Heavy Metal Mixture Exposure and Effects in Developing Nations: An Update
Abstract
1. Introduction
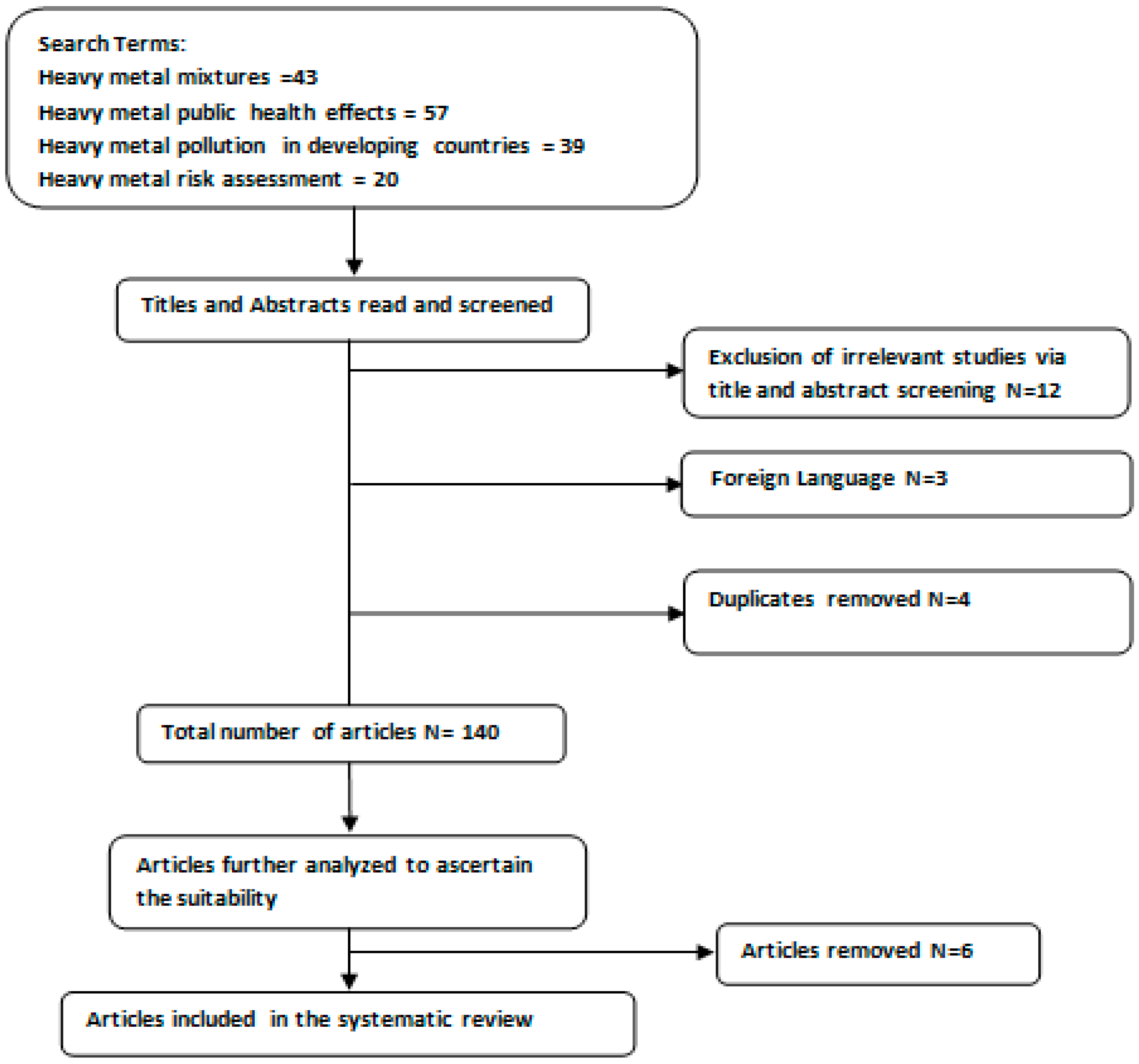
2. Materials and Methods
2.1. Database Searching and Search Strategy
2.2. Inclusion and Exclusion Criteria
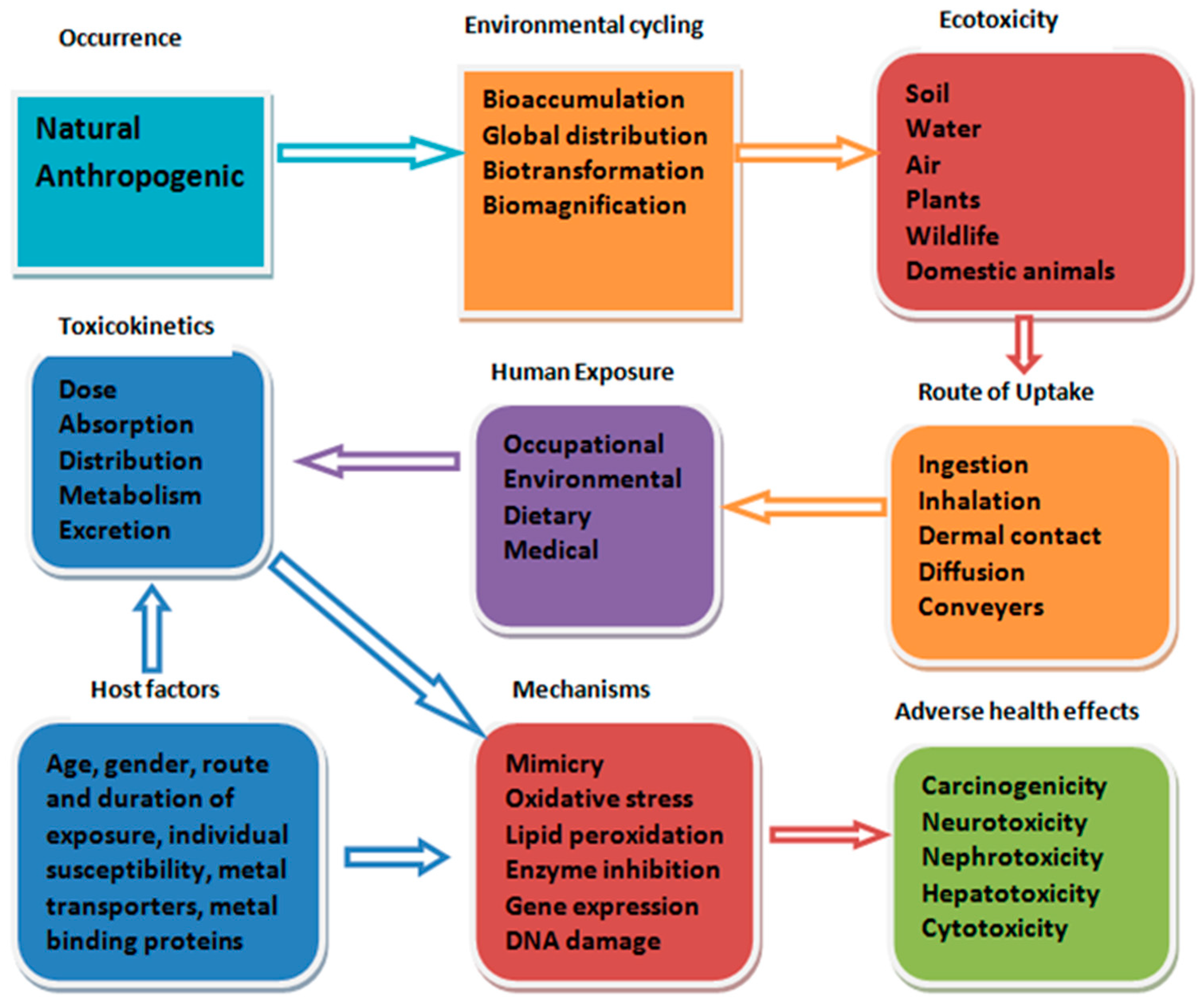
3. Exposure to Heavy Metal Mixture
4. Effects of Heavy Metal Mixture to Tissues and Organs
4.1. Cytotoxicity
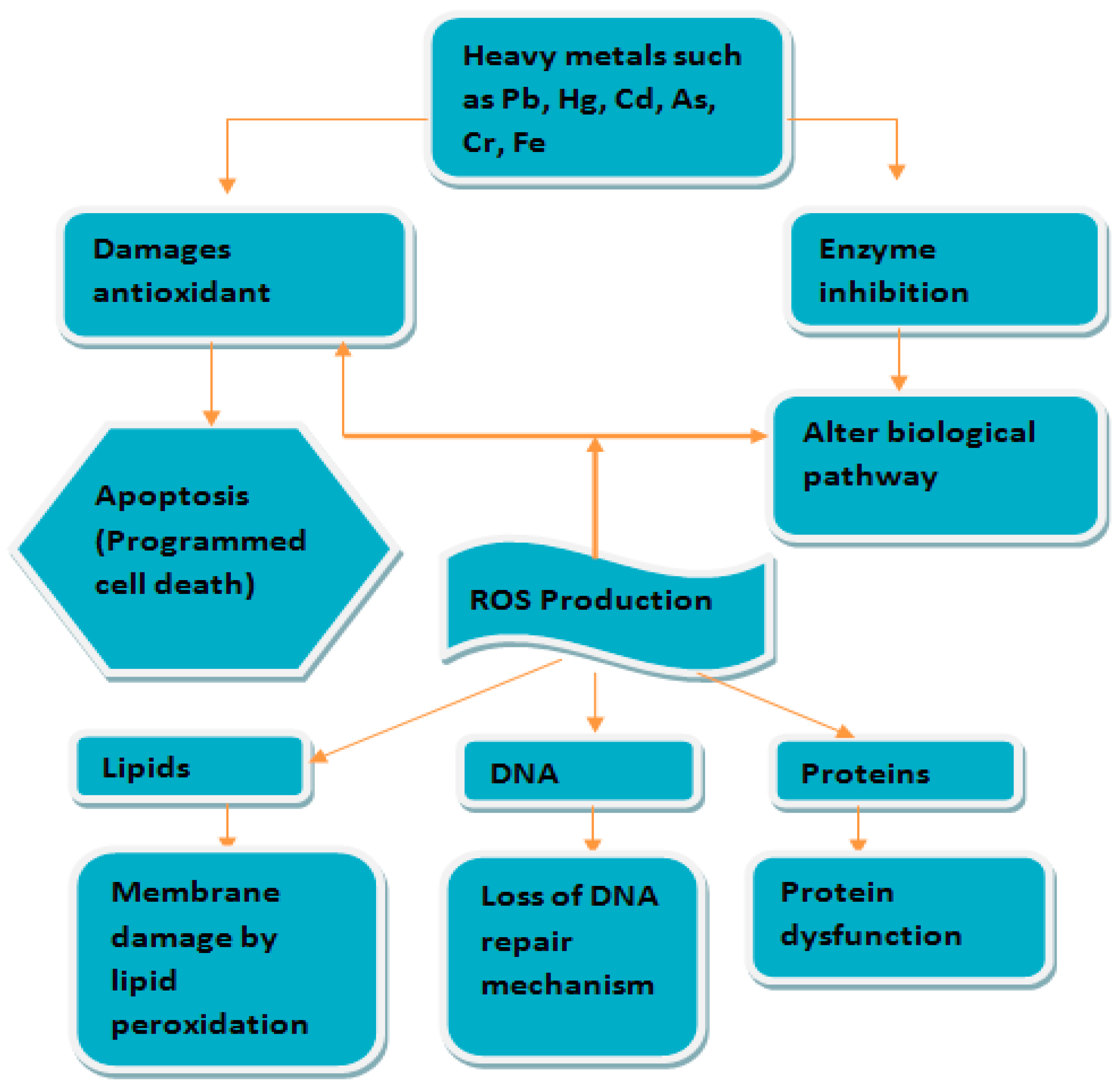
4.2. Oxidative Stress
4.3. Immunotoxicity
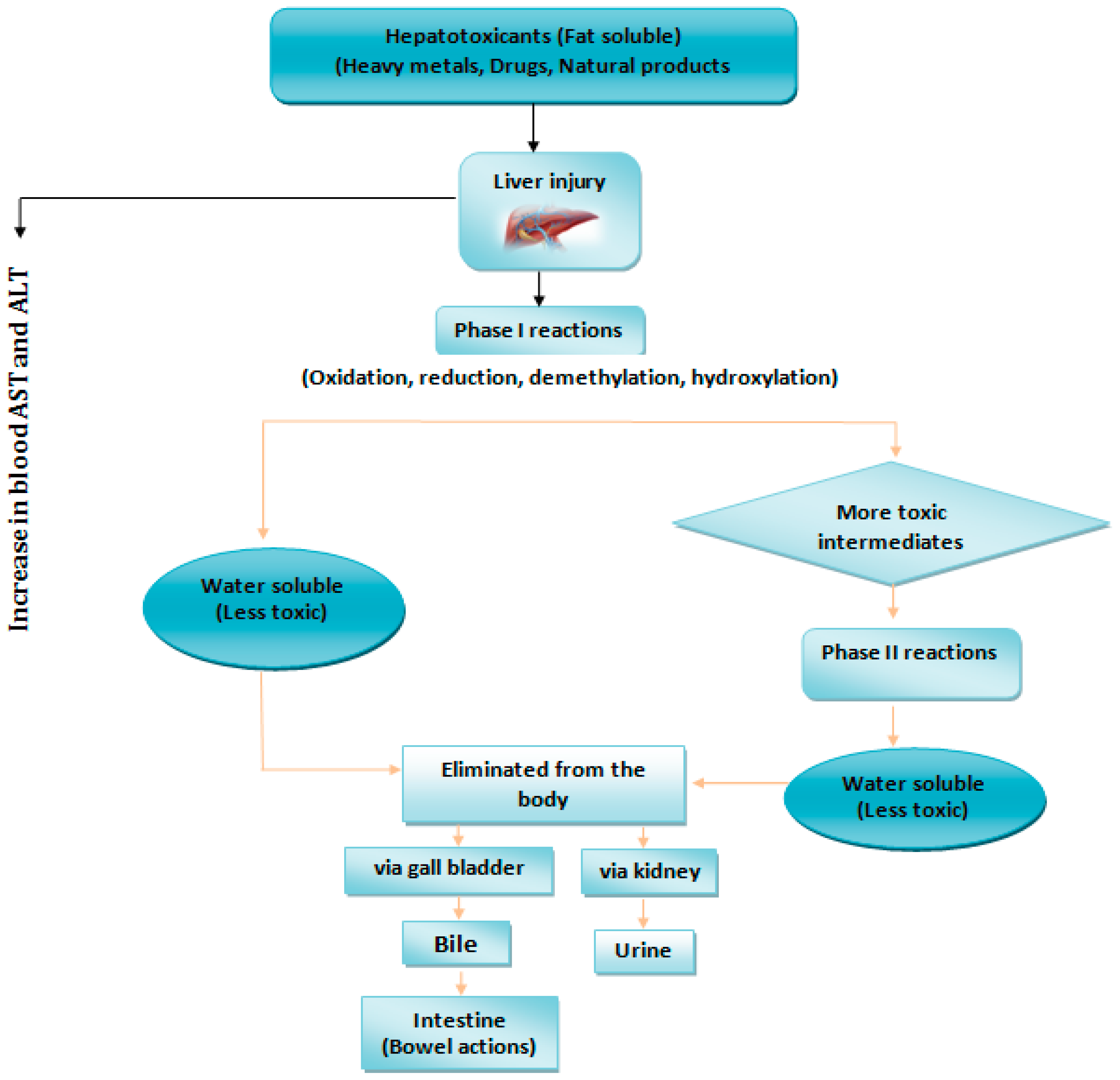
4.4. Hepatotoxicity
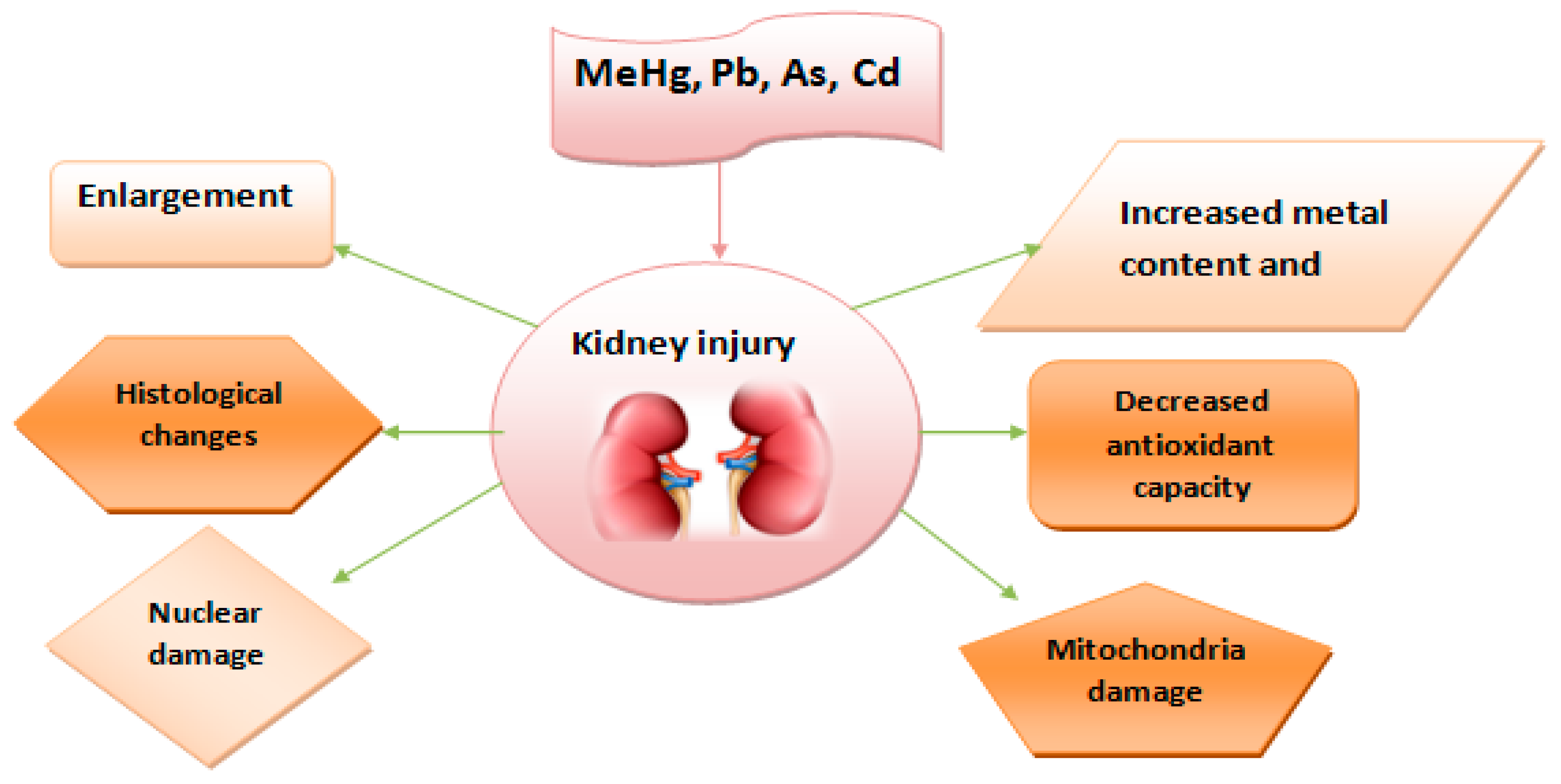
4.5. Nephrotoxicity
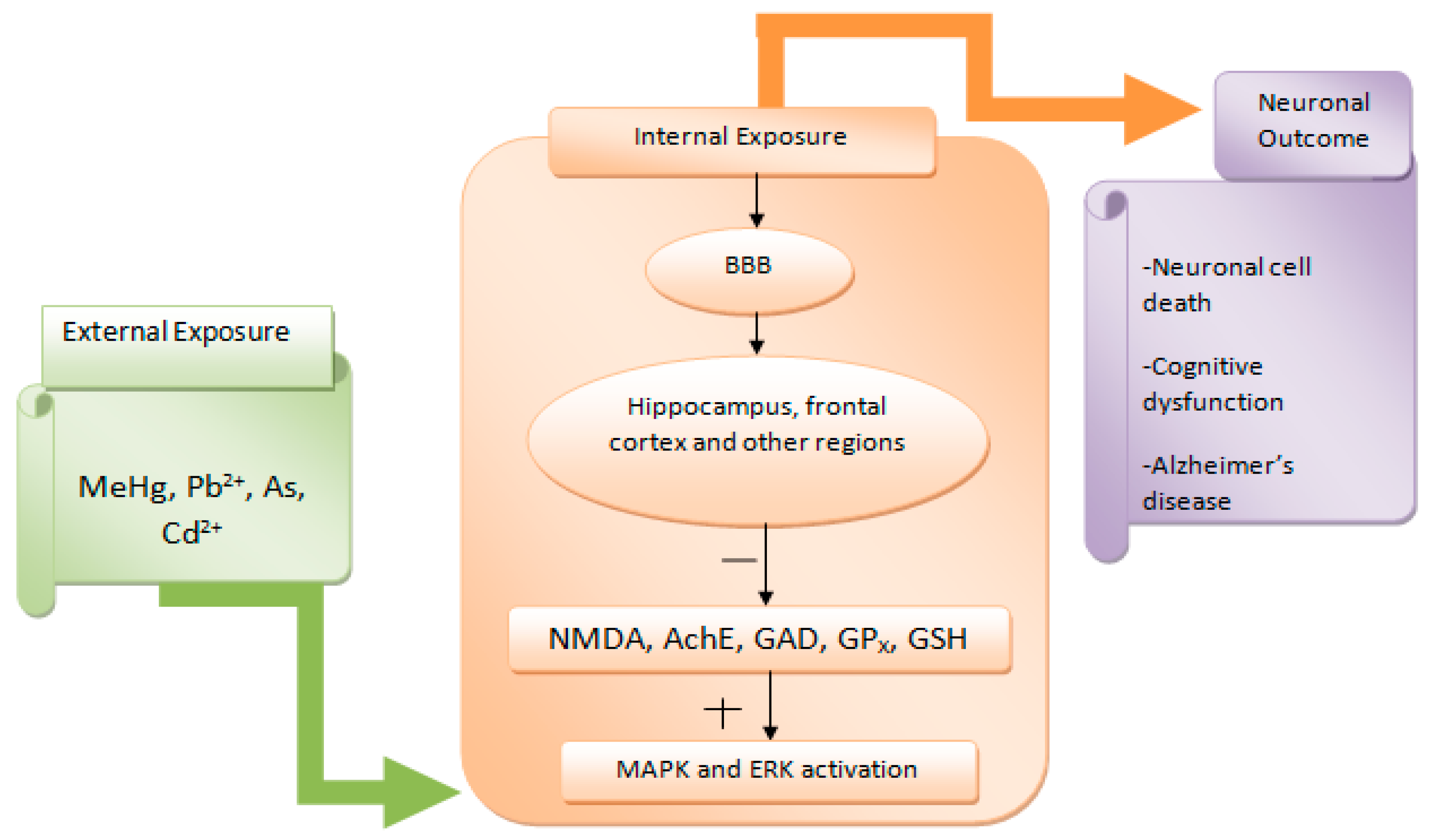
4.6. Neurotoxicity
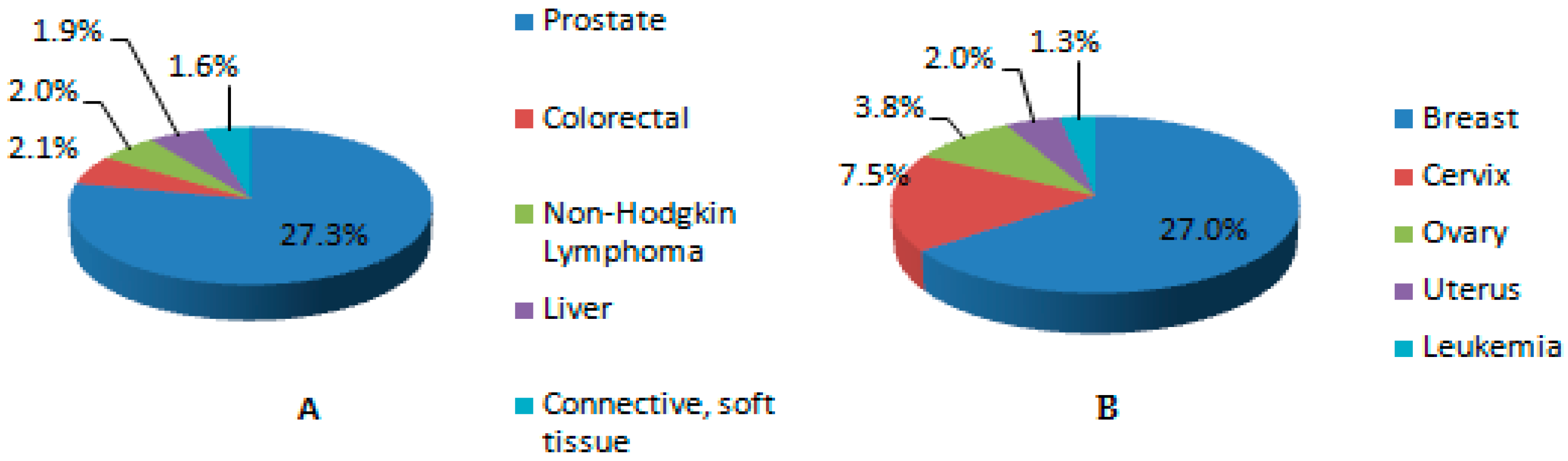
4.7. Development of Cancer
5. Public Health Effects of Heavy Metal Exposure in Sub Saharan Africa
6. Risk Assessment of Exposure to Mixtures of Heavy Metal
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Fasinu, P.S.; Orisakwe, O.E. Heavy metal pollution in sub-Saharan Africa and possible implications in cancer epidemiology. Asian Pac. J. Cancer Prev. 2013, 14, 3393–3402. [Google Scholar] [CrossRef] [PubMed]
- Jan, A.T.; Azam, M.; Siddiqui, K.; Ali, A.; Choi, I.; Haq, Q.M.R. Heavy metals and human health: Mechanistic insight into toxicity and counter defense system of antioxidants. Int. J. Mol. Sci. 2015, 16, 29592–29630. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Cobbina, S.J.; Mao, G.; Xu, H.; Zhang, Z.; Yang, L. A review of toxicity and mechanisms of individual and mixtures of heavy metals in the environment. Environ. Sci. Pollut. Res. 2016, 23, 8244–8259. [Google Scholar] [CrossRef] [PubMed]
- Jaishankar, M.; Mathew, B.B.; Shah, M.S.; Murthy, K.T.P.; Gowda, K.R.S. Biosorption of few heavy metal ions using agricultural wastes. J. Environ. Pollut. Hum. Health 2014, 2, 1–6. [Google Scholar]
- Nagajyoti, P.C.; Lee, K.D.; Sreekanth, T.V.M. Heavy metals, occurrence andtoxicity for plants: A review. Environ. Chem. Lett. 2010, 8, 199–216. [Google Scholar] [CrossRef]
- Jan, A.T.; Ali, A.; Haq, Q.M.R. Glutathione as an Antioxidant in Inorganic Mercury Induced Nephrotoxicity. J. Postgrad. Med. 2011, 57, 72–77. [Google Scholar] [PubMed]
- Martinez-Finley, E.J.; Chakraborty, S.; Fretham, S.J.; Aschner, M. Cellular transport and homeostasis of essential and nonessential metals. Metallomics 2012, 4, 593–605. [Google Scholar] [CrossRef] [PubMed]
- Fairbrother, A.; Wenstel, R.; Sappington, K.; Wood, W. Framework for metals risk assessment. Ecotoxicol. Environ. Saf. 2007, 68, 145–227. [Google Scholar] [CrossRef] [PubMed]
- Yabe, J.; Ishizuka, M.; Umemura, T. Current levels of heavy metal pollution in Africa. J. Vet. Med. Sci. 2010, 72, 1257–1263. [Google Scholar] [CrossRef] [PubMed]
- United Nation Environment Programme (UNEP). Environmental Assessment of Ogoniland. 2011. Available online: http://postconflict.unep.ch/publications/OEA/UNEP_OEA.pdf (accessed on 4 August 2018).
- Njoku, C.O.; Orisakwe, O.E. Higher blood lead levels in rural than urban pregnant women in eastern Nigeria. Occup. Environ. Med. 2012, 69, 850–851. [Google Scholar] [CrossRef]
- Ibeto, C.N.; Okoye, C.O.B. High levels of Heavy metals in Blood of Urban population in Nigeria. Res. J. Environ. Sci. 2010, 4, 371–382. [Google Scholar] [CrossRef]
- Röllin, H.B.; Rudge, C.V.; Thomassen, Y.; Mathee, A.; Odland, J.Ø. Levels of toxicand essential metals in maternal and umbilical cord blood from selected areas of South Africa—Results of a pilot study. J. Environ. Monit. 2009, 11, 618–627. [Google Scholar] [CrossRef] [PubMed]
- Cheremisinoff, N.P. Agency for Toxic Substances and Disease Registry (Atsdr); John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2016; pp. 83–93. [Google Scholar]
- Løkke, H.; Ragas, A.M.; Holmstrup, M. Tools and perspectives for assessing chemical mixture sand multiple stressors. Toxicology 2013, 313, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Hernández-García, A.; Romero, D.; Gómez-Ramírez, P.; María-Mojica, P.; Martínez-López, E.; García-Fernández, A.J. In vitro evaluation of cell death induced by cadmium, lead and their binary mixtures on erythrocytes of Common buzzard (Buteo buteo). Toxicol. Vitro 2014, 28, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Jadhav, S.H.; Sarkar, S.N.; Tripathi, H.C. Cytogenetic Effects of a Mixture of Selected Metals Following Subchronic Exposure through Drinking Water in Male rats. Indian J. Exp. Biol. 2006, 44, 997–1005. [Google Scholar] [PubMed]
- Dantzer, R.; O’Connor, J.C.; Freund, G.G.; Johnson, R.W.; Kelley, K.W. From inflammation to sickness and depression: When the immune system subjugates the brain. Nat. Rev. Neurosci. 2008, 9, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Heyen, J.R.; Ye, S.M.; Finck, B.N.; Johnson, R.W. Interleukin (IL)-10 inhibits IL-6 production in microglia by preventing activation of NF-κB. Mol. Brain Res. 2000, 77, 138–147. [Google Scholar] [CrossRef]
- Bluthé, R.M.; Castanon, N.; Pousset, F.; Bristow, A.; Ball, C.; Lestage, J.; Dantzer, R. Central injection of IL-10 antagonizes the behavioural effects of lipopolysaccharide in rats. Psychoneuroendocrinology 1999, 24, 301–311. [Google Scholar] [CrossRef]
- Dantzer, R.; Gheusi, G.; Johnson, R.W.; Kelley, K.W. Central administration of insulin-like growth factor-1 inhibits lipopolysaccharide-induced sickness behavior in mice. NeuroReport 1999, 10, 289–292. [Google Scholar] [CrossRef] [PubMed]
- Ye, S.M.; Johnson, R.W. Increased interleukin-6 expression by microglia from brain of aged mice. J. Neuroimmunol. 1999, 93, 139–148. [Google Scholar] [CrossRef]
- Ye, S.M.; Johnson, R.W. An age-related decline in interleukin-10 may contribute to the increased expression of interleukin-6 in brain of aged mice. Neuroimmunomodulation 2001, 9, 183–192. [Google Scholar] [CrossRef] [PubMed]
- Fowler, B.A.; Whittaker, M.H.; Lipsky, M.; Wang, G.; Chen, X.Q. Oxidative stress induced by lead, cadmium and arsenic mixtures: 30-day, 90-day, and 180-day drinking water studies in rats: An overview. Biometals 2004, 17, 567–568. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, T.; Naito, Y. What is oxidative stress? Jpn. Med. Assoc. J. 2002, 45, 271–276. [Google Scholar]
- Tan, S.; Sagara, Y.; Liu, Y.; Maher, P.; Schubert, D. The regulation of reactive oxygen species production during programmed cell death. J. Cell Boil. 1998, 141, 1423–1432. [Google Scholar] [CrossRef]
- Jadhav, S.H.; Sarkar, S.N.; Patil, R.D.; Tripathi, H.C. Effects of subchronic exposure via drinking water to a mixture of eight water-contaminating metals: A biochemical and histopathological study in male rats. Arch. Environ. Contam. Toxicol. 2007, 53, 667–677. [Google Scholar] [CrossRef] [PubMed]
- Aykin-Burns, N.; Laegeler, A.; Kellogg, G.; Ercal, N. Oxidative effects of lead in young and adult Fisher 344 rats. Arch. Environ. Contam. Toxicol. 2003, 44, 417–420. [Google Scholar] [CrossRef] [PubMed]
- Santra, A.; Maiti, A.; Das, S.; Lahiri, S.; Charkaborty, S.K.; Guha Mazumder, D.N.; GuhaMazumder, D. Hepatic damage caused by chronic arsenic toxicity inexperimentalanimals. J. Toxicol. Clin. Toxicol. 2000, 38, 395–405. [Google Scholar] [CrossRef] [PubMed]
- Bagchi, D.; Vuchetich, P.J.; Bagchi, M.; Hassoun, E.A.; Tran, M.X.; Tang, L.; Stohs, S.J. Induction of oxidative stress by chronic administration of sodium dichromate [chromium VI] and cadmium chloride [cadmium II] to rats. Free Radic. Biol. Med. 1997, 22, 471–478. [Google Scholar] [CrossRef]
- Bagchi, D.; Stohs, S.J.; Downs, B.W.; Bagchi, M.; Preuss, H.G. Cytotoxicity and oxidative mechanisms of different forms of chromium. Toxicology 2002, 180, 5–22. [Google Scholar] [CrossRef]
- Chen, C.Y.; Wang, Y.F.; Huang, W.R.; Huang, Y.T. Nickel induces oxidative stress and genotoxicity in human lymphocytes. Toxicol. Appl. Pharmacol. 2003, 189, 153–159. [Google Scholar] [CrossRef]
- Zhang, Z.; Wei, T.; Hou, J.; Li, G.; Yu, S.; Xin, W. Iron-induced oxidative damage and apoptosis in cerebellar granule cells: Attenuation by tetramethylpyrazine and ferulic acid. Eur. J. Pharmacol. 2003, 467, 41–47. [Google Scholar] [CrossRef]
- Mathew, B.B.; Tiwari, A.; Jatawa, S.K. Free radicals and antioxidants: A review. J. Pharm. Res. 2011, 4, 4340–4343. [Google Scholar]
- Valko, M.; Morris, H.; Cronin, M.T.D. Metals, toxicity and oxidative stress. Curr. Med. Chem. 2005, 12, 1161–1208. [Google Scholar] [CrossRef] [PubMed]
- Mates, J.M. Effects of antioxidant enzymes in the molecular control of reactive oxygen species toxicology. Toxicology 2000, 153, 83–104. [Google Scholar] [CrossRef]
- Bhattacharyya, A.; Chattopadhyay, R.; Mitra, S.; Crowe, S.E. Oxidative stress: An essential factor in the pathogenesis of gastrointestinal mucosal diseases. Physiol. Rev. 2014, 94, 329–354. [Google Scholar] [CrossRef] [PubMed]
- Carter, J.D.; Ghio, A.J.; Samet, J.M.; Devlin, R.B. Cytokine production by human airway epithelial cells after exposure to an air pollution particle is metal-dependent. Toxicol. Appl. Pharmacol. 1997, 146, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Quay, J.L.; Reed, W.; Samet, J.; Devlin, R.B. Air pollution particles induce IL-6 gene expression in human airway epithelial cells via NF-κ B activation. Am. J. Respir. Cell Mol. Biol. 1998, 19, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Gilliland, F.D.; McConnell, R.; Peters, J.; Gong, H., Jr. A theoretical basis for investigating ambient air pollution and children’s respiratory health. Environ. Health Perspect. 1999, 107, 403. [Google Scholar] [CrossRef] [PubMed]
- Samet, J.M.; Graves, L.M.; Quay, J.; Dailey, L.A.; Devlin, R.B.; Ghio, A.J.; Reed, W. Activation of MAPKs in human bronchial epithelial cells exposed to metals. Am. J. Physiol. Lung Cell. Mol. Physiol. 1998, 275, L551–L558. [Google Scholar] [CrossRef]
- Gulati, K.; Ray, A. Immunotoxicity. In Handbook of Toxicology of Chemical Warfare Agents; Elsevier Science: Amsterdam, The Netherlands, 2009; pp. 595–609. [Google Scholar]
- Rajeshkumar, S.; Liu, Y.; Ma, J.; Duan, H.Y.; Li, X. Effects of exposure to multipleheavy metals on biochemical and histopathological alterations in common carp, Cyprinus carpio L. Fish Shellfish Immunol. 2017, 70, 461–472. [Google Scholar] [CrossRef] [PubMed]
- Jung, D.; Bolm-Audorff, U.; Faldum, A.; Hengstler, J.G.; Attia, D.I.; Janssen, K.; Konietzko, J. Immunotoxicity of co-exposures to heavy metals: In vitro studies and results from occupational exposure to cadmium, cobalt and lead. EXCLI J. 2003, 2, 31–44. [Google Scholar]
- Miyai, K.; Meeks, R.G.; Harrison, S.D.; Bull, R.J. Structural organization of the liver. In Hepatotoxicology; CRC Press: London, UK, 1991; Volume 1. [Google Scholar]
- Navarro, V.J.; Senior, J.R. Drug-related hepatotoxicity. N. Engl. J. Med. 2006, 354, 731–739. [Google Scholar] [CrossRef] [PubMed]
- Bleibel, W.; Kim, S.; D’Silva, K.; Lemmer, E.R. Drug-induced liver injury. Dig. Dis. Sci. 2007, 52, 2463–2471. [Google Scholar] [CrossRef] [PubMed]
- Naruse, K.; Tang, W.; Makuuchi, M. Artificial and bioartificial liver support: A review of perfusion treatment for hepatic failure patients. World J. Gastroenterol. 2007, 13, 1516. [Google Scholar] [CrossRef] [PubMed]
- Saukkonen, J.J.; Cohn, D.L.; Jasmer, R.M.; Schenker, S.; Jereb, J.A.; Nolan, C.M.; Bernardo, J. An official ATS statement: Hepatotoxicity of antituberculosis therapy. Am. J. Respir. Crit. Care Med. 2006, 174, 935–952. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharjee, T.; Bhattacharjee, S.; Choudhuri, D. Hepatotoxic and nephrotoxic effects of chronic low dose exposure to a mixture of heavy metals-lead, cadmium and arsenic. Int. J. Pharm. Chem. Biol. Sci. 2016, 6, 39. [Google Scholar]
- Yuan, G.; Dai, S.; Yin, Z.; Lu, H.; Jia, R.; Xu, J.; Zhao, X. Toxicological assessment of combined lead and cadmium: Acute and sub-chronic toxicity study in rats. Food Chem. Toxicol. 2014, 65, 260–268. [Google Scholar] [CrossRef] [PubMed]
- Kedderis, G.L. Biochemical basis of hepatocellular injury. Toxicol. Pathol. 1996, 24, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.M. Drug-induced hepatotoxicity. N. Engl. J. Med. 1995, 333, 1118–1127. [Google Scholar] [CrossRef] [PubMed]
- Anders, M.W.; Lash, L.; Dekant, W.; Elfarra, A.A.; Dohn, D.R.; Reed, D.J. Biosynthesis and biotransformation of glutathione S-conjugates to toxic metabolites. CRC Crit. Rev. Toxicol. 1988, 18, 311–341. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.; Gu, Y.; Zhou, Q.; Mao, G.; Zou, B.; Zhao, J. Combined toxicity of heavy metal mixtures in liver cells. J. Appl. Toxicol. 2016, 36, 1163–1172. [Google Scholar] [CrossRef] [PubMed]
- Zalups, R.K.; Klotzbach, J.M.; Diamond, G.L. Enhanced accumulation of injected inorganic mercury in renal outer medulla after unilateral nephrectomy. Toxicol. Appl. Pharmacol. 1987, 89, 226–236. [Google Scholar] [CrossRef]
- Price, R.G. Urinary enzymes, nephrotoxicity and renal disease. Toxicology 1982, 23, 99–134. [Google Scholar] [CrossRef]
- Orr, S.E.; Bridges, C.C. Chronic kidney disease and exposure to nephrotoxic metals. Int. J. Mol. Sci. 2017, 18, 1039. [Google Scholar] [CrossRef]
- Hall, R.L.; Wilke, W.L.; Fettman, M.J. Renal resistance to mercuric chloride toxicity during prolonged exposure in rats. Vet. Hum. Toxicol. 1986, 28, 305–307. [Google Scholar] [PubMed]
- Hambach, R.; Lison, D.; D’haese, P.C.; Weyler, J.; De Graef, E.; De Schryver, A.; Van Sprundel, M. Co-exposure to lead increases the renal response to low levels of cadmium in metallurgy workers. Toxicol. Lett. 2013, 222, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Houser, M.T.; Berndt, W.O. Unilateral nephrectomy in the rat: Effects on mercury handling and renal cortical subcellular distribution. Toxicol. Appl. Pharmacol. 1988, 93, 187–194. [Google Scholar] [CrossRef]
- Costa, L.G. Biochemical and molecular neurotoxicology: Relevance to biomarker development, neurotoxicity testing and risk assessment. Toxicol. Lett. 1998, 102, 417–421. [Google Scholar] [CrossRef]
- Lucchini, R.; Zimmerman, N. Lifetime cumulative exposure as a threat forneurodegeneration: Need for prevention strategies on a globalscale. NeuroToxicology 2009, 30, 1144–1148. [Google Scholar] [CrossRef] [PubMed]
- Kakkar, P.; Jaffery, F.N. Biological markers for metal toxicity. Environ. Toxicol. Pharmacol. 2005, 19, 335–349. [Google Scholar] [CrossRef] [PubMed]
- Landrigan, P.; Nordberg, M.; Lucchini, R.; Nordberg, G.; Grandjean, P.; Iregren, A.; Alessio, L. The declaration of Brescia on prevention of the neurotoxicity of metals. Am. J. Ind. Med. 2007, 50, 709–711. [Google Scholar] [CrossRef] [PubMed]
- Pohl, H.R.; Roney, N.; Abadin, H.G. Metal ions affecting the neurological system. Met. Ions Life Sci. 2011, 8, 247–262. [Google Scholar] [PubMed]
- Kaur, A.; Joshi, K.; Minz, R.W.; Gill, K.D. Neurofilament phosphorylation and disruption: A possible mechanism of chronic aluminium toxicity in Wistarrats. Toxicology 2006, 219, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Flora, S.J. Arsenic-induced oxidative stress and its reversibility. Free Radic. Biol. Medic. 2011, 51, 257–281. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.Y.; Hsiao, W.C.; Huang, C.J.; Kao, C.F.; Hsu, G.S.W. Heme oxygenase-1induction by the ROS–JNK pathway plays a role in aluminum-induced anemia. J. Inorg. Biochem. 2013, 128, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Freitas Fonseca, M.; De Souza Hacon, S.; Grandjean, P.; Choi, A.; Rodrigues Bastos, W. Iron status as a covariate in methylmercury-associated neurotoxicity risk. Chemosphere 2014, 100, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Clarkson, T.W. Metal toxicity in the central nervous system. Environ. Health Perspect. 1987, 75, 59. [Google Scholar] [CrossRef] [PubMed]
- Cecil, K.M.; Brubaker, C.J.; Adler, C.M.; Dietrich, K.N.; Altaye, M.; Egelhoff, J.C.; Lanphear, B.P. Decreased brain volume in adults with childhood lead exposure. PLoS Med. 2008, 5, 112. [Google Scholar] [CrossRef] [PubMed]
- Kuhlmann, A.C.; McGlothan, J.L.; Guilarte, T.R. Developmental lead exposurecauses spatial learning deficits in adult rats. Neurosci. Lett. 1997, 233, 101–104. [Google Scholar] [CrossRef]
- Martinez-Finley, E.J.; Goggin, S.L.; Labrecque, M.T.; Allan, A.M. Reduced expression of MAPK/ERK genes in perinatal arsenic-exposed offspring induced by glucocorticoid receptor deficits. Neurotoxicol. Teratol. 2011, 33, 530–537. [Google Scholar] [CrossRef] [PubMed]
- Bellinger, D.C. Inorganic arsenic exposure and children’s neurodevelopment: A review of the evidence. Toxics 2013, 1, 2–17. [Google Scholar] [CrossRef]
- Crespo-López, M.E.; Macêdo, G.L.; Pereira, S.I.; Arrifano, G.P.; Picanço-Diniz, D.L.; do Nascimento, J.L.M.; Herculano, A.M. Mercury and human genotoxicity: Critical considerations and possible molecular mechanisms. Pharmacol. Res. 2009, 60, 212–220. [Google Scholar] [CrossRef] [PubMed]
- Leong, C.C.; Syed, N.I.; Lorscheider, F.L. Retrograde degeneration of neurite membrane structural integrity of nerve growth cones following in vitro exposure tomercury. NeuroReport 2001, 12, 733–737. [Google Scholar] [CrossRef] [PubMed]
- Czarnecki, L.A.; Moberly, A.H.; Turkel, D.J.; Rubinstein, T.; Pottackal, J.; Rosenthal, M.C.; McGann, J.P. Functional rehabilitation of cadmium-induced neurotoxicity despite persistent peripheral pathophysiology in the olfactory system. Toxicol. Sci. 2012, 126, 534–544. [Google Scholar] [CrossRef] [PubMed]
- Viaene, M.K.; Masschelein, R.; Leenders, J.; De Groof, M.; Swerts, L.J.V.C.; Roels, H.A. Neurobehavioural effects of occupational exposure to cadmium: A cross sectionalepidemiological study. Occup. Environ. Med. 2000, 57, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Rai, A.; Maurya, S.K.; Khare, P.; Srivastava, A.; Bandyopadhyay, S. Characterizationof developmental neurotoxicity of As, Cd, and Pb mixture: Synergistic action of metal mixture in glial and neuronal functions. Toxicolo. Sci. 2010, 118, 586–601. [Google Scholar] [CrossRef] [PubMed]
- Ankley, G.T.; Bennett, R.S.; Erickson, R.J.; Hoff, D.J.; Hornung, M.W.; Johnson, R.D.; Serrrano, J.A. Adverse outcome pathways: A conceptual framework to support ecotoxicology research and risk assessment. Environ. Toxicol. Chem. 2010, 29, 730–741. [Google Scholar] [CrossRef] [PubMed]
- Desai, V.; Kaler, S.G. Role of copper in human neurological disorders. Am. J. Clin. Nutr. 2008, 88, 855S–858S. [Google Scholar] [CrossRef] [PubMed]
- Shaw, C.A.; Tomljenovic, L. Aluminum in the central nervous system (CNS): Toxicity in humans and animals, vaccine adjuvants, and autoimmunity. Immunol. Res. 2013, 56, 304–316. [Google Scholar] [CrossRef] [PubMed]
- Authier, F.J.; Cherin, P.; Creange, A.; Bonnotte, B.; Ferrer, X.; Abdelmoumni, A.; Maisonobe, T. Central nervous system disease in patients with macrophagic myofasciitis. Brain 2001, 124, 974–983. [Google Scholar] [CrossRef] [PubMed]
- Strausak, D.; Mercer, J.F.; Dieter, H.H.; Stremmel, W.; Multhaup, G. Copper indisorders with neurological symptoms: Alzheimer’s, Menkes, and Wilson diseases. Brain Res. Bull. 2001, 55, 175–185. [Google Scholar] [CrossRef]
- Okuda, B.; Iwamoto, Y.; Tachibana, H.; Sugita, M. Parkinsonism after acute cadmium poisoning. Occup. Health Ind. Med. 1998, 5, 232–243. [Google Scholar] [CrossRef]
- Chen, P.; Chakraborty, S.; Peres, T.V.; Bowman, A.B.; Aschner, M. Manganese-induced neurotoxicity: From C. elegans to humans. Toxicol. Res. 2015, 4, 191–202. [Google Scholar] [CrossRef] [PubMed]
- Sanders, A.P.; Henn, B.C.; Wright, R.O. Perinatal and childhood exposure tocadmium, manganese, and metal mixtures and effects on cognition and behavior: A review of recent literature. Curr. Environ. Health Rep. 2015, 2, 284–294. [Google Scholar] [CrossRef] [PubMed]
- McDermott, S.; Wu, J.; Cai, B.; Lawson, A.; Aelion, C.M. Probability of intellectual disability is associated with soil concentrations of arsenic and lead. Chemosphere 2011, 84, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Ha, E.H.; Park, H.; Ha, M.; Kim, Y.; Hong, Y.C.; Kim, B.N. Prenatal lead and cadmium co-exposure and infant neurodevelopment at 6 months of age: Mothers and Children’s Environ Health (MOCEH) study. Neurotoxicology 2013, 35, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Karri, V.; Schuhmacher, M.; Kumar, V. Heavy metals (Pb, Cd, As and MeHg) as risk factors for cognitive dysfunction: A general review of metal mixture mechanism in brain. Environ. Toxicol. Pharmacol. 2016, 48, 203–213. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.D.; Yan, C.H.; Shen, X.M.; Tian, Y.; Cao, L.L.; Yu, X.G.; Liu, J.X. Prenatal exposure to multiple toxic heavy metals and neonatal neurobehavioral development in Shanghai, China. Neurotoxicol. Teratol. 2011, 33, 437–443. [Google Scholar] [CrossRef] [PubMed]
- Antonio, M.T.; Corredor, L.; Leret, M.L. Study of the activity of several brain enzymes like markers of the neurotoxicity induced by perinatal exposure to lead and/or cadmium. Toxicol. Lett. 2003, 143, 331–340. [Google Scholar] [CrossRef]
- Farina, M.; Rocha, J.B.; Aschner, M. Mechanisms of methylmercury-induced neurotoxicity: Evidence from experimental studies. Life Sci. 2011, 89, 555–563. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Du, Y. Cadmium and its neurotoxic effects. Oxid. Med. Cell. Longev. 2013, 2013, 898034. [Google Scholar] [CrossRef] [PubMed]
- Orisakwe, O.E. Lead and cadmium in public health in Nigeria: Physicians neglect and pitfall in patient management. N. Am. J. Med. Sci. 2014, 6, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Shin, H.R.; Bray, F.; Forman, D.; Mathers, C.; Parkin, D.M. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int. J. Cancer 2010, 127, 2893–2917. [Google Scholar] [CrossRef] [PubMed]
- Boyle, P.; Levin, B. World Cancer Report 2008; IARC Press, International Agency for Research Cancer: Lyon, France, 2008; Available online: https://www.cabdirect.org/cabdirect/search/?q=bn%3a%229789283204237%22 (accessed on 15 August 2018).
- Lyerly, H.K.; Abernethy, A.P.; Stockler, M.R.; Koczwara, B.; Aziz, Z.; Nair, R.; Seymour, L. Need for global partnership in cancer care: Perceptions of cancer care researchers attending the 2010 Australia and Asia Pacific Clinical Oncology Research Development Workshop. J. Oncol. Pract. 2011, 7, 324–329. [Google Scholar] [CrossRef] [PubMed]
- Sylla, B.S.; Wild, C.P. A million Africans a year dying from cancer by 2030: What can cancer research and control offer to the continent? Int. J. Cancer 2012, 130, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Sassman, E. Types of cancer prevalent in South Africa: Cancer Association of South Africa. Health-e News. 2011. Available online: http://www.health-e.org.za/uploaded/00ceee132303b08c2e8a6f73622517e6.pdf (accessed on 3 August 2018).
- Hopenhayn-Rich, C.; Biggs, M.L.; Fuchs, A.; Bergoglio, R.; Tello, E.E.; Nicolli, H.; Smith, A.H. Bladder cancer mortality associated with arsenic in drinking water in Argentina. Epidemiology 1996, 7, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Song, H.; Chen, W.Q.; Lu, C.; Hu, Q.; Ren, Z.; Ling, W. Cancer mortality in a Chinese population surrounding a multi-metal sulphide mine in Guangdong province: An ecologic study. BMC Public Health 2011, 11, 319. [Google Scholar] [CrossRef] [PubMed]
- Carrigan, P.E.; Hentz, J.G.; Gordon, G.; Morgan, J.L.; Raimondo, M.; Anbar, A.D.; Miller, L.J. Distinctive heavy metal composition of pancreatic juice in patients with pancreatic carcinoma. Cancer Epidemiol. Biomark. Prev. 2007, 16, 2656–2663. [Google Scholar] [CrossRef] [PubMed]
- Lo, Y.C.; Dooyema, C.A.; Neri, A.; Durant, J.; Jefferies, T.; Medina-Marino, A.; Samson, M.Y. Childhood lead poisoning associated with gold ore processing: A village-level investigation—Zamfara State, Nigeria, October–November 2010. Environ. Health Perspect. 2012, 120, 1450–1455. [Google Scholar] [CrossRef] [PubMed]
- Von Lindern, I.H.; Von Braun, M.C.; Tirima, S.; Bartrem, C. Zamfara, Nigeria Lead Poisoning Epidemic Emergency Environmental Response, May 2010–March 2011, Final Report to the United Nations Childrens Fund (UNICEF). UNICEF Programme cooperation Agreement: YW-303 (01). 2011. Available online: http://www. blacksmithinstitute.org/files/FileUpload/files/Additional% 20Reports/Zamfara-Nigeria-Report. pdf (accessed on 7 August 2018).
- Dooyema, C.A.; Neri, A.; Lo, Y.C.; Durant, J.; Dargan, P.I.; Swarthout, T.; Biya, O.; Gidado, S.O.; Haladu, S.; Sani-Gwarzo, N.; et al. Outbreak of fatal childhood lead poisoning related to artisanal gold mining in northwestern Nigeria, 2010. Environ. Health Perspect. 2012, 120, 601–607. [Google Scholar] [CrossRef] [PubMed]
- CDC (Centers for Disease Control and Prevention). Response to the Advisory Committee on Childhood Lead Poisoning Prevention Report, Low Level Lead Exposure Harms Children: A Renewed Call for Primary Prevention; Morb Mortal Wkly Rep MMWR 61:383; CDC: Atlanta, GA, USA, 2012.
- Orisakwe, O.E.; Oladipo, O.O.; Ajaezi, G.C.; Udowelle, N.A. Horizontal and Vertical Distribution of Heavy Metals in Farm Produce and Livestock around Lead-Contaminated Goldmine in Dareta and Abare, Zamfara State, Northern Nigeria. J. Environ. Public Health 2017, 2017, 3506949. [Google Scholar] [CrossRef] [PubMed]
- Agency for Toxic Substances and Disease Registry. Toxicological Profile for Lead; US Department of Health and Human Services, Agency for Toxic Substances and Disease Registry: Atlanta, GA, USA, 2007.
- ATSDR Agency for Toxic Substances and Disease Registry. Toxicological Profile for Cadmium; Agency for Toxic Substances Disease Registry: Atlanta, GA, USA, 2008.
- Chindah, A.C.; Braide, A.S.; Sibeudu, O.C. Distribution of hydrocarbons and heavy metals in sediment and a crustacean(shrimps- Penaeus notialis) from the Bonny/New Calabar River Estuary, Niger Delta. Afr. J. Environ. Assess. Manag. 2004, 9, 1–17. [Google Scholar]
- Oloruntegbe, K.O.; Akinsete, M.A.; Odutuyi, M.O. Fifty years of oil exploration in Nigeria: Physico-chemical impacts and implication for environmental accounting and development. J. Appl. Sci. Res. 2009, 5, 2131–2137. [Google Scholar]
- Adekola, F.A.; Eletta, O.A.A. A study of heavy metal pollution of ASA River, Ilorin. Nigeria; trace metal monitoring and geochemistry. Environ. Monit. Assess. 2007, 125, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Fianko, J.R.; Osae, S.; Adomako, D.; Adotey, D.K.; Serfor-Armah, Y. Assessment of heavy metal pollution of the Iture Estuary in the central region of Ghana. Environ. Monitor. Assess. 2007, 131, 467–473. [Google Scholar] [CrossRef] [PubMed]
- Odai, S.N.; Mensah, E.; Sipitey, D.; Ryo, S.; Awuah, E. Heavy metals uptake by vegetables cultivated on urban waste dumpsites: Case study of Kumasi, Ghana. Res. J. Environ. Toxicol. 2008, 2, 92–99. [Google Scholar] [CrossRef]
- USEPA (US Environmental Protection Agency). Regulated Drinking Water Contaminants; Online database; USEPA: Washington, DC, USA, 2015.
- ATSDR (Agency for Toxic Substances and Disease Registry). Toxicologial Profiles, Toxic Substances Portal; ATSDR: Atlanta, GA, USA, 2015.
- Smith, A.H.; Lingas, E.O.; Rahman, M. Contamination of drinking- water by arsenic in Bangladesh: A public health emergency. Bull. World Health Organ. 2000, 78, 1093–1103. [Google Scholar] [PubMed]
- Ahsan, H.; Chen, Y.; Parvez, F.; Zablotska, L.; Argos, M.; Hussain, I.; Van Geen, A. Arsenic exposure from drinking water and risk of premalignant skin lesions in Bangladesh: Baseline results from the Health Effects of Arsenic Longitudinal Study. Am. J. Epidemiol. 2006, 163, 1138–1148. [Google Scholar] [CrossRef] [PubMed]
- Rosado, J.L.; Ronquillo, D.; Kordas, K.; Rojas, O.; Alatorre, J.; Lopez, P.; Stoltzfus, R.J. Arsenic exposure and cognitive performance in Mexican schoolchildren. Environ. Health Perspect. 2007, 115, 1371–1375. [Google Scholar] [CrossRef] [PubMed]
- Bawaskar, H.S.; Himmatrao Bawaskar, P.; Himmatrao Bawaskar, P. Chronic renal failure associated with heavy metal contamination of drinking water: A clinical report from a small village in Maharashtra. Clin. Toxicol. 2010, 48, 768. [Google Scholar] [CrossRef] [PubMed]
- Bernard, A. Cadmium & its adverse effects on human health. Indian J. Med. Res. 2008, 128, 557–564. [Google Scholar] [PubMed]
- Armah, F.A.; Quansah, R.; Luginaah, I. A systematic review of heavy metals of anthropogenic origin in environmental media and biota in the context of gold mining in Ghana. Int. Sch. Res. Not. 2014, 2014, 252148. [Google Scholar] [CrossRef] [PubMed]
- Alatise, O.I.; Schrauzer, G.N. Lead exposure: A contributing cause of the current breast cancer epidemic in Nigerian women. Biol. Trace Elem. Res. 2010, 136, 127–139. [Google Scholar] [CrossRef] [PubMed]
- Hnizdo, E.; Sluis-Cremer, G.K. Silica exposure, silicosis, and lung cancer: A mortality study of South African gold miners. Occup. Environ. Med. 1991, 48, 53–60. [Google Scholar] [CrossRef]
- McGlashan, N.D.; Harington, J.S.; Chelkowska, E. Changes in the geographical and temporal patterns of cancer incidence among black gold miners working in South Africa, 1964–1996. Br. J. Cancer 2003, 88, 1361–1369. [Google Scholar] [CrossRef] [PubMed]
- Hnizdo, E.; Murray, J.; Klempman, S. Lung cancer in relation to exposure to silica dust, silicosis and uranium production in South African gold miners. Thorax 1997, 52, 271–275. [Google Scholar] [CrossRef] [PubMed]
- Parkin, D.M.; Vizcaino, A.P.; Skinner, M.E.; Ndhlovu, A. Cancer patterns and risk factors in the African population of southwestern Zimbabwe, 1963–1977. Cancer Epidemiol. Prev. Biomark. 1994, 3, 537–547. [Google Scholar]
- Georgescu, B.; Georgescu, C.; Dărăban, S.; Bouaru, A.; Paşcalău, S. Heavy metals acting as endocrine disrupters. Sci. Pap. Anim. Sci. Biotechnol. 2011, 44, 89–93. [Google Scholar]
- Rahman, A.; Kumarathasan, P.; Gomes, J. Infant and mother related outcomes from exposure to metals with endocrine disrupting properties during pregnancy. Sci. Total Environ. 2016, 569, 1022–1031. [Google Scholar] [CrossRef] [PubMed]
- Gundacker, C.; Hengstschläger, M. The role of the placenta in fetal exposure to heavy metals. Wien. Med. Wochenschr. 2012, 162, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Needham, L.L.; Grandjean, P.; Heinzow, B.; Jørgensen, P.J.; Nielsen, F.; Patterson, D.G., Jr.; Weihe, P. Partition of environmental chemicals between maternal and fetal blood and tissues. Environ. Sci. Technol. 2010, 45, 1121–1126. [Google Scholar] [CrossRef] [PubMed]
- McDermott, S.; Bao, W.; Aelion, C.M.; Cai, B.; Lawson, A. When are fetuses and young children most susceptible to soil metal concentrations of arsenic, lead and mercury? Spat. Spatio-Temporal Epidemiol. 2012, 3, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Ajayi, O.O.; Charles-Davies, M.A.; Arinola, O.G. Progesterone, selected heavy metals and micronutrients in pregnant Nigerian women with a history of recurrent spontaneous abortion. Afr. Health Sci. 2012, 12, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Otebhi, O.E.; Osadolor, H.B. Select toxic metals status of pregnant women with history of pregnancy complications in Benin City, South-South Nigeria. J. Appl. Sci. Environ. Manag. 2016, 20, 5–10. [Google Scholar] [CrossRef]
- Coulam, C.B.; Stephenson, M.; Stern, J.J.; Clark, D.A. Immunotherapy for recurrent pregnancy loss: Analysis of results from clinical trials. Am. J. Reprod. Immunol. 1996, 35, 352–359. [Google Scholar] [CrossRef] [PubMed]
- Orisakwe, O.E.; Asomugha, R.; Obi, E.; Afonne, O.J.; Anisi, C.N.; Dioka, C.E. Impact of effluents from a car battery manufacturing plant in Nnewi, Nigeria onwater, soil and food qualities. Arch. Environ. Health Int. J. 2004, 59, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Amadi, C.N.; Orisakwe, O.E.; Roberts, I.I. Elemental impurities in registered herbal supplements in Nigeria. A. look at Mercury, Antimony and Tin. Rasayan J. Chem. 2012, 5, 220–228. [Google Scholar]
- Amadi, C.N.; Orisakwe, O.E. Herb-induced liver injuries in developing nations: An update. Toxics 2018, 6, 24. [Google Scholar] [CrossRef] [PubMed]
- Ideriah, T.J.K.; David-Omiema, S.; Ogbonna, D.N. Distribution of heavy metals in water and sediment along Abonnema Shoreline, Nigeria. Res. Environ. 2012, 2, 33–40. [Google Scholar]
- Owamah, H.I. Heavy metals determination and assessment in a petroleum impacted River in the Niger Delta Region of Nigeria. J. Pet. Environ. Biotechnol. 2013, 4, 135–138. [Google Scholar] [CrossRef]
- Ansari, T.M.; Marr, I.L.; Tariq, N. Heavy metals in marine pollution perspective—Amini review. J. Appl. Sci. 2004, 4, 1–20. [Google Scholar] [CrossRef]
- Akporido, S.O.; Onianwa, P.C. Heavy Metals and Total Petroleum Hydrocarbon Concentrations in Surface Water of ESI River, Western Niger Delta. Res. J. Environ. Sci. 2015, 9, 88–100. [Google Scholar] [CrossRef]
- Cooper, Z.; Bringolf, R.; Cooper, R.; Loftis, K.; Bryan, A.L.; Martin, J.A. Heavy metal bioaccumulation in two passerines with differing migration strategies. Sci. Total Environ. 2017, 592, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Egboh, S.H.O.; Nwajei, G.E.; Adaikpoh, E.O. Selected Heavy metals concentration in sediments from major roads and gutters in Warri, Delta State, Nigeria. Niger. J. Sci. Environ. 2000, 2, 105–111. [Google Scholar]
- Osakwe, S.A.; Otuya, B.O.; Adaikpoh, E.O. Determination of Pb, Cu, Ni, Fe and Hgin the Soils of Okpai Delta State, Nigeria. Niger. J. Sci. Environ. 2003, 3, 45–49. [Google Scholar]
- Ndubuisi, O.L.; Asia, I.O. Environmental pollution in oil producing areas of the Niger Delta Basin, Nigeria: Empirical assessment of trends and people’s perception. Environ. Res. J. 2007, 1, 18–26. [Google Scholar]
- Oze, G.; Oze, R.; Anunuso, C.; Ogukwe, C.; Nwanjo, H.; Okorie, K. Heavy metal pollution of fish of Qua-Iboe river estuary: Possible implications for neurotoxicity. Int. J. Toxicol. 2006, 3, 56–59. [Google Scholar]
- Farombi, E.O.; Adelowo, O.A.; Ajimoko, Y.R. Biomarkers of oxidative stress and heavy metal levels as indicators of environmental pollution in African cat fish (Clarias gariepinus) from Nigeria Ogun River. Int. J. Environ. Res. Public Health 2007, 4, 158–165. [Google Scholar] [CrossRef] [PubMed]
- Malik, R.N.; Zeb, N. Assessment of environmental contamination using feathers of Bubulcus ibis L., as a biomonitor of heavy metal pollution, Pakistan. Ecotoxicology 2009, 18, 522–536. [Google Scholar] [CrossRef] [PubMed]
- Avenant-Oldewage, A.; Marx, H.M. Bioaccumulation of chromium, copper and iron in the organs and tissues of Clarias gariepinus in the Olifants River, Kruger National Park. Water SA 2000, 26, 569–582. [Google Scholar]
- Abadin, H.; Ashizawa, A.; Stevens, Y.W.; Llados, F.; Diamond, G.; Sage, G.; Swarts, S.G. Health Effects in Toxicological Profile for Lead; ATSDR: Atlanta, GA, USA, 2007.
- Faroon, O.; Ashizawa, A.; Wright, S.; Tucker, P.; Jenkins, K.; Ingerman, L.; Rudisill, C. Toxicological Profile for Cadmium; Agency for Toxic Substances and Disease Registry: Atlanta, GA, USA, 2012.
- Kortenkamp, A. Ten years of mixing cocktails: A review of combination effects of endocrine-disrupting chemicals. Environ. Health Perspect. 2007, 115, 98–105. [Google Scholar] [CrossRef] [PubMed]
- Dopp, E.; Von Recklinghausen, U.; Diaz-Bone, R.; Hirner, A.V.; Rettenmeier, A.W. Cellular uptake, subcellular distribution and toxicity of arsenic compounds in methylating and non-methylating cells. Environ. Res. 2010, 110, 435–442. [Google Scholar] [CrossRef] [PubMed]
- Marsano, F.; Boatti, L.; Ranzato, E.; Cavaletto, M.; Magnelli, V.; Dondero, F.; Viarengo, A. Effects of mercury on Dictyostelium discoideum: Proteomics reveals the molecular mechanisms of physiological adaptation and toxicity. J. Proteome Res. 2010, 9, 2839–2854. [Google Scholar] [CrossRef] [PubMed]
- Naujokas, M.F.; Anderson, B.; Ahsan, H.; Aposhian, H.V.; Graziano, J.H.; Thompson, C.; Suk, W.A. The broad scope of health effects from chronic arsenic exposure: Update on a worldwide public health problem. Environ. Health Perspect. 2013, 121, 295–302. [Google Scholar] [CrossRef] [PubMed]
- Kenston, S.S.F.; Su, H.; Li, Z.; Kong, L.; Wang, Y.; Song, X.; Li, Z. The systemic toxicity of heavy metal mixtures in rats. Toxicol. Res. 2018, 7, 396–407. [Google Scholar] [CrossRef] [PubMed]
- Su, H.; Li, Z.; Kenston, S.S.F.; Shi, H.; Wang, Y.; Song, X.; Ding, M. Joint toxicity of different heavy metal mixtures after a short-term oral repeated-administration in rats. Int. J. Environ. Res. Public Health 2017, 14, 1164. [Google Scholar] [CrossRef] [PubMed]
- Silva, E.; Rajapakse, N.; Kortenkamp, A. Something from “nothing”—Eight weak estrogenic chemicals combined at concentrations below NOECs produce significant mixture effects. Environ. Sci. Technol. 2002, 36, 1751–1756. [Google Scholar] [CrossRef] [PubMed]
- Whittaker, M.H.; Wang, G.; Chen, X.Q.; Lipsky, M.; Smith, D.; Gwiazda, R.; Fowler, B.A. Exposure to Pb, Cd, and As mixtures potentiates the production of oxidative stress precursors: 30-day, 90-day, and 180-day drinking water studies in rats. Toxicol. Appl. Pharmacol. 2011, 254, 154–166. [Google Scholar] [CrossRef] [PubMed]
- Le, T.H.; Lim, E.S.; Hong, N.H.; Lee, S.K.; Shim, Y.S.; Hwang, J.R.; Min, J. Proteomic analysis in Daphnia magna exposed to As (III), As (V) and Cd heavy metals and their binary mixtures for screening potential biomarkers. Chemosphere 2013, 93, 2341–2348. [Google Scholar] [CrossRef] [PubMed]
- Varotto, L.; Domeneghetti, S.; Rosani, U.; Manfrin, C.; Cajaraville, M.P.; Raccanelli, S.; Venier, P. DNA damage and transcriptional changes in the gills of Mytilus galloprovincialis exposed to nanomolar doses of combined metal salts (Cd, Cu, Hg). PLoS ONE 2013, 8, e54602. [Google Scholar] [CrossRef] [PubMed]
- Vellinger, C.; Parant, M.; Rousselle, P.; Usseglio-Polatera, P. Antagonistic toxicity of arsenate and cadmium in a freshwater amphipod (Gammarus pulex). Ecotoxicology 2012, 21, 1817–1827. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, S.A.; Folorunsho, J.O. Heavy metals concentration in soil and Amaranthus retroflexus grown on irrigated farmlands in the Makera Area, Kaduna, Nigeria. J. Geogr. Reg. Plan. 2015, 8, 210–217. [Google Scholar] [CrossRef]
- WHO/UNEP. Water Pollution Control—A Guide to the Use of Water Quality Management Principles; Case Study of Nigeria; WHO: Geneva, Switzerland; UNEP: Nairobi, Kenya, 1997; 14p. [Google Scholar]
- Asonye, C.C.; Okolie, N.P.; Okenwa, E.E.; Iwuanyanwu, U.G. Some physico-chemical characteristics and heavy metal profiles of Nigerian rivers, streams and waterways. Afr. J. Biotechnol. 2007, 6, 617–624. [Google Scholar]
- Ogedengbe, K.; Akinbile, C.O. Impact of industrial pollutants on quality of ground and surface waters at Oluyole Industrial Estate, Ibadan, Nigeria. Niger. J. Technol. Dev. 2004, 4, 139–144. [Google Scholar]
- Prabu, P.C. Impact of heavy metal contamination of Akaki river of Ethiopia on soil and metal toxicity on cultivated vegetable crops. Electron. J. Environ. Agric. Food Chem. 2009, 8, 818–827. [Google Scholar]
- Abdullahi, M.S.; Uzairu, A.; Harrison, G.F.S.; Balarabe, M.L. Trace metals screening of tomatoes and onions from irrigated farmlands on the bank of river Challawa, Kano, Nigeria. EJEA Che. 2007, 6, 1869–1878. [Google Scholar]
- Muchuweti, M.; Birkett, J.W.; Chinyanga, E.; Zvauya, R.; Scrimshaw, M.D.; Lester, J.N. Heavy metal content of vegetables irrigated with mixtures of wastewater and sewage sludge in Zimbabwe: Implications for human health. Agric. Ecosyst. Environ. 2006, 112, 41–48. [Google Scholar] [CrossRef]
- Nabulo, G.; Oryem-Origa, H.; Diamond, M. Assessment of lead, cadmium, and zinc contamination of roadside soils, surface films, and vegetables in Kampala City, Uganda. Environ. Res. 2006, 101, 42–52. [Google Scholar] [CrossRef] [PubMed]
- United Nations Environment Program, UNEP. Environmental Pollution and Impacts on Public Health: Implications of the Dandora Municipal Dumping Site in Nairobi, Kenya. 2007. Available online: http://www.unep.org/urban_environment/PDFs/DandoraWasteDump-ReportSummary.pdf (accessed on 4 August 2018).
- Fakayode, S.O.; Olu-Owolabi, B.I. Heavy metal contamination of roadside topsoil in Osogbo, Nigeria: Its relationship to traffic density and proximity to highways. Environ. Geol. 2003, 44, 150–157. [Google Scholar] [CrossRef]
- Mireji, P.O.; Keating, J.; Hassanali, A.; Mbogo, C.M.; Nyambaka, H.; Kahindi, S.; Beier, J.C. Heavy metals in mosquito larval habitats in urban Kisumu and Malindi, Kenya, and their impact. Ecotoxicol. Environ. Saf. 2008, 70, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Emoyan, O.O.; Ogban, F.E.; Akarah, E. Evaluation of heavy metals loading of River Ijana in Ekpan–Warri, Nigeria. J. Appl. Sci. Environ. Manag. 2006, 10, 121–127. [Google Scholar] [CrossRef]
- Meck, M.; Love, D.; Mapani, B. Zimbabwean mine dumps and their impacts on river water quality—A reconnaissance study. Phys. Chem. Earth Parts A/B/C 2006, 31, 797–803. [Google Scholar] [CrossRef]
- Fatoki, O.S.; Mathabatha, S. An assessment of heavy metal pollution in the East London and Port Elizabeth harbours. Water SA 2001, 27, 233–240. [Google Scholar] [CrossRef]
- Naidja, L.; Ali-Khodja, H.; Khardi, S. Sources and levels of particulate matter in North African and Sub-Saharan cities: A literature review. Environ. Sci. Pollut. Res. 2018, 25, 12303–12328. [Google Scholar] [CrossRef] [PubMed]
- Park, S.S.; Sim, S.Y.; Bae, M.S.; Schauer, J.J. Size distribution of water-soluble components in particulate matter emitted from biomass burning. Atmos. Environ. 2013, 73, 62–72. [Google Scholar] [CrossRef]
- Planchart, A.; Green, A.; Hoyo, C.; Mattingly, C.J. Heavy metal exposure and metabolic syndrome: Evidence from human and model system studies. Curr. Environ. Health Rep. 2018, 5, 110–124. [Google Scholar] [CrossRef] [PubMed]
- Goyer, R.A.; Clarkson, T.M. Toxic effects of metals. In Casarett and Doull’s Toxicology; Klaassen, C.D., Ed.; McGraw-Hill: New York, NY, USA, 2001; pp. 811–868. [Google Scholar]
- Klaassen, C.D. Heavy metals. In Goodman and Gilman’s Pharmacological Basis of Therapeutics, 9th ed.; Hardman, J., Limbird, L., Eds.; McGraw Hill: New York, NY, USA, 1995; pp. 1049–1073. [Google Scholar]
- Institute of Environmental Conservation and Research, INECAR. Position Paper against Mining in Rapu-Rapu; INECAR, Ateneo de Naga University: Naga, Philippines, 2000; Available online: www.adnu.edu.ph/institutes/inecar/pospaper1.asp (accessed on 29 August 2018).
- Ogwuegbu, M.O.; Ijioma, M.A. Effects of certain heavy metals on the population due to mineral exploitation. In International Conference on Scientific and Environmental Issues in the Population, Environment and Sustainable Development in Nigeria; University of Ado Ekiti: Ado Ekiti, Nigerian, 2003; pp. 8–10. [Google Scholar]
- Velma, V.; Vutukuru, S.S.; Tchounwou, P.B. Ecotoxicology of hexavalent chromium in freshwater fish: A critical review. Rev. Environ. Health 2009, 24, 129–146. [Google Scholar] [CrossRef] [PubMed]
- Tchounwou, P.B.; Yedjou, C.G.; Patlolla, A.K.; Sutton, D.J. Heavy metal toxicity and the environment. In Molecular, Clinical and Environmental Toxicology; Springer: Basel, Switzerland, 2012; Volume 22, pp. 546–550. [Google Scholar] [CrossRef]
- Jiang, L.F.; Yao, T.M.; Zhu, Z.L.; Wang, C.; Ji, L.N. Impacts of Cd (II) on the conformation and self-aggregation of Alzheimer’s tau fragment corresponding to the hird repeat of microtubule-binding domain. Biochim. Biophys. Acta 2007, 1774, 1414–1421. [Google Scholar] [CrossRef] [PubMed]
- Nagata, C.; Nagao, Y.; Shibuya, C.; Kashiki, Y.; Shimizu, H. Urinary cadmium and serum levels of estrogens and androgens in postmenopausal Japanese women. Cancer Epidemiol. Biomark. Prev. 2005, 14, 705–708. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, G.G.; Il’yasova, D.; Ivanova, A. Urinary cadmium, impaired fasting glucose, and diabetes in the NHANES III. Diabetes Care 2003, 26, 468–470. [Google Scholar] [CrossRef] [PubMed]
- Su, C.C.; Yang, H.F.; Huang, S.J.; Lian, I.B. Distinctive features of oral cancer in Changhua County: High incidence, buccal mucosa preponderance, and a close relation to betel quid chewing habit. J. Formos. Med. Assoc. 2007, 106, 225–233. [Google Scholar] [CrossRef]
- Salnikow, K.; Zhitkovich, A. Genetic and epigenetic mechanisms in metal carcinogenesis and cocarcinogenesis: Nickel, arsenic, and chromium. Chem. Res. Toxicol. 2007, 21, 28–44. [Google Scholar] [CrossRef] [PubMed]
- Chiou, Y.H.; Wong, R.H.; Chao, M.R.; Chen, C.Y.; Liou, S.H.; Lee, H. Nickel accumulation in lung tissues is associated with increased risk of p53 mutation in lung cancer patients. Environ. Mol. Mutagen. 2014, 55, 624–632. [Google Scholar] [CrossRef] [PubMed]
- Adewuyi, G.O.; Olowu, R.A. Assessment of oil and grease, total petroleum hydrocarbons and some heavy metals in surface and groundwater within the vicinity of NNPC oil depot in Apata, Ibadan metropolis, Nigeria. Int. J. Recent Res. Appl. Stud. 2012, 13, 166–174. [Google Scholar]
- Akporido, S.O.; Asagba, S.O. Quality characteristics of soil close to the Benin River in the vicinity of a lubricating oil producing factory, Koko, Nigeria. Int. J. Soil Sci. 2013, 8, 1–16. [Google Scholar] [CrossRef]
- Akpanyung, E.O.; Akanemesang, U.M.; Akpakpan, E.I.; Anodoze, N.O. Levels of heavy metals in fish obtained from two fishing sites in Akwa Ibom State, Nigeria. Afr. J. Environ. Sci. Technol. 2014, 8, 416–421. [Google Scholar] [CrossRef]
- Olowoyo, J.O.; Okedeyi, O.O.; Mkolo, N.M.; Lion, G.N.; Mdakane, S.T.R. Uptake and translocation of heavy metals by medicinal plants growing around a waste dump site in Pretoria, South Africa. S. Afr. J. Bot. 2012, 78, 116–121. [Google Scholar] [CrossRef]
- Mutune, A.N.; Makobe, M.A.; Abukutsa-Onyango, M.O.O. Heavy metal content of selected African leafy vegetables planted in urban and peri-urban Nairobi, Kenya. Afr. J. Environ. Sci. Technol. 2014, 8, 66–74. [Google Scholar] [CrossRef]
- Fosu-Mensah, B.Y.; Addae, E.; Yirenya-Tawiah, D.; Nyame, F. Heavy metals concentration and distribution in soils and vegetation at Korle Lagoon area in Accra, Ghana. Cogent Environ. Sci. 2017, 3, 1405887. [Google Scholar] [CrossRef]
- Malan, M.; Müller, F.; Cyster, L.; Raitt, L.; Aalbers, J. Heavy metals in the irrigation water, soils and vegetables in the Philippi horticultural area in the Western Cape Province of South Africa. Environ. Monit. Assess. 2015, 187, 4085. [Google Scholar] [CrossRef] [PubMed]
- Teta, C.; Ncube, M.; Naik, Y.S. Heavy metal contamination of water and fish in peri-urban dams around Bulawayo, Zimbabwe. Afr. J. Aquat. Sci. 2017, 42, 351–358. [Google Scholar] [CrossRef]
- Ekengele, N.L.; Myung, C.; Ombolo, A.; Ngatcha, N.; Georges, E.; Lape, M. Metal pollution in freshly deposited sediments from river Mingoa, main tributary to the Municipal lake of Yaounde, Cameroon. Geosci. J. 2008, 12, 337–347. [Google Scholar] [CrossRef]
- Essumang, D.K. Analysis and human health risk assessment of arsenic, cadmium, and mercury in Manta birostris (manta ray) caught along the Ghanaian coastline. Hum. Ecol. Risk Assess. 2009, 15, 985–998. [Google Scholar] [CrossRef]
- Kwadwo, A.A.; Tetsuro, A.; Annamalai, S.; Ansa-Asare, O.D.; Biney, C.A.; Tanabe, S. Contamination status of arsenic and other trace elements in drinking water and residents from Tarkwa, a historic mining township in Ghana. Chemosphere 2007, 66, 1513–1522. [Google Scholar]
- Taylor, M.P.; Kesterton, R.G. Heavy metal contamination of an arid river environment: Gruben River, Namibia. Geomorphology 2002, 42, 311–327. [Google Scholar] [CrossRef]
- Obi, E.; Akunyili, D.N.; Ekpo, B.; Orisakwe, O.E. Heavy metal hazards of Nigerianherbal remedies. Sci. Total Environ. 2006, 369, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Ntekim, E.E.U.; Ekwere, S.J.; Ukpong, E.E. Heavy metal distribution in sediments from Calabar River, southeastern Nigeria. Environ. Geol. 1993, 21, 237–241. [Google Scholar] [CrossRef]
- Mombeshora, C.; Ajayi, S.O.; Osibanjo, O. Pollution studies on Nigerian rivers: Toxicheavy metal status of surface waters in Ibadan city. Environ. Int. 1981, 5, 49–53. [Google Scholar] [CrossRef]
- Fakayode, S.; Onianwa, P. Heavy metal contamination of soil, and bioaccumulation in Guinea grass (Panicum maximum) around Ikeja Industrial Estate, Lagos, Nigeria. Environ. Geol. 2002, 43, 145–150. [Google Scholar]
- Bahemuka, T.E.; Mubofu, E.B. Heavy metals in edible green vegetables grown along the sites of the Sinza and Msimbazi rivers in Dar es Salaam, Tanzania. Food Chem. 1999, 66, 63–66. [Google Scholar] [CrossRef]
- Kishe, M.A.; Machiwa, J.F. Distribution of heavy metals in sediments of Mwanza Gulf of Lake Victoria, Tanzania. Environ. Int. 2003, 28, 619–625. [Google Scholar] [CrossRef]
- Muwanga, A.; Barifaijo, E. Impact of industrial activities on heavy metal loading and their physico-chemical effects on wetlands of Lake Victoria basin (Uganda). Afr. J. Sci. Technol. 2006, 7. [Google Scholar] [CrossRef]
- Pettersson, U.T.; Ingri, J. The geochemistry of Co and Cu in the Kafue River as it drains the Copperbelt mining area, Zambia. Chem. Geol. 2001, 177, 399–414. [Google Scholar] [CrossRef]
- Von der Heyden, C.J.; New, M.G. Sediment chemistry: A history of mine contaminant remediation and an assessment of processes and pollution potential. J. Geochem. Explor. 2004, 82, 35–57. [Google Scholar] [CrossRef]
- Papanikolaou, N.C.; Hatzidaki, E.G.; Belivanis, S.; Tzanakakis, G.N.; Tsatsakis, A.M. Lead toxicity update. A brief review. Med. Sci. Monit. 2005, 11, RA329–RA336. [Google Scholar] [PubMed]
- Tong, S.; Schirnding, Y.E.V.; Prapamontol, T. Environmental lead exposure: A public health problem of global dimensions. Bull. World Health Organ. 2000, 78, 1068–1077. [Google Scholar] [PubMed]
- Sowers, M.; Jannausch, M.; Scholl, T.; Li, W.; Kemp, F.W.; Bogden, J.D. Blood lead concentrations and pregnancy outcomes. Arch. Environ. Health Int. J. 2002, 57, 489–495. [Google Scholar] [CrossRef] [PubMed]
- Rossi, E. Low level environmental lead exposure—A continuing challenge. Clin. Biochem. Rev. 2008, 29, 63–70. [Google Scholar] [PubMed]
- Nash, D.; Magder, L.; Lustberg, M.; Sherwin, R.W.; Rubin, R.J.; Kaufmann, R.B.; Silbergeld, E.K. Blood lead, blood pressure, and hypertension in perimenopausal and postmenopausal women. JAMA 2003, 289, 1523–1532. [Google Scholar] [CrossRef] [PubMed]
- National Research Council. Measuring Lead Exposure in Infants, Children, and Other Sensitive Populations; National Academies Press: Washington, DC, USA, 1993. [Google Scholar]
- Gulson, B.L.; Mizon, K.J.; Palmer, J.M.; Korsch, M.J.; Taylor, A.J.; Mahaffey, K.R. Blood lead changes during pregnancy and postpartum with calcium supplementation. Environ. Health Perspect. 2004, 112, 1499–1507. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.R.; Osterloh, J.D.; Flegal, A.R. Use of endogenous, stable lead isotopes to determine release of lead from the skeleton. Environ. Health Perspect. 1996, 104, 60. [Google Scholar] [CrossRef] [PubMed]
- Falcon, M.; Vinas, P.; Luna, A. Placental lead and outcome of pregnancy. Toxicology 2003, 185, 59–66. [Google Scholar] [CrossRef]
- FaikoĞLu, R.; Savan, K.; Utku, Ç.; Takar, N.; Zebitay, A.G. Significance of maternal plasma lead level in early pregnancy loss. J. Environ. Sci. Health Part A 2006, 41, 501–506. [Google Scholar] [CrossRef] [PubMed]
- Ngueta, G.; Ndjaboue, R. Blood lead concentrations in sub-Saharan African children below 6 years: Systematic review. Trop. Med. Int. Health 2013, 18, 1283–1291. [Google Scholar] [CrossRef] [PubMed]
- Adekunle, I.M.; Olorundare, O.; Nwange, C. Assessments of lead levels and daily intakes from green leafy vegetables of southwest Nigeria. Nutr. Food Sci. 2009, 39, 413–422. [Google Scholar] [CrossRef]
- Ugwuja, E.I.; Ibiam, U.A.; Ejikeme, B.N.; Obuna, J.A.; Agbafor, K.N. Blood Pb levels in pregnant Nigerian women in Abakaliki, South-Eastern Nigeria. Environ. Monit. Assess. 2013, 185, 3795–3801. [Google Scholar] [CrossRef] [PubMed]
- Naicker, N.; Norris, S.A.; Mathee, A.; von Schirnding, Y.E.; Richter, L. Prenatal and adolescent blood lead levels in South Africa: Child, maternal and household risk factors in the Birth to Twenty cohort. Environ. Res. 2010, 110, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Sanders, A.P.; Flood, K.; Chiang, S.; Herring, A.H.; Wolf, L.; Fry, R.C. Towards prenatal biomonitoring in North Carolina: Assessing arsenic, cadmium, mercury, and lead levels in pregnant women. PLoS ONE 2012, 7, e31354. [Google Scholar] [CrossRef] [PubMed]
- Miranda, M.L.; Edwards, S.E.; Swamy, G.K.; Paul, C.J.; Neelon, B. Blood lead levels among pregnant women: Historical versus contemporaneous exposures. Int. J. Environ. Res. Public Health 2010, 7, 1508–1519. [Google Scholar] [CrossRef] [PubMed]
- Rhainds, M.; Levallois, P.; Dewailly, É.; Ayotte, P. Lead, mercury, and organochlorine compound levels in cord blood in Quebec, Canada. Arch. Environ. Health Int. J. 1999, 54, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Adeniyi, F.A.; Anetor, J.I. Lead-poisoning in two distant states of Nigeria: An indication of the real size of the problem. Afr. J. Med. Med. Sci. 1999, 28, 107–112. [Google Scholar] [PubMed]
- Ademuyiwa, O.; Arowolo, T.; Ojo, D.A.; Odukoya, O.O.; Yusuf, A.A.; Akinhanmi, T.F. Lead levels in blood and urine of some residents of Abeokuta, Nigeria. Trace Elem. Electrolytes 2002, 19, 63–69. [Google Scholar]
- Orisakwe, O.E.; Nwachukwu, E.; Osadolor, H.B.; Afonne, O.J.; Okocha, C.E. Liver and kidney function tests amongst paint factory workers in Nkpor, Nigeria. Toxicol. Ind. Health 2007, 23, 161–165. [Google Scholar] [CrossRef] [PubMed]
- Adekunle, I.M.; Ogundele, J.A.; Oguntoke, O.; Akinloye, O.A. Assessment of blood and urine lead levels of some pregnant women residing in Lagos, Nigeria. Environ. Monit. Assess. 2010, 170, 467–474. [Google Scholar] [CrossRef] [PubMed]
- Osuntogun, B.A.; Koku, C.A. Environmental impacts of urban road transportation in South-Western states of Nigeria. J. Appl. Sci. 2007, 7, 2356–2360. [Google Scholar] [CrossRef]
- Alasia, D.; Emem-chioma, P.; Wokoma, F.; Okojaja, R.; Bellgam, H.; Iyagba, A.; Akobo, D. NDT Plus. In Proceedings of the World Congress of Nephrology, Milan, Italy, 22–26 May 2009; Volume 2, p. Ii1733. [Google Scholar]
- Nriagu, J.; Afeiche, M.; Linder, A.; Arowolo, T.; Ana, G.; Sridhar, M.K.; Adesina, A. Lead poisoning associated with malaria in children of urban areas of Nigeria. Int. J. Hyg. Environ. Health 2008, 211, 591–605. [Google Scholar] [CrossRef] [PubMed]
- Babalola, O.O.; Ojo, L.O.; Aderemi, M.O. Lead levels in some biological samples of auto-mechanics in Abeokuta, Nigeria. 2005. Available online: http://nopr.niscair.res.in/handle/123456789/3547 (accessed on 4 August 2018).
- Arinola, O.G.; Nwozo, S.O.; Ajiboye, J.A.; Oniye, A.H. Evaluation of trace elements and total antioxidant status in Nigerian cassava processors. Pak. J. Nutr. 2008, 7, 770–772. [Google Scholar] [CrossRef]
- Bodeau-Livinec, F.; Glorennec, P.; Cot, M.; Dumas, P.; Durand, S.; Massougbodji, A.; LeBot, B. Elevated blood lead levels in infants and mothers in Benin and potential sources of exposure. Int. J. Environ. Res. Public Health 2016, 13, 316. [Google Scholar] [CrossRef] [PubMed]
- Tuakuila, J.; Mbuyi, F.; Kabamba, M.; Lantin, A.C.; Lison, D.; Hoet, P. Blood lead levels in the Kinshasa population: A pilot study. Arch. Public Health 2010, 68, 30–41. [Google Scholar] [CrossRef]
- Balistrieri, L.S.; Mebane, C.A. Predicting the toxicity of metal mixtures. Sci. Total Environ. 2014, 466, 788–799. [Google Scholar] [CrossRef] [PubMed]
- Charles, J.; Crini, G.; Degiorgi, F.; Sancey, B.; Morin-Crini, N.; Badot, P.M. Unexpected toxic interactions in the freshwater amphipod Gammarus pulex (L.) exposed to binary copper and nickel mixtures. Environ. Sci. Pollut. Res. 2014, 21, 1099–1111. [Google Scholar] [CrossRef] [PubMed]
- Borgert, C.J.; Quill, T.F.; McCarty, L.S.; Mason, A.M. Can mode of action predict mixture toxicity for risk assessment? Toxicol. Appl. Pharmacol. 2004, 201, 85–96. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Jiang, Y.; Xu, C.; Yu, L.; Sun, D.; Xu, L.; Li, H. Comparison of two mathematical prediction models in assessing the toxicity of heavy metal mixtures to the feeding of the nematode Caenorhabditis elegans. Ecotoxicol. Environ. Saf. 2013, 94, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Qin, L.T.; Liu, S.S.; Zhang, J.; Xiao, Q.F. A novel model integrated concentration addition with independent action for the prediction of toxicity of multi-component mixture. Toxicology 2011, 280, 164–172. [Google Scholar] [CrossRef] [PubMed]
- Berenbaum, M.C. The expected effect of a combination of agents: The general solution. J. Theor. Biol. 1985, 114, 413–431. [Google Scholar] [CrossRef]
- Backhaus, T.; Altenburger, R.; Boedeker, W.; Faust, M.; Scholze, M.; Grimme, L.H. Predictability of the toxicity of a multiple mixture of dissimilarly acting chemicals to Vibrio fischeri. Environ. Toxicol. Chem. 2000, 19, 2348–2356. [Google Scholar] [CrossRef]
- Jonker, M.J.; Svendsen, C.; Bedaux, J.J.; Bongers, M.; Kammenga, J.E. Significance testing of synergistic/antagonistic, dose level-dependent, or dose ratio-dependent effects in mixture dose-response analysis. Environ. Toxicol. Chem. 2005, 24, 2701–2713. [Google Scholar] [CrossRef] [PubMed]
- Jonker, M.J.; Gerhardt, A.; Backhaus, T.; van Gestel, C.A. Test design, mixture characterization, and data evaluation. In Mixture Toxicity: Linking Approaches from Ecological and Human Toxicology; CRC Press: Boca Raton, FL, USA, 2011; pp. 121–156. [Google Scholar]
- Norwood, W.P.; Borgmann, U.; Dixon, D.G.; Wallace, A. Effects of metal mixtures on aquatic biota: A review of observations and methods. Hum. Ecol. Risk Assess. 2003, 9, 795–811. [Google Scholar] [CrossRef]
- Vijver, M.G.; Elliott, E.G.; Peijnenburg, W.J.; De Snoo, G.R. Response predictions for organisms water-exposed to metal mixtures: A meta-analysis. Environ. Toxicol. Chem. 2011, 30, 1482–1487. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Vijver, M.G.; Qiu, H.; Baas, J.; Peijnenburg, W.J. Statistically significant deviations from additivity: What do they mean in assessing toxicity of mixtures? Ecotoxicol. Environ. Saf. 2015, 122, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.S.; Schat, H.; Vooijs, R.; Van Heerwaarden, L.M. Combination toxicology of copper, zinc, and cadmium in binary mixtures: Concentration-dependent antagonistic, nonadditive, and synergistic effects on root growth in Silene vulgaris. Environ. Toxicol. Chem. Int. J. 1999, 18, 348–355. [Google Scholar] [CrossRef]
- Van Genderen, E.; Adams, W.; Dwyer, R.; Garman, E.; Gorsuch, J. Modeling and interpreting biological effects of mixtures in the environment: Introduction to the metal mixture modeling evaluation project. Environ. Toxicol. Chem. 2015, 34, 721–725. [Google Scholar] [CrossRef] [PubMed]
- Meyer, J.S.; Farley, K.J.; Garman, E.R. Metal mixtures modeling evaluation project: Background. Environ. Toxicol. Chem. 2015, 34, 726–740. [Google Scholar] [CrossRef] [PubMed]
- Spurgeon, D.J.; Jones, O.A.; Dorne, J.L.C.; Svendsen, C.; Swain, S.; Stürzenbaum, S.R. Systems toxicology approaches for understanding the joint effects of environmental chemical mixtures. Sci. Total Environ. 2010, 408, 3725–3734. [Google Scholar] [CrossRef] [PubMed]
- Koedrith, P.; Kim, H.; Weon, J.I.; Seo, Y.R. Toxicogenomic approaches forunderstanding molecular mechanisms of heavy metal mutagenicity and carcinogenicity. Int. J. Hyg. Environ. Health 2013, 216, 587–598. [Google Scholar] [CrossRef] [PubMed]
- Beyer, J.; Petersen, K.; Song, Y.; Ruus, A.; Grung, M.; Bakke, T.; Tollefsen, K.E. Environmental risk assessment of combined effects in aquatic ecotoxicology: A discussion paper. Mar. Environ. Res. 2014, 96, 81–91. [Google Scholar] [CrossRef] [PubMed]
- Suk, W.A.; Olden, K.; Yang, R.S. Chemical mixtures research: Significance and future perspectives. Environ. Health Perspect. 2002, 110, 891–892. [Google Scholar] [CrossRef] [PubMed]
- Sarigiannis, D.A.; Hansen, U. Considering the cumulative risk of mixtures of chemicals—A challenge for policy makers. Environ. Health 2012, 11, S18. [Google Scholar] [CrossRef] [PubMed]
| Country/Region | Pollution Source | Pb | Cd | Mn | Cu | Fe | Zn | Cr | Ni | As | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Nigeria/Kaduna | Soil | 0.87–1.41 | 0.0014–8.02 | 13.21–42.15 | 1425.0–1981.6 | 10.10–112.04 | 21.35–358.00 | [166] | |||
| Nigeria/Kaduna | Vegetable | 0.0014–0.001 | 0.013–2.12 | 0.582–8.65 | 331.6–1252 | 14.19–69.07 | 0.058–2.80 | [166] | |||
| Nigeria/Niger Delta | Water | 39 ± 33 | 2.4 ± 3.1 | 16.1 ± 7.4 | 107 ± 7.9 | 42 ± 17 | [144] | ||||
| Nigeria/Ibadan | Water | 0.162–0.195 | 0.279–0.315 | 8.744–10.307 | 5.063–5.096 | 0.052–0.059 | 0.103–0.133 | [195] | |||
| Nigeria/Benin | Soil | 227 ± 160 | 2.0 ± 2.9 | 562 ± 510 | 94.5 ± 150 | [196] | |||||
| Nigeria/Akwa Ibom | Fish | 0.013 ± 0.003 | 0.011 ± 0.004 | 81.36 ± 5.06 | 223.0 ± 23.47 | 0.044 ± 0.05 | 0.017 ± 0.02 | [197] | |||
| South Africa/Pretoria | Soil | 12.8–145 | 0.09–0.98 | 33.9–140 | 39.3–97.6 | 43.6–101 | [198] | ||||
| Kenya/Nairobi | Vegetable | 0–2.4 | 0–3.02 | 0.52–21.34 | 20.13–89.85 | 0–1.24 | [199] | ||||
| Kenya/Nairobi | Soil | 0.57–20 | 0–2.6 | 3.59–75.37 | 14.62–198.3 | 0.03–1.4 | [199] | ||||
| Ghana/Accra | Soil | 184.44 | 103.66 | 202.99 | 72.00 | [200] | |||||
| South Africa/Philippi horticultural area | Water | 0.04 ± 0.006 | 0.01 ± 0.002 | 0.02 ± 0.002 | 0.02 ± 0.003 | 0.02 ± 0.003 | 0.06 ± 0.009 | 0.02 ± 0.002 | [201] | ||
| South Africa/Philippi horticultural area | Soil | 19.24 ± 2.91 | 0.74 ± 0.18 | 96.74 ± 12.29 | 14.53 ± 2.02 | 30.13 ± 3.93 | 1.71 ± 0.40 | [201] | |||
| South Africa/Philippi horticultural area | Vegetable | 2.32 ± 0.91 | 0.22 ± 0.09 | 41.64 ± 5.21 | 5.55 ± 0.57 | 54.12 ± 9.24 | 2.68 ± 0.52 | 0.34 ± 0.25 | [202] | ||
| Zimbabwe/Bulawayo | Water | 0.19 ± 0.03 | 0.06 ± 0.03 | [202] | |||||||
| Zimbabwe/Bulawayo | Sediment | 51.67 ± 2.36 | 7.33 ± 0.76 | 79.17 ± 7.64 | 108.33 ± 17.02 | [202] | |||||
| Zimbabwe/Bulawayo | Fish | 35 | 5 | 120 | 10 | [202] | |||||
| Cameroon/Yaounde | River sediment | 20.3–249 | 2.8–15.6 | 42.8–142 | 26.8–341 | 94.7–199 | 2.68–32.7 | [203] | |||
| Ethiopia | Vegetable | 0.345 | 130 | 130 | 24.11 | [170] | |||||
| Ghana | Fish | 0.028 | 2.31 | [204] | |||||||
| Ghana/Iture | Water | 0.075 | 0.041 | 2.45 | 2.45 | [115] | |||||
| Ghana/Kumasi | Soil | 54.6 | 2.87 | 2606 | 2606 | [116] | |||||
| Ghana/Tarkwa | Water | 1.3 | [205] | ||||||||
| Kenya | Water | 0.496 | 0.01 | 1.95 | [176] | ||||||
| Kenya/Nairobi | Soil | 264 | 40 | 105 | 462 | 157 | [174] | ||||
| Namibia | Sediment | 10500 | 205 | 1950 | [206] | ||||||
| Nigeria | Herbal medicines | 27 | 4.75 | 97.5 | 25.5 | 78 | [207] | ||||
| Nigeria/Calabar | River sediment | 20 | 0.2 | 64 | 15 | 184 | 65 | 67 | [208] | ||
| Nigeria/Ibadan | River surface water | 0.046 | 0.0044 | 0.0033 | 0.018 | 0.14 | 0.0031 | [209] | |||
| Nigeria/Kano | Vegetable | 13.19 | 0.735 | 12.89 | [171] | ||||||
| Nigeria/Lagos | Soil | 67.5–426 | 1.61–5.31 | [210] | |||||||
| Nigeria/Niger Delta | Water | 0.025–0.064 | 0.01–0.11 | 0.03–0.081 | 0.03–0.09 | [177] | |||||
| Nigeria/Niger Delta | Fish | 0.3 | 0.03 | 0.53 | 0.21 | [149] | |||||
| Nigeria/Ogun | Fish | 3.4 | 2.1 | 5 | 20.35 | 2.3 | [150] | ||||
| Nigeria/Osogbo | Soil | 92.07 | 3.6 | 37.9 | 71.9 | 17.3 | [175] | ||||
| South Africa | Water | 16.3 | 72 | 42.6 | 27.6 | [175] | |||||
| Tanzania | Vegetable | 4.9 | 0.3 | [211] | |||||||
| Tanzania, along Lake Victoria | Water sediment | 54.6 | 7 | 26.1 | 83.7 | 12.9 | [212] | ||||
| Uganda | Vegetable | 18.7 | 1.87 | [173] | |||||||
| Uganda, along Lake Victoria | Water | 1.44 | 0.02 | 0.16 | 0.02 | 0.13 | [213] | ||||
| Zambia | Sediment | 12,855 ± 1445 | 1030 ± 58 | [214] | |||||||
| Zambia | Sediment | 9.75 | 0.8 | 125 | 130 | 220 | [215] | ||||
| Zimbabwe | Water | 1.02 | 0.12 | 2.48 | 2.37 | [178] | |||||
| Zimbabwe Harare | Vegetable | 6.77 | 3.68 | 0.05 | 111 | 221 | 16.1 | [172] |
| S/N | Age Groups | Sex | Place of Study (City/Country) | Condition | Reported Concentrations | Disease Scenarios | Exposure Scenarios | Ref. |
|---|---|---|---|---|---|---|---|---|
| 1 | Adults | F | Abeokuta, Southwest Nigeria | Pregnant | 54.50 ± 4.4 | Spontaneous abortion, premature delivery, pregnancy complications, still birth, hypertension, low birth weight | Non-occupationally exposed | [234] |
| 2 | Adults | M | Nkpor, Nigeria | N/A | 39.00 ± 4.00 | Increases risk of hypertension and liver damage | Occupationally exposed | [235] |
| 3 | Adults | M | Nkpor, Nigeria | N/A | 17.00 ± 4.00 | Increases risk of hypertension and liver damage | Non-occupationally exposed | [235] |
| 4 | Adults | F | Niger Delta, Nigeria | Pregnant | 40.00 ± 16.50 | Spontaneous abortion, premature delivery, pregnancy complications, still birth, hypertension, low birth weight | Non-occupationally exposed | [228] |
| 5 | Adults | F | Niger Delta, Nigeria | Non-pregnant | 27.7 ± 1.10 | Hypertension, increased risk of renal failure, cardiovascular attacks | Non-occupationally exposed | [236] |
| 6 | Adults | M/F | Oshodi, Dopemu, & Ojota in Southwest Nigeria | N/A | 155.42 148.56 122.6 | Increases risk of hypertension and liver damage, endocrine disorder, reproductive disorder | Occupationally exposed | [237] |
| 7 | Adults | M | Port Harcourt, Nigeria | N/A | 50.37 ± 24.58 | Increases risk of hypertension and liver damage | Occupationally exposed | [238] |
| 8 | Adults | M | Port Harcourt, Nigeria | N/A | 41.40 ± 26.85 | Increases risk of hypertension and liver damage | Non-exposed | [238] |
| 9 | Children | M/F | Nigeria | N/A | >10 or >20 | Increases risk of hypertension and liver damage | Non-exposed | [239] |
| 10 | Adults | M | Abeokuta, Nigeria | N/A | 48.50 ± 9.08 | Increases risk of hypertension and liver damage | Exposed | [240] |
| 11 | Adults | M | Southwest, Nigeria | N/A | 63.00 ± 9.00 | Increases risk of hypertension and liver damage | Exposed | [241] |
| 12 | Adults | M | Southwest, Nigeria | N/A | 61.00 ± 11.00 | Increases risk of hypertension and liver damage | Non-exposed | [241] |
| 13 | Children | M/F | Allada, Benin Republic | N/A | 46.6 | Lowers intelligent quotient scores, aggressive and violent behaviours | Environmentally exposed | [242] |
| 14 | Adults | F | Allada, Benin Republic | Non-pregnant | 55.1 | Increases risk of hypertension and liver damage | Environmentally exposed | [242] |
| 15 | Adults | M | Kinshasa (Democratic Republic of Congo) | N/A | 127 | Increases risk of hypertension and liver damage | Environmentally exposed | [243] |
| 16 | Children | M/F | Kinshasa(Democratic Republic of Congo) | N/A | 11.5 | Lowers intelligent quotient scores, aggressive and violent behaviours | Environmentally exposed | [243] |
| 17 | Children | M/F | Northwest, Nigeria | N/A | 143.8 | Lowers intelligent quotient scores, aggressive and violent behaviours | Environmentally exposed | [107] |
| 18 | Adults | F | Abakaliki, Nigeria | Pregnant | 40.0 ± 16.5 | Spontaneous abortion, premature delivery, pregnancy complications | Environmentally exposed | [228] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anyanwu, B.O.; Ezejiofor, A.N.; Igweze, Z.N.; Orisakwe, O.E. Heavy Metal Mixture Exposure and Effects in Developing Nations: An Update. Toxics 2018, 6, 65. https://doi.org/10.3390/toxics6040065
Anyanwu BO, Ezejiofor AN, Igweze ZN, Orisakwe OE. Heavy Metal Mixture Exposure and Effects in Developing Nations: An Update. Toxics. 2018; 6(4):65. https://doi.org/10.3390/toxics6040065
Chicago/Turabian StyleAnyanwu, Brilliance Onyinyechi, Anthonet Ndidiamaka Ezejiofor, Zelinjo Nkeiruka Igweze, and Orish Ebere Orisakwe. 2018. "Heavy Metal Mixture Exposure and Effects in Developing Nations: An Update" Toxics 6, no. 4: 65. https://doi.org/10.3390/toxics6040065
APA StyleAnyanwu, B. O., Ezejiofor, A. N., Igweze, Z. N., & Orisakwe, O. E. (2018). Heavy Metal Mixture Exposure and Effects in Developing Nations: An Update. Toxics, 6(4), 65. https://doi.org/10.3390/toxics6040065






