Assessment of Per- and Poly-Fluoroalkyl Substances (PFAS) and Polybrominated Diphenyl Ethers (PBDEs) in Surface Waters Used for Urban Water Supply in Brazil
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Sample Collection
2.3. Sample Preparation and Extraction
2.4. Instrumental Conditions
2.5. Quality Control
2.6. Statistical Analysis
3. Results
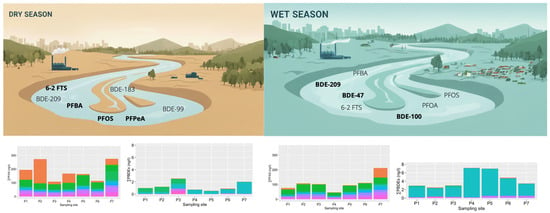
3.1. Occurrence and Concentrations of PFAS in Itupararanga Reservoir and in the Sorocaba River
3.2. Occurrence and Concentrations of PBDEs in the Sorocaba River
3.3. Current Regulations and Potential Risks of Persistent Pollutants on the Environment and Human Health
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- OECD (The Organisation for Economic Co-operation and Development). Toward a New Comprehensive Global Database of Per- and Polyfluoroalkyl Substances (PFASs): Summary Report on Updating the OECD 2007 List of Per- and Polyfluoroalkyl Substances (PFASs); OECD Publishing: Paris, France, 2018; Available online: https://www.oecd.org/content/dam/oecd/en/publications/reports/2018/05/summary-report-on-the-new-comprehensive-global-database-of-per-and-polyfluoroalkyl-substances-pfass_73d72705/1a14ad6c-en.pdf (accessed on 29 December 2025).
- Podder, A.; Sadmani, A.H.M.A.; Reinhart, D.; Chang, N.-B.; Goel, R. Per and polyfluoroalkyl substances (PFAS) as a contaminant of emerging concern in surface water: A transboundary review of their occurrences and toxicity effects. J. Hazard. Mater. 2021, 419, 126361. [Google Scholar] [CrossRef]
- Abdallah, M.A.-E.; Drage, D.S.; Sharkey, M.; Berresheim, H.; Harrad, S. A rapid method for the determination of brominated flame retardant concentrations in plastics and textiles entering the waste stream. J. Sep. Sci. 2018, 40, 3749–3922. [Google Scholar] [CrossRef]
- Kuang, J.; Abdallah, M.A.-E.; Harrad, S. Brominated flame retardants in black plastic kitchen utensils: Concentrations and human exposure implications. Sci. Total Environ. 2018, 610–611, 1138–1146. [Google Scholar] [CrossRef]
- Sunderland, E.M.; Hu, X.C.; Dassuncao, C.; Tokranov, A.K.; Wagner, C.C.; Allen, J.G. A review of the pathways of human exposure to poly- and perfluoroalkyl substances (PFASs) and present understanding of health effects. J. Expo. Sci. Environ. Epidemiol. 2019, 29, 131–147. [Google Scholar] [CrossRef]
- Starling, A.P.; Adgate, J.L.; Hamman, R.F.; Kechris, K.; Calafat, A.M.; Ye, X.; Dabelea, D. Perfluoroalkyl substances during pregnancy and offspring weight and adiposity at birth: Examining mediation by maternal fasting glucose in the Healthy Start Study. Environ. Health Perspect. 2017, 125, 067016. [Google Scholar] [CrossRef]
- Kashino, I.; Sasaki, S.; Okada, E.; Matsuura, H.; Goudarzi, H.; Miyashita, C.; Ito, Y.M.; Araki, A.; Kishi, R. Prenatal exposure to 11 perfluoroalkyl substances and fetal growth: A large-scale, prospective birth cohort study. Environ. Int. 2020, 136, 105355. [Google Scholar] [CrossRef]
- Coperchini, F.; Awwad, O.; Rotondi, M.; Santini, F.; Imbriani, M.; Chiovato, L. Thyroid disruption by perfluorooctane sulfonate (PFOS) and perfluorooctanoate (PFOA). J. Endocrinol. Investig. 2017, 40, 105–121. [Google Scholar] [CrossRef] [PubMed]
- IARC (International Agency for Research on Cancer). Perfluorooctanoic Acid; IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, No. 110; IARC: Lyon, France, 2016; pp. 1–74.
- Grandjean, P.; Landrigan, P.J. Neurobehavioural effects of developmental toxicity. Lancet Neurol. 2014, 13, 330–338. [Google Scholar] [CrossRef] [PubMed]
- Martin, O.V.; Evans, R.M.; Faust, M.; Kortenkamp, A. A human mixture risk assessment for neurodevelopmental toxicity associated with polybrominated diphenyl ethers used as flame retardants. Environ. Health Perspect. 2017, 125, 087008. [Google Scholar] [CrossRef] [PubMed]
- Sheikh, I.A.; Beg, M.A. Structural studies on the endocrine-disrupting role of polybrominated diphenyl ethers (PBDEs) in thyroid diseases. Environ. Sci. Pollut. Res. 2020, 27, 37866–37876. [Google Scholar] [CrossRef]
- Meeker, J.D.; Stapleton, H.M. House dust concentrations of organophosphate flame retardants in relation to hormone levels and semen quality parameters. Environ. Health Perspect. 2010, 118, 318–323. [Google Scholar] [CrossRef] [PubMed]
- Arowolo, O.; Pilsner, J.R.; Sergeyev, O.; Suvorov, A. Mechanisms of male reproductive toxicity of polybrominated diphenyl ethers. Int. J. Mol. Sci. 2022, 23, 14229. [Google Scholar] [CrossRef]
- Schreder, E.; Zheng, G.; Sathyanarayana, S.; Gunaje, N.; Hu, M.; Salamova, A. Brominated flame retardants in breast milk from the United States: First detection of bromophenols in U.S. breast milk. Environ. Pollut. 2023, 334, 122028. [Google Scholar] [CrossRef]
- Torres-Moreno, A.C.; Mejia-Grau, K.; Puente-DelaCruz, L.; Codling, G.; Villa, A.L.; Ríos-Marquez, O.; Patequiva-Chauta, L.; Cobo, M.; Johnson-Restrepo, B. Polybrominated diphenyl ethers (PBDEs), polychlorinated biphenyls (PCBs), organochlorine pesticides (OCPs) in human breast milk from Colombia: A probabilistic risk assessment approach. Chemosphere 2023, 339, 139597. [Google Scholar] [CrossRef]
- Mach, V.; Straková, J. Forever Chemicals Round and Round: Contamination of Water Bodies with Perfluorinated Substances and Brominated Flame Retardants in the Prague Area; Arnika—Toxics Waste Program: Prague, Czech Republic, 2020; 42p. [Google Scholar]
- AMAP (Arctic Monitoring and Assessment Programme). POPs and Chemicals of Emerging Arctic Concern: Influence of Climate Change; Summary for Policy-Makers; AMAP: Tromsø, Norway, 2021; 16p.
- Bai, X.; Son, Y. Perfluoroalkyl substances (PFAS) in surface water and sediments from two urban watersheds in Nevada, USA. Sci. Total Environ. 2021, 751, 141622. [Google Scholar] [CrossRef] [PubMed]
- Harrad, S.; Abou Elwafa Abdallah, M.; Rose, N.L.; Turner, S.D.; Davidson, T.A. Current use brominated flame retardants in water, sediment, and fish from English lakes. Environ. Sci. Technol. 2009, 43, 9077–9083. [Google Scholar] [CrossRef]
- Yu, G.; Bu, Q.; Cao, Z.; Du, X.; Xia, J.; Wu, M.; Huang, J. Brominated flame retardants (BFRs): A review on environmental contamination in China. Chemosphere 2016, 150, 479–490. [Google Scholar] [CrossRef]
- Garg, S.; Kumar, P.; Mishra, V.; Guijt, R.; Singh, P.; Dumée, L.F.; Sharma, R.S. A review on the sources, occurrence and health risks of per /poly fluoroalkyl substances (PFAS) arising from the manufacture and disposal of electric and electronic products. J. Water Process Eng. 2020, 38, 101683. [Google Scholar] [CrossRef]
- USEPA (United States Environmental Protection Agency). Navigation Panel to PFAS Structure Lists. 2025. Available online: https://comptox.epa.gov/dashboard/chemical-lists/pfasstruct (accessed on 29 December 2025).
- Torres, F.B.M.; Guida, Y.; Weber, R.; Torres, J.P.M. Brazilian overview of per- and polyfluoroalkyl substances listed as persistent organic pollutants in the Stockholm Convention. Chemosphere 2022, 291, 132674. [Google Scholar] [CrossRef]
- Quinete, N.; Wu, Q.; Zhang, T.; Yun, S.H.; Moreira, I.; Kannan, K. Specific profiles of perfluorinated compounds in surface and drinking waters and accumulation in mussels, fish, and dolphins from southeastern Brazil. Chemosphere 2009, 77, 863–869. [Google Scholar] [CrossRef] [PubMed]
- Löfstedt Gilljam, J.; Leonel, J.; Cousins, I.T.; Benskin, J.P. Is ongoing sulfluramid use in South America a significant source of perfluorooctanesulfonate (PFOS)? Production inventories, environmental fate, and local occurrence. Environ. Sci. Technol. 2016, 50, 653–659. [Google Scholar] [CrossRef]
- Schwanz, T.G.; Llorca, M.; Farré, M.; Barceló, D. Perfluoroalkyl substances assessment in drinking waters from Brazil, France and Spain. Sci. Total Environ. 2016, 539, 143–152. [Google Scholar] [CrossRef]
- Nascimento, R.A.; Nunoo, D.B.O.; Bizkarguenaga, E.; Schultes, L.; Zabaleta, I.; Benskin, J.P.; Spanó, S.; Leonel, J. Sulfluramid use in Brazilian agriculture: A source of per and polyfluoroalkyl substances (PFASs) to the environment. Environ. Pollut. 2018, 242, 1436–1443. [Google Scholar] [CrossRef]
- Stefano, P.H.P.; Roisenberg, A.; D’Anna Acayaba, R.; Pereira Roque, A.; Rodrigues Bandoria, D.; Soares, A.; Montagner, C.C. Occurrence and distribution of per and polyfluoroalkyl substances (PFAS) in surface and groundwaters in an urbanized and agricultural area, southern Brazil. Environ. Sci. Pollut. Res. 2023, 30, 6159–6169. [Google Scholar] [CrossRef]
- Rodrigues, D.A.S.; Starling, M.C.V.M.; Barros, A.L.C.; Santos, M.C.; da Silva, E.S.; Viana, G.C.C.; Ribeiro, L.F.S.; Simcik, M.F.; Amorim, C.C. Occurrence of antibiotics, hormones and PFAs in surface water from a Nile tilapia aquaculture facility in a Brazilian hydroelectric reservoir. Chemosphere 2024, 352, 141444. [Google Scholar] [CrossRef]
- Starling, M.C.V.M.; Rodrigues, D.A.S.; Miranda, G.A.; Jo, S.; Amorim, C.C.; Ankley, G.T.; Simcik, M. Occurrence and potential ecological risks of PFAS in Pampulha Lake, Brazil, a UNESCO World Heritage site. Sci. Total Environ. 2024, 948, 174586. [Google Scholar] [CrossRef] [PubMed]
- Dorneles, P.R.; Lailson Brito, J.; Dirtu, A.C.; Weijs, L.; Azevedo, A.F.; Torres, J.P.M.; Malm, O.; Neels, H.; Blust, R.; Das, K.; et al. Anthropogenic and naturally produced organobrominated compounds in marine mammals from Brazil. Environ. Int. 2010, 36, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Quinete, N.; Lavandier, R.; Dias, P.; Taniguchi, S.; Montone, R.; Moreira, I. Specific profiles of polybrominated diphenylethers (PBDEs) and polychlorinated biphenyls (PCBs) in fish and tucuxi dolphins from the estuary of Paraíba do Sul River, Southeastern Brazil. Mar. Pollut. Bull. 2011, 62, 440–446. [Google Scholar] [CrossRef] [PubMed]
- Yogui, G.T.; Santos, M.C.O.; Bertozzi, C.P.; Sericano, J.L.; Montone, R.C. PBDEs in the blubber of marine mammals from coastal areas of São Paulo, Brazil, southwestern Atlantic. Mar. Pollut. Bull. 2011, 62, 2666–2670. [Google Scholar] [CrossRef]
- Leonel, J.; Taniguchi, S.; Sasaki, D.K.; Cascaes, M.J.; Dias, P.S.; Botta, S.; Santos, M.C.O.; Montone, R.C. Contamination by chlorinated pesticides, PCBs and PBDEs in Atlantic spotted dolphin (Stenella frontalis) in western South Atlantic. Chemosphere 2012, 86, 741–746. [Google Scholar] [CrossRef]
- Leonel, J.; Sericano, J.L.; Secchi, E.R.; Bertozzi, C.; Fillmann, G.; Montone, R.C. PBDE levels in franciscana dolphin (Pontoporia blainvillei): Temporal trend and geographical comparison. Sci. Total Environ. 2014, 493, 405–410. [Google Scholar] [CrossRef]
- Magalhães, C.A.; Taniguchi, S.; Cascaes, M.J.; Montone, R.C. PCBs, PBDEs and organochlorine pesticides in crabs Hepatus pudibundus and Callinectes danae from Santos Bay, State of São Paulo, Brazil. Mar. Pollut. Bull. 2012, 64, 662–667. [Google Scholar] [CrossRef]
- Magalhães, C.A.; Taniguchi, S.; Lourenço, R.A.; Montone, R.C. Organochlorine pesticides, PCBs, and PBDEs in liver and muscle tissues of Paralonchurus brasiliensis, Trichiurus lepturus and Cathorops spixii in Santos Bay and surrounding area, São Paulo, Brazil. Reg. Stud. Mar. Sci. 2017, 16, 42–48. [Google Scholar] [CrossRef]
- Cipro, C.V.Z.; Colabuono, F.I.; Taniguchi, S.; Montone, R.C. Polybrominated diphenyl ethers in fat samples from White chinned Petrels (Procellaria aequinoctialis) and Spectacled Petrels (P. conspicillata) off southern Brazil. Mar. Pollut. Bull. 2013, 77, 396–399. [Google Scholar] [CrossRef]
- Da Silva, S.F.G.; Brüning, I.M.R.A.; Montone, R.C.; Taniguchi, S.; Cascaes, M.J.; Dias, P.S.; Lavandier, R.C.; Hauser Davis, R.A.; Moreira, I. Polybrominated Diphenyl Ethers (PBDEs) and Polychlorinated Biphenyls (PCBs) in mussels and two fish species from the estuary of the Guanabara Bay, Southeastern Brazil. Bull. Environ. Contam. Toxicol. 2013, 91, 261–266. [Google Scholar] [CrossRef]
- Lavandier, R.; Arêas, J.; Dias, P.S.; Taniguchi, S.; Montone, R.C.; de Moura, J.F.; Quinete, N.; Siciliano, S.; Moreira, I. An assessment of PCB and PBDE contamination in two tropical dolphin species from the Southeastern Brazilian coast. Mar. Pollut. Bull. 2015, 101, 947–953. [Google Scholar] [CrossRef]
- Lavandier, R.; Arêas, J.; Quinete, N.; de Moura, J.F.; Taniguchi, S.; Montone, R.C.; Siciliano, S.; Moreira, I. PCB and PBDE levels in a highly threatened dolphin species from the Southeastern Brazilian coast. Environ. Pollut. 2016, 208, 442–449. [Google Scholar] [CrossRef]
- Lavandier, R.; Quinete, N.; Hauser Davis, R.A.; Dias, P.S.; Taniguchi, S.; Montone, R.C.; Moreira, I. Polychlorinated biphenyls (PCBs) and polybrominated diphenyl ethers (PBDEs) in three fish species from an estuary in the southeastern coast of Brazil. Chemosphere 2013, 90, 2435–2443. [Google Scholar] [CrossRef]
- Cascaes, M.J.; Oliveira, R.T.; Ubarana, M.M.; Sato, R.M.; Baldassin, P.; Colabuono, F.I.; Leonel, J.; Taniguchi, S.; Weber, R.R. Persistent organic pollutants in liver of Brazilian sharpnose shark (Rhizoprionodon lalandii) from southeastern coast of Brazil. Mar. Pollut. Bull. 2014, 86, 591–593. [Google Scholar] [CrossRef] [PubMed]
- Burgos Melo, H.D.; de Souza Araujo, J.; Benavides Garzón, L.G.; Macedo, J.C.; Cardoso, R.; Mancini, S.D.; Harrad, S.; Rosa, A.H. Concentrations and legislative aspects of PBDEs in plastic of waste electrical and electronic equipment in Brazil. Sci. Total Environ. 2024, 906, 167349. [Google Scholar] [CrossRef]
- Smith, W.S.; Salmazzi, B.A.; Possomato, H.M.; Oliveira, L.C.A.; Almeida, M.A.G.; Pupo, R.H.; Tavares, T.A. A bacia do rio Sorocaba: Caracterização e principais impactos. Rev. Cient. Imapes 2005, 3, 51–57. [Google Scholar]
- Li, X.; Fatowe, M.; Cui, D.; Quinete, N. Assessment of per and polyfluoroalkyl substances in Biscayne Bay surface waters and tap waters from South Florida. Sci. Total Environ. 2022, 806, 150393. [Google Scholar] [CrossRef] [PubMed]
- Cristale, J.; Katsoyiannis, A.; Sweetman, A.J.; Jones, K.C.; Lacorte, S. Occurrence and risk assessment of organophosphorus and brominated flame retardants in the River Aire (UK). Environ. Pollut. 2013, 179, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Cristale, J.; Quintana, J.; Chaler, R.; Ventura, F.; Lacorte, S. Gas chromatography/mass spectrometry comprehensive analysis of organophosphorus, brominated flame retardants, by products and formulation intermediates in water. J. Chromatogr. A 2012, 1241, 1–12. [Google Scholar] [CrossRef]
- Harrad, S.; Drage, D.S.; Sharkey, M.; Berresheim, H. Perfluoroalkyl substances and brominated flame retardants in landfill-related air, soil, and groundwater from Ireland. Sci. Total Environ. 2020, 705, 135834. [Google Scholar] [CrossRef]
- Ackerman Grunfeld, D.; Gilbert, D.; Hou, J.; Jones, A.M.; Lee, M.J.; Kibbey, T.C.; O’Carroll, D.M. Underestimated burden of per and polyfluoroalkyl substances in global surface waters and groundwaters. Nat. Geosci. 2024, 17, 340–346. [Google Scholar] [CrossRef]
- Riaz, R.; Junaid, M.; Rehman, M.Y.A.; Iqbal, T.; Khan, J.A.; Dong, Y.; Yue, L.; Chen, Y.; Xu, N.; Malik, R.N. Spatial distribution, compositional profile, sources, ecological and human health risks of legacy and emerging per and polyfluoroalkyl substances (PFASs) in freshwater reservoirs of Punjab, Pakistan. Sci. Total Environ. 2023, 856, 159144. [Google Scholar] [CrossRef]
- Möller, A.; Ahrens, L.; Surm, R.; Westerveld, J.; van der Wielen, F.; Ebinghaus, R.; de Voogt, P. Distribution and sources of polyfluoroalkyl substances (PFAS) in the River Rhine watershed. Environ. Pollut. 2010, 158, 3243–3250. [Google Scholar] [CrossRef]
- Labadie, P.; Chevreuil, M. Biogeochemical dynamics of perfluorinated alkyl acids and sulfonates in the River Seine (Paris, France) under contrasting hydrological conditions. Environ. Pollut. 2011, 159, 3634–3639. [Google Scholar] [CrossRef]
- Ahrens, L.; Gerwinski, W.; Theobald, N.; Ebinghaus, R. Sources of polyfluoroalkyl compounds in the North Sea, Baltic Sea and Norwegian Sea: Evidence from their spatial distribution in surface water. Mar. Pollut. Bull. 2010, 60, 255–260. [Google Scholar] [CrossRef]
- Konwick, B.J.; Tomy, G.T.; Ismail, N.; Peterson, J.T.; Fauver, R.J.; Higginbotham, D.; Fisk, A.T. Concentrations and patterns of perfluoroalkyl acids in Georgia, USA surface waters near and distant to a major use source. Environ. Toxicol. Chem. 2008, 27, 2011–2018. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Xie, Q.; Liu, X.; Wang, J. Occurrence, distribution and risk assessment of polychlorinated biphenyls and polybrominated diphenyl ethers in nine water sources. Ecotoxicol. Environ. Saf. 2015, 115, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Pei, J.; Yao, H.; Wang, H.; Li, H.; Lu, S.; Zhang, X.; Xiang, X. Polybrominated diphenyl ethers (PBDEs) in water, surface sediment, and suspended particulate matter from the Yellow River, China: Levels, spatial and seasonal distribution, and source contribution. Mar. Pollut. Bull. 2018, 129, 106–113. [Google Scholar] [CrossRef]
- Trinh, M.M.; Tsai, C.L.; Chang, M.B. Characterization of polybrominated diphenyl ethers (PBDEs) in various aqueous samples in Taiwan. Sci. Total Environ. 2019, 649, 388–395. [Google Scholar] [CrossRef] [PubMed]
- Daso, A.P.; Fatoki, O.S.; Odendaal, J.P. Occurrence of polybrominated diphenyl ethers (PBDEs) and 2,2′,4,4′,5,5′ hexabromobiphenyl (BB 153) in water samples from the Diep River, Cape Town, South Africa. Environ. Sci. Pollut. Res. Int. 2013, 20, 5168–5176. [Google Scholar] [CrossRef]
- Yang, C.; Harrad, S.; Abdallah, M.A.-E.; Desborough, J.; Rose, N.L.; Turner, S.D.; Davidson, T.A.; Goldsmith, B. Polybrominated diphenyl ethers (PBDEs) in English freshwater lakes, 2008–2012. Chemosphere 2014, 110, 41–47. [Google Scholar] [CrossRef]
- Ruge, Z.; Muir, D.; Helm, P.; Lohmann, R. Concentrations, Trends, and Air–Water Exchange of PAHs and PBDEs Derived from Passive Samplers in Lake Superior in 2011. Environ. Sci. Technol. 2015, 49, 13777–13786. [Google Scholar] [CrossRef]
- Olutona, G.O.; Oyekunle, J.A.; Ogunfowokan, A.O.; Fatoki, O.S. Concentrations of Polybrominated Diphenyl Ethers (PBDEs) in Water from Asunle Stream, Ile-Ife, Nigeria. Toxics 2017, 5, 13. [Google Scholar] [CrossRef]
- Tongu, S.M.; Sha’Ato, R.; Okonkwo, J.O.; Olukunle, O.I.; Tor-Anyiin, T.A.; Eneji, I.S. Levels of Polybrominated Diphenyl Ethers (PBDEs) in Water and Sediment from Open City Drains in Makurdi Metropolitan Area, North Central Nigeria. Environ. Monit. Assess. 2021, 193, 244. [Google Scholar] [CrossRef]
- Ohoro, C.R.; Adeniji, A.O.; Okoh, A.I.; Okoh, O.O. Polybrominated Diphenyl Ethers in the Environmental Systems: A Review. J. Environ. Health Sci. Eng. 2021, 19, 1229–1247. [Google Scholar] [CrossRef]
- Hwang, I.K.; Kang, H.H.; Lee, I.S.; Oh, J.E. Assessment of Characteristic Distribution of PCDD/Fs and BFRs in Sludge Generated at Municipal and Industrial Wastewater Treatment Plants. Chemosphere 2012, 88, 888–894. [Google Scholar] [CrossRef]
- Ju, T.; Ge, W.; Jiang, T.; Chai, C. Polybrominated Diphenyl Ethers in Dissolved and Suspended Phases of Seawater and in Surface Sediment from Jiaozhou Bay, North China. Sci. Total Environ. 2016, 557, 571–578. [Google Scholar] [CrossRef] [PubMed]
- Li, W.L.; Ma, W.L.; Jia, H.L.; Hong, W.J.; Moon, H.B.; Nakata, H.; Minh, N.H.; Sinha, R.K.; Chi, K.H.; Kannan, K.; et al. Polybrominated Diphenyl Ethers (PBDEs) in Surface Soils across Five Asian Countries: Levels, Spatial Distribution, and Source Contribution. Environ. Sci. Technol. 2016, 50, 12779–12788. [Google Scholar] [CrossRef] [PubMed]
- Souza Araujo, J.; dos Santos, I.C.; Burgos Melo, H.D.; Rosa, A.H. Polybrominated diphenyl ethers in indoor dust from Brazil: Assessing demographic differences and human health exposure. Environ. Sci. Pollut. Res. Int. 2024, 31, 48122–48134. [Google Scholar] [CrossRef] [PubMed]
- Burgos Melo, H.D.; de Souza Araujo, J.; Cardoso, R.; Frascareli, D.; Gontijo, E.S.J.; Donnini Mancini, S.; Harrad, S.; Rosa, A.H. PBDE concentrations in wastes from construction and demolition, soft furnishings, and end of life vehicles in Brazil. Environ. Res. 2025, 287, 122963. [Google Scholar] [CrossRef]
- USEPA (United States Environmental Protection Agency). Per and Polyfluoroalkyl Substances (PFAS) Final PFAS National Primary Drinking Water Regulation; U.S. EPA: Washington, DC, USA, 2024. Available online: https://www.epa.gov/sdwa/and-polyfluoroalkyl-substances-pfas (accessed on 29 December 2025).
- EU (European Union). Commission Delegated Regulation (EU) 2020/784 of 8 April 2020 Amending Annex I to Regulation (EU) 2019/1021 of the European Parliament and of the Council as Regards the Listing of Perfluorooctanoic Acid (PFOA), Its Salts and PFOA Related Compounds. Official Journal of the European Union: L 188I. 15 June 2020. Available online: https://eur-lex.europa.eu/eli/reg_del/2020/784/oj (accessed on 29 December 2025).
- EU (European Union). Regulation (EU) 2019/1021 of the European Parliament and of the Council of 20 June 2019 on Persistent Organic Pollutants (recast). Official Journal of the European Union: L 169. 25 June 2019. Available online: https://eur-lex.europa.eu/eli/reg/2019/1021/oj/eng (accessed on 29 December 2025).
- HEPA (Heads of EPA Australia and New Zealand). Draft PFAS National Environmental Management Plan Version 3.0; HEPA: Melbourne, Australia, 2022. Available online: https://www.waternz.org.nz/Attachment?Action=Download&Attachment_id=5498 (accessed on 29 December 2025).





| Compound | DRY | WET | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| P1 | P2 | P3 | P4 | P5 | P6 | P7 | P1 | P2 | P3 | P4 | P5 | P6 | P7 | |
| 4-2 FTS | 0.46 ± 0.07 | 1.6 ± 1.81 | 0.11 ± 0.05 | 0.48 ± 0.13 | 0.02 ± 0 | 2.78 ± 3.81 | 0.27 ± 0.03 | 0.05 ± 0.02 | 0.01 ± 0.02 | 0.05 ± 0 | 0.01 ± 0.01 | 0.01 ± 0.02 | <MDL | 0.3 ± 0.21 |
| 6-2FTS | 69.82 ± 1.3 | 155.44 ± 18.86 | 16.79 ± 11.08 | 64.32 ± 15.55 | 6.53 ± 0.02 | 4.39 ± 0.22 | 39.46 ± 0.66 | 8.77 ± 0.04 | 1.55 ± 0.06 | 1.19 ± 0.05 | 0.8 ± 0.09 | 1.39 ± 0.04 | 1.41 ± 0.09 | 63.01 ± 39.16 |
| 8-2 FTS | 0.68 ± 0.09 | 1.58 ± 1.07 | 0.22 ± 0.04 | 0.42 ± 0.07 | 0.4 ± 0.11 | 1.84 ± 2.42 | 1.19 ± 0.24 | 0.05 ± 0.01 | <MDL | <MDL | <MDL | 0.01 ± 0.01 | 0.02 ± 0.03 | 0.33 ± 0.2 |
| Adona | <MDL | <MDL | <MDL | <MDL | <MDL | <MDL | <MDL | <MDL | <MDL | <MDL | <MDL | <MDL | <MDL | <MDL |
| FBSA | 0.06 ± 0.03 | 0.03 ± 0 | 0.11 ± 0.06 | 0.16 ± 0.02 | 0.15 ± 0.11 | 0.14 ± 0.06 | 0.18 ± 0.09 | 0.05 ± 0.02 | 0.18 ± 0.07 | 0.27 ± 0.1 | 0.09 ± 0.06 | 0.26 ± 0.02 | 0.21 ± 0 | 0.41 ± 0.19 |
| FHxSA | 0.03 ± 0.04 | <MDL | 0.01 ± 0.01 | 0.01 ± 0.01 | 0.02 ± 0.01 | <MDL | 0.08 ± 0.08 | 0.01 ± 0.01 | 0.05 ± 0.07 | 0.04 ± 0.01 | 0.01 ± 0 | 0.02 ± 0 | 0.01 ± 0.01 | 0.14 ± 0.09 |
| FOSA | 2.03 ± 0.41 | 2.88 ± 0.82 | 2.56 ± 0.85 | 2.92 ± 1.41 | 2.01 ± 0.55 | 2.53 ± 0.04 | 3.58 ± 0.21 | 1.47 ± 0.09 | 1.18 ± 0.32 | 1.1 ± 0.32 | 0.8 ± 0.28 | 1.41 ± 0.27 | 0.99 ± 0.21 | 0.88 ± 0.16 |
| GenX | 0.1 ± 0.1 | <MDL | <MDL | <MDL | <MDL | 0.11 ± 0.16 | 0.13 ± 0.22 | 0.4 ± 0.15 | 4.81 ± 6.43 | 0.87 ± 0.53 | 0.66 ± 0.55 | 0.53 ± 0.2 | 0.54 ± 0.76 | 0.35 ± 0.36 |
| N-EtFOSAA | 0.07 ± 0.12 | 0.22 ± 0.38 | 0.06 ± 0.08 | 0.06 ± 0.03 | 0.23 ± 0.14 | 0.2 ± 0.17 | 0.67 ± 0.24 | <MDL | <MDL | 0.04 ± 0.03 | 0.03 ± 0.05 | 0.14 ± 0.08 | 0.15 ± 0.05 | 0.09 ± 0.08 |
| N-MeFOSAA | 0.07 ± 0.01 | 0.04 ± 0.07 | 0.05 ± 0.04 | 0.06 ± 0.06 | 0.24 ± 0.03 | 0.01 ± 0.01 | 1.26 ± 0.38 | 0.05 ± 0 | 0.03 ± 0.04 | 0.09 ± 0.01 | <MDL | <MDL | <MDL | 0.04 ± 0.04 |
| PFBA | 48.77 ± 0.49 | 39 ± 3.65 | 37.61 ± 1.89 | 44.53 ± 0.29 | 72.25 ± 62.4 | 44.68 ± 0.7 | 44.01 ± 1.29 | 34 ± 11.34 | 47.07 ± 0.63 | 48.77 ± 0.46 | 21.13 ± 0.87 | 41.06 ± 0.84 | 34.99 ± 0.93 | 41.05 ± 0.66 |
| PFBS | 4.55 ± 0.08 | 4.25 ± 0.17 | 4.59 ± 0.05 | 3.83 ± 0.14 | 4.76 ± 0.02 | 4.05 ± 0.03 | 9.20 ± 1.05 | 2.45 ± 1 | 4.68 ± 0.37 | 3.51 ± 0.1 | 2.05 ± 0.1 | 3.6 ± 0.01 | 4.78 ± 0.01 | 6.1 ± 0.3 |
| PFDA | 1.06 ± 0.03 | 1.15 ± 0.14 | 0.68 ± 0.07 | 0.73 ± 0.07 | 1.04 ± 0.1 | 0.76 ± 0.15 | 1.83 ± 0.23 | 0.43 ± 0.12 | 0.62 ± 0.02 | 0.38 ± 0 | 0.22 ± 0 | 0.39 ± 0 | 0.56 ± 0 | 1.03 ± 0.07 |
| PFDoA | 0.17 ± 0.01 | 0.84 ± 0.24 | 0.2 ± 0.14 | 0.13 ± 0.11 | 0.28 ± 0.05 | 0.23 ± 0.05 | 0.3 ± 0.02 | 0.04 ± 0.04 | 0.18 ± 0.15 | 0.11 ± 0.02 | 0.03 ± 0.04 | 0.3 ± 0.03 | 0.06 ± 0 | 0.14 ± 0.06 |
| PFDS | 0.01 ± 0.02 | 0.04 ± 0.05 | 0.57 ± 0.01 | <MDL | <MDL | <MDL | <MDL | <MDL | <MDL | 0.16 ± 0.28 | 0.08 ± 0.14 | 0.03 ± 0.05 | 0.91 ± 0.35 | <MDL |
| PFHpA | 4.49 ± 0.06 | 2.98 ± 0.01 | 4.46 ± 0.04 | 3.79 ± 0.01 | 5.26 ± 0.02 | 3.35 ± 0 | 7.04 ± 0.07 | 2.37 ± 1.19 | 3.63 ± 0.37 | 3.64 ± 0.09 | 1.59 ± 0.05 | 3.82 ± 0.05 | 5.48 ± 0.06 | 11.83 ± 0.02 |
| PFHpS | <MDL | <MDL | <MDL | <MDL | 0.1 ± 0.17 | 0.15 ± 0.21 | 0.15 ± 0.25 | 0.13 ± 0.04 | 0.04 ± 0.06 | <MDL | 0.04 ± 0.07 | 0.07 ± 0.12 | 0.12 ± 0.17 | 0.13 ± 0.11 |
| PFHxA | 11.33 ± 0.02 | 7.31 ± 0.22 | 5.47 ± 0.02 | 6.15 ± 0.03 | 11.21 ± 0.06 | 6.25 ± 0.03 | 17.6 ± 0.05 | 4.02 ± 1.42 | 5.89 ± 0.46 | 5.2 ± 0.07 | 2.77 ± 0.26 | 6.85 ± 0.1 | 10.96 ± 0.06 | 15.87 ± 2.29 |
| PFHxS | 1.65 ± 0.03 | 0.98 ± 0.02 | 1.03 ± 0.06 | 0.92 ± 0 | 3.08 ± 0.09 | 0.99 ± 0 | 5.87 ± 0.14 | 0.76 ± 0.31 | 0.95 ± 0 | 0.87 ± 0 | 0.4 ± 0.06 | 1.22 ± 0 | 1.69 ± 0.06 | 4.42 ± 0.03 |
| PFNA | 1.19 ± 0.08 | 1.09 ± 0.07 | 1 ± 0.04 | 1.19 ± 0.02 | 1.36 ± 0.2 | 1.37 ± 0.43 | 1.81 ± 0.16 | 0.69 ± 0.24 | 0.98 ± 0.15 | 0.86 ± 0.03 | 0.49 ± 0.05 | 1.01 ± 0 | 1.1 ± 0.01 | 1.79 ± 0.24 |
| PFNS | 0.21 ± 0.31 | <MDL | 1.8 ± 0.53 | 1.08 ± 0.37 | <MDL | 2.08 ± 1.61 | 0.42 ± 0.49 | 0.03 ± 0.05 | 0.22 ± 0.27 | 5.44 ± 8.92 | 0.08 ± 0.1 | 0.03 ± 0.01 | 4.17 ± 5.83 | 0.13 ± 0.08 |
| PFOA | 10.67 ± 0.02 | 10.03 ± 0.22 | 7.94 ± 0.11 | 9.27 ± 0.11 | 12.35 ± 0.36 | 9.42 ± 0.14 | 14.78 ± 0.43 | 4.96 ± 1.68 | 12.71 ± 4.17 | 9.01 ± 0.25 | 5.11 ± 0.32 | 10.03 ± 0.61 | 15.65 ± 6.59 | 16.05 ± 4.98 |
| PFONS | 0.01 ± 0.01 | 0.04 ± 0.04 | <MDL | <MDL | 0.01 ± 0.02 | <MDL | 0.01 ± 0.02 | <MDL | <MDL | 0.01 ± 0.02 | 0.01 ± 0.01 | <MDL | <MDL | 0.03 ± 0.01 |
| PFOS | 18 ± 0.77 | 16.49 ± 4.5 | 14.64 ± 0.81 | 16.05 ± 0.59 | 21.01 ± 2.45 | 17.46 ± 2.54 | 34.3 ± 4.92 | 9.91 ± 4.41 | 16.26 ± 3.66 | 12.81 ± 0.72 | 6.17 ± 1 | 14.35 ± 0.63 | 14.79 ± 0.89 | 19.54 ± 2.75 |
| PFOUDS | 0.01 ± 0.02 | <MDL | 0.19 ± 0.17 | <MDL | 0.01 ± 0.02 | <MDL | <MDL | <MDL | <MDL | 0.07 ± 0.13 | <MDL | 0.23 ± 0.2 | <MDL | <MDL |
| PFPeA | 18.01 ± 0.51 | 6.55 ± 0.05 | 7.02 ± 0.12 | 8.02 ± 0.02 | 20.72 ± 0.5 | 7.7 ± 0.17 | 29.2 ± 0.48 | 5.19 ± 1.81 | 5.68 ± 0.03 | 6.84 ± 0.06 | 3.72 ± 0.11 | 7.47 ± 0.22 | 11.51 ± 1.67 | 27.02 ± 4.16 |
| PFPeS | 0.45 ± 0 | 0.31 ± 0.01 | 0.25 ± 0 | 0.37 ± 0.03 | 0.42 ± 0.01 | 0.31 ± 0 | 1.41 ± 0.1 | 0.2 ± 0.06 | 0.31 ± 0.17 | 0.28 ± 0.02 | 0.1 ± 0 | 0.45 ± 0.01 | 0.33 ± 0 | 0.67 ± 0.03 |
| PFTeDA | 0.11 ± 0.01 | 0.04 ± 0.07 | 0.08 ± 0.02 | 0.12 ± 0.02 | 0.13 ± 0.05 | 0.12 ± 0.17 | 0.07 ± 0 | 0.05 ± 0.04 | 0.1 ± 0.06 | 0.08 ± 0.05 | 0.01 ± 0.01 | 0.13 ± 0.08 | 0.05 ± 0.02 | 0.06 ± 0.02 |
| PFTrDA | 0.08 ± 0.05 | 0.2 ± 0.19 | 0.03 ± 0.04 | 0.1 ± 0.07 | 0.1 ± 0.01 | 0.61 ± 0.16 | 0.13 ± 0.03 | 0.12 ± 0.11 | 0.05 ± 0.03 | 0.14 ± 0.07 | 0.03 ± 0.02 | 0.27 ± 0.04 | 0.04 ± 0.01 | 0.11 ± 0.04 |
| PFUdA | 0.31 ± 0.53 | 3.34 ± 0.85 | 0.6 ± 1.03 | 2.09 ± 0.05 | 0.12 ± 0.11 | 2.13 ± 0.09 | 1.55 ± 0.99 | <MDL | 0.34 ± 0.38 | 0.81 ± 0.02 | 0.15 ± 0.03 | <MDL | 0.43 ± 0.36 | 0.26 ± 0.22 |
| ∑30PFAS | 194.41 ± 1.99 | 256.42 ± 29.19 | 108.07 ± 9.77 | 166.81 ± 15.74 | 163.81 ± 66.3 | 113.66 ± 7.03 | 216. 52 ± 5.85 | 76.24 ± 23.61 | 103.76 ± 8.48 | 102.66 ± 8.74 | 46.6 ± 2.8 | 95.08 ± 1.56 | 110.96 ± 1.91 | 211.79 ± 31.34 |
| DRY | WET | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| P1 | P2 | P3 | P4 | P5 | P6 | P7 | P1 | P2 | P3 | P4 | P5 | P6 | P7 | |
| BDE-28 | <MDL | <MDL | <MDL | <MDL | <MDL | <MDL | <MDL | 0.02 ± 0 | 0.02 ± 0 | 0.01 ± 0.01 | 0.02 ± 0 | 0.07 ± 0.08 | 0.03 ± 0.01 | 0.02 ± 0.01 |
| BDE-47 | 0.02 ± 0 | 0.02 ± 0 | 0.36 ± 0.12 | 0.01 ± 0 | 0.03 ± 0 | 0.12 ± 0 | 0.06 ± 0.01 | 0.19 ± 0.02 | 0.14 ± 0 | 0.24 ± 0.03 | 0.16 ± 0.01 | 0.52 ± 0.01 | 0.52 ± 0.01 | 0.51 ± 0.01 |
| BDE-99 | 0.02 ± 0 | 0.02 ± 0 | 0.54 ± 0.2 | 0.01 ± 0 | <MDL | <MDL | 0.01 ± 0 | 0.01 ± 0 | 0.21 ± 0.02 | 0.01 ± 0 | 0.01 ± 0 | <MDL | <MDL | <MDL |
| BDE-100 | 0.01 ± 0 | 0 ± 0 | 0.04 ± 0 | 0.01 ± 0 | 0.01 ± 0 | 0.07 ± 0 | 0.02 ± 0 | 0.03 ± 0.01 | 0.12 ± 0.02 | 0.02 ± 0.01 | 0.06 ± 0.02 | 0.05 ± 0.01 | 0.16 ± 0.03 | 0.03 ± 0.01 |
| BDE-153 | <MDL | <MDL | <MDL | 0.01 ± 0 | <MDL | <MDL | <MDL | 0.01 ± 0 | <MDL | 0.01 ± 0 | <MDL | <MDL | 0.01 ± 0 | 0.01 ± 0 |
| BDE-154 | <MDL | <MDL | 0.01 ± 0 | <MDL | 0.01 ± 0 | 0.01 ± 0 | <MDL | <MDL | <MDL | <MDL | 0.01 ± 0 | <MDL | 0.01 ± 0 | 0.01 ± 0 |
| BDE-183 | 0.6 ± 0.08 | 0.81 ± 0.06 | 0.8 ± 0.18 | 0.02 ± 0 | 0.03 ± 0 | 0.03 ± 0 | 0.03 ± 0 | 0.18 ± 0.02 | 0.07 ± 0.01 | 0.25 ± 0.02 | <MDL | <MDL | 0.02 ± 0 | <MDL |
| BDE-209 | 0.34 ± 0.04 | 0.34 ± 0.05 | 0.8 ± 0.04 | 0.69 ± 0.07 | 0.45 ± 0.16 | 0.68 ± 0.2 | 1.89 ± 0.03 | 2.48 ± 0.28 | 1.93 ± 0.03 | 2.43 ± 0.37 | 6.9 ± 1.12 | 6.35 ± 1.62 | 4.09 ± 0.39 | 2.86 ± 0.7 |
| ∑8BDEs | 1 ± 0.06 | 1.21 ± 0.11 | 2.56 ± 0.31 | 0.74 ± 0.07 | 0.55 ± 0.16 | 0.92 ± 0.21 | 2.02 ± 0.02 | 2.92 ± 0.27 | 2.51 ± 0.02 | 2.97 ± 0.39 | 7.15 ± 1.12 | 6.99 ± 1.55 | 4.84 ± 0.4 | 3.44 ± 0.7 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Souza-Araujo, J.d.; Santos, I.C.d.; Melo, H.D.B.; Lemos, L.S.; Quinete, N.; Rosa, A.H. Assessment of Per- and Poly-Fluoroalkyl Substances (PFAS) and Polybrominated Diphenyl Ethers (PBDEs) in Surface Waters Used for Urban Water Supply in Brazil. Toxics 2026, 14, 148. https://doi.org/10.3390/toxics14020148
Souza-Araujo Jd, Santos ICd, Melo HDB, Lemos LS, Quinete N, Rosa AH. Assessment of Per- and Poly-Fluoroalkyl Substances (PFAS) and Polybrominated Diphenyl Ethers (PBDEs) in Surface Waters Used for Urban Water Supply in Brazil. Toxics. 2026; 14(2):148. https://doi.org/10.3390/toxics14020148
Chicago/Turabian StyleSouza-Araujo, Juliana de, Isadhora Camargo dos Santos, Hansel David Burgos Melo, Leila Soledade Lemos, Natalia Quinete, and André Henrique Rosa. 2026. "Assessment of Per- and Poly-Fluoroalkyl Substances (PFAS) and Polybrominated Diphenyl Ethers (PBDEs) in Surface Waters Used for Urban Water Supply in Brazil" Toxics 14, no. 2: 148. https://doi.org/10.3390/toxics14020148
APA StyleSouza-Araujo, J. d., Santos, I. C. d., Melo, H. D. B., Lemos, L. S., Quinete, N., & Rosa, A. H. (2026). Assessment of Per- and Poly-Fluoroalkyl Substances (PFAS) and Polybrominated Diphenyl Ethers (PBDEs) in Surface Waters Used for Urban Water Supply in Brazil. Toxics, 14(2), 148. https://doi.org/10.3390/toxics14020148