Toxicological, Chemical, Social, and Economic Challenges Associated with PFAS and Replacement Aqueous Film-Forming Foams (AFFF)
Abstract
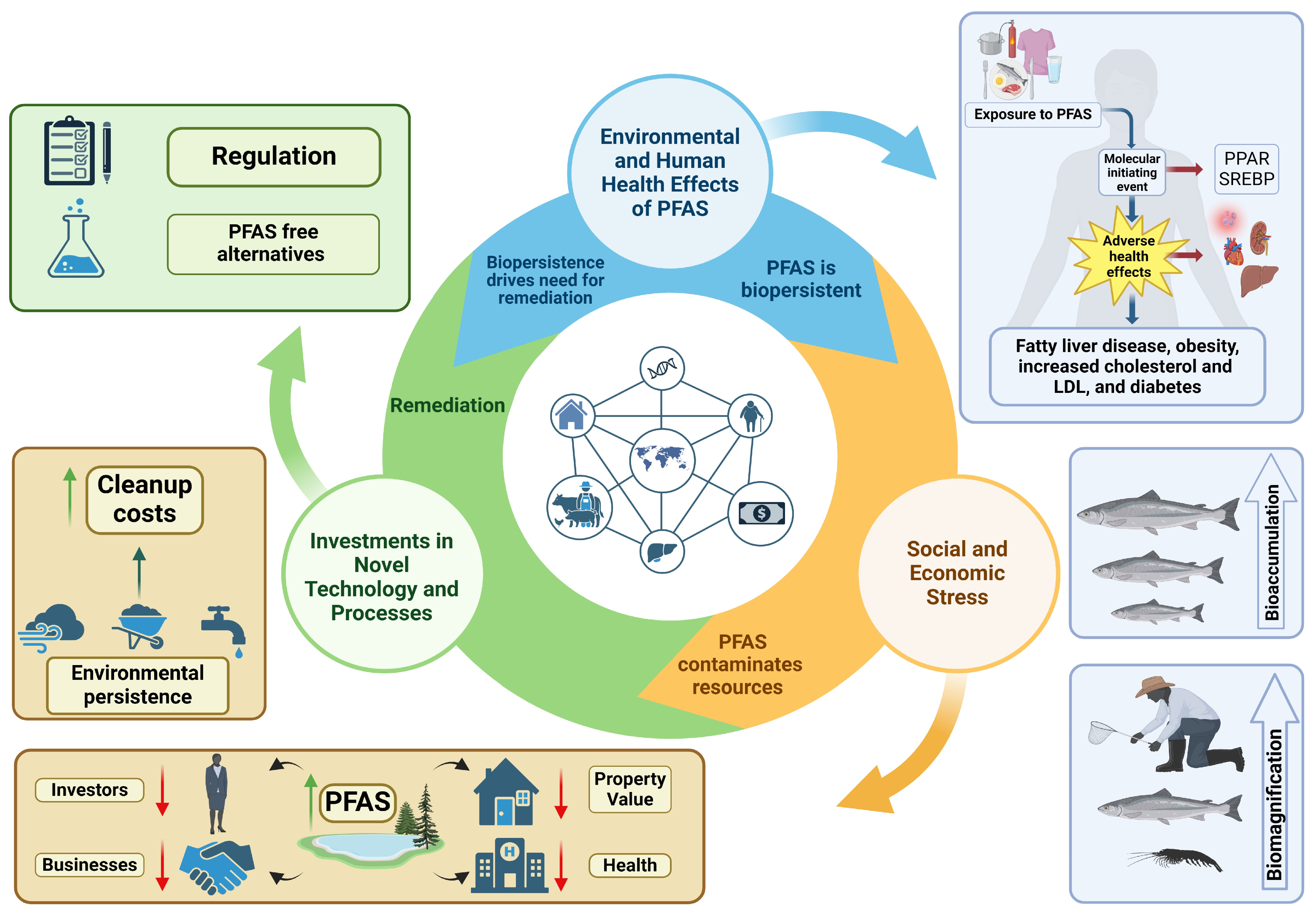
1. Background
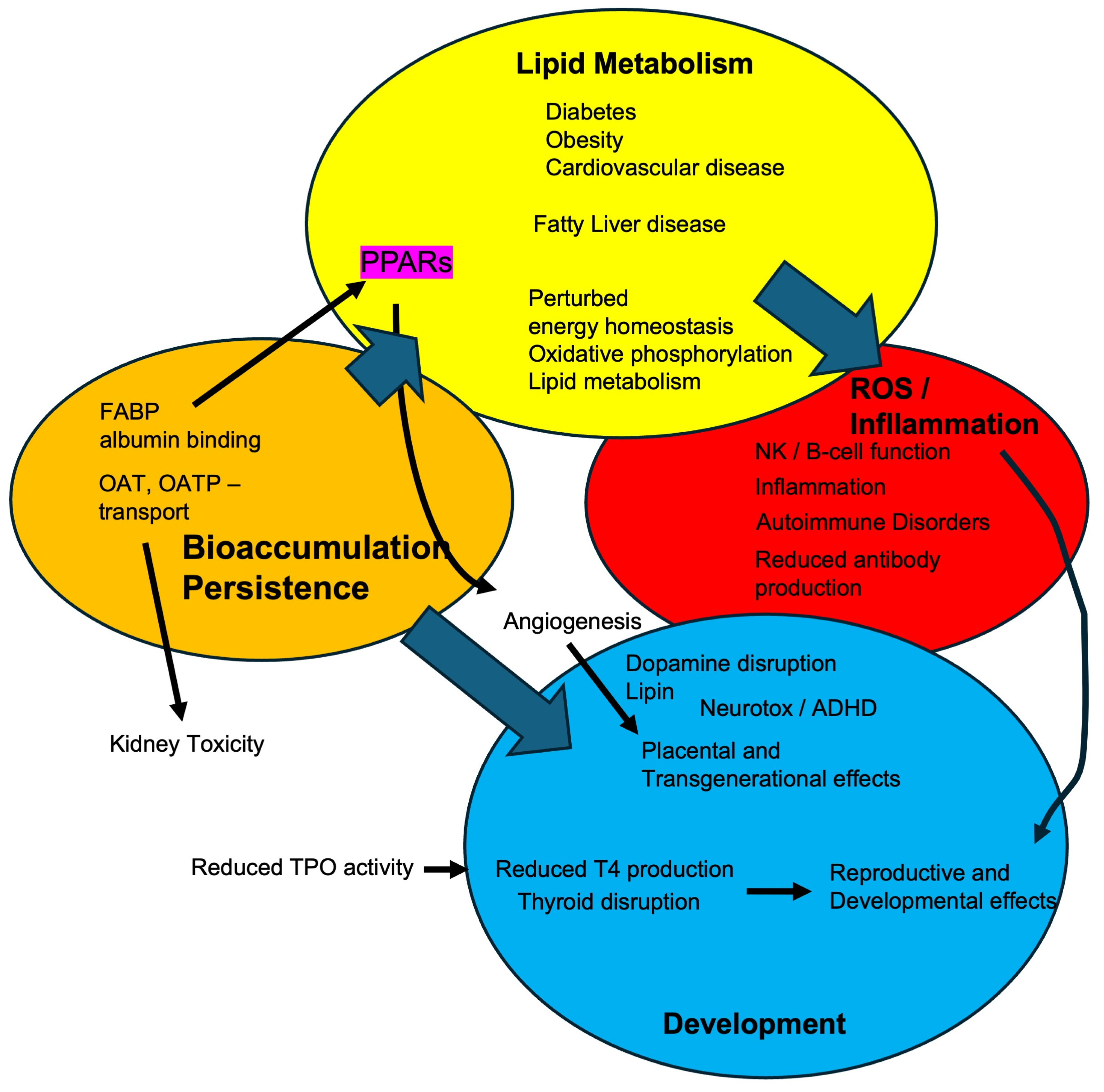
2. PFAS Exposure and Bioaccumulation
3. Bioanalytical Challenges for Identification and Quantitation of PFAS
4. PFAS and the One Health Concept
5. Human Health Effects and Systemic Toxicity of PFASs
6. Regulation of PFAS
7. Impacts of PFAS on Economic Development, Business, and Property Value
8. PFAS and Healthcare Costs
9. Clean-Up and Compliance Costs Due to PFAS
10. Economic Impacts Lead to PFAS-Free Alternatives—AFFF as an Example
11. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Naval History and Heritage Command. This Day in Naval History, 29 July 1967. 2025. Available online: https://www.history.navy.mil/today-in-history/july-29.html (accessed on 18 June 2025).
- Cox, S.J. H-008-6: Uss Forrestal Disaster, 29 July 1967; Naval History and Heritage Command: Washington, DC, USA, 2019. [Google Scholar]
- GZA GeoEnvironmental, Inc. Per- and Polyfluoroalkyl Substances–Part i–A Focus on the History of Aqueous Film-Forming Foams. Available online: https://www.gza.com/insights/per-and-polyfluoroalkyl-substances-part-i-focus-history-aqueous-film-forming-foams#:~:text=The%20Forrestal%20disaster%20drove%20the,vessels%20by%20the%20late%201960s (accessed on 18 June 2025).
- Buck, R.C.; Franklin, J.; Berger, U.; Conder, J.M.; Cousins, I.T.; de Voogt, P.; Jensen, A.A.; Kannan, K.; Mabury, S.A.; van Leeuwen, S.P.J. Perfluoroalkyl and Polyfluoroalkyl Substances in the Environment: Terminology, Classification, and Origins. Integr. Env. Assess. Manag. 2011, 7, 513–541. [Google Scholar] [CrossRef]
- Prevedouros, K.; Cousins, I.T.; Buck, R.C.; Korzeniowski, S.H. Sources, Fate and Transport of Perfluorocarboxylates. Environ. Sci. Technol. 2006, 40, 32–44. [Google Scholar] [CrossRef]
- Baldwin, W.S.; Davis, T.T.; Eccles, J.A. Per- and Polyfluoroalkylsubstances (PFAS) and Their Toxicology as Evidenced Through Disease and Biomarkers. In Biomarkers in Toxicology. Biomarkers in Disease: Methods, Discoveries and Applications; Patel, V.B., Preedy, V.R., Rajendram, R., Eds.; Springer: Berlin/Heidelberg, Germany, 2023; pp. 989–1016. [Google Scholar]
- Burkhard, L.P. Evaluation of published bioconcentration factor (BCF) and bioaccumulation factor (BAF) data for per-and polyfluoroalkyl substances across aquatic species. Environ. Toxicol. Chem. 2021, 40, 1530–1543. [Google Scholar] [CrossRef]
- Schymanski, E.L.; Zhang, J.; Thiessen, P.A.; Chirsir, P.; Kondic, T.; Bolton, E.E. Per- and Polyfluoroalkyl Substances (PFAS) in PubChem: 7 Million and Growing. Environ. Sci. Technol. 2023, 57, 16918–16928. [Google Scholar] [CrossRef]
- Bartell, S.M.; Calafat, A.M.; Lyu, C.; Kato, K.; Ryan, P.B.; Steenland, K. Rate of Decline in Serum PFOA Concentrations after Granular Activated Carbon Filtration at Two Public Water Systems in Ohio and West Virginia. Environ. Health Perspect. 2010, 118, 222–228. [Google Scholar] [CrossRef]
- Olsen, G.W.; Burris, J.M.; Ehresman, D.J.; Froelich, J.W.; Seacat, A.M.; Butenhoff, J.L.; Zobel, L.R. Half-Life of Serum Elimination of Perfluorooctanesulfonate, Perfluorohexanesulfonate, and Perfluorooctanoate in Retired Fluorochemical Production Workers. Environ. Health Perspect. 2007, 115, 1298–1305. [Google Scholar] [CrossRef] [PubMed]
- United National Environmental Programme (UNEP). Stockholm Convention on Persistent Organic Pollutants (POPs); UNEP/POPS/COP.10/33. United National Environmental Programme (UNEP): Chatelaine, Switzerland, 2022. [Google Scholar]
- Levin, R.; Villanueva, C.M.; Beene, D.; Cradock, A.L.; Donat-Vargas, C.; Lewis, J.; Martinez-Morata, I.; Minovi, D.; Nigra, A.E.; Olson, E.D.; et al. US drinking water quality: Exposure risk profiles for seven legacy and emerging contaminants. J. Exp. Sci. Environ. Epidemiol. 2024, 34, 3–22. [Google Scholar] [CrossRef]
- Forster, A.L.B.; Geiger, T.C.; Pansari, G.O.; Justen, P.T.; Richardson, S.D. Identifying PFAS hotspots in surface waters of South Carolina using a new optimized total organic fluorine method and target LC-MS/MS. Water Res. 2024, 256, 121570. [Google Scholar] [CrossRef] [PubMed]
- Evich, M.G.; Davis, M.J.B.; McCord, J.P.; Acrey, B.; Awkerman, J.A.; Knappe, D.R.U.; Lindstrom, A.B.; Speth, T.F.; Tebes-Stevens, C.; Strynar, M.J.; et al. Per- and polyfluoroalkyl substances in the environment. Science 2022, 375, eabg9065. [Google Scholar] [CrossRef]
- Cordner, A.; Goldenman, G.; Birnbaum, L.S.; Brown, P.; Miller, M.F.; Mueller, R.; Patton, S.; Salvatore, D.H.; Trasande, L. The true cost of PFAS and the benefits of acting now. Environ. Sci. Technol. 2021, 55, 9630–9633. [Google Scholar] [CrossRef] [PubMed]
- Goldenman, G.; Fernandes, M.; Holland, M.; Tugran, T.; Nordin, A.; Schoumacher, C.; McNeill, A. The Cost of Inaction: A Socioeconomic Analysis of Environmental and Health Impacts Linked to Exposure to PFAS; Nordic Council of Ministers: Copenhagen, Denmark, 2019. [Google Scholar]
- Bell, R.; Tachovsky, M. Real estate damage economics: The impact of PFAS “Forever chemicals” on real estate valuation. Environ. Claims J. 2022, 34, 136–155. [Google Scholar] [CrossRef]
- Fellows, N.; Wetzstein, D.; Lee, S.; Stockinger, J. PFCS and Class B Firefighting Foam; Minnesota Pollution Control Agency: St. Paul, MN, USA, 2009. [Google Scholar]
- Conder, J.; Zodrow, J.; Arblaster, J.; Kelly, B.; Gobas, F.; Suski, J.; Osborn, E.; Frenchmeyer, M.; Divine, C.; Lesson, A. Strategic resources for assessing PFAS ecological risks at AFFF sites. Integr. Environ. Assess. Manag. 2021, 17, 746–752. [Google Scholar] [CrossRef]
- Dinges, E.; Jones, J. FDA, Industry Actions End Sales of PFAS Used in Us Food Packaging. 2024. Available online: https://www.fda.gov/news-events/press-announcements/fda-industry-actions-end-sales-pfas-used-us-food-packaging (accessed on 6 September 2024).
- U.S. Environmental Protection Agency. PFAS National Primary Drinking Water Regulation; Volume EPA-HQ-OW-2022-0114, FRL 8543-02-OW; U.S. Environmental Protection Agency: Washington, DC, USA, 2024; pp. 32532–32757. [Google Scholar]
- Zarghamee, R.; Halter, A.G.; Marullo, J. EPA to Reconsider Drinking Water Standards for Several PFAS. 2025. Available online: https://pfas.pillsburylaw.com/epa-reconsider-drinking-water-standards-pfas/ (accessed on 21 August 2025).
- Hu, X.C.; Andrews, D.Q.; Lindstrom, A.B.; Bruton, T.A.; Schaider, L.A.; Grandjean, P.; Lohmann, R.; Carignan, C.C.; Blum, A.; Balan, S.A.; et al. Detection of Poly- and Perfluoroalkyl Substances (PFASs) in U.S. Drinking Water Linked to Industrial Sites, Military Fire Training Areas, and Wastewater Treatment Plants. Environ. Sci. Technol. Lett. 2016, 3, 344–350. [Google Scholar] [CrossRef]
- Kotlarz, N.; McCord, J.; Collier, D.; Lea, C.S.; Strynar, M.; Lindstrom, A.B.; Wilkie, A.A.; Islam, J.Y.; Matney, K.; Tarte, P.; et al. Measurement of Novel, Drinking Water-Associated PFAS in Blood from Adults and Children in Wilmington, North Carolina. Environ. Health Perspect. 2020, 128, 77005. [Google Scholar] [CrossRef]
- Smalling, K.L.; Romanok, K.M.; Bradley, P.M.; Morriss, M.C.; Gray, J.L.; Kanagy, L.K.; Gordon, S.E.; Williams, B.M.; Breitmeyer, S.E.; Jones, D.K.; et al. Per- and polyfluoroalkyl substances (PFAS) in United States tapwater: Comparison of underserved private-well and public-supply exposures and associated health implications. Environ. Int. 2023, 178, 108033. [Google Scholar] [CrossRef]
- Center for Disease Control. CDC National Report on Human Exposure to Environmental Chemicals—Biomonitoring Data Tables for Environmental Chemicals. 2024. Available online: https://www.cdc.gov/exposurereport/index.html (accessed on 15 August 2024).
- Botelho, J.C.; Kayoko, K.; Lee-Yang, W.; Calafat, A.M. Per- and Polyfluoroalkyl Substances (PFAS) Exposure in the U.S. Population: NHANES 1999–March 2020. Environ. Res. 2025, 270, 120916. [Google Scholar] [CrossRef] [PubMed]
- Pearce, J.L.; Neelon, B.; Bloom, M.S.; Buckley, J.P.; Ananth, C.V.; Perera, F.; Vena, J.; Hunt, K. Exploring associations between prenatal exposure to multiple endocrine disruptors and birth weight with exposure continuum mapping. Environ. Res. 2021, 200, 111386. [Google Scholar] [CrossRef]
- Scheringer, M.; Trier, X.; Cousins, I.T.; de Voogt, P.; Fletcher, T.; Wang, Z.; Webster, T.F. Helsingor Statement on Poly- and Perfluorinated Alkyl Substances (PFASs). Chemosphere 2014, 114, 337–339. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.Y.; Cousins, I.T.; Scheringer, M.; Buck, R.C.; Hungerbuhler, K. Global Emission Inventories for C-4-C-14 Perfluoroalkyl Carboxylic Acid (PFCA) Homologues from 1951 to 2030, Part I: Production and Emissions from Quantifiable Sources. Environ. Int. 2014, 70, 62–75. [Google Scholar] [CrossRef] [PubMed]
- Kato, K.; Wong, L.-Y.; Jia, L.T.; Kuklenyik, Z.; Calafat, A.M. Trends in Exposure to Polyfluoroalkyl Chemicals in the U.S. Population: 1999−2008. Environ. Sci. Technol. 2011, 45, 8037–8045. [Google Scholar] [CrossRef]
- Ríos-Bonilla, K.M.; Aga, D.S.; Lee, J.; Konig, M.; Qin, W.; Cristobal, J.R.; Atilla-Gokcumen, G.E.; Escher, B.I. Neurotoxic Effects of Mixtures of Perfluoroalkyl Substances (PFAS) at Environmental and Human Blood Concentrations. Environ. Sci. Technol. 2024, 58, 16774–16784. [Google Scholar] [CrossRef] [PubMed]
- Tanabe, P.; Key, P.B.; Chung, K.W.; Pisarski, E.C.; Reiner, J.L.; Rodowa, A.E.; Magnuson, J.T.; DeLorenzo, M.E. Mixture Effects of Per- and Polyfluoroalkyl Substances on Embryonic and Larval Sheepshead Minnows (Cyprinodon variegatus). Toxics 2024, 12, 91. [Google Scholar] [CrossRef] [PubMed]
- Giesy, J.P.; Kannan, K. Global distribution of perfluorooctane sulfonate in wildlife. Environ. Sci. Technol. 2001, 35, 1339–1342. [Google Scholar] [CrossRef]
- Houde, M.; De Silva, A.O.; Muir, D.C.G.; Letcher, R.J. Monitoring of perfluorinated compounds in aquatic biota: An updated review. Environ. Sci. Technol. 2011, 45, 7962–7973. [Google Scholar] [CrossRef]
- de Vos, M.G.; Huijbregts, M.A.; van den Heuvel-Greve, M.J.; Vethaak, A.D.; Van de Vijver, K.I.; Leonards, P.E.; van Leeuwen, S.P.; de Voogt, P.J.H.A. Accumulation of perfluorooctane sulfonate (PFOS) in the food chain of the Western Scheldt estuary: Comparing field measurements with kinetic modeling. Chemosphere 2008, 70, 1766–1773. [Google Scholar] [CrossRef] [PubMed]
- Kannan, K.; Tao, L.; Sinclair, E.; Pastva, S.D.; Jude, D.J.; Giesy, J.P. Perfluorinated compounds in aquatic organisms at various trophic levels in a Great Lakes food chain. Arch. Environ. Contam. Toxicol. 2005, 48, 559–566. [Google Scholar] [CrossRef]
- Martin, J.W.; Smithwick, M.M.; Braune, B.M.; Hoekstra, P.F.; Muir, D.C.; Mabury, S.A. Identification of long-chain perfluorinated acids in biota from the Canadian Arctic. Environ. Sci. Technol. 2004, 38, 373–380. [Google Scholar] [CrossRef]
- Penland, T.N.; Cope, W.G.; Kwak, T.J.; Strynar, M.J.; Grieshaber, C.A.; Heise, R.J.; Sessions, F.W. Trophodynamics of Per- and Polyfluoroalkyl Substances in the Food Web of a Large Atlantic Slope River. Environ. Sci. Technol. 2020, 54, 6800–6811. [Google Scholar] [CrossRef]
- Powley, C.R.; George, S.W.; Russell, M.H.; Hoke, R.A.; Buck, R.C. Polyfluorinated chemicals in a spatially and temporally integrated food web in the Western Arctic. Chemosphere 2008, 70, 664–672. [Google Scholar] [CrossRef]
- Tomy, G.T.; Tittlemier, S.A.; Palace, V.P.; Budakowski, W.R.; Braekevelt, E.; Brinkworth, L.; Friesen, K. Biotransformation of N-Ethyl Perfluorooctanesulfonamide by Rainbow Trout (Onchorhynchus mykiss) Liver Microsomes. Environ. Sci. Technol. 2004, 38, 758–762. [Google Scholar] [CrossRef]
- Xu, J.; Guo, C.S.; Zhang, Y.; Meng, W. Bioaccumulation and trophic transfer of perfluorinated compounds in a eutrophic freshwater food web. Environ. Pollut. 2014, 184, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Loi, E.; Yeung, I.L.W.; Taniyasu, S.; Lam, P.K.; Kannan, K.; Yamashita, N. Trophic Magnification of Poly- and Perfluorinated Compounds in a Subtropical Food Web. Environ. Sci. Technol. 2011, 45, 5506–5513. [Google Scholar] [CrossRef]
- Lassen, C.; Jensen, A.A.; Potrykus, A.; Christensen, F.; Kjølholt, J.; Jeppesen, C.N.; Mikkelsen, S.H.; Innanen, S. Survey of PFOS, PFOA and Other Perfluoroalkyl and Polyfluoroalkyl Substances; Part of the LOUS-Review; Environmental Project No. 1475; Danish Ministry of the Environment, Environmental Protection Agency: Copenhagen, Denmark, 2013. [Google Scholar]
- Fair, P.A.; House, M.; Hulsey, T.C.; Bossart, G.D.; Adams, J.; Balthis, L.; Muir, D.C.G. Assessment of perfluorinated compounds (PFCs) in plasma of bottlenose dolphins from two southeast US estuarine areas: Relationship with age, sex and geographic locations. Mar. Pollut. Bull. 2012, 64, 66–74. [Google Scholar] [CrossRef]
- Fair, P.A.; Wolf, B.; White, N.D.; Arnott, S.A.; Kannan, K.; Karthikraj, R.; Vena, J.E. Perfluoroalkyl substances (PFASs) in edible fish species from Charleston Harbor and tributaries, South Carolina, United States: Exposure and risk assessment. Environ. Res. 2019, 171, 266–277. [Google Scholar] [CrossRef]
- Aquilina-Beck, A.A.; Reiner, J.L.; Chung, K.W.; DeLise, M.J.; Key, P.B.; DeLorenzo, M.E. Uptake and biological effects of perfluorooctane sulfonate exposure in the adult Eastern oyster Crassostrea virginica. Arch. Environ. Contam. Toxicol. 2020, 79, 333–342. [Google Scholar] [CrossRef]
- Baldwin, W.S.; Bain, L.J.; Di Giulio, R.; Kullman, S.; Rice, C.D.; Ringwoo, A.H.; van den Hurk, P. 20th pollutant responses in marine organisms (PRIMO): Global issues and fundamental mechanisms caused by pollutant stress in marine and freshwater organisms. Aquat. Toxicol. 2020, 227, 105620. [Google Scholar] [CrossRef] [PubMed]
- Dhore, R.; Murthy, G.S. Per/polyfluoroalkyl substances production, applications and environmental impacts. Bioresour. Technol. 2021, 341, 125808. [Google Scholar] [CrossRef]
- MacMillan, D.K.; Wetmore, B.A.; Dasgupta, S.; Baldwin, W.S. Closing the Gaps in Understanding PFAS Toxicology and Metabolism. Toxics 2025, 13, 19. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Teng, M.; Zhao, X.; Li, Y.; Sun, J.; Zhao, W.; Ruan, Y.; Leung, K.M.Y.; Wu, F. Insight into the binding model of per- and polyfluoroalkyl substances to proteins and membranes. Environ. Int. 2023, 175, 107951. [Google Scholar] [CrossRef]
- Pfohl, M.; Marques, E.; Auclair, A.; Barlock, B.; Jamwal, R.; Goedken, M.; Akhlaghi, F.; Slitt, A.L. An ‘Omics Approach to Unraveling the Paradoxical Effect of Diet on Perfluorooctanesulfonic Acid (PFOS) and Perfluorononanoic Acid (PFNA)-Induced Hepatic Steatosis. Toxicol. Sci. 2021, 180, 277–294. [Google Scholar] [CrossRef]
- Williams, L.A.; Hamilton, M.C.; Edin, M.L.; Lih, F.B.; Eccles-Miller, J.A.; Tharayil, T.; Leonard, E.; Baldwin, W.S. Increased Perfluorooctanesulfonate (PFOS) Toxicity and Accumulation Is Associated with Perturbed Prostaglandin Metabolism and Increased Organic Anion Transport Protein (OATP) Expression. Toxics 2024, 12, 106. [Google Scholar] [CrossRef]
- Niu, S.; Cao, Y.; Chen, R.; Bedi, M.; Sanders, A.P.; Ducatman, A.; Ng, C. A State-of-the-Science Review of Interactions of Per- and Polyfluoroalkyl Substances (PFAS) with Renal Transporters in Health and Disease: Implications for Population Variability in PFAS Toxicokinetics. Environ. Health Perspect. 2023, 131, 076002. [Google Scholar] [CrossRef]
- Ryu, S.; Burchett, W.; Zhang, S.; Modaresi, S.M.S.; Areiza, J.A.; Fischer, F.C.; Slitt, A.L. Species-Specific Unbound Fraction Differences in Highly Bound PFAS: A Comparative Study across Human, Rat, and Mouse Plasma and Albumin. Toxics 2024, 12, 253. [Google Scholar] [CrossRef]
- Zhao, J.; Shi, X.; Wang, Z.; Xiong, S.; Lin, Y.; Wei, X.; Li, Y.; Tang, X. Hepatotoxicity assessment investigations on PFASs targeting L-FABP using binding affinity data and machine learning-based QSAR model. Ecotox. Environ. Saf. 2023, 262, 115310. [Google Scholar] [CrossRef] [PubMed]
- Dawson, D.E.; Lau, C.; Pradeep, P.; Sayre, R.R.; Judson, R.S.; Tornero-Velez, R.; Wambaugh, J.F. A Machine Learning Model to Estimate Toxicokinetic Half-Lives of Per- and Polyfluoro-Alkyl Substances (PFAS) in Multiple Species. Toxics 2023, 11, 98. [Google Scholar] [CrossRef] [PubMed]
- Corton, J.C.; Cunningham, M.L.; Hummer, B.T.; Lau, C.; Meek, B.; Peters, J.M.; Popp, J.A.; Rhomberg, L.; Seed, J.; Klaunig, J.E. Mode of action framework analysis for receptor-mediated toxicity: The peroxisome proliferator-activated receptor alpha (PPARα) as a case study. Crit. Rev. Toxicol. 2014, 44, 1–49. [Google Scholar] [CrossRef]
- Li, X.; Wang, Z.; Klaunig, J.E. The effects of perfluorooctanoate on high fat diet induced non-alcoholic fatty liver disease in mice. Tociology 2019, 416, 1–14. [Google Scholar] [CrossRef]
- Robarts, D.R.; Dai, J.; Lau, C.; Apte, U.; Corton, J.C. Hepatic Transcriptome Comparative In Silico Analysis Reveals Similar Pathways and Targets Altered by Legacy and Alternative Per- and Polyfluoroalkyl Substances in Mice. Toxics 2023, 11, 963. [Google Scholar] [CrossRef] [PubMed]
- Mello, T.; Materozzi, M.; Galli, A. PPARs and Mitochondrial Metabolism: From NAFLD to HCC. PPAR Res. 2016, 7403230. [Google Scholar] [CrossRef]
- Eccles, J.A.; Baldwin, W.S. Detoxification Cytochrome P450s (CYPs) in Families 1-3 Produce Functional Oxylipins from Polyunsaturated Fatty Acids. Cells 2023, 12, 82. [Google Scholar] [CrossRef]
- Cheng, W.; Doering, J.A.; LaLone, C.; Ng, C. Integrative computational approaches to inform relative bioaccumulation potential of per- and polyfluoroalkyl substances (PFAS) across species. Toxicol. Sci. 2022, 180, 212–223. [Google Scholar] [CrossRef]
- Yi, S.; Chen, P.; Yang, L.; Zhu, L. Probing the hepatotoxicity mechanisms of novel chlorinated polyfluoroalkyl sulfonates to zebrafish larvae: Implication of structural specificity. Environ. Int. 2019, 133, 105262. [Google Scholar] [CrossRef]
- Cao, H.; Zhou, Z.; Hu, Z.; Wei, C.L.; Li, J.; Wang, L.; Liu, G.; Zhang, J.; Wang, Y.; Wang, T.; et al. Effect of Enterohepatic Circulation on the Accumulation of Per- and Polyfluoroalkyl Substances: Evidence from Experimental and Computational Studies. Environ. Sci. Technol. 2022, 56, 3214–3224. [Google Scholar] [CrossRef]
- Zhao, W.; Zitzow, J.D.; Ehresman, D.J.; Chang, S.C.; Butenhoff, J.L.; Forster, J.; Hagenbuch, B. Na+/Taurocholate Cotransporting Polypeptide and Apical Sodium-Dependent Bile Acid Transporter Are Involved in the Disposition of Perfluoroalkyl Sulfonates in Humans and Rats. Toxicol. Sci. 2015, 146, 363–373. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Zitzow, J.D.; Weaver, Y.; Ehresman, D.J.; Chang, S.-C.; Butenhoff, J.L.; Hagenbuch, B. Organic Anion Transporting Polypeptides Contribute to the Disposition of Perfluoroalkyl Acids in Humans and Rats. Toxicol. Sci. 2016, 156, 84–95. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, M.C.; Heintz, M.M.; Pfohl, M.; Marques, E.; Ford, L.; Slitt, A.L.; Baldwin, W.S. Increased toxicity and retention of perflouroctane sulfonate (PFOS) in humanized CYP2B6-transgenic mice compared to Cyp2b-null mice is relieved by a high-fat diet (HFD). Food Chem. Toxicol. 2021, 152, 112175. [Google Scholar] [CrossRef]
- Loccisano, A.E.; Campbel, J.L., Jr.; Butenhoff, J.L.; Andersen, M.E.; Clewell, H.J.I. Comparison and evaluation of pharmacokinetics of PFOA and PFOS in the adult rat using a phsiologically based pharmacokinetic model. Reprod. Toxicol. 2012, 33, 452–467. [Google Scholar] [CrossRef]
- Worley, R.R.; Fisher, J. Application of physiologically-based pharmacokinetic modeling to explore the role of kidney transporters in renal reabsorption of perfluorooctanoic acid in the rat. Toxicol. Appl. Pharmacol. 2015, 289, 428–441. [Google Scholar] [CrossRef]
- Chung, K.W.; Key, P.B.; Tanabe, P.; DeLorenzo, M.E. Effects of Temperature and Salinity on Perfluorooctane Sulfonate (PFOS) Toxicity in Larval Estuarine Organisms. Toxics 2024, 12, 267. [Google Scholar] [CrossRef]
- United States Environmental Protection Agency (USEPA). PFAS Analytical Methods Development and Sampling Research; United States Environmental Protection Agency (USEPA): Washington, DC, USA, 2024. [Google Scholar]
- Sunderland, E.M.; Hu, X.C.; Dassuncao, C.; Tokranov, A.K.; Wagner, C.C.; Allen, J.G. A review of the pathways of human exposure to poly- and perfluoroalkyl substances (PFASs) and present understanding of health effects. J. Expo. Sci. Environ. Epidemiol. 2019, 29, 131–147. [Google Scholar] [CrossRef] [PubMed]
- Cao, D.; Rericha, Y.; Powley, C.; Truong, L.; Tanguay, R.L.; Field, J.A. Background Per- and Polyfluoroalkyl Substances (PFAS) in Laboratory Fish Diet: Implications for Zebrafish Toxicological Studies. Sci. Total Environ. 2022, 842, 156831. [Google Scholar] [CrossRef]
- United States Food and Drug Administration; USFDA. Determination of 30 Per and Polyfluoroalkyl Substances (PFAS) in Food and Feed Using Liquid Chromatography-Tandem Mass Spectrometry (LC-MS/MS), C-010.03; United States Food and Drug Administration: Washington, DC, USA, 2024. [Google Scholar]
- Lath, S.; Knight, E.R.; Navarro, D.A.; Kookana, R.S.; McLaughlin, M.J. Sorption of PFOA onto different laboratory materials: Filter membranes and centrifuge tubes. Chemosphere 2019, 222, 671–678. [Google Scholar] [CrossRef]
- ISO 25101; Water Quality—Determination of Perfluorooctanesulfonate (PFOS) and Perfluorooctanoate (PFOA)—Method for Unfiltered Samples Using Solid Phases Extraction and Liquid Chromatography/Mass Spectrometry. International Organization for Standardization: Geneva, Switzerland, 2009.
- Shoemaker, J.; Boutin, B.; Grimmett, P. Development of a U. S. EPA drinking water method for the analysis of selected perfluoroalakl acids by solid=phase extraction and LC-MS-MS. J. Chromatogr. Sci. 2009, 47, 3–11. [Google Scholar] [CrossRef]
- Hellsing, M.S.; Josefsson, S.; Hughes, A.V.; Ahrens, L. Sorption of perfluoroalkyl substances to two types of minerals. Chemosphere 2016, 159, 385–391. [Google Scholar] [CrossRef]
- Higgins, C.P.; Luthy, R.G. Sorption of perfluorinated surfactants on sediments. Environ. Sci. Technol. 2006, 40, 7251–7256. [Google Scholar] [CrossRef] [PubMed]
- Johansson, J.H.; Berger, U.; Cousins, I.T. Can the use of deactivated glass fibre filters eliminate sorption artifacts associated with active air sampling of perfluorooctanoicacid? Environ. Pollut. 2017, 224, 779–786. [Google Scholar] [CrossRef] [PubMed]
- Sorengard, M.; Franke, V.; Troger, R.; Ahrens, L. Losses of poly- and perfluoroalkyl substances to syringe filter materials. J. Chromatogr. A 2020, 1690, 460430. [Google Scholar] [CrossRef] [PubMed]
- Leung, S.C.E.; Wanninayake, D.; Chen, D.; Nguyen, N.-T. Physiochemical properties and interactions of perfluoroalkyl substances (PFAS)—Challenges and opportunities in sensing and remediation. Sci. Total Environ. 2023, 905, 166764. [Google Scholar] [CrossRef]
- Martin, J.W.; Kannan, K.; Berger, U.; De Voogt, P.; Field, J.; Franklin, J.; Giesy, J.P.; Harner, T.; Muir, D.C.G.; Scott, B.; et al. Analytical Challenges Hamper Perfluoroalkyl Research. Environ. Sci. Technol. 2004, 38, 248A–255A. [Google Scholar] [CrossRef]
- Liberatore, H.K.; Jackson, S.R.; Strynar, M.J.; McCord, J.P. Solvent Suitability for HFPO-DA (“GenX” Parent Acid) in Toxicological Studies. Environ. Sci. Technol. Lett. 2020, 7, 477–481. [Google Scholar] [CrossRef]
- Bangma, J.T.; Reiner, J.; Fry, R.C.; Manuck, T.; McCord, J.; Strynar, M.J. Identification of an Analytical Method Interference for Perfluorobutanoic Acid in Biological Samples. Environ. Sci. Technol. Lett. 2021, 8, 1085–1090. [Google Scholar] [CrossRef]
- Karrman, A.; Domingo, J.L.; Llebaria, X.; Nadal, M.; Bigas, E.; van Bavel, B.; Lindstrom, G. Biomonitoring perfluorinated compounds in Catalonia, Spain: Concentrations and trends in human liver and milk samples. Environ. Sci. Pollut. Res. 2010, 17, 750–758. [Google Scholar] [CrossRef] [PubMed]
- Scherer, M.; Smith, J.; Steed, J.; McMillan, D.; Stahl-Zeng, J. Quantitation of Per- and Polyfluoroalkyl Substances (PFAS) in Foodstuffs; Volume Sciex Technical Note MKT-28758-B2023; Sciex: Marlborough, MA, USA, 2023. [Google Scholar]
- Young, W.; Wiggins, S.; Limm, W.; Fisher, C.M.; DeJager, L.; Genualdi, S. Analysis of Per- and Poly(fluoroalkyl) Substances (PFASs) in Highly Consumed Seafood Products from U. S. Markets. J. Agric. Food Chem. 2022, 70, 13535–13553. [Google Scholar] [CrossRef]
- Van der Veen, I.; Fielder, H.; de Boer, J. Assessment of the per- and polyfluoroalkyl substances analysis under the Stockholm Convention—2018/2019. Chemosphere 2023, 313, 137549–137556. [Google Scholar] [CrossRef]
- EPA 821-R-24-001; Method 1633 Analysis of Per- and Polyfluoroalkyl Substances (PFAS) in Aqueous, Solid, Biosolids, and Tissue Samples by LC-MS/MS. United States Environmental Protection Agency (USEPA): Washington, DC, USA, 2024.
- Nhanes 2017–2018, Method 6304.09; Perfluoroalkyl and Polyfluoroalkyl Substances, Online Solid Phase Extraction-High Performance Liquid Chromatography-Turbo Ion Spray-Tandem Mass Spectrometry. Center for Disease Control and Prevention (CDC): Atlanta, Georgia, 2017–2018. Available online: https://wwwn.cdc.gov/nchs/data/nhanes/public/2013/labmethods/PFAS_H_MET.pdf (accessed on 18 June 2025).
- Baker, N.; Knudsen, T.; Williams, A. Abstract Sifter: A comprehensive front-end system to PubMed. F1000Research 2017, 6, 2164. [Google Scholar] [CrossRef]
- Comito, R.; Porru, E.; Violante, F.S. Analytical methods employed in the identification and quantification of per-and polyfluoroalkyl substances in human matrices—A scoping review. Chemosphere 2023, 345, 140433. [Google Scholar] [CrossRef]
- Conley, J.; Lambright, C.S.; Evans, N.; McCord, J.; Strynar, M.J.; Hill, D.; Medlock-Kakaley, E.; Wilson, V.; Gray, L.E., Jr. Hexafluorpropylene oxide-dimer acid (HFPO-DA or GenX) alters maternal and fetal glucose and lipid metabolism and produces neonatal mortality, low birthweight, and hepatomegaly in the Sprague-Dawley rat. Environ. Int. 2021, 146, 106204. [Google Scholar] [CrossRef]
- Conley, J.; Lambright, C.S.; Evans, N.; Medlock-Kakaley, E.; Hill, D.; McCord, J.; Strynar, M.J.; Wehmas, L.C.; Hester, S.; MacMillan, D.K.; et al. Developmental toxicity of Nafion byproduct 2 (NBP2) in the Sprague-Dawley rat with comparisons to hexafluoropropylene oxide-dimer acid (HFPO-DA or GenX) and perfluorooctane sulfonate (PFOS). Environ. Int. 2022, 160, 107056. [Google Scholar] [CrossRef] [PubMed]
- Renyer, A.; Ravindra, K.; Wetmore, B.A.; Ford, J.L.; DeVito, M.; Hughes, M.F.; Wehmas, L.C.; MacMillan, D.K. Dose Response, Dosimetric, and Metabolic Evaluations of Replacement PFAS Perfluoro-(2,5,8-trimethyl-3,6,9-trioxadodecanoic) Acid (HFPO-TeA). Toxics 2023, 11, 951. [Google Scholar] [CrossRef]
- Huang, M.C.; Dzierlenga, A.L.; Robinson, V.G.; Waidyanatha, S.; DeVito, M.J.; Elfrid, M.A.; Granville, C.A.; Gibbs, S.T.; Blystone, C.R. Toxicokinetics of perfluorobutane sulfonate (PFBS), perfluorohexane-1-sulphonic acid (PFHxS), and perfluorooctane sulfonic acid in male and female Hsd:Sprague SD rats after intravenous and gavage administration. Toxicol. Rep. 2019, 6, 645–655. [Google Scholar] [CrossRef] [PubMed]
- Dukes, D.A.; McDonough, C.A. N-glucuronidation and Excretion of Perfluoroalkyl Sulfonamides in Mice Following Ingestion of Aqueous Film-Forming Foam. Environ. Toxicol. Chem. 2024, 43, 2274–2284. [Google Scholar] [CrossRef]
- Kreutz, A.; Clifton, M.S.; Henderons, W.M.; Smeltz, M.G.; Phillips, M.; Wambaugh, J.F.; Wetmore, B.A. Category-Based Toxicokinetic Evaluations of Data-Poor Per- and Polyfluoroalkyl Substances (PFAS) using Gas Chromatography Coupled with Mass Spectrometry. Toxics 2023, 11, 463. [Google Scholar] [CrossRef]
- Bangma, J.; Barry, K.M.; Fisher, C.M.; Genualdi, S.; Guillette, T.C.; Huset, C.A.; McCord, J.; Ng, B.; Place, B.J.; Reiner, J.L.; et al. PFAS Ghosts: How to identify, evaluate, and exorcise new and existing analytical interference. Anal. Bioanal. Chem. 2024, 416, 1777–1785. [Google Scholar] [CrossRef] [PubMed]
- Chan, E.; Sandhu, M.; Benskin, J.P.; Ralitsch, M.; Thibault, N.; Birkholz, D.; Martin, J.W. Endogenous high performance liquid chromatography/tandem mass spectrometry interferences and the case of perfluorohexane sulfonate (PFHxS) in human serum; are we overestimating exposure? Rapid. Comm. Mass. Spectrom. 2009, 23, 1405–1410. [Google Scholar] [CrossRef]
- Benskin, J.P.; Bataineh, M.; Martin, J.W. Simultaneous Characterization of Perfluoroalkyl Carboxylate, Sulfonate, and Sulfonamide Isomers by Liquid Chromatography–Tandem Mass Spectrometry. Anal. Chem. 2007, 79, 6455–6464. [Google Scholar] [CrossRef]
- Kolanczyk, R.C.; Saley, M.R.; Serrano, J.A.; Daley, S.M.; Tapper, M.A. PFAS Biotransformation Pathways: A Species Comparison Study. Toxics 2023, 11, 74. [Google Scholar] [CrossRef] [PubMed]
- Ateia, M.; Chiang, D.; Cashman, M.; Acheson, C. Total Oxidizable Precursor (TOP) Assay—Best Practices, Capabilities and Limitations for PFAS Site Investigation and Remediation. Environ. Sci. Technol. Lett. 2024, 10, 292–301. [Google Scholar] [CrossRef]
- European Commission. A9-0238; Proposal for a Directive of the European Parliament and of the Council Amending Directive 2000/60/Ec Establishing a Framework for Community Action in the Field of Water Policy, Directive 2006/118/Ec on the Protection of Groundwater Against Pollution and Deterioration and Directive 2008/105/Ec on Environmental Quality Standards in the Field of Water Policy. European Commission: Brussels, Belgium, 2023.
- United States Environmental Protection Agency (USEPA). Per- and Polyfluoroalkyl Substances (PFAS) Final PFAS National Primary Drinking Water Regulation; United States Environmental Protection Agency (USEPA): Washington, DC, USA, 2024. [Google Scholar]
- Licul-Kucera, V.; Ragnarsdottir, O.; Fromel, T.; van Wezel, A.P.; Knepper, T.P.; Harrad, S.; Abdallah, M.A.-E. Interspecies comparison of metabolism of two novel prototype PFAS. Chemosphere 2024, 351, 141237. [Google Scholar] [CrossRef]
- Vestergren, R.; Cousins, I.T.; Trudel, D.; Wormuth, M.; Scheringer, M. Estimating the contribution of precursor compounds in consumer exposure to PFOS and PFOA. Chemosphere 2008, 73, 1617–1624. [Google Scholar] [CrossRef] [PubMed]
- McCord, J.; Strynar, M. Identification of Per- and Polyfluoroalkyl Substances in the Cape Fear River by High Resolution Mass Spectrometry and Nontargeted Screening. Environ. Sci. Technol. 2019, 53, 4717–4727. [Google Scholar] [CrossRef]
- Pu, S.; McCord, J.; Bangma, J.; Sobus, J. Establishing performance metrics for quantitative non-targeted analysis: A demonstration using per- and polyfluoroalkyl substances. Anal. Bioanal. Chem. 2024, 416, 1249–1267. [Google Scholar] [CrossRef] [PubMed]
- Idjaton, B.I.T.; Togola, A.; Ghestem, J.P.; Kastler, L.; Bristeau, S.; Ronteltap, M.; Colombano, S.; Devau, N.; Lions, J.; van Hullebusch, E.D. Determination of organic fluorinated compounds content in complex samples through combustion ion chromatography methods: A way to define a “Total Per- and Polyfluoroalkyl Substances (PFAS)” parameter? Sci. Total Environ. 2024, 932, 172589. [Google Scholar] [CrossRef]
- Bidaisee, S.; Macpherson, C.N.L. Zoonoses and One Health: A Review of the Literature. J. Parasitol. Res. 2014, 2014, 874345. [Google Scholar] [CrossRef]
- Bečanová, J.; Komprdová, K.; Vrana, B.; Klánová, J. Annual dynamics of perfluorinated compounds in sediment: A case study in the Morava River in Zlín district, Czech Republic. Chemosphere 2016, 151, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Gribble, M.O.; Bartell, S.M.; Kannan, K.; Wu, Q.; Fair, P.A.; Kamen, D.L. Longitudinal measures of perfluoroalkyl substances (PFAS) in serum of Gullah African Americans in South Carolina: 2003–2013. Environ. Res. 2015, 143, 82–88. [Google Scholar] [CrossRef]
- Reif, J.S.; Schaefer, A.M.; Bossart, G.D.; Fair, P.A. Health and Environmental Risk Assessment Project for bottlenose dolphins Tursiops truncatus from the southeastern USA. II. Environmental aspects. Dis. Aquat. Org. 2017, 125, 155. [Google Scholar] [CrossRef] [PubMed]
- Bossart, G.D.; Fair, P.; Schaefer, A.M.; Reif, J.S. Health and Environmental Risk Assessment Project for bottlenose dolphins Tursiops truncatus from the southeastern USA. I. Infectious diseases. Dis. Aquat. Org. 2017, 125, 141. [Google Scholar] [CrossRef]
- Wang, Y.; Chang, W.; Wang, L.; Zhang, Y.; Zhang, Y.; Wang, M.; Wang, Y.; Li, P. A review of sources, multimedia distribution and health risks of novel fluorinated alternatives. Ecotox. Environ. Saf. 2019, 182, 109402. [Google Scholar] [CrossRef]
- Boiteux, V.; Dauchy, X.; Bach, C.; Colin, A.; Hemard, J.; Sagres, V.; Rosin, C.; Munoz, J.-F. Concentrations and patterns of perfluoroalkyl and polyfluoroalkyl substances in a river and three drinking water treatment plants near and far from a major production source. Sci. Total Environ. 2017, 583, 393–400. [Google Scholar] [CrossRef]
- Ma, T.; Pan, X.; Wang, T.; Li, X.; Luo, Y. Toxicity of Per- and Polyfluoroalkyl Substances to Nematodes. Toxics 2023, 11, 593. [Google Scholar] [CrossRef]
- Beccacece, L.; Costa, F.; Pascali, J.P.; Giorgi, F.M. Cross-species transcriptomics analysis highlights conserved molecular responses to per- and polyfluoroalkyl substances. Toxics 2023, 11, 567. [Google Scholar] [CrossRef]
- Rock, K.D.; Polera, M.E.; Guillette, T.C.; Starnes, H.M.; Dean, K.; Watters, M.; Stevens-Stewart, D.; Belcher, S.M. Domestic Dogs and Horses as Sentinels of Per- and Polyfluoroalkyl Substance Exposure and Associated Health Biomarkers in Gray’s Creek North Carolina. Environ. Sci. Technol. 2023, 57, 9567–9579. [Google Scholar] [CrossRef]
- Kirkwood, K.I.; Fleming, J.; Nguyen, H.; Reif, D.M.; Baker, E.S.; Belcher, S.M. Utilizing Pine Needles to Temporally and Spatially Profile Per- and Polyfluoroalkyl Substances (PFAS). Environ. Sci. Technol. 2022, 56, 3441–3451. [Google Scholar] [CrossRef] [PubMed]
- Jian, J.-M.; Chen, D.; Han, F.-J.; Guo, Y.; Zeng, L.; Lu, X.; Wang, F. A short review on human exposure to and tissue distribution of per- and polyfluoroalkyl substances (PFASs). Sci. Total Environ. 2018, 636, 1058–1069. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.-S.; Wen, L.-L.; Chu, P.-L.; Lin, C.-Y. Association among total serum isomers of perfluorinated chemicals, glucose homeostasis, lipid profiles, serum protein and metabolic syndrome in adults: Nhanes, 2013–2014. Environ. Pollut. 2018, 232, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Greaves, A.K.; Letcher, R.J.; Sonne, C.; Dietz, R. Brain region distribution and patterns of bioaccumulative perfluoroalkyl carboxylates and sulfonates in East Greenland polar bears (Ursus maritimus). Environ. Toxicol. Chem. 2013, 32, 713–722. [Google Scholar] [CrossRef]
- Guillette, T.C.; Jackson, T.W.; Guillette, M.; McCord, J.; Belcher, S.M. Blood concentrations of per- and polyfluoroalkyl substances are associated with autoimmune-like effects in American alligators from Wilmington, North Carolina. Front. Toxicol. 2022, 4, 1010185. [Google Scholar] [CrossRef]
- Bost, P.C.; Strynar, M.J.; Reiner, J.L.; Zweigenbaum, J.A.; Secoura, P.L.; Lindstrom, A.B.; Dye, J.A. U.S. domestic cats as sentinels for perfluoroalkyl substances: Possible linkages with housing, obesity, and disease. Environ. Res. 2016, 151, 145–153. [Google Scholar] [CrossRef]
- De Silva, A.O.; Spencer, C.; Ho, K.C.D.; Tarhuni, M.A.; Go, C.; Houde, M.; de Solla, S.R.; Lavoie, R.A.; King, L.E.; Muir, D.C.G.; et al. Perfluoroalkylphosphinic Acids in Northern Pike (Esox lucius), Double-Crested Cormorants (Phalacrocorax auritus), and Bottlenose Dolphins (Tursiops truncatus) in Relation to Other Perfluoroalkyl Acids. Environ. Sci. Technol. 2016, 50, 10903–10913. [Google Scholar] [CrossRef]
- United States Environmental Protection Agency (EPA). Health Effects Support Document for Perfluorooctanoic Acid (PFOA); United States Environmental Protection Agency (EPA): Washington, DC, USA, 2016. [Google Scholar]
- Steenland, K.; Winquist, A. PFAS and cancer, a scoping review of the epidemiologic evidence. Environ. Res. 2021, 194, 110690. [Google Scholar] [CrossRef]
- Fenton, S.E.; Ducatman, A.; Boobis, A.; DeWitt, J.C.; Lau, C.; Ng, C.; Smith, J.S.; Roberts, S.M. Per- and Polyfluoroalkyl Substance Toxicity and Human Health Review: Current State of Knowledge and Strategies for Informing Future Research. Environ. Toxicol. Chem. 2021, 40, 606–630. [Google Scholar] [CrossRef]
- Rappazzo, M.K.; Coffman, E.; Hines, P.E. Exposure to Perfluorinated Alkyl Substances and Health Outcomes in Children: A Systematic Review of the Epidemiologic Literature. Int. J. Environ. Res. Public Health 2017, 14, 691. [Google Scholar] [CrossRef] [PubMed]
- Appel, M.; Forsthuber, M.; Ramos, R.; Widhalm, R.; Granitzer, S.; Uhl, M.; Hengstschlaeger, M.; Stamm, T.; Gundacker, C. The Transplacental Transfer Efficiency of Per- and Polyfluoroalkyl Substances (PFAS): A First Meta-Analysis. J. Toxicol. Environ. Healtth-Part B-Crit. Rev. 2022, 25, 23–42. [Google Scholar] [CrossRef] [PubMed]
- Gui, S.-Y.; Chen, Y.-N.; Wu, K.-J.; Liu, W.; Wang, W.-J.; Liang, H.-R.; Jiang, Z.-X.; Li, Z.-L.; Hu, C.-Y. Association between Exposure to Per- and Polyfluoroalkyl Substances and Birth Outcomes: A Systematic Review and Meta-Analysis. Front. Public Health 2022, 10, 855348. [Google Scholar] [CrossRef]
- Wright, J.M.; Lee, A.L.; Rappazzo, K.M.; Ru, H.; Radke, E.G.; Bateson, T.F. Systematic Review and Meta-Analysis of Birth Weight and PFNA Exposures. Environ. Res. 2023, 222, 115357. [Google Scholar] [CrossRef]
- Coperchini, F.; Croce, L.; Ricci, G.; Magri, F.; Rotondi, M.; Imbriani, M.; Chiovato, L. Thyroid Disrupting Effects of Old and New Generation PFAS. Front. Endocrinol. 2021, 11, 612320. [Google Scholar] [CrossRef]
- Kim, M.J.; Moon, S.; Oh, B.-C.; Jung, D.; Ji, K.; Choi, K.; Park, Y.J. Association between Perfluoroalkyl Substances Exposure and Thyroid Function in Adults: A Meta-Analysis. PLoS ONE 2018, 13, e0197244. [Google Scholar] [CrossRef]
- Dzierlenga, M.W.; Allen, B.C.; Clewell, H.J.; Longnecker, M.P. Pharmacokinetic Bias Analysis of an Association between Clinical Thyroid Disease and Two Perfluoroalkyl Substances. Environ. Int. 2020, 141, 105784. [Google Scholar] [CrossRef]
- Dzierlenga, M.W.; Moreau, M.; Song, G.; Mallick, P.; Ward, P.L.; Campbell, J.L.; Housand, C.; Yoon, M.; Allen, B.C.; Clewell, H.J.I.; et al. Quantitative Bias Analysis of the Association between Subclinical Thyroid Disease and Two Perfluoroalkyl Substances in a Single Study. Environ. Res. 2020, 182, 109017. [Google Scholar] [CrossRef] [PubMed]
- Hall, A.M.; Braun, J.M. Per- and Polyfluoroalkyl Substances and Outcomes Related to Metabolic Syndrome: A Review of the Literature and Current Recommendations for Clinicians. Am. J. Lifestyle Med. 2023, 19, 15598276231162802. [Google Scholar] [CrossRef]
- Stratakis, N.; Rock, S.; La Merrill, M.A.; Saez, M.; Robinson, O.; Fecht, D.; Vrijheid, M.; Valvi, D.; Conti, D.V.; McConnell, R.; et al. Prenatal Exposure to Persistent Organic Pollutants and Childhood Obesity: A Systematic Review and Meta-Analysis of Human Studies. Obes. Rev. 2022, 23, e13383. [Google Scholar] [CrossRef]
- Gui, S.-Y.; Qiao, J.-C.; Xu, K.-X.; Li, Z.-L.; Chen, Y.-N.; Wu, K.-J.; Jiang, Z.-X.; Hu, C.-Y. Association between Per- and Polyfluoroalkyl Substances Exposure and Risk of Diabetes: A Systematic Review and Meta-Analysis. J. Exp. Sci. Environ. Epidemiol. 2023, 33, 40–55. [Google Scholar] [CrossRef] [PubMed]
- Radke, E.G.; Wright, J.M.; Christensen, K.; Lin, C.J.; Goldstone, A.E.; Lemeris, C.; Thayer, K.A. Epidemiology Evidence for Health Effects of 150 Per- and Polyfluoroalkyl Substances: A Systematic Evidence Map. Environ. Health Perspect. 2022, 130, 096003. [Google Scholar] [CrossRef]
- Marques, E.; Pfohl, M.; Wei, W.; Tarantola, G.; Ford, L.; Amaeze, O.; Alesio, J.; Ryu, S.; Jia, X.; Zhu, H.; et al. Replacement per- and polyfluoroalkyl substances (PFAS) are potent modulators of lipogenic and drug metabolizing gene expression signatures in primary human hepatocytes. Toxicol. Appl. Pharmacol. 2022, 442, 115991. [Google Scholar] [CrossRef]
- Liu, S.H.; Kuiper, J.R.; Chen, Y.; Feuerstahler, L.; Teresi, J.; Buckley, J.P. Developing an Exposure Burden Score for Chemical Mixtures Using Item Response Theory, with Applications to PFAS Mixtures. Environ. Health Perspect. 2022, 130, 117001. [Google Scholar] [CrossRef]
- Zeng, Z.T.; Song, B.; Xiao, R.; Zeng, G.M.; Gong, J.L.; Chen, M.; Xu, P.A.; Zhang, P.; Shen, M.C.; Yi, H. Assessing the Human Health Risks of Perfluorooctane Sulfonate by in Vivo and in Vitro Studies. Environ. Int. 2019, 126, 598–610. [Google Scholar] [CrossRef] [PubMed]
- Negri, E.; Metruccio, F.; Guercio, V.; Tosti, L.; Benfenati, E.; Bonzi, R.; La Vecchia, C.; Moretto, A. Exposure to PFOA and PFOS and Fetal Growth: A Critical Merging of Toxicological and Epidemiological Data. Crit. Rev. Toxicol. 2017, 47, 482–508. [Google Scholar] [CrossRef]
- Mokra, K. Endocrine Disruptor Potential of Short- and Long-Chain Perfluoroalkyl Substances (PFASs)—A Synthesis of Current Knowledge with Proposal of Molecular Mechanism. Int. J. Mol. Sci. 2021, 22, 2148. [Google Scholar] [CrossRef]
- Gupta, R.; Kumar, P.; Fahmi, N.; Garg, B.; Dutta, S.; Sachar, S.; Matharu, A.S.; Vimaleswaran, K.S. Endocrine disruption and obesity: A current review on environmental obesogens. Curr. Res. Green. Sustain. Chem. 2020, 3, 100009. [Google Scholar] [CrossRef]
- Behr, A.-C.; Plinsch, C.; Braeuning, A.; Buhrke, T. Activation of human nuclear receptors by perfluoroalkylated substances (PFAS). Toxicology 2020, 62, 104700. [Google Scholar] [CrossRef] [PubMed]
- Heindel, J.J.; Blumberg, B. Environmental Obesogens: Mechanisms and Controversies. Annu. Rev. Pharmacol. Toxicol. 2019, 59, 89–106. [Google Scholar] [CrossRef]
- LeBlanc, G.A.; Norris, D.O.; Kloas, W.; Kullman, S.W.; Baldwin, W.S.; Greally, J.M. Detailed Review Paper on the State of the Science on Novel In Vitro and In Vivo Screening and Testing Methods and Endpoints for Evaluating Endocdrine Disruptors; Organisation for Economic Co-Operation and Development: Paris, France, 2012; p. 213. [Google Scholar]
- National Institute of Environmental Health Sciences. National Toxicology Program (NTP). Monograph on Immunotoxicity Associated with Exposure to Perfluorooctanoic Acid (PFOA) and Perfluorooctane Sulfonate (PFOS); National Institute of Environmental Health Sciences: Durham, NC, USA, 2016. [Google Scholar]
- Chang, E.T.; Adami, H.-O.; Boffetta, P.; Wedner, H.J.; Mandel, J.S. A critical review of perfluorooctanoate and perfluorooctanesulfonate exposure and immunological health conditions in humans. Crit. Rev. Toxicol. 2016, 46, 279–331. [Google Scholar] [CrossRef]
- Kim, M.; Kim, S.H.; Choi, J.Y.; Park, Y.J. Investigating fatty liver disease-associated adverse outcome pathways of perfluorooctane sulfonate using a systems toxicology approach. Food Chem. Toxicol. 2023, 176, 113781. [Google Scholar] [CrossRef]
- Sadrabadi, F.; Alarcan, J.; Sprenger, H.; Braeuning, A.; Buhrke, T. Impact of perfluoroalkyl substances (PFAS) and PFAS mixtures on lipid metabolism in differentiated HepaRG cells as a model for human hepatocytes. Arch. Toxicol. 2024, 98, 507–524. [Google Scholar] [CrossRef]
- Frangione, B.; Birk, S.; Benzouak, T.; Rodriguez-Villamizar, L.A.; Karim, F.; Dugandzic, R.; Villeneuve, P.J. Exposure to perfluoroalkyl and polyfluoroalkyl substances and pediatric obesity: A systematic review and meta-analysis. Int. J. Obes. 2024, 48, 131–146. [Google Scholar] [CrossRef] [PubMed]
- Stratakis, N.; Vrijheid, M. Invited Perspective: PFAS and the Childhood Obesity Phenotype—Challenges and Opportunities. Environ. Health Perspect. 2023, 131, 061301. [Google Scholar] [CrossRef]
- Rosen, M.B.; Das, K.P.; Wood, C.R.; Wolf, C.J.; Abbott, B.D.; Lau, C. Evaluation of perfluoroalkyl acid activity using primary mouse and human hepatocytes. Toxicology 2013, 308, 129–137. [Google Scholar] [CrossRef]
- McDonough, C.A.; Choyke, S.; Ferguson, P.L.; DeWitt, J.C.; Higgins, C.P. Bioaccumulation of Novel Per- and Polyfluoroalkyl Substances in Mice Dosed with an Aqueous Film-Forming Foam. Environ. Sci. Technol. 2020, 54, 5700–5709. [Google Scholar] [CrossRef] [PubMed]
- United States Environmental Protection Agency (USEPA). PFAS National Primary Drinking Water Regulation Rulemaking; United States Environmental Protection Agency (USEPA): Washington, DC, USA, 2023. [Google Scholar]
- Takeda, K.; Saito, T.; Sasaki, S.; Eguchi, A.; Sugiyama, M.; Eto, S.; Suzuki, K.; Kamata, R. Toxicity Assessment of Mixed Exposure of Nine Perfluoroalkyl Substances at Concentrations Relevant to Daily Intake. Toxics 2024, 12, 52. [Google Scholar] [CrossRef]
- Shearer, J.J.; Callahan, C.L.; Calafat, A.M.; Huang, W.-Y.; Jones, R.R.; Sabbisetti, V.S.; Freedman, N.D.; Sampson, J.N.; Silverman, D.T.; Purdue, M.P.; et al. Serum concentrations of per- and polyfluoroaklyl substances and risk of renal cell carcinoma. J. Natl. Cancer Inst. 2021, 113, 580–587. [Google Scholar] [CrossRef] [PubMed]
- Kalmendulis, L.M.; Hocevar, J.M.; Stephens, M.; Sandusky, G.E.; Hacevar, B.A. Exposure to perflurooctanoic acid leads to promotion of pancreatic cancer. Carcinogenesis 2022, 43, 469–478. [Google Scholar] [CrossRef]
- Eriksen, K.T.; Sørensen, M.; McLaughlin, J.K.; Lipworth, L.; Tjønneland, A.; Overvad, K.; Raaschou-Nielsen, O. Perfluorooctanoate and perfluorooctanesulfonate plasma levels and risk of cancer in the general Danish population. J. Natl. Cancer Inst. 2009, 101, 605–609. [Google Scholar] [CrossRef]
- Nelson, J.W.; Scammell, M.K.; Hatch, E.E.; Webster, T.F. Social disparities in exposures to bisphenol A and polyfluoroalkyl chemicals: A cross-sectional study within Nhanes 2003–2006. Environ. Health 2012, 11, 10. [Google Scholar] [CrossRef]
- Ding, N.; Karvonen-Gutierrez, C.A.; Zota, A.R.; Mukherjee, B.; Harlow, S.D.; Park, S.K. The role of exposure to per and polyfluroalkyl substances in racial/ethic disparities in hypertension: Results from the study of women’s health across the nation. Environ. Res. 2023, 227, 115813. [Google Scholar] [CrossRef] [PubMed]
- Rhee, J.; Chang, V.C.; Cheng, I.; Calafat, A.M.; Botelho, J.C.; Shearer, J.J.; Sampson, J.N.; Setiawan, V.W.; Wilkens, L.R.; Silverman, D.T.; et al. Serum concentrations of per- and polyfluoroalkyl substances and risk of renal cell carcinoma in the multiethnic cohort study. Environ. Int. 2023, 180, 108197. [Google Scholar] [CrossRef]
- Calafat, A.M.; Wong, L.Y.; Kuklenyik, Z.; Reidy, J.A.; Needham, L.L. Polyfluoroalkyl Chemicals in the U.S. Population: Data from the National Health and Nutrition Examination Survey (Nhanes) 2003–2004 and Comparisons with Nhanes 1999–2000. Environ. Health Perspect. 2007, 115, 1596–1602. [Google Scholar] [CrossRef]
- Nguyen, V.K.; Kahana, A.; Heidt, J.; Polemi, K.; Kvasnicka, J.; Jolliet, O.; Colacino, J.A. A Comprehensive Analysis of Racial Disparities in Chemical Biomarker Concentrations in United States Women, 1999–2014. Environ. Int. 2020, 137, 105496. [Google Scholar] [CrossRef]
- Britton, K.N.; Judson, R.S.; Hill, B.N.; Jarema, K.A.; Olin, J.K.; Knapp, B.R.; Lowery, M.; Feshuk, M.; Brown, J.; Padilla, S. Using Zebrafish to Screen Developmental Toxicity of Per- and Polyfluoroalkyl Substances (PFAS). Toxics 2024, 12, 501. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Quan, X.; Lei, S.; Huang, Z.; Wang, Q.; Xu, P. PFOS inhibited normal functional development of placenta cells via PPARg signaling. Biomedicines 2021, 9, 677. [Google Scholar] [CrossRef] [PubMed]
- Olsen, G.W.; Zobel, L.R. Assessment of lipid, hepatic, and thyroid parameters with serum perfluorooctanoate (PFOA) concentrations in fluorochemical production workers. Int. Arch. Occup. Environ. Health 2007, 81, 231–246. [Google Scholar] [CrossRef]
- Fletcher, T.; Galloway, T.S.; Melzer, D.; Holcroft, P.; Cipelli, R.; Pilling, L.C.; Mondal, D.; Luster, M.; Harries, L.W. Associations between PFOA, PFOS and changes in the expression of genes involved in cholesterol metabolism in humans. Environ. Int. 2013, 57–58, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Klaunig, J.E.; Hocevar, B.A.; Kamendulis, L.M. Mode of Action analysis of perfluorooctanoic acid (PFOA) tumorigenicity and Human Relevance. Reprod. Toxicol. 2012, 33, 410–418. [Google Scholar] [CrossRef]
- Yang, C.-H.; Glover, K.P.; Han, X. Characterization of cellular uptake of Perfluorooctanoate via organic anion-transporting polypeptide 1A2, organic anion transporter 4, and urate transporter 1 for their potential roles in mediating human renal reabsorption of perflurocarboxylates. Toxicol. Sci. 2010, 117, 294–302. [Google Scholar] [CrossRef]
- Albers, J.; Mylroie, J.; Kimble, A.; Steward, C.; Chapman, K.; Wilbanks, M.; Perkins, E.; Garcia-Reyero, N. Per- and Polyfluoroalkyl Substances: Impacts on Morphology, Behavior and Lipid Levels in Zebrafish Embryos. Toxics 2024, 12, 192. [Google Scholar] [CrossRef]
- Sobolewski, M.; Conrad, K.; Allen, J.L.; Weston, H.; Martin, K.; Lawrence, B.P.; Cory-Slechta, D.A. Sex-specific enhanced behavioral toxicity induced by maternal exposure to a mixture of low dose endocrine-disrupting chemicals. Neurotoxicology 2014, 45, 121–130. [Google Scholar] [CrossRef]
- Corsini, E.; Avogadro, A.; Galbiati, V.; dell’Agli, M.; Marinovich, M.; Galli, C.L.; Germolec, D.R. In vitro evaluation of the immunotoxic potential of perfluorinated compounds (PFCs). Toxicol. Appl. Pharmacol. 2011, 250, 108–116. [Google Scholar] [CrossRef]
- Timmermann, C.A.G.; Jensen, K.J.; Nielson, F.; Budtz-Jorgensen, E.; van der Klis, F.; Benn, C.S.; Grandjean, P.; Fisker, A.B. Serum Perfluoroalkyl Substances, Vaccine Responses, and Morbidity in a Cohort of Guinea-Bissau Children. Environ. Health Perspect. 2020, 128, 087002. [Google Scholar] [CrossRef]
- Louisse, J.; Dallafiora, L.; van den Heuvel, J.J.M.W.; Rijkers, D.; Leenders, L.; Dorne, J.-L.C.M.; Punt, A.; Russell, F.G.M.; Koenderink, J.B. Perfluoroalkyl substances (PFASs) are substrates of the renal human organic anion transporter 4 (OAT4). Arch. Toxicol. 2022; 97, 685–696. [Google Scholar]
- Han, X.; Nabb, D.L.; Russell, M.H.; Kennedy, G.L.; Rickard, R.W. Renal Elimination of Perfluorocarboxylates (PFCAs). Chem. Res. Toxicol. 2012, 25, 35–46. [Google Scholar] [CrossRef] [PubMed]
- Wen, L.-L.; Lin, C.-Y.; Chou, H.-C.; Chang, C.-C.; Lo, H.-Y.; Juan, S.-H. Perfluorooctanesulfonate Mediates Renal Tubular Cell Apoptosis through PPARgamma Inactivation. PLoS ONE 2016, 11, e0155190. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Yan, S.; Wang, P.; Chen, Q.; Liu, Y.; Cui, J.; Liang, Y.; Ren, S.; Gao, Y. Perfluorooctanoic acid (PFOA) exposure in relation to the kidneys: A review of current available literature. Front. Physiol. 2023, 14, 1103141. [Google Scholar] [CrossRef]
- Hampson, H.E.; Li, S.; Walker, D.I.; Wang, H.; Jia, Q.; Rock, S.; Costello, E.; Bjornstad, P.; Pyle, L.; Nelson, J.; et al. The potential mediating role of the gut microbiome and metabolites in the association between PFAS and kidney function in young adults: A proof-of-concept study. Sci. Total Environ. 2024, 954, 176519. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Fu, J.; Sun, J.; Hall, D.R.; Yang, D.; Blatz, D.; Houck, K.A.; Ng, C.; Doering, J.A.; LaLone, C.; et al. Quantitative chemical proteomics reveals interspecies variations on binding schemes of L-FABP with perfluorooctanesulfonate. Environ. Sci. Technol. 2022, 55, 9012–9023. [Google Scholar] [CrossRef] [PubMed]
- Khazaee, M.; Christie, E.; Cheng, W.; Michalsen, M.; Field, J.; Ng, C. Perfluoroalkyl Acid Binding with Peroxisome Proliferator-Activated Receptors α, γ, and δ, and Fatty Acid Binding Proteins by Equilibrium Dialysis with a Comparison of Methods. Toxics 2021, 9, 45. [Google Scholar] [CrossRef]
- Conway, B.N.; Badders, A.N.; Costacou, T.; Arthur, J.M.; Innes, K.E. Perfluoroalkyl substances and kidney function in chronic kidney disease, anemia, and diabetes. Diab Met. Syn. Obes Targets Ther. 2018, 11, 707–716. [Google Scholar] [CrossRef]
- Kaye, E.; Marques, E.; Areiza, J.A.; Modaresi, S.M.S.; Slitt, A. Exposure to a PFOA, PFOS and PFHxS Mixture during Gestation and Lactation Alters the Liver Proteome in Offspring of CD-1 Mice. Toxics 2024, 12, 348. [Google Scholar] [CrossRef]
- Wan, H.T.; Zhao, Y.G.; Wei, X.; Hui, K.Y.; Giesey, J.P.; Wong, C.K.C. PFOS-induced hepatic steatosis, the mechanistic actions on β-oxidation and lipid transport. Biochim. Biophys. Acta 2012, 1820, 1092–1101. [Google Scholar] [CrossRef]
- Takacs, M.L.; Abbott, B.D. Activation of mouse and human peroxisome proliferator-activated receptor (a,b/d,g) perfluorooctanoic acid and perfluorooctane sulfonate. Toxicol. Sci. 2007, 95, 108–117. [Google Scholar] [CrossRef]
- Pham, A.; Zhang, J.; Feng, L. Exposure to perfluorobutane sulfonate and perfluorooctanesulfonic acid disrupts the production of angiogenesis factors and stress responses in human placental syncytiotrophoblast. Reprod. Toxicol. 2020, 98, 269–277. [Google Scholar] [CrossRef] [PubMed]
- Bloom, M.S.; Varde, M.; Newman, R.B. Environmental Toxicants and Placental Function. Best Pract. Res. Clin. Obstet. Gynaecol. 2022, 85, 105–120. [Google Scholar] [CrossRef]
- Abbott, B.D.; Wolf, C.J.; Das, K.P.; Zehr, R.D.; Schmid, J.E.; Lindstrom, A.B.; Strynar, M.J.; Lau, C. Developmental toxicity of perfluorooctane sulfonate (PFOS) is not dependent on expression of peroxisome proliferator activated receptor-alpha (PPAR alpha) in the mouse. Reprod. Toxicol. 2008, 27, 258–265. [Google Scholar] [CrossRef]
- Ehrlich, V.; Bil, W.; Vandebriel, R.; Granum, B.; Luijten, M.; Lindeman, B.; Grandjean, P.; Kaiser, A.-M.; Hauzenberger, I.; Hartmann, C.; et al. Consideration of pathways for immunotoxicity of per- and polyfluoroalkyl substances (PFAS). Environ. Health 2023, 22, 19. [Google Scholar] [CrossRef]
- Kirk, A.B.; Michelsen-Correa, S.; Rosen, C.; Martin, C.F.; Blumberg, B. PFAS and Potential Adverse Effects on Bone and Adipose Tissue Through Interactions With PPARγ. Endocrinology 2021, 162, bqab194. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.-Q.; Liu, T.; Yang, S.; Sun, L.; Zhao, Z.-Y.; Li, L.-Y.; She, Y.-C.; Zheng, Y.-Y.; Bao, Q.; Dong, G.-H.; et al. Perfluoroalkyl substance pollutants activate the innate immune system through the AIM2 inflammasome. Nat. Commun. 2021, 12, 2915. [Google Scholar] [CrossRef]
- Bline, A.P.; DeWitt, J.C.; Kwiatkowski, C.F.; Pelch, K.E.; Reade, A.; Varshavsky, J.R. Public Health Risks of PFAS-Related Immunotoxicity Are Real. Curr. Environ. Health Rep. 2024, 11, 118–127. [Google Scholar] [CrossRef]
- Rudzanová, B.; Thon, V.; Vespalcová, H.; Martyniuk, C.J.; Piler, P.; Zvonař, M.; Klánová, J.; Bláha, L.; Adamovsky, O. Altered Transcriptome Response in PBMCs of Czech Adults Linked to Multiple PFAS Exposure: B Cell Development as a Target of PFAS Immunotoxicity. Environ. Sci. Technol. 2023, 58, 90–98. [Google Scholar] [CrossRef]
- Beans, C. How “forever chemicals” might impair the immune system. Proc. Natl. Acad. Sci. USA 2021, 118, e2105018118. [Google Scholar] [CrossRef]
- von Holst, H.; Nayak, P.; Dembek, Z.; Buehler, S.; Echeverria, D.; Fallacara, D.; John, L. Perfluoroalkyl substances exposure and immunity, allergic response, infection, and asthma in children: Review of epidemiologic studies. Heliyon 2021, 7, e08160. [Google Scholar] [CrossRef] [PubMed]
- Kamen, D.L.; Peden-Adams, M.M.; Vena, J.E.; Gilkeson, G.S.; Hulsey, T.C.; Moultrie, L.; Stevens, B.E. Seafood consumption and persistent organic pollutants as triggers of autoimmunity among Gullah African Americans. Arthritis Res. Ther. 2012, 14, A19. [Google Scholar] [CrossRef]
- Grønnestad, R.; Johanson, S.M.; Müller, M.H.B.; Schlenk, D.; Tanabe, P.; Krøkje, A.; Jaspers, V.L.B.; Jenssen, B.M.; Ræder, E.M.; Lyche, J.L.; et al. Effects of an environmentally relevant PFAS mixture on dopamine and steroid hormone levels in exposed mice. Toxicol. Appl. Pharmacol. 2021, 428, 115670. [Google Scholar] [CrossRef] [PubMed]
- Running, L.; Cristobal, J.R.; Karageorgiou, C.; Camdzic, M.; Aguilar, J.M.N.; Gokcumen, O.; Aga, D.S.; Atilla-Gokcumen, G.E. Investigating the Mechanism of Neurotoxic Effects of PFAS in Differentiated Neuronal Cells through Transcriptomics and Lipidomics Analysis. ACS Chem. Neurosci. 2024, 15, 4568–4579. [Google Scholar] [CrossRef]
- Kim, J.I.; Kim, B.-N.; Lee, Y.A.; Shin, C.H.; Hong, Y.-C.; Døssing, L.D.; Dalgaard, L.; Hildebrandt, G.; Lim, Y.-H. Association between early-childhood exposure to perfluoroalkyl substances and ADHD symptoms: A prospective cohort study. Sci. Environ. 2023, 879, 163081. [Google Scholar] [CrossRef] [PubMed]
- Bharal, B.; Ruchitha, C.; Kmar, P.; Pandey, R.; Rachamalla, M.; Niyogi, S.; Naidu, R.; Kaundal, R.V. Neurotoxicity of per- and polyfluoroalkyl substances: Evidence and future directions. Sci. Environ. 2024, 955, 176941. [Google Scholar] [CrossRef] [PubMed]
- Brennan, N.M.; Evans, A.T.; Fritz, M.K.; Peak, S.A.; von Holst, H.E. Trends in the Regulation of Per- and Polyfluoroalkyl Substances (PFAS): A Scoping Review. Int. J. Environ. Res. Public Health 2021, 18, 10900. [Google Scholar] [CrossRef] [PubMed]
- Cousins, I.T.; DeWitt, J.C.; Gluge, J.; Goldenman, G.; Herzke, D.; Lohmann, R.; Miller, M.; Ng, C.A.; Scheringer, M.; Vierkej, L.; et al. Strategies for grouping per- and polyfluoroalkyl substances (PFAS) to protect human and environmental health. Environ. Sci. Process Impacts 2020, 22, 1444–1460. [Google Scholar] [CrossRef]
- Thomas, T.; Malek, A.; Arokianathar, J.; Haddad, E.; Matthew, J. Global Regulations Around PFAS: The Past, the Present and the Future. Int. Chem. Regul. Law Rev. 2023, 6, 3–17. [Google Scholar]
- United States Environmental Protection Agency (USEPA). Perfluoroalkyl Sulfonates; Significant New Use Rule; United States Environmental Protection Agency (USEPA): Washington, DC, USA, 2002. [Google Scholar]
- United States Environmental Protection Agency (USEPA). Fact Sheet: 2010/2015 PFOA Stewardship Program; United States Environmental Protection Agency (USEPA): Washington, DC, USA, 2016. [Google Scholar]
- Cousins, I.T.; DeWitt, J.C.; Gluge, J.; Goldenman, G.; Herzke, D.; Lohmann, R.; Ng, C.A.; Scheringer, M.; Wang, Z. The high persistence of PFAS is sufficient for their management as a chemical class. Environ. Sci. Process Impacts 2020, 22, 2307–2312. [Google Scholar] [CrossRef]
- Environmental Protection Agency Press Office. EPA Announces It Will Keep Maximum Contaminant Levels for PFOA, PFOS: EPA Intends to Provide Regulatory Flexibility and Holistically Address These Contaminants in Drinking Water. 2025. Available online: https://www.epa.gov/newsreleases/epa-announces-it-will-keep-maximum-contaminant-levels-pfoa-pfos (accessed on 21 August 2025).
- Kwiatkowski, C.F.; Andrews, D.Q.; Birnbaum, L.S.; Bruton, T.A.; DeWitt, J.C.; Knappe, D.R.U.; Maffini, M.V.; Miller, M.F.; Pelch, K.E.; Reade, A.; et al. Scientific Basis for Managing PFAS as a Chemical Class. Environ. Sci. Technol. Lett. 2020, 7, 532–543. [Google Scholar] [CrossRef]
- U.S. Chamber of Commerce. Impacts of the PFAS Restriction on Trade Between the U.S. and the European Union. 2023. Available online: https://www.uschamber.com/assets/documents/Impacts-of-PFAS-Restriction-on-Trade-Between-the-U.S.-and-the-European-Union_9.25.2023.pdf (accessed on 3 November 2023).
- Legg, R.; Prior, J. Toxic torts as compensation: Legal geographies of environmental contamination litigation. Geogr. Res. 2023, 61, 234–247. [Google Scholar] [CrossRef]
- Mok, K.; Salvatore, D.; Powers, M.; Brown, P.; Poehlein, M.; Conroy-Ben, O.; Cordner, A. Federal PFAS Testing and Tribal Public Water Systems. Environ. Health Perspect. 2022, 130, 127701. [Google Scholar] [CrossRef]
- Fedinick, K.; Taylor, S.; Roberts, M.; Moore, R.; Olson, E. Watered Down Justice; National Research Development Corporation (NRDC): Washington, DC, USA, 2019; Available online: https://www.nrdc.org/sites/default/files/watered-down-justice-report.pdf (accessed on 3 November 2023).
- Hinshaw. Insurers Face Large PFAS-Related Losses: A Primer on Forever Chemical Regulation, Liabilities, and Insurance Coverage Issues. 2023. Available online: https://www.hinshawlaw.com/newsroom-updates-insights-for-insurers-insurers-face-large-pfas-related-losses.html (accessed on 3 November 2023).
- United States Environmental Protection Agency. Economic Analysis for the Proposed Per- and Polyfluoroalkyl Substances National Primary Drinking Water Regulation; Division, Office of Ground Water and Drinking Water Standards and Risk Management Division Eds.; United States Environmental Protection Agency: Washington, DC, USA, 2023; Available online: https://www.epa.gov/system/files/documents/2023-03/Proposed%20PFAS%20NPDWR%20EA_final_03_09_2023_0.pdf (accessed on 3 November 2023).
- Pepper, S. Transitioning from PFAS Firefighting Foam to Fluorine-Free Alternatives; Montrose Environmental: Little Rock, AR, USA, 2023; Available online: https://montrose-env.com/blog/transitioning-from-pfas-firefighting-foam-to-fluorine-free-alternatives/ (accessed on 3 November 2023).
- Jahura, F.T.; Mazumder, N.-U.S.; Hossain, M.T.; Kasebi, A.; Girase, A.; Ormond, R.B. Exploring the prospects and challenges of fluorine-free firefighting foams (F3) as alternatives to aqueous film-forming foams (AFFF): A review. ACS Omega 2024, 9, 37430–37444. [Google Scholar] [CrossRef]
- Holden, L.A.; East, A.G.; Narizzano, A.M.; Quinn, M.J., Jr. Toxicology assessment for six per- and polyfluoroalkyl (PFAS)-free aqueous film forming foam (AFFF) products. Integr. Environ. Assess. Manag. 2023, 19, 1609–1618. [Google Scholar] [CrossRef]
- Ateia, M.; Van Buren, J.; Barrett, W.; Martin, T.; Back, G.G. Sunrise of PFAS Replacements: A Perspective on Fluorine-Free Foams. ACS Sustain. Chem. Eng. 2023, 11, 7986–7996. [Google Scholar] [CrossRef]
- Marvel, S.W.; To, K.; Grimm, F.A.; Wright, F.A.; Rusyn, I.; Reif, D.M. ToxPi Graphical User Interface 2.0: Dynamic exploration, visualization, and sharing of integrated data models. BMC Bioinform. 2018, 19, 80. [Google Scholar] [CrossRef]
- Gharehveran, M.M.; Walus, A.M.; Anderson, T.A.; Subbiah, S.; Guelfo, J.; Frigon, M.; Longwell, A.; Suski, J.G. Per- and polyfluoroalkyl substances (PFAS)-free aqueous film forming foam formulations: Chemical composition and biodegradation in an aerobic environment. J. Environ. Chem. Eng. 2022, 10, 108953. [Google Scholar] [CrossRef]
- Jones, D.K.; Quinlin, K.A.; Wigren, M.A.; Choi, Y.J.; Sepúlveda, M.S.; Lee, L.S.; Haskins, D.L.; Lotufo, G.R.; Kennedy, A.; May, L.; et al. Acute toxicity of eight aqueous film-forming foams to 14 aquatic species. Environ. Sci. Technol. 2022, 56, 6078–6090. [Google Scholar] [CrossRef]
- Fuller, N.; Suski, J.G.; Lanasa, S.; Chanov, M.K.; Jones, D.K.; Haskins, D.L.; Quinlin, K.A.; Wigren, M.A.; Hoverman, J.T.; Choi, Y.J.; et al. Chronic Toxicity of Per- and Polyfluoroalkyl Substance–Free Firefighting Foams to Aquatic Organisms. Environ. Toxicol. Chem. 2024, 43, 2436–2454. [Google Scholar] [CrossRef] [PubMed]
- Cordner, A.; De la Rosa, V.Y.; Schaider, L.A.; Rudel, R.A.; Richter, L.; Brown, P. Guideline levels for PFOA and PFOS in drinking water: The role of scientific uncertainty, risk assessment decisions, and social factors. J. Expo. Sci. Environ. Epidemiol. 2019, 29, 157–171. [Google Scholar] [CrossRef] [PubMed]
- Office of the Under Secretary of Defense for Acquisition and Sustainment. Report on Per- and Polyfluoroalkyl Substances Active Sites Cleanup Costs; United States Department of Defense: Washington, DC, USA, 2022; p. 16. Available online: https://media.defense.gov/2022/Jul/21/2003039883/-1/-1/1/REPORT-ON-PER-AND-POLYFLUOROALKYL-SUBSTANCES-ACTIVE-SITES-CLEANUP-COSTS-June-2022.PDF (accessed on 18 June 2025).
- Environmental Working Group. The Pentagon’s contamination time bomb: Cleanup backlog outpaces funding. News Insights, 15 May 2023. Available online: https://www.ewg.org/news-insights/news-release/2023/05/pentagons-contamination-time-bomb-cleanup-backlog-outpaces (accessed on 18 June 2025).
- Wolohan, K.; Ling, A.; McCabe, A.; Vermace, B.; Richard, D.; Gantzer, C.; Nelson, S.; Muson, A.; Dursun, D.; Blate, M.; et al. Evluation of Current Alternatives and Estimateed Cost Curves for PFAS Removal and Destruction from Municipal Watewater, Biosolids, Landfill Leachate, and Compost Contact Water; Minnesota Pollution Control Agency: St. Paul, MN, USA, 2023. [Google Scholar]
- AutoinsuranceEZ. Gas vs. Electric Car Fires. Available online: https://www.autoinsuranceez.com/gas-vs-electric-car-fires/ (accessed on 31 July 2025).
- Kalhoff, J.; Bresser, D.; Bolloli, M.; Alloin, F.; Sanchez, J.-Y.; Passerini, S. Enabling LiTFSI-based Electrolytes for Safer Lithium-Ion Batteries by Using Linear Fluorinated Carbonates as (Co)Solvent. ChemSusChem 2014, 7, 2939–2946. [Google Scholar] [CrossRef]
- Owens, E.O.; Lizarraga, L.; Lambert, J.C.; Ahirke, A. ORD Human Health Toxicity Value for Lithium bis[(trifluoromethyl)sulfonyl]azanide (HQ-115) (CASRN 90076-65-6|DTXSID8044468); EPA IRIS Assessments; United States Environmental Protection Agency: Cincinnati, OH, USA, 2023. [Google Scholar]
- Boyd, R.I.; Shokry, D.; Fazal, Z.; Rennels, B.C.; Freemantle, S.J.; La Frano, M.R.; Prins, G.S.; Erdogan, Z.M.; Irudayaraj, J.; Singh, R.; et al. Perfluorooctanesulfonic Acid Alters Pro-Cancer Phenotypes and Metabolic and Transcriptional Signatures in Testicular Germ Cell Tumors. Toxics 2024, 12, 232. [Google Scholar] [CrossRef] [PubMed]
- Sands, M.; Zhang, X.; Gal, A.; Laws, M.; Spinella, M.; Erdogan, Z.-M.; Irudayaraj, J. Comparative hepatotoxicity of novel lithium bis(trifluoromethanesulfonyl)imide (LiTFSI, ie. HQ-115) and legacy Perfluorooctanoic acid (PFOA) in male mice: Insights into epigenetic mechanisms and pathway-specific responses. Environ. Int. 2024, 185, 108556. [Google Scholar] [CrossRef]
- Sands, M.; Zhang, X.; Irudayaraj, J. Kidney toxicology of a novel compound Lithium Bis (trifluoromethanesulfonyl)imide (LiTFSI, ie. HQ-115) used in energy applications: An epigenetic perspective. Sci. Environ. 2024, 955, 177019. [Google Scholar] [CrossRef] [PubMed]
- Dewapriya, P.; Chadwick, L.; Gorji, S.G.; Schulze, B.; Valsecchi, S.; Samanipour, S.; Thomas, K.V.; Kaserzon, S.L. Per- and polyfluoroalkyl substances (PFAS) in consumer products: Current knowledge and research gaps. J. Hazard. Mat Lett. 2023, 4, 100086. [Google Scholar] [CrossRef]
- Lendewig, M.; Marquez, R.; Franco, J.; Vera, R.E.; Vivas, K.A.; Forfora, N.; Venditti, R.A.; Gonzalez, R. PFAS regulations and economic impact: A review of U.S. pulp & paper and textiles industries. Chemosphere 2025, 377, 144301. [Google Scholar] [CrossRef]
- BCLPemerging.com. PFAS Drinking Water Standards: STATE-by-State Regulations Updated: June 2025. Available online: https://www.bclplaw.com/en-US/events-insights-news/pfas-drinking-water-standards-state-by-state-regulations.html#:~:text=For%20example%2C%20Florida%2C%20Illinois%2C,(PWS)%20in%20various%20states (accessed on 21 August 2025).
- Source Intelligence. PFAS Regulations by State for Consumer Products. Available online: https://blog.sourceintelligence.com/us-pfas-regulations-by-state#:~:text=PFAS%20are%20widely%20used%20in,gear%2C%20to%20enhance%20their%20durability (accessed on 21 August 2025).
- Destoumieux-Garzón, D.; Mavingui, P.; Boetsch, G.; Boissier, J.; Darriet, F.; Duboz, P.; Fritsch, C.; Giraudoux, P.; Le Roux, F.; Morand, S.; et al. The One Health Concept: 10 Years Old and a Long Road Ahead. Front. Vet. Sci. 2018, 5, 14. [Google Scholar] [CrossRef] [PubMed]
- Ankley, G.T.; Cureton, P.; Hoke, R.A.; Houde, M.; Kumar, A.; Kurias, J.; Lanno, R.; McCarthy, C.; Newsted, J.; Salice, C.J.; et al. Assessing the Ecological Risks of Per- and Polyfluoroalkyl Substances: Current State-of-the Science and a Proposed Path Forward. Environ. Toxicol. Chem. 2021, 40, 564–605. [Google Scholar] [CrossRef] [PubMed]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baldwin, W.S.; Bloom, M.S.; Chung, K.W.; Dasgupta, S.; DeLorenzo, M.E.; Hunt, K.J.; Key, P.B.; Pearce, J.L.; Rock, K.D.; Tanabe, P.; et al. Toxicological, Chemical, Social, and Economic Challenges Associated with PFAS and Replacement Aqueous Film-Forming Foams (AFFF). Toxics 2025, 13, 732. https://doi.org/10.3390/toxics13090732
Baldwin WS, Bloom MS, Chung KW, Dasgupta S, DeLorenzo ME, Hunt KJ, Key PB, Pearce JL, Rock KD, Tanabe P, et al. Toxicological, Chemical, Social, and Economic Challenges Associated with PFAS and Replacement Aqueous Film-Forming Foams (AFFF). Toxics. 2025; 13(9):732. https://doi.org/10.3390/toxics13090732
Chicago/Turabian StyleBaldwin, William S., Michael S. Bloom, Katy W. Chung, Subham Dasgupta, Marie E. DeLorenzo, Kelly J. Hunt, Peter B. Key, John L. Pearce, Kylie D. Rock, Philip Tanabe, and et al. 2025. "Toxicological, Chemical, Social, and Economic Challenges Associated with PFAS and Replacement Aqueous Film-Forming Foams (AFFF)" Toxics 13, no. 9: 732. https://doi.org/10.3390/toxics13090732
APA StyleBaldwin, W. S., Bloom, M. S., Chung, K. W., Dasgupta, S., DeLorenzo, M. E., Hunt, K. J., Key, P. B., Pearce, J. L., Rock, K. D., Tanabe, P., Jacobellis, M. A., Garcia, M. M., & Bain, L. J. (2025). Toxicological, Chemical, Social, and Economic Challenges Associated with PFAS and Replacement Aqueous Film-Forming Foams (AFFF). Toxics, 13(9), 732. https://doi.org/10.3390/toxics13090732









