Schütz, A.; Skerfving, S. Lead concentrations in cortical and trabecular bones in deceased smelter workers. J. Trace Elem. Med. Biol. 2005, 19, 209–215. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gerhardsson, L.; Englyst, V.; Lundström, N.G.; Nordberg, G.; Sandberg, S.; Steinvall, F. Lead in tissues of deceased lead smelter workers. J. Trace Elem. Med. Biol. 1995, 9, 136–143. [Google Scholar] [CrossRef]
- Hernandez-Avila, M.; Smith, D.; Meneses, F.; Sanin, L.H.; Hu, H. The influence of bone and blood lead on plasma lead levels in environmentally exposed adults. Environ. Health Perspect. 1998, 106, 473–477. [Google Scholar] [CrossRef]
- Hu, H.; Shih, R.; Rothenberg, S.; Schwartz, B.S. The epidemiology of lead toxicity in adults: Measuring dose and consideration of other methodologic issues. Environ. Health Perspect. 2007, 115, 455–462. [Google Scholar] [CrossRef]
- Shih, R.A.; Hu, H.; Weisskopf, M.G.; Schwartz, B.S. Cumulative lead dose and cognitive function in adults: A review of studies that measured both blood lead and bone lead. Environ. Health Perspect. 2007, 115, 483–492. [Google Scholar] [CrossRef] [PubMed]
- Farooqui, Z.; Bakulski, K.M.; Power, M.C.; Weisskopf, M.G.; Sparrow, D.; Spiro, A., III; Vokonas, P.S.; Nie, L.H.; Hu, H.; Park, S.K. Associations of cumulative Pb exposure and longitudinal changes in mini-mental status exam scores, global cognition and domains of cognition: The VA normative aging study. Environ. Res. 2017, 152, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Wright, R.O.; Tsaih, S.W.; Schwartz, J.; Spiro, A., III; McDonald, K.; Weiss, S.T.; Hu, H. Lead exposure biomarkers and mini-mental status exam scores in older men. Epidemiology 2003, 14, 713–718. [Google Scholar] [CrossRef] [PubMed]
- Zheutlin, A.R.; Hu, H.; Weisskopf, M.G.; Sparrow, D.; Vokonas, P.S.; Park, S.K. Low-level cumulative lead and resistant hypertension: A prospective study of men participating in the Veterans Affairs normative aging study. J. Am. Heart Assoc. 2018, 7, e010014. [Google Scholar] [CrossRef] [PubMed]
- Hirata, M.; Yoshida, T.; Miyajima, K.; Kosaka, H.; Tabuchi, T. Correlation between lead in plasma and other indicators of lead exposure among lead-exposed workers. Int. Arch. Occup. Environ. Health 1995, 68, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Schütz, A.; Olsson, M.; Jensen, A.; Gerhardsson, L.; Börjesson, J.; Mattsson, S.; Skerfving, S. Lead in finger bone, whole blood, plasma and urine in lead-smelter workers: Extended exposure range. Int. Arch. Occup. Environ. Health 2005, 78, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Fukui, Y.; Miki, M.; Ukai, H.; Okamoto, S.; Takada, S.; Higashikawa, K.; Ikeda, M. Urinary lead as a possible surrogate of blood lead among workers occupationally exposed to lead. Int. Arch. Occup. Environ. Health 1999, 72, 516–520. [Google Scholar] [CrossRef]
- Bai, Y.; Laenen, A.; Haufroid, V.; Nawrot, T.S.; Nemery, B. Urinary lead in relation to combustion-derived air pollution in urban environments. A longitudinal study of an international panel. Environ. Int. 2019, 125, 75–81. [Google Scholar] [CrossRef]
- Wang, X.; Jin, P.; Zhou, Q.; Liu, S.; Wang, F.; Xi, S. Metal biomonitoring and comparative assessment in urine of workers in lead-zinc and steel-iron mining and smelting. Biol. Trace Elem. Res. 2019, 189, 1–9. [Google Scholar] [CrossRef]
- Li, S.; Wang, J.; Zhang, B.; Liu, Y.; Lu, T.; Shi, Y.; Shan, G.; Dong, L. Urinary lead concentration is an independent predictor of cancer mortality in the U.S. general population. Front. Oncol. 2018, 8, 242. [Google Scholar] [CrossRef]
- Dudley, R.E.; Gammal, L.M.; Klaassen, C.D. Cadmium-induced hepatic and renal injury in chronically exposed rats: Likely role of hepatic cadmium-metallothionein in nephrotoxicity. Toxicol. Appl. Pharmacol. 1985, 77, 414–426. [Google Scholar] [CrossRef]
- Chan, H.M.; Zhu, L.F.; Zhong, R.; Grant, D.; Goyer, R.A.; Cherian, M.G. Nephrotoxicity in rats following liver transplantation from cadmium-exposed rats. Toxicol. Appl. Pharmacol. 1993, 123, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Shaikh, Z.A.; Vu, T.T.; Zaman, K. Oxidative stress as a mechanism of chronic cadmium-induced hepatoxicity and renal toxicity and protection by antioxidants. Toxicol. Appl. Pharmacol. 1999, 154, 256–263. [Google Scholar] [CrossRef] [PubMed]
- Goyer, R.A.; Miller, C.R.; Zhu, S.-Y.; Victery, W. Non-metallothionein-bound cadmium in the pathogenesis of cadmium nephrotoxicity in the rat. Toxicol. Appl. Pharmacol. 1989, 101, 232–244. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, J.; Habeebu, S.S.M.; Klaasen, C.D. Metallothionein protects against the nephrotoxicity produced by chronic CdMT exposure. Toxicol. Sci. 1999, 50, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Vestergaard, P.; Shaikh, Z.A. The nephrotoxicity of intravenously administered cadmium-metallothionein: Effect of dose, mode of administration, and preexisting renal cadmium burden. Toxicol. Appl. Pharmacol. 1994, 126, 240–247. [Google Scholar] [CrossRef] [PubMed]
- Min, K.S.; Onosaka, S.; Tanaka, K. Renal accumulation of cadmium and nephropathy following long-term administration of cadmium-metallothionein. Toxicol. Appl. Pharmacol. 1996, 141, 102–109. [Google Scholar] [CrossRef]
- Price, R.G. The role of NAG (N-acetyl-β-D-glucosaminidase) in the diagnosis of kidney disease including the monitoring of nephrotoxicity. Clin. Nephrol. 1992, 38, S14–S19. [Google Scholar]
- Bernard, A.; Thielemans, N.; Roels, H.; Lauwerys, R. Association between NAG-B and cadmium in urine with no evidence of a threshold. Occup. Environ. Med. 1995, 52, 177–180. [Google Scholar] [CrossRef]
- Jin, T.; Nordberg, G.; Wu, X.; Kong, Q.; Wang, Z.; Zhuang, F.; Cai, S. Urinary N-acetyl-beta-D-glucosaminidase isoenzymes as biomarker of renal dysfunction caused by cadmium in a general population. Environ. Res. 1999, 81, 167–173. [Google Scholar] [CrossRef]
- Tassi, C.; Abbritti, G.; Mancuso, F.; Morucci, P.; Feligioni, L.; Muzi, G. Activity and isoenzyme profile of N-acetyl-beta-D-glucosaminidase in urine from workers exposed to cadmium. Clin. Chim. Acta 2000, 299, 55–64. [Google Scholar] [CrossRef]
- Prozialeck, W.C.; Edwards, J.R. Early biomarkers of cadmium exposure and nephrotoxicity. Biometals 2010, 23, 793–809. [Google Scholar] [CrossRef] [PubMed]
- Prozialeck, W.C.; Vaidya, V.S.; Liu, J.; Waalkes, M.P.; Edwards, J.R.; Lamar, P.C.; Bernard, A.M.; Dumont, X.; Bonventre, J.V. Kidney injury molecule-1 is an early biomarker of cadmium nephrotoxicity. Kidney Int. 2007, 72, 985–993. [Google Scholar] [CrossRef]
- Prozialeck, W.C.; Edwards, J.R.; Vaidya, V.S.; Bonventre, J.V. Preclinical evaluation of novel urinary biomarkers of cadmium nephrotoxicity. Toxicol. Appl. Pharmacol. 2009, 238, 301–305. [Google Scholar] [CrossRef] [PubMed]
- Prozialeck, W.C.; Edwards, J.R.; Lamar, P.C.; Liu, J.; Vaidya, V.S.; Bonventre, J.V. Expression of kidney injury molecule-1 (Kim-1) in relation to necrosis and apoptosis during the early stages of Cd-induced proximal tubular injury. Toxicol. Appl. Pharmacol. 2009, 238, 306–314. [Google Scholar] [CrossRef]
- Pennemans, V.; De Winter, L.M.; Munters, E.; Nawrot, T.S.; Van Kerhove, E.; Rigo, J.-M.; Reynders, C.; Dewitte, H.; Carleer, R.; Penders, J.; et al. The association between urinary kidney injury molecule 1 and urinary cadmium in elderly during long-term, low-dose cadmium exposure: A pilot study. Environ. Health 2011, 10, 77. [Google Scholar] [CrossRef]
- Ruangyuttikarn, W.; Panyamoon, A.; Nambunmee, K.; Honda, R.; Swaddiwudhipong, W.; Nishijo, M. Use of the kidney injury molecule-1 as a biomarker for early detection of renal tubular dysfunction in a population chronically exposed to cadmium in the environment. Springerplus 2013, 2, 533. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, P.; Liang, X.; Chuen, S.; Tan, J.; Wang, J.; Huang, Q.; Huang, R.; Li, Z.; Chen, W.; et al. Associations between urinary excretion of cadmium and renal biomarkers in nonsmoking females: A cross-sectional study in rural areas of South China. Int. J. Environ. Res. Public Health 2015, 12, 11988–12001. [Google Scholar] [CrossRef]
- Chaumont, A.; Voisin, C.; Deumer, G.; Haufroid, V.; Annesi-Maesano, I.; Roels, H.; Thijs, L.; Staessen, J.; Bernard, A. Associations of urinary cadmium with age and urinary proteins: Further evidence of physiological variations unrelated to metal accumulation and toxicity. Environ. Health Perspect. 2013, 121, 1047–1053. [Google Scholar] [CrossRef]
- Nomiyama, K.; Foulkes, E.C. Reabsorption of filtered cadmium-metallothionein in the rabbit kidney. Proc. Soc. Exp. Biol. Med. 1977, 156, 97–99. [Google Scholar] [CrossRef]
- Tanimoto, A.; Hamada, T.; Koide, O. Cell death and regeneration of renal proximal tubular cells in rats with subchronic cadmium intoxication. Toxicol. Pathol. 1993, 21, 341–352. [Google Scholar] [CrossRef] [PubMed]
- Roels, H.A.; Lauwerys, R.R.; Buchyet, J.-P.; Bernard, A.; Chettle, D.R.; Harvey, T.C.; Al-Haddad, I.K. In vivo measurement of liver and kidney cadmium in workers exposed to this metal: Its significance with respect to cadmium in blood and urine. Environ. Res. 1981, 26, 217–240. [Google Scholar] [CrossRef]
- Weaver, V.M.; Kim, N.-S.; Jaar, B.G.; Schwartz, B.S.; Parsons, P.J.; Steuerwald, A.J.; Todd, A.C.; Simon, D.; Lee, B.-K. Associations of low-level urine cadmium with kidney function in lead workers. Occup. Environ. Med. 2011, 68, 250–256. [Google Scholar] [CrossRef] [PubMed]
- Buser, M.C.; Ingber, S.Z.; Raines, N.; Fowler, D.A.; Scinicariello, F. Urinary and blood cadmium and lead and kidney function: NHANES 2007–2012. Int. J. Hyg. Environ. Health 2016, 219, 261–267. [Google Scholar] [CrossRef]
- Jin, R.; Zhu, X.; Shrubsole, M.J.; Yu, C.; Xia, Z.; Dai, Q. Associations of renal function with urinary excretion of metals: Evidence from NHANES 2003–2012. Environ. Int. 2018, 121, 1355–1362. [Google Scholar] [CrossRef]
- Kawada, T.; Koyama, H.; Suzuki, S. Cadmium, NAG activity, and β2-microglobulin in the urine of cadmium pigment workers. Br. J. Ind. Med. 1989, 46, 52–55. [Google Scholar]
- Kawada, T.; Shinmyo, R.R.; Suzuki, S. Urinary cadmium and N-acetyl-β-D-glucosaminidase excretion of inhabitants living in a cadmium-polluted area. Int. Arch. Occup. Environ. Health 1992, 63, 541–546. [Google Scholar] [CrossRef]
- Koyama, H.; Satoh, H.; Suzuki, S.; Tohyama, C. Increased cadmium excretion and its relationship to urinary N-acetyl-β-D-glucosaminidase activity in smokers. Arch. Toxicol. 1992, 66, 598–601. [Google Scholar] [CrossRef]
- Thomas, L.D.; Hodgson, S.; Nieuwenhuijsen, M.; Jarup, L. Early kidney damage in a population exposed to cadmium and other heavy metals. Environ. Health Perspect. 2009, 117, 181–184. [Google Scholar] [CrossRef]
- Wang, D.; Sun, H.; Wu, Y.; Zhou, Z.; Ding, Z.; Chen, X.; Xu, Y. Tubular and glomerular kidney effects in the Chinese general population with low environmental cadmium exposure. Chemosphere 2016, 147, 3–8. [Google Scholar] [CrossRef]
- Akesson, A.; Lundh, T.; Vahter, M.; Bjellerup, P.; Lidfeldt, J.; Nerbrand, C.; Samsioe, G.; Strömberg, U.; Skerfving, S. Tubular and glomerular kidney effects in Swedish women with low environmental cadmium exposure. Environ. Health Perspect. 2005, 113, 1627–1631. [Google Scholar] [CrossRef] [PubMed]
- Eom, S.-Y.; Seo, M.-N.; Lee, Y.-S.; Park, K.-S.; Hong, Y.-S.; Sohn, S.-J.; Kim, Y.-D.; Choi, B.-S.; Lim, J.-A.; Kwon, H.-J.; et al. Low-level environmental cadmium exposure induces kidney tubule damage in the general population of Korean adults. Arch. Environ. Contam. Toxicol. 2017, 73, 401–409. [Google Scholar] [CrossRef] [PubMed]
- Swaddiwudhipong, W.; Limpatanachote, P.; Mahasakpan, P.; Krintratun, S.; Punta, B.; Funkhiew, T. Progress in cadmium-related health effects in persons with high environmental exposure in northwestern Thailand: A five-year follow-up. Environ. Res. 2012, 112, 194–198. [Google Scholar] [CrossRef] [PubMed]
- Piscator, M. Long-term observations on tubular and glomerular function in cadmium-exposed persons. Environ. Health Perspect. 1984, 54, 175–179. [Google Scholar] [CrossRef] [PubMed]
- Roels, H.A.; Lauwerys, R.R.; Buchyet, J.P.; Bernard, A.M.; Vos, A.; Oversteyns, M. Health significance of cadmium induced renal dysfunction: A five year follow up. Br. J. Ind. Med. 1989, 46, 755–764. [Google Scholar] [CrossRef] [PubMed]
- Jarup, L.; Persson, B.; Elinder, C.G. Decreased glomerular filtration rate in solderers exposed to cadmium. Occup. Environ. Med. 1995, 52, 818–822. [Google Scholar] [CrossRef] [PubMed]
- Mason, H.J.; Davison, A.G.; Wright, A.L.; Guthrie, C.J.G.; Fayers, P.M.; Venables, K.M.; Smith, N.J.; Chettle, D.R.; Franklin, D.M.; Scott, M.C.; et al. Relations between liver cadmium, cumulative exposure, and renal function in cadmium alloy workers. Br. J. Ind. Med. 1988, 45, 793–802. [Google Scholar] [CrossRef] [PubMed]
- Satarug, S.; Moore, M.R. Adverse health effects of chronic exposure to low-level cadmium in foodstuffs and cigarette smoke. Environ. Health Perspect. 2004, 112, 1099–1103. [Google Scholar] [CrossRef] [PubMed]
- Schnaper, H.W. The tubulointerstitial pathophysiology of progressive kidney disease. Adv. Chronic Kidney Dis. 2017, 24, 107–116. [Google Scholar] [CrossRef]
- Schardijn, G.H.C.; Statius van Eps, L.W. β2-microglobulin: Its significance in the evaluation of renal function. Kidney Int. 1987, 32, 635–641. [Google Scholar] [CrossRef]
- Hall, P.W., III; Chung-Park, M.; Vacca, C.V.; London, M.; Crowley, A.Q. The renal handling of beta2-microglobulin in the dog. Kidney Int. 1982, 22, 156–161. [Google Scholar] [CrossRef] [PubMed]
- Gauthier, C.; Nguyen-Simonnet, H.; Vincent, C.; Revillard, J.-P.; Pellet, M.V. Renal tubular absorption of β2-microglobulin. Kidney Int. 1984, 26, 170–175. [Google Scholar] [CrossRef] [PubMed]
- Norden, A.G.W.; Lapsley, M.; Lee, P.J.; Pusey, C.D.; Scheinman, S.J.; Tam, F.W.K.; Thakker, R.V.; Unwin, R.J.; Wrong, O. Glomerular protein sieving and implications for renal failure in Fanconi syndrome. Kidney Int. 2001, 60, 1885–1892. [Google Scholar] [CrossRef] [PubMed]
- Wibell, L.; Evrin, P.-E.; Berggard, J. Serum β2-microglobulin in renal disease. Nephron 1973, 10, 320–331. [Google Scholar] [CrossRef]
- Wibell, L.B. Studies on β2-microglobulin in patients and normal subjects. Acta Clin. Belg. 1976, 31, 14–26. [Google Scholar]
- Wibell, L. The serum level and urinary excretion of β2-microglobulin in health and renal disease. Pathol. Biol. (Paris) 1978, 26, 295–301. [Google Scholar]
- Hall, P.W., III; Ricanati, E.S. Renal handling of β2-microglobulin in renal disorders with special reference to hepatorenal syndrome. Nephron 1981, 27, 62–66. [Google Scholar] [CrossRef]
- Portman, R.J.; Kissane, J.M.; Robson, A.M. Use of β2-microglobulin to diagnose tubulo-interstitial renal lesions in children. Kidney Int. 1986, 30, 91–98. [Google Scholar] [CrossRef]
- Bernard, A. Renal dysfunction induced by cadmium: Biomarkers of critical effects. Biometals 2004, 17, 519–523. [Google Scholar] [CrossRef]
- Nogawa, K.; Kobayashi, E.; Honda, R. A study of the relationship between cadmium concentrations in urine and renal effects of cadmium. Environ. Health Perspect. 1979, 28, 161–168. [Google Scholar] [CrossRef]
- Ikeda, M.; Ezaki, T.; Moriguchi, J.; Fukui, Y.; Ukai, H.; Okamoto, S.; Sakurai, H. The threshold cadmium level that causes a substantial increase in β2-microglobulin in urine of general populations. Tohoku J. Exp. Med. 2005, 205, 247–261. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Peterson, P.A.; Evrin, P.-E.; Berggard, I. Differentiation of glomerular, tubular, and normal proteinuria: Determinations of urinary excretion of β2-microglobulin, albumin, and total protein. J. Clin. Investig. 1969, 48, 1189–1198. [Google Scholar] [CrossRef] [PubMed]
- Elinder, C.G.; Edling, C.; Lindberg, E.; Agedal, B.K.; Vesterberg, A. Assessment of renal function in workers previously exposed to cadmium. Br. J. Ind. Med. 1985, 42, 754–760. [Google Scholar] [CrossRef]
- Norden, A.G.W.; Lapsley, M.; Unwin, R.J. Urine retinol-binding protein 4: A functional biomarker of the proximal renal tubule. Adv. Clin. Chem. 2014, 63, 85–122. [Google Scholar] [PubMed]
- Blaner, W.S. Retinol-binding protein: The serum transport protein for vitamin A. Endocri. Rev. 1989, 10, 308–316. [Google Scholar] [CrossRef]
- Pallet, N.; Chauvet, S.; Chasse, J.-F.; Vincent, M.; Avilloach, P.; Levi, C.; Meas-Yedid, V.; Olivo-Marin, J.-C.; Nga-Matsogo, D.; Beaune, P.; et al. Urinary retinol binding protein is a marker of the extent of interstitial kidney fibrosis. PLoS ONE 2014, 9, e84708. [Google Scholar] [CrossRef]
- Bernard, A.; Vyskocyl, A.; Mahieu, P.; Lauwerys, R. Effect of renal insufficiency on the concentration of free retinol-binding protein in urine and serum. Clin. Chim. Acta 1988, 171, 85–94. [Google Scholar] [CrossRef]
- Jarup, L.; Akesson, A. Current status of cadmium as an environmental health problem. Toxicol. Appl. Pharmacol. 2009, 238, 201–208. [Google Scholar] [CrossRef]
- Heymsfield, S.B.; Arteaga, C.; McManus, C.; Smith, J.; Moffitt, S. Measurement of muscle mass in humans: Validity of the 24-hour urinary creatinine method. Am. J. Clin. Nutr. 1983, 37, 478–494. [Google Scholar] [CrossRef]
- Jenny-Burri, J.; Haldimann, M.; Bruschweiler, B.J.; Bochuyd, M.; Burnier, M.; Paccaud, F.; Dudler, V. Cadmium body burden of the Swiss population. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2015, 32, 1265–1272. [Google Scholar] [CrossRef]
- Chevalier, R.L.; Forbes, M.S. Generation and evolution of atubular glomeruli in the progression of renal disorders. J. Am. Soc. Nephrol. 2008, 19, 197–206. [Google Scholar] [CrossRef]
- Ferenbach, D.A.; Bonventre, J.V. Acute kidney injury and chronic kidney disease: From the laboratory to the clinic. Nephrol. Ther. 2016, 12 (Suppl. 1), S41–S48. [Google Scholar] [CrossRef]
- Zammouri, A.; Barbouch, S.; Najjar, M.; Aoudia, R.; Jaziri, F.; Kaaroud, H.; Hedri, H.; Abderrahim, E.; Goucha, R.; Hamida, F.B.; et al. Tubulointerstitial nephritis due to sarcoidosis: Clinical, laboratory, and histological features and outcome in a cohort of 24 patients. Saudi J. Kidney Dis. Transpl. 2019, 30, 1276–1284. [Google Scholar] [CrossRef]
- Goules, A.; Geetha, D.; Arend, L.J.; Baer, A.N. Renal involvement in primary Sjogren’s syndrome: Natural history and treatment outcome. Clin. Exp. Rheumatol. 2019, 37 (Suppl. 118), S123–S132. [Google Scholar]
- Jasiek, M.; Karras, A.; Le Guern, V.; Krastinova, E.; Mesbah, R.; Faguer, S.; Jourde-Chiche, N.; Fauchais, A.-L.; Chiche, L.; Dernis, E.; et al. A multicentre study of 95 biopsy-proven cases of renal disease in primary Sjogren’s syndrome. Rheumatology 2017, 56, 362–370. [Google Scholar] [CrossRef] [PubMed]
- Kelly, C.J.; Neilson, E.G. Tubulointerstitial diseases. In Brenner & Rector’s The Kidney, 10th ed.; Taal, M.W., Chertow, G.M., Marsden, P.A., Skorecki, K., Alan, S.L., Brenner, B.M., Eds.; Elsevier: Philadelphia, PA, USA, 2011; pp. 1209–1230. [Google Scholar]
- Nath, K.A. Tubulointerstitial changes as a major determinant in the progression of renal damage. Am. J. Kidney Dis. 1992, 20, 1–17. [Google Scholar] [CrossRef]
- Risdon, R.A.; Sloper, J.C.; De Wardener, H.E. Relationship between renal function and histological changes found in renal-biopsy specimens from patients with persistent glomerular nephritis. Lancet 1968, 292, 363–366. [Google Scholar] [CrossRef]
- Schainuck, L.I.; Striker, G.E.; Cutler, R.E.; Benditt, E.P. Structural-functional correlations in renal disease: Part II: The correlations. Human Pathol. 1970, 1, 631–641. [Google Scholar] [CrossRef]
- Bohle, A.; von Gise, H.; Mackensen-Haen, S.; Stark-Jakob, B. The obliteration of the postglomerular capillaries and its influence upon the function of both glomeruli and tubuli. Functional interpretation of morphologic findings. Klin. Wochenschr. 1981, 59, 1043–1051. [Google Scholar] [CrossRef]
- Baba, H.; Tsuneyama, K.; Kumada, T.; Aoshima, K.; Imura, J. Histopathological analysis for osteomalacia and tubulopathy in itai-itai disease. J. Toxicol. Sci. 2014, 39, 91–96. [Google Scholar] [CrossRef]
- Yasuda, M.; Miwa, A.; Kitagawa, M. Morphometric studies of renal lesions in itai-itai disease: Chronic cadmium nephropathy. Nephron 1995, 69, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Saito, H.; Shioji, R.; Hurukawa, Y.; Nagai, K.; Arikawa, T. Cadmium-induced proximal tubular dysfunction in a cadmium-polluted area. Contrib. Nephrol. 1977, 6, 1–12. [Google Scholar] [PubMed]
- Nogawa, K.; Ishizaki, A.; Fukushima, M.; Shibata, I.; Hagino, N. Studies on the women with acquired Fanconi syndrome observed in the Ichi River basin polluted by cadmium. Environ. Res. 1975, 10, 280–307. [Google Scholar] [CrossRef]
- Rappaport, S.M.; Smith, M.T. Environment and disease risks. Science 2010, 330, 460–461. [Google Scholar] [CrossRef]
- Lee, J.; Oh, S.; Kang, H.; Kim, S.; Lee, G.; Li, L.; Kim, C.T.; An, J.N.; Oh, Y.K.; Lim, C.S.; et al. Environment-wide association study of CKD. Clin. J. Am. Soc. Nephrol. 2020, 15, 766–775. [Google Scholar] [CrossRef]
- Soderland, P.; Lovekar, S.; Weiner, D.E.; Brooks, D.R.; Kaufman, J.S. Chronic kidney disease associated with environmental toxins and exposures. Adv. Chronic Kidney Dis. 2010, 17, 254–264. [Google Scholar] [CrossRef]
- Chevalier, R.L. The proximal tubule is the primary target of injury and progression of kidney disease: Role of the glomerulotubular junction. Am. J. Physiol. Renal. Physiol. 2016, 311, F145–F161. [Google Scholar] [CrossRef]
- Crowley, S.D.; Coffman, T.M. The inextricable role of the kidney in hypertension. J. Clin. Investig. 2014, 124, 2341–2347. [Google Scholar]
- Nakhoul, N.; Batuman, V. Role of proximal tubules in the pathogenesis of kidney disease. Contrib. Nephrol. 2011, 169, 37–50. [Google Scholar]
- De Nicola, L.; Zoccali, C. Chronic kidney disease prevalence in the general population: Heterogeneity and concerns. Nephrol. Dial. Transplant. 2016, 31, 331–335. [Google Scholar] [CrossRef]
- Glassock, R.J.; David, G.; Warnock, D.G.; Delanaye, P. The global burden of chronic kidney disease: Estimates, variability and pitfalls. Nat. Rev. Nephrol. 2017, 13, 104–114. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.K.; Knicely, D.H.; Grams, M.E. Chronic kidney disease diagnosis and management: A review. JAMA 2019, 322, 1294–1304. [Google Scholar] [CrossRef] [PubMed]
- Lees, J.S.; Welsh, C.E.; Celis-Morales, C.A.; Mackay, D.; Lewsey, J.; Gray, S.R.; Lyall, D.M.; Cleland, J.G.; Gill, J.M.R.; Jhund, P.S.; et al. Glomerular filtration rate by differing measures, albuminuria and prediction of cardiovascular disease, mortality and end-stage kidney disease. Nat. Med. 2019, 25, 1753–1760. [Google Scholar] [CrossRef] [PubMed]
- Sarnak, M.J.; Amann, K.; Bangalore, S.; Cavalcante, J.L.; Charytan, D.M.; Craig, J.C.; Gill, J.S.; Hlatky, M.A.; Jardine, A.G.; Landmesser, U.; et al. Chronic kidney disease and coronary artery disease: JACC state-of-the-art review. J. Am. Coll. Cardiol. 2019, 74, 1823–1838. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Lv, P.; Jin, H.; Cui, W.; Niu, C.; Zhao, M.; Fan, C.; Teng, Y.; Pan, B.; Peng, Q.; et al. Association between low estimated glomerular filtration rate and risk of cerebral small-vessel diseases: A meta-analysis. J. Stroke Cerebrovasc. Dis. 2016, 25, 710–716. [Google Scholar] [CrossRef] [PubMed]
- Kelly, D.M.; Rothwell, P.M. Does chronic kidney disease predict stroke risk independent of blood pressure? A systematic review and meta-regression. Stroke 2019, 50, 3085–3092. [Google Scholar] [CrossRef] [PubMed]
- Akoudad, S.; Sedaghat, S.; Hofman, A.; Koudstaal, P.J.; van der Lugt, A.; Ikram, M.A.; Vernooij, M.W. Kidney function and cerebral small vessel disease in the general population. Int. J. Stroke 2015, 10, 603–608. [Google Scholar] [CrossRef] [PubMed]
- Sedaghat, S.; Ding, J.; Eiriksdottir, G.; van Buchem, M.A.; Sigurdsson, S.; Ikram, M.A.; Meirelles, O.; Gudnason, V.; Levey, A.S.; Launer, L.J. The AGES-Reykjavik study suggests that change in kidney measures is associated with subclinical brain pathology in older community-dwelling persons. Kidney Int. 2018, 94, 608–615. [Google Scholar] [CrossRef]
- White, C.A.; Allen, C.M.; Akbari, A.; Collier, C.P.; Holland, D.C.; Day, A.G.; Knoll, G.A. Comparison of the new and traditional CKD-EPI GFR estimation equations with urinary inulin clearance: A study of equation performance. Clin. Chim. Acta 2019, 488, 189–195. [Google Scholar] [CrossRef]
- George, C.; Mogueo, A.; Okpechi, I.; Echouffo-Tcheugui, J.B.; Kengne, A.P. Chronic kidney disease in low-income to middle-income countries: The case for increased screening. BMJ Glob. Health 2017, 2, e000256. [Google Scholar] [CrossRef]
- Payton, M.; Hu, H.; Sparrow, D.; Weiss, S.T. Low-level lead exposure and renal function in the Normative Aging Study. Am. J. Epidemiol. 1994, 140, 821–829. [Google Scholar] [CrossRef] [PubMed]
- Navas-Acien, A.; Tellez-Plaza, M.; Guallar, E.; Muntner, P.; Silbergeld, E.; Jaar, B.; Weaver, V. Blood cadmium and lead and chronic kidney disease in US adults: A joint analysis. Am. J. Epidemiol. 2009, 170, 1156–1164. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.J.; Wang, J.J.; Mao, J.H.; Shu, Q.; Du, L.Z. Relationships between cadmium, lead and mercury levels and albuminuria: Results from the National Health and Nutrition Examination Survey Database 2009−2012. Am. J. Epidemiol. 2019, 188, 1281–1287. [Google Scholar] [CrossRef] [PubMed]
- Harari, F.; Sallsten, G.; Christensson, A.; Petkovic, M.; Hedblad, B.; Forsgard, N.; Melander, O.; Nilsson, P.M.; Borné, Y.; Engström, G.; et al. Blood lead levels and decreased kidney function in a population-based cohort. Am. J. Kidney Dis. 2018, 72, 381–389. [Google Scholar] [CrossRef] [PubMed]
- Sommar, J.N.; Svensson, M.K.; Björ, B.M.; Elmståhl, S.I.; Hallmans, G.; Lundh, T.; Schön, S.M.; Skerfving, S.; Bergdahl, I.A. End-stage renal disease and low level exposure to lead, cadmium and mercury; a population-based, prospective nested case-referent study in Sweden. Environ. Health 2013, 12, 9. [Google Scholar] [CrossRef] [PubMed]
- Satarug, S.; Swaddiwudhipong, W.; Ruangyuttikarn, W.; Nishijo, M.; Ruiz, P. Modeling cadmium exposures in low- and high-exposure areas in Thailand. Environ. Health Perspect. 2013, 121, 531–536. [Google Scholar] [CrossRef] [PubMed]
- Swaddiwudhipong, W.; Nguntra, P.; Kaewnate, Y.; Mahasakpan, P.; Limpatanachote, P.; Aunjai, T.; Jeekeeree, W.; Punta, B.; Funkhiew, T.; Phopueng, I. Human health effects from cadmium exposure: Comparison between persons living in cadmium-contaminated and non-contaminated areas in northwestern Thailand. Southeast Asian J. Trop. Med. Public Health 2015, 46, 133–142. [Google Scholar]
- Sun, Y.; Sun, D.; Zhou, Z.; Zhu, G.; Lei, L.; Zhang, H.; Chang, X.; Jin, T. Estimation of benchmark dose for bone damage and renal dysfunction in a Chinese male population occupationally exposed to lead. Ann. Occup. Hyg. 2008, 52, 527–533. [Google Scholar]
- Chen, X.; Zhu, G.; Wang, Z.; Zhou, H.; He, P.; Liu, Y.; Jin, T. The association between lead and cadmium co-exposure and renal dysfunction. Ecotoxicol. Environ. Saf. 2019, 173, 429–435. [Google Scholar] [CrossRef]
- Kim, N.H.; Hyun, Y.Y.; Lee, K.B.; Chang, Y.; Ryu, S.; Oh, K.H.; Ahn, C. Environmental heavy metal exposure and chronic kidney disease in the general population. J. Korean Med. Sci. 2015, 30, 272–277. [Google Scholar] [CrossRef]
- Myong, J.P.; Kim, H.R.; Baker, D.; Choi, B. Blood cadmium and moderate-to-severe glomerular dysfunction in Korean adults: Analysis of KNHANES 2005−2008 data. Int. Arch. Occup. Environ. Health 2012, 85, 885–893. [Google Scholar] [CrossRef] [PubMed]
- Chung, S.; Chung, J.H.; Kim, S.J.; Koh, E.S.; Yoon, H.E.; Park, C.W.; Chang, Y.S.; Shin, S.J. Blood lead and cadmium levels and renal function in Korean adults. Clin. Exp. Nephrol. 2014, 18, 726–734. [Google Scholar] [CrossRef]
- Lim, H.; Lim, J.A.; Choi, J.H.; Kwon, H.J.; Ha, M.; Kim, H.; Park, J.D. Associations of low environmental exposure to multiple metals with renal tubular impairment in Korean adults. Toxicol. Res. 2016, 32, 57–64. [Google Scholar] [CrossRef]
- Hambach, R.; Lison, D.; D’Haese, P.C.; Weyler, J.; De Graef, E.; De Schryver, A.; Lamberts, L.V.; van Sprundel, M. Co-exposure to lead increases the renal response to low levels of cadmium in metallurgy workers. Toxicol. Lett. 2013, 222, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Ferraro, P.M.; Sturniolo, A.; Naticchia, A.; D’Alonzo, S.; Gambaro, G. Temporal trend of cadmium exposure in the United States population suggests gender specificities. Intern. Med. J. 2012, 42, 691–697. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, S.; Zaman, T.; Tuzcu, E.M.; Kapadia, S.R. Heavy metals and cardiovascular disease: Results from the National Health and Nutrition Examination Survey (NHANES) 1999–2006. Angiology 2011, 62, 422–429. [Google Scholar] [CrossRef]
- Hecht, E.M.; Arheart, K.L.; Lee, D.J.; Hennekens, C.H.; Hlaing, W.M. Interrelation of cadmium, smoking, and cardiovascular disease (from the National Health and Nutrition Examination Survey). Am. J. Cardiol. 2016, 118, 204–209. [Google Scholar] [CrossRef]
- Hecht, E.M.; Arheart, K.L.; Lee, D.J.; Hennekens, C.H.; Hlaing, W.M. Interrelationships of cadmium, smoking, and angina in the National Health and Nutrition Examination Survey, a cross-sectional study. Cardiology 2018, 141, 177–182. [Google Scholar] [CrossRef]
- Chen, C.; Xun, P.; Tsinovoi, C.; McClure, L.A.; Brockman, J.; MacDonald, L.; Cushman, M.; Cai, J.; Kamendulis, L.; Mackey, J.; et al. Urinary cadmium concentration and the risk of ischemic stroke. Neurology 2018, 91, e382–e391. [Google Scholar] [CrossRef]
- Gallagher, C.M.; Chen, J.J.; Kovach, J.S. Environmental cadmium and breast cancer risk. Aging (Albany NY) 2010, 2, 804–814. [Google Scholar] [CrossRef]
- Tellez-Plaza, M.; Navas-Acien, A.; Menke, A.; Crainiceanu, C.M.; Pastor-Barriuso, R.; Guallar, E. Cadmium exposure and all-cause and cardiovascular mortality in the U.S. general population. Environ. Health Perspect. 2012, 120, 1017–1022. [Google Scholar] [CrossRef]
- Menke, A.; Muntner, P.; Silbergeld, E.K.; Platz, E.A.; Guallar, E. Cadmium levels in urine and mortality among U.S. adults. Environ. Health Perspect. 2009, 117, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Adams, S.V.; Passarelli, M.N.; Newcomb, P.A. Cadmium exposure and cancer mortality in the Third National Health and Nutrition Examination Survey cohort. Occup. Environ. Med. 2012, 69, 153–156. [Google Scholar] [CrossRef] [PubMed]
- Hyder, O.; Chung, M.; Cosgrove, D.; Herman, J.M.; Li, Z.; Firoozmand, A.; Gurakar, A.; Koteish, A.; Pawlik, T.M. Cadmium exposure and liver disease among US adults. J. Gastrointest. Surg. 2013, 17, 1265–1273. [Google Scholar] [CrossRef] [PubMed]
- Min, J.Y.; Min, K.B. Blood cadmium levels and Alzheimer’s disease mortality risk in older US adults. Environ. Health 2016, 15, 69. [Google Scholar] [CrossRef]
- Peng, Q.; Bakulski, K.M.; Nan, B.; Park, S.K. Cadmium and Alzheimer’s disease mortality in U.S. adults: Updated evidence with a urinary biomarker and extended follow-up time. Environ. Res. 2017, 157, 44–51. [Google Scholar] [CrossRef]
- Moberg, L.; Nilsson, P.M.; Samsioe, G.; Sallsten, G.; Barregard, L.; Engström, G.; Borgfeldt, C. Increased blood cadmium levels were not associated with increased fracture risk but with increased total mortality in women: The Malmö Diet and Cancer Study. Osteoporos Int. 2017, 28, 2401–2408. [Google Scholar] [CrossRef]
- Deering, K.E.; Callan, A.C.; Prince, R.L.; Lim, W.H.; Thompson, P.L.; Lewis, J.R.; Hinwood, A.L.; Devine, A. Low-level cadmium exposure and cardiovascular outcomes in elderly Australian women: A cohort study. Int. J. Hyg. Environ. Health 2018, 221, 347–354. [Google Scholar] [CrossRef]
- Suwazono, Y.; Nogawa, K.; Morikawa, Y.; Nishijo, M.; Kobayashi, E.; Kido, T.; Nakagawa, H.; Nogawa, K. All-cause mortality increased by environmental cadmium exposure in the Japanese general population in cadmium non-polluted areas. J. Appl. Toxicol. 2015, 35, 817–823. [Google Scholar] [CrossRef]
- Watanabe, Y.; Nogawa, K.; Nishijo, M.; Sakurai, M.; Ishizaki, M.; Morikawa, Y.; Kido, T.; Nakagawa, H.; Suwazono, Y. Relationship between cancer mortality and environmental cadmium exposure in the general Japanese population in cadmium non-polluted areas. Int. J. Hyg. Environ. Health 2020, 223, 65–70. [Google Scholar] [CrossRef]
- Maruzeni, S.; Nishijo, M.; Nakamura, K.; Morikawa, Y.; Sakurai, M.; Nakashima, M.; Kido, T.; Okamoto, R.; Nogawa, K.; Suwazono, Y.; et al. Mortality and causes of deaths of inhabitants with renal dysfunction induced by cadmium exposure of the polluted Jinzu River basin, Toyama, Japan; a 26-year follow-up. Environ. Health 2014, 13, 18. [Google Scholar] [CrossRef] [PubMed]
- Nishijo, M.; Nakagawa, H.; Suwazono, Y.; Nogawa, K.; Sakurai, M.; Ishizaki, M.; Kido, T. Cancer mortality in residents of the cadmium-polluted Jinzu River basin in Toyama, Japan. Toxics 2018, 6, 23. [Google Scholar] [CrossRef] [PubMed]
- van Bemmel, D.M.; Li, Y.; McLean, J.; Chang, M.H.; Dowling, N.F.; Graubard, B.; Rajaraman, P. Blood lead levels, ALAD gene polymorphisms, and mortality. Epidemiology 2011, 22, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Lanphear, B.P.; Rauch, S.; Auinger, P.; Allen, R.W.; Hornung, R.W. Low-level lead exposure and mortality in US adults: A population-based cohort study. Lancet Public Health 2018, 3, e177–e184. [Google Scholar] [CrossRef]
- Aoki, Y.; Brody, D.J.; Flegal, K.M. Blood lead and other metal biomarkers as risk factors for cardiovascular disease mortality. Medicine 2016, 95, e2223. [Google Scholar] [CrossRef]
- Kim, M.G.; Ryoo, J.H.; Chang, S.J.; Kim, C.B.; Park, J.K.; Koh, S.B.; Ahn, Y.S. Blood lead levels and cause-specific mortality of inorganic lead-exposed workers in South Korea. PLoS ONE 2015, 10, e0140360. [Google Scholar] [CrossRef]
- Min, Y.S.; Ahn, Y.S. The association between blood lead levels and cardiovascular diseases among lead-exposed male workers. Scand. J. Work Environ. Health 2017, 43, 385–390. [Google Scholar] [CrossRef]
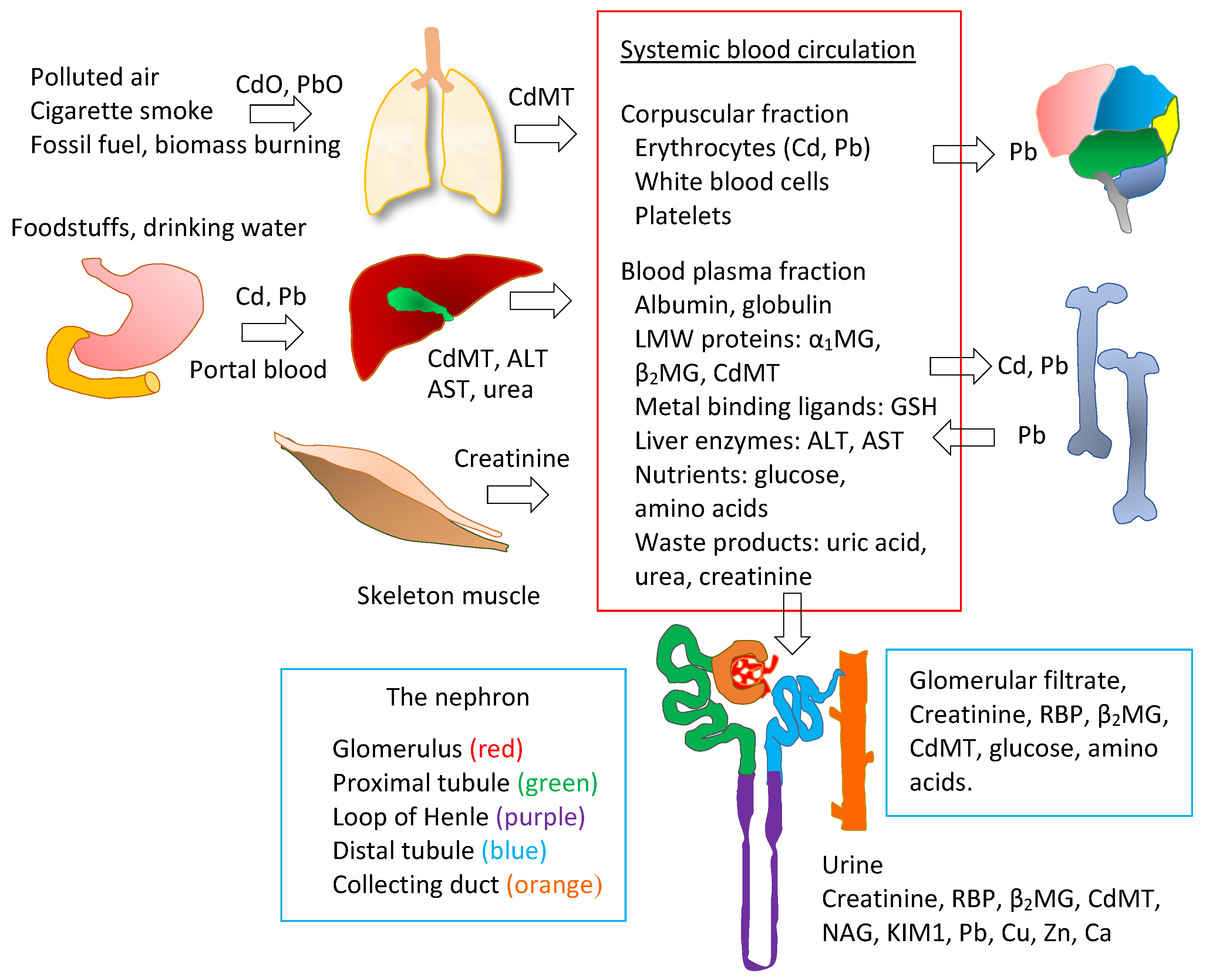
| Countries | Estimated Intake Levels as μg Per Day and Dietary Sources | |
|---|---|---|
| Cadmium (Atomic Weight 112.4) | Lead (Atomic Weight 207.2) | |
| China [89,90] 67% of population | Average consumers: 32.7 μg/day. Rice and vegetables as the main sources for most Chinese. Potato was the main source in Mongolia. High Cd foods: Nori, peanuts, squid, cuttlefish, and mushrooms. | Average consumers: 35.1 μg/day. Cereals, meats, vegetables, and beverages and water together contributed to 73.26% of total intake. High Pb foods: Kelp, nori, processed and preserved soybean, meat, and fungus. products. |
| Korea [92] n = 4867 | Average consumers: 12.6 μg/day. Sources: Grain and grain-based products (40.4%), vegetables and vegetable products (16.5%), and fish and shellfish (17.9%). High Cd foods: Seaweed, shellfish and crustaceans, molluscs, nuts and seeds, and flavourings, with median values of 594, 186, 155, 15.7, and 6.23 μg/kg, respectively. | Average consumers: 9.8 μg/day. High Pb foods: Seaweed, shellfish and crustaceans, molluscs, fish, and sugar and sugar products, with respective median values of 94.2, 91.4, 62.4, 8.13, and 4.61 μg/kg, while the median value for beverages (fruit juice, carbonated fruit juice, carbonated drinks, sports drinks, and coffee) was 11.0 μg/kg. |
| Germany [94,100] n = 15,371 | Average consumers: 14.6 μg/day. High consumers: 23.5 μg/day. Sources: Cereals and vegetables, beverages, fruits and nuts, and dairy products (milk included). High Cd foods: Cereals, oily seeds and fruits, and vegetables. | Average consumers: 37.1 μg/day. High consumers: 50.4 μg/day. Sources: Beverages, vegetables, fruits and nuts and cereals. High Pb foods: Meat (offal included), fish (seafood), vegetables and cereals. |
| Spain [95] n = 1281 | Average consumers: 7.7 μg/day. Sources: Cereals and fish contributed to 38% and 29% of total intake. High Cd foods: Cereals (16.25 μg/kg), fish group (11.40 μg/kg). | Average consumers: 14.7 μg/day. Cereals contributed to 49% of total intake. High Pb foods: Sweeteners and condiments, vegetable oils, meat, and fish, with respective median levels of 32.5, 15.25, 14.90 and 13.21 μg/kg. |
| U.S. [84,97] n = 14,614 FDA 2014–2016 total diet study | Average consumers: 4.63 μg/day. Sources: Cereals and bread, leafy vegetables, potatoes, legumes and nuts, stem/root vegetables, and fruits contributed to 34%, 20%, 11%, 7% and 6% of total intake, respectively. High Cd foods: Spaghetti, bread, potatoes and potato chips contributed the most to total Cd intake, followed by lettuce, spinach, tomatoes, and beer. Lettuce was a main Cd source for whites and blacks. Tortillas and rice were main Cd sources for Hispanic Americans, and Asians plus other ethnicities. Cd concentration of raw leaf lettuce and iceberg lettuce were 0.066 and 0.051 mg/kg, respectively. | Average consumers: 1.7−5.3 μg/day. High consumers: 3.2−7.8 μg/day. Sources: Grains, beverages, vegetables, dairy, fruits, meat, and poultry plus fish contributed to 24.1%, 14.3%, 10.7%, 9.7%, 9.3% and 3.4% to total intake, respectively. High Pb foods: Chocolate syrup, liver, canned sweet potatoes, brownies, low-calorie buttermilk, salad dressing, raisins, English muffins, canned apricots, milk chocolate, candy bars, chocolate cake, chocolate chip cookies, wine and oat ring cereal with respective median levels of 14, 14, 14, 13, 13, 12, 10, 10, 9, 8, 8, 7 and 7 μg/kg. |
| Countries | Exposure Levels and Estimates of Disease and Mortality Risks |
|---|---|
| U.S. [44,45,46,289] | [Cd]u ≥ 1 µg/L, [Cd]b ≥ 0.6 μg/L, [Pb]b ≥ 2.4 μg/dL and [Cd]b ≥ 0.6 plus [Pb]b ≥ 2.4 μg/dL were associated with 1.48-, 1.32-,1.56- and 2.34-fold increment in CKD risk, respectively. [Cd]b of >0.53 to >0.61 μg/L were associated with 1.80- to 2.2-fold increases in CKD risk. |
| U.S. [302,308,309,310,311,312,313,319,320,321] | ECd of ≥0.37 to ≥0.65 μg/g creatinine were associated with increased mortality from heart disease. ECd of >0.48 and ≥0.58 µg/g creatinine were linked to 4.29-fold and 3.22-fold increments in cancer mortality and lung cancer mortality in men, respectively. [Cd]b > 0.6 μg/L was linked to a 3.83-fold increase in mortality from Alzheimer’s disease. [Pb]b ≥ 5 μg/dL was linked to a 1.48-fold increment of cancer mortality[Pb]b 1.0−6.7 μg/dL were linked to 1.37-, 1.70- and 2.08-fold increments of morality from all causes and cardiovascular and ischaemic heart diseases. [Pb]u levels >1.26 μg/L were linked to 1.79- and 6.60-fold increments of mortality from all causes and cancer, respectively. |
| Sweden [291,292,314] | [Pb]b ≥ 3.3 μg/dL was associated with a 1.49-fold rise of incidence of CKD and erythrocyte Pb was associated with developing ESKD. [Cd]b ≥ 0.69 μg/L was linked to a 2.06-fold increase in mortality from all causes. |
| Australia [315] | A 2.7-fold higher [Cd]u were linked to a 36% increase in mortality from heart failure and a 17% increase in the risk of having a heart failure event. |
| Japan [316,317,318,319] | ECd of ≥3.23 and ≥4.66 µg/g creatinine were linked to increased mortality by 64%, and 49% in men and women, respectively. The mortality from pancreatic cancer in women rose by 13% for every 1 μg/g creatinine increase in ECd. In women with signs of Cd-related kidney pathologies, there were 3.85-, 7.71- and 10.1-fold increases in mortality from cancer of the uterus, kidney, and kidney plus urinary tract, respectively. |
| China [15,16] | Cd intake levels of 23.2, 29.6 and 36.9 μg/day were associated with 1.73-, 2.93- and 4.05-fold increments of CKD risk, For every 30 μg/day intake of Pb, all-cause mortality rose by 25%. Pb intake levels of 111.4 and 147 μg/day were linked to 1.52- and 3-fold increases in cancer mortality. |
| Thailand [53] | Eβ2MG of 100–299, 300–999 and ≥1000 μg/g creatinine were associated with 4.66-, 6.16-, and 11.47-fold increases in CKD risk, compared with Eβ2MG < 100 μg/g creatinine. An inverse association of Eβ2MG with eGFR was seen only in those with eGFR below 60 mL/min/1.73 m2, indicative of nephron loss. Eβ2MG did not show an association with eGFR in those with normal eGFR. |
| Belgium [301] | Associations of [Cd]u with [NAG]u and [RBP]u were seen in workers who had [Pb]b ≥ 21.9 μg/dL, corresponding to the 75th percentile or higher. |
| Korea [300] | A correlation between [Cd]b and [β2MG]u was strengthened in those who had [Pb]b above the median of 2.20 μg/dL. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Satarug, S.; C. Gobe, G.; A. Vesey, D.; Phelps, K.R. Cadmium and Lead Exposure, Nephrotoxicity, and Mortality. Toxics 2020, 8, 86. https://doi.org/10.3390/toxics8040086
Satarug S, C. Gobe G, A. Vesey D, Phelps KR. Cadmium and Lead Exposure, Nephrotoxicity, and Mortality. Toxics. 2020; 8(4):86. https://doi.org/10.3390/toxics8040086
Chicago/Turabian StyleSatarug, Soisungwan, Glenda C. Gobe, David A. Vesey, and Kenneth R. Phelps. 2020. "Cadmium and Lead Exposure, Nephrotoxicity, and Mortality" Toxics 8, no. 4: 86. https://doi.org/10.3390/toxics8040086
APA StyleSatarug, S., C. Gobe, G., A. Vesey, D., & Phelps, K. R. (2020). Cadmium and Lead Exposure, Nephrotoxicity, and Mortality. Toxics, 8(4), 86. https://doi.org/10.3390/toxics8040086







