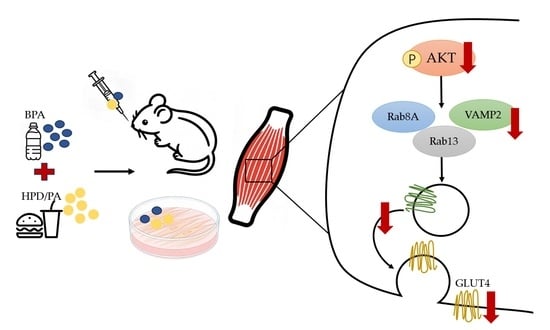
Co-Exposure to Bisphenol A and a High-Fat Diet Induces Insulin Resistance via Suppression of Insulin Signaling Molecule Expression and GLUT4 Translocation
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics
2.2. Animal Experimental Design
2.3. Fasting Blood Glucose Measurement
2.4. Fasting Plasma Insulin Measurement
2.5. Mouse HOMA-IR Measurement
2.6. Glucose Tolerance Test
2.7. Immunohistochemistry for Protein Expression
2.8. Cell Experiment Design
2.9. Cell Survival Assay
2.10. 2-NBDG Glucose Uptake Assays
2.11. Cellular Immunofluorescence Staining for GLUT4
2.12. Western Blotting for AKT-GLUT4 Signaling Molecules
2.13. Cell Membrane Isolation
2.14. Statistical Analysis
3. Results
3.1. BPA in Combination with an HFD Exacerbates IR
3.2. BPA and HFD Co-Exposure Affects Insulin Signaling Molecule Expression in Gastrocnemius Tissue
3.3. BPA in Combination with Palmitic Acid (PA) Significantly Decreases Glucose Uptake Levels in C2C12 Cells
3.4. Co-Exposure to BPA and PA Inhibits Insulin Signaling and GLUT4 Translocation in C2C12 Cells
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Huang, Y.; Karuranga, S.; Malanda, B.; Williams, D.R.R. Call for data contribution to the IDF Diabetes Atlas 9th Edition 2019. Diabetes Res. Clin. Pract. 2018, 140, 351–352. [Google Scholar] [CrossRef]
- Al Mansour, M.A. The Prevalence and Risk Factors of Type 2 Diabetes Mellitus (DMT2) in a Semi-Urban Saudi Population. Int. J. Environ. Res. Public Health 2019, 17, 7. [Google Scholar] [CrossRef]
- Lu, X.; Xie, Q.; Pan, X.; Zhang, R.; Zhang, X.; Peng, G.; Zhang, Y.; Shen, S.; Tong, N. Type 2 diabetes mellitus in adults: Pathogenesis, prevention and therapy. Signal Transduct. Target. Ther. 2024, 9, 262. [Google Scholar] [CrossRef]
- Woods, S.C.; Seeley, R.J.; Rushing, P.A.; D’Alessio, D.; Tso, P. A controlled high-fat diet induces an obese syndrome in rats. J. Nutr. 2003, 133, 1081–1087. [Google Scholar] [CrossRef] [PubMed]
- Kahn, S.E.; Hull, R.L.; Utzschneider, K.M. Mechanisms linking obesity to insulin resistance and type 2 diabetes. Nature 2006, 444, 840–846. [Google Scholar] [CrossRef] [PubMed]
- Small, L.; Brandon, A.E.; Turner, N.; Cooney, G.J. Modeling insulin resistance in rodents by alterations in diet: What have high-fat and high-calorie diets revealed? Am. J. Physiol. Endocrinol. Metab. 2018, 314, E251–E265. [Google Scholar] [CrossRef] [PubMed]
- Hinault, C.; Caroli-Bosc, P.; Bost, F.; Chevalier, N. Critical Overview on Endocrine Disruptors in Diabetes Mellitus. Int. J. Mol. Sci. 2023, 24, 4537. [Google Scholar] [CrossRef]
- Weiss, M.C.; Shih, Y.H.; Bryan, M.S.; Jackson, B.P.; Aguilar, D.; Hanis, C.L.; Argos, M.; Sargis, R.M. Relationships Between Urinary Metals and Diabetes Traits Among Mexican Americans in Starr County, Texas, USA. Biol. Trace Elem. Res. 2023, 201, 529–538. [Google Scholar] [CrossRef]
- Vandenberg, L.N.; Hauser, R.; Marcus, M.; Olea, N.; Welshons, W.V. Human exposure to bisphenol A (BPA). Reprod. Toxicol. 2007, 24, 139–177. [Google Scholar] [CrossRef]
- Geens, T.; Goeyens, L.; Covaci, A. Are potential sources for human exposure to bisphenol-A overlooked? Int. J. Hyg. Environ. Health 2011, 214, 339–347. [Google Scholar] [CrossRef]
- Vandenberg, L.N.; Chahoud, I.; Heindel, J.J.; Padmanabhan, V.; Paumgartten, F.J.; Schoenfelder, G. Urinary, circulating, and tissue biomonitoring studies indicate widespread exposure to bisphenol A. Cienc. Saude Coletiva 2012, 17, 407–434. [Google Scholar] [CrossRef] [PubMed]
- Russo, G.; Barbato, F.; Mita, D.G.; Grumetto, L. Occurrence of Bisphenol A and its analogues in some foodstuff marketed in Europe. Food Chem. Toxicol. 2019, 131, 110575. [Google Scholar] [CrossRef] [PubMed]
- Cimmino, I.; Fiory, F.; Perruolo, G.; Miele, C.; Beguinot, F.; Formisano, P.; Oriente, F. Potential Mechanisms of Bisphenol A (BPA) Contributing to Human Disease. Int. J. Mol. Sci. 2020, 21, 5761. [Google Scholar] [CrossRef] [PubMed]
- Hafezi, S.A.; Abdel-Rahman, W.M. The Endocrine Disruptor Bisphenol A (BPA) Exerts a Wide Range of Effects in Carcinogenesis and Response to Therapy. Curr. Mol. Pharmacol. 2019, 12, 230–238. [Google Scholar] [CrossRef]
- Freeman, A.M.; Acevedo, L.A.; Pennings, N. Insulin Resistance. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2023. [Google Scholar]
- Lee, K. Usefulness of the metabolic syndrome criteria as predictors of insulin resistance among obese Korean women. Public Health Nutr. 2010, 13, 181–186. [Google Scholar] [CrossRef]
- Ferrannini, E.; Simonson, D.C.; Katz, L.D.; Reichard, G., Jr.; Bevilacqua, S.; Barrett, E.J.; Olsson, M.; DeFronzo, R.A. The disposal of an oral glucose load in patients with non-insulin-dependent diabetes. Metab. Clin. Exp. 1988, 37, 79–85. [Google Scholar] [CrossRef]
- Foroutani, L.; Ashraf Ganjouei, A.; Wang, J.; Aburayya, B.I.; Corvera, C.; Alseidi, A.; Adam, M.A. Robotic-Assisted Endoluminal Resection of Gastroesophageal Junction Leiomyoma with Transoral Specimen Extraction: Technique, Outcome, and Safety. Ann. Surg Oncol. 2025, 32, 1218–1219. [Google Scholar] [CrossRef]
- DeFronzo, R.A.; Tripathy, D. Skeletal muscle insulin resistance is the primary defect in type 2 diabetes. Diabetes Care 2009, 32, S157–S163. [Google Scholar] [CrossRef]
- Zhu, Y.; Pereira, R.O.; O’Neill, B.T.; Riehle, C.; Ilkun, O.; Wende, A.R.; Rawlings, T.A.; Zhang, Y.C.; Zhang, Q.; Klip, A.; et al. Cardiac PI3K-Akt impairs insulin-stimulated glucose uptake independent of mTORC1 and GLUT4 translocation. Mol. Endocrinol. 2013, 27, 172–184. [Google Scholar] [CrossRef]
- Sharma, B.R.; Kim, H.J.; Rhyu, D.Y. Caulerpa lentillifera extract ameliorates insulin resistance and regulates glucose metabolism in C57BL/KsJ-db/db mice via PI3K/AKT signaling pathway in myocytes. J. Transl. Med. 2015, 13, 62. [Google Scholar] [CrossRef]
- Li, W.; Liang, X.; Zeng, Z.; Yu, K.; Zhan, S.; Su, Q.; Yan, Y.; Mansai, H.; Qiao, W.; Yang, Q.; et al. Simvastatin inhibits glucose uptake activity and GLUT4 translocation through suppression of the IR/IRS-1/Akt signaling in C2C12 myotubes. Biomed. Pharmacother. 2016, 83, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Bilan, P.J.; Liu, Z.; Klip, A. Rab8A and Rab13 are activated by insulin and regulate GLUT4 translocation in muscle cells. Proc. Natl. Acad. Sci. USA 2010, 107, 19909–19914. [Google Scholar] [CrossRef] [PubMed]
- Foster, L.J.; Klip, A. Mechanism and regulation of GLUT-4 vesicle fusion in muscle and fat cells. Am. J. Physiol. Cell Physiol. 2000, 279, C877–C890. [Google Scholar] [CrossRef] [PubMed]
- Mullainadhan, V.; Viswanathan, M.P.; Karundevi, B. Effect of Bisphenol-A (BPA) on insulin signal transduction and GLUT4 translocation in gastrocnemius muscle of adult male albino rat. Int. J. Biochem. Cell Biol. 2017, 90, 38–47. [Google Scholar] [CrossRef]
- Pessin, J.E.; Thurmond, D.C.; Elmendorf, J.S.; Coker, K.J.; Okada, S. Molecular basis of insulin-stimulated GLUT4 vesicle trafficking: Location! Location! Location! J. Biol. Chem. 1999, 274, 2593–2596. [Google Scholar] [CrossRef]
- Duman, J.G.; Forte, J.G. What is the role of SNARE proteins in membrane fusion? Am. J. Physiol. Cell Physiol. 2003, 285, C237–C249. [Google Scholar] [CrossRef]
- Chamberlain, L.H.; Gould, G.W. The vesicle- and target-SNARE proteins that mediate Glut4 vesicle fusion are localized in detergent-insoluble lipid rafts present on distinct intracellular membranes. J. Biol. Chem. 2002, 277, 49750–49754. [Google Scholar] [CrossRef]
- Garvey, W.T.; Maianu, L.; Zhu, J.H.; Brechtel-Hook, G.; Wallace, P.; Baron, A.D. Evidence for defects in the trafficking and translocation of GLUT4 glucose transporters in skeletal muscle as a cause of human insulin resistance. J. Clin. Investig. 1998, 101, 2377–2386. [Google Scholar] [CrossRef]
- Hu, F.B. Globalization of diabetes: The role of diet, lifestyle, and genes. Diabetes Care 2011, 34, 1249–1257. [Google Scholar] [CrossRef]
- Carnagarin, R.; Dharmarajan, A.M.; Dass, C.R. Molecular aspects of glucose homeostasis in skeletal muscle--A focus on the molecular mechanisms of insulin resistance. Mol. Cell. Endocrinol. 2015, 417, 52–62. [Google Scholar] [CrossRef]
- Tahapary, D.L.; Pratisthita, L.B.; Fitri, N.A.; Marcella, C.; Wafa, S.; Kurniawan, F.; Rizka, A.; Tarigan, T.J.E.; Harbuwono, D.S.; Purnamasari, D.; et al. Challenges in the diagnosis of insulin resistance: Focusing on the role of HOMA-IR and Tryglyceride/glucose index. Diabetes Metab. Syndr. 2022, 16, 102581. [Google Scholar] [CrossRef] [PubMed]
- Miyata, S.; Yada, T.; Ishikawa, N.; Taheruzzaman, K.; Hara, R.; Matsuzaki, T.; Nishikawa, A. Insulin-like growth factor 1 regulation of proliferation and differentiation of Xenopus laevis myogenic cells in vitro. Vitr. Cell. Dev. Biol. Anim. 2017, 53, 231–247. [Google Scholar] [CrossRef] [PubMed]
- Welshons, W.V.; Nagel, S.C.; vom Saal, F.S. Large effects from small exposures. III. Endocrine mechanisms mediating effects of bisphenol A at levels of human exposure. Endocrinology 2006, 147, S56–S69. [Google Scholar] [CrossRef] [PubMed]
- Reagan-Shaw, S.; Nihal, M.; Ahmad, N. Dose translation from animal to human studies revisited. FASEB J. 2008, 22, 659–661. [Google Scholar] [CrossRef]
- Chen, D.; Kannan, K.; Tan, H.; Zheng, Z.; Feng, Y.L.; Wu, Y.; Widelka, M. Bisphenol Analogues Other Than BPA: Environmental Occurrence, Human Exposure, and Toxicity-A Review. Environ. Sci. Technol. 2016, 50, 5438–5453. [Google Scholar] [CrossRef]
- Luo, W.; Ai, L.; Wang, B.F.; Zhou, Y. High glucose inhibits myogenesis and induces insulin resistance by down-regulating AKT signaling. Biomed. Pharmacother. 2019, 120, 109498. [Google Scholar] [CrossRef]
- Fryer, L.G.; Foufelle, F.; Barnes, K.; Baldwin, S.A.; Woods, A.; Carling, D. Characterization of the role of the AMP-activated protein kinase in the stimulation of glucose transport in skeletal muscle cells. Biochem. J. 2002, 363, 167–174. [Google Scholar] [CrossRef]
- Mora, S.; Pessin, J.E. An adipocentric view of signaling and intracellular trafficking. Diabetes/Metab. Res. Rev. 2002, 18, 345–356. [Google Scholar] [CrossRef]
- Ding, S.; Fan, Y.; Zhao, N.; Yang, H.; Ye, X.; He, D.; Jin, X.; Liu, J.; Tian, C.; Li, H.; et al. High-fat diet aggravates glucose homeostasis disorder caused by chronic exposure to bisphenol A. J. Endocrinol. 2014, 221, 167–179. [Google Scholar] [CrossRef]
- Ropero, A.B.; Soria, B.; Nadal, A. A nonclassical estrogen membrane receptor triggers rapid differential actions in the endocrine pancreas. Mol. Endocrinol. 2002, 16, 497–505. [Google Scholar] [CrossRef]
- Bryant, N.J.; Gould, G.W. SNARE proteins underpin insulin-regulated GLUT4 traffic. Traffic 2011, 12, 657–664. [Google Scholar] [CrossRef]
- Jaldin-Fincati, J.R.; Pavarotti, M.; Frendo-Cumbo, S.; Bilan, P.J.; Klip, A. Update on GLUT4 Vesicle Traffic: A Cornerstone of Insulin Action. Trends Endocrinol. Metab. 2017, 28, 597–611. [Google Scholar] [CrossRef]
- Kahn, B.B.; Flier, J.S. Regulation of glucose-transporter gene expression in vitro and in vivo. Diabetes Care 1990, 13, 548–564. [Google Scholar] [CrossRef]
- Kawaguchi, T.; Tamori, Y.; Kanda, H.; Yoshikawa, M.; Tateya, S.; Nishino, N.; Kasuga, M. The t-SNAREs syntaxin4 and SNAP23 but not v-SNARE VAMP2 are indispensable to tether GLUT4 vesicles at the plasma membrane in adipocyte. Biochem. Biophys. Res. Commun. 2010, 391, 1336–1341. [Google Scholar] [CrossRef] [PubMed]
- Saltiel, A.R.; Kahn, C.R. Insulin signalling and the regulation of glucose and lipid metabolism. Nature 2001, 414, 799–806. [Google Scholar] [CrossRef] [PubMed]
- Cohen, P.; Frame, S. The renaissance of GSK3. Nat. Rev. Mol. Cell Biol. 2001, 2, 769–776. [Google Scholar] [CrossRef] [PubMed]
- Gulli, G.; Ferrannini, E.; Stern, M.; Haffner, S.; DeFronzo, R.A. The metabolic profile of NIDDM is fully established in glucose-tolerant offspring of two Mexican-American NIDDM parents. Diabetes 1992, 41, 1575–1586. [Google Scholar] [CrossRef]
- Cohen, P. The Croonian Lecture 1998. Identification of a protein kinase cascade of major importance in insulin signal transduction. Philos. Trans. R. Soc. London. Ser. B Biol. Sci. 1999, 354, 485–495. [Google Scholar] [CrossRef]
- Cohen, P.; Goedert, M. GSK3 inhibitors: Development and therapeutic potential. Nat. Rev. Drug Discov. 2004, 3, 479–487. [Google Scholar] [CrossRef]
- Nikoulina, S.E.; Ciaraldi, T.P.; Mudaliar, S.; Mohideen, P.; Carter, L.; Henry, R.R. Potential role of glycogen synthase kinase-3 in skeletal muscle insulin resistance of type 2 diabetes. Diabetes 2000, 49, 263–271. [Google Scholar] [CrossRef]
- Cross, D.A.; Alessi, D.R.; Cohen, P.; Andjelkovich, M.; Hemmings, B.A. Inhibition of glycogen synthase kinase-3 by insulin mediated by protein kinase B. Nature 1995, 378, 785–789. [Google Scholar] [CrossRef]
- Henriksen, E.J.; Dokken, B.B. Role of glycogen synthase kinase-3 in insulin resistance and type 2 diabetes. Curr. Drug Targets 2006, 7, 1435–1441. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Lu, Z.; Cao, M.; Zhang, J.; Qi, C.; Huang, B.; Li, W.; Li, J.; Yang, G.; Zhang, Y.; Wu, J.; et al. Co-Exposure to Bisphenol A and a High-Fat Diet Induces Insulin Resistance via Suppression of Insulin Signaling Molecule Expression and GLUT4 Translocation. Toxics 2026, 14, 146. https://doi.org/10.3390/toxics14020146
Lu Z, Cao M, Zhang J, Qi C, Huang B, Li W, Li J, Yang G, Zhang Y, Wu J, et al. Co-Exposure to Bisphenol A and a High-Fat Diet Induces Insulin Resistance via Suppression of Insulin Signaling Molecule Expression and GLUT4 Translocation. Toxics. 2026; 14(2):146. https://doi.org/10.3390/toxics14020146
Chicago/Turabian StyleLu, Zeqi, Min Cao, Jiaoxiang Zhang, Congzheng Qi, Bing Huang, Wenxue Li, Juntao Li, Guangyu Yang, Yan Zhang, Jinyin Wu, and et al. 2026. "Co-Exposure to Bisphenol A and a High-Fat Diet Induces Insulin Resistance via Suppression of Insulin Signaling Molecule Expression and GLUT4 Translocation" Toxics 14, no. 2: 146. https://doi.org/10.3390/toxics14020146
APA StyleLu, Z., Cao, M., Zhang, J., Qi, C., Huang, B., Li, W., Li, J., Yang, G., Zhang, Y., Wu, J., Liu, W., & Zhu, W. (2026). Co-Exposure to Bisphenol A and a High-Fat Diet Induces Insulin Resistance via Suppression of Insulin Signaling Molecule Expression and GLUT4 Translocation. Toxics, 14(2), 146. https://doi.org/10.3390/toxics14020146