Occurrence of Polychlorinated Dibenzo-p-Dioxins and Dibenzofurans and Polychlorinated Biphenyls in Fruit and Vegetables from the “Land of Fires” Area of Southern Italy
Abstract
1. Introduction
2. Materials and Methods
2.1. Selection of Vegetables and Sampling Area
2.2. Chemicals and Standards
2.3. Sample Processing and Analysis
2.4. Quality Control Criteria
3. Results and Discussion
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References and Notes
- Alcock, R.E.; Gemmill, R.; Jones, K.C. Improvements to the UK PCDD/PCDF and PCB atmospheric emission inventory following an emission measurement programme. Chemosphere 1999, 38, 759–770. [Google Scholar] [CrossRef]
- USEPA (United States Environmental Protection Agency). Health Assessment for 2,3,7,8-TCDD and Related Compounds; EPA/600/EP-92/001 (public review draft); Exposure Assessment Group: Washington, DC, USA, 1994.
- PCDD/Fs and Their Effects on Human Health; Fact Sheet No. 225; WHO (World Health Organization): Geneva, Switzerland, 1999.
- Fattore, E.; Fanelli, R.; Turrini, A.; Di Domenico, A. Current dietary exposure to polychlorodibenzo-p-dioxins, polychlorodibenzofurans, and dioxin-like polychlorobiphenyls in Italy. Mol. Nutr. Food Res. 2006, 50, 915–921. [Google Scholar] [CrossRef] [PubMed]
- Health & Consumer Protection Directorate, European Commission. General Opinion of the SCF on the Risk Assessment of Dioxins and Dioxin-Like PCBs in Food. 2000. Available online: http://europa.eu.int/comm/food/fs/sc/scf/index_en.html (accessed on 21 October 2016).
- Zuccato, E.; Calvarese, S.; Mariani, G.; Mangiapan, S.; Grasso, P.; Guzzi, A.; Fanelli, R. Level, sources and toxicity of polychlorinated biphenyls in the Italian diet. Chemosphere 1999, 38, 2753–2765. [Google Scholar] [CrossRef]
- Lorán, S.; Conchello, P.; Bayarri, S.; Herrera, A. Evaluation of daily intake of PCDD/Fs and indicator PCBs in formula-fed Spanish children. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2009, 26, 1421–1431. [Google Scholar] [CrossRef] [PubMed]
- Turrio-Baldassarri, L.; Alivernini, S.; Carasi, S.; Casella, M.; Fuselli, S.; Iacovella, N.; Iamiceli, A.L.; La Rocca, C.; Scarcella, C.; Battistelli, C.L. PCB, PCDD and PCDF contamination of food of animal origin as the effect of soil pollution and the cause of human exposure in Brescia. Chemosphere 2009, 76, 278–285. [Google Scholar] [CrossRef] [PubMed]
- Freijer, J.I.; Hoogerbrugge, R.; Van Klaveren, J.D.; Traag, W.A.; Hoogenboom, L.A.P.; Liem, A.K.D. Dioxins and Dioxin-Like PCBs in Foodstuffs: Occurence and Dietary Intake in The Netherlands at the End of the 20th Century; Report Number 639102022; National Institute for Public Health and the Environment: Bilthoven, The Netherlands, 2001. [Google Scholar]
- Grassi, P.; Fattore, E.; Generoso, C.; Fanelli, R.; Arvati, M.; Zuccato, E. Polychlorobiphenyls (PCBs), polychlorinated dibenzo-p-dioxins (PCDDs) and dibenzofurans (PCDFs) in fruit and vegetables from an industrial area in northern Italy. Chemosphere 2010, 79, 292–298. [Google Scholar] [CrossRef] [PubMed]
- Aslan, S.; Kemal Korucu, M.; Karademir, A.; Durmusoglu, E. Levels of PCDD/Fs in local and non-local food samples collected from a highly polluted area in Turkey. Chemosphere 2010, 80, 1213–1219. [Google Scholar] [CrossRef] [PubMed]
- Van Dijk, C.; van Doorn, W.; van Alfen, B. Long term plant biomonitoring in the vicinity of waste incinerators in The Netherlands. Chemosphere 2015, 122, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Mazza, A.; Piscitelli, P.; Neglia, C.; Della Rosa, G.; Iannuzzi, L. Illegal Dumping of Toxic Waste and Its Effect on Human Health in Campania, Italy. Int. J. Environ. Res. Public Health 2015, 12, 6818–6831. [Google Scholar] [CrossRef] [PubMed]
- Esposito, M.; Serpe, F.P.; Neugebauer, F.; Cavallo, S.; Gallo, P.; Colarusso, G.; Baldi, L.; Iovane, G.; Serpe, L. Contamination levels and congener distribution of PCDDs, PCDFs and dioxin-like PCBs in buffalo’s milk from Caserta province (Italy). Chemosphere 2010, 79, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Esposito, M.; Serpe, F.P.; Cavallo, S.; Pellicanò, R.; Gallo, P.; Colarusso, G.; D’ambrosio, R.; Baldi, L.; Iovane, G.; Serpe, L. A survey of dioxins (PCDDs and PCDFs) and dioxin-like PCBs in sheep and goat milk from Campania, Italy. Food Addit. Contam. Part B Surveill. 2010, 3, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Fattore, E.; Fanelli, R.; Dellatte, E.; Turbini, A.; Di Domenico, A. Assessment of the dietary exposure to non-dioxin-like PCBs of the Italian general population. Chemosphere 2008, 73, 278–283. [Google Scholar] [CrossRef] [PubMed]
- Commission Regulation (2014/589/EU) of 2 June 2014 laying down methods of sampling and analysis for the control of levels of dioxins, dioxin-like PCBs and non-dioxin-like PCBs in certain foodstuffs and repealing Regulation (EU) No 252/2012 3 June 2014, Official Journal of the European Union, L 164/18.
- Diletti, G.; Ceci, R.; De Benedictis, A.; Migliorati, G.; Scortichini, G. Determination of dioxin-like polychlorinated biphenyls in feed and foods of animal origin by gas chromatography and high resolution mass spectrometry. Vet. Ital. 2007, 43, 115–128. [Google Scholar]
- Hoogerbrugge, R.; Bakker, M.I.; Hijman, W.C.; den Boer, A.C.; den Hartog, R.S.; Baumann, R.A. Dioxins in Dutch Vegetables; RIVM Report No. 310305003; RIVM: Bilthoven, The Netherlands, 2004. [Google Scholar]
- Breitweg-Lehmann, E.; Mathar, W.; Rottler, H.; Solbach, C. Background contamination of fruit and vegetables with PCDDs, PCDFs, dioxin-like PCBs and non-dioxin-like PCBs in Germany. Organohalog. Compd. 2006, 68, 201–204. [Google Scholar]
- Domingo, J.L.; Perelló, G.; Nadal, M.; Schuhmacher, M. Dietary intake of polychlorinated dibenzo-p-dioxins and dibenzofurans (PCDD/Fs) by a population living in the vicinity of a hazardous waste incinerator. Assessment of the temporal trend. Environ. Int. 2012, 50, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Schuhmacher, M.; Rodriguez-Larena, M.C.; Agramunt, M.C.; Diaz-Ferrero, J.; Domingo, J.L. Environmental impact of a new hazardous waste incinerator in Catalonia, Spain: PCDD/PCDF levels in herbage samples. Chemosphere 2002, 48, 187–193. [Google Scholar] [CrossRef]
- Schuhmacher, M.; Agramunt, M.C.; Bocio, A.; Domingo, J.L.; de Kok, H.A. Annual variation in the levels of metals and PCDD/PCDFs in soil and herbage samples collected near a cement plant. Environ. Int. 2003, 29, 415–421. [Google Scholar] [CrossRef]
- Commission Recommendation (2014/663/EU) of 11 September 2014 amending the Annex to Recommendation 2013/711/EU on the reduction of the presence of dioxins, furans and PCBs in feed and food Text with EEA relevance. OJ L 272, 13.9.2014, pp. 17–18.
- Neumann, G.; Hülster, A.; Römheld, V. PCDD/PCDF-mobilizing compounds in root exudates of Zucchini. Organohalog. Compd. 1999, 41, 331–334. [Google Scholar]
- Esposito, M.; Cavallo, S.; Rosato, G.; Chiaravalle, E.; Medico, O.; Pellicanò, R.; Soprano, V.; Baldi, L. Levels of trace elements in the fruits and vegetables from the so-called Land of fires in Campania Region (Southern Italy). Epidemiol. Prev. 2016, 40, 316–324. [Google Scholar] [PubMed]
- Gergen, I.; Harmanescu, M. Application of principal component analysis in the pollution assessment with heavy metals of vegetable food chain in the old mining areas. Chem. Cent. J. 2012, 6, 156. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, H.F. The application of electronic computers to factor analysis. Educ. Psychol. Meas. 1960, 20, 141–151. [Google Scholar] [CrossRef]
- Nowak, B. Contents and relationship of elements in human hair for a non-industrialised population in Poland. Sci. Total Environ. 1998, 209, 59–68. [Google Scholar] [CrossRef]
- INRAN. Exposure Estimation Data Set. Available online: http://www.inran.it/710/I consumi alimentari INRAN-SCAI 2005-06.html (accessed on 13 July 2016). (Updated tables 2012).
- ATSDR (Agency for Toxic Substances and Disease Registry). Toxicological Profile for Polychlorinated Biphenyls (PCBs). Prepared for US Department of Health and Human Service, November 2000.
- Lambiase, S.; Serpe, F.P.; Cavallo, S.; Rosato, G.; Baldi, L.; Neri, B.; Esposito, M. Occurrence of polychlorinated dibenzo-p-dioxins (PCDDs), dibenzofurans (PCDFs) and polychlorinated biphenyls (PCBs) in eggs from free-range hens in Campania (southern Italy) and risk evaluation. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2017, 34, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Serpe, F.P.; Scaramuzzo, A.; Maglio, P.; Lambiase, S.; Esposito, M. Monitoring plan on PCDD/Fs and DL-PCBs milk contamination in Campania region (Italy). Int. J. Anal. Mass Spectrom. Chromatogr. 2015, 3, 32–36. [Google Scholar] [CrossRef][Green Version]
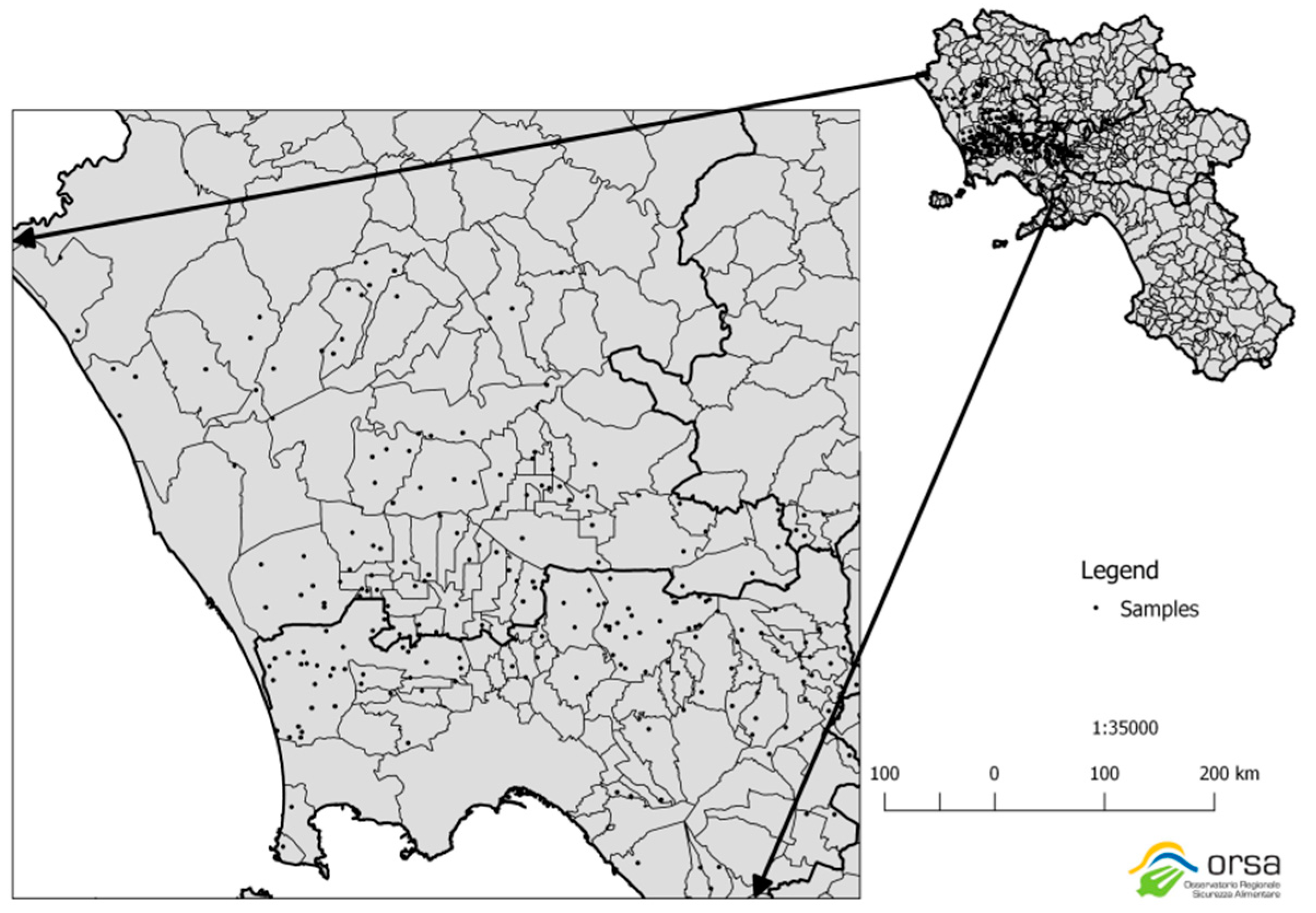
| Fruits | Group | Sample Size * |
|---|---|---|
| Ebenaceae | kaki | 2 |
| Vitaceae | grape | 4 |
| Juglandaceae | walnut | 5 |
| Betulaceae | hazelnut | 16 |
| Rosaceae | strawberry, apple, apricot, cherry, plum, peach, pear | 50 |
| Rutaceae | clementine, mandarin, lemon, orange | 8 |
| Vegetables | Group | Sample Size * |
| Cucurbitaceae | watermelon, melon, pumpkin, courgette | 11 |
| Amaryllidaceae | onion, garlic | 3 |
| Apiaceae | fennel | 3 |
| Compositae | lettuce | 3 |
| Asteraceae | chicory, endive, artichoke | 5 |
| Oleaceae | olive | 6 |
| Fabaceae | green bean, pea, broad bean | 9 |
| Poaceae | wheat, corn | 15 |
| Brassicaceae | broccoli, cabbage, kohlrabi, turnip tops, turnip, radish | 13 |
| Solanaceae | pepper, tomato, aubergine, potato, chili pepper | 75 |
| Total | 228 | |
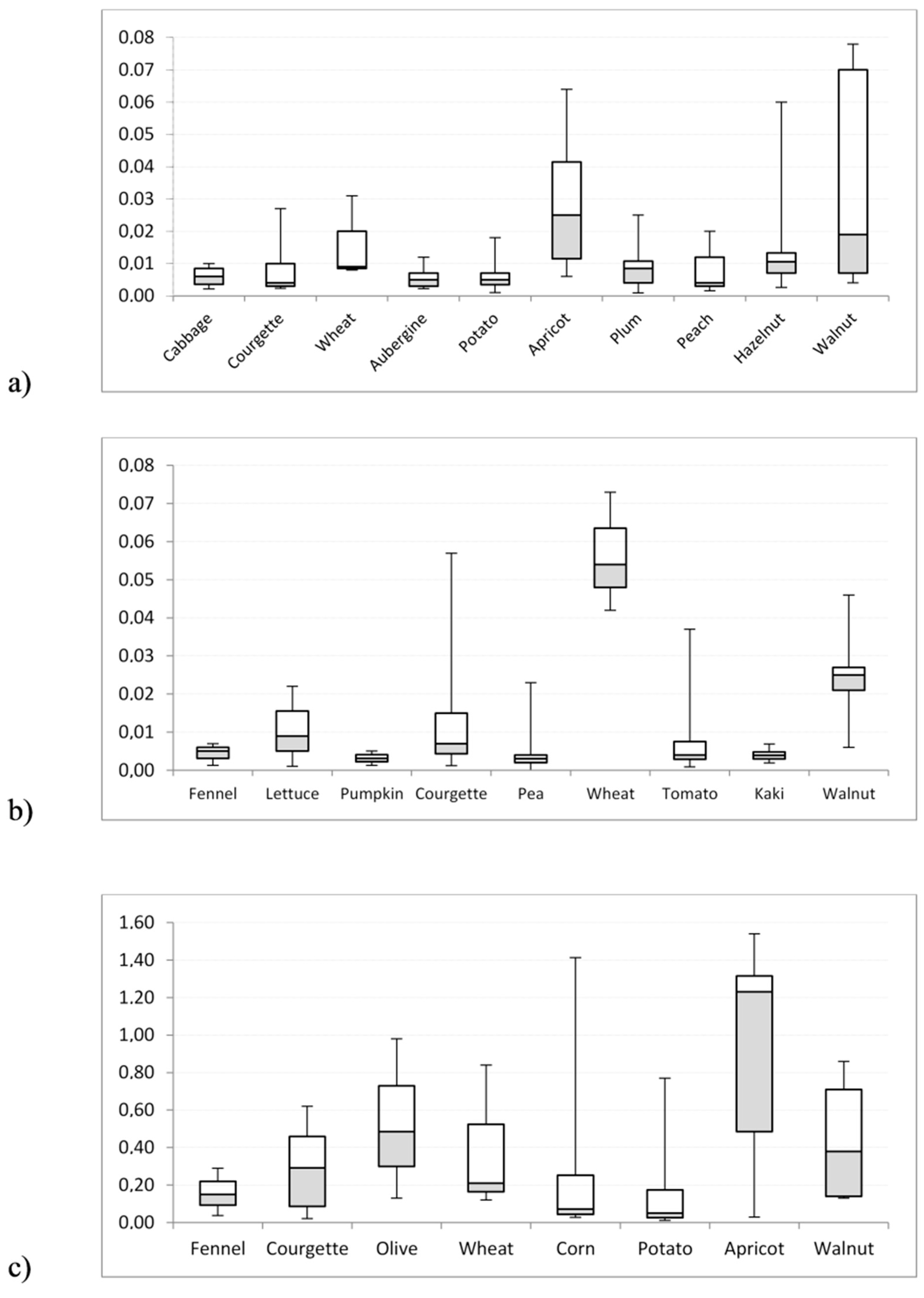
| DL-PCB (pg WHO-TEQ/g) | PCDD/F (pg WHO-TEQ/g) | NDL-PCB (ng/g) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| min | mean | median | max | min | mean | median | max | min | mean | median | max | |
| Vegetables | ||||||||||||
| Amaryllidaceae (3) * | 0.001 | 0.003 | 0.004 | 0.004 | 0.003 | 0.025 | 0.003 | 0.069 | 0.019 | 0.056 | 0.040 | 0.110 |
| Fennel (3) | 0.001 | 0.004 | 0.005 | 0.007 | 0.004 | 0.036 | 0.007 | 0.096 | 0.037 | 0.159 | 0.150 | 0.290 |
| Asteraceae (5) | 0.001 | 0.007 | 0.008 | 0.013 | 0.003 | 0.007 | 0.007 | 0.012 | 0.023 | 0.063 | 0.074 | 0.087 |
| Brassicaceae (13) | 0.002 | 0.008 | 0.006 | 0.051 | 0.002 | 0.010 | 0.008 | 0.034 | 0.026 | 0.166 | 0.064 | 1.260 |
| Lettuce (3) | 0.001 | 0.011 | 0.009 | 0.022 | 0.003 | 0.004 | 0.004 | 0.005 | 0.079 | 0.089 | 0.080 | 0.108 |
| Pumpkin (2) | 0.001 | 0.003 | 0.003 | 0.005 | 0.004 | 0.005 | 0.005 | 0.006 | 0.034 | 0.046 | 0.046 | 0.058 |
| Courgette (7) | 0.001 | 0.015 | 0.007 | 0.057 | 0.002 | 0.008 | 0.004 | 0.027 | 0.022 | 0.293 | 0.291 | 0.620 |
| Fabaceae (9) | 0.004 | 0.004 | 0.004 | 0.004 | 0.002 | 0.002 | 0.002 | 0.002 | 0.034 | 0.070 | 0.070 | 0.106 |
| Olive (6) | 0.003 | 0.024 | 0.021 | 0.055 | 0.008 | 0.040 | 0.050 | 0.072 | 0.130 | 0.522 | 0.485 | 0.980 |
| Wheat (3) | 0.042 | 0.056 | 0.054 | 0.073 | 0.008 | 0.016 | 0.009 | 0.031 | 0.120 | 0.390 | 0.210 | 0.840 |
| Corn (12) | 0.001 | 0.013 | 0.006 | 0.066 | 0.004 | 0.006 | 0.006 | 0.012 | 0.028 | 0.229 | 0.072 | 1.413 |
| Peppers (8) | 0.001 | 0.004 | 0.004 | 0.008 | 0.003 | 0.006 | 0.006 | 0.008 | 0.033 | 0.051 | 0.020 | 0.095 |
| Tomato (31) | 0.001 | 0.007 | 0.004 | 0.037 | 0.001 | 0.005 | 0.004 | 0.020 | 0.019 | 0.049 | 0.042 | 0.120 |
| Aubergine (13) | 0.001 | 0.004 | 0.003 | 0.007 | 0.002 | 0.006 | 0.005 | 0.012 | 0.019 | 0.046 | 0.042 | 0.101 |
| Potato (23) | 0.001 | 0.008 | 0.005 | 0.028 | 0.001 | 0.006 | 0.005 | 0.018 | 0.012 | 0.140 | 0.051 | 0.770 |
| Fruits | ||||||||||||
| Rosaceae (6) | 0.002 | 0.004 | 0.003 | 0.006 | 0.002 | 0.007 | 0.005 | 0.019 | 0.011 | 0.064 | 0.026 | 0.230 |
| Apricot (7) | 0.003 | 0.045 | 0.034 | 0.086 | 0.006 | 0.029 | 0.025 | 0.064 | 0.029 | 0.914 | 1.230 | 1.540 |
| Plum (22) | 0.001 | 0.007 | 0.006 | 0.023 | 0.001 | 0.008 | 0.009 | 0.025 | 0.012 | 0.050 | 0.040 | 0.098 |
| Peach (15) | 0.001 | 0.006 | 0.005 | 0.023 | 0.002 | 0.008 | 0.004 | 0.020 | 0.017 | 0.041 | 0.038 | 0.067 |
| Rutaceae (8) | 0.008 | 0.003 | 0.001 | 0.008 | 0.003 | 0.006 | 0.006 | 0.008 | 0.028 | 0.065 | 0.037 | 0.024 |
| Grape (4) | 0.002 | 0.009 | 0.009 | 0.017 | 0.004 | 0.012 | 0.007 | 0.032 | 0.140 | 0.158 | 0.150 | 0.190 |
| Melons (2) | 0.007 | 0.032 | 0.032 | 0.058 | 0.001 | 0.011 | 0.011 | 0.021 | 0.039 | 0.534 | 0.534 | 1.030 |
| Hazelnut (16) | 0.003 | 0.020 | 0.008 | 0.106 | 0.003 | 0.014 | 0.011 | 0.060 | 0.049 | 0.445 | 0.185 | 2.600 |
| Kaki (2) | 0.002 | 0.004 | 0.004 | 0.006 | 0.002 | 0.003 | 0.003 | 0.003 | 0.036 | 0.043 | 0.043 | 0.050 |
| Walnut (5) | 0.006 | 0.035 | 0.030 | 0.078 | 0.004 | 0.036 | 0.019 | 0.078 | 0.130 | 0.444 | 0.380 | 0.860 |
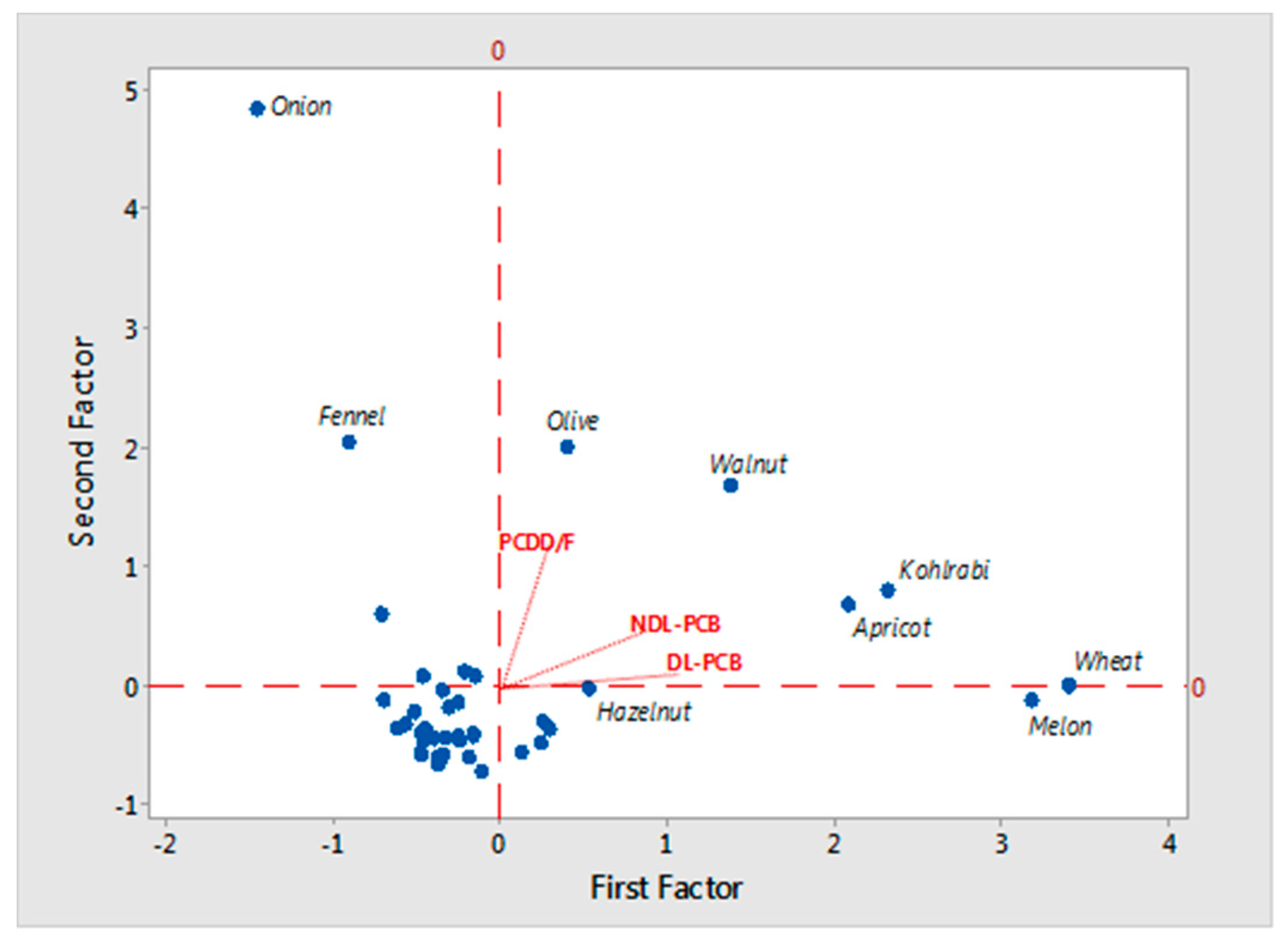
| Variable | Factor 1 | Factor 2 | Communality |
|---|---|---|---|
| DL-PCB | 0.976 | 0.207 | 1.00 |
| PCDD/F | 0.222 | 0.969 | 1.00 |
| NDL-PCB | 0.813 | 0.295 | 1.00 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Esposito, M.; De Roma, A.; Cavallo, S.; Diletti, G.; Baldi, L.; Scortichini, G. Occurrence of Polychlorinated Dibenzo-p-Dioxins and Dibenzofurans and Polychlorinated Biphenyls in Fruit and Vegetables from the “Land of Fires” Area of Southern Italy. Toxics 2017, 5, 33. https://doi.org/10.3390/toxics5040033
Esposito M, De Roma A, Cavallo S, Diletti G, Baldi L, Scortichini G. Occurrence of Polychlorinated Dibenzo-p-Dioxins and Dibenzofurans and Polychlorinated Biphenyls in Fruit and Vegetables from the “Land of Fires” Area of Southern Italy. Toxics. 2017; 5(4):33. https://doi.org/10.3390/toxics5040033
Chicago/Turabian StyleEsposito, Mauro, Antonella De Roma, Stefania Cavallo, Gianfranco Diletti, Loredana Baldi, and Giampiero Scortichini. 2017. "Occurrence of Polychlorinated Dibenzo-p-Dioxins and Dibenzofurans and Polychlorinated Biphenyls in Fruit and Vegetables from the “Land of Fires” Area of Southern Italy" Toxics 5, no. 4: 33. https://doi.org/10.3390/toxics5040033
APA StyleEsposito, M., De Roma, A., Cavallo, S., Diletti, G., Baldi, L., & Scortichini, G. (2017). Occurrence of Polychlorinated Dibenzo-p-Dioxins and Dibenzofurans and Polychlorinated Biphenyls in Fruit and Vegetables from the “Land of Fires” Area of Southern Italy. Toxics, 5(4), 33. https://doi.org/10.3390/toxics5040033






